MIRA
INFORM REPORT
|
Report Date : |
14.02.2014 |
IDENTIFICATION DETAILS
|
Name : |
PANACEA BIOTEC LIMITED (w.e.f. 07.09.2003) |
|
|
|
|
Formerly Known
As : |
PANACEA DRUGS LIMITED (w.e.f. 09.09.1993) PANACEA DRUGS PRIVATE LIMITED |
|
|
|
|
Registered
Office : |
Ambala-Chandigarh Highway, Lalru – 140 501, Punjab |
|
|
|
|
Country : |
|
|
|
|
|
Financials (as
on) : |
31.03.2013 |
|
|
|
|
Date of
Incorporation : |
02.02.1984 |
|
|
|
|
Com. Reg. No.: |
16-022350 |
|
|
|
|
Capital Investment
/ Paid-up Capital : |
Rs.61.300 Millions |
|
|
|
|
CIN No.: [Company Identification
No.] |
L33117PB1984PLC022350 |
|
|
|
|
TAN No.: [Tax Deduction &
Collection Account No.] |
PNEP07035A |
|
|
|
|
PAN No.: [Permanent Account No.] |
AAACP5335J |
|
|
|
|
Legal Form : |
Public Limited Liability Company. The Company’s Shares are Listed on the Stock Exchanges. |
|
|
|
|
Line of Business
: |
Manufacturer and Marketer of Pharmaceutical Formulations viz. Tablets, Syrups/ Liquids, Capsules, Gels and Vaccines. |
|
|
|
|
No. of Employees
: |
3600 (Approximately) |
RATING & COMMENTS
|
MIRA’s Rating : |
B (29) |
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
26-40 |
B |
Capability to overcome financial difficulties seems comparatively
below average. |
Small |
|
Maximum Credit Limit : |
USD 22400000 |
|
|
|
|
Status : |
Moderate |
|
|
|
|
Payment Behaviour : |
Slow but correct |
|
|
|
|
Litigation : |
Clear |
|
|
|
|
Comments : |
Subject is an established company having moderate track record. The ratings continue to remain constrained by its weak financial
profile marked by the decline in its sales and losses reported at the
operating level. However, trade relations are fair. Business is active. Payment terms
are slow but correct. The company can be considered for business dealing with some caution. |
NOTES :
Any query related to this report can be made
on e-mail : infodept@mirainform.com
while quoting report number, name and date.
ECGC Country Risk Classification List – December 1, 2013
|
Country Name |
Previous Rating (30.09.2013) |
Current Rating (01.12.2013) |
|
India |
A1 |
A1 |
|
Risk Category |
ECGC
Classification |
|
Insignificant |
A1 |
|
Low |
A2 |
|
Moderate |
B1 |
|
High |
B2 |
|
Very High |
C1 |
|
Restricted |
C2 |
|
Off-credit |
D |
INDIAN ECONOMIC OVERVIEW
The worst is over for India’s economy with gross domestic product likely
to expand 5 %to 5.5 % this year and more than 6 % in 2015, according to Moody’s
Analytics. Concerns over the rupee and current account deficit are under
control, said the agency. Ratings firm Crisil has forecast 6 % growth for
2014/15 up from the estimated 4.8 % for 2013/14. Total economic growth,
infrastructure bottlenecks and lack of transparency and consistency in foreign
direct investment policies seem to have taken a toll on India’s attractiveness
as an investment destination, says an Ernst & Young survey. Projects
with FDI component fell 16.4 % across the globe in 2012 from the previous
year. The drop in India was steeper at 21 %. State run carrier Air India
is doling out free tickets to its 24000 employees, even as it expects to incur
a loss of Rs 39000 mn this financial year and has a debt of Rs 350000 mn.
550000 number of jobs generated across India in 2013, a fall of 0.4 % as
compared to with a year earlier. The National Capital Region has a
one-fourth share in total jobs created, according to a study by industry lobby
group Assochem, Banks, real estate, automobile and telecommunications sectors
are showing a rise of job creation. $ 805 mn investments by venture capital firms
in India during 2013, registering a drop of about 18 % over the previous year.
The Information Technology and IT-Enabled Services Industry retained its
status as the favourable venture capital investors in 2013. Pakistan has
temporarily banned gold imports for the second time in six months, as it tries
to stem smuggling into India. India’s import duty on gold is 10 % and curbs on
purchases have dried up legal imports into what used to be the world’s biggest
bullion buyers. The World Gold Council puts the amount smuggled into India at
upto 200 tonnes in 2013. The Reserve Bank of India has proposed that unclaimed
bank deposits estimated to be about Rs 35000 mn be used for education and
awareness among depositors. According to the plan, deposits that have not
been claimed for at least 10 years will be transferred to the scheme.
EXTERNAL AGENCY RATING
|
Rating Agency Name |
CARE |
|
Rating |
Long Term Bank Facilities: B |
|
Rating Explanation |
Risk prone credit quality and high risk of default. |
|
Date |
October 09, 2013 |
|
Rating Agency Name |
CARE |
|
Rating |
Short Term Bank Facilities: A4 |
|
Rating Explanation |
Minimal degree of safety and very high credit risk. |
|
Date |
October 09, 2013 |
RBI DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available RBI Defaulters’ list.
EPF (Employee Provident Fund) DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available EPF (Employee Provident Fund) Defaulters’ list as of
31-03-2012.
LOCATIONS
|
Registered Office : |
Ambala-Chandigarh Highway, Lalru – 140 501, Punjab, India |
|
Tel. No.: |
Not Available |
|
Fax No.: |
Not Available |
|
E-Mail : |
|
|
Website : |
|
|
|
|
|
Head/ Corporate Office 1: |
B 1 Extension / A-27, Mohan Cooperative Industrial Estate, Mathura Road, New Delhi – 110 044, India |
|
Tel. No.: |
91–11–26945270/ 41679000 Extn. 2081 (D) 41578024 / 26974500 / 41678000 |
|
Fax No.: |
91–11–26940199/ 26940621 / 41679075 / 41679070 / 41679081 |
|
E-Mail : |
panbio.panbio@rme.sprintrpg.ems.vsnl.net.in |
|
Website : |
|
|
|
|
|
Administrative/ Secretarial/ Corporate Office 2: |
B – 1 Extension / G – 3, Mohan Co-Operation Industrial
Estate, |
|
Tel. No.: |
91-11-41679000 Extn. 2081, (D) 41578024/ 41679015 |
|
Fax No.: |
91-11-41679070/ 41679075 |
|
E-mail: |
|
|
|
|
|
Manufacturing Facilities : |
· Ambala-Chandigarh Highway, Lalru – 140 501, Punjab, India · A-239 -242, Okhla Indl. Area, Phase–I, New Delhi – 110 020, India · Malpur, Baddi, Dist. Solan, Himachal Pradesh-173 205, India · B-1/E-12, Mohan Co-operative Industrial Estate, Mathura Road, New Delhi – 110 044, India · Plot No. 72/3, Gen Block, T.T.C. Industrial Area, Mahape, Navi Mumbai - 400 710, Maharashtra, India |
|
|
|
|
Branch/ Sales and Marketing Office : |
701, |
|
|
|
|
R & D Centers : |
·
· B-1/E-xtn. A-24-25,, Mohan Co-operative Industrial Estate Mathura Road, New Delhi – 110044, India ·
Plot No. E-4, Phase II, Industrial Area Mohali
– 160055, · Plot No. 72/3, Gen Block, T.T.C. Industrial Area, Mahape, Navi Mumbai – 400710, Maharashtra, India |
DIRECTORS
AS ON 31.03.2012
|
Name : |
Mr. Soshil Kumar Jain |
|
Designation : |
Whole-time director |
|
Address : |
18/56, East Park Area, Karol Bagh, New Delhi – 110005, India |
|
Date of Birth/Age : |
04.04.1933 |
|
Qualification : |
Pharmacist |
|
Date of Appointment : |
02.02.1984 |
|
DIN No.: |
00012812 |
|
|
|
|
Name : |
Mr. Ravinder Jain |
|
Designation : |
Managing Director |
|
Address : |
18/56, East Park Area, Karol Bagh, New Delhi – 110005, India |
|
Date of Birth/Age : |
03.08.1957 |
|
Qualification : |
Matriculate |
|
Date of Appointment : |
15.11.1984 |
|
DIN No.: |
00010101 |
|
|
|
|
Name : |
Dr. Rajesh Jain |
|
Designation : |
Managing Director |
|
Address : |
18/56, East Park Area, Karol Bagh, New Delhi – 110005, India |
|
Date of Birth/Age : |
26.04.1964 |
|
Qualification : |
B.Sc., MBA, PGDBM,
Advanced Management Diploma in Market Research |
|
Date of Appointment : |
25.11.2006 |
|
DIN No.: |
00013053 |
|
|
|
|
Name : |
Mr. Sandeep Jain |
|
Designation : |
Managing Director |
|
Address : |
18/56, East Park Area, Karol Bagh, New Delhi – 110005, India |
|
Date of Birth/Age : |
17.07.1966 |
|
Qualification : |
B. Com |
|
Date of Appointment : |
15.11.1984 |
|
DIN No.: |
00012973 |
|
|
|
|
Name : |
Mr. Sumit Jain |
|
Designation : |
Whole-time director |
|
Address : |
18/56, East Park Area, Karol Bagh, New Delhi – 110005, India |
|
Date of Birth/Age : |
07.02.1981 |
|
Qualification : |
Post Graduate Diploma in Business Management |
|
Date of Appointment : |
27.07.2005 |
|
DIN No.: |
00014236 |
|
|
|
|
Name : |
Mr. Raghava Lakshmi Narasimhan |
|
Designation : |
Director |
|
Address : |
Flat No.6, Plot No.858, New No.3, Paneer Selvam Salai, K K nagar, Chennai – 600078, Tamilnadu, India
|
|
Date of Birth/Age : |
01.10.1940 |
|
Date of Appointment : |
31.01.2001 |
|
DIN No.: |
00073873 |
|
|
|
|
Name : |
Mr. Namdeo Narayan Khamitkar |
|
Designation : |
Director |
|
Address : |
3, Krishna Kunj Apartments, 42 Shantisheela Society, Law College Road,
Pune – 411004, Maharashtra, India |
|
Date of Birth/Age : |
02.12.1940 |
|
Date of Appointment : |
31.01.2006 |
|
DIN No.: |
00017154 |
|
|
|
|
Name : |
Mr. Sunil Kapoor |
|
Designation : |
Director |
|
Address : |
W-155, Greater Kailash, Part – II, New Delhi – 110045, India |
|
Date of Birth/Age : |
25.01.1957 |
|
Date of Appointment : |
31.01.2001 |
|
DIN No.: |
00029133 |
|
|
|
|
Name : |
Mr. Krishna Murari Lal |
|
Designation : |
Director |
|
Address : |
706-A, Sector – 23, Huda, Gurgaon – 122017, Haryana, India |
|
Date of Birth/Age : |
22.06.1940 |
|
Date of Appointment : |
28.04.2006 |
|
DIN No.: |
00016166 |
|
|
|
|
Name : |
Mr. Aditya Narain Saksena |
|
Designation : |
Director |
|
Address : |
Flat No. B-9012, B-Block, Guar Green City, Indirapuram, Ghaziabad –
201010, Uttar Pradesh, India |
|
Date of Birth/Age : |
07.05.1938 |
|
Date of Appointment : |
22.12.2005 |
|
DIN No.: |
00016107 |
KEY EXECUTIVES
|
Name : |
Mr. Vinod Goel |
|
Designation : |
Secretary and General Manager Legal |
|
Address : |
House No.173, Sector - 4, Vaishali, Ghaziabad – 201010, Uttar Pradesh,
India |
|
Date of Birth/Age : |
08.02.1965 |
|
Date of Appointment : |
13.01.1999 |
|
PAN No.: |
AHBPG9784H |
SHAREHOLDING PATTERN
AS ON 31.12.2013
|
Category
of Shareholders |
No. of Shares |
Percentage of Holding |
|
(A) Shareholding of Promoter and Promoter Group |
|
|
|
|
|
|
|
|
43510100 |
71.04 |
|
|
2313454 |
3.78 |
|
|
2313454 |
3.78 |
|
|
45823554 |
74.81 |
|
|
|
|
|
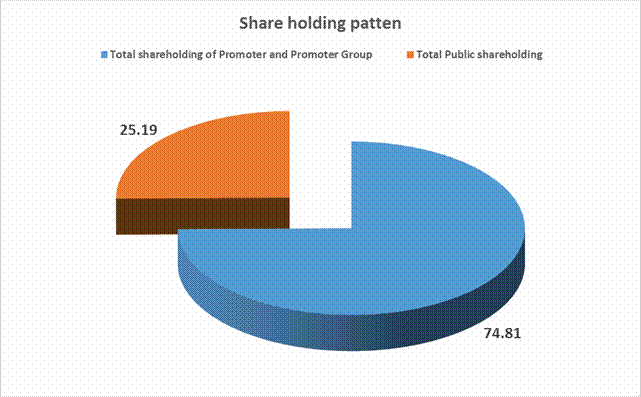
Total shareholding of Promoter and Promoter Group (A) |
45823554 |
74.81 |
|
(B) Public Shareholding |
|
|
|
|
|
|
|
|
22984 |
0.04 |
|
|
1236647 |
2.02 |
|
|
1259631 |
2.06 |
|
|
|
|
|
|
9709910 |
15.85 |
|
|
|
|
|
|
3093587 |
5.05 |
|
|
135000 |
0.22 |
|
|
1229064 |
2.01 |
|
|
45606 |
0.07 |
|
|
85178 |
0.14 |
|
|
50825 |
0.08 |
|
|
1045000 |
1.71 |
|
|
2455 |
0.00 |
|
|
14167561 |
23.13 |
|
Total Public shareholding (B) |
15427192 |
25.19 |
|
Total (A)+(B) |
61250746 |
100.00 |
|
(C) Shares held by Custodians and against which Depository Receipts
have been issued |
0 |
0.00 |
|
|
0 |
0.00 |
|
|
0 |
0.00 |
|
|
0 |
0.00 |
|
Total (A)+(B)+(C) |
61250746 |
0.00 |
BUSINESS DETAILS
|
Line of Business : |
Manufacturer and Marketer of Pharmaceutical Formulations
viz. Tablets, Syrups/ Liquids, Capsules, Gels and Vaccines. |
||||||||
|
|
|
||||||||
|
Products : |
|
PRODUCTION STATUS
AS ON 31.03.2011
Licensed Capacity
Per annum
Recombinant Bulk Vaccine – 18 Millions doses
|
Particulars |
Unit |
Installed
Capacity per annum |
|
Tables |
Nos./Million |
1684.000 |
|
Capsules |
Nos. / Million |
370.000 |
|
Syrups / Liquids |
Bottles /
Million |
15.800 |
|
Gels |
Tubes/ Million |
21.200 |
|
Vaccines (Finished Doses) |
Doses / Million |
878.000 |
|
Pre-filled Syringes |
Doses / Million |
17.000 |
|
Recombinant Bulk Vaccines ** |
Doses / Million |
18.000 |
|
Tetanus Bulk Vaccines |
Doses / Million |
75.000 |
|
Bacterial Bulk Vaccines *** |
Doses / Million |
68.800 |
|
Block - IV **** |
|
|
|
Cell Culture Block |
Doses / Million |
8.000 |
|
Recombinant Bulk Vaccine |
Doses / Million |
18.000 |
|
Flu Bulk Vaccine Block |
Doses / Million |
63.500 |
NOTE
*As Certified by the management.
**This facility is capable of manufacturing various Bulk Vaccines and Antigens including Hepatitis B (Hep B), Haemophilus Influenza Type B (HIB-TT) and other vaccines.
***Bacterial Bulk Vaccine facility is capable of manufacturing various bulk vaccines including Diptheria (D), Whole Cell Pertussis (wP), Acellular Pertussis (aP). Installed Capacity reduces by 5 million doses to 63.8 million doses in case of production of Acellular Pertussis (aP).
****Cell Culture facility is capable of manufacturing various bulk vaccines and biopharmaceuticals. Recombinant Bulk Vaccine facility is capable of manufacturing of Hepatitis B (Hep-B), Haemophilus Influenza Type B (HIB-TT) and other vaccines. Flu Bulk Vaccine facility is capable of manufacturing of H1N1 and other flu bulk vaccines.
|
Particulars |
Unit |
Actual
Production |
|
Tables ** |
Nos. |
625.600 |
|
Capsules |
Nos. |
95.000 |
|
Syrups / Liquids |
Ml |
334.700 |
|
Gels |
Gms |
52.900 |
|
Vaccines |
Vials |
60.300 |
|
Pre-filled Syringes |
PFS |
2.100 |
|
Injection |
Nos. |
0.500 |
|
Other Products |
Gms. |
43.300 |
Note:
* Actual production includes production at
loan licensee locations meant for sale by the company
** Actual production includes 258.7 Million
Tables manufactured for other under loan licenses basis.
GENERAL INFORMATION
|
No. of Employees : |
3600 (Approximately) |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Bankers : |
·
Axis Bank Limited ·
Bank of India ·
IDBI Bank Limited ·
Indian Overseas Bank ·
State Bank of Mysore ·
State Bank of Travancore ·
Union Bank of India ·
Canara Bank ·
State Bank of India, Industrial Finance Branch,
14th Floor, J. V. Building, 1, Tolstoy Marg, New Delhi – 110001,
India |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Facilities : |
Notes : Long term Borrowings: a)
Foreign currency term loans from State Bank of
India [loan - I and II] carrying interest @ LIBOR plus 7.5% and 5.75%
respectively, have been re-scheduled and converted into Indian rupee term
loans (loan I, II and III) during the year. These Indian rupee term loans
carry interest @ 5.9%, 5.2% and 5.5% above base rate respectively and are
repayable in twelve quarterly installments commencing from June’14. b)
Foreign currency term loan from State Bank of
Travancore carrying interest @ 6 months LIBOR plus 7.5% has been re-scheduled
and converted into Indian rupee term loan during the year. The Indian rupee
term loan carries interest @ 7.45% above base rate and is repayable in twelve
quarterly installments commencing from June’14. c)
Foreign currency term loan from Bank of India
carries interest @ 6 months LIBOR plus 4.75%. The loan is repayable in three
equal yearly installments commencing at the end of sixth year from the date
of first drawdown [i.e. in financial year 2017-18]. d)
Indian rupee term loan from Indian Overseas Bank
carries interest @ 1.75% above base rate. This loan is repayable in eight
equal quarterly installments commencing from January’14. e)
Above Foreign currency term loans and Indian
rupee term loans taken from banks are secured by way of first pari-passu
charge by hypothecation of the Company’s entire movable fixed assets, both
present and future and mortgage of immovable properties of the Company being
land admeasuring 96 bighas, 19 biswas and 93 bighas 12 biswas and 10 biswas
situated at village Samalheri, Tehsil Dera Bassi, District S.A.S. Nagar
(Mohali), Punjab and land admeasuring 26 bighas, 3 biswas situated at Village
Manpura, Tehsil Nalagarh, District Solan and land admeasuring 91 bighas, 1
biswas situated at Village Malpura, Tehsil Nalagarh, District Solan in the
state of Himachal Pradesh and land admeasuring 9435.66 sq yards situated at
Industrial Plot No.E-4, PH-2, Industrial Area, S.A.S Nagar, (Mohali), Punjab
. These loans are also collaterally secured by personal guarantees of the
promoter-directors of the Company, viz. Mr. Soshil Kumar Jain, Mr. Ravinder
Jain, Dr. Rajesh Jain and Mr. Sandeep Jain. f)
Indian rupee term loans from Government of India
through Department of Biotechnology are project specific loans which carry
interest @ 2.00% p.a. These loans are repayable in ten equal half-yearly
installments. The repayment of these loans would commence from one year after
the completion of the respective projects. g)
Indian rupee term loans from Government of India
through Technology Development Board is a project specific loan which carry
interest @ 5.00% p.a. The loan is repayable in ten equal half-yearly
installments commencing from January’14. h)
Secured term loan from Government of India is
secured by way of hypothecation of the company’s all equipment’s, apparatus,
machineries, machineries spares, tools and other accessories, goods and/or
other movable property present and future by way of first charge on
pari-passu basis. Short
Term Borrowings: a)
Cash credit from bank carries interest @ 12.5% to
14% p.a. b)
Buyers’ credit from bank carries interest @ 1.6%
to 5% p.a. c)
Working capital loan from bank carries interest @
13.5% p.a. |
|
|
|
|
Banking
Relations : |
-- |
|
|
|
|
Statutory Auditors : |
|
|
Name: |
S. R. Batiliboi and Associates Chartered Accountants |
|
Address: |
1st Floor, Tower A. Building No.8, DLF Cyber city, Phase III, Sector 25, Gurgoan-122001, Haryana, India |
|
PAN No.: |
AALFS0506L |
|
|
|
|
Cost Auditors : |
|
|
Name: |
J. P. Gupta and Associates Cost Accountants |
|
Address: |
|
|
|
|
|
Joint Ventures : |
·
Chiron Panacea
Vaccines Private Limited ·
Cambridge
Biostability Limited (liquidated
on September 16, 2011) ·
Adveta
Power Private Limited, w.e.f. July 4, 2011 |
|
|
|
|
Subsidiaries : |
· Best On Health Limited (BOH) (Wholly-owned subsidiary (WOS)) · Radicura and company Limited (Indirect WOS through BOH), CIN No.: U74899DL1993PLC056682 · Panacea Hospitality Services Private Limited, (Indirect WOS through BOH) CIN NO.: U55101DL2007PTC166763 · Panacea Educational Institute Private Limited, (Indirect WOS through BOH) CIN No.:U80904DL2007PTC166661 · Sunanda Steel Company Limited, (Indirect WOS through BOH) CIN No.: U13209DL2007PLC163082 · Nirmala Organic Farms and Resorts Private Limited, (Indirect WOS through BOH) w.e.f. February 23, 2011 CIN No.: U01403DL2010PTC198194 · Best On Health Foods Limited (Indirect WOS through BOH) w.e.f. December 6, 2010 CIN No.: U15122DL2007PLC170999 · Rees Investments Limited, (Rees) (Guernsey): WOS · Kelisia Holdings Limited, (Cyprus): Indirect WOS through Rees · Kelisia Investment Holding AG (KIH) (Switzerland): Indirect WOS through Kelisia Holdings Limited, · Panacea Biotec (International) SA (PBS) (Switzerland) (Indirect WOS through KIH) · Panacea Biotec GmbH (Germany) (Indirect WOS through PBS) · Panacea Biotec (Europe) AG, (Switzerland): Indirect WOS through PBS · Panacea Biotec FZE, (UAE): WOS ·
Panacea Biotec Inc. (USA): WOS (liquidated on March 30, 2011) · NewRise Healthcare Private Limited (Formerly known as Umkal Medical Institute Private Limited) CIN No.: U85110DL2002PTC114987 · Lakshmi & Manager Holdings Limited (“LMH”) WOS w.e.f. November 24, 2011* *Associate Company prior to becoming WOS. · Trinidhi Finance Private Limited (Indirect WOS through LMH) w.e.f. November 24, 2011** ** Subsidiary of LMH w.e.f. 6th August, 2011 and became WOS of LMH on October 7, 2011 · Best General Insurance Company Limited ( indirect subsidiary through LMH)) w.e.f. November 24, 2011 |
|
|
|
|
Associates : |
·
PanEra Biotec
Private Limited |
CAPITAL STRUCTURE
AS ON 31.03.2013
Authorised Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
125000000 |
Equity Shares |
Re.1/- Each |
Rs.125.000 millions |
|
110000000 |
Preference Shares |
Rs.10/- Each |
Rs.1100.000 millions |
|
|
Total |
|
Rs.1225.000
millions |
Issued, Subscribed & Paid-up Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
61250746 |
Equity Shares |
Re.1/- Each |
Rs.61.300
millions |
|
|
|
|
|
NOTE
a) Terms/right attached to equity shares:
The Company has
only one class of equity shares having a par value of Re.1 per share. Each holder
of equity shares is entitled to one vote per share. The Company declares and
pays dividends in Indian Rupees. The dividend if any, proposed by the Board of
Directors is subject to the approval of the shareholders in the ensuing Annual
General Meeting.
In the event of
liquidation of the Company, the holders of equity shares will be entitled to
receive remaining assets of the Company, after distribution of all preferential
amounts. The distribution will be in proportion to the number of equity shares held
by the shareholders.
b) Reconciliation
of the shares outstanding at the beginning and at the end of the reporting
financial year:
|
Particulars
|
No.
of shares |
Rs.
In Millions |
|
|
|
|
|
At the beginning of the year |
61,250,746 |
61.300 |
|
Less: Buy back of equity shares during the year |
-- |
-- |
|
Outstanding at the end of the year |
61,250,746 |
61.300 |
c) Aggregate no.
of equity shares bought back during the period of five years immediately
preceding the reporting date:
|
Name of Persons |
No.
of shares |
|
Equity shares bought back by the Company during the financial year
2010-11 |
5592000 |
d) Equity shares
bought back by the Company during the financial year 2010-11:
|
Name of Persons |
No. of shares |
% age of holding |
|
|
|
|
|
Mr. Soshil Kumar Jain |
5000000 |
8.16 |
|
Mr. Ravinder Jain |
5712300 |
9.33 |
|
Dr. Rajesh Jain |
6213500 |
10.14 |
|
Mr. Sandeep Jain |
4792100 |
7.82 |
|
Soshil Kumar Jain (HUF) |
2380700 |
3.89 |
|
Ravinder Jain (HUF) |
4135000 |
6.75 |
|
Rajesh Jain (HUF) |
4368500 |
7.13 |
|
Sandeep Jain (HUF) |
4105000 |
6.70 |
|
Serum Institute of India Limited |
8911632 |
14.55 |
FINANCIAL DATA
[all figures are
in Rupees Millions]
ABRIDGED BALANCE
SHEET
|
SOURCES OF FUNDS |
31.03.2013 |
31.03.2012 |
31.03.2011 |
|
I.
EQUITY AND LIABILITIES |
|
|
|
|
|
|
|
|
|
(1)Shareholders' Funds |
|
|
|
|
(a) Share Capital |
61.300 |
61.300 |
61.300 |
|
(b) Reserves & Surplus |
5,551.300 |
8,079.600 |
6,306.800 |
|
(c) Money received against
share warrants |
0.000 |
0.000 |
0.000 |
|
|
|
|
|
|
(2) Share Application money
pending allotment |
0.000 |
0.000 |
0.000 |
|
Total
Shareholders’ Funds (1) + (2) |
5,612.600 |
8,140.900 |
6,368.100 |
|
|
|
|
|
|
(3) Non-Current Liabilities |
|
|
|
|
(a) long-term borrowings |
6,072.000 |
4,830.300 |
4,237.200 |
|
(b) Deferred tax liabilities
(Net) |
0.000 |
205.000 |
756.600 |
|
(c) Other long term
liabilities |
23.600 |
23.800 |
0.000 |
|
(d) long-term provisions |
25.200 |
20.600 |
23.600 |
|
Total
Non-current Liabilities (3) |
6,120.800 |
5,079.700 |
5,017.400 |
|
|
|
|
|
|
(4) Current Liabilities |
|
|
|
|
(a) Short term borrowings |
2,991.400 |
2,693.400 |
3,824.400 |
|
(b) Trade payables |
1,539.900 |
1,181.400 |
826.900 |
|
(c) Other current liabilities |
434.700 |
1,339.800 |
1,010.000 |
|
(d) Short-term provisions |
98.800 |
93.600 |
131.100 |
|
Total
Current Liabilities (4) |
5,064.800 |
5,308.200 |
5,792.400 |
|
|
|
|
|
|
TOTAL |
16,798.200 |
18,528.800 |
17,177.900 |
|
|
|
|
|
|
II.
ASSETS |
|
|
|
|
(1) Non-current assets |
|
|
|
|
(a) Fixed Assets |
|
|
|
|
(i) Tangible assets |
9,169.800 |
9,543.400 |
5,346.300 |
|
(ii) Intangible Assets |
418.600 |
469.700 |
124.500 |
|
(iii) Capital work-in-progress |
169.400 |
120.700 |
229.400 |
|
(iv) Intangible assets under
development |
106.300 |
349.900 |
823.400 |
|
(b) Non-current Investments |
2,653.500 |
2,584.700 |
2,329.400 |
|
(c) Deferred tax assets (net) |
0.000 |
0.000 |
0.000 |
|
(d) Long-term Loan and Advances |
1,026.800 |
1,063.100 |
1,046.500 |
|
(e) Other Non-current assets |
0.000 |
1.200 |
0.900 |
|
Total
Non-Current Assets |
13,544.400 |
14,132.700 |
9,900.400 |
|
|
|
|
|
|
(2) Current assets |
|
|
|
|
(a) Current investments |
0.000 |
0.000 |
0.000 |
|
(b) Inventories |
2,247.400 |
3,397.300 |
3,680.200 |
|
(c) Trade receivables |
687.500 |
664.500 |
2,788.400 |
|
(d) Cash and cash equivalents |
125.900 |
113.300 |
393.800 |
|
(e) Short-term loans and
advances |
163.500 |
170.300 |
334.800 |
|
(f) Other current assets |
29.500 |
50.700 |
80.300 |
|
Total
Current Assets |
3,253.800 |
4,396.100 |
7,277.500 |
|
|
|
|
|
|
TOTAL |
16,798.200 |
18,528.800 |
17,177.900 |
PROFIT & LOSS
ACCOUNT
|
|
PARTICULARS |
31.03.2013 |
31.03.2012 |
31.03.2011 |
|
|
|
SALES |
|
|
|
|
|
|
|
Income |
5959.200 |
7005.800 |
11498.300 |
|
|
|
Other Income |
54.300 |
74.600 |
156.800 |
|
|
|
TOTAL (A) |
6013.500 |
7080.400 |
11655.100 |
|
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
|
|
Cost of Materials Consumed |
2363.700 |
3021.200 |
4473.900 |
|
|
|
Purchases of Stock-in-Trade |
232.600 |
245.200 |
244.400 |
|
|
|
Changes in inventories of finished goods, work-in-progress
and Stock-in-Trade |
2.900 |
205.600 |
588.700 |
|
|
|
Employees benefits expense |
1372.300 |
1504.500 |
1543.100 |
|
|
|
Other expenses |
2828.600 |
2968.400 |
1961.500 |
|
|
|
TOTAL (B) |
6800.100 |
7944.900 |
8811.600 |
|
|
|
|
|
|
|
|
Less |
PROFIT
BEFORE INTEREST, TAX, DEPRECIATION AND AMORTISATION (A-B) (C) |
(786.600) |
(864.500) |
2843.500 |
|
|
|
|
|
|
|
|
|
Less |
FINANCIAL
EXPENSES (D) |
1057.400 |
1011.100 |
557.500 |
|
|
|
|
|
|
|
|
|
|
PROFIT
BEFORE TAX, DEPRECIATION AND AMORTISATION (C-D) (E) |
(1844.000) |
(1875.600) |
2286.000 |
|
|
|
|
|
|
|
|
|
Less |
DEPRECIATION/
AMORTISATION (F) |
835.400 |
753.900 |
731.100 |
|
|
|
|
|
|
|
|
|
Less |
EXCEPTIONAL
ITEMS |
173.100 |
0.000 |
0.000 |
|
|
|
|
|
|
|
|
|
|
PROFIT/ (LOSS)
BEFORE TAX (E-F) (G) |
(2506.300) |
(2629.500) |
1554.900 |
|
|
|
|
|
|
|
|
|
Less |
TAX (H) |
205.000 |
551.600 |
204.400 |
|
|
|
|
|
|
|
|
|
|
PROFIT/ (LOSS)
AFTER TAX (G-H) (I) |
(2301.300) |
(2077.900) |
1350.500 |
|
|
|
|
|
|
|
|
|
|
PREVIOUS
YEARS’ BALANCE BROUGHT FORWARD |
1941.600 |
4019.500 |
2856.100 |
|
|
|
|
|
|
|
|
|
|
APPROPRIATIONS |
|
|
|
|
|
|
|
Dividend proposed on Equity Shares |
NA |
0.000 |
45.900 |
|
|
|
Dividend Distribution Tax |
NA |
0.000 |
7.500 |
|
|
|
Transfer to General Reserve |
NA |
0.000 |
135.000 |
|
|
BALANCE CARRIED
TO THE B/S |
NA |
1941.600 |
4019.500 |
|
|
|
|
|
|
|
|
|
|
EARNINGS IN
FOREIGN CURRENCY |
|
|
|
|
|
|
|
F.O.B. value of exports |
905.200 |
3436.500 |
7103.900 |
|
|
|
Income from distribution rights |
2.100 |
0.500 |
0.000 |
|
|
|
Research and license fees income |
299.400 |
0.000 |
0.000 |
|
|
|
Interest income from subsidiary company |
0.000 |
36.200 |
42.000 |
|
|
TOTAL EARNINGS |
1206.700 |
3473.200 |
7145.900 |
|
|
|
|
|
|
|
|
|
|
IMPORTS |
|
|
|
|
|
|
|
Raw Materials and Packing Materials |
310.900 |
2184.000 |
2839.900 |
|
|
|
Capital Goods |
74.300 |
226.900 |
91.200 |
|
|
TOTAL IMPORTS |
385.200 |
2410.900 |
2931.100 |
|
|
|
|
|
|
|
|
|
|
Earnings/ (Loss)
Per Share (Rs.) |
(37.57) |
(33.92) |
21.35 |
|
QUARTERLY RESULTS
|
PARTICULARS |
30.06.2013 (Unaudited) |
30.09.2013 (Unaudited) |
31.12.2013 (Unaudited) |
|
|
1st Quarter |
2nd Quarter |
3rd
Quarter |
|
|
|
|
|
|
Net Sales |
1176.300 |
1100.900 |
1116.200 |
|
Total Expenditure |
1277.200 |
1112.400 |
1508.5000 |
|
PBIDT (Excl OI) |
(100.900) |
(11.500) |
(392.300) |
|
Other Income |
2.500 |
2.000 |
1777.200 |
|
Operating Profit |
(98.400) |
(9.500) |
1384.900 |
|
Interest |
321.200 |
368.200 |
397.900 |
|
Exceptional Items |
0.000 |
0.000 |
0.000 |
|
PBDT |
(419.600) |
(377.700) |
987.000 |
|
Depreciation |
189.600 |
185.000 |
159.100 |
|
Profit Before Tax |
(609.200) |
(562.700) |
827.900 |
|
Tax |
0.000 |
0.000 |
0.000 |
|
Provisions and contingencies |
0.000 |
0.000 |
0.000 |
|
Profit After Tax |
(609.200) |
(562.700) |
827.900 |
|
Extraordinary Items |
0.000 |
0.000 |
0.000 |
|
Prior Period Expenses |
0.000 |
0.000 |
0.000 |
|
Other Adjustments |
0.000 |
0.000 |
0.000 |
|
Net Profit |
(609.200) |
(562.700) |
827.900 |
KEY RATIOS
|
PARTICULARS |
|
31.03.2013 |
31.03.2012 |
31.03.2011 |
|
PAT / Total Income |
(%) |
(38.27) |
(29.35) |
11.59 |
|
|
|
|
|
|
|
Net Profit Margin (PBT/Sales) |
(%) |
(42.06) |
(37.53) |
13.52 |
|
|
|
|
|
|
|
Return on Total Assets (PBT/Total Assets} |
(%) |
(18.07) |
(16.99) |
11.27 |
|
|
|
|
|
|
|
Return on Investment (ROI) (PBT/Networth) |
|
(0.45) |
(0.32) |
0.24 |
|
|
|
|
|
|
|
Debt Equity Ratio (Total Debt/Networth) |
|
1.61 |
0.92 |
1.27 |
|
|
|
|
|
|
|
Current Ratio (Current Asset/Current Liability) |
|
0.64 |
0.83 |
1.26 |
FINANCIAL ANALYSIS
[all figures are
in Rupees Millions]
DEBT EQUITY RATIO
|
Particular |
31.03.2011 |
31.03.2012 |
31.03.2013 |
|
|
(Rs.
In Millions) |
(Rs.
In Millions) |
(Rs.
In Millions) |
|
Share Capital |
61.300 |
61.300 |
61.300 |
|
Reserves & Surplus |
6,306.800 |
8,079.600 |
5,551.300 |
|
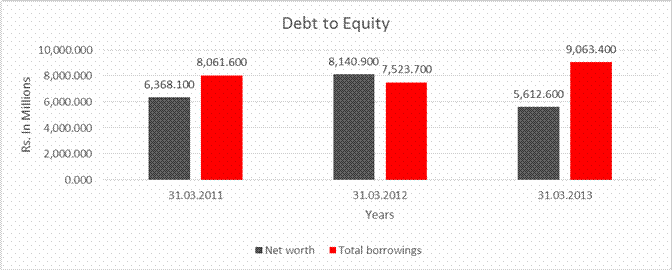
Net
worth |
6,368.100 |
8,140.900 |
5,612.600 |
|
|
|
|
|
|
long-term borrowings |
4,237.200 |
4,830.300 |
6,072.000 |
|
Short term borrowings |
3,824.400 |
2,693.400 |
2,991.400 |
|
Total
borrowings |
8,061.600 |
7,523.700 |
9,063.400 |
|
Debt/Equity
ratio |
1.266 |
0.924 |
1.615 |
YEAR-ON-YEAR GROWTH
|
Year
on Year Growth |
31.03.2011 |
31.03.2012 |
31.03.2013 |
|
|
(Rs.
In Millions) |
(Rs.
In Millions) |
(Rs.
In Millions) |
|
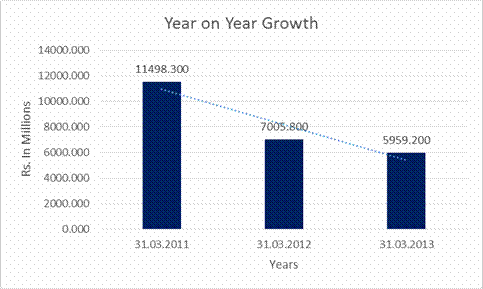
Sales |
11498.300 |
7005.800 |
5959.200 |
|
|
|
(39.071) |
(14.939) |
NET PROFIT MARGIN
|
Net
Profit Margin |
31.03.2011 |
31.03.2012 |
31.03.2013 |
|
|
(Rs.
In Millions) |
(Rs.
In Millions) |
(Rs.
In Millions) |
|
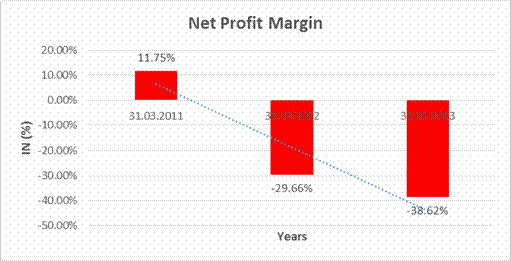
Sales |
11498.300 |
7005.800 |
5959.200 |
|
Profit |
1350.500 |
(2077.900) |
(2301.300) |
|
|
11.75% |
(29.66%) |
(38.62%) |
LOCAL AGENCY FURTHER INFORMATION
|
Sr. No. |
Check List by Info Agents |
Available in
Report (Yes / No) |
|
1] |
Year of Establishment |
Yes |
|
2] |
Locality of the firm |
Yes |
|
3] |
Constitutions of the firm |
Yes |
|
4] |
Premises details |
No |
|
5] |
Type of Business |
Yes |
|
6] |
Line of Business |
Yes |
|
7] |
Promoter's background |
Yes |
|
8] |
No. of employees |
Yes |
|
9] |
Name of person contacted |
No |
|
10] |
Designation of contact
person |
No |
|
11] |
Turnover of firm for last
three years |
Yes |
|
12] |
Profitability for last
three years |
Yes |
|
13] |
Reasons for variation
<> 20% |
-- |
|
14] |
Estimation for coming
financial year |
No |
|
15] |
Capital in the business |
Yes |
|
16] |
Details of sister
concerns |
Yes |
|
17] |
Major suppliers |
No |
|
18] |
Major customers |
No |
|
19] |
Payments terms |
No |
|
20] |
Export / Import details
(if applicable) |
No |
|
21] |
Market information |
-- |
|
22] |
Litigations that the firm
/ promoter involved in |
-- |
|
23] |
Banking Details |
Yes |
|
24] |
Banking facility details |
Yes |
|
25] |
Conduct of the banking
account |
-- |
|
26] |
Buyer visit details |
-- |
|
27] |
Financials, if provided |
Yes |
|
28] |
Incorporation details, if
applicable |
Yes |
|
29] |
Last accounts filed at
ROC |
Yes |
|
30] |
Major Shareholders, if
available |
No |
|
31] |
Date of Birth of
Proprietor/Partner/Director, if available |
Yes |
|
32] |
PAN of Proprietor/Partner/Director,
if available |
No |
|
33] |
Voter ID No of
Proprietor/Partner/Director, if available |
No |
|
34] |
External Agency Rating,
if available |
Yes |
NOTE:
The registered office address of the company has been shifted from
“Derabassi, Tehsil Rajpura, Patiala-140 501, Punjab, India” to the present
address w.e.f. 07.06.2011
UNSECURED LOANS:
|
Particulars |
31.03.2013 Rs. In Millions |
31.03.2012 Rs. In Millions |
|
Long Term Borrowings |
|
|
|
Through
Department of Science and Technology (refer note a) |
16.000 |
18.000 |
|
Other long term borrowings |
|
|
|
Deposits from
public and related party (refer note b) |
0.000 |
9.000 |
|
|
|
|
|
Short Term Borrowings |
|
|
|
Deposits from
public and related parties (refer note a) |
339.600 |
271.500 |
|
Loan from related parties (refer note a) |
362.400 |
198.000 |
|
|
|
|
|
Total |
718.000 |
496.500 |
NOTES:
Long Term Borrowings:
a)
Indian rupee term loan from Government of India,
through Department of Science & Technology is a project specific loan which
carries interest @ 3.00% p.a. The loan is repayable in ten equal annual
installments which would commence from one year after the completion of the
project.
b)
Deposits from public and related party is caring
interest @ 9%. These deposits are repayable in one to two years from the date
of receipt of deposit.
c)
Finance lease obligation is repayable into three
equal annual installments. Last installment has been repaid during the current
financial year.
Short Term Borrowings:
a)
Deposits and Loan from public and related parties
carries interest @ 9% to 10% p.a.
b)
Cash credit, Buyers’ credits & Working capital
loan from bank are secured by way of first pari passu charge by hypothecation
of all current assets and also by way of second paripassu charge on all the
movable fixed assets (including machinery and spares) of the Company and
existing immovable properties of the Company being land admeasuring 96 bighas,
19 biswas & 93 bighas 12 biswas & 10 biswas situated at village
Samalheri, Tehsil Dera Bassi, District S.A.S. Nagar (Mohali), Punjab and land
admeasuring 26 bighas, 3 biswas situated at Village Manpura, Tehsil Nalagarh,
District Solan and land admeasuring 91 bighas, 1 biswas situated at Village
Malpura, Tehsil Nalagarh, District Solan in the state of Himachal Pradesh,and
land admeasuring 9435.66 sq yards situated at Indl Plot No.E-4, PH-2, Indl
Area, S.A.S. Nagar, (Mohali ), Punjab. These are also collaterally secured by
personal guarantees of the promoter- directors of the Company, viz Mr. Soshil
Kumar Jain, Mr. Ravinder Jain, Dr. Rajesh Jain and Mr. Sandeep Jain.
INDEX OF CHARGE:
|
Sr. No. |
Charge ID |
Date of Charge
Creation/Modification |
Charge amount
secured |
Charge Holder |
Address |
Service Request
Number (SRN) |
|
1 |
10404560 |
24/12/2012 |
269,300,000.00 |
STATE
BANK OF INDIA |
INDUSTRIAL
FINANCE BRANCH, 14TH FLOOR, J V BUILDING, 1, TOLSTOY MARG, NEW DELHI, DELHI -
110001, INDIA |
B68639822 |
|
2 |
10376668 |
23/10/2012
* |
50,000,000.00 |
TECHNOLOGY
DEVELOPMENT BOARD |
WING
- A, GROUND FLOOR, VISHWAKARMA BHAWAN, SHAHEED JEET SINGH MARG, NEW DELHI,
DELHI - 110016, INDIA |
B64342330 |
|
3 |
10355025 |
20/04/2012 |
10,990,000.00 |
DEPARTMENT
OF BIOTECHNOLOGY |
6-8TH
FLOOR, BLOCK NO.2, CGO COMPLEX, LODHI ROAD, NEW DELHI, DELHI - 110003, INDIA |
B39421623 |
|
4 |
10355027 |
20/04/2012 |
32,830,000.00 |
DEPARTMENT
OF BIOTECHNOLOGY |
6-8TH
FLOOR, BLOCK NO.2, CGO COMPLEX, LODHI ROAD, NEW DELHI, DELHI - 110003, INDIA |
B39421870 |
|
5 |
10330445 |
18/01/2012
* |
1,290,000,000.00 |
BANK
OF INDIA |
HEAD
OFFICE; STAR HOUSE, C-5, G BLOCK, BANDRA KURLA COMPLEX, MUMBAI, MAHARASHTRA -
400051, INDIA |
B30279731 |
|
6 |
10266434 |
16/01/2012
* |
1,000,000,000.00 |
INDIAN
OVERSEAS BANK |
RAJIV
CIRCLE, D-28-29, CONNAUGHT PLACE, NEW DELHI, DELHI - 110001, INDIA |
B31260458 |
|
7 |
10230748 |
06/07/2010 |
109,400,000.00 |
DEPARTMENT
OF BIOTECHNOLOGY |
6-8TH
FLOOR, BLOCK NO. 2, CGO COMPLEX, LODHI ROAD, NEW DELHI, DELHI - 110003, INDIA |
A90678137 |
|
8 |
10224547 |
24/12/2012
* |
1,000,000,000.00 |
STATE
BANK OF INDIA |
INDUSTRIAL
FINANCE BRANCH, 14TH FLOOR, J V BUILDING, 1, TOLSTOY MARG, NEW DELHI, DELHI -
110001, INDIA |
B68640507 |
|
9 |
10089746 |
24/12/2012
* |
2,000,000,000.00 |
STATE
BANK OF INDIA |
INDUSTRIAL
FINANCE BRANCH, 14TH FLOOR, J V BUILDING, 1, TOLSTOY MARG, NEW DELHI, DELHI -
110001, INDIA |
B68641133 |
|
10 |
10069827 |
08/11/2012
* |
1,096,700,000.00 |
STATE
BANK OF TRAVANCORE |
COMMERCIAL
BRANCH, TRAVANCORE HOUSE, K.G. MARG, NEW DELHI, DELHI - 110001, INDIA |
B63220693 |
|
11 |
10056735 |
05/10/2009
* |
1,500,000,000.00 |
IDBI
BANK LIMITED |
3RD FLOOR,
INDIAN RED CROSS SOCIETY BUILDING, 1, RED CROSS ROAD, NEW DELHI -
110001,INDIA |
A72025109 |
|
12 |
80008672 |
24/12/2012
* |
3,621,800,000.00 |
STATE
BANK OF INDIA |
INDUSTRIAL
FINANCE BRANCH, 14TH FLOOR, J V BUILDING, 1, TOLSTOY MARG, NEW DELHI, DELHI -
110001, INDIA |
B68639061 |
* Date of charge modification
MANAGEMENT
DISCUSSION AND ANALYSIS:
Industry
Structure and Developments
Global
Vaccine Industry
In the
last decade, vaccines have emerged as a strong part of the global healthcare
markets. Vaccines, which are now preventing over 3 million deaths annually,
have also helped in improving the quality of life of millions across the globe.
The pharmaceutical companies worldwide have increased their focus on vaccines
which appears to be a key source of future growth. The growth of the vaccine
industry can be largely attributed to increased awareness worldwide supported
by increased availability of vaccines at affordable prices.
The
global vaccine market was valued at US$ 27 billion in the year 2012 and is
expected to grow to around US$ 32 billion by the end of year 2013. It is
expected that it will continue to grow at a CAGR of 11% to reach US$ 84 billion
by year 2022. Its future growth will be backed by vaccines developed through
technical advancements for emerging infectious diseases, cancers and allergies
etc. The market is also expected to see continuous improvement not only in
vaccine design but also in the delivery methodologies. There are several
critical vaccines which are currently under different stages of research and
development across the globe including vaccines for HIV, malaria, tuberculosis,
dengue etc. While the developed world is looking for these novel vaccines, the
developing world still requires increased availability of currently available
vaccines at affordable
prices.
Indian
Vaccine Market
India
continues to be a major supplier of vaccines. Leveraging its low cost vaccines,
India now caters for more than 40% of the global vaccine supply. Its own
vaccine market is estimated at around US$900 million and is expected to grow at
a CAGR of 23% from 2011 to 2013.
India is on the verge of being declared polio free country
as it has not recorded any case of polio since January 2011. The country has
now started focusing on increasing the immunization of pentavalent vaccine as
it has already included the same in the immunization schedule of eight states
by 2012. With its remarkable track record for producing low cost vaccines and
increasingly available funds for R&D, India is well positioned to take the
lead in the creation of innovative affordable high quality vaccines for the developing world and
cater to an ever growing demand of domestic as well as international market.
Global
Pharmaceutical Market
The
global pharmaceutical industry is going through a challenging time driven by
trends such as increasing regulatory compliance requirements, patents going on, lower R&D productivity,
pricing pressures from generic players and governments, globalization and
demographics.
The
global pharmaceuticals market reached US$962 billion, with a 2.4% growth in
year 2012, its lowest growth in the last ten years. It is expected to grow at a
CAGR of 5.3% to reach US$ 1.25 trillion by 2017. While the US, Japan and other
developed markets, coping with patent expiries are expected to grow by 1-4%,
the major growth will be driven by the pharmerging markets i.e. countries in
Asia, Africa, Latin America and Australia. The major factor for this growth is
due to the contribution made by 'pharmerging' markets, generics and the
biologics space. Amongst all developed markets, the US still continues to be
the largest, followed by Japan and Germany. However, what's noteworthy is that by
growing at a significantly
higher rate, the pharmerging markets are expected to account for over 30% of
the global market in 2017, up from 25% in 2012. In doing so, the pharmerging
markets will, in the aggregate, exceed the size of the US market in 2017. In
the year 2011 and 2012, few biggest blockbuster drugs lost patent protection.
The impact of patent expiration or the “patent cliff” will
continue by 2016. It is estimated that medicines that currently generate US$133
billion in US alone will lose patent protection and face generic competition.
Indian
Pharmaceutical Market
India
is one of the top five
pharmaceutical emerging markets globally. It has positioned itself as a front
runner in a wide range of specialties involving complex drugs' manufacture, development,
and technology. The Indian pharmaceutical industry is a highly knowledge based
industry which is growing steadily and plays a major role in the Indian
economy. The number of pharmaceutical companies are increasing their operations
in India.
The
Indian Pharmaceutical Industry is now ranked globally the 3rd largest in terms
of volume and 13th largest in terms of value. The total market size of around
Rs.1,233 bn includes domestic consumption market of around Rs.600 bn
(contributing around 49%) and the exports market being around Rs.633 bn
(contributing around 51%). The industry grew at a CAGR of 13% during the past five years and is expected to grow
at a robust CAGR of 15% during FY2012-17 given huge export potential coupled
with steady growth in the domestic formulation market.
The
future growth in the domestic pharma market will be driven mainly due to
increase in the penetration of medical facilities, increase in the prevalence
of chronic diseases, rising per capita income and increase in the health
insurance coverage. Growth in the exports of pharmaceutical products from India
will be driven by patent expiries of the major branded drugs across the world,
particularly in the US market. The growth in the US market will be led by
increasing generic penetration and healthy ANDA (Abbreviated New Drug
Application) pipeline of Indian pharma players.
Transplant
SBU
The
Transplant SBU contributes in prolonging the life of organ transplant recipients
and is responsible for marketing of the immuno-suppressive drugs in the therapy
area of post multi-organ transplantation primarily kidney, liver, heart
transplant etc. The SBU has carved a niche in superspecialty segment and
created a scientific
image and has achieved clear leadership in these segments.
Transplant
SBU's brand portfolio includes PanGraf (Tacrolimus), Panimun Bioral
(Cyclosporine), Mycept (Mycophenolate Mofetil), Mycept-S (Mycophenolate
Sodium), Siropan (Sirolimus), EverGraf (Everolimus), Imuza (Azathioprine),
VagaCyte (Valganciclovir) and Amphoject (Liposomal Amphotericin B).
This
SBU is planning to enter newer segments and introduce many new products. There
are plans to enter segments like anti-rejection segment to provide an end-to-end
solution for the organ transplant recipients. It is also exploring the
autoimmune segment in Hematology, Bone Marrow Transplant and Rheumatology where
there is immense scope for the use of the entire immunosuppressants and
anti-infective range.
Nephrology
SBU
Chronic
kidney disease (CKD) is a worldwide health problem and incidence of kidney
failure are rising significantly.
People suffering
from high blood pressure or diabetes are more prone to CKD. According to recent
trends, population of people suffering
from hypertension and diabetes is on a steep rise leading to rise in number of
patients diagnosed with CKD. Dialysis is primarily used as an optimal therapy
to compensate the lost kidney function in such patients.
The
Nephrology SBU is focused on providing “End to End solution in CKD management” thereby catering to need of
every CKD patient. The SBU is focused on renal anemia management, renal
nutrition and CKD-MBD (Mineral Bone Disorders) therapy.
The
SBU currently has a brand portfolio of Epotrust (Erythropoietin), Overcom (Iron
Sucrose), Alphadol (Alfacalcidol), SevBait and SevBait-DT (Sevelamer Carbonate),
Fosbait (Lanthanum Carbonate), Mimcipar (Cinacalcet Hydrochloride), K-bait
(Calcium Polystyrene Sulphonate), Proseventy and Renhold.
The
renal anemia segment is the largest contributor to the overall Nephrology
business having a share of 52% to the total business. Epotrust is amongst the
fastest growing brand of Erythropoietinin anemia market & oers a
wide range of SKU's (from PFS to Vials) catering the need of individual
patients.
OncoTrust
SBU
Cancer
is the second leading cause of death worldwide. Like the western world, the
peril of cancer has reached huge proportions in India with nearly 1 million new
cancer cases getting added every year to the existing cancer burden. The
increasing cancer incidence has witnessed the Indian oncology community fighting this battle against
cancer with more dedication.
Oncotrust,
the third Super Specialty SBU, is working with an object to make existing
cancer treatment more affordable
and also to develop NDDS cytotoxic drugs that enable the patients to get high
quality and affordable
medicine for better cancer management along with highest order of safety.
The
state-of-the-art Cytotoxic (anti-cancer) products facility at Baddi is going to
be fully operational in FY13-14 and demonstrates organizational commitment to
serve more patients in fighting
this dreadful disease. The new product launch by this SBU during the year
included Neupokine (Filgrastim PFS) and re-launch of PeggTrust (PegFilgrastim)
with new formulation matching international standards.
The
OncoTrust SBU has further consolidated its position as a profitable BU by continuing to grow
year on year. This SBU has 15 products encompassing the major therapy area in
cancer like lung, breast & colorectal cancer, gliomas, haematology and
supportive care.
Diacar
Alpha SBU
Today,
India is the diabetes and hypertension capital of the world and Indians are
further heading towards becoming the CAD (coronary artery disease) capital of
the world. WHO estimates that diabetes related mortality may increase up to 35%
by 2015. Today, India is poised at the ascending limb of epidemic diabetes. By
the end of year 2025, 70 million will suffer from diabetes and associated complications. Similarly,
cardiovascular segment is consistently showing high growth every year.
Diacar
Alpha SBU is the highest revenue contributing SBU of the company with dedicated
marketing and sales infrastructure for Diabetes and Cardiovascular therapy
management.
Diacar
Alpha focuses on Endocrinologists, Diabetologists, Cardiologists and Physicians
in a fiercely
competitive scenario and has achieved signicant leadership position in oral
anti-diabetic segment.
The flagship brand of Diacar Alpha is
Glizid-M (Gliclazide and Metformin) which is the No. 1 brand within the Company
across all SBUs.
The
brand portfolio of Diacar Alpha includes:
Oral
Hypoglycemic agents: Glizid, Glizid-MR & Glizid-MOD (Gliclazide Modied
release), Betaglim (Glimepiride), Betaglim-M (Glimepiride and Metformin),
Metlong and Metlong- DS (Metformin), Glizid Total and GlimTotal (Glimepiride,
Metformin and Pioglitazone);
Cardiovascular
agents: World's 1st Modified
Release and patented Ramipril - RAMY
(modified
release Ramipril with double peaks) developed through in-house patented
technology, Lower-A (Atorvastatin), Lower TG (Atorvastatin and Fenofibrate) and Tecbeta (Metoprolol
Succinate); and
Co-prescriptives:
Myelogen Forte (Methylcobalamin 1500 mcg, Alpha Lipoic Acid 100 mg, Enzogenol
50 mg, Vitamin B 6 3 mg and Folic Acid 1.5 mg) and Myelogen PG (Methylcobalamin
1500 mcg, Pregabalin 75 mg SR, Folic Acid 5 mg and Pyridoxine Hydrochloride 20
mg) which has gained wide spread usage and is growing fast. In co-morbid
conditions like Diabetic Peripheral Neuropathy.
Procare
SBU
Procare
SBU caters to chronic care segment of Orthopedic and Gastroenterology therapy
through focus on specific disease management with deep rooting in
osteoarthritis management. Within orthopedics, SBU's focus is on osteoporosis,
osteoarthritis and rheumatoid arthritis management and within the
gastroenterology focus is on constipation, anorectal disorders, acid-peptic
disorders and product range in liver disease management.
This
SBU promotes a portfolio of brands with special focus on Orthopaedicians,
Surgeons and Gastroenterologists along with Consulting and General Physicians.
The brand portfolio of Procare includes:
·
Gastrointestinal: Sitcom (Euphorbia
Prostrata) Tablets and Cream, Sitcom Forte (Euphorbia Prostrata & Calcium
Dobesilate) tablets, Sitcom LD (Euphorbia Prostrata and Lidocaine) cream,
Livoluk (Lactulose), Livoluk Fibre (Lactulose and Ispaghula husk), Gush
(Lactitol Monohydrate and Ispaghula Husk), ILAone (Ilaprazole) and ODpep
(Pantoprazole and Domperidone);
·
Chronic liver diseases: Uciro (Ursodeo
Xycholic Acid);
·
Anti-osteoporosis: Vacosteo (Zoledronic
acid, 3rd generation injectable bisphosphonate), Alphadol-C,
(Alfacalcidol), Calcom (Calcium Carbonate, Calcitriol and Zinc) and Monthiba
(ibandronic Acid);
·
Anti-arthritis : Willgo (Nimesulide),
Kondro OD (Glucosamine) and Kondro Acute (Glucosamine and Diacerein), Kondroex
(Collagen peptide, Glucosamine and Vitamin C) andKondrogen (Collagen peptide);
·
Pain Management: Nimulid-MR (Nimesulide
and Tizanidine), Delupa-P (Aceclofenac & Paracetamol), Delupa-TH
(Aceclofenac and Thiocolchicoside), Delupa-SP (Aceclofenac and Serratiopeptidase),
Delupa-CR (Aceclofenac) and Dolzero; and
·
Gout and Hyperuricemia: Febarto
(Febuxostat) launched during the year.
Sitcom
has evolved as the first
choice among antihemorrhoidals within Gastroenterologists and Surgeons. This
success has motivated the SBU to launch innovative formulations in the form of
Sitcom Forte tablets and Sitcom LD cream for the first time in India.
Growcare
SBU
Growcare
SBU focuses on gastrointestinal (GI), anti hemorrhoidal, cough & cold, vitamins
and minerals and pain management therapies.
Committed
to reduce the burden of these diseases, Growcare marks the Company's presence
in therapy areas like anorectal disorders (piles and hemorrhoids),
gastrointestinal, respiratory (cough, cold and allergy), anti-infectives, pain
relievers, vitamins and minerals. The dierent specialties serviced by Growcare
SBU are General Physicians, Consulting Physicians, ENT Surgeons, Pediatricians
and General
Surgeons.
The
brand portfolio of Growcare includes:
·
Anti hemorrhoidal: Thank OD Tablet
& Cream, Thank OD Forte and LD Cream;
·
Anti-infective: Ocimix (Ornidazole),
ValueCef (Cefixime),
ValueCef-O (Cefixime
and Ofioxacin);
·
Cough, Cold and Fever: Cough syrups
range Toff MD,
Toff DC and Toff expectorant, Orangemol
Suspension; Tecpara (technological advanced Paracetamol) an in house R&D
patented product), Tecpara-AC and Tecpara-D;
·
Pain Management: Nimulid &
Nimulid-MD (Mouth dissolving), Nimulid SP and Nimulid-HF; Gastrointestinal:
EnBa, EnBa-Rab, FiberFOS, HiFibre and Livoluk kid;
·
Vitamins and Minerals: Wholesum; and
·
Anti TB: Myser (Cycloserine) &
Myobid (Ethionamide).
Brands
Review
Over
the years, Panacea Biotec has established leading brands that enjoy top of the
mind recall by the medical fraternity. The Company's brands command excellent
market share in their therapeutic segments. According to AIOCD AWACS (MAT
JUN'13) Sales value, Panacea Biotec is among Top 50 companies in the Indian
Pharmaceutical Market with Nephrologists, Dentists, Orthopaedicians and Diabetologists
giving the best support. As per Stockist Secondary Audit of AIOCD AWACS (MAT
JUN'13), Glizid-M stands at 206th rank among top brands in the Indian
Pharmaceutical market and retain number one position within its category.
Glizid
group grew by 18% during the year maintaining its leadership among Gliclazide
sulphonyl urea market.
Euphorbia
prostrate (Sitcom and Thank OD) family also grew by 18% and is number one
prescribed therapy in Hemorrhoids management. Mycept and PanGraf grew by 12%
and 13% respectively in FY 12-13, are also leading brands in the Organ
Transplantation segment where the Company enjoys market leadership position.
FIXED ASSTES
· Land – Leasehold
· Land – Freehold
· Buildings
· Leasehold
Improvements
· Plant and
Machinery
· Vehicles
· Furniture and
Fixtures
· Office Equipments
· Computer
Equipments
UNAUDITED FINANCIAL
RESULTS FOR THE QUARTER / HALF YEAR ENDED DECEMBER 30.09.2013
(Rs.
In Millions)
|
Particulars |
Quarter Ended ( Unaudited) |
Half Year Ended ( Unaudited) |
|
|
|
30.09.2013 |
30.06.2013 |
30.09.2013 |
|
PART – I |
|
|
|
|
1. Income from
operations |
|
|
|
|
a. Net sales / income from operations (net of excise duty) |
1007.300 |
1044.600 |
2051.900 |
|
b. Other operating income |
93.600 |
131.700 |
225.300 |
|
Total income from operations (net) |
1100.900 |
1176.300 |
2277.200 |
|
2. Expenditure |
|
|
|
|
a. Cost of materials consumed |
721.200 |
639.000 |
1360.200 |
|
b. Purchase of stock in trade |
59.400 |
68.900 |
128.300 |
|
c. (Increase)/Decrease in inventories |
(632.800) |
(313.900) |
(946.700) |
|
d. Employees benefits expense |
359.300 |
367.200 |
726.500 |
|
e. Depreciation and amortisation expense |
185.000 |
189.600 |
374.600 |
|
g. Other expenses |
587.100 |
506.400 |
702.700 |
|
Total expenses |
1279.200 |
1457.200 |
2736.400 |
|
3. (Loss)/Profit from operations before other income, finance cost, foreign exchange fluctuation gain/ (loss) & exceptional items (1-2) |
(178.300) |
(280.900) |
(459.200) |
|
4. Other income |
2.000 |
2.500 |
4.500 |
|
5. (Loss)/Profit from ordinary activities before finance cost, foreign exchange fluctuation gain / (loss) & exceptional items (3+4) |
(176.300) |
(278.400) |
(454.700) |
|
6. Finance cost |
368.200 |
321.200 |
689.400 |
|
7. Foreign exchange fluctuation loss/ (gain) |
18.200 |
9.600 |
27.800 |
|
8. (Loss)/Profit
from ordinary activities after finance cost before exceptional items (5-6-7) |
(562.700) |
(609.200) |
(1171.900) |
|
9. Exceptional income/(exp) - Refer note 7(i) & (ii) |
-- |
-- |
-- |
|
10. Profit / (loss)
from ordinary activities before tax (8+9) |
(562.700) |
(609.200) |
(1171.900) |
|
11. Tax expenses |
-- |
-- |
-- |
|
12. Net profit /
(loss) from ordinary activities after tax (10-11) |
(562.700) |
(609.200) |
(1171.900) |
|
13. Extraordinary items (net of tax expenses) |
|
|
|
|
14. Net profit /
(loss) for the period (12-13) |
|
|
|
|
15. Paid up equity share capital (face value of Re.1 per share) |
61.300 |
61.300 |
61.300 |
|
16.Reserves excluding revaluation reserves |
-- |
-- |
-- |
|
17.Earning per
share (EPS) |
|
|
|
|
- Basic (in Rs.) |
(9.18) |
(9.95) |
(19.13) |
|
- Diluted (in Rs.) |
(9.18) |
(9.95) |
(19.13) |
|
A. Particulars of shareholding |
|
|
|
|
1. Public
shareholding |
|
|
|
|
- No. of shares |
15,427,192 |
15,426,992 |
15,427,192 |
|
- Percentage of shareholding |
25.19 |
25.19 |
25.19 |
|
2. Promoters and
promoter group Shareholding |
|
|
|
|
a) Pledge /
encumbered |
|
|
|
|
- No. of shares |
- |
- |
- |
|
- % of Shares (as a % of the total shareholding of promoter & promot |
- |
|
|
|
- % of Shares (as a % of the total share capital of the Company) |
- |
|
|
|
b) Non-encumbered |
|
|
|
|
- Number of shares |
45,823,554 |
45,823,754 |
45,823,554 |
|
- % of Shares (as a % of the total shareholding of promoter & promoter group) |
100.00 |
100.00 |
100.00 |
|
- % of Shares (as a % of the total share capital of the Company) |
74.81 |
74.81 |
74.81 |
|
B. Investor
complaints |
|
|
|
Pending at the beginning of the quarter |
|
- |
|
Received during the quarter |
|
- |
|
Disposed of during the quarter |
|
- |
|
Remaining unresolved at the end of the quarter |
|
- |
UNAUDITED FINANCIAL RESULTS
FOR THE QUARTER / HALF YEAR ENDED DECEMBER 30.09.2013
|
Particulars |
30.09.2013 (Unaudited) |
30.06.2013 (Unaudited) |
30.09.2013 (Unaudited) |
|
|
|
|
|
|
1. Segment revenue |
|
|
|
|
(a) Vaccines |
57.300 |
280.400 |
337.700 |
|
(b) Formulations |
986.400 |
815.800 |
1802.200 |
|
(c) Research & development |
57.500 |
79.800 |
137.300 |
|
(d) Unallocated |
(0.300) |
0.300 |
-- |
|
Gross sale/Income
from operation |
1100.900 |
1176.300 |
2277.200 |
|
Less : Inter segment revenue |
-- |
-- |
-- |
|
Net sales/income
from operations |
1100.900 |
1176.300 |
2277.200 |
|
2.Segment results |
|
|
|
|
Profit (+)/ loss
(-) before tax and interest from each segment |
|
|
|
|
(a) Vaccines |
(50.200) |
(141.100) |
(289.600) |
|
(b) Formulations |
225.300 |
141.900 |
367.200 |
|
(c) Research & development |
(203.800) |
(146.000) |
(349.800) |
|
Total |
(28.700) |
(145.200) |
(173.900) |
|
Less : i) Finance cost |
368.200 |
321.200 |
689.400 |
|
ii) Other un-allocated expenditure net off un-allocated income |
165.800 |
142.800 |
308.600 |
|
Total profit before
tax |
(562.700) |
(609.200) |
(1171.900) |
|
3. Capital Employed |
|
|
|
|
(Segment
assets-segment liabilities) |
|
|
|
|
(a) Vaccines |
5919.600 |
5535.400 |
5919.600 |
|
(b) Formulations |
2509.600 |
2522.600 |
2509.600 |
|
(c) Research & development |
1784.500 |
1853.300 |
1784.500 |
|
(d) Unallocated |
(6001.100) |
(5049.100) |
(6001.100) |
|
Total capital
employed |
4212.600 |
4862.200 |
4212.600 |
STANDALONE STATEMENT OF ASSETS AND
LIABILITIES
(Rs. In Millions)
|
SOURCES OF FUNDS |
30.09.2013 (Unaudited) |
|
I.
EQUITY
AND LIABILITIES |
|
|
(1)Shareholders' Funds |
|
|
(a) Share Capital |
61.300 |
|
(b) Reserves & Surplus |
4151.300 |
|
Total Shareholders’
Funds |
4212.600 |
|
|
|
|
(2)
Non-Current Liabilities |
|
|
(a) long-term borrowings |
5571.300 |
|
(b) Deferred tax liabilities (Net) |
0.000 |
|
(c) Other long term liabilities |
25.600 |
|
(d) long-term provisions |
22.400 |
|
Total Non-current Liabilities (3) |
5619.300 |
|
|
|
|
(3) Current Liabilities |
|
|
(a) Short term borrowings |
3693.900 |
|
(b) Trade payables |
1970.500 |
|
(c) Other current
liabilities |
1309.400 |
|
(d) Short-term provisions |
114.300 |
|
Total Current Liabilities (4) |
7088.100 |
|
|
|
|
TOTAL |
16920.000 |
|
|
|
|
II.
ASSETS |
|
|
(1) Non-current assets |
|
|
(a) Fixed Assets |
|
|
1)
Tangible
Assets |
8809.200 |
|
2)
Intangible
Assets |
362.800 |
|
3)
Capital
work-in-progress |
190.100 |
|
4)
Intangible
assets under development |
85.800 |
|
(b) Non-current Investments |
2648.100 |
|
(c) Deferred tax assets (net) |
0.000 |
|
(d) Long-term Loan and Advances |
915.200 |
|
(e) Other Non-current assets |
0.700 |
|
Total Non-Current Assets |
13011.900 |
|
|
|
|
(2) Current assets |
|
|
(a) Current investments |
0.000 |
|
(b) Inventories |
2888.700 |
|
(c) Trade receivables |
732.800 |
|
(d) Cash and cash
equivalents |
52.100 |
|
(e) Short-term loans and
advances |
210.700 |
|
(f) Other current assets |
23.800 |
|
Total Current Assets |
3908.100 |
|
|
|
|
TOTAL |
16920.000 |
NOTES:
1.
The above financial results were
reviewed by the Audit Committee of the Board and approved by the Board of Directors
at their meetings held on November 13, 2013 and November 14, 2013,
respectively.
2.
Tax expense includes income tax and
deferred tax.
3.
During the quarter, the Company has
launched one new product namely Kondrogen Powder (Osteoarthiritis).
4.
In view of lack of any business
activity, the Company’s wholly owned subsidiary (WOS), viz. Panacea Biotec FZE
in UAE has been closed down and amount of Rs 4.91 lacs as investment written
off, during the quarter.
5.
During the quarter, a net amount of
Rs.240 Lac has been received back from Company's subsidiary NewRise Healthcare
Private Limited, as refund of advance share application money.
6.
During the quarter, the Company has
decided to dispose of its entire shareholding in its WOS, Lakshmi and Manager
Holdings Limited (LMH) at its fair value of Rs. 12.37 Crore and the process for
such disposal of shares has been initiated.
7.
As regards Auditors’ observations in
their report on the audited accounts for the Financial Year 2012-13 and in
their limited review report on the above results.
i.
Due to the absence of profits during
the financial year 2012-13, the total remuneration of the Managing/Joint
Managing and Whole Time Director had exceeded the ceiling prescribed in Section
II of Part II of Schedule XIII to the Companies Act, 1956. Accordingly,
applications for protection/approval of the Central Government in respect of
excess remuneration for financial years 2012-13 have been filed and requisite
approval is awaited.
ii.
Due to the absence of profits for the current financial
year, the managerial remuneration of Rs 92.4 lac paid during the period, may
exceed the limits specified under relevant provisions of the Companies Act,
1956. The Company has already filed the requisite applications for obtaining
approval from Central Government for payment of remuneration.
8.
During the quarter ended September 30,
2011, World Health Organization (WHO) had delisted Company’s DTP-based
combination vaccines from its list of pre-qualified vaccines. During the month
of February/March, 2013 Auditors from WHO and UNICEF visited the Company's
vaccine facilities at Lalru (Punjab) and Baddi (H.P.) with the objective of
re-evaluation of the acceptability in principle of Pentavalent Vaccine (DTP-Hep
B-Hib) produced by Panacea Biotec for purchase by United Nations Agencies. The
company has made substantive efforts since September 2011 and has revamped the
whole Quality Management System at its Lalru and Baddi sites enabling it to get
pre-qualified by WHO once again
During the quarter, WHO completed
evaluation process of pre-qualification (PQ) of Pentavalent Vaccine
(Easyfive-TT) and has been pre-qualified by WHO for supply of such vaccine on
October 2, 2013. WHO has also informed UNICEF of its decision to pre-qualify
such vaccine for considering the Company for giving commercial orders. The
Company is now in discussion with UNICEF to secure the business of such vaccine
at the earliest and is expecting to receive orders shortly and start supply of
such vaccine in due course. This approval would also now enable the company to
participate in future bids by UNICEF and attempt to regain market share for its
vaccine business. The Company has also started participating in tenders of
various other countries which procure pentavalent vaccine directly. The Company
is confident that it will be able to secure supplies of pentavalent vaccine
from UNICEF; and other customers and the stock of Rs.157.200 Millions and
Rs.1149.200 Millions of raw and packing material and finished goods (including
work in progress), respectively as at September 30, 2013 pertaining to these
vaccines will be utilised/sold In due course.
9.
The Company's accumulated losses have
resulted in erosion of more than fifty percent of its peak net worth calculated
as per the provisions of Sick Industrial Companies (Special Provisions) Act,
1985 (SICA). As a result, the Company falls under the category of’ Potentially
Sick Industrial Company ‘in terms of Section 23 of the SICA. Accordingly, an
Extra-Ordinary General Meeting of the members is scheduled to be held on
November 22, 2013 for considering such erosion and the fact of such erosion and
measures initiated to improve financial condition shall be reported to the
Board for Industrial and Financial Restructuring (“BIFR”) within the stipulated
period. The measures taken by the company which mitigate risk of going concern
include the following.
i.
Supply to UNICEF/other customers of
pentavalent vaccine (as explained in note no. 8 above),
ii.
Certain strategic alliances with
foreign collaborators for supply of vaccines and pharma products, etc.
iii.
Certain concessions being proposed to
lenders towards its debt liabilities.
iv.
Launching its first product Tacrolimus
in USA in Dec 2012 and filing of four more ANDAs in USFDA.
Management is confident that
with this above measures and continuous efforts to improve the business, it
would be able to generate sustainable cash flow, and recover and recoup the
erosion in its net worth through profitable operations, discharge its
short-term and long term liabilities and continue as a going concern.
10.
As at March 31, 2013, an amount of
Rs.694.700 Millions (previous year Rs.654.300 Millions) including interest of
Nil (previous year Rs.36.200 Millions) is receivable from Rees Investments Ltd.
Pursuant to the diminution in the value of investment and losses in its
subsidiaries, an amount of Rs.114.800 Millions (Previous year Rs.421.400
Millions) has been provided for as ‘Provision for bad and doubtful advances’
which has been shown as an exceptional expense.
11.
In terms of the Accounting Standard -16
“Borrowing Costs”, the foreign exchange differences arising from foreign
currency borrowings to the extent regarded as an adjustment to interest cost
were treated as borrowing cost. In pursuance of the clarification issued by
Ministry of Corporate Affairs vide its circular dated August 9, 2012, the
Company accounted for the aforesaid foreign exchange differences arising from
foreign currency borrowings as per AS-11 - “The Effects of Changes in Foreign
Exchange Rates” in the current year. Consequent to the above, exchange
differences of Rs.173.100 Millions which was earlier recognized as borrowing
cost pertaining to the financial year 2011-12 were reversed and shown as an
exceptional income in the financial year 2012-13. Out of the aforesaid amount
of exchange differences of Rs.173.100 Millions, Rs.135.200 Millions were
capitalized to the cost of fixed assets and Rs.37.900 Millions were accumulated
in the “foreign currency monetary item translation difference account”.
12.
CARE has maintained credit rating
pertaining to Long term bank facilities as 'CARE B1 (Single B) (under credit
watch) and 'CARE A4' (A four) (under credit watch) in regard to Short term bank
facilities.
13.
The necessary certificate in respect of
above results in terms of requirement of clause 41 of the listing agreement,
has been placed before the Board of Directors.
14.
The figure of the quarter ended
September 30, 2013 are the balancing figures between half year ended and first
quarter ended figures of the financial year 2013-14.
15.
Previous period / year figures have
been regrouped/ reclassified to make them comparable with those of current
Quarter.
CMT REPORT (Corruption, Money Laundering & Terrorism]
The Public Notice information has been collected from various sources
including but not limited to: The Courts,
1] INFORMATION ON
DESIGNATED PARTY
No exist designating subject or any of its beneficial owners, controlling
shareholders or senior officers as terrorist or terrorist organization or whom
notice had been received that all financial transactions involving their assets
have been blocked or convicted, found guilty or against whom a judgement or
order had been entered in a proceedings for violating money-laundering,
anti-corruption or bribery or international economic or anti-terrorism sanction
laws or whose assets were seized, blocked, frozen or ordered forfeited for
violation of money laundering or international anti-terrorism laws.
2] Court Declaration :
No exist to suggest that subject is or was
the subject of any formal or informal allegations, prosecutions or other
official proceeding for making any prohibited payments or other improper
payments to government officials for engaging in prohibited transactions or
with designated parties.
3] Asset Declaration :
No records exist to suggest that the property or assets of the subject
are derived from criminal conduct or a prohibited transaction.
4] Record on Financial
Crime :
Charges or conviction
registered against subject: None
5] Records on Violation of
Anti-Corruption Laws :
Charges or
investigation registered against subject: None
6] Records on Int’l
Anti-Money Laundering Laws/Standards :
Charges or
investigation registered against subject: None
7] Criminal Records
No
available information exist that suggest that subject or any of its principals
have been formally charged or convicted by a competent governmental authority
for any financial crime or under any formal investigation by a competent
government authority for any violation of anti-corruption laws or international
anti-money laundering laws or standard.
8] Affiliation with
Government :
No record
exists to suggest that any director or indirect owners, controlling
shareholders, director, officer or employee of the company is a government
official or a family member or close business associate of a Government
official.
9] Compensation Package :
Our market
survey revealed that the amount of compensation sought by the subject is fair
and reasonable and comparable to compensation paid to others for similar
services.
10] Press Report :
No press reports / filings exists on
the subject.
CORPORATE GOVERNANCE
MIRA INFORM as part of its Due Diligence do provide comments on
Corporate Governance to identify management and governance. These factors often
have been predictive and in some cases have created vulnerabilities to credit
deterioration.
Our Governance Assessment focuses principally on the interactions
between a company’s management, its Board of Directors, Shareholders and other
financial stakeholders.
CONTRAVENTION
Subject is not known to have contravened any existing local laws,
regulations or policies that prohibit, restrict or otherwise affect the terms
and conditions that could be included in the agreement with the subject.
FOREIGN EXCHANGE RATES
|
Currency |
Unit
|
Indian Rupees |
|
US Dollar |
1 |
Rs.62.28 |
|
|
1 |
Rs.103.67 |
|
Euro |
1 |
Rs.85.18 |
INFORMATION DETAILS
|
Report Prepared
by : |
NKT |
SCORE & RATING EXPLANATIONS
|
SCORE FACTORS |
RANGE |
POINTS |
|
HISTORY |
1~10 |
4 |
|
PAID-UP CAPITAL |
1~10 |
3 |
|
OPERATING SCALE |
1~10 |
4 |
|
FINANCIAL CONDITION |
|
|
|
--BUSINESS SCALE |
1~10 |
4 |
|
--PROFITABILIRY |
1~10 |
- |
|
--LIQUIDITY |
1~10 |
4 |
|
--LEVERAGE |
1~10 |
3 |
|
--RESERVES |
1~10 |
4 |
|
--CREDIT LINES |
1~10 |
3 |
|
--MARGINS |
-5~5 |
- |
|
DEMERIT POINTS |
|
|
|
--BANK CHARGES |
YES/NO |
YES |
|
--LITIGATION |
YES/NO |
NO |
|
--OTHER ADVERSE INFORMATION |
YES/NO |
NO |
|
MERIT POINTS |
|
|
|
--SOLE DISTRIBUTORSHIP |
YES/NO |
NO |
|
--EXPORT ACTIVITIES |
YES/NO |
NO |
|
--AFFILIATION |
YES/NO |
YES |
|
--LISTED |
YES/NO |
YES |
|
--OTHER MERIT FACTORS |
YES/NO |
YES |
|
DEFAULTERS |
|
|
|
--RBI |
YES/NO |
NO |
|
--EPF |
YES/NO |
NO |
|
|
|
|
|
TOTAL |
|
29 |
This score serves as a reference to assess SC’s credit risk and
to set the amount of credit to be extended. It is calculated from a composite
of weighted scores obtained from each of the major sections of this report. The
assessed factors and their relative weights (as indicated through %) are as
follows:
Financial
condition (40%) Ownership
background (20%) Payment
record (10%)
Credit history
(10%) Market trend
(10%) Operational
size (10%)
RATING EXPLANATIONS
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
>86 |
Aaa |
Possesses an extremely sound financial base with the strongest
capability for timely payment of interest and principal sums |
Unlimited |
|
71-85 |
Aa |
Possesses adequate working capital. No caution needed for credit
transaction. It has above average (strong) capability for payment of interest
and principal sums |
Large |
|
56-70 |
A |
Financial & operational base are regarded healthy. General
unfavourable factors will not cause fatal effect. Satisfactory capability for
payment of interest and principal sums |
Fairly Large |
|
41-55 |
Ba |
Overall operation is considered normal. Capable to meet normal
commitments. |
Satisfactory |
|
26-40 |
B |
Capability to overcome financial difficulties seems comparatively below
average. |
Small |
|
11-25 |
Ca |
Adverse factors are apparent. Repayment of interest and principal sums
in default or expected to be in default upon maturity |
Limited with
full security |
|
<10 |
C |
Absolute credit risk exists. Caution needed to be exercised |
Credit not
recommended |
|
-- |
NB |
New Business |
-- |
This report is issued at your request without any
risk and responsibility on the part of MIRA INFORM PRIVATE LIMITED (MIPL)
or its officials.