MIRA
INFORM REPORT
|
Report Date : |
05.05.2014 |
IDENTIFICATION DETAILS
|
Name : |
STRIDES ARCOLAB LIMITED |
|
|
|
|
Registered
Office : |
201, Devavrata, Sector 17, Vashi,
Navi Mumbai – 400705, Maharashtra |
|
|
|
|
Country : |
India |
|
|
|
|
Financials (as
on) : |
31.12.2012 |
|
|
|
|
Date of
Incorporation : |
28.06.1990 |
|
|
|
|
Com. Reg. No.: |
11-057062 |
|
|
|
|
Capital
Investment / Paid-up Capital : |
Rs. 588.040
Millions |
|
|
|
|
CIN No.: [Company Identification
No.] |
L24230MH1990PLC057062 |
|
|
|
|
TAN No.: [Tax Deduction &
Collection Account No.] |
MUMS36534B |
|
|
|
|
PAN No.: [Permanent Account No.] |
AADCS8104P |
|
|
|
|
Legal Form : |
A Public Limited Liability Company. The Company’s Shares
are Listed on the Stock Exchanges. |
|
|
|
|
Line of Business
: |
Manufacturing and Marketing of all types of Bulk Drugs,
Pharmaceuticals, etc. |
|
|
|
|
No. of Employees
: |
2700 (Approximately) |
RATING & COMMENTS
|
MIRA’s Rating : |
Ba (49) |
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
41-55 |
Ba |
Overall operation is considered normal. Capable to meet normal
commitments. |
Satisfactory |
|
Maximum Credit Limit : |
USD 54860000 |
|
|
|
|
Status : |
Good |
|
|
|
|
Payment Behaviour : |
Regular |
|
|
|
|
Litigation : |
Exist |
|
|
|
|
Comments : |
Subject is an established company having a fine track record. Management of the company has failed to file its financial for the
year 2013 with the government department. As per available financials (2012), There are dip in profit of the
company. However, liquidity position of the company seems to be strong. Trade relations are reported to be fair. Business is active. Payment
terms are reported to be regular and as per commitments. The company can be considered for normal business dealings at usual
trade terms and conditions. |
NOTES :
Any query related to this report can be made
on e-mail : infodept@mirainform.com
while quoting report number, name and date.
ECGC Country Risk Classification List – March 31, 2014
|
Country Name |
Previous Rating (31.12.2013) |
Current Rating (31.03.2014) |
|
India |
A1 |
A1 |
|
Risk Category |
ECGC
Classification |
|
Insignificant |
A1 |
|
Low |
A2 |
|
Moderate |
B1 |
|
High |
B2 |
|
Very High |
C1 |
|
Restricted |
C2 |
|
Off-credit |
D |
INDIAN ECONOMIC OVERVIEW
US investment bank
Goldman Sachs has upgraded its outlook on Indian markets as it expects
positive impact of the election cycle.
India’s economy may
grow 4.7 % in the current financial year, lower than the official estimate of 4.9
%, Fitch Rating said. The global rating agency expects the economy to pick up
in the next two financial years.
Global ratings
agency Standard & Poor said increasing focus by India Inc on lowering debt
is likely to improve their credit profiles.
Singapore (1.1
million Indian tourists in 2012), Thailand (one million), the United Arab
Emirates ().98 million) and Malaysia ().82 million) emerged as the preferred
holidays hotspots for Indians. The total figure is expected to increase to 1.93
million by 2017, according to the latest Eurmonitor international report.
There is a $29.34 bn
outward foreign direct investment by domestic companies between April and
January of 2013/14 which has seen some signs of recovery according to a Care
Ratings report.
There are 264 number
of new companies being set up every day on average during 2014. Most of them
are registered in Mumbai. India had 1.38 million registered companies at the
end of January, 2014.
Twitter like
messaging service Weibo Corporation has filed to raise $ 500 million via a US
initial public offering. Alibaba, which owns a stake in Weibo is expected to
raise about $ 15 billion New York this year in the highest profile Internet IPO
since Facebook’s in 2012.
Bharti Airtel has
raised Rs.2,453.2 crore (350 million Swiss Francs) by selling six-year bonds at
a coupon rate of three per cent and maturing in 2020. This is the largest ever
bond offering by an Indian company in Swiss Francs. Bharat Petroleum
Corporation raised 175 million Swiss Francs by selling five year bonds at 2.98
% coupon rate in February.
Indian Oil
Corporation plans to invest Rs 7650 crore in setting up a petrochemical complex
at its almost complete Paradip refinery in Odhisha in three to four years. The
company board is set to consider the setting up of a 700000 tonne per annum
polypropylene plant at an estimated cost at Rs.3150 crore.
Global chief
information officers at gathering in Bangalore in April to meet Indian startups
at an event called Tech50 Watchout for Little Eye Labs-Facebook type deals in
the making.
EXTERNAL AGENCY RATING
|
Rating Agency Name |
ICRA |
|
Rating |
Term Loan Facilities = A+ |
|
Rating Explanation |
Having adequate degree of safety and carry low credit risk |
|
Date |
March, 2014 |
|
Rating Agency Name |
ICRA |
|
Rating |
Fund Based Facilities = A4+ |
|
Rating Explanation |
Having very strong degree of safety and carry lowest credit risk. |
|
Date |
March, 2014 |
RBI DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available RBI Defaulters’ list.
EPF (Employee Provident Fund) DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available EPF (Employee Provident Fund) Defaulters’ list as of
31-03-2012.
INFORMATION DECLINED BY
|
Name : |
Ms. Manjula |
|
Designation : |
Company Secretary |
|
Contact No.: |
91-22-27893199 |
|
Date : |
20.03.2014 |
LOCATIONS
|
Registered Office : |
201, Devavrata, Sector 17, Vashi,
Navi Mumbai – 400705, Maharashtra, India |
|
Tel. No.: |
91-22-27892924 / 27893199 |
|
Fax No.: |
91-22-27892942 |
|
E-Mail : |
|
|
Website : |
|
|
|
|
|
Corporate Office : |
Strides House, Bilekahalli, Bannerghatta Road, Bangalore –
560076, Karnataka, India |
|
Tel. No.: |
91-80-26581343/ 44/ 67580738/
39/ 67580000/ 66580751/ 66580000/ 66580600 |
|
Fax No.: |
91-80-26583538/ 4330/ 67580700/
800/ 66580800 |
|
E-Mail : |
|
|
|
|
|
R and D Centre |
Strides Technology And Research, Bilekahalli, Bannerghatta
Road, Bangalore 560076, Karnataka, India |
|
Tel. No.: |
91-80-67840290 |
|
Fax No.: |
91-80-66580200/300 |
|
|
|
|
GLOBAL PLANTS : |
|
|
Factory 1 : |
Sterile Products Division – I Bilekahalli, Bannerghatta
Road, Bangalore - 560076, Karnataka, India |
|
|
|
|
Factory 2 : |
Penicillins Facility Estrada Doutor
Lorival Martins Beda, 926 – 968 28110-000- Donana – Campos dos, Goytacazes-
Rio de Janeiro- Brazil |
|
|
|
|
Factory 3 : |
Sterile Products Division – II Plot No. 284-A,
Bommasandra Jigani Link Road, Industrial Area, Jigani Village, Jigani, Hobli,
Anekal Taluk, Bangalore - 562106, Karnataka, India |
|
|
|
|
Factory 4 : |
Strides Arcolab Polska Sp.Zo.o ul. Daniszewska
10 03-230 Warszawa NIP-813-34-15-000, Poland |
|
|
|
|
Factory 5 : |
Oral Dosage Form Facility – III Plot No. 9-12,
Dewan and Sons Industrial Area, Veroor, Palghar, District Thane - 401404,
Maharashtra, India |
|
|
|
|
Factory 6 : |
Onco Therapies Limited Plot No. 284-B,
Bommasandra Jigani Link Road, Industrial Area, Jigani Village, Jigani Hobli,
Anekal Taluk, Bangalore - 562106, Karnataka, India |
|
|
|
|
Factory 7 : |
Strides Vital Nigeria Limited Gate No. 02,
Ladipo Oluwole Avenue, Opposite Cocoa Warehouse, Off Oba Akran Road, Ikeja
Industrial Area, Ikeja Lagos, Nigeria |
|
|
|
|
Factory 8 : |
Beta-lactams
Facility Bilekahalli, Bannerghatta Road, Bangalore-560076,
Karnataka, India |
|
|
|
|
Factory 9 : |
Beltapharm SpA 20095 Cusano MIL.
(MI) – Via Stelvio, 66 Italy. |
|
|
|
|
Factory 10 : |
Penems Facility Estrada Doutor
Lorival Martins Beda, 926 – 968 28110-000- Donana – Campos dos, Goytacazes-
Rio de Janeiro- Brazil |
|
|
|
|
Factory 11 : |
Oral Dosage Form Facility – II 'KRS Gardens', Suragajakanahalli, Anekal
Taluk, Bangalore - 560106, Karnataka, India |
|
|
|
|
Factory 12 : |
Oral Dosage Form Facility – I 124, Sipcot Industrial Complex, Hosur - 635126, Tamilnadu, India |
|
|
|
|
Factory 13 : |
Beta-lactams Facility Bilekahalli, Bannerghatta Road, Bangalore - 560076, Karnataka, India |
|
|
|
|
Factory 14 : |
Star Drugs and Research Labs Limited Plot No. 14, Sipcot-II, Hosur -
635109, Tamilnadu, India |
|
|
|
|
Factory 15 : |
Cephalosporins Facility Bilekahalli, Bannerghatta Road,
Bangalore - 560076, Karnataka, India |
|
|
|
|
Factory 16 : |
Penems Facility Estrada DoutorLorival Martins
Beda, 926 - 968 28110-000- Donana - Campos
dos Goytacazes- Rio de Janeiro- Brazil |
|
|
|
|
Warehouse : |
Plot No. 62, Sector – 1, Nerul, Navi Mumbai – 400706, Maharashtra,
India |
|
|
|
|
Global Offices : |
Located at : USA
South Africa 4, Angus Cresent, Longmeadow East, Modderfontein-1644, Norway Sorkedalsveien,
10B 0369, Oslo, Norway. United
Kingdom Unit 4,
Metro Centre, Tolpits Lane,Watford, Hertfordshire, WD18 9SS, UK Singapore 8 Cross Street, No. 17-00 Singapore 048424 |
DIRECTORS
AS ON 31.12.2012
|
Name : |
Mr. Deepak Vaidya |
|
Designation : |
Chairman and Independent Director |
|
|
|
|
Name : |
Mr. Arun Kumar |
|
Designation : |
Executive Vice Chairman and Managing Director |
|
Qualification |
B.Com., PGDBM |
|
|
|
|
Name : |
Mr. K.R. Ravishankar |
|
Designation : |
Non-Executive Director |
|
Qualification |
B.Sc. (Part) |
|
|
|
|
Name : |
Mr. Mukul Sarkar |
|
Designation : |
Independent Director |
|
|
|
|
Name : |
Mr. P M Thampi |
|
Designation : |
Independent Director |
|
|
|
|
Name : |
Mr. Venkat S Iyer |
|
Designation : |
Director |
|
|
|
|
Name : |
Mr. M.R. Umarji |
|
Designation : |
Independent Director |
|
|
|
|
Name : |
Mr. A.K. Nair |
|
Designation : |
Independent Director |
|
|
|
|
Name : |
Mr. S Sridhar |
|
Designation : |
Independent Director |
KEY EXECUTIVES
|
Name : |
Mr. Arun Kumar |
|
Designation : |
Founder and Group Chief Executive Officer |
|
|
|
|
Name : |
Mr. Venkat S Iyer |
|
Designation : |
Executive Director and Chief Executive
Officer – Agila |
|
|
|
|
Name : |
Mr. T. S. Rangan |
|
Designation : |
Group Chief Executive
Officer |
|
|
|
|
Name : |
Mr. Adam Levitt |
|
Designation : |
Chief Executive Officer Americas Operations |
|
|
|
|
Name : |
Dr. Anand Iyer |
|
Designation : |
Chief Executive Officer, Agila Biotech Division |
|
|
|
|
Name : |
Mr. D P Shrivastava |
|
Designation : |
Chief Executive Officer, Brazil
|
|
|
|
|
Name : |
Mr. Manish Gupta |
|
Designation : |
Chief Executive Officer – pharma |
|
|
|
|
Name : |
Mr. Subroto Banerjee |
|
Designation : |
President, Agila (India Region) |
|
|
|
|
Name : |
Mr. Sihue B Noronha |
|
Designation : |
Chief Executive Officer – Africa |
|
|
|
|
Name : |
Mr. Joe Thomas |
|
Designation : |
Chief Corporate Development Officer |
|
|
|
|
Name : |
Mr. Venkatesh |
|
Designation : |
Account Manager |
|
|
|
|
Name : |
Ms. Manjula |
|
Designation : |
Company Secretary |
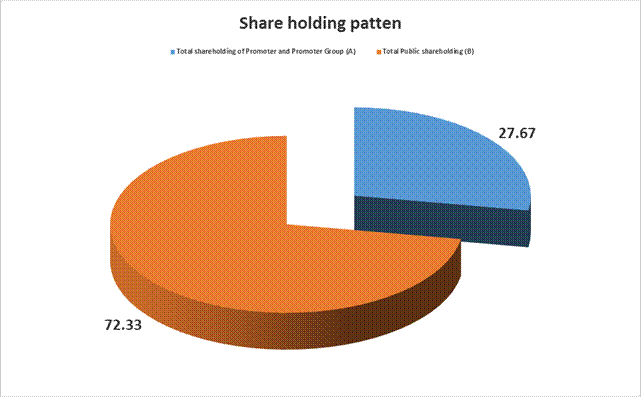
MAJOR SHAREHOLDERS / SHAREHOLDING PATTERN
AS ON 31.03.2014
|
Category
of Shareholder |
No. of Shares |
Percentage of
Holding |
|
(A) Shareholding of Promoter and Promoter Group |
|
|
|
|
|
|
|
|
3387326 |
5.69 |
|
|
13096876 |
21.99 |
|
|
16484202 |
27.67 |
|
|
|
|
|
Total shareholding of Promoter and Promoter Group (A) |
16484202 |
27.67 |
|
(B) Public Shareholding |
|
|
|
|
|
|
|
|
1372429 |
2.30 |
|
|
26386 |
0.04 |
|
|
105554 |
0.18 |
|
|
29343041 |
49.26 |
|
|
97263 |
0.16 |
|
|
30944673 |
51.95 |
|
|
|
|
|
|
2198471 |
3.69 |
|
|
|
|
|
|
4340495 |
7.29 |
|
|
1864023 |
3.13 |
|
|
5000 |
0.01 |
|
|
3728757 |
6.26 |
|
|
2475284 |
4.16 |
|
|
254509 |
0.43 |
|
|
290750 |
0.49 |
|
|
381834 |
0.64 |
|
|
305000 |
0.51 |
|
|
21005 |
0.04 |
|
|
375 |
0.00 |
|
|
12136746 |
20.38 |
|
Total Public shareholding (B) |
43081419 |
72.33 |
|
Total (A)+(B) |
59565621 |
100.00 |
|
(C) Shares held by Custodians and against which Depository
Receipts have been issued |
0 |
0.00 |
|
|
0 |
0.00 |
|
|
0 |
0.00 |
|
|
0 |
0.00 |
|
Total (A)+(B)+(C) |
59565621 |
0.00 |
BUSINESS DETAILS
|
Line of Business : |
Manufacturing and Marketing of all types of Bulk Drugs,
Pharmaceuticals, etc. |
||||||||
|
|
|
||||||||
|
Products : |
|
PRODUCTION STATUS (AS ON 31.12.2011)
|
Particulars |
Unit |
Installed
Capacity |
Actual
Production |
|
Soft Gelatin Plant Softgel Capsules |
Numbers
in Millions |
2645 |
-- |
|
Hard Gelatin Plant Capsules |
Numbers
in Millions |
450 |
699734 |
|
Tablet Plant Tablets |
Numbers
in Millions |
2160 |
2115452 |
|
Others |
Numbers
in Millions |
-- |
2105 |
Note:
Installed Capacities
are as certified by the management and relied upon by the Auditors. The
installed capacities serve multiple purposes and will vary according to product
mix.
** Not applicable as the products have been de-licensed.
GENERAL INFORMATION
|
No. of Employees : |
2700 (Approximately) |
|||||||||||||||||||||||||||
|
|
|
|||||||||||||||||||||||||||
|
Bankers : |
· Axis Bank Limited · Central Bank of India · HDFC Bank Limited · Indian Overseas Bank · Ratnakar Bank Limited · Syndicate Bank · Yes Bank Limited · Citi Bank · Exim Bank |
|||||||||||||||||||||||||||
|
|
|
|||||||||||||||||||||||||||
|
Facilities : |
|
|||||||||||||||||||||||||||
|
|
|
|
Banking
Relations : |
-- |
|
|
|
|
Statutory Auditors : |
|
|
Name : |
Deloitte Haskins and Sells Chartered Accountants |
|
Address : |
Deloitte Centre, Anchorage II, 100/2, Richmond Road,
Bangalore – 560025, Karnataka, India |
|
Tel. No.: |
91-80-66276000 |
|
Fax No.: |
91-80-66276011 |
|
|
|
|
Internal Auditors : |
|
|
Name : |
Grant Thornton International Chartered Accountants |
|
Address : |
Wings, 1st Floor, 16/1, Cambridge Road, Halasuru,
Bangalore-560008, Karnataka, India |
|
|
|
|
Wholly Owned Subsidiaries : |
Direct Holding · Arcolab Limited SA, Switzerland · Agila Biotech Private Limited, India (w.e.f. May 16, 2012) · Agila Specialties Asia Pte. Limited, Singapore (w.e.f. August 4, 2012) · Agila Specialties Private Limited, India · Agila Specialties Limited (formerly Starsmore Limited), Cyprus · Strides Africa Limited, British Virgin Islands (upto December 25, 2012) · Strides Arcolab International Limited, U.K (SAIL) · Strides Emerging Markets Private Limited, India (w.e.f. June 1, 2012 upto November7, 2012) · Strides Pharma International Limited, Cyprus (formerly Strides Specialty (Cyprus) Limited) · Strides Technology and Research Private Limited, India (Under the process of striking off) Indirect Holding · Agila Australasia Pty Limited, Australia (w.e.f. March 22, 2012) · Agila Biotech (Malaysia) SDN BHD, Malaysia (Formerly Agila Specialties (Malaysia) SDN BHD) · Agila (NZ) Pty Limited, New Zealand (w.e.f. February 8, 2012) · Agila Pharma Canada Corporation, Canada (formerly Pharma Strides Canada Corporation) · Agila Specialties Americas Limited, Cyprus (w.e.f. September 28, 2012) (Formerly Agila Specialties Latina Limited) · Agila Specialties Asia Pte. Limited, Singapore (upto August 3, 2012) · Agila Specialties Global Pte. Limited, Singapore (w.e.f. September 28, 2012) · Agila Specialties UK Limited, UK (w.e.f. December 14, 2012) · Agila Specialties Polska Sp. Z.o.o, Poland (Formerly Strides Arcolab Polska Sp.Z.o.o.) · Co Pharma Limited, UK · Farma Plus AS , Norway · Onco Laboratories Limited, Cyprus · Onco Therapies Limited, India · Plus Farma ehfi, Iceland · Scentia Pharmaceuticals Pty Limited, Australia (formerly Linkace Investments Pty Limited), · Strides Africa Limited, British Virgin Islands (w.e.f. December 26, 2012) Strides Australia Pty Limited, Australia · Strides Emerging Markets Private Limited, India (w.e.f. November 8, 2012) · Strides Inc., USA · Strides Pharma (Cyprus) Limited, Cyprus (upto June 28, 2012) Strides Pharma Limited, Cyprus (formerly Linkace Limited) · Strides Pharmaceuticals (Holdings) Limited, Mauritius · Strides Pharmaceuticals (Mauritius) Limited, Mauritius · Strides S.A. Pharmaceuticals Pty. Limited, South Africa (w.e.f. December31, 2012) · Strides Specialties (Holdings) Cyprus Limited, Cyprus · Strides Specialties (Holdings) Limited, Mauritius |
|
|
|
|
Other Subsidiaries : |
Indirect Holding · African Pharmaceuticals Development Company, Cameroon · Agila Marketing e distribicao de Productos Hospitalaries Limited. (formerly Ephos – 106 Produtos Hospitalaries Limited Me), Brazil · Beltapharm S.p.A., Italy · Congo Pharma SPRL, Congo · Inbiopro Solutions Private Limited, India · Sorepharm SA, Burkinofaso · SPC Company Limited, Sudan · Strides CIS Limited, Cyprus · Strides Farmaceutica Participacoes Limited, Brazil · Agila Jamp Canada Inc., Canada (w.e.f. March 20, 2012) · Strides Pharma Cameroon Limited, Cameroon · Strides Pharma (Cyprus) Limited, Cyprus · Strides Pharma Namibia (Pty) Limited, Namibia · Strides S.A. Pharmaceuticals Pty. Limited, South Africa · Strides Vital Nigeria Limited, Nigeria · Ascent Pharmahealth Asia Pte., Limited, Singapore · Ascent Pharma Pty Limited (formerly Genepharm Pty Limited), Australia · Ascent Pharmacy Services Pty Limited, Australia · Ascent Pharmaceuticals Limited (formerly Genepharm (New Zealand) Limited), New Zealand · Ascent Pharmahealth Asia (Hong Kong) Limited (formerly Strides Arcolab Hong Kong Limited), Hong Kong · Ascent Pharmahealth Asia (Malaysia) SDN BHD (formerly Strides Arcolab Malaysia SDN. BHD) , Malaysia · Drug Houses of Australia (Asia) Pte. Limited, Singapore · Pharmasave Australia Pty Limited, Australia |
|
|
|
|
Joint Ventures : |
· Akorn Strides LLC, USA · Sagent Agila LLC, USA |
|
|
|
|
Enterprises owned or significantly influenced by
key management personnel and relative of key management personnel : |
· Atma Projects, India · Agnus Holdings Private Limited, India · Agnus Global Holdings Pte Limited, Singapore · Agnus IPCO Limited, BVI · Mandala Valley Vineyards Private Limited, India · Nous Infosytems Private Limited, India · Patsys Consulting Private Limited, India · Santo Properties Private Limited, India · Sequent Scientific Limited, India · Sequent Research Limited, India · Sequent Penems Private Limited, India · Sequent Global Holdings Limited, Mauritius · Sequent Antibiotics (Private) Limited, India · Sequent Oncolytics (Private) Limited, India · Skanray Healthcare Private Limited, India (Formerly known as Triumph Fincap Ventures Private Limited) · Karuna Ventures Private Limited, India · Paradime Infrastructure Development Company · Deesha Properties, India · Agnus Capital LLP, India · Atma Enterprises LLP India · Chayadeep Ventures LLP India · Qualichem Remedies LLP India · Triumph Venture Holdings LLP, India · Chayadeep Properties Private Limited, India · Higher Pharmatech Private Limited, India · Pronomz Ventures LLP, India |
CAPITAL STRUCTURE
AS ON 31.12.2012
Authorised Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
89750000 |
Equity Shares |
Rs.10/- each |
Rs. 897.500 Millions |
|
620000 |
Cumulative Redeemable Preference Shares |
Rs.1000/- each |
Rs. 620.000 Millions |
|
|
TOTAL |
|
Rs. 1517.500
Millions |
Issued, Subscribed & Paid-up Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
58803721 |
Equity Shares |
Rs.10/- each |
Rs. 588.040
Millions |
|
|
|
|
|
NOTE:
Reconciliation of the number of shares and amount outstanding at the beginning
and at the end of the reporting period:
|
PARTICULARS |
31.12.2012 |
|
|
|
No. of shares |
(Rs. In Millions) |
|
Equity share of Rs.10/- each |
|
|
|
Opening balance |
58380171 |
583.800 |
|
Issued pursuant to employee stock option
plan |
423550 |
4.240 |
|
Closing balance |
58803721 |
588.040 |
Detail of the rights, preferences and restrictions attaching
to each class of shares outstanding Equity shares of Rs. 10/- each:
The Company has only one class
of equity shares, having a par value of Rs.10/-. The holder of equity shares is
entitled to one vote per share. The Company declares and pays dividends in
Indian rupees. The dividend proposed by the Board of Directors is subject to
approval by the shareholders at the ensuing Annual General Meeting. In the
event of liquidation of the Company, the holders of the equity shares will be
entitled to receive any of the remaining assets of the Company, after
distribution to all other parties concerned. The distribution will be in
proportion to number of equity shares held by the shareholders.
Details of equity shares held by each
shareholder holding more than 5% of shares:
|
PARTICULARS |
31.12.2012 |
|
|
|
No. of shares |
% |
|
Pronomz Ventures LLP |
12665000 |
21.54% |
Details of aggregate number of equity shares allotted
as fully paid-up pursuant to contract without payment being received in cash
for the period of five year immediately preceding the balance sheet date:
|
PARTICULARS |
31.12.2012 |
|
|
No. of shares |
|
Equity shares of Rs.10- issued pursuant to a scheme of amalgamation in
2009 |
13524 |
Details of equity shares ofRs.10/- each reserved for issuance:
|
PARTICULARS |
31.12.2012 |
|
|
No. of shares |
|
Towards Employee stock options under the various Strides Stock option
plans |
2702350 |
FINANCIAL DATA
[all figures are
in Rupees Millions]
ABRIDGED BALANCE
SHEET
|
SOURCES OF FUNDS |
|
31.12.2012 |
31.12.2011 |
|
I.
EQUITY
AND LIABILITIES |
|
|
|
|
(1)Shareholders' Funds |
|
|
|
|
(a) Share Capital |
|
588.040 |
583.800 |
|
(b) Reserves & Surplus |
|
13126.100 |
13008.360 |
|
(c) Money
received against share warrants |
|
0.000 |
0.000 |
|
|
|
|
|
|
(2) Share Application money pending allotment |
|
0.000 |
0.000 |
|
Total
Shareholders’ Funds (1) + (2) |
|
13714.140 |
13592.160 |
|
|
|
|
|
|
(3)
Non-Current Liabilities |
|
|
|
|
(a) long-term borrowings |
|
2846.610 |
3639.350 |
|
(b) Deferred tax liabilities (Net) |
|
0.000 |
0.000 |
|
(c) Other long term liabilities |
|
79.940 |
45.860 |
|
(d) long-term provisions |
|
504.290 |
588.640 |
|
Total Non-current Liabilities (3) |
|
3430.840 |
4273.850 |
|
|
|
|
|
|
(4) Current Liabilities |
|
|
|
|
(a) Short term borrowings |
|
2935.810 |
4142.280 |
|
(b) Trade payables |
|
1429.370 |
2096.120 |
|
(c) Other current
liabilities |
|
1104.260 |
7684.170 |
|
(d) Short-term provisions |
|
373.390 |
833.840 |
|
Total Current Liabilities (4) |
|
5842.830 |
14756.410 |
|
|
|
|
|
|
TOTAL |
|
22987.810 |
32622.420 |
|
|
|
|
|
|
II.
ASSETS |
|
|
|
|
(1) Non-current assets |
|
|
|
|
(a) Fixed Assets |
|
|
|
|
(i) Tangible assets |
|
2285.870 |
2354.360 |
|
(ii) Intangible Assets |
|
776.740 |
884.660 |
|
(iii) Capital
work-in-progress |
|
81.200 |
54.710 |
|
(iv)
Intangible assets under development |
|
214.470 |
15.420 |
|
(b) Non-current Investments |
|
12953.230 |
7868.180 |
|
(c) Deferred tax assets (net) |
|
0.000 |
0.000 |
|
(d) Long-term Loan and Advances |
|
922.580 |
5835.170 |
|
(e) Other Non-current assets |
|
0.000 |
0.000 |
|
Total Non-Current Assets |
|
17234.090 |
17012.500 |
|
|
|
|
|
|
(2) Current assets |
|
|
|
|
(a) Current investments |
|
0.380 |
0.000 |
|
(b) Inventories |
|
1043.540 |
1303.190 |
|
(c) Trade receivables |
|
1930.960 |
2642.830 |
|
(d) Cash and cash
equivalents |
|
293.300 |
814.610 |
|
(e) Short-term loans and
advances |
|
2343.170 |
10520.270 |
|
(f) Other current assets |
|
142.370 |
329.020 |
|
Total Current Assets |
|
5753.720 |
15609.920 |
|
|
|
|
|
|
TOTAL |
|
22987.810 |
32622.420 |
|
SOURCES OF FUNDS |
|
|
31.12.2010 |
|
|
SHAREHOLDERS FUNDS |
|
|
|
|
|
1] Share Capital |
|
|
577.450 |
|
|
2] Share Application Money |
|
|
0.000 |
|
|
3] Reserves & Surplus |
|
|
13462.740 |
|
|
4] Employees stock options outstanding account |
|
|
20.860 |
|
|
5] (Accumulated Losses) |
|
|
0.000 |
|
|
NETWORTH |
|
|
14061.050 |
|
|
LOAN FUNDS |
|
|
|
|
|
1] Secured Loans |
|
|
6461.360 |
|
|
2] Unsecured Loans |
|
|
5969.340 |
|
|
TOTAL BORROWING |
|
|
12430.700 |
|
|
DEFERRED TAX LIABILITIES |
|
|
0.000 |
|
|
|
|
|
|
|
|
TOTAL |
|
|
26491.750 |
|
|
|
|
|
|
|
|
APPLICATION OF FUNDS |
|
|
|
|
|
|
|
|
|
|
|
FIXED ASSETS [Net Block] |
|
|
3115.520 |
|
|
Capital work-in-progress |
|
|
375.280 |
|
|
|
|
|
|
|
|
INVESTMENT |
|
|
8645.200 |
|
|
DEFERREX TAX ASSETS |
|
|
0.000 |
|
|
|
|
|
|
|
|
CURRENT ASSETS, LOANS & ADVANCES |
|
|
|
|
|
|
Inventories |
|
|
1293.080 |
|
|
Sundry Debtors |
|
|
1597.310 |
|
|
Cash & Bank Balances |
|
|
810.290 |
|
|
Other Current Assets |
|
|
145.280 |
|
|
Loans & Advances |
|
|
14210.230 |
|
Total
Current Assets |
|
|
18056.190 |
|
|
Less : CURRENT
LIABILITIES & PROVISIONS |
|
|
|
|
|
|
Sundry Creditors |
|
|
1882.820 |
|
|
Other Current Liabilities |
|
|
326.790 |
|
|
Provisions |
|
|
1490.830 |
|
Total
Current Liabilities |
|
|
3700.440 |
|
|
Net Current Assets |
|
|
14355.750
|
|
|
|
|
|
|
|
|
MISCELLANEOUS EXPENSES |
|
|
0.000 |
|
|
|
|
|
|
|
|
TOTAL |
|
|
26491.750 |
|
PROFIT & LOSS
ACCOUNT
|
|
PARTICULARS |
31.12.2012 |
31.12.2011 |
31.12.2010 |
|
|
|
SALES |
|
|
|
|
|
|
|
Revenue from operations |
7120.070 |
7496.200 |
5046.610 |
|
|
|
Other Income |
1189.080 |
195.990 |
561.030 |
|
|
|
TOTAL |
8309.150 |
7692.190 |
5607.640 |
|
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
|
|
Cost of materials consumed |
2815.420 |
3178.380 |
|
|
|
|
Purchase of stock in trade |
1003.070 |
1072.110 |
|
|
|
|
(Increase)/Decrease in stock |
66.760 |
66.180 |
4347.240 |
|
|
|
Employees benefits expenses |
709.190 |
629.760 |
|
|
|
|
Other Expenses |
1568.570 |
1061.020 |
|
|
|
|
TOTAL |
6163.010 |
6007.450 |
4347.240 |
|
|
|
|
|
|
|
|
Less |
PROFIT
BEFORE INTEREST, TAX, DEPRECIATION AND AMORTISATION |
2146.140 |
1684.740 |
1260.400 |
|
|
|
|
|
|
|
|
|
Less |
FINANCIAL
EXPENSES |
712.200 |
805.900 |
733.740 |
|
|
|
|
|
|
|
|
|
|
PROFIT
BEFORE TAX, DEPRECIATION AND AMORTISATION |
1433.940 |
878.840 |
526.660 |
|
|
|
|
|
|
|
|
|
Less/ Add |
DEPRECIATION/ AMORTISATION |
190.990 |
176.150 |
150.820 |
|
|
|
|
|
|
|
|
|
|
EXCEPTIONAL ITEM
|
(644.090) |
559.060 |
514.990 |
|
|
|
|
|
|
|
|
|
|
PROFIT BEFORE
TAX |
598.860 |
1261.750 |
890.830 |
|
|
|
|
|
|
|
|
|
Less |
TAX |
39.000 |
82.500 |
155.210 |
|
|
|
|
|
|
|
|
|
|
PROFIT AFTER TAX
|
559.860 |
1179.250 |
735.620 |
|
|
|
|
|
|
|
|
|
|
EARNINGS IN
FOREIGN CURRENCY |
|
|
|
|
|
|
|
FOB Value of Exports of Goods |
5098.960 |
5404.610 |
3277.180 |
|
|
|
Development Income |
30.170 |
447.590 |
609.680 |
|
|
|
Management advisory service
fees |
507.150 |
332.64 |
312.970 |
|
|
|
Interest |
516.340 |
10.230 |
9.880 |
|
|
|
Profit on sale of investment |
308.460 |
0.000 |
94.400 |
|
|
|
Share of Profit on Sale of Product |
11.610 |
0.000 |
97.920 |
|
|
|
Other Income |
9.200 |
131.260 |
149.970 |
|
|
TOTAL EARNINGS |
6481.890 |
6326.330 |
4552.000 |
|
|
|
|
|
|
|
|
|
|
IMPORTS |
|
|
|
|
|
|
|
Raw Materials |
1379.150 |
1241.020 |
612.690 |
|
|
|
Capital Goods |
69.290 |
37.120 |
185.550 |
|
|
|
Others |
66.070 |
11.560 |
6.450 |
|
|
TOTAL IMPORTS |
1514.510 |
1289.700 |
804.690 |
|
|
|
|
|
|
|
|
|
|
Earnings Per
Share (Rs.) |
|
|
|
|
|
|
Basic |
9.55 |
20.30 |
15.69 |
|
|
|
Diluted |
5.84 |
20.13 |
11.98 |
|
KEY RATIOS
|
PARTICULARS |
|
31.12.2012 |
31.12.2011 |
31.12.2010 |
|
PAT / Total Income |
(%) |
6.74
|
15.33
|
13.12 |
|
|
|
|
|
|
|
Net Profit Margin (PBT/Sales) |
(%) |
8.41
|
16.83
|
17.65 |
|
|
|
|
|
|
|
Return on Total Assets (PBT/Total Assets} |
(%) |
6.15
|
5.11
|
4.21 |
|
|
|
|
|
|
|
Return on Investment (ROI) (PBT/Networth) |
|
0.04
|
0.09
|
0.06 |
|
|
|
|
|
|
|
Debt Equity Ratio (Total Debt /Networth) |
|
0.42
|
0.57
|
0.88 |
|
|
|
|
|
|
|
Current Ratio (Current Asset/Current Liability) |
|
0.98
|
1.06
|
4.88 |
FINANCIAL ANALYSIS
[all figures are in
Rupees Millions]
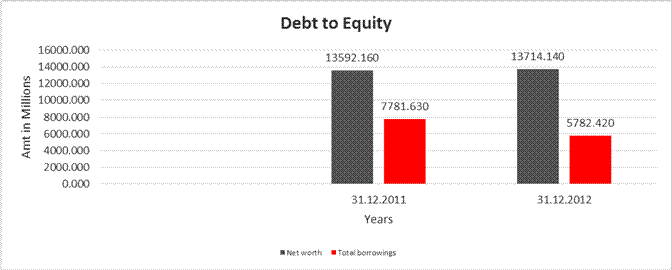
DEBT EQUITY RATIO
|
Particular |
31.12.2011 |
31.12.2012 |
|
|
(Rs.
In Millions) |
(Rs.
In Millions) |
|
Share Capital |
583.800 |
588.040 |
|
Reserves & Surplus |
13008.360 |
13126.100 |
|
Net
worth |
13592.160 |
13714.140 |
|
|
|
|
|
long-term borrowings |
3639.350 |
2846.610 |
|
Short term borrowings |
4142.280 |
2935.810 |
|
Total
borrowings |
7781.630 |
5782.420 |
|
Debt/Equity
ratio |
0.573 |
0.422 |
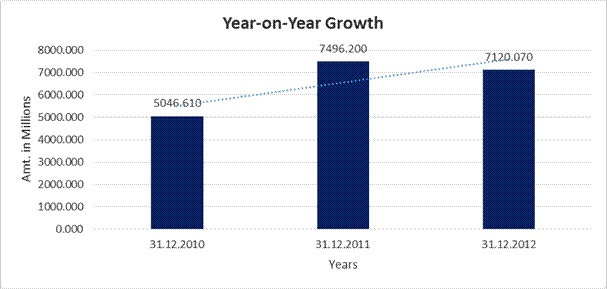
YEAR-ON-YEAR GROWTH
|
Year
on Year Growth |
31.12.2010 |
31.12.2011 |
31.12.2012 |
|
|
(Rs.
In Millions) |
(Rs.
In Millions) |
(Rs.
In Millions) |
|
Sales |
5046.610 |
7496.200 |
7120.070 |
|
|
|
48.539 |
(5.018) |
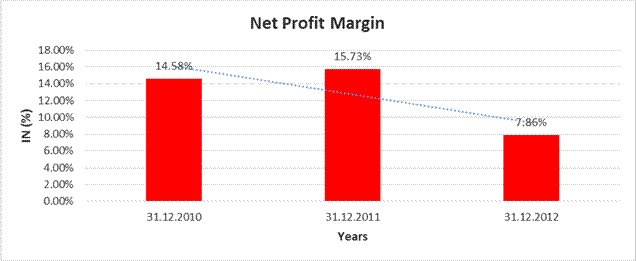
NET PROFIT MARGIN
|
Net
Profit Margin |
31.12.2010 |
31.12.2011 |
31.12.2012 |
|
|
(Rs.
In Millions) |
(Rs.
In Millions) |
(Rs.
In Millions) |
|
Sales |
5046.610 |
7496.200 |
7120.070 |
|
Profit |
735.620 |
1179.250 |
559.860 |
|
|
14.58% |
15.73% |
7.86% |
LOCAL AGENCY FURTHER INFORMATION
|
Sr. No. |
Check List by Info Agents |
Available in
Report (Yes / No) |
|
1] |
Year of Establishment |
Yes |
|
2] |
Locality of the firm |
Yes |
|
3] |
Constitutions of the firm |
Yes |
|
4] |
Premises details |
No |
|
5] |
Type of Business |
Yes |
|
6] |
Line of Business |
Yes |
|
7] |
Promoter's background |
No |
|
8] |
No. of employees |
Yes |
|
9] |
Name of person contacted |
No |
|
10] |
Designation of contact
person |
No |
|
11] |
Turnover of firm for last
three years |
Yes |
|
12] |
Profitability for last
three years |
Yes |
|
13] |
Reasons for variation
<> 20% |
----- |
|
14] |
Estimation for coming
financial year |
No |
|
15] |
Capital in the business |
Yes |
|
16] |
Details of sister
concerns |
Yes |
|
17] |
Major suppliers |
No |
|
18] |
Major customers |
No |
|
19] |
Payments terms |
No |
|
20] |
Export / Import details
(if applicable) |
No |
|
21] |
Market information |
----- |
|
22] |
Litigations that the firm
/ promoter involved in |
Yes |
|
23] |
Banking Details |
Yes |
|
24] |
Banking facility details |
Yes |
|
25] |
Conduct of the banking
account |
----- |
|
26] |
Buyer visit details |
----- |
|
27] |
Financials, if provided |
Yes |
|
28] |
Incorporation details, if
applicable |
Yes |
|
29] |
Last accounts filed at
ROC |
No |
|
30] |
Major Shareholders, if
available |
Yes |
|
31] |
Date of Birth of
Proprietor/Partner/Director, if available |
No |
|
32] |
PAN of
Proprietor/Partner/Director, if available |
No |
|
33] |
Voter ID No of
Proprietor/Partner/Director, if available |
No |
|
34] |
External Agency Rating,
if available |
Yes |
|
LITIGATION DETAILS |
|||||||
|
Bench:- Bombay |
|
||||||
|
Stamp No:- |
ITXAL/2454/2010 |
Failing Date:- |
07/12/2010 |
Reg. No.:- |
ITXA/46/2011 |
Reg. Date:- |
15/01/2011 |
|
|
|||||||
|
|
Main Matter |
|
|||||
|
Petitioner:- |
COMMISSIONER OF INCOME TAX |
Respondent:- |
STRIDES ARCOLAB LIMITED |
||||
|
Petn.Adv:- |
PADAM DIVAKAR |
||||||
|
District:- |
Mumbai |
||||||
|
|
|||||||
|
Bench:- |
Single |
Category:- |
TAX APPEALS |
||||
|
Status:- |
Admitted(Unready) |
Stage:- |
FOR ADMISSION - FRESH |
||||
|
Last Date:- |
23/11/2011 |
|
|||||
|
Last Coram:- |
HON'BLE SHRI JUSTICE J.P. DEVADHAR HON'BLE SHRI JUSTICE A.R. JOSHI |
||||||
|
|
|
||||||
|
Act:- |
Income Tax Act, 1961 |
||||||
UNSECURED LOANS
|
Unsecured Loans |
31.12.2012 |
31.12.2011 |
|
|
(Rs. In Millions) |
|
|
LONG-TERM BORROWINGS |
|
|
|
Term loan from banks |
2605.450 |
2663.500 |
|
SHORT-TERM BORROWINGS |
|
|
|
Unsecured loan repayable on demand from others |
0.000 |
100.000 |
|
Total |
2605.450 |
2763.500 |
TURNOVER AND
PROFITS
On a Standalone basis the total income during the year stood at Rs. 8309.150 Millions as against Rs. 7692.190 Millions in the previous year. The Standalone net profits Rs.559.806 Million as against a net profit ofRs.1179.250 Millions for the previous year.
BUSINESS OVERVIEW
2012 began and ended on an optimistic note for the Company. During the year, their revenues and EBIDTA increased considerably, compared to 2011, primarily driven by consistent new product launches and increase in operational scale. Besides, both Agila and Pharma divisions demonstrated significant operating leverage across all global manufacturing facilities and consistent regulatory filings. Besides, there were significant corporate initiatives round the year.
KEY BUSINESS HIGHLIGHTS FOR
2012
PHARMA
COLLABORATION WITH GILEAD
SCIENCES
The Company entered into an in-licensing agreement to collaborate with Gilead Sciences, Inc. to promote access to high-quality, low-cost generic versions of Gilead's HIV medicine emtricitabine (FTC) in developing countries - including fixed-dose combinations of emtricitabine co-formulated with other Gilead HIV medicines.
FUNDING FROM FRENCH
DEVELOPMENT
FINANCING INSTITUTION
During the year the French Development Financing Institution Proparco invested USD12.5 Million in the form of equity participation for a 20% stake in Strides' African front-end arm, valuing the African operations at about USD 60 Million. The proceeds will be used to create additional manufacturing infrastructure in key markets in Africa and to build a regional Company.
BIOTECH
CONSOLIDATION OF STAKE IN
INBIOPRO
During the year 2012, the Company consolidated its stake in Inbiopro Solutions Private Limited (Inbiopro) the Biotech arm of the Company from the initial holding of 70% to 96.79%.
As at the date of this report the Company further consolidated its stake to 100% and consequently Inbiopro is a wholly owned subsidiary of the Company.
CUSTOMISED BIOTECH FACILITY
IN MALAYSIA
In March 2013, the Company's wholly owned subsidiary Agila Biotech (Malaysia) SDN BHD, Malaysia, entered into an arrangement with Bio-XCell Sdn Bhd (Malaysian Government undertaking) for the establishment of a customised biotech facility located in the Bio-XCell ecosystem in Johor, Malaysia.
The Company plans to incorporate into this facility, the "next-generation" technology platforms which revolutionise the way biomolecules are developed, manufactured and commercialised.
SPECIALTIES (AGILA)
BRAZIL STERILE PENEMS
FACILITY - US FDA APPROVAL
In February 2012, the Brazilian Sterile Penems facility received US FDA approval. This state-of-the-art facility manufactures sterile dry powder injectables of Penems. The plant has already been approved by other international regulatory agencies like MHRA and ANVISA.
POLISH FACILITY - US-FDA
APPROVAL
During the year, the Company's polish sterile facility received US-FDA approval. This state-of-the-art facility located in Warsaw, Poland, manufactures vials, ampoules, pre-filled syringes and lyophilized injections. The approval offers significant flexibility to the manufacturing which is currently experiencing strong demand on a worldwide basis.
ACQUISITION OF USFDA
APPROVED STERILE MANUFACTURING FACILITY
During the year, the Company through its wholly owned subsidiary, Agila Specialties Private Limited acquired a USFDA approved Sterile Formulations facility situated at Hosur, Tamil Nadu from Star Drugs and Research Labs Limited.
JOINT VENTURE WITH JAMP
PHARMA CORPORATION
During the year, the Company through its wholly owned subsidiary Agila Specialties Pharma Corporation, Canada (part of injectable division of Strides), formed a joint venture with Jamp Pharma, a Canadian generic drug company, to introduce a variety of quality injectable generic drugs in Canada.
COLLABORATION WITH ELI LILLY
In December 2012, the Company along with its subsidiary Agila Specialties Private Limited collaborated with Eli Lilly to expand delivery of cancer medicines in the emerging markets. As a part of this arrangement, Lilly will in-license a portfolio of high-quality, branded generic injectable and oral cancer medicines from Agila Specialties, the specialties division of Strides.
MANAGEMENT DISCUSSION AND ANALYSIS
GLOBAL ECONOMY
The global
economy weathered unpredictable headwinds in 2012. The Euro zone had little to
cheer in 2012 as governments continued to look for ways and means to keep
financial markets afloat without promoting unsustainable debts, or continue
with austerity measures without impacting the economy. Emerging economies were
also impacted in a largely interdependent world.However, the world's largest
economy, the United States, seems to be recovering.
In 2012
the economy grew 2.2% against 1.8% in 2011.
Reliable
forecasts of the US economy for 2013 put growth upwards of 2% and for 2014,
above 3%. These estimates apparently look insignificant, but considering the
size of the economy, around US$ 15 Trillion, even a 2% growth would add an
output worth US$ 300 Billion. This will create a fresh wave of economic
activity not only in the US, but around the world, thanks to globalisation. The
global economy is expected to have grown by 3.2% in 2012. It is estimated to
grow at the rate of 3.5% in 2013.
INDIAN ECONOMY
India's GDP is estimated to have grown by 5% in FY 2012-13. The Reserve Bank of India infused Rs. 18,000 crore to its economy by reducing the cash reserve ratio (CRR) to 4% in its third monetary policy during 2012-13. Moreover, he Reserve Bank of India twice lowered the repo rate by 25 basis points in 2012-13 to help revive growth. Increasing FDI limits across major sectors have significantly improved India's capital inflows. The government is also taking various initiatives to keep the fiscal deficit within 5.2%. More financial reforms are expected to strengthen India's growth story.
GLOBAL PHARMACEUTICAL
INDUSTRY
PHARMACEUTICAL INDUSTRY
OVERVIEW
The global pharmaceutical market has witnessed a 6% CAGR from 2006 to 2012 to reach a US$ 956 Billion market size in 2012. According to IMS Health, the global pharmaceutical spend is estimated to touch US$ 1.2 Trillion by CY2016, growing at 4.5%annually. Growth will be primarilydriven by higher generic spending (accounting for 3/4th of the total increase) and increasing medical expenditure.
The pharmerging countries will be the primary growth driver with market share likely to rise to 30% by CY2016 from 20% in CY2011. Simultaneously, the share of developed markets is likely to decline to 57% by CY2016 from 66% in CY2011, led by around US$ 100 Billion of patent expiries in the next five years.
Gradually growth is gravitating from developed countries to emerging markets. This is owing to an enhanced focus on biopharmaceuticals, compared to small molecule drugs and higher preference for generics, compared to their branded counterparts.
INNOVATOR MARKET
As the innovator market emerges from a wave of patent expiries in the US, there has been a sharp increase in the number of NDA (new drug application) approvals. The year 2012 saw around 34 new drug approvals, highest in the last eight years.
GENERIC MARKET
Growing generics spending in the developed Market over the next five year will be fuelled by generic competition due to patent expiries, with some additional increases due to expanded generic use for off-patent molecules. In
pharmerging markets generic companies will increase most of the spending
GLOBAL
PHARMACEUTICAL MARKETS
REGULATED MARKET
USA
The US pharmaceutical market valued is US$ 322 Billion in 2011, is expected to grow at a CAGR of 1-4% over 2012-16, likely to reach a market value of 350-380 Billion by 2016 The year 2012 witnessed around US$ 35 Billion worth of drugs to go off patent The US generics market, worth US$ 100 Billion, is also estimated to register a CAGR of 8-9% in the medium term on account of patent expiries
JAPAN
Japan is the world’s second largest pharmaceutical market. Japan’s
pharmaceutical market valued at US$ 111 Billion in 2011, is likely to witness
CAGR of 1-4% over 2012-16, reaching a market size of US$ 105 – 135
Billion.Growth rate is marginally hindered by price cuts expected in 2014 and
2016. Rising healthcare costs and ageing population have forced the government
to initiate a shift towards generic drugs. The generics segment is the highest
growing of all with anestimated CAGR of ~14% (2010-2015).
EUROPE
Europe is one of the largest global pharmaceutical market (around 17%)
followed by the US and Japan. In Europe pharmaceutical market is likely to
witness growth in the range of -1% to 2% by 2016. Sluggish growth is expected
due to healthcare cost containment measures adopted in order to curtail
the debt crisis. The EU5* nations are likely to touch a market size of US$
125-175 Billion by 2016.
PHARMERGING
MARKETS
The pharmerging markets are likely to double their pharmaceutical
spending from US$ 151 Billion 2011 to around US$ 285-313 Billion by 2015.Growth
will be led by gradual economic growth and government efforts to expand
healthcare access. IMS expects the pharmerging markets to grow by 13%-plus CAGR
from 2011 to 2016, reaching US$ 357 Billion by 2016.
CHINA
China’s pharmaceutical market is expected to soar to 2.3 Trillion yuan
(US$ 369.2 Billion) by 2020, up from 926.1 Billion yuan (US$ 148. 66 Billion),
currently. The growth will be led by China’s ageing population and economic
development, driving social insurance and consumption capacities (Source: Life
Sciences Health Industry Group, 2013).
LATIN AMERICA
The outlook for the Latin American market is positive, given the sheer
size and the burgeoning population. Generics and biologics are among the
strongest areas. Even if standards vary, the tendency is for ‘similar drugs’
without proven bio-equivalence to be phased out.
According to the IMS Health, the Latin American market will double from
2011 to 2015.The Latin America’s pharmaceutical market is worth US$ 45 Billion
in 2011.
Brazil’s is the largest market in Latin America and ranks the 7th
globally. The market is growing at 14-15%, and is expected to reach US$ 25
Billion by 2014 (Source: IMS, Fortune).
INDIA
India’s pharmaceutical industry is expected to reach US$ 29 Billion,
growing at a CAGR of 15-16%. The country is expected to have registered a growth
of US$ 15.7 Billion in 2012(Source: Espicom). The key growth enablers
(disposable income, insurance penetration, growing prevalence of lifestyle
diseases) will continue to drive growth.
GLOBAL APPROACH
India is rapidly emerging as one of the most preferred outsourcing
destinationsfor pharmaceuticals. A favourable regulatory environment has
attracted significant foreign investment. The cumulative drugs and pharmaceutical
industry attracted a foreign direct investment (FDI) of US$ 9,596 Million between
April 2000 and May 2012.
STATEMENT OF
STANDALONE UNAUDITED RESULTS FOR THE QUARTER & TWELVE MONTHS ENDED DECEMBER
31, 2013
(Rs. In Millions)
|
Sl. No. |
Particulars |
3
Months ended 31.12.2013 |
Preceding3
Mounts ended 30.09.2013 |
Year
to date figures for the current period ended 31.12.2013 |
|
|
|
Unaudited |
Unaudited |
Unaudited |
|
1 |
Income from operations |
|
|
|
|
|
(a) Net Sales / Income
from Operations (Net of excise duty) |
2402.433 |
1991.813 |
7515.973 |
|
|
(b) Other Operating
Income |
131.325 |
97.607 |
555.172 |
|
|
Total Income from operations (net) |
2533.758 |
2089.420 |
8071.145 |
|
2 |
Expenses |
|
|
|
|
|
(a) Cost of material
consumed |
1321.732 |
835.806 |
3522.969 |
|
|
(b) Purchases of
stock-in-trade |
386.499 |
458.850 |
1318.160 |
|
|
(c) Changes in
inventories of finished goods, work-in-progress and stock-in-trade |
(131.698) |
(70.359) |
(138.092) |
|
|
(d) Employee benefit
expenses |
196.871 |
196.147 |
759.853 |
|
|
(e) Depreciation and
amortisation expense |
97.121 |
92.993 |
370.637 |
|
|
(f) Other expenses |
483.836 |
317.219 |
1438.127 |
|
|
Total expenses |
2354.361 |
1830.656 |
7271.654 |
|
3 |
Profit/(Loss) from Operations before Other Income, finance
cost & Exceptional Items (1 -2) |
179.397 |
258.764 |
799.49 |
|
4 |
Other Income |
13882.386 |
165.278 |
14390.585 |
|
5 |
Profit/ (Loss) from ordinary activities before finance cost
& Exceptional Items (3+4) |
14061.783 |
424.042 |
15190.076 |
|
6 |
Finance costs |
399.053 |
251.226 |
934.245 |
|
7 |
Profit/(Loss) from ordinary activities after finance cost but
before Exceptional Items (5-6) |
13662.730 |
172.816 |
14255.831 |
|
8 |
Exceptional Items |
|
|
|
|
|
Exchange Fluctuation
(Loss) / gain (Net) |
271.278 |
(118.666) |
36.630 |
|
|
Net gain on sale of
long term investments |
32655.267 |
-- |
32655.267 |
|
|
Write down of
investor |
(161.894) |
-- |
(161.894) |
|
9 |
Profit / (Loss) from Ordinary Activities before tax (7+ 8) |
46427.381 |
54.150 |
46785.834 |
|
10 |
Tax Expense / (credit) |
1174.968 |
32.000 |
11173.969 |
|
11 |
Net Profit / (Loss) after tax
(9-10) |
35252.413 |
22.150 |
35611.865 |
|
12 |
Paid-up Equity Share Capital (Face value of Rs.10/-each) |
595.656 |
591.614 |
595.656 |
|
13 |
Earnings per share
(face value of Rs. 10/- each) - not annualised |
|
|
|
|
|
Basic EPS (Rs.) |
597.00 |
0.38 |
602.79 |
|
|
Diluted EPS (Rs.) |
595.12 |
0.37 |
599.85 |
|
A |
PARTICULARS OF
SHAREHOLDING |
|
|
|
|
1 |
Public shareholding
: |
|
|
|
|
|
(a) Number of shares |
43,381,419 |
42,977,169 |
43,381,419 |
|
|
(b) Percentage of shareholding |
72.83% |
72.64% |
72.83% |
|
2 |
Promoters and Promoter
group shareholding : (a) Pledged /
Encumbered |
|
|
|
|
|
- Number of shares |
1,876,667 |
7,280,152 |
1,876,667 |
|
|
- Percentage of shareholding (as a % of the total share holding of promoter and promoter group) |
11.60% |
44.98% |
11.60% |
|
|
- Percentage of shareholding (as a % of the total share capital of the Company) |
3.15% |
12.31% |
3.15% |
|
|
(b) Non Pledged / Non Encumbered |
|
|
|
|
|
- Number of shares |
14,307,535 |
8,904,050 |
14,307,535 |
|
|
- Percentage of shareholding (as a % of the total share holding of promoter and promoter group) |
88.40% |
55.02% |
88.40% |
|
|
- Percentage of shareholding (as a % of the total share capital of the Company) |
24.02% |
15.05% |
24.02% |
|
|
|
|
|
|
|
B |
INVESTOR COMPLAINTS [Nos.] |
|
|
|
|
|
Pending at the beginning
of the quarter |
-- |
|
|
|
|
Received during the
quarter |
33 |
|
|
|
|
Disposed of during the
quarter |
30 |
|
|
|
|
Remaining unresolved at
the end of the quarter |
3 |
|
|
STANDALONE STATEMENT
OF ASSETS AND LIABILITIES
|
Particulars |
As
at 31.12.2013 |
|
|
A |
EQUITY AND LIABILITIES |
|
|
1 |
Shareholder’s Funds |
|
|
|
a) Share Capital |
595.656 |
|
|
b) Reserves & Surplus |
16461.443 |
|
|
Sub Total- Shareholders funds |
17057.099 |
|
2 |
Non-current liabilities |
|
|
|
(a) Long term borrowings |
2533.111 |
|
|
(b) Other long term liabilities |
61.632 |
|
|
(d) Deferred tax liabilities |
43.900 |
|
|
(c) Long term provisions |
151.849 |
|
|
Sub Total- Non Current Liabilities |
2790.492 |
|
3 |
Current liabilities |
|
|
|
(a) Short term borrowings |
3857.426 |
|
|
(b) Trade Payables |
2046.822 |
|
|
(c) Other current liabilities |
1388.961 |
|
|
(d) Short term provisions |
2482.555 |
|
|
Sub Total- Current Liabilities |
9775.764 |
|
|
TOTAL-EQUITY AND LIABILITIES |
29623.355 |
|
|
|
|
|
B |
ASSETS |
|
|
1 |
Non-current assets |
|
|
|
(a) Fixed assets |
5215.117 |
|
|
(b) Non-current Investment |
6363.496 |
|
|
(c) Long term loans and
advances |
1321.542 |
|
|
Sub-Total- Non current assets |
12900.155 |
|
2 |
Current assets |
|
|
|
a) Current Investments |
4717.444 |
|
|
b) Inventories |
1255.575 |
|
|
c) Trade Receivables |
2497.443 |
|
|
d) Cash and cash equivalents |
5736.790 |
|
|
(e) Short term loans and
advances |
2375.112 |
|
|
(f) Other current assets |
140.836 |
|
|
Sub-Total- current assets |
16723.200 |
|
|
TOTAL ASSETS |
29623.355 |
NOTE:
1. The above unaudited results of the Company has been reviewed by the Audit Committee and taken on record by the Board of Directors at their meeting held on February 7, 2014.
2. The statutory auditors have carried out limited review of the above standalone results.
3. The previous period's figures have been regrouped/ reclassified wherever necessary to conform to the classification of the current period.
4. In terms of the scheme of arrangement approved by the Hon’ble High Courts of Judicature during the year ended December 31, 2009, the Company had created a Reserve for Business Restructure (BRR) which has been utilised during prior years as mentioned below
|
|
Particulars |
3
Months ended 31.12.2013 |
Preceding3
Mounts ended 30.09.2013 |
Year
to date figures for the current period ended 31.12.2013 |
|
|
Utilisation of BRR : - Employee benefit expenses accrued / (reversed) - net - Depreciation and Amortisation - Other expenses - Interest on Fixed Loans Impact if the Company
followed the Accounting Standards instead of the accounting treatment
provided in the Scheme :- |
-- |
-- |
-- |
|
|
|
|
|
- |
|
|
Net Profit for the
period would have decreased by : |
|
|
- |
|
|
Earnings / (Loss) per
share (EPS) (Face value of Rs.10/-each) would have been : - Basic (not annualised) - Diluted (not annualised) |
Rs. 597.00 595.12 |
Rs. 0.38 0.37 |
Rs. 602.79 599.85 |
5. During the quarter, 404,250 equity shares were allotted by the Company [203,750 shares under Strides Arcolab ESOP 2006 Scheme, 160,500 shares under Strides Arcolab ESOP 2008 and 40,000 shares under Strides Arcolab ESOP 2008 (Director) Scheme] on exercising equal number of options. No options were granted in the current period.
6. During the quarter ended March 2013 (February 27, 2013), the Company and its subsidiary, Agila Specialties Asia Pte. Limited. (Agila Asia), had entered into definitive agreements for the sale of entities into Specialty products (a part of the Pharmaceutical business that the Group is into) by way of share sales to Mylan Inc. (the `Purchaser’). In the current quarter (on December 5, 2013), the Company has completed such sale of shares in Agila Specialties Private Limited (AGILA) and in Agila Specialties Global Asia Pte. Limited (Agila Global). Exceptional items in the above results include profit from sale of investments in AGILA to the extent of Rs. 32655.267 Millions net of transaction costs and special bonus of Rs. 1072.577 Millions.
7.
CONTINGENT
LIABILITIES (AS ON 31.12.2012)
The Company has given corporate guarantees upto Rs.26298.570 Millions (Previous year Rs.4572.940 Millions) to financial institutions and other parties, on behalf of its subsidiaries. At December 31, 2012, the subsidiaries had availed facilities from such financial institutions/ were obligated to the parties referred above for an aggregate amount of Rs.4068.850 Millions (Previous year Rs.3672.540 Millions). The Company has additionally provided its fixed assets (under a paripassu second charge) as security in respect of some of these facilities.
The Company has disputed tax liabilities arising from assessment proceedings relating to earlier years from the income tax authorities amounting to Rs.741.310 Millions (Previous year Rs.741.270). The outflow on account of disputed taxes is dependent on completion of assessments.
The Company has preferred an appeal with the CESTAT against the order of the Commissioner of Central Excise disallowing transfer of CENVAT credit of Rs.3.860 Million (Previous year Rs.3.860 Millions) as on the date of conversion of one of the units of the Company into a 100% EOU. The outflow on account of disputed taxes is dependent on completion of assessments.
FIXED ASSETS
·
Freehold Land
·
Leased Hold Land
·
Buildings
·
Furniture and Fixtures
·
Office Equipment and Computers
·
Plant and Machinery
·
Motor Vehicles
·
Registration and Brands
·
Software Licences
PRESS RELEASE
CONTINUED US FDA
APPROVAL STATUS FOR ORAL DOSAGE FACILITY
April 25, 2014: Strides Arcolab is pleased to announce that its Oral Dosage Forms manufacturing site (KRS Gardens) in Bangalore was recently inspected by the USFDA as part of GMP compliance audit and the facility continues to be approved.
The last USFDA inspection and approval for this facility was in the year 2011.
The KRS Gardens facility in Bangalore manufactures oral dosage forms such as tablets, capsules (both hard gelatine and soft gelatine) and sachets. The manufacturing plant supports important current and future submissions for the US Market.
About Strides Arcolab
Strides Arcolab, listed on the Bombay Stock Exchange Limited (532531) and National Stock Exchange of India Limited (STAR), is a global pharmaceutical Company headquartered in Bangalore, India that develops and manufactures a wide range of IP-led niche pharmaceutical products.
The Company has 5 manufacturing facilities presence in more than 75 countries in developed and emerging markets.
CMT REPORT (Corruption, Money Laundering
Terrorism]
The Public Notice information has been collected from various sources
including but not limited to: The Courts, India Prisons Service,
Interpol, etc.
1] INFORMATION ON
DESIGNATED PARTY
No exist designating subject or any of its beneficial owners,
controlling shareholders or senior officers as terrorist or terrorist
organization or whom notice had been received that all financial transactions
involving their assets have been blocked or convicted, found guilty or against
whom a judgement or order had been entered in a proceedings for violating
money-laundering, anti-corruption or bribery or international economic or
anti-terrorism sanction laws or whose assets were seized, blocked, frozen or
ordered forfeited for violation of money laundering or international
anti-terrorism laws.
2] Court Declaration :
No exist to suggest that subject is or was
the subject of any formal or informal allegations, prosecutions or other
official proceeding for making any prohibited payments or other improper
payments to government officials for engaging in prohibited transactions or
with designated parties.
3] Asset Declaration :
No records exist to suggest that the property or assets of the subject
are derived from criminal conduct or a prohibited transaction.
4] Record on Financial
Crime :
Charges or conviction
registered against subject: None
5] Records on Violation of
Anti-Corruption Laws :
Charges or
investigation registered against subject: None
6] Records on Int’l
Anti-Money Laundering Laws/Standards :
Charges or
investigation registered against subject: None
7] Criminal Records
No
available information exist that suggest that subject or any of its principals have
been formally charged or convicted by a competent governmental authority for
any financial crime or under any formal investigation by a competent government
authority for any violation of anti-corruption laws or international anti-money
laundering laws or standard.
8] Affiliation with
Government :
No record
exists to suggest that any director or indirect owners, controlling
shareholders, director, officer or employee of the company is a government
official or a family member or close business associate of a Government
official.
9] Compensation Package :
Our market
survey revealed that the amount of compensation sought by the subject is fair
and reasonable and comparable to compensation paid to others for similar
services.
10] Press Report : No
press reports / filings exists on the subject.
CORPORATE GOVERNANCE
MIRA INFORM as part of its Due Diligence do provide comments on
Corporate Governance to identify management and governance. These factors often
have been predictive and in some cases have created vulnerabilities to credit
deterioration.
Our Governance Assessment focuses principally on the interactions
between a company’s management, its Board of Directors, Shareholders and other
financial stakeholders.
CONTRAVENTION
Subject is not known to have contravened any existing local laws,
regulations or policies that prohibit, restrict or otherwise affect the terms
and conditions that could be included in the agreement with the subject.
FOREIGN EXCHANGE RATES
|
Currency |
Unit
|
Indian Rupees |
|
US Dollar |
1 |
Rs. |
|
|
1 |
Rs. |
|
Euro |
1 |
Rs. |
INFORMATION DETAILS
|
Information
Gathered by : |
PRT |
|
|
|
|
Analysis Done by
: |
RAS |
|
|
|
|
Report Prepared
by : |
NTH |
SCORE RATING EXPLANATIONS
|
SCORE FACTORS |
RANGE |
POINTS |
|
HISTORY |
1~10 |
6 |
|
PAID-UP CAPITAL |
1~10 |
5 |
|
OPERATING SCALE |
1~10 |
6 |
|
FINANCIAL CONDITION |
|
|
|
--BUSINESS SCALE |
1~10 |
6 |
|
--PROFITABILIRY |
1~10 |
5 |
|
--LIQUIDITY |
1~10 |
5 |
|
--LEVERAGE |
1~10 |
5 |
|
--RESERVES |
1~10 |
6 |
|
--CREDIT LINES |
1~10 |
5 |
|
--MARGINS |
-5~5 |
-- |
|
DEMERIT POINTS |
|
|
|
--BANK CHARGES |
YES/NO |
YES |
|
--LITIGATION |
YES/NO |
YES |
|
--OTHER ADVERSE INFORMATION |
YES/NO |
NO |
|
MERIT POINTS |
|
|
|
--SOLE DISTRIBUTORSHIP |
YES/NO |
NO |
|
--EXPORT ACTIVITIES |
YES/NO |
NO |
|
--AFFILIATION |
YES/NO |
YES |
|
--LISTED |
YES/NO |
YES |
|
--OTHER MERIT FACTORS |
YES/NO |
YES |
|
DEFAULTER |
|
|
|
--RBI |
YES/NO |
NO |
|
--EPF |
YES/NO |
NO |
|
TOTAL |
|
49 |
This score serves as a reference to assess SC’s credit risk
and to set the amount of credit to be extended. It is calculated from a
composite of weighted scores obtained from each of the major sections of this
report. The assessed factors and their relative weights (as indicated through
%) are as follows:
Financial
condition (40%) Ownership
background (20%) Payment
record (10%)
Credit history
(10%) Market trend
(10%) Operational
size (10%)
RATING EXPLANATIONS
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
>86 |
Aaa |
Possesses an extremely sound financial base with the strongest
capability for timely payment of interest and principal sums |
Unlimited |
|
71-85 |
Aa |
Possesses adequate working capital. No caution needed for credit transaction.
It has above average (strong) capability for payment of interest and
principal sums |
Large |
|
56-70 |
A |
Financial operational base are
regarded healthy. General unfavourable factors will not cause fatal effect. Satisfactory
capability for payment of interest and principal sums |
Fairly Large |
|
41-55 |
Ba |
Overall operation is considered normal. Capable to meet normal
commitments. |
Satisfactory |
|
26-40 |
B |
Capability to overcome financial difficulties seems comparatively
below average. |
Small |
|
11-25 |
Ca |
Adverse factors are apparent. Repayment of interest and principal sums
in default or expected to be in default upon maturity |
Limited with full
security |
|
<10 |
C |
Absolute credit risk exists. Caution needed to be exercised |
Credit not
recommended |
|
-- |
NB |
New Business |
-- |
This report is issued at your request without any
risk and responsibility on the part of MIRA INFORM PRIVATE LIMITED (MIPL) or
its officials.