MIRA
INFORM REPORT
|
Report Date : |
29.07.2014 |
IDENTIFICATION DETAILS
|
Name : |
MYLAN LABORATORIES LIMITED (w.e.f. 05.10.2011) |
|
|
|
|
Formerly Known
As : |
MATRIX LABORATORIES LIMITTED |
|
|
|
|
Registered
Office : |
Plot No.564/A/22 Road No.92, Jubilee Hills, |
|
|
|
|
Country : |
|
|
|
|
|
Financials (as
on) : |
31.03.2013 |
|
|
|
|
Date of
Incorporation : |
29.11.1984 |
|
|
|
|
Com. Reg. No.: |
01-005146 |
|
|
|
|
Capital Investment
/ Paid-up Capital : |
Rs.312.680 Millions |
|
|
|
|
CIN No.: [Company Identification
No.] |
U24231AP1984PLC005146 |
|
|
|
|
TAN No.: [Tax Deduction &
Collection Account No.] |
HYDM02247A |
|
|
|
|
PAN No.: [Permanent Account No.] |
AADCM3491M |
|
|
|
|
Legal Form : |
A Closely Held Public Limited Liability Company |
|
|
|
|
Line of Business
: |
Manufacturer of Pharmaceutical products. |
|
|
|
|
No. of Employees
: |
Not Divulged |
RATING & COMMENTS
|
MIRA’s Rating : |
A (66) |
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
56-70 |
A |
Financial & operational base are regarded healthy. General
unfavourable factors will not cause fatal effect. Satisfactory capability for
payment of interest and principal sums |
Fairly Large |
|
Maximum Credit Limit : |
USD 99470000 |
|
|
|
|
Status : |
Good |
|
|
|
|
Payment Behaviour : |
Regular |
|
|
|
|
Litigation : |
Clear |
|
|
|
|
Comments : |
Subject is a part of Mylan Inc, USA. It is an established company having
fine track record. The company has seen a better improvement in its sales volume as well
as net profitability during 2013. Financial as well as liquidity position
appears to be strong and healthy. Further, the company successfully maintains strong research and
development capabilities and low cost manufacturing base. The ratings also take into consideration the company’s working capital
intensive operations and increasing competition in the international generics
market. However, trade relations are fair. Business is active. Payment terms
are reported as regular and as per commitments. In view of strong business profile and its established market
position, the subject can be considered good for business dealings at usual
trade terms and conditions. |
NOTES :
Any query related to this report can be made
on e-mail : infodept@mirainform.com
while quoting report number, name and date.
ECGC Country Risk Classification List – June 1, 2014
|
Country Name |
Previous Rating (31.03.2014) |
Current Rating (01.06.2014) |
|
India |
A1 |
A1 |
|
Risk Category |
ECGC
Classification |
|
Insignificant |
A1 |
|
Low |
A2 |
|
Moderate |
B1 |
|
High |
B2 |
|
Very High |
C1 |
|
Restricted |
C2 |
|
Off-credit |
D |
INDIAN ECONOMIC OVERVIEW
N E W S
As per the latest IMF study, the total weigh of emerging markets in the
GDP of the world on a purchasing power parity basis has seen a sizeable shift. It
highlights how as against 51 % in 2005, the emerging economies now account for
close to 56 % of the global purchasing power GDP as per the latest survey. And
with the emerging economies growing at a faster rate than their developed
counterparts, there are every possibility that the their share goes up further
in the coming years. China may surpass the US over the next few years.
Politics and economics are very intricately connected. They tend to
influence each other in ways that could be very complex and far-reaching. The
prospects of the India’s economy have been seriously compromised due to
political corruption. High inflation, poor standard of living are to a great
extent a result of rampant corruption in the country. China on the other hand,
seems to be facing diametrically opposite challenge. American hedge fund
manager Jim Chanos has been keenly following the political and economic
development in the dragon economy and has figured out something that is quite
worrying. He is of the view that the Chinese economy could be heading toward
trouble on account of new Chinese President Xi Jingping’s very aggressive
anti-corruption drive. Chanos believes tat many things such as apartment sales,
luxury products, etc. were largely bought with dirty money. And it is now
beginning to impact consumption. This may indeed be bad news for an economy
that is struggling to transition from an investment-driven export-oriented
economy to a domestic consumption-driven economy.
A study published by Firstpost has revealed that asset classes like real
estate and equities were the biggest beneficiaries of the liberalization
policies. A firm called Ciane Analytics studied returns from assets
including equities, gold, fixed deposits, G-Secs and real estate since 1991.
Real estate outperformed every other asset classes during the 23-year period
with an annualized return of 20 % ! Equities came in second with annualized
return of 15.5 % ! However, while these returns may seem mouthwatering, the
fact is that the return from equities adjusted for inflation came down to just
7.1 %.
Some brief news are as under
. R-Power to buy Jaypee’s hydro assets
. Investors await justice in NSEL case
. India seeks MFN status from Pakistan ahead of meeting
. Ukrain’s clashes with rebels hinder MH17 crash investigation
. India exploring merger of state-owned hydro PSUs
..Higher costs weigh down profit growth to slowest in 9 quarters
..Wal-Mart to expand wholesale business in India
. GMR group moves to strengthen balance sheet
. Central Bank to sell 4 % stake to Life Insurance Corporation
. Tata Chemicals plans to raise up to Rs 10000 mn.
EXTERNAL AGENCY RATING
|
Rating Agency Name |
CRISIL |
|
Rating |
AA- (Long Term Rating) |
|
Rating Explanation |
High degree of safety and low credit risk. |
|
Date |
10.02.2014 |
|
Rating Agency Name |
CRISIL |
|
Rating |
A1+ (Short Term Debt) |
|
Rating Explanation |
Very strong degree of safety and lowest
credit risk. |
|
Date |
10.02.2014 |
RBI DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available RBI Defaulters’ list.
EPF (Employee Provident Fund) DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter in
the publicly available EPF (Employee Provident Fund) Defaulters’ list as of
31-03-2012.
INFORMATION PARTED BY
|
Name : |
Mr. Dattatray Deekshitula |
|
Designation : |
Accounts Manager |
|
Contact No.: |
91-40-630866363 |
|
Date : |
24.07.2014 |
LOCATIONS
|
Registered Office : |
Plot No.564/A/22 Road No.92, Jubilee Hills, Hyderabad – 500033, Andhra
Pradesh, India |
|
Tel. No.: |
91-40-30496666/ 23550543 |
|
Fax No.: |
91-40-27700343/ 30866699 |
|
E-Mail : |
nagaraj.bodige@matrixlabsindia.com apisales@maylan.in (APIs) fdfsales@mylan.in (Formulations) careers@mylan.in (Careers) |
|
Website : |
|
|
|
|
|
Factory: |
Located at ·
Hyderabad (4 Factories) ·
Maharashtra
(2 Factories) ·
Aurangabad (1 Factory) ·
Indore (1 Factory) ·
Andhra Pradesh (2 Factories) |
|
|
|
|
Branch Office 1 : |
7th Floor, Tower 2B, Indiabulls Centre, 841, Senapati Bapat Marg, Elphinstone Road (West), Mumbai – 400021, Maharashtra, India |
|
|
|
|
Branch Office 2 : |
Nirmal, 20th Floor, Nariman Point, Mumbai – 400021, Maharashtra, India |
DIRECTORS
As on: 24.09.2010
|
Name : |
Mr. Rajiv Malik |
|
Designation : |
Director |
|
Address : |
B- 6B, Gangotri Alaaknanda, |
|
Date of Birth/Age : |
12.03.1961 |
|
Qualification : |
Degree in Pharmaceutical Industry |
|
Date of Appointment : |
01.07.2008 |
|
DIN No.: |
00120557 |
|
|
|
|
Name : |
Mr. Susanto Banerjee |
|
Designation : |
Director |
|
Address : |
JI003 Spring Field Apartment, Sarjapura Road ,Belan Dooru, Bangalore –
560102, Karnataka, India |
|
Date of Birth/Age : |
28.11.1966 |
|
PAN No.: |
ACZPB6469M |
|
Date of Appointment : |
28.09.2012 |
|
DIN No.: |
00182743 |
|
|
|
|
Name : |
Dr. B Hari Babun |
|
Designation : |
Whole-Time Director |
|
Address : |
11, Sai Ansh Arcade, Duragavihar Nagar Coly, Trimulghery, Secunderabad
– 500 015, Andhra Pradesh, India |
|
Date of Birth/Age : |
01.07.1964 |
|
Qualification : |
Ph. D. and Master of Science |
|
PAN No.: |
ABYPB0113E |
|
Date of Appointment : |
17.08.2012 |
|
DIN No.: |
01119687 |
|
|
|
|
Name : |
Mr. Sanjeev Sethi |
|
Designation : |
Director |
|
Address : |
Plot No.24, Quitelands, Gatchibowli, Hyderabad – 500 008, Andhra
Pradesh, India |
|
Date of Birth/Age : |
22.07.1967 |
|
Qualification : |
Post Graduate |
|
Date of Appointment : |
24.09.2010 |
|
DIN No.: |
02168682 |
KEY EXECUTIVES
|
Name : |
Mr. B Nagaraj Goud |
|
Designation : |
Secretary |
|
Address : |
H.No.5-14-88, Indiranagar Colony II APHB Colony, Movalai, Hyderabad – 500
040, Andhra Pradesh, India |
|
Date of Birth/Age : |
02.12.1977 |
|
Date of Appointment : |
07.10.2009 |
|
Pan No.: |
AGYPB3840P |
|
|
|
|
Name : |
Mr. Dattatray Deekshitula |
|
Designation : |
Accounts Manager |
MAJOR SHAREHOLDERS / SHAREHOLDING PATTERN
As on: 24.09.2010
SHAREHOLDING DETAILS FILE ATTACHED
As on: 04.12.2013
|
Name of Allottee |
|
No. of Shares |
|
Mylan Luxembourg 2 S.a.r.i, Luxembourg |
|
28418047 |
|
|
|
|
|
Total |
|
28418047 |
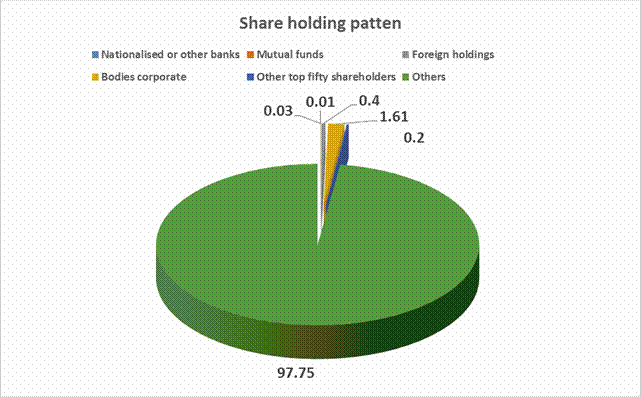
Equity Share Break up (Percentage of Total Equity)
As on: 24.09.2010
|
Category |
|
Percentage |
|
Nationalised or other banks |
|
0.01 |
|
Mutual funds |
|
0.03 |
|
Foreign holdings [Foreign institutional investors, Foreign Companies, Foreign Financial Institutions, Non-resident Indian or Overseas corporate bodies or others] |
|
0.40 |
|
Bodies corporate |
|
1.61 |
|
Other top fifty shareholders |
|
0.20 |
|
Others |
|
97.75 |
|
Total
|
|
100.00 |
BUSINESS DETAILS
|
Line of Business : |
Manufacturer of Pharmaceutical products. |
||||||||||
|
|
|
||||||||||
|
Products : |
|
||||||||||
|
|
|
||||||||||
|
Exports : |
|
||||||||||
|
Products : |
Finished Goods |
||||||||||
|
Countries : |
·
US ·
Europe |
||||||||||
|
|
|
||||||||||
|
Imports : |
|
||||||||||
|
Products : |
Raw Material |
||||||||||
|
Countries : |
China |
||||||||||
|
|
|
||||||||||
|
Terms : |
|
||||||||||
|
Selling : |
L/C and Credit |
||||||||||
|
|
|
||||||||||
|
Purchasing : |
L/C and Credit |
GENERAL INFORMATION
|
Customers : |
Distributor |
||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||
|
No. of Employees : |
Not Divulged |
||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||
|
Bankers : |
· The Bank of Nova Scotia, 6-3-346/1, Road No.1, Banjara Hills, Hyderabad - 500034, Andhra Pradesh, India · DBS Bank Limited, Salarpuria Windsor, No.3, No.10, Ulsoor Road, Ward 78, Bangalore - 560042, Karnataka, India · The Hongkong and Shanghai Banking Corporation Limited, 6-3-1107 and 1108, Raj Bhavan Road, Somajiguda, Hyderabad - 500082, Andhra Pradesh, India · The Royal Bank of Scotland NV, Maker Maxity, 4 North Avenue, Level 3, BKC, Mumbai - 400051, Maharashtra, India · Export - Import Bank of India, Centre One; World Trade Centre, Cuffe Parade; Mumbai - 400005, Andhra Pradesh, India |
||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||
|
Facilities : |
(Rs.
In Millions)
|
|
|
|
|
Banking
Relations : |
-- |
|
|
|
|
Auditors : |
|
|
Name : |
Deloitte Haskins and Sells Chartered Accounts |
|
Address : |
Gowra Grand, III Floor, 1-8-1-384- and 385, SP Road, Secunderabad – 500
003, Andhra Pradesh, India |
|
PAN No. : |
AACFD3771D |
|
|
|
|
Holding Company : |
MP Laboratories (Mauritius) Limited |
|
|
|
|
Ultimate Holding
Company: |
Mylan Inc. |
|
|
|
|
Subsidiary Company
: |
· Matrix Laboratories (Xiamen) Limited · Jiangsu Matrix Pharmaceutical Chemical Limited · Matrix Laboratories Singapore Pte Limited · Matrix Pharma Group (Xiamen) Limited · Mylan Laboratories Inc · Astrix Laboratories Limited CIN No.: U24239AP2005PLC047551 |
|
|
·
|
|
Fellow Subsidiary
Company : |
· Alphapharm Pty Limited · Mylan Pharma UK · Mylan SAS · Mylan SPA · Mylan Seiyaku Limited · Mylan New Zealand Limited · Arcana Arzneimittel GmbH · Mylan (UK) Limited · Mylan Pharmaceutical ULC · Mylan Technologies · Xixia Pharmaceuticals (Pty) Limited Inc · Docpharma BVBA · Gerard Laboratories Limited · Mylan Ireland Limited · Mylan Pharmaceuticals Private Limited CIN No.: U73100MH1997PTC106267 |
CAPITAL STRUCTURE
AFTER: 24.09.2010
Authorised Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
200000000 |
Equity Shares |
Rs.2/- each |
Rs.400.000 Millions |
|
|
|
|
|
Issued, Subscribed & Paid-up Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
184756858 |
Equity Shares |
Rs.2/- each |
Rs.369.514 Millions |
|
|
|
|
|
As on: 31.03.2013
Authorised Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
200000000 |
Equity Shares |
Rs.2/- each |
Rs.400.000 Millions |
|
|
|
|
|
Issued, Subscribed & Paid-up Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
156338811 |
Equity Shares |
Rs.2/- each |
Rs.312.680 Millions |
|
|
|
|
|
FINANCIAL DATA
[all figures are
in Rupees Millions]
ABRIDGED BALANCE
SHEET
|
SOURCES OF FUNDS |
31.03.2013 |
31.03.2012 |
31.03.2011 |
|
EQUITY AND LIABILITIES |
|
|
|
|
(1)Shareholders' Funds |
|
|
|
|
(a) Share Capital |
312.680 |
312.680 |
312.680 |
|
(b) Reserves & Surplus |
24554.940 |
18223.520 |
16020.810 |
|
(c) Money received against share warrants |
0.000 |
0.000 |
0.000 |
|
|
|
|
|
|
(2) Share Application money pending allotment |
0.000 |
0.000 |
0.000 |
|
Total Shareholders’
Funds (1) + (2) |
24867.620 |
18536.200 |
16333.490 |
|
|
|
|
|
|
(3) Non-Current
Liabilities |
|
|
|
|
(a) long-term borrowings |
6916.340 |
5218.080 |
2636.230 |
|
(b) Deferred tax liabilities (Net) |
2018.230 |
1628.280 |
1154.600 |
|
(c) Other long term liabilities |
0.000 |
0.000 |
23.480 |
|
(d) long-term provisions |
184.440 |
126.380 |
109.410 |
|
Total Non-current
Liabilities (3) |
9119.010 |
6972.740 |
3923.720 |
|
|
|
|
|
|
(4) Current
Liabilities |
|
|
|
|
(a) Short term borrowings |
10009.800 |
6828.490 |
5501.990 |
|
(b) Trade payables |
11399.810 |
8550.780 |
6043.480 |
|
(c) Other current liabilities |
4705.640 |
4724.910 |
4155.820 |
|
(d) Short-term provisions |
463.92 |
304.420 |
47.480 |
|
Total Current
Liabilities (4) |
26579.170 |
20408.600 |
15748.770 |
|
|
|
|
|
|
TOTAL |
60565.800 |
45917.540 |
36005.980 |
|
|
|
|
|
|
ASSETS |
|
|
|
|
(1) Non-current
assets |
|
|
|
|
(a) Fixed Assets |
|
|
|
|
(i) Tangible assets |
16954.720 |
13493.140 |
11495.540 |
|
(ii) Intangible Assets |
1044.410 |
54.520 |
38.890 |
|
(iii) Capital work-in-progress |
1314.260 |
1098.010 |
768.900 |
|
(iv) Intangible assets under development |
0.000 |
0.000 |
0.000 |
|
(b) Non-current Investments |
536.410 |
2451.210 |
2566.140 |
|
(c) Deferred tax assets (net) |
0.000 |
0.000 |
0.000 |
|
(d) Long-term Loan and Advances |
1765.830 |
898.200 |
620.660 |
|
(e) Other Non-current assets |
0.000 |
0.000 |
0.000 |
|
Total Non-Current
Assets |
21615.630 |
17995.080 |
15490.130 |
|
|
|
|
|
|
(2) Current assets |
|
|
|
|
(a) Current investments |
0.000 |
0.000 |
51.800 |
|
(b) Inventories |
17572.250 |
12967.350 |
9827.240 |
|
(c) Trade receivables |
12692.880 |
10502.480 |
5629.530 |
|
(d) Cash and cash equivalents |
2042.000 |
39.760 |
146.120 |
|
(e) Short-term loans and advances |
3111.900 |
1705.910 |
2044.750 |
|
(f) Other current assets |
3531.140 |
2706.960 |
2816.410 |
|
Total Current
Assets |
38950.170 |
27922.460 |
20515.850 |
|
|
|
|
|
|
TOTAL |
60565.800 |
45917.540 |
36005.980 |
PROFIT & LOSS
ACCOUNT
|
|
PARTICULARS |
31.03.2013 |
31.03.2012 |
31.03.2011 |
|
|
|
SALES |
|
|
|
|
|
|
|
Income |
53920.770 |
39523.870 |
28775.510 |
|
|
|
Other Income |
2059.810 |
448.900 |
169.750 |
|
|
|
TOTAL (A) |
55980.580 |
39972.770 |
28945.260 |
|
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
|
|
Cost of materials consumed |
29044.530 |
20724.650 |
15694.470 |
|
|
|
Purchases of stock-in-trade |
1321.230 |
848.850 |
359.040 |
|
|
|
Changes in inventories of finished goods, work-in-progress and stock-in-trade |
(1559.540) |
(867.610) |
(1858.650) |
|
|
|
Employee benefit expense |
2918.400 |
2225.330 |
1676.840 |
|
|
|
Other expenses |
12220.950 |
8876.130 |
6716.230 |
|
|
|
Exceptional items |
1914.800 |
130.640 |
(851.390) |
|
|
|
TOTAL (B) |
45860.370 |
31937.990 |
21736.540 |
|
|
|
|
|
|
|
|
Less |
PROFIT
BEFORE INTEREST, TAX, DEPRECIATION AND AMORTISATION (A-B) (C) |
10120.210 |
8034.780 |
7208.720 |
|
|
|
|
|
|
|
|
|
Less |
FINANCIAL
EXPENSES (D) |
630.160 |
575.690 |
432.660 |
|
|
|
|
|
|
|
|
|
|
PROFIT
BEFORE TAX, DEPRECIATION AND AMORTISATION (C-D) (E) |
9490.050 |
7459.090 |
6776.060 |
|
|
|
|
|
|
|
|
|
Less/ Add |
DEPRECIATION/
AMORTISATION (F) |
964.650 |
779.260 |
601.880 |
|
|
|
|
|
|
|
|
|
|
PROFIT BEFORE
TAX (E-F) (G) |
8525.400 |
6679.830 |
6174.180 |
|
|
|
|
|
|
|
|
|
Less |
TAX (H) |
3329.970 |
1987.400 |
1516.980 |
|
|
|
|
|
|
|
|
|
|
PROFIT AFTER TAX
(G-H) (I) |
5195.430 |
4692.430 |
4657.200 |
|
|
|
|
|
|
|
|
|
|
EARNINGS IN
FOREIGN CURRENCY |
|
|
|
|
|
|
|
Exports at F.O.B. value |
44478.270 |
34171.130 |
|
|
|
|
Other revenue |
34.080 |
3.240 |
|
|
|
|
Guarantee commission |
31.720 |
28.130 |
|
|
|
TOTAL EARNINGS |
44544.070 |
34202.500 |
23461.870 |
|
|
|
|
|
|
|
|
|
|
Earnings Per
Share (Rs.) |
33.23 |
30.01 |
29.79 |
|
|
Particulars |
|
|
31.03.2014 |
|
Sales Turnover (Approximately) |
|
|
60000.000 |
Expected Sales (2014-2015) : Rs.80000.000 Millions
The above information has been parted by Mr. Dattatray Deekshitula
(Accounts Manager)
KEY RATIOS
|
PARTICULARS |
|
31.03.2013 |
31.03.2012 |
31.03.2011 |
|
PAT / Total Income |
(%) |
9.28 |
11.74 |
16.09 |
|
|
|
|
|
|
|
Net Profit Margin (PBT/Sales) |
(%) |
15.81 |
16.90 |
21.46 |
|
|
|
|
|
|
|
Return on Total Assets (PBT/Total Assets} |
(%) |
14.52 |
15.77 |
18.90 |
|
|
|
|
|
|
|
Return on Investment (ROI) (PBT/Networth) |
|
0.34 |
0.36 |
0.38 |
|
|
|
|
|
|
|
Debt Equity Ratio (Total Debt/Networth) |
|
0.68 |
0.65 |
0.50 |
|
|
|
|
|
|
|
Current Ratio (Current Asset/Current Liability) |
|
1.47 |
1.37 |
1.30 |
FINANCIAL ANALYSIS
[all figures are
in Rupees Millions]
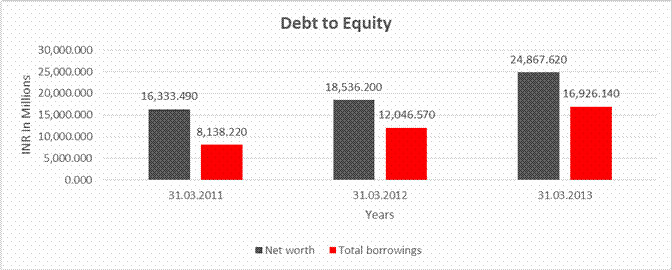
DEBT EQUITY RATIO
|
Particular |
31.03.2011 |
31.03.2012 |
31.03.2013 |
|
|
(Rs.
In Millions) |
(Rs.
In Millions) |
(Rs.
In Millions) |
|
Share Capital |
312.680 |
312.680 |
312.680 |
|
Reserves & Surplus |
16020.810 |
18223.520 |
24554.940 |
|
Net worth |
16,333.490 |
18,536.200 |
24,867.620 |
|
|
|
|
|
|
long-term borrowings |
2636.230 |
5218.080 |
6916.340 |
|
Short term borrowings |
5501.990 |
6828.490 |
10009.800 |
|
Total
borrowings |
8,138.220 |
12,046.570 |
16,926.140 |
|
Debt/Equity
ratio |
0.498 |
0.650 |
0.681 |
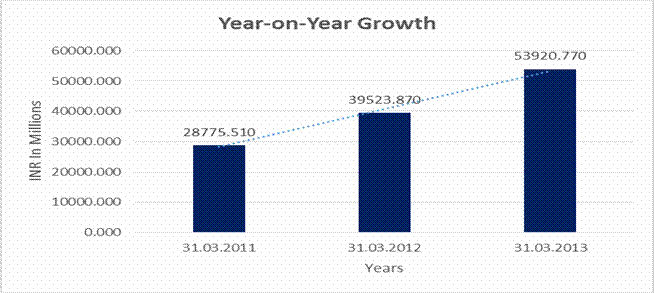
YEAR-ON-YEAR GROWTH
|
Year
on Year Growth |
31.03.2011 |
31.03.2012 |
31.03.2013 |
|
|
(Rs.
In Millions) |
(Rs.
In Millions) |
(Rs.
In Millions) |
|
Sales |
28775.510 |
39523.870 |
53920.770 |
|
|
|
37.352 |
36.426 |
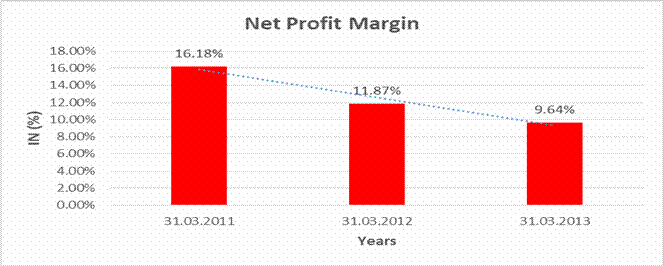
NET PROFIT MARGIN
|
Net
Profit Margin |
31.03.2011 |
31.03.2012 |
31.03.2013 |
|
|
(Rs.
In Millions) |
(Rs.
In Millions) |
(Rs.
In Millions) |
|
Sales |
28775.510 |
39523.870 |
53920.770 |
|
Profit |
4657.200 |
4692.430 |
5195.430 |
|
|
16.18% |
11.87% |
9.64% |
LOCAL AGENCY FURTHER INFORMATION
|
Sr. No. |
Check List by Info
Agents |
Available in Report
(Yes / No) |
|
1] |
Year of Establishment |
Yes |
|
2] |
Locality of the firm |
Yes |
|
3] |
Constitutions of the firm |
Yes |
|
4] |
Premises details |
No |
|
5] |
Type of Business |
Yes |
|
6] |
Line of Business |
Yes |
|
7] |
Promoter's background |
Yes |
|
8] |
No. of employees |
Yes |
|
9] |
Name of person contacted |
Yes |
|
10] |
Designation of contact person |
Yes |
|
11] |
Turnover of firm for last four years |
Yes |
|
12] |
Profitability for last three years |
Yes |
|
13] |
Reasons for variation <> 20% |
-- |
|
14] |
Estimation for coming financial year |
Yes |
|
15] |
Capital in the business |
Yes |
|
16] |
Details of sister concerns |
Yes |
|
17] |
Major suppliers |
No |
|
18] |
Major customers |
No |
|
19] |
Payments terms |
Yes |
|
20] |
Export / Import details (if applicable) |
Yes |
|
21] |
Market information |
-- |
|
22] |
Litigations that the firm / promoter involved in |
-- |
|
23] |
Banking Details |
Yes |
|
24] |
Banking facility details |
Yes |
|
25] |
Conduct of the banking account |
-- |
|
26] |
Buyer visit details |
-- |
|
27] |
Financials, if provided |
Yes |
|
28] |
Incorporation details, if applicable |
Yes |
|
29] |
Last accounts filed at ROC |
Yes |
|
30] |
Major Shareholders, if available |
Yes |
|
31] |
Date of Birth of Proprietor/Partner/Director, if available |
Yes |
|
32] |
PAN of Proprietor/Partner/Director, if available |
Yes |
|
33] |
Voter ID No of Proprietor/Partner/Director, if available |
No |
|
34] |
External Agency Rating, if available |
Yes |
NOTE:
The Registered Office of the company has been shifted from 1-1-151/1,
Sairam Towers, 4th Floor, Alexander Road, Secunderabad – 500003,
Andhra Pradesh, India to the present address w.e.f. 15.03.2011
REVIEW OF OPERATIONS
The Company posted yet another impressive year of performance. During the year, the turnover, on a standalone basis, increased by 37%, while on a consolidated basis, the sales increased by 36% over the previous year. The increase in sales was mainly due to the increase in the sales of Active Pharmaceutical Ingredients (APIs) and Finished Dosage Form (FDF) products. The net profit before tax and exceptional items also showed an impressive growth of 53% on a standalone basis, while on a consolidated basis, the increase was 51% over the previous year.
During the year, the Management has assessed the operations of its subsidiary group of companies in China, Matrix Pharma Group (Xiamen) Limited and its subsidiaries (held by global holding company Matrix Laboratories (Singapore) Pte Limited to identify indication of diminution, other than temporary, in the value of investment recorded in the books of account and, accordingly, has made a gross provision of Rs. 1914.800 Millions. The investment in the China Group of Companies was made by the Company in 2006 as part of a backward integration strategy of the Company to secure supply of Active Pharmaceuticals (APIs) and API intermediates. The China operations consist of an API and FDF manufacturing facility and an APIIntermediates manufacturing facility. The API intermediates manufacturing operations were shut down in December 2011. The API manufacturing operations had been shut down since January 2013. Given the substantial scale down in operations, as the API manufacturing is closed down and FDF manufacturing activity is currently very small, the cash flows from future operations are insufficient and accordingly the entire carrying value of investment has been written down.
During the year, the Company filed 7 US Drug Master Files (DMFs) and 18 EU DMFs / Certificates of Suitability to European Pharmacopoeia (CEPs). With these filings, as on March 31, 2013, the cumulative number of DMFs filed by the Company, together with its subsidiaries and associates is 174 U.S. DMFs and 180 EU DMFs / CEPs.
During the year, the Company has filed 20 Abbreivated New Drug Applications(ANDAs) with the U.S. Food and Drug Administration(FDA), 18 with European regulatory agencies, 4 with the World Health Organisation(WHO), 16 with Canada regulatory agencies, 11 with Australian regulatory agencies, 10 with New Zealand regulatory agencies and 18 with South Africa regulatory agencies. Aggregate filings covering Finished Dosage Forms during the year were 97 in numbers.
Cumulatively, the Company made the filings of 151 ANDAs with the FDA, 99 regulatory filings with European regulatory agencies, 36 filings with the WHO, 58 with Canadian regulatory agencies, 50 with Australian regulatory agencies, 31 with New Zealand, and 74 with RSA aggregating to 499 regulatory submissions. During the year, the Company secured approvals for 16 ANDAs from the FDA, 21 from Canadian regulatory agencies, 16 from the European regulatory agencies, 6 from Australian regulatory agencies (TGA), 5 from New Zealand regulatory agencies and 15 from South Africa regulatory agencies.
During the year 2012-13, the Company was selected as a leading supplier of antiretroviral (ARV) drugs toIndia’s National AIDS Control Organization (NACO).
During the year 2012-13, the Company entered into the South African market by participating in the South African ARV tender and was selected as one of the leading suppliers of ARV drugs to the South African National Department of Health for the tender period January 1, 2013 to December 31, 2014.
UNSECURED LOAN
(Rs.
In Millions)
|
Particular |
As on 31.03.2013 |
As on 31.03.2012 |
|
LONG-TERM BORROWINGS |
|
|
|
Deferred sales tax loan |
20.010 |
22.000 |
|
Loans and advances from others |
6896.330 |
5102.330 |
|
SHORT-TERM
BORROWINGS |
|
|
|
Working capital loans from banks |
5545.100 |
3087.120 |
|
|
|
|
|
Total |
12461.440 |
8211.450 |
INDEX OF CHARGES
|
S.No. |
Charge ID |
Date of Charge
Creation/Modification |
Charge amount
secured |
Charge Holder |
Address |
Service Request
Number (SRN) |
|
1 |
10296577 |
12/08/2013 * |
480,000,000.00 |
DBS BANK LIMITED |
SALARPURIA WINDSOR, NO.3, NO.10, ULSOOR ROAD, WARD 78, BANGALORE, KARNATAKA - 560042, INDIA |
B84460450 |
|
2 |
10268093 |
27/01/2011 |
480,000,000.00 |
DBS BANK LIMITED |
SALARPURIA WINDSOR, NO.3, (OLD NO.10), ULSOOR ROAD, WARD NO. 78, BANGALORE, KARNATAKA - 560042, INDIA |
B06623540 |
|
3 |
10198643 |
12/08/2013 * |
8,104,000,000.00 |
THE HONGKONG AND SHANGHAI BANKING CORPORATION LIMITED |
6-3-1107 AND 1108, RAJ BHAVAN ROAD, SOMAJIGUDA, HYDERABAD, ANDHRA PRADESH - 500082, INDIA |
B84182138 |
|
4 |
10198487 |
29/07/2010 * |
470,000,000.00 |
THE ROYAL BANK OF SCOTLAND N.V. |
74, SAKHAR BHAVAN, 7TH FLOOR, NARIMAN POINT, MUMBAI, MAHARASHTRA - 400021, INDIA |
A91987479 |
|
5 |
10179307 |
21/07/2009 |
300,000,000.00 |
INDUSIND BANK LIMITED |
2401 GEN THIMMAYYA ROAD, CONTONMENT, PUNE, MAHARASHTRA - 411001, INDIA |
A69552057 |
|
6 |
10179308 |
21/07/2009 |
700,000,000.00 |
INDUSIND BANK LIMITED |
2401 GEN THIMMAYYA ROAD, CONTONMENT, PUNE, MAHARASHTRA - 411001, INDIA |
A69552685 |
|
7 |
90128817 |
06/07/2007 * |
186,000,000.00 |
ANDHRA BANK |
R P. ROAD, SECUNDERABAD, SECUNDERABAD, ANDHRA PRADESH - 500003, INDIA |
A19287150 |
|
8 |
90130616 |
08/06/2007 * |
434,000,000.00 |
HDFC BANK LIMITED |
HDFC BANK HOUSESENAPATI BAPAT MARG, LOWER PAREL W, MUMBAI, MAHARASHTRA - 400013, INDIA |
A19336171 |
|
9 |
90130414 |
12/08/2013 * |
700,000,000.00 |
THE ROYAL BANK OF SCOTLAND NV |
MAKER MAXITY, 4 NORTH AVENUE, LEVEL 3, BKC, MUMBAI, MAHARASHTRA - 400051, INDIA |
B84032853 |
|
10 |
90262408 |
12/08/2013 * |
8,104,000,000.00 |
STATE BANK OF INDIA |
CAG BRANCH, 2ND FLOOR, OZONE BUILDING, 6-3-669, PUNJAGUTTA MAIN ROAD, HYDERABAD, ANDHRA PRADESH - 500082, INDIA |
B84033638 |
|
11 |
90130385 |
12/08/2013 * |
1,000,000,000.00 |
THE BANK OF NOVA SCOTIA |
6-3-346/1, ROAD NO.1, BANJARA HILLS, HYDERABAD, ANDHRA PRADESH - 500034, INDIA |
B85301729 |
|
12 |
90130372 |
01/12/2003 |
262,500,000.00 |
HDFC BANK LTD |
LAKDIKAPUL BRANCH, SADEED PLAZA, HYDERABAD, ANDHRA PRADESH, INDIA |
- |
|
13 |
90262350 |
19/09/2003 |
120,000,000.00 |
IDBI BANK LTD. |
MAHAVIR HOUSE, BASEERBAGH, HYDERABAD, ANDHRA PRADESH, INDIA |
- |
|
14 |
90262253 |
24/02/2003 |
5,000,000.00 |
COOPERATIVE
CENTRALE RAIFFEISEN - BOERENFEENBANK B |
SINGAPORE BRANCH; 77; ROBINSON ROAD - 08 - 00, SIA BUILDING, SINGAPORE, ANDHRA PRADESH - 068896, INDIA |
- |
|
15 |
90262220 |
14/11/2002 |
75,000,000.00 |
PUNJAB NATIONAL BANK |
RP ROAD, SECUNDRABAD, ANDHRA PRADESH, INDIA |
- |
|
16 |
90260738 |
25/11/1988 |
263,000.00 |
ANDHRA PRADESH STATE FINANCIAL CORPAORATION |
CHIRAG ALI LANE, ABIDS, HYDERABAD, ANDHRA PRADESH, INDIA |
- |
|
17 |
90260687 |
11/02/1988 |
3,635,000.00 |
AP STATE FINANCIAL CORPORATION |
CHIRAG ALI LANE, HYDERABAD, ANDHRA PRADESH, INDIA |
- |
* Date of charge modification
FIXED ASSETS
Tangible Assets
· Land
· Buildings
· Plant and Equipment
· Furniture and Fixtures
· Vehicles
· Office Equipment
· Electrical Equipment
· Lab Equipment
· Computer Hardware
Intangible Assets
· Computer Software
· Goodwill
PRESS RELEASES
MYLAN TO ACQUIRE ABBOTT'S NON-U.S. DEVELOPED MARKETS SPECIALTY AND
BRANDED GENERICS BUSINESS IN AN ALL-STOCK TRANSACTION
JULY 14, 2014
PITTSBURGH, July 14, 2014 /PRNewswire/ -- Mylan Inc. (NASDAQ: MYL) today announced that it has entered into a definitive agreement with Abbott (NYSE: ABT) whereby Mylan will acquire Abbott's non-U.S. developed markets specialty and branded generics business ("the Assets") in an all-stock transaction. Upon closing, Abbott will receive 105 million shares of the combined company worth approximately $5.3 billion based on Mylan's closing price of $50.20 on Friday, July 11, 2014, representing an approximately 21% ownership stake. The transaction will instantly further diversify Mylan's business and strengthen its commercial platform outside the U.S., building new opportunities for growth and additional sales channels in the acquired markets. It also is expected to provide Mylan with significant additional financial firepower to pursue future opportunities, an additional $600 million of annual post-close EBITDA, an optimized global tax structure and enhanced balance sheet capacity.
The Assets, which are being acquired on a debt-free basis, include an attractive portfolio of more than 100 specialty and branded generic pharmaceutical products in five major therapeutic areas (cardio/metabolic, gastrointestinal, anti-infective/respiratory, CNS/pain and women's and men's health) and include several patent protected, novel and/or hard-to-manufacture products with continued growth potential. With a strong presence in Europe, Japan, Canada, Australia and New Zealand, the Assets are expected to provide approximately $1.9 billion in annual additional revenues at deal close. The business includes an active sales organization of approximately 2,000 representatives in more than 40 non-U.S. markets, as well as two high-quality manufacturing facilities.
Following the transaction, Mylan expects to have approximately $10 billioni in pro forma 2014 sales, adjusted EBITDA of approximately $3 billion at transaction close, an attractive and diverse portfolio of more than 1,400 specialty and generic products, an enhanced global commercial infrastructure, and an expanded high-quality manufacturing platform.
Mylan Executive Chairman Robert J. Coury commented, "We have been actively looking at a wide range of opportunities, and the acquisition of this business is absolutely the right next strategic transaction for Mylan as it builds on our strong momentum, expands and further diversifies our business in our largest markets outside of the U.S., and clearly positions Mylan for the next phase of growth through enhanced financial flexibility and a more competitive global tax structure. In addition to maximizing our growth drivers, the transaction is expected to be immediately and significantly accretive, and to create significant additional cash financial flexibility at close, which we fully intend to put to use to fund future opportunities in this continually consolidating sector. The numerous strategic and financial benefits of this transaction will allow Mylan to potentially accelerate achievement of our long-term financial targets to the benefit of our shareholders."
Mylan CEO Heather Bresch said, "We targeted this differentiated business with a complementary portfolio of attractive specialty and branded generic products, many of which have strong continued growth potential. The Assets also have an impressive commercial infrastructure and capabilities, which provide us with reach in the physician and patient channels in the acquired markets, complementing our reach in pharmacies. This enhanced commercial platform will help us drive the continued expansion of EpiPen® Auto-Injector globally and enable us to more effectively launch important growth drivers, such as respiratory and biologics. We believe Mylan is uniquely positioned to realize improved financial performance and profitability from these assets by leveraging our integrated, efficient operating platform, more effectively distributing the portfolio across channels, and maintaining a greater strategic focus on key products. We have experience successfully integrating large, complex transactions such as this one, and we are confident in our ability to deliver the value inherent from this combination."
Bresch continued, "In addition to creating value for our shareholders, this transaction delivers on our mission of providing the world's 7 billion people access to high-quality medicine. The Abbott team associated with this business shares Mylan's uncompromising commitment to quality, our high-performance culture and our passion for making a difference. We look forward to welcoming them and their strong sales organization to Mylan and benefiting from their capabilities."
"Mylan is the right organization for our developed markets branded generics business," said Miles D. White, Chairman and Chief Executive Officer of Abbott. "Mylan has the scale and breadth across critical distribution channels and a complementary portfolio that will quickly position this business for success. Mylan also shares our commitment to patients and product quality."
Strategic Rationale
This transaction further diversifies Mylan's business outside of the U.S. by adding a differentiated and attractive portfolio of durable specialty and branded generic products and providing entry into the over-the-counter market. Key products include Creon®, Influvac®, Brufen®, Amitiza® and Androgel®, among others.
The addition of the Assets also enhances Mylan's geographic reach and provides Mylan with enhanced scale and critical mass in Mylan's largest markets outside of the U.S. The transaction is expected to approximately double Mylan's revenues in Europe by strengthening its presence in Italy, the United Kingdom, Germany, France, Spain and Portugal, among others. It also is expected to more than double Mylan's revenues in Canada and Japan, and build on Mylan's business in Australia and New Zealand. The transaction also provides Mylan with a meaningful presence in the specialty and branded generics market in Central and Eastern Europe.
The combination significantly expands Mylan's commercial platform and capabilities. The business's strong salesforce in key developed markets enhances Mylan's reach with physicians and patients and complements Mylan's existing strength in pharmacies. This platform provides Mylan with the enhanced infrastructure and expertise to more effectively execute on growth drivers that require access to the physician channel, such as the global expansion of EpiPen® Auto-Injector® and the launch of biologics and respiratory products, including generic Seretide® and generic Advair®.
Mylan expects to maximize the business's strong portfolio and attractive financial profile to drive enhanced financial performance from the business, including stabilizing revenues and growing EBITDA and EBITDA margins.
Financial Highlights
The transaction is expected to be immediately and significantly accretive to Mylan, with expected year one adjusted diluted EPS accretion of approximately $0.25, increasing thereafter through 2018. The combination is expected to deliver in excess of $200 million in pre-tax operational efficiencies by the end of year three post-close.
Mylan's pro forma leverage at close is expected to be approximately 2.3x debt-to-adjusted EBITDA, substantially below current levels, giving the company ample financial flexibility to pursue future opportunities. Strong cash flow generation will further enhance Mylan's balance sheet and financial flexibility and create additional shareholder value. Finally, the transaction is expected to lower Mylan's tax rate to approximately 20-21% in the first full year, and to the high teens thereafter, enhancing the company's competitiveness.
Mylan believes this transaction gives it the potential to accelerate its previously stated financial targets for 2018, including at least $6.00 in adjusted diluted EPS.ii
Transaction Structure
Abbott will carve out the Assets and transfer them to a new public company ("New Mylan") organized in the Netherlands. Immediately following the transfer, Mylan will merge with a wholly owned subsidiary of New Mylan, and New Mylan will become the parent company of Mylan. The new public company will be called Mylan N.V. and will be led by the current Mylan leadership team and headquartered in Pittsburgh.
Under the terms of the transaction agreement, Abbott will receive 105 million shares of New Mylan upon closing, resulting in Mylan shareholders owning approximately 79% of New Mylan and Abbott indirectly owning approximately 21% of New Mylan. Mylan shareholders will recognize gain for U.S. federal income tax purposes on the exchange of Mylan common shares for New Mylan ordinary shares.
Shares of New Mylan will continue to trade in the U.S. on the NASDAQ under Mylan's existing ticker symbol MYL.
The transaction has been unanimously approved by Mylan's Board of Directors and is expected to close in the first quarter of 2015, subject to certain closing conditions, including regulatory clearances and approval by Mylan's shareholders.
Centerview Partners served as financial advisor to Mylan, and Cravath, Swaine and Moore LLP served as its legal advisor.
Investor Call Information
Mylan's management team will hold an investor conference call and webcast this morning at 8:00 a.m. EDT to discuss the transaction. To participate in the conference call, please use the following dial-in:
|
Conference ID : |
74158970 |
|
Participant Toll-Free Dial-In Number : |
(800) 514-4861 |
|
Participant International Dial-In Number : |
(678) 809-2405 |
|
|
|
To access the live webcast, including the slide presentation, please go to the Investor Relations page of Mylan's at least 15 minutes before the event is scheduled to begin to register and download or install any necessary software.
Forward-Looking Statements
This press release contains "forward-looking statements." These statements are made pursuant to the safe harbor provisions of the Private Securities Litigation Reform Act of 1995 and may often be identified by the use of words such as "will", "may", "could", "should," "would", "project", "believe", "anticipate", "expect", "plan," "estimate", "forecast", "potential", "intend", "continue", "target" and variations of these words or comparable words. Such forward-looking statements include, without limitation, statements regarding the proposed acquisition of the Assets by Mylan, the expected timetable for completing the transaction, benefits and synergies of the transaction, future opportunities for the combined company and products and any other statements regarding Mylan's and the acquired business's future operations, anticipated business levels, future earnings, planned activities, anticipated growth, market opportunities, strategies, competition, and other expectations and targets for future periods. Because forward-looking statements inherently involve risks and uncertainties, actual future results may differ materially from those expressed or implied by such forward-looking statements. Factors that could cause or contribute to such differences include, but are not limited to: the parties' ability to meet expectations regarding the timing, completion and accounting and tax treatments of the transaction; changes in relevant tax and other laws; the parties' ability to consummate the transaction; the conditions to the completion of the transaction, including the receipt of approval of Mylan's shareholders; the regulatory approvals required for the transaction not being obtained on the terms expected or on the anticipated schedule; inherent uncertainties involved in the estimates and judgments used in the preparation of financial statements, and the providing of estimates of financial measures, in accordance with GAAP and related standards or on an adjusted basis; the integration of the acquired business by Mylan being more difficult, time-consuming or costly than expected; operating costs, customer loss and business disruption (including, without limitation, difficulties in maintaining relationships with employees, customers, clients or suppliers) being greater than expected following the transaction; the retention of certain key employees of the acquired business being difficult; Mylan's and the acquired business's expected or targeted future financial and operating performance and results; the combined company's capacity to bring new products to market, including but not limited to where it uses its business judgment and decides to manufacture, market, and/or sell products, directly or through third parties, notwithstanding the fact that allegations of patent infringement(s) have not been finally resolved by the courts (i.e., an "at-risk launch"); the scope, timing and outcome of any ongoing legal proceedings and the impact of any such proceedings on Mylan's and the acquired business's consolidated financial condition, results of operations or cash flows; Mylan's and the acquired business's ability to protect their intellectual property and preserve their intellectual property rights; the effect of any changes in customer and supplier relationships and customer purchasing patterns; the ability to attract and retain key personnel; changes in third-party relationships; the impacts of competition; changes in economic and financial conditions of Mylan's business or the acquired business; uncertainties and matters beyond the control of management; and the possibility that Mylan may be unable to achieve expected synergies and operating efficiencies in connection with the transaction within the expected time-frames or at all and to successfully integrate the acquired business. For more detailed information on the risks and uncertainties associated with Mylan's business activities, see the risks described in Mylan's Annual Report on Form 10-K for the year ended December 31, 2013 filed with the Securities and Exchange Commission ("SEC"). You can access Mylan's Form 10-K through the SECand Mylan strongly encourages you to do so. Mylan undertakes no obligation to update any statements herein for revisions or changes after the date of this press release.
Non-GAAP Financial Measures
Non-GAAP financial measures should be considered only as a supplement to, and not as a substitute for or as a superior measure to, financial measures prepared in accordance with GAAP.
Additional Information and Where to Find It
In connection with the proposed acquisition of the Assets by Mylan, New Mylan and Mylan intend to file relevant materials with the SEC, including a New Mylan registration statement on Form S-4 that will include a proxy statement of Mylan that also constitutes a prospectus of New Mylan. Investors And Security Holders Are Urged To Read The Proxy Statement/Prospectus and any Other Relevant Documents When They Become Available Because They Will Contain Important Information About Mylan, New Mylan, The Assets And The Proposed Transaction. A definitive proxy statement will be sent to shareholders of Mylan seeking approval of the proposed transaction. The proxy statement/prospectus and other documents relating to the proposed transaction (when they are available) can be obtained free of charge from the SEC's. These documents (when they are available) can also be obtained free of charge from Mylan upon written request to Mylan at 724.514.1813.
Participants in Solicitation
This press release is not a solicitation of a proxy from any investor or shareholder. However, Mylan, New Mylan and certain of their directors and executive officers may be deemed to be participants in the solicitation of proxies in connection with the proposed transaction under the rules of the SEC. Information regarding Mylan's directors and executive officers may be found in its definitive proxy statement relating to its 2014 Annual Meeting of Shareholders filed with the SEC on March 10, 2014. This document can be obtained free of charge from the sources indicated above. Additional information regarding the interests of these participants will also be included in the proxy statement/prospectus when it becomes available.
Non-Solicitation
This communication shall not constitute an offer to sell or the solicitation of an offer to sell or the solicitation of an offer to buy any securities, nor shall there be any sale of securities in any jurisdiction in which such offer, solicitation or sale would be unlawful prior to registration or qualification under the securities laws of any such jurisdiction. No offer of securities shall be made except by means of a prospectus meeting the requirements of Section 10 of the Securities Act of 1933, as amended.
Mylan is a global pharmaceutical company committed to setting new standards in health care. Working together around the world to provide 7 billion people access to high quality medicine, we innovate to satisfy unmet needs; make reliability and service excellence a habit; do what's right, not what's easy; and impact the future through passionate global leadership. We offer a growing portfolio of more than 1,300 generic pharmaceuticals and several brand medications. In addition, we offer a wide range of antiretroviral therapies, upon which approximately 40% of HIV/AIDS patients in developing countries depend. We also operate one of the largest active pharmaceutical ingredient manufacturers and currently market products in approximately 140 countries and territories. Our workforce of more than 20,000 people is dedicated to improving the customer experience and increasing pharmaceutical access to consumers around the world.
1 Stated 2018 target; targets beyond 2014 do not reflect Company guidance
i Based on the midpoint of Mylan's guidance range for 2014
ii Targets beyond 2014 do not reflect Company guidance
CMT REPORT (Corruption, Money Laundering & Terrorism]
The Public Notice information has been collected from various sources
including but not limited to: The Courts,
1] INFORMATION ON
DESIGNATED PARTY
No exist designating subject or any of its beneficial owners,
controlling shareholders or senior officers as terrorist or terrorist
organization or whom notice had been received that all financial transactions
involving their assets have been blocked or convicted, found guilty or against
whom a judgement or order had been entered in a proceedings for violating
money-laundering, anti-corruption or bribery or international economic or
anti-terrorism sanction laws or whose assets were seized, blocked, frozen or
ordered forfeited for violation of money laundering or international
anti-terrorism laws.
2] Court Declaration :
No exist to suggest that subject is or was
the subject of any formal or informal allegations, prosecutions or other
official proceeding for making any prohibited payments or other improper
payments to government officials for engaging in prohibited transactions or
with designated parties.
3] Asset Declaration :
No records exist to suggest that the property or assets of the subject
are derived from criminal conduct or a prohibited transaction.
4] Record on Financial
Crime :
Charges or conviction
registered against subject: None
5] Records on Violation of
Anti-Corruption Laws :
Charges or
investigation registered against subject: None
6] Records on Int’l
Anti-Money Laundering Laws/Standards :
Charges or
investigation registered against subject: None
7] Criminal Records
No
available information exist that suggest that subject or any of its principals
have been formally charged or convicted by a competent governmental authority
for any financial crime or under any formal investigation by a competent
government authority for any violation of anti-corruption laws or international
anti-money laundering laws or standard.
8] Affiliation with
Government :
No record
exists to suggest that any director or indirect owners, controlling
shareholders, director, officer or employee of the company is a government official
or a family member or close business associate of a Government official.
9] Compensation Package :
Our market
survey revealed that the amount of compensation sought by the subject is fair
and reasonable and comparable to compensation paid to others for similar
services.
10] Press Report :
No press reports / filings exists on
the subject.
CORPORATE GOVERNANCE
MIRA INFORM as part of its Due Diligence do provide comments on Corporate
Governance to identify management and governance. These factors often have been
predictive and in some cases have created vulnerabilities to credit
deterioration.
Our Governance Assessment focuses principally on the interactions
between a company’s management, its Board of Directors, Shareholders and other
financial stakeholders.
CONTRAVENTION
Subject is not known to have contravened any existing local laws,
regulations or policies that prohibit, restrict or otherwise affect the terms
and conditions that could be included in the agreement with the subject.
FOREIGN EXCHANGE RATES
|
Currency |
Unit
|
Indian Rupees |
|
US Dollar |
1 |
Rs.60.10 |
|
|
1 |
Rs.102.04 |
|
Euro |
1 |
Rs.80.74 |
INFORMATION DETAILS
|
Information Gathered
by : |
HTL |
|
|
|
|
Analysis Done by
: |
SUB |
|
|
|
|
Report Prepared
by : |
KVT |
SCORE & RATING EXPLANATIONS
|
SCORE FACTORS |
RANGE |
POINTS |
|
HISTORY |
1~10 |
8 |
|
PAID-UP CAPITAL |
1~10 |
7 |
|
OPERATING SCALE |
1~10 |
7 |
|
FINANCIAL CONDITION |
|
|
|
--BUSINESS SCALE |
1~10 |
8 |
|
--PROFITABILIRY |
1~10 |
7 |
|
--LIQUIDITY |
1~10 |
7 |
|
--LEVERAGE |
1~10 |
7 |
|
--RESERVES |
1~10 |
8 |
|
--CREDIT LINES |
1~10 |
7 |
|
--MARGINS |
-5~5 |
- |
|
DEMERIT POINTS |
|
|
|
--BANK CHARGES |
YES/NO |
YES |
|
--LITIGATION |
YES/NO |
NO |
|
--OTHER ADVERSE INFORMATION |
YES/NO |
NO |
|
MERIT POINTS |
|
|
|
--SOLE DISTRIBUTORSHIP |
YES/NO |
NO |
|
--EXPORT ACTIVITIES |
YES/NO |
NO |
|
--AFFILIATION |
YES/NO |
YES |
|
--LISTED |
YES/NO |
NO |
|
--OTHER MERIT FACTORS |
YES/NO |
YES |
|
DEFAULTER |
|
|
|
--RBI |
YES/NO |
NO |
|
--EPF |
YES/NO |
NO |
|
|
|
|
|
TOTAL |
|
66 |
This score serves as a reference to assess SC’s credit risk and
to set the amount of credit to be extended. It is calculated from a composite
of weighted scores obtained from each of the major sections of this report. The
assessed factors and their relative weights (as indicated through %) are as
follows:
Financial
condition (40%) Ownership
background (20%) Payment
record (10%)
Credit history
(10%) Market trend
(10%) Operational
size (10%)
RATING EXPLANATIONS
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
>86 |
Aaa |
Possesses an extremely sound
financial base with the strongest capability for timely payment of interest
and principal sums |
Unlimited |
|
71-85 |
Aa |
Possesses adequate
working capital. No caution needed for credit transaction. It has above average
(strong) capability for payment of interest and principal sums |
Large |
|
56-70 |
A |
Financial &
operational base are regarded healthy. General unfavourable factors will not
cause fatal effect. Satisfactory capability for payment of interest and principal
sums |
Fairly
Large |
|
41-55 |
Ba |
Overall operation is
considered normal. Capable to meet normal commitments. |
Satisfactory |
|
26-40 |
B |
Capability to overcome
financial difficulties seems comparatively below average. |
Small |
|
11-25 |
Ca |
Adverse factors are
apparent. Repayment of interest and principal sums in default or expected to
be in default upon maturity |
Limited
with full security |
|
<10 |
C |
Absolute credit risk
exists. Caution needed to be exercised |
Credit
not recommended |
|
-- |
NB |
New
Business |
-- |
This report is issued at your request without any
risk and responsibility on the part of MIRA INFORM PRIVATE LIMITED (MIPL)
or its officials.