
MIRA INFORM REPORT
|
Report Date : |
25th May, 2006 |
IDENTIFICATION DETAILS
|
Name : |
KLENZAIDS
CONTAMINATION CONTROLS PRIVATE LIMITED |
|
|
|
|
Registered Office : |
A-21-22,
MIDC Industrial Area, Street No. 3, Andheri (East), Mumbai-400093,
Maharashtra, India |
|
|
|
|
Country : |
India
|
|
|
|
|
Financials (as on) : |
31.03.2005 |
|
|
|
|
Date of Incorporation : |
1st
February, 1978 |
|
|
|
|
Com. Reg. No.: |
11-20112 |
|
|
|
|
CIN No.: [Company Identification No.] |
U33112MH1978PTC020112 |
|
|
|
|
IEC No.: |
0388050594 |
|
|
|
|
Legal Form : |
Private
Limited Liability Company |
|
|
|
|
Line of Business : |
Manufacturer
of and Dealer in Clean Room Equipments and Accessories, Surgical Equipments
and Facilities for parenterals including L.V.P., Ophthalmic ointments,
surgical suits, patient houses ad other equipments for turn-key sterile
facilities, laminar flow work stations, tailor made, to suit individual
application, needs, air showers, air barriers for insect contract, in line
modules and laminar air flow. |
RATING & COMMENTS
|
MIRA’s Rating : |
A |
RATING
|
STATUS |
PROPOSED CREDIT LINE |
|
|
56-70 |
A |
Financial
& operational base are regarded healthy. General unfavourable factors
will not cause fatal effect. Satisfactory capability for payment of interest
and principal sums |
Fairly Large |
|
Maximum Credit Limit : |
USD 175000 |
|
|
|
|
Status : |
Good |
|
|
|
|
Payment Behaviour : |
Regular |
|
|
|
|
Litigation : |
Clear |
|
|
|
|
Comments : |
Subject
is a well – established and reputed concern having fine track. Financial
position is good. Trade relations are fair. Payments are correct and as per
commitments. The
concern can be considered good for normal business dealings at usual trade
terms and conditions. |
LOCATIONS
|
Registered Office : |
A-21-22,
MIDC Industrial Area, Street No. 3, Andheri (East), Mumbai-400093,
Maharashtra, India |
|
Tel. No.: |
91-22-28218921 |
|
Fax No.: |
91-22-28377837/927 |
|
E-Mail : |
|
|
Website: |
|
|
|
|
|
Factory 1 : |
218,
GIDC Industrial Area, Ombergaon, District – Valsad |
|
Tel. No.: |
91-260-2562082 |
|
Fax No.: |
91-260-2563082 |
|
Area : |
30000
Sq. ft |
|
Location : |
Leased
|
|
|
|
|
Factory 2 : |
1/1,
½, 3/1, GIDC Industrial Area, District – Valsad |
|
Tel. No.: |
91-260-2562862 |
|
Area : |
2000
Sq. ft |
|
Location : |
Leased
|
DIRECTORS
|
Name : |
Mr.
Prabhakar V. Shetty |
|
Designation : |
Director
|
|
Address : |
Plot
No. 1, 4D, 1st Floor, R.D. Muljee Nagar, S.V. Road, Borivali
(West), Mumbai-400092, Maharashtra, India |
|
Date of Appointment : |
1st
May, 2003 |
|
|
|
|
Name : |
Mr.
Krishnamurthy H. |
|
Designation : |
Director
|
|
Address : |
D
– 7, Eavour, Goft Road |
|
Date of Birth : |
8th
September, 1953 |
|
Qualification : |
B.
Com |
|
Experience : |
28
Years |
|
Date of Appointment : |
1st
December, 1983 |
|
|
|
|
Name : |
Mrs.
Neena Y. Mathur |
|
Designation : |
Director
|
|
Address : |
Mumbai-400026,
Maharashtra, India |
|
Date of Appointment : |
30th
November, 1985 |
|
Date of Leasing : |
30th
November, 1990 |
|
|
|
|
Name : |
Mr.
C. R. Shah |
|
Designation : |
Director
|
|
Address : |
Prayag
C 15, Ayojananagar, New Sharda Mandir Road, Ahmedabad-380007, Gujarat, India |
|
Date of Appointment : |
5th
March, 1992 |
|
|
|
|
Name : |
Mr.
Hariprasad K. Iyer |
|
Designation : |
Director
|
|
Address : |
14,
Suphala Apartment, Bailbazar Road, Gandhi Nagar, Kalyan (West), Thane-421301 |
|
Date of Appointment : |
19th
June, 1992s |
|
|
|
|
Name : |
Mr.
Chandra M Sahani |
|
Designation : |
Director
|
|
Address : |
28,
Silver Sadan, Juhu Tara Road, Mumbai – 49 |
|
Date of Birth : |
70
Years |
|
Qualification : |
B.
E. |
|
Experience : |
46
Years |
|
|
|
|
Name : |
Mr.
Himesh C Sahani |
|
Designation : |
Director
|
|
Address : |
28,
Silver Sadan, Juhu Tara Road, Mumbai – 49 |
|
Date of Birth : |
38
Years |
|
Qualification : |
B.
E. |
|
Experience : |
15
Years |
|
|
|
|
Name : |
Mr.
Ramesh P Lala |
|
Designation : |
Director
|
|
Address : |
2,
Oiha Niwas, 5 Sector, Juhu Tara Road, Mumbai – 49 |
|
Date of Birth : |
54
Years |
|
Qualification : |
B.
Tech |
|
Experience : |
30
Years |
MAJOR SHAREHOLDERS
|
Names of Shareholders |
No. of Shares |
|
Mona C Sahani |
2251 |
|
Manish C Sahani |
4918 |
|
Mr. H P Sahani |
4980 |
|
M/s. Hamish Inv. Private
Limited |
12150 |
|
Otimfm |
56 |
BUSINESS DETAILS
|
Line of Business : |
Manufacturer
of and Dealer in Clean Room Equipments and Accessories, Surgical Equipments
and Facilities for parenterals including L.V.P., Ophthalmic ointments,
surgical suits, patient houses ad other equipments for turn-key sterile
facilities, laminar flow work stations, tailor made, to suit individual
application, needs, air showers, air barriers for insect contract, in line
modules and laminar air flow. |
|
|
|
|
Products : |
Clean
Room Equipments and Pharmaceutical Machineries, Filter Paper and Machinery
Components |
|
|
|
|
Exports to : |
Germany,
Developing Countries |
|
|
|
|
Imports from : |
Italy,
France and Germany |
|
|
|
|
Terms : |
|
|
Selling : |
Cash
& Credit (30 days) |
|
|
|
|
Purchasing : |
Cash
& credit (60 to 90 days) |
PRODUCTION STATUS
|
Particulars |
Unit |
Actual Production |
|
Clean Tech Machineries |
Sq mtrs |
819 |
|
Pharmaceuticals Machineries |
- |
7 |
|
Accessories |
-- |
107 |
GENERAL INFORMATION
|
Customers : |
End
Users |
|
|
|
|
No. of Employees : |
134 |
|
|
|
|
Bankers : |
v
Canara Bank, Mumbai v
Indusind Bank Limited,
Andheri (East), Mumbai |
|
|
|
|
Facilities : |
- |
|
|
|
|
Banking Relations : |
Good
|
|
|
|
|
Auditors : |
Natwarlal
Vepari and Company Chartered
Accountant |
|
Tel. No.: |
91-22-22073936/56315851 |
|
Fax. No.: |
91-22-56315852 |
|
Address: |
Jiji
House, 4th Floor, 15, Ravehine St., Fort, Mumbai 400001, India |
CAPITAL STRUCTURE
Authorised Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
30000 |
Equity
Shares |
Rs. 100 each |
Rs. 3.000 millions |
Issued, Subscribed
& Paid-up Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
24800 |
Equity
Shares |
Rs. 100 each |
Rs. 2.480 millions |
FINANCIAL
DATA
[all figures are in Rupees Millions]
ABRIDGED
BALANCE SHEET
|
SOURCES OF FUNDS |
|
31.03.2005 |
31.03.2004 |
|
|
SHAREHOLDERS FUNDS |
|
|
|
|
|
1] Share Capital |
|
2.480 |
2.480 |
|
|
2] Share Application Money |
|
0.000 |
0.000 |
|
|
3] Reserves & Surplus |
|
43.358 |
39.518 |
|
|
4] (Accumulated Losses) |
|
0.000 |
0.000 |
|
NETWORTH
|
|
45.838 |
41.998 |
|
|
LOAN FUNDS |
|
|
|
|
|
1] Secured Loans |
|
0.037 |
0.037 |
|
|
2] Unsecured Loans |
|
0.095 |
0.388 |
|
TOTAL
BORROWING
|
|
0.132 |
0.425 |
|
|
DEFERRED TAX LIABILITIES |
|
0.437 |
0.506 |
|
|
|
|
|
|
|
TOTAL
|
|
46.407 |
42.929 |
|
|
|
|
|
|
|
APPLICATION OF FUNDS
|
|
|
|
|
|
|
|
|
|
|
FIXED ASSETS [Net Block]
|
|
4.338 |
4.727 |
|
Capital work-in-progress
|
|
0.000 |
0.000 |
|
|
|
|
|
|
|
INVESTMENT
|
|
0.000 |
0.000 |
|
DEFERREX TAX ASSETS
|
|
0.000 |
0.000 |
|
|
|
|
|
|
|
CURRENT ASSETS, LOANS & ADVANCES
|
|
|
|
|
|
|
Inventories
|
|
11.800 |
66.674 |
|
|
Sundry Debtors
|
|
12.583 |
14.207 |
|
|
Cash & Bank Balances
|
|
6.818 |
15.186 |
|
|
Other Current Assets
|
|
13.762 |
13.761 |
|
|
Loans & Advances
|
|
85.672 |
94.318 |
Total Current Assets
|
|
130.635 |
204.146 |
|
Less : CURRENT LIABILITIES & PROVISIONS
|
|
|
|
|
|
|
Current Liabilities
|
|
79.312 |
158.990 |
|
|
Provisions
|
|
9.254 |
6.954 |
Total Current Liabilities
|
|
88.566 |
165.944 |
|
Net Current
Assets
|
|
42.069 |
38.202 |
|
|
|
|
|
|
|
MISCELLANEOUS EXPENSES
|
|
0.000 |
0.000 |
|
|
|
|
|
|
|
TOTAL
|
|
46.407 |
42.929 |
|
PROFIT & LOSS ACCOUNT
|
PARTICULARS |
|
31.03.2005 |
31.03.2004 |
Sales Turnover [including other income]
|
|
135.246 |
78.537 |
|
|
|
|
|
Profit/(Loss) Before Tax
|
|
6.086 |
1.410 |
Provision for Taxation
|
|
2.232 |
1.144 |
Profit/(Loss) After Tax
|
|
3.854 |
0.266 |
|
|
|
|
|
|
Export
Value |
|
6.262 |
71.857 |
|
|
|
|
|
|
Import
Value |
|
3.016 |
29.408 |
|
|
|
|
|
|
Total
Expenditure |
|
129.159 |
77.127 |
KEY RATIOS
|
PARTICULARS |
|
|
31.03.2005 |
31.03.2004 |
|
PAT / Total Income |
(%) |
|
2.84 |
0.33 |
|
|
|
|
|
|
|
Net
Profit Margin (PBT/Sales) |
(%) |
|
4.49 |
1.79 |
|
|
|
|
|
|
|
Return
on Total Assets (PBT/Total
Assets} |
(%) |
|
4.50 |
0.67 |
|
|
|
|
|
|
|
Return
on Investment (ROI) (PBT/Networth) |
|
|
0.13 |
0.02 |
|
|
|
|
|
|
|
Debt
Equity Ratio (Total
Liability/Networth) |
|
|
1.93 |
3.96 |
|
|
|
|
|
|
|
Current
Ratio (Current
Asset/Current Liability) |
|
|
1.47 |
1.23 |
LOCAL AGENCY FURTHER INFORMATION
The companies fixed assets of important value includes-
Factory Building, Staff Quarters, Motor Car,
Plant and Machinery, Furniture and Fixtures, Computers, and Office Equipments.
ABOUT US
Klenzaids Contamination
Controls
Today
Klenzaids due to its relentless efforts
and sustained investments in technology is recognized as world's pioneer in
MicroFlora Management and Contamination Control of enclosed spaces. Dedication
to that niche has made us major players in diverse industrial sectors
like Pharmaceuticals, Hospitals, Biolabs, Aerospace, Electronics.
Eliminating
Contamination is our DNA.
Klenzaids
has also been working in the
field of Aseptic Technologies providing engineering services that minimize and
control contamination from vectors like Biota, Personnel, Process, Room Shell,
Material and Clean-down.
Klenzaids
has broken through the
barrier from Contamination Control to Total Containment making advances in
Barrier Technologies.
Over
35 years - quality is our brand equity, reinforced by our day-to-day working
with ISO - we are periodically audited by TUV and are proud to
be an ISO 9001 : 2000 certified Company.
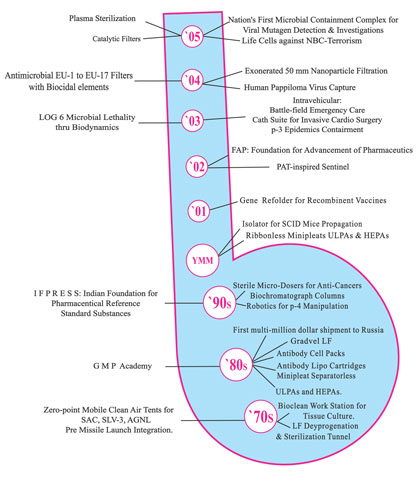
INNOVATIONS
Klenzaids Contamination Controls
LA
NOTE
EXACTEMENT
MANDATE
Klenzaids Contamination
Controls
![]()
To pioneer new particulate immobilizing
technologies in nanometer regime.
![]()
Make fair & equitable profit.
![]()
Provide sharpest cutting-edge creative
solutions.
![]()
Carry on constant research.
![]()
To satisfy our customers so completely
that they want to work with us as individuals and as a Company because they are
convinced that in us they have found the best partners.
![]()
Upgrade equipment and facilities.
![]()
To be responsible to our employees.
![]()
Experiment with new ideas and develop
innovative programmes and products.
![]()
Respect their dignity and recognize their
merit.
![]()
To be committed to the environment we
live in. Keep it pristine clean and ever green.
![]()
Give equal opportunity for development
and advancement.
![]()
We abide to our Quality Policy.
![]()
To provide competent, just and ethical
management.
![]()
In
no other industry is Quality as parametrically defined as in the pharmaceutical
industry. Regulations, Standards, limits-constrict manufacturing latitudes.
In
response to the regulatory compliance needs of industry, the Klenzaids GMP Academy was instituted.
The
aim of the Academy is to promote an understanding of cGMP at fundamental
levels, while divesting them of their coercive connotation.
Through
multi-pronged efforts (publications, colloquiums, learning modules and
residential focus workshops) the Academy aims to create an informed Health-Care
Industry, capable of initiating proactive responses to quality related issues.
Gene
Technology has gained momentum. It has to demonstrate higher levels of quality
consciousness than mainstream medicine manufacture. To make that happen,
preliminary guidelines have already been drafted. The Klenzaids GMP Academy will extend its
efforts to make cGMP integral to Biotechnology Derived Products.
Microbiological Evaluation
Aseptic Processing Areas
Micro Monograph # 01
Pharma-industry
in USA and EU have objected to the Regulatory Agencies insistence on
particle-counts to establish CLEANLINESS of aseptic processing areas.
Microbial
monitoring is only one limb of information needed to build a meaningful
environmental surveillance and control program. There has been much discussion
as to whether the standards should include numbers. i.e. limits or levels. At
present there exists no scientific rationale or standard methodologies upon
which to base such quantification. Each manufacturer should be responsible for
establishing its own microbial levels based on historical data, individual
facility operations and specific product considerations.
Bacterial De-contamination
Stainless Steel Equipment
Micro Monograph # 02
Stainless Steel is among the most widely used
materials in pharmaceutical equipment construction. The amount of Stainless
Steel utilized in this field has been increasing steadily. A major problem in
the use of stainless steel is the efficient removal of bacterial cells.
Bacterial adhesion and colonization on stainless steel is well known, but as
yet unclear phenomenon in the pharmaceutical industry. In this micro monograph,
general mechanisms of bacterial adhesion to stainless steel, and disinfection
of contaminated surfaces are described.
Pharmaceutical Facilities:
Integrated
Planning &Construction
Micro
Monograph # 03
Most pharmaceutical projects lack
integrated approaches. Although work places are increasingly becoming
networked, the effects are additive, parallel and at times even negative.
Generic—ready to use—solutions for pharmaceutical production buildings do not
and cannot exist. User-requirements, with reference to product qualities and
quantities, production-processes and boundary-conditions can vary greatly.
Templates for pharmaceutical plant projects cannot be taken off-the-shelf.
cGMP advocates the integrated planning
approach to pharmaceutical projects that yield interactive results. The essence
of that approach is making the process determine the space elements of the
facility, enabling high levels of flexibility, rather than cumbersome
infrastructure dictating how processes should be run.
HVAC Systems Validation
Micro Monograph # 04
VAC systems are an integral
component of a pharmaceutical facility's functionality and impacts on the
safety of scientists and technicians working in a lab or production facility,
the integrity of processes, and the environment outside. An important element
of successful HVAC validation is prevalidation design work. In this
micro-monograph, we explore the correlation between preliminary design and each
phase of validation.
Crossflow Filteration
Micro
Monograph # 05
Crossflow filtration is often the primary
process used for purification of biotechnology derived products. Crossflow
filtration is distinguished from conventional "dead-end" filtration,
in that the fluid to be filtered, flows perpendicular to the filtrate stream,
rather than parallel to the filtrate or permeate stream. The concept of
concentration polarization is examined as it affects filter performance,
retentivity and flux.
Air Filteration
Micro Monograph # 06
Efficiency, airflow Resistance and Arrestance
capability are primary criteria that differentiate diverse types of extended
surface aerosol filters. Measurable performance— meeting application needs— is
complicated by the many Standards defining panel filter classifications which
do provide means for comparing and ranking filters, but do not provide adequate
information needed by filtration system designers who have to meet specific
requirements; and, who also want to minimize system costs and energy use .
This micro-monograph correlates the unified EURO
Standard EN 779, to the American Standards ; and, reviews them historically. It
also distinguishes EN 1822, which covers the DIN Schwebstoff — HEPA and ULPA —
range from the IEST Institute of Environmental Science Technology Recommended
Practice 001 to 008.
Bioanalytical
Method Validation
Micro Monograph # 07
This micro-monograph represents the US Food and Drug
Administration's current thinking on this topic. An alternative approach may be
used if such approach satisfies the requirements of the applicable statutes and
regulations. The intent is to provide assistance to sponsors of investigational
new drug applications —INDs — new drug applications — NDAs — abbreviated new
drug applications — ANDAs — and supplements in developing bioanalytical method
validation information used in human clinical pharmacology, bioavailability —
BA — and bioequivalence — BE — studies requiring pharmacokinetic — PK —
evaluation. This guidance also applies to bioanalytical methods used for non-human
pharmacology and toxicology studies and preclinical studies. For studies
related to the veterinary drug approval process, this guidance applies only to
blood and urine BA, BE, and PK studies.
P-4 Biocontainment Facilities
Micro Monograph # 08
In aseptic and clean and hypobaric containment
facilities, the envelope and environment control directly affect egress
reliability. Safety and effectiveness cannot be tested or inspected into such a
facility as an afterthought.
The modular approach is contra-indicated. In a true
modular system; walls; ceilings; return air chases; utility access chases; and
even the heating; ventilating ; and air-conditioning system — HVAC — are
standardized components with clearly defined functions.
Hazop
Management
Micro Monograph # 09
The meaning of hazard is often confused with risk.
Hazard is defined as the inherent potential of a substance and activity to harm
people or the environment. Hazard does not have a probability component.
Hazop Analysis is a qualitative tool amongst a
sub-group that is covered by this narrative. Defining quantitative elements
that are pre-requisites for Hazop qualification of Klenzaids Isolators are also
presented.
Vaporized Hydrogen Peroxide
Micro Monograph # 10
As simple as it may seem, the treatment of
contaminated elements is as diverse and complicated as the operations from
which it comes. In today's environment, where merely transferring contaminants
from one medium to another is no longer acceptable, it is no surprise that a
powerful oxidizer that looks like water — in its appearance, chemical formula
and reaction products — should be so widely used. This is hydrogen peroxide —
H2O2 — a powerful yet versatile oxidant that is both safe and effective.
Instrument Calibration &
Certification
Micro
Monograph # 11
It is generally presumed that most instruments used
in the pharma industry, in both formulation and bulk sectors, function as
intended. That perception alone is not enough.
Proof of performance by way of calibration and
certification is GMP-mandated. That proof is derived from reference
measurements and calibration. The precision with which these results of
measurements are simulated, must be documented and witnessed by the end-user;
and, finally certified in accordance with International Standards; the more
important ones being traceable to ANSI — American National Standards Institute
— and ISO Guide 25 and 58.
The documentation of processes for the calibration of
instruments, making up the analytical and production equipment, is vital to the
Quality Systems that spell out compliance to cGMP. A series on instrument
calibration and certification is being brought to you in this micro-monograph.
The factors to be considered in instrument selection
within the context of calibration and certification are described.
Particulate Cleanliness Evaluation
Micro Monograph # 12
Importance of non-viable airborne particles is
introduced in this micro-monograph. This monograph intends to deal with the
measurement and analysis of particles in parenterals as they relate to
regulatory conformance. Particle Counters are, therefore, described here in
some detail.
Cleaning Validation
Mini Monograph # 01
A significant portion of this
Monograph is derived from round-table discussions on Cleaning Validation held
during various validation courses conducted by the PDA. The summary is
organized around the following series of questions:
What is being cleaned ?
When must cleaning validation be performed?
Which physical parameters must be evaluated?
How clean is clean? And many more!
cGMP
Validation of Aseptic
Pharmaceutical Facilities
Mini Monograph # 02
Since cGMP validation is a framework designed to
establish total control over process outcome, the documentation needed to
provide evidence that this has been achieved will vary with the specific
program. Depending on how and when the data is generated and utilized,
validation is said to be prospective, retrospective or concurrent. Documentary
requirements for prospective and retrospective validation — as currently
employed internationally in aseptic pharmaceutical production — are described
in this mini-monograph. By definition, process validation requires the
accumulation of documentary evidence that a specific process will constantly
produce a product meeting its predetermined specifications and quality
characteristics. Unfortunately, there is still much confusion as to what
constitutes process validation documentation.
Water in the Pharmaceutical Industry
Mini Monograph # 03
Water is a singular substance possessing unusual
properties that renders it extraordinarily useful as a drug vehicle. Water is
used in the Pharmaceutical Industry for Process and for Energies. In this
presentation the current processes used in the production of pharmaceutical
waters, are evaluated, regulatory limits operational for various categories are
presented and present technological trends that are becoming current GMP in the
production processes are described.
Pharmaceutical Clean Air Systems
Mini
Monograph # 04
Clean Air Systems are an integral aspect
of every pharmaceutical facility. In this mini-monograph the focus is on
mechanisms of air filtration and filtration qualification. The first part
describes deposition and adsorption mechanisms of particulate and vapor
contaminants.
Freeze
Drying of Sterile Products
Mini Monograph # 05
In this mini-monograph, we discuss the basic
principles of lyophilization along with guidelines for process validation and
sterilization. This freeze dry process is commonly referred to as
lyophilization from (Greek made solvent loving). Lyophilization is a means of
drying, achieved by freezing the wet substance and causing the ice to sublime
directly to vapour by exposing it to a low partial pressure of water vapor. At
these low pressures, water in the form of ice, can be turned directly into
water vapor without first becoming liquid, by adding heat energy.
cGMP for Pharma Premises
and Materials
Mini Monograph # 06
A decision to develop, manufacture, and or market a
medical device requires a basic understanding of the responsibility of the Food
and Drug Administration. In particular, individuals who are responsible for the
manufacture of devices, whether as part of a production or quality control
function, must recognize their potential exposure. This section is intended to
describe basic provisions. The general operation of the Food and Drug
Administration as it relates to the manufacture of devices; and the development
and current status of FDA regulations on device good manufacturing practices.
Moist Heat
Sterilization : Current
Principles and Practices
Technical Monograph
Efforts of personnel in many disciplines, familiar
with all aspects of the process from inception, are involved in the development
of an efficacious Moist Heat Sterilization Process and its execution. The
process is susceptible to a wide range of variables. The advent of new
heat-sensitive solutions, as well as more sophisticated container-closure
systems and materials, require a constant redefinition in the approach to moist
heat sterilization. Process parameters must be established for every
end-product container system that is developed.
This technical Monograph provides a cogent overview
of the current principles and practices in moist heat sterilization.
Sterilization
and Depyrogenation using Dry Heat
Technical Monograph
Dry Heat Sterilization is the preferred
method for treatment of heat stable materials that will not tolerate steam. A
significant advantage of dry heat processing is the destruction of bacterial
endotoxins that have a much greater heat resistance than bacterial spores.
In this Monograph, the current status of
sterilization and depyrogenation using dry heat are examined, and readers are
familiarised with the basic microbiological, statistical and engineering
concepts that are essential for effective development of the process.
Since depyrogenation is a
field characterised by rapid technological advances, an attempt is made to
acquaint the reader with some of the recent research findings in the area.
Kinetics of endotoxin inactivation have been specially emphasised.
Label
Design and Labelling of Drugs
R.S. Iyer & S M Mudda
Designing product labels is an art, as the label has
to reconcile the demands of the law, the medical claims, promotional efforts
and, in an increasingly competitive environ-ment, the demand for aesthetic
appeal.
In this Monograph, an effort has been made to put
together all the legal requirements for designing a drug label. These are
presented in the form of two appendices to this book. The Appendices form the
core of the publication and provide the rationale for it.
Aims & Objectives
The
academy conducts one/two day seminars held across various cities all over
India. It draws créme-de-la-créme faculty from the industry. Participants come
from various disciplines, like QA/QC, Production, Research at all levels and
that makes the colloquiums highly interactive and interesting.
Glimpse of held
Colloquiums :
• SOPs, Designed for
Doing
• Parenteral Problems
• Documentation Blues
•
Labels & Good Labelling Practice more than a legal requirement
• Sterile Product Manufacture (Entry level induction
programme)
• Scientific Storekeeping • Primary Packaging
• Taming Tableting (A focus workshop)
• Tablet Coating from obscure art to an exacting
Science
• Aseptic Processing from Conception to Compliance
• Sampling Skills
• Focus Workshop on Good Laboratory Practice
• Cleaning Validation - A current perspective
• The GMP Route to TQC
• GCP Compliances in Clinical Trials.
Your Only Global
Source ........
INDIAN FOUNDATION FOR PHARMACEUTICAL
REFERENCE STANDARD SUBSTANCES
The Pharmaceutical Industry always stands sky high in
producing Quality Medicines. IFPRESS is proud to provide REFERENCE STANDARD
SUBSTANCES IP INTEGRAL and Important part of QC, QA and R&D.
RELIABILITY AND AUTHENTICITY IS ASSURED by
Preparation and Standardization by reputed manufacturers, in accordance with
strict Standard Operating Procedures Tested and Certified by CDL, Calcutta.
Subdivision, Storage and Distribution is by qualified persons under directions
of an expert committee, in a model Class 100 aseptic facility.
IFPRESS (INDIAN FOUNDATION FOR PHARMACEUTICAL
REFERENCE STANDARD SUBSTANCES) is the custodian of the project, comprising
professionals from the Regulatory Agencies, Industry, Academia and Research
Disciplines.
AVAILABLE
. Acetazolamide
. Cloxacillin Sodium
. Mianserin Hydrochloride
. Albendazole
. Clofazimine
. Metoprolol Tartrate
. Amoxycillin Trihydrate
. Danazol
. Mebendazole
. Ampicillin Trihydrate
. Dithranol
. Nandrolone Phenylpropionate
. Ascorbic Acid
. Diphenhydramine Hydrochloride
. Nalidixic Acid
. Betamethasone Valerate 17
. Ethyloestranol
. Nicotinic Acid
. Betamethasone Sodium Phosphate
. Folic Acid
. Nitrofurazone
. Bromhexine Hydrochloride
. Furazolidone
. Norfloxacin
. Cephalexine
. Glibenclamide
. Piroxicam
. Chloroquine Phosphate
. Ibuprofen
. Salbutamol Sulphate
. Chloroquine Sulphate
. Lignocaine Hydrochloride
. Sodium Valproate
. Ciprofloxacin Hydrochloride
. Mefenamic Acid
. Terbutaline Sulphate
. Atenolol
. Dicyclomine Hydrochloride
. Oxytetracycline Dihydrate
. 6-APA*
. Diclofenac Sodium
. PHPA*
. 7-ADC*
. Dicloxacilliin Sodium*
. Procaine Penicillin
. Betamethasone
. Diazepam
. Pentazocine
. Betamethasone Valerate 21
. Doxycycline Hydrochloride
. Penicillin G. Pottasium
. Beclomethasone 17 Propionate
. Ethambutol Hydrochloride
. Ranitidine Hydrochloride
. Benzyl Penicillin Sodium
. Flurbiprofen
. Ranitidine B*
. Cefuroxime Sodium
. Flucloxacillin Sodium*
. Rifampicin
. Cefadroxil
. Metronidazole
. Rifampicin Quinone*
. Chlorpropamide
. Methdilazine Hydrochloride
. Terfenadine
. DCDA*
. Metformin Hydrochloride
. Thyroxine Sodium
. Dicyclomine Hydrochloride
SERVICES
Maintenance and Validation
services for all laminar Flow Work Stations, both supplied by us as well as
supplied by other organizations
Supply
and Replacement of all spares needed for the workstations
Renovation and Up gradations of Clean Air
Systems
Carrying out area validation to certify
cleanliness levels
Consultation
Pharmaceutical
Engineering services
Designing
of HVAC and Filtration System for Clean Rooms
Area
Layouts including support facility in selection of suitable equipments for the
projects
Segregated
services for Injectbles and Vaccines
Barrier
Isolation Technology
Designing
of Barrier isolators & manufacture
Provision
of Solutions & Designs for Containment Systems
Complete
turn key projects undertaking
Provision
of techno-economical services
Provision
of solutions for handling dry bulk pharma-chemicals
Provision
of complete know-how of Bio-technological systems
Aseptic
processes for requirements in Pharma & Research Units
GMP
issues & stimulated discussions on design, manufacture, control monitor and
technical audit of pharma systems
CMT REPORT [Corruption, Money
laundering & Terrorism]
The
Public Notice information has been collected from various sources including but
not limited to: The Courts, India Prisons Service, Interpol, etc.
1] INFORMATION ON DESIGNATED PARTY
No records exist designating subject or any of its beneficial owners,
controlling shareholders or senior officers as terrorist or terrorist
organization or whom notice had been received that all financial transactions
involving their assets have been blocked or convicted, found guilty or against
whom a judgement or order had been entered in a proceedings for violating
money-laundering, anti-corruption or bribery or international economic or
anti-terrorism sanction laws or whose assets were seized, blocked, frozen or
ordered forfeited for violation of money laundering or international
anti-terrorism laws.
2] Court Declaration :
No records exist to suggest that subject is or was
the subject of any formal or informal allegations, prosecutions or other
official proceeding for making any prohibited payments or other improper
payments to government officials for engaging in prohibited transactions or
with designated parties.
3] Asset Declaration :
No records exist to suggest that the property or assets of the subject
are derived from criminal conduct or a prohibited transaction.
4] Record on Financial Crime :
Charges or conviction registered
against subject: None
5] Records on Violation of Anti-Corruption Laws :
Charges or investigation registered
against subject: None
6] Records on Int’l Anti-Money Laundering Laws/Standards :
Charges or investigation registered
against subject: None
7] Criminal Records
No available information exist that suggest that
subject or any of its principals have been formally charged or convicted by a
competent governmental authority for any financial crime or under any formal
investigation by a competent government authority for any violation of
anti-corruption laws or international anti-money laundering laws or standard.
8] Affiliation with Government :
No record exists to suggest that any director or
indirect owners, controlling shareholders, director, officer or employee of the
company is a government official or a family member or close business associate
of a Government official.
9] Compensation Package :
Our market survey revealed that the amount of
compensation sought by the subject is fair and reasonable and comparable to
compensation paid to others for similar services.
10] Press Report :
No press reports / filings exists on
the subject.
CORPORATE GOVERNANCE
MIRA
INFORM as part of its Due Diligence do provide comments on Corporate Governance
to identify management and governance. These factors often have been predictive
and in some cases have created vulnerabilities to credit deterioration.
Our
Governance Assessment focuses principally on the interactions between a
company’s management, its Board of Directors, Shareholders and other financial
stakeholders.
CONTRAVENTION
Subject
is not known to have contravened any existing local laws, regulations or
policies that prohibit, restrict or otherwise affect the terms and conditions
that could be included in the agreement with the subject.
FOREIGN EXCHANGE RATES
|
Currency |
Unit
|
Indian Rupees |
|
US
Dollar |
1 |
Rs.45.85 |
|
UK
Pound |
1 |
Rs.85.68 |
|
Euro |
1 |
Rs.58.59 |
SCORE & RATING EXPLANATIONS
|
SCORE FACTORS |
RANGE |
POINTS |
|
HISTORY |
1~10 |
7 |
|
PAID-UP
CAPITAL |
1~10 |
7 |
|
OPERATING
SCALE |
1~10 |
7 |
|
FINANCIAL
CONDITION |
|
|
|
--BUSINESS
SCALE |
1~10 |
7 |
|
--PROFITABILIRY |
1~10 |
7 |
|
--LIQUIDITY |
1~10 |
7 |
|
--LEVERAGE |
1~10 |
7 |
|
--RESERVES |
1~10 |
7 |
|
--CREDIT
LINES |
1~10 |
7 |
|
--MARGINS |
-5~5 |
- |
|
DEMERIT
POINTS |
|
|
|
--BANK
CHARGES |
YES/NO |
YES |
|
--LITIGATION |
YES/NO |
NO |
|
--OTHER
ADVERSE INFORMATION |
YES/NO |
NO |
|
MERIT
POINTS |
|
|
|
--SOLE
DISTRIBUTORSHIP |
YES/NO |
NO |
|
--EXPORT
ACTIVITIES |
YES/NO |
YES |
|
--AFFILIATION |
YES/NO |
YES |
|
--LISTED |
YES/NO |
NO |
|
--OTHER
MERIT FACTORS |
YES/NO |
YES |
|
TOTAL |
|
63 |
This score serves as a reference to assess SC’s credit risk and to set
the amount of credit to be extended. It is calculated from a composite of
weighted scores obtained from each of the major sections of this report. The
assessed factors and their relative weights (as indicated through %) are as
follows:
Financial condition (40%) Ownership background (20%) Payment record (10%)
Credit history (10%) Market trend (10%) Operational size
(10%)
RATING EXPLANATIONS
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
>86 |
Aaa |
Possesses
an extremely sound financial base with the strongest capability for timely
payment of interest and principal sums |
Unlimited |
|
71-85 |
Aa |
Possesses
adequate working capital. No caution needed for credit transaction. It has
above average (strong) capability for payment of interest and principal sums |
Large |
|
56-70 |
A |
Financial
& operational base are regarded healthy. General unfavourable factors
will not cause fatal effect. Satisfactory capability for payment of interest
and principal sums |
Fairly Large |
|
41-55 |
Ba |
Overall
operation is considered normal. Capable to meet normal commitments. |
Satisfactory |
|
26-40 |
B |
Unfavourable
& favourable factors carry similar weight in credit consideration.
Capability to overcome financial difficulties seems comparatively below
average/normal. |
Small |
|
11-25 |
Ca |
Adverse
factors are apparent. Repayment of interest and principal sums in default or
expected to be in default upon maturity |
Limited with full
security |
|
<10 |
C |
Absolute
credit risk exists. Caution needed to be exercised |
Credit not recommended |