MIRA
INFORM REPORT
|
Report Date : |
11.04.2014 |
IDENTIFICATION DETAILS
|
Name : |
RPG LIFE SCIENCES LIMITED [w.e.f.
13.02.2008] |
|
|
|
|
Formerly Known
As : |
RPG PHARMACEUTICALS LIMITED |
|
|
|
|
Registered
Office : |
RPG House, 463, Dr. Annie Besant Road, Worli, Mumbai-400030, Maharashtra |
|
|
|
|
Country : |
India |
|
|
|
|
Financials (as
on) : |
31.03.2013 |
|
|
|
|
Date of
Incorporation : |
29.03.2007 |
|
|
|
|
Com. Reg. No.: |
11-169354 |
|
|
|
|
Capital Investment
/ Paid-up Capital : |
Rs.132.300
Millions |
|
|
|
|
CIN No.: [Company Identification
No.] |
L24232MH2007PLC169354 |
|
|
|
|
TAN No.: [Tax Deduction &
Collection Account No.] |
PNER01855A |
|
|
|
|
Legal Form : |
A Public Limited Liability Company. The Company’s Shares are Listed on The Stock Exchanges. |
|
|
|
|
Line of Business
: |
Manufacturing and Marketing of Pharmaceutical Products. |
|
|
|
|
No. of Employees
: |
Information denied by the management. |
RATING & COMMENTS
|
MIRA’s Rating : |
Ba (49) |
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
41-55 |
Ba |
Overall operation is considered normal. Capable to meet normal
commitments. |
Satisfactory |
|
Maximum Credit Limit : |
USD 3000000 |
|
|
|
|
Status : |
Satisfactory |
|
|
|
|
Payment Behaviour : |
Usually correct |
|
|
|
|
Litigation : |
Clear |
|
|
|
|
Comments : |
Subject is an established company having a satisfactory track record. Trade relations are fair. Business is active. Payment terms are
usually correct and as per commitment. The company can be considered normal for business dealings at usual
trade terms and conditions. |
NOTES :
Any query related to this report can be made
on e-mail : infodept@mirainform.com
while quoting report number, name and date.
ECGC Country Risk Classification List – December 1, 2013
|
Country Name |
Previous Rating (30.09.2013) |
Current Rating (01.12.2013) |
|
India |
A1 |
A1 |
|
Risk Category |
ECGC
Classification |
|
Insignificant |
A1 |
|
Low |
A2 |
|
Moderate |
B1 |
|
High |
B2 |
|
Very High |
C1 |
|
Restricted |
C2 |
|
Off-credit |
D |
INDIAN ECONOMIC OVERVIEW
India’s current account deficit for the fiscal third quarter ended
September 2013 narrowed to $4.2 billion or 0.9 % of the gross domestic product
from $31.9 billion or 6.5 % of GDP a year earlier, thanks to a pick-up in
exports and moderation in gold imports. Manufacturing activity and new orders
in India showed their strongest growth in a year in February. The news comes as
a relief after data showed Asia’s third largest economy grew by a
slower-than-expected 4.7 % annually in the three months through December. The
HSBC Manufacturing Purchasing Managers’ Index which gauges the business
activity of India’s factories but not its’ utilities, rose to 52.5 in February,
its highest in a year from 51.4 in January. Overall new orders for factory
goods which rose to a one-year high of 54.9 contributed to the surge. China has
emerged as India’s biggest trading partner in the current financial year
replacing the United Arab Emirates and pushing it to the third spot.
India-China trade has reached $49.5 billion with a 8.7 % share in India’s total
trade. The US comes second at $46 billion with 8.1 % share during the first
nine months of the current financial year.
The Reserve Bank of India has granted an additional nine months to the
public to exchange currency notes printed before 2005 including Rs 500 and Rs 1,000
denominations, pushing the deadline to January 1, 2015. A day before dates for
the Lok Sabha polls were
announced, the government decided to hike interest rates on fixed deposit
schemes offered by post offices up to 0.2 per cent. The new rates will be
effective April, 1. The Supreme Court will resume hearing on March, 11 Nokia’s
appeal against a ruling over transferring ownership of its local mobile phones
plant which is the subject of a tax dispute to Microsoft Corp.
In the last days of the current Government, another scam has surfaced.
The defence ministry has ordered a probe into
Hindustan Aeronautics Limited’s contracts from
Britain’s Rolls-Royce Holdings worth at least $ 1.2 billion. The Central Bureau
of Investigation will look into allegations that over $80 million was paid in
kickbacks in a deal signed in 2011. India has asked Boeing Co. to find a
solution for problems with state-owned Air India’s 787 Dreamliners.
The aircraft has experienced a series of malfunctions since its debut in 2011.
EXTERNAL AGENCY RATING
|
Rating Agency Name |
CARE |
|
Rating |
Long term bank facilities: BBB |
|
Rating Explanation |
Have moderate degree of safety and carry moderate credit risk. (withdrawn) |
|
Date |
03.03.2014 |
|
Rating Agency Name |
CARE |
|
Rating |
Short term bank facilities: A3+ |
|
Rating Explanation |
Have minimal degree safety and carry higher credit risk. (withdrawn) |
|
Date |
03.03.2014 |
RBI DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available RBI Defaulters’ list.
EPF (Employee Provident Fund) DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter in
the publicly available EPF (Employee Provident Fund) Defaulters’ list as of
31-03-2012.
INFORMATION DECLINED B
MANAGEMENT NON CO-OPERATIVE. [91-22-24981650]
LOCATIONS
|
Registered / Corporate Office : |
RPG House, 463, Dr. Annie Besant Road, Worli, Mumbai-400030, Maharashtra,
India |
|
Tel. No.: |
91-22-24981650/51/66606375/76/77/78 |
|
Fax No.: |
91-22-24970127 |
|
E-Mail : |
|
|
|
|
|
Factory 1 : |
BIOTECH Plot No. 2702/A, GIDC Industrial Estate, Ankleshwar-393002, District Bharuch, Gujarat, India |
|
Tel. No.: |
91-2646-652102/03/04/652074 |
|
Fax No.: |
91-2646-250104 |
|
|
|
|
Factory 2 : |
FORMULATION Plot No. 3102/A, GIDC Estate, Ankleshwar-393002, District Bharuch, Gujarat, India |
|
Tel. No.: |
91-2646-652062 to 652069 |
|
Fax No.: |
91-2646-250572 |
|
|
|
|
Purchase Office / Factory 3 : |
API 25, MIDC Land, Thane Belapur
Road, Navi Mumbai-400705, Maharashtra,
India |
|
Tel. No.: |
91-22-67955555/5398/5399/6795/5400 |
|
Fax No.: |
91-22-27672646/27631052 |
DIRECTORS
As on 31.03.2013
|
Name : |
Mr. H. V. Goenka |
|
Designation : |
Chairman |
|
|
|
|
Name : |
Mr. Ajit Singh Chouhan |
|
Designation : |
Managing Director |
|
|
|
|
Name : |
Mr. H.V. Goenka |
|
Designation : |
Chairman |
|
|
|
|
Name : |
Mr. R.A. Shah |
|
Designation : |
Alternate to Mr. C. Vinayaraghavan |
|
|
|
|
Name : |
Mr. C.L. Jain |
|
Designation : |
Director |
|
|
|
|
Name : |
Mr. Ajit Gulabchand |
|
Designation : |
Director |
|
|
|
|
Name : |
Dr. Lalit S. Kanodia |
|
Designation : |
Director |
|
|
|
|
Name : |
Mr. Mahesh S. Gupta |
|
Designation : |
Director |
|
|
|
|
Name : |
Mr. Manoj K. Maheshwari |
|
Designation : |
Director |
|
|
|
|
Name : |
Mr. P.K. Mohapatra |
|
Designation : |
Director |
|
|
|
|
Name : |
Mr. C. Vinayaraghavan |
|
Designation : |
Director |
|
|
|
|
Name : |
Mr. Ajit Singh Chouhan |
|
Designation : |
Managing Director |
KEY EXECUTIVES
|
Audit Committee: |
|
|
Name : |
· Mr. C.L. Jain · Mr. Mahesh S. Gupta · Mr. P.K. Mohapatra ·
Mr. Ajit Singh Chouhan |
|
|
|
|
Name : |
Mr. Rajindrra Patkar |
|
Designation : |
Chief Executive – Global Formulations |
|
|
|
|
Name : |
Mr. Vimalendu K. Singh |
|
Designation : |
Chief Executive – API, Global Generics and Biotech |
|
|
|
|
Name : |
Mr. Sachin Raole |
|
Designation : |
CFO and Sr. Vice President – Corporate Services |
|
|
|
|
Name : |
Dr. Uday R. Bapat |
|
Designation : |
Chief Scientific Officer |
|
|
|
|
Name : |
Ms. Suchitra Tiwari |
|
Designation : |
General Manager – QA/QC and Regulatory Affairs |
|
|
|
|
Name : |
Mr. Rabindranath Nandi |
|
Designation : |
Vice President - Corporate Quality |
|
|
|
|
Name : |
Mr. Anil Kumar Gupta |
|
Designation : |
Vice President - Manufacturing |
|
|
|
|
Name : |
Mr. Rajesh Shirambekar |
|
Designation : |
Head – Legal and Company Secretary |
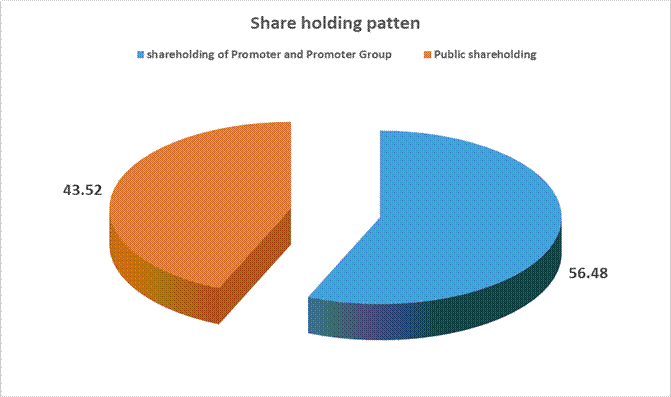
MAJOR SHAREHOLDERS / SHAREHOLDING PATTERN
As on 31.12.2013
|
Category of Shareholder |
Total
No. of Shares |
Total
Shareholding as a % of Total No. of Shares |
|
(A) Shareholding of Promoter and Promoter Group |
|
|
|
|
|
|
|
|
9338897 |
56.48 |
|
|
9338897 |
56.48 |
|
|
|
|
|
Total shareholding of Promoter and Promoter Group (A) |
9338897 |
56.48 |
|
(B) Public Shareholding |
|
|
|
|
|
|
|
|
1175 |
0.01 |
|
|
3217 |
0.02 |
|
|
289979 |
1.75 |
|
|
20650 |
0.12 |
|
|
315021 |
1.91 |
|
|
|
|
|
|
1827817 |
11.05 |
|
|
|
|
|
|
3214420 |
19.44 |
|
|
1656071 |
10.02 |
|
|
183015 |
1.11 |
|
|
79 |
0.00 |
|
|
182936 |
1.11 |
|
|
6881323 |
41.62 |
|
Total Public shareholding (B) |
7196344 |
43.52 |
|
Total (A)+(B) |
16535241 |
100.00 |
|
(C) Shares held by Custodians and against which Depository Receipts
have been issued |
0 |
0.00 |
|
|
0 |
0.00 |
|
|
0 |
0.00 |
|
|
0 |
0.00 |
|
Total (A)+(B)+(C) |
16535241 |
0.00 |
BUSINESS DETAILS
|
Line of Business : |
Manufacturing and Marketing of Pharmaceutical Products. |
||||||||
|
|
|
||||||||
|
Products : |
|
PRODUCTION STATUS [AS ON 31.03.2011]
|
Particulars |
Licensed
Capacity |
Installed
Capacity |
Actual
Production |
|
Tablets (Million Nos.) |
1420 |
2750 |
2241 |
|
Liquids (Kilolitres) |
1287 |
6000 |
592 |
|
Injections – Ampoules (Thousands) |
1050 |
-- |
2309 |
|
Capsules (Million Nos.) |
86 |
312 |
17 |
|
Powder Pouches (Thousands) |
10000 |
12000 |
233 |
|
Ointments (Thousands) |
100 |
-- |
20 |
|
Ointments (Kilolitres) |
30 |
-- |
-- |
|
Bulk Drugs and Chemicals (Tonnes) |
231 |
71 |
27 |
|
|
|
|
|
|
Consumer
Products |
|
|
|
|
Tablets (Million Nos.) |
120 |
120 |
@ |
|
Powder Pouches (Thousands) |
8000 |
8000 |
-- |
@0.13 Million Nos.
NOTE:
(i) Production includes manufacture of pharmaceutical
preparations by other parties and of chemicals for sale but excludes
manufacture of pharmaceutical preparations for other parties. Production of
pharmaceutical preparations includes production of physician samples.
(ii) The installed
capacities are as per the certificate given by the management of the company on
which the auditors have relied.
(iii) The licensed
capacity in respect of certain items has been converted into dosage / units to
make it comparable with installed capacity, production and stocks.
GENERAL INFORMATION
|
No. of Employees : |
Information denied by the management. |
|||||||||||||||||||||||||||||||||
|
|
|
|||||||||||||||||||||||||||||||||
|
Bankers : |
· Union Bank of India · State Bank of India · IDBI Bank ·
Export-Import Bank of India |
|||||||||||||||||||||||||||||||||
|
|
|
|||||||||||||||||||||||||||||||||
|
Facilities : |
|
|
|
|
|
Banking
Relations : |
-- |
|
|
|
|
Auditors : |
|
|
Name : |
Lovelock and Lewes Chartered Accountants |
|
|
|
|
Solicitors: |
|
|
Name : |
Crawford Bayley and Company |
|
|
|
|
Enterprise where
control exists: |
· Swallow Associates LLP (from 13th July, 2012)*@ Note: *No transactions during the year @Consequent to acquisition of additional shares of the company during the year by one of the subsidiaries of Swallow Associates Limited, a Promoter Group Company, the company became a subsidiary of Swallow Associates Limited with effect from 13th July, 2012 in terms of the provisions of sub-section (3) of Section 4 of the Act. However, on conversion of Swallow Associates Limited to Swallow Associates LLP ('SAL'), the company ceased to be a subsidiary of Swallow Associates Limited with effect from 31st October, 2012. SAL along with its subsidiaries, now holds 52.64% of the paid-up share capital of the company as at 31st March, 2013. |
CAPITAL STRUCTURE
As on 31.03.2013
Authorised Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
18750000 |
Equity Shares |
Rs.8/- each |
Rs.150.000 Millions |
Issued, Subscribed & Paid-up Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
16535241 |
Equity Shares |
Rs.8/- each |
Rs.132.300
Millions |
NOTE:
Reconciliation of Number of Shares
|
Particular |
As on 31.03.2013 |
|
|
|
No. of Shares |
Rs. in Millions |
|
Number of shares outstanding as at the beginning of the year |
16535241 |
132.300 |
The company has only one class of shares i.e. Equity Shares having a face value of Rs. 8 each. Each shareholder is eligible for one vote per share held. The dividend proposed by the Board of Directors is subject to the approval of the shareholders in the ensuing Annual General Meeting.
List of shareholders holding more than 5% shares as at the Balance Sheet date
|
Name of the
Shareholder |
As on 31.03.2013 |
|
|
|
No. of Shares |
% of Holding |
|
Swallow
Associates Limited |
4228089 |
25.57 |
|
Instant Holdings Limited |
2701200 |
16.34 |
|
Summit
Securities Limited |
1774708 |
10.73 |
Shares allotted as fully paid-up pursuant to contract(s) without payment being received in cash (during 5 years immediately preceding 31st March, 2013)
14,368,850 shares were allotted in the last 5 years as fully paid-up pursuant to a Scheme of Arrangement without payments being received in cash
FINANCIAL DATA
[all figures are
in Rupees Millions]
ABRIDGED BALANCE
SHEET
|
SOURCES
OF FUNDS |
31.03.2013 |
31.03.2012 |
31.03.2011 |
|
|
|
|
|
|
I.
EQUITY AND LIABILITIES |
|
|
|
|
(1)Shareholders' Funds |
|
|
|
|
(a) Share Capital |
132.300 |
132.300 |
132.300 |
|
(b) Reserves & Surplus |
630.000 |
609.100 |
616.000 |
|
(c) Money received against
share warrants |
0.000 |
0.000 |
0.000 |
|
|
|
|
|
|
(2) Share Application money
pending allotment |
0.000 |
0.000 |
0.000 |
|
Total
Shareholders’ Funds (1) + (2) |
762.300 |
741.400 |
748.300 |
|
|
|
|
|
|
(3) Non-Current Liabilities |
|
|
|
|
(a) long-term borrowings |
105.200 |
206.700 |
248.000 |
|
(b) Deferred tax liabilities
(Net) |
29.400 |
68.200 |
59.600 |
|
(c) Other long term
liabilities |
72.500 |
72.100 |
70.600 |
|
(d) long-term provisions |
12.500 |
11.800 |
7.600 |
|
Total
Non-current Liabilities (3) |
219.600 |
358.800 |
385.800 |
|
|
|
|
|
|
(4) Current Liabilities |
|
|
|
|
(a) Short term borrowings |
370.900 |
243.900 |
203.400 |
|
(b) Trade payables |
433.300 |
288.700 |
398.500 |
|
(c) Other current liabilities |
240.100 |
168.800 |
110.800 |
|
(d) Short-term provisions |
31.400 |
23.700 |
46.800 |
|
Total
Current Liabilities (4) |
1075.700 |
725.100 |
759.500 |
|
|
|
|
|
|
TOTAL |
2057.600 |
1825.300 |
1893.600 |
|
|
|
|
|
|
II.
ASSETS |
|
|
|
|
(1) Non-current assets |
|
|
|
|
(a) Fixed Assets |
|
|
|
|
(i)
Tangible assets |
828.300 |
875.400 |
888.100 |
|
(ii) Intangible Assets |
200.900 |
211.500 |
236.900 |
|
(iii) Capital work-in-progress |
12.600 |
12.800 |
2.500 |
|
(iv) Intangible assets under
development |
0.000 |
0.000 |
0.000 |
|
(b) Non-current Investments |
0.000 |
0.000 |
0.000 |
|
(c) Deferred tax assets (net) |
0.000 |
0.000 |
0.000 |
|
(d) Long-term Loan and Advances |
18.000 |
25.200 |
26.000 |
|
(e) Other Non-current assets |
5.500 |
6.700 |
5.900 |
|
Total
Non-Current Assets |
1065.300 |
1131.600 |
1159.400 |
|
|
|
|
|
|
(2) Current assets |
|
|
|
|
(a) Current investments |
0.000 |
0.000 |
0.000 |
|
(b) Inventories |
293.700 |
287.900 |
263.100 |
|
(c) Trade receivables |
487.400 |
300.900 |
383.500 |
|
(d) Cash and cash equivalents |
7.800 |
5.600 |
4.800 |
|
(e) Short-term loans and
advances |
119.300 |
99.300 |
82.800 |
|
(f) Other current assets |
84.100 |
0.000 |
0.000 |
|
Total
Current Assets |
992.300 |
693.700 |
734.200 |
|
|
|
|
|
|
TOTAL |
2057.600 |
1825.300 |
1893.600 |
PROFIT & LOSS
ACCOUNT
|
|
PARTICULARS |
31.03.2013 |
31.03.2012 |
31.03.2011 |
|
|
|
SALES |
|
|
|
|
|
|
|
Income |
2216.400 |
1928.200 |
1868.300 |
|
|
|
Other Income |
17.800 |
6.900 |
7.200 |
|
|
|
TOTAL (A) |
2234.200 |
1935.100 |
1875.500 |
|
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
|
|
Cost of Materials Consumed |
547.900 |
446.500 |
450.000 |
|
|
|
Purchases of Stock-in-Trade |
188.000 |
119.500 |
142.900 |
|
|
|
Employee Benefits Expense |
552.000 |
499.900 |
402.400 |
|
|
|
Other Expenses |
739.300 |
694.800 |
643.200 |
|
|
|
Changes in
Inventories of Finished Goods, Work-in-Progress and Stock-in-Trade |
28.500 |
(14.000) |
(95.700) |
|
|
|
TOTAL (B) |
2055.700 |
1746.700 |
1542.800 |
|
|
|
|
|
|
|
|
Less |
PROFIT
/ (LOSS) BEFORE INTEREST, TAX, DEPRECIATION AND AMORTISATION (A-B) (C) |
178.500 |
188.400 |
332.700 |
|
|
|
|
|
|
|
|
|
Less |
FINANCIAL
EXPENSES (D) |
74.400 |
68.800 |
62.800 |
|
|
|
|
|
|
|
|
|
|
PROFIT
/ (LOSS) BEFORE TAX, DEPRECIATION AND AMORTISATION (C-D) (E) |
104.100 |
119.600 |
269.900 |
|
|
|
|
|
|
|
|
|
Less/ Add |
DEPRECIATION/
AMORTISATION (F) |
98.700 |
102.200 |
95.200 |
|
|
|
|
|
|
|
|
|
|
PROFIT / (LOSS)
BEFORE TAX (E-F) (G) |
5.500 |
17.400 |
174.700 |
|
|
|
|
|
|
|
|
|
Less |
TAX (H) |
(38.700) |
8.900 |
47.600 |
|
|
|
|
|
|
|
|
|
|
PROFIT / (LOSS)
AFTER TAX (G-H) (I) |
44.100 |
8.500 |
127.100 |
|
|
|
|
|
|
|
|
|
Add |
PREVIOUS
YEARS’ BALANCE BROUGHT FORWARD |
233.500 |
240.400 |
153.600 |
|
|
|
|
|
|
|
|
|
Less |
APPROPRIATIONS |
|
|
|
|
|
|
|
Transfer to General Reserve |
2.200 |
0.000 |
9.500 |
|
|
|
Proposed Dividend |
19.800 |
13.200 |
26.500 |
|
|
|
Tax on Proposed Dividend |
3.400 |
2.200 |
4.300 |
|
|
BALANCE CARRIED
TO THE B/S |
252.200 |
233.500 |
240.400 |
|
|
|
|
|
|
|
|
|
|
EARNINGS IN
FOREIGN CURRENCY |
|
|
|
|
|
|
|
Export Earnings |
618.600 |
506.900 |
556.827 |
|
|
|
Freight and Insurance |
12.900 |
11.200 |
10.611 |
|
|
TOTAL EARNINGS |
631.500 |
518.100 |
567.438 |
|
|
|
|
|
|
|
|
|
|
IMPORTS |
|
|
|
|
|
|
|
Raw Materials |
107.200 |
92.800 |
108.655 |
|
|
|
Stores & Spares |
2.300 |
1.900 |
2.299 |
|
|
|
Capital Goods |
5.100 |
14.500 |
12.699 |
|
|
TOTAL IMPORTS |
114.600 |
109.200 |
123.653 |
|
|
|
|
|
|
|
|
|
|
Earnings /
(Loss) Per Share (Rs.) (Basic) |
2.67 |
0.51 |
8.04 |
|
|
|
Earnings /
(Loss) Per Share (Rs.) (Diluted) |
2.67 |
0.51 |
7.69 |
|
QUARTERLY RESULTS
|
Particulars |
30.06.2013 |
30.09.2013 |
31.12.2013 |
|
|
1st
Quarter |
2nd
Quarter |
3rd
Quarter |
|
Audited / UnAudited |
UnAudited |
UnAudited |
UnAudited |
|
Net Sales |
641.800 |
616.100 |
568.000 |
|
Total Expenditure |
557.600 |
578.700 |
596.800 |
|
PBIDT (Excl OI) |
84.200 |
37.400 |
(28.800) |
|
Other Income |
8.400 |
8.500 |
4.900 |
|
Operating Profit |
92.600 |
45.900 |
(23.900) |
|
Interest |
16.100 |
2.600 |
3.400 |
|
Exceptional Items |
642.600 |
0.000 |
0.000 |
|
PBDT |
719.100 |
43.300 |
(27.300) |
|
Depreciation |
25.400 |
26.300 |
27.100 |
|
Profit Before Tax |
693.700 |
17.000 |
(54.400) |
|
Tax |
146.800 |
(5.400) |
(24.200) |
|
Provisions and contingencies |
0.000 |
0.00 |
0.000 |
|
Profit After Tax |
546.900 |
22.400 |
(30.200) |
|
Extraordinary Items |
0.000 |
0.000 |
0.000 |
|
Prior Period Expenses |
0.000 |
0.000 |
0.000 |
|
Other Adjustments |
0.000 |
0.000 |
0.000 |
|
Net Profit |
546.900 |
22.400 |
(30.200) |
KEY RATIOS
|
PARTICULARS |
|
31.03.2013 |
31.03.2012 |
31.03.2011 |
|
PAT / Total Income |
(%) |
1.97
|
0.44 |
6.78 |
|
|
|
|
|
|
|
Net Profit Margin (PBT/Sales) |
(%) |
0.25
|
0.90 |
9.35 |
|
|
|
|
|
|
|
Return on Total Assets (PBT/Total Assets} |
(%) |
0.27
|
0.96 |
9.24 |
|
|
|
|
|
|
|
Return on Investment (ROI) (PBT/Networth) |
|
0.01
|
0.02 |
0.23 |
|
|
|
|
|
|
|
Debt Equity Ratio (Total Debt /Networth) |
|
0.62
|
0.61 |
0.60 |
|
|
|
|
|
|
|
Current Ratio (Current Asset/Current Liability) |
|
0.92
|
0.96 |
0.97 |
FINANCIAL ANALYSIS
[all figures are
in Rupees Millions]
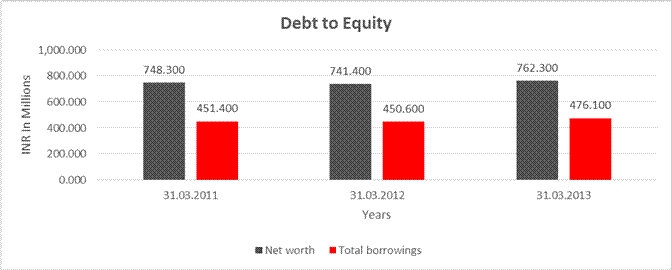
DEBT EQUITY RATIO
|
Particular |
31.03.2011 |
31.03.2012 |
31.03.2013 |
|
|
(Rs. In Millions) |
(Rs. In Millions) |
(Rs. In Millions) |
|
Share Capital |
132.300 |
132.300 |
132.300 |
|
Reserves & Surplus |
616.000 |
609.100 |
630.000 |
|
Net
worth |
748.300 |
741.400 |
762.300 |
|
|
|
|
|
|
long-term borrowings |
248.000 |
206.700 |
105.200 |
|
Short term borrowings |
203.400 |
243.900 |
370.900 |
|
Total
borrowings |
451.400 |
450.600 |
476.100 |
|
Debt/Equity
ratio |
0.603 |
0.608 |
0.625 |
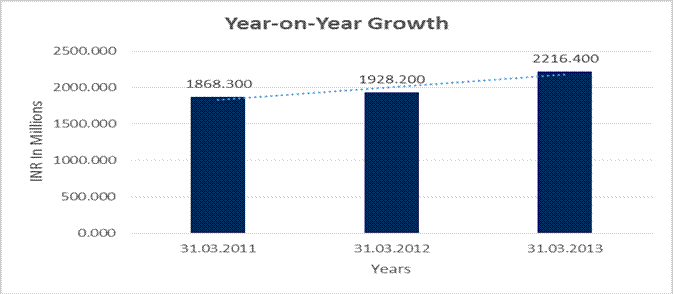
YEAR-ON-YEAR GROWTH
|
Year
on Year Growth |
31.03.2011 |
31.03.2012 |
31.03.2013 |
|
|
(Rs. In Millions) |
(Rs. In Millions) |
(Rs. In Millions) |
|
Sales |
1868.300 |
1928.200 |
2216.400 |
|
|
|
3.206 |
14.947 |
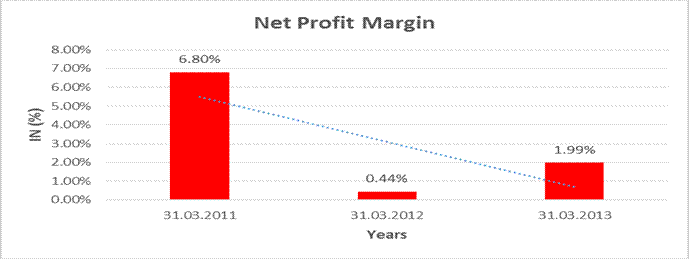
NET PROFIT MARGIN
|
Net
Profit Margin |
31.03.2011 |
31.03.2012 |
31.03.2013 |
|
|
(Rs. In Millions) |
(Rs. In Millions) |
(Rs. In Millions) |
|
Sales |
1868.300 |
1928.200 |
2216.400 |
|
Profit |
127.100 |
8.500 |
44.100 |
|
|
6.80% |
0.44% |
1.99% |
LOCAL AGENCY FURTHER INFORMATION
|
Sr. No. |
Check List by Info Agents |
Available in
Report (Yes / No) |
|
1] |
Year of Establishment |
Yes |
|
2] |
Locality of the firm |
Yes |
|
3] |
Constitutions of the firm |
Yes |
|
4] |
Premises details |
No |
|
5] |
Type of Business |
Yes |
|
6] |
Line of Business |
Yes |
|
7] |
Promoter's background |
Yes |
|
8] |
No. of employees |
No |
|
9] |
Name of person contacted |
No |
|
10] |
Designation of contact
person |
No |
|
11] |
Turnover of firm for last
three years |
Yes |
|
12] |
Profitability for last
three years |
Yes |
|
13] |
Reasons for variation
<> 20% |
-- |
|
14] |
Estimation for coming
financial year |
No |
|
15] |
Capital in the business |
Yes |
|
16] |
Details of sister
concerns |
Yes |
|
17] |
Major suppliers |
No |
|
18] |
Major customers |
No |
|
19] |
Payments terms |
No |
|
20] |
Export / Import details
(if applicable) |
No |
|
21] |
Market information |
-- |
|
22] |
Litigations that the firm
/ promoter involved in |
Yes |
|
23] |
Banking Details |
Yes |
|
24] |
Banking facility details |
Yes |
|
25] |
Conduct of the banking
account |
-- |
|
26] |
Buyer visit details |
-- |
|
27] |
Financials, if provided |
Yes |
|
28] |
Incorporation details, if
applicable |
Yes |
|
29] |
Last accounts filed at
ROC |
Yes |
|
30] |
Major Shareholders, if
available |
Yes |
|
31] |
Date of Birth of
Proprietor/Partner/Director, if available |
Yes |
|
32] |
PAN of
Proprietor/Partner/Director, if available |
No |
|
33] |
Voter ID No of
Proprietor/Partner/Director, if available |
No |
|
34] |
External Agency Rating,
if available |
Yes |
LITIGATION DETAILS
CASE DETAILS
BENCH:-BOMBAY
|
|
Lodging No.:- |
ITXAL/415/2010 |
Filing Date:- |
20/02/2010 |
Reg. No.:- |
ITXA/3427/2010 |
Reg. Date:- |
03/06/2010 |
|
|
|
Petitioner:- |
Commissioner
of income tax – 7, Mumbai - |
Respondent:- |
M/s.
RPG Life Sciences Limited, Mumbai |
|
|
|
Petn.Adv.:- |
M/S.
Padma Divakar |
Resp.Adv.:- |
B
V Jhaveri |
|
|
|
District:- |
MUMBAI |
|
|
|
Bench:- |
DIVISION |
|
|
|
|
|
Status:- |
Admitted(Unready) |
Category:- |
TAX APPEALS |
|
|
|
Last Date:- |
23/07/2012 |
Stage:- |
APPEALS FOR ADMISSION - FRESH [ORIGINAL
SIDE MATTERS] |
|
|
|
Last Coram:- |
HON'BLE SHRI JUSTICE S.J. VAZIFDAR |
|
|
|
|
HON'BLE SHRI JUSTICE M.S. SANKLECHA |
|
|
|
Act :- |
Income Tax Act, 1961 |
Under Section:- |
260A |
OPERATIONS
The Company earned a total income of Rs. 2234.200 Millions for the year as compared to Rs. 1935.100 Millions during the previous year. The Company earned a profit before tax of Rs.5.400 Millions as against Rs.17.400 Millions during the previous year. After writing back the deferred tax benefit of Rs.38.700 Millions, the profit after tax for the year stood at Rs.44.100 Millions as compared to Rs.8.500 Millions for the previous year.
During the year, the performance of the Company was adversely affected due to cancellation of orders by some key customers due to non-receipt of EU GMP certification for the Active Pharmaceutical Ingredient (API) facility at Navi Mumbai.
In April 2013, the Company has since received the EU GMP approval from the Ministry of Social and Family Affairs, Health and Consumer Protection, Hamburg, Germany for the API facility at Navi Mumbai, Maharashtra. The Company continues to hold a WHO GMP and TGA, Australia certification for the API facility at the Navi Mumbai plant.
ACTIVE PHARMACEUTICAL
INGREDIENTS (API/BULK DRUGS) BUSINESS:
PERFORMANCE
The API business achieved sales revenue of Rs. 279.500 Millions, a marginal growth of 2% over the last year. Due to the setback in the previous year with regard to the withdrawal of EUGMP certification, the performance continued to be adversely impacted during the current year as well. However, the re-audit of the API facility at Navi Mumbai took place during November 2012. The audit findings were favourable and consequently the company has received the certification in April 2013. The product specific certification (CEP) is expected in the first quarter of the next financial year. The traditional markets of Latin America where the Company enjoys a major market share for Quinfamide, Haloperidol and Lamotrigine continued to perform well, while sales of Pantoprazole in the domestic marketgrew by 175% over the previous year.
OUTLOOK
API business has a strategic importance in the overall growth of the company. The company has a backward integration for Di-phenoxylate and Azathioprine.
One of the key objectives of the Company is to build a strong and sustainable product portfolio. The primary focus during the current year was to achieve EU GMP certification. Going forward, efforts will be directed at new product introduction in various keysegments.
GLOBAL GENERICS BUSINESS:
PERFORMANCE
The Global Generics business achieved sales revenue of Rs. 173.100 Millions, with a growth of31% over the previous year. The key factor for the growth was the increased focus on existing clients in Canada and UK. Today, given the dedicated facility for manufacturing immunosup pressant APIs and also oral solid dosage forms, the company enjoys a large share of the Azathioprine market. But more generic companies are entering the market with Azathioprine formulations, increasing the competition in this product.
OUTLOOK
The company is constantly evaluating generic opportunities for both advanced markets like Europe and South Africa and major emerging markets like Russia and other CIS states. Opportunities are also being evaluated to develop and manufacture private label products for pharmacy chains, which have secured a large share of business of both generics and branded generics in markets like Canada.
The Company has a robust business plan for Azathioprine formulations. Globally the product generates revenues of about USD 180 mn, with the largest markets being Europe, Canada and USA. The Company has drawn up its entry strategy for all these markets for Azathioprine. The company has secured definitive agreements for launch of products in new markets like Australia, New Zealand, Germany and other EU Countries which will result in awider presence for the company's products.
In the coming year, the company expects to increase the footprint of its flagship product Azathioprine Tablets in all strengths and launch this product in Germany and certain other European countries for tenders as well as private business.
BIOTECH BUSINESS:
PERFORMANCE
The Biotech business achieved Rs. 240 .000 Millions in Sales and has done well as compared to the previous year. This business has shown a growth of 88% in spite of intense competition from domestic as well as Chinese and Korean companies. New tenders in the traditional markets such as Latin America and new geographies such as Egypt, Indonesia, Korea and Turkey helped international business to grow by 90% over the previous year. New clients in Korea and Indonesia were developed.
The Company continued to make several improvements and upgrades in its Biotech facility, to meet the requirements of various markets in emerging geographies. In line with its strategy to take the products to regulated markets, the Company has filed COS(Certificate of Suitability) for Doxorubicin with EDQM (European Directive Quality Medicines) at France and also has a plan to file DMFs (Drug Master File) in the current financial year.
OUTLOOK
The yields of the products were substantially improved by continuous RandD efforts. Both, Doxorubicin and Epirubicin, used in the treatment of cancer and which are manufactured by the Company compete with the best in class in their category.
The oncology market is one of the fastest growing markets. The Company has decided to add synthetic oncology products to its existing portfolio of fermentation oncology products and four synthetic oncology APIs are already under scale up. The APIs so manufactured will be sold to major Oncology players in emerging markets.
FORMULATIONS
BUSINESS:
PERFORMANCE
During the year, the Formulations business achieved sales revenue ofRs.1486.000 Millions, a growth of 9% over the previous year. The Company ranked 65th in the Indian Pharmaceutical market as per Pharmatrac with its Nephrocare Division, ranked 3rdin Nephrology therapy. Business Verticals of Oncology and Nephrology clocked growth of 50%and 21% respectively over last year. However, profitability was affected by substantially higher sales returns due to market conditions.
The concept of Focus Brands, which was introduced in the year has fuelled growth. Focus brands in Domestic Formulation have shown growth of about 25% with Impulse and Nephrology focus brands registering growth of 70% and 49% respectively.
OUTLOOK:
In the current year, the company's focus will be to leverage the strength of the legacy brands so as to build successful new products along with continued emphasis on Focus brands. Dedicated efforts on chronic therapies like Cardiology, Anti-Diabetic, Oncology and Nephrology will be made to accelerate the growth in the chronic therapy segment. With focus on Transplant and Dialysis therapy the company will consolidate its value in Nephrology therapy.
Existing brands like Aldactone, Naprosyn, Azoran and Tricane will continue to receive the necessary thrust as also other focus brands like RPO, Alfalog, Azopen andanti-diabetic products such as Cicoline, CVMET, Olrass and Glimetop. New launches Azistart-O and MinMin Tonic, Qugyl OP are showing promise.
MANAGEMENT
DISCUSSIONS AND ANALYSIS REPORT
INDUSTRY STRUCTURE
AND DEVELOPMENTS
The Indian healthcare industry has evolved rapidly from being a product centric industry to a service driven sector with delivery (hospitals) and medical insurance segments gaining prominence. India's pharmaceutical industry has been ranked 3rdin terms of volume and 10th in terms of value, globally.
Optimistic estimates predict India's pharmaceutical sector to touch US$ 74 billion in sales by 2020 from the current US$ 11 billion. The Indian pharmaceutical market is expected to grow at a compounded annual growth rate (CAGR) of 14-17 per cent over 2012-16.India is now among the top five emerging pharmaceutical markets.
On back of increasing sales of generic medicines, continued growth in chronic therapies and a greater penetration in rural markets, the domestic pharmaceutical market in India is expected to register a strong double-digit growth of 13-14 per cent in 2013. The year 2012closed with a growth of 12 per cent, according to data from research firm IMS Health.
In FY '13, growth of the Domestic Formulation industry slipped to 11.9% from 16% in the last year. Slowdown in the Domestic Pharmaceutical Industry has been attributed to several factors like slowdown in new therapies due to restrictive approvals for New Products by Regulators, down-stocking by distributors in fear of the impending pharma policy etc.
SEGMENT WISE
PERFORMANCE
The Company is exclusively engaged in pharmaceutical business.
Global Formulation division catering to domestic market and rest of the world market achieved sales of Rs. 1486.000 Millions registering a growth of 9%. The Company has strengthened its presence in the market by leveraging the existing brands to grow newer products launched. The company also took a host of other initiatives such as increasing the in-clinic effectiveness of the field force through extensive scientific training, emphasis on Focus brands, innovative promotional strategies. These initiatives are expected to reap benefits in the current year too. In the export market, the revenues grew to Rs. 120.400 Millions from Rs.90.400 Millions, where the emphasis has been the immunosuppressant products to Asian, African and Latin American markets. Focus brands achieved growth of24%.
The Global Generics business achieved net sales of Rs. 173.100 Millions registering a growth of 31% during the year. New agreements have been executed with customers in Australia and New Zealand for Azathioprine tablets. New products development has been undertaken for the regulated markets. The company is conceiving a pipeline of products for regulated markets in Global Generics vertical. The Bulk Drug business achieved net sales of Rs. 279.500 Millions registering a growth of 2% during the year. The Biotech business achieved net sales of Rs. 240 .000 Millions with a phenomenal growth of 88%.
OUTLOOK
The growth estimate for the domestic Formulation Industry is pegged at 11% to 12%. In comparison to the Industry growth prediction, the Company has taken a growth target of 20%for products promoted through trade channels. In light of the initiatives detailed above, the outlook of the business looks positive.
In the current year the Company plans to launch new bulk drugs and also plans to foray into new markets with existing products. With these initiatives the bulk drugs business is expected to perform better in the current year.
BACKGROUND
Subjectwas incorporated on 29th March, 2007 as RPG Pharmaceuticals Limited. The name of the company was subsequently changed to RPG Life Sciences Limited on 13th February, 2008.
Pursuant to a Scheme of Arrangement, the company acquired the pharmaceuticals business of Brabourne Enterprises Limited (formerly RPG Life Sciences Limited) with retrospective effect from the appointed date of 2nd April, 2007.
UNSECURED LOAN
|
PARTICULARS |
31.03.2013 (Rs. in Millions) |
31.03.2012 (Rs. in Millions) |
|
Long-term
Borrowings |
|
|
|
Fixed Deposits |
0.000 |
45.600 |
|
Finance Lease Obligations |
1.500 |
0.000 |
|
Total |
1.500 |
45.600 |
INDEX OF CHARGES
|
S.NO. |
CHARGE ID |
DATE OF CHARGE
CREATION/MODIFICATION |
CHARGE AMOUNT
SECURED |
CHARGE HOLDER |
ADDRESS |
SERVICE REQUEST
NUMBER (SRN) |
|
1 |
10181270 |
04/06/2013 * |
659,200,000.00 |
UNION BANK OF INDIA |
239, VIDHAN BHAVAN MARG,,
NARIMAN POINT, MUMBAI, |
B76526151 |
* Date of charge modification
STAMEN OF STANDALONE UNAUDITED RESULTS FOR THE QUARTER AND NINE MONTHS
ENDED 31ST DECEMBER, 2013
|
Sr. No. |
Particular |
3
Months Ended |
9
months Ended |
|
|
|
|
31.12.2013 |
30.09.2013 |
31.12.2013 |
|
|
|
Unaudited |
Unaudited |
Unaudited |
|
|
|
|
|
|
|
1. |
Net Sales/Income
from Operations |
547.900 |
605.500 |
1782.400 |
|
|
Other Operating
Income |
20.100 |
10.600 |
43.500 |
|
|
Total Income From Operations (Net) |
568.000 |
616.100 |
1825.900 |
|
|
|
|
|
|
|
2. |
Expenditure |
|
|
|
|
|
Cost
of materials consumed |
151.400 |
181.500 |
523.900 |
|
|
Purchase
of stock in trade |
50.700 |
61.700 |
157.300 |
|
|
Changes
in inventories of finished goods, work in progress and stock in trade |
(8.500) |
(54.200) |
(97.700) |
|
|
Employee
benefits expenses |
151.000 |
174.000 |
472.200 |
|
|
Depreciation
and amortization expenses |
27.100 |
26.300 |
78.800 |
|
|
Provision
for doubtful debts and advances |
68.100 |
1.600 |
71.700 |
|
|
Other
expenses |
184.100 |
214.100 |
605.700 |
|
|
Total Expenses |
623.900 |
605.000 |
1811.900 |
|
|
|
|
|
|
|
3. |
Profit
From Operations before Other Income, Interest and Exceptional Items (1-2) |
(55.900) |
11.100 |
14.000 |
|
|
|
|
|
|
|
4. |
Other
Income |
4.900 |
8.500 |
21.800 |
|
|
|
|
|
|
|
5. |
Profit
Before Interest and Exceptional Items (3+4) |
(51.000) |
19.600 |
35.800 |
|
|
|
|
|
|
|
6. |
Interest |
3.400 |
2.600 |
22.100 |
|
|
|
|
|
|
|
7. |
Profit
After Interest but before Exceptional Items (5-6) |
(54.400) |
17.000 |
13.700 |
|
|
|
|
|
|
|
8. |
Exceptional
Items |
-- |
-- |
642.600 |
|
|
|
|
|
|
|
9. |
Profit
from Ordinary Activities before Tax (7+8) |
(54.400) |
17.000 |
656.300 |
|
|
|
|
|
|
|
10. |
Tax
Expense |
(24.200) |
(5.400) |
117.200 |
|
|
|
|
|
|
|
11. |
Net
Profit from Ordinary Activities after Tax (9-10) |
(30.200) |
22.400 |
539.100 |
|
|
|
|
|
|
|
12. |
Extraordinary
Item (net of expense) |
-- |
-- |
-- |
|
|
|
|
|
|
|
13. |
Net
Profit for the period (11-12) |
(30.200) |
22.400 |
539.100 |
|
|
|
|
|
|
|
14. |
Paid-up
Equity Share Capital (Face Value of Rs.8/- Each) |
132.300 |
132.300 |
132.300 |
|
|
|
|
|
|
|
15. |
Reserves
Excluding Revaluation Reserve |
-- |
--- |
-- |
|
|
|
|
|
|
|
16. |
Basic and Diluted Earning Per
Share (EPS) (Rs.)-Not Annualised |
|
|
|
|
|
a)
Basic and diluted EPS before extraordinary items |
(1.83) |
1.36 |
32.60 |
|
|
b)
Basic and diluted EPS after extraordinary items |
(1.83) |
1.36 |
32.60 |
|
|
|
|
|
|
|
17. |
Public Shareholding |
|
|
|
|
|
-Number
of Shares |
7196344 |
7196344 |
7196344 |
|
|
-
Percentage of Shareholding |
43.52 |
43.52 |
43.52 |
|
|
|
|
|
|
|
18. |
Promoters and Promoter Group
Shareholding |
|
|
|
|
|
a) Pledged/Encumbered |
|
|
|
|
|
-
Number of Shares |
-- |
-- |
-- |
|
|
- Percentage
of Shares (as a % of the Total Shareholding of promoter and promoter group) |
-- |
-- |
-- |
|
|
-
Percentage of Shares (as a % of the Total Share Capital of the Company) |
-- |
-- |
-- |
|
|
|
|
|
|
|
|
b) Non Encumbered |
|
|
|
|
|
- Number
of Shares |
9338897 |
9338897 |
9338897 |
|
|
-
Percentage of Shares (as a % of the Total Shareholding of Promoter and
Promoter Group) |
100.00 |
100.00 |
100.00 |
|
|
-
Percentage of Shares (as a % of the Total Share Capital of the Company) |
56.48 |
56.48 |
56.48 |
|
Particulars |
3 Months Ended 31.12.2013 |
|
Pending at the beginning of the quarter |
Nil |
|
Received during the quarter |
6 |
|
Disposed of during the quarter |
6 |
|
Remaining unresolved at the end of the
quarter |
Nil |
NOTE:
The above results have been reviewed by the Audit Committee and approved by the Board of Directors at its meeting held on February 07, 2014.
The results for the quarter ended December 31, 2013 have been subjected to
limited review by the statutory auditors of the company.
The company is exclusively engaged in the pharmaceuticals segment,
Provision for Doubtful Debts and Advances (Net) for the quarter and nine months ended December 31, 2013 includes an amount of Rs. 67.400 Millions towards receivable from certain parties to whom pharmaceutical goods were supplied as per contracts and for which these parties have reneged on the contractual payment terms. The company has initiated legal action against these parties. Without prejudice to the position that Rs. 67.400 Millions is recoverable, the company has made provision for the same.
Exceptional Items for the nine months ended December 31, 2013 comprise of
profit of Rs. 618.400 Millions on sale of a portion
of leasehold land and building thereon and the company's share of interest
income of Rs. 24.200 Millions earned on the sale
consideration deposited by the buyer in an escrow account, jointly held in the
name of the company and the buyer, till the execution of the sale deed. The
amount of sale consideration and the company's share of interest income lying
in the escrow account has been transferred to the company on June 06, 2013, the
date of execution of the sale deed.
Figures for the prior periods have been regrouped where necessary.
FIXED ASSETS
·
Goodwill
·
Technical Knowhow
·
Computer Software
·
Leasehold Land
·
Building
·
Plant and Machinery
·
Furniture, Fittings and Office Equipments
·
Vehicles
CMT REPORT (Corruption, Money Laundering & Terrorism]
The Public Notice information has been collected from various sources
including but not limited to: The Courts,
1] INFORMATION ON
DESIGNATED PARTY
No exist designating subject or any of its beneficial owners,
controlling shareholders or senior officers as terrorist or terrorist
organization or whom notice had been received that all financial transactions
involving their assets have been blocked or convicted, found guilty or against
whom a judgement or order had been entered in a
proceedings for violating money-laundering, anti-corruption or bribery or
international economic or anti-terrorism sanction laws or whose assets were
seized, blocked, frozen or ordered forfeited for violation of money laundering
or international anti-terrorism laws.
2] Court Declaration :
No exist to suggest that subject is or was
the subject of any formal or informal allegations, prosecutions or other
official proceeding for making any prohibited payments or other improper
payments to government officials for engaging in prohibited transactions or
with designated parties.
3] Asset Declaration :
No records exist to suggest that the property or assets of the subject
are derived from criminal conduct or a prohibited transaction.
4] Record on Financial
Crime :
Charges or conviction
registered against subject: None
5] Records on Violation of
Anti-Corruption Laws :
Charges or
investigation registered against subject: None
6] Records on Int’l
Anti-Money Laundering Laws/Standards :
Charges or
investigation registered against subject: None
7] Criminal Records
No
available information exist that suggest that subject or any of its principals
have been formally charged or convicted by a competent governmental authority
for any financial crime or under any formal investigation by a competent
government authority for any violation of anti-corruption laws or international
anti-money laundering laws or standard.
8] Affiliation with
Government :
No record
exists to suggest that any director or indirect owners, controlling
shareholders, director, officer or employee of the company is a government
official or a family member or close business associate of a Government
official.
9] Compensation Package :
Our market
survey revealed that the amount of compensation sought by the subject is fair
and reasonable and comparable to compensation paid to others for similar
services.
10] Press Report :
No press reports / filings exists on
the subject.
CORPORATE GOVERNANCE
MIRA INFORM as part of its Due Diligence do provide comments on
Corporate Governance to identify management and governance. These factors often
have been predictive and in some cases have created vulnerabilities to credit
deterioration.
Our Governance Assessment focuses principally on the interactions
between a company’s management, its Board of Directors, Shareholders and other
financial stakeholders.
CONTRAVENTION
Subject is not known to have contravened any existing local laws,
regulations or policies that prohibit, restrict or otherwise affect the terms
and conditions that could be included in the agreement with the subject.
FOREIGN EXCHANGE RATES
|
Currency |
Unit
|
Indian Rupees |
|
US Dollar |
1 |
Rs.60.21 |
|
|
1 |
Rs.101.05 |
|
Euro |
1 |
Rs.83.33 |
INFORMATION DETAILS
|
Information
Gathered by : |
NYA |
|
|
|
|
Analysis Done by
: |
RAS |
|
|
|
|
Report Prepared
by : |
KVT |
SCORE & RATING EXPLANATIONS
|
SCORE FACTORS |
RANGE |
POINTS |
|
HISTORY |
1~10 |
5 |
|
PAID-UP CAPITAL |
1~10 |
5 |
|
OPERATING SCALE |
1~10 |
6 |
|
FINANCIAL CONDITION |
|
|
|
--BUSINESS SCALE |
1~10 |
6 |
|
--PROFITABILIRY |
1~10 |
6 |
|
--LIQUIDITY |
1~10 |
5 |
|
--LEVERAGE |
1~10 |
5 |
|
--RESERVES |
1~10 |
6 |
|
--CREDIT LINES |
1~10 |
5 |
|
--MARGINS |
-5~5 |
-- |
|
DEMERIT POINTS |
|
|
|
--BANK CHARGES |
YES/NO |
YES |
|
--LITIGATION |
YES/NO |
YES |
|
--OTHER ADVERSE INFORMATION |
YES/NO |
NO |
|
MERIT POINTS |
|
|
|
--SOLE DISTRIBUTORSHIP |
YES/NO |
NO |
|
--EXPORT ACTIVITIES |
YES/NO |
NO |
|
--AFFILIATION |
YES/NO |
NO |
|
--LISTED |
YES/NO |
YES |
|
--OTHER MERIT FACTORS |
YES/NO |
YES |
|
DEFAULTER |
|
|
|
--RBI |
YES/NO |
NO |
|
--EPF |
YES/NO |
NO |
|
TOTAL |
|
49 |
This score serves as a reference to assess SC’s credit risk
and to set the amount of credit to be extended. It is calculated from a
composite of weighted scores obtained from each of the major sections of this
report. The assessed factors and their relative weights (as indicated through
%) are as follows:
Financial
condition (40%) Ownership
background (20%) Payment
record (10%)
Credit history
(10%) Market trend
(10%) Operational
size (10%)
RATING EXPLANATIONS
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
>86 |
Aaa |
Possesses an extremely sound financial base with the strongest
capability for timely payment of interest and principal sums |
Unlimited |
|
71-85 |
Aa |
Possesses adequate working capital. No caution needed for credit
transaction. It has above average (strong) capability for payment of interest
and principal sums |
Large |
|
56-70 |
A |
Financial & operational base are regarded healthy. General unfavourable factors will not cause fatal effect.
Satisfactory capability for payment of interest and principal sums |
Fairly Large |
|
41-55 |
Ba |
Overall operation is considered normal. Capable to meet normal
commitments. |
Satisfactory |
|
26-40 |
B |
Capability to overcome financial difficulties seems comparatively
below average. |
Small |
|
11-25 |
Ca |
Adverse factors are apparent. Repayment of interest and principal sums
in default or expected to be in default upon maturity |
Limited with full
security |
|
<10 |
C |
Absolute credit risk exists. Caution needed to be exercised |
Credit not
recommended |
|
-- |
NB |
New Business |
-- |
This report is issued at your request without any
risk and responsibility on the part of MIRA INFORM PRIVATE LIMITED (MIPL) or
its officials.