MIRA
INFORM REPORT
|
Report Date : |
30.09.2014 |
IDENTIFICATION DETAILS
|
Name : |
BIOCON LIMITED |
|
|
|
|
Registered
Office : |
20th KM, |
|
|
|
|
Country : |
|
|
|
|
|
Financials (as
on) : |
31.03.2014 |
|
|
|
|
Date of
Incorporation : |
29.11.1978 |
|
|
|
|
Com. Reg. No.: |
08-003417 |
|
|
|
|
Capital
Investment / Paid-up Capital : |
Rs.1000.000 Millions |
|
|
|
|
CIN No.: [Company Identification
No.] |
L24234KA1978PLC003417 |
|
|
|
|
TAN No.: [Tax Deduction &
Collection Account No.] |
BLRB00214E |
|
|
|
|
PAN No.: [Permanent Account No.] |
AAACB7461R |
|
|
|
|
Legal Form : |
A Public Limited Liability Company. The Company’s Shares are Listed on
the Stock Exchanges. |
|
|
|
|
Line of Business
: |
Manufacturer of biotechnology products and also
engaged in research and development in the biotechnology sector. |
|
|
|
|
No. of Employees
: |
Not Divulged |
RATING & COMMENTS
|
MIRA’s Rating : |
Aa (72) |
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
71-85 |
Aa |
Possesses adequate working capital. No caution needed for credit
transaction. It has above average (strong) capability for payment of interest
and principal sums |
Large |
|
Status : |
Good |
|
|
|
|
Payment Behaviour : |
Regular |
|
|
|
|
Litigation : |
Exist |
|
|
|
|
Comments : |
Subject is a well-established company having fine track record. The rating reflects company’s healthy financial risk profile marked by
adequate liquidity position and fair profitability and decent profitability
of the company. Trade relations are reported as fair. Business is active. Payments are
reported to be regular and as per commitments. The company can be considered good for normal business dealings at
usual trade terms and conditions. |
NOTES :
Any query related to this report can be made
on e-mail : infodept@mirainform.com
while quoting report number, name and date.
ECGC Country Risk Classification List – June 1, 2014
|
Country Name |
Previous Rating (31.03.2014) |
Current Rating (01.06.2014) |
|
India |
A1 |
A1 |
|
Risk Category |
ECGC
Classification |
|
Insignificant |
A1 |
|
Low |
A2 |
|
Moderate |
B1 |
|
High |
B2 |
|
Very High |
C1 |
|
Restricted |
C2 |
|
Off-credit |
D |
INDIAN ECONOMIC OVERVIEW
N E W S
Verdict Implications
: Apex court order may alter coal import dynamics. Traders go slow on talks
over coal supply contracts, uncertainty over cancellation of blocks weigh on
stocks.
Recent arrest of the
Chennai head of the Registrar of Companies, the ministry of corporate affairs
arm that ensures that companies file all the information required by the
Companies Act is the latest manifestation of a messy fight between a father and
his adopted son for the control of Rs 40000 mn business empire. The Central
Bureau of Investigation arrested Manumeethi Cholan after he accepted Rs 10
lakhs as bribe from M A M Ramaswamy, a CBI official said.
Central Bureau of
Investigation books Electrotherm for cheating Central Bank of Rs 4360 mn.
Infosys maintains
revenue guidance. COO Rao says attrition still an area of concern and it would
take a few more quarters to bring down levels to 13-15 %.
DHL to invest
Euro 100 mn in India over next 2 years. The firm has chosen India to pilot its
e-commerce business model for the Asia-Pacific region.
Blackstone may buy
stake in BlueRidge SEZ in line with the fund’s real estate strategy in India.
Kingfisher Airlines
Ltd grounded in October 2012 under the weight of heavy debt and accumulated
losses, recently approached the Delhi high court for relief in two separate
cases. The airline challenged a notice by Punjab & National Bank alleging
that It had wilfully defaulted on Rs 7700 mn of loans and sought more time to
comply with the requirements under the listing agreements with the Stock
Exchanges.
OnMobile likely to
sack another 300 employees. The lay-offs follow a spate of senior-level exits
over the past two years, starting with of its founder. The overall lay-offs
could number around 600 and are driven by the need to cut costs, says a former
employee.
EXTERNAL AGENCY RATING
|
Rating Agency Name |
CRISIL |
|
Rating |
Long Term Rating : “AA+” |
|
Rating Explanation |
High degree of safety and very low credit risk. |
|
Date |
03.06.2014 |
|
Rating Agency Name |
CRISIL |
|
Rating |
Short Term Rating : “A1+” |
|
Rating Explanation |
Very strong degree of safety and lowest credit risk |
|
Date |
03.06.2014 |
RBI DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available RBI Defaulters’ list.
EPF (Employee Provident Fund) DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available EPF (Employee Provident Fund) Defaulters’ list as of
31-03-2014.
INFORMATION DENIED BY
|
Name : |
Mr. mayank verma |
|
Designation : |
Accounts head |
|
Contact No.: |
91-80-28082808 (Extension 2027) |
|
Date : |
29.09.2014 |
LOCATIONS
|
Registered Office
/Factory 1 / Corporate Headquarters: |
20th
KM, Hosur Main Road, Hebbagodi, Electronics City, Bangalore – 560100,
Karnataka, India |
|
Tel. No.: |
91-80-28422169/28523434/
28082808 / 40144014 |
|
Fax No.: |
91-80-28422623/25531662/28523423 |
|
E-Mail : |
|
|
Website : |
http://www.biocon.com |
|
Area : |
15000
sq. ft. |
|
Locations : |
Owned
|
|
|
|
|
Factory 2 : |
Plot No 113/C2,
Bommasandra Industrial Area, Bommasandra, |
|
|
|
|
Factory 3 : |
Plot No 2,3,4 and
5, Bommasandra – |
|
|
|
|
Factory 4 : |
Plot 213-215 IDA
Phase – II,pashamlaram Medak District – 502307, |
DIRECTORS
As on 31.03.2014
|
Name : |
Ms. Kiran Mazumdar-Shaw |
|
Designation : |
Chairman and Managing Director |
|
Address : |
874/1,
7th Cross III Block, Koramangala, |
|
Date of
Birth/Age : |
20.11.1978 |
|
Qualification
: |
B.Sc.
(Hons.), PG Diploma in Malting and Brewing |
|
Date Of
Appointment : |
01.12.1978 |
|
|
|
|
Name : |
Mr. John Shaw |
|
Designation : |
Vice Chairman |
|
Date of
Birth/Age : |
64 Years |
|
|
|
|
Name : |
Dr. Bala S. Manian |
|
Designation : |
Chairman and Founder |
|
Date of
Birth/Age : |
67 Years |
|
|
|
|
Name : |
Prof. Charles L. Cooney |
|
Designation : |
Director |
|
Address : |
35, |
|
Date of
Birth/Age : |
14.09.1961 |
|
Date of
Appointment : |
27.07.2001 |
|
|
|
|
Name : |
Ms. Mary Harney |
|
Designation : |
Director |
|
Date of
Birth/Age : |
60 Years |
|
|
|
|
Name : |
Prof. Ravi Mazumdar |
|
Designation : |
Director |
|
Address : |
706, |
|
Date of
Birth/Age : |
14.07.1940 |
|
Date of
Appointment : |
08.08.2000 |
|
|
|
|
Name : |
Mr. Russel Walls |
|
Designation : |
Director |
|
Date of
Birth/Age : |
69 Years |
|
|
|
|
Name : |
Mr. Suresh N. Talwar |
|
Designation : |
Director |
|
Date of
Birth/Age : |
74 Years |
|
|
|
|
Name : |
Prof. Catherine Rosenberg |
|
Designation : |
Director |
|
|
|
|
Name : |
Mr. Peter Bains |
|
Designation : |
Director |
|
|
|
|
Name : |
Mr. Daniel M. Bradbury |
|
Designation : |
Director` |
KEY EXECUTIVES
|
CORE
COMMITTEE |
|
|
|
|
|
Name : |
Ms. Kiran Mazumdar-Shaw |
|
Designation : |
Chairman and Managing Director |
|
|
|
|
Name : |
Mr. Murali Krishnan |
|
Designation : |
President |
|
|
|
|
Name : |
Mr. John Shaw |
|
Designation : |
Vice Chairman |
|
|
|
|
Name : |
Dr. Abhijit Barve |
|
Designation : |
President |
|
|
|
|
Name : |
Dr. Arun Chandavarkar |
|
Designation : |
Chief Operating Officer |
|
|
|
|
Name : |
Mr. Rakesh Bamzai |
|
Designation : |
President |
|
|
|
|
Name : |
Mr. Ravi Dasgupta |
|
Designation : |
Group Head |
|
|
|
|
Name : |
Mr. Rahul Agrawal |
|
Designation : |
Finance Department |
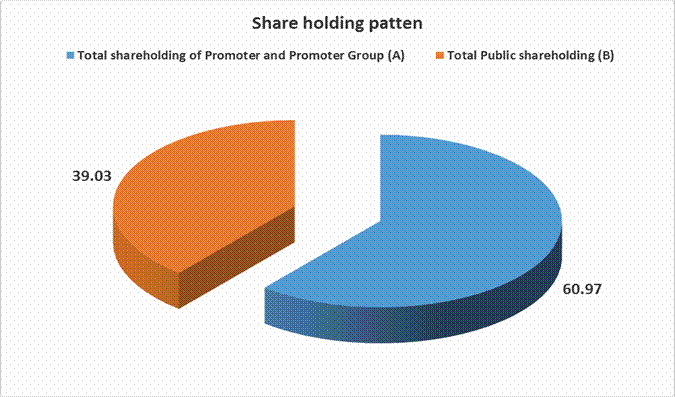
MAJOR SHAREHOLDERS / SHAREHOLDING PATTERN
As on 30.06.2014
|
Category of
Shareholder |
No. of Shares |
Percentage of
Holding |
|
(A) Shareholding of Promoter and Promoter Group |
|
|
|
|
|
|
|
|
80747694 |
40.37 |
|
|
80747694 |
40.37 |
|
|
|
|
|
|
1665558 |
0.83 |
|
|
39535194 |
19.77 |
|
|
41200752 |
20.60 |
|
Total shareholding of Promoter and Promoter Group (A) |
121948446 |
60.97 |
|
(B) Public Shareholding |
|
|
|
|
|
|
|
|
6560039 |
3.28 |
|
|
5495063 |
2.75 |
|
|
28053983 |
14.03 |
|
|
40109085 |
20.05 |
|
|
|
|
|
|
4855269 |
2.43 |
|
|
|
|
|
|
12872435 |
6.44 |
|
|
10728351 |
5.36 |
|
|
9486414 |
4.74 |
|
|
1019057 |
0.51 |
|
|
7834884 |
3.92 |
|
|
632473 |
0.32 |
|
|
37942469 |
18.97 |
|
Total Public shareholding (B) |
78051554 |
39.03 |
|
Total (A)+(B) |
200000000 |
100.00 |
|
(C) Shares held by Custodians and against which Depository
Receipts have been issued |
0 |
0.00 |
|
|
0 |
0.00 |
|
|
0 |
0.00 |
|
|
0 |
0.00 |
|
Total (A)+(B)+(C) |
200000000 |
0.00 |
|
Total (A)+(B)+(C) |
200000000 |
100.00 |
BUSINESS DETAILS
|
Line of Business : |
Manufacturer of biotechnology products and also
engaged in research and development in the biotechnology sector. |
||||||
|
|
|
||||||
|
Products : |
|
GENERAL INFORMATION
|
No. of Employees : |
Not Divulged |
||||||||||||||||||
|
|
|
||||||||||||||||||
|
Bankers : |
· State Bank of India, Overseas Branch, No. 65, St. Marks Road, Bangalore - 560001, Karnataka, India The Hongkong and Shanghai Banking Corporation Limited, 7
M.G.Road, Bangalore - 560001, Karnataka, India |
||||||||||||||||||
|
|
|
||||||||||||||||||
|
Facilities : |
NOTE: LONG TERM
BORROWINGS (a) On February 9, 2000, the Company obtained an
order from the Karnataka Sales Tax Authority for allowing an interest free
deferment of sales tax (including turnover tax) for a period up to 12 years
with respect to sales from its Hebbagodi manufacturing facility for an amount
not exceeding Rs. 649.000 millions. This is an interest free liability. The
amount is repayable in 10 equal half yearly installments of Rs. 65 each
starting from February 2012. (b) On March 31, 2005, the Company entered into
an agreement with the Council of Scientific and Industrial Research (‘CSIR’),
for an unsecured loan of Rs. 3.000 millions for carrying out part of the
research and development project under the New Millennium Indian Technology
Leadership Initiative (‘NMITLI’) Scheme. The loan is repayable over 10 equal
annual installments of Rs. 0.3 starting from April 2009 and carry an interest
rate of 3 percent per annum. (c) (i) On March 31, 2009, the Department of
Scientific and Industrial Research (‘DSIR’) sanctioned financial assistance
for a sum of Rs. 17.000 to the Company for part financing one of its research
projects. The assistance is repayable in the form of royalty payments for
three years post commercialisation of the project in five equal annual
installments of Rs. 3 each. The said projects have been completed during the
year ended March 31, 2010 and the repayments would commence from April 1,
2013. (ii) In addition, during the FY 2010-11, the
Company has further received Rs. 4.000 towards a development project out of
sanctioned amount of Rs. 12.000. The assistance is repayable in the form of
royalty payments for a period of five years post commercialisation of the
project in five equal annual installments of Rs. 3 each. The saidproduct has
not yet been commercialised as at March 31, 2013. (d) On November 3, 2009, the Department of
Biotechnology (‘DBT’) under the Biotechnology Industrial Partnership
Programme (‘BIPP’) has sanctioned financial assistance for a sum of Rs.
53.000 millions to the Company for financing one of its research projects. Of
the said sanctioned amount, the Company had received a sum of Rs. 37.000
millions during the year ended March 31, 2011 and the remaining amount of Rs.
16.000 millions during the previous year. The loan is repayable over 10 half
yearly installments of Rs. 5.000 millions after two years from date of
completion of the project and carries an interest rate of 2 percent per
annum. However, the Company has repaid the loan during the year end March 31,
2013. In addition, on May 23, 2011, the DBT under the
BIPP has sanctioned financial assistance of Rs. 40.000 millions to the
Company for financing another research project. Of thesanctioned amount, the
Company has received a sum of Rs. 12.000 millions during the previous year.
The loan is repayable over 10 half yearly installments of Rs. 4.000 after one
year from date of completion of the project and carries an interest rate of 2
percent per annum. However, the Company has repaid the loan during the year
end March 31, 2013. (e) On August 25, 2010, the Department of Science
and Technology (‘DST’) under the Drugs and Pharmaceutical Research Programme
(‘DPRP’) has sanctioned financial assistance for a sum of Rs. 70.000 millions
to the Company for financing one of its research projects. Of the said
sanctioned amount, the Company has received the first installment of Rs.
14.000 millions during the year ended March 31, 2011 and the remaining amount
during the year ended March 31, 2012. The loan is repayable over 10 annual
installments of Rs. 7 each starting from July 1, 2012, and carries an
interest rate of 3 percent per annum. (f) In respect of the financial assistance
received under the aforesaid programmes (refer notes (b) to (e) above), the
Company is required to utilize the funds for the specified projects and is
required to obtain prior approvals from the said authorities for disposal of
assets / Intellectual property rights acquired/developed under the above
programmes. SHORT TERM
BORROWINGS (i) The Company has obtained foreign currency
denominated loans of Rs. 491.000 (US$ 9 million) [March 31, 2012 - Rs.
812.000 millions (US$ 15.95 million)], carrying an interest rate of LIBOR
plus 0.5% to 1.50% p.a., from Bank/Financial institutions as at March 31,
2013. (ii) The Company has working
capital facilities with Banks carrying interest rate ranging from 11%-13% per
annum. These facilities are repayable on demand, secured by pari-passu first
charge on inventories and trade receivables. As on March 31, 2013, the Company
has utilized fund based limits of Rs. 282.000 millions (March 31, 2012 - Rs.
56.000 millions) |
|
|
|
|
Banking
Relations : |
--- |
|
|
|
|
Auditors : |
|
|
Name : |
S. R. Batliboi and Associates Chartered Accountants |
|
Address : |
Bangalore, Karnataka, India |
|
|
|
|
Joint Venture : |
NeoBiocon FZ LLC |
|
|
|
|
Associate : |
IATRICa Inc. |
|
|
|
|
Subsidiary : |
·
Syngene International Limited Clinigene International Limited Biocon Biopharmaceuticals
Limited Biocon Research Limited Biocon SA Biocon Sdn.Bhd. |
|
|
|
|
Enterprise owned by key management personnel : |
Glentec International |
CAPITAL STRUCTURE
As on 31.03.2014
Authorised Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
220000000 |
Equity shares |
Rs.5/- each |
Rs. 1100.000 millions |
|
|
|
|
|
Issued, Subscribed & Paid-up Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
200000000 |
Equity shares |
Rs.5/- each |
Rs. 1000.000 millions |
|
|
|
|
|
As on 31.03.2014
(a) Reconciliation of the shares outstanding at
the beginning and at the end of the reporting period
|
Equity Shares |
31.03.2014 |
|
|
|
No. |
Rs. In millions |
|
At the beginning of the year |
200,000,000 |
1000.000 |
|
Issued during the year |
-- |
-- |
|
Outstanding at the end of the year |
200,000,000 |
1000.000 |
(b) Terms/rights
attached to equity shares
The Company has only one class of equity shares
having a par value of Rs. 5 per share.
Each holder of equity shares is entitled to one vote per share. The Company
declares and pays dividends in Indian Rupees. The dividend proposed by the
Board of Directors is subject to the approval of the shareholders in the
ensuing Annual General Meeting.
During the year ended March 31, 2013, final
dividends proposed for distribution to equity shareholders was Rs. 7.5 (March 31, 2012 – Rs. 5) per share. In the event of liquidation of the Company,
the holders of equity shares will be entitled to receive remaining assets of
the Company, after distribution of all preferential amounts, if any. The
distribution will be in proportion to the number of equity shares held by the
shareholders.
(c) Aggregate
number of bonus shares issued during the period of five years immediately
preceding the reporting date
On September 15, 2008, the Company issued
100,000,000 equity shares of Rs. 5 each as fully paid bonus shares by
capitalization of balance in the securities premium account of Rs.500.
iv. Details of shareholders holding more than 5% shares in the Company
|
Equity Shares |
31.03.2014 |
|
|
Equity shares of
Rs. 5 each fully paid |
No. |
% holding |
|
Dr Kiran Mazumdar Shaw |
79,287,564 |
39.64% |
|
Glentec
International |
39,535,194 |
19.77% |
As per of the Company, including its register of
shareholders/members. The above shareholding represents both legal and
beneficial ownerships of shares.
(e) Shares reserved for issue under options for
details of shares reserved for issue under the employee stock option (ESOP)
plan of the Company.
FINANCIAL DATA
[all figures are
in Rupees Millions]
ABRIDGED BALANCE
SHEET
|
SOURCES OF FUNDS |
31.03.2014 |
31.03.2013 |
31.03.2012 |
|
I.
EQUITY
AND LIABILITIES |
|
|
|
|
(1)Shareholders' Funds |
|
|
|
|
(a) Share Capital |
1000.000 |
1000.000 |
1000.000 |
|
(b) Reserves & Surplus |
23177.000 |
21068.000 |
19964.000 |
|
(c) Money
received against share warrants |
0.000 |
0.000 |
0.000 |
|
|
|
|
|
|
(2) Share Application money pending allotment |
0.000 |
0.000 |
0.000 |
|
Total
Shareholders’ Funds (1) + (2) |
24177.000 |
22068.000 |
20964.000 |
|
|
|
|
|
|
(3)
Non-Current Liabilities |
|
|
|
|
(a) long-term borrowings |
259.000 |
400.000 |
605.000 |
|
(b) Deferred tax liabilities (Net) |
400.000 |
302.000 |
349.000 |
|
(c) Other long term liabilities |
1311.000 |
1083.000 |
649.000 |
|
(d) long-term provisions |
0.000 |
0.000 |
0.000 |
|
Total Non-current Liabilities (3) |
1970.000 |
1785.000 |
1603.000 |
|
|
|
|
|
|
(4) Current Liabilities |
|
|
|
|
(a) Short term borrowings |
815.000 |
773.000 |
868.000 |
|
(b) Trade payables |
2685.000 |
2650.000 |
2511.000 |
|
(c) Other current
liabilities |
899.000 |
679.000 |
769.000 |
|
(d) Short-term provisions |
1639.000 |
2177.000 |
1488.000 |
|
Total Current Liabilities (4) |
6038.000 |
6279.000 |
5636.000 |
|
|
|
|
|
|
TOTAL |
32185.000 |
30132
.000 |
28203.000 |
|
|
|
|
|
|
II.
ASSETS |
|
|
|
|
(1) Non-current assets |
|
|
|
|
(a) Fixed Assets |
|
|
|
|
(i) Tangible assets |
9410.000 |
8455.000 |
6757.000 |
|
(ii) Intangible Assets |
83.000 |
59.000 |
93.000 |
|
(iii) Capital
work-in-progress |
1018.000 |
512.000 |
825.000 |
|
(iv)
Intangible assets under development |
0.000 |
0.000 |
0.000 |
|
(b) Non-current Investments |
1449.000 |
1660.000 |
1664.000 |
|
(c) Deferred tax assets (net) |
0.000 |
0.000 |
0.000 |
|
(d) Long-term Loan and Advances |
5546.000 |
4713.000 |
5343.000 |
|
(e) Other Non-current assets |
6.000 |
0.000 |
0.000 |
|
Total Non-Current Assets |
17512.000 |
15399.000 |
14682.000 |
|
|
|
|
|
|
(2) Current assets |
|
|
|
|
(a) Current investments |
3483.000 |
4530.000 |
4906.000 |
|
(b) Inventories |
3576.000 |
3589.000 |
3404.000 |
|
(c) Trade receivables |
4946.000 |
4270.000 |
4450.000 |
|
(d) Cash and cash
equivalents |
2042.000 |
1792.000 |
400.000 |
|
(e) Short-term loans and
advances |
568.000 |
510.000 |
302.000 |
|
(f) Other current assets |
58.000 |
42.000 |
59.000 |
|
Total Current Assets |
14673.000 |
14733.000 |
13521.000 |
|
|
|
|
|
|
TOTAL |
32185.000 |
30132.000 |
28203.000 |
PROFIT & LOSS
ACCOUNT
|
|
PARTICULARS |
31.03.2014 |
31.03.2013 |
31.03.2012 |
|
|
|
SALES |
|
|
|
|
|
|
|
Income |
22025.000 |
19380.000 |
15558.000 |
|
|
|
Other Income |
606.000 |
515.000 |
666.000 |
|
|
|
TOTAL (A) |
22631.000 |
19895.000 |
16224.000 |
|
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
|
|
Cost of raw materials and packing materials consumed |
8876.000 |
8300.000 |
6971.000 |
|
|
|
Purchases of traded goods |
1039.000 |
857.000 |
857.000 |
|
|
|
Employee benefits expense |
2664.000 |
2276.000 |
1916.000 |
|
|
|
Other expenses |
4741.000 |
4110.000 |
2893.000 |
|
|
|
Exceptional
items |
0.000 |
139.000 |
0.000 |
|
|
|
(Increase)/Decrease in
inventories of finished goods, traded goods and work-in-progress |
13.000 |
(179.000) |
(414.000) |
|
|
|
Recovery of Product development
costs from co-development partner |
(41.000) |
(41.000) |
0.000 |
|
|
|
|
|
|
|
|
|
|
TOTAL (B) |
17292.000 |
15462.000 |
12223.000 |
|
|
|
|
|
|
|
|
|
PROFIT/
(LOSS) BEFORE INTEREST, TAX, DEPRECIATION
AND AMORTISATION (A-B) (C) |
5339.000 |
4433.000 |
4001.000 |
|
|
|
|
|
|
|
|
|
Less |
FINANCIAL
EXPENSES (D) |
9.000 |
12.000 |
17.000 |
|
|
|
|
|
|
|
|
|
|
PROFIT
/ (LOSS) BEFORE TAX, DEPRECIATION AND AMORTISATION (C-D) (E) |
5330.000 |
4421.000 |
3984.000 |
|
|
|
|
|
|
|
|
|
Less/ Add |
DEPRECIATION/
AMORTISATION (F) |
1244.000 |
951.000 |
940.000 |
|
|
|
|
|
|
|
|
|
|
PROFIT/
(LOSS) BEFORE TAX (E-F) (G) |
4086.000 |
3470.000 |
3044.000 |
|
|
|
|
|
|
|
|
|
Less |
TAX (H) |
842.000 |
713.000 |
489.000 |
|
|
|
|
|
|
|
|
|
|
PROFIT/
(LOSS) AFTER TAX (G-H) (I) |
3244.000 |
2757.000 |
2555.000 |
|
|
|
|
|
|
|
|
|
Add |
Impact of scheme of merger for earlier year (L) |
55.000 |
0.000 |
0.000 |
|
|
|
|
|
|
|
|
|
Add |
PREVIOUS
YEARS’ BALANCE BROUGHT FORWARD |
14476.000 |
13750.000 |
12613.000 |
|
|
|
|
|
|
|
|
|
Less |
APPROPRIATIONS |
|
|
|
|
|
|
|
Transfer to General Reserve |
330.000 |
276.000 |
256.000 |
|
|
|
Dividend |
170.000 |
1500.000 |
1000.000 |
|
|
|
Tax on Dividend |
1000.000 |
255.000 |
162.000 |
|
|
BALANCE CARRIED
TO THE B/S |
16275.000 |
14476.000 |
13750.000 |
|
|
|
|
|
|
|
|
|
|
EARNINGS IN FOREIGN
CURRENCY |
|
|
|
|
|
|
|
Export of goods on FOB basis |
10669.000 |
9450.000 |
6661.000 |
|
|
|
Licensing and development fees |
37.000 |
114.000 |
27.000 |
|
|
|
Other
operating revenue |
97.000 |
342.000 |
79.000 |
|
|
|
Other
income |
0.000 |
0.000 |
5.000 |
|
|
|
Interest
on foreign currency loan given to subsidiary company |
0.000 |
0.000 |
1.000 |
|
|
TOTAL EARNINGS |
10803.000 |
9906.000 |
6773.000 |
|
|
|
|
|
|
|
|
|
|
IMPORTS |
|
|
|
|
|
|
|
Raw Materials |
5239.000 |
4917.000 |
3833.000 |
|
|
|
Packing materials |
247.000 |
177.000 |
193.000 |
|
|
|
Traded goods |
408.000 |
250.000 |
0.000 |
|
|
|
Maintenance spares |
66.000 |
49.000 |
44.000 |
|
|
|
Capital goods |
613.000 |
168.000 |
411.000 |
|
|
TOTAL IMPORTS |
6573.000 |
5561.000 |
4481.000 |
|
|
|
|
|
|
|
|
|
|
Earnings /
(Loss) Per Share (Rs.) |
|
|
|
|
|
|
Basic |
16.81 |
14.08 |
13.04 |
|
|
|
Diluted
|
16.62 |
13.95 |
12.92 |
|
QUARTERLY /
SUMMARISED RESULTS
|
Particulars (Rs.
Millions) |
|
|
|
Jun 2014 |
|
Audited / UnAudited |
|
|
|
UnAudited |
|
Net Sales |
|
|
|
5586.900 |
|
Total Expenditure |
|
|
|
4422.900 |
|
PBIDT (Excl OI) |
|
|
|
1164.000 |
|
Other Income |
|
|
|
117.400 |
|
Operating Profit |
|
|
|
1281.400 |
|
Interest |
|
|
|
2.700 |
|
Exceptional Items |
|
|
|
0.000 |
|
PBDT |
|
|
|
1278.700 |
|
Depreciation |
|
|
|
307.400 |
|
Profit Before Tax |
|
|
|
971.300 |
|
Tax |
|
|
|
225.000 |
|
Provisions and contingencies |
|
|
|
0.000 |
|
Profit After Tax |
|
|
|
746.300 |
|
Extraordinary Items |
|
|
|
0.000 |
|
Prior Period Expenses |
|
|
|
0.000 |
|
Other Adjustments |
|
|
|
0.000 |
|
Net Profit |
|
|
|
746.300 |
KEY RATIOS
|
PARTICULARS |
|
31.03.2014 |
31.03.2013 |
31.03.2012 |
|
PAT / Total Income |
(%) |
14.33 |
13.86 |
15.75 |
|
|
|
|
|
|
|
Net Profit Margin (PBT/Sales) |
(%) |
18.55 |
17.91 |
19.57 |
|
|
|
|
|
|
|
Return on Total Assets (PBT/Total Assets} |
(%) |
13.75 |
12.41 |
11.84 |
|
|
|
|
|
|
|
Return on Investment (ROI) (PBT/Networth) |
|
0.17 |
0.16 |
0.15 |
|
|
|
|
|
|
|
Debt Equity Ratio (Total Debt/Networth) |
|
0.04 |
0.05 |
0.07 |
|
|
|
|
|
|
|
Current Ratio (Current Asset/Current Liability) |
|
2.43 |
2.35 |
2.40 |
FINANCIAL ANALYSIS
[all figures are
in Rupees Millions]
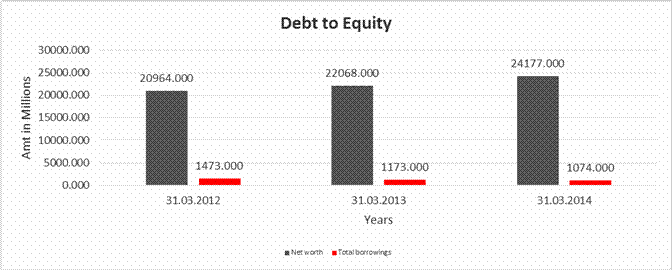
DEBT EQUITY RATIO
|
Particular |
31.03.2012 |
31.03.2013 |
31.03.2014 |
|
|
(Rs.
In Millions) |
(Rs.
In Millions) |
(Rs.
In Millions) |
|
Share Capital |
1000.000 |
1000.000 |
1000.000 |
|
Reserves & Surplus |
19964.000 |
21068.000 |
23177.000 |
|
Net
worth |
20964.000 |
22068.000 |
24177.000 |
|
|
|
|
|
|
long-term borrowings |
605.000 |
400.000 |
259.000 |
|
Short term borrowings |
868.000 |
773.000 |
815.000 |
|
Total
borrowings |
1473.000 |
1173.000 |
1074.000 |
|
Debt/Equity
ratio |
0.070 |
0.053 |
0.044 |
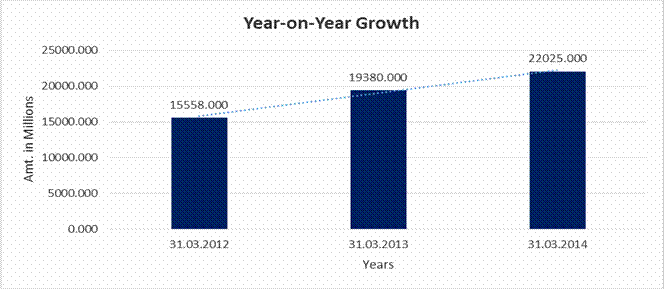
YEAR-ON-YEAR GROWTH
|
Year
on Year Growth |
31.03.2012 |
31.03.2013 |
31.03.2014 |
|
|
(Rs.
In Millions) |
(Rs.
In Millions) |
(Rs.
In Millions) |
|
Sales |
15558.000 |
19380.000 |
22025.000 |
|
|
|
24.566 |
13.648 |
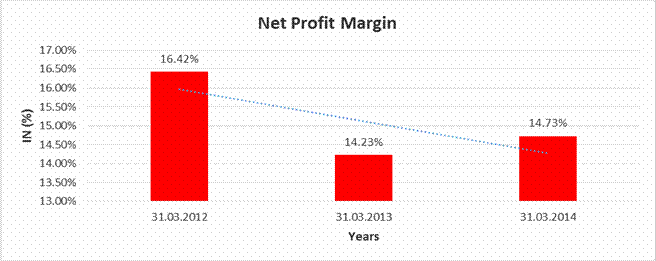
NET PROFIT MARGIN
|
Net
Profit Margin |
31.03.2012 |
31.03.2013 |
31.03.2014 |
|
|
(Rs.
In Millions) |
(Rs.
In Millions) |
(Rs.
In Millions) |
|
Sales |
15558.000 |
19380.000 |
22025.000 |
|
Profit |
2555.000 |
2757.000 |
3244.000 |
|
|
16.42% |
14.23% |
14.73% |
LOCAL AGENCY FURTHER INFORMATION
|
Sr. No. |
Check List by Info
Agents |
Available in Report (Yes / No) |
|
1] |
Year of Establishment |
Yes |
|
2] |
Locality of the firm |
Yes |
|
3] |
Constitutions of the firm |
Yes |
|
4] |
Premises details |
Yes |
|
5] |
Type of Business |
Yes |
|
6] |
Line of Business |
Yes |
|
7] |
Promoter's background |
Yes |
|
8] |
No. of employees |
Yes |
|
9] |
Name of person contacted |
Yes |
|
10] |
Designation of contact person |
Yes |
|
11] |
Turnover of firm for last three years |
Yes |
|
12] |
Profitability for last three years |
Yes |
|
13] |
Reasons for variation <> 20% |
----- |
|
14] |
Estimation for coming financial year |
No |
|
15] |
Capital in the business |
Yes |
|
16] |
Details of sister concerns |
Yes |
|
17] |
Major suppliers |
No |
|
18] |
Major customers |
No |
|
19] |
Payments terms |
No |
|
20] |
Export / Import details (if applicable) |
No |
|
21] |
Market information |
----- |
|
22] |
Litigations that the firm / promoter involved in |
Yes |
|
23] |
Banking Details |
Yes |
|
24] |
Banking facility details |
Yes |
|
25] |
Conduct of the banking account |
----- |
|
26] |
Buyer visit details |
----- |
|
27] |
Financials, if provided |
Yes |
|
28] |
Incorporation details, if applicable |
Yes |
|
29] |
Last accounts filed at ROC |
Yes |
|
30] |
Major Shareholders, if available |
Yes |
|
31] |
Date of Birth of Proprietor/Partner/Director, if available |
Yes |
|
32] |
PAN of Proprietor/Partner/Director, if available |
No |
|
33] |
Voter ID No of Proprietor/Partner/Director, if available |
No |
|
34] |
External Agency Rating, if available |
Yes |
LITIGATION DETAILS
|
High Court of Karnataka - Principal Bench
at Bangalore |
|
ITA 650/2013 |
|
Petitioner/Appnt. |
THE COMMISSIONER OF INCOME - TAX |
|
|
Respondent/Defnt.
Name |
M/S BIOCON LIMITED |
|
|
Petnr./Appnt.
Advocate |
ARAVIND KV |
|
|
Respnt./Defnt.
Advocate |
|
|
|
Date Filed |
13/12/2013 |
Classification |
|
District |
Bangalore City |
|
|
Stage |
Hearing
Last Posted for: ADMISSION |
|
Last Action Taken |
ADMIT/RULE Last
Date of Action 01/04/2014 Next hearing date |
|
Before Hon'ble
Judge/s |
DILIP B.BHOSALE B.MANOHAR |
|
Case No |
Court name |
Disposal Dt |
|
ITA 371/2010 |
INCOME TAX APPELLATE TRIBUNAL BANGALORE |
16/07/2013 |
|
Sl.No. |
Honble Judge |
Date of Order |
|
1 |
HONBLE DBBJ & BMJ |
01/04/2014 |
UNSECURED LOANS
|
PARTICULARS |
31.03.2014 (Rs
in Millions) |
31.03.2013 (Rs in Millions) |
|
LONG TERM BORROWINGS |
|
|
|
Deferred sales
tax Liability |
195.000 |
324.000 |
|
Other
loans and advance |
|
|
|
NMITU – CSIR
Loan |
1.000 |
2.000 |
|
Financial
Assistance From DSIR |
14.000 |
18.000 |
|
Financial Assistance
From DST |
49.000 |
56.000 |
|
|
|
|
|
SHORT TERM
BORROWINGS |
|
|
|
From
banks/Financial institutions |
|
|
|
Packing credit foreign currency
loan (unsecured) |
0.000 |
491.000 |
|
Total
|
259.000 |
891.000 |
INDEX OF CHARGES
|
S.No. |
Charge ID |
Date of Charge Creation/Modification |
Charge amount secured |
Charge Holder |
Address |
Service Request Number (SRN) |
|
1 |
10300393 |
29/06/2011 |
44,267,000.00 |
DEPARTMENT OF BIOTECHNOLOGY |
6-8TH FLOOR, BLOCK
NO. 2, CGO COMPLEX, NEW DELHI, |
B18143230 |
|
2 |
10255822 |
12/11/2010 |
57,081,000.00 |
DEPARTMENT OF BIOTECHNOLOGY |
6-8TH FLOOR, BLOCK NO. 2, CGO COMPLEX, LODHI ROAD,, NEW DELHI, DELHI - 110003, INDIA |
B01390137 |
|
3 |
10059940 |
17/02/2007 |
650,000,000.00 |
STATE BANK OF INDIA |
OVERSEAS BRANCH, NO. 65, ST. MARKS ROAD,, BANGALORE, KARNATAKA - 560001, INDIA |
A11660974 |
|
4 |
10060347 |
17/02/2007 |
650,000,000.00 |
STATE BANK OF INDIA |
OVERSEAS BRANCH, NO. 65, ST. MARKS ROAD,, BANGALORE, KARNATAKA - 560001, INDIA |
A11661360 |
|
5 |
80022593 |
23/07/2010 * |
1,773,500,000.00 |
THE HONGKONG AND
SHANGHAI BANKING CORPORATION LIMI |
7 M.G.ROAD, BANGALORE, KARNATAKA - 560001, INDIA |
A90645920 |
Biocon to Mark a Decade of Excellence in
Affordable Insulin Therapy
Bangalore, India,
August 10th, 2014
Biocon Ltd, Asia's premier biotechnology company, kicked off a series of patient outreach programs to mark a Decade of Excellence in Affordable Insulin Therapy, through which it has been touching the lives of lakhs of diabetic patients. Biocon was an early pioneer in the development of generic recombinant human Insulin (rh-Insulin) and introduced it to patients in India in 2004.
As a part of the initiative, special programs like health camps for senior citizens are being organized in several cities across India. In Bangalore, Dr. N. Prakash (General Practitioner, MBBS DNB) led the company’s efforts at the Shree Old Age Home in Mahalakshmipuram, where over 40 senior citizens underwent a comprehensive free health check-up. The camp included cardio-diabetic screening, health & eye checkups followed by doctor consultations and a session on diabetes awareness.
The company’s engagement with the senior citizens who underwent screening at the health camp will extend to subsidized/free lifetime medication (oral cardio, anti – diabetics & insulins ) and distribution of free optical devices.
The health camp was organized as a part of Biocon’s efforts to improve the quality of life of patients, especially the less privileged & neglected members of society who deserve that extra attention and care in the advancing years.
Commenting on this
noble initiative, Mr. Ravindra Limaye, President, Marketing, Biocon said, “The
instances of chronic morbidities like diabetes and hypertension among the
geriatric population are constantly increasing. Also, as age advances, this is
the population which is neglected worldwide. Proper diagnosis and timely treatment
are key in reducing the burden of illness and associated costs.”
“For the past decade,
Biocon has been hand holding patients through a holistic & highly effective
diabetes management program that reaches out to all stakeholders. As we
approach the milestone of a decade of excellence in affordable insulin therapy
in India, we are endeavoring to empower many more fellow Indians with increased
choices in healthcare management,” he added.
Dr. N. Prakash said:
“I am happy to be a part of this initiative from Biocon to bring healthcare
services to the doorsteps of less privileged members of the society. I feel
they will greatly benefit from the health interventions that we did today.”
Developing countries like India are challenged with a much larger disease burden due to ageing populations and rising incidence of non-communicable diseases (NCDs) like diabetes, cancer and autoimmune disorders.
Among the NCDs, diabetes represents a huge disease burden for India, where there were 65 million cases of diabetes in 2013. It is estimated that India could have over 85 million diabetes patients by 2030. (Source: ICMR; Research Society for the Study of Diabetes in India).
Over the past decade, Biocon has been offering cost-effective and easily accessible treatment for diabetes, which typically imposes a huge economic burden on patients that gets magnified because it leads to related complications, including heart, kidney, eye and foot disease.
Biocon has been addressing the large need for affordable Insulin therapy in India through its generic rh-Insulin and analogs. It has increased patient access to Insulin across India by ensuring affordability, resulting in improved diabetes management. Nearly a million patients have benefited from Biocon’s inclusive diabetes management programme till date.
Biocon's differentiated insulin products and devices and personalized medical support have given the company the distinction of being not only the largest Indian insulin’s company but the fastest growing insulin’s company in India as well. (Source: MAT June 2014, IMS).
‘Winning with Diabetes’ is an ongoing patient outreach initiative of Biocon, which is manned by around 100 diabetes care advisors working round-the-clock to improve patient adherence to therapy (medication, diet and exercise) through a structured six visit plan for counseling called ‘iTAP’ (insulin therapy assistance program). Other programs include ABIDE, a novel diabetes education initiative for medical practitioners and Sampark, a chemist education program.
Biocon announces Q1 FY15 results
Revenues: ₹ 7420.000 Millions; EBITDA: ₹
1910.000 Millions; PAT: ₹ 1030.000 Millions
Bengaluru, India: July 24, 2014
Commenting on the
quarterly performance and highlights, Chairman and Managing Director, Kiran
Mazumdar-Shaw stated, ““Their revenue growth this quarter has been muted. Their
business performance reflects the challenges that we are temporarily facing in
some of their key markets especially in the Middle East We are working towards
diversifying their regional dependencies to diminish the impact of such
externalities. However, we have sustained their operating margins and profits
despite increase in costs. Their business profitability has remained intact
indicating the benefits of portfolio optimization. Their development pipeline
across biosimilars and novel candidates continues to progress well. We remain
committed towards improving their performance in the coming quarters”.
Highlights:
· Biocon files its first set of ANDAs, targets the US generics market
· Extension of the research collaboration between Bristol Myers Squibb and Syngene
· Appointment of Mr. Siddharth Mittal as Chief Financial Officer, Biocon
Business Performance
Financial Highlights: Q1 FY15 (In ₹ Millions)
|
Revenue : 7420.000 R&D Expenses: 310.000
(6% of Biopharmaceutical sales) EBITDA: 191.000 (EBITDA
Margin: 26%) PAT: 1030.000
(PAT Margin: 14%) |
Revenue Breakup: • Biopharmaceuticals: 5470.000 • Research Services: 172.000 • Other Income: 230.000 |
Biopharmaceuticals
Biopharma
The biopharma segment
recorded revenues of 4360.000 Millions in Q1FY15.
The e biopharma business was impacted this quarter by various external factors including the ongoing geo political conflicts in the MENA region, affecting the growth of this business. We believe this is a temporary phasing issue as the underlying demand remains robust. We hope that stability in the region will restore business momentum. We are working towards mitigating the impact of these regional instabilities on their Business performance.
This quarter we filed their first ANDAs, targeting the US generics market. This effort is part of their communicated strategy to move up the pharma value chain to enter finished dosages with generic formulations. We continue working towards further optimization of their product portfolio in the small molecules vertical, with a clear focus on profitable growth.
We remain on track to initiate global phase III trials for their biosimilar glargine. Their upcoming insulin facility in Malaysia continues to progress well and we expect the plant to be commissioned, as per plan in the second half of this fiscal.
Branded Formulations
The branded formulations recorded revenues of 1110.000 Millions in Q1 FY15. The vertical grew at 10% YoY, in line with the industry. This quarter, we have reorganized certain divisions within this vertical. The reorganization largely focuses around the cardiology and diabetes divisions to drive synergies around key anchor brands and optimization of product portfolios with a focus on profitability.
Novel Molecules
The initial set of trials for their oral insulin molecule, IN-105, are on track. We continue to engage with potential partners to out-licensing discussions for their novel anti-CD6 molecule, Itolizumab.
Research Services
The research services segment grew by 12% YoY in Q1 FY15, recording revenues of 1720.000 Millions. This quarter witnessed the extension of their research collaboration with Bristol Myers Squibb, for 5 years. This extension of their longest standing collaboration validates their integrated drug discovery model and reiterates their commitment towards quality, world class research.
Commenting on this performance, Peter Bains, Director Syngene International, said, “They have made a steady start to the new fiscal, and will build upon this performance during the course of the year. They have been investing in capacity expansions, some of which will come into play in the latter half of FY15. The new capacities will help them expand their order book and build growth momentum. A key milestone this quarter was the extension of their partnership with Bristol Myers Squibb until the year 2020.”
Appointments:
Mr. Siddharth Mittal, President – Finance has been appointed as Chief Financial Officer of Biocon Limited effective August 01, 2014. He joined Biocon in May 2013 and takes over from Mr. Murali Krishnan, who retires after more than 30 years of distinguished service with the organization. Siddharth is a Chartered Accountant (India) and a CPA (US) and has more than 15 years of global and diversified experience. Prior to joining Biocon, he was Vice President-Finance and Corporate Controller with a leading US based multinational Information Technology Company based in Bangalore.
Outlook
The inherent growth drivers for their business remain intact and will play out over the course of this year. The progress in their development pipeline (across biosimilars and novel molecules) will help some of their molecules to enter the clinic. However, these milestones are subject to various external dynamics including the business and clinical trial environment in the country. They continue to make investments across infrastructure and people, to support their growth and work steadfastly to deliver strong, sustained value growth to their stakeholders.
CORPORATE INFORMATION
Subject
was incorporated at Bangalore in 1978 for manufacture of biotechnology
products. Subject is an integrated healthcare company engaged in manufacture of
biotechnology products for the pharmaceutical sector. The Company is also
engaged in research and development in the biotechnology sector. During the
year ended March 31, 2007, the Company had received an approval for operation
of SEZ Developer and for setting up SEZ Unit operations to be located within
Biocon SEZ.
Syngene
International Limited ('Syngene'), promoted by Dr Kiran Mazumdar Shaw, was
incorporated at Bangalore in 1993. In March 2002, Biocon acquired 99.99 per
cent of the equity shares of Syngene and, resultantly, Syngene became the
subsidiary of Biocon. As at March 31, 2014, 12.31% of the equity interest in
Syngene is held by third parties.
On
January 10, 2008, Biocon entered into an agreement with Dr. B.R. Shetty to set
up a joint venture Company NeoBiocon FZ-LLC, with a 50% equity interest
incorporated in Dubai (‘NeoBiocon’).
The
Company has also established Biocon Research Limited (‘BRL’), a subsidiary
of the Company to undertake research and development in novel and innovative
drug initiatives.
During
the year ended March 31, 2011, Biocon set up a wholly owned subsidiary company
in Malaysia, Biocon Sdn. Bhd. (‘Biocon Malaysia’) for
development and manufacture of bio-pharmaceuticals.
During the year ended March 31, 2014, the Company has established Biocon Academy, a not for profit company under Companies Act, 1956 to provide educational courses, training and research in the biosciences, life sciences and all fields of study.
MANAGEMENT DISCUSSION
AND ANALYSIS
Industry Landscape,
Opportunity and Outlook
Global pharmaceutical
market
Fiscal 2014 was an interesting phase in the evolution of the global pharma market; marked by increased regulatory oversight, continued pricing pressures and a sustained wave of pharma reforms in various developed markets. These externalities not only defined the growth momentum but also the strategies employed across the industry to return to sustainable growth. The slowdown in pharma growth in the developed markets due to various recessionary and fiscal prudence measures has largely been offset by the sustained momentum seen across developing markets. In fact, IMS expects this trend to continue for the next 5 years as well.
One of the key driver for this differential growth performance, beyond the respective economic engines, is the healthcare support system in these geographies. The developing markets have seen an increasing trend of government-sponsored healthcare initiatives aimed at decreasing the out-of-pocket (OOP) spend of patients. The developed markets on the other hand, are trying to do a balancing act between the current OOP spending from patients and the sustained pressure to decrease their healthcare spends. In both of these scenarios, the current drug pricing mechanisms have come under substantial scrutiny and criticism by various stakeholders.
To elaborate further: Barring a few exceptions, the list prices for high end medications including biologics does not vary substantially between the developed and developing markets. This is despite the disparity in income and the high OOP expenditure in developing markets, which makes these drugs unaffordable to a large section of the patient pool. As the above figure shows, the highest OOP expenditures take place in some of the countries with lowest GDP per capita. Several governments in these countries are looking to expand accessibility and affordability by expanding healthcare coverage, active patent regime management and encouraging local players to make generic versions of both small molecules and biologics.
This momentum in the developing markets has helped crystallise newer business models to help deliver affordable innovation with improved patient outcomes and value to the various stakeholders. The regulators are opening up further to the prospect of biosimilars, with regulatory guidelines gaining more clarity.
The scenario in the developed countries is not very different either. The increasing gap between drug prices and nationwide inflation rates has created an unaffordability gap. Given that the pharma companies have now begun to charge significant premiums for their latest novel drugs, the various stakeholders in the healthcare management process find themselves at crossroads when it comes to containing their healthcare expenditures.
Given that the next generics patent wave is at least 5 years away, countries are increasingly turning towards biosimilars to balance healthcare expenses. The European Union (EU) has been at the forefront of encouraging the uptake of biosimilars and shaping the regulatory landscape as well. A key event this year was the regulatory approval granted to a biosimilar monoclonal antibody (infliximab) for commercialization in EU.
The new biosimilar based business models, therefore focus on delivering quality biosimilars buoyed by the healthcare rationing initiatives being adopted across developed markets. The financial viability for a biosimilar based business model gains further veracity if we look at the uptake of biosimilars in the five key EU nations
We have seen differential rates of biosimilar uptake in various EU member nations, largely reflecting the variations in their local healthcare systems. On the one hand we have Germany, which saw quick adoption of biosimilars at launch; while other nations like Spain and Italy have slowly warmed up to biosimilars. This trajectory is remarkably similar to the adoption behaviour we saw when generic small molecules were introduced in the developed markets. This gives us further confidence that it is a matter of time and further experience, which will be the inflexion point for the global biosimilars market.
Despite these encouraging developments, the biggest caveat in the evolution of the pharma market still lies in the regulatory space. The cautious approach to biosimilars by certain regulators has helped delay millions of dollars worth of potential savings which could have accrued to both patients and healthcare systems worldwide. IMS predicts global pharma spending to exceed $1 Trillion in 2014, largely due to the delay in entry of biosimilars in the market place1.
The paradox for biosimilars lies in the fact that, in several cases, the clinical trial requirements for the biosimilar are significantly larger vis- à-vis what were used to approve the reference innovator biologic in the first place. A fine balance is hence needed between the value of additional information gained from extensive clinical trials vis-à-vis the level of safety, efficacy and biosimilarity data which would already be available from pre-Phase III analysis.
The next few years would be critical in shaping the debate around affordability and equality to access. The implementation of the Affordable Care Act in the US, coupled with the austerity measures across developed markets to support economic recovery should help open up further avenues of discussions between the various stakeholders in the pharma space. In addition, the expansion of healthcare coverage in developing nations along with the upward economic and social mobility will help propel more value-conscious pharma pricing decisions worldwide. The pharma landscape is now shifting to a more stringent cost-benefit analysis of drugs, thereby setting the stage for affordable innovation to make the value leap from developing to developed markets.
BUSINESS STRATEGY AND
OPERATIONAL PERFORMANCE
The year gone by
The shifting regulatory and business landscape in FY14 made for some interesting times for Biocon. While on the one hand we witnessed sustained momentum in Research Services, on the other we saw changing market dynamics impacting the Biopharmaceuticals segment as discussed further in the note below. We also made progress on their development pipeline encompassing their biosimilar and novel portfolio. In addition, the depreciation of the rupee vis-à-vis the dollar also aided their growth momentum.
Despite the headwinds, we delivered broad based growth across all their verticals. Their diversified growth strategy focussing on the 5 verticals, helped us deliver a healthy growth of 16% this year to reach Rs.29,332 in FY14 up from Rs.25,380 in FY13. While Research Services grew at 28% YoY, Branded Formulations and Biopharma delivered growth of 13% and 15% respectively.
FIXED ASSETS
Tangible Assets
Land
Buildings
Leasehold
Improvements
Plant
and Equipment
Research
and Development Equipments
Furniture
and Fixtures
Vehicles
Intangible Assets
Intellectual
Property Rights
Computer Software
Marketing
Rights
PRESS RELEASE
KIRAN
MAZUMDAR-SHAW AWARDED THE ‘2014 GLOBAL ECONOMY PRIZE’ BY THE KIEL INSTITUTE FOR
THE WORLD ECONOMY
Bangalore, India,
June 23, 2014
Ms Kiran Mazumdar-Shaw, Chairperson and Managing Director of Biocon Ltd, Asia’s leading Biopharmaceutical Company, has been awarded the Kiel Institute’s most coveted ‘2014 Global Economy Prize’ for Business during the 100th anniversary celebrations of the Institute held in Kiel, Germany on Sunday, June 22.
The Kiel Institute for the World Economy is an international center for research in global economic affairs, economic policy consulting and economic education. This prestigious award, established in 2005 by the Kiel Institute, is bestowed upon individuals who have been pioneers in finding solutions to global economic problems
by strongly influencing and implementing economic or trade systems based on individual initiative and responsibility.
This annual prize is awarded to three individuals: A high ranking policy maker, a renowned economist and an outstanding entrepreneur. This year’s winners are:
Politics: H.E. Madam Ellen Johnson-Sirleaf, President of Liberia
Economic Sciences: Prof Richard H. Thaler, the University of Chicago
Business: Ms Kiran Mazumdar-Shaw, Chairperson & Managing Director, Biocon, India
Sigmar Gabriel, German Vice Chancellor and Minister for Economic affairs, delivered a keynote address at the ceremony, which was attended by distinguished personalities from politics, academia, business and civil society.
Ms Mazumdar-Shaw is the first Indian woman and the fourth Indian to be conferred this prize. Previous honorees include Amartya Sen, Nobel laureate in Economics (2007); Baba N. Kalyani, Chairman of the Kalyani Group (2009); and Sunil Bharti Mittal, Chairman of the Bharti Group (2009).
Accepting the award from Prof. Dennis Snower, President, Kiel Institute for the World Economy, Ms Mazumdar-Shaw, CMD, Biocon, said, “I am honored to receive this prestigious award and consider it a great privilege to be in the company of great economists, world leaders and entrepreneurs. I thank the Kiel Institute for awarding me with ’The Global Economy Prize 2014’. ”
Ms Mazumdar-Shaw pioneered biotechnology in India and started Biocon as a novel enzymes company, which has evolved into India’s largest biopharmaceutical enterprise committed to affordable innovation. Biocon has the largest scientific talent pool in India, and is engaged in path-breaking research to develop novel and differentiated biopharmaceuticals aimed at reducing the cost of treatment for cancer, diabetes and autoimmune disorders for patients worldwide.
With this award, Ms Mazumdar-Shaw joins an elite list that includes Gro Harlem Brundtland, Norway’s former Prime Minister; Mary Robinson, former President of Ireland; Joseph E. Stiglitz, Nobel laureate in Economics; Paul R. Krugman, Nobel laureate in Economics; Pascal Lamy, former Director General of the World Trade Organization; Jean-Claude Trichet, former President of the European Central Bank; and Dietmar Hopp, Founder of SAP.
About the Global
Economy Prize:
The Kiel Institute Global Economy Prize is awarded to honor persons who have been pioneers in finding solutions to global economic problems, who have been willing to participate in a dialog with people from other walks of life, and who have championed a society based on individual initiative and responsibility. The prize is awarded to a
high-ranking policymaker, a renowned economist and an outstanding entrepreneur.
The Global Economy Prize is not endowed with prize money.
About Kiel Institute for the World Economy:
The Kiel Institute is an international centre for research in global economic affairs, economic policy consulting and economic education. The Institute engages especially in creating solutions to problems in global economic affairs. On basis of its research, the Institute advises decision takers in policy, business and society and informs the broader public about important developments in international economic policy. In 2013, it was ranked as one of the top 20 research centres in the world for international trade and one of the top four think tanks in the world for economic policy.
CMT REPORT (Corruption, Money Laundering & Terrorism]
The Public Notice information has been collected from various sources
including but not limited to: The Courts,
1] INFORMATION ON
DESIGNATED PARTY
No records exist designating subject or any of its beneficial owners,
controlling shareholders or senior officers as terrorist or terrorist
organization or whom notice had been received that all financial transactions
involving their assets have been blocked or convicted, found guilty or against
whom a judgement or order had been entered in a proceedings for violating
money-laundering, anti-corruption or bribery or international economic or
anti-terrorism sanction laws or whose assets were seized, blocked, frozen or
ordered forfeited for violation of money laundering or international
anti-terrorism laws.
2] Court Declaration :
No records exist to suggest that subject is
or was the subject of any formal or informal allegations, prosecutions or other
official proceeding for making any prohibited payments or other improper
payments to government officials for engaging in prohibited transactions or
with designated parties.
3] Asset Declaration :
No records exist to suggest that the property or assets of the subject
are derived from criminal conduct or a prohibited transaction.
4] Record on Financial
Crime :
Charges or conviction
registered against subject: None
5] Records on Violation of
Anti-Corruption Laws :
Charges or
investigation registered against subject: None
6] Records on Int’l
Anti-Money Laundering Laws/Standards :
Charges or
investigation registered against subject: None
7] Criminal Records
No available
information exist that suggest that subject or any of its principals have been
formally charged or convicted by a competent governmental authority for any
financial crime or under any formal investigation by a competent government
authority for any violation of anti-corruption laws or international anti-money
laundering laws or standard.
8] Affiliation with
Government :
No record
exists to suggest that any director or indirect owners, controlling
shareholders, director, officer or employee of the company is a government
official or a family member or close business associate of a Government
official.
9] Compensation Package :
Our market
survey revealed that the amount of compensation sought by the subject is fair
and reasonable and comparable to compensation paid to others for similar
services.
10] Press Report :
No press reports / filings exists on
the subject.
CORPORATE GOVERNANCE
MIRA INFORM as part of its Due Diligence do provide comments on Corporate
Governance to identify management and governance. These factors often have been
predictive and in some cases have created vulnerabilities to credit
deterioration.
Our Governance Assessment focuses principally on the interactions
between a company’s management, its Board of Directors, Shareholders and other
financial stakeholders.
CONTRAVENTION
Subject is not known to have contravened any existing local laws,
regulations or policies that prohibit, restrict or otherwise affect the terms
and conditions that could be included in the agreement with the subject.
FOREIGN EXCHANGE RATES
|
Currency |
Unit
|
Indian Rupees |
|
US Dollar |
1 |
Rs.61.43 |
|
|
1 |
Rs.97.73 |
|
Euro |
1 |
Rs.77.93 |
INFORMATION DETAILS
|
Information Gathered
by : |
PRT |
|
|
|
|
Analysis Done by
: |
KAR |
|
|
|
|
Report Prepared
by : |
JAY |
SCORE & RATING EXPLANATIONS
|
SCORE FACTORS |
RANGE |
POINTS |
|
HISTORY |
1~10 |
8 |
|
PAID-UP CAPITAL |
1~10 |
8 |
|
OPERATING SCALE |
1~10 |
8 |
|
FINANCIAL CONDITION |
|
|
|
--BUSINESS SCALE |
1~10 |
8 |
|
--PROFITABILIRY |
1~10 |
8 |
|
--LIQUIDITY |
1~10 |
8 |
|
--LEVERAGE |
1~10 |
8 |
|
--RESERVES |
1~10 |
8 |
|
--CREDIT LINES |
1~10 |
8 |
|
--MARGINS |
-5~5 |
-- |
|
DEMERIT POINTS |
|
|
|
--BANK CHARGES |
YES/NO |
YES |
|
--LITIGATION |
YES/NO |
YES |
|
--OTHER ADVERSE INFORMATION |
YES/NO |
NO |
|
MERIT POINTS |
|
|
|
--SOLE DISTRIBUTORSHIP |
YES/NO |
NO |
|
--EXPORT ACTIVITIES |
YES/NO |
NO |
|
--AFFILIATION |
YES/NO |
YES |
|
--LISTED |
YES/NO |
YES |
|
--OTHER MERIT FACTORS |
YES/NO |
YES |
|
DEFAULTER |
|
|
|
--RBI |
YES/NO |
NO |
|
--EPF |
YES/NO |
NO |
|
TOTAL |
|
72 |
This score serves as a reference to assess SC’s credit risk and
to set the amount of credit to be extended. It is calculated from a composite
of weighted scores obtained from each of the major sections of this report. The
assessed factors and their relative weights (as indicated through %) are as
follows:
Financial
condition (40%) Ownership
background (20%) Payment
record (10%)
Credit history
(10%) Market trend
(10%) Operational
size (10%)
RATING EXPLANATIONS
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
>86 |
Aaa |
Possesses an extremely sound financial base with the strongest
capability for timely payment of interest and principal sums |
Unlimited |
|
71-85 |
Aa |
Possesses adequate working capital. No caution needed for credit
transaction. It has above average (strong) capability for payment of interest
and principal sums |
Large |
|
56-70 |
A |
Financial & operational base are regarded healthy. General
unfavourable factors will not cause fatal effect. Satisfactory capability for
payment of interest and principal sums |
Fairly Large |
|
41-55 |
Ba |
Overall operation is considered normal. Capable to meet normal
commitments. |
Satisfactory |
|
26-40 |
B |
Capability to overcome financial difficulties seems comparatively below
average. |
Small |
|
11-25 |
Ca |
Adverse factors are apparent. Repayment of interest and principal sums
in default or expected to be in default upon maturity |
Limited with
full security |
|
<10 |
C |
Absolute credit risk exists. Caution needed to be exercised |
Credit not
recommended |
|
-- |
NB |
New Business |
-- |
This report is issued at your request without any
risk and responsibility on the part of MIRA INFORM PRIVATE LIMITED (MIPL)
or its officials.