![]()
MIRA
INFORM REPORT
|
Report Date : |
07.06.2007 |
IDENTIFICATION DETAILS
|
Name : |
CAPSUGEL [ |
|
|
|
|
Registered Office : |
1/82
Moo 5, Rojana
Industrial Estate, |
|
|
|
|
Country : |
|
|
|
|
|
Financials (as on) : |
30.11.2005 |
|
|
|
|
Date of Incorporation : |
1983 |
|
|
|
|
Com. Reg. No.: |
0105526006556 |
|
|
|
|
Legal Form : |
Private Limited Company |
|
|
|
|
Line of Business : |
Manufacturer and distributor
of gelatin capsule
and hard gelatin
capsules for pharmaceutical and
dietary supplement applications. |
RATING & COMMENTS
|
MIRA’s Rating : |
Ba |
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
41-55 |
Ba |
Overall operation is considered normal. Capable to meet normal
commitments. |
Satisfactory |
|
Status : |
Satisfactory |
|
|
|
|
Payment Behaviour : |
Regular |
|
|
|
|
Litigation : |
Clear |
name
CAPSUGEL [
FORMER
INTERNATIONAL CAPSULE CO., LTD.
SUMMARY
BUSINESS ADDRESS : 1/82
MOO 5, ROJANA
INDUSTRIAL ESTATE,
TELEPHONE : [66] 35
334-000, 35 334-050
FAX : [66] 35
334-001, 35 227-088
E-MAIL ADDRESS : kriengsak.piyathanavong@pfizer.com
REGISTRATION ADDRESS : SAME
AS BUSINESS ADDRESS
ESTABLISHED : 1983
REGISTRATION NO. : 0105526006556 [Former : 655/2526]
CAPITAL
REGISTERED : BHT.
170,000,000
CAPITAL
PAID-UP : BHT.
170,000,000
FISCAL
YEAR CLOSING DATE : NOVEMBER 30
LEGAL STATUS : PRIVATE LIMITED
COMPANY
EXECUTIVE : MR.
WANCHAI SANTIMANOCHAI, THAI
MANAGING DIRECTOR
NO. OF
STAFF : 200
LINES OF
BUSINESS : CAPSULE PRODUCTS
MANUFACTURER &
DISTRIBUTOR
CORPORATE PROFILE
OPERATING TREND : STABLE
PRESENT SITUATION : OPERATING NORMALLY
REPUTATION : GOOD
WITH
MANAGEMENT STANDARD : MANAGEMENT WITH
AVERAGE PERFORMANCE
HISTORY
The subject was
established on February
17, 1983 as
a private limited
company under the registered
name International Capsule
Co., Ltd., by Thai
and Dutch Groups.
On February 12,
2002, the subject was changed
its name to
be CAPSUGEL [THAILAND]
CO., LTD. It
currently employs approximately
200 staff.
The subject is
a joint venture
between “O.C.T. [
On February
1, 1999, the
subject received the
International Standard ISO
9002 certification to
manufacture of hard
gelatin capsules by
RWTUV, and ISO
14001 certificate for
environmental system by RWTUV on
March 7, 2003.
On October 25,
2000, the subject
was invested about
500 million baht
and 270 million
baht to produce
5,250 million of
gelatin capsules and
5,250 million of
hard gelatin capsules
per year respectively
with BOI promoted.
The subject adopts the Acceptable Quality Level (AQL) standard, in
addition to the ISO 9001:2000 quality management system, to create quality
consciousness.
The subject’s registered
address is 1/82
Moo 5, Rojana Rd.,
T. Kamham, A. Uthai, Ayutthya
13210, and this
is the subject’s
current operation address.
THE BOARD
OF DIRECTORS
Name Nationality Age
Mr. Kriengsak Piyatanawong
Thai 51
Mr. Charles R. Donjout
French
63
Ms. Vilaiporn Laochuwongse Thai 54
Mr. Wanchai Santimanochai
Thai 51
Mr. Guido Edouard
Driesen Belgian 51
AUTHORIZED PERSON
Two of the
above directors can
jointly sign on
behalf of the
subject with company’s
affixed.
MANAGEMENT
Mr. Wanchai
Santimanochai is the
Managing Director.
He is Thai
nationality with the
age of 51
years old.
Ms. Vilaiporn
Laochuwongse is the
Finance & Account
Manager.
She is Thai
nationality with the age of
54 years old.
Mr. Kriengsak
Piyatanawong is the
Marketing & Sales
Director.
He is Thai
nationality with the
age of 51
years old.
Ms. Lu
Jin Hua is
the Factory Manager.
She is Chinese
nationality.
Ms. Chotika
Pudpad is the
Human Resources Manager.
She is Thai
nationality.
BUSINESS OPERATIONS
The subject’s activities are
manufacturer and distributor
of gelatin capsule
and hard gelatin
capsules for pharmaceutical and
dietary supplement applications.
Its products include
Coni-snap capsules, Vcap
capsules, Licaps capsules,
PCcaps capsules, Press-fit
capsules, DBcaps capsules
& NP caps
capsules, as well
as dietary supplement
applications.
IMPORT [COUNTRIES]
Most of its raw
materials for processing
such as gelatin
are imported from
Japan and the
others are from Germany, United
States of America, Netherlands, France
and United Kingdom.
MAJOR SUPPLIER
Pfizer Inc. :
SALES [LOCAL]
70% of its
products is sold
locally to manufacturers, wholesalers
and end-users.
EXPORT [COUNTRY]
70% of its
products is exported
to Malaysia, Australia,
New Zealand, Vietnam,
India, Singapore and Pakistan.
MAJOR CUSTOMERS
Pharmaceutical Organizations and
manufactures
LITIGATION
Bankruptcy and Receivership
There are no
litigation on bankruptcy
and receivership cases
filed against the
subject found at
Legal Execution Department
for the past
five years.
Others
There are no
legal suits filed
against the subject for
the past two
years.
CREDIT
Sales are by
cash or on
the credits term
of 30-60 days.
Local bills are
paid by cash
or on the
credits term of
30-60-90 days.
Imports are by
L/C at sight
or T/T on
negotiated term.
Exports are against
L/C at sight
or T/T on
negotiated term.
BANKING
Bangkok Bank
Public Co., Ltd.
[Rojana Branch :
Citibank, NA
[
EMPLOYMENT
The subject employs
approximately 200 staff.
[office staff and
factory workers]
LOCATION DETAILS
The premise is
rented for administrative office
and factory at
the heading address.
Premise is located
in an industrial
area.
The subject was
relocated its office
from 15th Floor, Sino-Thai
Tower, 32/39 Sukhumvit
21 Rd. to the present
address in August
2001.
COMMENT
The subject is a production
facility of Capsugel, a leading global manufacturer of capsules for
pharmaceutical and dietary supplement applications. With its headquarters in
Capsugel (
The subject has become the group’s export center for the Asian market,
as well as
Local demand for
capsule products consumption
has significantly increased
due to pharmaceutical and
health food supplement
industry are growing
in high demand.
Local pharmaceutical and
food supplement industries
has shown strong
growth both local
and international markets.
The industry outlook is
soaring.
The subject has
maintained its leading
role in the industry
and expanding steadily.
FINANCIAL INFORMATION
The capital was
registered at Bht.
1,000,000 divided into
10,000 shares of
Bht. 100 each.
The capital was
increased later as
followings :
Bht. 30,000,000
on April 3,
1986
Bht. 70,000,000
on April 12,
1988
Bht. 90,000,000
on June 17,
1998
Bht. 170,000,000 on
June 17, 2004
The latest registered
capital was increased
to Bht. 170,000,000
divided into 1,700,000
shares of Bht.
100 each with
fully paid.
THE SHAREHOLDERS
LISTED WERE : [as at
March 31, 2006]
|
NAME |
HOLDING |
% |
|
|
|
|
|
Pfizer B.V. Nationality: Dutch Address : |
1,240,999 |
49.00 |
|
O.C.T. [ Nationality: Thai Address : 1/82 Moo 5,
A. Uthai, |
458,996 |
51.00 |
|
Warner -
Lambert [Thai] Ltd. Nationality: Thai Address : |
1 |
- |
|
Renroll LLC Nationality: American Address : |
1 |
- |
|
Solinor LLC Nationality: American Address : |
1 |
- |
|
International Affiliated
Corporation LLC Nationality: American Address : |
1 |
- |
|
Tebor Corporation Nationality: American Address : |
1 |
- |
Total Shareholders
7
NAME OF
AUDITOR & CERTIFIED
PUBLIC
ACCOUNTANT NO.
Mr. Vichien Thamtrakul
No. 3183
BALANCE SHEET [BAHT]
The
latest financial figures
published for November
30, 2005 & 2004 were:
ASSETS
|
Current Assets |
2005 |
2004 |
|
|
|
|
|
Cash in hand
& at Bank
|
159,318 |
2,300,504 |
|
Trade Accts. &
Notes Receivable |
248,629,420 |
239,332,474 |
|
Inventories [note 1] |
68,725,239 |
71,608,724 |
|
Account Receivable –
Related Company |
4,331,288 |
7,255,955 |
|
Refundable Value Added
Tax |
27,348,285 |
29,868,570 |
|
Other Current Assets
|
2,815,505 |
7,654,040 |
|
|
|
|
|
Total Current
Assets |
352,009,055 |
358,020,267 |
|
Fixed Assets [note 2] |
836,144,107 |
851,336,067 |
|
Deposit |
1,147,936 |
2,807,936 |
|
Total Assets
|
1,189,301,098 |
1,212,164,270 |
LIABILITIES &
SHAREHOLDERS' EQUITY [BAHT]
|
Current Liabilities |
2005 |
2004 |
|
|
|
|
|
Bank Overdraft |
- |
2,223,430 |
|
Trade Accounts Payable |
92,387,045 |
60,826,957 |
|
Account Payable -
Related Company |
193,050 |
1,375,041 |
|
Short-term Loan from
Related Company |
440,000,000 |
646,242,144 |
|
Accrued Expenses |
33,611,503 |
40,197,992 |
|
Accrued Income Tax |
4,770,982 |
77,049 |
|
Revenue Expenses |
7,406,457 |
2,494,427 |
|
Other Current Liabilities |
27,974,299 |
14,866,837 |
|
|
|
|
|
Total Current
Liabilities |
606,343,336 |
768,226,828 |
|
|
|
|
|
Shareholders'
Equity |
|
|
|
Share capital : Baht 100
value authorized, issued
and fully paid share
capital 1,700,000 shares |
170,000,000 |
170,000,000 |
|
|
|
|
|
Capital Paid |
170,000,000 |
170,000,000 |
|
Retained Earning Appropriated for
Statutory Reserve |
17,000,000 |
8,353,575 |
|
Unappropriated [Deficit] |
395,957,762 |
265,583,867 |
|
Total
Shareholders' Equity |
582,957,762 |
443,937,442 |
|
Total
Liabilities & Shareholders' Equity |
1,189,301,098 |
1,212,164,270 |
PROFIT &
LOSS ACCOUNT
|
Revenue |
2005 |
2004 |
|
|
|
|
|
Sales |
906,813,690 |
819,645,777 |
|
Other Income |
9,875,262 |
3,446,502 |
|
Total Revenues
|
916,688,952 |
823,092,279 |
|
Expenses |
|
|
|
|
|
|
|
Cost of Goods
Sold |
537,198,017 |
503,038,073 |
|
Selling & Administrative Expenses |
132,091,065 |
134,317,252 |
|
|
|
|
|
Total Expenses |
669,289,082 |
637,355,325 |
|
|
|
|
|
Profit / [Loss] before Interest
Expenses & Income Tax |
247,399,870 |
185,736,954 |
|
Interest Expenses |
[9,250,486] |
[219,958] |
|
Income Tax |
[9,129,064] |
[112,262] |
|
Net Profit / [Loss] |
229,020,320 |
185,404,734 |
FINANCIAL NOTE
NOTE 1
Inventories
|
|
2005 |
2004 |
|
|
|
|
|
Finished Goods |
29,482,437 |
42,544,862 |
|
Raw Material |
29,375,319 |
22,807,902 |
|
Works in Progress |
- |
3,486,750 |
|
Material Supplies |
- |
1,959,791 |
|
Good |
3,828,001 |
15,237,884 |
|
Goods in Transit |
16,654,358 |
- |
|
Total |
79,340,115 |
86,037,189 |
|
Less: Allowance
for Stock Obsolescence |
[10,614,876] |
[14,428,465] |
|
Total |
68,725,239 |
71,608,724 |
NOTE 2
Fixed Assets
|
|
2005 |
2004 |
|
|
|
|
|
Land |
38,247,010 |
39,421,335 |
|
Building & Construction |
156,432,956 |
156,432,956 |
|
Machine & Equipment |
1,131,066,533 |
1,041,461,797 |
|
Office Automation & Furniture |
27,122,213 |
24,658,501 |
|
Vehicle |
7,520,098 |
2,119,713 |
|
Assets under Installation |
6,225,198 |
7,378,707 |
|
Total |
1,366,614,008 |
1,271,473,009 |
|
Less: Depreciation |
[530,469,901] |
[420,136,942] |
|
Total |
836,144,107 |
851,336,067 |
FINANCIAL ANALYSIS
Annual Growth
& Profitability
|
Annual Growth |
2005 |
2004 |
|
Net Sales |
10.63 |
|
|
Operating Profit |
20.40 |
|
|
Net Profit |
23.52 |
|
|
Fixed Assets |
[1.78] |
|
|
Total Assets |
[1.89] |
|
|
Profitability |
|
|
|
Cost of Good
Sold |
59.24 |
61.37 |
|
Operating Profit Margin |
27.28 |
22.66 |
|
S & A Expenses |
14.57 |
16.39 |
|
Interest Paid |
1.02 |
0.03 |
|
Other Revenues |
1.09 |
0.42 |
|
Net Profit Margin |
25.26 |
22.62 |
|
Earning Per Shares |
134.72 |
109.06 |
|
No. of Shares |
1,700,000 |
1,700,000 |
Net Sales was
increased by 10.63%,
Operating Profit was
increased by 20.40%,
Net Profit was
increased by 23.52%,
Fixed Assets was
decreased by 1.78%,
Total Assets was
decreased by 1.89%.

Compare each cost
with sales, Cost of Good Sold
was decreased from
61.37% to 59.24%,
Operating Profit Margin
was increased from
22.66% to 27.28%,
S & A
Expenses was decreased
from 16.39% to
14.57%, Interest Paid was
increased from 0.03%
to 1.02%, Other
Revenues was increased
from 0.42% to
1.09%, Net Profit
Margin was increased
from 22.62% to
25.26%.
Earning Per Shares
was increased from
109.06 baht per
share to 134.72
baht per share.
Liquidity
|
Liquidity |
2005 |
2004 |
|
Current Ratio |
0.58 |
0.47 |
|
Quick-Assets Ratio |
0.41 |
0.31 |
Current Ratio was
increased from 0.47
to 0.58, show
Current Assets can
not cover Current
Liabilities, Liquidity Ratio
of the company
was not good,
Quick-Assets Ratio was
increased from 0.31
to 0.41, capacity
to pay Short
Term Loan was
not good too,
because Current Assets
without Inventory can
not cover Current
Liabilities.
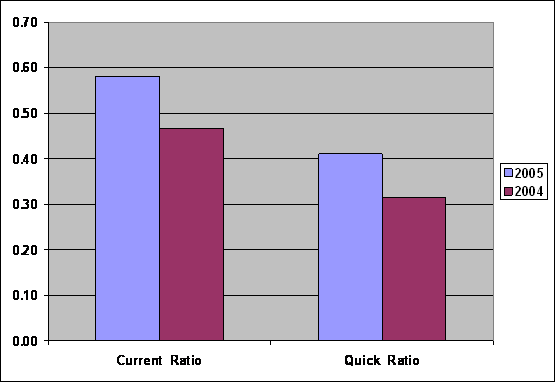
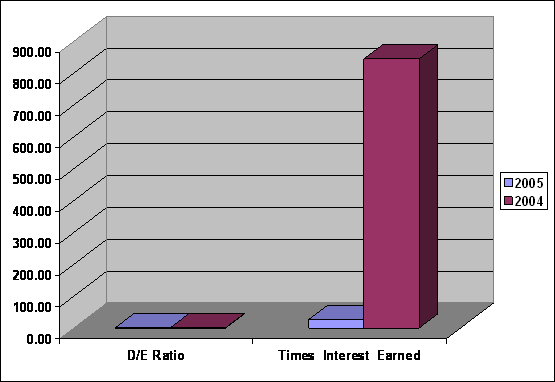
Leverage
|
Leverage |
2005 |
2004 |
|
D/E Ratio |
1.04 |
1.73 |
|
Times Interest Earned |
26.74 |
844.42 |
D/E Ratio was
decreased from 1.73
to 1.04, show
the company had
fund from Loan
more than fund
from Equity, investment
risked of the company is
medium, Times Interest
Earned was decreased
from 844.42 to
26.74, capacity to
pay interest paid
was good.
Efficiency
|
Efficiency |
2005 |
2004 |
|
Fixed Assets Turnover |
1.08 |
0.96 |
|
Total Assets Turnover |
0.76 |
0.68 |
|
Return on Assets |
19.26 |
15.30 |
|
Return on Equity |
39.29 |
41.76 |
|
Day's Payable |
36.68 |
26.72 |
|
Day’s Inventories |
27.28 |
31.45 |
|
Day’s Receivables |
98.70 |
105.12 |
Fixed Assets Turnover was increased from 0.96 times to 1.08 times, Total Assets Turnover was increased from 0.68 times to 0.76 times, show the company could use Assets so efficiently, Return on Assets was increased from 15.30 to 19.26, Return on Equity was decreased from 41.76 to 39.29, capacity to use Assets to make profit was good.
Day's Payable was
increased from 27
days to 37
days, while Day’s
Inventories was decreased
from 32 days
to 28 days,
and Day’s Receivables
was decreased from
106 days to
99 days.

RATING EXPLANATIONS
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
>86 |
Aaa |
Possesses an extremely sound financial base with the strongest capability
for timely payment of interest and principal sums |
Unlimited |
|
71-85 |
Aa |
Possesses adequate working capital. No caution needed for credit
transaction. It has above average (strong) capability for payment of interest
and principal sums |
Large |
|
56-70 |
A |
Financial & operational base are regarded healthy. General
unfavourable factors will not cause fatal effect. Satisfactory capability for
payment of interest and principal sums |
Fairly Large |
|
41-55 |
Ba |
Overall operation is considered normal. Capable to meet normal
commitments. |
Satisfactory |
|
26-40 |
B |
Unfavourable & favourable factors carry similar weight in credit
consideration. Capability to overcome financial difficulties seems
comparatively below average/normal. |
Small |
|
11-25 |
Ca |
Adverse factors are apparent. Repayment of interest and principal sums
in default or expected to be in default upon maturity |
Limited with
full security |
|
<10 |
C |
Absolute credit risk exists. Caution needed to be exercised |
Credit not
recommended |
|
NR |
In view of the lack of information, we have no basis upon which to
recommend credit dealings |
No Rating |
|
This score serves as a reference to assess SC’s credit risk
and to set the amount of credit to be extended. It is calculated from a
composite of weighted scores obtained from each of the major sections of this
report. The assessed factors and their relative weights (as indicated through
%) are as follows:
Financial
condition (40%) Ownership
background (20%) Payment
record (10%)
Credit history
(10%) Market trend
(10%) Operational
size (10%)