MIRA
INFORM REPORT
|
Report Date : |
27.02.2014 |
IDENTIFICATION DETAILS
|
Name : |
MSN LABORATORIES PRIVATE LIMITED (w.e.f.27.02.2013) |
|
|
|
|
Formerly Known
As : |
MSN LABORATORIES LIMITED |
|
|
|
|
Registered
Office : |
MSN House, Plot No.C-24, Sanath Nagar Industrial Estate, Sanath Nagar,
|
|
|
|
|
Country : |
|
|
|
|
|
Financials (as
on) : |
31.03.2012 |
|
|
|
|
Date of
Incorporation : |
27.08.2003 |
|
|
|
|
Com. Reg. No.: |
01-041583 |
|
|
|
|
Capital
Investment / Paid-up Capital : |
Rs.52.000 Millions |
|
|
|
|
CIN No.: [Company Identification
No.] |
U24239AP2003PTC041583 |
|
|
|
|
TAN No.: [Tax Deduction &
Collection Account No.] |
HYDM02459C |
|
|
|
|
PAN No.: [Permanent Account No.] |
AADCM6283F |
|
|
|
|
Legal Form : |
Private Limited Liability Company |
|
|
|
|
Line of Business
: |
Manufacturer, Exporter and Importer of Pharmaceutical Bulk Drugs and Organic Intermediates Salmetrol, Rosuvastatin Calcium, Ezetimibe etc |
|
|
|
|
No. of Employees
: |
60 (Approximately) |
RATING & COMMENTS
|
MIRA’s Rating : |
A (62) |
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
56-70 |
A |
Financial & operational base are regarded healthy. General
unfavourable factors will not cause fatal effect. Satisfactory capability for
payment of interest and principal sums |
Fairly Large |
|
Maximum Credit Limit : |
USD 8222000 |
|
|
|
|
Status : |
Good |
|
|
|
|
Payment Behaviour : |
Regular |
|
|
|
|
Litigation : |
Clear |
|
|
|
|
Comments : |
Subject is an established company having fine track record. Management has filed the latest financials of 2013, with government department,
however the same could not be made available from them. As per available financials, the company has achieved better increase
in its sales turnover a well as net profitability during 2012. Financial
position of the company appears to be sound. Directors are reported to be
well experienced and knowledgeable businessmen. Trade relations are reported as trustworthy. Business is active.
Payments are reported to be regular and as per commitments. The company can be considered good for business dealings at usual
trade terms and conditions. |
NOTES:
Any query related to this report can be made
on e-mail : infodept@mirainform.com
while quoting report number, name and date.
ECGC Country Risk Classification List – December 1, 2013
|
Country Name |
Previous Rating (30.09.2013) |
Current Rating (01.12.2013) |
|
India |
A1 |
A1 |
|
Risk Category |
ECGC
Classification |
|
Insignificant |
A1 |
|
Low |
A2 |
|
Moderate |
B1 |
|
High |
B2 |
|
Very High |
C1 |
|
Restricted |
C2 |
|
Off-credit |
D |
INDIAN ECONOMIC OVERVIEW
The worst is over for India’s economy with gross domestic product likely
to expand 5 %to 5.5 % this year and more than 6 % in 2015, according to Moody’s
Analytics. Concerns over the rupee and current account deficit are under
control, said the agency. Ratings firm Crisil has forecast 6 % growth for
2014/15 up from the estimated 4.8 % for 2013/14. Total economic growth,
infrastructure bottlenecks and lack of transparency and consistency in foreign
direct investment policies seem to have taken a toll on India’s attractiveness
as an investment destination, says an Ernst & Young survey. Projects
with FDI component fell 16.4 % across the globe in 2012 from the previous
year. The drop in India was steeper at 21 %. State run carrier Air India
is doling out free tickets to its 24000 employees, even as it expects to incur
a loss of Rs 39000 mn this financial year and has a debt of Rs 350000 mn.
550000 number of jobs generated across India in 2013, a fall of 0.4 % as
compared to with a year earlier. The National Capital Region has a
one-fourth share in total jobs created, according to a study by industry lobby
group Assochem, Banks, real estate, automobile and telecommunications sectors
are showing a rise of job creation. $ 805 mn investments by venture capital
firms in India during 2013, registering a drop of about 18 % over the previous
year. The Information Technology and IT-Enabled Services Industry retained
its status as the favourable venture capital investors in 2013. Pakistan has
temporarily banned gold imports for the second time in six months, as it tries
to stem smuggling into India. India’s import duty on gold is 10 % and curbs on
purchases have dried up legal imports into what used to be the world’s biggest
bullion buyers. The World Gold Council puts the amount smuggled into India at
upto 200 tonnes in 2013. The Reserve Bank of India has proposed that unclaimed
bank deposits estimated to be about Rs 35000 mn be used for education and
awareness among depositors. According to the plan, deposits that have not
been claimed for at least 10 years will be transferred to the scheme.
EXTERNAL AGENCY RATING
|
Rating Agency Name |
CRISIL SME |
|
Rating |
SME1 |
|
Rating Explanation |
Highest credit quality and negligible credit
risk. |
|
Date |
28.11.2013 |
RBI DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available RBI Defaulters’ list.
EPF (Employee Provident Fund) DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available EPF (Employee Provident Fund) Defaulters’ list as of
31-03-2012.
INFORMATION DENIED
Management Non Co-operative (91-40-30438600)
LOCATIONS
|
Registered
Office / Corporate Office: |
MSN House, Plot No.C-24, Sanath Nagar Industrial Estate, Sanath Nagar,
Hyderabad – 500 018, Andhra Pradesh, India |
|
Tel. No.: |
91-40-30438600 |
|
Fax No.: |
91-40-30438638 / 30438643 / 30438719 |
|
E-Mail : |
|
|
Website : |
|
|
|
|
|
Factory 1 : |
SY No 317 and 323, Rudraram Village, Patancheru Mandal, Medak – 502
329, Andhra Pradesh, India |
|
Tel. No.: |
91-84-55 220372 |
|
Fax No.: |
91-84-58 279938 |
|
E-Mail : |
|
|
|
|
|
Factory 2 : |
Plot No.42, Anrich Industrial Estate, Bollarum, Medak – 502 325, Andhra
Pradesh, India |
|
Tel No.: |
91-84-58 279936 /37 |
|
Fax No.: |
91-84-58 279938 |
|
Email: |
formulation_at_msnlabs.com
|
|
|
|
|
Factory 3 : |
SYS No. 14, Gaddapotharam Village, Jinnaram Mandal, Medak – 502 319
District, Andhra Pradesh, India |
|
Tel. No.: |
91-8458-277090 |
|
Fax No.: |
91-8458-277085 |
|
|
|
|
Factory 4 : |
Sy No-50, Kardanur (V), Patancheru (M), Medak – 502 300, Andhra
Pradesh, India |
|
Tel. No.: |
91-08458-331200 |
|
Email: |
api_at_msnlabs.com (replace _at_ with @ before emailing) |
DIRECTORS
AS ON 30.09.2013
|
Name : |
Dr. Manne Satyanarayana Reddy |
|
Designation : |
Managing Directors |
|
Address : |
8-3-167/D/16, Kalyan Nagar, Phase – I, Near A. G. Colony, Hyderabad – 500 038, Andhra Pradesh, India |
|
Date of Birth/Age : |
18.04.1957 |
|
Date of Appointment : |
27.08.2003 |
|
DIN No.: |
01657567 |
|
|
|
|
Name : |
Mrs. Manne Laxmi Prasuna |
|
Designation : |
Directors |
|
Address : |
8-3-167/D/16, Kalyan Nagar, Phase – I, Near A. G. Colony, Hyderabad – 500 038, Andhra Pradesh, India |
|
Date of Birth/Age : |
06.08.1968 |
|
Date of Appointment : |
27.08.2003 |
|
DIN No.: |
00954334 |
|
|
|
|
Name : |
Mr. Bharat Chintapally Bharat |
|
Designation : |
Directors |
|
Address : |
8-3-167/D/16, Kalyan Nagar, Phase – I, Near A. G. Colony, |
|
Date of Birth/Age : |
30.06.1969 |
|
Date of Appointment : |
01.02.2007 |
|
DIN No.: |
01591473 |
MAJOR SHAREHOLDERS
AS ON 30.09.2013
|
Names of Shareholders |
No. of Shares |
|
Manne Satyanarayana Reddy |
2526800 |
|
Manne Laxmi Prasuna |
427800 |
|
Manne Venkat Ram Reddy |
120000 |
|
Thatikonda Seetha |
1000 |
|
Thatikonda Srinivas |
1000 |
|
Chintapally Anjani |
1000 |
|
Chintapally Janardhan Reddy |
132000 |
|
Manne Venkat Pratap Reddy |
124000 |
|
Manne Jeevan Reddy |
120000 |
|
Manne Srinivasa Reddy |
120000 |
|
Manne Nirmala Devi |
66000 |
|
Manne Geetha |
55000 |
|
Thatikonda Sreedhar Reddy |
1000 |
|
Srinivasan Tirumalarajan |
72000 |
|
Sajja Eswaraiah |
72000 |
|
Challa N.V. Ravi Kumar |
72000 |
|
Muppa Kishore Kumar |
72000 |
|
Chintapally Bharat Reddy |
700000 |
|
Manne Madhavi |
1200 |
|
Thatikonda Varalakshmamma |
1000 |
|
Chintapally Vasudha |
1000 |
|
Thatikonda Raja Reddy |
1000 |
|
Manne Swathi |
1200 |
|
Chakilam Nagaraju |
456000 |
|
Manne Shrawya Reddy |
55000 |
|
Total |
5200000 |
Equity Share Break up (Percentage of Total Equity)
AS ON 30.09.2013
|
Category |
Percentage |
|
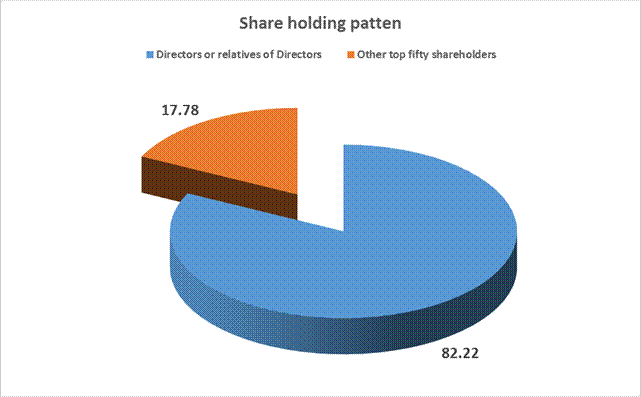
Directors or relatives of Directors |
88.22 |
|
Other top fifty shareholders |
17.78 |
|
Total |
100.00 |
BUSINESS DETAILS
|
Line of Business : |
Manufacturer, Exporter and Importer of Pharmaceutical Bulk Drugs and Organic Intermediates Salmetrol, Rosuvastatin Calcium, Ezetimibe etc |
||||||||
|
|
|
||||||||
|
Products : |
|
PRODUCTION STATUS [AS ON 31.03.2011]
|
Particulars |
Unit |
Installed
Capacity |
Actual
Production |
|
APIs and Intermediates |
Kgs. |
38000 |
37740 |
|
Formulations* [Tablets and Capsules] |
Nos. in Millions |
450 |
305561 |
NOTE:
·
Installed capacities as certified by the management
but not verified by the auditors.
·
* Sale quantities are after adjustment of samples.
GENERAL INFORMATION
|
No. of Employees : |
60 (Approximately) |
||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||
|
Bankers : |
·
Axis Bank Limited, Corporate Banking Branch, 1st
Floor, Pullareddy Buildings, Greenlands, Begumpet, Hyderabad – 500 016,
Andhra Pradesh, India ·
ICICI Bank Limited, Landmark Race Cource Circle,
Alkapuri, Baroda – 390 015, Gujarat, India |
||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||
|
Facilities : |
|
|
|
|
|
Banking
Relations : |
-- |
|
|
|
|
Financial Institution : |
3i Infotech
Trusteeship Services Limited, 3rd To 6th Floor, International
Infotech Park, Tower No.5, Vashi Railway Station Complex, Vashi, Navi Mumbai
- 400703, Maharashtra, India |
|
|
|
|
Auditors : |
|
|
Name : |
B.R.M. Reddy and Company Chartered Accountants |
|
Address : |
Lane Adj. To ITC Kakatiya, China Balreddy Building, Begumpet – 500
016, Hyderabad, India |
|
Pan No.: |
AADFB7698A |
|
|
|
|
Associates : |
·
MSN Pharmachem Private Limited ·
Maithri Laboratories Private Limited ·
MSN Organics Private Limited ·
MSN Research and Development Private Limited |
CAPITAL STRUCTURE
AS ON 30.09.2013
Authorised Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
10000000 |
Equity Shares |
Rs. 10/- each |
Rs.100.000 Millions |
|
|
|
|
|
Issued, Subscribed & Paid-up Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
5200000 |
Equity Shares |
Rs. 10/- each |
Rs.52.000 Millions |
|
|
|
|
|
FINANCIAL DATA
[all figures are
in Rupees Millions]
ABRIDGED BALANCE
SHEET
|
SOURCES OF FUNDS |
|
31.03.2012 |
31.03.2011 |
|
I.
EQUITY
AND LIABILITIES |
|
|
|
|
(1)Shareholders' Funds |
|
|
|
|
(a) Share Capital |
|
52.000 |
52.000 |
|
(b) Reserves & Surplus |
|
2003.685 |
1060.709 |
|
(c) Money
received against share warrants |
|
0.000 |
0.000 |
|
|
|
|
|
|
(2) Share Application money pending allotment |
|
0.000 |
0.000 |
|
Total
Shareholders’ Funds (1) + (2) |
|
2055.685 |
1112.709 |
|
|
|
|
|
|
(3) Non-Current
Liabilities |
|
|
|
|
(a) long-term borrowings |
|
7.709 |
9.818 |
|
(b) Deferred tax liabilities (Net) |
|
114.380 |
73.459 |
|
(c) Other long term liabilities |
|
9.259 |
8.159 |
|
(d) long-term provisions |
|
0.000 |
0.000 |
|
Total Non-current Liabilities (3) |
|
131.348 |
91.436 |
|
|
|
|
|
|
(4) Current Liabilities |
|
|
|
|
(a) Short term borrowings |
|
460.523 |
348.773 |
|
(b) Trade payables |
|
1153.671 |
1304.241 |
|
(c) Other current
liabilities |
|
85.902 |
121.887 |
|
(d) Short-term provisions |
|
24.512 |
75.199 |
|
Total Current Liabilities (4) |
|
1724.608 |
1850.100 |
|
|
|
|
|
|
TOTAL |
|
3911.641 |
3054.245 |
|
|
|
|
|
|
II.
ASSETS |
|
|
|
|
(1) Non-current assets |
|
|
|
|
(a) Fixed Assets |
|
|
|
|
(i) Tangible assets |
|
1168.545 |
881.833 |
|
(ii) Intangible Assets |
|
0.000 |
0.000 |
|
(iii) Capital
work-in-progress |
|
386.247 |
93.127 |
|
(iv) Intangible
assets under development |
|
0.000 |
0.000 |
|
(b) Non-current Investments |
|
0.000 |
0.000 |
|
(c) Deferred tax assets (net) |
|
0.000 |
0.000 |
|
(d) Long-term Loan and Advances |
|
175.788 |
404.246 |
|
(e) Other Non-current assets |
|
0.000 |
0.000 |
|
Total Non-Current Assets |
|
1730.580 |
1379.206 |
|
|
|
|
|
|
(2) Current assets |
|
|
|
|
(a) Current investments |
|
0.000 |
0.000 |
|
(b) Inventories |
|
684.159 |
401.052 |
|
(c) Trade receivables |
|
1199.718 |
1079.477 |
|
(d) Cash and cash
equivalents |
|
66.417 |
30.286 |
|
(e) Short-term loans and
advances |
|
230.767 |
164.224 |
|
(f) Other current assets |
|
0.000 |
0.000 |
|
Total Current Assets |
|
2181.061 |
1675.039 |
|
|
|
|
|
|
TOTAL |
|
3911.641 |
3054.245 |
|
SOURCES OF FUNDS |
|
|
31.03.2010 |
|
|
SHAREHOLDERS FUNDS |
|
|
|
|
|
1] Share Capital |
|
|
52.000 |
|
|
2] Share Application Money |
|
|
0.000 |
|
|
3] Reserves & Surplus |
|
|
491.715 |
|
|
4] (Accumulated Losses) |
|
|
0.000 |
|
|
NETWORTH |
|
|
543.715 |
|
|
LOAN FUNDS |
|
|
|
|
|
1] Secured Loans |
|
|
477.151 |
|
|
2] Unsecured Loans |
|
|
0.000 |
|
|
TOTAL BORROWING |
|
|
477.151 |
|
|
DEFERRED TAX LIABILITIES |
|
|
37.354 |
|
|
|
|
|
|
|
|
TOTAL |
|
|
1058.220 |
|
|
|
|
|
|
|
|
APPLICATION OF FUNDS |
|
|
|
|
|
|
|
|
|
|
|
FIXED ASSETS [Net Block] |
|
|
484.764 |
|
|
Capital work-in-progress |
|
|
53.017 |
|
|
|
|
|
|
|
|
INVESTMENT |
|
|
0.000 |
|
|
DEFERRED TAX ASSETS |
|
|
0.000 |
|
|
|
|
|
|
|
|
CURRENT ASSETS, LOANS & ADVANCES |
|
|
|
|
|
|
Inventories |
|
|
340.169
|
|
|
Sundry Debtors |
|
|
763.915
|
|
|
Cash & Bank Balances |
|
|
7.214
|
|
|
Other Current Assets |
|
|
0.000
|
|
|
Loans & Advances |
|
|
124.372
|
|
Total
Current Assets |
|
|
1235.670 |
|
|
Less : CURRENT
LIABILITIES & PROVISIONS |
|
|
|
|
|
|
Sundry Creditors |
|
|
687.818
|
|
|
Other Current Liabilities |
|
|
20.840
|
|
|
Provisions |
|
|
6.573
|
|
Total
Current Liabilities |
|
|
715.231 |
|
|
Net Current Assets |
|
|
520.439 |
|
|
|
|
|
|
|
|
MISCELLANEOUS EXPENSES |
|
|
0.000 |
|
|
|
|
|
|
|
|
TOTAL |
|
|
1058.220 |
|
PROFIT & LOSS
ACCOUNT
|
|
PARTICULARS |
31.03.2012 |
31.03.2011 |
31.03.2010 |
|
|
|
SALES |
|
|
|
|
|
|
|
Income |
3896.458 |
3125.325 |
2125.342 |
|
|
|
Other Income |
53.337 |
31.673 |
12.686 |
|
|
|
TOTAL (A) |
3949.795 |
3156.998 |
2138.028 |
|
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
|
|
Consumption Materials Changes Inventories |
|
1613.828 |
1236.189 |
|
|
|
Manufacturing Service Costs |
|
154.245 |
102.987 |
|
|
|
Employee Related Expenses |
|
172.827 |
123.693 |
|
|
|
Administrative Selling Others Expenses |
|
230.674 |
147.233 |
|
|
|
Research Development Expenditure |
|
79.678 |
56.380 |
|
|
|
TOTAL (B) |
2486.517 |
2251.252 |
1666.482 |
|
|
|
|
|
|
|
|
Less |
PROFIT
/ (LOSS) BEFORE INTEREST, TAX, DEPRECIATION AND AMORTISATION (A-B) (C) |
1463.278 |
905.746 |
471.545 |
|
|
|
|
|
|
|
|
|
Less |
FINANCIAL
EXPENSES (D) |
48.850 |
33.490 |
45.610 |
|
|
|
|
|
|
|
|
|
|
PROFIT
/ (LOSS) BEFORE TAX, DEPRECIATION AND AMORTISATION (C-D) (E) |
1414.428 |
872.256 |
425.935 |
|
|
|
|
|
|
|
|
|
Less |
DEPRECIATION/
AMORTISATION (F) |
55.414 |
30.495 |
19.339 |
|
|
|
|
|
|
|
|
|
|
PROFIT / (LOSS)
BEFORE TAX (E-F) (G) |
1359.014 |
841.761 |
406.596 |
|
|
|
|
|
|
|
|
|
Less |
TAX (H) |
397.908 |
254.516 |
140.811 |
|
|
|
|
|
|
|
|
|
|
PROFIT / (LOSS)
AFTER TAX (G-H) (I) |
961.106 |
587.245 |
265.785 |
|
|
|
|
|
|
|
|
|
Add |
PREVIOUS
YEARS’ BALANCE BROUGHT FORWARD |
1001.986 |
491.715 |
238.097 |
|
|
|
|
|
|
|
|
|
Less |
APPROPRIATIONS |
|
|
|
|
|
|
|
Dividend |
|
|
10.400 |
|
|
|
Tax on Dividend |
|
|
1.767 |
|
|
BALANCE CARRIED
TO THE B/S |
NA |
1001.986 |
491.715 |
|
|
|
|
|
|
|
|
|
|
EARNINGS IN
FOREIGN CURRENCY |
|
|
|
|
|
|
|
Export Earnings |
|
2234.012 |
11349.782 |
|
|
|
Other Earnings |
|
4.072 |
4.002 |
|
|
TOTAL EARNINGS |
NA |
2238.084 |
11353.784 |
|
|
|
|
|
|
|
|
|
|
IMPORTS |
|
|
|
|
|
|
|
Raw Materials |
|
212.546 |
168.624 |
|
|
|
Stores & Spares |
|
47.679 |
0.000 |
|
|
|
Capital Goods |
|
0.553 |
10.489 |
|
|
TOTAL IMPORTS |
NA |
260.778 |
179.113 |
|
|
|
|
|
|
|
|
|
|
Earnings /
(Loss) Per Share (Rs.) |
184.83 |
112.93 |
51.11 |
|
KEY RATIOS
|
PARTICULARS |
|
31.03.2012 |
31.03.2011 |
31.03.2010 |
|
PAT / Total Income |
(%) |
24.33
|
18.60
|
12.43
|
|
|
|
|
|
|
|
Net Profit Margin (PBT/Sales) |
(%) |
34.88
|
26.93
|
19.13
|
|
|
|
|
|
|
|
Return on Total Assets (PBT/Total Assets} |
(%) |
40.57
|
32.92 |
23.63 |
|
|
|
|
|
|
|
Return on Investment (ROI) (PBT/Networth) |
|
0.66
|
0.76 |
0.75 |
|
|
|
|
|
|
|
Debt Equity Ratio (Total Debt /Networth) |
|
0.23
|
0.32 |
0.88 |
|
|
|
|
|
|
|
Current Ratio (Current Asset/Current Liability) |
|
1.26
|
0.91 |
1.73 |
FINANCIAL ANALYSIS
[all figures are
in Rupees Millions]
DEBT EQUITY RATIO
|
Particular |
31.03.2011 |
31.03.2012 |
|
|
(INR
in Mlns) |
(INR
in Mlns) |
|
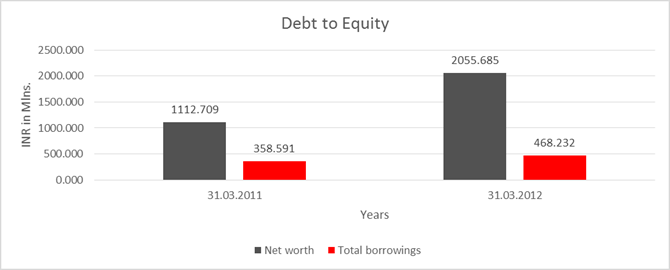
Share Capital |
52.000 |
52.000 |
|
Reserves & Surplus |
1060.709 |
2003.685 |
|
Net
worth |
1112.709 |
2055.685 |
|
|
|
|
|
long-term borrowings |
9.818 |
7.709 |
|
Short term borrowings |
348.773 |
460.523 |
|
Total
borrowings |
358.591 |
468.232 |
|
Debt/Equity
ratio |
0.322 |
0.228 |
YEAR-ON-YEAR GROWTH
|
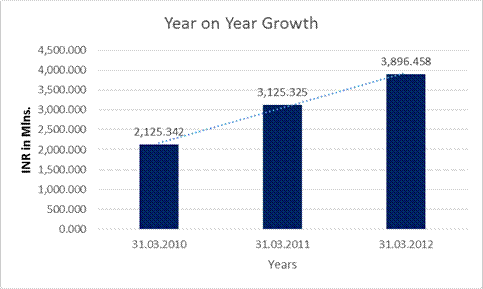
Year
on Year Growth |
31.03.2010 |
31.03.2011 |
31.03.2012 |
|
|
(INR
in Mlns) |
(INR
in Mlns) |
(INR
in Mlns) |
|
Sales |
2125.342 |
3125.325 |
3896.458 |
|
|
|
47.050 |
24.674 |
NET PROFIT MARGIN
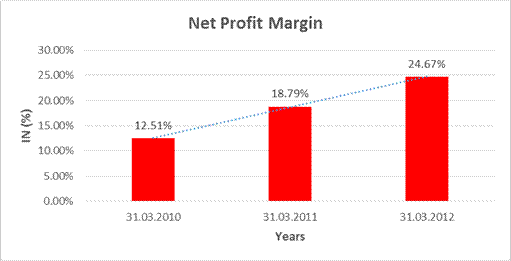
|
Net Profit
Margin |
31.03.2010 |
31.03.2011 |
31.03.2012 |
|
|
(INR
in Mlns) |
(INR
in Mlns) |
(INR
in Mlns) |
|
Sales
|
2125.342 |
3125.325 |
3896.458 |
|
Profit |
265.785 |
587.245 |
961.106 |
|
|
12.51% |
18.79% |
24.67% |
LOCAL AGENCY FURTHER INFORMATION
|
Sr. No. |
Check List by Info Agents |
Available in
Report (Yes / No) |
|
1] |
Year of Establishment |
Yes |
|
2] |
Locality of the firm |
Yes |
|
3] |
Constitutions of the firm |
Yes |
|
4] |
Premises details |
No |
|
5] |
Type of Business |
Yes |
|
6] |
Line of Business |
Yes |
|
7] |
Promoter's background |
Yes |
|
8] |
No. of employees |
Yes |
|
9] |
Name of person contacted |
No |
|
10] |
Designation of contact
person |
No |
|
11] |
Turnover of firm for last
three years |
Yes |
|
12] |
Profitability for last
three years |
Yes |
|
13] |
Reasons for variation
<> 20% |
----------- |
|
14] |
Estimation for coming
financial year |
No |
|
15] |
Capital in the business |
Yes |
|
16] |
Details of sister
concerns |
Yes |
|
17] |
Major suppliers |
No |
|
18] |
Major customers |
No |
|
19] |
Payments terms |
No |
|
20] |
Export / Import details
(if applicable) |
No |
|
21] |
Market information |
---------- |
|
22] |
Litigations that the firm
/ promoter involved in |
---------- |
|
23] |
Banking Details |
Yes |
|
24] |
Banking facility details |
Yes |
|
25] |
Conduct of the banking
account |
---------- |
|
26] |
Buyer visit details |
---------- |
|
27] |
Financials, if provided |
Yes |
|
28] |
Incorporation details, if
applicable |
Yes |
|
29] |
Last accounts filed at
ROC |
No |
|
30] |
Major Shareholders, if
available |
Yes |
|
31] |
Date of Birth of
Proprietor/Partner/Director, if available |
Yes |
|
32] |
PAN of
Proprietor/Partner/Director, if available |
No |
|
33] |
Voter ID No of Proprietor/Partner/Director,
if available |
No |
|
34] |
External Agency Rating,
if available |
Yes |
NOTE: The
registered address of the company has been shifted from H.No.8-3-167/D/16,
Kalyan Nagar, Phase-I, Near A. G. Colony, Hyderabad – 500 038, Andhra Pradesh,
India to the present address w.e.f. 28.09.2009.
INDEX OF CHARGE:
|
Sr. No. |
Charge ID |
Date of Charge
Creation/Modification |
Charge amount
secured |
Charge Holder |
Address |
Service Request
Number (SRN) |
|
1 |
10372144 |
15/11/2012
* |
200,000,000.00 |
ICICI
BANK LIMITED |
LANDMARKRACE
COURCE CIRCLE, ALKAPURI, BARODA – 390 015, GUJARAT, INDIA |
B63884381 |
|
2 |
10333818 |
15/11/2012
* |
600,000,000.00 |
ICICI
BANK LIMITED |
LANDMARKRACE
COURCE CIRCLE, ALKAPURI, BARODA – 390 015, GUJARAT, INDIA |
B63883599 |
|
3 |
10016973 |
24/09/2009
* |
65,000,000.00 |
AXIS
BANK LIMITED |
CREDIT
MANAGEMENT CENTRE, 5-2-183/184, 8751(OLD), IIIRD FLOOR, RP ROAD, SECUNDERABAD
– 500 003, ANDHRA PRADESH, INDIA |
A71194419 |
|
4 |
90126902 |
15/11/2012
* |
600,000,000.00 |
AXIS
BANK LIMITED |
CORPORATE
BANKING BRANCH, 1ST FLOOR, PULLAREDDY BUILDINGS, GREENLANDS, BEGUMPET,
HYDERABAD – 500 016, ANDHRA PRADESH, INDIA |
B63794192 |
* Date
of charge modification
OPERATIONS:
The Company presently has two world class bulk actives and chemicals
manufacturing facility situated at Rudraram (V), Medak Dist., and Kardanur (V),
Medak Dist. About 40 kms from the city of Hyderabad and one finished dosages
manufacturing facility at ANRICH Industrial Estate, Bollaram, Medak Dist., near
Hyderabad.
BULK ACTIVES:
Subject produces around 36 bulk actives in addition to the other chemicals
and intermediates in its bulk actives facility. Of this the majorproducts are
Clopidogrel, Ezetimibe, Rosuvastatin, Terbinafine and Voricanazole which
together contribute over 80% of the product revenues. The year saw launch of new molecules viz.,
Dabigatran; Desvenlafaxine; Dronedarone; Bicalutamide; Bimatoprost; Bortezomib;
Carboprost;Docetaxel; Epoprostonol; Erlotonib; Imatinib; Irinotecan;
Palonosetran.
Subject efforts to build a stronger customer base, both in domestic as
well as export markets. The share of export revenues contributes 62%of the
total revenues. The Company expects that
the share of export revenues would increase in the years to come. This being
propelled mainly through conscious efforts to increase its regulatory filings,
customer partnerships etc.
Subject has filed, in the Indian Patent Office, 139 intellectual
property rights applications. Similarly, 60 applications have been filed under
Patents Cooperation Treaty (PCT).
Subject has filed 86 DMFs including CEPS in US, Europe, Canada, Korea,
Japan etc.
FINISHED DOSAGES
BUSINESS:
Subject produces over 75 products in its finished dosage basket. The
company has ventured into chronic therapy-Lifestyle diseases which is the biggest
threat to the country. This segment, covering invasive and degenerative
diseases, is one of the fastest growing segments world over and India in
particular. The successful presence of
our finished dosages business enables the Company in moving up the value chain
in the pharmaceutical vertical.
During the year, subject has successfully expanded its market reach by
establishing a strong presence in the market. As Domestic Formulations Company
we have started making inroads in the market and are able to make a mark in the
Indian Market.
Subject is making necessary efforts in registering its products in
various countries in the semi-regulated markets of the world. Distribution partners have been identified in
many countries in Asia-Pacific, Latin America, Africa and some parts of
CIS. Over the next few years' subject
expects this business segment will make great progress in the market.
FIXED ASSETS:
·
Land
·
Building
·
Plant and Machinery
·
Electrical installations
·
Lab Equipments
·
Generator
·
Office Equipments
·
Computers
·
Furniture and Fixtures
·
Motor Vehicles
PRESS
RELEASE:
MSN LABORATORIES
LAUNCHES PAIN-KILLER DRUG
Hyderabad, May 17:
Generics and finished dosages manufacturer, MSN Laboratories Ltd, has launched
its analgesic drug Tapentadol under the brand ‘Tapal' in domestic market.
“With this launch, we are entering pain management market which is valued
at over Rs 30000.000 Millions,” Dr M.S.N. Reddy, Chairman and Managing
Director, MSN Laboratories, told newspersons here on Monday.
The drug is the generic version of Johnson & Johnson's Nucynta. “Our
product is priced at one-fifth of the innovator's drug,” Dr Reddy claimed.
Depending on dosage, Tapal is priced at Rs 10 to Rs19.
Tapentadol is indicated for management of moderate to severe and acute pain
in adult patients.
R&D FACILITY
The privately-held
company was in the process of setting up a R&D facility with Rs 1000.000
Millions at Pashmylaram here.
“The first phase is
expected to be completed by this year-end,” Dr Reddy said.
The company had a
turnover of Rs 5000.000 Millions during 2010-11. About 75 per cent of revenue
had come from exports to over 100 countries.
MSN LABS TO ENTER
PAIN MANAGEMENT MARKET
Hyderabad, May 16:
MSN Laboratories Ltd, a city-based research oriented pharma company, has
said it will foray into the pain management market with the launch of
‘Tapentadol’ a pain killer under the brand name Tapal in the Indian market.
According to Mr MSN Reddy, Chairman of MSN Labs, the drug was developed by
the company for the first time in India and the results of phase-III trials of
the drug have shown that it would work better than the current acute pain
relieving drugs.
“We expect to clock revenues of up to Rs 1000.000 Millionsin the next 3-4
years from Tapal,” Mr Reddy told media persons here.
He said the market for pain killers in the country is around Rs 35000.000
Millions.
The Chairman said currently 10 to 12 drugs are under research and
development and the company will centralise the R&D activities in a new
facility being developed near Hyderabad with an investment of Rs 1000.000
Millions.
MSN currently has five manufacturing units, one facility for finished
dosage and a dedicated R&D centre. The company so far has filed over 100
Indian and over 40 international patents.
The company has also filed nearly 80 drug master files for the US, Canada
and European markets, Mr Reddy said.
MSN LABS TO INVEST
RS.1500.000 MILLIONS FOR API, FORMULATIONS & CRAMS UNIT IN HYDERABAD
MSN Laboratories
Ltd, a fast growing active pharmaceutical ingredients and formulations company
of India is planning to invest Rs.1500.000 Millions for setting up an API,
formulations and CRAMS (Contract Research and Manufacturing Services) unit in
Hyderabad.
Started small with
a single unit in the year 2003, today the Hyderabad based MSN Group has
established 8 units across India of which 6 are manufacturing units catering to
APIs, 1 unit for formulations and 1 research and development centre. Out of the
6 manufacturing units 3 units are US FDA certified while the remaining 3 are
catering to ROW (Rest of the World) and Indian markets.
While sharing his
views with Pharmabiz, Dr MSN Reddy, CMD of MSN group of companies, revealed his
expansion plans and said that the MSN group is planning to invest a total of
Rs.1500.000 Millions in the first phase to set up an exclusive API,
Formulations and CRAMS unit in Hyderabad. “We are a vertically integrated
company and our main focus is global markets. At present we are growing
consistently with a year-on-year growth of 30-40 per cent. Our major strength
is our people, focused R&D and cGMP practices” said Dr Reddy.
The year 2012 was
a great year for the MSN Group. The Group achieved a growth target of 40 per
cent. The overall turnover of the Group by March 2012 was set at Rs.6000.000
Millions and by the end of the financial year march 2013 it is expected that it
will cross Rs.10000.000 Millions. So far the Group has filed 148 Drug Master
Files (DMFs) globally of which 74 DMFs are filed in USA and other 74 are filed
in EU.
Airing his future
plans Dr MSN Reddy said, “We are planning to launch 2 important molecules in EU
and USA by 2015. Our research is mainly focused on the development of molecules
and not on therapeutics. At present we are having 3-4 potential molecules which
can give us a competitive advantage and our success lies in winning new markets
with new molecules.”
The MSN Group of companies has been recognized globally and has been
approved and certified by all countries as quality and safety complier. Very
soon the group is planning to develop a Green field facility for its
formulations unit in a plot of over 40 acres in Hyderabad. The company is
already having a patient portfolio of 150 drugs in different countries. It is
also planning to file 35 more patents in USA and 40 patents in EU in the coming
days.
MSN LABORATORIES
SUCCESSFULLY COMPLETES USFDA AUDIT FOR THE 3RD SUCCESSIVE TIME
Hyderabad based MSN Laboratories has successfully completed US FDA
Audit. The group has 55 DMFs for the
US market. Out of which 29 DMFs are from this facility.
Commenting on the development, Dr MSN Reddy, chairman & managing
director, MSN Group, said, that the 3rd consecutive audit that has been
successfully completed by the MSN Group. Achieving this for the 2nd time for the same unit has reiterated our belief
and commitment towards quality.
MSN Group is one of the fastest growing pharmaceutical manufacturers of
APIs and finished dosages in India.
The group has six API (including an Oncology) and one finished dosage
facilities & an integrated R&D Center located in Hyderabad. Two more
finished dosage facilities including one for Oncology are being built. MSN group facilities are USFDA, WHO/EU GMP
approved and ISO 9001:2008 certified.
The MSN group be offering custom Synthesis for API, Dosage development
services for the global markets from its State-of-the-art R&D Center soon.
The world class research center has about 42 stand alone laboratories housing
400 scientists spread over a 300,000 sq.ft facility.
With more than 100 APIs and 120 finished dosages, MSN group offers a
strong portfolio of complex and niche products covering all the major
therapies. It has 120 filed DMFs & CEPs for the US, Europe, Canada, Korea
& Japan, and 800 dossiers for the finished dosages for the emerging
markets. With 137 Indian & 51 International
Patents under its` belt, MSN Group offers comprehensive product support
to more than 250 customers across 65 countries in the North America, Europe,
Latin America, Africa & Asia.
With an outstanding workforce, cutting-edge science, state-of-the-art
technology base and knowledge intensive
initiatives, MSN group is working on innovative solutions for tomorrow`s
healthcare problems
MSN LABORATORIES
TO SET UP A NEW R&D CENTRE IN HYDERABAD
Hyderabad-based Rs 3900.000 Millions formulation and contract research and
manufacturing services provider, MSN Laboratories, is setting up a new R&D
centre in Hyderabad with an investment of over Rs 400.000 Millions. The full
fledge commercial operation from the new centre will begin by first quarter of
2011. The company has arranged for funds from internal accruals.
The new R&D centre will be dedicated for
process chemistry, oncology chemicals, formulations and R&D, process
engineering etc. The company is involved in the manufacturing of high quality
Active Pharmaceutical Ingredients (APIs) and key pharmaceutical intermediates.
MSN Lab has strong R&D background supported by IPM team to develop
non-infringing and cost effective processes to cater to the specific
requirements of the customers worldwide.
Bharat Reddy, Executive Director, MSN
Laboratories said, "We have a very strong team and R&D backup. Looking
at the pharma generic products opportunity in the market we have finalized our
expansion plans. Initially, we will be building up a brand new integrated
R&D centre for finished dosages, APIs and CRAMs. For our new R&D centre
we will be hiring 350 scientists. We have already commenced construction work
and it should be completed by early next year."
At present, MSN Lab has total manpower
strength of 1,350 people, of which 20 percent staff is dedicated for R&D on
new product development and R&D process equally. The company is actively
working on generic APIs for oncology and anything related to synthetic organic
chemistry. MSN Lab is outsourcing required raw material from India, China and
some parts of Europe.
The company has 55 APIs globally, out of which
it has obtained 30 plus drug master files (DMFs) for US and Europe market. It
has filed seven COPs/CEP for Europe market. Overall, it has filled 85 patents
and soon it is targeting to get three to four product patents. "We have
been continuously working on novel root of synthesis and we are applying for
patent on process development," Reddy said.
MSN has 60 odd branded generic products for
anti hypertensive, anti-hyper lipoprotenemic, anti depressants,
anti-thrombotic, anti fungal, anti-bacterials, anti migraine, anti diabetic and
anti psychotic etc in its basket. It is in the process of product registration
of existing portfolio for ROW market. "We have already started
registration work and in next six months we will be having everything in
place."
Reddy comments on the company's inorganic
growth, "At present we do not have any activities in place, but we are
looking for marketing tie ups. We are entering into the Japanese market, while
also extending our presence in the Korean market."
From the financial year 2010-11, the company
is expecting a turnover of Rs 6500.000 Millions. The company has strong
expansion plans lined up for the next two years.
MSN SUCCESSFULLY COMPLETES THE AUDIT FROM FMHACA OF
ETHIOPIA. (30 DEC 2013)
The
Formulation Division of MSN Laboratories Private Limited successfully completed
the audit from Ethiopia on 29 Dec 2013.
MSN LABS TO INVEST RS.1500.000 MILLIONS FOR API,
FORMULATIONS & CRAMS UNIT IN HYDERABAD.
MSN
Laboratories Limited, a fast growing active pharmaceutical ingredients and formulations
company of India is planning to invest Rs.150 crore for setting up an API,
formulations and CRAMS (Contract Research and Manufacturing Services) unit in
Hyderabad.
Started
small with a single unit in the year 2003, today the Hyderabad based MSN Group
has established 8 units across India of which 6 are manufacturing units
catering to APIs, 1 unit for formulations and 1 research and development
Centre. Out of the 6 manufacturing units 3 units are US FDA certified while the
remaining 3 are catering to ROW (Rest of the World) and Indian markets.
While
sharing his views with Pharmabiz, Dr MSN Reddy, CMD of MSN group of companies,
revealed his expansion plans and said that the MSN group is planning to invest
a total of Rs.1500.000 Millions in the first phase to set up an exclusive API,
Formulations and CRAMS unit in Hyderabad. “We are a vertically integrated
company and our main focus is global markets. At present we are growing
consistently with a yearon- year growth of 30-40 per cent. Our major strength
is our people, focused R&D and cGMP practices” said Dr Reddy.
The
year 2012 was a great year for the MSN Group. The Group achieved a growth
target of 40 percent. The overall turnover of the Group by March 2012 was set
at Rs.6000.000 Millions and by the end of the financial year march 2013 it is
expected that it will cross Rs.10000.000 Millions. So far the Group has filed
148 Drug Master Files (DMFs) globally of which 74 DMFs are filed in USA and
other 74 are filed in EU.
Airing
his future plans Dr MSN Reddy said, “We are planning to launch 2 important
molecules in EU and USA by 2015. Our research is mainly focused on the
development of molecules and not on therapeutics. At present we are having 3-4
potential molecules which can give us a competitive advantage and our success
lies in winning new markets with new molecules.”
The
MSN Group of companies has been recognized globally and has been approved and
certified by all countries as quality and safety complier. Very soon the group
is planning to develop a Green field facility for its formulations unit in a
plot of over 40 acres in Hyderabad. The company is already having a patient
portfolio of 150 drugs in different countries. It is also planning to file 35
more patents in USA and 40 patents in EU in the coming days.
CPHI INDIA PHARMA AWARD 2012
MSN
Laboratories Limited has been awarded CPhI India Award 2012 for
"Innovation in Cost". It recognizes our contribution (1st-in-India
generics of Bosentan, Lacosamide, Silodosin, etc. at very affordable prices) to
Healthcare in India.
MSN LABORATORIES SUCCESSFULLY COMPLETES USFDA AUDIT FOR THE
3 RD SUCCESSIVE TIME
Hyderabad
based MSN Laboratories has successfully completed US FDA Audit. The group has
55 DMFs for the
US
market. Out of which 29 DMFs are from this facility. Commenting on the
development, Dr. MSN Reddy, chairman and managing director, MSN Group, said,
that the 3rd consecutive audit that has been successfully completed by the MSN
Group. Achieving this for the 2nd time for the same unit has reiterated our
belief and commitment towards quality.
MSN
Group is one of the fastest growing pharmaceutical manufacturers of APIs and
finished dosages in India. The group has six API (including an Oncology) and
one finished dosage facilities and an integrated R&D Center located in
Hyderabad. Two more finished dosage facilities including one for Oncology are
being built. MSN group facilities are USFDA, WHO/EU GMP approved and ISO
9001:2008 certified.
The MSN
group be offering custom Synthesis for API, Dosage development services for the
global markets from its State-of-the-art R&D Center soon. The world class
research center has about 42 standalone laboratories housing 400 scientists
spread over a 300,000 sq. ft. facility.
With
more than 100 APIs and 120 finished dosages, MSN group offers a strong
portfolio of complex and niche products covering all the major therapies. It
has 120 filed DMFs and CEPs for the US, Europe, Canada, Korea and Japan, and
800 dossiers for the finished dosages for the emerging markets. With 137 Indian
and 51 International Patents under its belt, MSN Group offers comprehensive
product support to more than 250 customers across 65 countries in the North
America, Europe, Latin America, Africa and Asia.
With
an outstanding workforce, cutting-edge science, state-of-the-art technology
base and knowledge intensive
initiatives, MSN group is working on innovative solutions
for tomorrow`s healthcare problems.
MSN LABS WINS THE FAPCCI OUTSTANDING EXPORT PERFORMANCE AWARD
PHARMEXCIL'S OUTSTANDING EXPORT PERFORMANCE AWARDS GIVEN TO
12 PHARMA MAJORS, 24 COS GOT PATENT AWARDS
Tuesday,
September 28, 2010 08:00 IST
Our
Bureau, Chennai
The
Outstanding Export Performance Awards and Patent Awards for 2009-10, instituted
by the Pharmaceuticals Export Promotion Council (Pharmexcil), were presented to
leading pharma companies in a ceremony organized by the Pharmaexcil, as part of
Pharma CEOs conclave in Hyderabad last week.
Twelve
pharma majors have bagged the Outstanding Export Performance Awards-2009-10 and
24 companies have won Patent Awards for the same year. The awards were
presented by the Chief Minister of Andhra Pradesh, K Rosaiah. Giving away the
awards, he said that his government has launched investor friendly industrial
policy in the state giving major thrust for pharma and biotechnology sectors.
While
the Mumbai based Ranbaxy Labs bagged the Outstanding Exports Award in Overall
Category, the Ahmedabad based Cadila Healthcare won the Award in emerging
category. Outstanding Exports Award Formulations in Overall category has gone
to Aurobindo Pharma Limited and Outstanding Exports Award Formulations in
emerging category was presented to Matrix Laboratories Limited.
Dr
Reddy's Lab in Hyderabad won the award in overall category for bulk drugs and
MSN Laboratories was given the award for bulk drugs in emerging category.
Outstanding Exports Award Biologicals in emerging category has gone to
Wockhardt Limited, and the award for neutraceuticals in emerging category has
bagged by two companies, Omniactive Health Technologies Limited and Divis
Laboratories Limited. Mission Vivacare Limited and Medo Pharma have won the
Outstanding Exports Award Small Company in Overall Category. The same award in
emerging category has tone to Titan Laboratories Private Limited and Smilax
Laboratories Limited
In
Ayush section, the outstanding exports award has gone to three companies,
Jyotindra International, Ban Labs Limited and Welex Laboratories Private
Limited The outstanding Exports award Merchant Exporters in emerging category
has bagged by Anagah Pharma Private Limited
The
companies that bagged the Patents Awards (Gold Awardees) for securing patents in
2009-10, are Suven Life Sciences (for drug discovery product category), Lupin
Limited (for drug discovery), Dr Reddy's Lab (for API/ bulk drugs), Hetero
Drugs Limited (for API/bulk drugs), IPCA Laboratories Ltd (for API/ bulk
drugs), Bicon Limited (for Biotech Products) and Wockhardt (for Medical
devices).
The
silver awardees are Glenmark Pharmaceuticals Limited (for drug discovery
category), Torrent Pharmaceuticals Ltd (for drug discovery), USV Limited (for
API/ bulk drugs), Symed Labs Limited (for API / bulk drugs), Lyka Labs limited
( for formulations), Rubican Research Pvt Limited (for formulations),
Polymedicure Limited (for medical devices), Laila Neutraceuticals (for herbal/
ayurvedic/ neutraceuticals), Bharath Biotech International Limited (for biotech
products) and Natco Pharma Limited (for API/ bulk drugs).
Certificate
of Appreciations were given to Matrix Laboratories Limited (for API / Bulk
drugs product category), Shasun Pharmaceuticals Limited (for API/ bulk drugs),
Ind-Swift Laboratories limited (for API/bulk drugs), Themis Medicare (for
formulations), Green Chem (for herbal/ ayurvedic / neutraceuticals) and Bilcare
(for others).
MSN LABORATORIES AWARDED EMERGING INDIA SME 2010
We
were awarded the Emerging India SME Award 2010 by ICICI/CRISIL/CNBC_TV18 in
Macau (Hongkong) on 10th November 2010.
MSN LABORATORIES: A MASTER IN THE MAKING
In
less than a decade, MSN Laboratories has consolidated its place as one of the
fast growing pharma companies in India. Sachin Jagdale analyses the growth trajectory
of the company it has barely been a decade since MSN Laboratories' inception,
but the company's achievements would make even the industry veterans give them
a standing ovation. It is never an easy thing to start your own company, and to
survive the grueling competition, is even more difficult; but when has a smooth
sea made a good mariner:
MSN
Reddy, Chairman and Managing Director, MSN Laboratories, strongly believes in
this philosophy. MSN Group is one of the fastest growing manufacturers of active
pharmaceutical ingredients (APIs) and finished dosages in India. Established in
the year 2003, MSN group comprises four API manufacturing plants, one finished
dosage facility and a dedicated technology development Centre.
From
employee to employer
"When
I worked as an employee in an Indian multinational company, one thought was
etched my mind-that instead of being an employee today, why can't I be an
entrepreneur “That’s how I started my own company," says an ambitious
Reddy. Prior to starting his own firm, Reddy was R&D head in one of India's
leading pharma companies. Today, if you go through the list of his colleagues,
at least 75-80 of them worked with him in his previous organisation, which
shows the trust that Reddy had garnered. He says, "When I was in the other
company, my responsibility was very wide, from product identification to Drug
Master File (DMF) filing, but basically I am an R&D person."
Quality
matters
MSN
group management comprises of top notch professionals, with extensive experience
in synthetic chemistry, process engineering, quality, IT, SCM, marketing,
regulatory requirements and environmental safety and a proven track record in
the pharma industry.
Thriving
on the platform of innovation and excellence, the company has grown into a
successful and profitable enterprise with a portfolio covering all the major
therapeutic areas. Speaking about the features of its facilities, Reddy
informs, "Our plants are ISO 9001-2000 certified, WHO: GMP, EU: GMP and US
FDA approved. Our manufacturing units are designed as per WHO- GMP standards
and confirm to the principles of quality, safety and sound environment. Our
outstanding team of well-educated, motivated and result-oriented employees has
contributed actively to our spectacular growth." He adds, "We have
very efficient systems driven supply chain management and a highly responsive
customer relationship management team. In fact, we are the youngest pharma
company in the world to have SAP in place for all our commercial operations. We
guarantee competitive edge to our customers because of our absolute cost
efficiency and aggressive product development timelines. Along with this
commitment comes an irrevocable promise of quality and consistency."
A
capable entrepreneur
From
an R&D head to an entrepreneur, there has been a tremendous shift in
Redyy's responsibilities. He recalls, "When I was working in the previous
company I used to handle R&D, IP, regulatory affairs, quality, as well as
part of the manufacturing. But I was oblivious to other functions. Today, I
need to look into corporate functions like finance, business development,
supply chain management, marketing as well. There was a time when as a pharma
professional I got the chance to complete only 60 percent of operations, but
today I have the chance to finish the rest of the 40 percent as well."
According to him, the business model that they chose ensured success as
presently, there is a difference between the products of MSN Labs and that of
other companies. "The size of our products is different from those in
other companies, which may be primarily due to the age factor. Our product
portfolio can be compared to only four to five companies in India, which has
brought us to the stage we are at today," avers Reddy.
MSN Laboratories
has shown a mind boggling growth. The company has clocked 150 percent
year-on-year (Y-O Y) growth since its inception, making it one of the fastest
growing pharma companies in India. Apart from financials, they have made
tremendous progress in improving themselves. The company received the
Outstanding Export Performance Award in 2008-09, and within less than 20 months
of inception, it received ISO 9001:2000 certifications. WHO: GMP approvals came
in less than 24 months of inception, whereas; EU: GMP approvals came in less
than 48 months of establishment, followed by an FDA approval. Other than having
a Department of Scientific and Industrial Research (DSIR) recognition to its
credit, MSN Laboratories also received an Udyog Ratna Award.
Smart
business
Reddy
has very interesting and intelligent business plans in mind. According to him,
as chemistry is going to remain a core area for the company, they can develop a
product in time with cost effective process. But to convert it into a business
they need to have expertise in other areas also. "For instance, we should
have a good marketing team, finance team etc.," opines Reddy. Being in the
pharma industry for more than two decades, Reddy is a familiar and reliable
face in the industry, which must have helped MSN to grow as well. Reddy says,
"Since the inception of this company, recognition of customers as well as
suppliers has been very different as compared to my previous company, and the
fact that I was recognised in the industry definitely helped us go
further."
MSN's
core strength is R&D, which is how they are converting this strength into
business by developing important molecules in time and supplying it to big
generic players in the global market. As far as revenue is concerned, 60
percent is exports and 40 percent is from domestic sales. Currently, the
company's R&D Centre is catering to the R&D needs of the group of
companies. In the future, the Centre will also look at contract research for
multinational companies. Biotechnology will be ruling the medicine world soon
and Reddy understands this fact very well, hence they may foray into this
category in the near future as well.
Caring
for future
All
the functions of MSN Laboratories are located in Andhra Pradesh, however, Reddy
is happy with his current operational locations. He says, "All our plants
are in close vicinity of each other. So it is very convenient for us to visit
any plant in case of any emergency." He adds, "By 2012-13 we
definitely want to cross the 1,000crore mark, and want to be the preferred API
source for global generic players."
Like a
good employer, Reddy wants his employees in future to improve in their work. He
thinks that work done in
As it
is always said-'well begun is half done'. MSN Laboratory's extraordinary beginning
has already put them in league with one of the greatest achievers in the Indian
pharma industry. However, the company is capable of doing much more, and with a
person like Reddy at the helm, every new challenge looks miniscule.
CMT REPORT (Corruption, Money Laundering & Terrorism]
The Public Notice information has been collected from various sources
including but not limited to: The Courts,
1] INFORMATION ON
DESIGNATED PARTY
No exist designating subject or any of its beneficial owners,
controlling shareholders or senior officers as terrorist or terrorist
organization or whom notice had been received that all financial transactions
involving their assets have been blocked or convicted, found guilty or against
whom a judgement or order had been entered in a proceedings for violating
money-laundering, anti-corruption or bribery or international economic or
anti-terrorism sanction laws or whose assets were seized, blocked, frozen or
ordered forfeited for violation of money laundering or international
anti-terrorism laws.
2] Court Declaration :
No exist to suggest that subject is or was
the subject of any formal or informal allegations, prosecutions or other official
proceeding for making any prohibited payments or other improper payments to
government officials for engaging in prohibited transactions or with designated
parties.
3] Asset Declaration :
No records exist to suggest that the property or assets of the subject
are derived from criminal conduct or a prohibited transaction.
4] Record on Financial
Crime :
Charges or conviction
registered against subject: None
5] Records on Violation of
Anti-Corruption Laws :
Charges or
investigation registered against subject: None
6] Records on Int’l
Anti-Money Laundering Laws/Standards :
Charges or
investigation registered against subject: None
7] Criminal Records
No
available information exist that suggest that subject or any of its principals
have been formally charged or convicted by a competent governmental authority
for any financial crime or under any formal investigation by a competent
government authority for any violation of anti-corruption laws or international
anti-money laundering laws or standard.
8] Affiliation with
Government :
No record
exists to suggest that any director or indirect owners, controlling
shareholders, director, officer or employee of the company is a government
official or a family member or close business associate of a Government
official.
9] Compensation Package :
Our market
survey revealed that the amount of compensation sought by the subject is fair
and reasonable and comparable to compensation paid to others for similar
services.
10] Press Report :
No press reports / filings exists on
the subject.
CORPORATE GOVERNANCE
MIRA INFORM as part of its Due Diligence do provide comments on
Corporate Governance to identify management and governance. These factors often
have been predictive and in some cases have created vulnerabilities to credit
deterioration.
Our Governance Assessment focuses principally on the interactions
between a company’s management, its Board of Directors, Shareholders and other
financial stakeholders.
CONTRAVENTION
Subject is not known to have contravened any existing local laws,
regulations or policies that prohibit, restrict or otherwise affect the terms
and conditions that could be included in the agreement with the subject.
FOREIGN EXCHANGE RATES
|
Currency |
Unit
|
Indian Rupees |
|
US Dollar |
1 |
Rs.61.94 |
|
|
1 |
Rs.103.31 |
|
Euro |
1 |
Rs.85.11 |
INFORMATION DETAILS
|
Information
Gathered by : |
PDT |
|
|
|
|
Report Prepared
by : |
NKT |
SCORE & RATING EXPLANATIONS
|
SCORE FACTORS |
RANGE |
POINTS |
|
HISTORY |
1~10 |
6 |
|
PAID-UP CAPITAL |
1~10 |
7 |
|
OPERATING SCALE |
1~10 |
7 |
|
FINANCIAL CONDITION |
|
|
|
--BUSINESS SCALE |
1~10 |
7 |
|
--PROFITABILIRY |
1~10 |
7 |
|
--LIQUIDITY |
1~10 |
7 |
|
--LEVERAGE |
1~10 |
7 |
|
--RESERVES |
1~10 |
8 |
|
--CREDIT LINES |
1~10 |
6 |
|
--MARGINS |
-5~5 |
-- |
|
DEMERIT POINTS |
|
|
|
--BANK CHARGES |
YES/NO |
YES |
|
--LITIGATION |
YES/NO |
NO |
|
--OTHER ADVERSE INFORMATION |
YES/NO |
NO |
|
MERIT POINTS |
|
|
|
--SOLE DISTRIBUTORSHIP |
YES/NO |
NO |
|
--EXPORT ACTIVITIES |
YES/NO |
NO |
|
--AFFILIATION |
YES/NO |
YES |
|
--LISTED |
YES/NO |
NO |
|
--OTHER MERIT FACTORS |
YES/NO |
YES |
|
DEFAULTER |
|
|
|
--RBI |
YES/NO |
NO |
|
--EPF |
YES/NO |
NO |
|
|
|
|
|
TOTAL |
|
62 |
This score serves as a reference to assess SC’s credit risk
and to set the amount of credit to be extended. It is calculated from a
composite of weighted scores obtained from each of the major sections of this
report. The assessed factors and their relative weights (as indicated through
%) are as follows:
Financial
condition (40%) Ownership
background (20%) Payment
record (10%)
Credit history
(10%) Market trend
(10%) Operational
size (10%)
RATING EXPLANATIONS
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
>86 |
Aaa |
Possesses an extremely sound financial base with the strongest
capability for timely payment of interest and principal sums |
Unlimited |
|
71-85 |
Aa |
Possesses adequate working capital. No caution needed for credit
transaction. It has above average (strong) capability for payment of interest
and principal sums |
Large |
|
56-70 |
A |
Financial & operational base are regarded healthy. General
unfavourable factors will not cause fatal effect. Satisfactory capability for
payment of interest and principal sums |
Fairly Large |
|
41-55 |
Ba |
Overall operation is considered normal. Capable to meet normal
commitments. |
Satisfactory |
|
26-40 |
B |
Capability to overcome financial difficulties seems comparatively
below average. |
Small |
|
11-25 |
Ca |
Adverse factors are apparent. Repayment of interest and principal sums
in default or expected to be in default upon maturity |
Limited with full
security |
|
<10 |
C |
Absolute credit risk exists. Caution needed to be exercised |
Credit not
recommended |
|
NB |
NEW BUSINESS |
||
This report is issued at your request without any
risk and responsibility on the part of MIRA INFORM PRIVATE LIMITED (MIPL) or
its officials.