![]()
MIRA
INFORM REPORT
|
Report Date : |
16.06.2008 |
IDENTIFICATION
DETAILS
|
Name : |
ANGES MC INC |
|
|
|
|
Registered Office : |
Mita Suzuki Bldg 5F, 5-20-14 Shiba Minatoku Tokyo 108-0014 |
|
|
|
|
Country : |
Japan |
|
|
|
|
Financials (as on) : |
31.12.2007 |
|
|
|
|
Date of Incorporation : |
December 1999 |
|
|
|
|
Legal Form : |
Limited Company (Kabushiki Kaisha) |
|
|
|
|
Line of Business : |
Development of Genome-Based Medicines |
RATING &
COMMENTS
|
MIRA’s Rating : |
B |
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
26-40 |
B |
Unfavourable & favourable factors carry similar weight in credit
consideration. Capability to overcome financial difficulties seems
comparatively below average. |
Small |
|
Maximum Credit Limit : |
YEN 244.8 Million |
|
|
|
|
Status : |
Moderate |
|
|
|
|
Payment Behaviour : |
Regular |
name & address
ANGES MC INC
REGD NAME: AnGes MG KK
MAIN OFFICE: Mita Suzuki Bldg 5F,
5-20-14 Shiba Minatoku Tokyo 108-0014 JAPAN
Tel:
03-5730-2630
*
Registered at: 7-7-15 Saitoasagi Ibaraki City Osaka-Pref 567-0085
E-Mail address: info@anges-mg.com
ACTIVITIES
Development of genome-based medicines
BRANCH(ES)
Osaka
OVERSEAS
USA, UK (--subsidiaries)
FACTORY(IES)
(subcontracted)
CHIEF EXEC
EI YAMADA, PRES
Yen Amount
In million Yen, unless otherwise stated
SUMMARY
FINANCES R/WEAK A/SALES Yen 1,720 M
PAYMENTS REGULAR CAPITAL Yen 5,693 M
TREND SLOW WORTH Yen 12,305 M
STARTED 1999 EMPLOYES 93
COMMENT
DEVELOPER
& MFR OF GENOME-BASED MEDICINES..
FINANCIAL
SITUATION COSIDERED RATHER WEAK BUT SHOULD BE GOOD FOR MODERATE BUSINESS
ENGAGEMENTS.
MAX CREDIT LIMIT
YEN 244.8 MILLION, 30 DAYS NORMAL TERMS.
Forecast
(or estimated) figures for 31/12/2008 fiscal term
HIGHLIGHTS
The subject company was established by Osaka University Assistant Professor Ryuichi Morishita in order to develop & manufacture genome-based medicines. This is the first genetic drug treatment developer in Japan. On 27/Mar/2008 the firm applied to the Ministry of Health, Labor & Welfare for approval to make and sell Collategene, which uses the human gene that controls the secretion of hepatocyte growth factor (HGF). When injected into diabetics with ischemia – or poor circulation in certain parts of the body – the genes will produce proteins and promote the generation of new blood vessels, according to the firm. In Jun 2005 formed partnership with Alfresa Pharma Corp on development of NF-kB decoy oligonucleotides in the field of atopic demantitis in Japan. In Aug 2005 formed partnership with Avontec GmbH, with cross license arrangement, granting NF-kB decoy for psoriasis in Europe. In May 2006 forme4d partnership with Vical Inc, investing into Vical’s Phase III program of a gene based drug. In Dec 2006 formed partnership with BioMarin Pharmaceutical Inc on marketing and distribution of product approved in the US for treatment of the genetic disease.
FINANCIAL INFORMATION
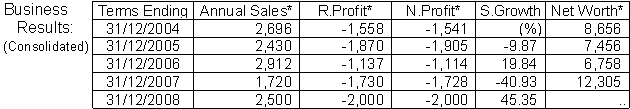
The sales volume for Dec/2007 fiscal term amounted to Yen 1,720 million, a 41% down from Yen 2,912 million in the previous term. New drug development delayed. The operations continued in the red to post Yen 1,730 million recurring loss and Yen 1,728 million net losses, respectively, compared with Yen 1,137 million recurring loss and Yen 1,114 million net losses, respectively, a year ago. Losses continued unabated since its listing in 2002, this being heavy development costs.
For the current term ending Dec 2008 the operations will continue in the red to post Yen 2,000 million recurring loss and Yen 2,000 million net losses, respectively, on a 45% rise in turnover, to Yen 2,500 million.
The financial situation is considered RATHER WEAK but should be good for MODERATE business engagements. Max credit limit is estimated at Yen 244.8 million, on 30 days normal terms.
REGISTRATION
Date Registered: Dec 1999
Legal Status: Limited Company (Kabushiki Kaisha)
Authorized: 370,464 shares
Issued: 117,313 shares
Sum: Yen 5,693 million
Major shareholders (%)
Ryuichi Morishita (11.7), Toshikazu Nakamura (6.7), ECS Ltd (3.7), Hitoshi Kotani (2.3), Shota Morishita (2.3), Bio Frontier Global Investment (2.1), Mayumi Morishita (2.1), Miwako Sakata (1.4), Kensuke Tomita (1.3), Morgan Stanley & Co (1.3); foreign owners (4.8).
No. of shareholders: 6,757
Listed on the S/Exchange (s) of
Tokyo (Mothers)
Managements
Shiro Enoki, ch; Ei Yamada, pres; Koji Nakamoto, dir; Takefumi Genba, dir; Ryuichi Morishita, dir; Ichiro Kitasato, dir
Nothing detrimental is known as to the commercial morality of executives.
Related companies
AnGes Inc (USA), AnGes Euro (Germany), other.
OPERATION
Activities: Develops & manufactures genome-based medical treatment using HGF (Hepatcyte Growth Factor) (--100%).
Clients
Pharmaceutical mfrs, wholesalers, other
No. of accounts
300
Domestic areas of activities
Nationwide
Suppliers
Laboratories, Mfrs, wholesalers, other..
Payment record
Regular
Location
Business area in Tokyo. Office premises at the caption address are leased and maintained satisfactorily.
Bank References
MUFG (Shinjuku-Nishi)
Mizuho Bank (Hamamatsucho)
Relations: Satisfactory
Financials
|
FINANCES: (Consolidated
in million yen) |
|
||||
|
|
|
Terms Ending: |
31/12/2007 |
31/12/2006 |
|
|
INCOME STATEMENT |
|
|
|||
|
|
Annual Sales |
|
1,720 |
2,912 |
|
|
|
Cost of Sales |
3,147 |
3,852 |
||
|
|
GROSS PROFIT |
-1,427 |
-940 |
||
|
|
Selling & Adm Costs |
612 |
583 |
||
|
|
OPERATING PROFIT |
-2,039 |
-1,523 |
||
|
|
Non-Operating P/L |
309 |
386 |
||
|
|
RECURRING PROFIT |
-1,730 |
-1,137 |
||
|
|
NET PROFIT |
-1,728 |
-1,114 |
||
|
BALANCE SHEET |
|
|
|
||
|
|
Cash |
|
6,576 |
3,975 |
|
|
|
Receivables |
|
99 |
129 |
|
|
|
Inventory |
|
572 |
443 |
|
|
|
Securities, Marketable |
3,401 |
802 |
||
|
|
Other Current Assets |
823 |
852 |
||
|
|
TOTAL CURRENT ASSETS |
11,471 |
6,201 |
||
|
|
Property & Equipment |
111 |
152 |
||
|
|
Intangibles |
|
389 |
352 |
|
|
|
Investments, Other Fixed Assets |
1,211 |
1,358 |
||
|
|
TOTAL ASSETS |
13,182 |
8,063 |
||
|
|
Payables |
|
108 |
100 |
|
|
|
Short-Term Bank Loans |
|
|
||
|
|
|
|
|
|
|
|
|
Other Current Liabs |
768 |
1,182 |
||
|
|
TOTAL CURRENT LIABS |
876 |
1,282 |
||
|
|
Debentures |
|
|
|
|
|
|
Long-Term Bank Loans |
|
|
||
|
|
Reserve for Retirement Allw |
|
|
||
|
|
Other Debts |
|
0 |
22 |
|
|
|
TOTAL LIABILITIES |
876 |
1,304 |
||
|
|
MINORITY INTERESTS |
|
|
||
|
|
Common
stock |
9,439 |
5,693 |
||
|
|
Additional
paid-in capital |
7,749 |
4,004 |
||
|
|
Retained
earnings |
(4,702) |
(2,973) |
||
|
|
Evaluation
p/l on investments/securities |
(225) |
32 |
||
|
|
Others |
|
44 |
2 |
|
|
|
Treasury
stock, at cost |
|
|
||
|
|
TOTAL S/HOLDERS` EQUITY |
12,305 |
6,758 |
||
|
|
TOTAL EQUITIES |
13,182 |
8,063 |
||
|
CONSOLIDATED CASH FLOWS |
|
|
|||
|
|
|
Terms ending: |
31/12/2007 |
31/12/2006 |
|
|
|
Cash Flows
from Operating Activities |
|
-1,976 |
-898 |
|
|
|
Cash
Flows from Investment Activities |
-3,668 |
-703 |
||
|
|
Cash
Flows from Financing Activities |
7,446 |
395 |
||
|
|
Cash,
Bank Deposits at the Term End |
|
6,276 |
4,478 |
|
|
ANALYTICAL RATIOS Terms
ending: |
31/12/2007 |
31/12/2006 |
|||
|
|
|
Net
Worth (S/Holders' Equity) |
12,305 |
6,758 |
|
|
|
|
Current
Ratio (%) |
1309.47 |
483.70 |
|
|
|
|
Net
Worth Ratio (%) |
93.35 |
83.81 |
|
|
|
|
Recurring
Profit Ratio (%) |
-100.58 |
-39.05 |
|
|
|
|
Net
Profit Ratio (%) |
-100.47 |
-38.26 |
|
|
|
|
Return
On Equity (%) |
-14.04 |
-16.48 |
|
RATING
EXPLANATIONS
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
>86 |
Aaa |
Possesses an extremely sound financial base with the strongest
capability for timely payment of interest and principal sums |
Unlimited |
|
71-85 |
Aa |
Possesses adequate working capital. No caution needed for credit transaction.
It has above average (strong) capability for payment of interest and
principal sums |
Large |
|
56-70 |
A |
Financial & operational base are regarded healthy. General unfavourable
factors will not cause fatal effect. Satisfactory capability for payment of
interest and principal sums |
Fairly Large |
|
41-55 |
Ba |
Overall operation is considered normal. Capable to meet normal
commitments. |
Satisfactory |
|
26-40 |
B |
Unfavourable & favourable factors carry similar weight in credit
consideration. Capability to overcome financial difficulties seems
comparatively below average. |
Small |
|
11-25 |
Ca |
Adverse factors are apparent. Repayment of interest and principal sums
in default or expected to be in default upon maturity |
Limited with
full security |
|
<10 |
C |
Absolute credit risk exists. Caution needed to be exercised |
Credit not
recommended |
|
NR |
In view of the lack of information, we have no basis upon which to
recommend credit dealings |
No Rating |
|
This score serves as a reference to assess SC’s credit risk
and to set the amount of credit to be extended. It is calculated from a composite
of weighted scores obtained from each of the major sections of this report. The
assessed factors and their relative weights (as indicated through %) are as
follows:
Financial
condition (40%) Ownership
background (20%) Payment
record (10%)
Credit history
(10%) Market trend
(10%) Operational
size (10%)