MIRA INFORM REPORT
|
Report Date : |
02.06.2014 |
IDENTIFICATION DETAILS
|
Name : |
BIOCON LIMITED |
|
|
|
|
Registered
Office : |
20th KM, |
|
|
|
|
Country : |
|
|
|
|
|
Financials (as
on) : |
31.03.2013 |
|
|
|
|
Date of
Incorporation : |
29.11.1978 |
|
|
|
|
Com. Reg. No.: |
08-003417 |
|
|
|
|
Capital
Investment / Paid-up Capital : |
Rs.1000.000 Millions |
|
|
|
|
CIN No.: [Company Identification No.] |
L24234KA1978PLC003417 |
|
|
|
|
TAN No.: [Tax Deduction &
Collection Account No.] |
BLRB00214E |
|
|
|
|
PAN No.: [Permanent Account No.] |
AAACB7461R |
|
|
|
|
Legal Form : |
A Public Limited Liability Company. The Company’s Shares are Listed on
the Stock Exchanges. |
|
|
|
|
Line of Business
: |
Manufacturer of biotechnology products and also
engaged in research and development in the biotechnology sector. |
|
|
|
|
No. of Employees
: |
6700 (Approximately) |
RATING & COMMENTS
|
MIRA’s Rating : |
A (68) |
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
56-70 |
A |
Financial & operational base are regarded healthy. General
unfavourable factors will not cause fatal effect. Satisfactory capability for
payment of interest and principal sums |
Fairly Large |
|
Maximum Credit Limit : |
USD 88000000 |
|
|
|
|
Status : |
Good |
|
|
|
|
Payment Behaviour : |
Regular |
|
|
|
|
Litigation : |
Exist |
|
|
|
|
Comments : |
Subject is a well-established company having fine track record. The rating reflects healthy financial risk profile marked by adequate
liquidity position and fair profitability of the company. Trade relations are reported as fair. Business is active. Payments are
reported to be regular and as per commitments. The company can be considered good for normal business dealings at
usual trade terms and conditions. |
NOTES :
Any query related to this report can be made
on e-mail : infodept@mirainform.com
while quoting report number, name and date.
ECGC Country Risk Classification List – March 31, 2014
|
Country Name |
Previous Rating (31.12.2013) |
Current Rating (31.03.2014) |
|
India |
A1 |
A1 |
|
Risk Category |
ECGC
Classification |
|
Insignificant |
A1 |
|
Low |
A2 |
|
Moderate |
B1 |
|
High |
B2 |
|
Very High |
C1 |
|
Restricted |
C2 |
|
Off-credit |
D |
INDIAN ECONOMIC OVERVIEW
US investment bank
Goldman Sachs has upgraded its outlook on Indian markets as it expects
positive impact of the election cycle.
India’s economy may
grow 4.7 % in the current financial year, lower than the official estimate of
4.9 %, Fitch Rating said. The global rating agency expects the economy to pick
up in the next two financial years.
Global ratings
agency Standard & Poor said increasing focus by India Inc on lowering debt
is likely to improve their credit profiles.
Singapore (1.1
million Indian tourists in 2012), Thailand (one million), the United Arab
Emirates ().98 million) and Malaysia ().82 million) emerged as the preferred
holidays hotspots for Indians. The total figure is expected to increase to 1.93
million by 2017, according to the latest Eurmonitor international report.
There is a $29.34 bn
outward foreign direct investment by domestic companies between April and
January of 2013/14 which has seen some signs of recovery according to a Care
Ratings report.
There are 264 number
of new companies being set up every day on average during 2014. Most of them
are registered in Mumbai. India had 1.38 million registered companies at the
end of January, 2014.
Twitter like
messaging service Weibo Corporation has filed to raise $ 500 million via a US
initial public offering. Alibaba, which owns a stake in Weibo is expected to
raise about $ 15 billion New York this year in the highest profile Internet IPO
since Facebook’s in 2012.
Bharti Airtel has
raised Rs.2,453.2 crore (350 million Swiss Francs) by selling six-year bonds at
a coupon rate of three per cent and maturing in 2020. This is the largest ever
bond offering by an Indian company in Swiss Francs. Bharat Petroleum
Corporation raised 175 million Swiss Francs by selling five year bonds at 2.98
% coupon rate in February.
Indian Oil
Corporation plans to invest Rs 7650 crore in setting up a petrochemical complex
at its almost complete Paradip refinery in Odhisha in three to four years. The
company board is set to consider the setting up of a 700000 tonne per annum
polypropylene plant at an estimated cost at Rs.3150 crore.
Global chief
information officers at gathering in Bangalore in April to meet Indian startups
at an event called Tech50 Watchout for Little Eye Labs-Facebook type deals in
the making.
EXTERNAL AGENCY RATING
|
Rating Agency Name |
ICRA |
|
Rating |
Fund based facilities: “AA+” |
|
Rating Explanation |
High degree of safety and very low credit risk. |
|
Date |
April, 2014 |
RBI DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter in
the publicly available RBI Defaulters’ list.
EPF (Employee Provident Fund) DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available EPF (Employee Provident Fund) Defaulters’ list as of
31-03-2012.
INFORMATION DECLINED BY
|
Name : |
Mr. Rahul Agrawal |
|
Designation : |
Finance Department |
|
Contact No.: |
91-80-28082808 |
|
Date : |
06.05.2014 |
LOCATIONS
|
Registered
Office /Factory 1 / Corporate Headquarters: |
20th
KM, Hosur Main Road, Hebbagodi, Electronics City, Bangalore – 560100,
Karnataka, India |
|
Tel. No.: |
91-80-28422169/28523434/
28082808 / 40144014 |
|
Fax No.: |
91-80-28422623/25531662/28523423 |
|
E-Mail : |
|
|
Website : |
|
|
Area : |
15000
sq. ft. |
|
Locations : |
Owned
|
|
|
|
|
Factory 2 : |
Plot No 113/C2,
Bommasandra Industrial Area, Bommasandra, |
|
|
|
|
Factory 3 : |
Plot No 2,3,4 and
5, Bommasandra – |
|
|
|
|
Factory 4 : |
Plot 213-215 IDA Phase
– II,pashamlaram Medak District – 502307, |
DIRECTORS
As on 31.03.2013
|
Name : |
Ms. Kiran Mazumdar-Shaw |
|
Designation : |
Chairman and Managing Director |
|
Address : |
874/1,
7th Cross III Block, Koramangala, |
|
Date of
Birth/Age : |
20.11.1978 |
|
Qualification
: |
B.Sc.
(Hons.), PG Diploma in Malting and Brewing |
|
Date Of
Appointment : |
01.12.1978 |
|
|
|
|
Name : |
Mr. John Shaw |
|
Designation : |
Vice Chairman |
|
Date of Birth/Age
: |
64 Years |
|
|
|
|
Name : |
Dr. Bala S. Manian |
|
Designation : |
Chairman and Founder |
|
Date of
Birth/Age : |
67 Years |
|
|
|
|
Name : |
Prof. Charles L. Cooney |
|
Designation : |
Director |
|
Address : |
35, |
|
Date of
Birth/Age : |
14.09.1961 |
|
Date of
Appointment : |
27.07.2001 |
|
|
|
|
Name : |
Ms. Mary Harney |
|
Designation : |
Director |
|
Date of
Birth/Age : |
60 Years |
|
|
|
|
Name : |
Prof. Ravi Mazumdar |
|
Designation : |
Director |
|
Address : |
706, |
|
Date of
Birth/Age : |
14.07.1940 |
|
Date of
Appointment : |
08.08.2000 |
|
|
|
|
Name : |
Mr. Russel Walls |
|
Designation : |
Director |
|
Date of
Birth/Age : |
69 Years |
|
|
|
|
Name : |
Mr. Suresh N. Talwar |
|
Designation : |
Director |
|
Date of
Birth/Age : |
74 Years |
|
|
|
|
Name : |
Prof. Catherine Rosenberg |
|
Designation : |
Director |
|
|
|
|
Name : |
Mr. Peter Bains |
|
Designation : |
Director |
KEY EXECUTIVES
|
CORE
COMMITTEE |
|
|
|
|
|
Name : |
Ms. Kiran Mazumdar-Shaw |
|
Designation : |
Chairman and Managing Director |
|
|
|
|
Name : |
Mr. Murali Krishnan |
|
Designation : |
President |
|
|
|
|
Name : |
Mr. John Shaw |
|
Designation : |
Vice Chairman |
|
|
|
|
Name : |
Dr. Abhijit Barve |
|
Designation : |
President |
|
|
|
|
Name : |
Dr. Arun Chandavarkar |
|
Designation : |
Chief Operating Officer |
|
|
|
|
Name : |
Mr. Rakesh Bamzai |
|
Designation : |
President |
|
|
|
|
Name : |
Mr. Ravi Dasgupta |
|
Designation : |
Group Head |
|
|
|
|
Name : |
Mr. Rahul Agrawal |
|
Designation : |
Finance Department |
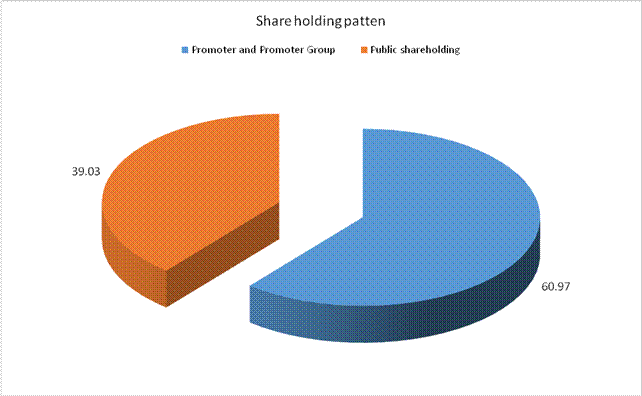
MAJOR SHAREHOLDERS / SHAREHOLDING PATTERN
As on 31.03.2014
|
Category of
Shareholder |
No. of Shares |
Percentage of
Holding |
|
(A) Shareholding of Promoter and Promoter Group |
|
|
|
|
|
|
|
|
80747694 |
40.37 |
|
|
80747694 |
40.37 |
|
|
|
|
|
|
1665558 |
0.83 |
|
|
39535194 |
19.77 |
|
|
41200752 |
20.60 |
|
Total shareholding of Promoter and Promoter Group (A) |
121948446 |
60.97 |
|
(B) Public Shareholding |
|
|
|
|
|
|
|
|
6563087 |
3.28 |
|
|
5523308 |
2.76 |
|
|
27080730 |
13.54 |
|
|
39167125 |
19.58 |
|
|
|
|
|
|
4835898 |
2.42 |
|
|
|
|
|
|
13411855 |
6.71 |
|
|
11381727 |
5.69 |
|
|
75 |
0.00 |
|
|
9254874 |
4.63 |
|
|
1049851 |
0.52 |
|
|
7651508 |
3.83 |
|
|
553515 |
0.28 |
|
|
38884429 |
19.44 |
|
Total Public shareholding (B) |
78051554 |
39.03 |
|
Total (A)+(B) |
200000000 |
100.00 |
|
(C) Shares held by Custodians and against which Depository Receipts
have been issued |
|
|
|
|
0 |
0.00 |
|
|
0 |
0.00 |
|
|
0 |
0.00 |
|
Total (A)+(B)+(C) |
200000000 |
100.00 |
BUSINESS DETAILS
|
Line of Business : |
Manufacturer of biotechnology products and also
engaged in research and development in the biotechnology sector. |
||||||
|
|
|
||||||
|
Products : |
|
GENERAL INFORMATION
|
No. of Employees : |
6700 (Approximately) |
|||||||||||||||
|
|
|
|||||||||||||||
|
Bankers : |
· State Bank of India, Overseas Branch, No. 65, St. Marks Road, Bangalore - 560001, Karnataka, India · The Hongkong and Shanghai Banking Corporation Limited, 7 M.G.Road, Bangalore - 560001, Karnataka, India |
|||||||||||||||
|
|
|
|||||||||||||||
|
Facilities : |
NOTE: LONG TERM
BORROWINGS (a) On February 9, 2000, the Company obtained an
order from the Karnataka Sales Tax Authority for allowing an interest free
deferment of sales tax (including turnover tax) for a period up to 12 years
with respect to sales from its Hebbagodi manufacturing facility for an amount
not exceeding Rs. 649.000 millions. This is an interest free liability. The
amount is repayable in 10 equal half yearly installments of Rs. 65 each
starting from February 2012. (b) On March 31, 2005, the Company entered into an
agreement with the Council of Scientific and Industrial Research (‘CSIR’),
for an unsecured loan of Rs. 3.000 millions for carrying out part of the
research and development project under the New Millennium Indian Technology
Leadership Initiative (‘NMITLI’) Scheme. The loan is repayable over 10 equal
annual installments of Rs. 0.3 starting from April 2009 and carry an interest
rate of 3 percent per annum. (c) (i) On March 31, 2009, the Department of
Scientific and Industrial Research (‘DSIR’) sanctioned financial assistance
for a sum of Rs. 17.000 to the Company for part financing one of its research
projects. The assistance is repayable in the form of royalty payments for
three years post commercialisation of the project in five equal annual
installments of Rs. 3 each. The said projects have been completed during the
year ended March 31, 2010 and the repayments would commence from April 1,
2013. (ii) In addition, during the FY 2010-11, the
Company has further received Rs. 4.000 towards a development project out of
sanctioned amount of Rs. 12.000. The assistance is repayable in the form of
royalty payments for a period of five years post commercialisation of the
project in five equal annual installments of Rs. 3 each. The saidproduct has
not yet been commercialised as at March 31, 2013. (d) On November 3, 2009, the Department of
Biotechnology (‘DBT’) under the Biotechnology Industrial Partnership
Programme (‘BIPP’) has sanctioned financial assistance for a sum of Rs.
53.000 millions to the Company for financing one of its research projects. Of
the said sanctioned amount, the Company had received a sum of Rs. 37.000
millions during the year ended March 31, 2011 and the remaining amount of Rs.
16.000 millions during the previous year. The loan is repayable over 10 half
yearly installments of Rs. 5.000 millions after two years from date of
completion of the project and carries an interest rate of 2 percent per
annum. However, the Company has repaid the loan during the year end March 31,
2013. In addition, on May 23, 2011, the DBT under the
BIPP has sanctioned financial assistance of Rs. 40.000 millions to the
Company for financing another research project. Of thesanctioned amount, the
Company has received a sum of Rs. 12.000 millions during the previous year. The
loan is repayable over 10 half yearly installments of Rs. 4.000 after one
year from date of completion of the project and carries an interest rate of 2
percent per annum. However, the Company has repaid the loan during the year
end March 31, 2013. (e) On August 25, 2010, the Department of Science
and Technology (‘DST’) under the Drugs and Pharmaceutical Research Programme
(‘DPRP’) has sanctioned financial assistance for a sum of Rs. 70.000 millions
to the Company for financing one of its research projects. Of the said
sanctioned amount, the Company has received the first installment of Rs.
14.000 millions during the year ended March 31, 2011 and the remaining amount
during the year ended March 31, 2012. The loan is repayable over 10 annual
installments of Rs. 7 each starting from July 1, 2012, and carries an
interest rate of 3 percent per annum. (f) In respect of the financial assistance
received under the aforesaid programmes (refer notes (b) to (e) above), the
Company is required to utilize the funds for the specified projects and is
required to obtain prior approvals from the said authorities for disposal of
assets / Intellectual property rights acquired/developed under the above
programmes. SHORT TERM
BORROWINGS (i) The Company has obtained foreign currency
denominated loans of Rs. 491.000 (US$ 9 million) [March 31, 2012 - Rs.
812.000 millions (US$ 15.95 million)], carrying an interest rate of LIBOR
plus 0.5% to 1.50% p.a., from Bank/Financial institutions as at March 31,
2013. (ii) The Company has working capital facilities
with Banks carrying interest rate ranging from 11%-13% per annum. These
facilities are repayable on demand, secured by pari-passu first charge on
inventories and trade receivables. As on March 31, 2013, the Company has
utilized fund based limits of Rs. 282.000 millions (March 31, 2012 - Rs.
56.000 millions) |
|
|
|
|
Banking
Relations : |
-- |
|
|
|
|
Auditors : |
|
|
Name : |
S. R. Batliboi and Associates Chartered Accountants |
|
Address : |
Bangalore, Karnataka, India |
|
|
|
|
Joint Venture : |
NeoBiocon FZ LLC |
|
|
|
|
Associate : |
IATRICa
Inc. |
|
|
|
|
Subsidiary : |
·
Syngene
International Limited ·
Clinigene
International Limited ·
Biocon
Biopharmaceuticals Limited ·
Biocon
Research Limited ·
Biocon
SA ·
Biocon
Sdn.Bhd. |
|
|
|
|
Enterprise owned by key
management personnel : |
Glentec
International |
CAPITAL STRUCTURE
As on 31.03.2013
Authorised Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
220000000 |
Equity shares |
Rs.5/- each |
Rs. 1100.000 millions |
|
|
|
|
|
Issued, Subscribed & Paid-up Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
200000000 |
Equity shares |
Rs.5/- each |
Rs. 1000.000 millions |
|
|
|
|
|
(a) Reconciliation of the shares outstanding at
the beginning and at the end of the reporting period
|
Equity Shares |
31.03.2013 |
|
|
|
No. |
Rs. In millions |
|
At the beginning of the year |
200,000,000 |
1000.000 |
|
Issued during the year |
-- |
-- |
|
Outstanding at the end of the year |
200,000,000 |
1000.000 |
(b) Terms/rights
attached to equity shares
The Company has only one class of equity shares
having a par value of Rs. 5 per share.
Each holder of equity shares is entitled to one vote per share. The Company
declares and pays dividends in Indian Rupees. The dividend proposed by the
Board of Directors is subject to the approval of the shareholders in the
ensuing Annual General Meeting.
During the year ended March 31, 2013, final
dividends proposed for distribution to equity shareholders was Rs. 7.5 (March 31, 2012 – Rs. 5) per share. In the event of liquidation of the
Company, the holders of equity shares will be entitled to receive remaining
assets of the Company, after distribution of all preferential amounts, if any.
The distribution will be in proportion to the number of equity shares held by
the shareholders.
(c) Aggregate
number of bonus shares issued during the period of five years immediately
preceding the reporting date
On September 15, 2008, the Company issued
100,000,000 equity shares of Rs. 5 each as fully paid bonus shares by
capitalization of balance in the securities premium account of Rs.500.
iv. Details of shareholders holding more than 5% shares in the Company
|
Equity Shares |
31.03.2013 |
|
|
Equity shares of
Rs. 5 each fully paid |
No. |
% holding |
|
Dr Kiran Mazumdar Shaw |
79,287,564 |
39.64% |
|
Glentec
International |
39,535,194 |
19.77% |
As per of the Company, including its register of shareholders/members.
The above shareholding represents both legal and beneficial ownerships of
shares.
(e) Shares reserved for issue under options for
details of shares reserved for issue under the employee stock option (ESOP)
plan of the Company.
FINANCIAL DATA
[all figures are
in Rupees Millions]
ABRIDGED BALANCE
SHEET
|
SOURCES OF FUNDS |
31.03.2013 |
31.03.2012 |
31.03.2011 |
|
I.
EQUITY
AND LIABILITIES |
|
|
|
|
(1)Shareholders' Funds |
|
|
|
|
(a) Share Capital |
1000.000 |
1000.000 |
1000.000 |
|
(b) Reserves & Surplus |
21068.000 |
19964.000 |
18468.000 |
|
(c) Money
received against share warrants |
0.000 |
0.000 |
0.000 |
|
|
|
|
|
|
(2) Share Application money pending
allotment |
0.000 |
0.000 |
0.000 |
|
Total Shareholders’
Funds (1) + (2) |
22068.000 |
20964.000 |
19468.000 |
|
|
|
|
|
|
(3)
Non-Current Liabilities |
|
|
|
|
(a) long-term borrowings |
400.000 |
605.000 |
658.000 |
|
(b) Deferred tax liabilities (Net) |
302.000 |
349.000 |
396.000 |
|
(c) Other long term
liabilities |
1083.000 |
649.000 |
695.000 |
|
(d) long-term
provisions |
0.000 |
0.000 |
0.000 |
|
Total Non-current
Liabilities (3) |
1785.000 |
1603.000 |
1749.000 |
|
|
|
|
|
|
(4)
Current Liabilities |
|
|
|
|
(a) Short
term borrowings |
773.000 |
868.000 |
963.000 |
|
(b) Trade
payables |
2650.000 |
2511.000 |
1997.000 |
|
(c) Other
current liabilities |
679.000 |
769.000 |
571.000 |
|
(d) Short-term
provisions |
2177.000 |
1488.000 |
1287.000 |
|
Total Current
Liabilities (4) |
6279.000 |
5636.000 |
4818.000 |
|
|
|
|
|
|
TOTAL |
30132
.000 |
28203.000 |
26035.000 |
|
|
|
|
|
|
II.
ASSETS |
|
|
|
|
(1)
Non-current assets |
|
|
|
|
(a) Fixed
Assets |
|
|
|
|
(i)
Tangible assets |
8455.000 |
6757.000 |
6662.000 |
|
(ii)
Intangible Assets |
59.000 |
93.000 |
134.000 |
|
(iii)
Capital work-in-progress |
512.000 |
825.000 |
1027.000 |
|
(iv)
Intangible assets under development |
0.000 |
0.000 |
0.000 |
|
(b) Non-current Investments |
1660.000 |
1664.000 |
920.000 |
|
(c) Deferred tax assets (net) |
0.000 |
0.000 |
0.000 |
|
(d) Long-term Loan and Advances |
4713.000 |
5343.000 |
4162.000 |
|
(e) Other
Non-current assets |
0.000 |
0.000 |
0.000 |
|
Total Non-Current
Assets |
15399.000 |
14682.000 |
12905.000 |
|
|
|
|
|
|
(2)
Current assets |
|
|
|
|
(a) Current
investments |
4530.000 |
4906.000 |
3939.000 |
|
(b)
Inventories |
3589.000 |
3404.000 |
2747.000 |
|
(c) Trade
receivables |
4270.000 |
4450.000 |
4181.000 |
|
(d) Cash
and cash equivalents |
1792.000 |
400.000 |
2103.0000 |
|
(e) Short-term
loans and advances |
510.000 |
302.000 |
160.000 |
|
(f) Other
current assets |
42.000 |
59.000 |
0.000 |
|
Total
Current Assets |
14733.000 |
13521.000 |
13130.000 |
|
|
|
|
|
|
TOTAL |
30132.000 |
28203.000 |
26035.000 |
PROFIT & LOSS
ACCOUNT
|
|
PARTICULARS |
31.03.2013 |
31.03.2012 |
31.03.2011 |
|
|
|
SALES |
|
|
|
|
|
|
|
Income |
19380.000 |
15558.000 |
15611.000 |
|
|
|
Other Income |
515.000 |
666.000 |
572.000 |
|
|
|
TOTAL (A) |
19895.000 |
16224.000 |
16183.000 |
|
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
|
|
Cost of raw materials and packing materials consumed |
8300.000 |
6971.000 |
6173.000 |
|
|
|
Purchases of traded goods |
857.000 |
857.000 |
503.000 |
|
|
|
Employee benefits expense |
2276.000 |
1916.000 |
1456.000 |
|
|
|
Other expenses |
4069.000 |
2893.000 |
2245.000 |
|
|
|
Exceptional
items |
139.000 |
0.000 |
0.000 |
|
|
|
(Increase)/Decrease in inventories
of finished goods, traded goods and work-in-progress |
(179.000) |
(414.000) |
(278.000) |
|
|
|
TOTAL (B) |
15462.000 |
12223.000 |
10099.000 |
|
|
|
|
|
|
|
|
|
PROFIT/
(LOSS) BEFORE INTEREST, TAX,
DEPRECIATION AND AMORTISATION (A-B) (C) |
4433.000 |
4001.000 |
6084.000 |
|
|
|
|
|
|
|
|
|
Less |
FINANCIAL
EXPENSES (D) |
12.000 |
17.000 |
10.000 |
|
|
|
|
|
|
|
|
|
|
PROFIT
/ (LOSS) BEFORE TAX, DEPRECIATION AND AMORTISATION (C-D) (E) |
4421.000 |
3984.000 |
6074.000 |
|
|
|
|
|
|
|
|
|
Less/ Add |
DEPRECIATION/
AMORTISATION (F) |
951.000 |
940.000 |
902.000 |
|
|
|
|
|
|
|
|
|
|
PROFIT/
(LOSS) BEFORE TAX (E-F)
(G) |
3470.000 |
3044.000 |
5172.000 |
|
|
|
|
|
|
|
|
|
Less |
TAX (H) |
713.000 |
489.000 |
579.000 |
|
|
|
|
|
|
|
|
|
|
PROFIT/
(LOSS) AFTER TAX (G-H) (I) |
2757.000 |
2555.000 |
4593.000 |
|
|
|
|
|
|
|
|
|
Add |
PREVIOUS
YEARS’ BALANCE BROUGHT FORWARD |
13750.000 |
12613.000 |
9470.267 |
|
|
|
|
|
|
|
|
|
Less |
APPROPRIATIONS |
|
|
|
|
|
|
|
Transfer to General Reserve |
276.000 |
256.000 |
459.000 |
|
|
|
Dividend |
1500.000 |
1000.000 |
900.000 |
|
|
|
Tax on Dividend |
255.000 |
162.000 |
91.000 |
|
|
BALANCE CARRIED
TO THE B/S |
14476.000 |
13750.000 |
12613.267 |
|
|
|
|
|
|
|
|
|
|
EARNINGS IN
FOREIGN CURRENCY |
|
|
|
|
|
|
|
Export of goods on FOB basis |
9449.000 |
6661.000 |
5244.000 |
|
|
|
Licensing and development fees |
114.000 |
27.000 |
1658.000 |
|
|
|
Other
operating revenue |
342.000 |
79.000 |
0.000 |
|
|
|
Other
income |
0.000 |
5.000 |
0.000 |
|
|
|
Interest
on foreign currency loan given to subsidiary company |
0.000 |
1.000 |
33.000 |
|
|
TOTAL EARNINGS |
9905.000 |
6773.000 |
6935.000 |
|
|
|
|
|
|
|
|
|
|
IMPORTS |
|
|
|
|
|
|
|
Raw Materials |
4917.000 |
3833.000 |
3822.000 |
|
|
|
Packing materials |
177.000 |
193.000 |
45.000 |
|
|
|
Maintenance spares |
49.000 |
44.000 |
30.000 |
|
|
|
Capital goods |
168.000 |
411.000 |
502.000 |
|
|
TOTAL IMPORTS |
5311.000 |
4481.000 |
4399.000 |
|
|
|
|
|
|
|
|
|
|
Earnings / (Loss)
Per Share (Rs.) |
|
|
|
|
|
|
Basic |
14.08 |
13.04 |
23.49 |
|
|
|
Diluted
|
13.95 |
12.92 |
23.27 |
|
QUARTERLY RESULTS
|
PARTICULARS |
30.06.2013 |
30.09.2013 |
31.12.2013 |
31.03.2014 |
|
Type |
1st
Quarter |
2nd
Quarter |
3rd
Quarter |
4th
Quarter |
|
Net Sales |
5410.900 |
5594.100 |
5263.000 |
5714.300 |
|
Total Expenditure |
4315.900 |
4304.300 |
4271.700 |
4492.500 |
|
PBIDT (Excl OI) |
1095.000 |
1289.800 |
991.300 |
1221.800 |
|
Other Income |
276.900 |
203.000 |
107.200 |
111.000 |
|
Operating Profit |
1371.900 |
1492.800 |
1098.500 |
1332.800 |
|
Interest |
2.200 |
2.400 |
1.800 |
2.500 |
|
Exceptional Items |
0.000 |
0.000 |
0.000 |
0.000 |
|
PBDT |
1369.700 |
1490.400 |
1096.700 |
1330.300 |
|
Depreciation |
265.200 |
302.100 |
313.100 |
339.400 |
|
Profit Before Tax |
1104.500 |
1188.300 |
783.600 |
990.900 |
|
Tax |
239.900 |
289.500 |
184.000 |
123.500 |
|
Provisions and contingencies |
0.000 |
0.000 |
0.000 |
0.000 |
|
Profit After Tax |
864.600 |
898.800 |
599.600 |
867.400 |
|
Extraordinary Items |
0.000 |
0.000 |
0.000 |
0.000 |
|
Prior Period Expenses |
0.000 |
55.400 |
0.000 |
0.000 |
|
Other Adjustments |
0.000 |
0.000 |
0.000 |
0.000 |
|
Net Profit |
864.600 |
954.200 |
599.600 |
867.400 |
KEY RATIOS
|
PARTICULARS |
|
31.03.2013 |
31.03.2012 |
31.03.2011 |
|
PAT / Total Income |
(%) |
13.86
|
15.75
|
28.38 |
|
|
|
|
|
|
|
Net Profit Margin (PBT/Sales) |
(%) |
18.42
|
19.57
|
33.13 |
|
|
|
|
|
|
|
Return on Total Assets (PBT/Total Assets} |
(%) |
12.44
|
11.88
|
21.59 |
|
|
|
|
|
|
|
Return on Investment (ROI) (PBT/Networth) |
|
0.16
|
0.15
|
0.27 |
|
|
|
|
|
|
|
Debt Equity Ratio (Total Debt/Networth) |
|
0.05
|
0.07
|
0.08 |
|
|
|
|
|
|
|
Current Ratio (Current Asset/Current Liability) |
|
2.35
|
2.40
|
2.73 |
FINANCIAL ANALYSIS
[all figures are
in Rupees Millions]
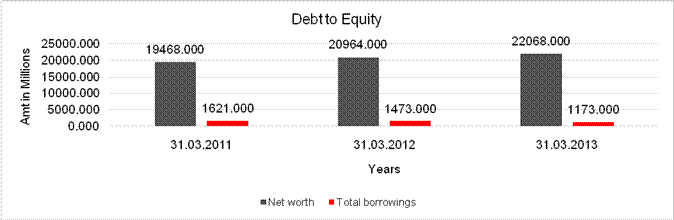
DEBT EQUITY RATIO
|
Particular |
31.03.2011 |
31.03.2012 |
31.03.2013 |
|
|
(Rs.
In Millions) |
(Rs.
In Millions) |
(Rs.
In Millions) |
|
Share Capital |
1000.000 |
1000.000 |
1000.000 |
|
Reserves & Surplus |
18468.000 |
19964.000 |
21068.000 |
|
Net
worth |
19468.000 |
20964.000 |
22068.000 |
|
|
|
|
|
|
long-term borrowings |
658.000 |
605.000 |
400.000 |
|
Short term borrowings |
963.000 |
868.000 |
773.000 |
|
Total
borrowings |
1621.000 |
1473.000 |
1173.000 |
|
Debt/Equity
ratio |
0.083 |
0.070 |
0.053 |
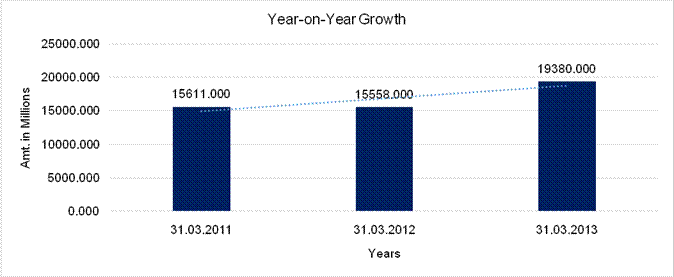
YEAR-ON-YEAR GROWTH
|
Year
on Year Growth |
31.03.2011 |
31.03.2012 |
31.03.2013 |
|
|
(Rs.
In Millions) |
(Rs.
In Millions) |
(Rs.
In Millions) |
|
Sales |
15611.000 |
15558.000 |
19380.000 |
|
|
|
-0.340 |
24.566 |
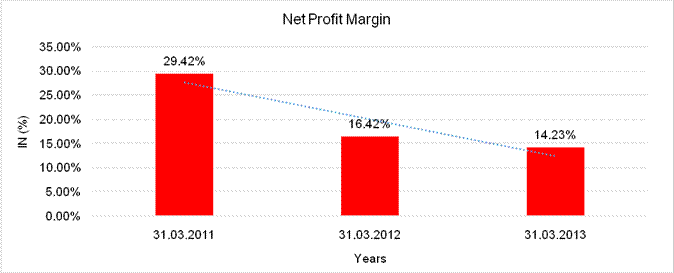
NET PROFIT MARGIN
|
Net
Profit Margin |
31.03.2011 |
31.03.2012 |
31.03.2013 |
|
|
(Rs.
In Millions) |
(Rs.
In Millions) |
(Rs.
In Millions) |
|
Sales |
15611.000 |
15558.000 |
19380.000 |
|
Profit |
4593.000 |
2555.000 |
2757.000 |
|
|
29.42% |
16.42% |
14.23% |
LOCAL AGENCY FURTHER INFORMATION
|
Sr. No. |
Check List by Info Agents |
Available in
Report (Yes / No) |
|
1] |
Year of Establishment |
Yes |
|
2] |
Locality of the firm |
Yes |
|
3] |
Constitutions of the firm |
Yes |
|
4] |
Premises details |
Yes |
|
5] |
Type of Business |
Yes |
|
6] |
Line of Business |
Yes |
|
7] |
Promoter's background |
Yes |
|
8] |
No. of employees |
Yes |
|
9] |
Name of person contacted |
Yes |
|
10] |
Designation of contact
person |
Yes |
|
11] |
Turnover of firm for last
three years |
Yes |
|
12] |
Profitability for last
three years |
Yes |
|
13] |
Reasons for variation
<> 20% |
----- |
|
14] |
Estimation for coming
financial year |
No |
|
15] |
Capital in the business |
Yes |
|
16] |
Details of sister
concerns |
Yes |
|
17] |
Major suppliers |
No |
|
18] |
Major customers |
No |
|
19] |
Payments terms |
No |
|
20] |
Export / Import details
(if applicable) |
No |
|
21] |
Market information |
----- |
|
22] |
Litigations that the firm
/ promoter involved in |
Yes |
|
23] |
Banking Details |
Yes |
|
24] |
Banking facility details |
Yes |
|
25] |
Conduct of the banking
account |
----- |
|
26] |
Buyer visit details |
----- |
|
27] |
Financials, if provided |
Yes |
|
28] |
Incorporation details, if
applicable |
Yes |
|
29] |
Last accounts filed at
ROC |
Yes |
|
30] |
Major Shareholders, if
available |
Yes |
|
31] |
Date of Birth of
Proprietor/Partner/Director, if available |
Yes |
|
32] |
PAN of Proprietor/Partner/Director,
if available |
No |
|
33] |
Voter ID No of
Proprietor/Partner/Director, if available |
No |
|
34] |
External Agency Rating,
if available |
Yes |
LITIGATION DETAILS
|
High Court of Karnataka - Principal Bench
at Bangalore |
|
ITA 650/2013 |
|
Petitioner/Appnt. |
THE COMMISSIONER OF INCOME - TAX |
|
|
Respondent/Defnt.
Name |
M/S BIOCON LIMITED |
|
|
Petnr./Appnt.
Advocate |
ARAVIND KV |
|
|
Respnt./Defnt.
Advocate |
|
|
|
Date Filed |
13/12/2013 |
Classification |
|
District |
Bangalore City |
|
|
Stage |
Hearing
Last Posted for: ADMISSION |
|
Last Action Taken |
ADMIT/RULE Last
Date of Action 01/04/2014 Next hearing date |
|
Before Hon'ble
Judge/s |
DILIP B.BHOSALE B.MANOHAR |
|
Case No |
Court name |
Disposal Dt |
|
ITA 371/2010 |
INCOME TAX APPELLATE TRIBUNAL BANGALORE |
16/07/2013 |
|
Sl.No. |
Honble Judge |
Date of Order |
|
1 |
HONBLE DBBJ & BMJ |
01/04/2014 |
UNSECURED LOANS
|
PARTICULARS |
31.03.2013 (Rs
in Millions) |
31.03.2012 (Rs
in Millions) |
|
LONG TERM BORROWINGS |
|
|
|
Deferred payment
Liability |
324.000 |
454.000 |
|
Other
loans and advance |
|
|
|
NMITU – CSIR
Loan |
2.000 |
2.000 |
|
Financial
Assistance From DSIR |
18.000 |
21.000 |
|
Financial Assistance
From DBT |
0.000 |
65.000 |
|
Financial
Assistance From DST |
56.000 |
63.000 |
|
|
|
|
|
SHORT TERM
BORROWINGS |
|
|
|
From
banks/Financial institutions |
|
|
|
Packing credit foreign currency
loan (unsecured) |
491.000 |
812.000 |
|
Total
|
891.000 |
1417.000 |
CORPORATE
INFORMATION
The Company was incorporated at Bangalore in 1978
for manufacture of biotechnology products. SyngeneInternational Limited (‘Syngene’),
promoted by Dr. Kiran Mazumdar-Shaw, was incorporated at Bangalore in 1993. In
March 2002, Biocon acquired 99.99 per cent of the equity shares of Syngene and,
resultantly, Syngene became the subsidiary of Biocon. Clinigene International
Limited (‘Clinigene’) was incorporated on August 4, 2000 at Bangalore and
became a wholly owned subsidiary of Biocon on March 31, 2001. In February 2012,
Biocon sold its shareholding in Clinigene to Syngene.
On January 10, 2008, Biocon entered into an
agreement with Dr. B. R. Shetty to set up a joint venture Company NeoBiocon
FZ-LLC, with a 50% equity interest incorporated in Dubai (‘NeoBiocon’).
The Company has also established Biocon Research
Limited (‘BRL’), a subsidiary of the Company to undertake research and
development in novel and innovative drug initiatives.
Effective April 30, 2008, Biocon acquired 71%
equity interest in AxiCorp GmbH, Germany (‘AxiCorp’) through its newly
incorporated wholly owned subsidiary company Biocon SA. Switzerland. In
February 2009, Biocon SA acquired an additional 7.4% equity interest in
AxiCorp. During the year ended March 31, 2012, Biocon SA sold its shareholding
in AxiCorp to third parties.
Biocon Biopharmaceuticals Limited (formerly Biocon Biopharmaceuticals
Private Limited), [‘BBL’] was originally incorporated on June 17, 2002 as a
Joint Venture between Biocon and CIMAB SA (‘CIMAB’) with Biocon holding 51 per
cent of the share capital. During the financial year ended March 31, 2011,
Biocon acquired the interest of the joint venture partner, CIMAB. Consequently
all the equity shares of BBL are held by Biocon.
During the year ended March 31, 2011, Biocon set up
a wholly owned subsidiary company in Malaysia, Biocon Sdn. Bhd. (‘Biocon
Malaysia’) for development and manufacture of bio-pharmaceuticals.
Biocon is an integrated healthcare company engaged
in manufacture of biotechnology products for the pharmaceutical sector. The
Company is also engaged in research and development in the biotechnology
sector. During the year ended March 31, 2007, the Company had received an
approval for operation of SEZ Developer and for setting up SEZ Unit operations
to be located within Biocon SEZ.
BUSINESS
OPERATIONS OVERVIEW
During the fiscal year, the group delivered a 18%
top line growth with revenues reaching 25,380 millions vis-à-vis 21,483
millions in FY12. This growth has been driven by a strong momentum in research
services and branded formulations which grew YoY at 36% and 34% respectively.
The biopharma segment excluding branded formulations grew by 10% YoY led by
strong Insulin sales in RoW markets, Immunosuppressants, speciality molecules
like Fidaxomicin and Orlistat.
Group net profits for FY 2013 grew over 50% to Rs. 5087.000 million on the back of exceptional income
recognized on the re-licensing of their generic insulin analogs portfolio.
Further, on a prudent basis, the Company has also made a provision in respect
of its investment in IATRICA Inc. a U.S. startup engaged in development of
molecules, on account of value erosion in its IP.
This fiscal year witnessed good traction in all
their business verticals viz. Small Molecules, Branded Formulations,
Biosimilars, Research Services and Novel Molecules with a firm focus on
emerging markets. Emerging markets are currently outpacing growth in developed
markets reiterating their emphasis in these geographies.
The construction of their new insulin manufacturing
facility in Malaysia is on track. A significant milestone during the fiscal has
been the extension of their partnership with Mylan for generic insulin analogs.
This partnership assumes importance given the strong co-development and
commercialization partner for their key growth vertical. The contract with
Mylan for biosimilar insulin analogues will enable them to optimize their
partnership approach to carve out a large slice of the global Insulin market in
the developed markets. They aim to leverage existing alliances in RoW markets
for penetration of their biosimilar molecules.
The year was also marked by significant advances in
their R&D initiatives in their insulins and biosimilar mAbs programmes
which added momentum to their journey up the value chain. Their biosimilar mAbs
programmes with Mylan are progressing well and are also due to enter clinics
over the course of the next couple of years. They expect FY14 to carry forward
the momentum of their R&D programs and substantiate their efforts of moving
up the value chain.
A detailed performance analysis is
provided in the Management Discussion and Analysis segment, which is annexed to
this report.
SUBSIDIARIES AND
JOINT VENTURES
SYNGENE
INTERNATIONAL LIMITED
Syngene International Limited (Syngene) is the
largest contract and custom research enterprise in India with extensive
competencies in chemistry and biologics. Syngene offers integrated research
services in the drug discovery and development space along with manufacturing
services in APIs, Intermediates and Biologics. The organization offers
value-added service models to complement the evolving needs of global Pharma,
Bio-pharma and Biotech players. Syngene’s clientele spans across industries
like pharmaceuticals, nutraceuticals, agri-chemicals, engineering and
speciality segments, and today includes 16 of the top 20 pharma companies of the
world.
During the year, Syngene had made a preferential
issue of 7.7% equity shares at Rs. 300/- per
share to GE Equity International for a total consideration of Rs. 125 Crores.
In this fiscal year, Syngene recorded a growth of 33%
in top line with revenues touching Rs. 5542.000
millions against Rs. 4182.000 millions in FY12. Syngene’s
EBIDTA margin for the year was 30%, with the operational margin at Rs. 1681.000 millions compared to Rs. 1,404 millions last year, a growth of 20%.
Clinigene International, a 100% subsidiary of
Syngene works across the clinical trial domain, conducting complex
bioavailability, bioequivalence and clinical trials required for validation of
drugs and pharmaceuticals in India. It also has competencies in medical
sciences for the development and enhancement of medical diagnostic, surgical
and therapeutic techniques. For the fiscal ended March 31, 2013, Clinigene
clocked revenues worth Rs. 385 millions
and turned the corner to deliver a net profit of Rs. 4
millions.
BIOCON
BIOPHARMACEUTICALS LIMITED
Biocon Biopharmaceuticals Limited (BBL) is a wholly
owned subsidiary engaged in the production of monoclonal antibodies and other
biologics. During the year, BBL earned revenues worth of Rs. 584.000 millions and generated a net profit of Rs. 55.000 millions. During the year, the Company
commissioned its state of the art biologics facility built with an investment
to the tune of Rs. 1500.000 millions.
In April 2012, the Board of BBL has approved the
merger of the Company with Biocon Limited. The merger application has been
filed with the Hon’ble High Court of Karnataka and the same is pending.
BIOCON RESEARCH
LIMITED
Biocon Research Limited (BRL), a 100% subsidiary of
Biocon, undertakes discovery and development research work in Biologics,
Monoclonal antibody molecules and Proteins. This fiscal year saw the
inauguration of a world class research facility which primarily houses the
operations of BRL. Known as Biocon Research Centre, this state of the art facility
is spread across 200,000 sq. ft. and houses cutting-edge technology
to enable the development of ‘best-in-class’
biologics and biosimilars. For the current year, BRL registered revenues of Rs.
254 millions largely on account of services rendered to other group companies.
The Biosimilar mAbs programme with Mylan undertaken by BRL is in development
stage and hence BRL has reported a net loss of Rs. 899 millions for the year
ended March 31, 2013.
BIOCON SA
Biocon SA is their wholly owned subsidiary based
out of Switzerland, engaged in development and commercialization of
biopharmaceuticals for the global markets. During the current year Biocon SA
entered into an agreement with Mylan for the co-development and
commercialization of insulin analogs. The added impetus from their partner
gives them reason to believe that there is a possibility of an early approval
for insulin products in the regulated markets.
The commitment of the company to the biosimilars
program stays in place as demonstrated by the progress of their molecules in
the clinics. Biosimilar rh-Insulin has completed EU phase III trial while
Biosimilar glargine is expected to enter global phase III trial for the
developed markets shortly. For the current year, at the back of exceptional
income Biocon SA registered a net profit of Rs. 2468.000 millions.
BIOCON SDN. BHD
Biocon SDN. BHD., a wholly owned subsidiary in
Malaysia is setting up the group’s first overseas manufacturing facility in BioXcell,
a biotechnology park being promoted by the Malaysian government. This facility
is expected to be operational with regulatory approvals in 2015. Biocon SDN BHD
is in the process of setting up the manufacturing facility and is yet to
commence commercial operations.
NEO BIOCON FZ LLC
Neo Biocon FZ LLC. is a research and marketing
pharmaceutical company, which was incorporated in January 2008 as a ‘50:50’
joint venture with Dr. B. R. Shetty of Neo Pharma. Based out of Abu Dhabi, Neo
Biocon helps them reach out to the Middle East and GCC with their veritable
portfolio of quality small molecules and biologics. During the current fiscal,
Neo Biocon earned Rs. 227.000 millions in revenues and reported a net profit of
Rs. 66.000 millions.
MANAGEMENT DISCUSSION AND ANALYSIS
The financial statements have been prepared in
compliance with the requirements of the Companies Act, 1956 and Generally
Accepted Accounting Principles (GAAP) in India. This discussion may contain
forward-looking statements that involve risks and uncertainties.
(All amounts in Indian Rupees Millions, except
share data including share price, holding details in a subsidiary company and
amounts expressed in foreign currency).
INDUSTRY LANDSCAPE, OPPORTUNITY AND OUTLOOK
GLOBAL PHARMACEUTICAL MARKET
The year 2012 was an inflection point in the global
pharma growth story. The Global Pharma Market (GPM) grew by 2% in 2012 to reach
$ 856 Billion compared to the median 5% growth seen in the last 5 years1. This
deceleration was largely due to 7 major patent expiries, where they have seen
significant price erosion due to genericization. Though the patent cliff will
continue in 2013, the earnings growth in GPM is expected to return, as a
smaller number of blockbuster drugs are on the patent expiry block going
forward.
Not only did 2012 mark the peak of the currently
ensuing patent cliff, but also witnessed significant strides made in the
biosimilar regulatory regime across major regulated markets. While US now has a
biosimilar bill under consideration which builds on the EU guidelines and
includes ideas of interchangeability and substitutability, EU itself is
revising its biosimilar guidelines to make it easier for quality biosimilar
players to enter the space. Japan has also taken key steps in this area and now
has a couple of biosimilar applications under review.
The thrust for clearer biosimilar regulatory
guidelines have been driven by the increasing concern over the healthcare
burden being shouldered by government and players alike. The experience of EU
with biosimilars has amply demonstrated that the presence of biosimilars enhances existing market competition, increases
access to medicine to a larger set of the population and helps stabilize
healthcare costs2. Biosimilars have been able to command 11% of the
total accessible market in EU since their introduction in
2006; despite the absence of
automatic substitution.
The increasing focus on rationalising healthcare spends
has thus prompted a number of “Big Pharma” companies to re-evaluate their
growth strategies. Consequently a flurry of partnerships and research programs
aimed at creating a foothold in the biosimilar space were announced this year.
These evolving strategies point towards the fact that the line demarcating ‘Big
Pharma’ and Generic players is blurring and they are now looking at the
emergence of a hybrid model where innovation and affordability could go
hand-in-hand.
Thus, 2012 marked the year of changing
dynamics in the global pharma market with the balance of power shifting towards
biologics.
INDIAN PHARMA
MARKET
The Indian Pharma Industry grew a healthy 16% in
FY13 to reach $ 38 Billion against a size of $ 33 Billion in FY12. This growth
was largely export-driven, supported by the domestic industry which grew by 12%
this fiscal. The growth in the domestic market has been led by the chronic
segments which grew by 14% YoY and today account for 30% of the total market up
from 25% in FY093.
The growth of the domestic pharma market has
decelerated over the last few quarters owing to a number of systemic
bottlenecks. The current policy paralysis with regards to the new drug pricing
mechanism, delays in approval of clinical trials, aggressive patent activism
and the evolving marketing guidelines have led to a wait and watch approach
being implemented by most pharma players. In addition macro-economic pressures
of high inflation and lower disposable income in the hands of payers has led to
the current economic slowdown extending to the pharma space as well.
The growth in the domestic market is therefore
expected to remain muted till the current bottlenecks are eased. There are some
signs of recovery at the policy level with clarity emerging from the National
Pharmaceutical Pricing Authority on the latest Essential Medicine List and
fresh regulations being put in place to better manage clinical trials. However,
it is still a long way to go before the policy measures bear fruit and the
domestic growth can sustainably resume. The 2013 sectoral update by Fitch
reiterates a stable outlook for the domestic Pharma market as the inherent
growth drivers are still in place, despite the delta emerging from the current
regulatory ambiguity.
BUSINESS STRATEGY
AND OPERATIONAL PERFORMANCE
THE YEAR GONE BY
The maturing patent cliff in FY13 coincided with
the beginning of Biocon’s gestational growth phase prior to the launch of
biosimilars in major regulated markets. The focus is firmly on reaching the
milestone of $ 1 Billion in revenues by 2018; by leveraging their core
competencies in fermentation, biologics and the India cost advantage.
The emphasis during this gestation period is on
timely execution of their development and manufacturing strategy supported
organically by their current portfolio. Staying true to this mandate, they
delivered a solid top line growth of 18% to reach Rs. 25,380 in FY13 up from Rs. 21,483 in FY12. This growth was broad-based across
their three major drivers: Research Services, Branded Formulations and Core
Biopharma which grew at 36%, 34% and 20% respectively. At Constant Exchange
Rates, group sales* grew by 12% YoY with research services growing by 22% YoY.
CORE BIOPHARMA
The 20% YoY growth in their traditional API
business reflects the strength of their differentiated product offerings that
have grown steadily despite the current headwinds in the Industry. The Core
Biopharma segment consists of their small molecule offerings and their
biosimilars portfolio.
The Small Molecules vertical
which encompasses statins, immunosuppressants, speciality molecules like
Fidaxomicin and other APIs, has seen good traction over the past year. The
growth mantle has been taken up by Immunosuppressants and Fidaxomicin while
statins have largely remained stable. The aggressive genericization of
atorvastatin has intensified the competition in the statins space. They have
been able to maintain their position in statins due to the sustained process
efficiency measures that have helped them stay cost competitive and the
marketing capabilities of their partners. The focus going forward is on
optimising their small molecules portfolio to ensure more marginaccretive
molecules can take over from statins as their flagship offering. They have also
commenced multiple programs under the ANDA initiative that aims to build a
robust pipeline of difficult-to-make, technology-intensive molecules which can
be commercialized in the regulated markets.
The Biosimilars vertical is expected to catalyse
the next wave of growth for them. This vertical comprises of generic
rh-Insulin, generic Insulin Glargine and a pipeline of 7 biologics under
development (2 insulin analogs, 3 Monoclonal Antibodies and 2 other biologics).
The biosimilar development pathway is still evolving as the regulators gain
greater experience and comfort with these molecules. Additionally, there is
intensive lobbying from the innovators to protect their domain of multi-billion
dollar drugs.
In such a dynamic scenario, the commercialization
success of their offerings hinge on the quality of their partnerships. They
extended their biosimilar partnership with Mylan this year to bring generic
insulin analogs into the fold. The partnership with Mylan builds on their
extensive co-development experience with biologics, endorses the quality of
their products and re-affirms their faith in their capabilities of bringing
these difficult to make molecules to the market. The current partnership
extension is on the same lines as their earlier arrangement for biosimilars
whereby both the partners share development and capital costs. Mylan gets
exclusive commercialization rights in select regulated markets and the two
partners enjoy co-marketing rights in all other regions. This partnership therefore
complements the regional partnership model that they have employed in other
geographies. They are today present
in over 40 countries with their generic rh-Insulin and over 5 countries with
generic Insulin Glargine.
The strategy going forward is to enhance their
emerging markets footprint while the development work of these molecules for
the regulated market progresses simultaneously. They achieved several
milestones this year on the biosimilar development front as listed below:
Completed the EU Phase III trial for generic
rh-Insulin and successfully established the efficacy endpoint with comparable
safety and efficacy against the innovator products
Successfully concluded the Global Phase I trial for
generic Insulin Glargine
Initiated the Global Phase III trial for Biosimilar
Trastuzumab post the successful completion of its Global Phase I trial
Recruitment completed for India Phase III trial of
Biosimilar Trastuzumab
Given the flux in the regulatory environment, they
have attempted to keep the regulators abreast at every stage of development.
The next steps would be to re-engage with the authorities to:
Determine the quality of their data, before
proceeding with the dossier filing of generic rh-Insulin in EU
Ascertain the development pathway for generic
rh-Insulin in US
Understand the requirements and expectations of the
EMEA and FDA prior to the initiation of a global Phase III trial for generic
Insulin Glargine
They continue to make progress on the 5 other biosimilar
molecules which are currently in pre-clinical development. They hope to bring
some of these molecules to the clinic this fiscal. The headway in their India
Phase III for Biosimilar Trastuzumab is encouraging and gives them the
confidence that they should be able to bring it to the markets soon.
The rapidly changing biosimilar landscape is
expected to fundamentally alter the balance of power in the GPM. Navigating this space not only requires
sustained investments in development and manufacturing but also in building a
conducive regulatory understanding to bring these products to the market. Being
one of the first few players in the biosimilar space for regulated markets,
they face greater hurdles in creating the requisite acceptance and the biosimilar
development pathway. Their strategy of commercializing the same quality product
across geographies, beginning with emerging markets has helped them gain
greater acceptance, confidence and build capabilities to take on the
goliaths of the biotech world.
BOARD OF
DIRECTORS:
PROFILE AND
COMPOSITION:
The Board of directors comprises eight members
including two executive directors, six non-executive directors, of which five
are independent directors. Ms. Kiran Mazumdar-Shaw is the Chairman and Managing
Director (‘CMD’) of the Company and Mr. John Shaw is the Vice- Chairman. Ms.
Kiran Mazumdar-Shaw and Mr. John Shaw conduct the day-to-day management of the
Company, subject to the supervision and control of the Board of Directors. The
independent directors on the Board are management professionals, scientists,
and technocrats who are senior, competent and highly respected persons from
their respective fields. A brief profile of the Board is as under:
Ms. Kiran
Mazumdar-Shaw, 60 years, CMD, is a first generation
entrepreneur with more than 37 years’ experience in the field of biotechnology.
She is a founder promoter and has led the Company since its inception in 1978.
After graduating in B.Sc. (Zoology Hons.) from Bangalore University in 1973,
she completed her post-graduate degree in malting and brewing from Ballarat
College, Melbourne University in 1975. She has been awarded with several
honorary degrees including Honorary Doctorate of Science from Ballarat
University, in recognition of pre-eminent contribution to the field of
Biotechnology - 2004, Doctor of Technology from the University of Abertay
Dundee - 2007, Doctor of Science from the University of Glasgow - 2008 and
Doctor of Science from the Heriot-Watt University, Edinburgh - 2008. She is the
recipient of several awards, the most noteworthy being the ‘Padmabhushan’ Award
(one of the highest civilian awards in India) in 2005 conferred by the
President of India, the Nikkei Asia Prize, 2009 for Regional Growth, Express
Pharmaceutical Leadership Summit Award 2009 for Dynamic Entrepreneur, the
Economic Times ‘Businesswoman of the Year’, the ‘Veuve Clicquot Initiative for
Economic Development For Asia), Ernst & Young’s Entrepreneur of the Year
Award for Life Sciences and Healthcare, ‘Technology Pioneer’ recognition by
World Economic Forum and The Indian Chamber of Commerce Lifetime Achievement
Award. She heads several biotechnology task forces including the Karnataka
Vision Group on Biotechnology, an initiative by the Government of Karnataka and
the National Taskforce on Biotechnology for the Confederation of Indian
Industry (CII). She is also a member of the Prime Minister’s Council on Trade
and Industry and is a Member, Governing Body and General Body of the Indian
Pharmacopoeia Commission, an Autonomous Body of the Government of India.
Recently she has been appointed on the Board of trustees of U.S. Pharmacopeial
Convention (USP), USA and trustee of Bangalore Political Action Committee
(BPAC).
Mr. John Shaw, 64 years, Vice-Chairman, is a foreign promoter and
a whole-time director of the Company. He is also a controlling shareholder and
director of Glentec International. He completed M.A. (Hons.) in History and
Political Economy from Glasgow University, U.K. in 1970. He had 27 years
experience with Coats Viyella plc. in various capacities including finance and
general administration and also served as Finance Director and the Managing
Director of Coats Viyella group companies across geographies, before he came on
the Board of Biocon
Limited in 1999.
Dr. Bala S.
Manian, 67 years, has been a part of the
Silicon Valley entrepreneurial community over the last three decades and is
responsible for successfully starting several life science companies. Dr.
Manian is a co-founder of Quantum Dot Corporation and a co-founder of SurroMed
Corporation. He was also chairman of Entigen Corporation, a Bioinformatics
Company. He was the founder and Chairman of Biometric Imaging, Inc. Prior to
founding Biometric Imaging, Inc., Dr. Manian founded Digital Optics
Corporation, an optical instrumentation and systems development Company in 1980
and two other Companies, Lumisys and Molecular Dynamics in June, 1987. Dr.
Manian is presently the CEO of ReaMetrix Inc. He has been recognized through
several awards for his contributions as an educator, inventor and an
entrepreneur. In February 1999, the Academy of Motion Picture Arts and Sciences
awarded a Technical Academy Award to Dr. Manian for advances in digital
cinematography. He has a B.S. in Physics from the University of Madras, a M.S.
in Applied Optics from the University of Rochester and a Ph.D. in mechanical
engineering from Purdue University. He was a faculty member of the University
of Rochester’s Institute of Optics for four years, teaching courses in optical
fabrication and testing, optical instrumentation and holography. Presently, he
also serves as a member of the Board of Trustees of University of Rochester.
Prof. Charles L.
Cooney, 69 years, is the Professor of
Chemical and Biochemical Engineering, Faculty Director of the Deshpande Center
for Technological Innovation, Massachusetts, U.S.A. He obtained his Bachelor’s
degree in Chemical Engineering from the University of Pennsylvania in 1966,
Master’s degree and Ph.D in Biochemical Engineering from MIT in 1967 and 1970
respectively. His research interests span topics in biochemical engineering and
pharmaceutical manufacturing. He is a recipient of several prestigious awards,
including Gold Medal of the Institute of Biotechnology Studies (London), the
Food, Pharmaceutical and Bioengineering Award from the American Institute of
Chemical Engineers and the James Van Lanen Distinguished Service Award from the
American Chemical Society. He serves as a consultant to and also as director of
a number of biotech and pharmaceutical companies globally. He is also on the
editorial board of several professional journals.
Ms. Mary Harney, 60
years, a graduate from Trinity College in BA (General Studies) 1976. Her
political career began when she was appointed to the Senate by the Taoiseach (Prime
Minister) in 1977. She was a member of the Dublin County Council from 1979 to
1990. She served as a member of the Dail Eireann (Irish Parliament) from 1981
to 2011 and was a Deputy Prime Minister for 9 years. She is the longest serving
woman ever in the Irish Parliament. She was President of the EU Council of
Enterprise Ministers during Ireland’s Presidency. She was a founder member of
the Progressive Democrats in 1985 and was the only woman to lead a political
party in Ireland when she succeeded Des O’Malley in 1993. She was the first
woman to be elected Auditor of the Trinity College Dublin College Historical
Society (Debating society). She has won many international awards for student
debating. She is also an Honorary Member of the International
Women’s Forum.
Prof. Ravi Mazumdar, 58 years, completed Ph.D from the University of
California, Los Angeles, USA in 1983. Prior to this, he obtained B.Tech from
the Indian Institute of Technology, Bombay in 1977 and Masters in Science from
the Imperial College of Science, London in 1978. He is a professor in
University of Waterloo, Canada and has been professor in several prestigious
universities including Purdue University, U.S.A., Columbia University, U.S.A.,
University of Essex, U.K., McGill University, Canada and the Indian Institute
of Science,
Bangalore. He has over 100 referred publications in
international journals in the area of applied probability and stochastic
processes, nonlinear dynamical systems, statistical signal processing, queuing
theory and in the control and design of high-speed networks. He has been a
member of several advisory committees and working groups, including the US
Congress Sub-Committee on Science and Technology. He is a Fellow of the Royal
Statistical Society, U.K. and Fellow of the Institute of Electrical and
Electronics Engineers, Inc., U.S.A. He is a younger brother of Ms. Kiran
Mazumdar Shaw.
Mr. Russell Walls, 69 years, is a Fellow Member of the Association of
Chartered Certified Accountants, U.K and brings to the Board his extensive
experience in the field of finance. He possesses experience as director across
a range of industries such as pharmaceuticals, textiles, transport and leisure.
He is presently on the board of Signet Jewelers Ltd., Treasurer and Trustee of
The British Red Cross and Member of the Finance Commission of The International
Federation of The Red Cross. He has formerly held positions as finance
director, chairman of audit committee and has held board positions in companies
such as BAA plc, Wellcome plc, Coats Viyella plc, Stagecoach Group plc, Hilton
Group plc and others.
Mr. Suresh N. Talwar, 74 years, is a partner in Talwar Thakore and
Associates, a law firm of repute. He completed his professional studies in Law from
the Government Law College, Bombay in 1961. He was a partner of Crawford Bayley
& Co., a reputed Indian law firm. His area of professional specialisation
includes corporate laws and other related matters. He has been the legal
counsel to numerous Indian companies, multinational corporations, Indian and
foreign banks. He is on the board of several leading companies in India.
CONTINGENT
LIABILITIES:
(Rs. in millions)
|
Particular |
31.03.2013 |
31.03.2012 |
|
(a) Claims against the Company not
acknowledged as debt Taxation matters under appeal (Direct and Indirect
taxes) |
812.000 |
287.000 |
|
(b) Guarantees |
|
|
|
(i) Corporate guarantees given in favour of the Central Excise Department in respect of certain performance obligations of the subsidiaries. |
|
|
|
Syngene |
218.000 |
218.000 |
|
BBPL |
131.000 |
131.000 |
|
Clinigene |
27.000 |
27.000 |
|
Total |
376.000 |
376.000 |
|
(ii) Corporate guarantee given by Syngene in favour of the CED in respect of certain performance obligations of Biocon. |
465.000 |
465.000 |
|
(iii) Corporate guarantees given in
favour of a bank towards loans obtained by Clinigene |
75.000 |
77.000 |
|
(iv) Guarantee given for securing loan
facilities granted to AxiCorp GmbH. |
0.000 |
271.000 |
|
(v) Guarantees given by banks on
behalf of the Company for financial and other contractual obligations of the
Company. The necessary terms and conditions have been complied with and no
liabilities have arisen. (refer note below) |
554.000 |
505.000 |
|
(vi) Corporate guarantees given in
favour of a bank towards loans obtained by Biocom Malaysia |
1240.000 |
51.000 |
INDEX OF CHARGES
|
S.NO. |
CHARGE ID |
DATE OF CHARGE CREATION/MODIFICATION |
CHARGE AMOUNT SECURED |
CHARGE HOLDER |
ADDRESS |
SERVICE REQUEST NUMBER (SRN) |
|
1 |
10300393 |
29/06/2011 |
44,267,000.00 |
DEPARTMENT OF BIOTECHNOLOGY |
6-8TH FLOOR, BLOCK NO. 2, CGO COMPLEX, NEW DELHI - 110003, INDIA |
B18143230 |
|
2 |
10255822 |
12/11/2010 |
57,081,000.00 |
DEPARTMENT OF BIOTECHNOLOGY |
6-8TH FLOOR, BLOCK NO. 2, CGO COMPLEX, LODHI ROAD, NEW DELHI - 110003, INDIA |
B01390137 |
|
3 |
10059940 |
17/02/2007 |
650,000,000.00 |
STATE BANK OF INDIA |
OVERSEAS BRANCH, NO. 65, ST. MARKS ROAD, BANGALORE - 560001, KARNATAKA, INDIA |
A11660974 |
|
4 |
10060347 |
17/02/2007 |
650,000,000.00 |
STATE BANK OF INDIA |
OVERSEAS BRANCH, NO. 65, ST. MARKS ROAD, BANGALORE - 560001, KARNATAKA, INDIA |
A11661360 |
|
5 |
80022593 |
23/07/2010 * |
1,773,500,000.00 |
THE HONGKONG AND SHANGHAI BANKING CORPORATION LIMITED |
7 M.G.ROAD, BANGALORE - 560001, KARNATAKA, INDIA |
A90645920 |
* Date of charge modification
FIXED ASSETS
Tangible Assets
· Land
· Buildings
· Leasehold Improvements
· Plant and Equipment
· Research and Development Equipments
· Furniture and Fixtures
·
Vehicles
Intangible Assets
· Intellectual Property Rights
·
Computer Software
·
Marketing Rights
PRESS
RELEASE
BIOCON DELIVER 16% REVENUE GROWTH IN FY14
Apr 24, 2014
FY14 Performance: Revenues Rs 2,9330.000 Millions; EBITDA Rs 7430.000 Millions; PAT Rs 4140.000 Millions
Commenting on the quarterly performance and highlights, Chairman and Managing
Director, Kiran Mazumdar-Shaw stated,"Biocon ended fiscal 2014 on a strong
note. We delivered 16% revenue growth along with a healthy EBITDA Margin of
25%. This fiscal we recorded robust performance, an outcome of our efforts
aimed at optimising our product mix, augmenting capacities and driving
operational efficiencies.
We have delivered on our promise of affordable innovation through
commercialization of Alzuma(TM), an anti-CD6 novel biologic for Psoriasis; and
CANMA(TM), the world's most affordable trastuzumab. We are pleased that a large
number of patients benefitted from these two products. We continue with our
innovation led business strategy and look forward to deliver superior value to
our stakeholders."
Highlights:
Diversified Revenue growth in FY14
-Biopharmaceuticals Segment: 14% YoY
- Research Services Segment: 28% YoY
Group EBITDA and PAT margins at 25% and 14% respectively
Research and Development investments of Rs.131 Crores (6% of Biopharma Segment
sales)
Commercial Launch of the world's most affordable trastuzumab, CANMAb(R)
Inauguration of Baxter's Global Research Centre at Syngene
Elevation of Dr. Arun Chandavarkar to Chief Executive Officer and Joint
Managing Director
Appointment of Mr. Ravi Limaye, as President - Marketing, Biocon
Business Performance
Financial Highlights: Q4 FY14 (In Rs in Millions)
Revenue : 7460.000 Millions
Research and Development Expenses: 290.000 Millions
EBITDA: 193 (EBITDA Margin: 26%)
PAT: 113 (PAT Margin: 15%)
Revenue Breakup:
Biopharmaceuticals: 5350.000 Millions
Research Services: 1880.000 Millions
Other Income: 230.000 Millions
Biopharma
The biopharma segment delivered a growth of 14% YoY and 15% YoY for FY14 and Q4
FY14 respectively.
Commenting on this performance, Ravi Limaye, President - Marketing, Biocon,
said "The healthy growth of 14% this fiscal from our biopharma segment
reflects our ability to manage the rapidly changing business environment. We
continue our efforts to rebalance our product portfolio in the biopharma
segment to ensure higher margin accretion despite the underlying current of
commoditization in some of our key product portfolios. The strong traction in
biosimilars, supported by capacity augmentation should hold us in good stead
till our Malaysian facility comes online. The return to growth of our branded
formulations segment is a positive sign, and we expect this vertical to deliver
strong growth going forward."
Small Molecules
Our sustained focus on optimizing our product portfolio in Small Molecules has
helped us deliver a healthy set of numbers this fiscal. The recent portfolio
realignment helped us offset the impact of continuous commoditization in the
statins space. We have witnessed good business traction in Immunosuppressants
and specialty products and expect it to sustain in FY15.
We have made investments this fiscal to progress from APIs to generic
formulations and ANDAs, thereby moving up the pharma value chain. These
investments will help us sustain our growth momentum in the coming years by
ensuring a healthier product mix. We expect to initiate our dossier filings
from this portfolio in FY15, which will bear dividends over the next few years.
Biosimilars
Our generic Insulins portfolio has delivered strong growth this fiscal through
our expanding geographical footprint and increasing market penetration. Our
generic rh-Insulin is now approved in over 55 countries. This growth has been
supported by our enhanced capacities over the course of fiscal 2014. Our
Malaysia project is on track to be commissioned in FY15. We continue to make
progress on our various developmental programs, and hope to bring some of them
to the clinic in FY15.
Branded Formulations
The branded formulations vertical grew at 9% YoY this quarter, vis-a-vis the
industry growth of 7% YoY, delivering revenues of Rs 930.000 Millions in Q4 FY14.
We closed FY14 with sales of Rs 3910.000 Millions and a growth of 13% vs. the
overall industry growth of 6%, driven by our flagship brands of BioMAb
EGFR9(R), Abraxane(TM), Insugen(R) and Basalog(R). We launched our trastuzumab
product, CANMAb(R) in India in Q4 FY14.
Novel Molecules
We continue with the clinical development of our novel oral insulin molecule,
IN 105, in USA, in partnership with BMS.
We have initiated the groundwork to explore expanded indications for our novel
Anti CD6 molecule, Itolizumab. We are in discussions with potential partners
for the out-licensing of this molecule.
Research Services
The research services segment grew at 28% YoY in FY14 and 14% in Q4 FY14. The
quarter also saw the inauguration of the Baxter Global Research Centre, another
multi-year dedicated services engagement with a global pharma company.
Commenting on this performance, Peter Bains, Director Syngene International,
said, "We are pleased to report a strong set of numbers for FY14 with 28%
YoY revenue growth which has also translated into a strong EBITDA growth. It is
encouraging to see strong, broad based business momentum across our chemistry,
biology, biologics and clinical service platforms. Our latest long term,
dedicated research Centre for Baxter reflects the strong value that we bring to
biopharma through wide ranging quality services and flexible business models.
Our order book and outlook for the coming year remains robust, and, in line
with our midterm outlook, we continue to invest in strengthening and enhancing
our discovery and development service platforms."
Appointments:
Mr. Ravi Limaye has joined Biocon as President, Marketing. He has over 25
years' experience with global pharma companies like Novartis, GSK and Roussel Uclaf
(now part of Sanofi). Prior to joining Biocon, Ravi was with Novartis, India,
responsible for their Specialty Business, Business Development and Licensing.
Outlook
We expect FY15 to reflect continued business momentum with biosimilars, branded
formulations and research services driving growth. The progress in our
development pipeline (across biosimilars and novel molecules) will see some of
our molecules enter the clinic. We continue to make investments in
infrastructure and people to support our growth.
DR ARUN CHANDAVARKAR APPOINTED AS CEO AND JOINT MANAGING DIRECTOR OF
BIOCON LTD
April 24, 2014
Biocon Ltd, Asia's premier biotechnology company, announced that the Board of Directors of the Company, in their meeting today, have approved the induction of Dr Arun Chandavarkar, on to the board of Biocon Limited. He has also been appointed as Chief Executive Officer and Joint Managing Director.
Welcoming Arun on the Board, Kiran Mazumdar-Shaw, CMD, Biocon, said, "I am
extremely happy to have Arun join the Board of Directors and take on the mantle
of CEO and Joint MD. Arun has been a core member of the leadership team at
Biocon and has worked very closely with me over the last 24 years. He has
played a pivotal role in the evolution of Biocon and I am confident that, in
his new role, he will build immense value for Biocon and its
stakeholders."
Dr Arun Chandavarkar, CEO and Joint MD, Biocon, said, "I am delighted and
honoured to join the Board of Biocon. These are exciting times and I look
forward to steering the Company to the next level."
Dr. Arun Chandavarkar has a B. Tech from IIT, Bombay and earned his Ph.D. in
Biochemical Engineering from the Massachusetts Institute of Technology,
Cambridge, USA
Dr Chandavarkar has been the Chief Operating Officer of Biocon since 2006. He
joined Biocon as General Manager - Operations in 1990. Under his leadership,
Biocon has established expertise in diverse technology platforms spanning
microbial fermentation, cell culture, chemical synthesis and purification to
develop a wide range of products from specialty enzymes to active
pharmaceutical ingredients to recombinant therapeutic proteins.
BIOCON INKS LICENSING PACT WITH QUARK PHARMA
December 18, 2013
Biotechnology major Biocon on Wednesday said it has inked a pact with Quark Pharmaceuticals, Inc to develop a range of siRNA (small interfering RNA) based novel therapeutics.
The companies have entered into a licensing and collaboration agreement for the development of a range of siRNA (small interfering RNA) based novel therapeutics, Biocon Ltd said in a statement.
“This collaboration will enable Biocon to co-develop, manufacture and commercialise QPI-1007, a novel siRNA drug candidate for ophthalmic conditions, for India and other key markets,” it added.
As part of the agreement, Biocon will have access to Quark’s innovative and proprietary siRNA technology platform that can be leveraged for the development of novel therapeutics for various unmet medical needs.
The company, however, did not disclose financial details.
Commenting on the development, Biocon Chairperson and Managing Director Kiran Mazumdar Shaw said Quark is the world leader in this technology and their joint development efforts on QPI—1007, targeting ocular neuroprotection, aims at providing relief to several patients suffering from serious ophthalmic conditions.
“This collaboration reinforces our commitment to develop and introduce innovative therapeutics to India to meet the unmet medical needs. We hope to use this technology for developing several other novel therapeutics,” she added.
QuarkPharma CEO Daniel Zurr said the collaboration will position Biocon as the leading siRNA company in India and as an international player in this new drug category.
BIOCON LAUNCHES
ALZUMAB™ - A ‘FIRST IN CLASS’ NOVEL BIOLOGIC TREATMENT FOR PSORIASIS PATIENTS
IN INDIA
• ALZUMAb™-World's
first novel anti-CD6 antibody developed by Biocon to address a large unmet need
for the treatment of Psoriasis in India
• Excellent safety
and efficacy profile with very low opportunistic infection rates and longer
remission period
• Offers a new
treatment option for Psoriasis with a less aggressive dosing regimen and a
longer treatment free period, ensuring better patient compliance and
convenience
• Is an innovative
affordable treatment option with a promise to offer a better quality of life
for the patient
• Will provide an effective biologic
treatment solution to 1-2% of Indian population suffering from Psoriasis
• ALZUMAb™- (Itolizumab)
has demonstrated preclinical and/clinical evidence in treating other autoimmune
diseases like rheumatoid arthritis, psoriatic arthritis and multiple sclerosis.
• Biocon is committed to offering this novel
anti -CD6 biologic from India to patients across the globe
Bangalore, (India): August 10, 2013
Biocon, Asia's premier biotechnology company,
today announced the launch of its ‘first in class’ novel biologic ALZUMAb™
(Itolizumab), the world’s first anti-CD6 monoclonal antibody to be introduced
for treating patients with chronic plaque psoriasis, in India.
ALZUMAb™ with a unique Mechanism of Action
(MOA) offers superior safety and similar efficacy profile compared to other
existing therapies, and has a long remission period with very low opportunistic
infection rate. Psoriasis is a socially debilitating disease affecting 2-3 % of
world population. The global Psoriasis market size is estimated to cross $8 bn
by 2016.
Commenting on this development Ms. Kiran Mazumdar-Shaw,
Chairperson and Managing Director, Biocon said, “Biocon’s ALZUMAb™ (Itolizumab)
is the first anti-CD6 monoclonal antibody to be commercialized, an outcome of
path breaking research in India .This new line of treatment will usher in a
paradigm shift in the management of psoriasis. We are proud that this will be
the first instance of a breakthrough innovation from India with a potential to
treat multiple autoimmune diseases and making a difference to a much larger
patient population across the world.”
Mr. Rakesh Bamzai President-Marketing, Biocon
said:
“Biocon is committed to address the
huge unmet need of patients suffering from Psoriasis through its ‘first in
class’ biologic ALZUMAb™. Compared
to existing therapies, ALZUMAb™ offers a better safety and efficacy profile to the patients with longer
remission periods and lower infection rates that will lead to better patient
compliance and overall reduction in the cost burden to the patient.”
Indicated for the treatment of
Moderate-to-Severe Psoriasis, ALZUMAb™ is being introduced in India by
Biocon's Immunotherapy Division. Formulated as an infusion drug it is
manufactured at Biocon's Biopharma manufacturing facility at Biocon Park,
Asia’s largest biotech hub in Bangalore.
Biocon: Front runner in introducing a
biologic based on TH 17 pathway
ALZUMAb™ is the world’s first anti -CD6
biologic addressing the Th 17 pathway that has completed its ‘Lab to Market’
journey. Emerging data shows that in addition to Th 1, Th17 cells, play a
critical role in autoimmune diseases, and Biocon is a front-runner in the race
to develop biologics targeting the Th17 pathway with ALZUMAb™ (Itolizumab).
This differentiated MOA positions Itolizumab
as a potentially 'best-in-class' treatment for psoriasis, and other autoimmune
diseases.
Itolizumab: A high potential biologic for a
range of autoimmune diseases
ALZUMAb™ is an outcome of breakthrough
innovation at Biocon which has the potential to transform the treatment options
for several autoimmune diseases besides psoriasis like rheumatoid arthritis,
multiple sclerosis and others. It is potentially a pipeline within a product.
Its launch in India for psoriasis is the
first milestone on a promising and exciting journey towards new treatment
options for life-changing autoimmune diseases. Biocon is committed to taking ALZUMAb™
(Itolizumab) from India to patients worldwide with a global partner.
Notes to the Editor:
ALZUMAb™ (Itolizumab) Unique MOA
ALZUMAb™ (Itolizumab) is the world’s
'first-in-class' humanized anti-CD6 MAb that has a unique mechanism of action
(MOA) targeting the CD6 pathway. CD6 is a pan T-Cell marker involved in
co-stimulation, adhesion and maturation of T-Cells, which have been found to
play a leading role in autoimmune diseases.
ALZUMAb™ (Itolizumab), by binding to CD6,
down regulates T cell activation, causes reduction in synthesis of
pro-inflammatory cytokines and possibly plays an important role by reducing T
cell infiltration at sites of inflammation.Unlike other biologics like cytokine
inhibitors which act downstream in the pro-inflammatory cascade, Itolizumab
acts upstream inhibiting the formation of cytokines, resulting in lower
infection rates and longer remission periods.
Clinical Development
In addition to the Phase 3 TREAT-PLAQ study,
two positive Phase 2a dose finding studies have been conducted one each in
rheumatoid arthritis and psoriasis. A total of over 300 patients have been
treated with ALZUMAb™ (Itolizumab), of these, about 100 patients have received
the drug for 52 weeks with encouraging safety and tolerability profile.
Itolizumab was approved by Drugs Controller General of India for treatment of
Psoriasis in India in 2013.
Kiran Mazumdar-Shaw
Receives Coveted 'Othmer Gold Medal 2014'
The first Indian and the 20th recipient of this award
Biocon Limited, Asia's premier biotechnology company, announced today that its
Chairperson and Managing Director, Kiran Mazumdar-Shaw, received
the 'Othmer Gold Medal 2014' on Thursday, May 15, in Philadelphia,
Pennsylvania, USA.
Established by the Chemical Heritage Foundation (CHF) in 1997, the annual award
honors outstanding individuals who have made multifaceted contributions to
chemical and scientific heritage through outstanding activity in such areas as
innovation, entrepreneurship, research, education, public understanding,
legislation or philanthropy.
Kiran Mazumdar-Shaw is the third woman to receive the Othmer Gold Medal and the
first Indian to make it to this prestigious group. Nineteen people have been
conferred with this honor till date, including Arnold O. Beckman, Carl
Djerassi, Mary Lowe Good, Harry B. Gray, Jon M. Huntsman, Kazuo Inamori, Robert
S. Langer, P. Roy Vagelos, James D. Watson, George Whitesides and Ahmed Zewail.
Carsten Reinhardt, President and CEO, CHF, handed over the Foundation's top
award to Ms. Mazumdar-Shaw during CHF'sChemical Heritage Day celebration
that culminated with the presentation of the Othmer Gold Medal.
The CHF lauded Kiran Mazumdar-Shaw's pioneering efforts in biotechnology and
for scripting a hugely successful entrepreneurial story by drawing on her
knowledge of fermentation science to build Biocon, India's first biopharma
company.
Accepting the award, Kiran Mazumdar-Shaw, CMD - Biocon, said, "I am
deeply grateful to the Chemical Heritage Foundation for awarding me the Othmer
Gold Medal 2014. This is an honour akin to being invited to join an
extraordinary league of scientists, engineers, entrepreneurs, thought leaders
and philanthropists, an emotion that is both proud and humbling at the same
time.
As an entrepreneur who is driven by a sense of purpose to make a difference to
global healthcare, I take pride in saying that Biocon is contributing to
affordable innovation which goes to the core of ensuring a global right to
healthcare. In this, we seem to be drawing lessons from Donald Othmer's
practical and low-cost approach to innovation."
A pioneer of the biotechnology industry in India, Ms. Mazumdar-Shaw has made
her country proud with a globally recognized biopharma enterprise -
Biocon.Under her stewardship, Biocon has evolved into India's largest biopharma
company committed to innovation and affordability to deliver world class
therapeutics to patients across the globe.
CMT REPORT (Corruption, Money Laundering & Terrorism]
The Public Notice information has been collected from various sources
including but not limited to: The Courts,
1] INFORMATION ON
DESIGNATED PARTY
No exist designating subject or any of its beneficial owners,
controlling shareholders or senior officers as terrorist or terrorist
organization or whom notice had been received that all financial transactions
involving their assets have been blocked or convicted, found guilty or against
whom a judgement or order had been entered in a proceedings for violating
money-laundering, anti-corruption or bribery or international economic or
anti-terrorism sanction laws or whose assets were seized, blocked, frozen or ordered
forfeited for violation of money laundering or international anti-terrorism
laws.
2] Court Declaration :
No records exist to suggest that subject is
or was the subject of any formal or informal allegations, prosecutions or other
official proceeding for making any prohibited payments or other improper
payments to government officials for engaging in prohibited transactions or
with designated parties.
3] Asset Declaration :
No records exist to suggest that the property or assets of the subject are
derived from criminal conduct or a prohibited transaction.
4] Record on Financial
Crime :
Charges or conviction
registered against subject: None
5] Records on Violation of
Anti-Corruption Laws :
Charges or investigation
registered against subject: None
6] Records on Int’l
Anti-Money Laundering Laws/Standards :
Charges or
investigation registered against subject: None
7] Criminal Records
No
available information exist that suggest that subject or any of its principals
have been formally charged or convicted by a competent governmental authority
for any financial crime or under any formal investigation by a competent
government authority for any violation of anti-corruption laws or international
anti-money laundering laws or standard.
8] Affiliation with
Government :
No record
exists to suggest that any director or indirect owners, controlling
shareholders, director, officer or employee of the company is a government
official or a family member or close business associate of a Government
official.
9] Compensation Package :
Our market
survey revealed that the amount of compensation sought by the subject is fair
and reasonable and comparable to compensation paid to others for similar
services.
10] Press Report :
No press reports / filings exists on
the subject.
CORPORATE GOVERNANCE
MIRA INFORM as part of its Due Diligence do provide comments on
Corporate Governance to identify management and governance. These factors often
have been predictive and in some cases have created vulnerabilities to credit
deterioration.
Our Governance Assessment focuses principally on the interactions
between a company’s management, its Board of Directors, Shareholders and other
financial stakeholders.
CONTRAVENTION
Subject is not known to have contravened any existing local laws,
regulations or policies that prohibit, restrict or otherwise affect the terms
and conditions that could be included in the agreement with the subject.
FOREIGN EXCHANGE RATES
|
Currency |
Unit
|
Indian Rupees |
|
US Dollar |
1 |
Rs. 59.03 |
|
|
1 |
Rs. 98.91 |
|
Euro |
1 |
Rs. 80.33 |
INFORMATION DETAILS
|
Information
Gathered by : |
PRT |
|
|
|
|
Analysis Done by
: |
KAR |
|
|
|
|
Report Prepared
by : |
DPH |
SCORE & RATING EXPLANATIONS
|
SCORE FACTORS |
RANGE |
POINTS |
|
HISTORY |
1~10 |
8 |
|
PAID-UP CAPITAL |
1~10 |
7 |
|
OPERATING SCALE |
1~10 |
8 |
|
FINANCIAL CONDITION |
|
|
|
--BUSINESS SCALE |
1~10 |
8 |
|
--PROFITABILIRY |
1~10 |
8 |
|
--LIQUIDITY |
1~10 |
7 |
|
--LEVERAGE |
1~10 |
7 |
|
--RESERVES |
1~10 |
7 |
|
--CREDIT LINES |
1~10 |
8 |
|
--MARGINS |
-5~5 |
-- |
|
DEMERIT POINTS |
|
|
|
--BANK CHARGES |
YES/NO |
YES |
|
--LITIGATION |
YES/NO |
YES |
|
--OTHER ADVERSE INFORMATION |
YES/NO |
NO |
|
MERIT POINTS |
|
|
|
--SOLE DISTRIBUTORSHIP |
YES/NO |
NO |
|
--EXPORT ACTIVITIES |
YES/NO |
NO |
|
--AFFILIATION |
YES/NO |
YES |
|
--LISTED |
YES/NO |
YES |
|
--OTHER MERIT FACTORS |
YES/NO |
YES |
|
DEFAULTER |
|
|
|
--RBI |
YES/NO |
NO |
|
--EPF |
YES/NO |
NO |
|
TOTAL |
|
68 |
This score serves as a reference to assess
SC’s credit risk and to set the amount of credit to be extended. It is
calculated from a composite of weighted scores obtained from each of the major
sections of this report. The assessed factors and their relative weights (as
indicated through %) are as follows:
Financial
condition (40%) Ownership
background (20%) Payment
record (10%)
Credit history
(10%) Market trend (10%) Operational size
(10%)
RATING EXPLANATIONS
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
>86 |
Aaa |
Possesses an extremely sound financial base with the strongest
capability for timely payment of interest and principal sums |
Unlimited |
|
71-85 |
Aa |
Possesses adequate working capital. No caution needed for credit
transaction. It has above average (strong) capability for payment of interest
and principal sums |
Large |
|
56-70 |
A |
Financial & operational base are regarded healthy. General
unfavourable factors will not cause fatal effect. Satisfactory capability for
payment of interest and principal sums |
Fairly Large |
|
41-55 |
Ba |
Overall operation is considered normal. Capable to meet normal
commitments. |
Satisfactory |
|
26-40 |
B |
Capability to overcome financial difficulties seems comparatively
below average. |
Small |
|
11-25 |
Ca |
Adverse factors are apparent. Repayment of interest and principal sums
in default or expected to be in default upon maturity |
Limited with
full security |
|
<10 |
C |
Absolute credit risk exists. Caution needed to be exercised |
Credit not
recommended |
|
-- |
NB |
New Business |
-- |
This report is issued at your request without any
risk and responsibility on the part of MIRA INFORM PRIVATE LIMITED (MIPL) or
its officials.