MIRA INFORM REPORT
|
Report Date : |
27.12.2014 |
IDENTIFICATION DETAILS
|
Name : |
PANACEA BIOTEC LIMITED (w.e.f. 07.09.2003) |
|
|
|
|
Formerly Known
As : |
PANACEA DRUGS LIMITED (w.e.f. 09.09.1993) PANACEA DRUGS PRIVATE LIMITED |
|
|
|
|
Registered
Office : |
|
|
|
|
|
Country : |
|
|
|
|
|
Financials (as
on) : |
31.03.2014 |
|
|
|
|
Date of Incorporation
: |
02.02.1984 |
|
|
|
|
Com. Reg. No.: |
16-022350 |
|
|
|
|
Capital
Investment / Paid-up Capital : |
Rs.61.300 Millions |
|
|
|
|
CIN No.: [Company Identification
No.] |
L33117PB1984PLC022350 |
|
|
|
|
TAN No.: [Tax Deduction & Collection
Account No.] |
PNEP07035A |
|
|
|
|
PAN No.: [Permanent Account No.] |
AAACP5335J |
|
|
|
|
Legal Form : |
Public Limited Liability Company. The Company’s Shares are Listed on the Stock Exchanges. |
|
|
|
|
Line of Business
: |
Manufacturer and Marketer of Pharmaceutical Formulations viz. Tablets, Syrups/ Liquids, Capsules, Gels and Vaccines. |
|
|
|
|
No. of Employees
: |
Not Available [We tried to
confirm the number of employees but no one is ready to part any information from
the company management] |
RATING & COMMENTS
|
MIRA’s Rating : |
Ca (21) |
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
11-25 |
Ca |
Adverse factors are apparent. Repayment of interest and principal sums
in default or expected to be in default upon maturity |
Limited with
full security |
|
Status : |
Moderate |
|
|
|
|
Payment Behaviour : |
Slow |
|
|
|
|
Litigation : |
Exist |
|
|
|
|
Comments : |
Subject is a well-established company having moderate track record. The company is continuously incurring losses from its operational
activities. The rating also takes into account the recent delays by the company in
servicing its debt obligation. However, Business is active. Payment terms are reported to be slow. The company can be considered for business dealings on a safe and
secured trade terms and conditions. |
NOTES:
Any query related to this report can be made
on e-mail : infodept@mirainform.com
while quoting report number, name and date.
ECGC Country Risk Classification List – September 30, 2014
|
Country Name |
Previous Rating (30.06.2014) |
Current Rating (30.09.2014) |
|
|
A1 |
A1 |
|
Risk Category |
ECGC
Classification |
|
Insignificant |
A1 |
|
Low |
A2 |
|
Moderate |
B1 |
|
High |
B2 |
|
Very High |
C1 |
|
Restricted |
C2 |
|
Off-credit |
D |
EXTERNAL AGENCY RATING
|
Rating Agency Name |
CARE |
|
Rating |
Long Term Bank Facilities: D |
|
Rating Explanation |
Lowest credit quality very low prospects of
recovery. |
|
Date |
11.02.2014 |
RBI DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available RBI Defaulters’ list.
EPF (Employee Provident Fund) DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available EPF (Employee Provident Fund) Defaulters’ list as of
31-03-2014.
INFORMATION DENIED
Management Non Co-operative (91-11-26945270)
LOCATIONS
|
Registered Office : |
|
|
Tel. No.: |
91-11-41578049 / 26945270 |
|
Fax No.: |
Not Available |
|
E-Mail : |
|
|
Website : |
|
|
|
|
|
Head/ Corporate Office 1: |
B-1 Extension / A-27, Mohan Cooperative Industrial Estate, Mathura Road, New Delhi – 110044, India |
|
Tel. No.: |
91–11–26945270/ 41679000 Extn. 2081 (D) 41578024 / 26974500 / 41678000 |
|
Fax No.: |
91–11–26940199/ 26940621 / 41679075 / 41679070 / 41679081 |
|
E-Mail : |
panbio.panbio@rme.sprintrpg.ems.vsnl.net.in |
|
Website : |
|
|
|
|
|
Administrative/ Secretarial/ Corporate Office 2: |
B – 1 Extension / G – 3, Mohan Co-Operation Industrial
Estate, |
|
Tel. No.: |
91-11-41679000 Extn. 2081, (D) 41578024/ 41679015 |
|
Fax No.: |
91-11-41679070/ 41679075 |
|
E-mail: |
|
|
|
|
|
Manufacturing Facilities : |
· Ambala-Chandigarh Highway, Lalru – 140 501, Punjab, India · A-239 -242, Okhla Indl. Area, Phase–I, New Delhi – 110 020, India · Malpur, Baddi, Dist. Solan, Himachal Pradesh-173 205, India · B-1/E-12, Mohan Co-operative Industrial Estate, Mathura Road, New Delhi – 110 044, India · Plot No. 72/3, Gen Block, T.T.C. Industrial Area, Mahape, Navi Mumbai - 400 710, Maharashtra, India |
|
|
|
|
Branch/ Sales and Marketing Office : |
701, |
|
|
|
|
R & D Centers : |
·
· B-1/E-xtn. A-24-25,, Mohan Co-operative Industrial Estate Mathura Road, New Delhi – 110044, India ·
Plot No. E-4, Phase II, Industrial Area Mohali
– 160055, · Plot No. 72/3, Gen Block, T.T.C. Industrial Area, Mahape, Navi Mumbai – 400710, Maharashtra, India |
DIRECTORS
AS ON 31.03.2014
|
Name : |
Mr. Soshil Kumar Jain |
|
Designation : |
Whole-time director |
|
Address : |
18/56, East Park Area, Karol Bagh, New Delhi – 110005, India |
|
Date of Birth/Age : |
04.04.1933 |
|
Qualification : |
Pharmacist |
|
Date of Appointment : |
02.02.1984 |
|
DIN No.: |
00012812 |
|
|
|
|
Name : |
Mr. Ravinder Jain |
|
Designation : |
Managing Director |
|
Address : |
18/56, East Park Area, Karol Bagh, New Delhi – 110005, India |
|
Date of Birth/Age : |
03.08.1957 |
|
Qualification : |
Matriculate |
|
Date of Appointment : |
15.11.1984 |
|
DIN No.: |
00010101 |
|
|
|
|
Name : |
Dr. Rajesh Jain |
|
Designation : |
Managing Director |
|
Address : |
18/56, East Park Area, Karol Bagh, New Delhi – 110005, India |
|
Date of Birth/Age : |
26.04.1964 |
|
Qualification : |
B.Sc., MBA, PGDBM,
Advanced Management Diploma in Market Research |
|
Date of Appointment : |
25.11.2006 |
|
DIN No.: |
00013053 |
|
|
|
|
Name : |
Mr. Sandeep Jain |
|
Designation : |
Managing Director |
|
Address : |
18/56, East Park Area, Karol Bagh, New Delhi – 110005, India |
|
Date of Birth/Age : |
17.07.1966 |
|
Qualification : |
B. Com |
|
Date of Appointment : |
15.11.1984 |
|
DIN No.: |
00012973 |
|
|
|
|
Name : |
Mr. Sumit Jain |
|
Designation : |
Whole-time director |
|
Address : |
18/56, East Park Area, Karol Bagh, New Delhi – 110005, India |
|
Date of Birth/Age : |
07.02.1981 |
|
Qualification : |
Post Graduate Diploma in Business Management |
|
Date of Appointment : |
27.07.2005 |
|
DIN No.: |
00014236 |
|
|
|
|
Name : |
Mr. Raghava Lakshmi Narasimhan |
|
Designation : |
Director |
|
Address : |
Flat No.6, Plot No.858, New No.3, Paneer Selvam Salai, K K nagar, Chennai – 600078, Tamilnadu, India
|
|
Date of Birth/Age : |
01.10.1940 |
|
Date of Appointment : |
31.01.2001 |
|
DIN No.: |
00073873 |
|
|
|
|
Name : |
Mr. Namdeo Narayan Khamitkar |
|
Designation : |
Director |
|
Address : |
3, Krishna Kunj Apartments, 42 Shantisheela Society, Law College Road,
Pune – 411004, Maharashtra, India |
|
Date of Birth/Age : |
02.12.1940 |
|
Date of Appointment : |
31.01.2006 |
|
DIN No.: |
00017154 |
|
|
|
|
Name : |
Mr. Sunil Kapoor |
|
Designation : |
Director |
|
Address : |
W-155, Greater Kailash, Part – II, New Delhi – 110045, India |
|
Date of Birth/Age : |
25.01.1957 |
|
Date of Appointment : |
31.01.2001 |
|
DIN No.: |
00029133 |
|
|
|
|
Name : |
Mr. Krishna Murari Lal |
|
Designation : |
Director |
|
Address : |
706-A, Sector – 23, Huda, Gurgaon – 122017, Haryana, India |
|
Date of Birth/Age : |
22.06.1940 |
|
Date of Appointment : |
28.04.2006 |
|
DIN No.: |
00016166 |
|
|
|
|
Name : |
Mr. Aditya Narain Saksena |
|
Designation : |
Director |
|
Address : |
Flat No. B-9012, B-Block, Guar Green City, Indirapuram, Ghaziabad –
201010, Uttar Pradesh, India |
|
Date of Birth/Age : |
07.05.1938 |
|
Date of Appointment : |
22.12.2005 |
|
DIN No.: |
00016107 |
KEY EXECUTIVES
|
Name : |
Mr. Vinod Goel |
|
Designation : |
Secretary and General Manager Legal |
|
Address : |
House No.173, Sector - 4, Vaishali, Ghaziabad – 201010, Uttar Pradesh,
India |
|
Date of Birth/Age : |
08.02.1965 |
|
Date of Appointment : |
13.01.1999 |
|
PAN No.: |
AHBPG9784H |
|
|
|
|
Name : |
Mr. Partha Sarathi De |
|
Designation : |
Chief Financial Officer and Head IT and BPR |
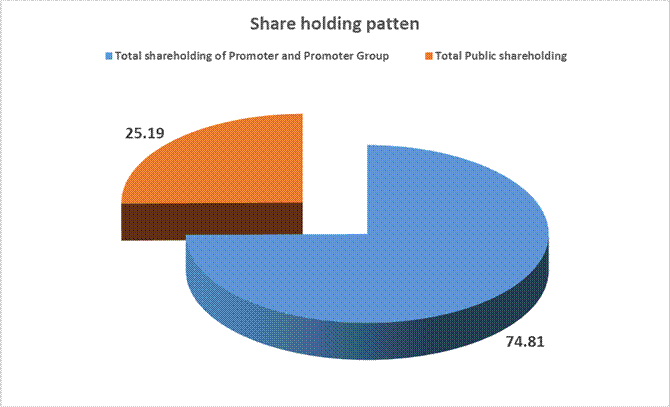
SHAREHOLDING PATTERN
AS ON 30.09.2014
|
Category
of Shareholders |
No. of Shares |
Percentage of Holding |
|
(A) Shareholding of Promoter and Promoter Group |
|
|
|
|
|
|
|
|
43509100 |
71.03 |
|
|
2313454 |
3.78 |
|
|
2313454 |
3.78 |
|
|
45822554 |
74.81 |
|
|
|
|
|
Total shareholding of Promoter and Promoter Group (A) |
45822554 |
74.81 |
|
(B) Public Shareholding |
|
|
|
|
|
|
|
|
469659 |
0.77 |
|
|
30808 |
0.05 |
|
|
844540 |
1.38 |
|
|
1345007 |
2.20 |
|
|
|
|
|
|
10084653 |
16.46 |
|
|
|
|
|
Individual shareholders holding nominal share capital up
to Rs.0.100 Million |
3032839 |
4.95 |
|
Individual shareholders holding nominal share capital in
excess of Rs.0.100 Million |
179572 |
0.29 |
|
|
786121 |
1.28 |
|
|
121387 |
0.20 |
|
|
74362 |
0.12 |
|
|
87917 |
0.14 |
|
|
500000 |
0.82 |
|
|
2455 |
0.00 |
|
|
14083185 |
22.99 |
|
Total Public shareholding (B) |
15428192 |
25.19 |
|
Total (A)+(B) |
61250746 |
100.00 |
|
(C) Shares held by Custodians and against which Depository
Receipts have been issued |
0 |
0.00 |
|
|
0 |
0.00 |
|
|
0 |
0.00 |
|
|
0 |
0.00 |
|
Total (A)+(B)+(C) |
61250746 |
0.00 |
BUSINESS DETAILS
|
Line of Business : |
Manufacturer and Marketer of Pharmaceutical Formulations
viz. Tablets, Syrups/ Liquids, Capsules, Gels and Vaccines. |
||||||||
|
|
|
||||||||
|
Products : |
|
||||||||
|
|
|
||||||||
|
Brand Names : |
-- |
||||||||
|
|
|
||||||||
|
Agencies Held : |
-- |
||||||||
|
|
|
||||||||
|
Exports : |
-- |
||||||||
|
|
|
||||||||
|
Imports : |
-- |
||||||||
|
|
|
||||||||
|
Terms : |
|
||||||||
|
Selling : |
-- |
||||||||
|
|
|
||||||||
|
Purchasing : |
-- |
GENERAL INFORMATION
|
Suppliers : |
-- |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Customers : |
-- |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
No. of Employees : |
Not Available [We tried to
confirm the number of employees but no one is ready to part any information from
the company management] |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Bankers : |
·
Axis Bank Limited ·
Bank of India ·
IDBI Bank Limited ·
Indian Overseas Bank ·
State Bank of Mysore ·
State Bank of Travancore ·
Union Bank of India ·
Canara Bank ·
State Bank of India, Industrial Finance Branch,
14th Floor, J. V. Building, 1, Tolstoy Marg, New Delhi – 110001,
India |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Facilities : |
|
|
|
|
|
Banking
Relations : |
-- |
|
|
|
|
Statutory Auditors : |
|
|
Name: |
S. R. Batiliboi and Associates Chartered Accountants |
|
Address: |
1st Floor, Tower A. Building No.8, DLF Cyber city, Phase III, Sector 25, Gurgoan-122001, Haryana, India |
|
PAN No.: |
AALFS0506L |
|
|
|
|
Cost Auditors : |
|
|
Name: |
J. P. Gupta and Associates Cost Accountants |
|
Address: |
|
|
|
|
|
Membership : |
-- |
|
|
|
|
Joint Ventures : |
·
Chiron Panacea Vaccines Private Limited ·
Adveta Power Private Limited |
|
|
|
|
Associates : |
·
PanEra Biotec
Private Limited |
|
|
|
|
Subsidiaries : |
·
Radhika Heights Limited (“RHL”)
(Formerly Best On Health Limited (Wholly-owned
subsidiary (WOS)) ·
Radicura lnfa Limited (formerly
Radicura & Co. Limited, (Indirect WOS (IWOS) through RHL), ·
Nirmala Buildwell Private Limited
(formerly Panacea Hospitality Services Private Limited. (IWOS through RHL) ·
Cabana Construction Private. Limited
(formerly Panacea Educational Institute Private Limited (IWOS through RHL) ·
Sunanda Infra Limited (formerly
Sunanda Steel Company Limited (IWOS through RHL) ·
Nirmala Organic Farms & Resorts
Private Limited (IWOS through RHL) ·
Cabana Structures Limited (formerly
Best On Health Foods Limited (IWOS through RHL) ·
Rees investments Limited
("Rees") (Guernsey): (WOS) ·
Kelisia Holdings Limited
("KHL") (Cyprus) (IWOS through Rees) ·
Kelisia Investment Holding AG
("KIH') (Switzerland) (IWOS through KHL) (under liquidation) ·
Panacea Biotec (International) SA
(“PBS”) (Switzerland) (WOS) ·
Panacea Biotec Germany GmbH (Germany)
(Indirect WOS through PBS) ·
Panacea Biotec GmbH (Germany) (WOS)
(under liquidation) ·
Panacea Biotec FZE, (UAE) (WOS) (liquidated on June
18,2013) ·
NewRise Healthcare Private Limited
(Formerly Umkal Medical Institute Private Limited: (Subsidiary) ·
Lakshmi & Manager Holdings
Limited ("LMH") WOS* ·
Trinidhi Finance Private Limited
("Trinidhi") (IWOS through LMH)* ·
Best General Insurance Company
Limited. ('Best General") (indirect subsidiary through LMH))' The
shares held in LMH have been sold to related parties during the year and LMH
and its subsidiaries (Trinidhi and Best General) cease to be Company's
subsidiary company w.e.f. 25.1.2014. |
|
|
|
|
Enterprises over which person(s) having control or
significant influence over the Company / Key management personnel), along
with their relatives, are able to exercise significant influence: |
· Neophar Alipro Limited · First Lucre Partnership Co.* · LMH, Trinidhi and Best General with effect from 25.01.2014 *This enterprise is also holding shares in the company. |
CAPITAL STRUCTURE
AS ON 31.03.2014
Authorised Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
125000000 |
Equity Shares |
Re.1/- Each |
Rs.125.000 millions |
|
110000000 |
Preference Shares |
Rs.10/- Each |
Rs.1100.000 millions |
|
|
Total |
|
Rs.1225.000 millions |
Issued, Subscribed & Paid-up Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
61250746 |
Equity Shares |
Re.1/- Each |
Rs.61.300
millions |
|
|
|
|
|
a) Terms/right attached to equity shares:
The Company has only one class of equity shares having a par value of Re.1 per share. Each holder of equity shares is entitled to one vote per share. The Company declares and pays dividends in Indian Rupees. The dividend if any, proposed by the Board of Directors is subject to the approval of the shareholders in the ensuing Annual General Meeting.
The Company has not declared any dividend for current year and previous year. In the event of liquidation of the Company, the holders of equity shares will be entitled to receive remaining assets of the Company, after distribution of all preferential amounts.
The distribution will be in proportion to the number of equity shares held by the shareholders.
b) Reconciliation of the shares outstanding at
the beginning and at the end of the reporting Financial year:
|
Particulars
|
No.
of shares |
Rs.
In Millions |
|
|
|
|
|
At the beginning of the year |
61,250,746 |
61.300 |
|
Movement during the year |
-- |
-- |
|
Outstanding at the end of the year |
61,250,746 |
61.300 |
c) Aggregate number of equity shares bought back
during the period of five years immediately preceding the reporting date:
|
Name of Persons |
No.
of shares |
|
Equity shares bought back by the Company during the financial year
2010-11 |
5592000 |
d) Detail of shareholders holding more than 5%
shares in the Company:
|
Name of Persons |
No. of shares |
% age of holding |
|
|
|
|
|
Mr. Soshil Kumar Jain |
5000000 |
8.16 |
|
Mr. Ravinder Jain |
5712300 |
9.33 |
|
Dr. Rajesh Jain |
6213500 |
10.14 |
|
Mr. Sandeep Jain |
4792100 |
7.82 |
|
Ravinder Jain (HUF) |
4135000 |
6.75 |
|
Rajesh Jain (HUF) |
4368500 |
7.13 |
|
Sandeep Jain (HUF) |
4105000 |
6.70 |
|
Serum Institute of India Limited |
8932632 |
14.58 |
FINANCIAL DATA
[all figures are
in Rupees Millions]
ABRIDGED BALANCE
SHEET
|
SOURCES OF FUNDS |
31.03.2014 |
31.03.2013 |
31.03.2012 |
|
I.
EQUITY
AND LIABILITIES |
|
|
|
|
(1)Shareholders' Funds |
|
|
|
|
(a) Share Capital |
61.300 |
61.300 |
61.300 |
|
(b) Reserves & Surplus |
5561.600 |
5551.300 |
8079.600 |
|
(c) Money
received against share warrants |
0.000 |
0.000 |
0.000 |
|
|
|
|
|
|
(2) Share Application money pending
allotment |
0.000 |
0.000 |
0.000 |
|
Total
Shareholders’ Funds (1) + (2) |
5622.900 |
5612.600 |
8140.900 |
|
|
|
|
|
|
(3)
Non-Current Liabilities |
|
|
|
|
(a) long-term borrowings |
4801.200 |
6072.000 |
4830.300 |
|
(b) Deferred tax liabilities (Net) |
0.000 |
0.000 |
205.000 |
|
(c) Other long term
liabilities |
20.200 |
23.600 |
23.800 |
|
(d) long-term
provisions |
17.300 |
25.200 |
20.600 |
|
Total Non-current
Liabilities (3) |
4838.700 |
6120.800 |
5079.700 |
|
|
|
|
|
|
(4)
Current Liabilities |
|
|
|
|
(a) Short
term borrowings |
3544.100 |
2991.400 |
2693.400 |
|
(b) Trade
payables |
2262.600 |
1539.900 |
1181.400 |
|
(c) Other
current liabilities |
2481.200 |
434.700 |
1339.800 |
|
(d) Short-term
provisions |
115.100 |
98.800 |
93.600 |
|
Total Current
Liabilities (4) |
8403.000 |
5064.800 |
5308.200 |
|
|
|
|
|
|
TOTAL |
18864.600 |
16798.200 |
18528.800 |
|
|
|
|
|
|
II.
ASSETS |
|
|
|
|
(1)
Non-current assets |
|
|
|
|
(a) Fixed
Assets |
|
|
|
|
(i)
Tangible assets |
10550.900 |
9169.800 |
9543.400 |
|
(ii)
Intangible Assets |
308.600 |
418.600 |
469.700 |
|
(iii)
Capital work-in-progress |
137.200 |
169.400 |
120.700 |
|
(iv)
Intangible assets under development |
123.600 |
106.300 |
349.900 |
|
(b) Non-current Investments |
3942.000 |
2653.500 |
2584.700 |
|
(c) Deferred tax assets (net) |
0.000 |
0.000 |
0.000 |
|
(d) Long-term Loan and Advances |
807.600 |
1026.800 |
1063.100 |
|
(e) Other
Non-current assets |
1.800 |
0.000 |
1.200 |
|
Total Non-Current
Assets |
15871.700 |
13544.400 |
14132.700 |
|
|
|
|
|
|
(2)
Current assets |
|
|
|
|
(a)
Current investments |
0.000 |
0.000 |
0.000 |
|
(b)
Inventories |
1767.600 |
2247.400 |
3397.300 |
|
(c) Trade
receivables |
905.800 |
687.500 |
664.500 |
|
(d) Cash
and cash equivalents |
96.800 |
125.900 |
113.300 |
|
(e)
Short-term loans and advances |
183.800 |
163.500 |
170.300 |
|
(f) Other
current assets |
38.900 |
29.500 |
50.700 |
|
Total
Current Assets |
2992.900 |
3253.800 |
4396.100 |
|
|
|
|
|
|
TOTAL |
18864.600 |
16798.200 |
18528.800 |
PROFIT & LOSS
ACCOUNT
|
|
PARTICULARS |
31.03.2014 |
31.03.2013 |
31.03.2012 |
|
|
|
SALES |
|
|
|
|
|
|
|
Income |
5030.300 |
5959.200 |
7005.800 |
|
|
|
Other Income |
116.400 |
54.300 |
74.600 |
|
|
|
TOTAL (A) |
5146.700 |
6013.500 |
7080.400 |
|
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
|
|
Cost of Materials Consumed |
1965.800 |
2363.700 |
3021.200 |
|
|
|
Purchases of Stock-in-Trade |
223.900 |
232.600 |
245.200 |
|
|
|
Changes in inventories of finished goods, work-in-progress
and Stock-in-Trade |
(108.900) |
2.900 |
205.600 |
|
|
|
Employees benefits expense |
1427.300 |
1372.300 |
1504.500 |
|
|
|
Other expenses |
2444.500 |
2828.600 |
2968.400 |
|
|
|
Exceptional items |
(2970.200) |
(173.100) |
0.000 |
|
|
|
TOTAL (B) |
2982.400 |
6800.100 |
7944.900 |
|
|
|
|
|
|
|
|
Less |
PROFIT
BEFORE INTEREST, TAX, DEPRECIATION AND AMORTISATION (A-B) (C) |
2164.300 |
(786.600) |
(864.500) |
|
|
|
|
|
|
|
|
|
Less |
FINANCIAL
EXPENSES (D) |
1480.500 |
1057.400 |
1011.100 |
|
|
|
|
|
|
|
|
|
|
PROFIT
BEFORE TAX, DEPRECIATION AND AMORTISATION (C-D) (E) |
683.800 |
(1844.000) |
(1875.600) |
|
|
|
|
|
|
|
|
|
Less |
DEPRECIATION/
AMORTISATION (F) |
688.000 |
835.400 |
753.900 |
|
|
|
|
|
|
|
|
|
|
PROFIT/ (LOSS)
BEFORE TAX (E-F) (G) |
(4.200) |
(2506.300) |
(2629.500) |
|
|
|
|
|
|
|
|
|
Less |
TAX (H) |
0.000 |
205.000 |
551.600 |
|
|
|
|
|
|
|
|
|
|
PROFIT/ (LOSS)
AFTER TAX (G-H) (I) |
(4.200) |
(2301.300) |
(2077.900) |
|
|
|
|
|
|
|
|
|
|
PREVIOUS
YEARS’ BALANCE BROUGHT FORWARD |
(359.700) |
1941.600 |
4019.500 |
|
|
|
|
|
|
|
|
|
|
BALANCE CARRIED
TO THE B/S |
(363.900) |
(359.700) |
1941.600 |
|
|
|
|
|
|
|
|
|
|
EARNINGS IN
FOREIGN CURRENCY |
|
|
|
|
|
|
|
F.O.B. value of exports |
1263.800 |
905.200 |
3436.500 |
|
|
|
Income from distribution rights |
2.300 |
2.100 |
0.500 |
|
|
|
Research and license fees income |
178.500 |
299.400 |
0.000 |
|
|
|
Interest income from subsidiary company |
94.600 |
0.000 |
36.200 |
|
|
TOTAL EARNINGS |
1539.200 |
1206.700 |
3473.200 |
|
|
|
|
|
|
|
|
|
|
IMPORTS |
|
|
|
|
|
|
|
Raw Materials and Packing Materials |
490.100 |
310.900 |
2184.000 |
|
|
|
Capital Goods |
34.700 |
74.300 |
226.900 |
|
|
TOTAL IMPORTS |
524.800 |
385.200 |
2410.900 |
|
|
|
|
|
|
|
|
|
|
Earnings/ (Loss)
Per Share (Rs.) |
(0.07) |
(37.57) |
(33.92) |
|
QUARTERLY RESULTS
|
PARTICULARS |
|
30.06.2014 (Unaudited) |
30.09.2014 (Unaudited) |
|
|
|
1st Quarter |
2nd Quarter |
|
Net Sales |
|
1509.100 |
1585.800 |
|
Total Expenditure |
|
1199.300 |
1448.700 |
|
PBIDT (Excl OI) |
|
309.800 |
137.100 |
|
Other Income |
|
03.100 |
293.000 |
|
Operating Profit |
|
312.900 |
430.100 |
|
Interest |
|
393.900 |
342.800 |
|
Exceptional Items |
|
0.000 |
0.000 |
|
PBDT |
|
(81.000) |
87.300 |
|
Depreciation |
|
154.400 |
155.500 |
|
Profit Before Tax |
|
(235.400) |
(68.200) |
|
Tax |
|
0.000 |
0.000 |
|
Provisions and contingencies |
|
0.000 |
0.000 |
|
Profit After Tax |
|
(235.400) |
(68.200) |
KEY RATIOS
|
PARTICULARS |
|
31.03.2014 |
31.03.2013 |
31.03.2012 |
|
Net Profit Margin PAT / Sales |
(%) |
(0.08) |
(38.62) |
(29.66) |
|
|
|
|
|
|
|
Operating Profit Margin (PBDIT/Sales) |
(%) |
43.03 |
(13.20) |
(12.34) |
|
|
|
|
|
|
|
Return on Total Assets (PBT/Total Assets} |
(%) |
(0.03) |
(18.07) |
(16.99) |
|
|
|
|
|
|
|
Return on Investment (ROI) (PBT/Networth) |
|
0.00 |
(0.45) |
(0.32) |
|
|
|
|
|
|
|
Debt Equity Ratio (Total Debt /Networth) |
|
1.48 |
1.61 |
0.92 |
|
|
|
|
|
|
|
Current Ratio (Current Asset/Current Liability) |
|
0.36 |
0.64 |
0.83 |
FINANCIAL ANALYSIS
[all figures are
in Rupees Millions]
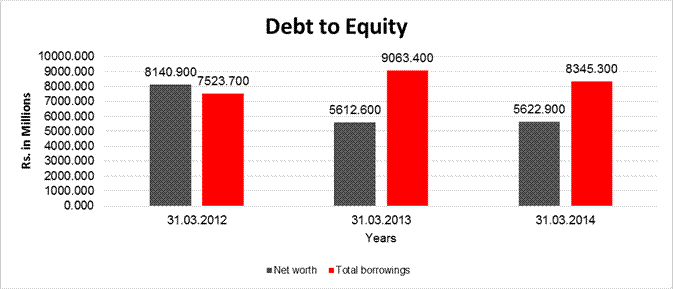
DEBT EQUITY RATIO
|
Particular |
31.03.2012 |
31.03.2013 |
31.03.2014 |
|
|
Rs.
In Millions |
Rs.
In Millions |
Rs.
In Millions |
|
Share Capital |
61.300 |
61.300 |
61.300 |
|
Reserves & Surplus |
8079.600 |
5551.300 |
5561.600 |
|
Net
worth |
8140.900 |
5612.600 |
5622.900 |
|
|
|
|
|
|
long-term borrowings |
4830.300 |
6072.000 |
4801.200 |
|
Short term borrowings |
2693.400 |
2991.400 |
3544.100 |
|
Total
borrowings |
7523.700 |
9063.400 |
8345.300 |
|
Debt/Equity
ratio |
0.924 |
1.615 |
1.484 |
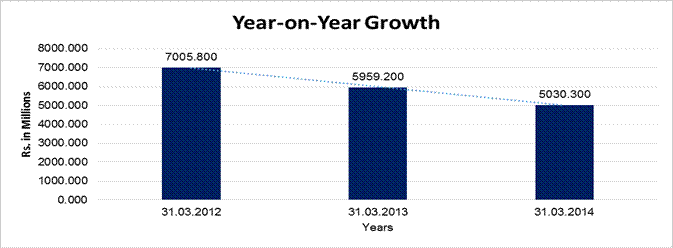
YEAR-ON-YEAR GROWTH
|
Year
on Year Growth |
31.03.2012 |
31.03.2013 |
31.03.2014 |
|
|
Rs.
In Millions |
Rs.
In Millions |
Rs.
In Millions |
|
Sales |
7005.800 |
5959.200 |
5030.300 |
|
|
|
(14.939) |
(15.588) |
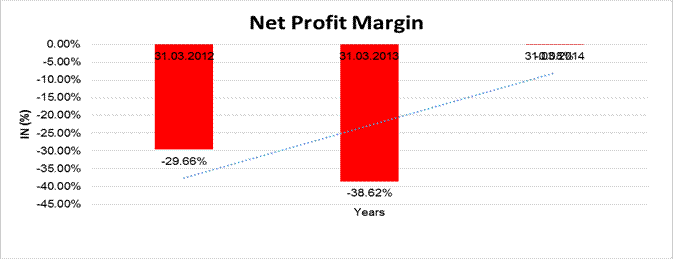
NET PROFIT MARGIN
|
Net
Profit Margin |
31.03.2012 |
31.03.2013 |
31.03.2014 |
|
|
Rs.
In Millions |
Rs.
In Millions |
Rs.
In Millions |
|
Sales
|
7005.800 |
5959.200 |
5030.300 |
|
Profit |
(2077.900) |
(2301.300) |
(4.200) |
|
|
(29.66%) |
(38.62%) |
(0.08%) |
LOCAL AGENCY FURTHER INFORMATION
|
Sr. No. |
Check List by Info Agents |
Available in
Report (Yes / No) |
|
1] |
Year of Establishment |
Yes |
|
2] |
Locality of the firm |
Yes |
|
3] |
Constitutions of the firm |
Yes |
|
4] |
Premises details |
No |
|
5] |
Type of Business |
Yes |
|
6] |
Line of Business |
Yes |
|
7] |
Promoter's background |
Yes |
|
8] |
No. of employees |
No |
|
9] |
Name of person contacted |
No |
|
10] |
Designation of contact
person |
No |
|
11] |
Turnover of firm for last
three years |
Yes |
|
12] |
Profitability for last
three years |
Yes |
|
13] |
Reasons for variation
<> 20% |
-- |
|
14] |
Estimation for coming
financial year |
No |
|
15] |
Capital in the business |
Yes |
|
16] |
Details of sister
concerns |
Yes |
|
17] |
Major suppliers |
No |
|
18] |
Major customers |
No |
|
19] |
Payments terms |
No |
|
20] |
Export / Import details
(if applicable) |
No |
|
21] |
Market information |
-- |
|
22] |
Litigations that the firm
/ promoter involved in |
Yes |
|
23] |
Banking Details |
Yes |
|
24] |
Banking facility details |
Yes |
|
25] |
Conduct of the banking
account |
-- |
|
26] |
Buyer visit details |
-- |
|
27] |
Financials, if provided |
Yes |
|
28] |
Incorporation details, if
applicable |
Yes |
|
29] |
Last accounts filed at
ROC |
Yes |
|
30] |
Major Shareholders, if
available |
No |
|
31] |
Date of Birth of
Proprietor/Partner/Director, if available |
Yes |
|
32] |
PAN of Proprietor/Partner/Director,
if available |
No |
|
33] |
Voter ID No of
Proprietor/Partner/Director, if available |
No |
|
34] |
External Agency Rating,
if available |
Yes |
LITIGATION
DETAILS:
|
High Court of Punjab and Haryana Chandigarh |
|||||
|
Case Details For
Case CP-189-2014 |
|||||
|
Diary Number |
1286448 |
District |
Other-Districts |
||
|
Category |
21-COMPANY
PETITIONS |
Main Case Detail |
-- |
||
|
Party Detail |
SUDHARMA METROPOLIS
HEALTH SERVICES PRIVATE LIMITED V/S PANACEA BIOTECH LIMITED |
||||
|
Advocate Name |
HARISH BHARDWAJ |
List Type |
|
||
|
Status |
PENDING |
Next Date |
09-JAN-2015 |
||
|
|
|||||
|
Case Listing
Details |
|||||
|
Case List Date |
List Type: Se.
No. |
Bench |
Order Link |
||
|
05-dec-2014 |
NA:110 |
HON’BLE MR.
JUSTICE RAJESH BINDAL |
---- |
||
|
Judgment Details
For Case: CP-189-2014 Party Detail:
SUDHARMA METROPOLIS HEALTH SERVICES PRIVATE LIMITED V/S PANACEA BIOTECH
LIMITED |
|||||
|
Order Date |
Order Type |
Bench |
Judgment Link |
||
|
No Judgment/Order Found. |
|||||
|
Designed and Developed by National Informatics
Centre
Contents Published and Managed by Punjab and Haryana High Court,
Chandigarh. Disclaimer |
|||||
NOTE:
The registered office address of the company has been shifted from
“Derabassi, Tehsil Rajpura, Patiala-140 501, Punjab, India” to the present
address w.e.f. 07.06.2011
UNSECURED LOANS:
|
Particulars |
31.03.2014 Rs.
In Millions |
31.03.2013 Rs.
In Millions |
|
Long Term
Borrowings |
|
|
|
Through Department of Science and Technology |
14.000 |
16.000 |
|
|
|
|
|
Short Term
Borrowings |
|
|
|
Deposits from public |
7.000 |
339.600 |
|
Deposits from related parties |
133.200 |
0.000 |
|
Loan from related parties |
627.600 |
362.400 |
|
|
|
|
|
Total |
781.800 |
718.000 |
OPERATING RESULTS AND PROFITS
During the year ended
March 31, 2013, subject registered a net turnover of Rs.5304.200 million as
against Rs.6883.800 million during the corresponding financial year.
The formulations
segment registered a growth of 8.4% with a net turnover of Rs.3581.300 million
as against Rs.3304.300 million during the previous financial year. The vaccines
segment registered a net turnover of Rs.1722.900 million as against Rs.3579.500
million during the previous financial year, a decline of 52% mainly on account
of delisting of its vaccines from the WHO’s list of prequalified vaccines for
supply to UNICEF and other UN Agencies in previous financial year, impacting
the performance negatively in the current financial year.
Since then, subject
has taken several corrective and preventive measures to ensure compliance with
the WHO pre-qualification guidelines. During the period, the auditors from WHO
and UNICEF visited the vaccine facilities at Lalru (Punjab) and Baddi (H.P.) in
Feb-Mar 2013 with the objective of re-evaluation of the acceptability in
principle of combination vaccine (DTP-Hep B-Hib) produced by Panacea Biotec for
purchase by United Nations Agencies. There were no critical observations and
the Audit Team acknowledged the continuing improvements that have been made in
this regard. Subject is confident that with the post audit activities, it will
be able to get re-listing of Pentavalent vaccine in the list of WHO
pre-qualified vaccines in due course of time.
Subject however,
continues to focus on cost optimization and efficient management of working
capital. Panacea Biotec is constantly striving to enhance its reputation as one
of the India’s leading research-based health management companies with
established research, manufacturing and marketing capabilities and is confident
of overcoming the current adverse situation.
UNAUDITED FINANCIAL RESULTS FOR THE QUARTER
/ HALF YEAR ENDED DECEMBER 30.09.2014
(Rs.
In Millions)
|
Particulars |
Quarter Ended ( Unaudited) |
Half Year Ended ( Unaudited) |
|
|
|
30.09.2014 |
30.06.2014 |
30.09.2014 |
|
PART – I |
|
|
|
|
1. Income from
operations |
|
|
|
|
a. Net sales / income from operations (net of excise duty) |
1556.400 |
1315.000 |
2871.400 |
|
b. Other operating income |
29.400 |
194.100 |
223.500 |
|
Total income from
operations (net) |
1585.800 |
1509.100 |
3094.900 |
|
2. Expenditure |
|
|
|
|
a. Cost of materials consumed |
551.200 |
306.300 |
857.500 |
|
b. Purchase of stock in trade |
63.000 |
54.700 |
117.700 |
|
c. (Increase)/Decrease in inventories |
37.100 |
112.800 |
149.900 |
|
d. Employees benefits expense |
334.000 |
332.400 |
666.400 |
|
e. Depreciation and amortization expense |
155.500 |
154.400 |
309.900 |
|
g. Other expenses |
463.400 |
393.100 |
856.500 |
|
Total expenses |
1604.200 |
1353.700 |
2957.900 |
|
3. (Loss)/Profit from operations before other income, finance cost, foreign exchange fluctuation gain/ (loss) & exceptional items (1-2) |
(18.400) |
155.400 |
137.000 |
|
4. Other income |
54.000 |
3.100 |
57.100 |
|
5. (Loss)/Profit from ordinary activities before finance cost, foreign exchange fluctuation gain / (loss) & exceptional items (3+4) |
35.600 |
158.500 |
194.100 |
|
6. Finance cost |
103.800 |
393.900 |
497.700 |
|
7. Foreign exchange fluctuation loss/ (gain) |
0.000 |
0.000 |
0.000 |
|
8. (Loss)/Profit from
ordinary activities after finance cost before exceptional items (5-6-7) |
(68.200) |
(235.400) |
(303.600) |
|
9. Exceptional income/(exp) - Refer note 7(i) & (ii) |
-- |
-- |
-- |
|
10. Profit / (loss)
from ordinary activities before tax (8+9) |
(68.200) |
(235.400) |
(303.600) |
|
11. Tax expenses |
-- |
-- |
-- |
|
12. Net profit /
(loss) from ordinary activities after tax (10-11) |
(68.200) |
(235.400) |
(303.600) |
|
13. Extraordinary items (net of tax expenses) |
-- |
-- |
-- |
|
14. Net profit /
(loss) for the period (12-13) |
(68.200) |
(235.400) |
(303.600) |
|
15. Paid up equity share capital (face value of Re.1 per share) |
61.300 |
61.300 |
61.300 |
|
16.Reserves excluding revaluation reserves |
|
|
|
|
17.Earning per share
(EPS) |
|
|
|
|
- Basic (in Rs.) |
(1.11) |
(3.84) |
(4.95) |
|
- Diluted (in Rs.) |
(1.11) |
(3.84) |
(4.95) |
|
A. Particulars of
shareholding |
|
|
|
|
1. Public
shareholding |
|
|
|
|
- No. of shares |
15428192 |
15428192 |
15428192 |
|
- Percentage of shareholding |
25.19 |
25.19 |
25.19 |
|
2. Promoters and
promoter group Shareholding |
|
|
|
|
a) Pledge /
encumbered |
|
|
|
|
- No. of shares |
- |
- |
- |
|
- % of Shares (as a % of the total shareholding of promoter & promot |
- |
- |
- |
|
- % of Shares (as a % of the total share capital of the Company) |
- |
- |
- |
|
b) Non-encumbered |
|
|
|
|
- Number of shares |
45822554 |
45822554 |
45822554 |
|
- % of Shares (as a % of the total shareholding of promoter & promoter group) |
100.00 |
100.00 |
100.00 |
|
- % of Shares (as a % of the total share capital of the Company) |
74.81 |
74.81 |
74.81 |
|
B. Investor
complaints |
|
|
Pending at the beginning of the quarter |
- |
|
Received during the quarter |
2 |
|
Disposed of during the quarter |
2 |
|
Remaining unresolved at the end of the quarter |
- |
UNAUDITED FINANCIAL RESULTS FOR THE QUARTER
/ HALF YEAR ENDED DECEMBER 30.09.2014
|
Particulars |
30.09.2014 (Unaudited) |
30.06.2014 (Unaudited) |
30.09.2014 (Unaudited) |
|
|
|
|
|
|
1. Segment revenue |
|
|
|
|
(a) Vaccines |
471.400 |
522.000 |
993.400 |
|
(b) Formulations |
1107.600 |
832.600 |
1940.200 |
|
(c) Research & development |
7.100 |
154.200 |
161.300 |
|
(d) Unallocated |
(0.300) |
0.300 |
0.000 |
|
Gross sale/Income
from operation |
1585.800 |
1509.100 |
3094.900 |
|
Less : Inter segment revenue |
-- |
-- |
-- |
|
Net sales/income
from operations |
1585.800 |
1509.100 |
3094.900 |
|
2.Segment results |
|
|
|
|
Profit (+)/ loss (-)
before tax and interest from each segment |
|
|
|
|
(a) Vaccines |
(111.400) |
19.000 |
(92.400) |
|
(b) Formulations |
450.000 |
252.900 |
702.900 |
|
(c) Research & development |
(145.300) |
20.300 |
(125.000) |
|
Total |
193.300 |
292.200 |
485.500 |
|
Less : i) Finance cost |
104.300 |
393.400 |
497.700 |
|
ii) Other un-allocated expenditure net off un-allocated income |
157.200 |
134.200 |
291.400 |
|
Total profit before
tax |
(68.200) |
(235.400) |
(303.600) |
|
3. Capital Employed |
|
|
|
|
(Segment
assets-segment liabilities) |
|
|
|
|
(a) Vaccines |
5776.800 |
5707.500 |
5776.800 |
|
(b) Formulations |
2427.200 |
2317.700 |
2427.200 |
|
(c) Research & development |
1899.600 |
1837.800 |
1899.600 |
|
(d) Unallocated |
(4823.300) |
(4480.700) |
(4823.300) |
|
Total capital
employed |
5280.300 |
5382.300 |
5280.300 |
STANDALONE
STATEMENT OF ASSETS AND LIABILITIES
(Rs. In Millions)
|
SOURCES OF FUNDS |
30.09.2014 (Unaudited) |
|
I.
EQUITY
AND LIABILITIES |
|
|
(1)Shareholders' Funds |
|
|
(a) Share Capital |
61.300 |
|
(b) Reserves & Surplus |
5219.000 |
|
Total
Shareholders’ Funds |
5280.300 |
|
|
|
|
(2)
Non-Current Liabilities |
|
|
(a) long-term borrowings |
4086.600 |
|
(b) Deferred tax liabilities (Net) |
0.000 |
|
(c) Other long term
liabilities |
120.200 |
|
(d) long-term
provisions |
12.100 |
|
Total Non-current
Liabilities (3) |
4218.900 |
|
|
|
|
(3)
Current Liabilities |
|
|
(a) Short
term borrowings |
3574.500 |
|
(b) Trade
payables |
2073.800 |
|
(c) Other
current liabilities |
3693.000 |
|
(d) Short-term
provisions |
17.500 |
|
Total Current
Liabilities (4) |
9358.800 |
|
|
|
|
TOTAL |
18858.000 |
|
|
|
|
II.
ASSETS |
|
|
(1)
Non-current assets |
|
|
(a) Fixed
Assets |
|
|
1)
Tangible
Assets |
10306.800 |
|
2)
Intangible
Assets |
253.100 |
|
3)
Capital
work-in-progress |
139.900 |
|
4)
Intangible
assets under development |
124.200 |
|
(b) Non-current Investments |
3946.000 |
|
(c) Deferred tax assets (net) |
0.000 |
|
(d) Long-term Loan and Advances |
906.500 |
|
(e) Other
Non-current assets |
1.000 |
|
Total Non-Current
Assets |
15677.500 |
|
|
|
|
(2)
Current assets |
|
|
(a)
Current investments |
0.000 |
|
(b)
Inventories |
1619.800 |
|
(c) Trade
receivables |
1085.300 |
|
(d) Cash
and cash equivalents |
332.200 |
|
(e)
Short-term loans and advances |
112.500 |
|
(f) Other
current assets |
30.700 |
|
Total
Current Assets |
3180.500 |
|
|
|
|
TOTAL |
18858.000 |
NOTES:
1.
The above financial results were reviewed
by the Audit Committee of the Board and approved by the Board of Directors at
their meetings held on October 30, 2014 and October 31, 2014 respectively.
2.
During the quarter, the Company has
launched two new products, namely Calcom Plus and RF Willgo (both for pain
management in Orthopaedic segment).
3.
The Company's Corporate Debt
Restructuring ('CDR') proposal has been approved in the CDR Empowered Group
(’CDR EG') meeting held on 9 September 2014 and letter of approval was received
by the Company on 24 September 2014. Final implementation of the CDR package is
subject to execution of Master Restructuring Agreement, which is required to be
executed within 120 days from the approval date.
4.
As regards Auditor's observations in
their limited review report on the above results:\
a. The Company has received research and development ('R&D') fee of INR
1,499 lacs from a customer and this has been accounted for as income in the
quarter ended 30 June 2014. Such R&D fee is non-refundable subject to
certain pre-conditions being met by the Company. As this product is already in
the domestic market, the Company is reasonably certain of meeting the
pre-conditions set out in the agreement and therefore the said fee has been
accounted for according])’.
b. During the six month ended 30 September 2014, the Company has incurred
losses of Rs. 3,036 lacs (Previous year Rs. 41.6 lacs after adjusting
exceptional income of Rs. 29,744 lacs). Further, the Company's accumulated
losses have resulted in erosion of more than fifty percent of its peak net
worth calculated as per the provisions of Sick Industrial Companies (Special
Provisions) Act, 1985 (SICA). The fact of such erosion and measures initiated
to improve financial condition have already been reported to the Board for Industrial
and Financial Restructuring (“BIFR”) within the stipulated period. These
continuous losses have also adversely affected the cash flows of the Company.
These conditions indicate the existence of a material uncertainty that may cast
significant doubt about the Company's ability to continue as going concern.
The
Company has undertaken several measures to mitigate the risk of going concern
which include supply to UNICEF/other customers of pentavalent vaccine, certain
strategic alliances with foreign collaborators for supply of vaccines and
pharma products; CDR proposal which has already been approved by CDR EG (as
explanned note 3 above); launch of its first product "[acrolimus' in USA
in December 2012 and filing of more ANDAs with USFDA. The management is
confident that with the above measures and continuous efforts to improve the
business, it would be able to generate sustainable cash flow, recover and
regroup the erosion in its net worth through profitable operations, discharge
its short-term and long term liabilities and continue as a going concern.
c. In view of absence of profits during financial year 2013-14 and 2012-13,
total remuneration to the Managing/Joint Managing and Whole time Director had exceeded
the ceiling prescribed in Section II of Part II of Schedule XIII to the
Companies Act, 1956 by Rs. 135 lacs and Rs. 132 lacs respectively. Accordingly,
applications for protection/approval of the Central Government of such excess
remuneration have been filed and requisite approvals arc awaited.
5.
Finance cost for the quarter and six
months ended 30 September 2014 are stated net of reversals of excess accrual of
interest of Rs. 2,390 lacs.
6.
The necessary certificate in respect of
the above results in terms of requirement of clause 41 of the listing
agreement, has been placed before the Board of Directors.
7.
Previous period / year figures have
been regrouped/ reclassified to make them comparable with those of current
Quarter.
INDEX OF CHARGE:
|
Sr. No. |
Charge ID |
Date of Charge
Creation/Modification |
Charge amount
secured |
Charge Holder |
Address |
Service Request
Number (SRN) |
|
1 |
10404560 |
24/12/2012 |
269,300,000.00 |
STATE BANK OF INDIA |
INDUSTRIAL FINANCE BRANCH, 14TH FLOOR, J V BUILDING, 1, TOLSTOY MARG, NEW DELHI, DELHI - 110001, INDIA |
B68639822 |
|
2 |
10376668 |
28/03/2014 * |
50,000,000.00 |
TECHNOLOGY DEVELOPMENT BOARD |
WING - A, GROUND FLOOR,, VISHWAKARMA BHAWAN, SHAHEED JEET SINGH MARG,, NEW DELHI, DELHI - 110016, INDIA |
C03659463 |
|
3 |
10355025 |
20/04/2012 |
10,990,000.00 |
DEPARTMENT OF BIOTECHNOLOGY |
6-8TH FLOOR, BLOCK NO.2, CGO COMPLEX, LODHI ROAD, NEW DELHI, DELHI - 110003, INDIA |
B39421623 |
|
4 |
10355027 |
20/04/2012 |
32,830,000.00 |
DEPARTMENT OF BIOTECHNOLOGY |
6-8TH FLOOR, BLOCK NO.2, CGO COMPLEX, LODHI ROAD, NEW DELHI, DELHI - 110003, INDIA |
B39421870 |
|
5 |
10330445 |
18/01/2012 * |
1,290,000,000.00 |
BANK OF INDIA |
HEAD OFFICE; STAR HOUSE, C-5, G BLOCK, BANDRA KURLA COMPLEX, MUMBAI, MAHARASHTRA - 400051, INDIA |
B30279731 |
|
6 |
10266434 |
16/01/2012 * |
1,000,000,000.00 |
INDIAN OVERSEAS BANK |
RAJIV
CIRCLE, D-28-29, CONNAUGHT PLACE, NEW DELHI |
B31260458 |
|
7 |
10230748 |
06/07/2010 |
109,400,000.00 |
DEPARTMENT OF BIOTECHNOLOGY |
6-8TH FLOOR, BLOCK NO. 2, CGO COMPLEX, LODHI ROAD, NEW DELHI, DELHI - 110003, INDIA |
A90678137 |
|
8 |
10224547 |
24/12/2012 * |
1,000,000,000.00 |
STATE BANK OF INDIA |
INDUSTRIAL FINANCE BRANCH, 14TH FLOOR, J V BUILDING, 1, TOLSTOY MARG, NEW DELHI, DELHI - 110001, INDIA |
B68640507 |
|
9 |
10089746 |
24/12/2012 * |
2,000,000,000.00 |
STATE BANK OF INDIA |
INDUSTRIAL FINANCE BRANCH, 14TH FLOOR, J V BUILDING, 1, TOLSTOY MARG, NEW DELHI, DELHI - 110001, INDIA |
B68641133 |
|
10 |
10069827 |
08/11/2012 * |
1,096,700,000.00 |
STATE BANK OF TRAVANCORE |
COMMERCIAL BRANCH, TRAVANCORE HOUSE, K.G. MARG, NEW DELHI, DELHI - 110001, INDIA |
B63220693 |
|
11 |
10056735 |
05/10/2009 * |
1,500,000,000.00 |
IDBI BANK LIMITED |
3RD FLOOR, INDIAN RED CROSS SOCIETY BUILDING, 1, RED CROSS ROAD,, NEW DELHI, DELHI - 110001, INDIA |
A72025109 |
|
12 |
80008672 |
24/12/2012 * |
3,621,800,000.00 |
STATE BANK OF INDIA |
INDUSTRIAL FINANCE BRANCH, 14TH FLOOR, J V BUILDING, 1, TOLSTOY MARG, NEW DELHI, DELHI - 110001, INDIA |
B68639061 |
*Date of
modification Charges
FIXED ASSTES
· Land – Leasehold
· Land – Freehold
· Buildings
· Leasehold
Improvements
· Plant and
Machinery
· Vehicles
· Furniture and
Fixtures
· Office Equipments
· Computer
Equipments
PRESS RELEASE:
PANACEA ENTERS INTO ALLIANCE WITH CANADA BASED APOTEX INC
Drug firm Panacea Biotec has entered into a partnership
with Canada based Apotex Inc for research, development, licence and supply of
two drug delivery based generic products to US, Canada, Australia and New
Zealand markets.
In addition to upfront and milestone research fee payments
from Apotex, the company shall receive a share of development cost and profit.
Post commercialisation the profit from the products shall be shared at a
pre-agreed ratio between the two companies, Panacea Biotec said in a filing to
BSE.
The current sales of the innovator products are to the tune
of USD 1 billion, it added.
Commenting on the development, Panacea Biotec Joint MD
Rajesh Jain said: "This strategic alliance with Apotex is one of the
cardinal milestones in our global generics strategy."
As per the agreement Panacea will undertake product
development, manufacture and supply of the products while Apotex will be
responsible for the regulatory affairs and commercialisation activities, the
company said. "The strategic alliance with Panacea Biotec is another example
of Apotex seeking to develop strategic partnerships with carefully selected
companies to enable access to products and technologies where Apotex does not
have its own manufacturing facilities," Apotex Inc Chief Business Officer
Steven Lydeamore said.
Both companies intend to expand the scope of the alliance by
adding new drug delivery based complex generic products to the collaboration,
Panacea said. Shares of Panacea Biotec were today trading at Rs 163.85 per
scrip in the afternoon trade on BSE, up 6.22 percent from it's previous close.
PANACEA BIO UP 13% ON STRATEGIC ALLIANCE WITH CANADIAN CO
Shares of Panacea Biotec climbed nearly 13 percent
intraday Friday on signing strategic alliance with Apotex, the largest
Canadian-owned pharmaceutical company.
The alliance is for research, development, license and
supply of two drugs delivery based high barrier to entry generic products for
US, Canada, Australia and New Zealand.
"Panacea will undertake product development and will
manufacture and supply the products to Apotex, while Apotex will be responsible
for regulatory affairs, Hatch Waxman litigation and commercialisation
activities in the territories," said the company in its filing to the
exchange.
Panacea already had a strategic alliance with Apotex for a
drug delivery based drug, which have now expanded by adding a second drug
delivery. The company said both the products have been developed using
Panacea's proprietary platform nanoparticle, liposomal and microparticle drug delivery
systems. The current sales of the innovator products are to the tune of about
USD 1 billion.
"Coupled with
Apotex's expertise in successfully commercialising such products makes this a
highly synergistic collaboration," said Rajesh Jain, joint managing
director, Panacea Biotec.
In addition to an upfront and milestone research fee
payments from Apotex, Panacea will receive a share of the development cost and
profit. This is after the commercialisation of the products will be shared at a
pre-agreed ratio between both companies.
GOVT NOD TO 15 FDI PLANS; CCEA TO CONSIDER HDFC BK PROPOSAL
The Finance Ministry today said it has cleared 15 FDI
applications, including that of Panacea Biotech and Sanofi-Synthelabo
(India), and recommended HDFC Bank 's proposal to hike foreign holding to
the Cabinet for consideration.
The proposals were cleared by Foreign Investment Promotion
Board (FIPB) headed by the Economic Affairs Secretary in its meeting held
earlier this month. The Ministry said HDFC Bank had sought approval for
maintaining the permissible foreign holding in the bank up to 74 per cent of
the total paid up capital, out of which the FII sub-limit would be 49 per cent
and the balance 25 per cent would be foreign direct investment (FDI).
"...(the) proposal has been recommended for the
consideration of CCEA, as the investment involved in the proposals is above Rs
1,200 crore," it said. It further said Panacea Biotech Limited has got the
government's nod for issuance of equity shares to Qualified Institutional
Buyers (QIBs) through proposed qualified institutional placement. Proposal of
the Delhi-based listed pharmaceutical firm entails investment of Rs 250 crore.
The FIPB cleared the Rs 90-crore proposal of
Sanofi-Synthelabo (India), a wholly owned subsidiary of Sanofi S.A., France. It
had sought approval for additional foreign investment of Rs 90 crore from its
parent company and inter-corporate loan from another Indian incorporated Sanofi
Group company to acquire 20 per cent equity in Apollo Sugar Clinics, Mumbai- a
company engaged in providing healthcare clinical services.
A Rs 184-crore investment proposal of GETIT Infoservices too
has been approved. The permission was given to increasing foreign equity
participation in its share capital from present 96.266 per cent to up to 100
per cent.
The FIPB also rejected proposals of Quikjet Cargo Airlines,
Bangalore and Phil Arthur Bedford, Dubai. Meanwhile, the Board deferred
decision on seven proposals, including of Holcim (India) and Augere Wireless
Broadband India Pvt Ltd
PANACEA BIOTEC SHOOTS UP 18% ON LAND DEVELOPMENT BUZZ
Delhi-based Panacea Biotec surged as much as 18 percent
intraday on Wednesday on media reports that the company will develop a township
project in Gurgaon.
The Economic Times reported that Panacea Biotec and builder
Bestech Group will jointly develop a township project over a 100-acre plot
owned by Panacea in Gurgaon, fetching the pharma company over Rs 750 crore in
revenue.
"The land was held by Best On Health (BOH), Panacea's
wholly owned subsidiary, which holds a substantial piece of land on Pataudi
Road, Gurgaon, Haryana. Panacea had initially planned to develop a biotech
special economic zone (SEZ) on this property. Both companies will hold an equal
stake in the project, which is expected to generate revenues of over Rs 1,500
crore," the report said. At 15:17 hours IST, shares rose 13.29 percent to
Rs 136.40 amid large volumes on Bombay Stock Exchange. Trading volumes
jumped 5 times to 2,40,248 equity shares as against five day average of 45,084
shares. Market capitalisation of the company currently stands at Rs 835.46
crore.
PANACEA BIOTEC FORMS RESEARCH ALLIANCE
WITH US-BASED APOTEX
Panacea biotec has entered
into a strategic alliance with ApotexInc for research, development, license and supply of
two drug delivery-based generic products for US, Canada, Australia and New
Zealand.Both companies intend to expand the scope of the alliance by adding new drug delivery based complex generic products to the collaboration.
Panacea Biotec will undertake product
development and will manufacture and supply the products to Apotex, while
Apotex will be responsible for regular affairs, Hatch Waxman litigation and
commercialisation activities in the territories, the company said in a
statement.
Panacea Biotec trades higher on the BSE
Panacea Biotec is currently trading at Rs. 164.30, up by 3.15
points or 1.95 % from its previous closing of Rs. 161.15 on the BSE.
The scrip opened at Rs. 165.00 and has touched a high and low of
Rs. 166.70 and Rs. 162.50 respectively. So far 14783 shares were traded on the
counter.
The BSE group 'B' stock of face value Rs. 1 has touched a 52 week
high of Rs. 218.95 on 22-Sep-2014 and a 52 week low of Rs. 88.60 on
30-Jan-2014.
Last one week high and low of the scrip stood at Rs. 174.10 and
Rs. 152.00 respectively. The current market cap of the company is Rs. 1001.14
crore.
The promoters holding in the company stood at 74.81%, while
Institutions and Non-Institutions held 2.20% and 22.99% respectively.
Panacea Biotec, India's highly progressive research based health
management company has entered into a strategic alliance with Apotex Inc, the
largest Canadian owned Pharmaceutical company for research, development,
license and supply of two drug delivery based high barrier to entry generic
products for US, Canada, Australia and New Zealand.
The strategic alliance was initially established for a drug
delivery based drug for which Panacea Biotec had received research fees and the
product development is progressing on priority. Panacea Biotec and Apotex have
now further expanded the scope of the collaboration by including a second drug
delivery based product to the collaboration. Both the products have been
developed using Panacea Biotec's proprietary platform nanoparticle, liposomal and
microparticle drug delivery systems. The current sales of the innovator
products are to the tune of about $1 billion.
Both companies intend to expand the scope of the alliance by
adding new drug delivery based complex generic products to the collaboration.
Panacea Biotec shall undertake product development and shall manufacture and
supply the products to Apotex, while Apotex shall be responsible for regulatory
affairs, Hatch Waxman litigation and commercialization activities in the
territories.
Panacea Biotec is one of India’s leading research based health
management companies with established research, manufacturing and marketing
capabilities.
DCVMN INTERNATIONAL DEVELOPS MULTIPLE
INITIATIVES TO ACCELERATE ACCESS TO AFFORDABLE HIGH QUALITY-VACCINES
09-December-2014
October 2014 — The Developing Countries
Vaccine Manufacturers Network (DCVMN) International has announced at its annual
gathering in Delhi, India, its intention to support access to affordable
high-quality vaccines by enabling a larger number of vaccine manufacturers to
achieve a more sustainable and secure supply of priority vaccines for
international procurement, particularly for GAVI-eligible countries, with the
common goal of protecting people against known and emerging infectious diseases
globally.
DCVMN, through its members, has been working together with
global health stakeholders since early this year, in identifying needs and
challenges of the vaccine industry in developing countries. Four areas of
action have been agreed to strengthen and foster sustainable vaccine supply:
(1) review of manufacturing facilities design, (2) provide adequate training on
evolving GMP requirements, quality management systems, and the WHO standards
and prequalification, (3) encourage dialogue on regulatory challenges, (4)
facilitate access to independent experts able to resolve vaccine industry
specific issues.The three years’ project costs of over 3.6 million dollars will
be sourced to 60 percent by international global health organizations and the
remaining jointly by DCVMN members and partners. The priority vaccines to be
targeted encompass Pentavalent/Hexavalent, Pneumococcal Conjugate, Rotavirus,
Typhoid Conjugate, Human Papillomavirus, Measles/Rubella and Inactivated Polio vaccines.
“Two out of three children in the world receive lifesaving
vaccines from emerging manufacturers, and as the world's population is growing
at the fastest rate in developing countries, it is important to ensure improved
manufacturing in every facility we can reach” said Mr. Mahendra Suhardono,
President of DCVMN.
All members involved in funding these initiatives share the
vision of developing countries free of suffering and disabilities from major
infectious diseases, and will work together to foster the development and
supply of safe, effective and affordable vaccines for the future generations of
world’s developing nations.
PANACEA INKS DRUG DEVELOPMENT PACT WITH
RISING PHARMA
Panacea Biotech has inked a pact with
Rising Pharmaceuticals Inc to develop and market a 'product' in the American
market.
The collaboration has been established for
an oral controlled release product utilising Panacea's platform of oral drug
delivery technology, Panacea Biotec Ltd said in a statement.
Rising specialises in the sales, marketing
and distribution of finished pharma products in the US market,
"Upon commercialisation of the product
both companies shall share the profits at a pre-agreed ratio. The product under
the collaboration currently has a market size of more than $300 million in the
US," it added.
As part of the pact, Panacea would receive
an upfront research fee on signing as well as a milestone payment on approval
of the "oral controlled release product" by Food & Drug
Administration (FDA), it added.
The dossier for the product is currently
under review by FDA.
Further, both companies intend to expand
the scope of the alliance by adding new drug delivery based complex generic
products to the collaboration, the statement said.
Under the collaboration, Panacea Biotec
would undertake product development and manufacture and supply of the products;
while Rising Pharmaceuticals shall be responsible for sales and distribution of
the products in US, it added.
"The collaboration...Represents a true
win-win strategic alliance wherein the commercialisation strength of Rising
Pharmaceuticals for high barrier to entry generics perfectly synergises with
Panacea Biotec's research, development and manufacturing capabilities for such
complex products," Panacea Biotec Joint Managing Director Rajesh Jain
said.
In the current fiscal, Panacea has inked
multiple long term collaborations for North America and cumulatively these
alliances translate to potential $13 million as milestone based research fee
payments upon successful accomplishment of milestones, he added.
"The company has already received $2.5
million in Q1 FY2015 and close to $1 million is expected in the current
quarter," Jain said.
CMT REPORT (Corruption, Money Laundering
& Terrorism]
The Public Notice
information has been collected from various sources including but not limited
to: The Courts,
1] INFORMATION ON DESIGNATED PARTY
No exist designating subject or any of its
beneficial owners, controlling shareholders or senior officers as terrorist or
terrorist organization or whom notice had been received that all financial
transactions involving their assets have been blocked or convicted, found
guilty or against whom a judgement or order had been entered in a proceedings
for violating money-laundering, anti-corruption or bribery or international
economic or anti-terrorism sanction laws or whose assets were seized, blocked,
frozen or ordered forfeited for violation of money laundering or international
anti-terrorism laws.
2] Court Declaration :
No exist to
suggest that subject is or was the subject of any formal or informal
allegations, prosecutions or other official proceeding for making any
prohibited payments or other improper payments to government officials for
engaging in prohibited transactions or with designated parties.
3] Asset Declaration :
No records exist to suggest that the
property or assets of the subject are derived from criminal conduct or a
prohibited transaction.
4] Record on Financial Crime :
Charges or conviction registered
against subject: None
5] Records on Violation of
Anti-Corruption Laws :
Charges or investigation
registered against subject: None
6] Records on Int’l Anti-Money
Laundering Laws/Standards :
Charges or investigation
registered against subject: None
7] Criminal Records
No available information exist that suggest
that subject or any of its principals have been formally charged or convicted
by a competent governmental authority for any financial crime or under any
formal investigation by a competent government authority for any violation of
anti-corruption laws or international anti-money laundering laws or standard.
8] Affiliation with Government :
No record exists to suggest that any
director or indirect owners, controlling shareholders, director, officer or
employee of the company is a government official or a family member or close
business associate of a Government official.
9] Compensation Package :
Our market survey revealed that the amount
of compensation sought by the subject is fair and reasonable and comparable to
compensation paid to others for similar services.
10] Press Report :
No
press reports / filings exists on the subject.
CORPORATE GOVERNANCE
MIRA INFORM as
part of its Due Diligence do provide comments on Corporate Governance to
identify management and governance. These factors often have been predictive
and in some cases have created vulnerabilities to credit deterioration.
Our Governance Assessment
focuses principally on the interactions between a company’s management, its
Board of Directors, Shareholders and other financial stakeholders.
CONTRAVENTION
Subject is not
known to have contravened any existing local laws, regulations or policies that
prohibit, restrict or otherwise affect the terms and conditions that could be
included in the agreement with the subject.
FOREIGN EXCHANGE RATES
|
Currency |
Unit
|
Indian Rupees |
|
US Dollar |
1 |
Rs.63.64 |
|
|
1 |
Rs.98.98 |
|
Euro |
1 |
Rs.77.74 |
INFORMATION DETAILS
|
Information
Gathered by : |
PRT |
|
|
|
|
Analysis Done by
: |
DIV |
|
|
|
|
Report Prepared
by : |
NKT |
SCORE & RATING EXPLANATIONS
|
SCORE FACTORS |
RANGE |
POINTS |
|
HISTORY |
1~10 |
3 |
|
PAID-UP CAPITAL |
1~10 |
3 |
|
OPERATING SCALE |
1~10 |
3 |
|
FINANCIAL
CONDITION |
|
|
|
--BUSINESS SCALE |
1~10 |
3 |
|
--PROFITABILITY |
1~10 |
-- |
|
--LIQUIDITY |
1~10 |
3 |
|
--LEVERAGE |
1~10 |
3 |
|
--RESERVES |
1~10 |
3 |
|
--CREDIT LINES |
1~10 |
-- |
|
--MARGINS |
-5~5 |
-- |
|
DEMERIT POINTS |
|
|
|
--BANK CHARGES |
YES/NO |
YES |
|
--LITIGATION |
YES/NO |
YES |
|
--OTHER ADVERSE INFORMATION |
YES/NO |
NO |
|
MERIT POINTS |
|
|
|
--SOLE DISTRIBUTORSHIP |
YES/NO |
NO |
|
--EXPORT ACTIVITIES |
YES/NO |
NO |
|
--AFFILIATION |
YES/NO |
YES |
|
--LISTED |
YES/NO |
YES |
|
--OTHER MERIT FACTORS |
YES/NO |
YES |
|
DEFAULTER
|
|
|
|
--RBI |
YES/NO |
NO |
|
--EPF |
YES/NO |
NO |
|
|
|
|
|
TOTAL |
|
21 |
This score serves as
a reference to assess SC’s credit risk and to set the amount of credit to be
extended. It is calculated from a composite of weighted scores obtained from
each of the major sections of this report. The assessed factors and their
relative weights (as indicated through %) are as follows:
Financial condition (40%) Ownership background (20%) Payment
record (10%)
Credit history (10%) Market trend (10%) Operational
size (10%)
RATING EXPLANATIONS
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
>86 |
Aaa |
Possesses an extremely sound financial base with the strongest
capability for timely payment of interest and principal sums |
Unlimited |
|
71-85 |
Aa |
Possesses adequate working capital. No caution needed for credit
transaction. It has above average (strong) capability for payment of interest
and principal sums |
Large |
|
56-70 |
A |
Financial & operational base are regarded healthy. General
unfavourable factors will not cause fatal effect. Satisfactory capability for
payment of interest and principal sums |
Fairly Large |
|
41-55 |
Ba |
Overall operation is considered normal. Capable to meet normal
commitments. |
Satisfactory |
|
26-40 |
B |
Capability to overcome financial difficulties seems comparatively below
average. |
Small |
|
11-25 |
Ca |
Adverse factors are apparent. Repayment of interest and principal sums
in default or expected to be in default upon maturity |
Limited with
full security |
|
<10 |
C |
Absolute credit risk exists. Caution needed to be exercised |
Credit not
recommended |
|
-- |
NB |
New Business |
-- |
This report is issued at your request without any
risk and responsibility on the part of MIRA INFORM PRIVATE LIMITED (MIPL)
or its officials.