
MIRA
INFORM REPORT
|
Report Date : |
28.03.2007 |
IDENTIFICATION
DETAILS
|
Name : |
GREATER PHARMA LIMITED PARTNERSHIP |
|
|
|
|
Registered Office : |
46, 46/1-2 Charansanitwong 40 Road, Bangyikhan, Bangplad, Bangkok 10700 |
|
|
|
|
Country : |
Thailand |
|
|
|
|
Financials (as on) : |
31.12.2005 |
|
|
|
|
Date of Incorporation : |
1967 |
|
|
|
|
Com. Reg. No.: |
0103510003763 |
|
|
|
|
Legal Form : |
Limited Partnership |
|
|
|
|
Line of Business : |
Manufacturing various types
of pharmaceutical products,
diagnostic products, medicated
cosmetics products. |
RATING &
COMMENTS
|
MIRA’s Rating : |
Ba |
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
41-55 |
Ba |
Overall operation is considered normal. Capable to meet normal
commitments. |
Satisfactory |
|
Status : |
Good |
|
|
|
|
Payment Behaviour : |
Regular |
|
|
|
|
Litigation : |
Clear |
name
GREATER PHARMA LIMITED PARTNERSHIP
SUMMARY
BUSINESS ADDRESS : 46, 46/1-2 CHARANSANITWONG 40 ROAD,
BANGYIKHAN, BANGPLAD,
BANGKOK 10700,
THAILAND
TELEPHONE : [66] 2433-0061-6,
883-1890-, 2433-0256
FAX : [66] 2433-0076
E-MAIL ADDRESS : greater@mozard.net.co.th
REGISTRATION ADDRESS : SAME
AS BUSINESS ADDRESS
ESTABLISHED : 1967
REGISTRATION NO.
: 0103510003763 [Former : 381/2510]
CAPITAL
REGISTERED : BHT.
60,000,000
CAPITAL
PAID-UP : BHT. 60,000,000
FISCAL
YEAR CLOSING DATE : DECEMBER 31
LEGAL STATUS : LIMITED PARTNERSHIP
EXECUTIVE : MR.
CHERNPORN TENGAMNUAY, THAI
MANAGING PARTNER
NO. OF
STAFF : 350
LINES OF
BUSINESS : PHARMACEUTICALS
MANUFACTURER, IMPORTER,
DISTRIBUTOR
& EXPORTER
CORPORATE PROFILE
OPERATING TREND : STABLE
PRESENT SITUATION : OPERATING NORMALLY
REPUTATION : GOOD
WITH NORMAL BUSINESS
ENGAGEMENT
MANAGEMENT STANDARD : MANAGEMENT WITH GOOD PERFORMANCE
HISTORY
The subject was
established on March
23, 1967 as
a limited partnership
under the name
style GREATER PHARMA
LIMITED PERTNERSHIP by
Mr. Kian Tengamnuay,
a Thai, who
is the founder and the
chairman of Ouiheng
Group, with the
business objective to
manufacture wide range
of pharmaceutical products and distribute to both
in domestic and
international markets. It
currently employs 350
staff.
At present, Mr.
Chernporn Tengamuay &
Miss Duangjai Tengamnuay
bear full financial
responsibilities by law.
The subject also
received awarded the
GMP Certificate from
the Thai Foods
& Drugs Administration [TFDA], Ministry of
Health.
The subject’s registered
address is 46, 46/1-2 Charansanitwong 40 Road, Bangyikhan,
Bangplad, Bangkok 10700,
and this is
the company’s current
operation address.
MANAGEMENT
Mr. Chernporn
Tengamnuay is the Managing Partner
& Factory Manager.
He is Thai nationality with
the age of
49 years old.
Mrs. Daojai
Tengamnuay is the Finance Manager.
She is Thai nationality with
the age of
43 years old.
BUSINESS OPERATIONS
The subject’s activity
is a manufacturing various types
of pharmaceutical products,
diagnostic products, medicated
cosmetics products, under
its owned trademark,
such as “Madaus”, “Daiichi”,
“Seba Med”, “Ulgastrin”, “Greater” and etc. The
company also produce
pharmaceutical products for
oversea companies as
the followings:
Daiichi Pharmaceutical Co.,
Ltd. : Japan
Dolorgiet GmbH &
Co., Ltd. : Germany
Lab. Therapeutique Moderne : France
Madaus A.G. : Germany
PRODUCTION CAPACITY
Tablet : 300
million tablet/year
Capsule : 30
million tablet/year
Liquid Drug : 60,000
litr/year
Balm & Cream
Drug : 12,000
kg./year
The subject is
also an agent
of finished
pharmaceutical products from
the companies as
followings:
Daiichi Pharmaceutical Co.,
Ltd. : Japan
Lab Therapeutique Moderne : France
Seratec GmbH : Germany
PRODUCT PROFILE
|
Product |
Product-Prescription |
Unit Size
|
|
|
|
|
|
A.C.V. Cream |
Acyclovir |
5.0 g. |
|
Aminophyline Tablet |
Aminophylline |
100 mg. |
|
Amoxcillin 125 Dry
Syrup |
Amoxcillin |
125 mg. |
|
Amoxcillin 250 Capsule |
Amoxcillin 250 mg. |
1000’s |
|
Amoxcillin 500
Capsule |
Amoxcillin 500 mg. |
500’s |
|
Amoxcillin 250 Capsule
|
Ampicillin 250 mg. |
1000’s |
|
|
Ampicillin 1000 mg. |
|
|
B-Feron Coated Tablet |
B-Complex Vitamins Plus Ferrous |
50x10’s |
|
Baby Cough Syrup |
Ammonium Choride |
0.05 g. |
|
Benzo Ointment |
Benzoic acid 6.0
g. Salicylic acid 4.0
g. |
Zinc Oxide 10.0 g. |
|
Bethasone-N Cream |
Betamethasone Valerater |
0.1% |
|
Brewers Yeast Tablet [300mg.] |
Dried Yeast 300
mg. |
1000’s |
|
Cefalec 500 Capsule |
Cephalexin Capsul |
10x10’s |
|
Ammonium Chloride 125.0 mg. |
Sodium Citrate 50.0
mg. |
160ml |
|
Cyheptine Tablet |
Cyproheptadine HCI 4 mg. |
1000’s |
|
Defanol Capsule |
Ethisterone 100 mg. |
|
|
|
Ethinyloestradiol 500 mcg. |
|
|
|
Cyanocobalamin 100 mcg. |
|
|
Terpin Hydrate 125
mg. |
Chlorpheniramine Maleate 2 mg. |
1000’s |
|
Diapam 2 Tablet |
Diazepam 2 mg. |
|
|
Dexpin Tablet |
Dextromethorphan HBr. 10 mg. |
|
|
Doremol Syrup |
Paracetamol 120 mg. |
|
|
Finisol Suspension |
Astemizole 5.0 mg. |
|
|
Finisol Tablet |
Astemizole 10 mg. |
500’s |
|
Flunarium Capsule |
Flunarizine 5 mg |
1000’s |
|
Fungiderm Cream |
Clotrimazole 1% |
5 g. |
|
Gastab Table |
|
|
|
Sodium Bicarbonate 195 mg |
Sodium Bicarbonate 65 mg |
1000’s |
|
Greatofen 400 |
Ibuprofen 400 mg. |
50x10’s |
|
Griseofuevin Tablet |
Griseofuevin 125 mg |
500’s |
|
Hadarax Tablet |
Hydroxyzine HCI 10 mg. |
500’s |
|
Hiscifed Syrup |
Triprolidine HCI 1.25 mg. |
60ml. |
|
Pseudoephedrine HCI 30.0 mg. |
1 Gallon |
|
|
Hiscolgen Syrup |
Paracetamol 300 mg. |
|
|
Indomed Capsule |
Indomethacin 25.0 mg |
|
|
Kenazole Tablet |
Ketoconazole 200 mg. |
|
|
Klinna Solution |
Clindamycin Phosphate 1% |
|
|
Liquimed Shampoo |
Ketoconazole 2 gm. |
|
|
Miformin Tablet |
Metformin HCI 500
mg. |
|
|
Mucolid Syrup |
Ambroxol 30 mg. |
|
|
Mybacin Dermic |
Neomycin base 0.50
gm. |
|
|
|
Bacitracin 25,000 IU |
|
|
Product |
Product-Prescription |
Unit Size
|
|
Mysocort Cream |
Miconazole Nitrate 2.0
g. |
|
|
Mysoven Granules 3 gm. |
Acetyleysteine 200 mg. |
|
|
Mytrocin Ointment |
Nitrofurazone 20 mg. |
|
|
Navamed Tablet |
Dimenhydrinate 50 mg. |
|
|
Nicotinic Acid Tablet |
Nicotinic acid 50
mg. |
|
|
Ohexine Tablet |
Bromhexine HCI 8
mg. |
|
|
Opredsone Capsule |
Prednisolone 5 mg. |
|
|
Oxymet |
Oxymetazoline 0.05% |
|
|
Oxytetracycline HCI Capsule
|
Oxytetracycline HCI 250
mg. |
|
|
Paramol Table |
Paracetamol 500 mg. |
|
|
Propylthioruacil Table |
Propylthiouracil 50 mg. |
|
|
Proxen 250 Tablet |
Naproxen 250 mg. |
|
|
Quinidine Sulfate Table |
Quinidine Sulfate 200
mg. |
|
|
Roxycam Capsule |
Piroxicam 10 mg. |
|
|
|
Piroxicam 20 mg. |
|
|
Semeth 40 Tablet |
Simethicone 40 mg. |
|
|
Silicin Tablet |
Cinarizine 25 mg. |
|
|
Stomac Tablet |
Atropine Sulfate 0.13
mg. |
|
|
|
Dried AI [OH] 3 Gel 325 mg. |
|
|
|
Magnesium Trisilicate 260
mg. |
|
IMPORT [COUNTRIES]
Raw materials &
finished products are
imported from Japan,
France and Germany.
SALES [LOCAL]
95% of the products is
sold locally to
wholesalers and Thai
Government.
EXPORT [COUNTRY]
5% of pharmaceutical
products is exported
to Vietnam, Myanmar,
Malaysia, Laos and
Middle East countries.
SUBSIDIARIES &
AFFILIATED COMPANY
Ouiheng Group
of Companies are consisted of
the Followings:
Ouiheng Import
Co., Ltd.
Importer of pharmaceutical
Greater Poly
Manufacturing Co., Ltd.
Manufacturer of medicated
cosmetic products
Grand Poly
Industry Co., Ltd.
Manufacturer of pharmaceutical plastic containers
Ouiheng Health
Consumer Co., Ltd.
Distributor of consumer
products
Grand Interprint
Co., Ltd.
Offset printing house
ASSOCIATIONS
Thai Pharmaceutical Manufacturer Association
Franco - Thai Chamber of
Commerce
The Federation of
Thai Industries
LITIGATION
Bankruptcy and Receivership
There are no
litigation on bankruptcy
and receivership cases
filed against the
subject found at
Legal Execution Department
for the past
five years.
Others
There are no
legal suits filed
against the subject for
the past two
years.
CREDIT
The subject sells
on the credits term of
30-60 days.
Local bills are
paid on the
credits term of
30-60-90 days.
Imports are by
L/C on the credits term
of 90-180 days or
D/A on the credits o f 90-180
days & T/T.
Exports are against
L/C at sight or T/T.
BANKING
Kasikornbank Public
Co., Ltd.
[Bangyikhan Branch, Charansanitwong Rd., Bangyikhan, Bangplad,
Bangkok]
Bangkok Bank
Public Co., Ltd.
[Pinklao Branch,
Charansanitwong Rd., Bangyikhan,
Bangplad, Bangkok]
EMPLOYMENT
The subject employs
approximately 350 staff.
LOCATION DETAILS
The premise is
owned for operating
office & factory
in a 5 storey building
of 3 row shop houses
at the heading address in
commercial/residential area.
NOTE
Refer to your
given name “Greather Pharma
Group Basngyikhan, please
be informed that
the correct subject’s
name is “Greater Pharma
Limited Partnership”
COMMENT
The company has
strong market position
in the pharmaceutical
business, professional management
term as well
as diversification into
the cosmetic business.
The plant is staffed by a
team of highly
qualified and well
experienced personnel. Quality
is in-built into
the products. Starting
with vendor approval,
analysis of raw materials,
in process checks
and finally finished
products analysis, various
tests are performed
to ensure that
the products meet
stringent international specifications. As well new
technology has always
been introduced and
developed, vice versa
the production process.
At the present the
company’s business became
well-know and grew
to be a leading pharmaceutical producers both for
local and international markets.
FINANCIAL INFORMATION
The initial registered
capitalization was Bht.
3.6 million.
Increased were : Bht. 9.62 million in
1984 [October]
Bht.
16.62 million in 1985
Bht.
20.62 million in 1987 [June]
It was held by 8
partners viz:
Name Age
Mr. Thienchai Tengamnuay
47 Bht.
2,636,000
Mrs. Pasana Tengamnuay 67 Bht.
3,970,000
Mr. Chernporn Tengamnuay 43 Bht.
2,472,000 [Unlimited Partner]
Mr. Kian Tengamnuay 75 Bht.
4,830,000
Mr. Pakphoom Tengamnuay 40 Bht.
2,274,000
Mr. Pruedsakorn Tengamnuay 39 Bht.
2,228,000
Miss Daowjai Tengamnuay
37 Bht.
2,090,000 [Unlimited Partner]
Mrs. Chatchada Tengamnuay 42 Bht. 120,000
On Mar 30, 2004, capital
was increased to
Bht. 60,000,000, which
was carried by
14 persons as
followed:
Name Age
Mrs. Pasana Tengamnuay 74 Bht. 21,000,000
Mr. Thienchai Tengamnuay 51 Bht. 3,000,000
Mr. Chernporn Tengamnuay 49 Bht. 16,200,000 [unlimited partner]
Mr. Pakphoom Tengamnuay 47 Bht. 9,000,000
Mr. Prusakorn Tengamnuay 46 Bht. 3,000,000
Ms. Daojai Tengamnuay 43 Bht.
3,000,000 [unlimited partner]
Mrs. Chatchada Tengamnuay 44 Bht. 600,000
Mrs. Suwannee Tengamnuay 43 Bht. 600,000
Mr. Paran Tengamnuay 23 Bht. 600,000
Mr. Thanach Tengamnuya 20 Bht. 600,000
Mr. Thanit Tengamnuay 19 Bht.
600,000
Ms. Thachaya Tengamnuay 16 Bht. 600,000
Ms. Chanya Tengamnuay 16 Bht. 600,000
Master Chanawin Tengamnuay 15 Bht. 600,000
NAME OF
AUDITOR & CERTIFIED
PUBLIC
ACCOUNTANT NO.
Mr. Poonsak Saengsan
No. 1910
BALANCE SHEET [BAHT]
The
latest financial figures
published for December
31, 2005 &
2004 were:
ASSETS
|
Current Assets |
2005 |
2004 |
|
|
|
|
|
Cash in hand
& at Bank
|
4,264,872.23 |
3,390,123.06 |
|
Trade Accts. & Notes Receivable |
2,439,537.97 |
2,246,385.62 |
|
Trade Accts.
Receivable-Related Company |
197,780,714.09 |
163,223,072.69 |
|
Inventories |
36,703,279.78 |
44,596,318.71 |
|
Other Current Assets
|
3,875,347.45 |
3,391,540.91 |
|
Total Current
Assets |
245,063,751.52 |
216,847,440.99 |
|
Cash at Bank
under Commitment |
1,831,794.27 |
1,831,794.27 |
|
Long Term Investment |
150,000.00 |
150,000.00 |
|
Investment in Related
Company |
800,000.00 |
800,000.00 |
|
Fixed Assets |
21,998,685.79 |
25,901,400.80 |
|
Other Assets |
1,098,170.45 |
4,493,352.32 |
|
Total Assets |
270,942,402.03 |
250,023,988.38 |
LIABILITIES &
SHAREHOLDERS' EQUITY [BAHT]
|
Current Liabilities |
2005 |
2004 |
|
|
|
|
|
Bank Overdraft &
Short Term Loan
from Financial Institute |
50,545,088.76 |
30,227,333.98 |
|
Trade Accts. Payable |
50,193,291.07 |
49,677,425.19 |
|
Short Term Loan
from Related Company |
104,381,182.72 |
119,681,182.72 |
|
Accrued Expenses |
7,208,224.27 |
8,827,683.70 |
|
Other Current Liabilities |
6,217,923.78 |
3,570,393.25 |
|
Total Current
Liabilities |
218,545,710.60 |
211,984,018.84 |
|
Long Term Loan
from Financial Institute |
2,326,854.00 |
3,682,854.00 |
|
Total Liabilities |
220,872,564.60 |
215,666,872.84 |
|
|
|
|
|
Shareholders'
Equity |
|
|
|
Capital Paid |
60,000,000.00 |
60,000,000.00 |
|
Retained Earning -
Unappropriated |
[9,930,162.57] |
[25,642,884.46] |
|
Total
Shareholders' Equity |
50,069,837.43 |
34,357,115.54 |
|
Total
Liabilities & Shareholders' Equity |
270,942,402.03 |
250,023,988.38 |
PROFIT &
LOSS ACCOUNT
|
Revenue |
2005 |
2004 |
|
|
|
|
|
Sales |
168,057,934.32 |
148,807,259.99 |
|
Services |
5,219,725.52 |
3,955,791.72 |
|
Interest Income |
43,938.51 |
11,063.61 |
|
Other Income |
66,220,825.16 |
77,760,231.04 |
|
Total Revenues
|
239,542,423.51 |
230,534,346.36 |
|
Expenses |
|
|
|
|
|
|
|
Cost of Goods
Sold |
124,607,200.41 |
113,941,412.90 |
|
Selling & Administrative Expenses |
95,249,944.21 |
110,106,429.43 |
|
Total Expenses |
219,857,144.62 |
224,047,842.33 |
|
Profit / [Loss] Before Interest
Expenses |
19,685,278.89 |
6,486,504.03 |
|
Interest Expenses |
[3,972,557.00] |
[5,602,494.46] |
|
Net Profit / [Loss] |
15,712,721.89 |
884,009.57 |
FINANCIAL ANALYSIS
Annual Growth
& Profitability
|
Annual Growth |
2005 |
2004 |
|
Net Sales |
13.43 |
|
|
Operating Profit |
167.55 |
|
|
Net Profit |
1,677.44 |
|
|
Fixed Assets |
[15.07] |
|
|
Total Assets |
8.37 |
|
|
Profitability |
|
|
|
Cost of Good Sold |
71.91 |
74.59 |
|
Operating Profit Margin |
11.36 |
4.25 |
|
S & A
Expenses |
54.97 |
72.08 |
|
Interest Paid |
2.29 |
3.67 |
|
Other Revenues |
38.24 |
50.91 |
|
Net Profit Margin |
9.07 |
0.58 |
Net Sales was increased by
13.43%, Operating Profit
was increased by
167.55%, Net Profit
was increased by
1,677.44%, Fixed Assets
was decreased by
15.07%, Total Assets
was increased by
8.37%,
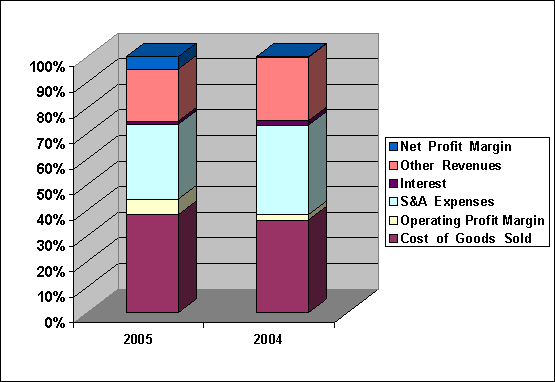
Compare each cost
with sales, Cost of Good Sold
was decreased from
74.59% to 71.91%,
Operating Profit Margin
was increased from
4.25% to 11.36%,
S & A
Expenses was decreased
from 72.08% to
54.97%, Interest Paid was
decreased from 3.67%
to 2.29%, Other
Revenues was decreased
from 50.91% to
38.24%, Net Profit
Margin was increased
from 0.58% to
9.07%.
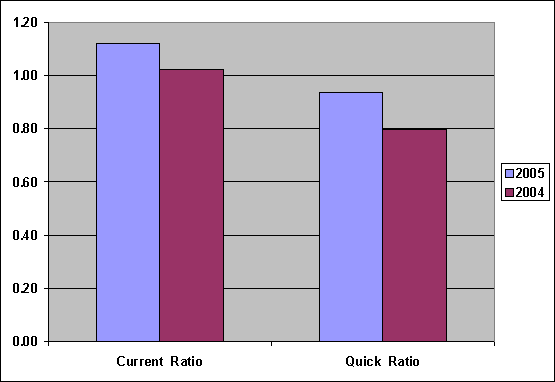
Liquidity
|
Liquidity |
2005 |
2004 |
|
Current Ratio |
1.12 |
1.02 |
|
Quick-Assets Ratio |
0.94 |
0.80 |
Current Ratio was
increased from 1.02
to 1.12, show
Current Assets can
cover Current Liabilities, Liquidity Ratio of
the company was
good, Quick-Assets Ratio
was increased from
0.80 to 0.94,
capacity to pay
Short Term Loan
was not good,
because Current Assets
without Inventory can
not cover Current
Liabilities.
Leverage
|
Leverage |
2005 |
2004 |
|
D/E Ratio |
4.41 |
6.28 |
|
Times Interest Earned |
4.96 |
1.16 |
D/E Ratio was decreased from
6.28 to 4.41,
show the company
had fund from
Loan more than
fund from Equity,
investment risked of
the company is
high, Times Interest
Earned was increased
from 1.16 to
4.96, capacity to
pay interest paid
was good.

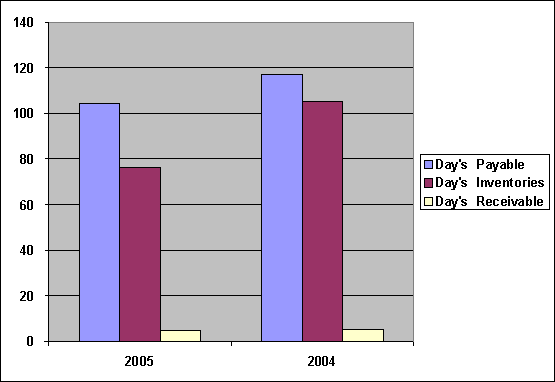
Efficiency
|
Efficiency |
2005 |
2004 |
|
Fixed Assets Turnover |
7.88 |
5.90 |
|
Total Assets Turnover |
0.64 |
0.61 |
|
Return on Assets |
5.80 |
0.35 |
|
Return on Equity |
31.38 |
2.57 |
|
Day's Payable |
104.28 |
117.07 |
|
Day’s Inventories |
76.25 |
105.10 |
|
Day’s Receivables |
5.07 |
5.29 |
Fixed Assets Turnover
was increased from
5.90 times to
7.88 times, Total
Assets Turnover was
increased from 0.61
times to 0.64
times, show the
company could use
Assets so efficiently, Return on Assets
was increased from
0.35 to 5.80,
Return on Equity
was increased from
2.57 to 31.38,
capacity to use
Assets to make
profit was good,
Day's Payable was
decreased from 118
days to 105
days, and Day’s
Inventories was decreased
from 106 days
to 77 days, while Day’s
Receivables was stable
at 6 days.
RATING
EXPLANATIONS
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
>86 |
Aaa |
Possesses an extremely sound financial base with the strongest
capability for timely payment of interest and principal sums |
Unlimited |
|
71-85 |
Aa |
Possesses adequate working capital. No caution needed for credit transaction.
It has above average (strong) capability for payment of interest and
principal sums |
Large |
|
56-70 |
A |
Financial & operational base are regarded healthy. General unfavourable
factors will not cause fatal effect. Satisfactory capability for payment of
interest and principal sums |
Fairly Large |
|
41-55 |
Ba |
Overall operation is considered normal. Capable to meet normal
commitments. |
Satisfactory |
|
26-40 |
B |
Unfavourable & favourable factors carry similar weight in credit
consideration. Capability to overcome financial difficulties seems
comparatively below average/normal. |
Small |
|
11-25 |
Ca |
Adverse factors are apparent. Repayment of interest and principal sums
in default or expected to be in default upon maturity |
Limited with
full security |
|
<10 |
C |
Absolute credit risk exists. Caution needed to be exercised |
Credit not
recommended |
|
NR |
In view of the lack of information, we have no basis upon which to
recommend credit dealings |
No Rating |
|
This score serves as a reference to assess SC’s credit risk
and to set the amount of credit to be extended. It is calculated from a composite
of weighted scores obtained from each of the major sections of this report. The
assessed factors and their relative weights (as indicated through %) are as
follows:
Financial
condition (40%) Ownership
background (20%) Payment
record (10%)
Credit history
(10%) Market trend
(10%) Operational
size (10%)