MIRA INFORM REPORT
|
Report Date : |
12.06.2014 |
IDENTIFICATION DETAILS
|
Name : |
JUBILANT LIFE SCIENCES LIMITED (w.e.f.01.10.2010) |
|
|
|
|
Formerly Known
As : |
JUBILANT ORGANOSYS LIMITED |
|
|
|
|
Registered
Office : |
Bhartiagram, Gajraula, Jyotiba Phoolay Nagar – 244223, Uttar Pradesh |
|
|
|
|
Country : |
India |
|
|
|
|
Financials (as
on) : |
31.03.2013 |
|
|
|
|
Date of
Incorporation : |
21.06.1978 |
|
|
|
|
Com. Reg. No.: |
20-004624 |
|
|
|
|
Capital
Investment / Paid-up Capital : |
Rs.159.300
Millions |
|
|
|
|
CIN No.: [Company Identification
No.] |
L24116UP1978PLC004624 |
|
|
|
|
TAN No.: [Tax Deduction &
Collection Account No.] |
MRTJ00275C |
|
|
|
|
PAN No.: [Permanent Account No.] |
AABCV0200H |
|
|
|
|
Legal Form : |
A Public Limited Liability Company. The Company’s Shares are Listed on
the Stock Exchanges. |
|
|
|
|
Line of Business
: |
Subject is a global
Pharmaceutical and Life Sciences player engaged in manufacture and supply of
Generics (Including Active Pharmaceuticals Ingredients (APIs) and Solid
Dosage Formulations) and Life Science Ingredients (Including Proprietary
Products and Exclusive Synthesis, Nutrition Ingredients and Life Sciences
Chemicals). |
|
|
|
|
No. of Employees
: |
6223 (Approximately) |
RATING & COMMENTS
|
MIRA’s Rating : |
Ba (54) |
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
41-55 |
Ba |
Overall operation is considered normal. Capable to meet normal
commitments. |
Satisfactory |
|
Status : |
Satisfactory |
|
|
|
|
Payment Behaviour : |
Regular |
|
|
|
|
Litigation : |
Clear |
|
|
|
|
Comments : |
Subject is a well-established integrated pharmaceutical and life
sciences company with presence in India, North America, Europe and China
having satisfactory track. The management has reported a consecutive loss for past two years due
to which the position of the reserves has seen a slight deterioration. However, the company possesses a healthy business profile, supported
by its strong domestic market position in custom research and manufacturing
services. The ratings also take into consideration adequate liquidity along with
increasing cash balance during 2013. Trade relations are fair. Business is active. Payment terms are
reported as regular and as per commitment. In view of leadership position in niche products, the subject can be considered
normal for business dealings at usual trade terms and conditions. |
NOTES:
Any query related to this report can be made
on e-mail : infodept@mirainform.com
while quoting report number, name and date.
ECGC Country Risk Classification List – March 31, 2014
|
Country Name |
Previous Rating (31.12.2013) |
Current Rating (31.03.2014) |
|
India |
A1 |
A1 |
|
Risk Category |
ECGC
Classification |
|
Insignificant |
A1 |
|
Low |
A2 |
|
Moderate |
B1 |
|
High |
B2 |
|
Very High |
C1 |
|
Restricted |
C2 |
|
Off-credit |
D |
INDIAN ECONOMIC OVERVIEW
US investment bank
Goldman Sachs has upgraded its outlook on Indian markets as it expects
positive impact of the election cycle.
India’s economy may grow
4.7 % in the current financial year, lower than the official estimate of 4.9 %,
Fitch Rating said. The global rating agency expects the economy to pick up in
the next two financial years.
Global ratings
agency Standard & Poor said increasing focus by India Inc on lowering debt
is likely to improve their credit profiles.
Singapore (1.1
million Indian tourists in 2012), Thailand (one million), the United Arab
Emirates ().98 million) and Malaysia ().82 million) emerged as the preferred
holidays hotspots for Indians. The total figure is expected to increase to 1.93
million by 2017, according to the latest Eurmonitor international report.
There is a $29.34 bn
outward foreign direct investment by domestic companies between April and
January of 2013/14 which has seen some signs of recovery according to a Care
Ratings report.
There are 264 number
of new companies being set up every day on average during 2014. Most of them
are registered in Mumbai. India had 1.38 million registered companies at the
end of January, 2014.
Twitter like
messaging service Weibo Corporation has filed to raise $ 500 million via a US
initial public offering. Alibaba, which owns a stake in Weibo is expected to
raise about $ 15 billion New York this year in the highest profile Internet IPO
since Facebook’s in 2012.
Bharti Airtel has
raised Rs.2,453.2 crore (350 million Swiss Francs) by selling six-year bonds at
a coupon rate of three per cent and maturing in 2020. This is the largest ever
bond offering by an Indian company in Swiss Francs. Bharat Petroleum
Corporation raised 175 million Swiss Francs by selling five year bonds at 2.98
% coupon rate in February.
Indian Oil
Corporation plans to invest Rs 7650 crore in setting up a petrochemical complex
at its almost complete Paradip refinery in Odhisha in three to four years. The
company board is set to consider the setting up of a 700000 tonne per annum
polypropylene plant at an estimated cost at Rs.3150 crore.
Global chief
information officers at gathering in Bangalore in April to meet Indian startups
at an event called Tech50 Watchout for Little Eye Labs-Facebook type deals in
the making.
EXTERNAL AGENCY RATING
|
Rating Agency Name |
INDIA RATINGS AND RESEARCH |
|
Rating |
Long Term Rating=A+ |
|
Rating Explanation |
Adequate degree of safety and low credit
risk. |
|
Date |
20.01.2014 |
|
Rating Agency Name |
INDIA RATINGS AND RESEARCH |
|
Rating |
Short Term Rating=A1+ |
|
Rating Explanation |
Very strong degree of safety and lowest
credit risk. |
|
Date |
20.01.2014 |
RBI DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available RBI Defaulters’ list.
EPF (Employee Provident Fund) DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter in
the publicly available EPF (Employee Provident Fund) Defaulters’ list as of
31-03-2012.
INFORMATION DENIED
Management Non Co-operative (91-120-4361000)
LOCATIONS
|
Registered Office/ Factory 1 : |
Bhartiagram, Gajraula, Jyotiba Phoolay Nagar – 244223, Uttar Pradesh,
India |
|
Tel. No.: |
91-5924-252351-60 |
|
Fax No.: |
91-5924-252352 |
|
E-Mail : |
|
|
Website : |
|
|
|
|
|
Corporate Office : |
Plot No.1A, Sector – 16-A, Noida–201301, Uttar Pradesh, India. |
|
Tel. No.: |
91-120-4361000 |
|
Fax No.: |
91-120-4234881/ 84/ 85/ 87/ 95/ 96 |
|
E-Mail : |
|
|
|
|
|
Mumbai Office: |
|
|
|
|
|
Factory 2 : |
Village
Nimbut, Near Nira Railway Station, District Pune, |
|
Tel. No.: |
91-2112-269155-57 |
|
Fax No.: |
91-2112-269154 |
|
|
|
|
Factory 3 : |
Block
No. 133, P. O. Samlaya, Savli Jarod Road, Taluka Savli, Vadodara–391520,
Gujarat, India |
|
Tel. No.: |
91-2667-251361/
251563-4 |
|
Fax No.: |
91-2667-251305 |
|
|
|
|
Factory 4 : |
Village
Sikanderpur Bhainswal Bhagwanpur, Roorkee, District Haridwar, Uttrakhand,
India |
|
Tel. No.: |
91-332-235161-66 |
|
Fax No.: |
91-332-235169 |
|
|
|
|
Factory 5 : |
N-34,
MIDC, Anand Nagar, Ambernath - 421506, Maharashtra, India |
|
Tel. No.: |
91-251-2620437/
438 |
|
Fax No.: |
91-251-2620439 |
|
|
|
|
Factory 6 : |
SEZ,
Bharuch Plot No.5, Vilayat GIDC, Taluka Vagra, Bharuch – 392012,
Gujarat, India |
|
Tel. No.: |
91-2641-281500 (30 Lines) |
|
Fax No.: |
91-2641-281515 |
|
|
|
|
International Manufacturing
Facilities : |
Add : Jubilant DraxImage, Inc 16751, Tel. : 91-514- 630 7030 Fax : 91-514 -694 9295 Add : Jubilant HollisterStier LLC 3525, N. Regal, Tel. : 91- 509 -489 5656 Fax : 91- 509 -484 4320 Add : Jubilant Cadista Pharmaceuticals Tel. : 91- 410- 860 8500 Fax : 91- 410- 860 8719
|
|
|
|
|
R and D
Facilities : |
Located at:
UTTAR PRADESH (CENTRAL R&D) · C-26, Sector 59, Noida - 201 301, India · D-12, Sector 59, Noida - 201 301, India · C- 46, Sector 62, Noida - 201 301, India UTTAR PRADESH (GAJRAULA R&D) Bhartiagram, Gajraula - 244 223, District Jyotiba Phoolay Nagar, India KARNATAKA (NANJANGUD R&D) 51-56, KIADB Industrial Area, Nanjangud, District Mysore - 571 302, India GUJARAT (SAVLI R&D) Block 133, Village Samalaya, Taluka Savli, District Vadodara - 391 520, India |
|
|
|
|
Branch Offices :
|
Located at: ·
Lucknow · Chennai · Hyderabad · Bangalore · Mumbai · Pune ·
Ahmedabad ·
Kolkata |
DIRECTORS
AS ON 31.03.2013
|
Name : |
Mr. Shyam S Bhartia |
|
Designation : |
Chairman and Managing Director |
|
|
|
|
Name : |
Mr. Hari S Bhartia |
|
Designation : |
Co-Chairman and Managing Director |
|
|
|
|
Name : |
Mr. Shyamsundar Bang |
|
Designation : |
Executive Director (Manufacturing and Supply Chain Operations) |
|
Qualification : |
M. Tech (Chem Engg.) |
|
|
|
|
Name : |
Mr. Abhay Havaldar |
|
Designation : |
Director |
|
|
|
|
Name : |
Mr. Shardul S Shroff |
|
Designation : |
Director |
|
|
|
|
Name : |
Dr. Inder Mohan Verma |
|
Designation : |
Director |
|
|
|
|
Name : |
Mr. Suresh Kumar |
|
Designation : |
Director |
|
|
|
|
Name : |
Mr. S Sridhar |
|
Designation : |
Director |
KEY EXECUTIVES
|
Name : |
Mr. Lalit Jain |
|
Designation : |
Company Secretary |
|
|
|
|
Name : |
Mr. Pramod Yadav |
|
Designation : |
CEO [Advance Intermediates and Nutritional Products] |
|
|
|
|
Name : |
Mr. Rajesh Srivastava |
|
Designation : |
CEO [Fine Chemicals and CRAMS] |
|
|
|
|
Name : |
Mr. Neeraj Agrawal |
|
Designation : |
CEO [Generics] |
|
|
|
|
Name : |
Mr. Marcelo Morales |
|
Designation : |
CEO [Contract Manufacturing and Services, Jubilant HollisterStier] |
|
|
|
|
Name : |
Mr. Scott Delaney |
|
Designation : |
CEO [Jubilant Cadista] |
|
|
|
|
Name : |
Mr. Chandan Singh |
|
Designation : |
President [Acetyls and Ethanol] |
|
|
|
|
Name : |
Mr. Martyn Coombs |
|
Designation : |
President [Jubilant DraxImage] |
|
|
|
|
Name : |
Mr. Kevin Garrity |
|
Designation : |
President [Allergy Business] |
|
|
|
|
Name : |
Dr. Vijayesh Kumar Gupta |
|
Designation : |
President [Branded Generics – India] |
|
|
|
|
Name : |
Dr. Subir Kumar Basak |
|
Designation : |
President [Jubilant Drug Discovery Services (Jubilant Biosys and
Jubilant Chemsys)] |
|
|
|
|
Name : |
Mr. Nayan Nanavati |
|
Designation : |
CEO [Jubilant Clinsys] |
|
|
|
|
Name : |
Dr. Ashutosh Agarwal |
|
Designation : |
Chief Scientific Officer [Chemicals and Life Science Ingredients] |
|
|
|
|
Name : |
Dr. Goutam Muhuri |
|
Designation : |
President [RandD - Dosage Forms] |
MAJOR SHAREHOLDERS / SHAREHOLDING PATTERN
AS ON 31.03.2014
|
Category of
Shareholder |
Total No. of Shares |
As a % |
|
(A) Shareholding of Promoter and Promoter Group |
|
|
|
|
|
|
|
Individuals / Hindu Undivided Family |
1903435 |
1.20 |
|
|
78577176 |
49.33 |
|
|
80480611 |
50.53 |
|
|
|
|
|
|
5570445 |
3.50 |
|
|
5570445 |
3.50 |
|
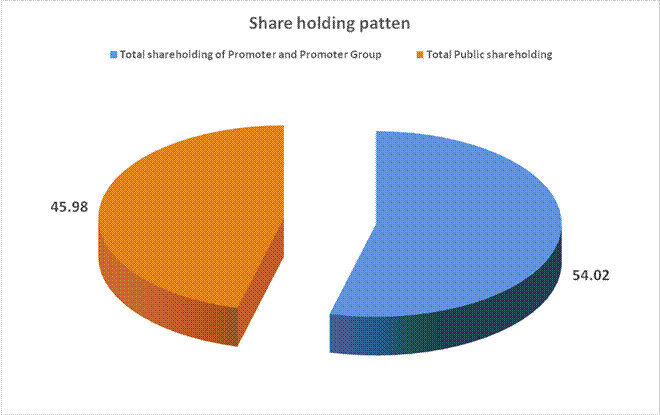
Total shareholding of Promoter and Promoter Group (A) |
86051056 |
54.02 |
|
(B) Public Shareholding |
|
|
|
|
|
|
|
|
2294538 |
1.44 |
|
|
1130204 |
0.71 |
|
|
27592231 |
17.32 |
|
|
11707200 |
7.35 |
|
|
11707200 |
7.35 |
|
|
42724173 |
26.82 |
|
|
|
|
|
|
10187881 |
6.40 |
|
|
|
|
|
|
13420931 |
8.43 |
|
|
870562 |
0.55 |
|
|
6026536 |
3.78 |
|
|
951491 |
0.60 |
|
|
5075045 |
3.19 |
|
|
30505910 |
19.15 |
|
Total Public shareholding (B) |
73230083 |
45.98 |
|
Total (A)+(B) |
159281139 |
100.00 |
|
(C) Shares held by Custodians and against which Depository
Receipts have been issued |
0 |
0.00 |
|
|
0 |
0.00 |
|
|
0 |
0.00 |
|
|
0 |
0.00 |
|
Total (A)+(B)+(C) |
159281139 |
0.00 |
BUSINESS DETAILS
|
Line of Business : |
Subject is a
global Pharmaceutical and Life Sciences player engaged in manufacture and
supply of Generics (Including Active Pharmaceuticals Ingredients (APIs) and
Solid Dosage Formulations) and Life Science Ingredients (Including
Proprietary Products and Exclusive Synthesis, Nutrition Ingredients and Life
Sciences Chemicals). |
||||||||
|
|
|
||||||||
|
Products/ Services : |
|
PRODUCTION STATUS (AS ON 31.03.2011)
|
Particulars |
Unit |
Installed
Capacity |
Actual
Production |
|
Alcohol |
KBL |
161000 |
23278 |
|
Organic including Specialty Chemicals and its
Intermediates |
MT |
656001 |
314727 |
|
Dry and Acqueous Choline
Chloride and Ethyoxylates |
MT |
21604 |
15246 |
|
Feed Premixes |
MT |
1800 |
1777 |
|
Active Pharmaceuticals Ingredients [API] |
MT |
680 |
414 |
|
Tablets and Capsules |
No. in Millions |
891 |
75 |
GENERAL INFORMATION
|
No. of Employees : |
6223 (Approximately) |
||||||||||||||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||||||||||||||
|
Bankers : |
· Central Bank of India · Corporation Bank · Export Import Bank of India · ICICI Bank Limited · ING Vysya Bank Limited · Punjab National Bank · State Bank of India · The Hong Kong and Shanghai Bank Corporation Limited ·
Yes Bank Limited |
||||||||||||||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||||||||||||||
|
Facilities : |
|
||||||||||||||||||||||||||||||||||||
|
|
|
|
Banking
Relations : |
-- |
|
|
|
|
Statutory Auditors : |
|
|
Name : |
K. N. Gutgutia and Company Chartered Accountants |
|
Address : |
11K, Gopala Tower,
25, Rajendra Place, New Delhi - 110008, India |
|
|
|
|
IFRS Auditors : |
|
|
Name : |
KPMG |
|
Address : |
Building No. 10,
8th Floor, Tower B, DLF Cyber City, Phase-II, Gurgaon - 122002 Haryana, India |
|
|
|
|
Cost Auditors : |
|
|
Name : |
J K Kabra and Company Cost Accountants |
|
Address : |
552/1B, Arjun
Street, Main Vishwas Road, Vishwas Nagar, Delhi - 110032, India |
|
|
|
|
Internal Auditors : |
|
|
Name : |
Ernst and Young Private Limited Chartered Accountants |
|
Address : |
Hindustan Times
Building, 6th Floor, 18-20, Kasturba Gandhi Marg, New Delhi - 110001, India |
|
|
|
|
Subsidiaries
including Step-down subsidiaries: |
· Jubilant Pharma Pte Limited · Draximage Limited Cyprus · Draximage Limited, Ireland · Draximage LLC · Jubilant DraxImage (USA) Inc. · Deprenyl Inc., USA · Jubilant DraxImage Inc. · 6963196 Canada Inc. · 6981364 Canada Inc · DAHI Animal Health (UK) Limited · Draximage (UK) Limited · Jubilant Life Sciences Holdings Inc. · Jubilant Clinsys Inc. · Cadista Holdings Inc. · Jubilant Cadista Pharmaceuticals Inc. · Jubilant Life Sciences International Pte. Limited · HSL Holdings Inc. · Jubilant HollisterStier LLC · Jubilant Life Sciences (Shanghai) Limited · Jubilant Pharma NV · Jubilant Pharmaceuticals NV · PSI Supply NV · Jubilant Life Sciences (USA) Inc. · Jubilant Life Sciences (BVI) Limited · Jubilant Biosys (BVI) Limited · Jubilant Biosys (Singapore) Pte. Limited · Jubilant Biosys Limited, · Jubilant Discovery Services, Inc. · Jubilant Drug Development Pte. Limited · Jubilant Chemsys Limited · Jubilant Clinsys Limited · Jubilant Infrastructure Limited · Jubilant First Trust Healthcare Limited · Asia Healthcare Development Limited · Jubilant Innovation (BVI) Limited · Jubilant Innovation Pte. Limited · Jubilant DraxImage Limited India · Jubilant Innovation (India) Limited · Jubilant Innovation (USA) Inc · Jubilant HollisterStier Inc. · Draxis Pharma LLC · Generic Pharmaceuticals Holdings, Inc. · Jubilant Life Sciences (Switzerland) AG · First Trust Medicare Private Limited · Jubilant Drug Discovery and Development Services Inc. · Vanthys Pharmaceutical Development Private Limited |
|
|
|
|
Other
Entities: |
· Jubilant HollisterStier General Partnership Canada, · Draximage General Partnership Canada (controlled through subsidiaries/step down subsidiaries). |
|
|
|
|
Enterprise
over which certain key management personnel have significant influence: |
· Jubilant Enpro Private Limited · Jubilant Oil and Gas Private Limited · Jubilant FoodWorks Limited · Tower Promoters Private Limited · B&M Hot Breads Private Limited · Jubilant Industries Limited · Jubilant Agri and Consumer Products Limited · Jubilant Motors Private Limited, · Jubilant Aeronautics Private Limited · Sankur Chalets Private Limited |
|
|
|
|
Others: |
· Vam Employees Provident Fund Trust · Jubilant Employee Welfare Trust · Jubilant Bhartia Foundation · Vam Officers Superannuation Fund |
CAPITAL STRUCTURE
AS ON 31.03.2013
Authorised Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
655,000,000 |
Equity Shares |
Re. 1/- each |
Rs. 655.000 Millions |
|
|
|
|
|
Issued and Subscribed Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
159,313,139 |
Equity Shares |
Re. 1/- each |
Rs. 159.310
Millions |
|
|
|
|
|
Paid-up Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
159,281,139 |
Equity Shares |
Re. 1/- each |
Rs. 159.280
Millions |
|
|
Add: Equity shares forfeited (paid up) |
|
Rs. 0.020
Million |
|
|
|
|
|
|
|
Total |
|
Rs. 159.300 Millions |
NOTES:
Paid up capital includes, 501,364, equity shares of Re.1 allotted and
issued pursuant to the Scheme of Amalgamation and Demerger, to the shareholders
of erstwhile Pace Marketing Specialities Limited for consideration other than
cash during the year 2010-11.
The Company has only one class of shares referred to as equity shares
having par value of ` 1. Each holder of equity shares is entitled to one vote
per share.
The details of shareholders holding more than 5% shares is set out
below:
|
Name of the shareholder |
31.03.2013 |
|
|
|
No of shares |
% held |
|
Jubilant Stock Holding Private Limited |
21,740,992 |
13.65% |
|
SSB Consultants and Management Services Private Limited |
21,007,665 |
13.19% |
|
HSB Corporate Consultants Private Limited |
15,824,979 |
9.94% |
|
Jubilant Capital Private Limited |
-- |
-- |
|
Jubilant Securities Private Limited |
-- |
-- |
|
GA Global Investments Limited |
11,707,200 |
7.35% |
The reconciliation of the number of shares outstanding is set out below:
|
Particulars |
31.03.2013 |
|
|
|
No of shares |
Rs. in Millions |
|
Numbers of shares at the beginning |
159,281,139 |
159.280 |
|
Numbers of shares at the end |
159,281,139 |
159.280 |
a)
114,835, equity shares of Re.1 each allotted on exercise
of the vested stock options in accordance with the terms of exercise under the
“Jubilant Employees Stock Option Plan”.
b)
Under the Jubilant Employees Stock Option 2005
Plan, as at 31st March, 2013 - 145,443 options are outstanding convertible into
727,215 shares.
c)
Under the Jubilant Employees Stock Option 2011
Plan, as at 31st March, 2013 - 1,585,055 options are outstanding convertible
into 1,585,055 shares.
In the event of liquidation of the Company, the holders of equity shares
will be entitled to receive any of the remaining assets of the Company, after
distribution of all preferential amounts. However, no such preferential amounts
exist currently. The distribution will be in proportion to the number of equity
shares held by the shareholders.
The Company declares and pays dividends in Indian rupees. The dividend
proposed by the Board of Directors is subject to the approval of the
shareholders in the ensuing Annual General Meeting.
FINANCIAL DATA
[all figures are
in Rupees Millions]
ABRIDGED BALANCE
SHEET
|
SOURCES OF FUNDS |
31.03.2013 |
31.03.2012 |
31.03.2011 |
|
I.
EQUITY AND LIABILITIES |
|
|
|
|
(1)Shareholders'
Funds |
|
|
|
|
(a) Share Capital |
159.300 |
159.300 |
159.300 |
|
(b) Reserves & Surplus |
18458.910 |
19011.680 |
21246.600 |
|
(c) Money received against share warrants |
0.000 |
0.000 |
0.000 |
|
|
|
|
|
|
(2) Share
Application money pending allotment |
0.000 |
0.000 |
0.000 |
|
Total
Shareholders’ Funds (1) + (2) |
18618.210 |
19170.980 |
21405.900 |
|
|
|
|
|
|
(3) Non-Current Liabilities |
|
|
|
|
(a) long-term borrowings |
20083.830 |
24222.120 |
22390.630 |
|
(b) Deferred tax liabilities (Net) |
2419.270 |
2114.130 |
1899.020 |
|
(c)
Other long term liabilities |
0.000 |
0.000 |
0.000 |
|
(d)
long-term provisions |
2273.500 |
1568.850 |
308.760 |
|
Total
Non-current Liabilities (3) |
24776.600 |
27905.100 |
24598.410 |
|
|
|
|
|
|
(4)
Current Liabilities |
|
|
|
|
(a)
Short term borrowings |
6325.630 |
4847.320 |
7412.140 |
|
(b)
Trade payables |
9347.640 |
7067.000 |
4158.850 |
|
(c)
Other current liabilities |
4712.890 |
1867.860 |
1432.250 |
|
(d)
Short-term provisions |
1136.020 |
883.540 |
3843.330 |
|
Total
Current Liabilities (4) |
21522.180 |
14665.720 |
16846.570 |
|
|
|
|
|
|
TOTAL |
64916.990 |
61741.800 |
62850.880 |
|
|
|
|
|
|
II.
ASSETS |
|
|
|
|
(1)
Non-current assets |
|
|
|
|
(a)
Fixed Assets |
|
|
|
|
(i)
Tangible assets |
20901.220 |
17663.100 |
14917.000 |
|
(ii)
Intangible Assets |
953.620 |
737.580 |
468.130 |
|
(iii)
Capital work-in-progress |
257.600 |
2520.780 |
2694.950 |
|
(iv) Intangible assets under development |
1875.970 |
1668.800 |
1475.180 |
|
(b) Non-current
Investments |
19785.060 |
19380.240 |
18523.050 |
|
(c) Deferred tax assets
(net) |
0.000 |
0.000 |
0.000 |
|
(d) Long-term Loan
and Advances |
4532.400 |
4632.240 |
4052.250 |
|
(e)
Other Non-current assets |
2.180 |
3.700 |
0.000 |
|
Total
Non-Current Assets |
48308.050 |
46606.440 |
42130.560 |
|
|
|
|
|
|
(2)
Current assets |
|
|
|
|
(a)
Current investments |
0.000 |
217.000 |
117.500 |
|
(b)
Inventories |
6112.140 |
5933.260 |
4047.000 |
|
(c)
Trade receivables |
3932.510 |
4038.250 |
3345.170 |
|
(d)
Cash and cash equivalents |
2558.850 |
2028.270 |
9852.590 |
|
(e)
Short-term loans and advances |
3412.390 |
2749.490 |
3316.030 |
|
(f)
Other current assets |
593.050 |
169.090 |
42.030 |
|
Total
Current Assets |
16608.940 |
15135.360 |
20720.320 |
|
|
|
|
|
|
TOTAL |
64916.990 |
61741.800 |
62850.880 |
PROFIT & LOSS
ACCOUNT
|
|
PARTICULARS |
31.03.2013 |
31.03.2012 |
31.03.2011 |
|
|
|
SALES |
|
|
|
|
|
|
|
Revenue from operations (net) |
31463.030 |
26410.670 |
22084.770 |
|
|
|
Other Income |
89.160 |
89.410 |
51.450 |
|
|
|
TOTAL (A) |
31552.190 |
26500.080 |
22136.220 |
|
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
|
|
Cost of
materials consumed |
15285.770 |
12399.520 |
9014.250 |
|
|
|
Purchase of
traded goods |
1917.380 |
2436.790 |
2248.190 |
|
|
|
Other
manufacturing expenses |
5446.030 |
4618.120 |
3498.420 |
|
|
|
Employee
benefits expenses |
2486.510 |
2072.320 |
1728.110 |
|
|
|
Other expenses |
2027.780 |
1700.320 |
1449.720 |
|
|
|
Exceptional
Items |
1525.230 |
1800.840 |
45.530 |
|
|
|
Change in inventories of finished goods, work-in-progress and traded
goods |
(241.750) |
(932.610) |
(313.070) |
|
|
|
TOTAL (B) |
28446.950 |
24095.300 |
17671.150 |
|
|
|
|
|
|
|
|
Less |
PROFIT/
(LOSS) BEFORE INTEREST, TAX, DEPRECIATION AND AMORTISATION (A-B) (C) |
3105.240 |
2404.780 |
4465.070 |
|
|
|
|
|
|
|
|
|
Less |
FINANCIAL
EXPENSES (D) |
1711.660 |
1544.210 |
466.520 |
|
|
|
|
|
|
|
|
|
|
PROFIT/
(LOSS) BEFORE TAX, DEPRECIATION AND AMORTISATION (C-D) (E) |
1393.580 |
860.570 |
3998.550 |
|
|
|
|
|
|
|
|
|
Less/ Add |
DEPRECIATION/
AMORTISATION (F) |
1521.670 |
1320.030 |
999.070 |
|
|
|
|
|
|
|
|
|
|
PROFIT/ (LOSS)
BEFORE TAX (E-F) (G) |
(128.090) |
(459.460) |
2999.480 |
|
|
|
|
|
|
|
|
|
Less |
TAX (H) |
305.140 |
349.690 |
203.220 |
|
|
|
|
|
|
|
|
|
|
PROFIT/ (LOSS)
AFTER TAX (G-H) (I) |
(433.230) |
(809.150) |
2796.260 |
|
|
|
|
|
|
|
|
|
|
EARNINGS IN FOREIGN
CURRENCY |
|
|
|
|
|
|
|
Export Earnings |
18008.460 |
13576.020 |
11356.890 |
|
|
|
Other Operating Income |
77.630 |
103.130 |
34.500 |
|
|
|
Interest Income |
0.000 |
0.210 |
1.130 |
|
|
TOTAL EARNINGS |
18086.090 |
13679.360 |
11392.520 |
|
|
|
|
|
|
|
|
|
|
IMPORTS |
|
|
|
|
|
|
|
Raw Materials |
5649.480 |
4761.490 |
3987.490 |
|
|
|
Traded Goods |
1153.160 |
1729.250 |
1544.990 |
|
|
|
Stores, spares, chemicals and packing material |
335.100 |
129.340 |
192.580 |
|
|
|
Capital Goods |
99.610 |
227.100 |
158.390 |
|
|
TOTAL IMPORTS |
7237.350 |
6847.180 |
5883.450 |
|
|
|
|
|
|
|
|
|
|
Earnings/ (Loss)
Per Share (Rs.) |
(2.72) |
(5.08) |
17.56 |
|
KEY RATIOS
|
PARTICULARS |
|
31.03.2013 |
31.03.2012 |
31.03.2011 |
|
PAT / Total Income |
(%) |
(1.37)
|
(3.05)
|
12.63 |
|
|
|
|
|
|
|
Net Profit Margin (PBT/Sales) |
(%) |
(0.41)
|
(1.74)
|
13.58 |
|
|
|
|
|
|
|
Return on Total Assets (PBT/Total Assets} |
(%) |
(0.30)
|
(1.20)
|
7.47 |
|
|
|
|
|
|
|
Return on Investment (ROI) (PBT/Networth) |
|
(0.01)
|
(0.02)
|
0.14 |
|
|
|
|
|
|
|
Debt Equity Ratio (Total Debt/Networth) |
|
1.42
|
1.52 |
1.39 |
|
|
|
|
|
|
|
Current Ratio (Current Asset/Current Liability) |
|
0.77
|
1.03 |
1.23 |
FINANCIAL ANALYSIS
[all figures are
in Rupees Millions]
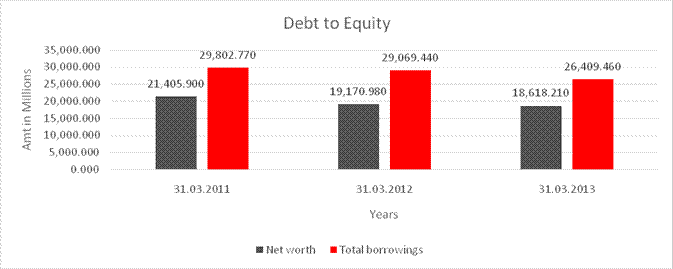
DEBT EQUITY RATIO
|
Particular |
31.03.2011 |
31.03.2012 |
31.03.2013 |
|
|
(Rs.
in Millions) |
(Rs.
in Millions) |
(Rs.
in Millions) |
|
Share Capital |
159.300 |
159.300 |
159.300 |
|
Reserves & Surplus |
21246.600 |
19011.680 |
18458.910 |
|
Net
worth |
21,405.900 |
19,170.980 |
18,618.210 |
|
|
|
|
|
|
long-term borrowings |
22390.630 |
24222.120 |
20083.830 |
|
Short term borrowings |
7412.140 |
4847.320 |
6325.630 |
|
Total
borrowings |
29,802.770 |
29,069.440 |
26,409.460 |
|
Debt/Equity
ratio |
1.392 |
1.516 |
1.418 |
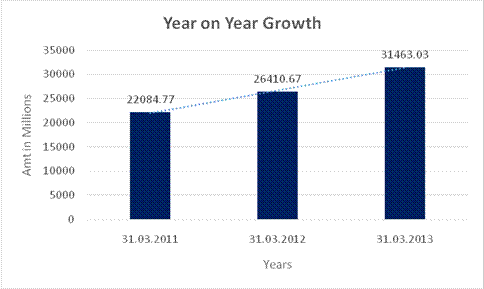
YEAR-ON-YEAR GROWTH
|
Year
on Year Growth |
31.03.2011 |
31.03.2012 |
31.03.2013 |
|
|
(Rs.
in Millions) |
(Rs.
in Millions) |
(Rs.
in Millions) |
|
Sales |
22084.770 |
26410.670 |
31463.030 |
|
|
|
19.588 |
19.130 |
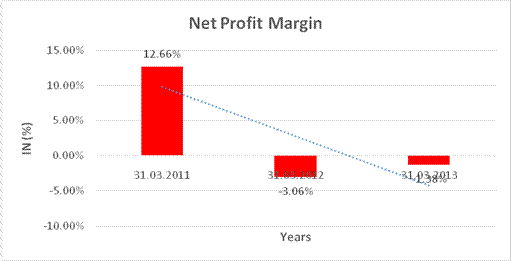
NET PROFIT MARGIN
|
Net
Profit Margin |
31.03.2011 |
31.03.2012 |
31.03.2013 |
|
|
(Rs.
in Millions) |
(Rs.
in Millions) |
(Rs.
in Millions) |
|
Sales |
22084.770 |
26410.670 |
31463.030 |
|
Profit/ (Loss) |
2796.260 |
(809.150) |
(433.230) |
|
|
12.66% |
(3.06%) |
(1.38%) |
LOCAL AGENCY FURTHER INFORMATION
|
Sr. No. |
Check List by Info Agents |
Available in
Report (Yes / No) |
|
1] |
Year of Establishment |
Yes |
|
2] |
Locality of the firm |
Yes |
|
3] |
Constitutions of the firm |
Yes |
|
4] |
Premises details |
No |
|
5] |
Type of Business |
Yes |
|
6] |
Line of Business |
Yes |
|
7] |
Promoter's background |
No |
|
8] |
No. of employees |
Yes |
|
9] |
Name of person contacted |
No |
|
10] |
Designation of contact
person |
No |
|
11] |
Turnover of firm for last
three years |
Yes |
|
12] |
Profitability for last
three years |
Yes |
|
13] |
Reasons for variation
<> 20% |
-- |
|
14] |
Estimation for coming
financial year |
No |
|
15] |
Capital in the business |
Yes |
|
16] |
Details of sister
concerns |
Yes |
|
17] |
Major suppliers |
No |
|
18] |
Major customers |
No |
|
19] |
Payments terms |
No |
|
20] |
Export / Import details
(if applicable) |
No |
|
21] |
Market information |
-- |
|
22] |
Litigations that the firm
/ promoter involved in |
-- |
|
23] |
Banking Details |
Yes |
|
24] |
Banking facility details |
Yes |
|
25] |
Conduct of the banking
account |
-- |
|
26] |
Buyer visit details |
-- |
|
27] |
Financials, if provided |
Yes |
|
28] |
Incorporation details, if
applicable |
Yes |
|
29] |
Last accounts filed at
ROC |
Yes |
|
30] |
Major Shareholders, if
available |
Yes |
|
31] |
Date of Birth of
Proprietor/Partner/Director, if available |
No |
|
32] |
PAN of Proprietor/Partner/Director,
if available |
No |
|
33] |
Voter ID No of
Proprietor/Partner/Director, if available |
No |
|
34] |
External Agency Rating,
if available |
Yes |
INDEX OF CHARGES
|
S.NO. |
CHARGE ID |
DATE OF CHARGE CREATION/MODIFICATION |
CHARGE AMOUNT SECURED |
CHARGE HOLDER |
ADDRESS |
SERVICE REQUEST NUMBER (SRN) |
|
1 |
10424562 |
25/04/2013 * |
1,600,000,000.00 |
INDIAN BANK |
47-48 PRAGATI
HOUSE, NEHRU PLACE, NEW DELHI - 110019, INDIA |
B74962739 |
|
2 |
10343200 |
25/04/2013 * |
2,504,900,000.00 |
DBS BANK LIMITED
|
ACTING ON BEHALF
OF DBS BANK LIMITED, SINGAPORE, CAPITOL POINT, BABA KHARAK SINGH MARG,
NEW DELHI - 110001, INDIA |
B74167784 |
|
3 |
10303869 |
20/03/2012 * |
2,400,000,000.00 |
CENTRAL BANK OF
INDIA |
PRESS AREA BRANCH,
LINK HOUSE, 3 BAHADUR SHAH MARG, NEW DELHI - 110002, INDIA |
B35922640 |
|
4 |
10274305 |
20/03/2012 * |
3,000,000,000.00 |
AXIS BANK
LIMITED |
B-2 & B-3,
SECTOR-16, NOIDA, NOIDA, UTTAR PRADESH - 201301, INDIA |
B35468982 |
|
5 |
10271462 |
20/03/2012 * |
1,000,000,000.00 |
CORPORATION BANK
INDUSTRIAL FINANCE BRANCH |
H.T. HOUSE, 10TH
FLOOR, K.G.MARG, CONNAUGHT PLACE, NEW DELHI - 110001, INDIA |
B35922079 |
|
6 |
10271469 |
20/03/2012 * |
2,700,000,000.00 |
CORPORATION BANK
INDUSTRIAL FINANCE BRANCH |
H.T. HOUSE, 10TH
FLOOR, K.G.MARG, CONNAUGHT PLACE, NEW DELHI - 110001, INDIA |
B35921576 |
|
7 |
10229540 |
12/07/2010 |
4,884,000,000.00 |
HOUSING
DEVELOPMENT FINANCE CORPORATION LIMITED |
RAMON HOUSE 169BACKBAY
RECLAMATION, H T PAREKH MARG, MUMBAI - 400020, MAHARASHTRA, INDIA |
A90177528 |
|
8 |
10217801 |
20/03/2012 * |
2,300,000,000.00 |
EXPORT-IMPORT
BANK OF INDIA |
CENTRE ONE
BUILDING, FLOOR 21, WORD TRADE CENTRE COMPLEX, CUFFE PARADE, MUMBAI - 400005,
MAHARASHTRA, INDIA |
B35908292 |
|
9 |
10173318 |
12/07/2010 * |
4,884,000,000.00 |
HOUSING
DEVELOPMENT FINANCE CORPORATION LIMITED |
RAMON HOUSE
169BACKBAY RECLAMATION, H T PAREKH MARG, MUMBAI - 400020, MAHARASHTRA, INDIA |
A89713697 |
|
10 |
80046092 |
20/09/2013 * |
15,000,000,000.00 |
ICICI BANK
LIMITED |
LANDMARK RACE
COURCE CIRCLE, ALKAPURI, VADODARA - 390007, GUJARAT, INDIA |
B87010690 |
* Date of charge modification
UNSECURED LOANS
|
UNSECURED LOANS |
31.03.2013 (Rs.
In Millions) |
31.03.2012 (Rs.
In Millions) |
|
SHORT TERM BORROWINGS |
|
|
|
Other working capital loans From Banks |
1514.970 |
2572.890 |
|
Loans from related parties |
522.200 |
65.000 |
|
Commercial Papers |
0.000 |
600.000 |
|
|
|
|
|
Total |
2037.170 |
3237.890 |
CORPORATE INFORMATION
Jubilant Life
Sciences Limited (the Company) is a public limited company domiciled in India
and incorporated under the provisions of Companies Act, 1956. Its shares are listed
on Bombay Stock Exchange and National Stock Exchange of India. The company is a
global Pharmaceutical and Life Sciences player engaged in manufacture and
supply of Generics (Including Active Pharmaceuticals Ingredients (APIs) and
Solid Dosage Formulations) and Life Science Ingredients (Including Proprietary
Products and Exclusive Synthesis, Nutrition Ingredients and Life Sciences
Chemicals). The company’s strength lies in its unique offerings of
Pharmaceuticals and Life Sciences products and services across the value chain.
It is well recognized as a ‘Partner of Choice’ by leading pharmaceuticals and
life sciences companies globally.
OVERVIEW
Jubilant Life Sciences Limited is a global Pharmaceutical and Life
Sciences Company engaged in manufacture and supply of Generics (including
Active Pharmaceutical Ingredients (APIs) and Solid Dosage Formulations),
Specialty Pharmaceuticals (including Radiopharmaceuticals, Allergy Therapy
Products and Sterlite Injectibles and Ointments, Creams and Liquids (OCL)) and
Life Science Ingredients (including Proprietary Products and Exclusive
Synthesis (PPES), Nutrition Ingredients (NI) and Life Science Chemicals (LSC).
It also provides Drug Discovery and Development Solutions (DDDS) and other
Healthcare services. The Company’s strength lies in its unique offerings of
Pharmaceuticals and Life Science products and services across the value chain.
With 10 worldclass manufacturing facilities in India, US and Canada and a team
of over 6,200 multicultural people across the globe, the Company is committed
to deliver value to its customers spread across 98 countries. The Company is
well recognized as a “Partner of Choice” by leading pharmaceuticals and life
sciences companies globally.
SUBSIDIARIES
As on March 31, 2013, the Company had 46 subsidiaries. Brief particulars
of principal subsidiaries are given below:
JUBILANT
HOLLISTERSTIER LLC
This subsidiary is based in Spokane, State of Washington, USA. It is a
wholly owned subsidiary of HSL Holdings Inc. It is a recognised contract
manufacturer of sterile injectables (vials and ampoules), lyophilized products,
liquid fills, biologics, suspensions, WFI/Diluents and provides a complete
range of services to support the pharmaceutical and biopharmaceutical
industries. Additionally, it is a manufacturer of allergenic extracts, targeted
primarily at treating allergies and asthma.
Its contract manufacturing capabilities include aseptic liquid fill /
finishing and lyophilization in three distinct cGMP areas designated as Small
Volume Parenteral (SVP), Small Lot Manufacturing (SLM) and Clinical Trial
Manufacturing (CTM). Its capabilities can be applied to a variety of projects
from pre-clinical through commercial scale across a multitude of dosage forms
including microspheres, suspensions, WFI/diluents, biologics (proteins),
lyophilized products and liposomes. Jubilant HollisterStier maintains an
outstanding regulatory record with the FDA (CBER and CDER), EMA and Japan’s and
Brazil’s regulatory agencies. Its contract manufacturing business serves
customers including innovators ranging from small biotechnology to large
pharmaceutical companies.
JUBILANT DRAXIMAGE
INC.
- This company is a wholly owned subsidiary of the Company through Jubilant
Pharma Pte. Limited. It deals in radiopharmaceuticals which is a niche, high
entry barrier business. DraxImage develops, manufactures and markets innovative
diagnostic imaging radiopharmaceuticals solutions and therapeutic
radiopharmaceutical products for the global market. The application of these products
extends to cardiology, thyroid uptake and scan, lung scan, kidney imaging, bone
scan etc.
This company is the major supplier of lyophilized radiopharmaceutical
kits for use with Technetium - 99m including DRAXIMAGE MAA, MDP, DTPA,
Glucoheptonte and Sestamibi. Its I-131 products are the market leaders in the
US with more than 70% market share. These 131 products are currently the major
revenue drivers. Radiopharmaceuticals are used for both therapeutic and
diagnostic molecular imaging applications to customers comprising hospitals,
imaging centres and cardiology / oncology clinics.
DraxImage also markets non-radioactive products, which are solid in
lyophilized form.
This company operates a US FDA and Health Canada approved manufacturing facility
in Montreal at Canada. It is recognised
globally for its quality and execution capabilities, strong regulatory track
record and has an established customer base comprising large innovator and
specialty pharmaceutical companies.
JUBILANT BIOSYS LIMITED – This company is
a subsidiary of the Company through Jubilant Biosys (Singapore) Pte. Limited, a
wholly owned subsidiary of the Company, which holds 66.98% of the equity of
this company. This company provides Drug Discovery Services to Global Pharmaceutical
and Biotech companies in:
- Standalone Service Model
• Functional services in area of Discovery Informatics, Structural
Biology and In Vivo and Invitro Biology and Insilico on FTE or Fee based Model
- Collaborative / Partnership Model
· Integrated discovery program across a single or a portfolio of molecules; and
· Risk / reward sharing option
- Research Funding
- Payments for scientific milestones including bonus achieved through
Discovery and Development phase
- Royalties on successful commercialisation of drug During 2012-13, this
company has:
· Continued to provide Drug Discovery Services in integrated drug discovery programmes, functional service in structural biology, High thru put screening, Insilco modeling and IN Vivo Biology and Invitro Biology;
· Expanded relationship with a couple of midsized biotech companies such as Norgine, Mnemosyne;
· Successfully engaged a number of potential big clients which once closed, would give a continued stream of revenue;
· Successfully received a developmental milestone from one of its customers named Endo Pharmaceuticals, culminating in its business model;
· Successfully delivered a number of scientific milestones in different Therapeutic areas; and
· Commenced internal research on new molecules which would be available for either partnering with existing clients or outlicencing to the interested potential clients.
JUBILANT DISCOVERY
SERVICES INC. - This Delaware based USA corporation, is a wholly owned subsidiary of
Jubilant Biosys Limited. This company apart from providing sales, marketing and
liaisoning services to Jubilant Biosys Limited for its US based customers is
also providing electrophysiology services to Jansen Pharmaceutical NV and
Mnemosyne.
During the year, this company has expanded its reach and a number of
potential clients have shown interest in this capability and has been a key
differential to the competitors.
Jubilant Discovery Services Inc. has completed an important step in the realisation
of a long term strategy to extend capabilities in prosecuting ion channel
targets and expanding the capabilities in other targets including GPCRS and
Kinases. As part of company’s strategy to extend its capabilities, company
started the ‘State of the Art Discovery Center’ in North America for ion
Channel targets. This center is enabled with comprehensive discovery biology
capabilities with a focus in Voltage gated and ligand gated ion channels, GPCRs
and Kinases.
JUBILANT CHEMSYS
LIMITED – This company is a subsidiary of the Company through Jubilant Drug
Development Pte. Limited, a wholly owned subsidiary of the Company, which holds
entire equity of this company. This company offers following services to drug
discovery companies based out of US, Europe and Japan on FullTime Equivalent
and Molecule basis:
· Discovery Chemistry Functions;
· Hit-to-Lead and Lead Optimisation;
· Medicinal Chemistry Services; and
· Scaling up from mg to kg in kilo lab and pilot plant.
It also works closely with Jubilant Biosys Limited in collaborative drug
discovery research services arena. During the year, the chemistry business has
been challenging and the same has been compounded due to certain customer
contract cancellations. The management has, however, put together a plan for
the revival of this business and expansion to other related areas such as
GMP scale up facility.
JUBILANT CLINSYS
LIMITED – This company is a subsidiary of the Company through Jubilant Drug
Development Pte. Limited, a wholly owned subsidiary of the Company, which holds
entire equity of this company. This company is a full service,
scientifically-focused contract research organisation that provides
pharmaceutical, biotechnology and medical device companies with a wide range of
services in support of Phase I-IV drug and device development. These services
range from bio-analytical, bio-equivalence and pharmacokinetic studies, all
phases of clinical trials, biostatistics, clinical data management, medical and
scientific support including medical writing, drug safety, regulatory, quality
assurance, end-to-end project management, clinical monitoring, site management,
investigator and site recruitment. This company operates an 80 bedded Clinical
Pharmacology Unit in Noida and is equipped with a bio-analytical as well as a
clinical laboratory accredited by College of American Pathologists (CAP) and
NABL, India.
During the year, this company has extended its reach to European clients
and signed contracts with NRIM (U.K) and Aristo Pharma (Germany).
JUBILANT CLINSYS
INC. – This New Jersey based USA corporation is a wholly owned subsidiary of
Jubilant Life Sciences Holdings Inc. and is a therapeutically focused full
service clinical research organisation.
This company has expertise in a wide range of highly specialised
therapeutic areas including oncology, cardiovascular, central nervous system,
respiratory, dermatology and allergy/immunology. It offers broad range of
clinical research services to pharmaceutical, biotechnology and medical device
companies in support of Phase II-IV drug and device development including
project management, clinical monitoring, scientific and medical support,
patient and investigator recruitment, site management, biostatistics, data
management, drug safety, quality assurance, regulatory affairs and medical
writing. This company expanded therapeutically, geographically and added
functional service offerings. It has operations in Bedminster, New Jersey,
Raleigh, North Carolina, Ottawa, Ontario, Canada and Dusseldorf, Germany.
JUBILANT
INFRASTRUCTURE LIMITED – This wholly owned subsidiary of the Company had
entered into a Memorandum of Understanding (MOU) with the Government of Gujarat
during the ‘Vibrant Gujarat’ conference in 2007 for development of Sector
Specific Special Economic Zone (SEZ) for Chemicals in Gujarat. About 107
hectares land was taken on lease from Gujarat Industrial Development
Corporation (GIDC) in Bharuch District, Gujarat.
This SEZ became operational last year and commenced commercial
production of Unit-1. The finished products from this facility would be fully
backward integrated and based on in-house developed innovative technologies,
making it a hub for world class quality offering value to all stakeholders.
The global scale plants of Vitamin B3 and 3-Cyanopyridine at SEZ make
the Company the largest producer of Vitamin B3 in India and second largest
globally.
During the year, the commercial production of Symtet, a crop science
ingredient for one of the world’s largest and safest low cost insecticide,
commenced in Unit-2 through an environment friendly process. This will make the
Company the world’s largest producer of the crop science ingredient for the
insecticide through green route.
JUBILANT FIRST TRUST
HEALTHCARE LIMITED – This company is a wholly owned subsidiary of the Company. 95.8% of
its capital is being held directly by the Company and 4.2% by First Trust
Medicare Private Limited. With a vision for providing quality healthcare at
affordable cost, this company has set up multi-specialty hospitals in the
district towns of West Bengal. It addresses vital disease segments like
critical care, neonatal care, high risk pregnancies, dialysis, neuro sciences,
plastic surgery to name a few. It has projected itself as a friendly
neighbourhood hospital with a firm belief in ethics.
ASIA HEALTHCARE DEVELOPMENT LIMITED - This
company is a subsidiary of the Company through Jubilant First Trust Healthcare
Limited, which holds its entire capital. This company runs a 50 bedded low cost
model multispecialty hospital in Behrampur on a Public-Private-Partnership
(PPP) with the Government of West Bengal. For years, it has been serving the
health needs and has stood as number one choice for the people of this region.
CADISTA HOLDINGS
INC. AND JUBILANT CADISTA PHARMACEUTICALS INC.
1.
Cadista Holdings Inc., a corporation incorporated
in Delaware, got registered with the Securities and Exchange Commission (SEC)
during the year ended March 31, 2012. The registration was obtained pursuant to
section 12(g) of the Securities and Exchange Act of 1934 according to which, a
company has to get registered with SEC on the number of shareholders exceeding
500. However, such registration did not constitute an offering of securities by
the Company and no fresh money was raised pursuant to such registration. The
Company, through its subsidiary, Generic Pharmaceuticals Holdings Inc., holds
82.38% of common stock of this company.
2.
Jubilant Cadista Pharmaceuticals Inc., a
corporation incorporated in Delaware, is a wholly owned subsidiary of Cadista
Holdings Inc. This company is in the business of manufacturing solid dosage
forms of generic pharmaceuticals at its US FDA approved manufacturing facility
in Salisbury, Maryland, USA. Its customer base includes all the large
wholesalers, retail and grocery chains. Besides manufacturing its own label
products, it also provides Product Development and Contract Manufacturing
services. As of March 31, 2013 there were 16 products commercialised in the US
with focus in the therapeutic areas of CVS, CNS, Anti Allergic, Steroids etc.
The company is the US market leader in 3 products and ranked in top 2 in 2
products and has a strong pipeline of product filings for future growth.
JUBILANT LIFE
SCIENCES (USA) INC. – This corporation in the State of Delaware, USA,
is a wholly owned subsidiary of the Company. It undertakes sales and
distribution of advance intermediates, nutrition ingredients, fine chemicals
and APIs in the USA.
JUBILANT LIFE
SCIENCES (SHANGHAI) LIMITED – This wholly owned subsidiary of the Company is
held through Jubilant Pharma Pte. Limited. It undertakes sales and distribution
of products in China. It is into trading of advance intermediates - pyridine
and its derivatives, vitamins, fine chemicals and crop protection chemicals. It
is catering to pharmaceutical, animal feed and agrochemical industries in
China. This subsidiary is also a sourcing hub of raw materials for the Company.
JUBILANT
PHARMACEUTICALS NV - This is a wholly owned subsidiary of the Company through Jubilant
Pharma NV, Belgium, which holds 99.8% of its shares and Jubilant Pharma Pte.
Limited which holds the balance shares, both of which are wholly owned
subsidiaries of the Company. This company is engaged in the business of
licensing of generic dosage forms providing regulatory services to generic
pharmaceutical companies and distribution of life science chemicals and
vitamins to European customers. PSI Supply NV - This is a wholly owned
subsidiary of the Company. 99.5% shares of this company are held by Jubilant
Pharma NV and balance by Jubilant Pharma Pte. Limited. This company is engaged
in the supply of generic dosage forms to European markets.
JUBILANT DRAXIMAGE
LIMITED -This is a wholly owned subsidiary of the Company through Draximage
Limited, Cyprus. This company is engaged in marketing of innovative diagnostic
imaging radiopharmaceutical solution and therapeutic radiopharmaceutical
products. It is providing wide range of radioisotopes which include Tc-99m
Generator (used in the diagnosis of Bone Cancer, Renal Imaging, Cerebral
Perfusion Imaging, Myocardial Perfusion Imaging), Thallium-201 and Iodine-131
capsules and solution for the diagnosis and treatment of Thyroid and its
related disease. It has also launched the Lyophilized kits MDP, MAA and
Sestamibi and would soon be launching DTPA. The target customers are Nuclear
Medicine physicians, Cardiologists and Oncologists of various hospitals and
imaging labs.
Other subsidiaries
as at the year end are as follows:
· First Trust Medicare Private Limited
· Jubilant Innovation (India) Limited
· Vanthys Pharmaceutical Development Private Limited
· Jubilant Innovation Pte. Limited
· Jubilant Biosys (Singapore) Pte. Limited
· Jubilant Drug Development Pte. Limited
· Jubilant Pharma Pte. Limited
· Jubilant Life Sciences International Pte. Limited
· Jubilant Innovation (BVI) Limited
· Jubilant Life Sciences (BVI) Limited
· Jubilant Biosys (BVI) Limited
· Jubilant Innovation (USA) Inc.
· Generic Pharmaceuticals Holdings, Inc.
· Jubilant Life Sciences Holdings Inc.
· HSL Holdings Inc.
· Draximage LLC.
· Jubilant DraxImage (USA) Inc.
· Deprenyl Inc., USA
· Draxis Pharma LLC
· Jubilant HollisterStier Inc.
· Draximage Limited, Cyprus
· Draximage Limited, Ireland
· Jubilant Pharma NV
· 6963196 Canada Inc.
· 6981364 Canada Inc.
· Jubilant Drug Discovery and Development Services Inc.
· DAHI Animal Health (UK) Limited
· Draximage (UK) Limited
· Jubilant Life Sciences (Switzerland) AG
AWARDS AND
ACCOLADES
During the year, various awards and accolades were received by the
Company/its management. These are:
· ‘FICCI Quality System Excellence Award 2012’ silver prize under large scale category won by Gajraula Plant, India;
· ‘AIMA Managing India Awards 2013: Entrepreneurs of the Year’ award won by Mr. Shyam S. Bhartia and Mr. Hari S. Bhartia, presented by the President of India, Mr. Pranab Mukherjee;
· NDTV Profit ‘Business Leadership Award 2012 under Corporate Social Responsibility category’ won by the Company presented by Dr. Montek Singh Ahluwalia, Deputy Chairman, Planning Commission, Government of India;
· ‘7 Star Category Certificate’ from Directorate of Industries, U.P., (valid for two years), won by Gajraula Plant, India;
· ‘ICC Award for Water Resource Management in Chemical Industry’ for the year 2011 won by Gajraula Plant, India;
· ‘The Economic Times - Frost and Sullivan India Manufacturing Excellence Gold Award – Process Sector’ for 2012 won by Gajraula Plant, India - second time in a row;
· CII ‘National Award for Excellence in Water Management 2012’ as Water Efficient Unit won by Gajraula Plant, India;
· ‘National Quality Excellence Award’ for best in Class manufacturing presented by Stars of the Industry Group, won by Gajraula Plant, India;
· ‘Golden Peacock Environment Management Award 2012’ won by Gajraula Plant, India; and
· ‘Golden Peacock Award for Sustainability 2012’ won by Gajraula Plant, India.
MANAGEMENT
DISCUSSION AND ANALYSIS
KEY ECONOMIC AND
INDUSTRY TRENDS
The world economy is once again on the growth path. With financial
conditions stabilising, there is greater hope of a sustained uptrend. The
emerging economies continue to support broader economic expansion and have
attracted robust capital inflows.
Last year has been a year of mixed trends for Pharmaceuticals and Life
Sciences companies. On one hand, these are exciting times for pharmaceutical
industry and it is on a growth path especially in emerging markets, on the
other there are challenges that the industry has to face due to patents cliff,
increased regulatory intervention and escalating healthcare costs.
The pharmaceuticals industry is riding on a growth wave in line with a
rapidly strengthening scientific base, growing demand for medicines due to
increasing and ageing global population, longer life expectancy, higher
prevalence of infectious and chronic diseases and the removal of former
impediments to free trade with the objective of providing lower cost healthcare
services and improved access for all sections of society.
Agricultural chemicals have been proven to be highly effective in
reducing crop losses caused by pests, diseases and weeds and to enable farmers
to grow crops that meet growing demand and consumer expectations at reasonable
prices.
They offer a substantial footprint in life science and pharmaceutical
products and services through their presence across the value chain, thereby
contributing to the needs of the environment, society and economy. Their
integrated operations make it feasible to deliver advantages of scale and
quality required by global clients in the chosen verticals. While the
opportunity in outsourcing is large, the requirements from products and service
solutions providers in this sector are often stringent. They continue to enjoy
a sterling reputation as a ‘Partner of Choice’ to almost all top players within
pharmaceuticals and life sciences.
Their strategy of continuously moving up the value chain into life
sciences and pharmaceuticals businesses with expanded geographic reach and
ongoing investments in RandD has yielded excellent results. This is exemplified
by their long standing relationships with 19 of the top 20 pharmaceutical and 6
of the top 10 agrochemical companies of the world. Over the years the Company
has consolidated its position and has truly transformed itself into a global
life sciences player.
THEIR BUSINESS
STRATEGY
Their strategic objective is to continue to maintain and establish
leading market positions in select key business lines to drive profitable
growth. As such, they have implemented the following core business strategies:
Global leadership in chosen lines of business and increasing market
share by continuing to grow their product portfolio - Their success is derived
from their ability to select attractive product candidates in niche markets and
to increase capacity utilisation for higher sales volume at optimum cost.
Capitalise on their strong customer relationships to creating and
pursuing growth opportunities – They believe in providing quality products with
high service levels which help in establishing long lasting relationships with
their customers. Their track record of compliance to global standards and
regulations is an important factor in obtaining timely regulatory approvals
helping in maintaining stable key accounts.
Optimise their margins while maintaining prudent financial policies - by
leveraging their existing sales capabilities and administrative functions
across an expanded revenue base, thereby gaining scale in operations. They
estimate that no major capital expenditure for their businesses is needed in
the short term as their existing capacities are sufficient to drive growth
until FY 2015 and they anticipate using their free cash flows towards overall
debt reduction.
The focus has now been on generating free cash flows from operations to
reduce the overall debt of the consolidated entity to further strengthen the
Balance Sheet. The board has formed a committee to explore various options of
raising foreign currency bonds up to US$ 250 million for the purpose of
prepayment of the existing debt and other general corporate purposes without
increasing the overall net debt levels of the company in the best interests of
the company and all its stakeholders.
I. PHARMACEUTICALS
The pharmaceuticals segment which accounted for 52% of their total
revenue from operations (net) in the FY ended March 31, 2013, had increased
revenue from operations (net) of 22% to Rs.26,580 million from Rs.21,764
million in the FY ended March 31, 2012.
ACTIVE PHARMACEUTICAL
INGREDIENTS (APIS)
Commonly known as bulk actives or bulk drugs, APIs are mixed with other
components to produce tablets, capsules or liquids. They have a clear focus on
production of APIs for Cardiovascular System (CVS) and Central Nervous System
(CNS) therapeutic areas besides few Anti-infective and Anti-ulcerants.
They are increasing their APIs product portfolio by entering first in
markets, improving their cost competitiveness through efficient manufacturing
processes and systems, accelerating Drug Master File (DMF) filings, entering
into and expanding relationships with major US, European and Indian generic
companies for sale of their APIs, and continuing to build on their previous
track record. Their APIs are exported worldwide, into emerging as well as
developed markets. Their key markets are North America, South America, Europe,
Japan, Korea, Commonwealth of Independent States (CIS) countries, the Middle
East and Australia. Their API customers are leading global generic companies.
As of March 31, 2013, they have 27 APIs available through commercial
scale plants, of which Carbamazepine, Oxcarbazepine, Citalopram, Lamotrigine,
Donepezil, Pinaverium Bromide, Meclizine and Azithromycin Monohydrate are the
most significant. They are constantly working to ensure that all plant lines
provide the desired turnover, with least downtime and optimal product mix.
They filed 50 DMFs during the year, out of which 7 were in the US, 15
across Europe, 1 Certificates of suitability to European Pharmacopoeia (CEPs),
4 in Canada and 23 in Rest of World (ROW). During the year, they launched
multiple products across regions. As of March 31, 2013, they have filed 65 DMFs
in the US, 29 CEPs in Europe, 33 in Canada, 6 in Japan and over 100 filings in
other countries.
APIs business witnessed 13% increase in revenue to Rs.5081 million in
the FY ended March 31, 2013, from Rs.4,486 million in the FY ended March 31,
2012, largely due to an increase in demand for their existing products. The
increase in sales volume revenue was partially offset by lower sales prices
achieved across most of the APIs.
To tap the opportunity of increased demand and to counter challenges
from reducing prices, they are aggressively optimising and de-bottlenecking
their operations by using existing infrastructure to maximise throughput. Their
future development will be driven by their strategic objective of focusing and
specialising in certain therapeutic areas and integrating vertically to the
formulation development, wherein lies their strength of APIs.
SOLID DOSAGE
FORMULATIONS
The Solid Dosage Formulations business is supported by their in-house
RandD facility for formulation development and regulatory filings, in-house
Clinical Research Organisation (CRO) for conducting bio-equivalence studies for
the generics RandD program and cost effective manufacturing from India, while
deriving benefit from backward integration from their API business. They focus
primarily on manufacture and sale of proprietary solid dosage formulations
including value-added formulations for CVS, CNS and Anti-allergy categories.
They are also expanding the sales reach in the United States directly to
government agencies and distributors through their subsidiary Jubilant Cadista
and continuing to award strategic licenses for their products to third parties
in various European countries with regulatory support from their subsidiaries.
As of March 31, 2013, they have 31 commercialised products across geographies
including the United States and Europe. They are one of the largest exporters
of oral solid formulations to Japan. This business develops first-to-market
generic drugs, innovative drugs, over-the-counter drugs and line extensions.
Their range of products also includes value-added formulations and special
formulations such as taste masking, flash tablets, oral dispersible forms,
chewable tablets and modified release forms.
They enjoy leadership in the US for Methylprednisolone, Terazosin and
Lamotrigine and figure among the top 3 in Meclizine, Cyclobenzaprine,
Prochlorperazin, Donepezil and HCTZ Caps. Their key strengths in Europe also
include regulatory affairs services, formulation development, licensing of
marketing authorisations in addition to supplies of Solid Dosage Formulations
to makers of generic products.
Revenues for FY 2013 witnessed 55% increase to Rs.8,315 million from
Rs.5,366 million in the FY ended March 31, 2012, driven primarily by volume
growth in existing commercial products. This was made possible by an increase
in capacity utilisation in their Roorkee plant and new product launches in
various regions during the FY ended March 31, 2013. During the year, they
launched multiple products across regions.
As of March 31, 2013, they have 58 ANDAs filings in the United States,
41 dossiers in Europe, 20 filings in Canada and over 500 filings in other
countries. 25 ANDAs in the United States and 35 dossiers in Europe have already
been approved and 33 ANDAs in the United States, 13 in Canada and 6 products in
Europe are pending approval.
While they see opportunity for growth in Canada through tie ups with
local Canadian companies for product supplies, in Europe, they are focusing on
distribution agreements and profit sharing agreements to help them get higher
penetration and an opportunity to recover development cost from licensing out,
life time business and cost sharing. They are also licensing out wherein they
invest in development of dossiers and then find customers. In South Africa,
they see opportunity by supplying to retail chains and have tied up with
various pharmacy chains and pharmaceuticals distributors. They are
out-licensing to local South African companies too. They plan to tap into
Russia and Commonwealth of Independent States by supplying to retail chains and
distributors, Out-licensing to local companies. In Ukraine, they look to
register the products in their brand names and tie up with sales and marketing
company. In Latin America, they target enhanced sales through new launches,
focus on filings in Brazil and commercialise countries like Chile, Peru and
Costa Rica for top line growth. They also plan to enter Mexico and launch
select generic products.
RADIOPHARMACEUTICALS
They offer a portfolio of products and complementary equipment in the
niche segment of nuclear imaging. The business exhibits excellent skills in R
and D, manufacturing, quality and regulatory affairs. It enjoys an established
presence in North America. Nuclear medicine imaging and therapeutic agents are
the focus of their Radiopharmaceuticals division, which develops, manufactures
and markets such products in the global marketplace. Applications for their
products include cardiology, oncology, thyroid uptake and scans, lung scans,
kidney and brain imaging and bone scans.
The products currently marketed by their Radiopharmaceuticals business
include a line of lyophilized Technetium-99m kits used in nuclear medicine
imaging procedures and a line of imaging and therapeutic products including
Sodium Iodide I-131 and Smart-Fill, a dispenser for I-131 for its therapeutic
application in the treatment of thyroid cancer. Sodium Iodide I-131 is
currently the main revenue contributor to this business segment. I-131 (used
for treatment of thyroid cancer) is the sole US FDA product in its class,
resulting in leadership for them. The diagnostic products in the portfolio
include Macro Aggregates of Albumin (MAA) used in lung imaging, Diethylene
Triamine Penta Acetic Acid (DTPA) suitable both for lung and renal imaging,
Methyl Di Phosphonate (MDP) used in bone scanning, Gluceptate used in kidney
and brain imaging and Sestamibi used in myocardial perfusion imaging. These
products are often sold along with the kit that is used to administer the
product. Products are directly retailed to radiopharmacies and hospitals with
which they have tie-ups.
Revenues in FY 2013 witnessed 26% increase in revenue to Rs.2089 million
in the FY ended March 31, 2013 from Rs.1659 million in the FY ended March 31,
2012, primarily because they launched existing products in new markets thereby
contributing to growth.
They intend to expand their range of product offerings and consolidate
their market share for Radiopharmaceuticals in North America. They are also
expanding in markets such as Europe and Asia through their collaborative and
contractual arrangements with partners and new distribution channels to drive
growth in their current and pipeline products. Their Radiopharmaceuticals
business has a number of other products in late stage development, including the
Rubidium Rb-82 Generator System, a next-generation Rubidium generator. The
system is currently under regulatory review in the United States, Europe and
Canada, and they expect to launch it subject to regulatory approval.
The launch of Ruby-fill, a paradigm changing product, will help them
become a leader in Nuclear Medicine Pet Cardiology. They see opportunity to
build machine for rapid development of generics, a center of excellence in
Montreal and to carefully forge partnerships with industry counterparts. Their
innovation endeavors continue and they have identified potential new product
targets and formulated a product launch plan upto 2020.
ALLERGY THERAPY
PRODUCTS
Their Allergy Therapy business provides products to the allergy specialty
industry with a range of over 200 different allergens and standard allergy
vaccine mixtures both in bulk and against customer prescriptions. They focus on
big 5 antigens plus skin test devices and the target user-base covers
conventional allergists, ENT, regular physicians and managed care/hospital
based clinics across the US and Canada besides other international markets.
Majority of their therapeutic and diagnostic vaccines are extracted from
pollens, animal pelt and stinging insects (venom).
Revenues in FY 2013 stood at Rs.1,767 million, up 22% from Rs.1,452
million in the previous year reflecting traction on account of their
consolidation in the North American market with the introduction of new
marketing and promotional tools.
During the year, they have brought improvements in their allergy sales
and marketing organisation. They developed and tested sales force
optimisation/profit maximisation model. They also validated research analysis
of the US Market for allergenic extracts and skin testing devices. In
international business, they completed a major supply agreement, expanded
distribution in France and other markets including India, Korea, Mexico and
Australia.
They aim to become leaders in US extract sales with improved margins by
improving operations, reducing costs, improving cash management, eliminating
product supply interruptions, increasing the sophistication of their sales and
marketing efforts, adding value to their current service offering, improving
Sales efficiency and capitalising on the emerging primary care segment. They
aim to capitalize on opportunities by establishing strategic alliances and
distributor relationships in order to expand their allergenic extracts product
portfolio, and to introduce new immunotherapy products focusing on alternative
routes of administration.
STERILE
INJECTABLES AND OCL (OINTMENTS, CREAMS AND LIQUIDS)
In Sterile Injectables and OCL, they are focused on servicing innovator
and branded pharmaceutical and biotechnology organisations, expanding their
business to include clinical trial manufacturing and sterile manufacturing
capabilities, leveraging on existing business relationships and cross-selling
opportunities within the pharmaceuticals segment.
In sterile portfolio, they offer services for a broad range of products
including Vial and Ampoule Liquid Fills, Freeze-Dried (Lyophilized)
Injectables, Biologics, Suspensions and water for Injection Diluents to
pharmaceutical companies. They are also capable of manufacturing products in
quantities suitable for clinical trials as well as for large scale commercial
requirements. The services they offer for non-sterile products include solid
oral and semi-solid dosage formulations, including antibiotic ointments,
dermatological cream and liquids (syrups and suspensions), capsules, tablets
and powder blends.
They follow a partnership approach to contract manufacturing of Sterile
Injectables and OCL where the primary clientele is innovator companies. They
are among top 5 Contract Manufacturing Outsourcing players in North America in
Sterile Injectables and have been strengthening their presence with
manufacturing facilities at two locations in US and Canada with multiple
service capabilities.
Revenues in FY 2013 stood at Rs.7,052 million with 14% growth compared
to Rs.6,211 million in the previous year excluding the onetime other operating
income of Rs.249 million in FY 2012). Backed by strong order execution, the
business exhibited improvement in revenues on a year on year basis building
superior channels for growth in the future. Focus on cost-saving helped
Jubilant to improve margins in the sterile injectibles space.
They have integrated operations at their US and Canadian facilities.
They had a successful launch of lab services across key customer segments in FY
2013. They now have a mechanism in place to monitor manufacturing effectiveness
to ensure execution as per their plans. Automation of vial inspection will
become a critical requirement from 2013 onwards. They are working towards
achieving this. The business benefits from a strong order book and there are
multiple contracts under execution for supplies to US and European geographies.
Necessary measures to improve capacity utilisation have been taken which in
turn will support growth in the future.
Their contract manufacturing facility at Canada had been issued a
warning letter by USFDA identifying violations of certain Current Good
Manufacturing Practice (cGMP) regulations. They have already carried out
certain improvements suggested by the regulators and they are continuously
engaged to provide all the necessary clarifications sought by the agency.
However, their normal operations are going on as usual though this warning
letter may have an impact on approval of any new applications.
DRUG DISCOVERY AND
DEVELOPMENT SOLUTIONS (DDDS)
They provide their discovery services such as bioinformatics (pathart),
chemoinformatics (ChemBioBase), crystallography structure directed molecular design
and information technology services; pre-clinical services such as medicinal
chemistry, analytical chemistry, custom synthesis, library design,
combinatorial and focused and lead optimisation; clinical development and
market launch services across Phase I (bioavailability studies, bioequivalence
studies, bioanalytics analysis, pharmacokinetic support, statistical support
and clinical materials management), Phase II, Phase III/ III B (clinical trial
management study feasibility, site identification, site initiation/close out
and medical monitoring) and Phase IV (data management, biostatistics, quality
assurance, regulatory affairs, drug safety, consulting services and staffing
solutions).
They also conduct collaborative and integrated drug discovery programs
under this business. The collaborative/ partnership model is an integrated
discovery program across a single or a portfolio of molecules. They share with
the collaborators the risks and rewards of the project and they receive
payments as research funding upon the achievement of agreed milestones, subject
to the fulfillment of certain criteria and also bonus amounts at each specified
stage. Continued development milestones and royalties for further development
and commercialisation of a successful molecule or portfolio of molecules may
also be agreed. Their partnering model allows collaborators to pursue
integrated pre-clinical and clinical development strategies by taking targets
and leads from research institutions, developing and taking them up to clinical
trial phase II and licensing them to large pharmaceutical companies for further
development and commercialisation. They offer an integrated play of Drug
Discovery and Development Solutions where the focus is on oncology, metabolic
disorders, pain and inflammation. There are certain projects being executed
with leading pharmaceutical companies which are complemented by delivery
capabilities across the US, European and Indian markets. The model is
inherently flexible and offers optimal solutions in terms of costing and
time-to-market.
The research facilities under the DDDS business are located in Malvern,
US and at Bengaluru and Noida in India. They also maintain a global presence in
the Clinical Trial operations and have facilities both in the US and in India
with presence in Canada as well as in Germany.
Revenues in FY 2013 stood at Rs.2,082 million from Rs.2,450 million in
the previous year. The performance in this business was muted due to decrease
in services volume, reflecting intense competition. The global pharmaceutical
industry continued to consolidate in the RandD space in the past year throwing
up challenges for the business growth.
OTHERS –
HEALTHCARE
Their Healthcare business is engaged in providing ‘Better Care at
Affordable Cost’ to the middle-income population in West Bengal. It operates
hospitals at Baharampur (with 50 beds) and at Barasat (with 120 beds), which
provide services under Neurosurgery, Neonatal and Paediatric Intensive Care.
This facility is operated by a team of full-time doctors representing major
medical disciplines. These doctors are also available on-call to extend
emergency care to patients.
Revenues in FY 2013 stood at Rs.194 million from Rs.140 million in the
previous year. We are looking at improving profitability by focusing on
enhancing productivity from current investments.
II. LIFE SCIENCE
INGREDIENTS
Their Life Science Ingredients segment comprises revenue lines of
Proprietary Products and Exclusive Synthesis, Nutrition Ingredients and Life
Science Chemicals businesses. Life Science Ingredients segment revenues
continued to chart their growth trajectory at Rs.25,030 million in FY 2013,
contributing over 48% of their total revenue mix on the back of 19% growth
year-on-year, mainly driven by volume uptick across businesses.
PROPRIETARY
PRODUCTS AND EXCLUSIVE SYNTHESIS (PPES)
Their PPES business unit develops, manufactures and provides products
which comprise basic building blocks, value-added intermediates, and
agro-actives used in the production of pharmaceutical and agrochemical and
other life science industries. Their Fine Chemicals and Crop Science Chemicals
products are mainly value-added intermediates that they develop from their
advance intermediates on the basis of customer requests.
They are focused on maintaining and enhancing their market position in
pyridine and derivatives, expanding their Fine Chemicals and Advance
Intermediate product portfolios by leveraging their capability in complex
chemical processes, and strengthening RandD and technology capabilities in
developing new value-added products through forward value added products. They
have developed manufacturing processes that enable us to produce more
efficiently.
Their Exclusive Synthesis business unit offers process research, development,
scale-up and optimisation services for intermediates of new chemical entities
and other in-market products to life science companies across the value chain.
They have the capability for seamless scale-up through their cGMP kilo lab,
pilot plant and commercial scale plants.
PPES business is a fully-integrated operation where Pyridine, Picolines
and a range of Pyridine derivatives are manufactured. The business benefits
from over 3 decades of experience in Pyridine chemistry. There is continuous development
of new products with pharmaceuticals and agrochemicals being the main focus
areas. They enjoy global leadership across range of products including
Pyridines, Beta Picolines and 14 other derivatives.
This business reported revenues of Rs.11,211 million in FY 2013 compared
to Rs.9,312 million in the previous year. The 20% sales growth that they
witnessed was primarily driven by a 14% increase in volume of PPES products
sold. Their Pyridines and Beta Picoline capacity utilisation was close to 90% in
FY 2013.
Advanced Intermediates global business is driven by dynamism of two
co-products with highly competitive market and concentrated supplier structure.
The low entrance barrier has paved way for many new Chinese players. This is
coupled with few big customers and then highly fragmented customer base. In
such a scenario, key differentiation is primarily based on manufacturing
efficiencies. The past year saw them achieve market share of over 33% for
Pyridine bases, helping them retain leadership position in the category.
They expect to maintain leadership position in Pyridine bases worldwide
by maximising co-product utilisation through supply to Vitamins business, being
the best in alpha chemistry and technological capabilities leading to be the
lowest cost manufacturer and ensuring 100% compliance to environmental measures
and Zero discharge.
They aim to become one of the Top 2 global supplier partners of chosen
Pyridine derivatives by adding new products and Advanced Intermediates,
achieving cost reduction, new product development and debottlenecking
capacities of few Fine Chemicals. They are actively trying to work with
innovator life science companies, for long term contracts of projects having
synergy with us. They will keep working on present pipeline of 14 projects and
keep developing new late phase pipeline for Exclusive Synthesis business
potential.
In Crop Science Chemicals, they have formulated a definitive plan for
their growth across regions. They plan to grow their agroactives business through
presence in critical markets of Europe, Brazil and the US to become a global
leader in the Pyridine based route of producing Chlorpyrifos by supplying
Symtet, they plan to validate the bigger size column trials for optimising
production and achieving longer batch time cycles. They also plan to ensure
global compliance for Symtet like China Registration, Evaluation, Authorisation
and Restriction of Chemical Substance notification, EU REACH and other
forthcoming regulations. They enjoy excellent relationships with the global
crop science active players.
NUTRITION
INGREDIENTS
Their Nutrition Ingredients business is a fully integrated operation and
primarily manufactures and markets Vitamin B3, which are formulated for human,
pharmacological, cosmetics and animal feed consumption, as well as choline
chloride (also referred to as Vitamin B4), an important feed additive for
poultry. Vitamin B3 is also used extensively in human nutrition such as flour
fortification, food enrichment, sports drinks, energy drinks, baby food and
multi-vitamins and in animal nutrition as feed additives for the poultry, dairy
and pork industry, and in pharmaceuticals such as diabetes and
cholesterol-related drugs in cosmetics for skin color and texture improvement
and the manufacture of other life sciences intermediates.
The biggest advantage they have is their integrated nature of
operations. Beta Picoline manufactured under the Proprietary Products is the
precursor to Niacin and Niacinamide (Vitamin B3) produced. This provides them
with the cost-advantage that is difficult for any player in the industry to
match.
Revenues in FY 2013 witnessed 26% increase to Rs.2,648 million in the FY
ended March 31, 2013 from Rs.2,108 million in the FY ended March 31, 2012,
driven by sales volume increase as a result of enhanced capacity utilisation
after the commissioning of the new facility at SEZ Bharuch in the FY ended
March 31, 2012.
This year saw consolidation at operational and management structure
level. They also divided the sales team into two teams (Straight Ingredients
and Specialty Products) to increase focus in the targeted segments. There were
a slew of such initiatives across Manufacturing, RandD, Supply Chain and Human
Resources. They are focused on improving their share in Vitamin B3 through
higher capacity utilisation. With the recent price increase announcement after
a challenged FY 2013 when prices were low, they see opportunity to benefit both
in terms of volumes and value, thereby improving their profitability. They are
working towards building a global leadership position in Vitamin B3. They plan
to capitalise on strength of Vitamin B3 positioning for the launch of new
products in Nutritional space. With their improved quality of Niacinamide,
their strategy is centered on aggressively building market share in regions
other than US, Europe and China. De-risking their business is high on agenda
and they continue to explore new products to diversify the Nutrition
Ingredients portfolio.
In Animal Nutrition business, they will focus on new product
development, new business development initiatives such as participation in
expositions and achieving optimum capacity utilisations going forward. New
launches as well as entry into new geographies for existing products will hold
the key to success of their strategy.
LIFE SCIENCE
CHEMICALS
Life Science Chemicals are organic intermediates, also known as Acetyls,
which are precursors to Advance Intermediates and Fine Chemicals used in a
range of applications such as pharmaceuticals, aromatics, adhesives, food,
packaging, beverages, crop protection chemicals, textiles and other solvents.
They produce various organic intermediates including Acetic Acid, Monochloro
Acetic Acid, Acetic Anhydride, Ethyl Acetate and Sodium Monochloracetate, which
are typically used in the manufacture of downstream products such as
pharmaceuticals, crop protection chemicals and solvents. Life Science Chemicals
is a capital intensive business in which scale of operations is imperative.
They have leadership position in Acetyls in India and sizeable presence
globally. They enjoy economies of scale. The business produces ‘Green Solvent’
Ethyl Acetate, which is being preferred by customers in all markets.
Their strength lies in some of these Acetyls being consumed by their
other business verticals in production of value added Fine Chemicals and APIs.
Strong integration and manufacturing efficiencies have helped them rank within
the top 10 in key Acetyl products across the world. They have created large
storage capacities at their plants and ports to ensure continued supplies of
feedstock to the operation and to benefit from lower feedstock prices which are
cyclical in nature, especially with respect to Ethyl Alcohol.
Revenues in FY 2013 witnessed 16% increase in Life Science Chemicals to
Rs.11,171 million in the FY ended March 31, 2013 from Rs.9,598 million in the
FY ended March 31, 2012, driven by volume growth largely as a result of more
efficient capacity utilisation.
This year saw them growing and further consolidating their presence in
Life Science Chemicals by enhancing utilisations from newly added capacities of
Ethyl Acetate and Anhydride in the previous year. They have started bringing
economies through alternate material usage and developing new suppliers for
other raw material. They successfully demonstrated developing Mundra as new
Exporting Port providing them opportunity for substantial savings in the
logistics area.
As part of their constant endeavour to enter into long term supply
contract with major ethanol suppliers in the region, they were able to develop
the same, covering more than 50% of alcohol purchase. This year they were able
to secure alcohol requirement from domestic market by avoiding high cost
imports (which were higher by Rs.7-8/lit). This resulted in positive impact on
profits. They successfully conducted pilot trials of feeding spent wash with
15% solid to bio-methanation reactor at Nira (normal spent wash has 10% soild).
This will help them implementing the initiative for reducing effluent at Nira.
In another initiative, they optimised alcohol cost by stopping slop fired
boiler for four months as purchase alcohol was cheaper compared to
manufacturing cost through this route, resulting in savings.
For them to achieve their objective of becoming a formidable Acetyls
player globally, they see opportunity by building a more robust marketing and
distribution platform to handle larger annual volumes. They are conscious of
the challenges that face them and have formulated a detailed business
development plan for the same. The risks they might face stem from changing
Acetic Acid balance in their region. They aim to increase the profitability by
utilising their existing capacity, by changing their product mix, targeting
higher share in Middle-East, African and Asian Markets by replicating their
hugely successful Europe model and looking at charting aggressive growth in
domestic market by entering untapped accounts.
BUSINESS
EXCELLENCE
In Jubilant, Business Excellence function is proactively creating the
framework for new improvement strategies which drives the competitive advantage
backed by a strong execution mechanism and capability. These improvement
strategies pertain to all three critical pillars of the organisation –
Customer, Process and People.
The continual efforts of Business Excellence function is to understand
processes and systems, model them by transfer functions and define crucial
measurements resulting in a superior co-ordination and integration of
processes. Learning, reconfiguration and transfiguration become source of
competitive advantage and can be effectively used to leverage Company’s
competitive strategy. During this journey of continual improvement, this function
has adopted various improvement methodologies in line with organisation
priorities like Six Sigma, Lean, Design for Six Sigma (DFSS), World Class
Manufacturing (WCM), Total Productivity Management (TPM), Supply Chain
Re-engineering (SCOR), Project Management (EPM), Operation Research (OR),
Business Intelligence (BI) etc. This year Business Excellence function has also
added competencies like Maynard Operation Sequencing Technique (MOST) for
manpower productivity enhancement and dynamic and steady state simulation
modelling for enhancing efficiencies of chemical processes using tools like
ASPEN and DYNOCHEM.
The scope of these improvement initiatives cover all facets of the
business like Manufacturing, Sales and Marketing, New Product Introduction
(RandD), Supply Chain, Corporate HR, Projects and other support functions which
helped creating a more efficient value chain. The Business Excellence
infrastructure element helps in creating self-driven / mission directed teams
which drive their operational area towards excellence in alignment to business
objective through right accountability and training. This sustained culture of
innovation and excellence is the result of deep commitment of the people at
Jubilant.
These varied businesses have specific challenges and require customised
innovative solutions to cater to these requirements. With the support of all
CEOs, Business Heads, Champions and urge of all Business Functions (including
foreign subsidiaries) towards internalisation of Business Excellence initiatives,
this improvement journey has gone a long way in firming the foundations for
sustained profitable growth.
The Company won award and recognition from Confederation of Indian
Industry (CII), Kaizen Institute and Frost and Sullivan for various accomplishments
in manufacturing excellence.
With respect to bringing about improvement in the Company, knowledge
based newsletters were shared across all businesses, yellow and green belt
trainings for corporate functions were undertaken covering many employees and a
Kaizen scheme was launched to generate number of ideas on cost reduction,
capacity enhancement and quality improvement.
Some of the key projects undertaken during the year are capacity
debottlenecking projects through application of Lean Six Sigma and process
simulation in APIs, Solid Dosage Formulations, Fine Chemicals and Exclusive
Synthesis business, Supply Chain reengineering solutions in the area planning
and sourcing by creating optimisation and business intelligence solutions in
the pharmaceutical businesses customer delight projects in contract
manufacturing businesses where critical cost savings and quality improvement
projects were done to resolve chronic issues using design for Six Sigma
techniques. In the last year an extensive emphasis was also created around
effluent load reduction where extensive Raffinate reduction in Advance
Intermediates, isolation of salts in APIs, reduction of fresh water usage in
Acetyls, were accomplished. Total Productive Maintenance has been taken to the
next level especially in Gajraula and Nanjangud (APIs) sites and significant
improvements have been achieved in overall asset effectiveness. More than 100
green belts completed their requirements for certification by undergoing the
Lean Six Sigma training and completing at least one Improvement project
sponsored by their Functional Head. Cash to Cash cycle time reduction and
working capital improvements were driven across all businesses by following
best in class Lean and Supply Chain practices.
STATEMENT OF UNAUDITED
STANDALONE RESULTS FOR THE
(Rs.
In Millions)
|
Particulars |
STANDALONE RESULTS |
||
|
Quarter Ended |
Year Ended |
||
|
31.03.2014 |
31.12.2013 |
31.03.2014 |
|
|
(Audited) |
(Unaudited) |
(Audited) |
|
|
PART
I |
|
|
|
|
Income
from operations |
|
|
|
|
(a) Net sales/Income from operations (Net
of excise duty) |
9599.200 |
9381.000 |
36275.100 |
|
(b) Other Operating Income |
109.300 |
126.100 |
452.800 |
|
Total
income from operations (net) |
9708.500 |
9507.100 |
36727.900 |
|
Expenses |
|
|
|
|
a)
Cost of materials consumed |
4741.000 |
4914.700 |
18330.500 |
|
b)
Purchase of traded goods |
433.100 |
460.200 |
1740.200 |
|
c)
Change in inventories of finished goods, work-in progress and traded
goods |
386.400 |
(377.400) |
(212.100) |
|
d)
Power & fuel |
870.500 |
926.600 |
3601.700 |
|
e)
Employee benefits expenses |
796.800 |
758.200 |
2982.300 |
|
f)
Depreciation and amortization expenses |
442.000 |
446.400 |
1752.900 |
|
g)
Other expenses |
1310.800 |
1119.600 |
4468.300 |
|
Total
Expenses |
8980.600 |
8248.300 |
32663.800 |
|
Profit/(Loss) from operations before other
income, finance cost and exceptional items |
727.900 |
1258.800 |
4064.100 |
|
Other Income |
81.100 |
67.000 |
298.800 |
|
Profit/(Loss) from ordinary activities
before finance costs & exceptional items |
809.000 |
1325.800 |
4352.900 |
|
Finance costs |
538.300 |
735.900 |
2775.900 |
|
Profit/(Loss) from ordinary activities
after finance costs but before exceptional items |
170.700 |
589.900 |
1587.000 |
|
Exceptional items |
(319.400) |
(63.600) |
2268.600 |
|
Profit/(Loss) from ordinary activities
before tax |
490.100 |
653.500 |
(681.600) |
|
Tax expense (Net) |
282.500 |
(991.700) |
(689.800) |
|
Net
Profit/(Loss) from ordinary activities after tax |
207.600 |
1645.200 |
8.200 |
|
Extraordinary items (net of tax expenses) |
-- |
-- |
-- |
|
Net
Profit/(Loss) for the period |
207.600 |
1645.200 |
8.200 |
|
Paid-up equity share capital (Face value per
share Re.1) |
159.300 |
159.300 |
159.300 |
|
Reserves (excluding revaluation reserve) |
|
|
17173.100 |
|
Earnings per share before and after
extraordinary items (Not annualized) |
|
|
|
|
Basic
(Rs.) |
1.30 |
10.33 |
0.05 |
|
Diluted
(Rs.) |
1.30 |
10.33 |
0.05 |
|
PART
II |
|
|
|
|
PARTICULARS
OF SHAREHOLDING |
|
|
|
|
Public
shareholding |
|
|
|
|
-
Number of shares (Re. 1 each) |
73230083 |
73355083 |
73230083 |
|
-
Percentage of shareholding |
45.98 |
46.05 |
45.98 |
|
Promoters
and promoter group shareholding |
|
|
|
|
a) Pledged/Encumbered |
|
|
|
|
-
Number of shares |
-- |
-- |
-- |
|
-
Percentage of shares (as a % of the total shareholding of promoter and
promoter group) |
-- |
-- |
-- |
|
- Percentage
of shares (as a % of the total share capital of the company) |
-- |
-- |
-- |
|
b) Non-Encumbered |
|
|
|
|
-
Number of shares |
86051056 |
85926056 |
86051056 |
|
-
Percentage of shares (as a % of the total shareholding of promoter and
promoter group) |
100.00 |
100.00 |
100.00 |
|
-
Percentage of shares (as a % of the total share capital of the
company) |
54.02 |
53.95 |
54.02 |
|
Particulars
|
31.03.2014 |
|
Investors
Complaints (Nos.) |
|
|
Pending at the beginning of the quarter |
Nil |
|
Received during the quarter |
3 |
|
Disposed of during the quarter |
3 |
|
Remaining unresolved at the end of the quarter |
Nil |
1. SEGMENT WISE REVENUE, RESULTS AND CAPITAL
EMPLOYED
|
Particulars |
STANDALONE RESULTS |
||
|
Quarter Ended |
Year Ended |
||
|
31.03.2014 |
31.12.2013 |
31.03.2014 |
|
|
(Audited) |
(Unaudited) |
(Audited) |
|
|
Segment revenue |
|
|
|
|
a. Pharmaceuticals |
2023.500 |
2021.200 |
8110.700 |
|
b. Life Sciences Ingredients |
7694.400 |
7495.200 |
28652.900 |
|
Total |
9717.900 |
9516.400 |
36763.600 |
|
Less: Inter segment revenue |
9.400 |
9.300 |
35.700 |
|
Net
Sales/Income from operations |
9708.500 |
9507.100 |
36727.900 |
|
a. Pharmaceuticals |
2023.500 |
2021.200 |
8110.700 |
|
b. Life Sciences Ingredients |
7685.000 |
7485.900 |
28617.200 |
|
Total |
9708.500 |
9507.100 |
36727.900 |
|
Segment
results (profit(+)/loss(-) before tax and interest from each segment) |
|
|
|
|
a. Pharmaceuticals |
268.500 |
570.400 |
1853.500 |
|
b.
Life Sciences Ingredients |
653.100 |
880.800 |
3119.100 |
|
Total |
921.600 |
1451.200 |
4972.600 |
|
Less: i. Interest (Finance costs) |
638.300 |
689.400 |
2775.900 |
|
ii. Other un-allocable expenditure
(including exceptional items) |
(125.700) |
128.800 |
3177.100 |
|
iii. Un-allocable Income (including
exceptional items) |
(81.100) |
(20.500) |
(298.800) |
|
Total
Profit/(Loss) before tax |
490.100 |
653.500 |
(681.600) |
|
Capital
Employed (Segment assets less Segment liabilities) |
|
|
|
|
a. Pharmaceuticals |
10906.700 |
11044.700 |
10906.700 |
|
b. Life Sciences Ingredients |
16302.300 |
15272.000 |
16302.300 |
|
Total
capital employed in segments |
27209.000 |
26316.700 |
27209.000 |
|
Add: Un-allocable corporate assets less
liabilities |
24992.200 |
22899.800 |
24992.200 |
|
Total
capital employed in the Company |
52201.200 |
49216.500 |
52201.200 |
2.
STANDALONE STATEMENT OF ASSETS AND
LIABILITIES
(Rs. In Millions)
|
SOURCES OF FUNDS |
31.03.2014 (Audited) |
|
I.
EQUITY
AND LIABILITIES |
|
|
(1)Shareholders' Funds |
|
|
(a) Share Capital |
154.500 |
|
(b) Reserves & Surplus |
17173.100 |
|
Total
Shareholders’ Funds |
17327.600 |
|
|
|
|
(2)
Non-Current Liabilities |
|
|
(a) long-term borrowings |
11410.500 |
|
(b) Deferred tax liabilities (Net) |
1734.200 |
|
(c) Other long term
liabilities |
104.100 |
|
(d) long-term
provisions |
2104.100 |
|
Total Non-current
Liabilities (3) |
15352.900 |
|
|
|
|
(3)
Current Liabilities |
|
|
(a) Short
term borrowings |
10971.200 |
|
(b) Trade
payables |
5992.400 |
|
(c) Other current
liabilities |
14682.500 |
|
(d) Short-term
provisions |
2146.000 |
|
Total Current
Liabilities (4) |
33792.100 |
|
|
|
|
TOTAL |
66472.600 |
|
|
|
|
II.
ASSETS |
|
|
(1)
Non-current assets |
|
|
(a) Fixed
Assets |
23930.800 |
|
(b) Non-current Investments |
20056.900 |
|
(c) Deferred tax assets (net) |
0.000 |
|
(d) Long-term Loan and Advances |
2989.700 |
|
(e) Other
Non-current assets |
5.000 |
|
Total Non-Current
Assets |
46982.400 |
|
|
|
|
(2)
Current assets |
|
|
(a)
Current investments |
3.700 |
|
(b)
Inventories |
7335.200 |
|
(c) Trade
receivables |
5296.000 |
|
(d) Cash
and cash equivalents |
1787.100 |
|
(e)
Short-term loans and advances |
5050.800 |
|
(f) Other
current assets |
17.400 |
|
Total
Current Assets |
19490.200 |
|
|
|
|
TOTAL |
66472.600 |
3.
The Board has recommended dividend of Rs.3 per equity shares of Re.1 fully paid up amounting to Rs.559.000 Millions, subject to
approval in the Annual General Meeting.
4.
As reported earlier,
the Company plan to consolidate its Pharmaceuticals business under its wholly
owned subsidiary Jubilant Pharma Limited Singapore (JPL) and evaluate the
option and opportunity to raise money to reduce the consolidated debt of the
Company. Accordingly, the Board in its meeting held on 4 October 2013 approved
transfer of Active Pharmaceutical Ingredients (API) and Dosage Forms business
of the Company by way of a slump sale on going concern basis and shares held by
it in Jubilant Pharma Holdings Inc USA and Jubilant Pharma NV Belgium, to a wholly
owned Indian subsidiary of JPL for a net consideration of 7 114510 lacs (net of debts). During the quarter the
shareholders of the Company have approved the aforesaid sale subject to the
concerned subsidiaries achieving financial closure to meet their obligation
under the said purchase, and authorized the Board to decide whether to make
this approval effective. JPL has received an approval from the Foreign
Investment Promotion Board in this regard and subsequent to the year end, the
board approved the hare purchase agreement between the parties fors8m mentioned sale of shares held
by the Company.
5.
The Company has applied hedge accounting in respect
of certain foreign currency transactions including forward contracts under
Accounting Standard ("AS") 30
"Financial Instruments: Recognition and measurement" and the
credit balance in Hedging Reserve (net) representing a portion of foreign
exchange loss/ gain on such transactions (after adjustment for related tax
impact) as at 31 Mac& 2014 and 2013 is 7 92 tars
and T 3542 lacs
respectively.
6.
Finance costs includes exchange difference arising
from foreign currency short-term borrowings regarded as an adjustment to
interest costs as per AS 16-"Borrowing Costs ", and is gross of credit
on the swap contracts as under.
|
Particulars |
STANDALONE RESULTS |
||
|
Quarter Ended |
Year Ended |
||
|
31.03.2014 |
31.12.2013 |
31.03.2014 |
|
|
(Audited) |
(Unaudited) |
(Audited) |
|
|
- Finance Cost net of credit on swap contracts |
536.300 |
533.200 |
2151.700 |
|
Add : Foreign Exchange differences and credit on swap contracts |
102.000 |
202.700 |
624.200 |
|
- Gross Finance Cost |
638.300 |
735.900 |
2775.900 |
7.
Exceptional items for each period presented
includes:
- Amortization of debit balance in foreign
currency Monetary Items Translation Difference Account (FCMITDA) of Rs.103.700 Millions, RS.254.000 Millions, Rs.175.600 Millions, Rs.1000.200 Millions and Rs.631.600 Millions for the
quarters ended 31 March 2014, 31 December 2013, 31 March 2013; and years ended 31 March 2014,031 March 2013; respectively,
representing exchange difference on long term foreign currency monetary
liabilities which has been used for the purpose other than acquiring fixed
assets.
- The remaining amount of exceptional items for
all periods presented primarily represents foreign exchange difference for
the period (excluding portion included in finance cost) and mark to market
gain/ loss (net of related contractual recoveries) In respect of currency
and interest rate swap contracts.
8.
Consequent to reevaluation of certain tax
provisions pertaining to earlier years (including deferred taxes), tax expense
/ (tax benefit), net of reversal
of deferred tax assets and true up of current tax, amounting to Rs.42.700 Millions, (Rs.615.200) Millions (excluding Rs.505.600 Millions towards current year
reevaluation) and (Rs.591.900 Millions)
has been recognized during the
quarter ended 31 March 2014. 31 December 2013 and Year ended 31 March 2014 respectively.
9.
Previous periods figures have been reclassified to
conform to the current period's classification.
10. The above audited
results were reviewed by the Audit Committee and approved by the Board of
Directors at its meeting held on 26 May
2014.
CONTINGENT
LIABILITIES (AS ON 31.03.2013):
A. Guarantees:
i. The Company has
given following corporate guarantee on behalf of its subsidiaries to secure
financial facilities :
|
Name of
Subsidiary |
Bank |
2013 |
2012 |
|
Jubilant
HollisterStier Inc. |
ICICI Bank Canada |
US$ 20.93 million |
US$ 37.66 million |
|
Jubilant Life
Sciences International Pte. Limited |
ICICI Bank Limited Singapore |
SGD 22.00 million |
-- |
Total effective guarantee
as on 31st March, 2013 in Rs.2098.11 million (previous year Rs.2234.09
million).
ii. Outstanding
guarantees furnished by banks on behalf of the Company/by the Company including
in respect of letters of credits is Rs.2794.99 million (Previous year
Rs.2101.03 million).
B. Claims against
Company, disputed by the Company, not acknowledged as debt:
|
Particulars |
31.03.2013 |
31.03.2012 |
|
Central Excise |
514.460 |
320.330 |
|
Customs |
31.630 |
12.590 |
|
Sales Tax |
7.080 |
45.540 |
|
Income Tax |
563.520 |
411.190 |
|
Service Tax |
175.740 |
105.830 |
|
Others |
47.030 |
41.830 |
Excluding demands
in respect of business transferred in earlier year to Jubilant Industries Limited
in terms of the scheme of demerger though the demands may be continuing in the
name of the Company.
Future cash
outflows in respect of the above matters as well as for matters listed under 33
(C) below are determinable only on receipt of judgments/decisions pending at
various stages/forums.
C. Other
contingent liabilities:
i. Liability in
respect of bills discounted with banks is Rs.1050 million (Previous year Rs.500
million).
ii. The Company’s
writ petition against the levy of transport fee by the State of Maharashtra on
consumption of rectified spirit and molasses within Nira factory has been
allowed by the Hon’ble Bombay High Court with consequential refund. The Company
has filed a refund claim for an amount of Rs.2.51 million deposited during the
period when the dispute was pending before the High Court. The total amount of
disputed transport fee is Rs.171.66 million. The State of Maharashtra has filed
a special leave petitionin the Supreme Court and has sought a stay on the
operation of the High Court order.
iii. The Company
has challenged before the Hon’ble Allahabad High Court, the increase in
denaturing fee by the State of Uttar Pradesh w.e.f 1st April, 2004 on
denaturing of rectified spirit in the Gajraula factory and the writ petition
has been admitted by the court. The Company has deposited Rs.22.52 million
under protest which is shown as deposits.
iv. Zila Panchayat
at J.P. Nagar (in respect of the Company’s Gajraula plant) served a notice
demanding a compensation of Rs.277.40 million allegedly for, percolation of
poisonous water stored in lagoons and flowing through the land of Zila
Panchayat resulting in loss of crops and cattle of the farmers and for putting
poisonous fly ash on national highway which caused loss to the health and
damages to eyes and skin of people. District Magistrate issued a recovery
certificate along with 10% collection charges inflating the demand to Rs.305.14
million. In the opinion of the Company, the Zila Panchayat has no jurisdiction
in raising this demand. The demand was challenged in Hon’ble Allahabad High
Court and the court stayed the demand till further orders.
v. The Company has
challenged, before the Hon’ble Allahabad High Court, the levy of license fees
of Rs.2.87 million by State of Uttar Pradesh, for grant of PD-2 license for
manufacture of ethyl alcohol for industrial use. The writ petition has been
admitted and is being listed for final hearing. Though the amount has been
deposited and shown as such, no provision against this has been made as the
issue is covered by the earlier favorable judgment of the Hon’ble Supreme Court
of India.
vi. The State of
Uttar Pradesh (UP) has imposed levy on import of denatured spirit into the
State of Uttar Pradesh (UP). The Company has imported denatured spirit into the
State of Uttar Pradesh and has challenged levy amounting to Rs.90 million
before Hon’ble Allahabad High Court. The writ petition has been allowed by the
High Court in favour of the Company. The State of Uttar Pradesh filed a special
leave petition (SLP) with Hon’ble Supreme Court. The SLP has been admitted but
the Hon’ble Supreme Court has declined the request of the State of Uttar
Pradesh (UP) to stay the operation of High Court Order.
vii. The Hon’ble
Supreme Court has quashed the levy of license fee by State of Uttar Pradesh on
captive consumption of denatured spirit in the Gajraula factory, and has
ordered the refund of the fee paid during the period of dispute subject to
condition that the amount has not been collected from the Company’s customers.
Further the Court has directed the State to investigate whether the Company has
collected the disputed fee from its customers to the extent bank guarantees
were furnished.
The Company is
entitled to a refund of Rs.84.06 million as the amount paid during the period
of dispute or secured by bank guarantees was not collected from its customers.
Accordingly the Company has approached the State of Uttar Pradesh for the
refund of the said amount. The amount paid has been shown as deposit.
viii. The State of
Uttar Pradesh (UP) has also levied trade tax (VAT) in addition to the
administrative fee on sale of
molasses within
the State. The Hon’ble Allahabad High Court had struck down the levy of VAT on
grounds that once an administrative fee, in the form of tax, has already been
charged under a Special Act, no Trade Tax or VAT can be charged under a general
act. The Hon’ble Supreme Court (SC) has also declined to grant stay in SLP
filed by the State of UP and the VAT authorities are currently treating
molasses as goods not subject to VAT. The Company discontinued paying VAT on
molasses purchased during FY 2011- FY 2013. The issue of applicability of VAT
on sale of molasses is pending before the Hon’ble S.C and in case it is decided
in favour of the State of UP, VAT amounting to Rs.228.40 million would become
payable on molasses purchased till FY 2013 and the net liability would be
Rs.24.70 million after adjustment of VAT credit to the extent of Rs.203.70
million.
FIXED ASSETS:
Tangible assets:
· Freehold Land
· Leasehold Land
· Buildings
· Plant and equipment
· Furniture and fixtures
· Vehicles
· Vehicles-leased
· Office equipments
·
Railway sidings
Intangible assets:
· Internally generated
· Product registration/market authorization
· Rights
·
Software
PRESS
RELEASES:
JUBILANT LIFE
SCIENCES JUMPS AFTER USFDA OKS CANADA PLANT
February 28, 2014
Jubilant Life Sciences jumped 6.39% to Rs 124 at 10:22 IST on BSE after the company said that the US drug regulator classified the company's pharmaceutical manufacturing facility at Montreal in Canada as acceptable.
Meanwhile, the BSE Sensex was up 95.45 points, or 0.45%, to 21,082.44.
On BSE, so far 75,000 shares were traded in the counter, compared with
an average volume of 75,324 shares in the past one quarter.
The stock hit a high of Rs 126 and a low of Rs 121.50 so far during the
day. The stock hit a 52-week high of Rs 190 on 12 March 2013. The stock hit a
52-week low of Rs 65.10 on 28 August 2013.
The stock had underperformed the market over the past one month till 26
February 2014, sliding 5.59% compared with the Sensex's 0.69% fall. The scrip
had also underperformed the market in past one quarter, falling 4.15% as
against Sensex's 2.75% rise.
The small-cap integrated pharmaceuticals and life sciences company has
an equity capital of Rs 159.300 Millions. Face value per share is Re 1.
Jubilant Life Sciences announced that it has received a communication
from the US Food and Drug Administration (FDA), classifying its pharmaceutical
manufacturing facility at Montreal in Canada as 'acceptable'. This resolves all
issues raised by the FDA on the facility in February 2013 and subsequent
communications, the company said.
The development follows completion of FDA's review of the company's
responses post the February letter and the subsequent re-inspection conducted
at Jubilant's Montreal facility in September, 2013. This development
successfully resolves the FDA issues at our Montreal facility, Jubilant Life
Sciences said.
In February 2013, Jubilant Life Sciences had informed that one of its
manufacturing facility, Jubilant HollisterStier General Partnership (JHS),
located at Kirkland, Quebec, Canada was issued a Warning Letter (WL) by US FDA identifying
significant violations of current Good Manufacturing Practices (cGMP)
Regulations.
Jubilant Life Sciences' consolidated net profit surged 437.39% to Rs
1434.300 Millions on 10.44% growth in total income from operations to Rs
14427.800 Millions in Q3 December 2013 over Q3 December 2012. The profit after
tax (PAT) during the quarter was boosted by exceptional items.
Consequent to re-evaluation of certain tax provisions pertaining to
earlier years (including deferred taxes), tax benefit (net of reversal of
deferred tax assets and true up of current tax) amounting to Rs 789.600
Millions and Rs 563.500 Millions has been recognized in the current quarter and
nine months ended 31 December 2013 respectively, the company said.
Amortization of debit balance in foreign currency monetary items
translation difference account (FCMITDA) of Rs 254.000 Millions, Rs 387.400
Millions, Rs 224.500 Millions, Rs 896.500 Millions, Rs 456.000 Millions and Rs
631.600 Millions for the quarter ended 31 December 2013, 30 September 2013, 31
December 2012; nine months ended 31 December 2013, 31 December 2012; and year
ended 31 March 2013 respectively, representing exchange difference on long-term
foreign currency monetary liabilities which has been used for the purpose other
than acquiring fixed assets, Jubilant Life Sciences said. The remaining amount
of exceptional items for all periods primarily represents foreign exchange
difference for the period (excluding portion included in finance cost) and mark
to market gain/loss (net of related contractual recoveries) in respect of
currency and interest rate swap contracts, the company said.
With regard to future business outlook, Jubilant Life Sciences said the
revenues and EBIDTA are expected to improve in the coming quarters led by
improved capacity utilisations in Sterile Injectables and OCL, Nutrition
Ingredients, Symtet and 3CP and backward integration of Pyridine and expansion
to newer markets. The company said it expects revenue growth due to strong
pipeline in APIs and Solid Dosage Formulations, new product launches, expansion
in newer geographies in API and Formulations, and robust order book in Sterile
Injectables.
Jubilant Life Sciences is a global pharmaceutical and life sciences
company engaged in manufacture and supply of APIs, generics, specialty
pharmaceuticals and life science ingredients. It also provides services in
contract manufacturing and drug discovery and development. The company's
strength lies in its unique offerings of pharmaceutical and life sciences products
and services across the value chain. With 10 world-class manufacturing
facilities in India, US and Canada and a team of over 6,300 multicultural
people across the globe, the company is committed to deliver value to its
customers spread across 100 countries.
JUBILANT BIOSYS ANNOUNCES EXPANSION OF DRUG
DISCOVERY ALLIANCE WITH JANSSEN PHARMACEUTICA
20 Feb 14
Jubilant Biosys, a Bengaluru based subsidiary of Jubilant Life Sciences, announced today that it has expanded its drug discovery alliance with Janssen Pharmaceutica N.V., Beerse, Belgium. The alliance was forged initially in the year 2011 and aims to deliver preclinical candidates to Janssen. The new agreement into multiple therapeutic areas has been expanded owing to Jubilant’s diversified skill sets in various therapeutic areas. Most of the research will be undertaken at Jubilant Biosys, India and some parts at Jubilant Discovery Center, USA.
Janssen
Pharmaceutica, Belgium is part of the ‘Janssen Pharmaceutical Companies of
Johnson & Johnson’. The collaboration between the two organizations has
been expanded to leverage Jubilant’s drug discovery capabilities on selected
drug discovery targets to advance programs in multiple disease areas. Jubilant
will carry out research services and will deliver preclinical candidates to
Janssen for potential development and commercialization. In addition to
research funding, Jubilant will also receive milestones and royalties should
Janssen successfully progress the assets into clinical development and commercialization.
“We are pleased to
announce the expansion of the Janssen- Jubilant collaboration and look forward
to discovering Novel Chemical Entities (NCE’s) that address the unmet medical
needs in these important disease areas", said Dr. Subir K. Basak, President, Jubilant
Biosys (Global Drug Discovery Services)”. Over the past several years, we have been strengthening our disease
franchises from a scientific perspective, and this collaboration serves as a validation
of those efforts. We look forward to a long, and mutually beneficial,
partnership.”
Jubilant provides
comprehensive drug discovery solutions from target discovery to IND in
partnership with leading healthcare companies worldwide, offering integrated
& collaborative drug discovery services across early discovery, synthetic
and medicinal chemistry including scale up and pre-clinical services. Jubilant
houses eminent scientists who carry with them rich experience from global
pharmaceutical companies and academic institutions. The company is one of the
leading service providers in Protein X-Ray crystallography in India, which in
combination with medicinal chemistry and computational chemistry provides a
powerful Structure Based Drug Design platform and a best niche platform to
build and drive Structure Activity Relationship (SAR) for various discovery
programs. Jubilant has been a partner to some of the major large and mid-size
pharma and Biotech companies.
About Jubilant Biosys Limited:
Jubilant Biosys
Limited is a Bengaluru based subsidiary of Jubilant Life Sciences Limited,
headquartered in Noida, India. Jubilant Biosys, is a leading provider of
integrated drug discovery solutions to the global pharmaceutical industry.
Biosys has pioneered the risk shared collaborative discovery model and has a
demonstrated track record of placing assets in the clinic via multiple
partnerships in the therapeutic areas of Oncology, Metabolic Disorders, Pain
& Inflammation, CNS and others.
About Jubilant Life Sciences:
Jubilant Life
Sciences Limited is a global Pharmaceutical and Life Sciences Company engaged
in manufacture and supply of APIs, Solid Dosage Formulations,
Radiopharmaceuticals, Allergy Therapy Products and Life Science Ingredients. It
also provides services in Contract Manufacturing of Sterile Injectables and
Ointments, Creams and Liquids and Drug Discovery and Development. The Company’s
strength lies in its unique offerings of Pharmaceutical and Life Sciences
products and services across the value chain. With 10 world-class manufacturing
facilities in India, US and Canada and a team of over 6300 multicultural people
across the globe, the Company is committed to deliver value to its customers
spread across over 100 countries. The Company is well recognized as a ‘Partner
of Choice’ by leading pharmaceuticals and life sciences companies globally.
About Janssen Pharmaceutica
Janssen
Pharmaceutica is a part of the worldwide Janssen Pharmaceutical Group of
Johnson and Johnson. Janssen Pharmaceutica is one of the most innovative
pharmaceutical companies in the world and the Janssen research and development
centre develops products for a wide range of disease areas, such as mental
disorders, neurological problems, infectious diseases, immunological disorders,
cancer, and cardiovascular and metabolic conditions.
JUBILANT LIFE
SCIENCES DECLARED WINNER OF 'BIO EXCELLENCE AWARD 2014' AT BANGALORE INDIA BIO
2014
21 Feb 14
Jubilant Life Sciences has been conferred with this year’s prestigious Bio-Excellence Award from the Department of IT, BT and S & T, Government of Karnataka. Jubilant was awarded under the category – BioServices.
Jubilant was chosen for this award for its exemplary services in the
Integrated Drug Discovery and functional services business, delivering
high-quality science with high efficiency for international clients that
include big pharma and small biotech. The award was received by Jubilant
Biosys, a Bengaluru based subsidiary of Jubilant Life Sciences Limited.
Conveying his pleasure on the award, Dr. Subir K.
Basak, President, Jubilant Biosys (Global Drug Discovery Services) said, ‘It is our honor
to receive the prestigious award in the category of BioServices in this highly
acclaimed event. The award is an encouragement of our work in comprehensive
drug discovery solutions, from target discovery to IND. The recognition
motivates our team of eminent scientists to deliver the best to our partners
who are large and mid-size pharma and Biotech companies.”
‘Bangalore India Bio 2014’ is India’s premier annual Biotech event
renowned for calibrating the pulse of the Biotechnology industry. The event is
organized by the Department of IT, BT and S & T, Government of Karnataka in
association with Vision Group on Biotechnology (an apex Advisory Body headed by
Dr. Kiran Mazumdar Shaw and consisting of members from both public and private
sectors). The focal theme of Bangalore India Bio 2014 was "Biotech for a
better tomorrow".
As part of the event, for the first time, Bio-Excellence Awards were
presented to biotech leaders for their outstanding achievements in the Biotech
sector. The Bio-Excellence Awardees are selected by a Jury of Industry Experts,
constituted by Association of Biotechnology Led Enterprises (ABLE). The
eminent Jury of Industry Experts selected winners who met three major criteria
of: increase in revenue, awards and achievements received by an individual/ an
institution and total number of Patents and Products.
The International Conference deliberated on issues of pressing interest
to the Biotech sector including Stem Cells and Regenerative Medicine,
Biosimilars, Personalised Medicine, Biomedical Devices and Digital Health,
Bioinformatics for Drug Discovery & Development, Key Issues in Clinical
Trials Development and Agri Biotech & Secondary Agriculture.
World Food Prize Laureate 2013 Em. Prof. Dr. Marc Van
Montagu, Founder and Chairman, Institute of Plant Biotechnology Outreach
(IPBO), Belgium; Shri S.R. Patil,
Hon’ble Minister for Information Technology, Biotechnology and Science &
Technology and Planning & Statistics, Govt. of Karnataka; Nobel
Laureate 2012 Prof. Sir John Gurdon, Kt DPhil DSc FRS,
Distinguished Group Leader, The Wellcome Trust / Cancer Research UK, The Gurdon
Institute, University of Cambridge, UK; Shri Srivatsa Krishna,
IAS, Secretary, Department of Information Technology, Biotechnology and Science
& Technology and DPAR (e-Governance), Govt. of Karnataka and Shri
S.S. Nakul, IAS, Director, IT & BT, Government of
Karnataka. The Award was received by Dr. Jayashree Aiyar, VP and Head,
Discovery Biology, Jubilant Biosys on behalf of Jubilant
Life Sciences.
About Bangalore
India Bio:
Bangalore INDIA BIO is an annual event organized by Department of IT, BT
and S & T, Government of Karnataka, under the guidance of Vision Group on
Biotechnology, an apex advisory body comprising of members from Government,
Public and Private Sectors including Industry and Academia. Since 2001,
Bangalore INDIA BIO has been instrumental in promoting the inherent strength of
the Indian Biotech Industry to the outside world and is now acknowledged as the
biggest and India’s National event on Life Sciences.
JUBILANT LIFE SCIENCES RECEIVES ANDA
APPROVALS FOR BUPROPION HYDROCHLORIDE
October 18, 2013
Jubilant Life Sciences Limited, an integrated Pharmaceuticals
and Life Sciences Company announced today that it has received Abbreviated New
Drug Application (ANDA) approvals from the US Food and Drug Administration (US
FDA) for:
Bupropion Hydrochloride
Extended-release Tablets USP (SR), 100 mg, 150 mg and 200 mg, the generic
version of GlaxoSmithKline’s antidepressant Wellbutrin SR(R).
Bupropion Hydrochloride
Extended-release Tablets USP (SR), 150 mg, the generic version
of GlaxoSmithKline’s smoking cessation aid, Zyban(R).
The current total market size for these products as per IMS
is US$ 518 Million per annum. We expect to launch these products in Q3 FY 14.
As on June 30, 2013, Jubilant Life Sciences had a total of
649 filings for formulations of which 189 have been approved in various regions
of the world. This includes 58 ANDAs filed in the U.S and 41 Dossier filings in
Europe.
About Jubilant Life Sciences
Jubilant Life Sciences Limited is a global Pharmaceutical and
Life Sciences Company engaged in manufacture and supply of APIs, Generics,
Specialty Pharmaceuticals and Life Science Ingredients. It also provides
Services in Contract Manufacturing and Drug Discovery and Development. The
Company’s strength lies in its unique offerings of Pharmaceutical and Life
Sciences products and services across the value chain. With 10 world-class
manufacturing facilities in India, US and Canada and a team of over 6,200
multicultural people across the globe, the Company is committed to deliver
value to its customers spread across 98 countries. The Company is well
recognized as a ‘Partner of Choice’ by leading pharmaceuticals and life
sciences companies globally.
Statements in this document relating to future status,
events, or circumstances, including but not limited to statements about plans
and objectives, the progress and results of research and development, potential
product characteristics and uses, product sales potential and target dates for
product launch are forward-looking statements based on estimates and the
anticipated effects of future events on current and developing circumstances.
Such statements are subject to numerous risks and uncertainties and are not
necessarily predictive of future results. Actual results may differ materially
from those anticipated in the forward-looking statements. Jubilant Life
Sciences may, from time to time, make additional written and oral forward
looking statements, including statements contained in the company’s filings
with the regulatory bodies and its reports to shareholders. The company assumes
no obligation to update forward-looking statements to reflect actual results,
changed assumptions or other factors.
JUBILANT RECEIVES
USFDA APPROVAL FOR TWO GENERIC DRUGS
New Delhi October 18, 2013
Drug firm Jubilant Life Sciences Limited today said it has received approval from the US Food and Drugs Administration (USFDA) to market generic versions of GlaxoSmithKline's anti-depressant Wellbutrin SR and smoking cessation aid, Zyban.
The abbreviated new drug applications (ANDAs) approval bupropion hydrochloride extended-release tablets, the generic version of Wellbutrin SR, is in multiple strengths of 100 mg, 150 mg and 200 mg, Jubilant Life Sciences said in a filing to the BSE.
For bupropion hydrochloride extended-release tablets, the generic version of Zyban, the approval is for 150 mg, the company added.
"We expect to launch these products in Q3 FY14," it said.
Quoting IMS data, the company said the current total market size of these
products in the US is $518 million per annum.
As on June 30, 2013, Jubilant Life Sciences had a total of 649 filings for
formulations, including 58 ANDAs filed in the US and 41 Dossier filings in
Europe.
Of these, 189 have been approved in various regions of the world, the company
said.
Shares of Jubilant Life Sciences were trading at Rs 75.25 a piece, down 0.13%
from their previous close on the BSE.
Jubilant Life stock
price
On November 26, 2013, Jubilant Life Sciences closed at Rs 122.50, up Rs 0.60, or 0.49 percent. The 52-week high of the share was Rs 248.25 and the 52-week low was Rs 65.10.
The latest book value of the company is Rs 116.89 per share. At current value, the price-to-book value of the company was 1.05.
JUBILANT LIFE SCIENCES
Noida, Tuesday October 29, 2013
The Board of Jubilant Life Sciences Limited, an integrated
pharmaceutical and life science industry player met today to approve financial
results for the quarter ended September 30, 2013.
Commenting on the Company’s performance, Mr. Shyam S Bhartia, Chairman
& Managing Director and Mr. Hari S Bhartia, Co-Chairman & Managing
Director, Jubilant Life Sciences said:
“Our performance is shaped by good volumes traction in Q2 FY2014. Growth
will accelerate in H2 on the back of product launches under Solid Dosage
Formulations, scale up in Speciality Pharmaceuticals and volume uptick visible
in Vitamins and Acetyl businesses due to higher capacity utilisation levels.
FY2014 will be marked by a strong business performance from all key business
verticals and the restructuring initiative aimed at de-leveraging the balance
sheet, which will drive a focussed reduction in debt.”
Q2 FY14 Highlights
· Revenue growth was driven by volume growth in both segments
· Consolidated revenue up 17% YoY
- Pharmaceuticals revenue up by 6% YoY
- LSI revenue up by 30% YoY
· International revenues at Rs. 10700.000 Millions, contributing 75% to the overall mix and up 21% YoY
· EBITDA margins at 19.2% and Normalized PAT margins at 4.9%
H1 FY14 Highlights
· Revenue growth on account of volume growth in both segments but prices remained under pressure for Pharmaceuticals business.
· Consolidated revenue up 13% YoY
- Pharmaceuticals revenue up by 4% YoY
- LSI revenue up by 24% YoY
· International revenues at Rs. 20690.000 Millions, contributing 74% to the overall revenues
· EBITDA margins at 18.5% and Normalized PAT margins at 4.6%
Business Performance Review
Pharmaceuticals Segment Review
In Q2 FY2014, Income from operations of the Pharma business stood at Rs.
6910.000 Millions, showing growth of 6% YoY and contributing 48% to the revenue mix. The
Pharma business EBITDA was at Rs. 1750.000 Millions with EBITDA
margins at 25.3%.
For H1 FY2014 the Income from operations stood at Rs. 13430.000 Millions, growing 4% YoY
and contributing 48% to the overall revenue mix. The segment EBITDA stood at
Rs. 3230.000 Millions with EBITDA margins at 24.1%.
Life Science Ingredients Segment Review
In Q2 FY2014, Income from operations for the Life Science Ingredients
segment was at Rs. 7440.000 Millions, thus improved by 30% YoY and having 52% of
share in total revenues. The segment EBITDA stood at Rs. 1250.000 Millions with EBITDA
margins at 16.8%.
In H1 FY2014 the Income from operations of the segment was at Rs.
14510.000 Millions, up 24% YoY and contributing 52% to the revenue mix. The segment EBITDA
came in at Rs. 2340.000 Millions with EBITDA margins at 16.1%.
Geographical Overview
Products and services of the Company reach out to clients in 99
countries of the world. International revenues contribute 75% of the revenue
mix at Rs. 10700.000 Millions with the share of regulated markets at Rs.
8650.000 Millions.
In Q2 FY2014 revenues from North America stood at 9% improved YoY at Rs.
5590.000 Millions, contributing 39% in the overall revenues; revenues from Europe and
Japan stood at Rs. 3060.000 Millions, up 28% with a share of 21% to the revenue
mix. Domestic revenues grew 9% YoY at Rs. 3660.000 Millions thus giving a 25%
share to the revenue mix. Revenues in ROW including China came in at Rs.
2050.000 Millions, better by 52%, with a 14% contribution to the revenue mix.
In H1 FY2013 the Revenue from North America was at Rs. 10790.000 Millions, up 6% with a
contribution of 39% to the revenue mix. Revenue from Europe and Japan was at
Rs. 5790.000 Millions contributing 21% to the revenue mix with a growth of 18% YoY. The
Domestic revenue was at Rs. 7240.000 Millions up 8% YoY, contributing 26% to the revenue
mix. Revenue from ROW including China was at Rs. 4110.000 Millions, up 44% and
giving 15% contribution to the revenue mix.
Corporate Actions
With the objective of consolidating the Pharmaceuticals business under
one entity and raise money to reduce the overall debt of the company, the board
has resolved to transfer the following to its wholly-owned subsidiary in
Singapore
1.
The API and Dosage Form businesses of the Company
in India by way of a slump sale on a going concern basis
2.
Also, part of the shares held by the Company
directly in US and European subsidiaries to enable the Singapore subsidiary to
hold 100% of the business in Europe and US
The consideration for above transfer is Rs 11450.000 Millions (net of debt of
Rs 5830.000 Millions) subject to the approval of the shareholders of the company and such
other approvals and permissions as may be deemed necessary. Jubilant Pharma
Limited, Singapore has received approval from the Foreign Investment Promotion
Board (FIPB) for above transfer. This will enable the Company to consolidate
its API, Solid Dosage Forms, Radiopharmaceuticals, Allergenic Extracts, Sterile
Injectibles and Ointment, Cream and Liquid business (Pharma Business) under the
Singapore subsidiary.
Outlook
The revenues and EBIDTA are expected to continue the uptrend and meet
the growth targets set for FY2014. This is on account of improved capacity
utilisations in Sterile Injctables and OCL, Nutrition Ingredients, Symtet and
3CP and backward integration of Pyridine and expansion to newer markets. We
expect the price uptrend in Pyridine and Nutrition Ingredients to continue. We
have a strong pipeline in API’s and Solid Dosage Formulations. We expect new
product launches, expansion in newer geographies and robust order book in
Sterile Injectables to improve outlook in H2FY2014, which we expect to be
better than H1 FY2014.
JUBILANT LIFE SCIENCES RECEIVES ANDA APPROVALS FOR BUPROPION
HYDROCHLORIDE
Noida (UP), India, Friday, October 18, 2013
Jubilant Life Sciences Ltd, an integrated Pharmaceuticals and Life
Sciences Company announced today that it has received Abbreviated New Drug
Application (ANDA) approvals from the US Food and Drug Administration (US FDA) for:
1.
Bupropion Hydrochloride Extended-release Tablets
USP (SR), 100 mg, 150 mg and 200 mg, the generic version of GlaxoSmithKline’s
antidepressant Wellbutrin SR(R).
2.
Bupropion Hydrochloride Extended-release Tablets
USP (SR), 150 mg, the generic version of GlaxoSmithKline’s smoking cessation aid,
Zyban(R).
The current total market size for these products as per IMS is US$ 518
Million per annum. We expect to launch these products in Q3 FY 14.
As on June 30, 2013, Jubilant
Life Sciences had a total of 649 filings for formulations of which 189 have
been approved in various regions of the world. This includes 58 ANDAs filed in
the U.S and 41 Dossier filings in Europe.
USFDA
ISSUES WARNING LETTER TO JUBILANT LIFE OVER CGMP NORMS
The US health regulator has found significant violations of
good manufacturing norms at the Indian drug company Jubilant Life Sciences' Washington facility for finished
pharmaceuticals.
The manufacturing facilities, Jubilant HollisterStier, LLC
(JHS) located at Spokane, Washington state, was inspected between April 15 and
May 10, 2013, the US Food and Drug Administration (USFDA) said in a warning letter to company's CEO Marcelo A
Morales.
Investigators from USFDA found significant violations of
current good manufacturing practice (CGMP) regulations for finished
pharmaceuticals, it added.
"The violations cause your drug products to be
adulterated within the meaning of Section 501(a)(2)(B) of the Federal Food,
Drug, and Cosmetic Act (the Act), 21 U.S.C. 351 (a)(2)(B), in that the methods
used in, or the facilities or controls used for, their manufacture, processing,
packing, or holding do not conform to, or are not operated or administered in
conformity with CGMP," USFDA said.
One of the specific violations which the investigators found
was failure to establish written procedures, that are designed to prevent microbiological
contamination of drug products purporting to be sterile, and that include
validation of all aseptic and sterilisation processes, it added.
The other violation was failure to adequately document all
work orders associated with the April 2013 shutdown and include appropriate
quality unit oversight.
"Your firm failed to assure an adequate system for
cleaning and disinfecting aseptic processing areas and equipment," USFDA
said.
"The company should take prompt action to correct the
violations cited in this letter. Failure to promptly correct these violations
may result in legal action without further notice including, without
limitation, seizure and injunction," it added.
"Within fifteen working days of receipt of this letter,
please notify this office in writing of the specific steps that you have taken
to correct and prevent the recurrence of violations, and provide copies of
supporting documentation," USFDA said.
According to the company's website, the Spokane plant contract
manufactures sterile injectables and allergenic extract.
Shares of Jubilant Life Sciences today ended at Rs 116.50 per scrip on BSE,
down 2.51% from its previous close.
JUBILANT
LIFE TO HIKE NIACIN PRICES BY 18 %
Drug
maker Jubilant Life Sciences on Thursday announced a price increase of 18 per
cent for Niacin feed-grade with immediate effect.
Jubilant
Life Sciences, ranked among the largest makers of vitamin B3 (Niacin &
Niacinamide) in the world, announced a price increase of 18 per cent for Niacin
feed-grade for non-contract customers and wherever existing contracts permit,
it said in a statement.
The
company's nutrition business is backward integrated up to the initial raw
aterial stage with global scale of operations.
Jubilant
Life Sciences is engaged in manufacturing and supply of Active Pharmaceutical
Ingredients (APIs), solid dosage formulations, radio pharmaceuticals, allergy
therapy products and life science ingredients.
It also
provides services in contract manufacturing of sterile injectables and
ointments, creams and liquids and drug discovery and development.
The
company has 10 manufacturing facilities in India, the US and Canada and a team
of over 6,300 people across the globe.
JUBILANT LIFE SCIENCES GETS WARNING
LETTER FROM USFDA
Jubilant Life Sciences Ltd (JLL) on Thursday said it has
received a warning letter from the USFDA over violation of manufacturing norms
at one of its plants located in Washington State.
Shares of Jubilant Life Sciences were trading at Rs 126.05
per scrip in the afternoon trade, down 9.96 per cent from the previous close on
the BSE.
In a statement, the company said one of its manufacturing
facilities, Jubilant HollisterStier, LLC (JHS) located at Spokane, Washington
state, US has been issued a warning letter by US Food and Drug Administration
(USFDA).
As required by USFDA, JHS-Spokane will respond to this warning letter (WL) on
or before December 12, 2013. The response will identify corrective actions
already been completed as well as some pending corrective actions to ensure
on-going cGMP (current good manufacturing practice) compliance, it said.
"(US)FDA specified in the WL that until all corrections
have been completed and that they have confirmed correction of the violations
and firm compliance to cGMPs, FDA may withhold approval of new applications or
supplements listing JHS-Spokane as the drug product manufacturer," the
statement added.
On the implications of the letter, the company clarified:
"We expect that the on-going manufacturing, distribution and sale of products
from this facility will not be impacted as the WL will affect new approvals
only."
JHS-Spokane is committed to implementing the necessary
corrective actions required to address the FDA concerns, and will work closely
with the FDA to bring resolution to this matter, it added.
According to the company's website, the Spokane plant
contract manufactures sterile injectables and allergenic extracts.
During the first half of FY14, the contract manufacturing
operations at JHS-Spokane contributed 7 per cent to consolidated sales, it
said.
The company had reported consolidates sales of Rs 2,742.34 crore in the first half
of the ongoing fiscal.
JUBILANT LIFE SCIENCES JUMPS AFTER
USFDA OKS CANADA PLANT
Capital Market/ 10:28 , Feb 28, 2014
Jubilant Life Sciences jumped 6.39% to Rs 124 at 10:22 IST
on BSE after the company said that the US drug regulator classified the
company's pharmaceutical manufacturing facility at Montreal in Canada as
acceptable.
Meanwhile, the BSE Sensex was up 95.45 points, or 0.45%, to
21,082.44.
On BSE, so far 75,000 shares were traded in the counter,
compared with an average volume of 75,324 shares in the past one quarter.
The stock hit a high of Rs.126 and a low of Rs.121.50 so far
during the day. The stock hit a 52-week high of Rs.190 on 12 March 2013. The
stock hit a 52-week low of Rs.65.10 on 28 August 2013.
The stock had underperformed the market over the past one
month till 26 February 2014, sliding 5.59% compared with the Sensex's 0.69%
fall. The scrip had also underperformed the market in past one quarter, falling
4.15% as against Sensex's 2.75% rise.
The small-cap integrated pharmaceuticals and life sciences
company has an equity capital of Rs 15.93 crore. Face value per share is Re 1.
Jubilant Life
Sciences announced that it has received a communication from the US Food and
Drug Administration (FDA), classifying its pharmaceutical manufacturing
facility at Montreal in Canada as 'acceptable'. This resolves all issues raised
by the FDA on the facility in February 2013 and subsequent communications, the
company said.
The development follows completion of FDA's review of the
company's responses post the February letter and the subsequent re-inspection
conducted at Jubilant's Montreal facility in September, 2013. This development successfully
resolves the FDA issues at our Montreal facility, Jubilant Life Sciences said.
In February 2013, Jubilant Life Sciences had informed that
one of its manufacturing facility, Jubilant HollisterStier General Partnership
(JHS), located at Kirkland, Quebec, Canada was issued a Warning Letter (WL) by
US FDA identifying significant violations of current Good Manufacturing
Practices (cGMP) Regulations.
Jubilant Life Sciences' consolidated net profit surged
437.39% to Rs 143.43 crore on 10.44% growth in total income from operations to
Rs 1442.78 crore in Q3 December 2013 over Q3 December 2012. The profit after
tax (PAT) during the quarter was boosted by exceptional items.
Consequent to re-evaluation of certain tax provisions
pertaining to earlier years (including deferred taxes), tax benefit (net of
reversal of deferred tax assets and true up of current tax) amounting to Rs
78.96 crore and Rs 56.35 crore has been recognized in the current quarter and
nine months ended 31 December 2013 respectively, the company said.
Amortization of debit balance in foreign currency monetary
items translation difference account (FCMITDA) of Rs 254.000 Millions , Rs
387.400 Millions , Rs 224.500 Millions , Rs 896.500 Millions , Rs 456.000
Millions and Rs 631.600 Millions for the quarter ended 31 December 2013, 30
September 2013, 31 December 2012; nine months ended 31 December 2013, 31
December 2012; and year ended 31 March 2013 respectively, representing exchange
difference on long-term foreign currency monetary liabilities which has been
used for the purpose other than acquiring fixed assets, Jubilant Life Sciences
said. The remaining amount of exceptional items for all periods primarily
represents foreign exchange difference for the period (excluding portion
included in finance cost) and mark to market gain/loss (net of related
contractual recoveries) in respect of currency and interest rate swap
contracts, the company said.
With regard to future business outlook, Jubilant Life
Sciences said the revenues and EBIDTA are expected to improve in the coming
quarters led by improved capacity utilisations in Sterile Injectables and OCL,
Nutrition Ingredients, Symtet and 3CP and backward integration of Pyridine and
expansion to newer markets. The company said it expects revenue growth due to
strong pipeline in APIs and Solid Dosage Formulations, new product launches,
expansion in newer geographies in API and Formulations, and robust order book
in Sterile Injectables.
Jubilant Life Sciences is a global pharmaceutical and life
sciences company engaged in manufacture and supply of APIs, generics, specialty
pharmaceuticals and life science ingredients. It also provides services in
contract manufacturing and drug discovery and development. The company's
strength lies in its unique offerings of pharmaceutical and life sciences
products and services across the value chain. With 10 world-class manufacturing
facilities in India, US and Canada and a team of over 6,300 multicultural
people across the globe, the company is committed to deliver value to its
customers spread across 100 countries.
IDRI and Jubilant Chemsys Extend Research for TB Drug
Discovery
24 Mar
14
Jubilant
Chemsys, a wholly-owned subsidiary of Jubilant Life Sciences, and one of the
most recognized drug discovery research services organizations from Asia today
announced an extension of collaborative partnership with Infectious Disease
Research Institute (IDRI) for TB drug discovery. Today marks World TB Day,
designated to build public awareness of the disease. Jubilant Chemsys offers
drug discovery research services on Full Time Equivalent and Fees- for-services
basis.
The
collaboration in chemistry support has been in existence since 2009 as part of
a joint effort with the Lilly TB Drug Discovery Initiative (LTI) and so far has
generated hundreds of Novel Chemical Entities (NCEs) – some of which have now
been identified for further exploration. Jubilant Chemsys, under the terms of
agreement, so far had been offering philanthropic support and expertise in
synthesis of NCEs to support the early research in TB.
Commenting
on the collaboration, Dr. Subir Basak, President, Jubilant Drug Discovery
Services “Jubilant is excited to extend the partnership with IDRI in
identifying and discovering novel chemical entities in the area of
tuberculosis. We are glad that the collaboration is leveraging our chemistry
talent and our experience in anti-bacterial research. The research outcome will
address the current unmet medical need in the field of TB therapy - a growing
concern in under developed and developing economies including India.”
“We
have been impressed with the commitment and engagement from Jubilant Chemsys in
support of TB drug discovery over the past few years. We are excited to expand
our efforts with them to access additional synthetic and medicinal chemistry
capabilities. This will take us another step closer to our goal of developing
much-needed new drugs to combat tuberculosis.” Tanya Parish, Ph.D., IDRI, Vice
President of Drug Discovery.
IDRI
is a founding member of both the LTI and the TB Drug Accelerator, a unique
partnership funded by the Bill & Melinda Gates Foundation that targets the
discovery of new TB drugs by collaborating on early-stage research. The
Foundation recently awarded IDRI $3.4 million in additional funding to Tanya
Parish, Ph.D., IDRI Vice President of Drug Discovery, and supplements an
earlier grant awarded in 2010, for a total of $7.8 million. The grant is
focused on identifying new leads and drug targets for tuberculosis with the
ultimate goal of producing new drugs to treat TB.
“Jubilant
has been extremely successful in advancing several validated hit series for the
Lilly Initiative, and we are very pleased to see this progress recognized
through the ability to expand the Jubilant collaboration with IDRI. We
are very grateful to Jubilant for all they have done to get us to this point,
and look forward to reaching our goals through this expanded
partnership.” Dr. Philip Hipskind, Ph.D., Lilly, Distinguished
Research Fellow and Leader of the Lilly Initiative.
With
this association, Jubilant will continue to provide chemistry and medicinal
chemistry support to IDRI. The company is also evaluating further liaisoning
with IDRI in key functional areas like computational chemistry and
Drug Metabolism and Pharmacokinetics (DMPK) Services.
Mycobacterium tuberculosis is
the causative agent of human tuberculosis, a devastating infectious disease
that kills about 1.5 million and infects more than eight million people each
year. There is an increasing threat from multi-drug resistant and extremely
drug resistant strains, demonstrating the need to develop more effective,
cheaper and faster-acting drugs – the prime focus of IDRI’s Discovery Program,
led by Parish.
First Trust Healthcare sells its
hospital business to Narayana Health
Jubilant First Trust Healthcare
(JFTH), a wholly owned subsidiary of Jubilant Life Sciences sells its hospital
business to Narayana Health. JFTH operates two hospitals in West Bengal,
Kalpataru in Barasat and Rabindranath Thakur in Berhampore. The business has
been transferred on a going concern basis as a slump sale which will enable
Jubilant Life Sciences to focus on its core businesses in Pharmaceuticals and
Life Sciences.
Commenting on the occasion, Ms. Aashti Bhartia, Director
Jubilant First Trust Healthcare said, "We entered the hospital business
with the vision to build high quality and affordable healthcare. We are happy
to hand over the hospitals to Narayana Health, whom we are confident of taking
the vision forward."
Speaking on the occasion, Dr. A. Raghuvanshi, Vice Chairman,
MD and Group CEO, Narayana Health said, "The acquisition of Jubilant First
Trust Healthcare's hospital business in West Bengal is part of our strategic
growth plan for East India. We have plans to upgrade the facilities at Jubilant
Kalpataru Hospital in Kolkata to offer tertiary level super-speciality care in
four to six months." He further added, "We are glad to include these
hospitals into our network and reinforce our commitment to West Bengal in
bringing high-class super-speciality healthcare services within the reach of
the common man."
Anand
Rathi Advisors were the Investment Bankers for the deal.
JUBILANT LIFE SCIENCES DECLARED WINNER OF 'BIO EXCELLENCE
AWARD 2014' AT BANGALORE INDIA BIO 2014
Jubilant
Life Sciences has been conferred with this year’s prestigious Bio-Excellence
Award from the Department of IT, BT and S & T, Government of Karnataka.
Jubilant was awarded under the category – BioServices.
Jubilant
was chosen for this award for its exemplary services in the Integrated Drug
Discovery and functional services business, delivering high-quality science
with high efficiency for international clients that include big pharma and
small biotech. The award was received by Jubilant Biosys, a Bengaluru based
subsidiary of Jubilant Life Sciences Ltd.
Conveying
his pleasure on the award, Dr. Subir K. Basak, President, Jubilant Biosys
(Global Drug Discovery Services) said, ‘It is our honor to receive
the prestigious award in the category of BioServices in this highly acclaimed
event. The award is an encouragement of our work in comprehensive drug
discovery solutions, from target discovery to IND. The recognition motivates
our team of eminent scientists to deliver the best to our partners who are large
and mid-size pharma and Biotech companies.”
‘Bangalore
India Bio 2014’ is India’s premier annual Biotech event renowned for
calibrating the pulse of the Biotechnology industry. The event is organized by
the Department of IT, BT and S & T, Government of Karnataka in association
with Vision Group on Biotechnology (an apex Advisory Body headed by Dr. Kiran
Mazumdar Shaw and consisting of members from both public and private sectors).
The focal theme of Bangalore India Bio 2014 was "Biotech for a better tomorrow".
As
part of the event, for the first time, Bio-Excellence Awards were presented to
biotech leaders for their outstanding achievements in the Biotech sector. The
Bio-Excellence Awardees are selected by a Jury of Industry Experts, constituted
by Association of Biotechnology Led Enterprises (ABLE). The eminent Jury
of Industry Experts selected winners who met three major criteria of: increase
in revenue, awards and achievements received by an individual/ an institution
and total number of Patents and Products.
The
International Conference deliberated on issues of pressing interest to the
Biotech sector including Stem Cells and Regenerative Medicine, Biosimilars,
Personalised Medicine, Biomedical Devices and Digital Health, Bioinformatics
for Drug Discovery & Development, Key Issues in Clinical Trials Development
and Agri Biotech & Secondary Agriculture.
USFDA ISSUES WARNING TO JUBILANT LIFE
OVER CGMP NORMS
The US
health regulator has found significant violations of good manufacturing norms
at the Indian drug company Jubilant Life Sciences ' Washington facility for
finished pharmaceuticals.
The
manufacturing facilities, Jubilant HollisterStier, LLC (JHS) located at
Spokane, Washington state, was inspected between April 15 and May 10, 2013, the
US Food and Drug Administration (USFDA) said in a warning letter to company's
CEO Marcelo A Morales.
Investigators
from USFDA found significant violations of current good manufacturing practice
(CGMP) regulations for finished pharmaceuticals, it added.
"The
violations cause your drug products to be adulterated within the meaning of
Section 501(a)(2)(B) of the Federal Food, Drug, and Cosmetic Act (the Act), 21
USC 351 (a)(2)(B), in that the methods used in, or the facilities or controls
used for, their manufacture, processing, packing, or holding do not conform to,
or are not operated or administered in conformity with CGMP," USFDA said.
One of
the specific violations which the investigators found was failure to establish
written procedures, that are designed to prevent microbiological contamination
of drug products purporting to be sterile, and that include validation of all
aseptic and sterilisation processes, it added.
The
other violation was failure to adequately document all work orders associated
with the April 2013 shutdown and include appropriate quality unit oversight.
"Your
firm failed to assure an adequate system for cleaning and disinfecting aseptic
processing areas and equipment," USFDA said.
"The
company should take prompt action to correct the violations cited in this
letter. Failure to promptly correct these violations may result in legal action
without further notice including, without limitation, seizure and
injunction," it added.
"Within
fifteen working days of receipt of this letter, please notify this office in
writing of the specific steps that you have taken to correct and prevent the
recurrence of violations, and provide copies of supporting documentation,"
USFDA said.
According
to the company's website, the Spokane plant contract manufactures sterile
injectables and allergenic extract.
Shares
of Jubilant Life Sciences today ended at Rs 116.50 per scrip on BSE, down 2.51
percent from its previous close.
USFDA WARNING MAY HIT JUBILANT LIFE'S VALUATIONS: EXPERT
Jubilant Life Sciences has received a
warning letter from the US Food and Drug Administration (US FDA) for its plant
is located at Spokane, Washington State, US. Shares of the company were locked
at 10 percent lower circuit on Thursday (Read More). In an interview to
CNBC-TV18, Surajit Pal, Pharma Analyst of Prabhudas Lilladher shared his views
on this development and outlook for the stock.
Below is the verbatim transcript of Surajit
Pal’s interview with CNBC-TV18
Q: What is your first take on this and whether
this is the second US-FDA warning letter that Jubilant Life Sciences has
received this year?
A: Yes and if this plant again receives a
warning letter along with the other one in Canada, then it is a dent in terms
of growth expectation for the company in same operations, which contributes
roughly around 14 percent of the total revenue.
At the time when the company is recovering
from downfall in price purchase agreements (PPAs) getting benefit of increasing
price over there, their pharmaceutical business is facing this kind of a
problem. Ultimately, what will happen is that the valuation increase that was
happening from a very low level could be impacted going forward.
Q: How much do you think would have been
factored in by the 8 percent fall in stock price today? Do you think going
forward, the stock will underperform?
A: Being a midcap company and a huge
underperformer over a period of time, people were expecting that return of
price range in PPAs, which contributes roughly around 52 percent in first half
mostly had contributed higher margin and pharmaceutical business, will at least
maintain their 24-25 percent margin. That will be hampered now and the company
will need a lot of capex, which is not good sign because it is already highly
leveraged.
Q: They did have plans of raising around
USD 250-300 million through the listing of their pharmaceutical business in
Singapore, something which they had just announced a few weeks or maybe a month
or so ago. Do you think that might be hampered now considering that the US-FDA
has clamped down now?
A: Yes, I think it would be tough to sell
the proposition when 14 percent of the total revenue is facing a scenario which
could be banned from production and supply into US market which is the main
market.
Q: How much do you think Jubilant Life
Sciences needs this money simply because of the debt that they will have to
service?
A: That is what management expressed that
they will reduce a big chunk of that money to reduce debts.
JUBILANT LIFE SCIENCES' US MANUFACTURING FACILITIES GETS
WARNING LETTER FROM USFDA
Jubilant Life Sciences Ltd has submitted to
BSE a copy of the Announcement being issued on December 05, 2013.
IFC’S $147.5 MILLION PACKAGE TO JUBILANT PHARMA TO EXPAND
ACCESS TO AFFORDABLE PHARMACEUTICALS
New Delhi, India, May 26, 2014—IFC, a member of the World Bank Group, is lending $147.5
million to Jubilant Pharma Limited to enable better access to quality and
affordable pharmaceuticals in underserved markets in India and across the
world. Jubilant Pharma, a wholly owned subsidiary of Jubilant Life Sciences
Limited, is incorporated in Singapore, with manufacturing operations in India,
USA, and Canada.
Of the total financing package, $110 million
is from IFC’s own account. The remaining $37.5 million is from IFC’s Managed
Co-Lending Portfolio Program, which provides additional long-term financing
through co-financing partners. The loan will help Jubilant Life Sciences
increase focus on the pharmaceutical sector and strengthen its generic drug
manufacturing facilities in India.
"We consider IFC a long-term partner
with significant healthcare expertise across emerging markets. IFC’s
contribution goes beyond financing. IFC will also help us strengthen our
quality assurance and risk mitigation mechanisms and make the company systems
more robust,” said Shyam S. Bhartia, Chairman and Managing Director, Jubilant
Life Sciences. “IFC’s long-term financing package will consolidate our entire
pharmaceuticals business under Jubilant Pharma and build global
competitiveness.”
Jubilant Life Sciences is a pharmaceutical
and life sciences company engaged in the manufacture and supply of active
pharmaceutical ingredients, generics, specialty pharmaceuticals, and life
science ingredients. It provides services in contract manufacturing and drug
discovery and development.
“Health is a priority sector for IFC in
India. There is an need to expand access to affordable and quality healthcare,
especially among low-income communities,” said Vipul Prakash, Director –
Manufacturing, Agribusiness and Services, Asia Pacific, IFC. “This investment
will contribute towards making the Indian pharmaceuticals sector globally
competitive, and improve access to affordable medicines to a wider population.”
IFC is the world’s largest multilateral
investor in the private health care sector in emerging markets, having provided
financing of over $2.2 billion to 164 private health care and life sciences
projects in 53 countries. IFC-supported health projects treat about 12 million
patients annually.
IDRI and Jubilant Chemsys Extend Research for TB Drug
Discovery
Jubilant
Chemsys, a wholly-owned subsidiary of Jubilant Life Sciences, and one of the
most recognized drug discovery research services organizations from Asia today
announced an extension of collaborative partnership with Infectious Disease
Research Institute (IDRI) for TB drug discovery. Today marks World TB Day,
designated to build public awareness of the disease. Jubilant Chemsys offers
drug discovery research services on Full Time Equivalent and Fees- for-services
basis.
The
collaboration in chemistry support has been in existence since 2009 as part of
a joint effort with the Lilly TB Drug Discovery Initiative (LTI) and so far has
generated hundreds of Novel Chemical Entities (NCEs) – some of which have now
been identified for further exploration. Jubilant Chemsys, under the terms of
agreement, so far had been offering philanthropic support and expertise in
synthesis of NCEs to support the early research in TB.
Commenting
on the collaboration, Dr. Subir Basak, President, Jubilant
Drug Discovery Services “Jubilant
is excited to extend the partnership with IDRI in identifying and discovering
novel chemical entities in the area of tuberculosis. We are glad that the
collaboration is leveraging our chemistry talent and our experience in
anti-bacterial research. The research outcome will address the current unmet
medical need in the field of TB therapy - a growing concern in under developed
and developing economies including India.”
“We have
been impressed with the commitment and engagement from Jubilant Chemsys in
support of TB drug discovery over the past few years. We are excited to expand
our efforts with them to access additional synthetic and medicinal chemistry
capabilities. This will take us another step closer to our goal of developing
much-needed new drugs to combat tuberculosis.” Tanya Parish, Ph.D., IDRI, Vice
President of Drug Discovery.
IDRI is a
founding member of both the LTI and the TB Drug Accelerator, a unique
partnership funded by the Bill & Melinda Gates Foundation that targets the
discovery of new TB drugs by collaborating on early-stage research. The
Foundation recently awarded IDRI $3.4 million in additional funding to Tanya
Parish, Ph.D., IDRI Vice President of Drug Discovery, and supplements an
earlier grant awarded in 2010, for a total of $7.8 million. The grant is
focused on identifying new leads and drug targets for tuberculosis with the
ultimate goal of producing new drugs to treat TB.
“Jubilant
has been extremely successful in advancing several validated hit series for the
Lilly Initiative, and we are very pleased to see this progress recognized
through the ability to expand the Jubilant collaboration with IDRI. We
are very grateful to Jubilant for all they have done to get us to this point,
and look forward to reaching our goals through this expanded
partnership.” Dr. Philip Hipskind, Ph.D., Lilly, Distinguished
Research Fellow and Leader of the Lilly Initiative.
With this
association, Jubilant will continue to provide chemistry and medicinal
chemistry support to IDRI. The company is also evaluating further liaisoning
with IDRI in key functional areas like computational chemistry and
Drug Metabolism and Pharmacokinetics (DMPK) Services.
Mycobacterium tuberculosis is the causative agent of human tuberculosis,
a devastating infectious disease that kills about 1.5 million and infects more than
eight million people each year. There is an increasing threat from multi-drug
resistant and extremely drug resistant strains, demonstrating the need to
develop more effective, cheaper and faster-acting drugs – the prime focus of
IDRI’s Discovery Program, led by Parish.
Jubilant Life Sciences receives ANDA approval
25 Apr 14
Jubilant Life Sciences Ltd, an integrated Pharmaceuticals and
Life Sciences Company announced today that it has received Abbreviated
New Drug Application (ANDA) approval from the US Food and Drug Administration
(US FDA) forSpironolactone Tablets, 25 mg, 50 mg and 100 mg, the generic version of Aldactone® (of GD Searle), which is used as a
diuretic to treat fluid retention (edema) caused by congestive heart failure and
cirrhosis of the liver. We expect to launch this product in Q1 FY15. The total
market size for Spironolactone Tablets as per IMS is US$ 87 Million per annum.
Jubilant has also received a tentative approval from the
USFDA for Memantine Tablets, 5 mg and 10 mg, the generic version of Namenda® (of Forest Labs), which is used for
treatment of moderate-to-severe Alzheimer’s disease. We expect to launch this
product post patent expiry in 2015. The total market size for Namenda® as per IMS is US$ 1.85 Billion per
annum.
As on December 31, 2013, Jubilant Life Sciences had a total of 689
filings for formulations of which 230 have been approved in various regions
globally. This includes 60 ANDAs filed in the US and 42 Dossier filings in
Europe.
Jubilant Life Sciences to participate in VIV India 2014
22 Apr 14
Jubilant Life Sciences, the worldwide second largest
manufacturer of Niacinamide and Niacin (Vitamin B3) will be participating in
VIV India 2014. The exhibition to be held over 2 days, 23rd-24th April at BIEC,
Bengaluru, India brings together different sectors in animal production and
processing. At VIV India, Jubilant aims to introduce several of its existing
products to the industry players in animal nutrition and feed category. Visit
Jubilant Stall at A062 to meet officials from the Company.
Jubilant’s Niacinamide & Niacin (Vitamin B3) finds
multiple applications in Animal Nutrition, Human Nutrition, Pharmaceuticals,
Cosmeceuticals, Technical Applications and Agrochemicals. The complete backward
integration, (Molasses – Alcohol – Acetaldehyde – Pyridine/Beta Picoline –
Niacinamide / Niacin) up to the basic feedstock, is one of the major advantages
that the Company has over the other manufacturers of Niacinamide and Niacin.
Jubilant’s Animal Nutrition products are widely used in Poultry, Dairy, Pet
Food and Veterinary Drugs industry. The manufacturing facilities of Jubilant
meet global standards and are certified for major regulatory certifications
such as KOSHER, Halal, FAMI-QS and cGMP regulations.
Jubilant Life Sciences is also India’s largest manufacturer
of Synthetic Choline Chloride. This compound is used to create products that
are offered in different forms for animal feed applications, all of which duly
meet international quality standards. Choline Chloride is used in: Poultry,
Dairy, Pet food and Pharmaceuticals with Choline enriched products.
In Speciality Products, Jubilant offers Specialty Feed
Supplements to farmers across the globe, thus helping them to continuously increase
their returns on investments, which are mainly Vitamin and Minerals Premixes,
Betaine, Organic Mineral Premix, Acidifiers, Toxin Binders, Chromium based
Growth Promoter, Feed Emulsifier and Liver Treatment Products.
Jubilant has a broad range of product portfolio in
Nutrition Ingredients. Portfolio includes:
- AciFeed - Feed Acidifier for mould & bacteria inhibition
- BGTMDS/MultiminPlus - Trace Mineral Premixes
- EnCroMix - Organic Tri-Chromium preparation for energy utilisation
- Hi Feed Phytase - Hi Performance Thermo-tolerant Micro-granulated
Phytase
- Hi-Pro-Min-Organic Mineral premix for poultry
- JubiDOL - Feed emulsifier
- OsmoBetaine – Betaine for osmo-regulation in livestock
- ToxiCOP/Protect Plus - Broad spectrum mould and Mycotoxin binder
- WinTox - Liquid formulation for nourishment, detoxification and
rejuvenation of vital organs
The Board of Jubilant Life Sciences Limited, an integrated
pharmaceutical and life sciences company met today to approve financial results
for the quarter ended March 31, 2014.
26 May 14
The Board of Jubilant Life Sciences Limited, an integrated
pharmaceutical and life sciences company met today to approve financial results
for the quarter ended March 31, 2014.
Commenting on the Company’s performance, Mr. Shyam S Bhartia,
Chairman & Managing Director and Mr. Hari S Bhartia, Co-Chairman &
Managing Director, Jubilant Life Sciences said:
“We have implemented the management consolidation of Pharmaceutical
and Life Science Ingredients businesses to enable faster and focussed growth
going forward. In pharmaceutical business we have strengthened our Quality
System for better compliance. We are happy to report that warning letter issue
in Montreal have been resolved and in Spokane, we have responded to all FDA
observations. We are confident of bringing back our growth in CMO of Sterile
injectibles business on the back of strong order book and better compliance.
Our financial arrangement with IFC enable us to de-couple our pharmaceutical
business from Life Science Ingredient and enhance shareholder value.”
Q4 FY2014 Highlights
·
Consolidated revenue
up 12% YoY
·
International revenues
at Rs. 1,160 crore, contributing 74% to the overall mix and up 8% YoY
·
EBITDA margins at
16.1% and Normalized PAT at 62 Crore
FY2014 Highlights
·
Consolidated revenue
up 12% YoY
·
International revenues
at Rs. 4,327 Crore, contributing 75% to the overall revenues
·
EBITDA margins at
17.7% and Normalized PAT at 324 Crore
Pharmaceuticals Segment Review
In Q4 FY2014, Income from operations of the Pharmaceuticals
business was at Rs. 705 crore, with a share of 45% to the revenue mix. The
Pharmaceutical business EBITDA stood at Rs. 132 crore with EBITDA margins at
18.8%.
For FY2014 the Income from operations was at Rs. 2,728 crore
with contribution of 47% to the overall revenue mix. The segment EBITDA came in
at Rs. 610 crore with EBITDA margins at 22.4%.
Life Science Ingredients Segment Review
In Q4 FY2014, Income from operations for the Life Science
Ingredients segment stood at Rs. 858 crore, improving 24% YoY and contributing
55% to total revenues. The segment EBITDA was at Rs. 131 crore with EBITDA
margins at 15.3%.
In FY2014 the Income from operations of the segment came in
at Rs. 3,076 crore, increased by 23% YoY and contributing 53% to the revenue
mix. The segment EBITDA came in at Rs. 483 crore with EBITDA margins at 15.7%.
Geographical Overview
International revenues contributed 74% to the revenue mix at
Rs. 1,160 crore with a share of 58% from regulated markets in Q4 FY2014.
Revenues from North America stood at Rs. 581 crore, contributing 37% share to
the overall revenues; revenues from Europe and Japan stood at Rs. 320 crore,
contributing 21% to the revenue mix. Domestic revenues grew 27% YoY at Rs. 403
crore, with a 26% contribution to the revenue mix. Revenues in ROW including
China were at Rs. 258 crore, up 33%, contributing 17% to the revenue mix.
International revenues contributed 75% to the revenue mix at
Rs. 4,327 crore with a share of 60% from regulated markets in FY2014. Revenue
from North America stood at Rs. 2,231 crore, up 5% and contributing a share of
38% to the revenue mix. Revenue from Europe and Japan was at Rs. 1,224 crore,
with a contribution of 21% to the revenue mix and growing 15% YoY. The Domestic
revenue was at Rs. 1,477 crore, up 11% YoY, with a share of 25% to the revenue
mix. Revenue from ROW including China stood at Rs. 871 crore, growing 37% and
contributing 15% to the revenue mix.
Outlook
Going forward, we expect strong growth momentum in both the
segments of our businesses. The Pharmaceuticals business is expected to deliver
on account of resolution of Warning Letter in Montreal, focussed attention to
resolve US FDA issues in Spokane, consolidation of global quality system for
compliance, new product launches in Generics business and better price
realizations and expected launch of Ruby-fill in Radiopharmaceuticals business.
Growth in Life Science Ingredients business is to be led by higher capacity
utilization, better pricing and entry into new geographies.
CMT REPORT (Corruption, Money Laundering & Terrorism]
The Public Notice information has been collected from various sources
including but not limited to: The Courts,
1] INFORMATION ON
DESIGNATED PARTY
No records exist designating subject or any of its beneficial owners,
controlling shareholders or senior officers as terrorist or terrorist organization
or whom notice had been received that all financial transactions involving
their assets have been blocked or convicted, found guilty or against whom a
judgement or order had been entered in a proceedings for violating
money-laundering, anti-corruption or bribery or international economic or
anti-terrorism sanction laws or whose assets were seized, blocked, frozen or
ordered forfeited for violation of money laundering or international
anti-terrorism laws.
2] Court Declaration :
No records exist to suggest that subject is
or was the subject of any formal or informal allegations, prosecutions or other
official proceeding for making any prohibited payments or other improper
payments to government officials for engaging in prohibited transactions or
with designated parties.
3] Asset Declaration :
No records exist to suggest that the property or assets of the subject
are derived from criminal conduct or a prohibited transaction.
4] Record on Financial
Crime :
Charges or conviction
registered against subject: None
5] Records on Violation of
Anti-Corruption Laws :
Charges or
investigation registered against subject: None
6] Records on Int’l
Anti-Money Laundering Laws/Standards :
Charges or
investigation registered against subject: None
7] Criminal Records
No
available information exist that suggest that subject or any of its principals
have been formally charged or convicted by a competent governmental authority
for any financial crime or under any formal investigation by a competent government
authority for any violation of anti-corruption laws or international anti-money
laundering laws or standard.
8] Affiliation with
Government :
No record
exists to suggest that any director or indirect owners, controlling
shareholders, director, officer or employee of the company is a government
official or a family member or close business associate of a Government
official.
9] Compensation Package :
Our market
survey revealed that the amount of compensation sought by the subject is fair
and reasonable and comparable to compensation paid to others for similar
services.
10] Press Report :
No press reports / filings exists on
the subject.
CORPORATE GOVERNANCE
MIRA INFORM as part of its Due Diligence do provide comments on
Corporate Governance to identify management and governance. These factors often
have been predictive and in some cases have created vulnerabilities to credit
deterioration.
Our Governance Assessment focuses principally on the interactions between
a company’s management, its Board of Directors, Shareholders and other
financial stakeholders.
CONTRAVENTION
Subject is not known to have contravened any existing local laws,
regulations or policies that prohibit, restrict or otherwise affect the terms
and conditions that could be included in the agreement with the subject.
FOREIGN EXCHANGE RATES
|
Currency |
Unit
|
Indian Rupees |
|
US Dollar |
1 |
Rs.59.26 |
|
|
1 |
Rs.99.63 |
|
Euro |
1 |
Rs.80.59 |
INFORMATION DETAILS
|
Information
Gathered by : |
GYT |
|
|
|
|
Analysis Done by
: |
SUB |
|
|
|
|
Report Prepared
by : |
NKT |
SCORE & RATING EXPLANATIONS
|
SCORE FACTORS |
RANGE |
POINTS |
|
HISTORY |
1~10 |
6 |
|
PAID-UP CAPITAL |
1~10 |
6 |
|
OPERATING SCALE |
1~10 |
6 |
|
FINANCIAL CONDITION |
|
|
|
--BUSINESS SCALE |
1~10 |
6 |
|
--PROFITABILIRY |
1~10 |
6 |
|
--LIQUIDITY |
1~10 |
6 |
|
--LEVERAGE |
1~10 |
6 |
|
--RESERVES |
1~10 |
6 |
|
--CREDIT LINES |
1~10 |
6 |
|
--MARGINS |
-5~5 |
-- |
|
DEMERIT POINTS |
|
|
|
--BANK CHARGES |
YES/NO |
YES |
|
--LITIGATION |
YES/NO |
NO |
|
--OTHER ADVERSE INFORMATION |
YES/NO |
NO |
|
MERIT POINTS |
|
|
|
--SOLE DISTRIBUTORSHIP |
YES/NO |
NO |
|
--EXPORT ACTIVITIES |
YES/NO |
NO |
|
--AFFILIATION |
YES/NO |
YES |
|
--LISTED |
YES/NO |
YES |
|
--OTHER MERIT FACTORS |
YES/NO |
YES |
|
DEFAULTERS |
|
|
|
--RBI |
YES/NO |
NO |
|
--EPF |
YES/NO |
NO |
|
|
|
|
|
TOTAL |
|
54 |
This score serves as a reference to assess
SC’s credit risk and to set the amount of credit to be extended. It is calculated
from a composite of weighted scores obtained from each of the major sections of
this report. The assessed factors and their relative weights (as indicated
through %) are as follows:
Financial
condition (40%) Ownership
background (20%) Payment record
(10%)
Credit history
(10%) Market trend (10%) Operational size
(10%)
RATING EXPLANATIONS
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
>86 |
Aaa |
Possesses an extremely sound financial base with the strongest capability
for timely payment of interest and principal sums |
Unlimited |
|
71-85 |
Aa |
Possesses adequate working capital. No caution needed for credit
transaction. It has above average (strong) capability for payment of interest
and principal sums |
Large |
|
56-70 |
A |
Financial & operational base are regarded healthy. General
unfavourable factors will not cause fatal effect. Satisfactory capability for
payment of interest and principal sums |
Fairly Large |
|
41-55 |
Ba |
Overall operation is considered normal. Capable to meet normal
commitments. |
Satisfactory |
|
26-40 |
B |
Capability to overcome financial difficulties seems comparatively
below average. |
Small |
|
11-25 |
Ca |
Adverse factors are apparent. Repayment of interest and principal sums
in default or expected to be in default upon maturity |
Limited with
full security |
|
<10 |
C |
Absolute credit risk exists. Caution needed to be exercised |
Credit not
recommended |
|
-- |
NB |
New Business |
-- |
This report is issued at your request without any
risk and responsibility on the part of MIRA INFORM PRIVATE LIMITED (MIPL)
or its officials.