MIRA INFORM REPORT
|
Report Date : |
28.02.2014 |
IDENTIFICATION DETAILS
|
Name : |
SHASUN PHARMACEUTICALS LIMITED (w.e.f. 11.08.2010) |
|
|
|
|
Formerly Known
As : |
SHASUN CHEMICALS AND DRUGS LIMITED |
|
|
|
|
Registered
Office : |
3rd and 4th Floor, The Batra Centre, Old No. 28,
Sardar Patel Road, Guindy Chennai-600032, Tamilnadu |
|
|
|
|
Country : |
|
|
|
|
|
Financials (as
on) : |
31.03.2013 |
|
|
|
|
Date of
Incorporation : |
19.04.1976 |
|
|
|
|
Com. Reg. No.: |
18–007122 |
|
|
|
|
Capital
Investment/ Paid-up Capital: |
Rs. 113.250 Millions |
|
|
|
|
CIN No.: [Company Identification
No.] |
L24117TN1976PLC007122 |
|
|
|
|
TAN No.: [Tax Deduction &
Collection Account No.] |
CHES03195D |
|
|
|
|
PAN No.: [Permanent Account No.] |
AAACS5031L |
|
|
|
|
Legal Form : |
A Public Limited Liability company. The company’s Shares are Listed on
the Stock Exchange. |
|
|
|
|
Line of Business
: |
Manufacturer and Exporter of Ibuprofen, Anti – inflammatory Drug and
Anti – Ulcerative Drug. |
|
|
|
|
No. of
Employees: |
2000 (Approximately) |
RATING & COMMENTS
|
MIRA’s Rating : |
Ba (47) |
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
41-55 |
Ba |
Overall operation is considered normal. Capable to meet normal
commitments. |
Satisfactory |
|
Maximum Credit Limit : |
USD 11400000 |
|
|
|
|
Status : |
Satisfactory |
|
|
|
|
Payment Behaviour : |
Usually Correct |
|
|
|
|
Litigation : |
Clear |
|
|
|
|
Comments : |
Subject is an established company having a satisfactory track record. There seems dip in the profitability of the company during the financial
year 2013. However, general financial position of the company seems to be sound
and healthy. Trade relations are fair. Business is active. Payment terms are
reported to be usually correct. The company can be considered for business dealings at usual trade
terms and condition. |
NOTES :
Any query related to this report can be made
on e-mail : infodept@mirainform.com
while quoting report number, name and date.
ECGC Country Risk Classification List – December 1, 2013
|
Country Name |
Previous Rating (30.09.2013) |
Current Rating (01.12.2013) |
|
India |
A1 |
A1 |
|
Risk Category |
ECGC
Classification |
|
Insignificant |
A1 |
|
Low |
A2 |
|
Moderate |
B1 |
|
High |
B2 |
|
Very High |
C1 |
|
Restricted |
C2 |
|
Off-credit |
D |
INDIAN ECONOMIC OVERVIEW
The worst is over for India’s economy with gross domestic product likely
to expand 5 %to 5.5 % this year and more than 6 % in 2015, according to Moody’s
Analytics. Concerns over the rupee and current account deficit are under
control, said the agency. Ratings firm Crisil has forecast 6 % growth for
2014/15 up from the estimated 4.8 % for 2013/14. Total economic growth,
infrastructure bottlenecks and lack of transparency and consistency in foreign
direct investment policies seem to have taken a toll on India’s attractiveness
as an investment destination, says an Ernst & Young survey. Projects
with FDI component fell 16.4 % across the globe in 2012 from the previous
year. The drop in India was steeper at 21 %. State run carrier Air India
is doling out free tickets to its 24000 employees, even as it expects to incur
a loss of Rs 39000 mn this financial year and has a debt of Rs 350000 mn.
550000 number of jobs generated across India in 2013, a fall of 0.4 % as
compared to with a year earlier. The National Capital Region has a
one-fourth share in total jobs created, according to a study by industry lobby
group Assochem, Banks, real estate, automobile and telecommunications sectors
are showing a rise of job creation. $ 805 mn investments by venture capital
firms in India during 2013, registering a drop of about 18 % over the previous
year. The Information Technology and IT-Enabled Services Industry retained
its status as the favourable venture capital investors in 2013. Pakistan has
temporarily banned gold imports for the second time in six months, as it tries
to stem smuggling into India. India’s import duty on gold is 10 % and curbs on
purchases have dried up legal imports into what used to be the world’s biggest
bullion buyers. The World Gold Council puts the amount smuggled into India at
upto 200 tonnes in 2013. The Reserve Bank of India has proposed that unclaimed
bank deposits estimated to be about Rs 35000 mn be used for education and
awareness among depositors. According to the plan, deposits that have not
been claimed for at least 10 years will be transferred to the scheme.
EXTERNAL AGENCY RATING
|
Rating Agency Name |
ICRA |
|
Rating |
BBB- (Fund Based) |
|
Rating Explanation |
Have moderate degree of safety and carry moderate credit risk. |
|
Date |
May 2013 |
|
Rating Agency Name |
ICRA |
|
Rating |
A3 (Non Fund Based) |
|
Rating Explanation |
Have moderate degree of safety and carry higher credit risk. |
|
Date |
May 2013 |
RBI DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available RBI Defaulters’ list.
EPF (Employee Provident Fund) DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available EPF (Employee Provident Fund) Defaulters’ list as of
31-03-2012.
INFORMATION PARTED BY
|
Name : |
Mr. Ramesh |
|
Designation : |
D. G. M. Finance |
|
Contact No.: |
91-44-24316700 |
|
Date : |
26.02.2014 |
LOCATIONS
|
Registered / Corporate Office : |
3rd and 4th Floor, The Batra Centre, Old No. 28,
|
|
Tel. No.: |
91-44-24316700 |
|
Fax No.: |
91-44-24348924 and 22350278 |
|
E-Mail : |
|
|
Web Site: |
|
|
Location: |
Owned |
|
|
|
|
Research Office: |
No.27, Vandalur –Keambakkam Road, Keezhakkottaiyur, Village, Chennai –
600048, Tamilnadu, India |
|
Tel. No.: |
91-44-47406100 |
|
Fax No.: |
91-44-47406190 |
|
E-Mail : |
|
|
|
|
|
API Facility – Puducherry / Bio –Tec Centre : |
|
|
Tel. No.: |
91-413-2655156/ 2655157 |
|
Fax No.: |
91-413-655154 |
|
E-Mail : |
|
|
|
|
|
Finished Dosage Facility : |
Pharmaceutical
Division |
|
Tel. No.: |
91-413-2655946 / 2655697 / 2655698 |
|
Fax No.: |
91-413-2656052 |
|
|
|
|
Multi Product Facility – Cuddalore |
A1/B SIPCOT Industrial Estate, Kudikadu, Cuddalore-607005, |
|
Tel. No.: |
91-4142-239701/ 239702/ 239703/ 239704 |
|
Fax No.: |
91-4142-239709 |
|
|
|
|
UK Branch Office : |
165 A, |
|
Tel. No.: |
+ 44 20 8560 9711 |
|
Fax No.: |
+ 44 20 8560 9455 |
|
E-Mail : |
|
|
|
|
|
Overseas Office
: |
Shasun USA
Inc, 197, Rt
18 South, Suite 102 East Brunswick, NJ 08816, USA |
|
Tel. No.: |
1-732-465-0700
* 116 |
|
Fax No.: |
1-732-465-0710 |
|
E-Mail : |
DIRECTORS
AS ON 31.03.2013
|
Name : |
Mr. R Kannan |
|
Designation : |
Chairman |
|
|
|
|
Name : |
Mr. R Sundara Rajan |
|
Designation : |
Independent Director |
|
|
|
|
Name : |
Mr. N Subramanian |
|
Designation : |
Independent Director |
|
|
|
|
Name : |
Dr Sunny Sharma |
|
Designation : |
Independent Director |
|
|
|
|
Name : |
Prof Indira J Parikh |
|
Designation : |
Independent Director |
|
|
|
|
Name : |
Mr. S Abhaya Kumar |
|
Designation : |
Managing Director |
|
|
|
|
Name : |
Dr S Devendra |
|
Designation : |
Whole Time Director |
|
|
|
|
Name : |
Mr. S Vimal Kumar |
|
Designation : |
Whole Time Director |
|
|
|
|
Name : |
Mr. M Mohan |
|
Designation : |
Whole Time Director |
KEY EXECUTIVES
|
Name : |
Mr. Mani |
|
Designation : |
Accounts Executive |
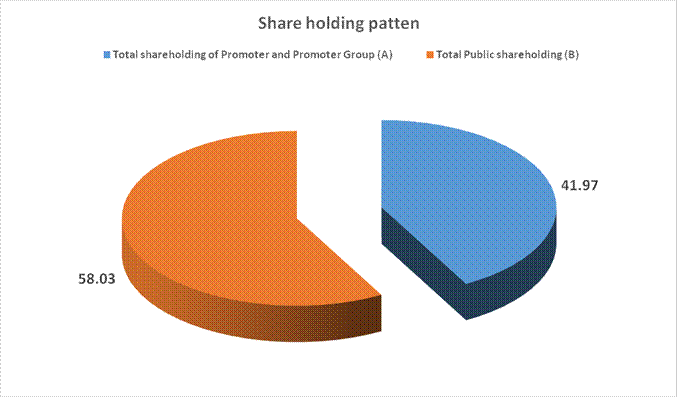
MAJOR SHAREHOLDERS / SHAREHOLDING PATTERN
AS ON 31.12.2013
|
Category of Shareholder |
No. of Shares |
% of No. of Shares |
|
(A) Shareholding of
Promoter and Promoter Group |
|
|
|
|
|
|
|
|
15999716 |
28.26 |
|
|
7762983 |
13.71 |
|
|
23762699 |
41.97 |
|
|
|
|
|
Total shareholding
of Promoter and Promoter Group (A) |
23762699 |
41.97 |
|
(B) Public
Shareholding |
|
|
|
|
|
|
|
|
3305156 |
5.84 |
|
|
134624 |
0.24 |
|
|
350950 |
0.62 |
|
|
50000 |
0.09 |
|
|
6578947 |
11.62 |
|
|
10419677 |
18.40 |
|
|
|
|
|
|
3717296 |
6.56 |
|
|
|
|
|
|
13027284 |
23.01 |
|
|
4934443 |
8.71 |
|
|
762453 |
1.35 |
|
|
759953 |
1.34 |
|
|
1500 |
0.00 |
|
|
1000 |
0.00 |
|
|
22441476 |
39.63 |
|
Total Public
shareholding (B) |
32861153 |
58.03 |
|
Total (A)+(B) |
56623852 |
100.00 |
|
(C) Shares held by Custodians
and against which Depository Receipts have been issued |
0 |
0.00 |
|
|
0 |
0.00 |
|
|
0 |
0.00 |
|
|
0 |
0.00 |
|
Total (A)+(B)+(C) |
56623852 |
0.00 |
BUSINESS DETAILS
|
Line of Business : |
Manufacturer and Exporter of Ibuprofen, Anti – inflammatory Drug and
Anti – Ulcerative Drug. |
|
|
|
|
Exports : |
|
|
Products : |
Finished Goods |
|
Countries : |
|
|
|
|
|
Imports : |
|
|
Products : |
Raw Materials |
|
Countries : |
|
|
|
|
|
Terms : |
|
|
Selling : |
L/C, Credit |
|
|
|
|
Purchasing : |
L/C, Credit |
GENERAL INFORMATION
|
Customers : |
Retailers and End Users |
||||||||||||||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||||||||||||||
|
No. of Employees : |
2000 (Approximately) |
||||||||||||||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||||||||||||||
|
Bankers : |
|
||||||||||||||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||||||||||||||
|
Facilities : |
Cash Credit: Rs. 2200.000 Millions
|
||||||||||||||||||||||||||||||||||||
|
|
|
|
Banking
Relations : |
-- |
|
|
|
|
Auditors : |
|
|
Name : |
BSR and Company Chartered Accountant |
|
Address : |
No.10, |
|
Tel. No.: |
91-44-3914 5000 |
|
Fax No.: |
91-44-3914 5999 |
|
|
|
|
Cost Auditor : |
|
|
Name : |
Geeyes and Company Chartered Accountant |
|
Address : |
A-3, III Floor,
56, Seventh Avenue, Ashok Nagar, Chennai-600083, Tamilnadu, India |
|
|
|
|
Wholly owned
subsidiaries : |
|
|
|
|
|
Wholly owned
step down subsidiaries : |
|
|
|
|
|
Joint Venture : |
|
|
|
|
|
Entities where Directors
have control or significant influence : |
|
CAPITAL STRUCTURE
AS ON 31.03.2013
Authorised Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
75000000 |
Equity Shares |
Rs.2/- each |
Rs. 150.000 Millions |
|
1000000 |
Redeemable Preference Shares |
Rs.100/- each |
Rs. 100.000 Millions |
|
|
TOTAL |
|
Rs. 250.000
Millions |
Issued, Subscribed & Paid-up Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
56623852 |
Equity Shares |
Rs.2/- each |
Rs. 113.250
Millions |
|
|
|
|
|
FINANCIAL DATA
[all figures are
in Rupees Millions]
ABRIDGED BALANCE
SHEET
|
SOURCES OF FUNDS |
31.03.2013 |
31.03.2012 |
31.03.2011 |
|
I.
EQUITY
AND LIABILITIES |
|
|
|
|
(1)Shareholders' Funds |
|
|
|
|
(a) Share Capital |
113.250 |
110.250 |
97.090 |
|
(b) Reserves & Surplus |
2759.400 |
2486.490 |
1617.810 |
|
(c) Money
received against share warrants |
0.000 |
28.130 |
0.000 |
|
|
|
|
|
|
(2) Foreign Currency monetary item
translation difference account |
(20.560) |
(9.950) |
0.000 |
|
Total
Shareholders’ Funds (1) + (2) |
2852.090 |
2614.920 |
1714.900 |
|
|
|
|
|
|
(3)
Non-Current Liabilities |
|
|
|
|
(a) long-term borrowings |
985.300 |
490.530 |
359.900 |
|
(b) Deferred tax liabilities (Net) |
0.000 |
15.030 |
129.840 |
|
(c) Other long term
liabilities |
10.400 |
0.800 |
0.000 |
|
(d) long-term
provisions |
59.680 |
51.370 |
156.280 |
|
Total Non-current
Liabilities (3) |
1055.380 |
557.730 |
646.020 |
|
|
|
|
|
|
(4) Current
Liabilities |
|
|
|
|
(a) Short
term borrowings |
2720.040 |
1652.510 |
1599.620 |
|
(b) Trade
payables |
1763.650 |
1552.660 |
1176.180 |
|
(c) Other
current liabilities |
637.070 |
516.780 |
351.160 |
|
(d) Short-term
provisions |
123.300 |
225.040 |
159.050 |
|
Total Current
Liabilities (4) |
5244.060 |
3946.990 |
3286.010 |
|
|
|
|
|
|
TOTAL |
9151.530 |
7119.640 |
5646.930 |
|
|
|
|
|
|
II.
ASSETS |
|
|
|
|
(1)
Non-current assets |
|
|
|
|
(a) Fixed
Assets |
|
|
|
|
(i)
Tangible assets |
2663.890 |
1938.500 |
1659.100 |
|
(ii)
Intangible Assets |
9.960 |
4.790 |
2.640 |
|
(iii)
Capital work-in-progress |
773.360 |
308.090 |
49.340 |
|
(iv)
Intangible assets under development |
35.210 |
0.000 |
0.000 |
|
(b) Non-current Investments |
509.000 |
50.400 |
23.490 |
|
(c) Deferred tax assets (net) |
11.410 |
0.000 |
0.000 |
|
(d) Long-term Loan and Advances |
701.080 |
1000.150 |
789.670 |
|
(e) Other
Non-current assets |
11.310 |
5.290 |
70.280 |
|
Total Non-Current
Assets |
4715.220 |
3307.220 |
2594.520 |
|
|
|
|
|
|
(2)
Current assets |
|
|
|
|
(a)
Current investments |
0.000 |
0.000 |
0.000 |
|
(b)
Inventories |
1453.330 |
1106.880 |
1048.740 |
|
(c) Trade
receivables |
2206.760 |
1970.550 |
1379.200 |
|
(d) Cash
and cash equivalents |
39.740 |
66.270 |
106.500 |
|
(e)
Short-term loans and advances |
680.100 |
593.710 |
441.140 |
|
(f) Other
current assets |
56.380 |
75.010 |
76.830 |
|
Total Current
Assets |
4436.310 |
3812.420 |
3052.410 |
|
|
|
|
|
|
TOTAL |
9151.530 |
7119.640 |
5646.930 |
PROFIT & LOSS
ACCOUNT
|
|
PARTICULARS |
31.03.2013 |
31.03.2012 |
31.03.2011 |
|
|
|
SALES |
|
|
|
|
|
|
|
Income |
7827.560 |
7359.090 |
5671.260 |
|
|
|
Other Income |
136.550 |
409.640 |
160.040 |
|
|
|
TOTAL (A) |
7964.110 |
7768.730 |
5831.300 |
|
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
|
|
Cost of Material Consumed |
5009.680 |
4655.130 |
3812.790 |
|
|
|
Purchase of Stock In Trade |
17.740 |
42.920 |
11.830 |
|
|
|
Employee benefits expenses |
767.430 |
595.240 |
516.460 |
|
|
|
Other Expenses |
1529.270 |
1562.020 |
1245.930 |
|
|
|
Changes in inventories of Finished goods, work in progress and stock
in trade |
(177.840) |
(11.400) |
(304.800) |
|
|
|
TOTAL (B) |
7146.280 |
6843.910 |
5282.210 |
|
|
|
|
|
|
|
|
Less |
PROFIT
/ (LOSS) BEFORE INTEREST, TAX, DEPRECIATION AND AMORTISATION (A-B) (C) |
817.830 |
924.820 |
549.090 |
|
|
|
|
|
|
|
|
|
Less |
FINANCIAL
EXPENSES (D) |
264.870 |
304.810 |
223.350 |
|
|
|
|
|
|
|
|
|
|
PROFIT
/ (LOSS) BEFORE TAX, DEPRECIATION AND AMORTISATION (C-D) (E) |
552.960 |
620.010 |
325.740 |
|
|
|
|
|
|
|
|
|
Less/ Add |
DEPRECIATION/
AMORTISATION (F) |
316.620 |
247.280 |
242.150 |
|
|
|
|
|
|
|
|
|
|
PROFIT / (LOSS)
BEFORE TAX (E-F) (G) |
236.340 |
372.730 |
83.590 |
|
|
|
|
|
|
|
|
|
Less |
TAX (H) |
(26.440) |
(157.850) |
34.820 |
|
|
|
|
|
|
|
|
|
|
PROFIT / (LOSS)
AFTER TAX (G-H) (I) |
262.780 |
530.580 |
48.770 |
|
|
|
|
|
|
|
|
|
|
Mark to Market loss
option contracts relating to prior
periods |
0.000 |
0.000 |
(405.320) |
|
|
|
|
|
|
|
|
|
|
NET
PROFIT / (LOSS) FOR THE YEAR |
262.780 |
530.580 |
(356.550) |
|
|
|
|
|
|
|
|
|
|
EARNINGS IN
FOREIGN CURRENCY |
|
|
|
|
|
|
|
Export Earnings |
5101.320 |
4981.450 |
3901.450 |
|
|
|
Product Development fee |
68.920 |
54.940 |
63.820 |
|
|
|
Contract research fees |
48.940 |
29.610 |
3.640 |
|
|
|
Interest and Dividend |
18.720 |
16.630 |
41.590 |
|
|
|
Miscellaneous Income |
20.470 |
0.000 |
0.000 |
|
|
TOTAL EARNINGS |
5258.370 |
5082.630 |
4010.500 |
|
|
|
|
|
|
|
|
|
|
IMPORTS |
|
|
|
|
|
|
|
Raw Materials |
1304.290 |
1362.260 |
1115.730 |
|
|
|
Capital Goods & Spares Parts |
179.910 |
172.850 |
30.840 |
|
|
TOTAL IMPORTS |
1484.200 |
1535.110 |
1146.570 |
|
|
|
|
|
|
|
|
|
|
Earnings /
(Loss) Per Share (Rs.) |
4.76 |
10.92 |
(7.34) |
|
Expected Sales (2013-2014): Rs. 9850.000
The above information has been parted by Mr. Ramesh
QUARTERLY RESULTS
|
PARTICULARS |
30.06.2013 |
|
|
1st
Quarter |
|
Net Sales |
1759.500 |
|
Total Expenditure |
1688.400 |
|
PBIDT (Excl OI) |
71.100 |
|
Other Income |
14.700 |
|
Operating Profit |
85.800 |
|
Interest |
77.000 |
|
Exceptional Items |
0.000 |
|
PBDT |
8.800 |
|
Depreciation |
86.400 |
|
Profit Before Tax |
(77.600) |
|
Tax |
(102.700) |
|
Provisions and contingencies |
0.00 |
|
Profit After Tax |
25.100 |
|
Extraordinary Items |
0.000 |
|
Prior Period Expenses |
0.000 |
|
Other Adjustments |
0.000 |
|
Net Profit |
25.100 |
KEY RATIOS
|
PARTICULARS |
|
31.03.2013 |
31.03.2012 |
31.03.2011 |
|
PAT / Total Income |
(%) |
3.30
|
6.83 |
0.84 |
|
|
|
|
|
|
|
Net Profit Margin (PBT/Sales) |
(%) |
3.02
|
5.06 |
1.47 |
|
|
|
|
|
|
|
Return on Total Assets (PBT/Total Assets} |
(%) |
3.02
|
5.51 |
1.50 |
|
|
|
|
|
|
|
Return on Investment (ROI) (PBT/Networth) |
|
0.08
|
0.14 |
0.05 |
|
|
|
|
|
|
|
Debt Equity Ratio (Total Debt /Networth) |
|
1.30
|
0.82 |
1.14 |
|
|
|
|
|
|
|
Current Ratio (Current Asset/Current Liability) |
|
0.85
|
0.97 |
0.93 |
FINANCIAL ANALYSIS
[all figures are
in Rupees Millions]
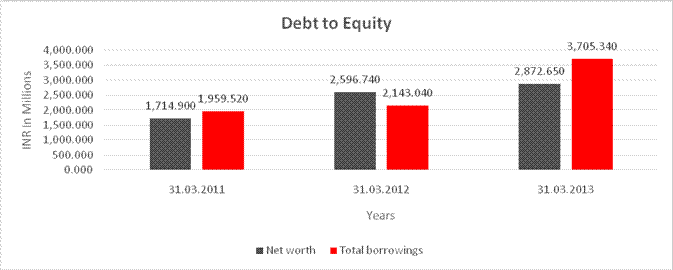
DEBT EQUITY RATIO
|
Particular |
31.03.2011 |
31.03.2012 |
31.03.2013 |
|
|
(Rs.
In Millions) |
(Rs.
In Millions) |
(Rs.
In Millions) |
|
Share Capital |
97.090 |
110.250 |
113.250 |
|
Reserves & Surplus |
1,617.810 |
2,486.490 |
2,759.400 |
|
Net
worth |
1,714.900 |
2,596.740 |
2,872.650 |
|
|
|
|
|
|
long-term borrowings |
359.900 |
490.530 |
985.300 |
|
Short term borrowings |
1,599.620 |
1,652.510 |
2,720.040 |
|
Total
borrowings |
1,959.520 |
2,143.040 |
3,705.340 |
|
Debt/Equity
ratio |
1.143 |
0.825 |
1.290 |
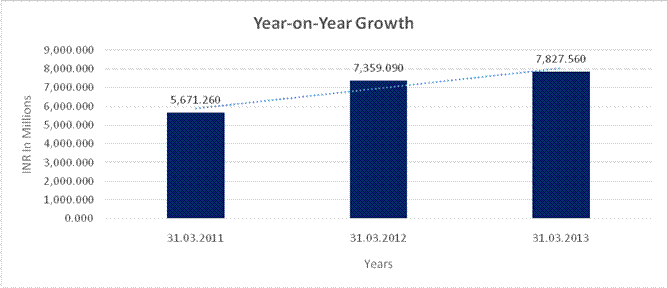
YEAR-ON-YEAR GROWTH
|
Year
on Year Growth |
31.03.2011 |
31.03.2012 |
31.03.2013 |
|
|
(Rs.
In Millions) |
(Rs.
In Millions) |
(Rs.
In Millions) |
|
Sales |
5,671.260 |
7,359.090 |
7,827.560 |
|
|
|
29.761 |
6.366 |
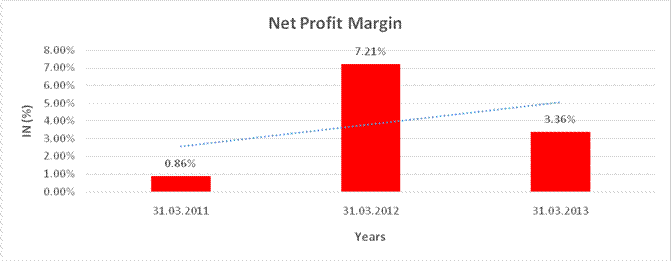
NET PROFIT MARGIN
|
Net
Profit Margin |
31.03.2011 |
31.03.2012 |
31.03.2013 |
|
|
(Rs.
In Millions) |
(Rs.
In Millions) |
(Rs.
In Millions) |
|
Sales |
5,671.260 |
7,359.090 |
7,827.560 |
|
Profit |
48.770 |
530.580 |
262.780 |
|
|
0.86% |
7.21% |
3.36% |
LOCAL AGENCY FURTHER INFORMATION
|
Sr. No. |
Check List by Info Agents |
Available in
Report (Yes / No) |
|
1] |
Year of Establishment |
Yes |
|
2] |
Locality of the firm |
Yes |
|
3] |
Constitutions of the firm |
Yes |
|
4] |
Premises details |
No |
|
5] |
Type of Business |
Yes |
|
6] |
Line of Business |
Yes |
|
7] |
Promoter's background |
No |
|
8] |
No. of employees |
Yes |
|
9] |
Name of person contacted |
Yes |
|
10] |
Designation of contact
person |
Yes |
|
11] |
Turnover of firm for last
three years |
Yes |
|
12] |
Profitability for last
three years |
Yes |
|
13] |
Reasons for variation
<> 20% |
----- |
|
14] |
Estimation for coming
financial year |
Yes |
|
15] |
Capital in the business |
Yes |
|
16] |
Details of sister
concerns |
Yes |
|
17] |
Major suppliers |
No |
|
18] |
Major customers |
No |
|
19] |
Payments terms |
Yes |
|
20] |
Export / Import details
(if applicable) |
Yes |
|
21] |
Market information |
----- |
|
22] |
Litigations that the firm
/ promoter involved in |
----- |
|
23] |
Banking Details |
Yes |
|
24] |
Banking facility details |
Yes |
|
25] |
Conduct of the banking
account |
----- |
|
26] |
Buyer visit details |
----- |
|
27] |
Financials, if provided |
Yes |
|
28] |
Incorporation details, if
applicable |
Yes |
|
29] |
Last accounts filed at
ROC |
Yes |
|
30] |
Major Shareholders, if
available |
No |
|
31] |
Date of Birth of
Proprietor/Partner/Director, if available |
No |
|
32] |
PAN of Proprietor/Partner/Director,
if available |
No |
|
33] |
Voter ID No of
Proprietor/Partner/Director, if available |
No |
|
34] |
External Agency Rating,
if available |
Yes |
|
Unsecured Loan |
Rs.
In Millions 31.03.2013 |
Rs.
In Millions 31.03.2012 |
|
LONG-TERM
BORROWINGS |
|
|
|
Loan from others |
10.740 |
0.000 |
|
SHORT TERM
BORROWINGS |
|
|
|
Other loans from banks |
547.540 |
312.840 |
|
|
|
|
|
TOTAL |
558.280 |
312.840 |
INDEX OF CHARGES
|
S. No |
Charge ID |
Date of Charge Creation /Modification |
Charge amount secured |
Charge Holder |
Address |
Service Request Number (SRN |
|
1 |
10464661 |
19/11/2013 |
300,000,000.00 |
STATE BANK OF MYSORE |
INDUSTRIAL FINANCE BRANCH, SECOND FLOOR, MOH
BLDG., 576 ANNA SALAI, CHENNAI, TAMIL NADU - 600006, INDIA |
B91325217 |
|
2 |
10454803 |
03/10/2013 |
500,000,000.00 |
STATE BANK OF TRAVANCORE |
COMMERCIAL BRANCH, JEEVAN ANAND, 556,
MOUNT ROAD, TEYNAMPET, CHENNAI, TAMIL NADU - 600018, INDIA |
B87450532 |
|
3 |
10445370 |
19/08/2013 |
300,000,000.00 |
STATE BANK OF INDIA |
COMMERCIAL BRANCH, NO.232 NSC BOSE ROAD,
CHENNAI, TAMIL NADU - 600001,
INDIA |
B83179440 |
|
4 |
10399371 |
02/01/2013 |
553,000,000.00 |
DBS BANK LIMITED. |
806, ANNA SALAI, CHENNAI, TAMIL NADU -
600002, INDIA |
B66396938 |
|
5 |
10374612 |
30/08/2012 |
52,000,000.00 |
STATE BANK OF INDIA |
COMMERCIAL BRANCH, NO.232 NSC BOSE ROAD,
CHENNAI, TAMIL NADU - 600001,
INDIA |
B57336117 |
|
6 |
10363032 |
05/09/2013 * |
753,500,000.00 |
AXIS BANK LIMITED |
CORPORATE BANKING BRANCH, KARUMUTHU
NILAYAM, NO.1 92, ANNA SALAI, CHENNAI, TAMIL NADU - 600002, INDIA |
B86735701 |
|
7 |
10363184 |
13/06/2012 |
450,000,000.00 |
STATE BANK OF INDIA |
COMMERCIAL BRANCH, NO.232 NSC BOSE ROAD,
CHENNAI, TAMIL NADU - 600001,
INDIA |
B42701110 |
|
8 |
10340690 |
30/08/2012 * |
50,000,000.00 |
STATE BANK OF TRAVANCORE |
COMMERCIAL BRANCH, JEEVAN ANAND, 556,
MOUNT ROAD, TEYNAMPET, CHENNAI,
TAMIL NADU - 600018, INDIA |
B57370173 |
|
9 |
10297652 |
30/11/2011 * |
270,000,000.00 |
ICICI BANK LIMITED |
LANDMARKRACE COURCE CIRCLE, ALKAPURI,
BARODA, GUJARAT - 390015, INDIA |
B26741348 |
|
10 |
10262659 |
09/07/2011 * |
246,300,000.00 |
STATE BANK OF INDIA |
COMMERCIAL BRANCH, 232, N.S.C.BOSE ROAD,
CHENNAI, TAMIL NADU - 600001,
INDIA |
B16868804 |
|
11 |
10245653 |
05/10/2012 * |
192,500,000.00 |
DBS BANK LIMITED |
806, ANNASALAI, CHENNAI, TAMIL NADU -
600002, INDIA |
B59780585 |
|
12 |
10240157 |
13/09/2010 |
145,400,000.00 |
AXIS BANK LIMITED |
CMC, KARUMUTHU NILAYAM, NO.192, ANNA
SALAI,, CHENNAI, TAMIL NADU - 600002, INDIA |
A94539111 |
|
13 |
10196450 |
09/01/2013 * |
279,000,000.00 |
STATE BANK OF INDIA |
COMMERCIAL BRANCH, NO.232 NSC BOSE ROAD,
CHENNAI, TAMIL NADU - 600001,
INDIA |
B68082387 |
|
14 |
90292831 |
15/04/2004 * |
92,800,000.00 |
STATE BANK OF INDIA |
COMMERCIAL BRANCH; 232; N.S.C. BOSE ROAD,
CHENNAI, TAMIL NADU - 600001,
INDIA |
- |
|
15 |
90292714 |
24/05/1999 * |
16,700,000.00 |
EXPORT IMPORT BANK OF INDIA |
CENTRE ONE; WORLD TRADE CENTRE, CUFFE
PARADE, MUMBAI, MAHARASHTRA - 400005, INDIA |
- |
|
16 |
90293805 |
23/02/1999 |
16,700,000.00 |
EXPORT IMPORT BANK OF INDIA |
CENTRE ONE; WORLD TRADE CENTRE, CUFFE
PARADE, MUMBAI, MAHARASHTRA - 400005, INDIA |
- |
|
17 |
90292397 |
16/07/1996 |
6,000,000.00 |
M/S. PONDICHERRY INDUSTRIAL PROMOTION
DEVLOPMENT C |
PONDICHEERY, PONDICHERRY, PONDICHERRY,
INDIA |
- |
|
18 |
90292330 |
24/05/1996 * |
80,000,000.00 |
EXPORT IMPORTBANK OF INDIA |
CENTRE ONE; WORLD TREAD CENTRE;, CUFFE
PARADE, BOMBAY, PONDICHERRY - 400005, INDIA |
- |
|
19 |
90295367 |
14/02/1995 |
32,900,000.00 |
INDIANRENEWABLE ENERGY DEVLOPMENTAGENCY
LIMITED. |
CORA 4A; EAST COURT; 1ST FLOOR, INDIAN
HABITAT CENTRE; LODI ROAD, NRE DELHI, DELHI - 110003, INDIA |
- |
|
20 |
90292172 |
30/08/2012 * |
3,700,000,000.00 |
STATE BANK OF TRAVANCORE |
STATE BANK OF TRAVANCORE, COMMERCIAL BRANCH,
JEEVAN ANAND, CHENNAI, TAMIL NADU - 600018, INDIA |
B57286189 |
|
21 |
90293342 |
17/06/1993 |
24,000,000.00 |
THE GOVT OF TAMILNADU |
COMMERCIAL TAXES, MADRAS, TAMIL NADU,
INDIA |
- |
|
22 |
90293511 |
07/04/1998 * |
24,000,000.00 |
THE GOVT. OF TAMILNADU |
DEPATMENT OF COMMERCIQL TAXES, CHENNAI,
TAMIL NADU, INDIA |
- |
|
23 |
90292114 |
30/05/1992 |
21,500,000.00 |
M/S.INTEGRATED FINANCE COMP. LIMITED. |
‘VAIRAMS'; 112; THYAGARAYA ROAD, T. NAGAR,
MADRAS, GUJARAT - 600017, INDIA |
- |
|
* Date of charge modification |
||||||
COMPANY OVERVIEW
Subject was
incorporated in 1976 having its registered office in Chennai, India. The
Company is primarily engaged in manufacturing of Active Pharmaceutical Ingredients
(APIs), their intermediates and finished dosage. The Company is also into
product development and provides contract research and manufacturing services.
MANAGEMENT
DISCUSSION AND ANALYSIS
COMPANY PERFORMANCE
The company is
cognizant of the various emerging trends in the global pharmaceutical landscape
and has taken a lot of initiatives to ensure that it is well positioned to
leverage the opportunities that arise out of such trends.
The following section discusses such initiatives on a divisional basis
API BUSINESS
On a consolidated
basis, API and its intermediate business contributed close to 52% of the
turnover of the company. On a standalone basis, they contributed close to 73%
of the total sales.
The Company’s
strategy with respect to this SBU is to enhance sales and profitability. This
can be achieved through the following measures:
Capacity
enhancement of the existing products to meet the growing demand Commercializing
new products which are New products development to improve pipeline of products
Process Improvement initiatives
Shasun’s current
API portfolio consists of Ibuprofen, Ranitidine, Nizatidine, Gabapentin,
Levetiracetan and Cycloserine, amongst others.
The Company has filed 5 new DMFs and 2 ANDAs in the current financial year.
In this current financial year, the API SBU has achieved a sales revenue
of Rs.5316.22 Mn.
OUTLOOK FOR API BUSINESS
The company continues to enjoy significant traction in its core APIs.
New products introduction is major initiative in FY 14 to enhance the
sales and profitability.
Currently, the
Company is investing in new facilities to manufacture three new APIs, which are
to be launched in FY15. This will bring significant volume and value to the
Company. The USDMFs filed by the company has been triggered by various
customers
This year the
Company is investing in Vizag SEZ to create manufacturing capacities for API
business Segment
While North
America and Europe continue to be key markets for the Company, other markets
such as Korea, Japan, and Brazil have presented new opportunities thereby
opening more avenues for the Company.
Contract Research
and Manufacturing Services Business
The Company has an active presence in CRAMS in both APIs and
Formulations
API CRAMS includes
- Shasun Pharma Solutions Limited, UK
- CRAMS SBU in India
FORMULATIONS CRAMS INCLUDES
Finished Dosages
facility in Pondicherry caters to contract manufacturing and contract
development of finished dosages for Innovator, Emerging and Generic pharma
companies.
Formulation research centre in Pondicherry provides support for
development of new formulation products.
During FY13, CRAMS
consolidated business registered the revenue of Rs 4,899 Mn, comprising 48% of
the total consolidated revenue of Shasun and registering a growth of 3% over
previous year.
SHASUN PHARMA
SOLUTIONS LIMITED, UK( SPSL)
OPERATIONS-SPSL
During FY13, SPSL
has continued to focus on its core strengths of safety and quality compliance, continuous
improvement, and innovation.
Safety performance
and compliance were maintained at an industry leading level. The year saw the
successful completion of several scheduled major hazard regulatory inspections
on key risk control systems with no major observations. In addition, their
safety performance and commitment to ensuring the safety of their employees has
been reinforced through a number of initiatives. Working with the Health and
Safety Executive on a competency project engaging all employees and a waste
minimization project working closely with the UK Environmental Agency. REACH
compliance was maintained and expert support and advise continues to be
provided to our customer base as part of their service offering.
Compliance with
cGMP standards was acknowledged through customer visits and eighteen quality
audits at their facility. The Company continued its excellent performance with
respect to quality performance indicators with a Process right first time of
98%. The company has invested around GBP 3 Million at Dudley facility to
capacity and capability enhancement and to infrastructure upgrades.
SALES-SPSL
Market interest
continues to be driven by their key strengths – technical expertise and
capability, speed of response, good facilities, their people and their
reliability.
SPSL has a unique
ability to handle both highly complex and hazardous projects. A world class
hazard evaluation capability which is fully integrated into their process
development group and project management process allows early understanding and
appropriate measures to ensure safe execution. This approach is seen as an
advantage over their competitors by several of their customers.
SPSL UK has
achieved sales GBP 35.23 Million and posted PAT of GBP 3.69 million. Focus on
high value added, technically challenging projects has enabled SPSL to return a
good PAT performance, despite pressure on sales.
In 2012, SPSL
hosted ninety customer visits to the Dudley site, of which 50% were new and
prospective clients. They have also seen improvements in other key performance
metrics including number of inquiries, number of proposals and proposal closure
rate, generating a number of good prospects for their future pipeline.
SCIENCE AND TECHNOLOGY – SPSL
Over the past year
they have continued to focus on science and technology as a key initiative to
differentiate their offering. They continue to develop our HKR (Hydrolytic
Kinetic Resolution) technology and broaden its application, In 2012 a key
milestone was achieved with the commercialization of their 4th generation Salen
catalyst and novel synthesis for Hyperzine A (natural product).
OUTLOOK FOR SPSL
In 2012 SPSL had a
pipeline of 24 launched products and grew its pre-launch pipeline to 27
projects spanning all clinical phases. The pipeline remains strong with 9
exciting projects in Phase III, 2 of which have been filed and are pending
regulatory approval. The company, through its excellent level of service
coupled with it’s proprietary technology and innovation, is well placed to take
advantage of the increased outsourcing opportunities presented by both
established and emerging pharma customers. The company remains confident that
based upon current orders and new business opportunities identified for 2013-14
it can sustain sales and coupled with planned plant efficiency improvements
ensure the company remains profitable.
CRAMS SBU IN INDIA
On the Contract
Manufacturing front in India, the Company has posted a growth of 26% by successfully
capitalising on the global growth in the contract manufacturing business
FINISHED DOSAGES BUSINESS – FORMULATIONS CRAMS
The company offers
the services of development and manufacture of finished dosages to both
Innovator and generic companies worldwide. The division achieved sales of
Rs.1472 Mn during FY13, a substantial growth of 75% over the previous year.
The Finished
Dosages facility has undergone successful inspections by various regulatory
bodies such as WHO Geneva, NDA Uganda, PMPB Malawi, TFDA Tanzania, MCAZ
Zimbabwe and ROA Ivory Coast during FY13.
During this
period, it has also five products for registration with various regulatory
agencies and has received one approval for a product previously filed for
registration.
OUTLOOK FOR FORMULATION CRAMS
Finished Dosages
Business remains a major opportunity for Shasun to increase and diversify its
revenue base. With the increased expansion capacity and expected approvals on
newer Products, the Formulation Division is planning to increase its revenues
from FY13.The development work on ANDAs have commenced with a goal of filing
atleast 10 ANDAs and 2 CTDs for the European market in FY 14. The outlook for
the Formulations division is robust and the Company is preparing plans to
initiate work on Phase-III expansion later part of this year so as to increase
the capacity to meet the growing needs of its existing customers, and it is
also preparing for new product approvals.
The service levels
to their existing customers have improved with the changing needs, taking their
partnership with customers to newer heights. Customers have started adding new
products from their formulation facility.
BIOTECHNOLOGY
The division
achieved Sales growth of 58% for its flagship recombinant streptokinase product
and continued to strengthen its position as a leading manufacturer for
streptokinase in India.
The product is also under export registration in some of the key
developing countries.
While its other
pipeline biosimilar molecules are progressing in development, the division is
also drawing up strategic plans for future growth on a sustainable and
differentiated basis.
NANOTECHNOLOGY
The company is one
of the first few pharmaceutical companies in India to have embarked on
nanotechnology based research and drug development. As the application and
potential of nanotechnology keeps validating through numerous research papers
and partnering deals worldwide, the company is well-positioned to leverage its
knowledge and skills in this sunrise area.
The company is
involved in a lead development compound based on the nanotechnology platform
with its joint venture partner – Nanoparticle Biochem Inc., USA and support from
the University of Missouri, USA. The compound is a radioactive gold
nanoparticle and is being investigated for treatment of solid tumours.
Extensive preclinical testing is underway in animals and these include studies
in both rodent and non-rodents like dogs and pigs.
A clinical study
has also been undertaken for treating prostate-cancer bearing patient dogs and
the compound has shown promising evidence of increase in survival time, tumour
regression and retention within the tumour.
The plan is for completing
the rest of the identified preclinical studies and take the product for
Investigational New Drug (IND) filing in both India and USA A Scientific
Advisory team comprising of well-known experts has also been formed in India
and they are guiding the team in the development of this molecule.
With its
nanotechnology foray, the company has also undertaken another area of research
into polymeric nanoparticles with the objective of developing a drug delivery
platform that can be applied across various molecules. This research is
currently underway with the target of establishing the preliminary proof-of
concept of the proposed technology platform.
CONSOLIDATED FINANCIAL REVIEW
The Company has
registered a consolidated Gross revenue of Rs.11030.52 Mn as compared to the
previous year figure of Rs.11073.23 Mn.
Raw material cost
as a proportion of total net income decreased to 50.33% in FY13 from 52.64% in
FY12. This is mainly due to product sales mix and process improvement in
various products.
Other operating expenses increase in line with increase in sales growth.
The Company has
achieved PAT of Rs.530.36 Mn. in FY13 as compared to the previous year figure
of Rs.1005.67 Mn.
FOREX FORWARD CONTRACTS
The Company has
completed all forex forward contracts during October, 2012 and there is no
pending contracts.
FIXED ASSETS
- Land
- Leasehold Land
- Buildings - Factory
- Building Non Factory
- Machinery
- Electrical installations
- Lab Equipments
- Office Equipments
- Factory Equipments
- R and D Equipments
- Fixtures and fittings
- Vehicles
- Computers Software
STATEMENT OF
STANDALONE UNAUDITED RESULTS FOR THE QUARTER AND NINE MONTHS ENDED DECEMBER 31,
2013
(Rs. in millions)
|
Sr. No. |
Particular |
Quarter Ended |
Half Year Ended |
|
|
|
|
31.12.2013 (Unaudited) |
30.09.2013 (Unaudited) |
31.12.2013 (Unaudited) |
|
1. |
Income from
Operations |
|
|
|
|
|
Net Sales |
2314.300 |
2115.700 |
6086.200 |
|
|
Other Operating Income |
151.100 |
116.700 |
371.100 |
|
|
Net Sales/Income
from Operations |
2465.400 |
2232.400 |
6457.300 |
|
|
|
|
|
|
|
2. |
Expenditure |
|
|
|
|
|
Cost of Material Consumed
|
1481.100 |
1352.000 |
3968.600 |
|
|
Purchase in Stock In Trade |
10.400 |
37.800 |
75.500 |
|
|
Change in Inventories of Finished Goods, Work-In-Progress
and Stock In Trade |
(100.300) |
(183.600) |
(365.500) |
|
|
Employee Benefits Expenses |
260.900 |
238.600 |
718.300 |
|
|
Depreciation and Amortization Expenses |
93.900 |
95.500 |
275.800 |
|
|
Foreign exchange loss |
38.600 |
23.000 |
57.200 |
|
|
Other Expenses |
501.900 |
499.700 |
1390.000 |
|
|
f) Total |
2286.500 |
2063.000 |
6119.900 |
|
|
|
|
|
|
|
3. |
Profit
From Operations before Other Income, Interest and Exceptional Items (1-2) |
178.900 |
169.400 |
337.400 |
|
4. |
Other Income |
31.900 |
34.900 |
77.100 |
|
5. |
Profit
Before Interest and Exceptional Items (3+4) |
210.800 |
204.300 |
414.500 |
|
6. |
Interest |
110.300 |
88.500 |
275.800 |
|
7. |
Profit
After Interest but before Exceptional Items (5-6) |
100.500 |
115.800 |
138.700 |
|
8. |
Exceptional Items |
-- |
-- |
-- |
|
9. |
Profit
from Ordinary Activities before Tax (7+8) |
100.500 |
115.800 |
138.700 |
|
10. |
Tax Expense |
0.700 |
(0.400) |
103.000 |
|
11. |
Net
Profit from Ordinary Activities after Tax (9-10) |
101.200 |
115.400 |
241.700 |
|
12. |
Extraordinary Item (net of expense) |
-- |
-- |
-- |
|
13. |
Net
Profit for the period (11-12) |
101.200 |
115.400 |
241.700 |
|
14. |
Paid-up Equity Share Capital (Face Value of Rs.10/- Each) |
113.300 |
113.300 |
113.300 |
|
15. |
Reserves Excluding Revaluation Reserve |
-- |
-- |
-- |
|
16. |
Basic
and Diluted Earning Per Share (EPS) (Rs.)-Not Annualised |
|
|
|
|
|
a) Basic and diluted EPS before extraordinary items |
1.79 |
2.04 |
4.27 |
|
|
b) Basic and diluted EPS after extraordinary items |
1.79 |
2.04 |
4.27 |
|
|
|
|
|
|
|
17. |
Public
Shareholding |
|
|
|
|
|
-Number of Shares |
32861153 |
32861153 |
32861153 |
|
|
- Percentage of Shareholding |
58.03% |
58.03% |
58.03% |
|
18. |
Promoters
and Promoter Group Shareholding |
|
|
|
|
|
a)
Pledged/Encumbered |
|
|
|
|
|
- Number of Shares |
7538000 |
5773000 |
7538000 |
|
|
- Percentage of Shares (as a % of the Total Shareholding
of promoter and promoter group) |
31.72% |
24.29% |
31.72% |
|
|
- Percentage of Shares (as a % of the Total Share Capital
of the Company) |
13.31% |
10.20% |
13.31% |
|
|
b)
Non Encumbered |
|
|
|
|
|
- Number of Shares |
16224699 |
17989699 |
16224699 |
|
|
- Percentage of Shares (as a % of the Total Shareholding
of Promoter and Promoter Group) |
68.28% |
75.71% |
68.28% |
|
|
- Percentage of Shares (as a % of the Total Share Capital
of the Company) |
28.66% |
31.77% |
28.66% |
|
Particulars
|
31.12.2013 |
|
Pending at the beginning of the quarter |
Nil |
|
Received during the quarter |
Nil |
|
Disposed of during the quarter |
Nil |
|
Remaining unresolved at the end of the quarter |
Nil |
The above results of the Company were reviewed by the Audit Committee and approved by the Board of Directors at their respective meetings held on February 07, 2014.
The statutory auditors have carried out a limited review of the results for the quarter and nine months ended December 31, 2013 and have expresses an unqualified opinion.
The Company has identified "Pharmaceuticals" as its single reportable business segment. Pharmaceuticals segment comprises manufacture of Active Pharmaceuticals Ingredients (API), Intermediates and Formulations.
Effective April 01, 2013 based on the recognition and measurement principles set out in the Accounting Standard (AS-30) on Financial Instruments: Recognition and Measurement, the charges in the derivative fair values relating to forward contracts that are designated as effective cash flow hedges, has been recognized directly in shareholders' funds until the hedged transactions occur. As a result of this change, current liabilities have increased by Rs. 10.700 Millions and Rs. 65.500 million and the shareholders funds have decreased by the corresponding amount as at December 31, 2013 and September 30, 2013.
Tax (expense) / benefit for the quarter ended December 31, 2013 and comprise current tax including Minimum alternate tax, deferred tax charge or benefit and reversal of excess provision arising on account of completed tax assessments of earlier years aggregating to Rs. 109.000 Millions.
The figures of the earlier periods have been regrouped to be in conformity with clause 41 of the listing agreement.
WEBSITE DETAILS
BOARD OF DIRECTORS
EXECUTIVE
DIRECTORS
DR. S. DEVENDRA WHOLE TIME DIRECTOR
Dr. S. Devendra,
Whole Time Director is a graduate in medicine and is one of the key architects
in developing the organization and was instrumental in transforming the company
from single product company to multi product service provider. He has gained a
worldwide reputation for his knowledge in Marketing, Customer Satisfaction,
Global Competition, and Strategic Thinking. He has been mentoring and
developing the marketing skills of the company.
S. ABHAYA KUMAR MANAGING DIRECTOR
Mr. S. Abhaya
Kumar is Managing Director. He is a chemical engineer and as such has been
instrumental in honing the technical aspects of the Shasun offering. His
leadership has ensured that our facilities and products meet the technical
challenge of the global market place. His drive for technical excellence and
quality manufacturing has taken Shasun to international heights. Well respected
within the national bodies of the Indian Chemical Industry Abhaya has extensive
experience of marketing in the USA and is a member of the Indo American Chamber
of Commerce.
S. VIMAL KUMAR WHOLE TIME DIRECTOR
Mr. S. Vimal
Kumar, Whole Time Director is a graduate in Commerce from Loyola College in
Chennai and a Fellow Member of the Institute of Chartered Accountants of India.
He started his career with Shasun Chemicals and Drugs Limited and became
Director Finance of the Company. As a Chartered Accountant, he heads the
accounting and finance functions of the company. He holds the Directorship for
the Shasun Group of Companies.
A M MOHAN DIRECTOR EHS
Mr. A M Mohan,
Director EHS, a chemical engineer has been one of the key management team
members involved in shaping the organization.
Mr. Mohan, has
headed the Operation and is currently responsible for heading the EHS
(Environment, Health and Safety) New Product Initiatives and Projects.
NON-EXECUTIVE
DIRECTORS
R. KANNAN CHAIRMAN
Mr. R. Kannan, a
Post Graduate in Mathematics from Madras University, PGDMS from Bombay
University and Certified Associate of Indian Institute of Bankers with
Industrial Finance.
R. SUNDARA RAJAN DIRECTOR
Mr. R. Sundara
Rajan, is a Mechanical Engineer and PGDM from IIM Ahmedabad. He is a Chartered
Engineer and Associate of Insurance Institute of India. He has a rich
experience of around 30 years in Pharmaceutical Industry Senior Management
positions.
N. SUBRAMANIAN DIRECTOR
Mr. N. Subramanian
is a Fellow Member of the Institute of the Chartered Accountants of India, New
Delhi and Associate Member of the Institute of Company Secretaries of India,
New Delhi.
DR. SUNNY SHARMA DIRECTOR
Dr. Sunny Sharma
is a Senior Managing Director on the Asia team of OrbiMed Advisors. Dr. Sharma
has over 12 years of life sciences experiences panning venture capital,
investment banking, and clinical practice.
PROF. INDIRA J PARIKH DIRECTOR
Prof. Indira J Parikh is the Founder
President of FLAME (Foundation for Liberal and Management Education). She was a
faculty at IIM Ahmedabad for over 30 years and Dean from 2002 to 2005. She has
taught at INSEAD (European Institute for Business Administration), Fontainebleau
(France) and Texas A & M University. She has specialized in organization
development and design and institution building. She has designed and offered
management and leadership development programs in public sector, private sector
and multinational organizations.
HISTORY
1976-77 - Incorporated in
Chennai, India. First production facility (for Analgin)
1986 - Second production facility at Pondicherry (for
Ibuprofen)
1991 - Third manufacturing facility in Cuddalore (for
Ranitidine HCl)
1992 - Established US subsidiary
1994 - Initial public offering raised approx Rs. 60.000
Millions
2005 - Commissioned new Research Centre and Formulations
facility
2006 - Successfully completed acquisition of Rhodia Pharma
Solutions business.
2009 - Commercial launch of recombinant streptokinase in
India (June 09)
2010 - Finished dosages sales grows 100% and reaches Rs.
500.000 Millions
2011 - Formed 50:50 JV Shasun NBI LLC in USA with
Nanoparticle Biochem
Major expansion of formulations facility
to 5 billion OSD
NEWS
PRESS RELEASE
SHASUN APPOINTS
KEVIN COOK TO ACCELERATE BUSINESS GROWTH
29 August 2013
Shasun has
appointed Kevin Cook as President - Business Growth from 1 August 2013 with a
remit to accelerate company growth and increase sales.
Kevin will be
based at UK manufacturing facility at Dudley, Northumberland and will report
directly to Shasun Managing Director, Abhaya Kumar. He will work closely with
the Shasun’s business development teams to promote the company’s manufacturing
offering. Shasun provide a unique “East-West” offering to the healthcare market
and Kevin’s key areas of focus will involve increasing the manufacturing
customer portfolio, identifying and creating new business lines and expanding
Shasun’s Active Pharmaceutical Ingredient (API) offerings.
Kevin is highly
experienced in both the operational and commercial disciplines within the
pharmaceutical manufacturing industry. Almost 30 years of experience in the
industry culminated in his being named President of Shasun Pharma Solutions
Limited, Dudley in 2008. Kevin held that role for three years before moving to
the pharmaceutical company Aesica in April 2011 as Managing Director, API
Business Unit. Shasun is delighted that he has now decided to rejoin their global
team.
Abhaya Kumar said
“I believe that appointing Kevin to this new and challenging role provides us
with a cutting edge advantage as we look to continue our worldwide growth
strategy. Kevin has a global reputation in the pharmaceutical and healthcare industry
and his knowledge of the healthcare business together with his worldwide
contacts will be vital as we work to increase our market share and sales
portfolio”.
SUCCESSFUL
COMPLETION OF MHRA EU INSPECTION
Chennai, 24 October 2013
Shasun Pharmaceuticals Limited, a global
pharmaceutical organization offering world class capabilities and facilities
has successfully completed MHRA inspection at its manufacturing facility in
Dudley, UK during the third week of October 2013. The main focus of the inspection
was to conduct a cGMP audit for its UK CRAMs Shasun Pharma Solutions Limited
(SPSL) facility.
The inspection concluded that the facility,
systems and practices comply with MHRA requirements.
Commenting on the inspections Mr. John Wiper, President SPSL added: “It gives us great pleasure on the successful completion of the MHRA
inspection. It is a good reflection of our people, systems and procedures and
our commitment to produce quality products at the required standard.”
About Shasun Pharmaceuticals Limited:
Shasun Pharmaceuticals Limited, listed on the
Bombay Stock Exchange Limited (stock code: 524552) and the National Stock
Exchange of India Limited (stock code: SHASUNPHAR) is a leading manufacturer of
API's & formulations, catering to global customers, with world class
capabilities and facilities. The company offers formulation and generic
manufacturing services at its India sites together with a contract research and
manufacturing service for APIs in India and the UK.
CMT REPORT (Corruption, Money Laundering & Terrorism]
The Public Notice information has been collected from various sources
including but not limited to: The Courts,
1] INFORMATION ON
DESIGNATED PARTY
No exist designating subject or any of its beneficial owners,
controlling shareholders or senior officers as terrorist or terrorist
organization or whom notice had been received that all financial transactions
involving their assets have been blocked or convicted, found guilty or against
whom a judgement or order had been entered in a proceedings for violating
money-laundering, anti-corruption or bribery or international economic or
anti-terrorism sanction laws or whose assets were seized, blocked, frozen or
ordered forfeited for violation of money laundering or international
anti-terrorism laws.
2] Court Declaration :
No exist to suggest that subject is or was
the subject of any formal or informal allegations, prosecutions or other
official proceeding for making any prohibited payments or other improper
payments to government officials for engaging in prohibited transactions or
with designated parties.
3] Asset Declaration :
No records exist to suggest that the property or assets of the subject
are derived from criminal conduct or a prohibited transaction.
4] Record on Financial
Crime :
Charges or conviction
registered against subject: None
5] Records on Violation of
Anti-Corruption Laws :
Charges or
investigation registered against subject: None
6] Records on Int’l
Anti-Money Laundering Laws/Standards :
Charges or
investigation registered against subject: None
7] Criminal Records
No
available information exist that suggest that subject or any of its principals
have been formally charged or convicted by a competent governmental authority
for any financial crime or under any formal investigation by a competent
government authority for any violation of anti-corruption laws or international
anti-money laundering laws or standard.
8] Affiliation with
Government :
No record
exists to suggest that any director or indirect owners, controlling
shareholders, director, officer or employee of the company is a government
official or a family member or close business associate of a Government
official.
9] Compensation Package :
Our market
survey revealed that the amount of compensation sought by the subject is fair
and reasonable and comparable to compensation paid to others for similar
services.
10] Press Report :
No press reports / filings exists on
the subject.
CORPORATE GOVERNANCE
MIRA INFORM as part of its Due Diligence do provide comments on
Corporate Governance to identify management and governance. These factors often
have been predictive and in some cases have created vulnerabilities to credit
deterioration.
Our Governance Assessment focuses principally on the interactions
between a company’s management, its Board of Directors, Shareholders and other
financial stakeholders.
CONTRAVENTION
Subject is not known to have contravened any existing local laws,
regulations or policies that prohibit, restrict or otherwise affect the terms
and conditions that could be included in the agreement with the subject.
FOREIGN EXCHANGE RATES
|
Currency |
Unit
|
Indian Rupees |
|
US Dollar |
1 |
Rs. 61.94 |
|
UK Pound |
1 |
Rs. 103.31 |
|
Euro |
1 |
Rs. 85.11 |
INFORMATION DETAILS
|
Information
Gathered by : |
PRT |
|
|
|
|
Report Prepared
by : |
DPH |
SCORE & RATING EXPLANATIONS
|
SCORE FACTORS |
RANGE |
POINTS |
|
HISTORY |
1~10 |
6 |
|
PAID-UP CAPITAL |
1~10 |
5 |
|
OPERATING SCALE |
1~10 |
6 |
|
FINANCIAL CONDITION |
|
|
|
--BUSINESS SCALE |
1~10 |
6 |
|
--PROFITABILIRY |
1~10 |
4 |
|
--LIQUIDITY |
1~10 |
5 |
|
--LEVERAGE |
1~10 |
5 |
|
--RESERVES |
1~10 |
5 |
|
--CREDIT LINES |
1~10 |
5 |
|
--MARGINS |
-5~5 |
-- |
|
DEMERIT POINTS |
|
|
|
--BANK CHARGES |
YES/NO |
YES |
|
--LITIGATION |
YES/NO |
NO |
|
--OTHER ADVERSE INFORMATION |
YES/NO |
NO |
|
MERIT POINTS |
|
|
|
--SOLE DISTRIBUTORSHIP |
YES/NO |
NO |
|
--EXPORT ACTIVITIES |
YES/NO |
YES |
|
--AFFILIATION |
YES/NO |
YES |
|
--LISTED |
YES/NO |
YES |
|
--OTHER MERIT FACTORS |
YES/NO |
YES |
|
DEFAULTER |
|
|
|
--RBI |
YES/NO |
NO |
|
--EPF |
YES/NO |
NO |
|
TOTAL |
|
47 |
This score serves as a reference to assess
SC’s credit risk and to set the amount of credit to be extended. It is
calculated from a composite of weighted scores obtained from each of the major
sections of this report. The assessed factors and their relative weights (as
indicated through %) are as follows:
Financial
condition (40%) Ownership
background (20%) Payment
record (10%)
Credit history
(10%) Market trend (10%) Operational size
(10%)
RATING EXPLANATIONS
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
>86 |
Aaa |
Possesses an extremely sound financial base with the strongest
capability for timely payment of interest and principal sums |
Unlimited |
|
71-85 |
Aa |
Possesses adequate working capital. No caution needed for credit transaction.
It has above average (strong) capability for payment of interest and
principal sums |
Large |
|
56-70 |
A |
Financial & operational base are regarded healthy. General
unfavourable factors will not cause fatal effect. Satisfactory capability for
payment of interest and principal sums |
Fairly Large |
|
41-55 |
Ba |
Overall operation is considered normal. Capable to meet normal
commitments. |
Satisfactory |
|
26-40 |
B |
Capability to overcome financial difficulties seems comparatively below
average. |
Small |
|
11-25 |
Ca |
Adverse factors are apparent. Repayment of interest and principal sums
in default or expected to be in default upon maturity |
Limited with
full security |
|
<10 |
C |
Absolute credit risk exists. Caution needed to be exercised |
Credit not
recommended |
|
- |
NB |
New Business |
- |
This report is issued at your request without any
risk and responsibility on the part of MIRA INFORM PRIVATE LIMITED (MIPL) or
its officials.