![]()
MIRA INFORM
REPORT
|
Report Date : |
16.07.2011 |
IDENTIFICATION DETAILS
|
Correct Name : |
THE
GOVERNMENT PHARMACEUTICAL ORGANIZATION |
|
|
|
|
Registered Office : |
|
|
|
|
|
Country : |
|
|
|
|
|
Financials (as on) : |
30.09.2009 |
|
|
|
|
Year of Establishment : |
1941 |
|
|
|
|
Com. Reg. No.: |
Not Available |
|
|
|
|
Legal Form : |
State
|
|
|
|
|
Line of Business : |
Manufacturer of pharmaceutical and medical
supplies |
RATING & COMMENTS
|
MIRA’s Rating : |
A |
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
56-70 |
A |
Financial & operational base are regarded healthy. General unfavourable
factors will not cause fatal effect. Satisfactory capability for payment of
interest and principal sums |
Fairly Large |
|
Status : |
Good |
|
Payment
Behaviour : |
No Complaints |
|
Litigation : |
Clear |
NOTES :
Any query related to this report can be made
on e-mail : infodept@mirainform.com
while quoting report number, name and date.
ECGC Country Risk Classification List – March 31st, 2011
|
Country Name |
Previous Rating (31.12.2010) |
Current Rating (31.03.2011) |
|
Thailand |
b1 |
b1 |
|
Risk Category |
ECGC
Classification |
|
Insignificant |
A1 |
|
Low |
A2 |
|
Moderate |
B1 |
|
High |
B2 |
|
Very High |
C1 |
|
Restricted |
C2 |
|
Off-credit |
D |
Company name
THE GOVERNMENT
PHARMACEUTICAL ORGANIZATION
SUMMARY
BUSINESS
ADDRESS :
TELEPHONE : [66]
2203-8000, 2354-8800-2
FAX :
[66] 2354-8780
E-MAIL
ADDRESS : dr-mkj@gpo.or.th
ESTABLISHED : 1941
FISCAL
YEAR CLOSING DATE : SEPTEMBER 30
LEGAL
STATUS : STATE
EXECUTIVE : DR. WITIT ARTHAVETKUL,
THAI
MANAGING DIRECTOR
NO.
OF STAFF : 2,445
LINES
OF BUSINESS : PHARMACEUTICAL AND
MEDICAL SUPPLIES
MANUFACTURER
CORPORATE PROFILE
OPERATING
TREND : STABLE
PRESENT
SITUATION : OPERATING
NORMALLY
REPUTATION : GOOD
FOR
MANAGEMENT
STANDARD : MANAGEMENT
WITH FAIR PERFORMANCE
HISTORY
The
subject was established
in 1941 as
a state enterprise
under the jurisdiction
of the Ministry
of Public Health,
standing as the
only pharmaceutical manufacturer
owned by the Government, has
responsibilities in producing
high quality drugs
at reasonable prices,
serving the Ministry
of Public Health’s
policy on the
“Health for All”
campaign and etc.
It currently employs
2,445 staff.
The
subject received ISO
9002 Certification certified
by the Bureau
Veritas Quality International
Co., Ltd. [BVQI], the
international standard recognizing
body, for Injection
Quality Systems with
effect from February
28, 1997. The
ISO 9002 Certificate
absolutely guarantees its
products’ standard quality.
The subject’s
business address is
75/1 Rama VI
Rd., Ratchathewi, Bangkok
10400.
THE BOARD OF DIRECTORS
|
Name |
|
Nationality |
Age |
|
|
|
|
|
|
Mr. Vichai
Chokewiwat |
: Chairman |
Thai |
- |
|
Mr. Chuchai Suphawong |
|
Thai |
- |
|
Mr. Paijit
Varachit |
|
Thai |
62 |
|
Mr. Raewat
Visarutwej |
|
Thai |
- |
|
Mr. Boonma
Taechavanich |
|
Thai |
- |
|
Mr. Boonjong Chuchaisaengrat |
|
Thai |
- |
|
Mr. Sombat
Thamrongthanyawong |
|
Thai |
- |
|
Mr. Narongsak Angkasuwapala |
|
Thai |
- |
|
Mr. Anuchart Kongmalai |
|
Thai |
- |
|
Mr. Sutthichai Eiamcharoenying |
|
Thai |
- |
|
Mr. Ch. Nant Petchayapaisit |
|
Thai |
- |
MANAGEMENT
Dr. Witit
Arthavetkul : Managing
Director
Ms.
Pisamorn Klinsuwan
: Deputy
Managing Director
Mr.
Soonthorn Vorakul Deputy
Managing Director
Mr.
Manoon Bupparenoo : Deputy
Managing Director
Mr. Somchai Srichainak
: Deputy
Managing Director
Mrs. Saengkamol Sriaranya
: Deputy
Managing Director
Mrs. Sujida
Chutima : Assistant
Managing Director
Ms.
Anchalee Utthangkorn : Director,
Internal Audit Office
Mr.
Pipat Niyomkarn : Director,
Office of the
Managing
: Department
Mrs.
Varunee Lekcharoenvong : Director,
Accounting & Finance
Department
Mr.
Somchai Anupong-ongart : Director,
Chemical Department
Mr.
Tumrongwut Kootiratrakarn : Director,
Production Department
BUSINESS OPERATIONS
The
subject’s main activities
are as follows:
-
Production of medicines
and medical supplies
-
Promotion of study
& research of
local raw materials
to be made
into
pharmaceutical & medicinal
productions.
-
Controlling the quality
of productions to
be standardized
-
Controlling price level
-
Reserve stocks to
prevent shortage in
case of emergencies
& disaster.
Production
The
cost of product
in the fiscal
year 2007 was valued
at Bht. 2,481.21
million, an increase
of Bht 126.63
million or 5.38%
higher than the
previous year. It
was a result
from the production
of Anti-AIDS medicines
[ARV] responsive to the
increasing demand.
The
production was increased
by over Bht.
399.24 million or
55.07%, mainly from the order of
the Public sectors.
Regarding medicines, the
production was reduced
by Bht. 214.95
million or 15.39% resulting
from the reduction
of penicillin production.
The test kits
and natural products
showed an increased
production by Bht.
23.63 million resulting
from the launch of two
new GPO Cumin
products namely Flash
Result Eye Gel and
Body Lotion. The
preventive medicine, one
the other hand,
showed the decrease
in production to
only Bht. 19.10
million due to
a partial closure
of production accommodating
the renovation of
manufacturing building to meet GMP
standard. The details
are as follows:-
Table
Comparing Cost of
Productions of Years 2008 – 2007 [categorized
by Product Group]
[Unit :
Million Baht]
|
|
Year |
Year |
Comparing to Year 2008/2007 |
|
|
|
Group of
Product |
2008 |
2007 |
Difference |
% |
|
|
|
|
|
|
|
|
|
Medicines |
1,597.27 |
1,181.59 |
415.68 |
35.18 |
|
|
Anti-AIDS [ARV] |
637.65 |
1,124.18 |
[486.53] |
[43.28] |
|
|
Test
Kits/Natural Products |
111.37 |
156.33 |
[44.96] |
[28.76] |
|
|
Preventive
Medicine |
92.42 |
19.10 |
73.32 |
383.87 |
|
|
Total |
2,438.71 |
2,481.21 |
[42.49] |
[1.71] |
|
Distribution
For fiscal year 2008, the Government
Pharmaceutical Organization has a total sale of THB
6,347 million, an increase of THB 902.27
million from previous year (16.57%). This amount includes THB 5,296.18 million of products of GPO, an
increase of THB 632.35 million (13.56%). As for
products manufactured by other manufacturers, the sales increased significantly. The total sale was THB 1,050.82 million, an increased of THB
269.92 million or 34.57%. This is due to the fact that there are medicines in the Compulsory
Licensing group according to the policy of the Ministry of Public Health. Also, the National Health
Security Office (NHSO) has a purchase policy for the GPO to be the provider of procurement.
Table
Comparing Total Sales
Volumes of Years
2007 – 2006 [categorized by
Product Group]
[Unit :
Million Baht]
|
|
Year |
Year |
Comparing to Year 2008/2007 |
|
|
|
Group of
Product |
2008 |
2007 |
Difference |
% |
|
|
|
|
|
|
|
|
|
GPO
Medicinal Products |
5,296.18 |
4,663.83 |
632.35 |
13.56 |
|
|
Non-GPO
Medicinal Products |
1,050.82 |
780.90 |
269.92 |
34.57 |
|
|
Total |
6,347.00 |
5,444.73 |
902.27 |
16.57 |
|
IMPORT
[Countries]
50%
of the chemicals
is imported from
Republic of China,
Japan, France, Singapore
and Spain, while
remaining 50% is
purchased from local suppliers.
SALES
[Local]
90%
of its products
is sold locally
to wholesalers, private
hospitals and Thai
Government such as
public hospitals.
EXPORT
[Countries]
10%
of its products
is also exported
to Myanmar, Laos, Singapore, Philippines,
Vietnam, Malaysia, Cambodia,
Africa and Middle
East countries.
JOINT VENTURE COMPANIES
General
Hospital Products Public
Co., Ltd.
Address
: 101/99
Soi Nava Nakorn
7,
Pathum Thani
12120
Core Business : Saline solution,
dialysis solution, dental
supplies
Holding : 49.6%
of investment by the subject.
United
Pharma Antibiotics Industry
Co., Ltd.
Address
: 101/52
Nava Nakorn Industrial
Estate,
District, Pathum
Thani 12120
Core Business : Raw materials
used for production
of anti-biotics: cloxacillin,
amoxicillin and dicloxacillin
Holding : 45%
of investment by
the subject.
Thai
Wattana Pharmaceutical Dextrose
Co., Ltd.
Address : 112
Moo
Chon Buri 20150
Core Business : Dextrose sugar
for production of
food and drugs
Holding : 30%
of investment by
the subject.
Thai
Herbal Products Co.,
Ltd.
Address :
Ayudhya
13170
Core Business : Herbal remedies
Holding : 49%
of investment by
the subject.
Gpo-Merieux
Biologicals Co., Ltd.
Address : 241
Moo 7, Huasamrong,
Plaengyao, Chachoengsao 24190.
Core
Business: Human vaccinations
Holding : 49%
of investment by
the subject
Medical
Device Manufacturer [
Address : In
the process of
building a plant
Core Business : Manufacture and
sells syringes and
safety hypodermic syringes
Holding : 13% of
investment by the
subject.
CREDIT
Local
bills are paid
by cash or
on the credits
term of 30-60
days.
Imports
are by L/C
at sight or
on the credits
term of 90-120
days and T/T.
Sales
are by cash
or on the
credits term of
60-90 days.
Exports
are against L/C
at sight and
T/T.
BANKING
Krung
Thai Bank Public
Co., Ltd.
[Head Office :
Bangkok
Bank Public Co.,
Ltd.
[Head Office :
Kasikornbank
Public Co., Ltd.
[Head Office :
EMPLOYMENt
The
subject employs 2,445 staff
[office, sales staff
and factory workers].
LOCATION DETIALS
The premise
is owned for
operating administrative office,
drug store, warehouse
and factory at
the heading address.
The ground space
is measured at
64,000 square meters
and the floor
space is around
45,500 square meters.
It is located
in commercial area.
Branches
are located at
following addresses:
Northeast
Regional Branch in
North
Regional Branch in
South
Regional Branch in
Moreover,
the subject has
11 drugs stores
under the name
“The Government Pharmaceutical Organization”
located at:
- Rama
6 Rd.,
- Rajthevee
Rd.,
- Yosse
Rd.,
- Charansanitwong Rd.,
- Theves
Rd.,
- Rangsit
Rd.,
- Bangrak,
- Wattana,
- Jomthong,
- Pravet,
- Nonthaburi
REMARK
CREDIT
OF US$ 125,000
AGAINST D/A TERMS
SHOULD BE IN
ORDER.
COMMENT
Though
the medicine market
tends to expand
continuously, there are
many factors that
could affect to
the growth of
the market, like
the high competition
among domestic manufacturers, the
market share taking
from import medicine,
and the effect
from FTA which
will be applied
gradually. Therefore, it
is necessary that
Thai manufacturers in
medical industry should
realize and prepare
themselves rapidly for
the situation that
could be changed
in the future.
Thus, GPO
increases the investment
in research and
development in order
to create the
new innovation for
the products and at the
same time, GPO
also upgrades the
manufacturing standard to
comply with the
world standard which
can be considered
as the important
factor that drives
the organization to
grow strongly and
can compete in
international level.
FINANCIAL INFORMATION
The
details of capital
and shareholders are
not available due
to the fact
that the subject
is regarded as
a state enterprise
and is funded
and operated under
the supervision of
the Ministry of
Public Heath.
NAME OF AUDITOR
& CERTIFIED PUBLIC
ACCOUNTANT NO.:
Office of the Auditor
General
BALANCE SHEET [BAHT]
The latest financial figures published as at September 30, 2009 & 2008 were:
ASSETS
|
Current Assets |
2009 |
2008 |
|
|
|
|
|
Cash and Cash
Equivalents |
3,340,542,275.06 |
1,536,735,526.21 |
|
Short-term Investment |
351,810,557.01 |
2,166,731,118.54 |
|
Trade Accounts Receivables, net |
2,266,061,606.65 |
1,915,240,733.49 |
|
Short-term Loan |
52,116,282.38 |
15,443,454.89 |
|
Inventories |
2,553,806,423.72 |
1,199,619,562.17 |
|
Other Current Assets
|
165,428,634.57 |
49,063,358.39 |
|
|
|
|
|
Total Current Assets
|
8,729,765,779.39 |
6,882,833,753.69 |
|
Investments in Join
Venture |
401,360,000.00 |
494,960,000.00 |
|
Long-term Investment |
400,000,000.00 |
- |
|
Long-term Loans |
61,196,161.44 |
60,367,306.87 |
|
Property, Plant Equipment |
1,923,654,376.96 |
1,875,735,887.96 |
|
Intangible Assets |
2,993,867.76 |
- |
|
Assets in Process |
240,359,079.74 |
107,984,647.36 |
|
Current Portion of
Drugs & Medical
Supply Reserve Project
|
8,509,784.66 |
22,590,845.20 |
|
Total Assets |
11,767,839,049.95 |
9,444,472,441.08 |
LIABILITIES &
SHAREHOLDERS' EQUITY [BAHT]
|
Current
Liabilities |
2009 |
2008 |
|
|
|
|
|
Trade Accounts Payable |
1,565,541,733.08 |
567,447,199.22 |
|
Other Payable |
571,926,128.86 |
398,255,242.65 |
|
|
|
|
|
Total Current Liabilities |
2,137,467,861.94 |
965,702,441.87 |
|
Employee’s Pension Fund |
258,658,257.20 |
245,985,018.60 |
|
GPO’s Loan Fund |
25,000,000.00 |
20,000,000.00 |
|
Total Liabilities |
2,421,126,119.14 |
1,231,687,460.47 |
|
|
|
|
|
Capital |
|
|
|
|
|
|
|
Paid-up Capital |
48,041,467.48 |
48,041,467.48 |
|
Capital Budget Allocations |
3,018,502.04 |
3,018,502.04 |
|
Transfer from Current
Capital |
3,526,548.46 |
3,526,548.46 |
|
|
54,586,517.98 |
54,586,517.98 |
|
|
|
|
|
Surplus Capital from Goodwill |
11,965,602.08 |
13,80,607.96 |
|
Drugs and Medical
Supply Reserve |
49,914,409.60 |
49,914,409.60 |
|
Retained Earnings |
9,230,246,401.15 |
8,094,480,445.07 |
|
Total Capital |
9,346,712,930.81 |
8,212,784,980.61 |
|
Total Liabilities & Capital |
11,767,839,049.95 |
9,444,472,441.08 |
PROFIT & LOSS ACCOUNT
|
Sales |
2009 |
2008 |
|
|
|
|
|
Sales of Pharmaceutical and
Medical Supplies |
8,127,957,550.05 |
6,347,001,230.97 |
|
Income from Hive
of Work |
1,924,612.96 |
2,346,445.75 |
|
Other Income |
187,655,748.52 |
134,228,911.46 |
|
Total Sales |
8,317,537,911.53 |
6,483,576,588.18 |
|
Expenses |
|
|
|
|
|
|
|
Cost of Sales
and Hive of
Work |
5,169,324,372.10 |
3,842,172,606.16 |
|
Selling Expenses |
481,307,653.41 |
494,071,276.60 |
|
Administration Expenses |
961,335,446.06 |
880,195,076.60 |
|
Other Expenses |
124,516,028.37 |
152,330,453.87 |
|
Total Expenses |
6,736,483,499.94 |
5,368,769,413.23 |
|
|
|
|
|
Net Profit |
1,581,054,411.59 |
1,114,807,174.95 |
|
Retained Earnings after
Adjustments at the Beginning of
the Year |
8,212,784,980.61 |
7,522,941,624.42 |
|
Increase [Decrease] Between
Payments |
[1,838,005.88] |
[34,785.80] |
|
Less Contribution to
State Income |
[445,288,455.51] |
[424,929,032.96] |
|
|
|
|
|
Retained Earnings at
the End of
the Year |
9,346,712,930.81 |
8,212,784,980.61 |
FINANCIAL ANALYSIS
|
ITEM |
UNIT |
2009 |
2008 |
|
|
|
|
|
|
LIQUIDITY RATIO |
|
|
|
|
CURRENT RATIO |
TIMES |
4.08 |
7.13 |
|
QUICK RATIO |
TIMES |
2.81 |
5.83 |
|
|
|
|
|
|
ACTIVITY RATIO |
|
|
|
|
FIXED ASSETS TURNOVER |
TIMES |
4.23 |
3.38 |
|
TOTAL ASSETS TURNOVER |
TIMES |
0.69 |
0.67 |
|
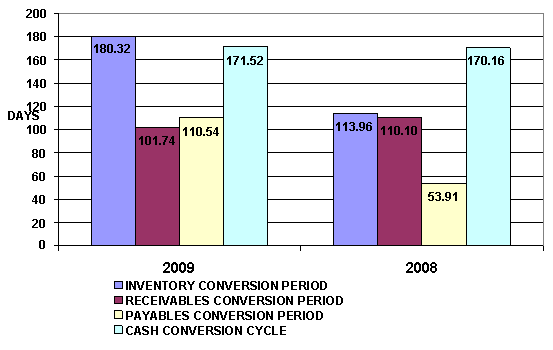
INVENTORY CONVERSION PERIOD |
DAYS |
180.32 |
113.96 |
|
INVENTORY TURNOVER |
TIMES |
2.02 |
3.20 |
|
RECEIVABLES CONVERSION PERIOD |
DAYS |
101.74 |
110.10 |
|
RECEIVABLES TURNOVER |
TIMES |
3.59 |
3.32 |
|
PAYABLES CONVERSION PERIOD |
DAYS |
110.54 |
53.91 |
|
CASH CONVERSION CYCLE |
DAYS |
171.52 |
170.16 |
|
|
|
|
|
|
PROFITABILITY
RATIO |
|
|
|
|
COST OF GOODS SOLD |
% |
63.58 |
60.51 |
|
SELLING & ADMINISTRATION |
% |
5.92 |
7.78 |
|
INTEREST |
% |
- |
- |
|
GROSS PROFIT MARGIN |
% |
38.72 |
41.60 |
|
NET PROFIT MARGIN BEFORE EX. ITEM |
% |
19.45 |
17.56 |
|
NET PROFIT MARGIN |
% |
19.45 |
17.56 |
|
RETURN ON EQUITY |
% |
16.92 |
13.57 |
|
RETURN ON ASSET |
% |
13.44 |
11.80 |
|
EARNING PER SHARE |
BAHT |
3,291.02 |
2,320.51 |
|
|
|
|
|
|
LEVERAGE RATIO |
|
|
|
|
DEBT RATIO |
TIMES |
0.21 |
0.13 |
|
DEBT TO EQUITY RATIO |
TIMES |
0.26 |
0.15 |
|
TIME INTEREST EARNED |
TIMES |
- |
- |
|
|
|
|
|
|
ANNUAL GROWTH |
|
|
|
|
SALES GROWTH |
% |
28.04 |
|
|
OPERATING PROFIT |
% |
41.82 |
|
|
NET PROFIT |
% |
41.82 |
|
|
FIXED ASSETS |
% |
2.55 |
|
|
TOTAL ASSETS |
% |
24.60 |
|
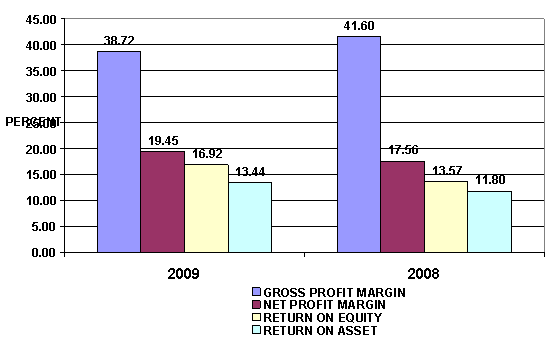
PROFITABILITY RATIO
|
Gross Profit Margin |
38.72 |
Deteriorated |
Industrial Average |
4,335.82 |
|
Net Profit Margin |
19.45 |
Impressive |
Industrial Average |
(10,176.78) |
|
Return on Assets |
13.44 |
Impressive |
Industrial Average |
(2,660.41) |
|
Return on Equity |
16.92 |
Impressive |
Industrial Average |
(2,146.46) |
Gross Profit Margin used to assess a firm's financial health by
revealing the proportion of money left over from revenues after accounting for
the cost of goods sold. Gross profit margin serves as the source for paying
additional expenses and future savings. The subject's figure is 38.72%. When
compared with the industry average, the ratio of the company was lower. This
indicated that subject may have the
problems with control over its costs.
Net Profit Margin is the indicator of the subject's efficiency in that
net profit takes into consideration all expenses of the subject. A low profit margin indicates a low
margin of safety, higher risk that a decline in sales will erase profits and
result in a net loss. Net Profit Margin is 19.45% compared with those of its
average competitors in the same industry, indicated that business was an
efficient operator in a dominant position within its industry.
Return on Assets measures how efficiently profits are being generated
from the assets employed in the business when compared with the ratios of firms
in a similar business. A low ratio in comparison with industry averages
indicates an inefficient use of business assets. Return on Assets is 13.44%,
higher figure when compared with those of its average competitors in the same
industry, indicated that business was an efficient profit in a dominant
position within its industry.
Return on Equity indicates how profitable a company is by comparing its
net income to its average shareholders' equity, ROE measures how much the
shareholders earned for their investment in the company. Return on Equity is
16.92%, higher figure when compared with those of its average competitors in
the same industry, indicated that business was an efficient profit in a
dominant position within its industry.
Trend of the
average competitors in the same industry for last 5 years
Return on Assets Downtrend
Return on Equity Downtrend
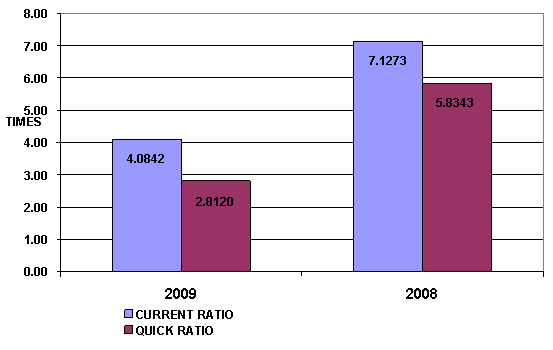
LIQUIDITY RATIO
|
Current Ratio |
4.08 |
Deteriorated |
Industrial Average |
15,055.26 |
|
Quick Ratio |
2.81 |
|
|
|
|
Cash Conversion Cycle |
171.52 |
|
|
|
The Current Ratio is to ascertain whether a subject's short-term assets
are readily available to pay off its short-term liabilities. The subject's figure
is 4.08 times in 2009, decrease from 7.13 times, then it is generally
considered to have good short-term financial strength. When compared with the
industry average, the ratio of the company was lower.
The Quick Ratio is a liquidity indicator that further refines the
current ratio by measuring the amount of the most liquid current assets there
are to cover current liabilities. The subject's figure is 2.81 times in 2009,
decrease from 5.83 times, although excluding inventory so the company still have
good short-term financial strength.
The Cash Conversion Cycle measures the number of days the
subject's cash is tied up in the production and sales process of its
operations and the benefit from payment terms from its creditors. It meant the
subject could survive when no cash inflow was received from sale for 172 days.
Trend of the
average competitors in the same industry for last 5 years
Current Ratio Uptrend
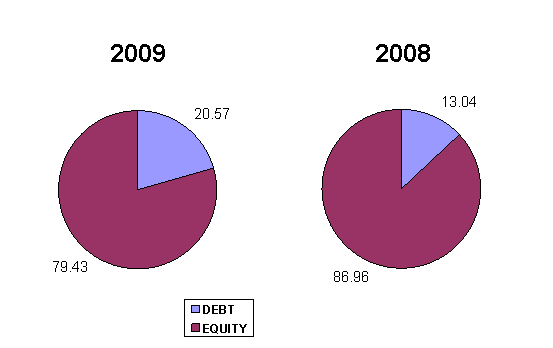
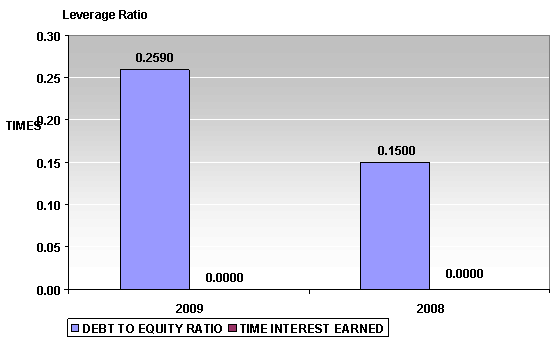
LEVERAGE RATIO
|
Debt Ratio |
0.21 |
Impressive |
Industrial Average |
60.90 |
|
Debt to Equity Ratio |
0.26 |
Impressive |
Industrial Average |
83.71 |
|
Times Interest Earned |
- |
|
Industrial Average |
121,468.14 |
Debt to Equity Ratio a measurement of how much suppliers, lenders,
creditors and obligors have committed to the subject versus what the
shareholders have committed. A lower the percentage means that the subject is
using less leverage and has a stronger equity position.
Debt Ratio shows the proportion of a subject's assets which are financed
through debt. The subject's figure is 0.21 less than 0.5, most of the company's
assets are financed through equity.
Trend of the average
competitors in the same industry for last 5 years
Debt Ratio Uptrend
Times Interest Earned Uptrend
ACTIVITY RATIO
|
Fixed Assets Turnover |
4.23 |
Deteriorated |
Industrial Average |
172,765.71 |
|
Total Assets Turnover |
0.69 |
Impressive |
Industrial Average |
(73.06) |
|
Inventory Conversion Period |
180.32 |
|
|
|
|
Inventory Turnover |
2.02 |
Deteriorated |
Industrial Average |
1,564.69 |
|
Receivables Conversion Period |
101.74 |
|
|
|
|
Receivables Turnover |
3.59 |
Deteriorated |
Industrial Average |
3,803.38 |
|
Payables Conversion Period |
110.54 |
|
|
|
Trend of the average
competitors in the same industry for last 5 years
Fixed Assets Turnover Uptrend
Total Assets Turnover Downtrend
Inventory Turnover Uptrend
Receivables Turnover Uptrend
FOREIGN EXCHANGE RATES
|
Currency |
Unit
|
Indian Rupees |
|
US Dollar |
1 |
Rs.44.53 |
|
|
1 |
Rs.71.91 |
|
Euro |
1 |
Rs.62.98 |
RATING EXPLANATIONS
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
>86 |
Aaa |
Possesses an extremely sound financial base with the strongest capability
for timely payment of interest and principal sums |
Unlimited |
|
71-85 |
Aa |
Possesses adequate working capital. No caution needed for credit
transaction. It has above average (strong) capability for payment of interest
and principal sums |
Large |
|
56-70 |
A |
Financial & operational base are regarded healthy. General
unfavourable factors will not cause fatal effect. Satisfactory capability for
payment of interest and principal sums |
Fairly Large |
|
41-55 |
Ba |
Overall operation is considered normal. Capable to meet normal
commitments. |
Satisfactory |
|
26-40 |
B |
Capability to overcome financial difficulties seems comparatively
below average. |
Small |
|
11-25 |
Ca |
Adverse factors are apparent. Repayment of interest and principal sums
in default or expected to be in default upon maturity |
Limited with
full security |
|
<10 |
C |
Absolute credit risk exists. Caution needed to be exercised |
Credit not
recommended |
|
---- |
NB |
New Business |
---- |
This score serves as a reference to assess SC’s credit risk
and to set the amount of credit to be extended. It is calculated from a
composite of weighted scores obtained from each of the major sections of this report.
The assessed factors and their relative weights (as indicated through %) are as
follows:
Financial
condition (40%) Ownership
background (20%) Payment
record (10%)
Credit history
(10%) Market trend
(10%) Operational
size (10%)
This report is issued at your request without any
risk and responsibility on the part of MIRA INFORM PRIVATE LIMITED (MIPL)
or its officials.