MIRA
INFORM REPORT
|
Report Date : |
20.08.2014 |
IDENTIFICATION DETAILS
|
Name : |
SUN PHARMACEUTICAL INDUSTRIES LIMITED |
|
|
|
|
Registered
Office : |
Sun Pharma Advance
Research Centre (SPARC), Tandalja, |
|
|
|
|
Country : |
|
|
|
|
|
Financials (as
on) : |
31.03.2014 |
|
|
|
|
Date of
Incorporation : |
01.03.1993 |
|
|
|
|
Com. Reg. No.: |
04-019050 |
|
|
|
|
Capital
Investment / Paid-up Capital : |
Rs.2071.200 Millions |
|
|
|
|
CIN No.: [Company Identification
No.] |
L24230GJ1993PLC019050 |
|
|
|
|
TAN No.: [Tax Deduction &
Collection Account No.] |
BRDS02426E |
|
|
|
|
PAN No.: [Permanent Account No.] |
AADCS3124K |
|
|
|
|
Legal Form : |
A Public Limited Liability Company. The Company’s Shares are Listed on
the Stock Exchanges. |
|
|
|
|
Line of Business
: |
Manufacturer of
Tablets, Capsules, Parenterals, Ointments, Bulk Drugs, Chemicals and Liquids. |
|
|
|
|
No. of Employees
: |
14000
(Approximately) |
RATING & COMMENTS
|
MIRA’s Rating : |
Aa (75) |
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
71-85 |
Aa |
Possesses adequate working capital. No caution needed for credit transaction.
It has above average (strong) capability for payment of interest and
principal sums |
Large |
|
Maximum Credit Limit : |
USD 323580000 |
|
|
|
|
Status : |
Good |
|
|
|
|
Payment Behaviour : |
Regular |
|
|
|
|
Litigation : |
Exist |
|
|
|
|
Comments : |
Subject is a well-established and reputed company having fine track
record. Financial position of the company is strong. Over all fundamentals of
the company is sound and healthy. Directors are reported to be experienced and respectable businessmen. Trade relations are reported a fair. Business is active. Payments are
reported to be regular and as per commitment. The company can be considered good for normal business dealings at
usual trade terms and conditions. |
NOTES :
Any query related to this report can be made
on e-mail : infodept@mirainform.com
while quoting report number, name and date.
ECGC Country Risk Classification List – June 1, 2014
|
Country Name |
Previous Rating (31.03.2014) |
Current Rating (01.06.2014) |
|
India |
A1 |
A1 |
|
Risk Category |
ECGC
Classification |
|
Insignificant |
A1 |
|
Low |
A2 |
|
Moderate |
B1 |
|
High |
B2 |
|
Very High |
C1 |
|
Restricted |
C2 |
|
Off-credit |
D |
INDIAN ECONOMIC OVERVIEW
N E W S
As per the latest IMF study, the total weigh of emerging markets in the
GDP of the world on a purchasing power parity basis has seen a sizeable shift.
It highlights how as against 51 % in 2005, the emerging economies now account
for close to 56 % of the global purchasing power GDP as per the latest survey.
And with the emerging economies growing at a faster rate than their developed
counterparts, there are every possibility that the their share goes up further
in the coming years. China may surpass the US over the next few years.
Politics and economics are very intricately connected. They tend to
influence each other in ways that could be very complex and far-reaching. The
prospects of the India’s economy have been seriously compromised due to
political corruption. High inflation, poor standard of living are to a great
extent a result of rampant corruption in the country. China on the other hand,
seems to be facing diametrically opposite challenge. American hedge fund manager
Jim Chanos has been keenly following the political and economic development in
the dragon economy and has figured out something that is quite worrying. He is
of the view that the Chinese economy could be heading toward trouble on account
of new Chinese President Xi Jingping’s very aggressive anti-corruption drive.
Chanos believes tat many things such as apartment sales, luxury products, etc.
were largely bought with dirty money. And it is now beginning to impact
consumption. This may indeed be bad news for an economy that is struggling to
transition from an investment-driven export-oriented economy to a domestic
consumption-driven economy.
A study published by Firstpost has revealed that asset classes like real
estate and equities were the biggest beneficiaries of the liberalization
policies. A firm called Ciane Analytics studied returns from assets
including equities, gold, fixed deposits, G-Secs and real estate since 1991.
Real estate outperformed every other asset classes during the 23-year period
with an annualized return of 20 % ! Equities came in second with annualized
return of 15.5 % ! However, while these returns may seem mouthwatering, the
fact is that the return from equities adjusted for inflation came down to just
7.1 %.
Some brief news are as under
. R-Power to buy Jaypee’s hydro assets
. Investors await justice in NSEL case
. India seeks MFN status from Pakistan ahead of meeting
. Ukrain’s clashes with rebels hinder MH17 crash investigation
. India exploring merger of state-owned hydro PSUs
..Higher costs weigh down profit growth to slowest in 9 quarters
..Wal-Mart to expand wholesale business in India
. GMR group moves to strengthen balance sheet
. Central Bank to sell 4 % stake to Life Insurance Corporation
. Tata Chemicals plans to raise up to Rs 10000 mn.
EXTERNAL AGENCY RATING
|
Rating Agency Name |
CRISIL |
|
Rating |
AAA = Long Term Rating |
|
Rating Explanation |
Highest degree of safety and carry lowest
risk |
|
Date |
April 8, 2014 |
|
Rating Agency Name |
CRISIL |
|
Rating |
A1+ = Short Term Rating |
|
Rating Explanation |
Strong degree of safety and carry lowest
credit risk. |
|
Date |
April 8, 2014 |
RBI DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter in
the publicly available RBI Defaulters’ list.
EPF (Employee Provident Fund) DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available EPF (Employee Provident Fund) Defaulters’ list as of
31-03-2014.
INFORMATION DENIED
Management Non Co-Operative (91-265-234001)
LOCATIONS
|
Registered Office/ Research Centre 1 : |
Sun Pharma
Advance Research Centre (SPARC), Tandalja,
Akota Road, Vadodara – 390020, Gujarat, India |
|
Tel. No.: |
91-265-2340001/
5515500/ 600/700 |
|
Fax No.: |
91-265-2339103/
2354897/ 2332664 |
|
E-Mail : |
|
|
Website : |
|
|
|
|
|
Corporate
Office : |
|
|
Tel. No.: |
91-22-28211822/1842/1917/1951/1953 |
|
Fax No.: |
91-22-28212010 |
|
E-Mail : |
|
|
|
|
|
Research
Centre 2 : |
· F.P 27, Part Survey No. 27, C. S. No. 1050, T. RS. Village, Tandalja, District Vadodara - 390020, Gujarat, India 17-B, Mahal Industrial Estate, Mahakali Caves Road,
Andheri (East), Mumbai - 400059, Maharashtra, India Tel No.:
91-2-66455645 Fax No.:
91-22-66455685 Chemistry and Discovery
Reaserch Israel, 14 Hakitor Street, P.O. Box, 10347 Haifa Boy 26110, Israel |
|
|
|
|
|
|
|
Factory 1 : |
Plot No. 214, Plot No. 20, Government Industrial Area, Phase II, Piparia, Silvassa – 396230, Gujarat, India |
|
|
|
|
Factory 2 : |
Halol-Baroda Highway, Near Anand Kendra, Halol, District Panchmahal - 389350, Gujarat, India |
|
|
|
|
Factory 3 : |
Plot No. 24/2 and 25, GIDC, Phase-IV, Panoli, District Bharuch - 394116, Gujarat, India |
|
|
|
|
Factory 4 : |
A-7 and A-8, MIDC Industrial Area, Ahmednagar - 414111, Maharashtra, India |
|
|
|
|
Factory 5 : |
Plot No. 4708, GIDC. Ankleshwar - 393002, Gujarat, India |
|
|
|
|
Factory 6 : |
Sathammai Village, Karunkuzhi Post, Maduranthakam TK, District Kanchipuram - 603303, Tamilnadu, India |
|
|
|
|
Factory 7 : |
Plot No. 817/A, Karkhadi, Taluka Padra, District Vadodara - 391450, Gujarat, India |
|
|
|
|
Factory 8 : |
Sun Pharma Drugs
Private Limited Plot No. 754, Nandok Block, Setipool, P.O. Ranipool – 737135, Sikkim, India |
|
|
|
|
Factory 9 : |
Sun Pharma Medication
Private Limited Survey No. 259/15, Dadra – 396191, Union Territory |
|
|
|
|
Factory 10 : |
Sun Pharma
Medication Private Limited 6-9 Export Promotion Industrial Park (EPIP), Kartholi, Bari Brahmana, Jammu - 181133, Jammu and Kashmir, India |
|
|
|
|
Factory 11 : |
Sun Pharma
Medication Private Limited I.G.C. Phase-I, Samba – 184121, Jammu and Kashmir, India |
|
|
|
|
Factory 12 : |
Sun Pharmaceutical
Industries Inc. 705, E. Mulberry Street, Bryan, Ohio – 43506, USA |
|
|
|
|
Factory 13 : |
Sun Pharmaceutical
Industries Inc. 270 Prospect Plains Road, Cranbury, New Jersey – 08512, USA |
|
|
|
|
Factory 14 : |
Caraco
Pharmaceutical Laboratories Limited 1150 Elijah McCoy Drive, Detroit – 48202, Michigan, USA |
|
|
|
|
Factory 15 : |
Sun Pharmaceutical
(Bangladesh) Limited Chandana, Joydevpur, Gazipur, Bangladesh |
|
|
|
|
Factory 16 : |
Alkaloida Chemical
Company Zrt H-4440 Tiszavasvari , Kabay, Janos u.29, Hungary |
|
|
|
|
Factory 17 : |
TKS Farmaceutica Rodovia GO-080, Km 02, Chacaras 01/02, Jardim Pompeia, Goiania/GO, Brazil CEP: 74690-170 |
|
|
|
|
Factory 18 : |
Sun Pharma de
Mexico S.A. de C.V, Av. Rio Churubusco No. 658, Col. El Sifon, Del. Iztapalapa, C.P 09400 Mexico, Distrito Federal |
|
|
|
|
Factory 19 : |
Chattem Chemicals,
Inc. 3708, St. Elmo Avenue, Chattanooga, TN 37409, USA |
|
|
|
|
Factory 20 : |
Taro
Pharmaceuticals Inc. 130 East Drive, Brampton, Ontario L6T 1C1, Canada |
|
|
|
|
Factory 21 : |
Taro Pharmaceutical
Industries Limited 14 Hakitor Street, P.O. Box 10347 Haifa Bay 26110, Israel |
|
|
|
|
Factory 22 : |
Dusa
Pharmaceuticals Inc., 25, Upton Drive, Wilmington, Massachusetts, 01887, USA |
|
|
|
|
Factory 23 : |
URL Pharmaceuticals Inc., 1100 Orthodox Street, Philadelphia, PA 19124,
USA |
|
|
|
|
Factory 24 : |
URL Pharmaceuticals Inc., 2500 Molitor Road, Aurora, IL 60502, USA |
DIRECTORS
As on 31.03.2013
|
Name : |
Mr. Israel Makov |
|
Designation : |
Chairman |
|
|
|
|
Name : |
Mr. Dilip S. Shanghvi |
|
Designation : |
Managing Director |
|
Qualification : |
B. Com. |
|
Date of Appointment
: |
01.04.1993 |
|
|
|
|
Name : |
Mr. Sudhir V. Valia |
|
Designation : |
Whole Time Director |
|
Qualification : |
FCA |
|
Date of Appointment
: |
01.04.1994 |
|
|
|
|
Name : |
Mr. Sailesh T. Desai |
|
Designation : |
Whole Time Director |
|
|
|
|
Name : |
Mr. S. Mohanchand Dadha |
|
Designation : |
Director |
|
|
|
|
Name : |
Mr. Hasmukh S. Shah |
|
Designation : |
Director |
|
|
|
|
Name : |
Mr. Ashwin Dani |
|
Designation : |
Director |
|
|
|
|
Name : |
Mr. Keki M. Mistry |
|
Designation : |
Additional Director |
KEY EXECUTIVES
|
Name : |
Mr. Sunil R. Ajmera |
|
Designation : |
Company Secretary |
|
|
|
|
Name : |
Ms. Sheetal |
|
Designation : |
Accounts Department |
MAJOR SHAREHOLDERS
As on 30.06.2014
|
Category of
Shareholder |
Total No. of
Shares |
% of Total No.
of Shares |
|
(A) Shareholding of Promoter and Promoter Group |
|
|
|
(1) Indian |
|
|
|
|
304042200 |
14.68 |
|
|
1013024000 |
48.91 |
|
|
1280200 |
0.06 |
|
|
1280200 |
0.06 |
|
|
1318346400 |
63.65 |
|
|
|
|
|
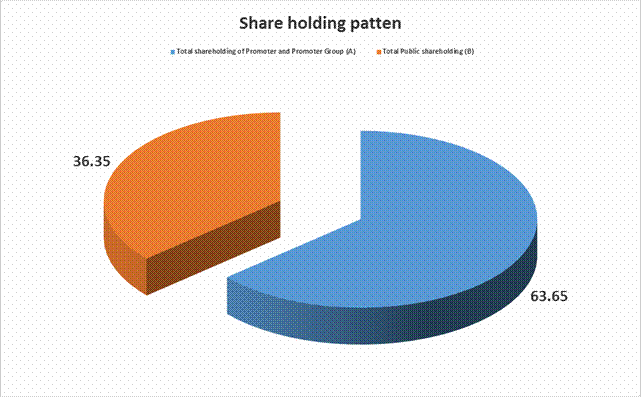
Total shareholding
of Promoter and Promoter Group (A) |
1318346400 |
63.65 |
|
(B) Public
Shareholding |
|
|
|
|
|
|
|
|
21660904 |
1.05 |
|
|
40953459 |
1.98 |
|
|
1312013 |
0.06 |
|
|
41805977 |
2.02 |
|
|
475295046 |
22.95 |
|
|
30000 |
0.00 |
|
|
581057399 |
28.05 |
|
|
|
|
|
|
52911529 |
2.55 |
|
|
|
|
|
|
67266031 |
3.25 |
|
|
41365740 |
2.00 |
|
|
10216811 |
0.49 |
|
|
2209623 |
0.11 |
|
|
2258104 |
0.11 |
|
|
1836434 |
0.09 |
|
|
3866650 |
0.19 |
|
|
46000 |
0.00 |
|
|
171760111 |
8.29 |
|
Total Public
shareholding (B) |
752817510 |
36.35 |
|
Total (A)+(B) |
2071163910 |
100.00 |
|
(C) Shares held by Custodians and against which Depository Receipts have been issued |
|
|
|
|
0 |
0.00 |
|
|
0 |
0.00 |
|
|
0 |
0.00 |
|
Total (A)+(B)+(C) |
2071163910 |
0.00 |
BUSINESS DETAILS
|
Line of Business : |
Manufacturer of
Tablets, Capsules, Parenterals, Ointments, Bulk Drugs, Chemicals and Liquids. |
||||||||||
|
|
|
||||||||||
|
Products : |
|
GENERAL INFORMATION
|
No. of Employees : |
14000 (Approximately) |
||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||
|
Bankers : |
· Bank of Baroda Bank of Nova Scotia Citibank N.A. ICICI Bank Limited Kotak Mahindra Bank Limited Standard Chartered Bank State Bank of India |
||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||
|
Facilities : |
Note : Long Term
Borrowing: Repayable in 10 (Previous
Year Nil) half-yearly installments of Rs. 4.600 Million each commencing from
31st March, 2017, Last installment is due on 30th September, 2021. |
|
Banking
Relations : |
-- |
|
|
|
|
Auditors : |
|
|
Name : |
Deloitte Haskins
and Sells Chartered
Accountants, |
|
Address : |
Mumbai, |
|
|
|
|
Subsidiaries : |
·
Alkaloida Chemical Company Zrt ·
Caraco
Pharmaceutical Laboratories Limited ·
Chattem Chemical
Inc ·
Green Eco
Development Centre Limited ·
OOO "Sun
Pharmaceutical Industries Limited ·
TKS Farmaceutica
Ltda ·
Sun Pharma De
Mexico S.A. DE C.V. ·
Sun Pharma De
Venezuela, CA ·
Sun Pharma Global
Inc ·
Sun Pharmaceutical
(Bangladesh) Limited ·
Sun Pharmaceutical
Industries (Europe) B.V. ·
Sun Pharmaceutical
Industries Inc (merged with Caraco Pharmaceutical Laboratories Limited w.e.f.
28th February, 2013) ·
Sun Pharmaceutical
Spain, S.L. ·
Sun Pharmaceuticals
France ·
Sun Pharmaceuticals
Germany GmbH ·
Sun Pharma Global
(FZE) ·
Sun Pharmaceuticals
Italia S.R.L. ·
Sun Pharmaceuticals
UK Limited ·
Taro Pharmaceutical
Industries Limited ·
Sun Pharmaceutical
Industries (Australia) Pty. Limited ·
Aditya Acquisition
Company Limited ·
Sun Pharmaceuticals
(SA) (Pty) Limited ·
Sun Global Canada
Pty Limited ·
Sun Pharmaceutical
Peru S.A.C. ·
Taro Development
Corporation ·
ZAO Sun Pharma Industriesx. ·
SPIL De Mexico S.A. DE C.V. ·
Caraco Pharma Inc ·
3 Skyline LLC ·
One Commerce Drive LLC ·
Taro Hungary Intellectual Property Licensing LLC. ·
Taro International Limited ·
Taro Pharmaceuticals Inc. ·
Taro Pharmaceutical India Private Limited ·
Taro Pharmaceutical Laboratories INC ·
Taro Pharmaceuticals U.S.A., Inc. ·
Taro Pharmaceuticals Europe B.V. ·
Taro Pharmaceuticals Ireland Limited ·
Taro Pharmaceuticals North America, Inc. ·
Taro Pharmaceuticals UK Limited ·
Taro Research Institute Limited (merged with Taro
Pharmaceutical Industries Limited w.e.f. 31st March , 2012) ·
Tarochem Limited ·
Morley and Company Inc ·
Sun Laboratories FZE ·
Taro Pharmaceuticals Canada, Limited ·
Sun Global Development FZE ·
Sun Pharmaceuticals Korea Limited ·
Sun Pharma Philippines Inc. ·
Caraco Pharmaceuticals Private Limited ·
Sun Pharma MEA JLT ·
Sun Pharma Healthcare FZE ·
Sun Pharma Japan Limited ·
Sun Pharma Laboratories Limited(Formerly, Sun
Resin and Polymers Private Limited) ·
Sun Pharma Medication Private Limited ·
Sun Pharma Drugs Private Limited ·
Universal Enterprise Private Limited (w.e.f. 31st
August, 2012) ·
** Faststone Mercantile Company Private Limited ·
** Neetnav Real Estate Private Limited ·
** Realstone Multitrade Private Limited ·
** Skisen Labs Private Limited ·
** Softdeal Trading Company Private Limited ·
** Orta Limited ·
** Sun Universal Limited ·
** Khyati Realty ME Limited ·
** Aditya Pharma limited Liability Company ·
** Alkaloida Sweden AB ·
** Dusa Pharmaceuticals Inc ·
** Dusa Pharmaceuticals New York Inc ·
** Sirius Laboratories Inc ·
** URL Pharma Inc ·
** AR Scientific.Inc ·
** Mutual Pharmaceutical Company, Inc. ·
** United Research Laboratories, Limited ·
** Dungan Mutual Associates, LLC ·
** URL PharmPro, LLC |
|
|
|
|
Controlled
Entity : |
·
Sun Pharmaceutical Industries ·
Sun Pharma Sikkim ·
Sun Pharma Drugs ·
Universal Enterprise Private Limited (upto 31st
August, 2012) |
|
|
·
|
|
Enterprise under
significant Influence of Key Management Personnel or their relative : |
·
Sun Petrochemicals Private Limited ·
Navjivan Rasayan (Gujarat) Private Limited ·
Sun Pharma Advanced Research Company Limited |
* Controlled entities converted into private limited companies under Part
IX of the Companies Act, 1956 w.e.f.31st August, 2012.
** Incorporated / Acquired during the year
CAPITAL STRUCTURE
As on: 31.03.2014
Authorised Capital : Not Available
Issued, Subscribed & Paid-up Capital : Rs.2071.200 Millions
As on 31.03.2013
Authorised Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
1500000000 |
Equity Shares |
Re. 1/- each |
Rs.1500.000 Millions |
|
|
|
|
|
Issued, Subscribed & Paid-up Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
1035581955 |
Equity Shares |
Re. 1/- each |
Rs.1035.600 Millions |
|
|
|
|
|
FINANCIAL DATA
[all figures are
in Rupees Millions]
ABRIDGED BALANCE
SHEET
|
SOURCES
OF FUNDS |
31.03.2014 |
31.03.2013 |
31.03.2012 |
|
|
|
|
|
|
I.
EQUITY AND LIABILITIES |
|
|
|
|
(1)Shareholders' Funds |
|
|
|
|
(a) Share Capital |
2071.200 |
1035.600 |
1035.600 |
|
(b) Reserves & Surplus |
78826.000 |
76853.200 |
77745.600 |
|
(c) Money received against
share warrants |
0.000 |
0.000 |
0.000 |
|
|
|
|
|
|
(2) Share Application money
pending allotment |
0.000 |
0.000 |
0.000 |
|
Total
Shareholders’ Funds (1) + (2) |
80897.200 |
77888.800 |
78781.200 |
|
|
|
|
|
|
(3) Non-Current Liabilities |
|
|
|
|
(a) long-term borrowings |
46.400 |
46.400 |
0.000 |
|
(b) Deferred tax liabilities
(Net) |
1929.500 |
1654.100 |
1339.100 |
|
(c) Other long term
liabilities |
13.800 |
10.100 |
20.300 |
|
(d) long-term provisions |
2080.000 |
1314.900 |
986.200 |
|
Total
Non-current Liabilities (3) |
4069.700 |
3025.500 |
2345.600 |
|
|
|
|
|
|
(4) Current Liabilities |
|
|
|
|
(a) Short term borrowings |
41.700 |
384.900 |
403.000 |
|
(b) Trade payables |
3800.600 |
3632.700 |
4001.900 |
|
(c) Other current liabilities |
2336.100 |
1451.500 |
625.400 |
|
(d) Short-term provisions |
95.400 |
6081.500 |
5154.900 |
|
Total
Current Liabilities (4) |
6273.800 |
11550.600 |
10185.200 |
|
|
|
|
|
|
TOTAL |
91240.700 |
92464.900 |
91312.000 |
|
|
|
|
|
|
II.
ASSETS |
|
|
|
|
(1) Non-current assets |
|
|
|
|
(a) Fixed Assets |
|
|
|
|
(i) Tangible assets |
|
11347.600 |
9759.300 |
|
(ii) Intangible Assets |
|
10.300 |
13.700 |
|
(iii) Capital work-in-progress |
|
3480.400 |
2488.700 |
|
(iv) Intangible assets under
development |
0.000 |
0.000 |
0.000 |
|
(b) Non-current Investments |
34176.900 |
33764.900 |
35928.000 |
|
(c) Deferred tax assets (net) |
0.000 |
0.000 |
0.000 |
|
(d) Long-term Loan and Advances |
6734.900 |
4910.900 |
3385.400 |
|
(e) Other Non-current assets |
1.100 |
78.300 |
28.300 |
|
Total
Non-Current Assets |
58367.200 |
53592.400 |
51603.400 |
|
|
|
|
|
|
(2) Current assets |
|
|
|
|
(a) Current investments |
8600.000 |
9410.500 |
8449.800 |
|
(b) Inventories |
9183.800 |
8687.600 |
6400.700 |
|
(c) Trade receivables |
9682.600 |
7375.300 |
7134.800 |
|
(d) Cash and cash equivalents |
1414.800 |
4311.200 |
13277.100 |
|
(e) Short-term loans and
advances |
3093.200 |
8495.000 |
3906.100 |
|
(f) Other current assets |
899.100 |
592.900 |
540.100 |
|
Total
Current Assets |
32873.500 |
38872.500 |
39708.600 |
|
|
|
|
|
|
TOTAL |
91240.700 |
92464.900 |
91312.000 |
PROFIT & LOSS
ACCOUNT
|
|
PARTICULARS |
31.03.2014 |
31.03.2013 |
31.03.2012 |
|
|
|
SALES |
|
|
|
|
|
|
|
Income |
27625.600 |
24321.400 |
40155.600 |
|
|
|
Other Income |
1030.200 |
2361.700 |
3428.500 |
|
|
|
TOTAL |
28655.800 |
26683.100 |
43584.100 |
|
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
|
|
Cost of Materials Consumed |
8946.200 |
7057.900 |
9517.100 |
|
|
|
Purchases of Stock-in-Trade |
1850.400 |
2009.600 |
1874.800 |
|
|
|
Changes in Inventories of Finished Goods, Work-in-Progress and Stock-in-Trade |
(145.300) |
(105.300) |
(817.300) |
|
|
|
Employee Benefits Expense |
2789.600 |
2348.700 |
3165.600 |
|
|
|
Other Expenses |
10017.200 |
7883.700 |
8829.200 |
|
|
|
TOTAL |
23458.100 |
|
22569.400 (Including
Financial Expenses) |
|
|
|
|
|
|
|
|
Less |
PROFIT
BEFORE INTEREST, TAX, DEPRECIATION AND AMORTISATION |
5197.700 |
|
|
|
|
|
|
|
|
|
|
|
Less |
FINANCIAL
EXPENSES |
25.600 |
|
|
|
|
|
|
|
|
|
|
|
|
PROFIT/(LOSS)
BEFORE TAX, DEPRECIATION AND AMORTISATION |
5172.100 |
7488.500 |
21014.700 |
|
|
|
|
|
|
|
|
|
Less/ Add |
DEPRECIATION/
AMORTISATION |
1008.300 |
858.200 |
757.200 |
|
|
|
|
|
|
|
|
|
|
PROFIT BEFORE
EXCEPTIONAL ITEMS AND TAX |
4163.800 |
6630.300 |
20257.500 |
|
|
|
|
|
|
|
|
|
Less |
EXCEPTIONAL
ITEMS |
0.000 |
0.000 |
2999.200 |
|
|
|
|
|
|
|
|
|
|
PROFIT/(LOSS)
BEFORE TAX |
4163.800 |
6630.300 |
17258.300 |
|
|
|
|
|
|
|
|
|
Less |
TAX |
1155.400 |
1464.800 |
283.400 |
|
|
|
|
|
|
|
|
|
|
PROFIT/(LOSS)
AFTER TAX |
3008.400 |
5165.500 |
16974.900 |
|
|
|
|
|
|
|
|
|
Add |
PREVIOUS
YEARS’ BALANCE BROUGHT FORWARD |
31964.400 |
33376.900 |
23517.000 |
|
|
|
|
|
|
|
|
|
Less |
APPROPRIATIONS |
|
|
|
|
|
|
|
Proposed Dividend on Equity Share |
NA |
5178.000 |
4401.000 |
|
|
|
Corporate Dividend Tax |
NA |
880.000 |
714.000 |
|
|
|
Transfer to General Reserve |
NA |
520.000 |
2000.000 |
|
|
BALANCE CARRIED
TO THE B/S |
NA |
31964.400 |
33376.900 |
|
|
|
|
|
|
|
|
|
|
EARNINGS IN
FOREIGN CURRENCY |
|
|
|
|
|
|
|
Exports (FOB basis) |
|
19238.000 |
14628.200 |
|
|
|
Interest |
|
4.100 |
0.000 |
|
|
|
Royalty |
|
3.300 |
3.000 |
|
|
|
Others |
|
369.800 |
14.700 |
|
|
TOTAL EARNINGS |
NA |
19615.200 |
14645.900 |
|
|
|
|
|
|
|
|
|
|
IMPORTS |
|
|
|
|
|
|
|
Raw Materials |
|
2692.000 |
2895.200 |
|
|
|
Packaging Material |
|
944.400 |
669.000 |
|
|
|
Capital Goods |
|
1217.600 |
907.800 |
|
|
|
Stores and Spares |
|
498.600 |
347.100 |
|
|
TOTAL IMPORTS |
NA |
5352.600 |
4819.100 |
|
|
|
|
|
|
|
|
|
|
Earnings Per
Share (Rs.) |
1.45 |
5.00 |
16.40 |
|
KEY RATIOS
|
PARTICULARS |
|
31.03.2014 |
31.03.2013 |
31.03.2012 |
|
PAT / Total Income |
(%) |
10.50 |
19.36 |
38.95 |
|
|
|
|
|
|
|
Net Profit Margin (PBT/Sales) |
(%) |
15.07 |
27.26 |
42.98 |
|
|
|
|
|
|
|
Return on Total Assets (PBT/Total Assets} |
(%) |
7.30 |
12.01 |
32.63 |
|
|
|
|
|
|
|
Return on Investment (ROI) (PBT/Networth) |
|
0.05 |
0.09 |
0.22 |
|
|
|
|
|
|
|
Debt Equity Ratio (Total Debt /Networth) |
|
0.00 |
0.01 |
0.01 |
|
|
|
|
|
|
|
Current Ratio (Current Asset/Current Liability) |
|
5.24 |
3.37 |
3.90 |
FINANCIAL ANALYSIS
[all figures are
in Rupees Millions]
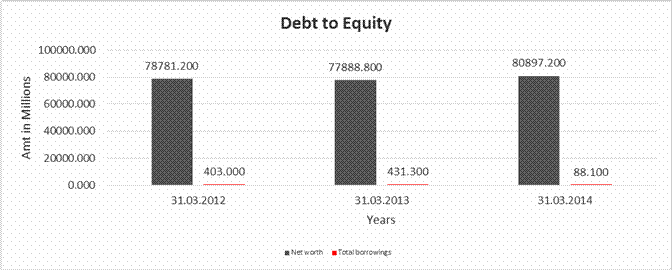
DEBT EQUITY RATIO
|
Particular |
31.03.2012 |
31.03.2013 |
31.03.2014 |
|
|
(Rs.
In Millions) |
(Rs.
In Millions) |
(Rs.
In Millions) |
|
Share Capital |
1035.600 |
1035.600 |
2071.200 |
|
Reserves & Surplus |
77745.600 |
76853.200 |
78826.000 |
|
Net
worth |
78781.200 |
77888.800 |
80897.200 |
|
|
|
|
|
|
long-term borrowings |
0.000 |
46.400 |
46.400 |
|
Short term borrowings |
403.000 |
384.900 |
41.700 |
|
Total
borrowings |
403.000 |
431.300 |
88.100 |
|
Debt/Equity
ratio |
0.005 |
0.006 |
0.001 |
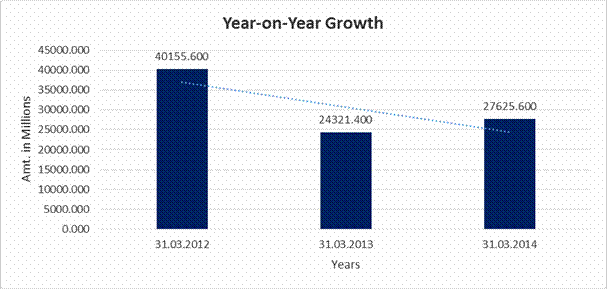
YEAR-ON-YEAR GROWTH
|
Year
on Year Growth |
31.03.2012 |
31.03.2013 |
31.03.2014 |
|
|
(Rs.
In Millions) |
(Rs.
In Millions) |
(Rs.
In Millions) |
|
Sales |
40155.600 |
24321.400 |
27625.600 |
|
|
|
(39.432) |
13.586 |
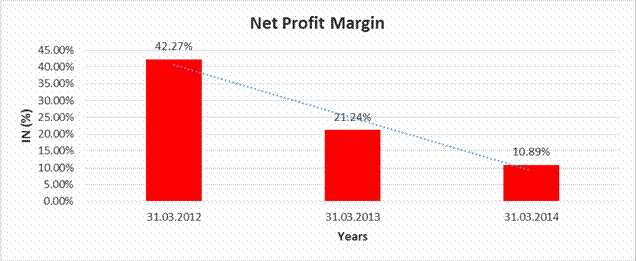
NET PROFIT MARGIN
|
Net
Profit Margin |
31.03.2012 |
31.03.2013 |
31.03.2014 |
|
|
(Rs.
In Millions) |
(Rs.
In Millions) |
(Rs.
In Millions) |
|
Sales |
40155.600 |
24321.400 |
27625.600 |
|
Profit |
16974.900 |
5165.500 |
3008.400 |
|
|
42.27% |
21.24% |
10.89% |
LOCAL AGENCY FURTHER INFORMATION
|
Sr. No. |
Check List by Info Agents |
Available in
Report (Yes / No) |
|
1] |
Year of Establishment |
Yes |
|
2] |
Locality of the firm |
Yes |
|
3] |
Constitutions of the firm |
Yes |
|
4] |
Premises details |
No |
|
5] |
Type of Business |
Yes |
|
6] |
Line of Business |
Yes |
|
7] |
Promoter's background |
Yes |
|
8] |
No. of employees |
Yes |
|
9] |
Name of person contacted |
No |
|
10] |
Designation of contact
person |
No |
|
11] |
Turnover of firm for last
three years |
Yes |
|
12] |
Profitability for last
three years |
Yes |
|
13] |
Reasons for variation
<> 20% |
----------- |
|
14] |
Estimation for coming
financial year |
No |
|
15] |
Capital in the business |
Yes |
|
16] |
Details of sister
concerns |
Yes |
|
17] |
Major suppliers |
No |
|
18] |
Major customers |
No |
|
19] |
Payments terms |
No |
|
20] |
Export / Import details
(if applicable) |
No |
|
21] |
Market information |
---------- |
|
22] |
Litigations that the firm
/ promoter involved in |
Yes |
|
23] |
Banking Details |
Yes |
|
24] |
Banking facility details |
Yes |
|
25] |
Conduct of the banking
account |
---------- |
|
26] |
Buyer visit details |
---------- |
|
27] |
Financials, if provided |
Yes |
|
28] |
Incorporation details, if
applicable |
Yes |
|
29] |
Last accounts filed at
ROC |
Yes |
|
30] |
Major Shareholders, if
available |
Yes |
|
31] |
Date of Birth of
Proprietor/Partner/Director, if available |
No |
|
32] |
PAN of
Proprietor/Partner/Director, if available |
No |
|
33] |
Voter ID No of
Proprietor/Partner/Director, if available |
No |
|
34] |
External Agency Rating,
if available |
Yes |
LITIGATION DETAILS
|
HIGH COURT OF GUJARAT TAX
APPEAL No. 1679 of 2007 |
||||||||||||||||||||||||||
|
Status: PENDING
(Converted from : ST/3691/2007) CCIN No : 001092200701679 Last Listing Date: 27/02/2008 Coram: HONOURABLE
MR.JUSTICE MOHIT S. SHAH HONOURABLE MR.JUSTICE
RAVI R.TRIPATHI |
||||||||||||||||||||||||||
|
S.N |
Name of the Petitioner |
Advocate On Record |
||||||||||||||||||||||||
|
1 |
THE COMMISSIONER
OF CENTRAL EXCISE AND CUSTOMS |
MR GAURANG H BHATT
for: Appellant(s) |
||||||||||||||||||||||||
|
S.N |
Name of the Respondant |
Advocate On Record |
||||||||||||||||||||||||
|
1 |
SUN PHARMACEUTICAL INDUSTRIES LIMITED |
RULE SERVED for
:Opponent(s) |
||||||||||||||||||||||||
|
Presented On : 03/12/2007
Registered On
: 03.12.2007 Bench Category : DIVISION BENCH District : NAVSARI Case Originated From: THROUGH ADVOCATE Listed : 119
times Stage Name : FOR ORDERS Act - TAXATION LAWS (CONTINUATION AND VALIDATION
OF RECOVERY PROCEEDINGS) ACT, 1964 |
||||||||||||||||||||||||||
|
Office Details |
||||||||||||||||||||||||||
|
|
||||||||||||||||||||||||||
|
S. No. |
Filing Date |
Document Name |
Advocate Name |
Court Fee on Document |
Document Details |
|||||||||||||||||||||
|
1 |
15/08/2007 |
VAKALATNAMA |
MR GAURANG H BHATT
ADVOCATE |
- |
MR GAURANG H
BHATT:1 |
|||||||||||||||||||||
|
2 |
15/08/2007 |
DOCUMENT |
RULE SERVED |
- |
RULE SERVED:1 |
|||||||||||||||||||||
|
3 |
03/02/2009 |
APPEARANCE NOTE |
MR GAURANG H BHATT
ADVOCATE |
- |
MR GAURANG H
BHATT:1 |
|||||||||||||||||||||
|
Court
Proceedings |
||||||||||||||||||||||||||
|
S. No. |
Notified Date |
Court Code |
Board Sr. No. |
Stage |
Action |
Coram |
||||||||||||||||||||
|
1 |
01/02/2008 |
2 |
- |
FOR FINAL HEARING
- TAX MATTERS |
NEXT DATE |
HONOURABLE THE
CHIEF JUSTICE Y.R.MEENA HONOURABLE
MR.JUSTICE J.C.UPADHYAYA |
||||||||||||||||||||
|
2 |
27/02/2008 |
2 |
- |
FOR ORDERS |
FIXED RULE / ADMIT |
HONOURABLE
MR.JUSTICE MOHIT S. SHAH HONOURABLE MR.JUSTICE RAVI R.TRIPATHI |
||||||||||||||||||||
|
Available Orders |
||||||||||||||||||||||||||
|
||||||||||||||||||||||||||
INDEX OF CHARGE:
|
Sr .No. |
Charge ID |
Date of Charge Creation/Modification |
Charge amount secured |
Charge Holder |
Address |
Service Request Number (SRN) |
|
1 |
10351563
|
31/03/2012
|
178,140,000.00
|
DEPARTMENT OF BIOTECHNOLOGY |
6-8TH FLOOR, BLOCK NO.2, CGO COMPLEX,
LODHI ROAD, NEW DELHI, DELHI - 110003, INDIA |
B38042354
|
|
2 |
90099243
|
27/03/2002
* |
500,000.00
|
THE BANK OF NOVA SCONA |
MITTAL TOWER; B WING, NARIMAN
POINT, MUMBAI, MAHARASHTRA - 400021, INDIA |
- |
|
3 |
90099216
|
26/09/2001
* |
82,500,000.00
|
THE BANK OF NOVA SCOTIA |
MAKER TOWER; B WING, NARIMAN
POINT, MUMBAI, MAHARASHTRA - 400021, INDIA |
- |
|
4 |
90099197
|
22/05/2000
|
218,000,000.00
|
ICICI BANK LIMITED |
FREE PRESS HOUSE, 215; NARIMAN
POINT, MUMBAI, MAHARASHTRA - 400021, INDIA |
- |
|
5 |
90095114
|
14/07/2004
* |
7,500,000.00
|
LAKSHDEEP INVESTMENT AND
FINANCE PRIVATE LIMITED |
3; NARAYAN BUILDING; 23; L.N.
ROAD, DADAR EAST, MUMBAI, MAHARASHTRA - 400014, INDIA |
- |
|
6 |
90095637
|
27/04/2000
* |
7,500,000.00
|
NVFC FINANCE PRIVATE LIMITED |
1843; 6TH CROSS 20TH MAIN,
J.P. NAGAR; II PHASE, BANGLORE, KARNATAKA - 560078, INDIA |
- |
|
7 |
90095112
|
05/08/2004
* |
15,000,000.00
|
RISK CAPITAL AND TECHNOLOGY FINANCE
CORPORATION LIMITED |
E-216; 3RD FLOOR, EAST
KAILASH, NEW DELHI, DELHI - 110065, INDIA |
- |
|
8 |
90095086
|
18/02/2005
* |
134,500,000.00
|
LAKSHDEEP INVESTMENT and
FINANCE PRIVATE LIMITED |
3; NARAYAN BUILDING; 23; L.N. ROAD,
DADAR EAST, MUMBAI, MAHARASHTRA - 400014, INDIA |
- |
|
9 |
90095083
|
09/07/2005
* |
32,500,000.00
|
INDUSTRIAL DEVELOPMENT BANK OF
INDIA |
IDBI TOWER, COLABA, MUMBAI,
MAHARASHTRA - 400005, INDIA |
- |
|
10 |
90101877
|
23/09/1998
|
250,000,000.00
|
ICICI LIMITED |
163, BACKBAY RECLAMATION,
MUMBAI, MAHARASHTRA - 400020, INDIA |
- |
* Date of charge modification
STATEMENT OF STANDALONE
UNAUDITED FINANCIAL RESULTS FOR THE QUARTER ENDED JUNE 30, 2014
(Rs.
In Millions)
|
|
Quarter Ended |
|
Particulars |
30.06.2014 Unaudited |
|
Income from
Operations |
|
|
Net Sales / Income from Operations (Net of
Excise Duty) |
5590.100 |
|
Other Operating Income |
165.700 |
|
Total Income
from Operations (Net) |
5755.800 |
|
Expenses |
|
|
Cost of materials consumed |
2388.400 |
|
Purchases of stock-in-trade |
313.600 |
|
Changes in inventories of finished goods, work-in-progress and stock-in-trade |
(451.400) |
|
Employee benefits expense |
876.800 |
|
Depreciation and amortisation expense |
463.900 |
|
Other expenses |
2329.300 |
|
Total Expenses |
5920.600 |
|
Profit from
Operations before Other Income and Finance Costs |
(164.800) |
|
Other income (net of impact of MTM loss on forward foreign exchange
contracts) |
842.100 |
|
Profit before
Finance Costs |
677.300 |
|
Finance costs |
80.900 |
|
Profit before
Tax |
596.400 |
|
Tax expense |
120.800 |
|
Net Profit for
the quarter / year |
475.600 |
|
Paid-up Equity
Share Capital |
2071.200 |
|
Equity Shares - Face Value Re. 1 each |
|
|
Reserves
excluding Revaluation Reserve |
|
|
Earnings Per
Share of Rs. 1 each (Basic and Diluted) |
0.023 |
|
Research and Development Expenses
incurred (included above) |
835.600 |
SELECT INFORMATION FOR THE QUARTER ENDED JUNE 30, 2014
|
Part II |
Quarter Ended |
|
Particulars |
30.06.2014 Unaudited |
|
|
|
|
Public
Shareholding |
|
|
Number of Equity Shares of Re. 1 each |
752822510 |
|
Percentage of Shareholding |
36.35 |
|
Promoters and
Promoter Group Shareholding |
|
|
Pledged / Encumbered |
|
|
Number of Equity Shares of Re. 1 each |
4370000 |
|
Percentage of Equity Shares (as a % of the total shareholding of promoter and promoter group) |
0.33 |
|
Percentage of Equity Shares (as a % of the total share capital of the Company) |
0.21 |
|
Non-encumbered |
|
|
Number of Equity Shares of Re. 1 each |
1313976400 |
|
Percentage of Equity Shares (as a % of the
total shareholding of promoter and promoter group) |
99.67 |
|
Percentage of Equity Shares (as a % of the
total share capital of the Company) |
63.44 |
|
|
|
|
|
5808 |
|
Investor
Complaints |
|
|
Pending at the beginning of the quarter |
-- |
|
Received during the quarter |
6 |
|
Disposed of during the quarter |
6 |
|
Remaining unresolved at the end of the quarter |
-- |
Notes:
1. The above financial results of the Company have been reviewed by the Audit Committee and approved by the Board of Directors at their respective meetings held on August 12, 2014 and have been subjected to a Limited Review by the Statutory Auditors of the Company.
2. The Company has only one reportable business segment namely 'Pharmaceuticals'.
3. In terms of the resolution passed by the shareholders by way of Postal Ballot on July 15, 2013, the Company has allotted 1,035,581,955 bonus equity shares on August 03, 2013 in the ratio of 1 equity share of Rs. 1 each fully paid up for every equity share of 1 each held. Consequently, the Earnings Per Share of Rs. 1 each for the quarter ended June 30, 2013 has been restated based on the number of equity shares post bonus issue, i.e. 2,071,163,910 equity shares, in accordance with Accounting Standard (AS-20) on 'Earnings Per Share'.
4. Pursuant to the scheme of arrangement in the nature of demerger, the specified undertaking of Sun Pharma Global FZE, a wholly owned subsidiary, has been transferred into the Company w.e.f May 01, 2013, without any consideration, on a going concern basis consisting of all the assets and liabilities pertaining to the said undertaking. The scheme of arrangement has been approved by shareholders of both the companies and is also approved by the Hon’ble High Court of Gujarat. Accordingly, the financial statements for the year ended March 31, 2014, earlier approved by the Board of Directors at their meeting held on May 29, 2014, have been revised and approved by them at their meeting held August 12, 2014. The effect of the Scheme was given in the financial results for the year ended March 31, 2014 only on an annual basis and approved by the Board of Directors at their meeting held on August 12, 2014. However, the financial results for quarterly periods during the year ended March 31, 2014 have not been restated in these results.
5. Consequent to the Company adopting the useful lives of fixed assets as indicated in Part C of Schedule II of the Companies Act, 2013, which has come into effect from April 01, 2014, the depreciation charge for the quarter ended June 30, 2014 is higher by Rs. 190.900 Millions and the adjustment by way of charge to retained earnings is Rs. 145.200 Millions (net of deferred tax effect of Rs. 74.800 Millions).
6. The Board of Directors of the Company at their meeting held on April 06, 2014 approved the proposed scheme of arrangement u/s 391 to 394 of the Companies Act. 1956 for amalgamation of Ranbaxy Laboratories Ltd into the Company with effect from April 01, 2014, the appointed date ("the Proposed Scheme"). The National Stock Exchange Ltd and BSE Ltd have conveyed their 'No Objection' in July 2014. Pending approval of the Proposed Scheme by the shareholders of the Company at the Court convened meeting to be held on August 22, 2014, the Hon’ble High Court of Gujarat and other statutory compliances, no effect of the Proposed Scheme has been given in these financial results.
7. Figures for the quarters / year have been regrouped wherever considered necessary, other than as referred in Note 4 above.
FIXED ASSETS
· Freehold Land
·
Leasehold
Land
·
Buildings
·
Buildings
– Leased
·
Plant
and Equipment
·
Vehicles
·
Office
Equipment
·
Furniture
and Fixtures
·
Trademarks
·
Designs
PRESS RELEASE:
SUN PHARMA REPORTS A
STRONG QUARTER
Mumbai, August 12,
2014: Sun Pharmaceutical Industries Limited (Reuters: SUN.BO, Bloomberg: SUNP
IN, NSE: SUNPHARMA, BSE: 524715)
reported financials for first quarter ending June 30th, 2014.
Highlights of Q1 FY15
consolidated financials
• Net sales / Income from operations at Rs. 39270.000 Millions, a growth of 13% over same quarter last year.
• Branded generic sales in India at Rs. 9920.000 Millions, up by 17% over Q1 last year.
• US finished dosage sales at US$ 389 million grew by 7% (in US$ terms) over Q1 last year.
• International formulation sales outside US at US$ 82 million, grew by 2% (in US$ terms) over Q1 last year.
• Overall international revenues accounted for more than 75% of total revenues for the quarter.
• EBITDA at Rs. 17240.000 Millions grew by 13%; resulting EBITDA margin of 44%, same as Q1 last year.
• Adjusted Net profit at Rs 13910.000 Millions witnessed a growth of 12% over Q1 last year; resulting margin of 35%.
• Reported Net profit at Rs. 13910.000 Millions, compared to Net loss of Rs. 12760.000 Millions in Q1 last year. The loss in Q1 last year was on account of a provision of Rs. 25170.000 Millions towards settlement for patent infringement litigation related to generic versions of ‘Protonix’.
These results were taken on record by the Board of Directors at a meeting held in Mumbai today.
Dilip Shanghvi, Managing Director of the Company said, “Our Q1 performance was in-line with our expectations. While we are intensively planning for the integration with Ranbaxy, we remain focused on strengthening our existing businesses and developing a differentiated and specialty driven product basket. We also continue to review opportunities to expand and strengthen our global footprint.”
India Branded Generics
– Chronic Therapy Leadership
Sale of branded prescription formulations in India was Rs. 9920.000 Millions, up by 17% from Q1 last year. As per AIOCD-AWACS report, the average industry growth was approximately 8% for Q1FY15. Seven products were launched in the Indian market during the quarter.
Sun Pharma is ranked 2nd and holds 5.4% market share in the Rs. 77,000 crore pharmaceutical market as per June-2014 AIOCD-AWACS report. It continues to be ranked no. 1 based on share of prescriptions with 7 classes of specialists: psychiatrists, neurologists, cardiologists, ophthalmologists, orthopedicians, nephrologists, and gastroenterologists.
US Formulations: Taro
price protection charges moderates overall growth
Sales in the US were US$ 389 million for the quarter, up by 7%, accounting for 58% of total sales.
Taro recently posted Q1 sales of US$ 130 million, down 15% from the corresponding quarter last year. Taro’s net profit for Q1 was US$ 46 million, a decline of 22% over Q1 last year. The decline in sales and profits is mainly due to the price protection charge taken by Taro during the quarter.
International ex-US
formulation sales
Formulation sales in international markets excluding US accounted for US$ 82 million in Q1FY15, registering a growth of 2% (in US$ terms). Excluding ex-US Taro sales, underlying sales growth in US$ terms for Sun Pharma business in these markets was 4% for Q1FY15.
Active Pharmaceutical
Ingredients (API): Strategic strength
External sales of API reached Rs. 174 Millions in Q1FY15, a decline of 10% over Q1 last year. On the other hand, we increased the API supply for captive consumption significantly for key products which enabled us to enjoy the benefits of strategic vertical integration. A cumulative of 259 DMF / CEP applications have been made, with 176 approved so far.
Research – Investing
for future
Consolidated R and D expense for Q1FY15 was Rs. 2570.000 Millions, or 6.6% of sales.
We now have a comprehensive product offering in the US market with approved ANDAs for 350 products while filings for 140 products await US FDA approval, including 12 tentative approvals. In the first quarter, ANDAs for 14 products were filed and 6 approvals were received.
The total number of patent applications submitted now stands at 575 with 349 patents granted so far.
SUN PHARMA UNIT RECALLS MUTIPLE LOTS OF CAPSULES FROM US
Caraco Pharmaceutical Laboratories, a unit of
Sun Pharma, has initiated a recall of multiple lots of Cephalexin capsules from
the US market.
According to a notification by the USFDA, the
recall of the 3,40,553 units of 500 mg and 1,13,677 units of 250 mg bottles is
voluntarily initiated by the company through a letter to the regulator in June
under 'Class-II' classification.
Cephalexin is an antibiotic that belongs to
the family of medications known as cephalosporins. It is used to treat certain
types of bacterial infections.
"CGMP Deviations: These products are
being recalled because they were manufactured with active pharmaceutical
ingredients (APIs) that were not manufactured with good manufacturing
practices," USFDA's website said citing the reason for recall.
When contacted, a Sun Pharma spokesperson
offered no comments.
The recalled drug bottles were distributed by
Caraco Pharmaceutical Laboratories, Ltd in the US while manufactured in India
by Sun Pharmaceutical Industries Ltd.
According to American health regulator USFDA,
Class II recall is a situation in which use of or exposure to a violative
product may cause temporary or medically reversible adverse health consequences
or where the probability of serious adverse health consequences is remote.
Recently Caraco Pharmaceutical had said that
it initiated a recall of some lots of Venlafaxine Hydrochloride extended-release
tablets from the US market for not meeting the drug release dissolution
specifications under 'Class-II' classification.
Meanwhile, in another notification FDA said
Wockhardt USA has initiated a recall of 840 bottles of Bupropion hydrochloride
extended-release tablets USP (SR), 100 mg, (500-count bottle) from USA market.
The reason for recall: "Out of specification levels of the impurity
m-chlorobenzoic acid were observed'.
Bupropion hydrochloride extended-release
tablets (SR) are indicated for the treatment of major depressive disorder.
SUN PHARMA SLIPS 2%,
RANBAXY MERGER MAY BE DELAYED
Shares of Sun Pharma slipped over 2 percent intraday on Friday as investors were worried that merger with Ranbaxy may still be stuck in some roadblocks. According to a media report its merger with Ranbaxy Laboratories may take longer than usual to secure all regulatory clearances.
Earlier in May, Andhra Pradesh High Court had cleared the decks for the USD 4-billion deal by lifting the stay it had imposed. The deal, however, needs to be approved by regulatory bodies such as CCI, BSE, NSE and Sebi which were investigating the insider-trading allegations.
On April 6, Sun Pharma had announced to fully acquire Ranbaxy in an all-stock transaction with a total equity value of USD 3.2 billion, along with debt of USD 800 million, taking the overall deal value to USD 4 billion.
At 12:32 hrs, Sun Pharma was quoting at Rs 629.40, down Rs 12.55, or 1.95 percent while Ranbaxy Laboratories was at Rs 468.55, down Rs 7.95, or 1.67 percent on the BS
SUN PHARMA, TARO CALL OFF PLANNED MERGER
Friday, Feb 08, 2013
Mumbai: Sun Pharmaceutical Industries Limited and Taro
Pharmaceutical Industries Limited have decided to terminate their August 2012
merger agreement, some three years after
With the termination of the agreement, announced in a joint statement on
Friday, Sun Pharma’s offer to all shareholders of Taro to buy their holdings at
$39.50 (around Rs.2120 today) per share stands cancelled. Taro will, however,
remain a subsidiary of the Indian company.
The move, announced the same day that Sun Pharma posted a 32% increase
in third quarter net profit, follows objections raised by a group of investors,
including minority shareholders, against the price offered by Sun Pharma, which
is lower than the prevailing market price.
Sun Pharma in August said it plans to buy more shares in Taro from
minority shareholders, including the public and a couple of institutional
investors, to raise its stake in the Israeli company beyond the existing 66%.
This offer was made to enable the merger of the company into Sun Pharma, which
has turned around Taro since acquiring it at a time when it was ailing.
A merger would have given the Indian firm full control of the Israeli
unit’s management. The proposed buyout of minority shareholders and the
delisting of Taro from the New York Stock Exchange (NYSE) would have improved
Sun Pharma’s cash flows because it would have saved on dividend payouts.
The firms said in their joint statement that they decided to call off
the merger “in the best interests of the companies and their shareholders”. The
decision was taken at the direction of the special committee formed to handle
the merger plan, said the statement made after the close of stock-market
trading in Mumbai.
Shares of Sun Pharma fell 0.63% to Rs.744.05 on BSE on Friday. The
Sensex lost 0.49% to 19,484.77 points. Taro shares were trading 2.87% up at $52
on NYSE at 9.15pm India time.
“The termination of the merger plan will not have any significant impact
on the revenue and the operations of Taro,” said Sun Pharma chairman Dilip Shanghvi in a conference call with media
and analysts on Friday.In August, the merger deal was approved by Taro’s board
of directors on the recommendation of the special committee.
Sun Pharma acquired a controlling stake in Taro in September 2010, after
a three-year-long court battle between the two firms. They signed the first
merger pact in May 2007.
The court battle was followed by a unilateral termination of this
agreement by the Taro board after the firm’s promoters, Barry Levitt and his family, and a few
institutional shareholders objected to the valuation. Following a series of
battles in Israeli and US courts, Sun Pharma received a favourable order from
the Supreme Court of Israel that allowed it to acquire a majority stake,
including the whole holding of the promoters, in 2010.
With a 25% increase in net profit, the subsidiary contributed
significantly to Sun Pharma’s third-quarter revenue and profit in fiscal 2013
that was announced on Friday.
“It (the termination of the merger) was quite expected as the
shareholders had not agreed to the price of $39.50 a unit. Since the shares of
Taro are currently traded at above $45 and also with the better performance, it
would seem difficult to get shareholder consent for the merger at a lower price
offer,” said Hitesh Mahida, an industry analyst with
brokerage Fortune Equity Brokers (India) Limited.
“I don’t think termination will have any impact on the company’s
business as Taro will remain a subsidiary of Sun Pharma till a consensus is
made on the offer price,” he said.
Sun Pharma’s net profit rose to Rs.8810.000 Millionse from the year-ago
quarter’s Rs.6680.000 Millions, it said on Friday. Exports grew 42% and margins
widened substantially, helping it beat analysts’ estimates. Sales rose by
one-third to Rs.28520.000 Millions in fiscal third quarter from Rs.21450.000 a
year ago.
SUN
PHARMACEUTICAL INDUSTRIES GETS FAVOURABLE US SUPREME COURT VERDICT IN PATENT
CASE
April 18, 2012
![]()
NEW DELHI: Sun Pharmaceutical Industries today said the US Supreme Court
has given a favourable verdict in its patent case against Novo Nordisk over
generic Prandin tablets used for treating diabetes.
US Supreme Court has ruled in favour of the company's subsidiary, Caraco
Pharmaceutical Laboratories, in it's patent litigation against Novo Nordisk
over Caraco's generic version of prandin, repaglinide tablets, Sun Pharma said
in a statement.
![]()
"The Supreme Court, in a unanimous opinion, concluded that Caraco
can seek correction of Novo Nordisk's inaccurate use code regarding the
combination use of repaglinide and metformin for the treatment of type II
diabetes," it added.
The judgement has said, "The text and context of the provision
demonstrate that a generic company can employ the counterclaim to challenge a
brand's overbroad use code. We accordingly hold that Caraco may bring a
counterclaim seeking to 'correct' Novo's use code..."
Prandin is a registered trademark of Denmark-based drug firm Novo
Nordisk.
The decision will help all generic companies prevent brand companies from
misrepresenting their patents to the USFDA and improperly delaying or
preventing generic companies from marketing their drugs, Sun Pharma said.
Caraco's abbreviated new drug application (ANDA) for generic Prandin is
still awaiting approval by the United States Food and Drug Administration
(USFDA), it added.
"Prandin has annual sales of approximately $ 230 million in the
US," Sun Pharma said.
A separate appeal concerning the validity of patents for Prandin is
pending before the Court of Appeals for the Federal Circuit after a lower court
ruled in favour of Caraco, it added.
Scrips of Sun Pharma were trading at Rs 596.50 apiece in the afternoon
on the BSE, up 2.11 per cent from its previous close.
SUN PHARMA REPORTS
STRONG QUARTER
Net Sales Rs.34820.000 Millions, up 31%, Adjusted Net Profit
Rs.12410.000 Millions, up 56%
Mumbai, August 09, 2013: Sun Pharmaceutical Industries Limited
(Reuters: SUN.BO, Bloomberg: SUNP IN,
Highlights of Q1FY14 consolidated financials
·
Net sales/Income from operations at Rs. 34820.000
Millions, a growth of 31% over same quarter last year. Adjusted for the impact
of one-time sales recorded in the domestic business in Q4FY12, which lowered
Q1FY13 sales, the net sales have grown by 23% over Q1FY13.
·
Branded generic sales in India, at Rs. 8490.000
Millions, grew by 44% over Q1 last year. Adjusted sales growth of the domestic
formulation business, as explained above, is 11%.
·
US finished dosages sale is US$ 364 million
recording a growth of 28% (in US$ terms) over Q1 last year.
·
International formulation sales is US$ 81 million,
and grew by 19% (in US$ terms) over same quarter last year.
·
EBITDA at Rs. 15310.000 Millions grew by 26% YoY
while EBITDA margins were at 44%, compared to 46% in Q1 last year.
·
Recurring Net profit at Rs 12410.000 Millions
witnessed a growth of 56% over Q1 last year; resulting margin of 36%.
·
Reported Net loss at Rs. 12760.000 Millions, on
account of a provision of Rs 25170.000 Millions towards settlement for patent infringement
litigation related to generic versions of ‘Protonix’.
These results were taken on record by the Board of Directors at a
meeting held in Mumbai today.
Dilip Shanghvi, Managing Director of the Company said, “All our
businesses continue to perform in-line with our expectations. We remain focused
on strengthening our existing businesses and developing a differentiated and
specialty driven product basket. We also continue to review opportunities to
expand and strengthen our global footprint.”
India Branded Generics – Chronic Therapy Leadership
Sales of branded prescription formulations in India were at Rs. 8490.000
Millions, grew by 44% from Q1 last year, accounting for 24% of total sales.
Adjusted sales growth for the quarter was 11%.
Sun Pharma is ranked 2rd and holds 5.1% market share in the Rs.72000.000
Millions Indian pharmaceutical market, as per June-2013 AIOCD-AWACS report.
Based on SMSRC data for Feb-2013, the company continues to be ranked no. 1
based on share of prescriptions with 7 classes of specialists: psychiatrists,
neurologists, cardiologists, ophthalmologists, orthopedicians, nephrologists
and gastroenterologists.
10 products were launched in the first quarter.
The company has completed the process of transferring its domestic formulations
business to Sun Pharma
Laboratories Limited, a wholly owned subsidiary.
US Formulations – Strong performance led by Sun portfolio
Sales in the US were at US$ 364 million for the quarter, up by 28%,
accounting for 58% of total sales.
Taro recently posted overall sales of US$ 153 million for Q1FY14, a
decline of 4% from the corresponding quarter
last year. Excluding the one-time charge related to price adjustments on
contractual obligations, sales would have
been higher by 10% YoY. Taro’s Net profit for Q1 was US$ 59 million.
During the quarter, Sun Pharma settled the ongoing litigation pending in
the US District Court, District of New
Jersey regarding Sun Pharma subsidiary’s generic pantoprazole. Under the
terms of the litigation settlement
between Sun Pharma, and Wyeth, (now a division of Pfizer Inc.) and
Altana Pharma AG, (now known as Takeda
GmbH) the parties have dismissed all their claims. Sun Pharma will pay a
lump-sum US$ 550 million as a part of
this settlement.
Sun Pharma had already provided Rs. 5840.000 Millions in Q2FY13 towards
this liability. For the quarter ended 30th Jun- 2013, the company has made a
provision of Rs. 25170.000 Millions towards the difference between total
settlement amount and past provision.
During the quarter, Sun Pharma’s subsidiary received a favourable
verdict from the US Federal Circuit Court regarding its on-going patent
litigation with Novo Nordisk for generic Prandin.
Rest of World – Growth traction continues
Formulation sales in rest of the world (ROW) markets outside of India
and US were US$ 81 million in Q1FY14 registering a growth of 19% (in US$
terms). Excluding ex-US Taro sales, underlying sales growth in US$ terms for
Sun Pharma’s business in these markets was 23% for Q1FY14.
Active Pharmaceutical Ingredients (API) – Focus on vertical integration
The API business continues to be largely used for vertical integration
on key products. A cumulative of 243 DMF /
CEP applications have been made, with 171 approved so far. External
sales of API, accounting for a fraction of the total API production, reached Rs
1930.000 Millions in Q1FY14 with marginal decrease of 4% over the same quarter
last year.
Research – Investing for future
Consolidated R and D expense for Q1FY14 was Rs. 2050.000 Millions, at 6%
of sales.
In the first quarter, ANDAs for 4 products were filed. After counting
these, and adjusting for filings that were dropped, cumulatively ANDAs for 453
products have been filed by Sun Pharma and Taro with the USFDA (as on
June 30, 2013). ANDAs for 9 products received approvals in the first
quarter, taking the total number of approvals to 320 (as on June 30, 2013).
ANDAs for 133 products now await USFDA approval, including 19 tentative
approvals. The above ANDA statistics exclude the discontinued/withdrawn
products of URL.
The total number of patent applications submitted now stands at 791,
with 503 patents granted so far.
CMT REPORT (Corruption, Money Laundering & Terrorism]
The Public Notice information has been collected from various sources
including but not limited to: The Courts, India Prisons Service,
Interpol, etc.
1] INFORMATION ON
DESIGNATED PARTY
No exist designating subject or any of its beneficial owners, controlling
shareholders or senior officers as terrorist or terrorist organization or whom
notice had been received that all financial transactions involving their assets
have been blocked or convicted, found guilty or against whom a judgement or
order had been entered in a proceedings for violating money-laundering,
anti-corruption or bribery or international economic or anti-terrorism sanction
laws or whose assets were seized, blocked, frozen or ordered forfeited for
violation of money laundering or international anti-terrorism laws.
2] Court Declaration :
No exist to suggest that subject is or was
the subject of any formal or informal allegations, prosecutions or other
official proceeding for making any prohibited payments or other improper payments
to government officials for engaging in prohibited transactions or with
designated parties.
3] Asset Declaration :
No records exist to suggest that the property or assets of the subject
are derived from criminal conduct or a prohibited transaction.
4] Record on Financial
Crime :
Charges or conviction
registered against subject: None
5] Records on Violation of
Anti-Corruption Laws :
Charges or
investigation registered against subject: None
6] Records on Int’l
Anti-Money Laundering Laws/Standards :
Charges or
investigation registered against subject: None
7] Criminal Records
No
available information exist that suggest that subject or any of its principals
have been formally charged or convicted by a competent governmental authority for
any financial crime or under any formal investigation by a competent government
authority for any violation of anti-corruption laws or international anti-money
laundering laws or standard.
8] Affiliation with
Government :
No record
exists to suggest that any director or indirect owners, controlling
shareholders, director, officer or employee of the company is a government
official or a family member or close business associate of a Government
official.
9] Compensation Package :
Our market
survey revealed that the amount of compensation sought by the subject is fair
and reasonable and comparable to compensation paid to others for similar
services.
10] Press Report :
No press reports / filings exists on
the subject.
CORPORATE GOVERNANCE
MIRA INFORM as part of its Due Diligence do provide comments on
Corporate Governance to identify management and governance. These factors often
have been predictive and in some cases have created vulnerabilities to credit
deterioration.
Our Governance Assessment focuses principally on the interactions
between a company’s management, its Board of Directors, Shareholders and other
financial stakeholders.
CONTRAVENTION
Subject is not known to have contravened any existing local laws,
regulations or policies that prohibit, restrict or otherwise affect the terms
and conditions that could be included in the agreement with the subject.
FOREIGN EXCHANGE RATES
|
Currency |
Unit
|
Indian Rupees |
|
US Dollar |
1 |
Rs.60.73 |
|
|
1 |
Rs.101.48 |
|
Euro |
1 |
Rs.81.04 |
INFORMATION DETAILS
|
Information
Gathered by : |
HTL |
|
|
|
|
Analysis Done by
: |
DIV |
|
|
|
|
Report Prepared
by : |
VRN |
SCORE & RATING EXPLANATIONS
|
SCORE FACTORS |
RANGE |
POINTS |
|
HISTORY |
1~10 |
8 |
|
PAID-UP CAPITAL |
1~10 |
9 |
|
OPERATING SCALE |
1~10 |
8 |
|
FINANCIAL CONDITION |
|
|
|
--BUSINESS SCALE |
1~10 |
9 |
|
--PROFITABILIRY |
1~10 |
8 |
|
--LIQUIDITY |
1~10 |
9 |
|
--LEVERAGE |
1~10 |
8 |
|
--RESERVES |
1~10 |
8 |
|
--CREDIT LINES |
1~10 |
8 |
|
--MARGINS |
-5~5 |
- |
|
DEMERIT POINTS |
|
|
|
--BANK CHARGES |
YES/NO |
YES |
|
--LITIGATION |
YES/NO |
YES |
|
--OTHER ADVERSE INFORMATION |
YES/NO |
NO |
|
MERIT POINTS |
|
|
|
--SOLE DISTRIBUTORSHIP |
YES/NO |
NO |
|
--EXPORT ACTIVITIES |
YES/NO |
NO |
|
--AFFILIATION |
YES/NO |
NO |
|
--LISTED |
YES/NO |
YES |
|
--OTHER MERIT FACTORS |
YES/NO |
YES |
|
DEFAULTER |
|
|
|
--RBI |
YES/NO |
NO |
|
--EPF |
YES/NO |
NO |
|
TOTAL |
|
75 |
This score serves as a reference to assess SC’s credit risk
and to set the amount of credit to be extended. It is calculated from a
composite of weighted scores obtained from each of the major sections of this
report. The assessed factors and their relative weights (as indicated through
%) are as follows:
Financial
condition (40%) Ownership
background (20%) Payment
record (10%)
Credit history
(10%) Market trend
(10%) Operational
size (10%)
RATING EXPLANATIONS
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
>86 |
Aaa |
Possesses an extremely sound financial base with the strongest
capability for timely payment of interest and principal sums |
Unlimited |
|
71-85 |
Aa |
Possesses adequate working capital. No caution needed for credit transaction.
It has above average (strong) capability for payment of interest and
principal sums |
Large |
|
56-70 |
A |
Financial & operational base are regarded healthy. General unfavourable
factors will not cause fatal effect. Satisfactory capability for payment of
interest and principal sums |
Fairly Large |
|
41-55 |
Ba |
Overall operation is considered normal. Capable to meet normal
commitments. |
Satisfactory |
|
26-40 |
B |
Capability to overcome financial difficulties seems comparatively
below average. |
Small |
|
11-25 |
Ca |
Adverse factors are apparent. Repayment of interest and principal sums
in default or expected to be in default upon maturity |
Limited with full
security |
|
<10 |
C |
Absolute credit risk exists. Caution needed to be exercised |
Credit not
recommended |
|
-- |
NB |
New Business |
-- |
This report is issued at your request without any
risk and responsibility on the part of MIRA INFORM PRIVATE LIMITED (MIPL)
or its officials.