![]()
MIRA
INFORM REPORT
|
Report Date : |
27.06.2007 |
IDENTIFICATION DETAILS
|
Name : |
MEDICAPS LIMITED |
|
|
|
|
Registered Office : |
|
|
|
|
|
Country : |
|
|
|
|
|
Financials (as on) : |
31.03.2006 |
|
|
|
|
Date of Incorporation : |
06.08.1983 |
|
|
|
|
Com. Reg. No.: |
10-2231 |
|
|
|
|
CIN No. : |
L24232MP1983PLC002231 |
|
|
|
|
TAN No.: [Tax
Deduction & Collection Account No.] |
BPLM01282B |
|
|
|
|
Legal Form : |
Public Limited Liability Company. Company’s shares are listed on the stock Exchanges. |
|
|
|
|
Line of Business : |
Manufacturing of Capsules to attain Zero – Defect in Production and to fully normalize operations. |
RATING & COMMENTS
|
MIRA’s Rating : |
Ba |
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
41-55 |
Ba |
Overall operation is considered normal. Capable to meet normal
commitments. |
Satisfactory |
|
Maximum Credit Limit : |
USD 1000000 |
|
|
|
|
Status : |
Satisfactory |
|
|
|
|
Payment Behaviour : |
Regular |
|
|
|
|
Litigation : |
Clear |
|
|
|
|
Comments : |
Subject is a well – established company having satisfactory track. Trade relations are fair. General financial position is satisfactory. Payments are usually correct and as per commitments. The company can be considered normal for business dealings at usual trade terms and conditions. |
LOCATIONS
|
Registered Office : |
|
|
Tel. No.: |
91-7292-2407445/2407446/253596 |
|
Fax No.: |
91-7292-407387 |
|
E-Mail : |
|
|
Website : |
|
|
|
|
|
Corporate Office : |
401, Chetal Center, 12/2, R. N. T. Marg, |
|
Tel. No.: |
91-731-24046321/2514062 |
|
Fax No.: |
91-731-2582269 |
|
E-Mail : |
DIRECTORS
|
Name : |
Mr. R.C. Mittal |
|
Designation : |
Chairman & Managing Director |
|
|
|
|
Name : |
Mr. Alok k. Garg |
|
Designation : |
Executive Director |
|
|
|
|
Name : |
Mrs. Trapti Gupta |
|
Designation : |
Director |
|
|
|
|
Name : |
Mrs. Kusum Mittal |
|
Designation : |
Director |
|
|
|
|
Name : |
Dr. S.K. Sharma |
|
Designation : |
Director |
|
|
|
|
Name : |
Dr. J.P. Srivastava |
|
Designation : |
Director |
|
|
|
|
Name : |
Dr. Shamsher Singh |
|
Designation : |
Director |
|
|
|
|
Name : |
Dr. Vishwanath B. Malkar |
|
Designation : |
Director |
|
|
|
|
Name : |
Mr. Ganesh Pawar |
|
Designation : |
Company Secretary |
|
|
|
|
Name : |
Dr. Vinay G. Nayak |
|
Designation : |
Director |
|
|
|
|
Name : |
Shri Ashok R. Pitaliya |
|
Designation : |
Manager – Accounts |
|
|
|
KEY EXECUTIVES
|
Name : |
D.K. Jain & Company |
|
Designation : |
Company Secretaries |
|
Address : |
4th floor, |
MAJOR SHAREHOLDERS / SHAREHOLDING PATTERN
|
Names of Shareholders |
No. of Shares |
Percentage of
Holding |
|
v Promoters Holding |
|
|
|
Indian Promoters |
1429800 |
45.864 |
|
Persons Acting in
Concert |
760 |
0.024 |
|
Sub Total |
1430560 |
45.888 |
|
v
Non Promoters
Holding |
|
|
|
Mutual Funds and UTI |
1400 |
0.045 |
|
Banks, Financial
Institutions, Companies
(Central/State Government/ Non-Government. Institutions) |
7300 |
0.234 |
|
Sub Total |
8700 |
0.279 |
|
v
Others |
|
|
|
Private Corporate Bodies |
146644 |
4.704 |
|
Indian Public |
1490458 |
47.810 |
|
NRI/OCBs |
41127 |
1.319 |
|
Sub Total |
1678229 |
53.833 |
|
|
|
|
|
Grand Total |
3117489 |
100.000 |
|
|
|
|
BUSINESS DETAILS
|
Line of Business : |
Manufacturing of Capsules to attain Zero – Defect in Production and to fully normalize operations. |
|
|
|
|
Products : |
Products
Description: Hard Gelatin Capsule Shells I.P. Item Code : 9602 0030 |
|
|
|
PRODUCTION STATUS
|
Particulars |
|
Unit |
Installed
Capacity |
Actual
Production |
|
|
|
|
|
|
|
Hard Gelatin Capsule Shells I.P. |
|
Nos in mins |
32,500 |
31,742 |
GENERAL INFORMATION
|
Customers: |
v
US
Vitamins v
SOL
Pharmaceuticals v
Dr
Reddy's Laboratories v
Lupin Laboratories v
Aristo Pharmaceuticals |
|
|
|
|
Bankers : |
v
IDBI Bank Limited. v
IndusInd Bank Limited. v
State-Bank of v ICICI Bank v HSBC Limited v Citibank N.A. |
|
|
|
|
Banking
Relations : |
Satisfactory |
|
|
|
|
Auditors : |
|
|
Name : |
Rawka Agrawal
& Company Chartered Accountants, |
|
Address : |
403, Arcade
Silver 56, 1, New Palasia, |
|
|
|
|
Associates/Subsidiaries : |
The Medi-Caps Group
Companies include: 1. Medi-Caps
Institute of Technology and Management (MITM) 2. Medi-Caps Finance Limited 3. International
Institute of Foreign Trade and Research
(IIFTR) 4. Medpak India
Limited 5. Trapti
Investments (P) Limited 6. Mittal
Enterprises 7. Akshit Finance (P) Limited |
CAPITAL STRUCTURE
Authorized Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
40,00,000 |
Equity Shares |
Rs. 10/- Each |
Rs. 40.000 Millions |
|
|
|
|
|
|
|
|
|
|
Issued, Subscribed & Paid-up Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
36,68,580 |
Equity Shares |
Rs. 10/- Each |
Rs. 36.685 Millions |
|
Subscribed |
|
|
|
|
31,17,489 |
Equity Shares |
Rs. 10/- Each |
Rs. 31.174 Millions |
|
Add: |
Forfeited Shares amount originally Paid Up |
|
Rs. 2.755 Millions |
|
|
Total |
|
Rs. 33.930
Millions |
FINANCIAL DATA
[all figures are
in Rupees Millions]
ABRIDGED BALANCE
SHEET
|
SOURCES OF FUNDS |
31.03.2006 |
31.03.2005 |
31.03.2004 |
|
|
|
SHAREHOLDERS FUNDS |
|
|
|
|
|
|
1] Share Capital |
33.930 |
33.930 |
33.930 |
|
|
|
2] Share Application Money |
0.000 |
0.000 |
0.000 |
|
|
|
3] Reserves & Surplus |
268.132 |
210.638 |
176.408 |
|
|
|
4] (Accumulated Losses) |
0.000 |
0.000 |
0.000 |
|
|
|
NETWORTH |
302.062 |
244.568 |
210.338 |
|
|
|
LOAN FUNDS |
|
|
|
|
|
|
1] Secured Loans |
1.551 |
0.000 |
0.000 |
|
|
|
2] Unsecured Loans |
0.000 |
4.255 |
5.921 |
|
|
|
TOTAL BORROWING |
1.551 |
4.255 |
5.921 |
|
|
|
DEFERRED TAX LIABILITIES |
10.305 |
11.645 |
12.410 |
|
|
|
|
|
|
|
|
|
|
TOTAL |
313.918 |
260.468 |
228.669 |
|
|
|
|
|
|
|
|
|
|
APPLICATION OF FUNDS |
|
|
|
|
|
|
|
|
|
|
|
|
|
FIXED ASSETS [Net Block] |
55.663 |
61.827 |
67.469 |
|
|
|
Capital work-in-progress |
0.000 |
0.000 |
0.006 |
|
|
|
|
|
|
|
|
|
|
INVESTMENT |
196.313 |
122.513 |
109.127 |
|
|
|
DEFERREX TAX ASSETS |
0.000 |
0.000 |
0.000 |
|
|
|
|
|
|
|
|
|
|
CURRENT ASSETS, LOANS & ADVANCES |
|
|
|
|
|
|
|
Inventories |
25.742 |
28.161
|
19.904 |
|
|
|
Sundry Debtors |
43.269 |
37.851
|
36.401 |
|
|
|
Cash & Bank Balances |
8.233 |
15.355
|
5.434 |
|
|
|
Other Current Assets |
0.000 |
0.000
|
0.000 |
|
|
|
Loans & Advances |
29.364 |
33.144
|
39.565 |
|
|
Total
Current Assets |
106.608
|
114.511
|
101.304 |
|
|
|
Less : CURRENT
LIABILITIES & PROVISIONS |
|
|
|
|
|
|
|
Current Liabilities |
26.875 |
22.335
|
29.436 |
|
|
|
Provisions |
18.357 |
17.182
|
21.499 |
|
|
Total
Current Liabilities |
45.232
|
39.517
|
50.935 |
|
|
|
Net Current Assets |
61.376
|
74.994
|
50.369 |
|
|
|
|
|
|
|
|
|
|
MISCELLANEOUS EXPENSES |
0.566 |
1.133 |
1.698 |
|
|
|
|
|
|
|
|
|
|
TOTAL |
313.918 |
260.468 |
228.669 |
|
|
PROFIT & LOSS
ACCOUNT
|
PARTICULARS |
31.03.2006 |
31.03.2005 |
31.03.2004 |
|
Sales Turnover (Including Other income ) |
240.335 |
240.000 |
247.263 |
|
|
|
|
|
|
Profit/(Loss) Before Tax |
75.195 |
51.218 |
55.621 |
|
Provision for Taxation |
11.853 |
11.085 |
14.064 |
|
Profit/(Loss) After Tax |
63.342 |
40.133 |
41.557 |
|
|
|
|
|
|
Export Value |
29.518 |
36.943 |
30.701 |
|
|
|
|
|
|
Import Value |
1.794 |
1.361 |
1.041 |
|
|
|
|
|
|
Expenditures : |
0.000 |
0.000 |
|
|
Cost of Goods Sold |
0.000 |
0.000 |
|
|
Manufacturing Expenses |
37.847 |
43.489 |
|
|
Administrative Expenses |
0.000 |
0.000 |
|
|
Raw Material Consumed |
53.538 |
57.352 |
|
|
Purchases made for re-sale |
0.000 |
0.000 |
|
|
Consumption of stores and spares parts |
0.000 |
0.000 |
|
|
Increase/(Decrease) in Finished Goods |
0.000 |
0.000 |
191.642 |
|
Salaries, Wages, Bonus, etc. |
25.154 |
25.122 |
|
|
Excise Duty |
19.441 |
24.579 |
|
|
Payment to Auditors |
0.000 |
0.000 |
|
|
Interest |
0.007 |
0.000 |
|
|
Right Issue Expenses |
0.566 |
0.566 |
|
|
Expenses of Previous Year |
0.139 |
0.603 |
|
|
Depreciation & Amortization |
7.699 |
7.869 |
|
|
Other Expenditure |
20.745 |
29.199 |
|
|
Total Expenditure |
165.136 |
188.779 |
191.642 |
SUMMERIZED RESULTS
|
PARTICULARS |
|
|
2007 (Full Year) |
|
Sales Turnover |
|
|
184.100 |
|
Other Income |
|
|
60.500 |
|
Total Income |
|
|
244.600 |
|
Total Expenditure |
|
|
144.600 |
|
Operating Profit |
|
|
100.000 |
|
Interest |
|
|
0.400 |
|
Gross Profit |
|
|
99.600 |
|
Depreciation |
|
|
8.100 |
|
Tax |
|
|
10.700 |
|
Reported PAT |
|
|
80.800 |
|
Dividend (%) |
|
|
0.000 |
KEY RATIOS
|
PARTICULARS |
31.03.2006 |
31.03.2005 |
31.03.2004 |
|
Debt Equity Ratio |
0.01 |
0.02 |
0.03 |
|
Long Term Debt Equity
Ratio |
0.01 |
0.02 |
0.03 |
|
Current Ratio |
2.07 |
1.88 |
1.68 |
|
TURNOVER RATIOS |
|
|
|
|
Fixed Assets |
1.24 |
1.32 |
1.51 |
|
Inventory |
7.22 |
8.54 |
11.56 |
|
Debtors |
4.79 |
5.53 |
6.27 |
|
Interest Cover
Ratio |
0.00 |
0.00 |
0.00 |
|
Operating Profit
Margin (%) |
22.97 |
23.62 |
27.71 |
|
Profit Before
Interest and Tax Margin (%) |
19.01 |
19.78 |
24.39 |
|
Cash Profit
Margin (%) |
20.25 |
19.43 |
21.54 |
|
Adjusted Net
Profit Margin (%) |
16.29 |
15.59 |
18.21 |
|
Return on Capital
Employed (%) |
13.56 |
17.80 |
28.65 |
|
Return on Net
Worth (%) |
11.71 |
14.26 |
21.92 |
STOCK PRICES
|
Face Value |
Rs. 10.00 |
|
High |
Rs. 69.50 |
|
Low |
Rs. 67.30 |
LOCAL AGENCY FURTHER INFORMATION
Fixed Assets
Ø
Free
Ø
Building
Ø
Plant & Machinery
Ø
Furniture & Fixture
Ø
Vehicles
Ø
Computers
History:
Medi-caps was incorporated
by S L Mittal in Aug.'83 as a private limited company and was converted into a public
limited company in Mar.'86. Pharmaceutical companies procure empty hard gelatin
capsules from Medi-caps for dosage medicines meant for oral administration. The
company is the second-largest manufacturer of empty hard gelatin capsules in
It takes several years for a company manufacturing capsules to attain
zero-defect in production and to fully normalize operations. Medi-caps can
claim to have achieved this. In Apr.'95, the company offered 0.611Millions
rights shares at a premium of Rs 116 to part-finance the Rs 91.9- Millions expansion of its manufacturing facilities
(from 2000 mln pa to 3440 mln pa) by installing four high-speed automatic
capsule-making machines.
The company's manufacturing unit is located in Pithampur, Madhya Pradesh..
At present, it has 40 clients. The other group company of Medi-caps is
Medi-caps Finance which is engaged in finance related activities. In 1993, R C
Mittal received the Glory of India International award.
In 1995-96, the company enhanced the capacity of empty hard gelatin
capsules by 250.0 Millions the company received ISO 9002 Certification during
1998-99. It developed six sizes of the Hard Gelatin Capsules in 2002-03. The
company is closely working on commissioning of 13th Machine with 100% atomization.
During 2002-03 the company established the export market in countries like
REVIEW OF
OPERATIONS:
During the under review Company could achieve a total income of
Rs.246.017 millions in comparison of previous year Rs.228.250 millions. Decline
in manufacturing turnover is purely due to various Government polices. For
example introduction of VAT, Excise on MRP for finished products etc. Due to
these issues major consumers of our product have cut down their production due
to which there was low demand.
The financial health of the country has shown significant growth
in all the sectors, and the company could able to earn Rs.46.821 millions
(previous year Rs.20.499 millions) this income includes dividend of Rs.10.087
millions and profit on sale of mutual funds Rs.32.192 millions. Due to strong
financial planning your company has maximized its value of investments and also
foresee the positive trends in the coming year. The overall profit for the year
is recorded to Rs.63.343 millions in comparison of previous year Rs.40.078
millions The Company has grown substantially the profitability despite of fall
in turnover.
MANAGEMENT DISCUSSION AND ANALYSIS REPORT:
Industry structure and developments:
The pharmaceuticals industry is a knowledge driven industry and is
heavily dependent on Research and Development of new products and growth.
However, basic research is a time consuming and expensive process and is thus
dominated by the huge local and global multinationals. Indian companies have
entered into the area and the results have been encouraging.
In the global pharmaceuticals market, western markets are growing largely
due to the introduction of the new molecules at the highest prices. A
well-established reimbursement and the insurance system imply that the per
capita drug expenditure is abnormally high in the western countries.
The Indian pharmaceuticals industry is highly fragmented, but has
grown rapidly due to the friendly patent regime and the low cost manufacturing
structure. The country production of the bulk drugs and the formulations grew
at CAGR of 18% over the past ten years
SWOT
ANALYSIS FOR THE COMPANY:
·
Strengths:
·
Strong Research & Development.* Integrated supply
chain.* Ability to deliver cost saving.* High quality of manpower resources at
a lower cost.* Centralized manufacturing activities at Pithampur Plant.* Zero
Debt Company.* Strong financial planning.
·
Weaknesses:
·
Scarcity of Technical Expertise.* Controlling of process
parameters is very critical.* Dependency on formulation Companies.* Frequent
fluctuation in market demand.* Very sensitive process of manufacturing.
·
Opportunities:
* In export there is a very good demand of gelatin
capsules and
* Due to improved capital market, there are good possibility
to invest the surplus funds in good securities and mutual funds.
·
Threats:
·
·
Aggressive price competition from local and multinational
player
·
Fast technology change in the manufacturing line of the
Company.
·
Frequent change in Govt. policy for pharmaceutical
industries.
·
Developed countries are very rigid in procuring capsules
from out of countries.
·
Competitions from the new entrants in the SEZ area, which
are enjoying duty exemption benefits.
Outlook:
While we cannot predict a further performance, we believe
considerable opportunities will exist for sustained and profitable growth, not
only in the developing countries but also in the developed western countries.
The Company is in continuous process to launch new variety of capsules and
variants to meet out the demands in the coming year and also to expand its
marketing reach in other countries for growth in the export as well as domestic
turnover. The company with its continued focus on exports stands to gain a lot
from the emerging scenario:
Marketing and Export:
In domestic market your Company had added many multinational
companies in its client lists with improved quality of products as well as
services. Due to that, company is forced to reduce the export basket.
ISO: 9001:2000 CERTIFICATION:
As you are aware that your Company is ISO: 9001:2000
complied companies which is the latest version of ISO series and this will give
further strength to the quality as well as international recognition and will
contribute to the export business of the company as it has done in the past
years.
As per website Details:
About Us:
Medi-Caps Limited flagship company of Medi-Caps Group, is one of the
largest manufacturer of empty hard gelatin capsules in terms of manufacturing
capacity as well as market capitalization in India, incorporated in 1986,
Medi-Caps Limited entered the highly competitive market with a team of
dedicated professionals, duly supported by competent individuals in every
aspect of operation.
At Medi-Caps Limited a philosophy of growth has been consistently upheld
with good business practices. Clear focus, excellent infrastructure, a powerful
quality policy and a well defined management information system have helped the
company to achieve consistent growth.
Their product range comprises of six sizes ranging from 00,0el, 0, 1el,
1,2,3,4 in more than 1000 shades, the same is increasing as rapidly as Pharma
Industry itself.
Today they offer printing features that are best in the industry. Medi-Caps
Limited has pioneered many advanced features in product development such as
“Pearlz Capsules” (Metallic Colored) as well as Python Printing & Li-Fill
Caps.
Medi-Caps Limited continuously strives to offer its customers excellent
product, technical advice, and a through professional service that makes their
product tailor made for their customers both in
Their professional goals and corporate personality have blended so well that
today Medi-Caps Limited name has become synonymous with the best in the
Industry.
Company
They implicitly believe that the corporate responsibility extends beyond
the ambit of a company's facilities and offices.
And that the true corporate citizenship must include common cause with
the society.
With this belief, Medi-Caps encourage funds and develop numerous
education and human capital initiatives.
These initiatives are now recognized in
Their mission is to offer product as well as services of the highest
professional standards in order to maintain their role as the customer's vendor
of first choice.”
Professional
Excellence
At Medi-Caps Ltd.
a philosophy of growth has been consistently upheld with good business
practices. Clear focus, excellent infrastructure, a powerful quality policy and
a well defined management information system have helped the company to achieve
constant success.
A team of dedicated
professionals is ably supported by competent colleagues in every area ranging
from manufacturing, through quality assurance, administration and management,
to sale.
Regular
interaction with vendors, dealer, customers, employees and financial
institutions, has helped to built a clear path to success.
PRODUCT INFORMATION
They manufacture Empty Hard Gelatin Capsules in sizes
00,0el,0,1el,1,2,3,4 elongated , fortified with more than thousand shades which
is growing as rapidly as pharma industries.
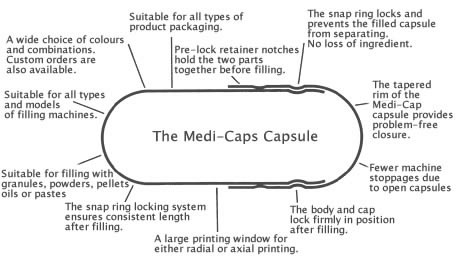
QUALITY
EXCELLENCE
A firm believer in the philosophy of Total Quality
Control Management, the company has evolved the
culture of systematic production that eliminates errors
in every stage of production.
Capsule production being a continuous process, representative samples
are regularly tested for physical defect, length, dome, double, wall thickness,
moisture content, colour shades, weight variation and other relevant
parameters.
The group's activities span manufacturing of Hard Gelatin Capsule
Shells, Education, Industrial Packaging & related products, Finance, Real
Estate and are a renowned name in central
The Flagship Company
Medi-Caps Limited has emerged as
CMT REPORT (Corruption, Money Laundering & Terrorism]
The Public Notice information has been collected from various sources
including but not limited to: The Courts,
1] INFORMATION ON
DESIGNATED PARTY
No records exist designating subject or any of its beneficial owners,
controlling shareholders or senior officers as terrorist or terrorist
organization or whom notice had been received that all financial transactions
involving their assets have been blocked or convicted, found guilty or against
whom a judgement or order had been entered in a proceedings for violating
money-laundering, anti-corruption or bribery or international economic or
anti-terrorism sanction laws or whose assets were seized, blocked, frozen or
ordered forfeited for violation of money laundering or international
anti-terrorism laws.
2] Court Declaration :
No records exist to suggest that subject is
or was the subject of any formal or informal allegations, prosecutions or other
official proceeding for making any prohibited payments or other improper
payments to government officials for engaging in prohibited transactions or
with designated parties.
3] Asset Declaration :
No records exist to suggest that the property or assets of the subject
are derived from criminal conduct or a prohibited transaction.
4] Record on Financial
Crime :
Charges or conviction registered
against subject: None
5] Records on Violation of
Anti-Corruption Laws :
Charges or
investigation registered against subject: None
6] Records on Int’l
Anti-Money Laundering Laws/Standards :
Charges or
investigation registered against subject: None
7] Criminal Records
No
available information exist that suggest that subject or any of its principals
have been formally charged or convicted by a competent governmental authority
for any financial crime or under any formal investigation by a competent
government authority for any violation of anti-corruption laws or international
anti-money laundering laws or standard.
8] Affiliation with
Government :
No record
exists to suggest that any director or indirect owners, controlling shareholders,
director, officer or employee of the company is a government official or a
family member or close business associate of a Government official.
9] Compensation Package :
Our market
survey revealed that the amount of compensation sought by the subject is fair
and reasonable and comparable to compensation paid to others for similar
services.
10] Press Report :
No press reports / filings exists on
the subject.
CORPORATE GOVERNANCE
MIRA INFORM as part of its Due Diligence do provide comments on Corporate
Governance to identify management and governance. These factors often have been
predictive and in some cases have created vulnerabilities to credit
deterioration.
Our Governance Assessment focuses principally on the interactions
between a company’s management, its Board of Directors, Shareholders and other
financial stakeholders.
CONTRAVENTION
Subject is not known to have contravened any existing local laws,
regulations or policies that prohibit, restrict or otherwise affect the terms
and conditions that could be included in the agreement with the subject.
FOREIGN EXCHANGE RATES
|
Currency |
Unit
|
Indian Rupees |
|
US Dollar |
1 |
Rs.41.01 |
|
|
1 |
Rs.81.74 |
|
Euro |
1 |
Rs.55.04 |
SCORE & RATING EXPLANATIONS
|
SCORE FACTORS |
RANGE |
POINTS |
|
HISTORY |
1~10 |
6 |
|
PAID-UP CAPITAL |
1~10 |
6 |
|
OPERATING SCALE |
1~10 |
6 |
|
FINANCIAL CONDITION |
|
|
|
--BUSINESS SCALE |
1~10 |
6 |
|
--PROFITABILIRY |
1~10 |
6 |
|
--LIQUIDITY |
1~10 |
6 |
|
--LEVERAGE |
1~10 |
6 |
|
--RESERVES |
1~10 |
6 |
|
--CREDIT LINES |
1~10 |
6 |
|
--MARGINS |
-5~5 |
- |
|
DEMERIT POINTS |
|
|
|
--BANK CHARGES |
YES/NO |
YES |
|
--LITIGATION |
YES/NO |
NO |
|
--OTHER ADVERSE INFORMATION |
YES/NO |
NO |
|
MERIT POINTS |
|
|
|
--SOLE DISTRIBUTORSHIP |
YES/NO |
NO |
|
--EXPORT ACTIVITIES |
YES/NO |
YES |
|
--AFFILIATION |
YES/NO |
YES |
|
--LISTED |
YES/NO |
YES |
|
--OTHER MERIT FACTORS |
YES/NO |
YES |
|
TOTAL |
|
54 |
This score serves as a reference to assess SC’s credit risk and
to set the amount of credit to be extended. It is calculated from a composite
of weighted scores obtained from each of the major sections of this report. The
assessed factors and their relative weights (as indicated through %) are as
follows:
Financial
condition (40%) Ownership
background (20%) Payment
record (10%)
Credit history
(10%) Market trend
(10%) Operational
size (10%)
RATING EXPLANATIONS
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
>86 |
Aaa |
Possesses an extremely sound financial base with the strongest
capability for timely payment of interest and principal sums |
Unlimited |
|
71-85 |
Aa |
Possesses adequate working capital. No caution needed for credit transaction.
It has above average (strong) capability for payment of interest and
principal sums |
Large |
|
56-70 |
A |
Financial & operational base are regarded healthy. General
unfavourable factors will not cause fatal effect. Satisfactory capability for
payment of interest and principal sums |
Fairly Large |
|
41-55 |
Ba |
Overall operation is considered normal. Capable to meet normal
commitments. |
Satisfactory |
|
26-40 |
B |
Unfavourable & favourable factors carry similar weight in credit consideration.
Capability to overcome financial difficulties seems comparatively below
average/normal. |
Small |
|
11-25 |
Ca |
Adverse factors are apparent. Repayment of interest and principal sums
in default or expected to be in default upon maturity |
Limited with
full security |
|
<10 |
C |
Absolute credit risk exists. Caution needed to be exercised |
Credit not
recommended |
|
NR |
In view of the lack of information, we have no basis upon which to
recommend credit dealings |
No Rating |
|