MIRA
INFORM REPORT
|
Report Date : |
19.04.2014 |
IDENTIFICATION DETAILS
|
Name : |
GLENMARK PHARMACEUTICALS LIMITED |
|
|
|
|
Registered
Office : |
B/2, Mahalaxmi
Chambers, 22, Bhulabhai Desai Road, Mumbai – 400026, Maharashtra |
|
|
|
|
Country : |
|
|
|
|
|
Financials (as
on) : |
31.03.2013 |
|
|
|
|
Date of
Incorporation : |
18.11.1977 |
|
|
|
|
Com. Reg. No.: |
11-19982 |
|
|
|
|
Capital
Investment / Paid-up Capital : |
Rs.270.850 Millions |
|
|
|
|
CIN No.: [Company Identification
No.] |
L24299MH1977PLC019982 |
|
|
|
|
TAN No.: [Tax Deduction &
Collection Account No.] |
MUMG07883B |
|
|
|
|
Legal Form : |
A Public Limited Liability Company. The Company’s Shares are Listed on
the Stock Exchange. |
|
|
|
|
Line of Business
: |
Manufacturing and
Marketing of Pharmaceutical Products |
|
|
|
|
No. of Employees
: |
7000 (Approximately) |
RATING & COMMENTS
|
MIRA’s Rating : |
A (68) |
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
56-70 |
A |
Financial & operational base are regarded healthy. General
unfavourable factors will not cause fatal effect. Satisfactory capability for
payment of interest and principal sums. |
Fairly Large |
|
Maximum Credit Limit : |
USD 101000000 |
|
|
|
|
Status : |
Good |
|
|
|
|
Payment Behaviour : |
Regular |
|
|
|
|
Litigation : |
Exists |
|
|
|
|
Comments : |
Subject is a well-established company having fine track. The rating reflects healthy financial risk profile marked by strong
liquidity position and decent profitability of the company. Trade relations are reported as fair. Business is active. Payments are
reported to be regular and as per commitments. The company can be considered good for normal business dealings at
usual trade terms and conditions. |
NOTES:
Any query related to this report can be made
on e-mail : infodept@mirainform.com
while quoting report number, name and date.
ECGC Country Risk Classification List – March 31, 2014
|
Country Name |
Previous Rating (31.12.2013) |
Current Rating (31.03.2014) |
|
India |
A1 |
A1 |
|
Risk Category |
ECGC
Classification |
|
Insignificant |
A1 |
|
Low |
A2 |
|
Moderate |
B1 |
|
High |
B2 |
|
Very High |
C1 |
|
Restricted |
C2 |
|
Off-credit |
D |
INDIAN ECONOMIC OVERVIEW
US investment bank Goldman
Sachs has upgraded its outlook on Indian markets as it expects positive impact
of the election cycle.
India’s economy may
grow 4.7 % in the current financial year, lower than the official estimate of
4.9 %, Fitch Rating said. The global rating agency expects the economy to pick
up in the next two financial years.
Global ratings
agency Standard & Poor said increasing focus by India Inc on lowering debt
is likely to improve their credit profiles.
Singapore (1.1 million
Indian tourists in 2012), Thailand (one million), the United Arab Emirates
().98 million) and Malaysia ().82 million) emerged as the preferred holidays
hotspots for Indians. The total figure is expected to increase to 1.93 million
by 2017, according to the latest Eurmonitor international report.
There is a $29.34 bn
outward foreign direct investment by domestic companies between April and
January of 2013/14 which has seen some signs of recovery according to a Care
Ratings report.
There are 264 number
of new companies being set up every day on average during 2014. Most of them
are registered in Mumbai. India had 1.38 million registered companies at the
end of January, 2014.
Twitter like
messaging service Weibo Corporation has filed to raise $ 500 million via a US
initial public offering. Alibaba, which owns a stake in Weibo is expected to
raise about $ 15 billion New York this year in the highest profile Internet IPO
since Facebook’s in 2012.
Bharti Airtel has
raised Rs.2,453.2 crore (350 million Swiss Francs) by selling six-year bonds at
a coupon rate of three per cent and maturing in 2020. This is the largest ever
bond offering by an Indian company in Swiss Francs. Bharat Petroleum
Corporation raised 175 million Swiss Francs by selling five year bonds at 2.98
% coupon rate in February.
Indian Oil
Corporation plans to invest Rs.7650 crore in setting up a petrochemical complex
at its almost complete Paradip refinery in Odhisha in three to four years. The
company board is set to consider the setting up of a 700000 tonne per annum
polypropylene plant at an estimated cost at Rs.3150 crore.
Global chief
information officers at gathering in Bangalore in April to meet Indian startups
at an event called Tech50 Watchout for Little Eye Labs-Facebook type deals in
the making.
EXTERNAL AGENCY RATING
|
Rating Agency Name |
CRISIL |
|
Rating |
Long Term Rating: AA- |
|
Rating Explanation |
High degree of safety and very low credit
risk |
|
Date |
18.03.2013 |
|
Rating Agency Name |
CRISIL |
|
Rating |
Short Term Rating: A1+ |
|
Rating Explanation |
Very strong degree of safety and lowest
credit risk. |
|
Date |
18.03.2013 |
RBI DEFAILTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available RBI Defaulters’ list.
EPF (Employee Provident Fund) DEFAILTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available EPF (Employee Provident Fund) Defaulters’ list as of
31-03-2012.
LOCATIONS
|
Registered/ Administrative Office : |
B/2, Mahalaxmi Chambers, 22, Bhulabhai
Desai Road, Mumbai – 400026, Maharashtra, India |
|
Tel. No.: |
91-22-24964893/ 24964894/ 24964895/
24964896/ 56549999/ 55902491/ 92 |
|
Fax No.: |
91-22-24932648/ 23512177/ 23519652 |
|
E-Mail : |
|
|
Website : |
|
|
|
|
|
Corporate Office : |
Gelnmark House, HDO – Corporate Building
Wing A, B D Sawant Marg, Chakala, Off Western Express Highway, Andheri
(East), Mumbai – 400099, Maharashtra, India |
|
Tel. No.: |
91-22-40189999 |
|
Fax No.: |
91-22-40189986 |
|
|
|
|
Manufacturing Facilities : |
Formulations ·
E – 37, MIDC Industrial Area, D-Road, Satpur,
Nasik – 422007, Maharashtra, India ·
Plot No. 7, Colvale Industrial Estate, Bardez –
403 115, ·
D-42, Plot No. 50, Kundaim Industrial Estate,
Kundaim – 403 115, Goa, India ·
Unit – I, Village-Kishanpura, Baddi Nalagarh
Road, Tehsil Nalagarh, District Solan, Baddi – 174 101, Himachal Pradesh,
India ·
Business Unit II, Village Bhattanwala, PO
Rajpura, Nalagarh District Solan, ·
Unit - III, Village Kishanpura,
Baddi-Nalagarh Road, Dist. - Solan – 174101, Himachal Pradesh, India ·
Plot No. 2, Phase-II, Pharma Zone, Special
Economic Zone Area, Pitampur, Indore – 454 775, Madhya Pradesh, India ·
Rua Assahi, 33-1, Andar CEP: 09633-0110, Rudge
Ramos ·
Rua Frei Liberato De Gries, 548, Jardim
Arpoadar, CEP: 05572-210, ·
Glenmark Pharmaceuticals S.R.O., Fibichova 143,
56617, Vysoke Myto, Czech Republic ·
Calle 9 Ing Meyer Oks N 593, Parque Industrial
Pilar, B1629MX Buenos Aires, Argentina
·
Growth Centre, Samlik-Marchak, District – East
Sikkim, Sikkim. API ·
3109-C, GIDC Industrial Estate, Ankleshwar,
District Bharuch - 393 002, ·
Plot No. 163-165/170-172, Chandramouli Industrial
Estate, Mohal Bazarpeth, ·
Plot No. A80, MIDC Area, Kurkumbh, Daund, Pune
– 413 802, ·
Z-103 I, Dahej SEZ, Dahej District, Bharuch,
Gujarat, Indi ·
Plot No.B-25, Five Star MIDC,
Shendra, District Aurangabad, Maharashtra, India |
|
|
|
|
R and D Centers : |
·
Plot
No. A-607, TTC Industrial Area, MIDC, Mahape, Vashi, Navi Mumbai - 400 705, ·
Chemin
de la Combeta 5, 2300 La Chaux-de-fonds, ·
Plot No.
C 152, MIDC Sinnar Industrial Area, ·
Plot
No. M4, Taloja Industrial Area, MIDC Taloja, Takula Panvel – 410 208,
District – Raigad, |
|
|
|
|
Clinical
Research Centre : |
·
Plot
No. D 508, TTC Industrial Estate, MIDC, Turbhe, Navi Mumbai – 400705,
Maharashtra, India ·
Building
2, Croxley Green Business Park, Merlins Meadow, Watford, Hertfordshire, UK |
DIRECTORS
(AS ON 31.03.2013)
|
Name:
|
Mr. Glenn
Saldanha |
|
Designation : |
Chairman and Managing Director |
|
Qualification: |
B. Pharma /
M.B.A. |
|
|
|
|
Name:
|
Mrs. Cheryl
Pinto |
|
Designation : |
Director -
Corporate Affairs |
|
Qualification: |
Graduate in
Pharmacy |
|
|
|
|
Name:
|
Mr. Rajesh V.
Desai |
|
Designation : |
Executive
Director and CFO |
|
|
|
|
Name |
Mrs. B. E. Saldanha |
|
Designation |
Non-Executive Director |
|
Qualification : |
Graduated in Arts and Law |
|
|
|
|
Name |
Mr. D. R. Mehta |
|
Designation |
Non-Executive Director |
|
|
|
|
Name |
Mr. Sridhar Gorthi |
|
Designation |
Non-Executive Director |
|
Qualification : |
B.A., L.L.B.
(Hons.) from |
|
|
|
|
Name |
Mr. Bernard
Munos |
|
Designation |
Non-Executive Director |
|
|
|
|
Name |
Mr. Hocine Sidi
Said |
|
Designation |
Non-Executive Director |
|
Qualification : |
B.A.
(International Marketing) |
|
|
|
|
Name |
Mr. J. F.
Ribeiro |
|
Designation |
Non-Executive Director |
|
Qualification : |
Ex-IPS |
|
|
|
|
Name |
Mr. N. B. Desai
|
|
Designation |
Non-Executive Director |
|
|
|
|
Name : |
Dr. Brian W
Tempest |
|
Designation : |
Non-Executive Director |
KEY EXECUTIVES
|
Name : |
Mr. Sanjay Kumar Chowdhary |
|
Designation : |
Company Secretary |
MAJOR SHAREHOLDERS / SHAREHOLDING PATTERN
(AS ON 31.04.2013)
|
Category of Shareholder |
Total No. of Shares |
Total Shareholding as a % of Total No. of Shares |
|
(A) Shareholding of
Promoter and Promoter Group |
|
|
|
|
|
|
|
|
2713681 |
1.00 |
|
|
128241936 |
47.28 |
|
|
|
|
|
|
128241936 |
47.28 |
|
|
130955617 |
48.28 |
|
|
|
|
|
|
|
|
|
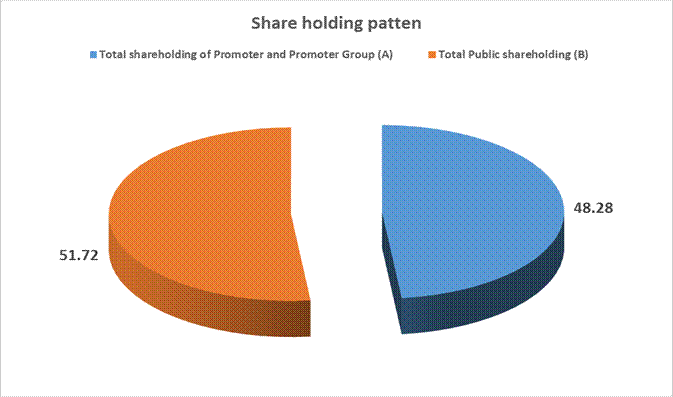
Total shareholding of
Promoter and Promoter Group (A) |
130955617 |
48.28 |
|
|
|
|
|
(B) Public Shareholding |
|
|
|
|
|
|
|
|
5413838 |
2.00 |
|
|
15743357 |
5.80 |
|
|
90133485 |
33.23 |
|
|
111290680 |
41.03 |
|
|
|
|
|
|
|
|
|
|
3786704 |
1.40 |
|
|
|
|
|
|
|
|
|
|
15000310 |
5.53 |
|
|
7786520 |
2.87 |
|
|
2403822 |
0.89 |
|
|
269026 |
0.10 |
|
|
380023 |
0.14 |
|
|
1229032 |
0.45 |
|
|
10004 |
0.00 |
|
|
514275 |
0.19 |
|
|
1462 |
0.00 |
|
|
28977356 |
10.68 |
|
|
|
|
|
Total Public shareholding
(B) |
140268036 |
51.72 |
|
|
|
|
|
Total (A)+(B) |
271223653 |
100.00 |
|
|
|
|
|
(C) Shares held by
Custodians and against which Depository Receipts have been issued |
|
|
|
|
0 |
0.00 |
|
|
0 |
0.00 |
|
|
0 |
0.00 |
|
|
|
|
|
Total (A)+(B)+(C) |
271223653 |
100.00 |
BUSINESS DETAILS
|
Line of Business : |
Manufacturing and
Marketing of Pharmaceutical Products |
||||||||
|
|
|
||||||||
|
Products : |
|
GENERAL INFORMATION
|
No. of Employees : |
7000 (Approximately) |
||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||
|
Bankers : |
· Bank of India Mahalaxmi Branch,
Mumbai – 400026, Maharashtra, India |
||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||
|
Facilities : |
|
||||||||||||||||||||||||
|
|
|
|
Banking
Relations : |
-- |
|
|
|
|
Auditors : |
|
|
Name : |
Walker, Chandiok and Company Chartered Accountants |
|
Address : |
Mumbai, Maharashtra, India |
|
|
|
|
Cost Auditors: |
|
|
Name: |
Sevekari Khare and Associates Ccost Accountants |
|
Address: |
Mumbai, Maharashtra, India |
|
|
|
|
Solicitor : |
· Kanga and Company, Mumbai, Maharashtra, India ·
Trilegal, Mumbai, Maharashtra, India |
|
|
|
|
Subsidiary
Companies: |
·
Glenmark Pharmaceuticals Europe Limited., U.K. ·
Glenmark Generics (Europe) Limited., U.K. ·
Glenmark Pharmaceuticals S.R.O., Czech Republic ·
Glenmark Pharmaceuticals SK, s.r.o., Slovak
Republic ·
Glenmark Pharmaceuticals S. A., Switzerland ·
Glenmark Holding S. A., Switzerland ·
Glenmark Generics Holding S. A., Switzerland ·
Glenmark Generics Finance S. A., Switzerland ·
Glenmark Pharmaceuticals S.R.L., Romania ·
Glenmark Pharmaceuticals Eood., Bulgaria ·
Glenmark Distributors SP z.o.o., Poland ·
Glenmark Pharmaceuticals SP z.o.o., Poland ·
Glenmark Generics Inc., USA ·
Glenmark Therapeutics Inc., USA ·
Glenmark Farmaceutica Ltda., Brazil ·
Glenmark Generics SA., Argentina ·
Glenmark Pharmaceuticals Mexico, S.A. DE C.V.,
Mexico ·
Glenmark Pharmaceuticals Peru SAC., Peru ·
Glenmark Pharmaceuticals Colombia Ltda., Colombia ·
Glenmark Uruguay S.A., Uruguay ·
Glenmark Pharmaceuticals Venezuela., C.A.,
Venezuela ·
Glenmark Dominicana, SRL, Dominican Republic ·
Glenmark Pharmaceuticals Egypt S.A.E., Egypt ·
Glenmark Pharmaceuticals FZE., United Arab
Emirates ·
Glenmark Impex L.L.C., Russia ·
Glenmark Philippines Inc., Philippines ·
Glenmark Pharmaceuticals (Nigeria) Limited.,
Nigeria ·
Glenmark Pharmaceuticals Malaysia Sdn Bhd.,
Malaysia ·
Glenmark Pharmaceuticals (Australia) Pty
Limited., Australia ·
Glenmark South Africa (Pty) Limited., South
Africa ·
Glenmark Pharmaceuticals South Africa (Pty)
Limited., South Africa ·
Glenmark Access Limited (formerly known as
Glenmark Exports Limited.) ·
Glenmark Generics Limited, India ·
Glenmark Generics B.V., Netherlands ·
Glenmark Arzneimittel Gmbh., Germany ·
Glenmark Generics Canada, Inc. ·
Glenmark Pharmaceuticals Kenya Limited, Kenya ·
Glenmark Therapeutics AG; Switzerland |
|
|
|
|
Investment in
Joint Venture: |
·
Glenmark Pharmaceuticals (Thailand) Company
Limited., Thailand |
|
|
|
|
Enterprise over which
key managerial personnel exercise significant influence: |
·
Glenmark Foundation, India |
CAPITAL STRUCTURE
(AS ON 02.08.2013)
Authorised Capital: Rs.750.000
Millions
Issued, Subscribed & Paid-up Capital: Rs.271.224 Millions
(AS ON 31.03.2013)
Authorised Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
350000000 |
Equity Shares |
Rs.1/- each |
Rs.350.000 Millions |
|
4000000 |
Cumulative Redeemable Non Convertible
Preference Shares |
Rs.100/- each |
Rs.400.000 Millions |
|
|
|
|
|
|
|
Total |
|
Rs.750.000
Millions |
Issued, Subscribed & Paid-up Capital:
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
270535503 |
Equity Shares |
Rs.10/- each |
Rs.270.530
Millions |
|
318150 |
Add: Issued during the year - Under the employee Stock Option Scheme,
2003 (ESOS) |
|
Rs.0.320
Millions |
|
|
|
|
|
|
|
Total |
|
Rs.270.850 Millions |
FINANCIAL DATA
[All figures are
in Rupees Millions]
ABRIDGED BALANCE SHEET
|
SOURCES OF FUNDS |
31.03.2013 |
31.03.2012 |
31.03.2011 |
|
I.
EQUITY AND LIABILITIES |
|
|
|
|
(1)Shareholders'
Funds |
|
|
|
|
(a) Share Capital |
270.850 |
270.530 |
270.270 |
|
(b) Reserves & Surplus |
24960.930 |
21586.550 |
19527.140 |
|
(c) Money
received against share warrants |
0.000 |
0.000 |
0.000 |
|
|
|
|
|
|
(2) Share Application money pending allotment |
0.000 |
0.000 |
0.000 |
|
Total
Shareholders’ Funds (1)+(2) |
25231.780 |
21857.080 |
19797.410 |
|
|
|
|
|
|
(3) Non-current liabilities |
|
|
|
|
(a) long-term borrowings |
0.000 |
2543.500 |
2745.700 |
|
(b) Deferred tax liabilities (Net) |
285.820 |
238.010 |
229.300 |
|
(c) Other long term liabilities |
849.450 |
778.420 |
31.230 |
|
(d) long-term provisions |
0.000 |
0.000 |
0.000 |
|
Total Non-current Liabilities (3) |
1135.270 |
3559.930 |
3006.230 |
|
|
|
|
|
|
(4) Current liabilities |
|
|
|
|
(a) Short term borrowings |
3088.490 |
2220.890 |
7973.830 |
|
(b) Trade payables |
4262.030 |
2704.990 |
1758.610 |
|
(c) Other current
liabilities |
4427.050 |
644.140 |
1007.990 |
|
(d) Short-term provisions |
710.760 |
703.580 |
198.000 |
|
Total Current Liabilities (4) |
12488.330 |
6273.600 |
10938.430 |
|
|
|
|
|
|
TOTAL |
38855.380 |
31690.610 |
33742.070 |
|
|
|
|
|
|
II. ASSETS |
|
|
|
|
(1) Non-current assets |
|
|
|
|
(a) Fixed Assets |
|
|
|
|
(i) Tangible assets |
2671.190 |
2162.680 |
2160.200 |
|
(ii) Intangible Assets |
85.390 |
74.900 |
77.590 |
|
(iii) Capital
work-in-progress |
1688.960 |
655.710 |
312.530 |
|
(iv)
Intangible assets under development |
35.240 |
33.700 |
23.400 |
|
(b) Non-current Investments |
12943.320 |
10832.690 |
10412.470 |
|
(c) Deferred tax assets (net) |
0.000 |
0.000 |
0.000 |
|
(d) Long-term Loan and Advances |
9228.010 |
9552.030 |
15401.460 |
|
(e) Other Non-current assets |
1809.160 |
1018.310 |
5430.610 |
|
Total Non-Current Assets |
28461.270 |
24330.020 |
33818.260 |
|
|
|
|
|
|
(2) Current assets |
|
|
|
|
(a) Current investments |
0.000 |
0.000 |
0.000 |
|
(b) Inventories |
1901.510 |
1759.270 |
1570.070 |
|
(c) Trade receivables |
4850.980 |
3587.430 |
1893.440 |
|
(d) Cash and cash
equivalents |
1677.860 |
475.140 |
309.490 |
|
(e) Short-term loans and
advances |
669.350 |
670.450 |
236.940 |
|
(f) Other current assets |
1294.410 |
868.300 |
813.870 |
|
Total Current Assets |
10394.110 |
7360.590 |
4823.810 |
|
|
|
|
|
|
TOTAL |
38855.380 |
31690.610 |
38642.070 |
PROFIT & LOSS ACCOUNT
|
|
PARTICULARS |
31.03.2013 |
31.03.2012 |
31.03.2011 |
|
|
|
SALES |
|
|
|
|
|
|
|
Revenue from Operations |
19493.040 |
15646.650 |
11629.390 |
|
|
|
Other Income |
1162.450 |
551.040 |
717.520 |
|
|
|
TOTAL (A) |
20655.490 |
16197.690 |
12346.910 |
|
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
|
|
Cost of materials consumed |
4157.500 |
3376.690 |
2404.910 |
|
|
|
Purchases of Stock-in-trade |
1410.880 |
1049.470 |
871.580 |
|
|
|
Changes in
inventories of finished goods, work-in-progress and Stock-in-trade |
(36.670) |
(128.630) |
139.890 |
|
|
|
Employee benefit expenses |
3030.170 |
2468.090 |
1741.570 |
|
|
|
Other expenses |
7606.720 |
5771.280 |
3613.650 |
|
|
|
TOTAL
(B) |
16168.600 |
12536.900 |
8771.600 |
|
|
|
|
|
|
|
|
Less |
PROFIT
BEFORE INTEREST, TAX, DEPRECIATION AND AMORTISATION (A-B) (C) |
4486.890 |
3660.790 |
3575.310 |
|
|
|
|
|
|
|
|
|
Less |
FINANCIAL
EXPENSES (D) |
436.940 |
608.690 |
857.500 |
|
|
|
|
|
|
|
|
|
|
PROFIT
BEFORE TAX, DEPRECIATION AND AMORTISATION (C-D) (E) |
4049.950 |
3052.100 |
2717.810 |
|
|
|
|
|
|
|
|
|
Less/ Add |
DEPRECIATION/
AMORTISATION (F) |
250.410 |
211.130 |
209.880 |
|
|
|
|
|
|
|
|
|
|
PROFIT BEFORE
TAX (E-F) (G) |
3799.540 |
2840.970 |
2507.930 |
|
|
|
|
|
|
|
|
|
Less |
TAX (H) |
(61.530) |
187.980 |
386.150 |
|
|
|
|
|
|
|
|
|
|
PROFIT AFTER TAX
(G-H) (I) |
3861.070 |
2652.990 |
2121.780 |
|
|
|
|
|
|
|
|
|
Add |
PREVIOUS
YEARS’ BALANCE BROUGHT FORWARD |
NA |
10294.140 |
8511.120 |
|
|
|
|
|
|
|
|
|
Less |
APPROPRIATIONS |
|
|
|
|
|
|
|
Proposed Dividend on Equity Shares |
NA |
NA |
108.110 |
|
|
|
Tax on Proposed Dividend on Equity Shares |
NA |
NA |
17.960 |
|
|
|
Residual Dividend and Dividend Tax |
NA |
NA |
0.500 |
|
|
|
Transfer to General Reserve |
NA |
NA |
212.190 |
|
|
BALANCE CARRIED
TO THE B/S |
NA |
NA |
10294.140 |
|
|
|
|
|
|
|
|
|
|
EARNINGS IN
FOREIGN CURRENCY |
|
|
|
|
|
|
|
Export of goods calculated on FOB basis |
6708.560 |
5405.840 |
3020.010 |
|
|
|
Guarantee Commission |
181.750 |
109.380 |
11.210 |
|
|
|
Interest on loan to subsidiaries |
115.360 |
144.800 |
308.040 |
|
|
|
Royalty Income |
1.130 |
1.850 |
0.000 |
|
|
TOTAL EARNINGS |
7006.800 |
5661.870 |
3339.260 |
|
|
|
|
|
|
|
|
|
|
IMPORTS |
|
|
|
|
|
|
|
Raw Materials |
290.090 |
244.680 |
158.840 |
|
|
|
Capital Goods |
567.820 |
102.580 |
166.850 |
|
|
TOTAL IMPORTS |
857.910 |
347.260 |
325.690 |
|
|
|
|
|
|
|
|
|
|
Earnings Per Share - Basic (Rs.) |
14.26 |
9.81 |
7.86 |
|
|
|
Earnings Per Share - Diluted (Rs.) |
14.25 |
9.80 |
7.85 |
|
KEY RATIOS
|
PARTICULARS |
|
31.03.2013 |
31.03.2012 |
31.03.2011 |
|
PAT / Total Income |
(%) |
18.69
|
16.38
|
17.18 |
|
|
|
|
|
|
|
Net Profit Margin (PBT/Sales) |
(%) |
19.49
|
18.16
|
21.57 |
|
|
|
|
|
|
|
Return on Total Assets (PBT/Total Assets} |
(%) |
15.71
|
14.09
|
8.99 |
|
|
|
|
|
|
|
Return on Investment (ROI) (PBT/Networth) |
|
0.15
|
0.13
|
0.13 |
|
|
|
|
|
|
|
Debt Equity Ratio (Total Debt/Networth) |
|
0.12
|
0.22
|
0.54 |
|
|
|
|
|
|
|
Current Ratio (Current Asset/Current Liability) |
|
0.83
|
1.17
|
1.29 |
FINANCIAL ANALYSIS
[All figures are
in Rupees Millions]
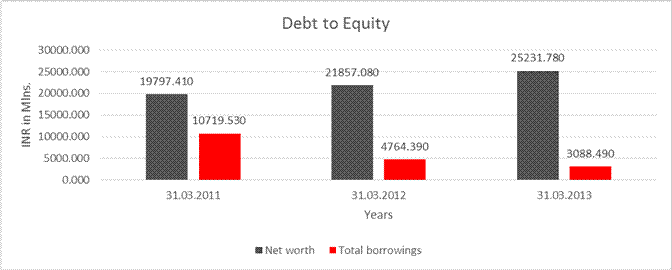
DEBT EQUITY RATIO
|
Particular |
31.03.2011 |
31.03.2012 |
31.03.2013 |
|
|
(INR in Mlns.) |
(INR in Mlns.) |
(INR in Mlns.) |
|
Share Capital |
270.270 |
270.530 |
270.850 |
|
Reserves & Surplus |
19527.140 |
21586.550 |
24960.930 |
|
Net worth |
19797.410 |
21857.080 |
25231.780 |
|
|
|
|
|
|
long-term borrowings |
2745.700 |
2543.500 |
0.000 |
|
Short term borrowings |
7973.830 |
2220.890 |
3088.490 |
|
Total borrowings |
10719.530 |
4764.390 |
3088.490 |
|
Debt/Equity ratio |
0.541 |
0.218 |
0.122 |
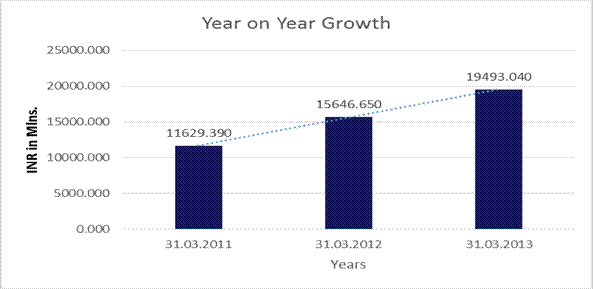
YEAR-ON-YEAR GROWTH
|
Year on Year Growth |
31.03.2011 |
31.03.2012 |
31.03.2013 |
|
|
(INR in Mlns) |
(INR in Mlns) |
(INR in Mlns) |
|
Revenue from Operations |
11629.390 |
15646.650 |
19493.040 |
|
|
|
34.544 |
24.583 |
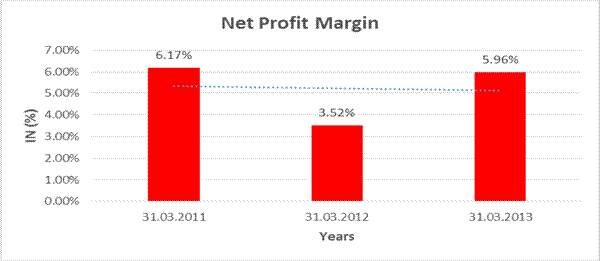
NET PROFIT MARGIN
|
Net Profit Margin |
31.03.2011 |
31.03.2012 |
31.03.2013 |
|
|
(INR in Mlns) |
(INR in Mlns) |
(INR in Mlns) |
|
Revenue from Operatoions |
11629.390 |
15646.650 |
19493.040 |
|
Profit |
717.520 |
551.040 |
1162.450 |
|
|
6.17% |
3.52% |
5.96% |
LOCAL AGENCY FURTHER INFORMATION
|
Sr. No. |
Check List by Info Agents |
Available in Report (Yes / No) |
|
1] |
Year of Establishment |
Yes |
|
2] |
Locality of the firm |
Yes |
|
3] |
Constitutions of the firm |
Yes |
|
4] |
Premises details |
No |
|
5] |
Type of Business |
Yes |
|
6] |
Line of Business |
Yes |
|
7] |
Promoter's background |
No |
|
8] |
No. of employees |
Yes |
|
9] |
Name of person contacted |
No |
|
10] |
Designation of contact person |
No |
|
11] |
Turnover of firm for last three years |
Yes |
|
12] |
Profitability for last three years |
Yes |
|
13] |
Reasons for variation <> 20% |
----- |
|
14] |
Estimation for coming financial year |
No |
|
15] |
Capital in the business |
Yes |
|
16] |
Details of sister concerns |
Yes |
|
17] |
Major suppliers |
No |
|
18] |
Major customers |
No |
|
19] |
Payments terms |
No |
|
20] |
Export / Import details (if applicable) |
No |
|
21] |
Market information |
----- |
|
22] |
Litigations that the firm / promoter
involved in |
Yes |
|
23] |
Banking Details |
Yes |
|
24] |
Banking facility details |
Yes |
|
25] |
Conduct of the banking account |
----- |
|
26] |
Buyer visit details |
----- |
|
27] |
Financials, if provided |
Yes |
|
28] |
Incorporation details, if applicable |
Yes |
|
29] |
Last accounts filed at ROC |
Yes |
|
30] |
Major Shareholders, if available |
Yes |
|
31] |
Date of Birth of
Proprietor/Partner/Director, if available |
Yes |
|
32] |
PAN of Proprietor/Partner/Director, if
available |
No |
|
33] |
Voter ID No of Proprietor/Partner/Director,
if available |
No |
|
34] |
External Agency Rating, if available |
Yes |
------------------------------------------------------------------------------------------------------------------------------
CASE DETAILS
BENCH:-BOMBAY
PRESENTATION
DATE: 08.04.2014
|
Lodging No.:- |
ITXAL/797/2014 |
Filing Date:- |
08.04.2014 |
|
Petitioner:- |
The commissioner of Income Tax - |
Respondent:- |
Glenmark Pharmaceuticals Limited |
|
Petn. Adv.:- |
Padma Divakar (I3287) |
|
District:- |
MUMBAI |
|
Bench:- |
Division |
|
Status:- |
Pre-Admission |
Category:- |
Tax Appeals |
|
Last Date:- |
15.04.2014 |
Stage:- |
-- |
|
Last Coram:- |
REGISTRAR (OS)/ PROTHONOTARY AND SR. MASTER |
|
Act :- |
Income Tax Act, 1961 |
Under
Section: |
260A |
------------------------------------------------------------------------------------------------------------------------------
UNSECURED LOANS
|
Particulars |
31.03.2013 |
31.03.2012 |
|
|
(Rs. In Millions) |
|
|
Long Term
Borrowings |
|
|
|
Other Loans |
|
|
|
- From Banks |
0.000 |
2543.500 |
|
|
|
|
|
Short Term
Borrowings |
|
|
|
Short-term loan from banks |
3088.490 |
1071.290 |
|
|
|
|
|
Total |
3088.490 |
3614.790 |
RESULTS OF OPERATIONS
On Standalone
basis the Company achieved a gross revenue of Rs.19,493.040 millions and the
Standalone operating profit before finance costs, depreciation and tax was
Rs.4,486.890 millions as compared to Rs.3,660.790 millions in the previous
year.
MANAGEMENT DISCUSSION
AND ANALYSIS
GLOBAL ENVIRONMENT
The global economy
continues to be challenging and global economic growth is projected to be lower
than the previous year Despite improved global financial conditions and reduced
short-term risks, the world economy continues to expand at a subdued pace.
After a marked downturn over the past two years, global economic activity is
expected to slowly gain momentum in the second half of 2013 and 2014 on the
back of accommodative monetary policies in developed and developing economies.
Short-term risks stemming from the euro area crisis, fiscal adjustment in the
United States and a further slowdown in large developing countries have
diminished, but not disappeared. Enhanced international policy coordination is
needed to mitigate negative policy spillovers and foster robust and balanced
growth. At the same time, new medium-term risks have emerged, including
possible adverse effects of unconventional monetary measures in developed
economies on global financial stability. These risks have the potential to once
again derail the feeble recovery of the world economy. International policy
coordination needs to be enhanced to mitigate negative policy spillovers,
promote cooperation in reforming the international financial system, and ensure
sufficient resource flows to developing economies, and in particular the least
developed countries.
GLOBAL PHARMA
SCENARIO
The Global
Pharma scenario remains dynamic and challenging. They are witnessing various new
developments that make one believe that the ensuing years for pharma companies
will be challenging. They would also witness significant changes in strategies
by Pharmaceutical companies to take into consideration the dynamic environment
that is surrounding the industry. An important trend that is being witnessed is
the regulatory environment in developed and developing countries. While
developed countries are constantly raising the bar, the developing countries
are rapidly changing guidelines to bring them on par with the developed
countries regulatory framework. This evolution has resulted in confusion in
many markets as Pharma companies operating in these markets are constantly
trying to assess the standards for getting their products approved. The increased
scrutiny from regulators will continue to enforce renewed commitment to quality
from the industry. Some of the other trends that are being witnessed are the
continuous shift of share of healthcare spends from treatment of disease to
prevention and diagnosis. Further, the disease burden shift towards chronic
diseases is rapidly happening. The patients are becoming increasingly empowered
and going ahead, will be responsible for an increased portion of healthcare
costs due to ever increasing pressure on governments. The value of patent
expiries will increase, but the composition of value will shift from small
molecule to biologics.
BUSINESS REVIEW
SPECIALTY BUSINESS
The
specialty formulations business is organized around four regions India, Latin
America, Central Eastern Europe and Markets of Africa/Asia/CIS.
During the year under review, the specialty formulations business performed well, with the business growing by 27.52% to 26514.43 (USD 486.06 Mn) as compared to X 20792.42 (USD 428.44 Mn) for the previous corresponding financial year (excluding out-licensing income). The specialty business is now 53.88% of the overall base business as compared to 58.02% for the previous financial year.
INDIA
The India
formulations business outperformed during the year registering revenue of X
13095.79 Mn (USD 240.07 Mn) as compared to 10021.30 Mn (USD 206.50 Mn) in the
previous corresponding year, recording growth of 30.68 % in X term.
As per
IMS-MAT Mar 2013, Glenmark gained 3 ranks from 23rd to 20th as compared to MAT
Mar 2012 exhibiting value growth of 18.0% vis-a-vis industry growth of 10.1%.
Market share for the India business moved up from 1.69% to 1.82% during the
same period and the India business was the 2nd fastest growing company among
top 20 players in the industry. During the last two financial years the India
business gained five ranks in the Indian market place. The growth is once again
driven by strong performance of leading brands resulting in market share
improvement across core therapeutic areas. To further strengthen its presence
in India, the company has entered into two new businesses i.e. the OTC business
and pure generics business.
PROGRESS
IN OPERATING THERAPEUTIC AREAS:
In Cardiology,
market share increased from 2.86% to 3.30%, Respiratory market share increased
from 2.84% to 3.33%, Anti-infective MS increased from 1.35% to 1.60% and
Gynaecology MS increased from 1.26% to 1.38% and in Dermatalogy market share
was maintained at 8.69%.
Market
share gains also led to two (2) ranks gain in the Cardiac and Respiratory
segments attaining 13th and 7th position in respective segments and one (1)
rank in Gynaecology segment with current rank at 22nd. In Dermatalogy, we
maintained 2nd rank and increased the value gap with respect to 3rd position.
ACTIVE
PHARMACEUTICAL INGREDIENTS (API)
The API
business continued with its strategy of focusing on differentiated API products
and also changing the business mix towards the regulated markets. During the
year under review, the API business also recorded its first sales in Japan.
Revenue
from sale of APIs globally wasRs.3976.410 Millions [USD 72.90 Mn] in FY13
against Rs.3094.440 Millions [USD 63.76 Mn] in FY12, recording an increase of
28.5% in Rs. term.
During the
year, Ankleshwar API facility received approval from the Regulatory Agency of
Japan (PMDA). The API business made inroads into Japan with 3 new product
filings. Glenmark s API facility at Ankleshwar receives European cGMP certification
post a joint inspection of USFDA and EMEA. Another seven new USDMFs were filed
including several first DMFs targeting FTF and three DMFs to support Europe
Market. The business continued its leadership position for Amiodarone,
Lercanidipine, Adapalene, Perindopril, combined with launches of new products
during the year viz, Atovaquone in Canada through partner and Levocetirizine in
Europe through partner.
The
development of a new state-of-the-art manufacturing facility of Glenmark is in
progress at Dahej, Gujarat. This facility will cater to the manufacturing of
intermediates and Active Pharmaceutical Ingredients for regulated markets and
is expected to be commissioned in FY 2014.
OUTLOOK
Glenmark's short-term and
long-term outlook is encouraging for several reasons. On the discovery front,
the pipeline is progressing well with 4 molecules in clinics.
The company will also
continue with its approach of out-licensing its molecules. On the generics
front, with high value patented drugs going off patent in the coming years,
there is huge potential for the generics business. Glenmark is actively
increasing its base in major generics markets of US and Western Europe.
At same time, the specialty
business will continue to build differentiated pipelines in rest of the world
markets, notably the 'Pharmerging' markets. Focus will be on building size and
scale organically. The Company has also put multiple systems and processes in place
to manage its complex operations and instill efficiencies across the value
chain. Glenmark will also continue to build capabilities and nurture a talent
pool with diverse skills sets to deliver continuous results.
CONTINGENT LIABILITIES:
|
Particulars |
31.03.2013 |
31.03.2012 |
|
|
Rs. in millions |
|
|
(a) Claims
against the Company not acknowledge as debts |
|
|
|
Labour Dispute |
0.060 |
0.090 |
|
Disputed Taxes and Duties |
105.780 |
154.470 |
|
(b) Guarantees |
|
|
|
Bank guarantees |
41.390 |
19.630 |
|
Letter of comfort on behalf of subsidiaries, to the extent of limits |
24286.730 |
15925.540 |
|
(c) Others |
|
|
|
Open letters of credit |
18.640 |
460.380 |
|
Indemnity Bond |
374.570 |
287.730 |
INDEX OF CHARGES
|
S.No. |
Charge ID |
Date of Charge
Creation/Modification |
Charge amount
secured |
Charge Holder |
Address |
Service Request
Number (SRN) |
|
1 |
80033325 |
23/09/2004 |
100,000,000.00 |
State Bank of
India |
ATLANTA BUILDING,
, NARIMAN POINT, MUMBAI, MAHARASHTRA - 400001, INDIA |
- |
|
2 |
80020353 |
26/09/2012 * |
4,900,000,000.00 |
Bank of India |
8-A, BHULABHAI
DESAI ROAD, MAHALAXMI, MUMBAI, MAH ARASHTRA - 400026, INDIA |
B60388204 |
* Date of charge modification
FIXED ASSETS
Tangible Assets
·
·
·
· Other buildings and premises
· Plant and Machinery
· Furniture and Fixtures
· Office Equipments
· Vehicles
Intangible Assets
·
Computer Software
·
Brands
PRESS RELEASE
GLENMARK ANNOUNCES
LAUNCH OF HYDROCORTISONE BUTYRATE CREAM IN THE US
Glenmark is entitled to 180 days of exclusivity as it is the
first generic company to file an ANDA for the product.
December 9, 2013: Glenmark Generics Inc., USA, the subsidiary of Glenmark
Generics Limited, announces the exclusive launch of Hydrocortisone Butyrate
cream USP, 0.1% in the United States. The Company received approval from the
United States Food and Drug Administration (U.S. FDA) for Abbreviated New Drug
Application (ANDA) for its generic version of Locoid Lipocream® on September
27, 2013.
Glenmark is entitled to 180 days of exclusivity with respect to its Hydrocortisone Butyrate cream, as it is the first generic company to file an ANDA for the product.
Hydrocortisone Butyrate cream is indicated for the relief of the inflammatory and pruritic manifestations of corticosteroid-responsive dermatoses in adults and the treatment of mild to moderate atopic dermatitis in patients 3 months to 18 years of age. According to IMS Health sales data for the 12 month period ending September 2013, Hydrocortisone Butyrate cream garnered annual sales of approximately USD 36.8 million.
Glenmark’s current portfolio consists of 90 products
authorized for distribution in the U.S. marketplace and 56 ANDA’s pending
approval with the U.S. FDA. In addition to these internal filings, GGI
continues to identify and explore external development partnerships to
supplement and accelerate the growth of the existing pipeline and portfolio.
About Glenmark
Generics Limited
Glenmark Generics Limited (GGL) is a subsidiary of Glenmark Pharmaceuticals Limited (Glenmark) and aims to be a global integrated Generic and API leader. GGL has an established presence in North America and developing an EU presence. It primarily sells its FDF products in the United States ("US") and the European Union ("EU"), as well as its oncology FDF products in South America. The Company supplies APIs to customers in approximately 80 countries, including the US, various countries in the EU, South America and India.
GLENMARK CONFIRMS PATENT CHALLENGE FOR BENDAMUSTINE HYDROCHLORIDE
This ANDA has been
filed from Glenmark's Argentina lyophilized injectable facility
December 31, 2013: Glenmark Pharmaceuticals Limited and Glenmark Generics Inc., USA ("Glenmark") confirms Cephalon Inc. has filed a patent infringement suit on 26 Dec 2013 in the U.S. District Court for the District of Delaware seeking to prevent Glenmark from commercializing its Abbreviated New Drug Application (ANDA) Bendamustine Hydrochloride product, their generic version of Treandae, prior to expiration of the Orange
Book patents. A complaint against Glenmark has been filed on US patent 8,445,524. This lawsuit was filed under the provisions af the Hatch-Waxman Act.
Bendamustine is indicated for the treatment of patients with Chronic Lymphocytic Leukemia. This ANDA has been filed from Glenmark's Argentina lyophilized injectable facility. For the twelve-month period ending September 2013, Treanda" achieved sales of USD 659 million according to IMS Health.
About Glenmark
Generics Limited
Glenmark Generics Limited (GGL) is a subsidiary of Glenmark Pharmaceuticals Limited (Glenmark) and aims to be a global integrated Generic and API leader. GGL has an established presence in North America and developing an EU presence, which primarily sells its FDF products in the United States ("US") and the European Union (L'EU"), as well as its oncology FDF products in South America. The Company supplies APIs to custorhers in approximately 80 countries, including the US, various countries in the EU, South America and.India.
CMT REPORT (Corruption, Money Laundering & Terrorism]
The Public Notice information has been collected from various sources
including but not limited to: The Courts,
1] INFORMATION ON
DESIGNATED PARTY
No exist designating subject or any of its beneficial owners,
controlling shareholders or senior officers as terrorist or terrorist
organization or whom notice had been received that all financial transactions
involving their assets have been blocked or convicted, found guilty or against
whom a judgement or order had been entered in a proceedings for violating money-laundering,
anti-corruption or bribery or international economic or anti-terrorism sanction
laws or whose assets were seized, blocked, frozen or ordered forfeited for
violation of money laundering or international anti-terrorism laws.
2] Court Declaration :
No records exist to suggest that subject is
or was the subject of any formal or informal allegations, prosecutions or other
official proceeding for making any prohibited payments or other improper
payments to government officials for engaging in prohibited transactions or
with designated parties.
3] Asset Declaration :
No records exist to suggest that the property or assets of the subject
are derived from criminal conduct or a prohibited transaction.
4] Record on Financial
Crime :
Charges or conviction
registered against subject: None
5] Records on Violation of
Anti-Corruption Laws :
Charges or
investigation registered against subject: None
6] Records on Int’l
Anti-Money Laundering Laws/Standards :
Charges or investigation
registered against subject: None
7] Criminal Records
No
available information exist that suggest that subject or any of its principals
have been formally charged or convicted by a competent governmental authority
for any financial crime or under any formal investigation by a competent
government authority for any violation of anti-corruption laws or international
anti-money laundering laws or standard.
8] Affiliation with
Government :
No record
exists to suggest that any director or indirect owners, controlling
shareholders, director, officer or employee of the company is a government
official or a family member or close business associate of a Government
official.
9] Compensation Package :
Our market
survey revealed that the amount of compensation sought by the subject is fair
and reasonable and comparable to compensation paid to others for similar
services.
10] Press Report :
No press reports / filings exists on
the subject.
CORPORATE GOVERNANCE
MIRA INFORM as part of its Due Diligence do provide comments on
Corporate Governance to identify management and governance. These factors often
have been predictive and in some cases have created vulnerabilities to credit
deterioration.
Our Governance Assessment focuses principally on the interactions
between a company’s management, its Board of Directors, Shareholders and other
financial stakeholders.
CONTRAVENTION
Subject is not known to have contravened any existing local laws,
regulations or policies that prohibit, restrict or otherwise affect the terms
and conditions that could be included in the agreement with the subject.
FOREIGN EXCHANGE RATES
|
Currency |
Unit
|
Indian Rupees |
|
US Dollar |
1 |
Rs.60.38 |
|
|
1 |
Rs.101.63 |
|
Euro |
1 |
Rs.83.57 |
INFORMATION DETAILS
|
Analysis Done by
: |
KAR |
|
|
|
|
Report Prepared
by : |
NIT |
SCORE & RATING EXPLANATIONS
|
SCORE FACTORS |
RANGE |
POINTS |
|
HISTORY |
1~10 |
8 |
|
PAID-UP CAPITAL |
1~10 |
7 |
|
OPERATING SCALE |
1~10 |
8 |
|
FINANCIAL CONDITION |
|
|
|
--BUSINESS SCALE |
1~10 |
8 |
|
--PROFITABILIRY |
1~10 |
8 |
|
--LIQUIDITY |
1~10 |
7 |
|
--LEVERAGE |
1~10 |
7 |
|
--RESERVES |
1~10 |
8 |
|
--CREDIT LINES |
1~10 |
7 |
|
--MARGINS |
-5~5 |
-- |
|
DEMERIT POINTS |
|
|
|
--BANK CHARGES |
YES/NO |
YES |
|
--LITIGATION |
YES/NO |
YES |
|
--OTHER ADVERSE INFORMATION |
YES/NO |
NO |
|
MERIT POINTS |
|
|
|
--SOLE DISTRIBUTORSHIP |
YES/NO |
NO |
|
--EXPORT ACTIVITIES |
YES/NO |
NO |
|
--AFFILIATION |
YES/NO |
YES |
|
--LISTED |
YES/NO |
YES |
|
--OTHER MERIT FACTORS |
YES/NO |
YES |
|
DEFAULTER |
|
|
|
--RBI |
YES/NO |
NO |
|
--EPF |
YES/NO |
NO |
|
TOTAL |
|
68 |
This score serves as a reference to assess SC’s credit risk
and to set the amount of credit to be extended. It is calculated from a
composite of weighted scores obtained from each of the major sections of this
report. The assessed factors and their relative weights (as indicated through
%) are as follows:
Financial
condition (40%) Ownership
background (20%) Payment
record (10%)
Credit history
(10%) Market trend
(10%) Operational
size (10%)
RATING EXPLANATIONS
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
>86 |
Aaa |
Possesses an extremely sound financial base with the strongest
capability for timely payment of interest and principal sums |
Unlimited |
|
71-85 |
Aa |
Possesses adequate working capital. No caution needed for credit
transaction. It has above average (strong) capability for payment of interest
and principal sums |
Large |
|
56-70 |
A |
Financial & operational base are regarded healthy. General
unfavourable factors will not cause fatal effect. Satisfactory capability for
payment of interest and principal sums |
Fairly Large |
|
41-55 |
Ba |
Overall operation is considered normal. Capable to meet normal
commitments. |
Satisfactory |
|
26-40 |
B |
Capability to overcome financial difficulties seems comparatively below
average. |
Small |
|
11-25 |
Ca |
Adverse factors are apparent. Repayment of interest and principal sums
in default or expected to be in default upon maturity |
Limited with
full security |
|
<10 |
C |
Absolute credit risk exists. Caution needed to be exercised |
Credit not
recommended |
|
-- |
NB |
New Business |
-- |
This report is issued at your request without any
risk and responsibility on the part of MIRA INFORM PRIVATE LIMITED (MIPL) or
its officials.