MIRA
INFORM REPORT
|
Report Date : |
13.05.2014 |
IDENTIFICATION DETAILS
|
Name : |
PIRAMAL ENTERPRISES LIMITED (w.e.f. 31.07.2012) |
|
|
|
|
Formerly Known
As : |
PIRAMAL HEALTHCARE LIMITED (w.e.f. 24.06.2008) NICHOLAS PIRAMAL
INDIA LIMITED |
|
|
|
|
Registered
Office : |
Peninsula Corporate Park,
Nicholas Piramal Tower, Ganpatrao Kadam Marg, Lower Parel, Mumbai – 400013,
Maharashtra |
|
|
|
|
Country : |
|
|
|
|
|
Financials (as
on) : |
31.03.2013 |
|
|
|
|
Date of
Incorporation : |
26.04.1947 |
|
|
|
|
Com. Reg. No.: |
11-005719 |
|
|
|
|
Capital Investment
/ Paid-up Capital : |
Rs. 345.100 Millions |
|
|
|
|
CIN No.: [Company Identification
No.] |
L24110MH1947PLC005719 |
|
|
|
|
TAN No.: [Tax Deduction &
Collection Account No.] |
MUMN07675D |
|
|
|
|
PAN No.: [Permanent Account No.] |
AAACN4538P |
|
|
|
|
Legal Form : |
A Public Limited Liability Company. The Company’s Shares are Listed on
the Stock Exchanges. |
|
|
|
|
Line of Business
: |
The Subject is engaged in pharmaceutical business including its research and development. |
|
|
|
|
No. of Employees
: |
2976 (Approximately) |
RATING & COMMENTS
|
MIRA’s Rating : |
A (60) |
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
56-70 |
A |
Financial & operational base are regarded healthy. General
unfavourable factors will not cause fatal effect. Satisfactory capability for
payment of interest and principal sums |
Fairly Large |
|
Maximum Credit Limit : |
USD 422000000 |
|
|
|
|
Status : |
Good |
|
|
|
|
Payment Behaviour : |
Regular |
|
|
|
|
Litigation : |
Clear |
|
|
|
|
Comments : |
Subject is a well-established company having fine track record. The company has incurred loss from its operational activities during
the financial year 2013. However, the rating takes into consideration company’s strong and
experienced management team with track record of successfully scaling up
businesses and decent general financial position of the company. Trade relations are reported as fair. Business is active. Payments are
reported to be regular and as per commitments. The company can be considered good for normal business dealings at
usual trade terms and conditions. |
NOTES :
Any query related to this report can be made
on e-mail : infodept@mirainform.com
while quoting report number, name and date.
ECGC Country Risk Classification List – March 31, 2014
|
Country Name |
Previous Rating (31.12.2013) |
Current Rating (31.03.2014) |
|
India |
A1 |
A1 |
|
Risk Category |
ECGC
Classification |
|
Insignificant |
A1 |
|
Low |
A2 |
|
Moderate |
B1 |
|
High |
B2 |
|
Very High |
C1 |
|
Restricted |
C2 |
|
Off-credit |
D |
INDIAN ECONOMIC OVERVIEW
US investment bank
Goldman Sachs has upgraded its outlook on Indian markets as it expects
positive impact of the election cycle.
India’s economy may
grow 4.7 % in the current financial year, lower than the official estimate of
4.9 %, Fitch Rating said. The global rating agency expects the economy to pick
up in the next two financial years.
Global ratings
agency Standard & Poor said increasing focus by India Inc on lowering debt
is likely to improve their credit profiles.
Singapore (1.1
million Indian tourists in 2012), Thailand (one million), the United Arab
Emirates ().98 million) and Malaysia ().82 million) emerged as the preferred
holidays hotspots for Indians. The total figure is expected to increase to 1.93
million by 2017, according to the latest Eurmonitor international report.
There is a $29.34 bn
outward foreign direct investment by domestic companies between April and
January of 2013/14 which has seen some signs of recovery according to a Care
Ratings report.
There are 264 number
of new companies being set up every day on average during 2014. Most of them
are registered in Mumbai. India had 1.38 million registered companies at the
end of January, 2014.
Twitter like
messaging service Weibo Corporation has filed to raise $ 500 million via a US
initial public offering. Alibaba, which owns a stake in Weibo is expected to raise
about $ 15 billion New York this year in the highest profile Internet IPO since
Facebook’s in 2012.
Bharti Airtel has
raised Rs.2,453.2 crore (350 million Swiss Francs) by selling six-year bonds at
a coupon rate of three per cent and maturing in 2020. This is the largest ever
bond offering by an Indian company in Swiss Francs. Bharat Petroleum
Corporation raised 175 million Swiss Francs by selling five year bonds at 2.98
% coupon rate in February.
Indian Oil
Corporation plans to invest Rs 7650 crore in setting up a petrochemical complex
at its almost complete Paradip refinery in Odhisha in three to four years. The
company board is set to consider the setting up of a 700000 tonne per annum
polypropylene plant at an estimated cost at Rs.3150 crore.
Global chief
information officers at gathering in Bangalore in April to meet Indian startups
at an event called Tech50 Watchout for Little Eye Labs-Facebook type deals in
the making
EXTERNAL AGENCY RATING
|
Rating Agency Name |
ICRA |
|
Rating |
Long term fund based limits: “AA” |
|
Rating Explanation |
High degree of safety and very low credit
risk. |
|
Date |
November, 2013 |
|
Rating Agency Name |
ICRA |
|
Rating |
Short term non-fund based limits: “A1+” |
|
Rating Explanation |
Very strong degree of safety and lowest
credit risk. |
|
Date |
November, 2013 |
RBI DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available RBI Defaulters’ list.
EPF (Employee Provident Fund) DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available EPF (Employee Provident Fund) Defaulters’ list as of
31-03-2012.
INFORMATION DECLINED
MANAGEMENT NON-COOPERATIVE (Tel. No.: 91-22-304666666)
LOCATIONS
|
Registered
Office : |
Peninsula Corporate Park,
Nicholas Piramal Tower, Ganpatrao Kadam Marg, Lower Parel, Mumbai – 400013,
Maharashtra, India |
|
Tel No.: |
91-22-30466666 |
|
Fax No.: |
Not Available |
|
Email : |
secretarial.department@piramal.com |
|
Website : |
|
|
|
|
|
Head Office : |
100, Centrepoint,
Dr. Ambedkar Road, Parel, Mumbai – 400012, Maharashtra, India |
|
Tel. No.: |
91-22-66636666/24134653/24102082 |
|
Fax No.: |
91-22-24163787/24172861/24163787/24144687/24902363 |
|
E-Mail : |
|
|
Website : |
|
|
|
|
|
Administrative
Office : |
Morarjee Mills
Compound, Administrative Building, Dr. Ambedkar Road, Parel, Mumbai - 400012,
Maharashtra, India |
|
Tel. No.: |
91-22-66636666 |
|
Fax No.: |
91-22-66636416 |
|
E-Mail : |
|
|
Website : |
|
|
|
|
|
Plant 1: |
Plot No. 67-70, Sector II, Pithampur - 454775, Madhya Pradesh, India |
|
|
|
|
Plant 2 : |
Plot No. K-I, Additional MIDC, Mahad, District Raigad, Maharashtra, India |
|
|
|
|
Plant 3: |
Ennore Express Highway, Chennai - 600057, Tamilnadu, India |
|
|
|
|
Plant 4: |
N.H.9, Digwal Village, Kohir Mandal, Medak District - 502321, Andhra Pradesh, India |
|
|
|
|
Plant 5: |
C-301/1 T.T.C. Industrial Area, Pawne Mahape, Navi Mumbai - 400705, Maharashtra, India |
|
|
|
|
Plant 6: |
Plot No. 6505 /3, Sachin – 394230, Surat, Gujarat, India |
|
|
|
|
Plant 7: |
Plot No.19 - PHARMEZ, Village Matoda, Sarkhej bawala, NH 8A,
Taluka Sanand, Ahmedabad - 382213, Gujarat, India |
|
|
|
|
Plant 8: |
Shirish Research Campus, Plot No – 18, PHARMEZ, Special Economic
Zone, Taluka Sanand, Ahmedabad, Gujarat, India |
|
|
|
|
Overseas Plant Location |
|
|
Plant 1: |
Piramal Healthcare UK
Limited Morpeth, Northumberland, UK |
|
|
|
|
Plant 2: |
Piramal Healthcare UK
Limited Grangemouth, Stirlingshire, UK |
|
|
|
|
Plant 3: |
Piramal Healthcare
(Canada) Limited 110, Industrial Parkway North, Aurora, Ontario, L4G 3H4, Canada |
|
|
|
|
Plant 4: |
Piramal Healthcare
(Canada) Limited 475, Boul, Armand-Frappier, Laval, Quebec, H7V 4B3, Canada |
|
|
|
|
Plant 5: |
Piramal Critical Care
Inc. Bethlehem, PA 18017, 3950 Schelden Circle, Pennsylvania State,
USA. |
|
|
|
|
Plant 6: |
Piramal Critical Care
Inc. 50 Cobham Dr, Orchard Park, New York - 14127 |
DIRECTORS
As on 31.03.2013
|
Name : |
Mr. Ajay G. Piramal |
|
Designation : |
Chairman |
|
Qualification : |
B. Sc, M.M.S., A.M.P. |
|
Date of Appointment : |
01.04.1997 |
|
|
|
|
Name : |
Dr. (Mrs.) Swati A. Piramal |
|
Designation : |
Vice Chairperson |
|
Qualification : |
M.B.B.S, D.I.M., M.P.B. (Harvard) |
|
Date of Appointment : |
01.10.1994 |
|
|
|
|
Name : |
Mr. Gautam Banerjee |
|
Designation : |
Director (w.e.f. April 1, 2013) |
|
|
|
|
Name : |
Mr. Amit Chandra |
|
Designation : |
Director |
|
|
|
|
Name : |
Mr. Keki Dadiseth |
|
Designation : |
Director |
|
|
|
|
Name : |
Mr. R.A. Mashelkar |
|
Designation : |
Director |
|
|
|
|
Name : |
Mr. Goverdhan Mehta |
|
Designation : |
Director |
|
|
|
|
Name : |
Mr. Siddharth Mehta |
|
Designation : |
Director (w.e.f. April 1, 2013) |
|
|
|
|
Name : |
Ms. Nandini Piramal |
|
Designation : |
Executive Director |
|
|
|
|
Name : |
Mr. S. Ramadorai |
|
Designation : |
Director |
|
|
|
|
Name : |
Mr. Deepak Satwalekar |
|
Designation : |
Director |
|
|
|
|
Name : |
Mr. Vijay Shah |
|
Designation : |
Executive Director and Chief Operating Officer |
|
|
|
|
Name : |
Mr. N. Vaghul |
|
Designation : |
Director |
MAJOR SHAREHOLDERS / SHAREHOLDING PATTERN
As on 31.12.2013
|
Category of
Shareholder |
Number
of Shares |
Percentage
of Holding |
|
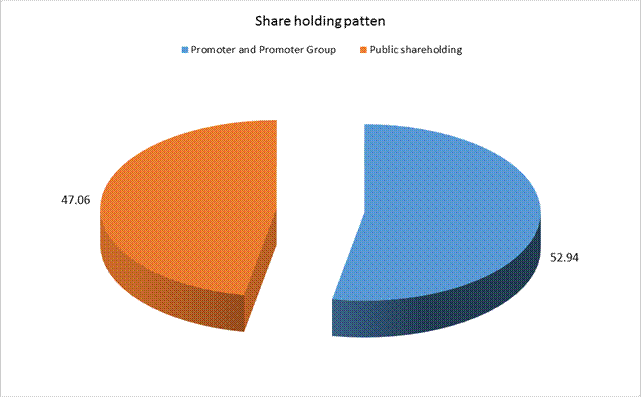
(A) Shareholding of Promoter and Promoter Group |
|
|
|
|
|
|
|
|
388753 |
0.23 |
|
|
88617771 |
51.35 |
|
|
2355234 |
1.36 |
|
|
2355234 |
1.36 |
|
|
91361758 |
52.94 |
|
|
|
|
|
Total shareholding of Promoter and Promoter Group (A) |
91361758 |
52.94 |
|
(B) Public Shareholding |
|
|
|
|
|
|
|
|
153606 |
0.09 |
|
|
5110692 |
2.96 |
|
|
213 |
0.00 |
|
|
1571602 |
0.91 |
|
|
45921177 |
26.61 |
|
|
52757290 |
30.57 |
|
|
|
|
|
|
2491131 |
1.44 |
|
|
|
|
|
|
18391204 |
10.66 |
|
|
2623889 |
1.52 |
|
|
4937828 |
2.86 |
|
|
507597 |
0.29 |
|
|
106834 |
0.06 |
|
|
2512 |
0.00 |
|
|
3946 |
0.00 |
|
|
28 |
0.00 |
|
|
4316911 |
2.50 |
|
|
28444052 |
16.48 |
|
Total Public shareholding (B) |
81201342 |
47.06 |
|
Total (A)+(B) |
172563100 |
100.00 |
|
(C) Shares held by Custodians and against which Depository Receipts
have been issued |
|
|
|
|
0 |
0.00 |
|
|
0 |
0.00 |
|
|
0 |
0.00 |
|
Total (A)+(B)+(C) |
172563100 |
100.00 |
BUSINESS DETAILS
|
Line of Business : |
The Subject is engaged in pharmaceutical business
including its research and development. |
||||||||
|
|
|
||||||||
|
Products : |
|
PRODUCTION STATUS (As on 31.03.2011)
|
Particulars |
Unit |
Installed
Capacity |
Actual
Production |
|
Tablets |
Mios |
7975.0 |
6373.5 |
|
Capsules |
Mios |
270.0 |
180.7 |
|
Liquids |
KLs |
9801.4 |
7340.1 |
|
Powders, creams and ointments |
MTs |
-- |
32.9 |
|
Bulk drug and intermediates |
MTs |
2090.3 |
1430.7 |
|
Vitamin A in various forms and combinations |
mmu |
276.0 |
136.8 |
Notes:
1.
Includes products processed by third parties.
2.
Includes production for captive consumption of Bulk Drugs 98328 kgs (PY 91850
kgs) and Vitamins 110.27 mmu (PY 138.33 mmu)
3.
Stocks are net of breakages and unsalable stock.
4.
Opening stocks, production, purchases and closing stocks are net of physician
samples.
5.
Licensed Capacity is not indicated as Industrial Licensing for all Bulk Drugs,
Intermediates and their Formulations stands abolished in terms of Press Note
No.4 (1994 series) dated 25th October, 1994 issued by the Department of
Industrial Development, Ministry of Industry Government of India.
6.
Excludes free samples issued.
7.
Variation in quantity/value is on account of change in product mix.
8. In
terms of Press Note No. 4 (1994 series) dated October 25, 1994 issued by the
Department of Industrial Development, Ministry of Industry, Government of
India, and Notification No. S.O 137 (E) dated March 1, 1999 issued by the
Department of Industrial Policy and Promotion, Ministry of Industry, Government
of India, industrial licensing has been abolished in respect of Bulk Drugs and
Formulations.
9. The
Pharmaceuticals business comprises of Manufacturing and trading of bulk drugs
and formulations.
10.
Installed capacities of the formulation factories of the Company (except where
continuous processes are involved) are on a triple shift basis are certified by
the Management and have not been verified by the Auditors, this being a
technical matter.
GENERAL INFORMATION
|
No. of Employees : |
2976
(Approximately) |
||||||||||||||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||||||||||||||
|
Bankers : |
· Allahabad Bank · Australia and New Zealand Banking Group Limited · BNP Paribas · Citibank N.A. · Credit Agricole Corporate and Investment Bank · HDFC Bank Limited · Kotak Mahindra Bank Limited · The Hongkong and Shanghai Banking Corporation Limited ·
Yes Bank Limited |
||||||||||||||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||||||||||||||
|
Facilities : |
|
||||||||||||||||||||||||||||||||||||
|
Banking
Relations : |
-- |
|
|
|
|
Auditors : |
|
|
Name : |
Price Waterhouse Chartered Accountants |
|
|
|
|
Solicitors: |
|
|
Name : |
Crawaford Bayely and Company |
|
|
|
|
Controlling
Companies : |
· The Swastik Safe Deposits and Investments Limited The Ajay G. Piramal Foundation* Paramount Pharma Private Limited* (upto October 30, 2012) BMK Laboratories Private Limited (upto October 30, 2012) Cavaal Fininvest Private Limited* (upto October 30, 2012) PHL Holdings Private Limited (formerly Known as Piramal
International Private Limited) Piramal Healthcare Limited – Senior Employee Option Scheme Piramal Enterprises Limited - Trustees of Piramal
Enterprises Executive Trust Piramal Life Sciences Limited - Senior Employees Stock
Option Trust |
|
|
|
|
Subsidiary
Companies/ step down Subsidiaries : |
Located in India · PHL Fininvest Private Limited (PHL Fininvest) Piramal Pharmaceutical Development Services Private Limited
(PPDSPL) Oxygen Bioresearch Private Limited PHL Capital Private Limited (PHL Capital) PHL Finance Private Limited (PHL Finance) PHL Infrastructure Finance Company Private Limited (PHL
Infra) Indiareit Fund Advisors Private Limited Piramal Systems and Technologies Private Limited (Piramal
Systems) Located Outside
India · Piramal International Piramal Holdings (Suisse) SA (Piramal Holdings) Piramal Pharma Inc (Formerly known as NPIL Pharma Inc,
USA) Piramal Healthcare Inc. Piramal Investment Holdings (Canada) Inc. Piramal Life Sciences (UK) Limited Piramal Healthcare UK Limited (Piramal Healthcare UK) Piramal Healthcare Pension Trustees Limited Piramal Healthcare (France) Limited (upto August 7, 2012) Piramal Healthcare (Canada) Limited (Piramal Healthcare,
Canada) Oxygen Healthcare Limited, UK (Oxygen Healthcare) Piramal Critical Care Italia, SPA Piramal Critical Care Inc (PCCI) Minrad EU Indiareit Investment Management Company, Mauritius Piramal Technologies SA Piramal Imaging SA Piramal Imaging GmbH (formerly known as “Piramal Molecular
Imaging Development GmbH”) Piramal Dutch Holdings N.V. (w.e.f October 17, 2012) Piramal Critical Care Deutschland GmbH (w.e.f April 16,
2012) Piramal Resources Inc. (w.e.f. May 22, 2012) DRI Holdco Inc (w.e.f. June 7, 2012) AMR/Arlington Medical Resources LLC (w.e.f. June 7, 2012) Arlington Medical International Inc (w.e.f. June 7, 2012) Biotrends Research Group LLC (w.e.f. June 7, 2012) Decision Resources Inc (w.e.f. June 7, 2012) Decision Resources LLC (w.e.f. June 7, 2012) (DRL) Decision Resources International Inc. (w.e.f. June 7,
2012) Decision Resources Group UK Limited (w.e.f. November 21,
2012) DR/ Decision Resources LLC (w.e.f. June 7, 2012) DR/MRG Holdings LLC (w.e.f. June 7, 2012) DRG UK Holdco Limited (w.e.f. November 21, 2012) Fingertip Formulary LLC (w.e.f. June 7, 2012) Healthleaders LLC (w.e.f. June 7, 2012) Manhattan Research LLC (w.e.f. June 7, 2012) Pharmastrat LLC (w.e.f. June 7, 2012) Millenium Research Group (w.e.f. June 7, 2012) Sigmatic Limited (w.e.f. December 03, 2012) |
|
|
|
|
Other related
parties where common control exists : |
· Piramal Glass Limited (PGL) Piramal Life Sciences Limited (PLSL) Piramal Corporate Services Private Limited (formerly known
as “Piramal Enterprises Limited”) (PCSPL) Piramal Estates Private Limited (formerly known as Piramal
Realty Limited) (Piramal Estates) India Venture Advisors Private Limited (India Venture) Allergan India Private Limited (Allergan) Piramal Foundation for Educational Leadership (PFEL) Health Management and Research Institute (HMRI) |
|
|
·
|
|
Investing parties
with whom the Company is a JV Partner: |
Allergan Inc. |
CAPITAL STRUCTURE
As on 31.03.2013
Authorised Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
250000000 |
Equity Shares |
Rs.2/- each |
Rs.500.000 Millions |
|
3000000 |
Preference Shares |
Rs.100/- each |
Rs.300.000 Millions |
|
24000000 |
Preference Shares |
Rs.10/- each |
Rs.240.000 Millions |
|
105000000 |
Unclassified Shares |
Rs.2/- each |
Rs.210.000 Millions |
|
|
|
|
|
|
|
Total |
|
Rs.1250.000 Millions |
Issued, Subscribed & Paid-up Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
172563100 |
Equity Shares |
Rs.2/- each |
Rs.345.100 Millions |
|
|
|
|
|
NOTE
1 Reconciliation of number of shares
Equity Shares
|
Particulars |
No. of
shares |
Rs. In
Millions |
|
At
the beginning of the year |
172563100 |
345.100 |
|
Less:
Shares bought back during the year |
- |
- |
|
Add:
Issued during the year |
- |
- |
|
At
the end of the year |
172563100 |
345.100 |
2 Details of shareholders holding more than 5%
shares in the Company
|
Particulars |
No. of shares |
% Holding |
|
PHL
Holdings Private Limited (formerly known as Piramal International Private
Limited) |
84092879 |
48.73% |
|
Aberdeen
Global Indian Equity Fund Mauritius Limited |
12401000 |
7.19% |
|
Paramount
Pharma Private Limited |
- |
- |
|
BMK
Laboratories Private Limited |
- |
- |
|
Cavaal
Fininvest Private Limited |
- |
- |
3 Aggregate number of shares issued for
consideration other than cash and shares bought back during the period of five
years immediately preceding reporting financial year
|
particulars |
Financial
Year |
No. of shares |
|
i.
Equity Shares allotted as fully paid-up pursuant to demerger of R&D NCE
division of Piramal Life Sciences Limited (PLSL) into the Company |
2011-12 |
5352585 |
|
ii. Equity shares bought back by the Company |
2011-12 |
705529 |
|
iii.
Equity shares bought back by the Company |
2010-11 |
41097100 |
4 Rights, preferences
and restrictions attached to shares
Equity Shares
The Company has one class of equity shares having a par value of Rs. 2/- per share. Each shareholder is eligible for one vote per share held. The dividend proposed by the Board of Directors is subject to the approval of the shareholders in the ensuing Annual General Meeting, except in case of interim dividend. In the event of liquidation, the equity shareholders are eligible to receive the remaining assets of the Company after distribution of all preferential amounts, in proportion to their shareholding.
FINANCIAL DATA
[all figures are
in Rupees Millions]
ABRIDGED BALANCE
SHEET
|
SOURCES OF FUNDS |
31.03.2013 |
31.03.2012 |
31.03.2011 |
|
EQUITY AND LIABILITIES |
|
|
|
|
(1)Shareholders' Funds |
|
|
|
|
(a) Share Capital |
345.100 |
345.100 |
335.800 |
|
(b) Reserves & Surplus |
105214.000 |
111062.700 |
116649.300 |
|
(c) Money received against share warrants |
0.000 |
0.000 |
0.000 |
|
|
|
|
|
|
(2) Share Application money pending allotment |
0.000 |
0.000 |
0.000 |
|
Total Shareholders’
Funds (1) + (2) |
105559.100 |
111407.800 |
116985.100 |
|
|
|
|
|
|
(3) Non-Current
Liabilities |
|
|
|
|
(a) long-term borrowings |
7333.300 |
2166.700 |
2392.000 |
|
(b) Deferred tax liabilities (Net) |
0.000 |
1005.300 |
923.300 |
|
(c) Other long term liabilities |
763.100 |
1328.400 |
1889.800 |
|
(d) long-term provisions |
198.500 |
199.700 |
155.100 |
|
Total Non-current
Liabilities (3) |
8294.900 |
4700.100 |
5360.200 |
|
|
|
|
|
|
(4) Current
Liabilities |
|
|
|
|
(a) Short term borrowings |
37292.600 |
9754.500 |
470.700 |
|
(b) Trade payables |
3316.300 |
3126.100 |
2761.300 |
|
(c) Other current liabilities |
5326.300 |
4856.000 |
3473.400 |
|
(d) Short-term provisions |
3634.800 |
3599.500 |
2404.400 |
|
Total Current
Liabilities (4) |
49570.000 |
21336.100 |
9109.800 |
|
|
|
|
|
|
TOTAL |
163424.000 |
137444.000 |
131455.100 |
|
|
|
|
|
|
ASSETS |
|
|
|
|
(1) Non-current
assets |
|
|
|
|
(a) Fixed Assets |
|
|
|
|
(i) Tangible assets |
6541.700 |
6319.000 |
5331.600 |
|
(ii) Intangible Assets |
1191.300 |
1285.200 |
1331.300 |
|
(iii) Capital work-in-progress |
454.400 |
230.300 |
292.500 |
|
(iv) Intangible assets under development |
1204.900 |
1434.800 |
5.900 |
|
(b) Non-current Investments |
88212.800 |
67636.000 |
4825.300 |
|
(c) Deferred tax assets (net) |
0.000 |
0.000 |
0.000 |
|
(d) Long-term Loan and Advances |
4323.000 |
1170.300 |
598.500 |
|
(e) Other Non-current assets |
21737.100 |
41255.800 |
57854.600 |
|
Total Non-Current
Assets |
123665.200 |
119331.400 |
70239.700 |
|
|
|
|
|
|
(2) Current assets |
|
|
|
|
(a) Current investments |
8018.200 |
3466.900 |
11007.200 |
|
(b) Inventories |
2617.100 |
2667.100 |
2302.000 |
|
(c) Trade receivables |
2430.300 |
2424.800 |
2090.900 |
|
(d) Cash and cash equivalents |
242.400 |
132.400 |
17540.300 |
|
(e) Short-term loans and advances |
14106.700 |
8579.600 |
9169.300 |
|
(f) Other current assets |
12344.100 |
841.800 |
19105.700 |
|
Total Current
Assets |
39758.800 |
18112.600 |
61215.400 |
|
|
|
|
|
|
TOTAL |
163424.000 |
137444.000 |
131455.100 |
PROFIT & LOSS
ACCOUNT
|
|
PARTICULARS |
31.03.2013 |
31.03.2012 |
31.03.2011 |
|
|
|
SALES |
|
|
|
|
|
|
|
Income |
14031.900 |
11534.800 |
8144.400 |
|
|
|
Other Income |
3760.100 |
5344.300 |
4722.900 |
|
|
|
TOTAL (A) |
17792.000 |
16879.100 |
12867.300 |
|
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
|
|
Cost of raw and packing materials consumed |
6333.300 |
5389.500 |
3545.200 |
|
|
|
Purchases of stock-in-trade |
792.600 |
809.600 |
807.800 |
|
|
|
Changes in inventories of finished
goods, work-in progress and stock-in-trade |
(20.400) |
(125.100) |
(377.600) |
|
|
|
Employee benefits expense |
1594.900 |
1517.500 |
1449.200 |
|
|
|
Other Expenses |
6592.500 |
5312.500 |
3986.600 |
|
|
|
Exceptional Items |
0.000 |
0.000 |
(162099.000) |
|
|
|
TOTAL (B) |
15292.900 |
12904.000 |
(152687.800) |
|
|
|
|
|
|
|
|
Less |
PROFIT/
(LOSS) BEFORE INTEREST, TAX, DEPRECIATION AND AMORTISATION (A-B) (C) |
2499.100 |
3975.100 |
165555.100 |
|
|
|
|
|
|
|
|
|
Less |
FINANCIAL
EXPENSES (D) |
4199.700 |
1999.000 |
800.600 |
|
|
|
|
|
|
|
|
|
|
PROFIT/
(LOSS) BEFORE TAX, DEPRECIATION AND AMORTISATION (C-D) (E) |
(1700.600) |
1976.100 |
164754.500 |
|
|
|
|
|
|
|
|
|
Less/ Add |
DEPRECIATION/
AMORTISATION (F) |
778.200 |
763.900 |
589.900 |
|
|
|
|
|
|
|
|
|
|
PROFIT/ (LOSS)
BEFORE TAX (E-F) (G) |
(2478.800) |
1212.200 |
164164.600 |
|
|
|
|
|
|
|
|
|
Less |
TAX (H) |
(163.200) |
(95.000) |
35195.500 |
|
|
|
|
|
|
|
|
|
|
PROFIT/ (LOSS)
AFTER TAX (G-H) (I) |
(2315.600) |
1307.200 |
128969.100 |
|
|
|
|
|
|
|
|
|
|
PREVIOUS
YEARS’ BALANCE BROUGHT FORWARD |
51974.000 |
57683.000 |
4606.000 |
|
|
|
|
|
|
|
|
|
|
APPROPRIATIONS |
|
|
|
|
|
|
|
Proposed dividend |
|
|
|
|
|
|
– Equity Shares |
3020.000 |
3020.000 |
2007.000 |
|
|
|
- Dividend Distribution Tax thereon |
513.000 |
490.000 |
326.000 |
|
|
|
Transfer to General Reserve |
0.000 |
131.000 |
73986.000 |
|
|
|
Transfer from Debenture Redemption Reserve |
0.000 |
0.000 |
500.000 |
|
|
|
Transfer to Debenture Redemption Reserve |
0.000 |
75.000 |
75.000 |
|
|
|
P&L Debit balance of Demerged R&D NCE Unit |
0.000 |
3300.000 |
0.000 |
|
|
|
|
|
|
|
|
|
BALANCE CARRIED
TO THE B/S |
46126.000 |
51974.000 |
57682.000 |
|
|
|
|
|
|
|
|
|
|
EARNINGS IN
FOREIGN CURRENCY |
|
|
|
|
|
|
|
Export Earnings |
7987.100 |
6515.300 |
4286.300 |
|
|
|
Interest on Loans |
772.100 |
334.300 |
284.400 |
|
|
|
Other Earnings |
131.500 |
196.100 |
63.200 |
|
|
TOTAL EARNINGS |
8890.700 |
7045.700 |
4633.900 |
|
|
|
|
|
|
|
|
|
|
IMPORTS |
|
|
|
|
|
|
|
Raw Materials |
3116.400 |
2285.000 |
1823.600 |
|
|
|
Capital Goods |
277.900 |
287.400 |
76.500 |
|
|
|
Traded Goods / reagents |
271.400 |
278.900 |
221.500 |
|
|
TOTAL IMPORTS |
3665.700 |
2851.300 |
2121.600 |
|
|
|
|
|
|
|
|
|
|
Earnings Per
Share (Rs.) |
(13.40) |
7.70 |
572.80 |
|
QUARTERLY RESULTS
|
Particular |
30.06.2013 |
30.09.2013 |
31.12.2013 |
31.03.2014 |
|
Type |
1st Quarter |
2nd Quarter |
3rd Quarter |
4th Quarter |
|
Net Sales |
4556.400 |
5424.800 |
5518.400 |
5447.100 |
|
Total Expenditure |
3990.200 |
4388.900 |
4517.500 |
4894.800 |
|
PBIDT (Excl OI) |
566.200 |
1035.900 |
1000.900 |
552.300 |
|
Other Income |
1146.100 |
680.100 |
142.600 |
(116.100) |
|
Operating Profit |
1712.300 |
1716.000 |
1143.500 |
436.200 |
|
Interest |
2561.700 |
1533.600 |
1772.100 |
2258.400 |
|
Exceptional Items |
0.000 |
180.000 |
0.000 |
0.000 |
|
PBDT |
(849.400) |
362.400 |
(628.600) |
(1822.200) |
|
Depreciation |
188.100 |
189.600 |
192.800 |
191.700 |
|
Profit Before Tax |
(1037.500) |
172.800 |
(821.400) |
(2013.900) |
|
Tax |
0.000 |
0.000 |
0.000 |
0.000 |
|
Provisions and contingencies |
0.000 |
0.000 |
0.000 |
0.000 |
|
Profit After Tax |
(1037.500) |
172.800 |
(821.400) |
(2013.900) |
|
Extraordinary Items |
0.000 |
0.000 |
0.000 |
0.000 |
|
Prior Period Expenses |
0.000 |
0.000 |
0.000 |
0.000 |
|
Other Adjustments |
0.000 |
0.000 |
0.000 |
0.000 |
|
Net Profit |
(1037.500) |
172.800 |
(821.400) |
(2013.900) |
KEY RATIOS
|
PARTICULARS |
|
31.03.2013 |
31.03.2012 |
31.03.2011 |
|
PAT / Total Income |
(%) |
(13.01) |
7.74 |
1002.30 |
|
|
|
|
|
|
|
Net Profit Margin (PBT/Sales) |
(%) |
(17.67) |
10.51 |
2015.67 |
|
|
|
|
|
|
|
Return on Total Assets (PBT/Total Assets} |
(%) |
(3.37) |
1.78 |
129.95 |
|
|
|
|
|
|
|
Return on Investment (ROI) (PBT/Networth) |
|
(0.02) |
0.01 |
1.40 |
|
|
|
|
|
|
|
Debt Equity Ratio (Total Debt/Networth) |
|
0.42 |
0.11 |
0.02 |
|
|
|
|
|
|
|
Current Ratio (Current Asset/Current Liability) |
|
0.80 |
0.85 |
6.72 |
FINANCIAL ANALYSIS
[all figures are
in Rupees Millions]
DEBT EQUITY RATIO
|
Particular |
31.03.2011 |
31.03.2012 |
31.03.2013 |
|
|
(Rs.
In Millions) |
(Rs.
In Millions) |
(Rs.
In Millions) |
|
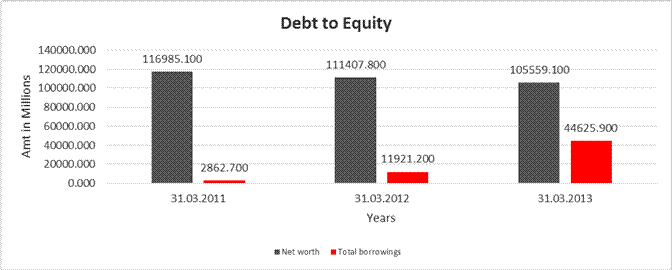
Share Capital |
335.800 |
345.100 |
345.100 |
|
Reserves & Surplus |
116649.300 |
111062.700 |
105214.000 |
|
Net
worth |
116985.100 |
111407.800 |
105559.100 |
|
|
|
|
|
|
long-term borrowings |
2392.000 |
2166.700 |
7333.300 |
|
Short term borrowings |
470.700 |
9754.500 |
37292.600 |
|
Total
borrowings |
2862.700 |
11921.200 |
44625.900 |
|
Debt/Equity
ratio |
0.024 |
0.107 |
0.423 |
YEAR-ON-YEAR GROWTH
|
Year
on Year Growth |
31.03.2011 |
31.03.2012 |
31.03.2013 |
|
|
(Rs.
In Millions) |
(Rs.
In Millions) |
(Rs.
In Millions) |
|
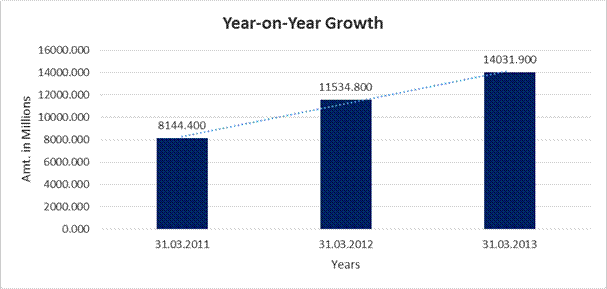
Sales |
8144.400 |
11534.800 |
14031.900 |
|
|
|
41.629 |
21.648 |
NET PROFIT MARGIN
|
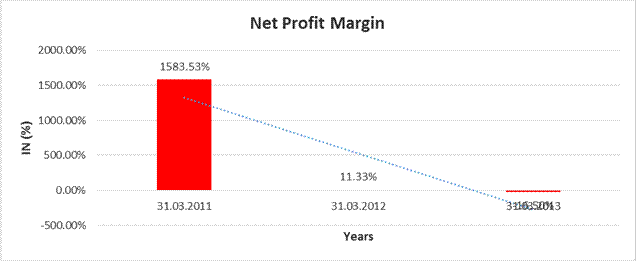
Net
Profit Margin |
31.03.2011 |
31.03.2012 |
31.03.2013 |
|
|
(Rs.
In Millions) |
(Rs.
In Millions) |
(Rs.
In Millions) |
|
Sales |
8144.400 |
11534.800 |
14031.900 |
|
Profit |
128969.100 |
1307.200 |
(2315.600) |
|
|
1583.53% |
11.33% |
(16.50%) |
LOCAL AGENCY FURTHER INFORMATION
|
Sr. No. |
Check List by Info
Agents |
Available in Report
(Yes / No) |
|
1] |
Year of Establishment |
Yes |
|
2] |
Locality of the firm |
Yes |
|
3] |
Constitutions of the firm |
Yes |
|
4] |
Premises details |
No |
|
5] |
Type of Business |
Yes |
|
6] |
Line of Business |
Yes |
|
7] |
Promoter's background |
Yes |
|
8] |
No. of employees |
Yes |
|
9] |
Name of person contacted |
No |
|
10] |
Designation of contact person |
No |
|
11] |
Turnover of firm for last three years |
Yes |
|
12] |
Profitability for last three years |
Yes |
|
13] |
Reasons for variation <> 20% |
-- |
|
14] |
Estimation for coming financial year |
No |
|
15] |
Capital in the business |
Yes |
|
16] |
Details of sister concerns |
Yes |
|
17] |
Major suppliers |
No |
|
18] |
Major customers |
No |
|
19] |
Payments terms |
No |
|
20] |
Export / Import details (if applicable) |
No |
|
21] |
Market information |
-- |
|
22] |
Litigations that the firm / promoter involved in |
-- |
|
23] |
Banking Details |
Yes |
|
24] |
Banking facility details |
Yes |
|
25] |
Conduct of the banking account |
-- |
|
26] |
Buyer visit details |
-- |
|
27] |
Financials, if provided |
Yes |
|
28] |
Incorporation details, if applicable |
Yes |
|
29] |
Last accounts filed at ROC |
Yes |
|
30] |
Major Shareholders, if available |
Yes |
|
31] |
Date of Birth of Proprietor/Partner/Director, if available |
No |
|
32] |
PAN of Proprietor/Partner/Director, if available |
No |
|
33] |
Voter ID No of Proprietor/Partner/Director, if available |
No |
|
34] |
External Agency Rating, if available |
Yes |
INDEX OF CHARGES
|
S.NO. |
CHARGE ID |
DATE OF CHARGE CREATION/MODIFICATION |
CHARGE AMOUNT SECURED |
CHARGE HOLDER |
ADDRESS |
SERVICE REQUEST NUMBER (SRN) |
|
1 |
10418435 |
11/12/2013 * |
3,500,000,000.00 |
IDBI TRUSTEESHIP SERVICES LIMITED |
ASIAN BUILDING, GROUND FLOOR, 17, R.K. KAMANI MARG, MUMBAI - 400001, MAHARASHTRA, INDIA |
B93656551 |
|
2 |
90218157 |
27/06/2012 * |
5,000,000,000.00 |
ALLAHABAD BANK |
INDUSTRIAL FINANCE BRANCH,ALLAHABAD BANK BUILDING, SECOND FLOOR, 37, MUMBAI SAMACHAR MARG, FORT, MUMBAI - 400023, MAHARASHTRA, INDIA |
B44005106 |
|
3 |
90218094 |
06/06/1997 |
6,500,000.00 |
DRESDNER BANK AG |
HOECHST HOUSE; 1ST
FLOOR, NARIMAN POINT, MUMBAI - 400021, |
- |
|
4 |
90217538 |
22/09/1993 * |
30,000,000.00 |
UNIT TRUST OF INDIA |
13, SIR VITHALDAS THACKERSEY MARG, MUMBAI - 400020, MAHARASHTRA - 400020, INDIA |
- |
|
5 |
90218748 |
07/12/1982 |
2,000,000.00 |
CANARA BANK |
CHEMBUR MAIN BRANCH,
MUMBAI - 400071, MAHARASHTRA, |
- |
* Date of charge modification
UNSECURED LOAN
|
PARTICULAR |
31.03.2013 (Rs. In
Millions) |
31.03.2012 (Rs. In
Millions) |
|
LONG TERM
BORROWINGS |
|
|
|
9.66% - Unsecured Redeemable Non Convertible Debentures (Redeemable at par at the end of 5th year from the date of allotment - February 26, 2013) |
3500.000 |
0.000 |
|
Term Loan From Banks |
333.300 |
666.700 |
|
SHORT-TERM
BORROWINGS |
|
|
|
Commercial Papers |
33500.000 |
9000.000 |
|
Loans repayable on demand - from banks |
2250.000 |
0.000 |
|
Total |
39583.300 |
9666.700 |
Note:
Terms of repayment and
rate of interest for secured borrowings (other than debentures)
|
|
As at March 31, 2013 |
|
|
|
Terms of Repayment |
Rate of Interest |
|
Loan from Central Bank of India |
Repayable in 12 equal Quarterly installments from June 2012 |
Interest to be paid monthly @ Bank Prime Lending Rate minus 2% p.a upto February 8, 2013. Interest to be paid monthly @ Bank Rate (floating) plus 1% p.a from February 9, 2013. |
GENERAL
INFORMATION
The Company is engaged in pharmaceutical business including its research and development. The Company has diversified into Financial Services and Information Management business through its subsidaries. The Company has manufacturing plants in India and sells in Domestic as well as International markets through various distribution channels. The Company is a public limited company and is listed on the Bombay Stock Exchange (BSE) and the National Stock Exchange (NSE).
SIGNIFICANT EVENTS
DURING THE FINANCIAL YEAR 2013:
ACQUISITION OF
DECISION RESOURCES GROUP (US) AND ABACUS INTERNATIONAL (UK)
Decision Resources Group (DRG), a US based provider of high quality, web-enabled research, predictive analytics via proprietary databases and consulting services to the global healthcare industry, was acquired in June 2012 for a consideration of approximately US$ 635 million (~Rs. 34000.000 Millions). It is one of the fastest growing companies with a CAGR of 20% for the last five years. It has 48 of the top 50 global pharmaceutical companies as its customers. The Company has retained 94% of its customers of 2012. DRG reported revenue of Rs. 6500.000 Millions in FY2013 since its acquisition. Abacus International, UK was acquired in December 2012 to help expand in the European markets.
ACQUISITION OF
BAYER’S MOLECULAR IMAGING DEVELOPMENT PORTFOLIO
Piramal Enterprises Limited (‘PEL’), through its newly established subsidiary – Piramal Imaging SA, acquired the molecular imaging research and development portfolio, including rights to Florbetaben, of Bayer Pharma AG in April 2012. Florbetaben, a PET tracer for the detection of beta-Amyloid plaque deposition in the brain, is the pathological hallmark of disease in probable Alzheimer’s disease patients. The molecule has been accepted for review by the European Medicines Agency (EMA) and the US Food and Drug Administration (USFDA) in March 2013.
EXPANSION OF
FINANCIAL SERVICES BUSINESS
PEL has started lending to the education sector and has explored new geographies like Bhopal, Coimbatore and NCR this year. The loan book for the Real Estate and Education Sector has increased to Rs. 15910.000 Millions as on March 31, 2013 from Rs. 3510.000 Millions as on March 31, 2012. PEL invested Rs. 4250.000 Millions in Optionally Convertible Debentures of Navayuga Road Projects Private Limited in March 2013. INDIAREIT, which manages funds for investments in real estate sector has Rs. 42570.000 Millions under management as on March 31, 2013. INDIAREIT raised Rs. 4000.000 Millions for Mumbai Redevelopment Fund during FY2013 and is currently raising a new domestic fund with targeted fund size of Rs. 7500.000 Millions along with green shoe option of Rs. 2500.000 Millions.
OPERATIONS REVIEW:
Total Operating Income on a standalone basis for the year grew by 15% to Rs. 16200.000 Millions against Rs. 14082.000 Millions in FY2012. EBIDTA on a standalone basis was lower at Rs. 2499.000 Millions against Rs. 3975.000 Millions in FY2012 due to increase in R and D expenses and lower foreign exchange gain. Loss for the year was at Rs. 2316.000 Millions as against profit after tax of Rs. 1307.000 Millions in FY2012. This is on account of an increase of 110.1% in interest cost due to increase in loans taken to fund the DRG acquisition and to fund the lending operations of the NBFC business. Earnings per share were Rs. (13.4) for the year.
MERGER OF PHL
HOLDINGS PRIVATE LIMITED INTO PEL
The Scheme of Amalgamation and Arrangement between PHL Holdings Private Limited and Piramal Enterprises Limited and their respective shareholders and creditors (“Scheme”), which was approved by the shareholders on March 13, 2013, is awaiting approval of the Hon’ble High Court of Judicature at Bombay. The Appointed Date of the Scheme is January 1, 2013. The Scheme inter alia provides for merger of PHL Holdings Private Limited into PEL and thereby cancellation of shares held by PHL Holdings Private Limited in PEL and consequent reissue of equivalent number of shares of PEL to the shareholders of PHL Holdings Private Limited.
MANAGEMENT DISCUSSION
AND ANALYSIS
FY2013 KEY
HIGHLIGHTS:
ACQUISITION OF
DECISION RESOURCES GROUP, INC. (US) AND ABACUS INTERNATIONAL (UK)
Piramal Enterprises Limited (PEL) completed acquisition of Decision Resources Group (DRG), a US based provider of high quality, web-enabled research, predictive analytics via proprietary databases and consulting services to the global healthcare industry, for a consideration of approximately US$ 635 million (~ Rs. 3,400 Millions) in June 2012. With 20% revenue CAGR for the last five years, it is one of the fastest growing companies in the US$ 5.7 billion global healthcare information industry. It has 48 of the top 50 global pharmaceutical companies as its customers. The overall customer retention rate is 94%. The company, now a subsidiary of PEL, generated revenues of Rs. 6496.000 Millions in FY2013. In December 2012, DRG acquired Abacus International, a UK based pioneer in evidence-based global market access solutions for many of the world’s leading healthcare companies.
ACQUISITION OF
BAYER’S MOLECULAR IMAGING DEVELOPMENT PORTFOLIO
PEL acquired worldwide rights to the molecular imaging research and development portfolio of Bayer Pharma AG through its newly created subsidiary – Piramal Imaging SA in April 2012. The portfolio includes rights to Florbetaben amongst a pipeline of other molecules. Florbetaben is a PET tracer for the detection of beta-Amyloid plaque deposition in the brain, which is the pathological hallmark of disease in probable Alzheimer’s disease patients. Phase III trial showed that PET imaging with Florbetaben reliably detects beta-Amyloid in the brain during life with great accuracy and thus shows value as a potential tool to aid in the diagnosis and assessment of Alzheimer’s disease. The molecule has been accepted for review by the US Food and Drug Administration (FDA) in March 2013 and by the European Medicines Agency (EMA) in January 2013.
EXPANSION OF
FINANCIAL SERVICES BUSINESS
During this year, PEL started lending to the education sector and has explored new geographies like Bhopal, Coimbatore and NCR. The loan book to Real Estate and Education Sectors has increased to Rs. 15910.000 Millions as on March 31, 2013 from Rs.3510.000 Millions as on March 31, 2012. PEL has also made two structured investments aggregating to Rs. 9250.000 Millions. The first investment of Rs. 4250.000 Millions was made in March 2013 through Optionally Convertible Debentures in Navayuga Road Projects Private Limited (NRPL), the road subsidiary of the Rs. 30000.000 Millions Navayuga Engineering Company (NECL). Another investment of Rs. 5000.000 Millions was made in April 2013 through Optionally Convertible Debentures in Green Infra Limited., one of the largest clean energy Independent Power Producers (IPPs) in the country funded by IDFC Private Equity.
INDIAREIT, which manages funds for investments in real estate sector, has Rs. 42570.000 Millions under management as on March 31, 2013. INDIAREIT raised Rs. 4000.000 Millions for Mumbai Redevelopment Fund during FY2013 and is currently raising a new domestic fund with targeted fund size of Rs. 7500.000 Millions along with green shoe option of Rs. 2500.000 Millions.
BUSINESS PERFORMANCE
PHARMACEUTICALS BUSINESSES
PHARMA SOLUTIONS
The market for Global Outsourcing is expected to grow to US$ 45 Bn by 2015 at a compounded annual growth rate of 12% per year. The global outsourcing trend is likely to continue, driven by the rising healthcare costs in developed countries and reducing profitability of pharmaceutical companies due to patent expiry of various blockbuster drugs. Long term prospects for the industry are backed by cost pressures for innovative companies and increasing genericisation. Established relationships and trust with innovators, quality infrastructure, good regulatory track record and low production and R and D costs should place PEL in a good position to take advantage of this trend.
In the context of this market outlook, PEL’s revenues from Pharma Solutions business grew by 14.7% to Rs. 15533.000 Millions in FY2013 as compared with Rs. 13545.000 Millions in FY2012. The revenues from both Indian assets and assets outside India grew by 14.7% to Rs. 9185.000 Millions against Rs. 8008.000 Millions in FY2012 and to Rs. 6348.000 Millions against Rs. 5537.000 Millions in FY2012, respectively.
Increase in demand for existing products was seen during the year. Order booking for the late phase commercial business increased in FY2013 as compared with FY2012. Long term commercial contracts were signed at Morpeth in FY2013. Their relationship with PEL’s key partner, Pfizer, at Morpeth, continued to grow. The year saw a strong pipeline of enquiries, especially for the ADCs business at Grangemouth. In October 2012, PEL entered into a strategic alliance with Fujifilm Diosynth Biotechnologies, whereby the two parties intend to offer seamless contract development and manufacture of ADCs, a growing niche in the development of biopharmaceuticals.
CRITICAL CARE
Revenue from Critical Care business was Rs. 6161.000 Millions as compared with Rs. 4126.00 Millions in FY2012, registering a growth of 49.3%. The high growth rate is on account of gaining greater share of the Sevoflurane market in the US and some other emerging mark0ets. PEL’s market share in terms of volume for Sevoflurane in US has increased from 25% in March 2012 to 30% in March 2013. Major Group Purchasing Organization (GPO) contracts have been renewed for next two years in the US. PEL has more than 50% market share in some of the emerging markets. There has been a meaningful increase in market share in countries such as Turkey and Indonesia. Several tenders were won this year in emerging markets including Mexico, Peru, Kazakhstan and South Africa. Sevoflurane was also launched in the European markets this year. The product will be marketed by direct field force in some countries while association with marketing partners will continue in most of the other countries. Isoflurane sales volumes were also higher as compared with last year.
OTC AND OPHTHALMOLOGY
Sales from OTC and Ophthalmology business grew by 23.3% to Rs. 2712.000 Millions in FY2013 as compared with Rs. 2200.000 Millions in FY2012. The OTC sales force and distribution network now covers 400,000 retailers in 485 ‘one-lakh+ towns’ in India. New products and brands were launched through FY2013 – Lacto Calamine Reneu, Lacto Calamine Sun Screen, Polycrol+, extensions of Jungle Magic brand of perfumes and Jungle Magic Mosquito Bandtiz. ‘Lacto Face wash’ was awarded the ‘Star 2012’ for excellence in packaging by Indian Institute of Packaging.We launched several new OTC products and brands in FY13,viz. Lacto Calamine Reneu, Lacto Calamine Sun Screen, Polycrol+, extensions of Jungle Magic brand of perfumes and Jungle Magic Mosquito Bandtiz.
Allergan India Limited (AIL) is a 51:49 Joint Venture for Ophthalmic products between Allergan Inc., USA and PEL. Total revenues of AIL were Rs. 2054.000 Millions (FY2012 Total Revenue: Rs. 1686.000 Millions). The Operating Profit for FY2013 was Rs. 456.000 Millions as compared with Rs. 403.000 Millions in FY2012. Profit after Tax for FY2013 was Rs. 292.000 Millions as compared with Rs. 256.000 Millions for FY2012.
LIFE SCIENCES
PEL is working on discovering and optimizing compounds to meet important unmet medical needs in the target markets of cancer, diabetes and inflammation. Spend on R and D expenses for FY2013 was Rs. 2867.000 Millions vis-à-vis Rs. 2331.000 Millions in FY2012.
NBFC OPERATIONS
During the year, PEL expanded its NBFC operations and started lending to the education sector along with the Real Estate Sector. The lending operations are currently focused in Mumbai, Pune, Bengaluru, Chennai, Bhopal, Coimbatore and NCR. A highly experienced and knowledgeable team, well defined systems and processes and robust credit approval and monitoring policies are in place. As of March 31, 2013, the total loan book for real estate and education sector stood at Rs. 15910.000 Millions as compared with Rs. 3510.000 Millions as on March 31, 2012. PEL also invested Rs. 4250.000 Millions in Optionally Convertible Debentures of Navayuga Road Projects Private Limited (NRPL) in March 2013. Similarly, another investment of Rs. 5000.000 Millions was made in Optionally Convertible Debentures of Green Infra Limited in April 2013. PEL is exploring opportunities either in yield assets (e.g. toll roads) or as mezzanine financing for a portfolio of cash generating and under execution projects.
BOARD OF DIRECTORS
Mr. N. Vaghul
Mr. N. Vaghul is a B. Com. (Hons.) from the University of Madras. He joined the State Bank of India in 1957 as a
Probationary Officer. He became a Director in the National Institute of Bank Management, Mumbai in 1976, before assuming charge as Executive Director in Central Bank of India in 1978. He became Chairman of Bank of India in 1981 and had the distinction of being the youngest ever Chairman in a Public Sector Bank. He joined ICICI Bank Limited as Chairman and CEO in 1985 and continued to head the group till April 2009. During his tenure in ICICI, he created several new institutions laying the foundation for the development of Universal Banking model (which he pioneered). He was also instrumental in setting up ICICI Securities, an Investment Banking company. He also pioneered the concept of Credit Rating in India by setting up CRISIL. He was the founder Chairman of CRISIL for close to ten years and helped in evolving the best practices of credit rating in the country.
He was the Chairman of “Pratham”, a leading NGO. He is associated with several foundations dedicated to the cause of primary education. He is also deeply committed to the cause of science and technology and was responsible for setting up the first Science and Technology Park in the country known as ICICI Knowledge Park. He is associated with the Institute of Technology in Jaipur. He has also been the Chairman of IFMR, a Business School with an array of research centers engaged in a variety of economic and social research.
Mr. Vaghul is the recipient of numerous awards and honours. He was chosen as the Business Man of the Year by ‘Business India’ in 1992. He received the Lifetime Achievement Award from Economic Times in 2006 and also the “Ernst and Young Entrepreneur of the Year Award Program” in 2009. He also received an award for his contribution to Corporate Governance from the Institute of Company Secretaries in 2007. He was awarded ‘Padma Bhushan’ by the Government of India in 2009. He was awarded Life Time Achievement Award by Bombay Management Association in March 2013.
Mr. Deepak Satwalekar
Mr. Deepak Satwalekar received a B. Tech. in Mechanical Engineering from IIT, Bombay, and a M.B.A. from The American University, Washington D.C.
He retired as the Managing Director and Chief Executive Officer (MD and CEO) of HDFC Standard Life Insurance Company Limited. in November 2008. Before taking on the responsibility of setting up and running the new Insurance company in the year 2000, he was the Managing Director of HDFC, the country’s largest mortgage lender. He has consulted for the World Bank, the Asian Development Bank and other bilateral and multilateral agencies in several countries.
Besides being a recipient of the “Distinguished Alumnus Award” from IIT, Bombay, he is on the Advisory Council of the IIT, Bombay. He has been a member of/chaired several industry, Reserve Bank of India and government expert groups. Inter alia, he has been/is a member of the Technical Advisory Committee of the RBI, the Committee for Policyholder Protection/Intermediaries, etc. of the Insurance Regulatory and Development Authority, Committee for Restructuring the NPS set up by the PFRDA. He also serves as an independent director on the boards of some large companies in India.
He serves on the India Advisory Board of a large European Bank and is currently active on the Board of Trustees of Isha Vidhya Gyan Prakash Foundation and Teach to Lead, which are engaged in the field of primary education for the low income and socially disadvantaged members of society in rural and urban India respectively. In the field of higher education, he is on the board of the Indian Institute for Human Settlements. He is on the Board of Trustees of the Infosys Science Foundation. He is an advisor to Private Equity and Venture Capital firms as also on the Board of Society for Innovation and Entrepreneurship, an early funding vehicle set up at IIT, Bombay. He is also advising a company which is establishing a network of BPO centers in rural areas across the country.
Dr. R.A. Mashelkar
Dr. R.A. Mashelkar is a B.E., Ph.D (Chemical Engineering) from the Institute of Chemical Technology, Mumbai. He is a National Research Professor and presently the President of Global Research Alliance, a network of publicly funded R and D institutes from Asia-Pacific, Europe and USA with over 60,000 scientists.
Dr. Mashelkar is only the third Indian engineer to have been elected (1998) as Fellow of Royal Society (FRS), London in the twentieth century. He was elected Foreign Associate of National Academy of Science (USA) in 2005, Associate Foreign Member, American Academy of Arts and Sciences (2011), Foreign Fellow of US National Academy of Engineering (2003), Fellow of Royal Academy of Engineering, U.K. (1996), Foreign Fellow of Australian Technological Science and Engineering Academy (2008) and Fellow of World Academy of Art and Science, USA (2000).
Thirty universities have honoured him with honorary doctorates, which include Universities of London, Salford, Pretoria, Wisconsin and Delhi.
The President of India honoured Dr. Mashelkar with Padmashree (1991) and with the Padmabhushan (2000), which are two of the highest civilian honours, in recognition of his contribution to nation building.
As Chairman of the Standing Committee on Information Technology of the World Intellectual Property Organization (WIPO), a member of the International Intellectual Property Rights Commission of the UK Government and as Vice Chairman of the Commission in Intellectual Property Rights, Innovation and Public Health (CIPIH) set up by World Health Organization (WHO), he brought new perspectives on the issue of IPR and the developing world concerns. He was a member of the Scientific Advisory Council to the Prime Minister and also of the Scientific Advisory Committee to the Cabinet set up by successive governments. He has chaired twelve high powered committees set up to look into diverse issues of higher education, national auto fuel policy, overhauling the Indian drug regulatory system, dealing with the menace of spurious drugs and reforming Indian agriculture research system, amongst others. Dr. Mashelkar has won over 50 awards and medals, which include S.S. Bhatnagar Prize (1982), Pandit Jawaharlal Nehru Technology Award (1991), G.D. Birla Scientific Research Award (1993), Material Scientist of Year Award (2000), IMC Juran Quality Medal (2002), HRD Excellence Award (2002), Lal Bahadur Shastri National Award for Excellence in Public Administration and Management Sciences (2002), World Federation of Engineering Organizations (WFEO) Medal of Engineering Excellence by WFEO, Paris (2003), Lifetime Achievement Award by Indian Science Congress (2004), the Science medal by the Academy of Science for the Developing World (2005) and Ashutosh Mookherjee Memorial Award by Indian Science Congress (2005).
Prof. Goverdhan Mehta
Prof. Goverdhan Mehta is a FRS, Ph.D., (D.Sc. h.c), National Research Professor and Bhartia-Jubilant Chair at the University of Hyderabad. He is an internationally acclaimed researcher in organic chemistry with wide ranging research interests. He has held faculty position at the Indian Institute of Technology, Kanpur and is a former Vice Chancellor of the University of Hyderabad. He was the Director of the Indian Institute of Science, a premier institution of the country and was a CSIR Bhatnagar Fellow at I. I. Sc, Bangalore. He has made many notable contributions in the area of organic synthesis. He has been exceptionally prolific in his scientific contributions, publishing more than 450 original research papers in international Journals of high repute and presented over 200 lectures in different parts of the world. He is on the Editorial Boards of many leading international journals in Chemical Sciences/Organic Chemistry and mentored over a hundred doctoral and post-doctoral students/scientists. He has also contributed to science education, science policy and planning and RandD efforts in India and abroad in many ways.
Professor Mehta has over fifty awards and honors to his credit, notable among these being the Humboldt Research Prize of Germany, Medals from the Third World Academy of Sciences, Trieste and Science Academies in India. He has been conferred ‘Chevalier de la Legion d’Honneur’ and Officier dans I’ordre Palmes Academiques by the President of France and Padma Shri by the President of India. He is a Fellow of Royal Society (FRS), Third World Academy of Sciences and other Science Academies. He has been President of the Indian National Science Academy (INSA) and of the Paris based International Council for Science (ICSU) as well as co-Chair of the Inter Academy Council.
Mr. Siddharth Mehta
Mr. Siddharth Mehta has an undergraduate degree from the London School of Economics and MBA from the University of Chicago. He is a director and the former President and Chief Executive Officer of TransUnion. He was also the Chief Executive Officer of HSBC Finance Corporation and HSBC North America Holdings, Inc. Prior to HSBC, he served as a Senior Vice President at the Boston Consulting Group in Los Angeles and as co-leader of the Boston Consulting Group’s Financial Services Practice where he developed retail, insurance and investment strategies for a variety of financial service clients. He also serves on the board of directors of TransUnion LLC, DataCard Group, The Chicago Public Education Fund, The Field Museum and the Myelin Repair Foundation. He has executive level experience and extensive knowledge of the banking industry and financial markets.
Mr. Mehta does not hold any directorship in any public limited companies or subsidiaries of public limited companies in
India.
Mr. Gautam Banerjee
Mr. Gautam Banerjee is a graduate from the University of Warwick, England and is a member of the Institute of Chartered Accountants in England and Wales and also a member of the Institute of Certified Public Accountants, Singapore. Mr. Banerjee is currently a Senior Advisor with Blackstone Group, Singapore. Mr. Banerjee has been associated with Pricewaterhouse Coopers (“PwC”), one of the Big 4 Auditing Firms worldwide, from 1982 to 2012, holding various positions in PwC during his long association. He was Chairman of PwC Singapore and the COO for PwC’s Eastern Cluster and a member of PwC Global Firm’s Strategy Council from October 2008 to December 2012. He was also the Interim Chairman of PwC India from December 2009 to December 2010.
Mr. Banerjee has been a member of various statutory boards / committees of the Government of Singapore and also a member of several Industry Associations in Singapore. Presently, he is a member of the Economic Development Board of Singapore and Vice Chairman of the Singapore Business Federation. He is also an Independent Director of Singapore Airlines Limited, The Government of Singapore Investment Company Limited. and the Straits Trading Company Limited. Mr. Banerjee has extensive knowledge and skills in the realm of finance, accounting and management. He was a nominated Member of Parliament in Singapore from 2007 to 2009.
Mr. Banerjee does not hold any directorship in any public limited companies or subsidiaries of public limited companies in India.
CONTINGENT
LIABILITIES
(Rs. In Millions)
|
Particular |
31.03.2013 |
31.03.2012 |
|
1 Claims against
the Company not acknowledged as debt: |
|
|
|
Demand dated June 5, 1984 the Government has asked for payment to the credit of the Drugs Prices Equalisation Account, the difference between the common sale price and the retention price on production of Vitamin ‘A’ Palmitate (Oily Form) from January 28, 1981 to March 31, 1985 not accepted by the Company. The Company has been legally advised that the demand is untenable. |
6.100 |
6.100 |
|
2 Guarantees issued to Government authorities and limited companies including guarantees issued on behalf of subsidiaries and performance guarantees. |
36441.400 |
10893.600 |
|
3 Others |
|
|
|
i. Appeals filed in respect of disputed demands: |
|
|
|
Income Tax |
|
|
|
- where the Company is in appeal |
3597.700 |
4207.100 |
|
- where the Department is in appeal |
1384.300 |
1710.800 |
|
Sales Tax |
123.600 |
143.700 |
|
Central / State Excise |
116.900 |
110.500 |
|
Labour Matters |
3.300 |
2.900 |
|
Stamp Duty |
40.500 |
40.500 |
|
Legal Cases |
70.700 |
70.700 |
|
ii. Bills Discounted |
539.400 |
240.000 |
|
iii. Unexpired Letters of Credit |
77.500 |
132.100 |
Note: Future cash outflows in respect of 1 and 3(i) above are determinable only on receipt of judgments/decisions pending with various forums/authorities.
STATEMENT OF STANDALONE AUDITED RESULTS FOR THE QUARTER AND YEAR ENDED
MARCH 31, 2014
(Rs. In Millions)
|
Particulars |
3 months ended |
Preceding 3 months ended |
Year to date figures for current period ended |
|
31/03/2014 |
31/12/2013 |
31/03/2014 |
|
|
(Unaudited) |
(Unaudited) |
(Audited) |
|
|
Income from Operations |
|
|
|
|
Net Sales / Income from Operations (Net of excise duty) |
4777.800 |
4791.100 |
18134.700 |
|
Other Operating Income |
669.300 |
727.300 |
2796.100 |
|
Total Income from Operations (Net) |
5447.100 |
5518.400 |
20930.800 |
|
|
|
|
|
|
Expenses |
|
|
|
|
Cost of Materials Consumed |
1945.100 |
1848.500 |
7006.700 |
|
Purchase of Stock-in-Trade |
196.100 |
223.900 |
849.700 |
|
Changes in Inventories of Finished Goods, work-in-progress and stock-in-trade |
(144.400) |
(59.200) |
(377.100) |
|
Employee Benefits Expenses |
496.200 |
577.500 |
2135.000 |
|
Depreciation and amortisation Expense |
191.700 |
192.800 |
762.200 |
|
Research and Development Expenses |
528.500 |
494.500 |
2401.200 |
|
Other Expenses |
1873.300 |
1432.300 |
5775.900 |
|
Total Expenses |
5086.500 |
4710.300 |
18553.600 |
|
|
|
|
|
|
Profit/ (Loss)
from Operations before
Other Income, Finance Costs and
Exceptional Items |
360.600 |
808.100 |
2377.200 |
|
Other Income |
(116.100) |
142.600 |
1868.600 |
|
Profit/ (Loss) from
ordinary activities before Finance Costs and Exceptional Items |
244.500 |
950.700 |
4245.800 |
|
Finance Costs |
2258.400 |
1772.100 |
8125.800 |
|
Profit/ (Loss) from
ordinary activities after Finance Costs but before exceptional items |
(2013.900) |
(821.400) |
(3880.000) |
|
Exceptional Items |
-- |
-- |
180.000 |
|
Profit/ (Loss) from
ordinary activities before Tax |
(2013.900) |
(821.400) |
(3700.000) |
|
Tax Expense (Including Deferred Tax) |
-- |
-- |
-- |
|
Net Profit/ (Loss)
from ordinary activities after tax |
(2013.900) |
(821.400) |
(3700.000) |
|
Extraordinary items (net of tax expense) |
-- |
-- |
-- |
|
Net Profit/ (Loss) for the period |
(2013.900) |
(821.400) |
(3700.000) |
|
Paid up Equity Share Capital (Face Value of Rs.2/- Each) |
345.100 |
345.100 |
345.100 |
|
Reserves (excluding Revaluation Reserves) |
|
|
90925.700 |
|
Paid-up Debt Capital |
|
|
3500.000 |
|
Debenture Redemption Reserve |
|
|
300.000 |
|
Earnings Per Share (EPS) |
|
|
|
|
a) Basic and diluted EPS before extraordinary items for the period (Rs.) |
(11.60) |
(4.80) |
(21.40) |
|
b) Basic and diluted EPS after extraordinary items for the period (Rs.) |
(11.60) |
(4.80) |
(21.40) |
|
Debt Service Coverage Ratio (Refer Footnote No.1) |
|
|
0.90 |
|
Interest Service Coverage Ratio (Refer Footnote No.2) |
|
|
0.40 |
|
|
|
|
|
|
Note: 1. Debt Service Coverage Ratio – Earnings before interest and Tax/ (Interest Expense + Principal Repayment) 2. Interest Service Coverage Ratio – Earnings before
interest and Tax / Interest Expense |
|||
|
|
|
|
|
|
A. PARTICULARS OF
SHAREHOLDING |
|
|
|
|
1.Public shareholding |
|
|
|
|
- Number of shares |
81201342 |
81198825 |
81201342 |
|
- Percentage of shareholding |
47.06% |
47.05% |
47.06% |
|
2. Promoters and promoter group shareholding |
|
|
|
|
a) Pledged/Encumbered |
|
|
|
|
- Number of shares |
-- |
-- |
-- |
|
- Percentage of shares (as a % of the total shareholding of promoter and promoter group) |
-- |
-- |
-- |
|
- Percentage of shares (as a % of the total share capital of the Company) |
-- |
-- |
-- |
|
b. Non-encumbered |
|
|
|
|
- Number of shares |
91361758 |
91364275 |
91361758 |
|
- Percentage of shares (as a % of the total shareholding of promoter and promoter group) |
100.00% |
100.00% |
100.00% |
|
- Percentage of shares (as a % of the total share capital of the Company) |
52.94% |
52.95% |
52.94% |
|
Particulars |
3 months ended 31/03/2014 |
|
B INVESTOR COMPLAINTS (Nos.) |
|
|
Pending at the beginning of the quarter |
2 |
|
Received during the quarter |
3 |
|
Disposed of during the quarter |
4 |
|
Remaining unresolved at the end of the quarter |
1 |
NOTE
1. The standalone financial results, for the quarter and nine months ended March 31, 2014, have been reviewed by the Audit Committee and approved by the Board of Directors at its meeting held on May 05, 2014.
2. Statement of
Assets and Liabilities:
(Rs. In Millions)
|
Particular |
Current
year end (31.03.2014) |
|
|
(Audited) |
|
EQUITY AND
LIABILITIES |
|
|
Shareholders’
funds |
|
|
Share capital |
345.100 |
|
Reserves and surplus |
90925.700 |
|
Sub-total
- Shareholders' funds |
91270.800 |
|
|
|
|
Non-current
liabilities |
|
|
Long-term borrowings |
6533.400 |
|
Deferred Tax Liabilities (Net) |
-- |
|
Other long-term liabilities |
234.200 |
|
Long Term Provisions |
529.400 |
|
Sub-total
- Non-current liabilities |
7297.000 |
|
|
|
|
Current
liabilities |
|
|
Short-term borrowings |
51597.800 |
|
Trade payables |
3838.200 |
|
Other current liabilities |
4439.600 |
|
Short-term provision |
10719.500 |
|
Sub-total - Current
liabilities |
70595.100 |
|
TOTAL - EQUITY AND
LIABILITIES |
|
|
|
|
|
ASSETS |
|
|
Non-current
assets |
|
|
Fixed assets |
9425.500 |
|
Non-current investments |
114848.700 |
|
Long-term loans and advances |
5778.100 |
|
Other Non-current assets |
1572.400 |
|
Sub-total
- Non-current assets Current assets |
131624.700 |
|
Current assets |
|
|
Current Investments |
1012.900 |
|
Inventories |
3004.900 |
|
Trade receivables |
2562.700 |
|
Cash and cash equivalents |
318.500 |
|
Short-term loans and advances |
22777.200 |
|
Other current assets |
7862.000 |
|
Sub-total
- Current assets |
37538.200 |
|
TOTAL
- ASSETS |
169162.900 |
3. In accordance with Accounting Standard-17 "Segment Reporting', segment Information has been given in the consolidated financial results of Piramal Enterprises Limited and therefore, no separate disclosure on segment information is given in these result.
4. A Dividend of Rs.52.50 per equity share (2625% of the face value of Rs.21- each) has been recommended by the Board of Directors which is subject to approval of the Shareholders.
5. Other Income includes the effect of Foreign Exchange Gain / (Loss) - (net) incurred during the respective quarter / year.
6. Subsequent to the year-end:
a) the Company has divested its entire equity stake, comprising of 45,425,328 shares, in Vodafone India Limited to Prime Metals Limited, an indirect subsidiary of Vodafone Group Plc., for a total consideration of Rs. 8,900 Crores;
b) The Company has acquired an effective 20 % equity stake in Shriram Capital Limited, a financial services company, for an aggregate consideration of Rs. 2,014 Crores; and
c) The Company has agreed to acquire 9.99 % of the post-diluted equity share capital of Shriram City Union Finance Limited, by way of subscription to fresh shares pursuant to a preferential allotment, for an aggregate consideration of Rs. 790 Crores.
7. The figures for the last quarter are balancing figures between the audited figures in respect of the full financial year and the published year to date figures upto the third quarter of the current financial year.
8. Previous quarter / year's figures have been regrouped / reclassified wherever necessary to correspond with the current quarter / year's classification / disclosure.
FIXED ASSETS
v
Tangible
Assets
Land
Leasehold
Land
Freehold
Building
Plant
and Equipment
Furniture
and Fixtures
Vehicles/Ships/Helicopter
Office
Equipment
v
Intangible Assets
Brands
and Trademarks
Copyrights,
Know-how and Intellectual Property Rights
Computer
Software
PRESS RELEASE
DR. RAMAN SINGH, CHIEF MINISTER OF CHHATTISGARH AND HMRI (PIRAMAL FOUNDATION)
INAUGURATES 104 HEALTH INFORMATION HELPLINE IN RAIPUR
HMRI, an initiative
under Piramal Foundation, has served over 66 million callers till date
Raipur Health
Helpline can handle 2,500 calls per day
Employs the use of
Piramal HMRI’s innovative algorithm and disease summary software
Raipur, Chattisgarh, September 23, 2013: Piramal Foundation’s primary healthcare initiative, Health Management and Research Institute (HMRI) and Dr. Raman Singh, the Chief Minister of Chhattisgarh inaugurated the 104 Health Information Helpline. The ceremony witnessed the presence of Shri. Amar Agarwal - Health Minister, Shri Ramesh Bias, MP – Raipur, Shri Brij Mohan Agarwal - Minister of Education, Smt Kiranmayee Nayak –Raipur Mayor, Shri M.K Raut (IAS) - Prl.Secretary – Health, Shri Pratap Singh (IFS) - Commissioner HM and FW, Dr. K.P.Singh – DHS.
The 30 seater Health Information Helpline would operate on 24 x7, throughout the year. The call centre is located in Raipur and can handle a call volume of 2,500 calls a day.
The call center is equipped with well trained paramedics, counselors, doctors, specialists and uses HMRI’s algorithm and disease summary. The paramedics aid in providing basic medical advice whereas the doctors provide expert advice at the call centre. Advice is also provided for long-term illness conditions such as diabetes, heart issues, cancer, common ailments and chronic and acute medical issues.
The team also shares much needed information during local epidemics and health scares, rehab counseling (alcohol, drugs, smoking) and psychological counseling (anxiety, depression, suicidal tendencies, chronic diseases like cancer etc.). Experts address issues related to family planning, stigmatized diseases (HIV, AIDS, and leprosy), birth disorders like cleft lip and club foot among the new born, childcare, pregnancy and safe birth
practices as well as sickle cell anemia.
In the future, the Raipur health helpline can be connected further to telemedicine node that would access and be connected to medical colleges and district hospitals.
Other services offered by the Piramal Foundation led HMRI initiative are 104 health helplines in Assam, Rajasthan, Maharasthra and Karnataka in partnership with the respective state Governments and has served over 66 million callers till date. Recently, HMRI has won the bid for the helpline in the state of Jharkhand. HMRI is poised for a growth with ambitious footprint plans across States.
PIRAMAL ENTERPRISES LIMITED RECEIVES
IND APPROVAL FOR P7435, ITS NOVEL DGAT1 INHIBITOR FOR TREATMENT OF LIPID
DISORDERS AND DIABETES
P7435 Investigational New Drug (IND) dossier receives USFDA
regulatory approval
Extended Phase I trial in the US to be initiated shortly
P7435 has shown a significant reduction in triglycerides,
blood glucose and insulin levels in preclinical studies
Mumbai, August 6, 2013: Piramal Enterprises Limited “PEL” (NSE: PEL, BSE:
500302) announced today that it has received approval from the US FDA for its
Investigational New Drug (IND) P7435. This is a novel, potent and highly
selective, oral diacylglycerol acyltransferase 1 (DGAT1) inhibitor.
P7435 has been developed by the NCE Research Division of PEL
for the management of metabolic disorders such as lipid abnormalities and
diabetes. It is well-established that increased lipid levels’ (including
triglycerides) is one of the major risk factors for cardiovascular disease
(CVD). It has been reported by the World Health Organisation, that CVD, is the
number one cause of deaths globally, representing approximately 30% of all
deaths. Currently, there is a significant medical need for effective and safe
drugs for the management of lipid abnormalities and metabolic disorders.
P7435 has demonstrated its lipid lowering potential in
various preclinical studies by showing significant reduction in triglyceride
levels, glucose and insulin levels, and decrease in food intake and body weight
gain - factors which are associated with lipid abnormalities and metabolic
disorders.
PEL has established the safety and tolerability of P7435 in
a Phase I trial recently completed in India. This extension trial in the US
will further evaluate the safety and efficacy of P7435 in a larger population.
Dr. Swati Piramal, Vice Chairperson, Piramal Enterprises Limited said, “The NCE
Research division of PEL continues its ambitious diabetes/metabolic disorders
programme to discover and develop NCEs to fight against diseases like diabetes
and lipid disorders. With P7435 we are looking at addressing a serious need for
effective and well-tolerated drugs that treat lipid disorders, which are
commonly associated with diabetes and CVDs. Expansion of this trial will allow
testing this NCE in a wider population, which is critical to the development of
this drug and will provide therapeutic solutions not just to India but also to
the rest of the world.”
PIRAMAL ENTERPRISES LIMITED (PEL) AND TATA MEMORIAL CENTRE (TMC) FORGE
ALLIANCE TO ADVANCE UNDERSTANDING OF CANCER BIOLOGY AND DEVELOP TOOLS FOR
PERSONALIZED MEDICINE
Focus on understanding disease biology and predicting response to
treatment
in preclinical models
Address critical need of personalized medicine in cancer care
Understand mechanism of drug action
April 1, 2013;
Mumbai: Piramal Enterprises Limited (PEL) and Tata Memorial Centre (TMC) today
announced that they will collaborate to enable the development of valuable
tools to better understand disease biology and predict responses to various
treatments for cancer patients.
The new alliance
between the two organizations will focus on the development of preclinical
cancer models to enhance the understanding of disease biology, treatment
response/resistance and biomarkers as they relate to diagnosis, prognosis and
response to drugs. These models will be based on tumor tissues from cancer patients and
are predicted to have better translational relevance than the currently used
human cancer cell line models. Scientists at TMC and PEL hope to gain a better
understanding of the mechanisms through which various drugs work on different
cancers. This research could lead to new therapies and predict the medicine
best suited to treat an individual cancer patient.
"Our
collaboration with Piramal Enterprises addresses a critical need in cancer
care, which is widely accessible and cost effective personalized medicine for
cancer patients," said Dr Rajendra Badwe, M.D., Director of Tata
Memorial Centre.
Dr Swati Piramal,
Vice Chairperson of Piramal Enterprises Ltd. commenting on the development said,
"We are excited to begin this collaboration with Tata Memorial Centre,
which is India’s leading centre associated with cancer treatment. We hope that
the insights we gain from this alliance will ultimately lead to new treatment
options for cancer patients."
The New Chemical
Entity (NCE) Research division of Piramal Enterprises focuses on the discovery
and development of innovative medicines to improve the lives of patients
suffering from cancer, diabetes, metabolic disorders and inflammatory conditions.
It has a state-of-the-art Research Centre in Mumbai and comprehensive
capabilities spanning target identification all the way through clinical
development.
PIRAMAL ENTERPRISES: UPDATES ON SCHEME OF AMALGAMATION AND ARRANGEMENT
July 03, 2013
Piramal Enterprises has informed that the Order of the Court sanctioning the scheme of Amalgamation and Arrangement between PHL Holdings and Piramal Enterprises and their respective shareholders and creditors has been filed with the Registrar of Companies on July 02, 2013 and accordingly, the Scheme has become effective.
With reference to the earlier announcement dated May 16, 2013, about the Hon'ble Bombay High Court sanctioning the scheme of Amalgamation and Arrangement between PHL Holdings Private Limited and Piramal Enterprises Limited and their respective shareholders and creditors (“the Scheme”) Piramal Enterprises Limited has informed BSE that the Order of the Court sanctioning the Scheme has been filed with the Registrar of Companies on July 02, 2013 and accordingly, the Scheme has become effective. The Appointed Date of the Scheme is January 1, 2013.In terms of the Scheme, 84092879 Equity Shares of the Company held by the Transferor i.e. PHL Holdings Private Limited (PHPL) stands cancelled and equivalent number of equity shares credited as fully paid up are to be issued and allotted by the Company to the equity shareholder of PHPL in lieu thereof. The Company is in the process of submitting necessary applications to the Stock Exchanges for seeking approval for listing of the 84092879 Equity Shares to be issued and allotted to the equity shareholders of PHPL.
PIRAMAL ENTERPRISES LIMITED RECEIVES IND APPROVAL FROM THE US FDA FOR
ITS GPR40 AGONIST P11187; AN ANTI-DIABETIC MOLECULE
· P11187 Investigational New Drug (IND) dossier receives US FDA regulatory approval
· Piramal's first US FDA approved Diabetes Phase I clinical trial; trials in the US to be initiated shortly
· P11187's anti-diabetic action is in a glucose-dependent manner, thus decreasing the potential risks of hypoglycemia
Mumbai, May 14, 2013: Piramal Enterprises Limited "PEL" (NSE: PEL, BSE: 500302) announced today that it has received approval from the US FDA for its Investigational New Drug (IND) P11187. This approval will enable PEL to initiate a Phase I clinical trial of P11187 in healthy volunteers in the US.
P11187 is an orally active, small molecule New Chemical Entity (NCE), discovered and developed by the NCE Research Division of PEL. P11187 selectively acts on GPR40; a potential therapeutic target for Type 2 Diabetes Mellitus (T2DM). T2DM is an emerging worldwide health crisis with an incidence rate of 300 million by 2025 as predicted by the WHO and accounts for about 90% of the diabetic population.
P11187 will be tested for safety and its glucose-lowering properties for the first time in humans; both properties having been well-established in our preclinical studies. Currently, the T2DM treatment space has limitations in terms of efficacy and adverse side-effect profiles. The advantage of P11187; as a GPR40 agonist, is the stimulation of insulin secretion in a glucose-dependent manner, thus reducing the potential risk of excess insulin production.
Dr. Swati Piramal, Vice Chairperson, Piramal Enterprises Limited said, "The NCE Research division of PEL is dedicated to finding new cures for metabolic disorders. It focuses upon nurturing innovation and break-through thinking to impact the lives of millions of people. P11187's IND approval by the US FDA; recognizes our untiring efforts to identify candidates that would translate into more efficacious drugs for the effective management of diabetes."
About NCE Research
The NCE Research Division of Piramal Enterprises Limited focuses on the discovery and development of innovative small molecule medicines to improve the lives of patients suffering from cancer, metabolic disorders and inflammatory conditions. The key elements of our strategy include capitalizing on Piramal's strengths, in particular the India advantage, and leveraging external partnerships to achieve high levels of RandD productivity. Piramal's state-of-the-art Research Centre in Mumbai has comprehensive capabilities spanning target identification all the way through clinical development. Our robust pipeline, including 9 compounds in clinical development, bears testimony to our innovative and rigorous drug discovery process.
Piramal Enterprises
Limited
Piramal Enterprises is one of India's largest diversified companies, with a presence in pharmaceutical, financial services and information management sectors. Piramal Enterprises had consolidated revenues of over $ 650 million in FY2013. In the pharmaceutical space, PEL is one of leading custom manufacturing player globally, has presence in the global critical care segment with a portfolio of inhalation and injectable anesthetics and its OTC business is ranked no. 7 in India. PEL is also engaged in drug discovery and research and has strong pipeline of development products. In the financial services space, PEL has a real estate focused PE fund - Indiareit and a NBFC that is focused on lending to real estate and education sector. PEL's information management business, Decision Resources Group is a leading provider of information based services to the healthcare industry.
PIRAMAL
ENTERPRISES ANNOUNCES Q1FY2014 RESULTS
Total
Operating Income for Q1FY2014 up by 29.8% to Rs. 9702.000 Millions, Operating
Profit was higher at Rs. 1461.000 Millions while net loss was Rs. 1467.000
Millions
Mumbai, July 25, 2013: Piramal Enterprises Limited (formerly Piramal Healthcare Limited) ('PEL', NSE: PEL, BSE: 500302) today announced results for Q1FY2014.
For the quarter ended June 30, 2013, the Total Operating Income grew by 29.8% to Rs. 9702.000 Millions against Rs. 7472.000 Millions in Q1FY2013. Operating Profit (OPBITDA) for the quarter was Rs. 1461.000 Millions as compared with Rs. 785.000 Millions during the same period last year. Net loss for the quarter was Rs. 1467.000 Millions as compared with a net profit of Rs. 41.000 Millions during Q1FY2013. Interest Expenses for Q1FY2014 are higher by 279.9% at Rs. 3325.000 Millions as against Rs. 875.000 Millions in Q1FY2013 as the company raised funds to invest in its NBFC operations and in shares of Shriram Transport Finance Company. The interest expenses for the quarter include one-time financing charges of Rs. 1628.000 Millions. Excluding these one-time financing charges, the PAT would have been a profit of Rs. 161.000 Millions instead of a loss of Rs. 1467.000 Millions.
During the quarter, PEL invested Rs.5000.000 Millions in Green Infra Limited through Optionally Convertible Debentures. The Company also acquired a minority stake of ~10% in Shriram Transport Finance Company Limited for Rs. 16340.000 Millions in May 2013.
FY2013 results also include the financial results of Decision Resources Group that was acquired in June 2012, Piramal Imaging operations in Germany acquired from Bayer in April 2012 and Abacus International acquired in December 2012.
THE CENTERS FOR MEDICARE AND MEDICAID SERVICES (CMS) DRAFT DECISION
COULD LIMIT PATIENT ACCESS TO NEW TECHNOLOGY THAT MAY HELP IN DIAGNOSING
DEMENTIA
Piramal Imaging Urges Public Participation During the CMS Public Comment Period on the use of Beta-Amyloid Imaging in Dementia
Berlin/Boston/Mumbai, July 25, 2013 - On Wednesday, July 3, 2013, the Centers for Medicare and Medicaid Services (CMS) released its proposed decision memorandum regarding the use of beta-amyloid imaging in dementia and neurodegenerative disease.1
Piramal Imaging is concerned about CMS' proposal, which appears to take an overly cautious approach to expanding Medicare coverage for positron emission tomography (PET) tracers to include beta-amyloid imaging agents. Limiting Medicare coverage only to patients enrolled in CMS-approved clinical trials imposes restrictions that reduce patient access to long-awaited diagnostic tools.
"We are concerned that the CMS draft coverage decision is too restrictive and, if finalized in its current form, will place an undue burden on physicians, patients and caregivers by delaying the definitive diagnosis of certain types of dementia and neurodegenerative disease, including Alzheimer's," said Dr. Ludger Dinkelborg, Director of the Board, Piramal Imaging SA.
In Piramal Imaging's view, the CMS decision needs to be reviewed for the following reasons:
The draft decision does not reference the Appropriate Use Criteria (AUC) that was developed by the Society for Nuclear Medicine and Molecular Imaging (SNMMI) and the Alzheimer's Association. The taskforce was comprised of a cross-section of experts including radiologists, nuclear medicine specialists and neurologists.2
It lacks clear guidance on clinical trial designs that should be practical and provide CMS with the requested evidence in a reasonable timeframe.
There is general disagreement with the relevance of autopsy as an appropriate endpoint to demonstrate clinical utility in the intended population.
The use of new beta-amyloid imaging agents should help to reduce uncertainties in the diagnosis of Alzheimer's disease and related dementias, which is of importance for patients and their caregivers. Piramal Imaging is hopeful that CMS will moderate its position when it renders its final decision by finding less restrictive ways to gather the information it seeks regarding health outcomes.
CMS is accepting public comments on its proposed decision from all stakeholder groups and is accepting public comments now through August 2, 2013. Comments can be posted at:
"We urge anyone concerned with Alzheimer's disease—patients, caregivers, healthcare professionals, patient advocacy groups and the general public—to share opinions on the value of beta-amyloid PET imaging during the initial 30-day public comment period," said Dr. Dinkelborg. "Every letter will have an impact on CMS' final decision in September."
About the value of
beta-amyloid PET imaging
Today, Alzheimer's disease is usually diagnosed after a person with a cognitive impairment undergoes an extensive clinical examination which typically includes family and medical history, physical and neurological examinations, laboratory tests, and imaging procedures such as computed tomography (CT) and magnetic resonance imaging (MRI) scans. Still, a definitive diagnosis of Alzheimer's disease can only be made after death where an autopsy can reveal the presence of beta-amyloid plaques and neurofibrillary tangles in the brain. However, post-mortem studies looking for accumulations of beta-amyloid in the brain have shown that 10 to 30 percent of diagnoses based on clinical examinations are incorrect.
Beta-amyloid PET imaging holds the promise to detect beta-amyloid plaques in live patients. Because beta-amyloid progresses in the brain for as long as a decade before classic signs and symptoms Alzheimer's disease become clear to diagnosticians, early detection of the pathophysiology in symptomatic patients is crucial to a more accurate assessment of the patient's condition, which may lead to a targeted and accurate therapeutic approach for those affected.
CMT REPORT (Corruption, Money Laundering & Terrorism]
The Public Notice information has been collected from various sources
including but not limited to: The Courts,
1] INFORMATION ON
DESIGNATED PARTY
No exist designating subject or any of its beneficial owners,
controlling shareholders or senior officers as terrorist or terrorist organization
or whom notice had been received that all financial transactions involving
their assets have been blocked or convicted, found guilty or against whom a
judgement or order had been entered in a proceedings for violating
money-laundering, anti-corruption or bribery or international economic or
anti-terrorism sanction laws or whose assets were seized, blocked, frozen or
ordered forfeited for violation of money laundering or international
anti-terrorism laws.
2] Court Declaration :
No exist to suggest that subject is or was
the subject of any formal or informal allegations, prosecutions or other
official proceeding for making any prohibited payments or other improper
payments to government officials for engaging in prohibited transactions or
with designated parties.
3] Asset Declaration :
No records exist to suggest that the property or assets of the subject
are derived from criminal conduct or a prohibited transaction.
4] Record on Financial
Crime :
Charges or conviction
registered against subject: None
5] Records on Violation of
Anti-Corruption Laws :
Charges or
investigation registered against subject: None
6] Records on Int’l
Anti-Money Laundering Laws/Standards :
Charges or
investigation registered against subject: None
7] Criminal Records
No
available information exist that suggest that subject or any of its principals
have been formally charged or convicted by a competent governmental authority
for any financial crime or under any formal investigation by a competent government
authority for any violation of anti-corruption laws or international anti-money
laundering laws or standard.
8] Affiliation with
Government :
No record
exists to suggest that any director or indirect owners, controlling
shareholders, director, officer or employee of the company is a government
official or a family member or close business associate of a Government
official.
9] Compensation Package :
Our market
survey revealed that the amount of compensation sought by the subject is fair
and reasonable and comparable to compensation paid to others for similar
services.
10] Press Report :
No press reports / filings exists on
the subject.
CORPORATE GOVERNANCE
MIRA INFORM as part of its Due Diligence do provide comments on
Corporate Governance to identify management and governance. These factors often
have been predictive and in some cases have created vulnerabilities to credit
deterioration.
Our Governance Assessment focuses principally on the interactions between
a company’s management, its Board of Directors, Shareholders and other
financial stakeholders.
CONTRAVENTION
Subject is not known to have contravened any existing local laws,
regulations or policies that prohibit, restrict or otherwise affect the terms
and conditions that could be included in the agreement with the subject.
FOREIGN EXCHANGE RATES
|
Currency |
Unit
|
Indian Rupees |
|
US Dollar |
1 |
Rs.59.73 |
|
|
1 |
Rs.100.82 |
|
Euro |
1 |
Rs.82.22 |
INFORMATION DETAILS
|
Information
Gathered by : |
PLA |
|
|
|
|
Analysis Done by
: |
KAR |
|
|
|
|
Report Prepared
by : |
MRI |
SCORE & RATING EXPLANATIONS
|
SCORE FACTORS |
RANGE |
POINTS |
|
HISTORY |
1~10 |
8 |
|
PAID-UP CAPITAL |
1~10 |
7 |
|
OPERATING SCALE |
1~10 |
8 |
|
FINANCIAL CONDITION |
|
|
|
--BUSINESS SCALE |
1~10 |
7 |
|
--PROFITABILIRY |
1~10 |
-- |
|
--LIQUIDITY |
1~10 |
7 |
|
--LEVERAGE |
1~10 |
7 |
|
--RESERVES |
1~10 |
8 |
|
--CREDIT LINES |
1~10 |
8 |
|
--MARGINS |
-5~5 |
- |
|
DEMERIT POINTS |
|
|
|
--BANK CHARGES |
YES/NO |
YES |
|
--LITIGATION |
YES/NO |
NO |
|
--OTHER ADVERSE INFORMATION |
YES/NO |
NO |
|
MERIT POINTS |
|
|
|
--SOLE DISTRIBUTORSHIP |
YES/NO |
NO |
|
--EXPORT ACTIVITIES |
YES/NO |
NO |
|
--AFFILIATION |
YES/NO |
YES |
|
--LISTED |
YES/NO |
YES |
|
--OTHER MERIT FACTORS |
YES/NO |
YES |
|
DEFAULTER |
|
|
|
--RBI |
YES/NO |
NO |
|
--EPF |
YES/NO |
NO |
|
TOTAL |
|
60 |
This score serves as a reference to assess SC’s credit risk
and to set the amount of credit to be extended. It is calculated from a
composite of weighted scores obtained from each of the major sections of this
report. The assessed factors and their relative weights (as indicated through
%) are as follows:
Financial
condition (40%) Ownership
background (20%) Payment
record (10%)
Credit history
(10%) Market trend
(10%) Operational
size (10%)
RATING EXPLANATIONS
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
>86 |
Aaa |
Possesses an extremely sound financial base with the strongest
capability for timely payment of interest and principal sums |
Unlimited |
|
71-85 |
Aa |
Possesses adequate working capital. No caution needed for credit
transaction. It has above average (strong) capability for payment of interest
and principal sums |
Large |
|
56-70 |
A |
Financial & operational base are regarded healthy. General
unfavourable factors will not cause fatal effect. Satisfactory capability for
payment of interest and principal sums |
Fairly Large |
|
41-55 |
Ba |
Overall operation is considered normal. Capable to meet normal
commitments. |
Satisfactory |
|
26-40 |
B |
Capability to overcome financial difficulties seems comparatively
below average. |
Small |
|
11-25 |
Ca |
Adverse factors are apparent. Repayment of interest and principal sums
in default or expected to be in default upon maturity |
Limited with
full security |
|
<10 |
C |
Absolute credit risk exists. Caution needed to be exercised |
Credit not
recommended |
|
-- |
NB |
New Business |
-- |
This report is issued at your request without any
risk and responsibility on the part of MIRA INFORM PRIVATE LIMITED (MIPL)
or its officials.