MIRA INFORM REPORT
|
Report Date : |
28.03.2014 |
IDENTIFICATION DETAILS
|
Name : |
GLOCHEM INDUSTRIES LIMITED |
|
|
|
|
Registered
Office : |
G.V. Chambers, 7-2-C8 and C8/2, IDA Santnagar,
Hyderabad-500018, Andhra Pradesh |
|
|
|
|
Country : |
India |
|
|
|
|
Financials (as
on) : |
31.03.2013 |
|
|
|
|
Date of
Incorporation : |
20.11.1995 |
|
|
|
|
Com. Reg. No.: |
01-022303 |
|
|
|
|
Capital Investment
/ Paid-up Capital : |
Rs.36.291
Millions |
|
|
|
|
CIN No.: [Company Identification
No.] |
U24110AP1995PLC022303 |
|
|
|
|
TAN No.: [Tax Deduction &
Collection Account No.] |
HYDG00852F |
|
|
|
|
PAN No.: [Permanent Account No.] |
AAACG9134E |
|
|
|
|
Legal Form : |
A Closely Held
Public Limited Liability Company |
|
|
|
|
Line of Business
: |
Subject is engaged in the business of manufacturing and marketing of
Active Pharma Ingredients (APIs), drug intermediates, finished dosage forms
(FDF). |
|
|
|
|
No. of Employees
: |
Information declined by the management
|
RATING & COMMENTS
|
MIRA’s Rating : |
B (33) |
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
26-40 |
B |
Capability to overcome financial difficulties seems comparatively below
average. |
Small |
|
Maximum Credit Limit : |
USD 2100000 |
|
|
|
|
Status : |
Moderate |
|
|
|
|
Payment Behaviour : |
Slow but correct |
|
|
|
|
Litigation : |
Clear |
|
|
|
|
Comments : |
Subject is a well-established company having moderate track record.
The company has achieved a better growth in its sales turnover and net
profitability during current year. However, the profit margin seems to be
low. The ratings also take into consideration the small scale of operations
carried out by the company. Trade relations are fair. Business is active. Payment terms are slow
but correct. In view of experienced management and long track record of operations,
the subject can be considered for business dealings with some caution. |
NOTES :
Any query related to this report can be made
on e-mail : infodept@mirainform.com
while quoting report number, name and date.
ECGC Country Risk Classification List – December 1, 2013
|
Country Name |
Previous Rating (30.09.2013) |
Current Rating (01.12.2013) |
|
India |
A1 |
A1 |
|
Risk Category |
ECGC
Classification |
|
Insignificant |
A1 |
|
Low |
A2 |
|
Moderate |
B1 |
|
High |
B2 |
|
Very High |
C1 |
|
Restricted |
C2 |
|
Off-credit |
D |
INDIAN ECONOMIC OVERVIEW
India’s current account deficit for the fiscal third quarter ended
September 2013 narrowed to $4.2 billion or 0.9 % of the gross domestic product from
$31.9 billion or 6.5 % of GDP a year earlier, thanks to a pick-up in exports
and moderation in gold imports. Manufacturing activity and new orders in India
showed their strongest growth in a year in February. The news comes as a relief
after data showed Asia’s third largest economy grew by a slower-than-expected
4.7 % annually in the three months through December. The HSBC Manufacturing
Purchasing Managers’ Index which gauges the business activity of India’s
factories but not its’ utilities, rose to 52.5 in February, its highest in a
year from 51.4 in January. Overall new orders for factory goods which rose to a
one-year high of 54.9 contributed to the surge. China has emerged as India’s
biggest trading partner in the current financial year replacing the United Arab
Emirates and pushing it to the third spot. India-China trade has reached $49.5
billion with a 8.7 % share in India’s total trade. The US comes second at $46
billion with 8.1 % share during the first nine months of the current financial
year.
The Reserve Bank of India has granted an additional nine months to the
public to exchange currency notes printed before 2005 including Rs 500 and Rs
1,000 denominations, pushing the deadline to January 1, 2015. A day before
dates for the Lok Sabha polls were announced, the government decided to hike
interest rates on fixed deposit schemes offered by post offices up to 0.2 per
cent. The new rates will be effective April, 1. The Supreme Court will resume
hearing on March, 11 Nokia’s appeal against a ruling over transferring
ownership of its local mobile phones plant which is the subject of a tax
dispute to Microsoft Corp.
In the last days of the current Government, another scam has surfaced.
The defence ministry has ordered a probe into Hindustan Aeronautics Limited’s
contracts from Britain’s Rolls-Royce Holdings worth at least $ 1.2 billion. The
Central Bureau of Investigation will look into allegations that over $80
million was paid in kickbacks in a deal signed in 2011. India has asked Boeing
Co. to find a solution for problems with state-owned Air India’s 787
Dreamliners. The aircraft has experienced a series of malfunctions since its
debut in 2011.
EXTERNAL AGENCY RATING
|
Rating Agency Name |
CARE |
|
Rating |
Long term Bank Facilities: BB+ |
|
Rating Explanation |
Have moderate risk of default. |
|
Date |
10.01.2014 |
|
Rating Agency Name |
CARE |
|
Rating |
Short term Bank Facilities: A4 |
|
Rating Explanation |
Have minimal degree of safety and carry very high credit risk. |
|
Date |
10.01.2014 |
RBI DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available RBI Defaulters’ list.
EPF (Employee Provident Fund) DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available EPF (Employee Provident Fund) Defaulters’ list as of
31-03-2012.
INFORMATION DECLINED
MANAGEMENT NON – COOPERATIVE (91-40-39219900)
LOCATIONS
|
Registered
Office/ Corporate Office : |
G.V. Chambers, 7-2-C8 and C8/2, IDA Santnagar, Hyderabad-500018,
Andhra Pradesh, India |
|
Tel. No.: |
91-40-23713041 / 42 / 43 / 23816951/39219900 |
|
Fax No.: |
91-40-23711483/23813850 |
|
E-Mail : |
|
|
Website: |
|
|
|
|
|
Factory 1 : |
174/176, I.D.A.,
Bollaram, Medak-502325 District, Andhra Pradesh, India |
|
Tel No.: |
91-8458-279003/
280021 91-8452-302944 |
|
Fax No.: |
91-8458-279058 |
|
E Mail: |
|
|
|
|
|
Factory 2 : |
Survey No. 36, 37 and 46, Plot No. 77, |
|
Tel No.: |
91-892-4247132 91-891-3016504 |
|
Fax No.: |
91-892-4247269 |
|
E Mail: |
|
|
|
|
|
Factory 3 : |
Plot No.S16 and S17,
Green Industrial Park, Polapally, Jedcheria, Mahaboob Nagar District –
509302, Andhra Pradesh, India |
|
Tel No.: |
91-8542-304654 |
DIRECTORS
AS ON 28.09.2012
|
Name : |
Mr. Kattamuri Subbarao |
|
Designation : |
Chief
Promoter and Managing Director |
|
Address : |
Plot
No. 48, Navanirman Nagar, Road No. 71, Jubilee Hills, |
|
Date of Birth/Age : |
10.03.1953 |
|
Date of Appointment : |
01.07.2011 |
|
DIN No.: |
00107121 |
|
Name : |
Mr. Hazarath Basha Shaik |
|
Designation : |
Whole-time director |
|
Address : |
Hausen
Im Wiesental, Am Sport Platz 6, Deutschland – 79688, Germany |
|
Date of Birth/Age : |
26.03.1956 |
|
Date of Appointment : |
01.07.2011 |
|
DIN No.: |
01321868 |
|
Name : |
Mr. Vikram Ravindra Mamidipudi |
|
Designation : |
Director
|
|
Address : |
68,
Gun Rock Encalve, Secunderabad –
500009, Andhra Pradesh, India |
|
Date of Birth/Age : |
18.05.1956 |
|
Date of Appointment : |
24.03.1999 |
|
DIN No.: |
00008241 |
|
Name : |
Mr. Gopakumar Gopalan Nair |
|
Designation : |
Additional Director |
|
Address : |
C-002/3, Gokul Plaza, Thakur Complex, Kandivli (East), Mumbai-400101,
Maharashtra, India |
|
Date of Birth/Age : |
11.06.1941 |
|
Date of Appointment : |
30.06.2010 |
|
DIN No.: |
00092637 |
|
Name : |
Mr. Mohan Krishna Reddy |
|
Designation : |
Director
|
|
Address : |
8-2-293/82/w/78,
New No. 123, Road No. 5, Women Co-operative Society, Jubilee Hills, Hyderabad – 500034, Andhra Pradesh, India |
|
Date of Birth/Age : |
03.05.1956 |
|
Date of Appointment : |
24.12.2003 |
|
DIN No.: |
00093185 |
|
Name : |
Mr. Peter Arend William Burema |
|
Designation : |
Nominee Director |
|
Address : |
Delsbergstrasse 58, 4253, Liesberg, Netherlands-4253 |
|
Date of Appointment : |
30.06.2010 |
|
DIN No.: |
03054636 |
|
|
|
|
Name : |
Mr. Sundeep Kattamuri |
|
Designation : |
Whole Time Director |
|
Address : |
Plot No.48, Navanirman Nagar, Jubilee Hills, Road No.71,
Hyderabad-50003, Andhra Pradesh, India |
|
Date of Birth/Age : |
17.11.1983 |
|
Date of Appointment : |
05.09.2011 |
|
PAN No.: |
BRMPK2131G |
|
DIN No.: |
03128742 |
MAJOR SHAREHOLDERS / SHAREHOLDING PATTERN
AS ON 28.09.2012
NOTE: SHAREHOLDING DETAILS FILE ATTACHED.
AS ON 28.09.2012
|
Equity Share Breakup |
|
Percentage of Holding |
|
Category |
|
|
|
Foreign holdings [Foreign institutional investors, Foreign Companies, Foreign Financial Institutions, Non-resident Indian or Overseas corporate bodies or others] |
|
24.50 |
|
Bodies
corporate |
|
0.55 |
|
Directors
or relatives of directors |
|
66.07 |
|
Other
top fifty shareholders |
|
8.88 |
|
TOTAL |
|
100.00 |

BUSINESS DETAILS
|
Line of Business : |
Subject is engaged in the business of manufacturing and marketing of Active
Pharma Ingredients (APIs), drug intermediates, finished dosage forms (FDF). |
||||||||||||||||
|
|
|
||||||||||||||||
|
Products : |
|
||||||||||||||||
|
|
|
PRODUCTION STATUS [AS ON 31.03.2011]
|
Particulars |
Unit |
Installed
Capacity |
Actual
Production |
|
|
|
|
|
|
Bulk Drugs, Intermediates and Spent Solvents |
MT |
478 |
726.155 |
NOTES:
- (Industrial Licensing has been abolished in respect of Bulk Drugs
and formulations in terms of Press Note No.4 (1994 series) dated October
25th 1994 issued by the Department of the Industrial
Development, Ministry of Industry, Government of India and Notification
No. S.O.137 (E) dated 1st March 1999 issued by the Department
of Industrial Policy and Promotion, Ministry of Industry, Government of
India). Licensed Capacities for bulk drugs and formulations are not
applicable.
- Installed Capacities varies based on product mix and utilization of
manufacturing facilities given the nature of the production.
- Installed capacities are certified by the management and have not been
verified by the Auditors due to technical in nature.
GENERAL INFORMATION
|
No. of Employees : |
Information declined by the management
|
|||||||||||||||||||||
|
|
|
|||||||||||||||||||||
|
Bankers : |
|
|||||||||||||||||||||
|
|
|
|||||||||||||||||||||
|
Facilities : |
|
|
|
|
|
Banking
Relations : |
-- |
|
|
|
|
Auditors : |
|
|
Name : |
Deloitte Haskins
and Sells Chartered
Accountants |
|
Address : |
1-8-384 and 385,
Gowra Grand, 3rd Floor, SP Road, Secunderabad-500003, Andhra
Pradesh, India |
|
PAN N Income-tax PAN of auditor or auditor's firm : |
AACFD3771D |
CAPITAL STRUCTURE
As on 31.03.2013
Authorised Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
5000000 |
Equity Shares |
Rs.10/- each |
Rs.50.000 Millions |
Issued, Subscribed & Paid-up Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
3629080 |
Equity Shares |
Rs.10/- each
|
Rs.36.291
Millions |
FINANCIAL DATA
[all figures are
in Rupees Millions]
ABRIDGED BALANCE
SHEET
|
SOURCES
OF FUNDS |
31.03.2013 |
31.03.2012 |
31.03.2011 |
|
I.
EQUITY
AND LIABILITIES |
|
|
|
|
(1)Shareholders' Funds |
|
|
|
|
(a) Share Capital |
36.291 |
36.291 |
34.261 |
|
(b) Reserves & Surplus |
476.871 |
460.899 |
403.117 |
|
(c) Money received against
share warrants |
0.000 |
0.000 |
0.000 |
|
|
|
|
|
|
(2) Share Application
money pending allotment |
0.000 |
0.000 |
0.000 |
|
Total Shareholders’ Funds (1) + (2) |
513.162 |
497.190 |
437.378 |
|
|
|
|
|
|
(3) Non-Current
Liabilities |
|
|
|
|
(a) long-term
borrowings |
52.277 |
38.098 |
24.975 |
|
(b) Deferred tax liabilities (Net) |
3.661 |
21.017 |
20.194 |
|
(c) Other long term
liabilities |
141.878 |
169.425 |
221.970 |
|
(d) long-term
provisions |
3.457 |
6.444 |
3.414 |
|
Total Non-current
Liabilities (3) |
201.273 |
234.984 |
270.553 |
|
|
|
|
|
|
(4) Current Liabilities |
|
|
|
|
(a) Short term
borrowings |
221.154 |
142.116 |
45.131 |
|
(b) Trade payables |
215.406 |
200.602 |
86.847 |
|
(c) Other current
liabilities |
244.107 |
120.927 |
71.054 |
|
(d) Short-term
provisions |
10.353 |
8.018 |
3.916 |
|
Total Current
Liabilities (4) |
691.020 |
471.663 |
206.948 |
|
|
|
|
|
|
TOTAL |
1405.455 |
1203.837 |
914.879 |
|
|
|
|
|
|
II.
ASSETS |
|
|
|
|
(1) Non-current assets |
|
|
|
|
(a) Fixed Assets |
|
|
|
|
(i) Tangible assets |
679.243 |
664.259 |
299.504 |
|
(ii) Intangible Assets |
1.177 |
1.993 |
2.649 |
|
(iii) Capital
work-in-progress |
1.641 |
0.031 |
318.316 |
|
(iv) Intangible assets under development |
63.554 |
23.789 |
0.000 |
|
(b) Non-current
Investments |
0.000 |
0.000 |
0.000 |
|
(c) Deferred tax
assets (net) |
0.499 |
0.000 |
0.000 |
|
(d) Long-term Loan and Advances |
22.683 |
16.398 |
67.276 |
|
(e) Other
Non-current assets |
0.000 |
0.000 |
0.000 |
|
Total Non-Current
Assets |
768.797 |
706.470 |
687.745 |
|
|
|
|
|
|
(2) Current assets |
|
|
|
|
(a) Current investments |
0.000 |
0.000 |
0.001 |
|
(b) Inventories |
370.792 |
275.224 |
147.556 |
|
(c) Trade receivables |
158.584 |
146.592 |
61.255 |
|
(d) Cash and cash
equivalents |
3.049 |
1.139 |
1.770 |
|
(e) Short-term loans
and advances |
68.695 |
39.853 |
16.389 |
|
(f) Other current
assets |
35.538 |
34.559 |
0.163 |
|
Total Current Assets |
636.658 |
497.367 |
227.134 |
|
|
|
|
|
|
TOTAL |
1405.455 |
1203.837 |
914.879 |
PROFIT & LOSS
ACCOUNT
|
|
PARTICULARS |
31.03.2013 |
31.03.2012 |
31.03.2011 |
|
|
|
SALES |
|
|
|
|
|
|
|
Income |
1014.437 |
540.307 |
358.617 |
|
|
|
Other Income |
NA |
13.767 |
4.156 |
|
|
|
TOTAL (A) |
NA |
554.074 |
362.773 |
|
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
|
|
Cost of materials consumed |
|
323.685 |
177.177 |
|
|
|
Purchases of stock in trade |
|
1.697 |
10.883 |
|
|
|
Employee benefits expense |
|
74.141 |
64.640 |
|
|
|
Manufacturing, administrative and other expense |
|
173.402 |
137.970 |
|
|
|
Changes in inventories of finished goods and work in progress |
|
(74.415) |
(23.837) |
|
|
|
TOTAL (B) |
NA |
498.510 |
366.833 |
|
|
|
|
|
|
|
|
Less |
PROFIT
/ (LOSS) BEFORE INTEREST, TAX, DEPRECIATION AND AMORTISATION (A-B) (C) |
. |
55.564 |
(4.060) |
|
|
|
|
|
|
|
|
|
Less |
FINANCIAL
EXPENSES (D) |
|
14.272 |
5.666 |
|
|
|
|
|
|
|
|
|
|
PROFIT
/ (LOSS) BEFORE TAX, DEPRECIATION AND AMORTISATION (C-D) (E) |
50.630 |
41.292 |
(9.726) |
|
|
|
|
|
|
|
|
|
Less/ Add |
DEPRECIATION/
AMORTISATION (F) |
49.090 |
34.651 |
28.801 |
|
|
|
|
|
|
|
|
|
|
PROFIT / (LOSS)
BEFORE TAX (E-F) (G) |
1.540 |
6.641 |
(38.527) |
|
|
|
|
|
|
|
|
|
Less |
TAX (H) |
(17.130) |
0.987 |
1.416 |
|
|
|
|
|
|
|
|
|
|
PROFIT / (LOSS)
AFTER TAX (G-H) (I) |
18.670 |
5.654 |
(39.943) |
|
|
|
|
|
|
|
|
|
|
PREVIOUS
YEARS’ BALANCE BROUGHT FORWARD |
10.160 |
4.508 |
44.451 |
|
|
|
|
|
|
|
|
|
|
BALANCE CARRIED
TO THE B/S |
28.830 |
10.162 |
4.508 |
|
|
|
|
|
|
|
|
|
|
EARNINGS IN
FOREIGN CURRENCY |
|
|
|
|
|
|
|
FOB Value of Exports |
NA |
175.776 |
181.018 |
|
|
TOTAL EARNINGS |
NA |
175.776 |
181.018 |
|
|
|
|
|
|
|
|
|
|
IMPORTS |
|
|
|
|
|
|
|
Raw Materials |
NA |
68.027 |
49.548 |
|
|
|
Capital Goods |
NA |
15.173 |
4.506 |
|
|
TOTAL IMPORTS |
NA |
83.200 |
54.054 |
|
|
|
|
|
|
|
|
|
|
Earnings /
(Loss) Per Share (Rs.) |
5.14 |
1.63 |
(11.66) |
|
KEY RATIOS
|
PARTICULARS |
|
31.03.2013 |
31.03.2012 |
31.03.2011 |
|
PAT / Total Income |
(%) |
NA
|
1.20 |
(10.62) |
|
|
|
|
|
|
|
Net Profit Margin (PBT/Sales) |
(%) |
NA
|
10.62 |
(10.62) |
|
|
|
|
|
|
|
Return on Total Assets (PBT/Total Assets} |
(%) |
0.11
|
0.56 |
(6.46) |
|
|
|
|
|
|
|
Return on Investment (ROI) (PBT/Networth) |
|
0.00
|
0.01 |
(0.09) |
|
|
|
|
|
|
|
Debt Equity Ratio (Total Debt /Networth) |
|
0.53
|
0.36 |
0.16 |
|
|
|
|
|
|
|
Current Ratio (Current Asset/Current Liability) |
|
0.92
|
1.05 |
1.10 |
FINANCIAL ANALYSIS
[all figures are
in Rupees Millions]
DEBT EQUITY RATIO
|
Particular |
31.03.2011 |
31.03.2012 |
31.03.2013 |
|
|
(Rs.
In Millions) |
(Rs.
In Millions) |
(Rs.
In Millions) |
|
Share Capital |
34.261 |
36.291 |
36.291 |
|
Reserves & Surplus |
403.117 |
460.899 |
476.871 |
|
Net
worth |
437.378 |
497.190 |
513.162 |
|
|
|
|
|
|
long-term borrowings |
24.975 |
38.098 |
52.277 |
|
Short term borrowings |
45.131 |
142.116 |
221.154 |
|
Total
borrowings |
70.106 |
180.214 |
273.431 |
|
Debt/Equity
ratio |
0.160 |
0.362 |
0.533 |

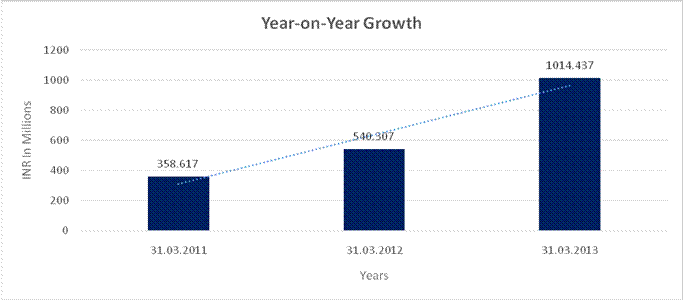
YEAR-ON-YEAR GROWTH
|
Year
on Year Growth |
31.03.2011 |
31.03.2012 |
31.03.2013 |
|
|
(Rs.
In Millions) |
(Rs.
In Millions) |
(Rs.
In Millions) |
|
Sales |
358.617 |
540.307 |
1014.437 |
|
|
|
50.664 |
87.752 |
NET PROFIT MARGIN
|
Net
Profit Margin |
31.03.2011 |
31.03.2012 |
31.03.2013 |
|
|
(Rs.
In Millions) |
(Rs.
In Millions) |
(Rs.
In Millions) |
|
Sales |
358.617 |
540.307 |
1014.437 |
|
Profit |
(39.943) |
5.654 |
18.67 |
|
|
(11.14%) |
1.05% |
1.84% |

LOCAL AGENCY FURTHER INFORMATION
|
Sr. No. |
Check List by Info Agents |
Available in
Report (Yes / No) |
|
1] |
Year of Establishment |
Yes |
|
2] |
Locality of the firm |
Yes |
|
3] |
Constitutions of the firm |
Yes |
|
4] |
Premises details |
No |
|
5] |
Type of Business |
Yes |
|
6] |
Line of Business |
Yes |
|
7] |
Promoter's background |
Yes |
|
8] |
No. of employees |
No |
|
9] |
Name of person contacted |
No |
|
10] |
Designation of contact
person |
No |
|
11] |
Turnover of firm for last
three years |
Yes |
|
12] |
Profitability for last
three years |
Yes |
|
13] |
Reasons for variation
<> 20% |
-- |
|
14] |
Estimation for coming
financial year |
No |
|
15] |
Capital in the business |
Yes |
|
16] |
Details of sister
concerns |
No |
|
17] |
Major suppliers |
No |
|
18] |
Major customers |
No |
|
19] |
Payments terms |
No |
|
20] |
Export / Import details
(if applicable) |
No |
|
21] |
Market information |
-- |
|
22] |
Litigations that the firm
/ promoter involved in |
-- |
|
23] |
Banking Details |
Yes |
|
24] |
Banking facility details |
Yes |
|
25] |
Conduct of the banking
account |
-- |
|
26] |
Buyer visit details |
-- |
|
27] |
Financials, if provided |
Yes |
|
28] |
Incorporation details, if
applicable |
Yes |
|
29] |
Last accounts filed at
ROC |
Yes |
|
30] |
Major Shareholders, if
available |
Yes |
|
31] |
Date of Birth of
Proprietor/Partner/Director, if available |
Yes |
|
32] |
PAN of Proprietor/Partner/Director,
if available |
No |
|
33] |
Voter ID No of
Proprietor/Partner/Director, if available |
No |
|
34] |
External Agency Rating,
if available |
Yes |
NOTE: Registered office of the company has been shifted from 120 Bhanu Enclave, Sundar Nagar, Esi, Hyderabad – 500038, Andhra Pradesh, India to the present address w.e.f 03.06.2006
CORPORATE INFORMATION:
Subject was
incorporated on November 20, 1995 and is engaged in the business of manufacturing
and marketing of Active Pharma Ingredients (APIs), drug intermediates, finished
dosage forms (FDF), and contract manufacturing of FDF.
The Company has three manufacturing facilities at Bollaram near
Hyderabad, at Vishakhapatnam and at Jadcherla.
BUSINESS REVIEW:
T Company has
achieved a turnover of Rs.1014.437 Millions for the year ended 31st
March 2013 compared to the previous year’s turnover of Rs. 587.404 Millions
representing a growth of 76%.
The Company has
made an EBITDA of Rs. 89 Millions (9 % of Turnover) for the year.
Besides the
manufacturing and marketing of Products developed in the own R N D, the company
has also taken up a new business model of contract manufacturing of APIs and
intermediates for the regulatory (US and Europe) markets for some of the
reputed Indian Companies.
The steps taken by
the management in expanding the product and customer base; and exploring new
business tie ups with new companies have started yielding results in the form
of business and revenues only in the last quarters of the year. The full impact
of these steps and the resultant revenues can be seen in the financial year
2013 - 14.
OUTLOOK:
APIs:
Two of the APIs,
for which the Company has received the EU approval, are going off patented in
Feb/March 2014. Considering the Company’s strength in these two APIs, the
Company expects substantial revenues from these two APIs in 2014-15. Company
continues to increase its market share in its two old APIs namely Amlodipine
and Cetrizine and the Company expects to increase the revenues from these two
products further in the coming years.
Company has
developed a new process for the manufacture of Clopidrel in the in house RND
and will be filing the DMF for Clopidogrel with the new process developed, in
the current year, and with this the Company expects to regain the market share
for this product in the Regulatory Markets.
FINISHED DOSAGE FORMS:
Company has
received the market authorization for its first product Levo-Cetrizine during
the year. Market Authorization/Approvals are expected to be received for some
more products in 2013-14 and 2014-15. Revenues from these products will be
significant in 2014-15.
The site variation
applications filed by few of its customers in UK have been approved during the
year. The Company’s major customer from Ukraine has also received approval for
the site variation application filed by them for several of its products. The
contracts signed with all these customers are expected to generate significant
revenues to the Company from Sept/Oct 2013 and are expected to grow by more
than 20% every year for the next 2/3 years.
Sale of
Formulations Plant at Jadcherla
The Company has
entered into a Business Transfer Agreement (BTA) with Mylan Laboratories Limited
(Mylan) on February 1, 2013 to transfer its formulation plant situated at Plot
no. S 16, S 17A, Green Industrial Park - APIIC, SEZ Jedcherla, Mahaboobnagar
District, AndhraPradesh herein after referred as Jadcherla unit subject to
various approvals. Foreign Investment Promotion Board (India) has approved the
transaction in its meeting held on May 10, 2013, and approvals from other
authorities have also been obtained. As a part of the agreement, the Company
shall transfer the leasehold land, the manufacturing facility, fixed assets and
all the employees of the formulations plant except R and D employees,
Intangible assets i.e., dossiers namely exclusive license for Raloxifene (US)
and non-exclusive license for Raloxifene (Europe) and Levocetirizine (Europe)
to Mylan The total consideration for the transfer is Rs. 850.000 Millions. The
Company has received an amount of Rs 85.000 Millions as an advance from Mylan
before March 31, 2013 and is disclosed as an advance for sale of unit.
Subsequent to the
end of the financial year, on August 13, 2013, the Company has signed
conveyance deed and the transfer of the plant has been completed. Upon
execution of conveyance deed, Gross book value of fixed assets amounting to Rs
405.900 Millions and all employees except Research and development employees
were transferred to Mylan and the Company received the balance consideration
amount.
Investment in
Optimus Generics Limited A Joint Venture
(JV) formed for the Formulations manufacturing
The Company has
set up a 51% subsidiary, Optimus Generics Limited (the Company), to carry on
the business of manufacturing oral dosageforms (tablets/capsules/sachets) for
generic products and also engage in research and development activities. The
total project cost of setting up a new facility is estimated to be about Rs
800.000 Millions. The project implementation has started in July, 2013 and may
end by December, 2014.
Commercial
production is expected to commence by 2 nd half of 2015 after receiving all
regulatory approvals.
Continuity of
Formulations business and the liquidity to the Company For the short to medium
term, till the new facility under the JV is ready, Glochem Industries Limited.
has an arrangement with Mylan Laboratories Limited. to manufacture at the sold
FDF The sold FDF division contributed to the revenues from the last financial
year only, and the economics of the Formulations Business will be continued at
Glochem till the new plant at the JV is operational as per the terms of the JV.
After the new plant is operational, the expected revenues from the JV will see
a significant jump, and the share in the JV economics will more than offset the
loss from discontinuing FDF business at Glochem.
Hence, the sale of
the FDF plant plus the manufacturing arrangement with Mylan, and the new JV for
a new FDF facility, will together ensure that the company has immediate
liquidity to pursue its R and D plans, plus secure continuity of the FDF
business.
Fire accident at
API Plant located at Vishakhapatnam
A fire accident
occurred at the Company’s API facility situated at Vishakhapatnam on May 30,
2013. The fire accident at Vizag unit has resulted in substantial loss of stock
and damaged 2 out of the 5 production blocks.
The estimated loss
from the accident is about Rs. 340.000 Millions of which loss on account of
inventories is estimated to be Rs. 270.000 Millions and loss of plant and
machinery and buildings is estimated to be Rs. 70.000 Millions. There is no
major loss of customers / orders for the Company because of fire.
Loss on fixed
assets and inventories are adequately covered and do not expect any material
deductibles. The Company has lodged a claim with the insurance company and
requested for an on account payment. The Claim has been admitted by the Insurer
(Oriental Insurance Company). The estimates of loss will be revised for changes
in valuation of stocks and once the actual purchase orders for various
equipments (being replaced) are finalised, a final claim will be lodged along
with other documents required by the insurer.
Resumption of
Operations at the Company’s Vizag Facility
The construction
work has begun at the Vizag plant and the Company expects to begin
manufacturing in two of the blocks by the end of September, 2013 and gradually
the entire plant will become operational by November. As such, it is a
short-term break in operating capacity and therefore the financial statement of
the company has been prepared under going concern basis.
Capital work in
Progress and Intangible assets (Dossier Development Expenses) Capital
Work-in-Progress and Intangible assets under development amount to Rs. 63.600
Millions The Company has commercialized one dossier (Nortrypoyline HCL EU) and
the product sales were made to NRIM Limited amounting to Rs. 0.470 Millions. As
per the accounting policy of the company, the dossier on commercialization is
being amortized over the period of 5 years. The total cost incurred on this
dossier development is Rs.0.100 Million.
INDEX
OF CHARGES
|
S.NO. |
CHARGE ID |
DATE OF CHARGE CREATION/MODIFICATION
|
CHARGE AMOUNT
SECURED |
CHARGE HOLDER |
ADDRESS |
SERVICE REQUEST
NUMBER (SRN) |
|
1 |
10396629 |
13/12/2012 |
13,000,000.00 |
PUNJAB NATIONAL BANK |
HI-TECH.CITY BRANCH,
1Q4-A3,1ST FLOOR,CYBER TOWERS, MADHAPUR, HYDERABAD, ANDHRA PRADESH - 500081,
I |
B65771081 |
|
2 |
10357060 |
04/10/2013 * |
243,800,000.00 |
PUNJAB NATIONAL BANK (ON BEHALF OF PNB
CONSORTIUM |
HI-TEC.CITY
BRANCH, 1Q4-A3,1ST FLOOR,CYBER TOWERS, |
B89481824 |
|
3 |
90116056 |
04/10/2013 * |
388,000,000.00 |
PUNJAB NATIONAL BANK (ON BEHALF OF PNB
CONSORTIUM |
HI-TEC.CITY
BRANCH, 1Q4-A3,1ST FLOOR,CYBER TOWERS, |
B89484067 |
|
4 |
90119034 |
15/12/2002 * |
7,600,000.00 |
PUNJAB NATIONAL BANK |
1Q4 A3; CYBER TOWERS,
HITECH CITY; MADHAPUR, HAYD |
- |
|
5 |
90259724 |
27/07/2013 * |
253,000,000.00 |
AXIS BANK LIMITED |
GROUND AND IST
FLOOR,6-3-879/B,G.PULLAREDDY BUILDING, GREENLANDS,BEGUMPET ROAD,, HYDERABAD, ANDHRA
PRADESH - 500016, INDIA |
B80562804 |
|
6 |
90115356 |
29/12/2003 * |
6,000,000.00 |
UTI BANK LIMITED |
BEGUMPET,
HAYDERABAD, ANDHRA PRADESH, INDIA |
- |
Note: * Date of charge modification
FIXED ASSETS
Tangible assets
- Land
- Buildings
- Office building
- Plant and
equipment
- Factory
equipment’s
- Furniture and
fixtures
- Vehicles
- Motor
vehicles
- Office
equipment
- Computer
equipment’s
Intangible assets
- Computer
software
CMT REPORT (Corruption, Money Laundering & Terrorism]
The Public Notice information has been collected from various sources
including but not limited to: The Courts,
1] INFORMATION ON
DESIGNATED PARTY
No exist designating subject or any of its beneficial owners,
controlling shareholders or senior officers as terrorist or terrorist
organization or whom notice had been received that all financial transactions
involving their assets have been blocked or convicted, found guilty or against
whom a judgement or order had been entered in a proceedings for violating
money-laundering, anti-corruption or bribery or international economic or
anti-terrorism sanction laws or whose assets were seized, blocked, frozen or
ordered forfeited for violation of money laundering or international
anti-terrorism laws.
2] Court Declaration :
No exist to suggest that subject is or was
the subject of any formal or informal allegations, prosecutions or other
official proceeding for making any prohibited payments or other improper
payments to government officials for engaging in prohibited transactions or
with designated parties.
3] Asset Declaration :
No records exist to suggest that the property or assets of the subject
are derived from criminal conduct or a prohibited transaction.
4] Record on Financial
Crime :
Charges or conviction
registered against subject: None
5] Records on Violation of
Anti-Corruption Laws :
Charges or
investigation registered against subject: None
6] Records on Int’l
Anti-Money Laundering Laws/Standards :
Charges or investigation
registered against subject: None
7] Criminal Records
No
available information exist that suggest that subject or any of its principals
have been formally charged or convicted by a competent governmental authority
for any financial crime or under any formal investigation by a competent
government authority for any violation of anti-corruption laws or international
anti-money laundering laws or standard.
8] Affiliation with
Government :
No record
exists to suggest that any director or indirect owners, controlling
shareholders, director, officer or employee of the company is a government
official or a family member or close business associate of a Government
official.
9] Compensation Package :
Our market
survey revealed that the amount of compensation sought by the subject is fair
and reasonable and comparable to compensation paid to others for similar
services.
10] Press Report :
No press reports / filings exists on
the subject.
CORPORATE GOVERNANCE
MIRA INFORM as part of its Due Diligence do provide comments on
Corporate Governance to identify management and governance. These factors often
have been predictive and in some cases have created vulnerabilities to credit
deterioration.
Our Governance Assessment focuses principally on the interactions
between a company’s management, its Board of Directors, Shareholders and other
financial stakeholders.
CONTRAVENTION
Subject is not known to have contravened any existing local laws,
regulations or policies that prohibit, restrict or otherwise affect the terms
and conditions that could be included in the agreement with the subject.
FOREIGN EXCHANGE RATES
|
Currency |
Unit
|
Indian Rupees |
|
US Dollar |
1 |
Rs.60.13 |
|
|
1 |
Rs.99.65 |
|
Euro |
1 |
Rs.82.91 |
INFORMATION DETAILS
|
Information
Gathered by : |
NYA |
|
|
|
|
Report Prepared
by : |
KVT |
SCORE & RATING EXPLANATIONS
|
SCORE FACTORS |
RANGE |
POINTS |
|
HISTORY |
1~10 |
4 |
|
PAID-UP CAPITAL |
1~10 |
4 |
|
OPERATING SCALE |
1~10 |
4 |
|
FINANCIAL CONDITION |
|
|
|
--BUSINESS SCALE |
1~10 |
4 |
|
--PROFITABILIRY |
1~10 |
3 |
|
--LIQUIDITY |
1~10 |
3 |
|
--LEVERAGE |
1~10 |
4 |
|
--RESERVES |
1~10 |
4 |
|
--CREDIT LINES |
1~10 |
3 |
|
--MARGINS |
-5~5 |
|
|
DEMERIT POINTS |
|
|
|
--BANK CHARGES |
YES/NO |
YES |
|
--LITIGATION |
YES/NO |
NO |
|
--OTHER ADVERSE INFORMATION |
YES/NO |
NO |
|
MERIT POINTS |
|
|
|
--SOLE DISTRIBUTORSHIP |
YES/NO |
NO |
|
--EXPORT ACTIVITIES |
YES/NO |
NO |
|
--AFFILIATION |
YES/NO |
NO |
|
--LISTED |
YES/NO |
NO |
|
--OTHER MERIT FACTORS |
YES/NO |
YES |
|
DEFAULTER |
|
|
|
--RBI |
YES/NO |
NO |
|
--EPF |
YES/NO |
NO |
|
TOTAL |
|
33 |
This score serves as a reference to assess
SC’s credit risk and to set the amount of credit to be extended. It is calculated
from a composite of weighted scores obtained from each of the major sections of
this report. The assessed factors and their relative weights (as indicated
through %) are as follows:
Financial
condition (40%) Ownership
background (20%) Payment record
(10%)
Credit history
(10%) Market trend (10%) Operational size
(10%)
RATING EXPLANATIONS
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
>86 |
Aaa |
Possesses an extremely sound financial base with the strongest capability
for timely payment of interest and principal sums |
Unlimited |
|
71-85 |
Aa |
Possesses adequate working capital. No caution needed for credit
transaction. It has above average (strong) capability for payment of interest
and principal sums |
Large |
|
56-70 |
A |
Financial & operational base are regarded healthy. General
unfavourable factors will not cause fatal effect. Satisfactory capability for
payment of interest and principal sums |
Fairly Large |
|
41-55 |
Ba |
Overall operation is considered normal. Capable to meet normal
commitments. |
Satisfactory |
|
26-40 |
B |
Capability to overcome financial difficulties seems comparatively
below average. |
Small |
|
11-25 |
Ca |
Adverse factors are apparent. Repayment of interest and principal sums
in default or expected to be in default upon maturity |
Limited with
full security |
|
<10 |
C |
Absolute credit risk exists. Caution needed to be exercised |
Credit not
recommended |
|
-- |
NB |
New Business |
-- |
This report is issued at your request without any
risk and responsibility on the part of MIRA INFORM PRIVATE LIMITED (MIPL)
or its officials.