MIRA INFORM REPORT
|
Report No. : |
318506 |
|
Report Date : |
22.04.2015 |
IDENTIFICATION DETAILS
|
Name : |
SUN PHARMACEUTICAL INDUSTRIES LIMITED |
|
|
|
|
Registered
Office : |
Sparc Tandalja, Vadodara – 390020, Gujarat |
|
Contact No.: |
91-265-6615500/ 600/ 700 |
|
|
|
|
Country : |
India |
|
|
|
|
Financials (as
on) : |
31.03.2014 |
|
|
|
|
Date of
Incorporation : |
01.03.1993 |
|
|
|
|
Com. Reg. No.: |
04-019050 |
|
|
|
|
Capital
Investment / Paid-up Capital : |
Rs. 2071.200 Million |
|
|
|
|
CIN No.: [Company Identification
No.] |
L24230GJ1993PLC019050 |
|
|
|
|
IEC No.: |
Not Available |
|
|
|
|
TAN No.: [Tax Deduction &
Collection Account No.] |
Not Available |
|
|
|
|
PAN No.: [Permanent Account No.] |
Not Available |
|
|
|
|
Legal Form : |
A Public Limited Liability Company. The Company’s Shares are Listed on
the Stock Exchanges. |
|
|
|
|
Line of Business
: |
Manufacturing, Selling, Trading, Marketing and Exporting
of various Pharmaceutical Products. |
|
|
|
|
No. of Employees
: |
14000 (Approximately) |
RATING & COMMENTS
|
MIRA’s Rating : |
A (65) |
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
56-70 |
A |
Financial & operational base are regarded healthy. General unfavourable
factors will not cause fatal effect. Satisfactory capability for payment of
interest and principal sums |
Fairly Large |
|
Maximum Credit Limit : |
USD 210000000 |
|
|
|
|
Status : |
Good |
|
|
|
|
Payment Behaviour : |
Regular |
|
|
|
|
Litigation : |
Not Available |
|
|
|
|
Comments : |
Subject is a well-established and reputed company having fine track
record. The company has incurred a heavy loss during F.Y.2014. However, general
financial of the company is sound and healthy. Fundamentals of the company
are strong. Share price are quoted high on stock exchange. Directors are reported to be experienced and respectable businessmen. Trade relations are reported as fair. Business is active. Payments are
reported to be regular and as per commitment. The company can be considered good for normal business dealings at
usual trade terms and conditions. |
NOTES:
Any query related to this report can be made
on e-mail : infodept@mirainform.com
while quoting report number, name and date.
ECGC Country Risk Classification List – December 31, 2014
|
Country Name |
Previous Rating (30.09.2014) |
Current Rating (31.12.2014) |
|
India |
A1 |
A1 |
|
Risk Category |
ECGC
Classification |
|
Insignificant |
A1 |
|
Low |
A2 |
|
Moderate |
B1 |
|
High |
B2 |
|
Very High |
C1 |
|
Restricted |
C2 |
|
Off-credit |
D |
EXTERNAL AGENCY RATING
|
Rating Agency Name |
CRISIL |
|
Rating |
Long-term rating = AAA |
|
Rating Explanation |
Highest degree of safety and carry lowest
credit risk. |
|
Date |
April 08, 2014 |
|
Rating Agency Name |
CRISIL |
|
Rating |
Short-term rating = A1+ |
|
Rating Explanation |
Very strong degree of safety and carry
lowest credit risk. |
|
Date |
April 08, 2014 |
RBI DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available RBI Defaulters’ list.
EPF (Employee Provident Fund) DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available EPF (Employee Provident Fund) Defaulters’ list as of
31-03-2014
INFORMATION PARTED BY
|
Name : |
Mr. Dhrunal Trivedi |
|
Designation : |
Export Logistical Manager |
|
Contact No.: |
91-22-66969696 |
|
Date : |
01.04.2015 |
LOCATIONS
|
Registered Office : |
Sparc Tandalja, Vadodara – 390020, Gujarat, India |
|
Tel. No.: |
91-265-6615500/ 600/ 700 |
|
Fax No.: |
91-265-2354897 |
|
E-Mail : |
|
|
Website : |
|
|
Area : |
10000 sq. ft. |
|
Location : |
Owned |
|
|
|
|
Corporate Office 1: |
Acme Plaza, Andheri-Kurla Road, Andheri (East), Mumbai – 400059,
Maharashtra, India |
|
Tel. No.: |
91-22-66969696 91-22-66969696 |
|
Fax No.: |
91-22-28212010 |
|
|
|
|
Corporate Office 2: |
Sun House, CTS No. 201 B/1, Western Express Highway, Goregaon (East), Mumbai 400063, Maharashtra, India |
|
Tel. No.: |
91-22-43244324 |
|
Fax No.: |
91-22-43244343 |
|
|
|
|
Research Centre 2 : |
F.P.27, Part Survey No. 27, C.S. No. 1050, TPS No. 24, Village Tandalja,
District Vadodara - 390020, Gujarat, India |
|
|
|
|
Research Centre 3 : |
17-B, Mahal Industrial Estate, Mahakali Caves Road, Andheri (East),
Mumbai - 400093, Maharashtra, India |
|
|
|
|
Research Centre 4 : |
Chemistry and Discovery Research Israel, 14 Hakitor Street, P.O. Box
10347 Haifa Bay 2624761, Israel |
|
|
|
|
Plants : |
·
Survey No. 214, Plot No. 20, Govt. Industrial
Area, Phase II, Piparia. Silvassa - 396230, Union Territory of Dadra and Nagar
Haveli, India ·
Halol-Baroda Highway, Near Anand Kendra, Halol,
District Panchmahal - 389350, Gujarat, India ·
Plot No. 24/2 & 25, GIDC, Phase-IV, Panoli,
Dist. Bharuch - 394116, Gujarat, India
·
A-7 & A-8, MIDC Ind. Area, Ahmednagar -
414111, Maharashtra, India ·
Plot No. 4708, GIDC. Ankleshwar - 393002,
Gujarat, India ·
Sathammai Village, Karunkuzhi Post, Maduranthakam
TK, Kanchipuram District - 603303, Tamilnadu, India ·
Plot No. 817/A, Karkhadi, Taluka Padra, District
Vadodara - 391450, Gujarat, India ·
Survey No. 259/15, Dadra - 396191, Union
Territory of Dadra and Nagar Haveli, India ·
Sun Pharma Laboratories Limited, Plot No.754,
Nandok Block, Setipool, P.O. Ranipool, Sikkim – 737135, India ·
Sun Pharma Laboratories Limited, Plot No. 107/108,
Namli Block, Setipool, P.O. Ranipool, Sikkim – 737135, India ·
Sun Pharma Laboratories Limited, 6-9 Export
Promotion Industrial Park (EPIP), Kartholi, Bari Brahmana, Jammu - 181133,
Jammu Kashmir, India ·
Sun Pharma Laboratories Limited, I.G.C. Phase-I,
Samba 184121, Jammu Kashmir, India ·
Sun Pharmaceutical Industries Inc., 705, E.
Mulberry Street, Bryan, Ohio – 43506, USA ·
Sun Pharmaceutical Industries Inc., 270 Prospect
Plains Road, Cranbury, New Jersey – 08512, USA ·
Sun Pharmaceutical (Bangladesh) Limited,
Chandana, Joydevpur, Gazipur, Bangladesh ·
Alkaloida Chemical Company Zrt, H-4440
Tiszavasvari , Kabay, Janos u.29, Hungary ·
TKS Farmaceutica, Rodovia GO-080, Km 02, Chacaras
01/02, Jardim Pompeia, Goiania/GO, Brazil CEP: 74690-170 ·
Sun Pharma de Mexico S.A. de C.V, Av. Rio
Churubusco No. 658, Col. El Sifon, Del. Iztapalapa, C.P 09400 Mexico,
Distrito Federal ·
Chattem Chemicals, Inc., 3708, St. Elmo Avenue,
Chattanooga, TN 37409, USA ·
Taro Pharmaceuticals Inc., 130 East Drive,
Brampton, Ontario L6T 1C1, Canada ·
Taro Pharmaceutical Industries Limited, 14
Hakitor Street, P.O. Box 10347 Haifa Bay 2624761, Israel ·
Dusa Pharmaceuticals Inc., 25, Upton Drive,
Wilmington, Massachusetts, 01887, USA ·
URL Pharmaceuticals Inc., 1100 Orthodox Street, Philadelphia,
PA 19124, USA ·
URL Pharmaceuticals Inc., 2500 Molitor Road,
Aurora, IL 60502, USA |
DIRECTORS
AS ON 31.03.2014
|
Name : |
Mr. Israel Makov |
|
Designation : |
Chairman |
|
|
|
|
Name : |
Mr. Dilip S. Shanghvi |
|
Designation : |
Managing Director |
|
Date of Birth/Age : |
58 Years |
|
Qualification : |
Graduate in commerce from the Kolkata University |
|
Profile : |
He had launched Sun Pharmaceutical Industries in 1982. He is
the Managing Director of the Company and Chairman and Managing Director of
Sun Pharma Advanced Research Company Limited. He has also been appointed as
the Chairman and Director of Taro Pharmaceutical Industries Limited. He has
extensive experience in the pharmaceutical industry. Under his leadership,
Sun Pharmaceutical Industries Limited has recorded an all-round growth in
business. |
|
|
|
|
Name : |
Mr. Sudhir V. Valia |
|
Designation : |
Whole-time Director |
|
Date of Birth/Age : |
58 Years |
|
Qualification : |
Chartered Accountant with more than three decades of
experience in Finance and Taxation |
|
Profile : |
He is representing the Board since 31st January, 1994 and
at present, he is Whole-time Director of the Company. |
|
|
|
|
Name : |
Mr. Sailesh T. Desai |
|
Designation : |
Whole-time Director |
|
Date of Birth/Age : |
60 Years |
|
Qualification : |
Science graduate from Kolkata University |
|
Profile : |
He is a successful entrepreneur with more than three decades
of wide industrial experience including more than two decades in the
pharmaceutical industry itself. |
|
|
|
|
Name : |
Mr. S. Mohanchand Dadha |
|
Designation : |
Director |
|
|
|
|
Name : |
Mr. Hasmukh S. Shah |
|
Designation : |
Director |
|
|
|
|
Name : |
Mr. Ashwin Dani |
|
Designation : |
Director |
|
|
|
|
Name : |
Mr. Keki M. Mistry |
|
Designation : |
Director |
|
|
|
|
Name : |
Ms. Rekha Sethi |
|
Designation : |
Director |
|
Date of Appointment : |
13.02.2014 |
KEY EXECUTIVES
|
Name : |
Mr. Sunil R. Ajmera |
|
Designation : |
Company Secretary |
|
|
|
|
Name : |
Mr. Uday V. Baldota |
|
Designation : |
Chief Financial Officer |
|
|
|
|
Name : |
Mr. Dhrunal Trivedi |
|
Designation : |
Export Logistical Manager |
MAJOR SHAREHOLDERS / SHAREHOLDING PATTERN
AS ON 31.12.2014
|
(A) Shareholding of Promoter
and Promoter Group |
||
|
|
|
|
|
|
302192200 |
12.56 |
|
|
1013024000 |
42.10 |
|
|
1280200 |
0.05 |
|
|
1280200 |
0.05 |
|
|
1316496400 |
54.71 |
|
|
|
|
|
Total shareholding of Promoter
and Promoter Group (A) |
1316496400 |
54.71 |
|
(B) Public Shareholding |
||
|
|
|
|
|
|
28274791 |
1.18 |
|
|
53667321 |
2.23 |
|
|
1945763 |
0.08 |
|
|
40448219 |
1.68 |
|
|
452764676 |
18.82 |
|
|
32846 |
0.00 |
|
|
32846 |
0.00 |
|
|
577133616 |
23.99 |
|
|
|
|
|
|
64546340 |
2.68 |
|
|
|
|
|
|
108364713 |
4.50 |
|
|
31011743 |
1.29 |
|
|
308567862 |
12.82 |
|
|
4198032 |
0.17 |
|
|
921846 |
0.04 |
|
|
2524989 |
0.10 |
|
|
3817386 |
0.16 |
|
|
79927985 |
3.32 |
|
|
59440 |
0.00 |
|
|
13842 |
0.00 |
|
|
217104342 |
9.02 |
|
|
512490658 |
21.30 |
|
Total Public shareholding (B) |
1089624274 |
45.29 |
|
Total (A)+(B) |
2406120674 |
100.00 |
|
(C) Shares held by Custodians
and against which Depository Receipts have been issued |
0 |
0.00 |
|
|
0 |
0.00 |
|
|
0 |
0.00 |
|
|
0 |
0.00 |
|
Total (A)+(B)+(C) |
2406120674 |
100.00 |

BUSINESS DETAILS
|
Line of Business : |
Manufacturing, Selling, Trading, Marketing and Exporting
of various Pharmaceutical Products. |
|
|
|
|
Brand Names : |
· Pantocid · Gemer · Susten · Levipil · Pantocid-D · Aztor · Glucored Group · Istamet · Rozavel ·
Montek-LC |
|
|
|
|
Agencies Held : |
Not Available |
|
|
|
|
Exports : |
Not Divulged |
|
|
|
|
Imports : |
Not Divulged |
|
|
|
|
Terms : |
Not Divulged |
GENERAL INFORMATION
|
Suppliers : |
|
||||||||||||||||||||||
|
|
|
||||||||||||||||||||||
|
Customers : |
|
||||||||||||||||||||||
|
|
|
||||||||||||||||||||||
|
No. of Employees : |
14000 (Approximately) |
||||||||||||||||||||||
|
|
|
||||||||||||||||||||||
|
Bankers : |
·
Bank of Baroda ·
Bank of Nova Scotia ·
Citibank N.A. ·
ICICI Bank Limited ·
Kotak Mahindra Bank Limited ·
Standard Chartered Bank ·
State Bank of India |
||||||||||||||||||||||
|
|
|
||||||||||||||||||||||
|
Facilities : |
|
||||||||||||||||||||||
|
Auditors : |
|
|
Name : |
Deloitte Haskins and Sells Chartered Accountants |
|
Address : |
Mumbai, Maharashtra, India |
|
|
|
|
Subsidiaries : |
· 3 Skyline LLC · Aditya Acquisition Company Limited · Aditya Pharma Private Limited · Alkaloida Chemical Company Zrt. · Alkaloida Sweden AB · AR Scientific Inc · Caraco Pharma Inc · Caraco Pharmaceutical Laboratories Limited · Caraco Pharmaceuticals Private Limited · Chattem Chemicals lnc · Dungan Mutual Associates, LLC · Dusa Pharmaceuticals lnc · Dusa Pharmaceuticals New York lnc · Faststone Mercantile Company Private Limited · Green Eco Development Centre Limited · Khyati Realty ME Limited · Morley and Company lnc · Mutual Pharmaceutical Company, lnc. · Neetnav Real Estate Private Limited · ** Nogad Holdings · One Commerce Drive LLC · OOO "Sun Pharmaceutical Industries" Limited · Orta Limited · Realstone Multitrade Private Limited · Sirius Laboratories lnc · Skisen Labs Private Limited · Softdeal Trading Company Private Limited · SPIL De Mexico S.A. DE C.V. · Sun Clobal Canada Pty. Limited · Sun Global Development FZE · Sun Laboratories FZE · Sun Pharma De Mexico 5.A. DE C.V. · Sun Pharma De Venezuela, CA. · Sun Pharma Global (FZE) · Sun Pharma Global lnc · Sun Pharma Healthcare FZE · * Sun Pharma Medication Private Limited (SPDPL) · * Sun Pharma Drugs Private Limited (SPMPL) · Sun Pharma Japan Limited · Sun Pharma Laboratories Limited · Sun Pharma MEAJLT · Sun Rharma Philippines Inc · ** Sun Pharma Switzerland · Sun Pharmaceutical Industries (Europe) B.V · Sun Pharmaceutical (Bangladesh) Limited · Sun Pharmaceutical Industries (Australia) Pty. Limited · Sun Pharmaceutical Peru S.A.C. · Sun Pharmaceutical Spain, S.L.U · Sun Pharmaceutical UK Limited · Sun Pharmaceuticals (SA) (Pty) Limited · Sun Pharmaceuticals France · Sun Pharmaceuticals Germany GmbH · Sun Pharmaceuticals Italia S.R.L. · ** Silverstreet Developers LLP · Sun Pharmaceuticals Korea Limited · Sun Universal Limited · Taro Development Corporation · Taro Hungary Intellectual Property Licensing LLC. · Taro International Limited · Taro Pharmaceutical India Private Limited · Taro Pharmaceutical Industries Limited · Taro Pharmaceutical Laboratories INC · Taro Pharmaceuticals Canada, Limited · Taro Pharmaceuticals Europe B.V. · Taro Pharmaceuticals lnc. · Taro Pharmaceuticals Ireland Limited · Taro Pharmaceuticals North America, lnc · Taro Pharmaceuticals U.S.A., lnc · Taro Pharmaceuticals UK Limited · Tarochem Limited · TKS Farmaceutica Limiteda · United Research Laboratories, Limited · Universal Enterprises Private Limited (w.e.f.31st August, 2012) · URL Pharma Inc · URL PharmPro, LLC · ZAO Sun Pharma Industries Limited [Liquidated during the year] |
|
|
|
|
Controlled Entity : |
· * Sun Pharmaceutical Industries · * Sun Pharma Sikkim · Sun Pharma Drugs ·
Universal Enterprise Private Limited [upto
31.08.2012] |
|
|
|
|
Enterprise under significant influence of
Key Management Personnel or their relatives : |
· Sun Petrochemicals Private Limited · Navjivan Rasayan (Gujarat) Private Limited · Sun Pharma Advanced Research Company Limited |
NOTE:
** Controlled entities converted into private
limited companies under Part IX of the Companies Act, 1956 w.e.f. 31.08.2012,
Amalgamated in to Sun Pharma Laboratories Limited w.e.f. 01.09.2012.
* Incorporated / Acquired during the year
CAPITAL STRUCTURE
AS ON 31.03.2014
Authorised Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
3000000000 |
Equity Shares |
Re. 1/- each |
Rs. 3000.000 Million |
|
|
|
|
|
Issued, Subscribed & Paid-up Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
2071163910 |
Equity Shares |
Re. 1/- each |
Rs. 2071.200 Million |
|
|
|
|
|
FINANCIAL DATA
[all figures are
in Rupees Million]
ABRIDGED BALANCE
SHEET
|
SOURCES OF FUNDS |
31.03.2014 |
31.03.2013 |
31.03.2012 |
|
I.
EQUITY
AND LIABILITIES |
|
|
|
|
(1)Shareholders'
Funds |
|
|
|
|
(a) Share Capital |
2071.200 |
1035.600 |
1035.600 |
|
(b) Reserves & Surplus |
72007.600 |
76853.200 |
77745.600 |
|
(c) Money
received against share warrants |
0.000 |
0.000 |
0.000 |
|
|
|
|
|
|
(2) Share Application money pending allotment |
0.000 |
0.000 |
0.000 |
|
Total
Shareholders’ Funds (1) + (2) |
74078.800 |
77888.800 |
78781.200 |
|
|
|
|
|
|
(3) Non-Current
Liabilities |
|
|
|
|
(a) Long-term borrowings |
46.400 |
46.400 |
0.000 |
|
(b) Deferred tax liabilities (Net) |
1928.400 |
1654.100 |
1339.100 |
|
(c) Other long term
liabilities |
13.800 |
10.100 |
20.300 |
|
(d) long-term
provisions |
25241.900 |
1314.900 |
986.200 |
|
Total Non-current
Liabilities (3) |
27230.500 |
3025.500 |
2345.600 |
|
|
|
|
|
|
(4) Current Liabilities |
|
|
|
|
(a) Short term
borrowings |
24043.700 |
384.900 |
403.000 |
|
(b) Trade payables |
3800.600 |
3625.100 |
4001.900 |
|
(c) Other current
liabilities |
2336.100 |
1451.500 |
625.400 |
|
(d) Short-term
provisions |
6880.300 |
6089.100 |
5154.900 |
|
Total Current
Liabilities (4) |
37060.700 |
11550.600 |
10185.200 |
|
|
|
|
|
|
TOTAL |
138370.000 |
92464.900 |
91312.000 |
|
|
|
|
|
|
II.
ASSETS |
|
|
|
|
(1) Non-current assets |
|
|
|
|
(a) Fixed Assets |
|
|
|
|
(i) Tangible assets |
12575.600 |
11347.600 |
9759.300 |
|
(ii) Intangible Assets |
96.100 |
10.300 |
13.700 |
|
(iii) Capital
work-in-progress |
4804.600 |
3480.400 |
2488.700 |
|
(iv)
Intangible assets under development |
0.000 |
0.000 |
0.000 |
|
(b) Non-current Investments |
61557.300 |
33764.900 |
35928.000 |
|
(c) Deferred tax assets (net) |
0.000 |
0.000 |
0.000 |
|
(d) Long-term Loan and Advances |
7614.900 |
4910.900 |
3385.400 |
|
(e) Other
Non-current assets |
1.100 |
78.300 |
28.300 |
|
Total Non-Current
Assets |
86649.600 |
53592.400 |
51603.400 |
|
|
|
|
|
|
(2) Current assets |
|
|
|
|
(a) Current investments |
8600.000 |
9410.500 |
8449.800 |
|
(b) Inventories |
9183.800 |
8687.600 |
6400.700 |
|
(c) Trade receivables |
4527.500 |
7375.300 |
7134.800 |
|
(d) Cash and cash
equivalents |
1414.800 |
4311.200 |
13277.100 |
|
(e) Short-term loans
and advances |
3093.200 |
8495.000 |
3906.100 |
|
(f) Other current
assets |
24901.100 |
592.900 |
540.100 |
|
Total Current Assets |
51720.400 |
38872.500 |
39708.600 |
|
|
|
|
|
|
TOTAL |
138370.000 |
92464.900 |
91312.000 |
PROFIT & LOSS
ACCOUNT
|
|
PARTICULARS |
31.03.2014 |
31.03.2013 |
31.03.2012 |
|
|
|
SALES |
|
|
|
|
|
|
|
Revenue from Operations |
28287.900 |
24321.400 |
40155.600 |
|
|
|
Other Income |
1593.800 |
2361.700 |
3428.500 |
|
|
|
TOTAL |
29881.700 |
26683.100 |
43584.100 |
|
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
|
|
Cost of Materials Consumed |
8946.200 |
7057.900 |
9517.100 |
|
|
|
Purchases of Stock-in-Trade |
1850.400 |
2009.600 |
1874.800 |
|
|
|
Changes in inventories of finished goods, work-in-progress
and Stock-in-Trade |
(145.300) |
(105.300) |
(817.300) |
|
|
|
Employees benefits expense |
2796.300 |
2348.700 |
3165.600 |
|
|
|
Other expenses |
14669.600 |
7883.700 |
8829.200 |
|
|
|
Exception Item |
28756.000 |
0.000 |
2999.200 |
|
|
|
TOTAL |
56873.200 |
19194.600 |
25568.600 |
|
|
|
|
|
|
|
|
|
PROFIT/
(LOSS) BEFORE TAX, DEPRECIATION AND AMORTISATION |
(26991.500) |
7488.500 |
18015.500 |
|
|
|
|
|
|
|
|
|
Less/ Add |
DEPRECIATION/
AMORTISATION |
1019.400 |
858.200 |
757.200 |
|
|
|
|
|
|
|
|
|
|
PROFIT/ (LOSS)
BEFORE TAX |
(28010.900) |
6630.300 |
17258.300 |
|
|
|
|
|
|
|
|
|
Less |
TAX |
274.300 |
1464.800 |
283.400 |
|
|
|
|
|
|
|
|
|
|
PROFIT/ (LOSS)
AFTER TAX |
(28285.200) |
5165.500 |
16974.900 |
|
|
|
|
|
|
|
|
|
|
EARNINGS IN
FOREIGN CURRENCY |
|
|
|
|
|
|
|
Exports (FOB basis) |
22713.300 |
19238.000 |
14628.200 |
|
|
|
Interest |
0.000 |
4.100 |
0.000 |
|
|
|
Royalty |
50.300 |
3.300 |
3.000 |
|
|
|
Others |
404.400 |
369.800 |
14.700 |
|
|
TOTAL EARNINGS |
23168.000 |
19615.200 |
14645.900 |
|
|
|
|
|
|
|
|
|
|
Earnings/ (Loss)
Per Share (Rs.) |
(13.70) |
2.50 |
16.40 |
|
QUARTERLY RESULTS
|
Particulars
(Rs.In Million) |
30.06.2014 |
30.09.2014 |
31.12.2014 |
|
UnAudited |
Unaudited |
Unaudited |
Unaudited |
|
Net Sales |
(5755.800) |
(5974.500) |
5650.300 |
|
Total Expenditure |
(5456.700) |
(8489.600) |
6124.300 |
|
PBIDT (Excl OI) |
299.100 |
(2515.100) |
(474.000) |
|
Other Income |
842.100 |
0.000 |
0.000 |
|
Operating Profit |
1141.200 |
(2515.100) |
(474.000) |
|
Interest |
80.900 |
(40.200) |
(0.800) |
|
Exceptional Items |
0.000 |
0.000 |
0.000 |
|
PBDT |
1060.300 |
(2555.300) |
(474.800) |
|
Depreciation |
463.900 |
(675.700) |
(408.600) |
|
Profit Before Tax |
596.400 |
(3231.000) |
(883.400) |
|
Tax |
120.800 |
(54.800) |
(27.600) |
|
Provisions and contingencies |
0.000 |
0.000 |
0.000 |
|
Profit After Tax |
475.600 |
(3176.200) |
(911.000) |
|
Extraordinary Items |
0.000 |
0.000 |
0.000 |
|
Prior Period Expenses |
0.000 |
0.000 |
0.000 |
|
Other Adjustments |
0.000 |
0.000 |
0.000 |
|
Net Profit |
475.600 |
(3176.200) |
(911.000) |
KEY RATIOS
|
PARTICULARS |
|
31.03.2014 |
31.03.2013 |
31.03.2012 |
|
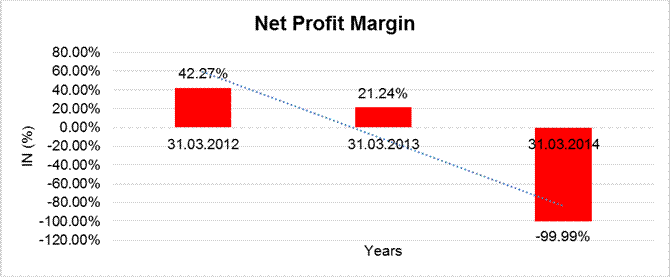
Net Profit Margin (PAT/Sales) |
(%) |
(99.99) |
21.24 |
42.27 |
|
|
|
|
|
|
|
Return on Total Assets (PBT/Total Assets} |
(%) |
(38.90) |
12.01 |
32.63 |
|
|
|
|
|
|
|
Return on Investment (ROI) (PBT/Networth) |
|
(0.38) |
0.09 |
0.22 |
|
|
|
|
|
|
|
Debt Equity Ratio (Total Debt/Networth) |
|
0.33 |
0.01 |
0.01 |
|
|
|
|
|
|
|
Current Ratio (Current Asset/Current Liability) |
|
1.40 |
3.37 |
3.90 |
FINANCIAL ANALYSIS
[all figures are
in Rupees Million]
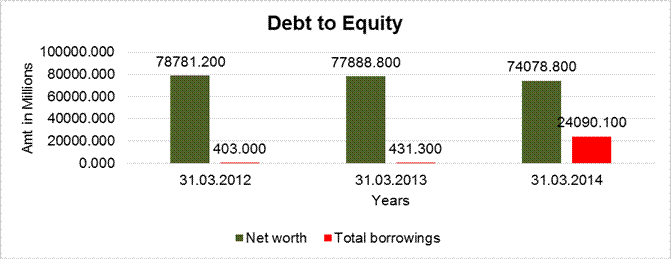
DEBT EQUITY RATIO
|
Particulars |
31.03.2012 |
31.03.2013 |
31.03.2014 |
|
|
(Rs.
In Million) |
(Rs.
In Million) |
(Rs.
In Million) |
|
Share Capital |
1035.600 |
1035.600 |
2071.200 |
|
Reserves & Surplus |
77745.600 |
76853.200 |
72007.600 |
|
Net
worth |
78781.200 |
77888.800 |
74078.800 |
|
|
|
|
|
|
Long-term borrowings |
0.000 |
46.400 |
46.400 |
|
Short term borrowings |
403.000 |
384.900 |
24043.700 |
|
Total
borrowings |
403.000 |
431.300 |
24090.100 |
|
Debt/Equity ratio |
0.005 |
0.006 |
0.325 |
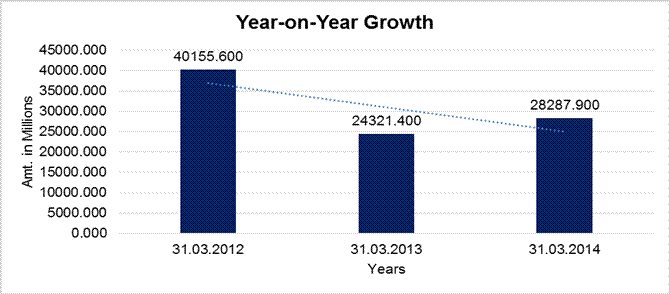
YEAR-ON-YEAR GROWTH
|
Year on Year Growth |
31.03.2012 |
31.03.2013 |
31.03.2014 |
|
|
(Rs.
In Million) |
(Rs.
In Million) |
(Rs.
In Million) |
|
Sales |
40155.600 |
24321.400 |
28287.900 |
|
|
|
-39.432 |
16.309 |
NET PROFIT MARGIN
|
Net Profit Margin |
31.03.2012 |
31.03.2013 |
31.03.2014 |
|
|
(Rs.
In Million) |
(Rs.
In Million) |
(Rs.
In Million) |
|
Sales |
40155.600 |
24321.400 |
28287.900 |
|
Profit/ (Loss) |
16974.900 |
5165.500 |
(28285.200) |
|
|
42.27% |
21.24% |
(99.99%) |
LOCAL AGENCY FURTHER INFORMATION
|
Sr. No. |
Check List by Info Agents |
Available in
Report (Yes / No) |
|
1] |
Year of Establishment |
Yes |
|
2] |
Locality of the firm |
Yes |
|
3] |
Constitutions of the firm |
Yes |
|
4] |
Premises details |
No |
|
5] |
Type of Business |
Yes |
|
6] |
Line of Business |
Yes |
|
7] |
Promoter's background |
Yes |
|
8] |
No. of employees |
Yes |
|
9] |
Name of person contacted |
Yes |
|
10] |
Designation of contact
person |
Yes |
|
11] |
Turnover of firm for last
three years |
Yes |
|
12] |
Profitability for last
three years |
Yes |
|
13] |
Reasons for variation
<> 20% |
-- |
|
14] |
Estimation for coming
financial year |
No |
|
15] |
Capital in the business |
Yes |
|
16] |
Details of sister
concerns |
Yes |
|
17] |
Major suppliers |
No |
|
18] |
Major customers |
No |
|
19] |
Payments terms |
No |
|
20] |
Export / Import details
(if applicable) |
No |
|
21] |
Market information |
-- |
|
22] |
Litigations that the firm
/ promoter involved in |
-- |
|
23] |
Banking Details |
Yes |
|
24] |
Banking facility details |
Yes |
|
25] |
Conduct of the banking
account |
-- |
|
26] |
Buyer visit details |
-- |
|
27] |
Financials, if provided |
Yes |
|
28] |
Incorporation details, if
applicable |
Yes |
|
29] |
Last accounts filed at
ROC |
Yes |
|
30] |
Major Shareholders, if
available |
Yes |
|
31] |
Date of Birth of
Proprietor/Partner/Director, if available |
Yes |
|
32] |
PAN of Proprietor/Partner/Director,
if available |
No |
|
33] |
Voter ID No of
Proprietor/Partner/Director, if available |
No |
|
34] |
External Agency Rating,
if available |
Yes |
Note: Registered office of
the company has been shifted from Sparc Tandalja, Vadodara – 390020, Gujarat,
India to the present address but the same has not been updated in the
government registry.
INDEX OF CHARGES
|
S.No. |
Charge ID |
Date of Charge Creation/Modification |
Charge amount secured |
Charge Holder |
Address |
Service Request Number (SRN) |
|
1 |
10351563 |
31/03/2012 |
178,140,000.00 |
DEPARTMENT OF BIOTECHNOLOGY |
6-8th FLOOR, BLOCK
NO.2,, CGO COMPLEX, LODHI ROAD INDIA |
B38042354 |
|
2 |
90095637 |
27/04/2000 * |
7,500,000.00 |
NVFC FINANCE PRIVATE LTD |
1843; 6TH CROSS
20TH MAIN, J.P. NAGAR; II PHASE, |
- |
|
3 |
90095112 |
05/08/2004 * |
15,000,000.00 |
RISK CAPITAL AND TECHNOLOGY FINANCE CORPORATION LTD |
E-216; 3RD FLOOR, EAST KAILASH, NEW DELHI, Delhi - 110065, INDIA |
- |
|
4 |
90095083 |
09/07/2005 * |
32,500,000.00 |
INDUSTRIAL DEVELOPMENT BANK OF INDIA |
IDBI TOWER, COLABA,
MUMBAI, Maharashtra - 400005, |
- |
|
5 |
90098599 |
07/05/1997 |
60,000,000.00 |
ICICI BANKING CORPORATION LTD. |
LAND MARK, RACE
COURSE CIRCLE, BARODA, Gujarat - |
- |
|
6 |
90098478 |
21/09/1996 |
75,000,000.00 |
ANZ GRINDLAYS BANK LTD. |
90; M. G. ROAD, MUMBAI, Maharashtra - 400001, INDIA |
- |
|
7 |
90094937 |
28/11/1997 * |
92,000,000.00 |
INDUSTRIAL DEVELOPMENT BANK OF INDIA |
IDBI TOWER, COLABA,
MUMBAI, Maharashtra - 400005, |
- |
|
8 |
90098193 |
26/08/2013 * |
1,260,000,000.00 |
ICICI BANK LIMITED |
ICICI BANK TOWERS, BANDRA KURLA COMPLEX, BANDRA (EAST),MUMBAI, Maharashtra - 400051, INDIA |
B84811918 |
|
9 |
90101587 |
03/10/2001 * |
252,500,000.00 |
BANK OF BARODA |
ALKAPURI BRANCH;
OPP. PETROL PUMP, LAKAPURI, BARO |
- |
|
10 |
90102599 |
08/05/1998 * |
252,500,000.00 |
BANK OPF BARODA |
ALKAPURI BRANCH, ALKAPURI, Gujarat, INDIA |
- |
* Date of charge modification
UNSECURED LOANS
|
PARTICULARS |
31.03.2014 (Rs.
In Million) |
31.03.2013 (Rs.
In Million) |
|
SHORT TERM BORROWINGS |
|
|
|
Term Loan from Bank |
24002.000 |
0.000 |
|
|
|
|
|
Total |
24002.000 |
0.000 |
FINANCE
CRISIL continued to reaffirm its highest rating of "AAA/
Stable" and"A1+", for the Company's Banking Facilities throughout
the year enabling the Company to avail facilities from banks at attractive
rates. The Company does not offer any Fixed Deposit Scheme.
MANAGEMENT
DISCUSSION AND ANALYSIS REPORT
GLOBAL
PHARMACEUTICAL INDUSTRY
The market size of the global pharmaceutical industry is estimated to
reach US$ 1.2 trillion by 2017 growing at a Compound Annual Growth Rate (CAGR)
of 3-6% and the emerging markets are likely to be the key growth drivers.
Several factors like economic growth, demographic changes, transition in
community health and policy responses and focus on healthcare funding are
expected to lead to double-digit growth in the pharmerging markets.
On the other hand, economic and healthcare austerity measures and the
savings realized from the growing availability of generic drugs, following
their patent expiry, may see developed markets record low single-digit growth.
GLOBAL PHARMA
INDUSTRY – GROWTH DRIVERS
Increase in life expectancy and ageing population: Life expectancy is
expected to reach 73.7 years by 2017 from an estimated 72.6 years in 2012,
bringing more than 10% of the total global population to over the age of 65
years.
Moreover, the global population aged 60 or above has almost tripled over
the period 1950-2000 and is expected to reach nearly two billion by 2050 –
prompting the need for more medical care, a key demand driver for the
pharmaceuticals industry.
Rising income of households: It
is forecasted that the number of high-income households (annual earnings of
over US$ 25,000) will rise by about 10%, taking the count to over 500 million
by 2017 - almost over 50% of such growth will come from Asia. Rising income
will make expensive medicines affordable, providing a thrust to growth of the
pharmaceuticals industry.
Growing incidence of chronic diseases: At
present, chronic diseases, including heart disease, cancer, stroke, diabetes
and respiratory illnesses top the global health agenda, accounting for over 63%
of all deaths worldwide. Sedentary lifestyles, diet changes and rising obesity levels
are likely causes. Healthcare demand for these diseases will contribute to the
industry's growth.
Improved healthcare access reforms: With
more than one billion people lacking access to a health care system across the
world, different countries are introducing healthcare reforms, including
increases in government funding and broader insurance coverage. For example,
the US extended health insurance to more than 30 million uninsured citizens
under the Patient Protection and Affordable Care Act (PPACA or ACA), making
medicines affordable and driving the growth potential and industry outlook.
OUTLOOK
Developed markets: Patent expiries,
impact of the global economic crisis and the increasing specialist nature of new
drugs may cause a slide in the share of the developed countries (US, Europe,
Japan) in total pharmaceutical spending to 53% in 2017 from 61% in2012.
Spending on generics is estimated to rise from 16% of the total spending
in 2012 to 21%of the overall developed market spend in 2017, reaching a market
value of US$ 136-143billion by 2017.
USA: The US was the
largest pharmaceutical market globally, with a market size of US$ 328 billion
in 2012 and it is estimated to grow at a compound annual growth rate (CAGR) of
1-4% during the year 2013-2017 to reach US$ 350-380 billion by 2017. But, USA's
contribution to the global spending pie is expected to decrease from 34% in
2012 to31% by 2017. Overall growth will continue to be impacted by patent
expiries and low cost generics.
ANDA Approvals
The rate of ANDA approvals at the US FDA has remained stagnant over the
past five years, intact, it even declined in 2013, despite a slight improvement
in 2010-2012. The USFDA, is in the process of implementing the 'Generic Drug
User Fee Amendments of 2012(GDUFA)' programme, which is designed to speed
access to safe and effective generic drugs.
Patent Expiries
After hitting a peak in 2012, patent expiries in the US have normalised
to more moderate levels. Drugs going off-patent contribute to incremental
growth of the US generic market. While patent expiries are lessening, the low
share of Indian generic players in the US implies potential for future growth.
EU5*: The EU5
pharmaceutical market size was around US$ 149 billion in 2012, butEU5's
contribution to the global spending pie is likely to come down from 15% in 2012
to13% in 2017. Loss of patent protection, government's austerity measures due
to the economic crisis and restricted use of innovative launches impacted the
overall growth in these markets in the last five years. Pharmaceutical spending
growth in the EU5 is expected to be 0-3% for the period 2013-2017, in
comparison to 2.4%during 2008-2012. The market size is estimated to reach US$
140-170 billion in 2017.
Japan: Japan's
pharmaceutical market contributed around 12% of the global pie in2012. In 2010,
the Japanese government embarked on healthcare reforms, with the objective of
increasing the penetration of generic drugs in the country.
The market size is projected to reach US$ 90-120 billion by 2017.
Spending growth will be in the range of 2-5% with gradual increases, but partly
impacted by the expected gradual increase in generic volumes due to the
government's biennial price cuts. Premium pricing is expected to sustain only
for drugs delivering substantial innovation over existing therapies or for
drugs which target complex/unmet medical needs.
Pharmerging markets**: While
the emerging markets are likely to be the main growth drivers of the global
pharmaceutical industry over 2012-17, the pharmerging markets are to be the
main contributors to this growth. The size of the pharmerging markets is
expected to reach US$ 370-400 billion by 2017 from US$ 224 billion in 2012,
growing at a rate of CAGR of 10-13%. China, Brazil, and India will be the key
contributing countries, driven by factors like rising income levels, healthcare
reforms and increased access to medicines.
The pharmerging markets are forecasted to contribute around 31% to the
total market share of the industry by 2017, driven by economic growth, coupled
with changes in public health policy responses and demography. Spending on
generics in these markets is estimated to rise from 58% in 2012 to 63% of the
overall pharmerging market spend in 2017, reaching a market value of US$
233-252 billion by 2017.
ACTIVE
PHARMACEUTICAL INGREDIENTS (API)
The global API market size stood at US$ 113 billion in 2012 as against
US$ 91 billion in 2008. It is expected to grow further at a CAGR of around 8% during
2012-2017, owing to patent expiries, increase in outsourcing and demand for
potent and biogeneric APIs.
With stiff competition in the global API market, a significant
proportion of API production is outsourced to China and India - two of the
largest API markets in the world.
INDIAN
PHARMACEUTICAL SECTOR
The Indian pharmaceutical market is estimated to reach US$ 22-32 billion
by 2017compared to US$ 14 billion in 2012, establishing India as the 11th
largest market by 2017,compared to its 13th position in 2012.
Crisp
facts
· Ranked thirteenth in terms of value in the global pharmaceutical industry in2012
· Fourth largest among the pharmerging markets in terms of market size in 2012
· Projected to grow at a CAGR of 11-14% during 2013-2017
· One of the key exporters to the US and other markets -the highest number of USFDA approved manufacturing facilities outside USA.
· Indian pharmaceutical companies received over 150 ANDA approvals from US FDA during 2013, accounting for approximately 38% of the total approvals.
The Indian pharmaceutical industry received foreign direct investments
(FDI) worth around US$ 11.95 billion during April, 2000 to September, 2013
EXPORTS
The pharmaceutical exports from India during 2012-13 stood at US$ 14.6 billion, witnessing an increase of US$ 1.4 billion from 2011-12. Indian pharmaceutical sector exports are likely to reach US$ 25 billion by 2016.
DEMAND
DRIVERS
Rising spend on healthcare: Total
annual healthcare spending is expected to more than double to US$ 201.4 billion,
growing at an average annual rate of 15.8% during2012-2017. Healthcare spending
is estimated to be around 0.5% of GDP in 2013.
Growing health insurance coverage: The
Indian government plans to bring 80% of India's population under health
insurance cover under its Health Insurance Vision 2020. This will lead to
higher volumes for the pharmaceuticals industry.
Growing incidence of chronic diseases: Chronic
therapies have grown at a faster pace than that of traditional acute therapies
over the past four years. Their contribution in the Indian pharmaceutical
market escalated from 27% in 2010 to 30% in 2013. Lifestyle changes, rapid
urbanisation and increasing affluence are factors which are expected to drive
it further.
Rapid urbanisation: An
increase in urban population from 31% to 40% or more by 2030 will see better
accessibility, with which will come with rapid urbanisation and the growth of
the pharmaceutical industry.
INDUSTRY CONCERNS
REGULATORY CHALLENGES
The Indian pharmaceutical market has its own set of regulatory challenges in the form of:
· Government-mandated price controls
· Delay in new product approvals
· Delay in clinical trial approvals
· Uncertainties over FDI policy
· These concerns act as a deterrent to the growth of the industry.
MANUFACTURING
QUALITY
India is attracting greater scrutiny from the US FDA in relation to cGMP
compliance, owing to the fact that it is the largest drugs supplier to the US.
Indian companies will have to conform to standards at par with the global
benchmarks. This will involve continuous improvement in systems and processes
and training of the workforce to ensure compliance to such standards.
SUN
PHARMA - RANBAXY MERGER
A LANDMARK
TRANSACTION
At Sun Pharma, they have taken a significant initiative to enhance
shareholder value for the future. They are in the process of acquiring Ranbaxy
Laboratories Limited, India's leading company in sales, in one of India's
largest MandA transactions. The deal, anall-stock transaction valued at US$ 4
billion, is expected to be completed by December2014. Ranbaxy shareholders will
receive 0.8 share of Sun Pharma for each Ranbaxy share.
ENHANCING
STAKEHOLDER VALUE
The combination has the potential to generate significant value for
shareholders:
The new entity will emerge as the world's fifth largest specialty
generic pharmaceutical company with a diverse, highly complementary portfolio
of specialty and generics (with minimal overlap) targeting chronic and acute
treatments globally. The entity's global presence across 55 markets will be
supported by over 40 manufacturing facilities and capabilities across multiple
dosage forms, including specialty branded products and complex generics.
In the US, the merged entity will become No.1 in the generic dermatology
market and No. 3 in the branded dermatology market with products to treat
Actinic Keratosis, Anti-Fungal, Acne and steroids for other treatments.
Post-merger, the overall pro-forma US revenues of the Company will be about US$
2.2 billion, with strong capabilities in developing complex products through a
broad portfolio of 184 ANDAs pending US FDA approval, including high-value FTF
(First-to-file) opportunities.
In India, the
merger will lead to Sun Pharma becoming the largest pharmaceutical company with
over 9% market share, enhancing value share across product offering sand market
territories. It will be ranked No.1 by prescriptions across 13different classes
of specialist doctors in India. Besides, the Ranbaxy acquisition will give it a
competitive edge in acute care, hospitals and OTC businesses with robust
brands. It will have 31 brands among India's top 300 brands and a greater
distribution reach. The merger will also create a foundation for the OTC business
in India. Pro-forma revenues of the merged entity in India will be about US$
1.1 billion.
With Sun Pharma's proven complex product capabilities and Ranbaxy's
strong global footprint, it will have a strong product pipeline and established
presence in key high-growth pharmerging economies like Russia, Romania, South
Africa, Brazil and Malaysia. Along with this the merged entity will have the
combined pro-forma revenues of about US$0.9 billion in emerging markets. In
some of these markets, the combined entity will have sales exceeding US$ 100
million each. The enhanced footprint across multiple markets will offer
opportunities for cross-selling and better brand building.
On a pro-forma basis, the merged company's revenues are estimated at US$
4.2 billion for calendar year CY 2013. The overall business will be much more
balanced with 47% of sales contributed by the US, 22% of it coming from India
and around 31% coming from the rest of the world and other businesses.
Pro-forma EBITDA will be US$ 1.2 billion for the twelvemonth period
ended 31st December, 2013.
Transaction value implies a revenue multiple of 2.2x, based on 12 months
ended 31st December, 2013.
Post-deal closure, the merged entity targets to generate synergy
benefits, of about US$ 250 million by the third year - driven by a combination
of revenue, procurement, supply chain and other cost synergies.
After deal closure, Daiichi Sankyo will become the second largest
shareholder in SunPharma (owning approximately 9% stake in Sun Pharma) and will
have the right to nominate one Director to Sun Pharma's Board of Directors. It
has agreed to indemnify Sun Pharma and Ranbaxy for, among other things, certain
costs and expenses that may arise from the recent subpoena, which Ranbaxy has
received from the United States Attorney for the Toansa facility.
Daiichi Sankyo (which holds approximately 63.4% of the outstanding
shares of Ranbaxy)and the Promoters of Sun Pharma (who hold approximately 63.7%
of the outstanding shares)have irrevocably agreed to vote in favour of the
transaction. Both the Ranbaxy and the SunPharma Boards have recommended
approval of the transaction to their respective shareholders.
The transaction closure will be subject to the usual closing conditions,
including approval by the following authorities: the Government of India; the
High Courts of Gujarat,
Punjab and Haryana; anti-competition authorities in India and a few
other markets; the National Stock Exchange of India; The Bombay Stock Exchange
and expiration of the waiting period under the Hart-Scott-Rodino Anti-trust
Improvement Act in the United States.
CREDIBLE TRACK
RECORD OF SUCCESSFUL TURNAROUNDS
One of the major challenges for Sun Pharma in this acquisition will be
to improve Ranbaxy's overall growth and profitability.
Sun Pharma has a robust track record of turning around its acquisitions
into success stories by enabling business and operational strategies and
building supply chain efficiencies, and Ranbaxy is likely to be a major
challenge. Sun Pharma's ability to juggle different businesses and multiple
cultures is likely to help in this transformation.
The Company leverages complementary functional strengths to achieve top
line growth andgains through both revenue enhancement and cost efficiencies
-translating into highermargins, greater market share and more operating
profits.
There were eight successful acquisitions in the period 1996-07 till
2000-01. Sharp increases in net sales and EBITDA across its acquisitions like
Taro, DUSA and URL marked the last decade. The Company acquired Taro in 2010
and was able to enhance the EBITDA from US$ 105 million to US$ 400 million
within three years by focusing on top line growth and becoming more efficient.
The payback from Sun Pharma's URL acquisition has been much faster than
expected.
SUN PHARMACEUTICAL
INDUSTRIES LIMITED (SUN PHARMA)
Sun Pharmaceutical Industries Limited is currently the fifth largest
global specialty pharmaceutical company manufacturing and marketing a variety
of pharmaceutical formulations as branded generics, as well as generics in the
US, India and several other global markets.
In India, the Company is a leader in niche therapy areas of psychiatry,
neurology, cardiology, nephrology, gastroenterology, orthopaedics and
ophthalmology. It has expertise in product development, process chemistry and
manufacture of complex dosage forms and APIs.
Major business
segments
· US Generics
· Indian Branded Generics
· International Branded Generics (Rest of the world, except the US)
· Active Pharmaceutical Ingredients (API)
STRATEGY AND
APPROACH
Sun Pharma's strategy and business approach is underpinned by the
following:
· Create sustainable revenue and cash flow stream:
Ensuring sustainable growth in revenues and cash flows is one of the key objectives. The Company achieves this by targeting complex/differentiated products in key markets; focusing on fast-growing chronic therapies and timely product launches
· Balance profitability and future investments:
Achieving a pragmatic balance between current profitability and future investments through its unwavering focus on developing complex products coupled with a strong track record of acquiring and turning around underperforming businesses.
· Cost leadership: Rationalising cost through vertical integration, optimisation of operational expenses and strengthening back-end and supply chain linkages
OPERATIONAL
PERFORMANCE, FY14
· Strong performance: Consolidated revenues for FY14 grew 42% over FY13 to Rs162 billion, while EBITDA grew by 45% to Rs 71 billion. The constant currency revenue growth guidance for FY14 was upgraded twice during the year from 18-20% to 29%. The strong performance was mainly driven by:
· A significant escalation in US revenues, which grew 59% and contributed about60% to overall revenues - was led by increased contribution from complex generics, strong profitability at Taro, favourable pricing for certain products and contribution from the180-day exclusivity for generic Prandin. The Ranbaxy acquisition will further strengthen Sun Pharma's positioning in the US branded market as well as enhance its overall product portfolio in the US.
· The India formulations business recorded 25% growth, despite the implementation of the new pricing policy and related trade channel disruptions. The Ranbaxy acquisitionis likely to further strengthen Sun Pharma's pan-India presence, as it will enable the merged entity to be ranked no.1 by prescriptions with 13 different classes of specialist doctors and facilitating its entry into India's OTC business with a few strong brands.
· Their Rest of World (RoW) business recorded 25% growth over FY13, led by a strengthening presence in key markets. The Ranbaxy acquisition is likely to significantly enhance Sun Pharma's presence in these markets and enable its entry into new markets.
· Ramp-up in specialty revenues: The Company continues to build its specialty revenues in the US, aided by a ramp-up in sales of generic Doxil and DUSA revenues - being the only company in the US market, with a US FDA approved version of generic Doxil. It also benefited from the supply constraints faced by the innovator.
· Taro: Taro reported good performance despite increasing competition. ForFY14, Taro's top line grew by 13% to US$ 759 million, while EBITDA grew by 29% to US$ 447million. EBITDA margins have expanded by 730 bps to 59% for the year. Taro's net profit for FY14 improved by 35% to US$ 360 million. The good performance was catalysed mainly by favourable sale prices throughout the year, which also witnessed a gradual increase in competition for some of Taro's products. The competition for some of Taro's products may intensify in future.
· 9 DUSA business scaling up: Sun Pharma had acquired DUSA in the US in December2012, giving it access to a branded patented product. DUSA revenues have gradually started scaling up, led by increasing penetration with dermatologists and gradual price increases.
· Sun-intrexon joint venture: As part of its efforts to establish a long-term specialty portfolio, Sun Pharma has entered into a joint venture with Intrexon Corporation (USA) to develop controllable gene-based therapies to treat ocular diseases that cause partial or total blindness in Million of people worldwide. Initial targets are dryage-related macular degeneration (AMD), glaucoma and retinitis pigmentosa. The joint venture will leverage Sun Pharma's global capabilities and experience in developing and manufacturing specialty pharmaceuticals for niche therapy areas.
· Ramp-up in URL: FY14 was the first full year of consolidation of the US-based URL acquisition. Favourable product pricing enabled significant ramp-up in URL's revenues with the relaunch of some of the discontinued products from URL's portfolio. The February 2013 acquisition broadens Sun Pharma's US product portfolio, besides giving access to two US FDA approved facilities.
· Generic Prandin exclusivity in the US: Sun Pharma's US business benefited from the one-time upside of 180-day exclusivity on generic Prandin, which expired in January 2014. By virtue of its successful patent challenge, it enjoyed the First-to-File (FTF) status and was the only generic player in the US market for 180 days in FY14. Post exclusivity, other generic players have also launched their versions of the product.
· Generic Gleevec settlement: In May 2014, one of Sun Pharma's subsidiaries executed a settlement agreement with Novartis Pharmaceuticals Corporation, stipulating adismissal of the lawsuits filed in the US against the Company regarding submission of an Abbreviated New Drug Application (ANDA) for a generic version of Gleevec, Imatinib Mesylate tablets. Indicated for the treatment of chronic myeloid leukemia and having annual sales of about US$ 2 billion in the US market, a generic version of these Gleevec tablets is to be launched by Sun Pharma's subsidiary in February, 2016, under the terms of the settlement.
· US FDA Approvals: Received a total of 26 Abbreviated New Drug Applications (ANDA) approvals from the US FDA, including approvals for Repaglinide, Testosterone Cypionate Injections, Topotecan HCl Injection, Duloxetine HCl capsules, Temozolomide capsules, and a few controlled substances.
· Strengthening senior management team: Sun Pharma consistently nurtures internal talent and is in the process of expanding the pool of capable people to drive growth. In FY14, the Company reinforced its senior management team by attracting national and global talent for its key functions.
· Strengthening the Board of Directors: In February 2014, Sun Pharma strengthened its Board of Directors by appointing Ms. Rekha Sethi as an additional Independent Director. Ms. Sethi is the Director General of the All India Management Association (AIMA), India's apex body for management. She is associated with the following organizations: Indo-Netherlands Joint Working Group on Corporate Governance and Corporate Social Responsibility, under the Ministry of Corporate Affairs, Government of India Advisory Board of the Switzerland-based St Gallen Foundation think tank, Leaders of Tomorrow - Knowledge Pool. She had also worked with the Confederation of Indian Industry (CII) for over 17 years before joining AIMA.
· Settling the generic Protonix patent litigation in the US: The Company has settled the patent litigation in the US regarding generic Protonix with Wyeth and Atlanta Pharma AG and paid US$ 550 million to Pfizer as part of the settlement. Sun Pharma can continue to sell its generic Protonix in the US.
· Karkhadi facility: In May 2014, the Company received a warning letter from the US FDA for its cephalosporin facility located at Karkhadi, Gujarat, India. This letter was a follow-up to the import alert issued by the US FDA for this facility in March, 2014, identifying some practises at the facility, which are non-compliant with current Good Manufacturing Practice (cGMP) regulations. The Company remains fully committed to compliance and has already initiated several corrective steps to address the US FDA's observations. It is committed to working cooperatively and expeditiously with the USFDA to resolve matters indicated in its letter. However, the USFDA might withhold approval of pending new drug applications from the facility until resolution of the issue. However, the contribution of this facility to Sun Pharma's consolidated revenues is negligible.
STATEMENT
OF STANDALONE UNAUDITED FINANCIAL RESULTS FOR THE QUARTER AND HALF YEAR ENDED
SEPTEMBER 30, 2014
(RS.
IN MILLION)
|
PARTICULARS |
Quarter ended |
Half Year ended |
|
|
|
30.09.2014 Unaudited |
30.06.2014 Unaudited |
30.09.2014 Unaudited |
|
Income from
Operations |
|
|
|
|
Net Sales / Income from Operations (Net of Excise Duty) |
5727.800 |
5590.100 |
11317.900 |
|
Other Operating Income |
246.700 |
165.700 |
412.400 |
|
Income from
Operations [Net] |
5974.500 |
5755.800 |
11730.300 |
|
|
|
|
|
|
|
|
|
|
|
Expenses |
|
|
|
|
Cost of materials consumed |
2315.500 |
2388.400 |
4703.900 |
|
Purchases of stock-in-trade |
416.900 |
313.600 |
730.500 |
|
Changes in inventories of finished goods, work-in-progress and stock-in-trade |
78.600 |
(451.400) |
(372.800) |
|
Employee benefits expense |
934.300 |
876.800 |
1811.100 |
|
Depreciation and amortisation expense |
675.700 |
463.900 |
1139.600 |
|
Other expenses |
4175.200 |
2329.300 |
6504.500 |
|
Total Expenses |
8596.200 |
5920.600 |
14516.800 |
|
|
|
|
|
|
Profit / (Loss) from Operations before Other Income, Finance Costs and Exceptional Item |
(2621.700) |
(164.800) |
(2786.500) |
|
Other income |
(569.100) |
842.100 |
273.000 |
|
Profit / (Loss) before Finance Costs and Exceptional Item |
(3190.800) |
677.300 |
(2513.500) |
|
Finance costs |
40.200 |
80.900 |
121.100 |
|
Profit / (Loss) after Finance Costs but before Exceptional Item |
(3231.000) |
596.400 |
(2634.600) |
|
Exceptional item |
-- |
-- |
-- |
|
Profit / (Loss) after Exceptional Item but before tax |
(3231.000) |
596.400 |
(2634.600) |
|
Tax expense |
(54.800) |
120.800 |
66.000 |
|
Net Profit / (Loss)
for the period / year |
(3176.200) |
475.600 |
(2700.600) |
|
Paid-up Equity Share Capital |
|
|
|
|
Equity Shares - Face Value Re. 1 each |
2071.200 |
2071.200 |
2071.200 |
|
Reserves excluding Revaluation Reserve |
-- |
-- |
-- |
|
Earnings Per Share of Re. 1 each - in ` (Basic and Diluted) |
(1.53) |
0.23 |
(1.30) |
|
|
|
|
|
|
Research and Development Expenses incurred (included above) |
1317.900 |
835.600 |
2153.500 |
|
|
|
|
|
|
PUBLIC
SHAREHOLDING |
|
|
|
|
Number of Equity Shares of Re. 1 each |
752817510 |
752817510 |
752817510 |
|
Percentage of Shareholding |
36.35 |
36.35 |
36.35 |
|
|
|
|
|
|
PROMOTERS AND
PROMOTER GROUP SHAREHOLDING |
|
|
|
|
a) Pledged / Encumbered |
|
|
|
|
Number of Equity Shares of Re. 1 each |
7405000 |
4370000 |
7405000 |
|
Percentage of Equity Shares (as a % of the total shareholding of
promoter and promoter group) |
0.56 |
0.33 |
0.56 |
|
Percentage of Equity Shares (as a % of the total share capital of the
Company) |
0.36 |
0.21 |
0.56 |
|
|
|
|
|
|
b) Non-encumbered |
|
|
|
|
Number of Equity Shares of Re. 1 each |
1310941400 |
1313976400 |
1310941400 |
|
Percentage of Equity Shares (as a % of the total shareholding of
promoter and promoter group) |
99.44 |
99.67 |
99.44 |
|
Percentage of Equity Shares (as a % of the total share capital of the
Company) |
63.29 |
63.44 |
63.29 |
|
Investor
Complaints [Nos.] |
30.09.2014 |
|
Pending at the beginning of the quarter |
- |
|
Received during the quarter |
2 |
|
Disposed of during the quarter |
2 |
|
Remaining unresolved at the end of the
quarter |
- |
NOTES
1) Statement of Assets and Liabilities
|
SR. NO. |
PARTICULARS |
30.09.2014 Unaudited |
|
A |
EQUITY AND
LIABILITIES |
|
|
i |
Shareholders'
Funds |
|
|
|
(a) Share Capital |
2071.200 |
|
|
(b) Reserves and Surplus |
69307.000 |
|
|
Total
|
71378.200 |
|
|
|
|
|
ii |
Non-current
Liabilities |
|
|
|
(a) Long-term Borrowings |
77.300 |
|
|
(b) Deferred Tax Liabilities (Net) |
1984.500 |
|
|
(c) Other Long-term Liabilities |
20.700 |
|
|
(d) Long-term Provisions |
25166.700 |
|
|
Total
|
27249.200 |
|
|
|
|
|
iii |
Current
Liabilities |
|
|
|
(a) Short-term Borrowings |
358.700 |
|
|
(b) Trade Payables |
5651.800 |
|
|
(c) Other Current Liabilities |
9633.000 |
|
|
(d) Short-term Provisions |
4054.900 |
|
|
Total
|
19698.400 |
|
|
|
|
|
|
TOTAL - EQUITY
AND LIABILITIES |
118325.800 |
|
|
|
|
|
B |
ASSETS |
|
|
i |
Non-current
Assets |
|
|
|
(a) Fixed Assets |
21327.700 |
|
|
(b) Non-Current Investments |
60940.000 |
|
|
(c) Long-term Loans and Advances |
8951.400 |
|
|
(d) Other Non-current Assets |
0.000 |
|
|
Total
|
91219.100 |
|
|
|
|
|
ii |
Current Assets |
|
|
|
(a) Current Investments |
1240.200 |
|
|
(b) Inventories |
10974.000 |
|
|
(c) Trade Receivables |
8574.500 |
|
|
(d) Cash and Cash Equivalents |
1072.200 |
|
|
(e) Short-term Loans and Advances |
4165.900 |
|
|
(f) Other Current Assets |
1079.900 |
|
|
Total
|
27106.700 |
|
|
|
|
|
|
TOTAL - ASSETS |
118325.800 |
2)
The above financial results of the Company have
been reviewed by the Audit Committee and approved by the Board of Directors at
their respective meetings held on November 13, 2014 and have been subjected to
a Limited Review by the Statutory Auditors of the Company.
3)
The Company has only one reportable business
segment namely 'Pharmaceuticals'.
4)
Pursuant to the scheme of arrangement in the nature
of demerger, the specified undertaking of Sun Pharma Global FZE, a wholly owned
subsidiary, has been transferred into the Company w.e.f May 1, 2013, without
any consideration, on a going concern basis consisting of all the assets and
liabilities pertaining to the said undertaking. The scheme of arrangement has
been approved by shareholders of both the companies and is also approved by the
Hon’ble High Court of Gujarat. Accordingly, the financial statements for the
year ended March 31, 2014, earlier approved by the Board of Directors at their
meeting held on May 29, 2014, have been revised and approved by them at their
meeting held on August 12, 2014 and the effect of the Scheme was given in the
financial results for the year ended March 31, 2014 only on an annual basis.
However, the financial results for the quarter and half year ended September
30, 2013 have not been restated in these results.
5)
The Company has adopted the useful lives of fixed
assets as indicated in Part C of Schedule II of the Companies Act, 2013 and
amendment thereto vide notification dated August 29, 2014 issued by Ministry of
Corporate Affairs. Consequently, the depreciation charge for the quarter ended
September 30, 2014 is higher by Rs.371.400 Million (including Rs.220.000
Million in respect of the quarter ended June 30, 2014) and the depreciation
charge for the half year ended September 30, 2014 is higher by Rs.562.300
Million.
6)
Other Income is net of impact of MTM loss on
forward foreign exchange contracts and loss on foreign currency translations
(other than those included in purchases and sales).
7)
The Board of Directors of the Company at their
meeting held on April 6, 2014 approved the proposed scheme of arrangement u/s
391 to 394 of the Companies Act, 1956 for amalgamation of Ranbaxy Laboratories
Ltd into the Company with effect from April 1, 2014, the appointed date
("the Proposed Scheme"). The National Stock Exchange Limited and BSE
Limited have conveyed their 'No Objection' in July 2014. The said Scheme has
also been approved by the shareholders of the Company at the Court convened meeting
held on August 22, 2014. Pending approval of the Proposed Scheme by the Hon'ble
High Court of Gujarat and other statutory compliances, no effect of the
Proposed Scheme has been given in these financial results.
8)
Figures for the previous periods / year have been regrouped
wherever considered necessary, other than as referred in Note 4 above.
CONTINGENT
LIABILITIES:
|
PARTICULARS |
31.03.2014 (Rs.
In Million) |
31.03.2013 (Rs.
In Million) |
|
A Claims against the Company not acknowledged as debts |
24.000 |
34.600 |
|
B Guarantees Guarantees Given by the bankers on behalf of the Company |
374.200 |
227.400 |
|
Corporate Guarantees |
86.000 |
149.200 |
|
C Others |
|
|
|
Letters of Credit for Imports |
2103.200 |
463.200 |
|
Liabilities Disputed - Appeals filed with respect to : |
|
|
|
Income Tax on account of Disallowances / Additions |
4927.800 |
2771.200 |
|
Sales Tax on account of Rebate / Classification |
48.500 |
48.400 |
|
Excise Duty on account of Valuation / Cenvat Credit |
93.600 |
322.200 |
|
ESIC Contribution on account of applicability |
0.200 |
0.200 |
|
Drug Price Equalisation Account [DPEA] on account of demand towards unintended
benefit, including interest thereon, enjoyed by the Company |
14.000 |
14.000 |
|
Demand by JDGFT import duty with respect to import alleged to be in
excess of entitlement as per the Advanced Licence Scheme |
14.600 |
13.900 |
FIXED ASSETS:
TANGIBLE ASSETS
· Freehold Land
· Leasehold Land
· Buildings
· Plant and Machinery
· Vehicles
· Office Equipment
· Furniture and Fixture
INTANGIBLE ASSETS
· Trademarks, Designs and Other Intangible Assets
CMT REPORT (Corruption, Money Laundering & Terrorism]
The Public Notice information has been collected from various sources
including but not limited to: The Courts,
1] INFORMATION ON
DESIGNATED PARTY
No exist designating subject or any of its beneficial owners, controlling
shareholders or senior officers as terrorist or terrorist organization or whom
notice had been received that all financial transactions involving their assets
have been blocked or convicted, found guilty or against whom a judgement or
order had been entered in a proceedings for violating money-laundering,
anti-corruption or bribery or international economic or anti-terrorism sanction
laws or whose assets were seized, blocked, frozen or ordered forfeited for
violation of money laundering or international anti-terrorism laws.
2] Court Declaration :
No exist to suggest that subject is or was
the subject of any formal or informal allegations, prosecutions or other
official proceeding for making any prohibited payments or other improper
payments to government officials for engaging in prohibited transactions or
with designated parties.
3] Asset Declaration :
No records exist to suggest that the property or assets of the subject
are derived from criminal conduct or a prohibited transaction.
4] Record on Financial
Crime :
Charges or conviction
registered against subject: None
5] Records on Violation of
Anti-Corruption Laws :
Charges or
investigation registered against subject: None
6] Records on Int’l
Anti-Money Laundering Laws/Standards :
Charges or
investigation registered against subject: None
7] Criminal Records
No
available information exist that suggest that subject or any of its principals
have been formally charged or convicted by a competent governmental authority for
any financial crime or under any formal investigation by a competent government
authority for any violation of anti-corruption laws or international anti-money
laundering laws or standard.
8] Affiliation with
Government :
No record
exists to suggest that any director or indirect owners, controlling
shareholders, director, officer or employee of the company is a government
official or a family member or close business associate of a Government
official.
9] Compensation Package :
Our market
survey revealed that the amount of compensation sought by the subject is fair
and reasonable and comparable to compensation paid to others for similar
services.
10] Press Report : No
press reports / filings exists on the subject
CORPORATE GOVERNANCE
MIRA INFORM as part of its Due Diligence do provide comments on
Corporate Governance to identify management and governance. These factors often
have been predictive and in some cases have created vulnerabilities to credit
deterioration.
Our Governance Assessment focuses principally on the interactions
between a company’s management, its Board of Directors, Shareholders and other
financial stakeholders.
CONTRAVENTION
Subject is not known to have contravened any existing local laws,
regulations or policies that prohibit, restrict or otherwise affect the terms
and conditions that could be included in the agreement with the subject.
FOREIGN EXCHANGE RATES
|
Currency |
Unit
|
Indian Rupees |
|
US Dollar |
1 |
Rs. 62.92 |
|
|
1 |
Rs. 93.57 |
|
Euro |
1 |
Rs. 67.34 |
INFORMATION DETAILS
|
Information
Gathered by : |
SAN |
|
|
|
|
Analysis Done by
: |
DIV |
|
|
|
|
Report Prepared
by : |
MTN |
SCORE & RATING EXPLANATIONS
|
SCORE FACTORS |
RANGE |
POINTS |
|
HISTORY |
1~10 |
7 |
|
PAID-UP CAPITAL |
1~10 |
7 |
|
OPERATING SCALE |
1~10 |
8 |
|
FINANCIAL
CONDITION |
|
|
|
--BUSINESS SCALE |
1~10 |
8 |
|
--PROFITABILITY |
1~10 |
5 |
|
--LIQUIDITY |
1~10 |
7 |
|
--LEVERAGE |
1~10 |
7 |
|
--RESERVES |
1~10 |
8 |
|
--CREDIT LINES |
1~10 |
8 |
|
--MARGINS |
-5~5 |
-- |
|
DEMERIT POINTS |
|
|
|
--BANK CHARGES |
YES/NO |
YES |
|
--LITIGATION |
YES/NO |
NO |
|
--OTHER ADVERSE INFORMATION |
YES/NO |
NO |
|
MERIT POINTS |
|
|
|
--SOLE DISTRIBUTORSHIP |
YES/NO |
NO |
|
--EXPORT ACTIVITIES |
YES/NO |
NO |
|
--AFFILIATION |
YES/NO |
YES |
|
--LISTED |
YES/NO |
YES |
|
--OTHER MERIT FACTORS |
YES/NO |
YES |
|
DEFAULTER |
|
|
|
--RBI |
YES/NO |
NO |
|
--EPF |
YES/NO |
NO |
|
TOTAL |
|
65 |
This score serves as a reference to assess SC’s
credit risk and to set the amount of credit to be extended. It is calculated
from a composite of weighted scores obtained from each of the major sections of
this report. The assessed factors and their relative weights (as indicated
through %) are as follows:
Financial
condition (40%) Ownership
background (20%) Payment
record (10%)
Credit history
(10%) Market trend (10%) Operational size
(10%)
RATING EXPLANATIONS
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
>86 |
Aaa |
Possesses an extremely sound financial base with the strongest
capability for timely payment of interest and principal sums |
Unlimited |
|
71-85 |
Aa |
Possesses adequate working capital. No caution needed for credit transaction.
It has above average (strong) capability for payment of interest and
principal sums |
Large |
|
56-70 |
A |
Financial & operational base are regarded healthy. General
unfavourable factors will not cause fatal effect. Satisfactory capability for
payment of interest and principal sums |
Fairly Large |
|
41-55 |
Ba |
Overall operation is considered normal. Capable to meet normal
commitments. |
Satisfactory |
|
26-40 |
B |
Capability to overcome financial difficulties seems comparatively below
average. |
Small |
|
11-25 |
Ca |
Adverse factors are apparent. Repayment of interest and principal sums
in default or expected to be in default upon maturity |
Limited with
full security |
|
<10 |
C |
Absolute credit risk exists. Caution needed to be exercised |
Credit not
recommended |
|
-- |
NB |
New Business |
-- |
This report is issued at your request without any
risk and responsibility on the part of MIRA INFORM PRIVATE LIMITED (MIPL) or
its officials.