
MIRA
INFORM REPORT
|
Report Date : |
28.03.2007 |
IDENTIFICATION
DETAILS
|
Name : |
THE GOVERNMENT PHARMACEUTICAL ORGANIZATION |
|
|
|
|
Registered Office : |
75/1 Rama Vi
Road, Ratchathewi,
Bangkok 10400 |
|
|
|
|
Country : |
Thailand |
|
|
|
|
Financials (as on) : |
30.09.2005 |
|
|
|
|
Date of Incorporation : |
1941 |
|
|
|
|
Legal Form : |
State Enterprise |
|
|
|
|
Line of Business : |
Production of medicines and
medical supplies Promotion
of study &
research of local
raw materials to
be made into
pharmaceutical & medicinal productions. |
RATING &
COMMENTS
|
MIRA’s Rating : |
Ba |
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
41-55 |
Ba |
Overall operation is considered normal. Capable to meet normal
commitments. |
Satisfactory |
|
Status : |
Satisfactory |
|
|
|
|
Payment Behaviour : |
Usually correct |
|
|
|
|
Litigation : |
Clear |
name
THE GOVERNMENT PHARMACEUTICAL ORGANIZATION
SUMMARY
BUSINESS ADDRESS : 75/1
RAMA VI ROAD,
RATCHATHEWI,
BANGKOK 10400,
THAILAND
TELEPHONE : [66] 2203-8000,
2354-8800-2
FAX : [66] 2203-8929
E-MAIL ADDRESS : dr-mkj@gpo.or.th
ESTABLISHED : 1941
FISCAL YEAR
CLOSING DATE : SEPTEMBER 30
LEGAL STATUS : STATE ENTERPRISE
EXECUTIVE : LT. GEN. MONGKOL
JIVASANTIKARN, THAI
MANAGING DIRECTOR
NO. OF
STAFF : 1,956
LINES OF
BUSINESS : MEDICINE
AND MEDICAL SUPPLIES
MANUFACTURER
CORPORATE PROFILE
OPERATING TREND : STABLE
PRESENT SITUATION : OPERATING
NORMALLY
REPUTATION : GOOD
FOR NORMAL BUSINESS
ENGAGEMENT
MANAGEMENT STANDARD : MANAGEMENT
WITH FAIR PERFORMANCE
HISTORY
The
subject was established
in 1941 as
a state enterprise
under the jurisdiction of the Ministry
of Public Health,
standing as the
only pharmaceutical manufacturer owned by the Government, has responsibilities in
producing high quality
drugs at reasonable
prices, serving the
Ministry of Public
Health’s policy on
the “Health for
All” campaign etc.
It currently employs
1,956 staff.
The
subject has received
ISO 9002 Certification certified by the
Bureau Veritas Quality
International Co., Ltd. [BVQI], the international standard
recognizing body, for
Injection Quality Systems
with effect from
February 28, 1997.
The ISO 9002
Certificate absolutely guarantees
its products’ standard
quality.
The subject’s business address is at 75/1 Rama VI Rd., Ratchathewi, Bangkok 10400.
THE BOARD OF DIRECTORS
|
Name |
|
Nationality |
Age |
|
|
|
|
|
|
Mr. Vithit Leenutaphong |
: Chairman |
Thai |
53 |
|
Dr. Vichai Tienthavorn |
|
Thai |
60 |
|
Dr. Prat
Boonyawongvirot |
|
Thai |
58 |
|
Dr. Narongsakdi Aungkasuvapala |
|
Thai |
59 |
|
Prof. [Special] Pakdee Pothisiri |
|
Thai |
61 |
|
Dr. Thawat Suntrajarn |
|
Thai |
60 |
|
Dr. Vichai Chokevivat |
|
Thai |
61 |
|
Dr. Paijit Warachit |
|
Thai |
56 |
|
Pro. Piyasakol Sakolsatayadorn |
|
Thai |
60 |
|
Dr. Satian Pooprasert |
|
Thai |
56 |
|
Dr. Siriporn Kanshana |
|
Thai |
58 |
|
Mrs. Benja Louicharoen |
|
Thai |
55 |
|
Dr. Apichai Mongkol |
|
Thai |
52 |
|
Mr. Thatree Kienthong |
|
Thai |
54 |
|
Lt. Gen. Mongkol Jivasantikarn |
|
Thai |
59 |
MANAGEMENT
Lt. Gen. Mongkol Jivasantikarn : Managing
Director
Mr.
Wanchai Subhachaturus : Deputy
Managing Director
Mrs.
Indanima Kunjara Na
Ayudhya : Deputy Managing
Director
Mr.
Dhanavadhana Thongpradist : Deputy
Managing Director
Mr.
Manoon Bupparenoo : Deputy
Managing Director
Mr.
Somchai Srichainak : Deputy
Managing Director
Mr.
Sirisak Vipavaphanich : Senior
Expert 11
Mr.
Kamchorn Balangura : Acting
Senior Export.
Mr.
Tamnu Chantorn : Assistant
Managing Director
Ms.
Anchalee Utthangkorn : Director, Internal Audit Office
Mr.
Pipat Niyomkarn : Human
Resources Department
Ms.
Valairat Anuketra : Expert
10
Mrs.
Saisumpun Suwachrungoon : Director,
Biological Production Department
Mr. Jenjob Srisak : Director, Engineering & Technology Department
Mrs.
Varunee Lekcharoenvong : Director,
Accounting & Finance
Department
Mr.
Suttipan Vanasup : Director,
Chemical Production Department
Mr.
Soontorn Vorakul : Director,
Production Department
Ms.
Pisamorn Klinsuiwan : Director,
Research & Development
Institute
Ms.
Viyada Jurt-Amphai : Director,
Quality Assurance Department
Mrs.
Suchada Subhachaturus : Director,
Logistics & Supply
Chain
Management Department
Mrs.
Sujida Chutima : Director,
Marketing & Sales
Department
BUSINESS OPERATIONS
The
subject’s main activities
are as follows:
-
Production of medicines
and medical supplies
-
Promotion of study
& research of
local raw materials
to be made into pharmaceutical & medicinal
productions.
-
Controlling the quality
of productions to
be standardized
-
Controlling price level
-
Reserve stocks to
prevent shortage in
case of emergencies
& disaster.
Production
The
subject has total
amount of production cost
at Bht. 2,105.20
million, which was Bht.
37.36 million more
compared to 2002, or
1.81%. The products
are categorized as
follows:
Type of
Products Cost of
Production Amount
1. Household Medicines : Bht.
162.50 Million
2. Medicines : Bht.
1,501.50 Million
3. Biological Products : Bht.
367.11 Million
4. Chemical Products : Bht.
74.09 Million
The
subject’s total sales
for 2003 were
Bht. 3,677.02 million,
an increase of Bht.
349.85 million or
10.51% from 2002.
Sales of products
manufactured by the
subject amounted to
Bht. 3,487.37 million,
a Bht. 364.66 million increase
from 2002, or 11.68% increase.
IMPORT [COUNTRIES]
50%
of the chemicals is imported
from Republic of
China, Japan, France,
Singapore and Spain,
while remaining 50%
is purchased from
local suppliers.
SALES [LOCAL]
90%
of its products is sold
to wholesalers, private
hospitals and Thai
Government such as
public hospitals.
EXPORT [COUNTRIES]
10%
of its products is exported
to Myanmar, Laos,
Singapore, Philippines, Vietnam,
Malaysia and Cambodia.
JOINT VENTURE
COMPANIES
General Hospital
Products Public Co.,
Ltd.
Address
: 101/99
Soi Nava Nakorn
7, Paholyothin Rd.,
Khlong Luang District,
Pathum
Thani 12120
Core Business : Saline solution,
dialysis solution, dental
supplies
Holding :
49.6%
of investment by
the subject.
United Pharma
Antibiotics Industry Co.,
Ltd.
Address
: 101/52
Nava Nakorn Industrial
Estate, Phaholyothin Rd., Khlong
Luang
District, Pathum Thani 12120
Core Business : Raw materials
used for production
of anti-biotics: cloxacillin,
amoxicillin and dicloxacillin
Holding : 45%
of investment by
the subject.
Thai Wattana
Pharmaceutical Dextrose Co.,
Ltd.
Address : 112
Moo 1 Banglamung-Rayong
Rd., Banglamung District,
Chon
Buri 20150
Core Business : Dextrose sugar
for production of
food and drugs
Holding : 30%
of investment by
the subject.
Thai Herbal
Products Co., Ltd.
Address : Factory
Land Wangnoi, 130/49
Moo 3, Wangnoi District,
Ayudhya
13170
Core Business : Herbal remedies
Holding : 49%
of investment by
the subject.
Government Pharmaceutical Organization – Merieux
Biological Products Co., Ltd.
Address : Gateway
City Industrial Estate,
241 Moo 7,
Tambon Hua Samrong,
Plangyao District,
Chachoengsao 24190
Core Business : Human vaccinations
Holding : 49%
of investment by
the subject.
Medical Device
Manufacturer [Thailand]
Co., Ltd.
Address : In
the process of
building a plant
Core Business : Manufacture and
sells syringes and
safety hypodermic syringes
Holding : 13% of
investment by the
subject.
Gpo-Merieux Biologicals
Co., Ltd.
Address : 241
Moo 7, Huasamrong, Plaengyao, Chachoengsao 24190.
Core
Buisess : Human
vaccinations
Holding : 49%
of investment by
the subject
CREDIT
Local
bills are paid
by cash or
on the credits term of
30-60 days.
Imports
are by L/C at sight
or on the credits term
of 90-120 days
and T/T.
Sales
are by cash or on
the credits term
of 60-90 days.
Exports
are against L/C
at sight and
T/T.
BANKING
Krung Thai
Bank Public Co.,
Ltd.
[Head Office :
35 Sukhumvit Rd.,
Klongtoeynua, Watana, Bangkok
10110]
Bangkok Bank
Public Co., Ltd.
[Head Office :
333 Silom Rd.,
Bangrak, Bangkok 10500]
Kasikornbank Public
Co., Ltd.
[Head Office :
1 Kasikorn Lane,
Rajburana Rd., Rajburana,
Bangkok 10140]
EMPLOYMENT
The
subject employs 1,956
staff [office staff,
sales staff and
factory workers].
LOCATION DETIALS
The
premise is owned
for operating an
office, drug store,
warehouse and factory
at the heading address in
commercial area. The
ground space is
measured at 64,000
square meters and
the floor space
is at 45,500 square meters.
Its
branches are located
at follows:
Northeast
Regional Branch in
Udon Thani Province
North
Regional Branch in
Chiang Mai Province
South
Regional Branch in
Songkla Province
Moreover,
the subject has
11 drugs stores
under the name
“The Government Pharmaceutical Organization” located at;
Rama
6 Rd.,
Rajthevee
Rd.,
Yosse
Rd.,
Charansanitwong Rd.,
Theves
Rd.,
Rangsit
Rd.,
Bangrak
Wattana
Jomthong
Pravet
Nonthaburi
COMMENT
Though
the medicine market
tends to expand
continuously, there are
many factors that
could affect to
the growth of
the market, like
the high competition
among domestic manufacturers, the market share
taking from import
medicine, and the
effect from FTA
which will be
applied gradually. Therefore, it is necessary
that Thai manufacturers in medical industry
should realize and
prepare themselves rapidly
for the situation
that could be
changed in the
future.
Thus, GPO
increases the investment
in research and
development in order
to create the
new innovation for
the products and
at the same time, GPO
also upgrades the
manufacturing standard to
comply with the
world standard which
can be considered as the
important factor that
drives the organization to grow strongly
and can compete
in international level.
FINANCIAL INFORMATION
The
details of capital
and shareholders are
not available due
to the fact that the
subject is regarded
as a state enterprise and
is funded and
operated under the
supervision of the
Ministry of Public
Heath.
NAME OF
AUDITOR & CERTIFIED
PUBLIC
ACCOUNTANT NO.:
Mrs. Sukhon Suksri
: Director of
Auditors-in-Charge No. 8,
Office of the Auditor
General [acting]
Ms. Fuengfa Thienprapasith
: Auditor-in-Charge No. 8
BALANCE SHEET [BAHT]
The latest financial figures published as at
September 30, 2005
& 2004 were:
ASSETS
|
Current Assets |
2005 |
2004 |
|
|
|
|
|
Cash and Cash
Equivalents |
2,041,923,745.31 |
1,399,379,630.29 |
|
Temporary Investment |
1,011,410,966.52 |
787,153,858.60 |
|
Trade Accounts Receivables |
1,009,859,064.66 |
1,167,446,906.74 |
|
Other Accounts Receivables |
10,821,617.22 |
12,054,746.20 |
|
Inventories |
1,050,504,333.85 |
1,116,808,067.80 |
|
Other Current Assets
|
57,035,757.66 |
35,652,184.92 |
|
|
|
|
|
Total Current
Assets |
5,181,555,485.22 |
4,518,495,394.55 |
|
Investments in Affiliated Companies |
494,960,000.00 |
504,956,146.43 |
|
Loans |
56,350,145.00 |
64,684,954.00 |
|
Fixed Assets |
1,403,301,260.29 |
1,410,076,436.97 |
|
Assets in Progress |
17,747,150.47 |
15,893,556.45 |
|
Drugs & Medical
Supply Reserve Project
|
20,697,422.95 |
28,966,980.22 |
|
Total Assets |
7,174,611,463.93 |
6,543,073,468.62 |
LIABILITIES &
SHAREHOLDERS' EQUITY [BAHT]
|
Current Liabilities |
2005 |
2004 |
|
|
|
|
|
Trade Accounts Payable |
380,773,918.59 |
408,911,537.37 |
|
Accrued Liabilities |
144,378,034.57 |
179,093,235.90 |
|
Withholding Income Tax |
3,816,742.71 |
5,045,593.09 |
|
Contract Guarantee |
24,588,452.93 |
18,272,440.00 |
|
Other Payable |
13,489,231.46 |
39,157,482.04 |
|
|
|
|
|
Total Current
Liabilities |
567,046,380.26 |
650,480,288.40 |
|
Employee Provident Fund |
285,095,332.25 |
260,379,542.50 |
|
GPO Financing Fund |
5,000,000.00 |
- |
|
Total Liabilities |
857,141,712.51 |
910,859,830.90 |
|
|
|
|
|
Capital |
|
|
|
|
|
|
|
Paid-up Capital |
48,041,467.48 |
48,041,467.48 |
|
Capital Budget Allocations |
3,018,502.04 |
3,018,502.04 |
|
Capital Transfer Payments
from Current Capital |
3,526,548.46 |
3,526,548.46 |
|
|
54,586,517.98 |
54,586,517.98 |
|
|
|
|
|
Premium of Share
Capital from Donations |
16,284,640.96 |
16,615,538.45 |
|
Drugs & Medical
Supply Reserve Program |
49,914,409.60 |
49,914,409.60 |
|
Accumulated Profit |
6,196,684,182.88 |
5,511,097,171.69 |
|
Total Capital |
6,317,469,751.42 |
5,632,213,637.72 |
|
Total
Liabilities & Capital |
7,174,611,463.93 |
6,543,073,468.62 |
PROFIT &
LOSS ACCOUNT
|
Operating Revenues |
2005 |
2004 |
|
|
|
|
|
Sales of Pharmaceutical and Medical Supplies |
4,463092,766.79 |
3,918,274,509.15 |
|
Income from Outsourcing |
2,704,065.38 |
2,304,277.97 |
|
Total Operating
Revenues |
4,465,796,832.17 |
3,920,578,787.12 |
|
Cost of
Sales & Operating Expenses |
|
|
|
|
|
|
|
Cost of Sales
and Outsourcing |
2,424,489,795.60 |
2,296,426,319.77 |
|
Cost of Sales
and Administration |
1,061,890,053.95 |
866,325,776.15 |
|
Other Costs |
45,225,138.54 |
11,412,690.81 |
|
Total Cost of
Sales & Operating Expenses |
3,531,604,988.09 |
3,174,164,786.73 |
|
|
|
|
|
Operating Profit |
934,191,844.08 |
746,414,000.39 |
|
Other Income |
54,825,167.11 |
37,567,203.58 |
|
|
|
|
|
Net Profit |
989,017,011.19 |
783,981,203.97 |
|
Retained Earnings Before
Adjustments at the Beginning
of the Year |
5,497,179,051.51 |
4,954,204,479.84 |
|
Less: Adjustments
after Accumulated Impacts on Changes
in Accounting |
13,918,120.18 |
13,491,487.88 |
|
Retained Earnings
after Adjustments at
the Beginning
of the Year |
5,511,097,171.69 |
4,967,695,967.72 |
|
Less: Allocation of
Net Profit for
Contribution as
State Income |
[303,430,000.00] |
[240,580,000.00] |
|
Net Profit
for the Year |
989,017,011.19 |
783,981,203.97 |
|
|
|
|
|
Retained Earnings
at the End
of the Year |
6,196,684,182.88 |
5,511,097,171.69 |
FINANCIAL ANALYSIS
Annual Growth
& Profitability
|
Annual Growth |
2005 |
2004 |
|
Net Sales |
13.90 |
|
|
Operating Profit |
9.88 |
|
|
Net Profit |
26.15 |
|
|
Fixed Assets |
[0.48] |
|
|
Total Assets |
9.65 |
|
|
Profitability |
|
|
|
Cost of Good
Sold |
54.32 |
58.61 |
|
Operating Profit Margin |
20.93 |
19.05 |
|
S & A
Expenses |
24.81 |
22.40 |
|
Other Revenues |
0.06 |
0.06 |
|
Net Profit Margin |
22.16 |
20.01 |
Net Sales was
increased by 13.90%,
Operating Profit was
increased by 9.88%,
Net Profit was
increased by 26.15%,
Fixed Assets was decreased by
0.48%, Total Assets
was increased by
9.65%,
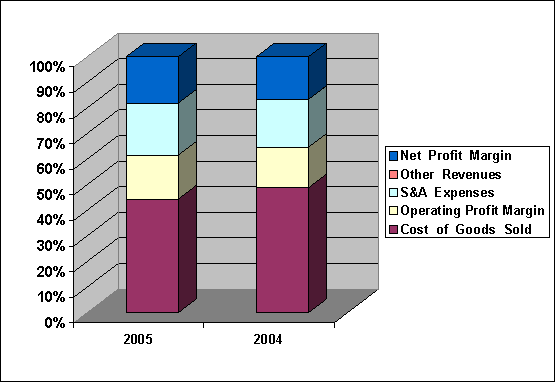
Compare each cost
with sales, Cost of
Good Sold was
decreased from 58.61%
to 54.32%, Operating
Profit Margin was
increased from 19.05%
to 20.93%, S
& A Expenses
was increased from
22.40% to 24.81%,
Other Revenues was
increased from 0.06%
to 0.06%, Net
Profit Margin was
increased from 20.01%
to 22.16%.
Liquidity
|
Liquidity |
2005 |
2004 |
|
Current Ratio |
9.14 |
6.95 |
|
Quick-Assets Ratio |
7.18 |
5.17 |
Current Ratio was
increased from 6.95
to 9.14, show
Current Assets can
cover Current Liabilities, Liquidity Ratio of
the company was
good, Quick-Assets Ratio
was increased from
5.17 to 7.18,
capacity to pay
Short Term Loan
was good too,
because Current Assets
without Inventory can
cover Current Liabilities.
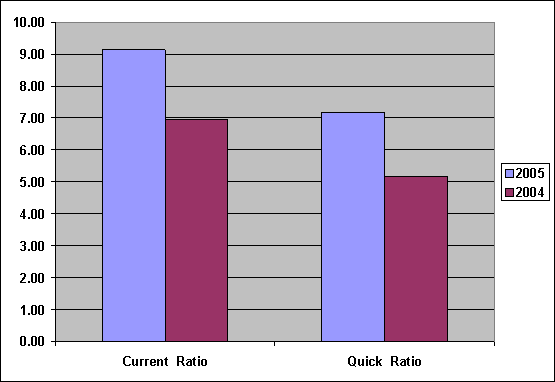
Leverage
|
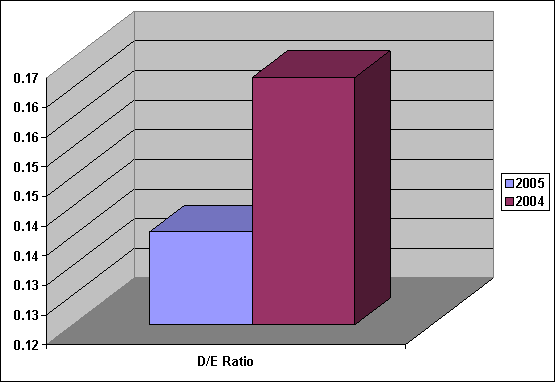
Leverage |
2005 |
2004 |
|
D/E Ratio |
0.14 |
0.16 |
D/E Ratio was
decreased from 0.16
to 0.14, show
the company had
fund from Equity
more than fund
from Loan, investment
risked of the
company is low.
Efficiency
|
Efficiency |
2005 |
2004 |
|
Fixed Assets Turnover |
3.18 |
2.78 |
|
Total Assets Turnover |
0.62 |
0.60 |
|
Return on Assets |
13.78 |
11.98 |
|
Return on Equity |
15.66 |
13.92 |
|
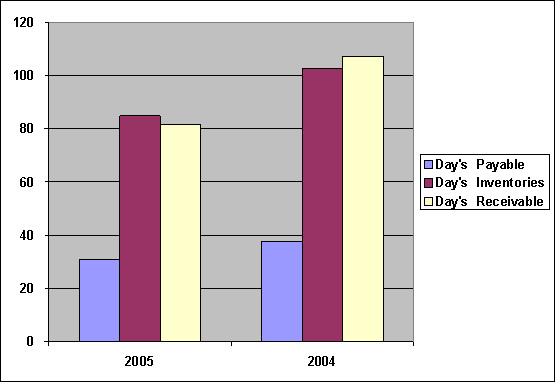
Day's Payable |
30.71 |
37.57 |
|
Day’s Inventories |
84.74 |
102.61 |
|
Day’s Receivables |
81.46 |
107.26 |
Fixed Assets Turnover
was increased from
2.78 times to
3.18 times, Total
Assets Turnover was
increased from 0.60
times to 0.62
times, show the
company could use
Assets so efficiently, Return on Assets
was increased from
11.98 to 13.78,
Return on Equity
was increased from
13.92 to 15.66,
capacity to use
Assets to make
profit was good,
Day's Payable was
decreased from 38
days to 31
days, Day’s Inventories
was decreased from
103 days to
85 days, and
Day’s Receivables was
decreased from 108
days to 82
days.
RATING
EXPLANATIONS
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
>86 |
Aaa |
Possesses an extremely sound financial base with the strongest
capability for timely payment of interest and principal sums |
Unlimited |
|
71-85 |
Aa |
Possesses adequate working capital. No caution needed for credit transaction.
It has above average (strong) capability for payment of interest and
principal sums |
Large |
|
56-70 |
A |
Financial & operational base are regarded healthy. General
unfavourable factors will not cause fatal effect. Satisfactory capability for
payment of interest and principal sums |
Fairly Large |
|
41-55 |
Ba |
Overall operation is considered normal. Capable to meet normal
commitments. |
Satisfactory |
|
26-40 |
B |
Unfavourable & favourable factors carry similar weight in credit consideration.
Capability to overcome financial difficulties seems comparatively below
average/normal. |
Small |
|
11-25 |
Ca |
Adverse factors are apparent. Repayment of interest and principal sums
in default or expected to be in default upon maturity |
Limited with
full security |
|
<10 |
C |
Absolute credit risk exists. Caution needed to be exercised |
Credit not
recommended |
|
NR |
In view of the lack of information, we have no basis upon which to
recommend credit dealings |
No Rating |
|
This score serves as a reference to assess SC’s credit risk
and to set the amount of credit to be extended. It is calculated from a
composite of weighted scores obtained from each of the major sections of this
report. The assessed factors and their relative weights (as indicated through
%) are as follows:
Financial
condition (40%) Ownership
background (20%) Payment
record (10%)
Credit history
(10%) Market trend
(10%) Operational
size (10%)