MIRA
INFORM REPORT
|
Report Date : |
14.05.2014 |
IDENTIFICATION DETAILS
|
Name : |
GRANULES INDIA LIMITED |
|
|
|
|
Registered
Office : |
Second Floor, Block III, My Home Hub, Madhapur, Cyberabad,
Hyderabad – 500 081, Andhra Pradesh |
|
|
|
|
Country : |
|
|
|
|
|
Financials (as
on) : |
31.03.2013 |
|
|
|
|
Date of
Incorporation : |
18.03.1991 |
|
|
|
|
Com. Reg. No.: |
01-012471 |
|
|
|
|
Capital
Investment / Paid-up Capital : |
Rs.201.262
millions |
|
|
|
|
CIN No.: [Company Identification
No.] |
L24110AP1991PLC012471 |
|
|
|
|
TAN No.: [Tax Deduction &
Collection Account No.] |
HYDG00432F |
|
|
|
|
PAN No.: [Permanent Account No.] |
AAACG7369K |
|
|
|
|
Legal Form : |
Public Limited Liability Company. The Company’s Shares are
Listed on the Stock Exchanges. |
|
|
|
|
Line of Business
: |
Subject is engaged in the manufacturing and selling of Active
Pharmaceutical Ingredients (APIs), Pharmaceutical Formulation Intermediates
(PFIs) and Finished Dosages (FDs). |
|
|
|
|
No. of Employees
: |
1500 (Approximately) |
RATING & COMMENTS
|
MIRA’s Rating : |
Ba (51) |
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
41-55 |
Ba |
Overall operation is considered normal. Capable to meet normal
commitments. |
Satisfactory |
|
Maximum Credit Limit : |
USD 10832000 |
|
|
|
|
Status : |
Satisfactory |
|
|
|
|
Payment Behaviour : |
Regular |
|
|
|
|
Litigation : |
Exist |
|
|
|
|
Comments : |
Subject is an established company having satisfactory track record. Profit margin of the company has increased. And financial performance
of the company appears to be fair. Trade relations are fair. Business is active. Payment terms are reported
to be regular. The company can be considered for business dealing at usual trade
terms and conditions. |
NOTES :
Any query related to this report can be made
on e-mail : infodept@mirainform.com
while quoting report number, name and date.
ECGC Country Risk Classification List – March 31, 2014
|
Country Name |
Previous Rating (31.12.2013) |
Current Rating (31.03.2014) |
|
India |
A1 |
A1 |
|
Risk Category |
ECGC
Classification |
|
Insignificant |
A1 |
|
Low |
A2 |
|
Moderate |
B1 |
|
High |
B2 |
|
Very High |
C1 |
|
Restricted |
C2 |
|
Off-credit |
D |
INDIAN ECONOMIC OVERVIEW
US investment bank
Goldman Sachs has upgraded its outlook on Indian markets as it expects positive
impact of the election cycle.
India’s economy may
grow 4.7 % in the current financial year, lower than the official estimate of
4.9 %, Fitch Rating said. The global rating agency expects the economy to pick
up in the next two financial years.
Global ratings
agency Standard & Poor said increasing focus by India Inc on lowering debt
is likely to improve their credit profiles.
Singapore (1.1
million Indian tourists in 2012), Thailand (one million), the United Arab
Emirates ().98 million) and Malaysia ().82 million) emerged as the preferred
holidays hotspots for Indians. The total figure is expected to increase to 1.93
million by 2017, according to the latest Eurmonitor international report.
There is a $29.34 bn
outward foreign direct investment by domestic companies between April and
January of 2013/14 which has seen some signs of recovery according to a Care
Ratings report.
There are 264 number
of new companies being set up every day on average during 2014. Most of them
are registered in Mumbai. India had 1.38 million registered companies at the
end of January, 2014.
Twitter like
messaging service Weibo Corporation has filed to raise $ 500 million via a US
initial public offering. Alibaba, which owns a stake in Weibo is expected to
raise about $ 15 billion New York this year in the highest profile Internet IPO
since Facebook’s in 2012.
Bharti Airtel has
raised Rs.2,453.2 crore (350 million Swiss Francs) by selling six-year bonds at
a coupon rate of three per cent and maturing in 2020. This is the largest ever
bond offering by an Indian company in Swiss Francs. Bharat Petroleum
Corporation raised 175 million Swiss Francs by selling five year bonds at 2.98
% coupon rate in February.
Indian Oil
Corporation plans to invest Rs 7650 crore in setting up a petrochemical complex
at its almost complete Paradip refinery in Odhisha in three to four years. The
company board is set to consider the setting up of a 700000 tonne per annum
polypropylene plant at an estimated cost at Rs.3150 crore.
Global chief
information officers at gathering in Bangalore in April to meet Indian startups
at an event called Tech50 Watchout for Little Eye Labs-Facebook type deals in
the making.
EXTERNAL AGENCY RATING
|
Rating Agency Name |
CARE |
|
Rating |
Long Term Bank Facilities: BBB+ |
|
Rating Explanation |
Moderate degree of safety and moderate credit risk. |
|
Date |
March 10, 2014 |
|
Rating Agency Name |
CARE |
|
Rating |
Short Term Bank Facilities: A2 |
|
Rating Explanation |
Strong degree of safety and low credit risk. |
|
Date |
March 10, 2014 |
RBI DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available RBI Defaulters’ list.
EPF (Employee Provident Fund) DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available EPF (Employee Provident Fund) Defaulters’ list as of
31-03-2012.
INFORMATION PARTED BY
|
Name : |
Mr. B. Chandrashekhar |
|
Designation : |
General Manager – Finance |
|
Contact No.: |
91-9963027747 |
|
Date : |
13.05.2014 |
LOCATIONS
|
Registered Office/ Corporate Office : |
Second Floor, Block III, My Home Hub, Madhapur, Cyberabad,
Hyderabad – 500 081, Andhra Pradesh, India |
|
Tel. No.: |
91-40-66760000 |
|
Mobile No.: |
91-9963027747 (Mr. B. Chandrashekhar) |
|
Fax No.: |
91-40-23115145 |
|
E-Mail : |
investorrelations@granulesindia.com |
|
Website: |
|
|
Area : |
10000 sq. ft. |
|
Location : |
Rented |
|
|
|
|
Factory 1 : |
Plot No.15/A/1, Phase-III,
I.D.A. Jeedimetla, Hyderabad – 500 055, Andhra Pradesh, India. |
|
|
|
|
Factory 2 : |
Temple Road, Bonthapally, Medak District – 502 313, Andhra Pradesh, India |
|
|
|
|
Factory 3 : |
Plot No.160/A and 161/E, Gagillapur Village, Qutubullapur Mandal, R.R. District – 500043, Andhra Pradesh, India |
|
|
|
|
R&D Centre 1 : |
Formulations Gagillapur, Qutubullapur Mandal, Ranga Reddy District – 500 043, Andhra Pradesh, India |
|
|
|
|
R&D Centre 2 : |
API Plot No.15/A/1, Phase-III, I.D.A. Jeedimetla, Hyderabad – 500 055, Andhra Pradesh, India |
|
|
|
|
R&D Centre 3 : |
API Gat No.258,
Shreeram Building, Lawale Phata, Pirangut, Taluka Mulshi, District Pune – 412
108, Maharashtra, India |
|
|
|
|
Overseas Offices : |
Located at: · Europe · U.S. · Canada ·
· Asia · Middle East · Africa |
DIRECTORS
AS ON 31.03.2013
|
Name : |
Dr. C. Nageswara Rao |
|
Designation : |
Chairman – Non-Executive, Non-Independent |
|
Date of Birth/Age : |
15.08.1926 |
|
Qualification : |
M. S. (Surgery and Urology) |
|
Date of Appointment : |
18.03.1991 |
|
|
|
|
Name : |
Mr. C. Krishna Prasad |
|
Designation : |
Managing Director – Non-Independent |
|
Date of Birth/Age : |
02.10.1954 |
|
Qualification : |
B. Sc. |
|
Date of Appointment : |
31.08.1994 |
|
|
|
|
Name : |
Mr. L.S. Sarma |
|
Designation : |
Director – Non-Executive, Independent |
|
|
|
|
Name : |
Mr. A.P. Kurian |
|
Designation : |
Director – Non-Executive, Independent |
|
|
|
|
Name : |
Mr. C. Parthasarathy |
|
Designation : |
Director – Non-Executive, Independent |
|
Date of Birth/Age : |
07.07.1955 |
|
Qualification : |
B. Sc., LLB, FCA, FCS |
|
Date of Appointment : |
27.05.2009 |
|
|
|
|
Name : |
Dr. Krishna Murthy Ella |
|
Designation : |
Director – Non-Executive, Independent |
|
|
|
|
Name : |
Mr. Arun Rao Akinepally |
|
Designation : |
Director – Non-Executive, Independent |
|
|
|
|
Name : |
Mr. Harsha Chigurupati |
|
Designation : |
Executive Director – Non-Independent |
|
|
|
|
Name : |
Mrs. Uma Chigurupati |
|
Designation : |
Executive Director – Non-Independent |
|
|
|
|
Name : |
Mr. K.B. Sankara Rao |
|
Designation : |
Additional Director |
KEY EXECUTIVES
|
Name : |
Mr. B. Madhusudan Rao |
|
Designation : |
Chief Operating Officer |
|
|
|
|
Name : |
Mr. VVS Murthy |
|
Designation : |
Chief Financial Officer |
|
|
|
|
Name : |
Mr. Stefan Lohle |
|
Designation : |
Chief Marketing Officer |
|
|
|
|
Name : |
Ms. Shivangi Sharma |
|
Designation : |
Company Secretary |
|
|
|
|
Name : |
Mr. B. Chandrashekhar |
|
Designation : |
General Manager – Finance |
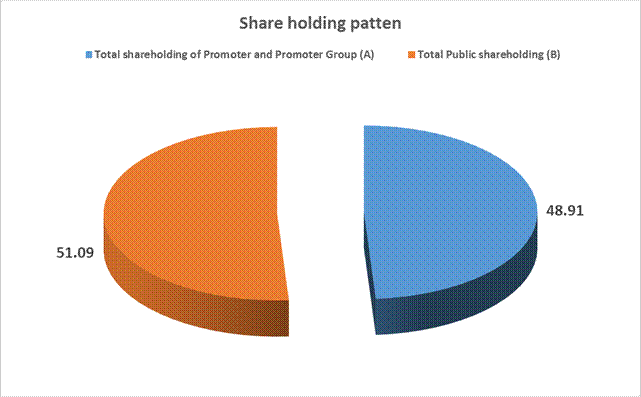
MAJOR SHAREHOLDERS / SHAREHOLDING PATTERN
AS ON 31.03.2014
|
Category of Shareholders |
No. of Shares |
Percentage of Holding |
|
(A)
Shareholding of Promoter and Promoter Group |
|
|
|
|
|
|
|
|
8983128 |
44.34 |
|
|
749127 |
3.70 |
|
|
9732255 |
48.03 |
|
|
|
|
|
|
177911 |
0.88 |
|
|
177911 |
0.88 |
|
Total
shareholding of Promoter and Promoter Group (A) |
9910166 |
48.91 |
|
(B)
Public Shareholding |
|
|
|
|
|
|
|
|
36505 |
0.18 |
|
|
342540 |
1.69 |
|
|
379045 |
1.87 |
|
|
|
|
|
|
1134345 |
5.60 |
|
|
|
|
|
|
3077396 |
15.19 |
|
|
1515508 |
7.48 |
|
|
4244694 |
20.95 |
|
|
3774067 |
18.63 |
|
|
378569 |
1.87 |
|
|
91958 |
0.45 |
|
|
100 |
0.00 |
|
|
9971943 |
49.22 |
|
Total
Public shareholding (B) |
10350988 |
51.09 |
|
Total
(A)+(B) |
20261154 |
100.00 |
|
(C)
Shares held by Custodians and against which Depository Receipts have been
issued |
0 |
0.00 |
|
|
0 |
0.00 |
|
|
20000 |
0.00 |
|
|
20000 |
0.00 |
|
Total
(A)+(B)+(C) |
20281154 |
0.00 |
BUSINESS DETAILS
|
Line of Business : |
Subject is engaged in the manufacturing and selling of Active
Pharmaceutical Ingredients (APIs), Pharmaceutical Formulation Intermediates
(PFIs) and Finished Dosages (FDs). |
||||
|
|
|
||||
|
Products : |
|
||||
|
|
|
||||
|
Exports : |
|
||||
|
Products : |
·
Pharmaceutical Formulation |
||||
|
Countries : |
·
USA ·
European Countries ·
African Countries |
||||
|
|
|
||||
|
Imports : |
|
||||
|
Products : |
·
Raw Materials |
||||
|
Countries : |
·
China |
||||
|
|
|
||||
|
Terms : |
|
||||
|
Selling : |
L/C, Cash and Credit |
||||
|
|
|
||||
|
Purchasing : |
L/C, Cash and Credit |
PRODUCTION STATUS (AS ON 31.03.2011):
|
Particulars |
Unit |
Licensed
Capacity |
Installed
Capacity |
Actual
Production |
|
APIs |
MT |
NA |
10400.00 |
9934.58 |
|
PFIs |
MT |
NA |
8400.00 |
5469.65 |
|
Formulations (Tablets) |
MT |
NA |
6000.00 |
2000.70 |
GENERAL INFORMATION
|
Customers : |
Wholesalers and Retailers |
||||||||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||||||||
|
No. of Employees : |
1500 (Approximately) |
||||||||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||||||||
|
Bankers : |
· Andhra Bank, Somajiguda Branch, 6-3-352/2, Astral Heights, Road No.1, Banjara Hills, Hyderabad – 500 034, Andhra Pradesh, India · Andhra Bank, Somajiguda Branch, Hyderabad – 500 177, Andhra Pradesh, India · State Bank of India · Standard Chartered Grindlays Bank Limited, Hyderabad – 500 177, Andhra Pradesh , India · Citibank NA, Hyderabad-500 177, Andhra Pradesh , India · State Bank of Hyderabad, Gun Foundry, Hyderabad – 500 177, Andhra Pradesh, India · ING Vysya Bank · IndusInd Bank · Bank of Baroda ·
Union Bank of ·
Export-Import Bank of · State Bank of Travancore |
||||||||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||||||||
|
Facilities : |
Total Fund and Non Fund Based Facilities: Rs.4000.000 millions
Notes: LONG TERM
BORROWINGS All secured term
loans are secured by a pari-passu first charge on fixed assets and a pari-passu
second charge of the current assets of the Company. Of the Indian rupee loans
from banks and foreign currency loans from banks, loans of Rs. Nil as on
31-March-2013 (Rs.46.714 millions as on 31-March-2012) are further guaranteed
by the personal guarantee of the Managing Director. Of the foreign
currency loans from Financial Institutions on account of Rs.1097.400 millions
as on 31-March-2013 (Rs.517.975 millions as on 31-March-2012) is further
guaranteed by the personnel guarantee of the Managing Director. |
|
|
|
|
Banking
Relations : |
-- |
|
|
|
|
Financial Institution : |
International Finance Corporation |
|
|
|
|
Statutory Auditors : |
|
|
Name : |
Kumar and Giri Chartered Accountants |
|
Address : |
D.No.1-11-126/D, Opposite Aeroview Towers, Begumpet,
Hyderabad – 500 016, Andhra Pradesh, India |
|
|
|
|
Internal Auditors : |
|
|
Name : |
Dhanunjaya and Prabhakar Chartered Accountants |
|
Address : |
302, Wings, 8-3-960/6/2,
Srinagar Colony, Hyderabad – 500 073, Andhra Pradesh, India |
|
|
|
|
Wholly Owned Subsidiary Companies : |
·
Granules USA Inc. ·
GIL Life Sciences Private Limited ·
Granules Singapore Pte Limited |
|
|
|
|
Joint Venture: |
·
Granules-Biocause Pharmaceutical Co. Limited ·
Granules Omnichem Private Limited |
CAPITAL STRUCTURE
AS ON 19.08.2013
Authorised Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
30000000 |
Equity Shares |
Rs.10/- each |
Rs.300.000 millions |
|
|
|
|
|
Issued, Subscribed & Paid-up Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
20313154 |
Equity Shares |
Rs.10/- each
|
Rs.203.132
millions |
|
|
|
|
|
AS ON 31.03.2013
Authorised Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
30000000 |
Equity Shares |
Rs.10/- each |
Rs.300.000 millions |
|
|
|
|
|
Issued, Subscribed & Paid-up Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
20126154 |
Equity Shares |
Rs.10/- each
|
Rs.201.262
millions |
|
|
|
|
|
Reconciliation of
the number of shares outstanding and the amount of share capital as at March
31, 2013
|
Particulars |
As at 31st March, 2013 |
|
|
No. of Shares |
Amount (Rs. in
millions) |
|
|
Number of shares
at the beginning of the year |
20061654 |
200.617 |
|
Add: Shares
issued on exercise of employee stock options |
64500 |
0.645 |
|
Number of shares
at the end of the year |
20126154 |
201.262 |
Terms/Rights
attached to equity shares:
The Company has
only one class of equity shares having a par value of Rs.10/-. Each holder of
equity shares is entitled to one vote per share. The Company declares and pays
dividends in Indian rupees. The dividend proposed by the Board of Directors is
subject to the approval of the shareholders in the ensuing Annual General
Meeting.
During the year
end 31-March-2013, the amount of per share dividend recognized as distribution
to equity shareholders was Rs.2.00/-
In the event of
liquidation of the Company, the holders of equity shares will be entitled to
receive remaining assets of the Company after distribution of all preferential
amounts. The distribution will be in proportion to the number of equity shares
held by the shareholders.
Details of
shareholders holding more than 5% shares in the Company
|
Particulars |
As at 31st March, 2013 |
|
|
No. of Shares |
% |
|
|
C. Krishna Prasad |
6535663 |
32.47 |
|
Investco Management LLC |
2211200 |
10.99 |
|
Ridgeback Capital Asia Limited |
2072504 |
10.30 |
|
International Finance Corporation |
1715301 |
8.52 |
FINANCIAL DATA
[all figures are
in Rupees Millions]
ABRIDGED BALANCE
SHEET
|
SOURCES OF FUNDS |
31.03.2013 |
31.03.2012 |
31.03.2011 |
|
I.
EQUITY AND LIABILITIES |
|
|
|
|
(1) Shareholders' Funds |
|
|
|
|
(a) Share Capital |
201.262 |
200.617 |
200.572 |
|
(b) Reserves & Surplus |
2503.668 |
2246.752 |
2020.163 |
|
(c) Money received against share warrants |
0.000 |
0.000 |
0.000 |
|
|
|
|
|
|
(2) Share Application money pending allotment |
3.150 |
0.000 |
0.000 |
|
Total
Shareholders’ Funds (1) + (2) |
2708.080 |
2447.369 |
2220.735 |
|
|
|
|
|
|
(3) Non-Current
Liabilities |
|
|
|
|
(a) Long-term borrowings |
1407.237 |
880.534 |
378.327 |
|
(b) Deferred tax liabilities (Net) |
250.987 |
230.918 |
199.138 |
|
(c) Other long
term liabilities |
0.000 |
0.000 |
0.000 |
|
(d) Long-term
provisions |
21.238 |
15.303 |
11.898 |
|
Total Non-current
Liabilities (3) |
1679.462 |
1126.755 |
589.363 |
|
|
|
|
|
|
(4) Current Liabilities |
|
|
|
|
(a)
Short term borrowings |
767.148 |
614.127 |
477.963 |
|
(b)
Trade payables |
838.475 |
642.269 |
352.467 |
|
(c)
Other current liabilities |
130.257 |
193.628 |
185.805 |
|
(d) Short-term
provisions |
47.093 |
46.632 |
34.966 |
|
Total Current
Liabilities (4) |
1782.973 |
1496.656 |
1051.201 |
|
|
|
|
|
|
TOTAL |
6170.515 |
5070.780 |
3861.299 |
|
|
|
|
|
|
II.
ASSETS |
|
|
|
|
(1) Non-current assets |
|
|
|
|
(a) Fixed Assets |
|
|
|
|
(i)
Tangible assets |
2324.008 |
2215.306 |
2041.751 |
|
(ii)
Intangible Assets |
124.338 |
156.233 |
188.128 |
|
(iii)
Capital work-in-progress |
916.026 |
290.752 |
61.364 |
|
(iv)
Intangible assets under development |
0.000 |
0.000 |
0.000 |
|
(b) Non-current Investments |
412.960 |
327.960 |
226.025 |
|
(c) Deferred tax assets (net) |
0.000 |
0.000 |
0.000 |
|
(d) Long-term Loan and Advances |
125.276 |
203.488 |
106.759 |
|
(e) Other
Non-current assets |
36.754 |
4.024 |
1.180 |
|
Total Non-Current
Assets |
3939.362 |
3197.763 |
2625.207 |
|
|
|
|
|
|
(2) Current assets |
|
|
|
|
(a)
Current investments |
94.592 |
0.000 |
0.000 |
|
(b)
Inventories |
1084.907 |
885.736 |
601.337 |
|
(c)
Trade receivables |
680.966 |
530.280 |
375.451 |
|
(d) Cash
and cash equivalents |
61.597 |
236.520 |
68.235 |
|
(e) Short-term
loans and advances |
56.479 |
26.394 |
20.339 |
|
(f)
Other current assets |
252.612 |
194.087 |
170.730 |
|
Total
Current Assets |
2231.153 |
1873.017 |
1236.092 |
|
|
|
|
|
|
TOTAL |
6170.515 |
5070.780 |
3861.299 |
PROFIT & LOSS
ACCOUNT
|
|
PARTICULARS |
31.03.2013 |
31.03.2012 |
31.03.2011 |
|
|
|
SALES |
|
|
|
|
|
|
|
Revenue from operations |
6797.970 |
5626.777 |
4056.770 |
|
|
|
Other Income |
17.181 |
12.082 |
7.117 |
|
|
|
TOTAL (A) |
6815.151 |
5638.859 |
4063.887 |
|
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
|
|
Cost of
Materials consumed |
4252.584 |
3613.993 |
2527.002 |
|
|
|
Changes in
inventories of finished goods and work-in- progress |
(74.062) |
(59.224) |
(14.284) |
|
|
|
Employee
benefits expense |
504.205 |
379.572 |
283.442 |
|
|
|
Other expenses |
1324.668 |
966.445 |
719.552 |
|
|
|
TOTAL (B) |
6007.395 |
4900.786 |
3515.712 |
|
|
|
|
|
|
|
|
Less |
PROFIT
/ (LOSS) BEFORE INTEREST, TAX, DEPRECIATION AND AMORTISATION (A-B) (C) |
807.756 |
738.073 |
548.175 |
|
|
|
|
|
|
|
|
|
Less |
FINANCIAL
EXPENSES (D) |
164.949 |
154.941 |
109.810 |
|
|
|
|
|
|
|
|
|
|
PROFIT
/ (LOSS) BEFORE TAX, DEPRECIATION AND AMORTISATION (C-D) (E) |
642.807 |
583.132 |
438.365 |
|
|
|
|
|
|
|
|
|
Less/ Add |
DEPRECIATION/
AMORTISATION (F) |
202.089 |
179.760 |
164.382 |
|
|
|
|
|
|
|
|
|
|
PROFIT / (LOSS)
BEFORE TAX (E-F) (G) |
440.718 |
403.372 |
273.983 |
|
|
|
|
|
|
|
|
|
Less |
TAX (H) |
138.965 |
130.309 |
56.800 |
|
|
|
|
|
|
|
|
|
|
PROFIT / (LOSS)
AFTER TAX (G-H) (I) |
301.753 |
273.063 |
217.183 |
|
|
|
|
|
|
|
|
|
Add |
PREVIOUS YEARS’
BALANCE BROUGHT FORWARD |
993.796 |
787.845 |
616.489 |
|
|
|
|
|
|
|
|
|
Less |
APPROPRIATIONS |
|
|
|
|
|
|
|
Provision for Dividend |
40.252 |
40.123 |
30.086 |
|
|
|
Provision for Dividend tax |
6.841 |
6.509 |
4.881 |
|
|
|
Transfer to General Reserve |
22.631 |
20.480 |
10.860 |
|
|
|
|
69.724 |
67.112 |
45.827 |
|
|
BALANCE CARRIED
TO THE B/S |
1225.825 |
993.796 |
787.845 |
|
|
|
|
|
|
|
|
|
|
EARNINGS IN FOREIGN
CURRENCY |
|
|
|
|
|
|
|
Export of goods
calculated on F.O.B. basis |
5457.763 |
4821.839 |
3155.989 |
|
|
TOTAL EARNINGS |
5457.763 |
4821.839 |
3155.989 |
|
|
|
|
|
|
|
|
|
|
IMPORTS |
|
|
|
|
|
|
|
Raw Materials |
2759.834 |
2168.052 |
1749.380 |
|
|
|
Components and
Spare Parts |
12.277 |
9.495 |
5.070 |
|
|
|
Capital Goods |
150.829 |
92.709 |
120.565 |
|
|
TOTAL IMPORTS |
2922.940 |
2270.256 |
1875.015 |
|
|
|
|
|
|
|
|
|
|
Earnings /
(Loss) Per Share (Rs.) |
|
|
|
|
|
|
- Basic |
15.02 |
13.61 |
10.83 |
|
|
|
- Diluted |
14.62 |
13.56 |
10.78 |
|
QUARTERLY RESULTS
|
PARTICULARS |
|
|
30.06.2013 |
|
Type |
|
|
1st
Quarter |
|
Net Sales |
|
|
2087.100 |
|
Total Expenditure |
|
|
1798.500 |
|
PBIDT (Excl OI) |
|
|
288.600 |
|
Other Income |
|
|
17.000 |
|
Operating Profit |
|
|
305.700 |
|
Interest |
|
|
34.100 |
|
Exceptional Items |
|
|
0.000 |
|
PBDT |
|
|
271.600 |
|
Depreciation |
|
|
50.100 |
|
Profit Before Tax |
|
|
221.400 |
|
Tax |
|
|
74.100 |
|
Provisions and contingencies |
|
|
0.000 |
|
Profit After Tax |
|
|
147.300 |
|
Extraordinary Items |
|
|
0.000 |
|
Prior Period Expenses |
|
|
0.000 |
|
Other Adjustments |
|
|
0.000 |
|
Net Profit |
|
|
147.300 |
KEY RATIOS
|
PARTICULARS |
|
31.03.2013 |
31.03.2012 |
31.03.2011 |
|
PAT / Total Income |
(%) |
4.43
|
4.84 |
5.34
|
|
|
|
|
|
|
|
Net Profit Margin (PBT/Sales) |
(%) |
6.48
|
7.17 |
6.75
|
|
|
|
|
|
|
|
Return on Total Assets (PBT/Total Assets} |
(%) |
9.10
|
9.06 |
7.67
|
|
|
|
|
|
|
|
Return on Investment (ROI) (PBT/Networth) |
|
0.16
|
0.16 |
0.12
|
|
|
|
|
|
|
|
Debt Equity Ratio (Total Debt/Networth) |
|
0.80
|
0.61 |
0.39
|
|
|
|
|
|
|
|
Current Ratio (Current Asset/Current Liability) |
|
1.25
|
1.25 |
1.18
|
FINANCIAL ANALYSIS
[all figures are
in Rupees Millions]
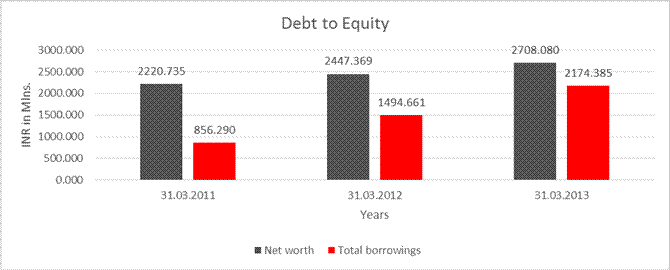
DEBT EQUITY RATIO
|
Particular |
31.03.2011 |
31.03.2012 |
31.03.2013 |
|
|
(INR in Mlns.) |
(INR in Mlns.) |
(INR in Mlns.) |
|
Share Capital |
200.572 |
200.617 |
201.262 |
|
Reserves & Surplus |
2020.163 |
2246.752 |
2503.668 |
|
Share Application money pending allotment |
0.000 |
0.000 |
3.150 |
|
Net worth |
2220.735 |
2447.369 |
2708.080 |
|
|
|
|
|
|
Long-term borrowings |
378.327 |
880.534 |
1407.237 |
|
Short term borrowings |
477.963 |
614.127 |
767.148 |
|
Total borrowings |
856.290 |
1494.661 |
2174.385 |
|
Debt/Equity ratio |
0.386 |
0.611 |
0.803 |
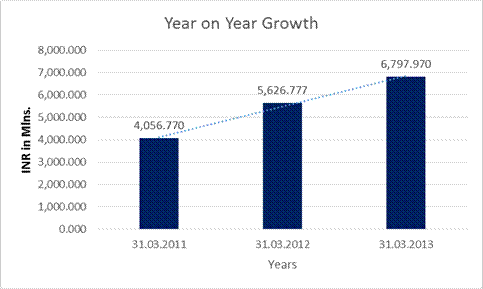
YEAR-ON-YEAR GROWTH
|
Year on Year Growth |
31.03.2011 |
31.03.2012 |
31.03.2013 |
|
|
(INR in Mlns) |
(INR in Mlns) |
(INR in Mlns) |
|
Revenue
from operations |
4,056.770 |
5,626.777 |
6,797.970 |
|
|
|
38.701 |
20.815 |
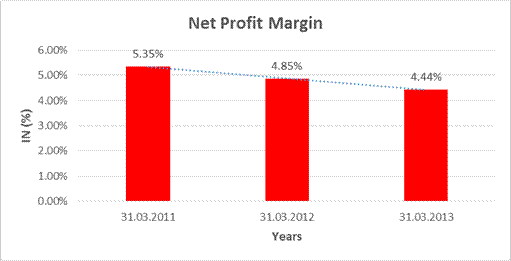
NET PROFIT MARGIN
|
Net Profit Margin |
31.03.2011 |
31.03.2012 |
31.03.2013 |
|
|
(INR in Mlns) |
(INR in Mlns) |
(INR in Mlns) |
|
Revenue
from operations |
4,056.770 |
5,626.777 |
6,797.970 |
|
Profit |
217.183 |
273.063 |
301.753 |
|
|
5.35% |
4.85% |
4.44% |
LOCAL AGENCY FURTHER INFORMATION
|
Check
List by Info Agents |
Available
in Report (Yes / No) |
|
1) Year of Establishment |
Yes |
|
2) Locality of the firm |
Yes |
|
3) Constitutions of the firm |
Yes |
|
4) Premises details |
Yes |
|
5) Type of Business |
Yes |
|
6) Line of Business |
Yes |
|
7) Promoter’s background |
Yes |
|
8) No. of employees |
Yes |
|
9) Name of person contacted |
Yes |
|
10) Designation of contact person |
Yes |
|
11) Turnover of firm for last three years |
Yes |
|
12) Profitability for last three years |
Yes |
|
13) Reasons for variation <> 20% |
-- |
|
14) Estimation for coming financial year |
No |
|
15) Capital in the business |
Yes |
|
16) Details of sister concerns |
Yes |
|
17) Major suppliers |
No |
|
18) Major customers |
No |
|
19) Payments terms |
Yes |
|
20) Export / Import details (if
applicable) |
Yes |
|
21) Market information |
-- |
|
22) Litigations that the firm / promoter involved
in |
Yes |
|
23) Banking Details |
Yes |
|
24) Banking facility details |
Yes |
|
25) Conduct of the banking account |
-- |
|
26) Buyer visit details |
-- |
|
27) Financials, if provided |
Yes |
|
28) Incorporation details, if applicable |
Yes |
|
29) Last accounts filed at ROC |
Yes |
|
30) Major Shareholders, if available |
Yes |
|
31)
Date of Birth of Proprietor/Partner/Director, if available |
Yes |
|
32)
PAN of Proprietor/Partner/Director, if available |
No |
|
33)
Voter ID No of Proprietor/Partner/Director, if available |
No |
|
34)
External Agency Rating, if available |
Yes |
LITIGATION
DETAILS:
|
ITTA 34 / 2010 |
ITTASR 92 / 2008 |
CASE IS:PENDING |
|
||||||||||||
|
||||||||||||
|
INDEX OF CHARGES:
|
S. No. |
Charge ID |
Date of Charge Creation/Modification |
Charge amount secured |
Charge Holder |
Address |
Service Request Number
(SRN) |
|
1 |
10476418 |
08/02/2014 |
1,645,000,000.00 |
ANDHRA BANK |
SOMAJIGUDA BRANCH, 6-3-352/2,
ASTRAL HEIGHTS, ROAD NO. 1, BANJARA HILLS, HYDERABAD, ANDHRA PRADESH |
B95849865 |
|
2 |
10389714 |
30/11/2012 |
527,500,000.00 |
DEG - DEUTSCHE
INVESTITIONS- UND ENTWICKLUNGSGESEL |
KAMMERGASSE 22, COLOGNE, -
50676, GERMANY |
B63236012 |
|
3 |
10389717 |
30/11/2012 |
527,500,000.00 |
INTERNATIONAL FINANCE
CORPORATION |
2121 PENNSYLVANIA AVENUE,
N.W., WASHINGTON DC, WASHINGTON DC, - 20433, UNITED STATES OF AMERICA |
B63236624 |
|
4 |
10319592 |
22/11/2011 |
49,150,000.00 |
INTERNATIONAL FINANCE
CORPORATION |
2121 PENNSYLVANIA AVENUE,
N.W., WASHINGTON DC, 20 |
B26012047 |
|
5 |
10224217 |
15/04/2010 |
87,500,000.00 |
BANK OF BARODA |
CORPORATE FINANCIAL SERVICES
HYDERABAD BRANCH, 3-6-262/6, TIRUMALA ESTATES, HIMAYAT NAGAR, HYDERABA |
A85176519 |
|
6 |
10082831 |
22/12/2007 |
360,000,000.00 |
INTERNATIONAL FINANCE
CORPORATION |
IFC'S SOUTH ASIA DEPARTMENT,
GATE NO.3, NITI MAG, 50-M, SHANTHIPATH, CHANAKYAPURI,, NEW DELHI, DELHI -
110021, INDIA |
A30260624 |
|
7 |
90139379 |
15/05/2013 * |
4,674,300,000.00 |
ANDHRA BANK |
SOMAJIGUDA BRANCH, 6-3-352/2,
ASTRAL HEIGHTS, ROAD NO.1. BANJARA HILLS, HYDERABAD, ANDHRA PRADESH -
500034, INDIA |
B76574565 |
* Date of charge modification
|
Unsecured Loan |
31.03.2013 (Rs.
in Millions) |
31.03.2012 (Rs.
in Millions) |
|
LONG TERM
BORROWINGS |
|
|
|
Deferred sales tax loan (Deferred sales
tax loan is interest free and payable in 14 yearly installments commencing
from June 2013 onwards.) |
6.473 |
7.524 |
|
Total
|
6.473 |
7.524 |
CORPORATE INFORMATION:
The Company is a
public domiciled in India and incorporated under the Companies Act, 1956. Its
shares are listed on two Stock Exchanges in India. The Company is engaged in
the manufacturing and selling of Active Pharmaceutical Ingredients (APIs),
Pharmaceutical Formulation Intermediates (PFIs) and Finished Dosages (FDs). The
Company caters to both domestic and international markets.
BOARD OF DIRECTORS
Dr. C. Nageswara
Rao – Chairman
Dr. C. Nageswara
Rao is one of the promoters of
Granules India Limited. Dr.
Nageswara Rao has an M.S. in Surgery and Urology and was an active surgeon in
Guntur for five decades. Dr. Nageswara Rao was the Chairman of Andhra Pradesh
Medical Council and a member of the All India Medical Council, a Syndicate
Member of Nagarjuna University and a Director of Hindustan Antibiotic Limited.
Mr. C. Krishna
Prasad – Managing Director
Mr. Prasad is the
Founder of Granules and has more than three decades of experience in the
pharmaceutical industry. In 1984, he set up a paracetamol manufacturing
facility, which has become one of the world’s reputed manufacturers of
paracetamol in the regulated markets. Mr. Prasad pioneered and popularized the
concept of Pharmaceutical Formulations Intermediates (PFIs) as a cost-efficient
product for global formulations manufacturers.
Mr. L.S. Sarma - Director
Mr. L.S.Sarma, is
a retired bank executive. Mr. Sarma was a General Manager at the Industrial
Development Bank of India (IDBI), as well as the Director of ECGC and Dena
Bank. He worked for International Trade Centre, Geneva, ITC (UNCTAD/GATT) as an
Export Credit Consultant. He is on the Board of several companies including
Hexaware Technologies Limited.
Mr. A.P. Kurian - Director
Mr. Kurian served
as the Chairman of the Association of Mutual Funds in India. Mr. Kurian has a
rich career in the financial services area spread over four decades. During
1975-1993, Mr. Kurian was with Unit Trust of India and held several positions
including Director-Investments, Director- Planning and Development and as an Executive
Trustee. Since 1998, he has been the Executive Chairman of Association of
Mutual Funds in India. He is on the Board of National Stock Exchange, Executive
Committee of NSDL and several other committees associated with mutual funds and
capital market.
Mr. C.
Parthasarathy - Director
Mr. C.
Parthasarathy is one of the founders of Karvy. As the Chairman of Karvy, he has
been responsible for building Karvy as one of India’s truly integrated
financial services organisations. He oversees the group’s operations and is
responsible for the vision, business direction and technology value addition to
the overall business. Karvy employees over 10,500 personnel and has a network
encompassing 583 offices in 391 cities/towns spread across the country,
providing a complete range of services. He is the fellow member of the
Institute of Chartered Accountants of India and the Institute of Company
Secretaries of India.
Dr. Krishna Murthy
Ella - Director
Dr. Krishna Murthy
Ella founded and established Bharat Biotech International Limited in 1996 along
with his wife Ms. Suchitra Ella. The Company today, is on the forefront of
Indian Biotechnology engaged in R&D, manufacturing and marketing of
vaccines and bio-therapeutics. Dr. Ella was awarded his doctorate from the University
of Wisconsin- Madison in Molecular Biology. He received the National Research
Service Award from the National Institute of Health, Bethesda, Maryland and
became a part of the Research faculty at the Medical University of South
Carolina at Charleston.
Mr. Arun Rao
Akinepally - Director
Mr. Akinepally
Arun Rao is the Executive Director of Akin Laboratories Private Limited, a
formulation manufacturing Company. Mr. Arun Rao is on the Board of ESPI
Industries and Chemicals Private Limited, a leading manufacturer of antacids in
India. Mr. Arun Rao is a member of the Central Executive Council of the Indian
Pharmaceutical Association. He was the Vice President and currently, a member
of the Executive Committee of the Indian Pharmaceutical Association (Andhra
Pradesh Branch). He is also a member of Executive Committee of Organisation of
Pharmaceutical Manufacturers, Hyderabad.
Mr. Harsha
Chigurupati - Executive Director
Mr. Chigurupati
has been with Granules since 2005 and served as CMO from 2006-2010. As CMO, Mr.
Chigurupati was instrumental in commercialising the Company’s Finished Dosage
Division and also changed the Company’s focus to marquee customers in the
regulated markets. As the Executive Director, Mr. Chigurupati is responsible
for the standalone operations of Granules India including the P&L. Mr.
Chigurupati has a Bachelor of Science in Business Management from Boston
University.
Mrs. Uma
Chigurupati – Executive Director
Mrs. Chigurupati
is Director of KRSMA Estates Private Limited, one of India’s premier boutique
wineries. Under her tenure, she has established a vineyard in Karnataka and has
been overseeing the ongoing operations at the site. In addition, Mrs.
Chigurupati is the Chairman of the Hyderabad 10K Foundation, which promotes
health awareness campaigns in Andhra Pradesh through multiple initiatives
including hosting several races in Hyderabad including the Hyderabad Heritage
Marathon. Mrs. Chigurupati has a post-graduate degree in soil microbiology from
Nagarjuna University.
Mr. K.B. Sankara
Rao – Additional Director
Mr. K. B. Sankar
Rao is post graduate from Andhra University and has rich experience of about 33
years in various domains. Mr. K. B. Sankar Rao was associated with various
reputed organisation like Warner Hindustan, Cipla Limited and Dr. Reddy’s
Laboratories Limited. He has varied experience in the field of production,
quality, formulations, R&D, supply chain, development and launch of API and
finished dosages for global markets and business strategy. Mr. Kolli is also
Managing Director of Raje Retail Private Limited, a pharmacy retail chain under
the brand name- “My Health Pharmacy” in Hyderabad.
REVIEW OF
OPERATIONS
During the year,
there were many challenges in the macro-environment including a weak global
economy. In spite of adverse conditions, the Company posted good results. The
net sales of the Company in FY13 stood at Rs.6797.970 millions compared to the
net sales of Rs.5626.777 millions in FY12, registering a growth of 20.8% in the
current financial year. The EBITDA stood at Rs.807.756 millions in FY13
compared to Rs.738.072 millions in FY12, registering a growth of 9.4%. The
profit after tax for FY13 stood at Rs.301.753 millions compared to Rs.273.063
millions in FY12, registering a growth of 10.5% in the current financial year.
The Company continued to strengthen its position in the market and aims to
increase productivity gains, volume growth and profit margins.
EXPANSIONS
During FY12, the
Company commenced work on an expansion at its Gagillapur facility. The
expansion involved a capacity expansion in the PFI and Finished Dosage
facilities. The expansion mainly focused on efficient design and output in
terms of material transfer and automation.
Upon completion of
the PFI module, the Company faced initial scale up issues. The problems were
addressed by the technical team along with outside consultants. The Finished
Dosage (FD) facility was expanded from an existing capacity of 6 billion doses
to 18 billion doses. Both expansions were done in an existing facility and did
not require further regulatory approvals and will be used for manufacturing
products for the regulated markets after completion of trials and necessary
approvals from relevant customers.
In addition,
during FY13, the Company completed an upgrade of its existing warehouse and
also commenced construction of a new warehouse at its Gagillapur facility. The
existing Finished Goods and Raw Material Warehouses were modified with a mobile
racking system which has increased capacity. The construction of a new
warehouse commenced in FY13 at Gagillapur; construction is expected to be
completed in FY14.
SUBSIDIARY
COMPANIES
Granules USA Inc
Granules USA Inc,
a wholly-owned subsidiary Company, operates for the marketing requirements of
the Company in the U.S market. The Share Capital of the Company as on March 31,
2013 is Rs.11.631 millions. During FY13, the Company achieved a turnover of
Rs.751.000 millions and the profit after tax is Rs.27.096 millions.
GIL Lifesciences Private Limited
The Company is yet
to commence its operations. As on March 31, 2013 the Authorised Share Capital
of the Company is Rs.35.000 millions divided into 3500000 equity shares of
Rs.10/- each and the Paid Up Share Capital of the Company is Rs.29.462 millions
divided into 2946176 equity shares of Rs.10 each.
Granules Singapore Pte Limited
The Company has
not commenced any activity so far. The Share Capital of the Company as on March
31, 2013 is Rs.0.500 million.
JOINT VENTURE
COMPANIES
Granules-Biocause Pharmaceutical Co. Limited
The Share Capital
of the Company as on March 31, 2013 is Rs.181.903 millions. During the FY13,
the Company achieved a turnover of Rs.1024.463 millions.
Granules OmniChem Private Limited
The Share Capital
of the Company as on March 31, 2013 is Rs.187.550 millions. The Company has not
yet commenced its commercial activity during the period hence there was no
income during FY13. However the Company incurred a loss of Rs.3.694 millions.
Granules OmniChem Private Limited is a 50:50 joint venture that will
manufacture pharmaceutical intermediates and APIs in a greenfield facility in
Visakhapatnam (AP) and mainly focus on high-value, low-volume APIs for the
regulated markets. The Company will initially cater to Ajinomoto OmniChem’s
(one of the joint venture partners and shareholders) existing customers and
will focus on oncology, cardiovascular and central nervous system (CNS)
products. Granules India Limited will also purchase APIs from the joint venture
Company and will offer finished dosages.
The joint venture
Company is setting up 100% export oriented unit at Jawaharlal Nehru Pharma City
(JNPC), Parwada Mandal, Visakhapatnam (AP) under the APIC special economic zone
to manufacture active pharmaceutical ingredients. The construction of the unit
(including manufacturing block, administration block, warehouse and utility
electric substation block) and various installations are ongoing and expected
to finish construction in mid- 2013. The trial and commercial production is
expected to commence by December 2013. The Company plans to obtain all
necessary regulatory approvals from the U.S. Food and Drug Administration
(“FDA”) or relevant European regulatory authorities by March 31, 2016 and all
Good Manufacturing Practice (“GMP”) standards in relation to the unit by March
31, 2015.
MANAGEMENT DISCUSSION AND ANALYSIS
The Pharmaceutical
Market
Global: Global medical spending is estimated to be
around $1,200 billion in 2016, a growth rate of 3%-6% over the next few years.
Developed markets are expected to experience their lowest annual growth this
year, at less than 1% or $3 billion, and then rebound to $18-20 billion in
annual growth in the 2014-16 period.
The market for
branded medicines is likely to experience 0%-3% annual growth through 2016 to
$615-$645 billion, up from $596 billion in 2011. In the major developed
markets, branded medicine growth could be severely constrained at only $10
billion over the five-year period due to patent expiries, increased
costcontainment actions by payers and modest spending on newly launched
products. Global generic spending is expected to increase from $242 billion in
2011 to $400-430 billion by 2016, fueled by volume growth in pharmerging
markets and the ongoing transition to generics in developed nations. The impact
of patent expiries will be felt largely in the US whereas in Europe, limited
savings from expiring patents are prompting policy shifts to encourage greater
use of generics and lower reimbursement for these products.
The U.S. share of
global spending is expected to decline from 41% in 2006 to 31% in 2016, while
the share of spending from the top five European countries is expected to
decline from 19% to 13%. Meanwhile, 17 high growth emerging markets are likely
to contribute 30% of the total spending by 2016 as compared to 14% in 2006.
Granules’ Product
Overview
Granules focuses
on pharmaceutical products with high API and/or finished dosage volume
requirements. In many high-volume pharmaceutical products, there are dozens of
suppliers leading to oversupply. While there are large surpluses, high quality
material for the regulated markets is in short supply and can only be serviced
by a handful of suppliers.
Customers in the
regulated markets and an increasingly growing number of customers in the
semi-regulated markets require high-quality supplies. These customers value
supply-based security and quality over pricing. Customers generally work
closely with their suppliers’ regulatory and quality control departments; once they
select a supplier, the customer usually stays with the supplier for years and
only periodically reviews alternatives.
Due to increased
accountability and consumer pressure, countries in the semi-regulated markets
are implementing tighter controls and demanding increased stringent quality
parameters.
Analgesics market
The analgesics
market, which focuses on pain relief, is one of the largest segments of the
healthcare industry with sales worth approximately $31 billion. The market is
growing at a 2.7% CAG R. Going ahead, growth will be driven by an ageing
population suffering chronic ailments and lifestyle diseases arising out of
sedentary jobs.
Granules is among
the leading producers of Paracetamol and Ibuprofen; the Company also competes
in the Naproxen market. Paracetamol accounts for 58% of the analgesic market by
volume and is widely used all over the world. Ibuprofen is the fastest growing
product in the sector and is expected to surpass Aspirin, which is not growing
as fast as it used to be. There are no analgesic products expected to replace
the current leaders.
Paracetamol
Paracetamol, also
known as acetaminophen, is used to reduce body pains, headaches and lower
fevers. The Paracetamol market continues to grow but there are signs of consolidation
within the industry and increasing challenges for Chinese Paracetamol
manufacturers. While many of these manufacturers targeted the emerging markets,
which offer lower margins, manufacturers were able to gain market share and
grow profits. However, many of the advantages that Chinese manufacturers had
including an undervalued Chinese currency, low employment costs and favourable
interest rates are diminishing which has added pressures. In order to combat
inflation, the Chinese government has let the RMB appreciate. During FY13, the
RMB appreciated 1.5% which is slightly lower than the 3.0% appreciation in
FY12. Due to the appreciation, the cost advantage from Chinese manufacturers
has been greatly diminished which has added pressure for many manufacturers. In
addition, labour and energy costs in China have increased by double-digits,
which are adding further pressure for companies. Also, there is an increasing
focus on quality from companies in the emerging markets which is resulting in
extra costs for many Chinese manufacturers since they must adjust their
processes to adhere to more stringent standards. Due to these pressures,
several Chinese manufacturers have shut down while others have sold their
facilities to larger players. Due to the diminishing value proposition from
Chinese manufacturers, many customers in the emerging markets are qualifying
Indian manufacturers which has resulted in higher capacity utilization in FY13.
Ibuprofen
Ibuprofen is
primarily used for arthritis relief and fever reduction. The drug is popular in
North America and Western Europe, which account for nearly 60% of global sales.
Ibuprofen is a more complex analgesic to manufacture compared to Paracetamol
which is why there are not as many suppliers. There are six primary
manufacturers in the Ibuprofen market, which is growing in the mid-to-high
single digits. The drug is becoming more popular due to an ageing population
that wants to maintain their lifestyle. The Ibuprofen market, which faced
pricing pressures in FY11 due to new capacity, has rebounded. Due to strong
demand, pricing has increased in FY13 and appears to have stabilised.
Anti-Diabetic
Market
The anti-diabetic
market is extremely lucrative due to the growing number of people
with diabetes. The number of people with diabetes is expected to
grow from 246 million in 2008 to 380 million by 2025. The emerging markets
are expected to be a major source of new diabetes cases as they adapt
Western lifestyles. There are multiple classes of drugs to treat diabetes
which range from cheap, first-line therapy to expensive, advanced
therapies
Ø Biguanides: The most popular drug in this category is
Metformin, which lowers glucose levels. This is often used as the first
response for Type II diabetes
Ø DPP-4 Inhibitors: This is the latest generation of diabetes
drugs and over the next decade, several products will be released
Diabetic cases are
spread evenly throughout the world and there is a large opportunity for cost
effective medication
Metformin
Metformin, a
prescription drug, is the first biguanide oral anti-diabetic agent to be
approved by the U.S. FDA after phenformin (phenethylbiguanide) was banned in
the U.S. in 1977. Due to its relatively low cost and higheffectiveness,
metformin is often used as a first-line therapy for patients with type-II
diabetes
Annual production
capacity for metformin is approximately 65,000 tonnes. Due the rising number of
diabetic patients in the world, demand for Metformin is increasing and multiple
suppliers are increasing capacity in order to meet global demand. The market is
growing in the mid-teens and is expected to maintain the growth rate for the
foreseeable future.
BUSINESS OPERATIONS
Granules has a
presence across the pharmaceutical manufacturing value chain. The Company’s operations
are spread across four facilities – three in India and one in China.
ACTIVE
PHARMACEUTICAL INGREDIENTS (API)
The Company’s API
vertical continued to report double-digit growth despite the markets for most of
these products reporting single-digit growth. Granules entered this segment in
1984 through the manufacture of Paracetamol APIs and expanded into other
products like Ibuprofen and Metformin. Over the decades, the Company emerged
among premier global API manufacturers due to a combination of scale, quality,
compliance and reliability.
The Company has
three API facilities; two located in India and a third in China. Due to a delay
in the Gagillapur expansion, the Company marketed a larger quantity of APIs in
FY13 but anticipates that its APIs will be progressively consumed largely for
captive consumption in the future. The API vertical will continue to be
critical to the Company’s success since it will continue to represent the
starting point of the Company’s PFI and FD verticals.
FY13 Highlights,
Ø API revenues
increased 40% from Rs.1810.000 millions in FY12 to Rs.2520.000 millions.
Ø Several
initiatives to increase API capacity were implemented, a number of these
finishing in late Q4FY12. The additional capacity was utilised in FY13.
Ø The delay in the
Gagillapur Formulation expansion resulted in an increase API sales; APIs will
be used more for captive consumption in FY14.
PHARMACEUTICAL
FORMULATION INTERMEDIATES (PFI)
Granules pioneered
the concept of commercialising PFIs, saving customers the need to manufacture
their own PFIs and leaving them free to focus on finished dosage manufacture
and marketing. Granules entered this business segment in the early-90s
following an insight that most APIs were not suitable for compression in their
crystalline or amorphous forms because they lacked the necessary binding,
lubricating and disintegrating properties. This made it necessary for APIs to
be granulated first to enhance functionality. Manufacturers would be able to
granulate but not able to derive operational efficiencies if they focused on
dozens of products or if they only produced the product for only a few days at
a time. This pulled down the overall return from their investments especially
because PFI manufacture accounts for 80% of the total cost of a finished
dosage.
The Company,
through its PFI facilities at Gagillapur and Jeedimetla, is a leader in
manufacturing PFIs and has the world’s largest capacity. The manufacturing
facility uses high-shear and fluid-bed granulation processes with a 6 ton batch
size, the largest in the industry.
FY13 Highlights
Ø Revenue increased
10% from Rs.1970.000 millions in FY12 to Rs.2160.000 millions.
Ø The Company
commercialized its Gagillapur facility expansion in March 2013; customer
validations commenced in March 2013 and are expected to be completed in FY14.
FINISHED DOSAGES
(FD)
At Granules, the
manufacture of finished dosages represents the apex of the value chain. The
Company entered this business in FY09 with an installed capacity of six billion
tablets. The business accounts for 31% of the Company’s revenues.
Granules’ finished
dosage facility in Gagillapur comprises automated processes, robust
infrastructure and superior quality systems that efficiently produce finished
dosages. Granules offers multiple finished dosage forms comprising tablets,
caplets and press fit capsules in bulk, blister packs and bottles. The Company
is the only Indian pharmaceutical player to manufacture press-fit (rapid
release tablets) dosages and among the few in India to manufacture bi-layered
tablets.
FY13 Highlights
Ø Revenue increased
14% from Rs.1850.000 millions in FY12 to Rs.2130.000 millions.
Ø The Company
commercialised the expansion of its Gagillapur facility, which substantially
increased capacity from March 2013 onwards. Customer validations started in
March 2013 and are expected to finish in FY14.
CONTINGENT
LIABILITIES:
|
Particulars |
31.03.2013 (Rs. in Millions) |
31.03.2012 (Rs. in Millions) |
|
a) Claims against the company not acknowledgment as debt |
|
|
|
Income Tax |
123.038 |
167.021 |
|
Excise |
20.555 |
20.555 |
|
Service Tax |
8.937 |
8.937 |
|
Customs |
4.347 |
4.347 |
|
b) Bank Guarantee and LC |
201.483 |
386.736 |
|
d) Bills discounted with banks |
1271.087 |
1146.961 |
|
Total |
1629.447 |
1734.557 |
STATEMENT
OF STANDALONE AUDITED RESULTS FOR THE 3RD QUARTER AND YEAR ENDED 31ST
MARCH, 2014
(Rs.
in millions)
|
Sr. No. |
Particulars |
3 Months ended 31.03.2014 (Audited) |
Preceding 3 Months Ended
31.12.2013 (Unaudited) |
Current year ended 31.03.2014 (Audited) |
|
|
|
|
||
|
1 |
Income from
Operations |
|
|
|
|
|
(a) Net sales / Income from Operation (Net of
Excise duty) |
2918.444 |
2621.245 |
10016.799 |
|
|
(b) Other operating income |
-- |
-- |
-- |
|
|
Total Income from
Operations |
2918.444 |
2621.245 |
10016.799 |
|
2 |
Expenses |
|
|
|
|
a |
Cost of material Consumed |
1534.827 |
1479.209 |
5873.564 |
|
b |
Changes in Inventories of Finished goods, Work in progress and Stock in
trade |
182.633 |
(7.955) |
(23.762) |
|
c |
Employee benefits Expense |
183.390 |
172.404 |
692.738 |
|
d |
Depreciation |
93.212 |
66.623 |
263.979 |
|
e |
Manufacturing Expenses |
137.015 |
161.847 |
551.825 |
|
f |
Freight outward & clearing charges |
160.848 |
122.589 |
518.414 |
|
g |
R & D Expenses |
27.464 |
8.738 |
56.764 |
|
h |
Other expenditure |
152.886 |
234.371 |
717.174 |
|
|
Total |
2472.276 |
2237.825 |
8650.696 |
|
3 |
Profit from
Operations before Other Income, Interest & Exceptional Items (1- 2) |
446.168 |
383.420 |
1366.103 |
|
4 |
Other Income |
12.198 |
3.125 |
36.475 |
|
5 |
Profit from
Ordinary activities before Finance Cost and exceptional items (3 - 4) |
458.366 |
386.545 |
1402.578 |
|
6 |
Finance Costs |
70.691 |
43.039 |
189.270 |
|
7 |
Profit from
Ordinary activities after Finance Cost but before exceptional items (5 - 6) |
387.675 |
343.506 |
1213.308 |
|
8 |
Exceptional Items |
-- |
-- |
-- |
|
9 |
Profit from
Ordinary activities before Tax (7 - 8) |
387.675 |
343.506 |
1213.308 |
|
10 |
Tax expense |
127.250 |
113.573 |
403.194 |
|
11 |
Profit
from Ordinary activities after Tax (9 - 10) |
260.426 |
229.933 |
810.114 |
|
12 |
Extraordinary Items (net of tax
expense) |
-- |
-- |
-- |
|
13
|
Net
Profit for the period (11 - 12) |
260.426 |
229.933 |
810.114 |
|
14 |
Paid-up share
capital (Face Value of
Rs.10/- each) |
202.812 |
202.812 |
202.812 |
|
15 |
Reserves
excluding Revaluation Reserve |
3240.315 |
3062.937 |
3240.315 |
|
16 |
Earnings per
Share |
|
|
|
|
|
(a) Basic Earning per share (Rs.)* |
12.88 |
11.39 |
40.06 |
|
|
(b) Diluted Earning per share (Rs)* |
12.63 |
11.17 |
39.30 |
|
|
|
|
|
|
|
|
Part II A. Particulars of
Shareholding |
|
|
|
|
1 |
Public
Shareholding |
|
|
|
|
|
- No. of
Shares |
10370988 |
10371180 |
10370988 |
|
|
-% of
Shareholding |
51.14% |
51.14% |
51.14% |
|
2 |
Promoters and
Promoter group Shareholding |
|
|
|
|
a |
Pledged /
Encumbered |
|
|
|
|
|
-Number
of Shares |
2445830 |
2445830 |
2445830 |
|
|
- Percentage of Shares (as a % of the
total shareholding of promoter and promoter group) |
24.68% |
24.68% |
24.68% |
|
|
- Percentage of Shares (as a % of the
total Share capital of the Company) |
12.06% |
12.06% |
12.06% |
|
b |
Non-encumbered |
|
|
|
|
|
- Number of Shares |
7464336 |
7464144 |
7464336 |
|
|
- Percentage of Shares (as a % of the
total shareholding of promoter and promoter group) |
75.32% |
75.32% |
75.32% |
|
|
- Percentage of Shares (as a % of the
total Share capital of the Company) |
36.80% |
36.80% |
36.80% |
|
B |
INVESTOR COMPLAINTS |
3 Months ended 31.03.2014 |
|
|
Pending
at the beginning of the quarter |
Nil |
|
|
Received
during the quarter |
20 |
|
|
Disposed
of during the quarter |
20 |
|
|
Remaining
unresolved at the end of the quarter |
Nil |
STANDALONE STATEMENT OF ASSETS AND LIABILITIES
(Rs. in millions)
|
Particulars |
As at current
year end 31.03.2014 |
|
I.
EQUITY AND LIABILITIES |
|
|
(1) Shareholders' Funds |
|
|
(a) Share Capital |
202.812 |
|
(b) Reserves & Surplus |
3240.315 |
|
Sub-total
- Shareholders' Funds |
3443.127 |
|
|
|
|
(2) Share Application money pending allotment |
2.548 |
|
|
|
|
(3) Non-Current
Liabilities |
|
|
(a) Long-term borrowings |
2220.918 |
|
(b) Deferred tax liabilities (Net) |
358.081 |
|
(c) Long-term
provisions |
36.990 |
|
Sub-total -
Non-current Liabilities |
2615.989 |
|
|
|
|
(4) Current Liabilities |
|
|
(a)
Short term borrowings |
774.862 |
|
(b)
Trade payables |
1262.480 |
|
(c)
Other current liabilities |
376.049 |
|
(d) Short-term
provisions |
83.048 |
|
Sub-total - Current Liabilities |
2496.439 |
|
|
|
|
TOTAL - EQUITY AND LIABILITIES |
8558.103 |
|
|
|
|
II.
ASSETS |
|
|
(1) Non-current assets |
|
|
(a) Fixed Assets |
|
|
(i)
Tangible assets |
3264.286 |
|
(ii)
Intangible Assets |
92.443 |
|
(iii)
Capital work-in-progress |
391.421 |
|
|
3748.150 |
|
(b) Non-current Investments |
1454.461 |
|
(c) Long-term Loan and Advances |
31.466 |
|
(d)
Other Non-current assets |
25.558 |
|
Sub-total - Non-Current Assets |
5259.634 |
|
|
|
|
(2) Current assets |
|
|
(a)
Current investments |
0.000 |
|
(b)
Inventories |
1202.873 |
|
(c)
Trade receivables |
1218.197 |
|
(d) Cash
and cash equivalents |
219.258 |
|
(e)
Short-term loans and advances |
31.434 |
|
(f)
Other current assets |
626.706 |
|
Sub-total - Current Assets |
3298.468 |
|
|
|
|
TOTAL - ASSETS |
8558.103 |
FIXED ASSETS:
Tangible Assets
·
Land
·
Buildings
·
Plant and Machinery
·
Computers
·
Office Equipment
·
R&D Equipment
·
Furniture and Fixtures
·
Vehicles
Intangible Assets
·
Technical Knowhow
WEBSITE DETAILS:
NEWS/ PRESS RELEASES:
GRANULES
INDIA TO CLOSE ITS SINGAPORE SUBSIDIARY
October
30, 2013,
Granules India to close its Singapore subsidiary
Granules India Limited has informed that the Board of Directors of the Company
at its meeting held on October 30, 2013, has decided to close M/s Granules
Singapore PTE LIMITED, wholly owned subsidiary of the Company, located at
Singapore.
GRANULES INDIA - GRANULES INDIA ANNOUNCES ACQUISITION OF AUCTUS PHARMA AND OPENING OF A
NEW R&D FACILITY
5th November, 2013
Granules India Limited has informed BSE regarding a Press
Release dated November 04, 2013 titled "Granules India announces
acquisition of Auctus Pharma and opening of a new R&D Facility".
Granules India announced the signing of definite agreement for the acquisition
of Auctus Pharma. The acquisition process is expected to be completed in the
next three to six months.
GRANULES INDIA’S FY14
SALES INCREASE 43% TO RS.10960.000 MILLIONS; NET PROFIT JUMPS 131% TO
RS.750.000 MILLIONS
Hyderabad, April 24, 2014: Granules India Limited, a fast growing pharmaceutical manufacturing company, announced financial results for its fiscal year ended March 31, 2014. Granules consolidated net sales increased 43% to Rs.10960.000 millions while consolidated net profit increased 131% to Rs.750.000 millions.
Financial Highlights
for Fiscal Year ended March 31, 2014
• Net Sales: Rs.10960.000 millions, an increase of 43% compared to Rs.7640.000 millions in FY13
• EBITDA: Rs.1630.000 millions, an increase of 87% compared to Rs.870.000 millions in FY13
• PBT: Rs.1120.000 millions, an increase of 143% compared to Rs.460.000 millions in FY13
• Net Profit: Rs.750.000 millions, an increase of 131% compared to Rs.330.000 millions in FY13
• Basic EPS: Rs.37.20 per share as compared to Rs.16.21 per share in FY13.
The Company reported record sales of Rs.10960.000 millions including Rs.3170.000 millions in Q4FY14, its highest revenue in a quarter. The Q4FY14 revenue was 55% higher than the corresponding quarter in FY13 and marks the sixth consecutive quarter of higher sequential sales.
Growth was driven by strong performance across all manufacturing facilities including the Company’s Chinese JV. The primary growth driver in FY14 was led by the Company’s commercialization of its PFI and Finished Dosage expansion at the Gagillapur facility. The Company improved utilization in the newly expanded facility throughout the year and expects to continue improving utilization in the future. In addition, the API facilities continued to increase production which contributed to strong sales.
On a standalone basis, the Finished Dosage business contributed the largest share of revenue at 35% while PFI and API contributed 33% and 32%, respectively. This is compared to 31%, 32% and 37% for Finished Dosages, PFI and API, respectively in FY13.
In addition to strong revenue growth, profitability margins expanded in the fiscal year. EBITDA grew 87% to Rs.1630.000 millions in FY14; the margin expanded 345 basis points to 14.8% During Q4FY14, the EBITDA margin was 16.2%, the highest for the year and an improvement of 227 basis points as compared to the corresponding quarter in FY13. Net Profit was Rs.750.000 millions, the highest in the Company’s history and an increase of 131% as compared to FY13. The net profit margin improved by 261 basis points to 6.9%
Profitability outpaced revenue growth due to several reasons. First, the Company improved its economy of scale by increasing production utilization in its newly expanded PFI and Finished Dosage capacity at the Gagillapur facility. Due to the increased utilization, the product mix shifted more towards finished dosages which bolstered profitability. Also, due to the Company’s focus on operational excellence, the API units also increased capacity through de-bottlenecking which drove profitability. Granules believes the profitability margins from the standalone operations are sustainable.
Due to the Company’s strong performance, the Board of Directors has recommended to increase the dividend to 35% of equity value or Rs.3.50/share from the rate of 20% (Rs.2.00 per share) that was paid in fiscal year 2013.
“The past fiscal year was exciting for Granules. We were able to increase sales while improving profitability due to our relentless focus on delivering high-quality material at a cost-effective price. We believe our product portfolio offers compelling opportunities and we will continue to strengthen our leadership position. In addition to growing our core business, we have been looking at opportunities to diversify our sales by leveraging our core competency of efficient manufacturing. In FY14, Granules made its first acquisition by purchasing Auctus Pharma. The acquisition of Auctus fits into our strategy of being a fully integrated manufacturer while diversifying our product portfolio by adding high-value products with significant market demand. In the short-to-midterm, we will focus on selling APIs from the Auctus portfolio to customers in the regulated markets. Over the long-term, we will add value by offering finished dosages from select APIs in the same portfolio. While we’re certainly happy with the
progress we made in FY14, we have our eyes toward the future. We will continue to strengthen our model and build systems that are sustainable as we continue to scale-up” said Krishna Prasad, Managing Director of Granules India.
About Granules India
Limited
(BSE: 532482, NSE: GRANULES)
Granules is a fast growing pharmaceutical manufacturing company with world class facilities and is committed to
manufacturing excellence, quality and customer service. The Company produces Finished Dosages (FDs), Pharmaceutical Formulation Intermediates (PFIs) and Active Pharmaceutical Ingredients (APIs) for quality conscious customers in the regulated and semi-regulated markets. Granules support customers with unique value, extensive product range, proactive solutions and a global network of associates. The Company’s global presence extends to over 300 customers in 60 countries through offices in India, U.S., U.K., China and Colombia. Granules offer all three components of the pharmaceutical value chain which gives the customers flexibility and choice.
CMT REPORT (Corruption, Money Laundering & Terrorism]
The Public Notice information has been collected from various sources
including but not limited to: The Courts,
1] INFORMATION ON
DESIGNATED PARTY
No exist designating subject or any of its beneficial owners, controlling
shareholders or senior officers as terrorist or terrorist organization or whom
notice had been received that all financial transactions involving their assets
have been blocked or convicted, found guilty or against whom a judgement or
order had been entered in a proceedings for violating money-laundering,
anti-corruption or bribery or international economic or anti-terrorism sanction
laws or whose assets were seized, blocked, frozen or ordered forfeited for
violation of money laundering or international anti-terrorism laws.
2] Court Declaration :
No exist to suggest that subject is or was
the subject of any formal or informal allegations, prosecutions or other
official proceeding for making any prohibited payments or other improper
payments to government officials for engaging in prohibited transactions or
with designated parties.
3] Asset Declaration :
No records exist to suggest that the property or assets of the subject
are derived from criminal conduct or a prohibited transaction.
4] Record on Financial
Crime :
Charges or conviction
registered against subject: None
5] Records on Violation of
Anti-Corruption Laws :
Charges or
investigation registered against subject: None
6] Records on Int’l
Anti-Money Laundering Laws/Standards :
Charges or
investigation registered against subject: None
7] Criminal Records
No
available information exist that suggest that subject or any of its principals
have been formally charged or convicted by a competent governmental authority
for any financial crime or under any formal investigation by a competent
government authority for any violation of anti-corruption laws or international
anti-money laundering laws or standard.
8] Affiliation with
Government :
No record
exists to suggest that any director or indirect owners, controlling
shareholders, director, officer or employee of the company is a government
official or a family member or close business associate of a Government
official.
9] Compensation Package :
Our market
survey revealed that the amount of compensation sought by the subject is fair
and reasonable and comparable to compensation paid to others for similar
services.
10] Press Report :
No press reports / filings exists on
the subject.
CORPORATE GOVERNANCE
MIRA INFORM as part of its Due Diligence do provide comments on
Corporate Governance to identify management and governance. These factors often
have been predictive and in some cases have created vulnerabilities to credit
deterioration.
Our Governance Assessment focuses principally on the interactions
between a company’s management, its Board of Directors, Shareholders and other
financial stakeholders.
CONTRAVENTION
Subject is not known to have contravened any existing local laws,
regulations or policies that prohibit, restrict or otherwise affect the terms
and conditions that could be included in the agreement with the subject.
FOREIGN EXCHANGE RATES
|
Currency |
Unit
|
Indian Rupees |
|
US Dollar |
1 |
Rs.59.88 |
|
UK Pound |
1 |
Rs.100.98 |
|
Euro |
1 |
Rs.82.40 |
INFORMATION DETAILS
|
Information
Gathered by : |
HNA |
|
|
|
|
Analysis Done by
: |
SUB |
|
|
|
|
Report Prepared
by : |
SMN |
SCORE & RATING EXPLANATIONS
|
SCORE FACTORS |
RANGE |
POINTS |
|
HISTORY |
1~10 |
6 |
|
PAID-UP CAPITAL |
1~10 |
5 |
|
OPERATING SCALE |
1~10 |
6 |
|
FINANCIAL CONDITION |
|
|
|
--BUSINESS SCALE |
1~10 |
6 |
|
--PROFITABILIRY |
1~10 |
6 |
|
--LIQUIDITY |
1~10 |
6 |
|
--LEVERAGE |
1~10 |
5 |
|
--RESERVES |
1~10 |
6 |
|
--CREDIT LINES |
1~10 |
5 |
|
--MARGINS |
-5~5 |
- |
|
DEMERIT POINTS |
|
|
|
--BANK CHARGES |
YES/NO |
YES |
|
--LITIGATION |
YES/NO |
YES |
|
--OTHER ADVERSE INFORMATION |
YES/NO |
NO |
|
MERIT POINTS |
|
|
|
--SOLE DISTRIBUTORSHIP |
YES/NO |
NO |
|
--EXPORT ACTIVITIES |
YES/NO |
YES |
|
--AFFILIATION |
YES/NO |
YES |
|
--LISTED |
YES/NO |
YES |
|
--OTHER MERIT FACTORS |
YES/NO |
YES |
|
DEFAULTERS |
|
|
|
--RBI |
YES/NO |
NO |
|
--EPF |
YES/NO |
NO |
|
TOTAL |
|
51 |
This score serves as a reference to assess SC’s credit risk
and to set the amount of credit to be extended. It is calculated from a
composite of weighted scores obtained from each of the major sections of this
report. The assessed factors and their relative weights (as indicated through
%) are as follows:
Financial
condition (40%) Ownership
background (20%) Payment
record (10%)
Credit history
(10%) Market trend
(10%) Operational
size (10%)
RATING EXPLANATIONS
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
>86 |
Aaa |
Possesses an extremely sound financial base with the strongest
capability for timely payment of interest and principal sums |
Unlimited |
|
71-85 |
Aa |
Possesses adequate working capital. No caution needed for credit
transaction. It has above average (strong) capability for payment of interest
and principal sums |
Large |
|
56-70 |
A |
Financial & operational base are regarded healthy. General
unfavourable factors will not cause fatal effect. Satisfactory capability for
payment of interest and principal sums |
Fairly Large |
|
41-55 |
Ba |
Overall operation is considered normal. Capable to meet normal
commitments. |
Satisfactory |
|
26-40 |
B |
Capability to overcome financial difficulties seems comparatively below
average. |
Small |
|
11-25 |
Ca |
Adverse factors are apparent. Repayment of interest and principal sums
in default or expected to be in default upon maturity |
Limited with
full security |
|
<10 |
C |
Absolute credit risk exists. Caution needed to be exercised |
Credit not
recommended |
|
-- |
NB |
New Business |
-- |
This report is issued at your request without any
risk and responsibility on the part of MIRA INFORM PRIVATE LIMITED (MIPL) or
its officials.