
MIRA INFORM REPORT
|
Report
Date : |
19th June, 2006. |
IDENTIFICATION
DETAILS
|
Name : |
DR REDDY’S LABORATORIES LIMITED |
|
|
|
|
Registered
Office : |
7-1-27, Ameerpet, Hyderabad – 500 016, Andhra Pradesh,
India |
|
|
|
|
Country
: |
India |
|
|
|
|
Financials
(as on) : |
31.03.2005 |
|
|
|
|
Date
of Incorporation : |
24.02. 1984 |
|
|
|
|
Com.
Reg. No.: |
01-4507 |
|
|
|
|
CIN
No.: [Company
Identification No.] |
L85195AP1984PLC004507 |
|
|
|
|
TAN
No.: [Tax
Deduction & Collection Account No.] |
HYDD00080D |
|
|
|
|
Legal
Form : |
Public
limited liability company. The company’s shares are listed on the Stock
Exchanges. |
|
|
|
|
Line
of Business : |
Manufacturers
and Sellers of Bulk Drugs, Formulations and Diagnostic Reagents and Kits. |
RATING & COMMENTS
|
MIRA’s
Rating : |
A |
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
56-70 |
A |
Financial & operational base
are regarded healthy. General unfavourable factors will not cause fatal
effect. Satisfactory capability for payment of interest and principal sums |
Fairly Large |
|
Maximum
Credit Limit : |
USD
80000000 |
|
|
|
|
Status
: |
Good |
|
|
|
|
Payment
Behaviour : |
Regular |
|
|
|
|
Litigation
: |
Clear |
|
|
|
|
0Comments
: |
Subject
is an old, well-established and reputed company engaged in manufacturing and
marketing of pharmaceuticals. The
company manufactures wide range of pharmaceutical products in India and
overseas. The company is making
satisfactory progress in its business and profitability. Directors are well-experienced and
resourceful businessmen. Their trade
relations are fair. Payments are
usually correct and as per commitments.
It can be
considered good for business dealings at usual trade terms and conditions. |
LOCATIONS
|
Registered
Office : |
7-1-27,
Ameerpet, Hyderabad – 500 016, Andhra Pradesh, India |
|
Tel.
No.: |
91-40-23731946/23731397/26511723 |
|
Fax
No.: |
91-40-23731955/23734504 |
|
E-Mail
: |
|
|
Website
: |
|
|
|
|
|
Corporate
Office : |
7-1-27, Ameerpet, Hyderabad – 500 016, Andhra Pradesh,
India |
|
Tel.
No.: |
91-40-23731946 |
|
Fax
No.: |
91-40-23731955 |
|
Website
|
|
|
|
|
|
Plants
(In India) : |
Bulk Drugs – I, II, III and IV v
Plot
Nos. 137, 138 & 146, IDA Bollarum, Jinnaram Mandal, Medak District - 502 320, Andhra Pradesh v
Plot
Nos. 110 & 111, IDA Bollarum, Jinnaram Mandal, Medak District - 502 320, Andhra Pradesh v
Plot
Nos. 116, IDA Bollarum, Jinnaram Mandal, Medak District - 502 320, Andhra Pradesh v
Plot
No. 9/A, Phase III, IDA Jeedimetla Ranga Reddy District – 500 055, Andhra
Pradesh Bulk Drugs – V v
Peddadevulapally,
Tripuraram Mandal, Nalgonda District – 508207, Andhra Pradesh, India Bulk Drugs – VI v IDA Pydibheemavaram,
Ransthal Mandal, Srikakulam District – 532409, Andhra Pradesh Formulations I – IDA Bollaram Jinnaram Mandal,
Medak District – 502320, Andhra Pradesh, India II- Survey No. 42, Bachupally
Quthbullapur Mandal, Ranga Reddy District – 500123, Andhra Pradesh, India III – R S No. 63/3 and 63/4,
Thiruvandarkoil Mannvipet, Pondicherry – 605102, Tamil Nadu, India IV – Ward – F, Block –4, Adavipolam,
Yanam, Pondicherry – 533465, Tamil Nadu, India V – Plot No. A-3 to A-6, Phase 1-A,
Verna Industiral Estate, Verna, Goa – 403722 Generics Survey
No. 41, Bachupally Quthbullapur Mandal, Ranga Reddy District – 500043, Andhra
Pradesh, India Boitech/Critical Care/Diagnostics Survey No.47, Bachupally Quthbullapur Mandal, Ranga Reddy District –
500043, Andhra Pradesh, India Custom Chemical Services/Discovery
Research Bollaram
Road, Miyapure, Hyderabad – 500050, Andhra Pradesh, India |
|
|
|
|
Plants
(Outside India) : |
v
Dr.
Reddy’s Laboratories (UK) Limited Riverview Road, Beverly, East
Yorkshire, HU 17 Old, United Kingdom. v
Kunshan
Rotam Reddy Pharmaceuticals Company Limited Huangpujiangzhonglu Kunshan Economic
and Technological Development Zone, Jiangsu province, China v
Dr.
Reddy’s Laboratories (UK) Limited 208-214, York
road, Battersea, London, SW11 3SD, United
Kingdom |
|
|
|
|
Overseas
Offices : |
v
Riverview
Road, Beverly, East Yorkshire, HU 17 Old United Kingdom v
Huangpujiangzhonglu
Kunshan Economic and Technologica Development Zone, Jiangsu Province, China v
208-214,
York Road, Battersea, London, SW 11-3SD, United Kingdom |
DIRECTORS
|
Dr. K.
Anji Reddy |
Executive Chairman |
|
Mr. G. V.
Prasad |
Executive Vice-Chairman & Chief Executive
Officer |
|
Mr.
Satish Reddy |
Managing Director & Chief Operating Officer |
|
Dr. P.
Satyanarayana Rao |
Director |
|
Dr. V.
Mohan |
Director |
|
Dr. Omkar
Goswami |
Director |
|
Mr. Ravi
Bhoothalingam |
Director |
|
Mr. P. N.
Devarajan |
Director |
|
Dr. A.
Venkateswarlu |
Director |
|
Mr.
Krishna G. Palepu |
Additional Director |
|
Mr.
Anupam Puri |
Non –executive director |
|
|
|
OTHER PERSONNEL:-
|
|
|
Mr.
Santosh Kumar Nair |
Company Secretary |
|
|
|
MANAGEMENT
|
|
|
Dr. K.
Anij Reddy |
Executive Director |
|
Mr. G. V.
Prasad |
Executive Vice Chairman & CEO |
|
Mr. K.
Satish Reddy |
Managing Director and Chief Operation Officer |
|
Mr. V. S.
Vasudevan |
Chief Financial Officer |
|
Dr. R.
Rajagopalan |
President |
|
Mr. Arun
Sawhney |
President |
|
Mr.
Abhijit Mukherjee |
President |
|
Mr. K. B.
Sankara Rao |
Executive Vice President |
|
Mr.
Saumen Chakraborthy |
Executive Vice President |
|
Mr. S.
Venkatraman |
Senior Vice President |
|
Mr. Vilas
M. Dholye |
Senior Vice President |
|
Mr.
Ashwani Kumar Malhotra |
Senior Vice President |
|
Mr. C. V.
Narayana Rao |
Vice President |
|
Mr.
Ranjan Chakraborthy |
Vice President |
|
Dr. N. R.
Srinivas |
Vice President |
|
Dr. Javed
Iqbal |
Distinguish Research Scientist |
|
Mr. Jaspal
Singh Bajwa |
President |
|
Dr.
Jayaram Chigurupati |
Executive Vice President |
|
Dr. G. Om
Reddy |
Senior Vice President |
|
Mr. B. R.
Reddy |
Senior Vice President |
|
MR.
Arvind Vasudeva |
Vice President |
|
Dr. M.
Satyanarayana Reddy |
Vice President |
|
Dr. R.
Buchi Reddy |
General Manager |
KEY EXECUTIVES
|
Name |
Mr. K. Satish Reddy |
|
Designation |
Managing Director & Chief Operating Officer |
|
Age |
33 years |
|
Qualification |
B. Tech., M. S. |
|
Experience |
9 years |
|
Date
of Joining |
18th January, 1993 |
|
Previous
Employment |
Director – Globe Organics Limited |
|
Other
Directorships |
1.
Diana Hotels Limited 2.
DRL Investments Limited 3.
Compact Electric Limited 4.
Cheminor Investments Limited |
|
Name |
Dr. K. Anji Reddy |
|
Designation |
Executive Chairman |
|
Age |
61 years |
|
Qualification |
B. Sc. (Tech.), Ph. D. |
|
Experience |
31 years |
|
Date
of Joining |
1st September, 1986 |
|
Previous
Employment |
Managing Director – Standard Organics Limited |
|
Other
Directorships |
1.
Diana Hotels Limited 2.
ICICI Venture Funds 3.
Deccan Hospitals Corporation Limited 4.
Biotech Consortium India Limited 5.
Viral Therapeutic, Inc. |
Brief Profile of Dr. K. Anji Reddy:
He is the
founder and the Executive Chairman of Dr. Reddy’s Laboratories Limited. He is
also the founder of the Dr. Reddy’s Group, Dr. Reddy’s Research Foundation and
Dr. Reddy’s Foundation for Human and Social Development. He is the chairman of
the Academy of Human Resources Development and chairman of the Research and
Development Committee of the Federation of Indian Chamber of Commerce and
Industry (FICCI). He is a member of both the Board of Trade and the Task Force
on pharmaceuticals and knowledge-based industries, which was instituted by the
Prime Minister. He has been recently honoured with the Padmashree by the
Government of India, for his distinguished service in the field of trade and commerce.
MAJOR SHAREHOLDERS
Category |
No. of shares |
% of shareholding |
promoters' holdings
|
|
|
|
Individuals
|
2246894 |
2.94 |
|
Companies
|
17461730 |
22.82 |
|
Directors |
700 |
-- |
|
Sub Total |
19709324 |
25.76 |
|
|
|
|
|
Indian
Financial Institutions |
5785844 |
7.56 |
|
Banks |
89458 |
0.12 |
|
Mutual
funds |
2061063 |
2.69 |
|
|
|
|
|
Foreign Holdings |
|
|
|
Foreign
Institutional Investors |
15964503 |
20.86 |
|
NRIs |
1933438 |
2.53 |
|
GDRs/ADSs |
20490993 |
26.78 |
|
Others |
850000 |
-- |
|
Indian
Public and Corporates |
10483226 |
13.70 |
|
Sub Total |
76518949 |
100.00 |
|
|
|
|
|
Indian
Public and Corporate |
11912687 |
15.57 |
|
|
|
|
|
Grand Total |
76515948 |
100.00 |
BUSINESS DETAILS
|
Line
of Business : |
Manufacturers and Sellers of Bulk Drugs, Formulations and
Diagnostic Reagents and Kits. |
|
|
|
|
Products
: |
Item Code No. [ITC Code] 29419003 Product Description Ciprofloxacin
Hydrochloride Item Code No. [ITC Code] 29420001 Product Description Norfloxacin Item Code No. [ITC Code] 30049038 Product Description Omerprazole |
PRODUCTION STATUS
|
Class of Goods |
Unit |
Licensed Capacity |
Installed Capacity |
Actual Production |
|
|
|
|
|
|
|
Formulations |
Million
Units |
2806.95 |
2806.95 |
2117.72 |
|
Active Pharmaceutical ingredients and
intermediates [API] |
Tones |
6941 |
4068 |
3153 |
|
Generics |
Million
Units |
5550 |
5550 |
1285.28 |
|
Diagnostic reagents and kits |
Units |
600000 |
600000 |
10667 |
|
Biotechnology – on single shift basis |
Grams |
370 |
340 |
14.79 |
GENERAL INFORMATION
|
No. of
Employees : |
1449 |
|
|
|
|
Bankers
: |
v Allahabad Bank, Industrial Finance
Branch, Secunderabad, Andhra Pradesh, India v Bank of Baroda, Khairatabad
Branch, Hyderabad, Andhra Pradesh, India v Canara Bank, Basheerbagh,
Hyderabad, Andhra Pradesh, India v Citibank, Hyderabad, Andhra
Pradesh, India v Global Trust Bank, Secunderabad,
Andhra Pradesh, India v HDFC Bank, Hyderabad, Andhra
Pradesh, India v The Hongkong & Shanghai
Banking Corporation Limited, Hyderabad, Andhra Pradesh, India v State Bank of Hyderabad, Overseas
Branch, Hyderabad, Andhra Pradesh, India v State Bank of India, Industrial
Finance Branch, Hyderabad, Andhra Pradesh, India v State Bank of Mysore, Industrial
Finance Branch, Hyderabad, Andhra Pradesh, India v Standard Chartered Grindlays Bank
Limited, Hyderabad, Andhra Pradesh, India |
|
|
|
|
Facilities : |
Secured loan Others Cash Credit 319.394 Loan from Indian Renewable Energy Development Agency Limited 36.985 356.379 Unsecured loan Foreign currency loan notes 146.738 Sales Tax Deferment Loan from the Government of Andhra Pradesh (Interest Free) 79.108 Short –
term loans from Banks Overdraft -- Total
225.846 |
|
|
|
|
Banking Relations : |
Good |
|
|
|
|
Auditors
: |
Bharat S.
Raut & Company Chartered
Accountants |
|
|
|
|
Associates
: |
v
Pathnet
India Private Limited v
Aurantis
Farmaceutica Ltda v
Compact
Electric Limited v
APR
LLC v
Standard
Organics Limited v
Dr.
Reddy’s Exports Limited v
Sol
Pharmaceuticals Limited |
|
|
|
|
Subsidiaries
: |
v
OOO
JV Reddy Biomed Limited, Russia v
Reddy
Pharmaceuticals Hong Kong Limited, Hong Kong v
Dr.
Reddy’s Laboratories Inc., USA v
Reddy’s
Cheminor S.A., France v
Reddy
Antilles N.V. v
Aurigene
Discovery Technologies Limited, India v
Dr.
Reddy’s Laboratories {EU} Limited, UK [Formerly known as BMS Laboratories
Limited, UK] v
Dr.
Reddy’s Laboratories {EU} Limited, UK [Formerly known as Meridian Healthcare,
UK] v
Chemnior
Investments Limited, India v
DRL
Investments Limited, India v
OOO
Dr. Reddy’s Laboratories Limited v
Dr.
Reddy’s Laboratories [Proprietory] Limited, South Africa v
Dr.
Reddy’s Biosciences Limited v
Reddy
Netherlands B.V. , Netherlands v
Reddy
Pharmaceuticals Singapore Pte. Limited, Singapore v
Reddy
US Therapeutics Inc., USA v
AMPNH
Inc. v
Zenovus
Biotech Private Limited, India v
Compact
Electric Limited, India v
Dr.
Reddy’s Pharmaceutical Do Brasil LTDA v
Kunshan
Rotam Reddy Pharmaceutical Company Limited v
Globe
Enterprises [a partnership firm in India] v
BMU
Laboratories Limited v
Meridian
Healthcare (U.K.) Limited |
|
|
|
|
Membership
: |
|
CAPITAL STRUCTURE
Authorised
Capital :
|
No. of
Shares |
Type |
Value |
Amount |
|
100000000 |
Equity
Shares |
Rs. 5/- each |
Rs. 500.000 millions |
Issued,
Subscribed & Paid-up Capital :
|
No. of
Shares |
Type |
Value |
Amount |
|
76519149 |
Equity
Shares |
Rs. 5/- each |
Rs. 382.596 millions |
FINANCIAL DATA
[all figures are in Rupees Millions]
ABRIDGED
BALANCE SHEET
|
SOURCES
OF FUNDS |
31.03.2005 |
31.03.2004 |
31.03.2003 |
|
SHAREHOLDERS FUNDS |
|
|
|
|
1] Share Capital |
382.600 |
382.595 |
382.580 |
|
2] Reserves & Surplus |
20358.200 |
20087.566 |
17686.622 |
|
NETWORTH |
20740.800 |
20470.161 |
18069.202 |
|
|
|
|
|
|
LOAN FUNDS |
|
|
|
|
1] Secured Loans |
32.700 |
356.379 |
42.905 |
|
2] Unsecured Loans |
2699.700 |
225.846 |
244.654 |
TOTAL BORROWING
|
2732.400 |
582.225 |
287.559 |
|
DEFERRED TAX LIABILITIES |
0.000 |
422.582 |
423.468 |
|
|
|
|
|
GRAND
TOTAL
|
23473.200 |
21474.968 |
18780.229 |
|
|
|
|
|
|
APPLICATION OF FUNDS |
|
|
|
|
|
|
|
|
|
FIXED ASSETS [Net Block] |
5625.400 |
4580.983 |
3957.636 |
|
Capital work-in-progress |
601.300 |
1052.469 |
514.067 |
|
|
|
|
|
|
INVESTMENTS |
3584.600 |
6120.511 |
1566.374 |
|
|
|
|
|
|
CURRENT ASSETS, LOANS & ADVANCES |
|
|
|
|
Inventories |
3038.100 |
2580.110 |
2401.168 |
|
Sundry Debtors |
4176.400 |
4440.462 |
4324.515 |
|
Cash & Bank Balances |
8917.200 |
4080.832 |
6884.002 |
|
Loans & Advances |
2679.000 |
2114.856 |
1868.143 |
|
Total Current Assets |
18810.700 |
13216.260 |
15477.828 |
|
Less : |
|
|
|
|
Current Liabilities |
4515.000 |
2913.942 |
2183.378 |
Provisions
|
633.800 |
581.313 |
552.298 |
Total Current Liabilities
|
5148.800 |
3495.255 |
2735.676 |
|
Net
Current Assets |
13661.900 |
9721.005 |
12742.152 |
|
|
|
|
|
|
MISCELLANEOUS EXPENSES |
0.000 |
0.000 |
0.000 |
|
|
|
|
|
GRAND
TOTAL
|
23473.200 |
21474.968 |
18780.229 |
PROFIT & LOSS ACCOUNT
|
PARTICULARS |
31.03.2005 |
31.03.2004 |
31.03.2003 |
|
Sales Turnover [including other income] |
17299.700 |
17423.711 |
15750.606 |
|
|
|
|
|
|
Profit/(Loss) Before Tax |
443.600 |
3033.438 |
4311.603 |
|
Provision for Taxation |
(211.000) |
201.475 |
390.677 |
|
Profit/(Loss) After Tax |
654.600 |
2831.960 |
3920.926 |
|
|
|
|
|
|
Export Value |
NA |
9854.318 |
9252.582 |
|
|
|
|
|
Import Value
|
NA |
2186.677 |
1634.434 |
|
|
|
|
|
Total Expenditure
|
16424.100 |
14390.273 |
11436.003 |
SUMMARISED RESULTS
|
Particulars |
|
|
31.03.2006 ( Full Year) |
|
|
|
|
|
|
Sales Turnover |
|
|
20058.500 |
|
Other Income |
|
|
1230.700 |
|
Total Income |
|
|
21289.200 |
|
Total Expenditure |
|
|
17324.000 |
|
Operating Profit |
|
|
3965.200 |
|
Interest |
|
|
214.200 |
|
Gross Profit |
|
|
3751.000 |
|
Depreciation |
|
|
1113.300 |
|
Tax |
|
|
207.100 |
|
Reported PAT |
|
|
2111.300 |
KEY
RATIOS
|
PARTICULARS |
31.03.2005 |
31.03.2004 |
31.03.2003 |
|
Debt Equity Ratio |
0.08 |
0.02 |
0.01 |
|
Long Term Debt Equity Ratio |
0.01 |
0.01 |
0.01 |
|
Current Ratio |
2.64 |
3.73 |
4.86 |
|
TURNOVER RATIOS |
|
|
|
|
Fixed Assets |
1.79 |
2.33 |
2.55 |
|
Inventory |
5.79 |
6.99 |
7.44 |
|
Debtors |
3.78 |
3.97 |
3.64 |
|
Interest Cover Ratio |
4.48 |
72.71 |
72.27 |
|
Operating Profit Margin (%) |
9.19 |
21.80 |
31.16 |
|
Profit Before Interest and Tax Margin (%) |
3.51 |
17.67 |
27.35 |
|
Cash Profit Margin (%) |
9.71 |
20.40 |
28.34 |
|
Adjusted Net Profit Margin (%) |
4.02 |
16.27 |
24.53 |
|
Return on Capital Employed (%) |
2.56 |
15.61 |
26.44 |
|
Return on Net Worth (%) |
3.18 |
14.70 |
24.02 |
STOCK PRICES
|
Face Value |
Rs. 5.00 |
|
High |
Rs. 929.00 |
|
Low |
Rs. 922.00 |
LOCAL AGENCY FURTHER INFORMATION
HISTORY
The company
was incorporated on 24th February 1984 as a Private Limited
Liability Company at Hyderabad in Andhra Pradesh having Company Registration
Number 4507 and was converted into a Public Limited Liability Company on 2nd
November 1985.
Subject is a leading
Indian pharmaceutical company with vertically integrated operations. The
company develops, manufactures and markets a wide range of pharmaceutical
products in India and overseas. It produces finished dosage forms, active
pharmaceutical ingredients, diagnostic kits, critical care and biotechnology
products. The company has over 190 finished dosage brands and 60 active
pharmaceutical ingredients currently in production.
The company
actively pursues a basic research programme under the aegis of Dr. Reddy’s
Research Foundation (DRF). DRF focuses on cancer, diabetes, bacterial
infections and pain management. The company has several pharmaceutical products
in development, three of which are in clinical trials and two had completed
pre-clinical testing.
The merger
with Cheminor Drugs (the swap ratio at nine shares of company for 25 shares of
Cheminor), had made DRL the third largest pharmaceutical company in India with
participation in every element of the value chain. DRL is a major player in the
domestic finished dosages market and many of its brands are leaders. The
company has a formidable presence in the highly regulated markets of the
U.S.A., the Europe and Japan and it exports its’ products to 60 countries.
The company
has two US-FDA approved plants. It has been exporting its products to the UK,
Switzerland, Germany, Spain, Italy and The Netherlands. It also started
exporting its formulations in a big way to Russia and has set up an office
there. DRL has signed a joint venture agreement with the Khetan Group, Nepal,
for setting up a joint venture for the manufacture and marketing of finished
formulations in Nepal and other neighbouring countries. It also signed a
marketing and distribution agreement with Organics, Israel, for a wide range of
sophisticated diagnostic kits. The products are recognised by World Health
Organisation and other leading organisations in the healthcare industry.
In May
2002, DRL has completed phase I clinical trials on its anti-cancer compound
DRF-1042. This is company’s first new
chemical entity (NCE) in the cancer area.
The company
issued 4,301,076 GDSs representing 4,301,076 equity shares of the company, par
value Rs.10 ("Shares"), in a private placement in 1994 pursuant to
Regulation S and Rule 144A under the Securities Act of 1933 (the
"Securities Act"). The GDSs
are listed on the Luxembourg Stock Exchange and each GDS represents one Share.
As of May 4, 2001, there were 1,789,285 GDSs outstanding representing 1,789,285
Shares.
The company entered the global generic market with exports of Ranitidine-75 mg
and Fluoxetine to North America.
The company has entered into an exclusive co-marketing and development
agreement with Par Pharmaceuticals Inc. covering fourteen generic
pharmaceutical products. This will strengthen the company's position in the US
generic market and it will get a substantial cost advantage on account of its
vertical integration capabilities. The company unveiled its new corporate
identity and philosophy - "Life, Research, Hope"- reinforcing its
commitment of bringing hope to life through research. Its new identity also
highlights the company's ethos - a caring organisation that leverages its
expertise in research for a healthier life.
In April
2001, as a first step towards taking its molecules through clinical development
on its own, the company had selected Simbec Research Limited, a well-known
U.K.-based Clinical Research Organization (CRO), for conducting clinical trials
of DRF 4832. DRF 4832 was a PPAR against for treatment of cardiovascular
complications.
In April
2001, the company began trading on the New York Stock Exchange (code : RDY).
The price to the public per ADS was $ 10.04. Total amount raised (net) was $
124 million.
In May
2001, Novartis Pharma AG and the company announced that they had entered a
licensing agreement for a novel anti-diabetes agent. Under terms of the
agreement, the company will grant Novartis worldwide exclusive right to
development and commercialisation of their insulin sensitiser DRF 4158 in type 2 diabetes, in return for upto
USD 55 million in upfront and milestone payments for specific clinical and
regulatory end points, as well as royalties. The Company would have
co-promotion rights for DRF 4158 in India. The agreement has received US
regulatory clearance and had become effective from July 30, 2001. This event
had triggered an upfront payment of 5 millions US$ from Novarties. The company
had received this payment.
The
company’s 9 products were awaiting USFDA approval with brandmarket value of US$
10.4 billion and it plans to file 11 ANDAs in FY 02 with brand market value of
$ 8.9 billion. In May 2002, The company has completed phase I clinical trials
on its anti-cancer compound DRF-1042. This was companies first new chemical
entity (NCE) in the cancer area.
With the
approval of shareholders the face value of the company shares has been reduced
to Rs. 5/- per share. The scheme of merger with erstwhile American Remedies
Limited was fully completed and shares were exchanged for one share of the
company for every 12 shares in erstwhile ARL.
The company
has expanded the installed capacity of formulations, bulk drugs and generics
during the year 2002-03 by 1.3 million units, 546 tonnes and 1950 million
(units), respectively. With this
expansion the total capacity had been risen to 2137.3 million (units), 3859
tonnes, 5550 million (units) respectively.
The year
2003 was significant to the company as it was poured by Awards. The company has got many awards during the
year which includes the National Award for best presented accounts in Annual
Reports for the financial year 2001-02 from ICAI.
The company
has expanded the installed capacity of Formulations and Bulk Drugs during the
year 2003-04 by 669650000 (Nos) and 209 Tonnes respectively. Consequent of this
expansion the total capacity has risen to 2806950000 (Nos) in respect of
Formulations and 4068 Tonnes in respect of Bulk Drugs.
It is in
trade terms with :-
v
Godavari
Plasto Containers Private Limited
v
Super
Olefins (Private) Limited
v
Murthy’s
Lab Glass Works
v
Ability
Engineering Equipments
v
Vinayak
Metal Labs
v
Hyderabad
Security and Offset Printers
v
Lisa
Ampoules and Vials (Private) Limited
v
Tirumula
Comprints Private Limited
v
Secunderabad
Printed Cartons
v
Vivala
Cartons Private Limited
v
Apex
Drugs and Intermediates Limited
v
Paper
Pack Industries
v
Walnut
Packaging Private Limited
v
Regal
Packaging
v
Sigachi
Chloro Chemicals Private Limited
v
PCR
Metacaps
v
Sree
Deepti Packaging Industries
v
Susheel
Enterprises
v
Milan
Art Printers
v
Temple
Packaging (Private) Limited
v
Madhavi
Engineering Company
v
Nagoor
Services
v
Sree
Industrial Services
v
Veer
Chemi And Aromatics
v
Tarus
Chemicals
v
Surya
Industrial Equipments
v
Sree
Deepthi Packing Industries
v
Class
Packaging
v
Dakshin
Packaging Private Limited
v
Indras
Agencies Limited
v
Srikals
Graphics
v
Esjay
Polyproducts Private Limited
The company
has joint venture with :-
v
Kunshan
rotam Reddy Pharmaceutical Company Limited
The
company’s fixed assets of important value include Land, Factory Buildings,
Leasehold Buildings, Plant & Machinery, Electrical Equipments, Laboratory
Equipments, Furniture & Fixtures, Patents & Trademarks, Vehicles and
Library.
PRESS RELEASES:
Hyderabad, India, November 8, 2005
Dr. Reddy’s signs definitive agreement to acquire Roche’s API business
at it’s Mexico facility
Hyderabad, India, November 8, 2005: Dr. Reddy’s Laboratories
Limited (NYSE: RDY) announced today that the Company has entered into a
definitive agreement to acquire Roche’s API business at the state-of-the-art
manufacturing site in Cuernavaca, Mexico including all employees and business
supply contracts. The total investment outlay is about USD 59 million including
working capital.
This business involves the manufacture and sale of
APIs including intermediates to Roche and other Innovator Companies. The
product portfolio currently comprises about 18 products including mature APIs
and a range of intermediates and steroids. This acquisition also adds unique
steroids manufacturing capabilities to Dr. Reddy’s. The Cuernavaca site at
Mexico currently employs nearly 340 people and has been inspected by the
U.S.FDA and other international regulatory agencies.
Commenting on the acquisition, GV Prasad, CEO, Dr.
Reddy's Laboratories, said, “With the acquisition of Roche’s API business at
the Mexico site, Dr. Reddy’s will emerge as a leading player in Custom
Pharmaceutical Services (CPS) business and position itself as a partner of
choice for Innovator Companies across the globe with service offerings spanning
the entire value chain of pharmaceutical services. This strategic acquisition
provides an opportunity for their CPS business to grow from the current base of
USD10 million to USD100 million over the next 18 months. The integration of
these businesses - people and technology; the expertise of the management team
and distinctive manufacturing capability at the Mexico site, with that of Dr.
Reddy’s strengths in process chemistry and world class research capabilities
will provide a strong foundation to drive the next wave of growth in their CPS
business.
The acquisition is expected to be completed by end of
December 2005. The Company expects the acquisition to be EPS accretive to Dr.
Reddy’s and the full year financial impact of the acquisition will be realized
from the year 2006-07 onwards.
About Dr. Reddy’s
Established in 1984, Dr. Reddy's Laboratories (NYSE: RDY) is an emerging global
pharmaceutical company with proven research capabilities. The Company is
vertically integrated with a presence across the pharmaceutical value chain. It
produces finished dosage forms, active pharmaceutical ingredients and
biotechnology products and markets them globally, with focus on India, US,
Europe and Russia. The Company conducts research in the areas of diabetes,
cardiovascular, anti-infectives, inflammation and cancer.
About Roche
Headquartered in Basel, Switzerland, Roche is one of the world’s leading
research-focused healthcare groups in the fields of pharmaceuticals and
diagnostics. As a supplier of innovative products and services for the early
detection, prevention, diagnosis and treatment of disease, the Group
contributes on a broad range of fronts to improving people’s health and quality
of life. Roche is a world leader in diagnostics, the leading supplier of
medicines for cancer and transplantation and a market leader in virology. In
2004 sales by the Pharmaceuticals Division totalled 21.7 billion Swiss francs,
while the Diagnostics Division posted sales of 7.8 billion Swiss francs. Roche
employs roughly 65,000 people in 150 countries and has R&D agreements and
strategic alliances with numerous partners, including majority ownership
interests in Genentech and Chugai. Additional information about the Roche Group
is available on the Internet (www.roche.com).
Disclaimer
This press release includes forward-looking statements, as defined in the U.S.
Private Securities Litigation Reform Act of 1995. They have based these
forward-looking statements on their current expectations and projections about
future events. Such statements involve known and unknown risks, uncertainties
and other factors that may cause actual results to differ materially. Such
factors include, but are not limited to, changes in local and global economic
conditions, their ability to successfully implement their strategy, the market
acceptance of and demand for their products, their growth and expansion,
technological change and their exposure to market risks. By their nature, these
expectations and projections are only estimates and could be materially
different from actual results in the future.
Hyderabad, India, October 29, 2005
Dr. Reddy’s reports Q2 FY06
revenue of Rs. 5,803 million; YoY growth of 7%
Net income increases by 72% to Rs. 890 million
Hyderabad,
India, October 29, 2005: Dr. Reddy’s Laboratories Ltd. (NYSE: RDY) today
announced its unaudited financial results for the second quarter ended
September 30, 2005.
Key highlights
- Revenues at Rs 5.8 billion, a growth of 7% over Q2
FY05. Highest revenue base in the previous sixteen quarters.
- Net profit increases by 72% to Rs 890 million. This
translates to a diluted EPS of Rs 11.61 as against Rs 6.75 in Q2 FY05.
- Overall revenue growth driven by increase in revenues
from all businesses excluding North America Generics
- Revenues from Active Pharmaceutical Ingredients (API)
business increase by 17% to Rs 2,130 million. This increase was driven by
the 22% growth in international segment led by higher sales from
sertraline & terbinafine.
- Revenues from Branded Formulations business increase by
11% to Rs 2,576 million. Revenues in India increase by 12% to Rs 1,507
million. Revenues in International segment increase by 10% to Rs 1,069
million, driven by 39% growth in Other CIS markets.
- Revenues from Europe generics finished dosage segment
increase by 46% to Rs 473 million led by higher price realizations
for key products of omeprazole and amlodipine maleate as well as the
launch of terbinafine.
- The pricing pressure in the U.S. Generics market
continued in the second quarter. As a result of the intense competition,
revenues from fluoxetine and tizanidine declined sharply leading to
an overall decline in revenues from North America generics segment
to Rs 299 million in Q2 FY06 from Rs 715 million in Q2 FY05.
- Revenues in the high margin Custom Pharmaceutical
Services business increase by 24% to Rs 121 million in Q2 FY06.
INCOME STATEMENT FOR THE QUARTER ENDED SEPTEMBER 30, 2005
All figures in millions, except EPS
All dollar figures based on convenience translation rate of 1USD = Rs 43.94
EXTRACT FROM THE UNAUDITED
INCOME STATEMENT
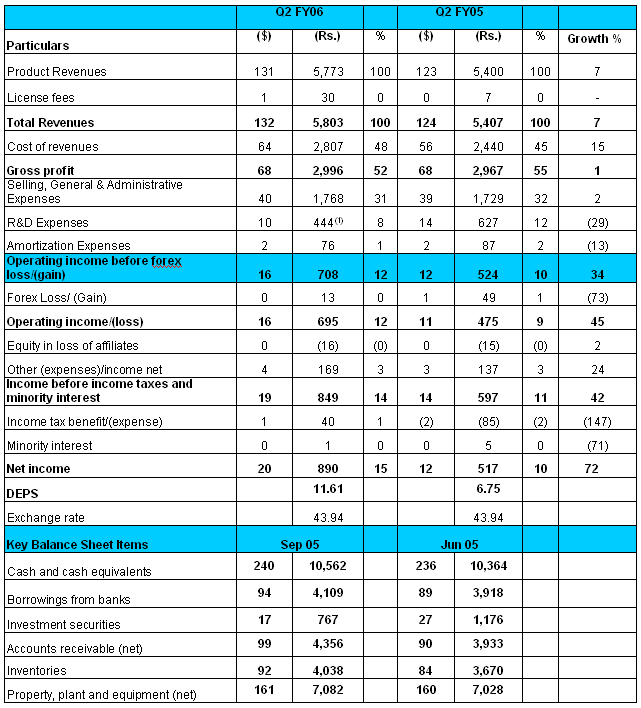
(1) Reduction of Rs 155 million
under the Generics R&D partnership deal with ICICI Venture
Active
Pharmaceutical Ingredients (API)
·
Revenues increase by 17% to Rs 2,130 million as against Rs
1,821 million in Q2 FY05.
·
Revenues in India increase by 5% to Rs 578 million as
against Rs 553 million in Q2 FY05.
·
Revenues outside India increase by 22% to Rs 1.6 billion.
Contributes 73% to overall segment revenues as against 70% in Q2 FY05.
·
Sales from Europe increase by 55% to Rs 338 million. This
growth was primarily driven by launch of terbinafine contributing to sales of
Rs 65 million.
·
Sales from North America decline by 6% to Rs 490 million
from Rs 523 million in Q2 FY05. This decrease was on account of decline in
sales of key products offset by higher sales of sertraline.
·
Sales from other international markets (excluding North
America and Europe) increase by 37% to Rs 724 million. This increase was driven
by higher sales of sertraline and terbinafine partially offset by decrease in
sales of amlodipine maleate.
·
The Company filed 1 US DMF during the quarter taking the
total filings to 78. In addition to this, the Company filed 2 Europe DMF’s and
4 Canada PMF’s
Generic Finished Dosages
·
Revenues in this segment at Rs 773 million as against Rs
1,043 million in Q2 FY05.
·
Europe contributed 61% to the total revenues and North
America contributed the balance 39%.
·
Revenues in Europe increase by 46% to Rs 473 million as
against Rs 324 million in Q2 FY05. The growth was primarily driven by higher
price realizations for omeprazole and amlodipine maleate as well as launch of
terbinafine. Combined revenues from omeprazole and amlodipine maleate at Rs 295
million as against Rs 148 million in Q2 FY05. Launch sales of terbinafine at Rs
65 million.
·
Revenues in North America at Rs 299 million as against Rs
715 million in Q2 FY05. On account of the intense competition, combined
revenues from fluoxetine capsules and tizanidine tablets declined to Rs 93
million from Rs 436 million in Q2 FY05. Products launched after September 2004
contributed revenues of Rs 34 million.
·
During the quarter, the Company filed 5 ANDAs. The Company
also received approval for two ANDAs during the quarter. This takes the total
ANDAs pending at the USFDA to 50.
Branded Finished Dosages -
International
·
Revenues at Rs 1.1 billion, an increase of 10% over Q2 FY05.
This increase was primarily driven by growth in Russia & CIS markets.
·
Revenues in other CIS markets increase by 39% to Rs 203
million as against Rs 146 million in Q2 FY05. This growth was primarily driven
by Ukraine and Kazakhstan.
·
Revenues in Russia increase by 10% to Rs 644 million as
against Rs 587 million in Q2 FY05. This growth was driven by the performance of
key brands of Nise, Ketorol and Omez.
·
Revenues in Central Eastern Europe increase by 19% to Rs 51
million as against Rs 43 million in Q2 FY05. This increase was mainly on
account of growth in Romania partially offset by decline in revenues from
Albania.
·
Revenues in other international markets decrease by 12% to
Rs 171 million as against Rs 195 million in Q2 FY05. This was primarily on
account of decrease in sales from Venezuela and United Arab Emirates.
Branded Finished Dosages-
India
·
Revenues at Rs 1.5 billion, an increase of 12% over Q2 FY05.
·
This growth was led by the performance of key brands of
Stamlo Beta, Atocor and Razo as well as revenues from new products launched
during the quarter.
·
During the quarter, the Company launched eight new products
contributing revenues of Rs 12 million.
Other Businesses
·
Revenues from Custom Pharmaceutical Services business
increase by 24% to Rs 121 million from Rs 98 million in Q2 FY05.
·
Revenues in the Oncology segment increase by 56% to Rs 203
million.
Income Statement Highlights
·
Gross profit margins for Q2 FY06 at 52% are comparable with
the gross profit margins in Q1 FY06. However, compared to Q2 FY05, gross profit
margins on total revenues at 52% as against 55% in Q2 FY05. This decline in
gross margins is on account of decrease in contribution of revenues from North
America Generics to the total revenues.
·
Investments in R&D at 8% of total revenues as against
12% in Q2 FY05. R&D investments for Q2 FY06 decrease by 29% to Rs 444
million from Rs 627 million in Q2 FY05. During the quarter, the Company
recognized Rs 155 million as income under the R&D partnership deal with
ICICI Venture. Excluding this benefit, R&D investments decreased by Rs 28
million. This decrease is on account of lower bio-study expenses in Generics.
·
Selling, General & Administration (SG&A) expenses
increased by 2% to Rs 1,768 million. As a % to revenues, SG&A expenses are
at 30% of total revenues as against 32% in Q2 FY05.
·
Other income (net) increased to Rs 169 million from Rs 137
million in Q2 FY05. This includes net interest income of Rs 140 million.
·
Depreciation for the quarter was at Rs 279 million as
against Rs 238 million for Q2 FY05.
·
Net income at Rs 890 million (15% of total revenues) as
against Rs 517 million (10% of total revenues) in Q2 FY05. This translates to a
diluted EPS of Rs 11.61 as against Rs 6.75 in Q2 FY05.
About Dr. Reddy’s
Established in 1984, Dr. Reddy's
Laboratories (NYSE: RDY) is an emerging global pharmaceutical company with
proven research capabilities. The Company is vertically integrated with a
presence across the pharmaceutical value chain. It produces finished dosage
forms, active pharmaceutical ingredients and biotechnology products and markets
them globally, with focus on India, US, Europe and Russia. The Company conducts
research in the areas of diabetes, cardiovascular, anti-infectives,
inflammation and cancer.
Disclaimer
This press release includes
forward-looking statements, as defined in the U.S. Private Securities
Litigation Reform Act of 1995. They have based these forward-looking statements
on their current expectations and projections about future events. Such
statements involve known and unknown risks, uncertainties and other factors
that may cause actual results to differ materially. Such factors include, but
are not limited to, changes in local and global economic conditions, their
ability to successfully implement their strategy, the market acceptance of and
demand for their products, their growth and expansion, technological change and
their exposure to market risks. By their nature, these expectations and
projections are only estimates and could be materially different from actual
results in the future.
Contact Information
Investors and Financial
Analysts:
Nikhil Shah at nikhilshah@drreddys.com
or on +91-40-55511532
Media
M Mythili at mythilim@drreddys.com
or on +91-40-55511620
Notes
1. In line with global disclosure standards, the company commenced reporting
its financials on a consolidated basis since Q2
FY03.
2. Current quarter financial discussions below are on a consolidated basis as
per the US GAAP.
3. Detailed analysis of the financials is available on the Company’s website at
www.drreddys.com
CMT REPORT
[Corruption, Money laundering & Terrorism]
The Public Notice information has been collected from
various sources including but not limited to: The Courts, India Prisons
Service, Interpol, etc.
1] INFORMATION ON DESIGNATED PARTY
No
exist designating subject or any of its beneficial owners, controlling
shareholders or senior officers as terrorist or terrorist organization or whom
notice had been received that all financial transactions involving their assets
have been blocked or convicted, found guilty or against whom a judgement or
order had been entered in a proceedings for violating money-laundering,
anti-corruption or bribery or international economic or anti-terrorism sanction
laws or whose assets were seized, blocked, frozen or ordered forfeited for
violation of money laundering or international anti-terrorism laws.
2] Court Declaration :
No records exist to suggest that
subject is or was the subject of any formal or informal allegations,
prosecutions or other official proceeding for making any prohibited payments or
other improper payments to government officials for engaging in prohibited
transactions or with designated parties.
3] Asset Declaration :
No
records exist to suggest that the property or assets of the subject are derived
from criminal conduct or a prohibited transaction.
4] Record on Financial Crime :
Charges or
conviction registered against subject: None
5] Records on Violation of
Anti-Corruption Laws :
Charges or
investigation registered against subject: None
6] Records on Int’l Anti-Money
Laundering Laws/Standards :
Charges or
investigation registered against subject: None
7] Criminal Records
No available
information exist that suggest that subject or any of its principals have been
formally charged or convicted by a competent governmental authority for any
financial crime or under any formal investigation by a competent government
authority for any violation of anti-corruption laws or international anti-money
laundering laws or standard.
8] Affiliation with Government :
No record exists to
suggest that any director or indirect owners, controlling shareholders,
director, officer or employee of the company is a government official or a
family member or close business associate of a Government official.
9] Compensation Package :
Our market survey
revealed that the amount of compensation sought by the subject is fair and
reasonable and comparable to compensation paid to others for similar services.
10] Press Report
:
No press reports / filings exists on the subject.
CORPORATE GOVERNANCE
MIRA INFORM as part of its Due Diligence do provide comments
on Corporate Governance to identify management and governance. These factors
often have been predictive and in some cases have created vulnerabilities to
credit deterioration.
Our Governance Assessment focuses principally on the
interactions between a company’s management, its Board of Directors,
Shareholders and other financial stakeholders.
CONTRAVENTION
Subject is not known to have contravened any existing local
laws, regulations or policies that prohibit, restrict or otherwise affect the
terms and conditions that could be included in the agreement with the subject.
FOREIGN EXCHANGE RATES
|
Currency |
Unit
|
Indian Rupees |
|
US Dollar |
1 |
Rs. 45.92 |
|
UK Pound |
1 |
Rs. 84.76 |
|
Euro |
1 |
Rs. 57.74 |
SCORE & RATING EXPLANATIONS
|
SCORE FACTORS |
RANGE |
POINTS |
|
HISTORY |
1~10 |
7 |
|
PAID-UP
CAPITAL |
1~10 |
7 |
|
OPERATING
SCALE |
1~10 |
6 |
|
FINANCIAL
CONDITION |
|
|
|
--BUSINESS
SCALE |
1~10 |
6 |
|
--PROFITABILIRY |
1~10 |
6 |
|
--LIQUIDITY |
1~10 |
7 |
|
--LEVERAGE |
1~10 |
6 |
|
--RESERVES |
1~10 |
8 |
|
--CREDIT
LINES |
1~10 |
7 |
|
--MARGINS |
-5~5 |
- |
|
DEMERIT
POINTS |
|
|
|
--BANK
CHARGES |
YES/NO |
YES |
|
--LITIGATION |
YES/NO |
NO |
|
--OTHER
ADVERSE INFORMATION |
YES/NO |
NO |
|
MERIT
POINTS |
|
|
|
--SOLE
DISTRIBUTORSHIP |
YES/NO |
NO |
|
--EXPORT ACTIVITIES |
YES/NO |
YES |
|
--AFFILIATION |
YES/NO |
YES |
|
--LISTED |
YES/NO |
YES |
|
--OTHER
MERIT FACTORS |
YES/NO |
YES |
|
TOTAL |
|
60 |
This
score serves as a reference to assess SC’s credit risk and to set the amount of
credit to be extended. It is calculated from a composite of weighted scores
obtained from each of the major sections of this report. The assessed factors
and their relative weights (as indicated through %) are as follows:
Financial
condition (40%) Ownership
background (20%) Payment
record (10%)
Credit
history (10%) Market
trend (10%) Operational
size (10%)
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
>86 |
Aaa |
Possesses an extremely sound
financial base with the strongest capability for timely payment of interest
and principal sums |
Unlimited |
|
71-85 |
Aa |
Possesses adequate working
capital. No caution needed for credit transaction. It has above average
(strong) capability for payment of interest and principal sums |
Large |
|
56-70 |
A |
Financial & operational base
are regarded healthy. General unfavourable factors will not cause fatal
effect. Satisfactory capability for payment of interest and principal sums |
Fairly Large |
|
41-55 |
Ba |
Overall operation is considered
normal. Capable to meet normal commitments. |
Satisfactory |
|
26-40 |
B |
Unfavourable & favourable
factors carry similar weight in credit consideration. Capability to overcome
financial difficulties seems comparatively below average/normal. |
Small |
|
11-25 |
Ca |
Adverse factors are apparent.
Repayment of interest and principal sums in default or expected to be in
default upon maturity |
Limited with full security |
|
<10 |
C |
Absolute credit risk exists.
Caution needed to be exercised |
Credit not recommended |