MIRA INFORM REPORT
|
Report No. : |
356656.2 |
|
Report Date : |
26.12.2015 |
IDENTIFICATION DETAILS
|
Name : |
GLAXOSMITHKLINE PHARMACEUTICALS LIMITED |
|
|
|
|
Registered
Office : |
|
|
Tel. No.: |
91-22-24959595 / 24959779 |
|
|
|
|
Country : |
India |
|
|
|
|
Financials (as
on) : |
31.03.2015 |
|
|
|
|
Date of
Incorporation : |
13.11.1924 |
|
|
|
|
Com. Reg. No.: |
11-001151 |
|
|
|
|
Capital
Investment / Paid-up Capital : |
Rs.847.030 Million |
|
|
|
|
CIN No.: [Company Identification
No.] |
L24239MH1924PLC001151 |
|
|
|
|
IEC No.: |
0388040092 |
|
|
|
|
TAN No.: [Tax Deduction &
Collection Account No.] |
MUMG00196A |
|
|
|
|
PAN No.: [Permanent Account No.] |
AAACG4414B |
|
|
|
|
Legal Form : |
A Public Limited Liability Company. The Company’s Shares are Listed on
the Stock Exchanges. |
|
|
|
|
Line of Business
: |
Subject
is engaged interalia, in the business of manufacturing, distributing and
trading in pharmaceuticals. (Registered activity) |
|
|
|
|
No. of Employees
: |
Information declined by the management |
RATING & COMMENTS
|
MIRA’s Rating : |
Aa (85) |
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
71-85 |
Aa |
Possesses adequate working capital. No caution needed for credit transaction.
It has above average (strong) capability for payment of interest and
principal sums |
Large |
|
Maximum Credit Limit : |
USD 52000000 |
|
|
|
|
Status : |
Excellent |
|
|
|
|
Payment Behaviour : |
Regular |
|
|
|
|
Litigation : |
Clear |
|
|
|
|
Comments : |
Subject is a subsidiary of GlaxoSmithKline plc, U.K. It is a
well-established and reputed company having excellent track. For the financial year ended 2015, the company has reported top line
growth of 29.5% along with profitability margin of 12.04%. Rating takes into consideration the company’s leading market position
in the industry supported by well established distribution networks,
diversified market presence globally and strong financial base along with
zero debt balance sheet profile. Rating reflects company’s establish product range, some of them are
Iodex, Ostoclacium, Betnovate. Trade relations are trustworthy. Business is active. Payments are
regular and as per commitments. In view of long business track records and strong holding company, it
can be considered for business dealings at usual trade terms and conditions. |
NOTES :
Any query related to this report can be made
on e-mail : infodept@mirainform.com
while quoting report number, name and date.
ECGC Country Risk Classification List – March 31, 2015
|
Country Name |
Previous Rating (31.12.2014) |
Current Rating (31.03.2015) |
|
India |
A1 |
A1 |
|
Risk Category |
ECGC
Classification |
|
Insignificant |
A1 |
|
Low |
A2 |
|
Moderate |
B1 |
|
High |
B2 |
|
Very High |
C1 |
|
Restricted |
C2 |
|
Off-credit |
D |
EXTERNAL AGENCY RATING
|
Rating Agency Name |
Not Available |
|
Rating |
Not Available |
|
Rating Explanation |
Not Available |
|
Date |
Not Available |
RBI DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available RBI Defaulters’ list.
EPF (Employee Provident Fund) DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter in
the publicly available EPF (Employee Provident Fund) Defaulters’ list as of
31-03-2015.
INFORMATION DECLINED
Management Non-Cooperative (Tel No.: 91-22-24959595 / 24959779)
LOCATIONS
|
Registered Office / Factory : |
|
|
Tel. No.: |
91-22-24959595 / 24959779 |
|
Fax No.: |
91-22-24935358/ 24959494 |
|
E-Mail : |
|
|
Website : |
|
|
|
|
|
Factory 1 : |
2nd
|
|
|
|
|
Factory 2 : |
Ambad,
Nashik, |
DIRECTORS
As on 31.03.2015
|
Name : |
Mr. Deepak Shantilal Parekh |
|
Designation : |
Director |
|
Address : |
9/B, Darbhanga Mansion, 12, Carmichael Road, Mumbai - 400026, Maharashtra, India |
|
Date of Appointment : |
28.09.1994 |
|
DIN No.: |
00009078 |
|
|
|
|
Name : |
Mr. Nihal Kaviratne |
|
Designation : |
Director |
|
Address : |
A-5, Seaface Park, 2nd Floor, Breach Candy, Bhulabhai Desai Road, Mumbai - 400026, Maharashtra, India |
|
Date of Appointment : |
26.037.2005 |
|
DIN No.: |
00032473 |
|
|
|
|
Name : |
Mr. Rajeshwar Raj Bajaaj |
|
Designation : |
Director |
|
Address : |
Flat No. 2, Ajanta Apartments, 11th Floor, 75, Colaba, Mumbai - 400005, Maharashtra, India |
|
Date of Appointment : |
10.10.2002 |
|
DIN No.: |
00419623 |
|
|
|
|
Name : |
Mr. Damodarannair Sundaram |
|
Designation : |
Director |
|
Address : |
Flat No.1901, Tower-A, Beaumonde, Appasaheb Marathe Marg, Prabhadevi, Mumbai - 400025, Maharashtra, India |
|
Date of Appointment : |
27.07.2009 |
|
DIN No.: |
00016304 |
|
|
|
|
Name : |
Mr. Pradeep Vasudeo Bhide |
|
Designation : |
Director |
|
Address : |
D - 1/48 ,( First Floor ), Vasant Vihar, New Delhi - 110057, India |
|
Date of Appointment : |
28.10.2010 |
|
DIN No.: |
03304262 |
|
|
|
|
Name : |
Mr. Anami Narayan Prema Roy |
|
Designation : |
Director |
|
Address : |
62, Sagar Tarang, Worli Seaface, Mumbai - 400025, Maharashtra, India |
|
Date of Appointment : |
04.11.2011 |
|
DIN No.: |
01361110 |
|
|
|
|
Name : |
Mr. Raju Krishanaswamy |
|
Designation : |
Director |
|
Address : |
Plot No. 44, Sector F-1, N-4, CIDCO, Aurangabad - 431003, Maharashtra, India |
|
Date of Appointment : |
01.08.2011 |
|
DIN No.: |
03043004 |
|
|
|
|
Name : |
Ms. Anjali Bansal |
|
Designation : |
Director |
|
Address : |
3202, A-Wing, Vivarea, Sane Guruji Marg, Jacob Circle, Mahalaxmi, Mumbai - 400011, Maharashtra, India |
|
Date of Appointment : |
19.02.2013 |
|
DIN No.: |
00207746 |
|
|
|
|
Name : |
Andrew Antrinkos Aristidou |
|
Designation : |
Director |
|
Address : |
Flat No 2405 North The Imperial Condominium, The Imperial Towers M P Mill Compound, B B Nakashe Marg Tardeo Mumbai - 400034, Maharashtra, India |
|
Date of Appointment : |
01.12.2014 |
|
DIN No.: |
07034424 |
|
|
|
|
Name : |
Regis Jean Simard |
|
Designation : |
Director |
|
Address : |
52 Adams Quarter Brentford Lock, Tallow Road, Brentford, TW88ER, United Kingdom |
|
Date of Appointment : |
18.05.2015 |
|
DIN No.: |
07186737 |
|
|
|
|
Name : |
Mr. Vaidheesh Annaswamy |
|
Designation : |
Director |
|
Address : |
1501-Lodha Grandeur, Sayani Road, Prabhadevi, Mumba - 400028, Maharashtra, India |
|
Date of Appointment : |
03.08.2015 |
|
DIN No.: |
01444303 |
|
|
|
|
Name : |
Ronald Canute Sequeira |
|
Designation : |
Director |
|
Address : |
19 Hill Park, A G Bell Marg Malabar Hill, Mumbai - 400006, Maharashtra, India |
|
Date of Appointment : |
25.10.2007 |
|
DIN No.: |
01549120 |
KEY EXECUTIVES
|
Name : |
Mr. Ajay Avinash Nadkarni |
|
Designation : |
Secretary |
|
Address : |
303/304 Vilas Apartment, Navghar Road Mulund (East), Mumbai - 400081, Maharashtra, India |
|
Date of Appointment : |
01.11.2001 |
|
PAN No.: |
AAAPN4414B |
|
|
|
|
Name : |
Andrew Antrinkos Aristidou |
|
Designation : |
Chief Finance Officer |
|
Address : |
Flat No 2405 North The Imperial Condominium, The Imperial Towers M P Mill Compound, B B Nakashe Marg Tardeo Mumbai - 400034, Maharashtra, India |
|
Date of Appointment : |
01.12.2014 |
|
PAN No.: |
BOZPA5080C |
MAJOR SHAREHOLDERS / SHAREHOLDING PATTERN
As on 30.09.2015
|
Category of
Shareholder |
Total No. of
Shares |
Total
Shareholding as a % of Total No. of Shares |
|
As a
% of (A+B) |
||
|
(A)
Shareholding of Promoter and Promoter Group |
||
|
|
|
|
|
|
|
|
|
|
63527262 |
75.00 |
|
|
63527262 |
75.00 |
|
Total
shareholding of Promoter and Promoter Group (A) |
63527262 |
75.00 |
|
(B)
Public Shareholding |
||
|
|
|
|
|
|
1140548 |
1.35 |
|
|
6804531 |
8.03 |
|
|
1870763 |
2.21 |
|
|
9815842 |
11.59 |
|
|
|
|
|
|
812282 |
0.96 |
|
|
|
|
|
|
10139081 |
11.97 |
|
|
179641 |
0.21 |
|
|
228909 |
0.27 |
|
|
18315 |
0.02 |
|
|
200135 |
0.24 |
|
|
6616 |
0.01 |
|
|
294 |
0.00 |
|
|
2937 |
0.00 |
|
|
612 |
0.00 |
|
|
11359913 |
13.41 |
|
Total
Public shareholding (B) |
21175755 |
25.00 |
|
Total
(A)+(B) |
84703017 |
100.00 |
|
(C)
Shares held by Custodians and against which Depository Receipts have been
issued |
0 |
0.00 |
|
|
0 |
0.00 |
|
|
0 |
0.00 |
|
|
0 |
0.00 |
|
Total
(A)+(B)+(C) |
84703017 |
0.00 |
BUSINESS DETAILS
|
Line of Business : |
Subject
is engaged interalia, in the business of manufacturing, distributing and
trading in pharmaceuticals. (Registered activity) |
|
|
|
|
Products : |
Not Divulged |
|
|
|
|
Brand Names : |
Not Divulged |
|
|
|
|
Agencies Held : |
Not Divulged |
|
|
|
|
Exports : |
Not Divulged |
|
|
|
|
Imports : |
Not Divulged |
|
|
|
|
Terms : |
Not Divulged |
PRODUCTION STATUS NOT AVAILABLE
GENERAL INFORMATION
|
Suppliers : |
|
||||||||||||||
|
|
|
||||||||||||||
|
Customers : |
|
||||||||||||||
|
|
|
||||||||||||||
|
No. of Employees : |
Information declined by the management |
||||||||||||||
|
|
|
||||||||||||||
|
Bankers : |
|
||||||||||||||
|
|
|
|
Auditors : |
|
|
Name : |
Price Waterhouse and Company Bangalore LLP Chartered Accountants |
|
Address : |
252, Veer Savarkar Marg, Shivji Park, Dadar (West), Mumbai – 400028,
Maharashtra, India |
|
Tel. No.: |
91-22-66691500 |
|
Fax No.: |
91-22-66547804 / 07 |
|
|
|
|
Cost Auditors : |
R. Nanabhoy and Company Chartered Accountants |
|
|
|
|
Memberships : |
Not Available |
|
|
|
|
Collaborators : |
Not Available |
|
|
|
|
Shareholders (the
GlaxoSmithKline (GSK) Group shareholding) in the Company |
|
|
|
|
|
Subsidiary of the Company : |
|
|
|
|
|
Other related parties in the
GlaxoSmithKline (GSK) Group where common control exists and with whom the
Company had transactions during the
period: |
|
CAPITAL STRUCTURE
As on 31.03.2015
Authorised Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
90000000 |
Equity Shares |
Rs.10/- each |
Rs.900.000 Million |
|
|
|
|
|
Issued Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
84707710 |
Equity Shares |
Rs.10/- each |
Rs.847.077
Million |
|
|
|
|
|
Subscribed & Paid-up Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
84703017 |
Equity Shares |
Rs.10/- each |
Rs.847.030
Million |
|
|
|
|
|
* excludes 4,693 equity shares of Rs. 10 each of the Company (3,352 equity shares of Rs. 10 each of erstwhile Burroughs Wellcome (India) Limited) held in abeyance.
NOTE
(a) Reconciliation of the
number of shares:
|
PARTICULARS |
As at 31st, March 2015 |
|
|
Number of Shares |
Rs. in Million |
|
|
Balance at the beginning of the year |
84,703,017 |
847.030 |
|
Issued during the year |
-- |
-- |
|
Balance at the end of the year |
84,703,017 |
847.030 |
(b) Rights, preferences and
restrictions attached to equity shares:
The
Company has one class of equity shares having a par value of Rs. 10 per share. Each
shareholder is eligible for one vote per share held. The dividend proposed by
the Board of Directors is subject to the approval of the shareholders in the
ensuing Annual General Meeting, except in case of interim dividend. In the
event of liquidation, the equity shareholders are eligible to receive the
remaining assets of the Company after distribution of all preferential amounts,
in proportion to their shareholding.
(c) Shares held by
subsidiaries of ultimate holding company in aggregate:
|
PARTICULARS |
As at 31st, March 2015 |
|
|
Number of Shares |
Rs. in Million |
|
|
Equity shares of Rs. 10 each (representing 50.67% of total shareholding) |
63527262 |
635.273 |
(d) Details of equity shares held
by shareholders holding more than 5% shares of the aggregate shares in the
Company:
|
PARTICULARS |
As at 31st December, 2013 |
|
|
Number of Shares |
Percentage of Holding |
|
|
Glaxo Group Limited, U.K. |
30,485,250 |
35.99% |
|
GlaxoSmithKline
Pte Limited, Singapore |
20609774 |
24.33% |
|
Eskaylab Limited, U.K. |
5,880,000 |
6.94% |
|
Life Insurance Corporation of India |
4749658 |
5.61% |
LISTING DETAILS:
|
|
BSE : 500660 NSE : GLAXO |
|
Stock Exchange Place : |
The Stock Exchange, Mumbai, National Stock Exchange of India Limited |
|
Listed Date : |
Not Available |
FINANCIAL DATA
[all figures are
in Rupees Million]
ABRIDGED
BALANCE SHEET
|
SOURCES
OF FUNDS |
31.03.2015 (15
Months) |
31.12.2013 |
31.12.2012 |
|
|
|
|
|
|
I.
EQUITY AND LIABILITIES |
|
|
|
|
(1)Shareholders' Funds |
|
|
|
|
(a) Share Capital |
847.030 |
847.030 |
847.030 |
|
(b) Reserves & Surplus |
17669.651 |
19324.871 |
19253.122 |
|
(c) Money received against share
warrants |
0.000 |
0.000 |
0.000 |
|
|
|
|
|
|
(2) Share Application money
pending allotment |
0.000 |
0.000 |
0.000 |
|
Total
Shareholders’ Funds (1) + (2) |
18516.681 |
20171.901 |
20100.152 |
|
|
|
|
|
|
(3) Non-Current Liabilities |
|
|
|
|
(a) long-term borrowings |
26.260 |
36.050 |
41.424 |
|
(b) Deferred tax liabilities
(Net) |
0.000 |
0.000 |
0.000 |
|
(c) Other long term
liabilities |
52.886 |
49.672 |
49.672 |
|
(d) long-term provisions |
2709.587 |
2410.404 |
2347.929 |
|
Total
Non-current Liabilities (3) |
2788.733 |
2496.126 |
2439.025 |
|
|
|
|
|
|
(4) Current Liabilities |
|
|
|
|
(a) Short term borrowings |
0.000 |
0.000 |
0.000 |
|
(b) Trade payables |
3058.863 |
2780.124 |
2351.889 |
|
(c) Other current liabilities |
1315.553 |
862.207 |
883.532 |
|
(d) Short-term provisions |
6684.192 |
5188.323 |
5122.067 |
|
Total
Current Liabilities (4) |
11058.608 |
8830.654 |
8357.488 |
|
|
|
|
|
|
TOTAL |
32364.022 |
31498.681 |
30896.665 |
|
|
|
|
|
|
II.
ASSETS |
|
|
|
|
(1) Non-current assets |
|
|
|
|
(a) Fixed Assets |
|
|
|
|
(i) Tangible assets |
1230.596 |
1000.718 |
894.021 |
|
(ii) Intangible Assets |
0.000 |
0.000 |
0.000 |
|
(iii) Capital work-in-progress |
1152.244 |
618.601 |
437.875 |
|
(iv) Intangible assets under
development |
0.000 |
0.000 |
0.000 |
|
(b) Non-current Investments |
476.697 |
476.697 |
576.751 |
|
(c) Deferred tax assets (net) |
829.143 |
921.065 |
865.409 |
|
(d) Long-term Loan and Advances |
2883.103 |
2328.739 |
1896.868 |
|
(e) Other Non-current assets |
140.403 |
135.538 |
101.788 |
|
Total
Non-Current Assets |
6712.186 |
5481.358 |
4772.712 |
|
|
|
|
|
|
(2) Current assets |
|
|
|
|
(a) Current investments |
0.000 |
100.025 |
449.092 |
|
(b) Inventories |
3688.559 |
3423.973 |
2820.429 |
|
(c) Trade receivables |
1003.179 |
963.932 |
1158.980 |
|
(d) Cash and cash equivalents |
18980.175 |
20271.361 |
20387.791 |
|
(e) Short-term loans and
advances |
1197.432 |
699.137 |
570.279 |
|
(f) Other current assets |
782.491 |
558.895 |
737.382 |
|
Total
Current Assets |
25651.836 |
26017.323 |
26123.953 |
|
|
|
|
|
|
TOTAL |
32364.022 |
31498.681 |
30896.665 |
PROFIT
& LOSS ACCOUNT
|
|
PARTICULARS |
31.03.2015 (15
Months) |
31.12.2013 |
31.12.2012 |
|
|
SALES |
|
|
|
|
|
Revenue from Operations |
32875.780 |
25461.474 |
26264.347 |
|
|
Other Income |
1986.717 |
2009.686 |
1973.470 |
|
|
TOTAL
|
34862.497 |
27471.160 |
28237.817 |
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
Cost of Materials Consumed |
7267.346 |
5385.674 |
4378.090 |
|
|
Purchases of Stock-in-Trade |
7798.939 |
6700.011 |
6074.549 |
|
|
Changes in inventories of finished
goods, work-in-progress and Stock-in-Trade |
(13.869) |
(507.471) |
525.864 |
|
|
Employees benefits expense |
4930.223 |
3620.485 |
2955.155 |
|
|
Exceptional Items |
518.814 |
(261.546) |
1482.170 |
|
|
Other expenses |
6621.290 |
5041.990 |
4178.001 |
|
|
TOTAL
|
27122.743 |
19979.143 |
19593.829 |
|
|
|
|
|
|
|
|
PROFIT
/ (LOSS) BEFORE TAX, DEPRECIATION AND AMORTISATION |
7739.754 |
7492.017 |
8643.988 |
|
|
|
|
|
|
|
Less/
Add |
DEPRECIATION/
AMORTISATION |
253.525 |
198.814 |
178.393 |
|
|
|
|
|
|
|
|
PROFIT/ (LOSS)
BEFORE TAX |
7486.229 |
7293.203 |
8465.595 |
|
|
|
|
|
|
|
Less |
TAX |
2769.771 |
2274.379 |
2693.006 |
|
|
|
|
|
|
|
|
PROFIT/ (LOSS)
AFTER TAX |
4716.458 |
5018.824 |
5772.589 |
|
|
|
|
|
|
|
Add |
PREVIOUS
YEARS’ BALANCE BROUGHT FORWARD |
10999.327 |
11429.460 |
11105.915 |
|
|
|
|
|
|
|
Less |
APPROPRIATIONS |
|
|
|
|
|
Transfer to General Reserve |
471.646 |
501.882 |
577.259 |
|
|
Dividend |
6371.678 |
4947.075 |
4871.785 |
|
|
Total
|
6843.324 |
5448.957 |
5449.044 |
|
|
|
|
|
|
|
|
Balance
Carried to the B/S |
8872.461 |
10999.327 |
11429.460 |
|
|
|
|
|
|
|
|
EARNINGS
IN FOREIGN CURRENCY |
|
|
|
|
|
F.O.B. value of exports including through merchant
exporters |
63.118 |
94.688 |
195.929 |
|
|
Recovery of expenses |
1.310 |
3.686 |
4.643 |
|
|
Clinical research and data
management |
520.272 |
436.602 |
454.118 |
|
|
Others |
114.564 |
90.501 |
60.467 |
|
|
TOTAL
EARNINGS |
699.264 |
625.477 |
715.157 |
|
|
|
|
|
|
|
|
IMPORTS |
|
|
|
|
|
Raw and packing materials |
1161.449 |
957.543 |
602.491 |
|
|
Traded goods |
3013.910 |
2045.585 |
1423.377 |
|
|
Components and spare parts for
Machinery |
6.986 |
0.000 |
0.000 |
|
|
Capital Goods |
151.291 |
155.966 |
126.433 |
|
|
TOTAL
IMPORTS |
4333.636 |
3159.094 |
2152.301 |
|
|
|
|
|
|
|
|
Earnings
/ (Loss) Per Share (Rs.) |
55.68 |
59.25 |
68.15 |
CURRENT MATURITIES OF LONG TERM DEBT DETAILS
|
Particulars |
31.03.2015 (15
Months) |
31.12.2013 |
31.12.2012 |
|
Current Maturities of Long term debt |
4.895 |
5.374 |
4.509 |
|
Cash generated from operations |
5979.395 |
5244.447 |
8547.615 |
|
Net cash flow from operating activity |
3478.064 |
2823.914 |
5409.556 |
QUARTERLY RESULTS
|
Particulars |
30.06.2015 |
30.09.2015 |
|
Audited / Unaudited |
Unaudited |
Unaudited |
|
Net Sales |
6283.100 |
7000.900 |
|
Total Expenditure |
5183.000 |
5712.200 |
|
PBIDT (Excl OI) |
1100.100 |
1288.700 |
|
Other Income |
398.800 |
313.400 |
|
Operating Profit |
1498.900 |
1602.100 |
|
Interest |
NA |
0.000 |
|
Exceptional Items |
(23.900) |
(78.400) |
|
PBDT |
1475.000 |
1523.700 |
|
Depreciation |
49.200 |
53.300 |
|
Profit Before Tax |
1425.800 |
1470.400 |
|
Tax |
493.000 |
509.900 |
|
Provisions and contingencies |
NA |
NA |
|
Profit After Tax |
932.800 |
960.500 |
|
Extraordinary Items |
NA |
NA |
|
Prior Period Expenses |
NA |
NA |
|
Other Adjustments |
NA |
NA |
|
Net Profit |
932.800 |
960.500 |
KEY
RATIOS
|
PARTICULARS |
|
31.03.2015 (15
Months) |
31.12.2013 |
31.12.2012 |
|
Net Profit Margin (PAT / Sales) |
(%) |
14.35 |
19.71 |
21.98 |
|
|
|
|
|
|
|
Operating Profit Margin (PBIDT/Sales) |
(%) |
23.54 |
29.42 |
32.91 |
|
|
|
|
|
|
|
Return on Total Assets (PBT/Total Assets} |
(%) |
25.03 |
24.74 |
29.17 |
|
|
|
|
|
|
|
Return on Investment (ROI) (PBT/Networth) |
|
0.40 |
0.36 |
0.42 |
|
|
|
|
|
|
|
Debt Equity Ratio (Total Debt /Networth) |
|
0.00 |
0.00 |
0.00 |
|
|
|
|
|
|
|
Current Ratio (Current Asset/Current Liability) |
|
2.32 |
2.95 |
3.13 |
STOCK
PRICES
|
Face Value |
Rs.10.00/- |
|
Market Value |
Rs.3264.70/- |
FINANCIAL ANALYSIS
[all figures are in
Rupees Million]
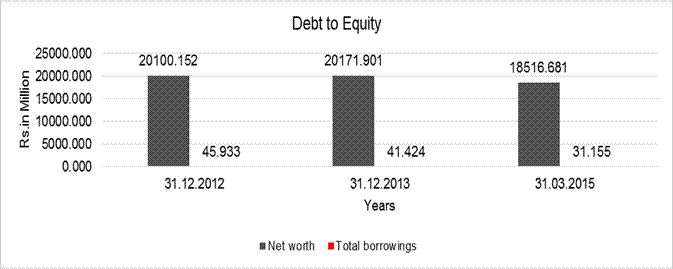
DEBT EQUITY RATIO
|
Particular |
31.12.2012 |
31.12.2013 |
31.03.2015 |
|
|
(Rs.
In Million) |
(Rs.
In Million) |
(Rs.
In Million) |
|
Share Capital |
847.030 |
847.030 |
847.030 |
|
Reserves & Surplus |
19253.122 |
19324.871 |
17669.651 |
|
Net
worth |
20100.152 |
20171.901 |
18516.681 |
|
|
|
|
|
|
long-term borrowings |
41.424 |
36.050 |
26.260 |
|
Short term borrowings |
0.000 |
0.000 |
0.000 |
|
Current Maturities Of
Long-Term Debts |
4.509 |
5.374 |
4.895 |
|
Total
borrowings |
45.933 |
41.424 |
31.155 |
|
Debt/Equity
ratio |
0.002 |
0.002 |
0.002 |
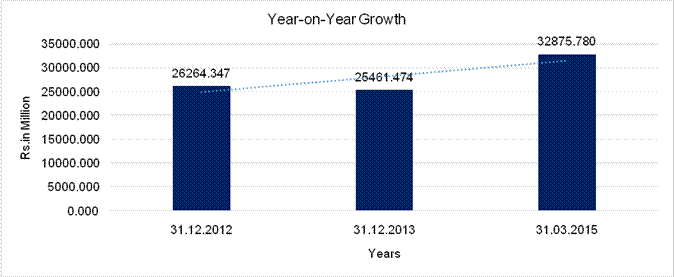
YEAR-ON-YEAR GROWTH
|
Year
on Year Growth |
31.12.2012 |
31.12.2013 |
31.03.2015 |
|
|
(Rs.
In Million) |
(Rs.
In Million) |
(Rs.
In Million) |
|
Sales |
26264.347 |
25461.474 |
32875.780 |
|
|
|
(3.057) |
29.120 |
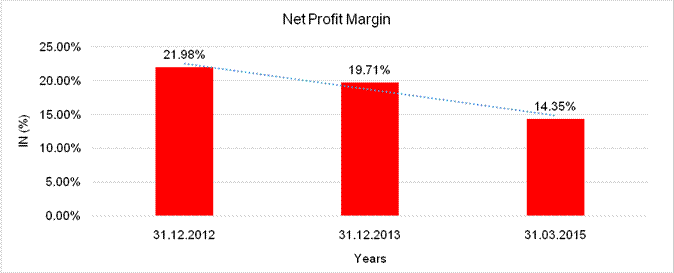
NET PROFIT MARGIN
|
Net
Profit Margin |
31.12.2012 |
31.12.2013 |
31.03.2015 |
|
|
(Rs.
In Million) |
(Rs.
In Million) |
(Rs.
In Million) |
|
Sales |
26264.347 |
25461.474 |
32875.780 |
|
Profit |
5772.589 |
5018.824 |
4716.458 |
|
|
21.98% |
19.71% |
14.35% |
LOCAL AGENCY FURTHER INFORMATION
|
Sr. No. |
Check list by
info agents |
Available in
Report (Yes/No) |
|
1 |
Year of establishment |
Yes |
|
2 |
Constitution of the entity -Incorporation
details |
Yes |
|
3 |
Locality of the entity |
Yes |
|
4 |
Premises details |
No |
|
5 |
Buyer visit details |
-- |
|
6 |
Contact numbers |
Yes |
|
7 |
Name of the person contacted |
Yes |
|
8 |
Designation of contact person |
Yes |
|
9 |
Promoter’s background |
Yes |
|
10 |
Date of Birth of Proprietor / Partners /
Directors |
Yes |
|
11 |
Pan Card No. of Proprietor / Partners |
No |
|
12 |
Voter Id Card No. of Proprietor / Partners |
No |
|
13 |
Type of business |
Yes |
|
14 |
Line of Business |
Yes |
|
15 |
Export/import details (if applicable) |
No |
|
16 |
No. of employees |
No |
|
17 |
Details of sister concerns |
Yes |
|
18 |
Major suppliers |
No |
|
19 |
Major customers |
No |
|
20 |
Banking Details |
Yes |
|
21 |
Banking facility details |
Yes |
|
22 |
Conduct of the banking account |
-- |
|
23 |
Financials, if provided |
Yes |
|
24 |
Capital in the business |
Yes |
|
25 |
Last accounts filed at ROC, if applicable |
Yes |
|
26 |
Turnover of firm for last three years |
Yes |
|
27 |
Reasons for variation <> 20% |
-- |
|
28 |
Estimation for coming financial year |
No |
|
29 |
Profitability for last three years |
Yes |
|
30 |
Major shareholders, if available |
Yes |
|
31 |
External Agency Rating, if available |
No |
|
32 |
Litigations that the firm/promoter
involved in |
-- |
|
33 |
Market information |
-- |
|
34 |
Payments terms |
No |
|
35 |
Negative Reporting by Auditors in the
Annual Report |
No |
MANAGEMENT DISCUSSION
AND ANALYSIS
Finance and Accounts
Sale
of Products (net of Excise Duty) increased by 29% for the fifteen months period
31st March 2015 as compared with the previous year twelve months period ending
31st December 2013. The fifteen months period was impacted by the full year
effect of the Prices Control Order introduced in mid 2013. The Pharmaceuticals
business continues to be affected by supply constraints mainly from local
supplies during the period to meet the demands of the market. The Gross Margin
has remained flat compared to last year mainly driven by the full period
absorption of the Price Control Order. Other expenses such as field force
recruitment were restricted to segments that required resourcing for growth and
significant investments continued to be made for the consumer brands lodex and
Ostocalcium which are marketed by GlaxoSmithKline Consumer Healthcare Limited.
Profit before Exceptional items and Tax amounted to 25% of Net Sales for the fifteen
months period, compared to the twelve months previous year of 28%. Profit after
Exceptional items and Tax for the corresponding periods amounted to 14% of Net
Sales for the fifteen months period, as compared to 20% in 2013. This is mainly
due to the profit on disposal of property in 2013 of Rs. 21.18 crores (pre-tax)
as compared to Exceptional costs of Rs. 52 crores in the current fifteen months
period. These costs include Rs. 21.51 crores relating to actuarial loss on
employee benefits and a charge of Rs. 30.37 crores for the rationalization of
capital assets for one dosage form at the Nashik facility.
Cash
generation from Operations continued to remain favourable this year and is in
line with business performance. The Company continues to look for ways and
means of deploying accumulated cash balances which remain invested largely in
bank deposits.
The
Company has not accepted any fixed deposits during the year. There was no
outstanding towards unclaimed deposit payable to depositors as on 31st March 2015.
Pharmaceuticals
Business performance and outlook
The
Company continues to enjoy a leadership position in the therapy areas in which
it provides health care solutions to patients.
The
speciality business continued to make rapid strides during the year with strong
leadership position in therapy areas like Dermatology. Within the Clinical
dermatology area, brands like Betnovate, TBact and Tenovate sustained strong
growth while the Cosmetology portfolio of brands like Ansolar, Oilatum and
Physiogel continued to benefit increasing number of patients requiring these
therapeutic options. The cornerstone of the Dermatology business continues to
be a strong patient focused approach. This division created unique platforms
for patients and physicians with programs like SKIN SENSE (aimed at enabling
patients and physicians for early diagnosis of skin problems) and SPEAK
campaign (aimed at spreading awareness for conditions like Psoriasis and
Eczema).
The
Oncology team articulated vision to improve patient care with affordability
benefited over 7000 patients during the year. Apart from the innovative
solutions in the form of key brands like Tykerb, Revolade, Votrient &
Hycamtin used to treat various types of cancer, the year saw this division
driving unique and unstinted commitment to patient care for brands like Xgeva
(indicated for Skeletal related disorders) with programs like “SWASTI” aimed at
improving patient access. This division also launched VECTIBIX (in licensed from
AMGEN), a novel innovative monoclonal Antibody for patients with certain types
of metastatic colorectal cancer.
The
Respiratory team made new inroads for patients with asthma/Chronic Obstructive
Pulmonary Disorder (COPD) with products like Seretide and Ventorlin. The “EVOHALER” device
continues to create a new mark in the category for patients with asthma with
dose counter. The Central Nervous System (CNS) team created new and unique
awareness campaigns for patients and physicians in areas like Epilepsy and
Bipolar Disorders. “Lamictal” became the 2nd largest
prescribed brand of Lamotrigine in Epilepsy. The Cardiology team continued to
focus on areas like hypertension and dyslipidemia with brands like Benitec,
Rosutec, Carzec and Volibris. This team deployed screening camps across the
country as a patient awareness initiative to enable physicians and patients to
diagnose conditions like dyslipidemia early and thereby improve treatment
outcomes.
The
Mass Speciality business continued its growth trajectory and “Augmentin” the
flagship brand of the Company regained the No. 1 position in the “Amoxy
Clav” market, showing a remarkable recovery after it was bought
under the NLEM (National List of Essential Medicines List) list and under DPCO
2013 with an annual prescription growth of 46%. The Company used the
opportunity of the price control induced downward correction to improve access
to patients and improve outcomes as is evidenced by the growth of
prescriptions. The team remains focused to enable Augmentin achieve the status
of volume leader in the market and be the highest prescribed brand in the “Amoxy
Clav” market. Eltroxin, the Company’s flagship therapeutic
solution for Thyroid management achieved an annual growth of 5%. Many recent
advances in prevention and management of Hypothyrodism need to be translated
into practice for Health Care Professionals (HCPs) in Tier 1 and Tier 2 cities.
To
help rapid and early diagnosis of Thyroid disorders, the Company undertook
several 1:1 Scientific Promotion meetings on “Maternal Thyrodism” and “Translating
of Thyroid guidelines”. As many as 2600 such
meetings took place during the year. The ‘One to One’ SPMs
were a good example in their pursuit of being a patient focused organisation
and taking a lead to ‘door deliver’
knowledge to equip HCPs for appropriate disease management. The Disease
Awareness initiative on Congenital Hypothyroidism(CH) was conducted in many
cities during the year. Feedback from doctors was encouraging as they believed
that the Company is spreading awareness in the society and thus improving the
future of children with CH. Ceftum also registered a double digit prescription
growth in major specialities areas. The new launch, “Avamys” showed a
positive value growth and became No. 6 brand amongst 52 brands in INCS market
with a market share of 5.8%.
The
key brands in Mass markets like Zinetac, Calpol, Neosporin, Betnesol, Zyloric,
Zentel, and Cetzine all have maintained dominant market leadership positions.
Brands like Betnesol and Zinetac sell almost double the number of units as
compared to total population of the country. The Company’s
strategy was to expand the access opportunities to patients by targeting the
adjacent and direct category competition and drive unit Sales consistent with
the Company’s policy of improving access to their medicines to as many
patients as possible. The rural initiative is being expanded further which will
ensure prescription leadership and support to patients in rural markets.
In
the area of preventive healthcare, GSK Vaccines continues to be the No. 1
vaccines Company in the private vaccines market in India. Again, consistent
with the Company philosophy of offering access to enable as many patients as
possible to use them, a more cost effective presentation of a flagship
pneumococcal vaccines in the form of vials was launched which will help protect
more kids from pneumococcal diseases which is one of the leading causes of
infant mortality in the country. Sudden changes in interpretation of licensing
requirements coupled with constraints of supplies from the Parent’s
facility led to shortages of number of vaccines through the year.
Notwithstanding that, the measure of the ability of the Company’s
vaccines to benefit children and women health is indicated in the healthy
growth registered. The vaccines team is gearing up for the launch of the
combined MMRV vaccine- the first of its kind to be launched in India in 2016.
Exports
recorded a sales turnover of Rs. 6.3 crores for the fifteen months period ended
31st March 2015.
GENERAL INFORMATION
Subject
is a public limited company and is listed on the BSE Ltd. (Bombay Stock
Exchange) and the National Stock Exchange of India Ltd. (NSE). The Company is
engaged interalia, in the business of manufacturing, distributing and trading
in pharmaceuticals.
UNSECURED LOAN
|
Unsecured Loan |
31.03.2015 (Rs.
in Million) |
31.03.2014 (Rs.
in Million) |
|
Long-term
Borrowings |
|
|
|
Interest free sales tax loan from SICOM Limited |
26.260 |
36.050 |
|
Total |
26.260 |
36.050 |
|
Note
: Terms of repayment Interest
free Sales Tax Loan from SICOM Limited as at 31st March, 2015 of Rs. 31.155
Million (Previous year - Rs. 41.424 Million) includes NIL (Previous year -
Rs. 0.0.480 Million repayable in one installment) availed under the 1988
Sales Tax deferment Scheme, closing on 31st January, 2014 and Rs. 31.155
Million (Previous year - Rs. 40.944 Million) under the 1993 Sales Tax
deferment Scheme repayable in twenty one instalments (previous year twenty
seven instalments) closing on 30th April, 2021. The current maturity amount
of Rs. 4.895 Million (Previous year - Rs. 5.374 Million) of the loan has been
disclosed under Note 8 - Other Current Liabilities. |
||
INDEX
OF CHARGES
|
S.No. |
Charge ID |
Date of Charge Creation/Modification |
Charge amount secured |
Charge Holder |
Address |
Service Request Number (SRN) |
|
1 |
90227070 |
20/09/2004 |
1,300,000,000.00 |
UTI BANK LTD. |
ROYAL ACCORD - IV; LOKAHANDWALA COMPLEX, ANDHERI |
- |
|
2 |
90231657 |
13/07/1989 |
218,700,000.00 |
THE HONGKONG AND SHANGHAI BANKING CORPORATION LTD. |
52/60; MG ROAD, MUMBAI, Maharashtra, INDIA |
- |
CONTINGENT
LIABILITIES:
(Rs. in million)
|
PARTICULARS |
31.03.2015 |
31.03.2014 |
|
A. Contingent Liabilities
not provided for: |
|
|
|
(i) Cheques discounted with banks |
37.400 |
15.684 |
|
(ii)
In respect of claims made against the Company not acknowledged as debts by the Company |
|
|
|
Sales tax matters |
343.935 |
341.665 |
|
Excise matters |
59.330 |
59.330 |
|
Service tax matters |
12.920 |
12.920 |
|
Labour matters |
693.123 |
628.149 |
|
Other legal matters |
201.155 |
220.155 |
|
which net of current tax amount to |
877.579 |
833.191 |
|
(iii) Income-tax matters in
respect of which appeals are pending |
|
|
|
Tax
on matters in dispute |
1774.683 |
1997.077 |
|
|
3999.692 |
4108.171 |
|
Notes: Future
cash outflows in respect of (i) above are dependant on the return of cheques
by banks. Future
cash outflows in respect of (ii) and (iii) above are determinable on receipt
of decisions / judgements pending with various forums / authorities. |
||
UNAUDITED FINANCIAL
RESULTS FOR THE QUARTER ENDED 30th SEPTEMBER 2015
(Rs. In Million)
|
|
|
Particulars |
3 Months ended
30.09.2015 |
Preceding 3 Months
ended 30.06.2015 |
6 Months ended
30.09.2015 |
|
1 |
|
Income from
Operations |
|
|
|
|
|
|
Sales/Income from Operations (Gross) |
6919.000 |
6218.500 |
13137.500 |
|
|
|
b) Other Operating Income |
81.900 |
64.600 |
146.500 |
|
|
Total Income from
Operations (Net) |
70000.900 |
6283.100 |
13284.000 |
|
|
2 |
Expenses |
|
|
|
|
|
|
a) |
Cost of Materials consumed |
1488.300 |
1355.300 |
2843.600 |
|
|
b) |
Purchase of Stock-in-trade |
2141.100 |
1825.900 |
3967.000 |
|
|
c) |
Changes in inventories of finished goods, work-in-progress and
stock-in-trade |
(614.700) |
(380.600) |
(995.300) |
|
|
d) |
Employee benefit expenses |
3014.700 |
2800.600 |
5815.300 |
|
|
e) |
Depreciation and amortization expense |
53.300 |
49.2000 |
102.500 |
|
|
f) |
Other expenses |
1664.400 |
1407.400 |
3071.800 |
|
|
g) |
Expenses relating to services income |
(93.900) |
(107.500) |
(201.400) |
|
|
Total Expenses |
2750.800 |
2431.600 |
5182.400 |
|
|
|
|
|
|
|
|
|
3 |
|
Profit /(Loss) from
operations before other income, finance costs and exceptional items |
1235.400 |
1050.900 |
2286.300 |
|
4 |
Other Income |
313.400 |
398.800 |
712.200 |
|
|
5 |
Profit /(Loss) from
ordinary activities before finance costs and exceptional items |
1548.800 |
1449.700 |
2998.500 |
|
|
6 |
Finance Costs |
-- |
-- |
-- |
|
|
7 |
Profit /(Loss) from
ordinary activities after finance costs but before exceptional items |
1548.800 |
1449.700 |
2998.500 |
|
|
8 |
Exceptional Items |
(78.400) |
(23.900) |
(102.300) |
|
|
9 |
Profit /(Loss) from
ordinary activities before tax |
1470.400 |
1425.800 |
2896.200 |
|
|
10 |
Tax Expense |
509.900 |
493.000 |
1002.900 |
|
|
11 |
Net Profit /(Loss)
from ordinary activities after tax |
960.500 |
932.800 |
1893.300 |
|
|
12 |
Paid up equity share capital (Eq. shares of Rs.10/- each) |
847.000 |
947.00 |
847.000 |
|
|
13 |
Reserve excluding revaluation reserves |
- |
- |
- |
|
|
14 |
|
Earnings per share (before/after extraordinary items) of Rs.10/- each |
|
|
|
|
|
|
Basic & Diluted |
11.30 |
11.00 |
22.40 |
|
|
|
|
|
|
|
|
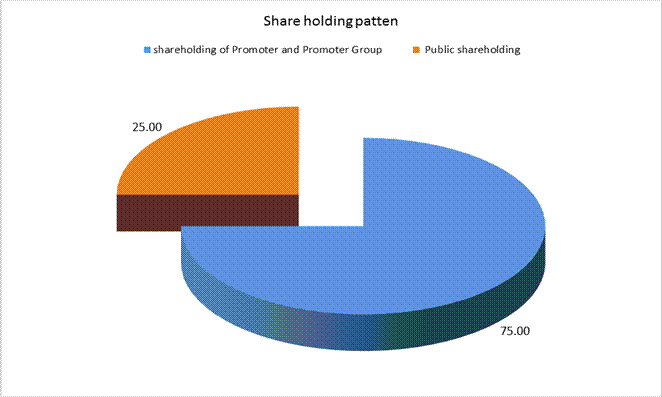
A |
|
PARTICULARS OF SHAREHOLDING |
|
|
|
|
1 |
|
Public Shareholding |
|
|
|
|
|
|
- No. of Shares |
21175755 |
21175755 |
21175755 |
|
|
|
- Percentage of Shareholding |
25.00% |
25.00% |
25.00% |
|
2 |
|
Promoters and promoter group shareholding |
|
|
|
|
|
|
a) Pledged/Encumbered |
|
|
|
|
|
|
- Number of shares |
Nil |
Nil |
Nil |
|
|
|
- Percentage of shares ( as a % of the total shareholding of the promoter and promoter group) |
Nil |
Nil |
Nil |
|
|
|
- Percentage of shares (as a % of the total share capital of the Company) |
Nil |
Nil |
Nil |
|
|
|
b) Non- encumbered |
|
|
|
|
|
|
- Number of shares |
63527262 |
63527262 |
63527262 |
|
|
|
- Percentage of shares ( as a % of the total shareholding of the promoter and promoter group) |
100.00% |
100.00% |
100.00% |
|
|
|
- Percentage of shares (as a % of the total share capital of the Company) |
75.00% |
75.00% |
75.00% |
|
Particulars |
3 Month Ended 30.09.2015 |
|
Pending at the beginning of the quarter |
3 |
|
Received during the quarter |
4 |
|
Disposed of during the quarter |
4 |
|
Remaining unresolved at the end of the
quarter |
3 |
STATEMENT OF ASSETS ANS LIABILITIES AS ON 30TH SEPTEMBER
2015
|
SOURCES
OF FUNDS |
30.09.2015 |
|
|
(Half
Year ended) |
|
|
|
|
I.
EQUITY AND LIABILITIES |
|
|
(1)Shareholders' Funds |
|
|
(a) Share Capital |
847.000 |
|
(b) Reserves & Surplus |
19562.900 |
|
(c) Money received against
share warrants |
0.000 |
|
|
|
|
(2) Share Application money
pending allotment |
0.000 |
|
Total
Shareholders’ Funds (1) + (2) |
20409.900 |
|
|
|
|
(3) Non-Current Liabilities |
|
|
(a) long-term borrowings |
19.900 |
|
(b) Deferred tax liabilities
(Net) |
0.000 |
|
(c) Other long term
liabilities |
44.700 |
|
(d) long-term provisions |
2691.700 |
|
Total
Non-current Liabilities (3) |
2756.300 |
|
|
|
|
(4) Current Liabilities |
|
|
(a) Short term borrowings |
0.000 |
|
(b) Trade payables |
3099.100 |
|
(c) Other current liabilities |
1517.700 |
|
(d) Short-term provisions |
110.400 |
|
Total
Current Liabilities (4) |
4727.200 |
|
|
|
|
TOTAL |
27893.400 |
|
|
|
|
II.
ASSETS |
|
|
(1) Non-current assets |
|
|
(a) Fixed Assets |
|
|
(i) Tangible assets |
3627.400 |
|
(ii) Intangible Assets |
0.000 |
|
(iii) Capital work-in-progress |
0.000 |
|
(iv) Intangible assets under
development |
0.000 |
|
(b) Non-current Investments |
476.700 |
|
(c) Deferred tax assets (net) |
820.500 |
|
(d) Long-term Loan and Advances |
2247.800 |
|
(e) Other Non-current assets |
137.800 |
|
Total
Non-Current Assets |
7310.200 |
|
|
|
|
(2) Current assets |
|
|
(a) Current investments |
0.000 |
|
(b) Inventories |
4638.400 |
|
(c) Trade receivables |
1156.100 |
|
(d) Cash and cash equivalents |
12916.400 |
|
(e) Short-term loans and
advances |
1304.600 |
|
(f) Other current assets |
567.700 |
|
Total
Current Assets |
20583.200 |
|
|
|
|
TOTAL |
27893.400 |
- There was a decline in Net Sales of the Pharmaceuticals business by 8.4% during the quarter ended 30th September 2015, as compared to the quarter ended 30th September 2014. The quarterly performance was Impacted by the supply constraints.
- On 22nd April 2014, GlaxoSmithKline Plc (GSK), London, UK, entered into an inter-conditional agreement with Novartis AG (Novartis), Basel, Switzerland where GSK (i) will acquire the Novartis's vacones Business and manufacturing capabilities and facilities from Novartis, and (ii) GSK sell the rights to Its Marketed Oncology Portfolio, related R&D activities and AKT Inhibitors currently In development to Novartis.
Pursuant to the global deal, the Board In Its meeting held on 12th February 2015, had approved the transactions on an Asset Sale basis with Novartis Healthcare Private Limited ('Novartis India'), a private unlisted Company incorporated under the Companies Act 1956 subject to the receipt of all applicable legal and regulatory approvals, consent, permissions and sanctions as may be necessary from concerned authorities. The Company has completed this transaction on September 30, 2015 on receipt of all applicable legal and regulatory approvals. The transaction is not material and is profit neutral for the Company.
3. Exceptional Items for the current quarter ended September 30, 2015 pertains to a net charge of Rs. 217 Lakhs for the rationalisation of manufacturing activities and a net charge of Rs. 566 Lakhs mainly relating to accounting of the distribution rights for the Asset Sale transactions with Novartis India, as referred to in Note 2 above.
4. As the Company's accounting year has been changed from January - December to April - March, the financial results for the current period are for six months ended 30th September, 2015. The comparable results for the previous period are for six months ended 30th September, 2014 which are balancing figures between the unaudited financial results for the nine months ended 30th September, 2014 and quarter ended 31st March, 2014.
5. The Company has only one reportable segment which is Pharmaceuticals. Accordingly, no separate disclosures of segment Information have been made.
6. The above Results were reviewed by the Audit Committee and were thereafter approved by the Board of Directors at their respective meetings held on 31st October, 2015.
7. The statutory auditors have carried out a limited review of the standalone
results for the quarter ended 30th September 2015.
8. The figures for 2014 have been regrouped wherever necessary to facilitate
comparison.
FIXED ASSETS
CMT REPORT (Corruption, Money Laundering & Terrorism]
The Public Notice information has been collected from various sources
including but not limited to: The Courts,
1] INFORMATION ON
DESIGNATED PARTY
No records exist designating subject or any of its beneficial owners,
controlling shareholders or senior officers as terrorist or terrorist
organization or whom notice had been received that all financial transactions
involving their assets have been blocked or convicted, found guilty or against
whom a judgement or order had been entered in a proceedings for violating
money-laundering, anti-corruption or bribery or international economic or
anti-terrorism sanction laws or whose assets were seized, blocked, frozen or
ordered forfeited for violation of money laundering or international
anti-terrorism laws.
2] Court Declaration :
No records exist to suggest that subject is
or was the subject of any formal or informal allegations, prosecutions or other
official proceeding for making any prohibited payments or other improper
payments to government officials for engaging in prohibited transactions or
with designated parties.
3] Asset Declaration :
No records exist to suggest that the property or assets of the subject
are derived from criminal conduct or a prohibited transaction.
4] Record on Financial
Crime :
Charges or conviction
registered against subject: None
5] Records on Violation of
Anti-Corruption Laws :
Charges or
investigation registered against subject: None
6] Records on Int’l
Anti-Money Laundering Laws/Standards :
Charges or
investigation registered against subject: None
7] Criminal Records
No available
information exist that suggest that subject or any of its principals have been
formally charged or convicted by a competent governmental authority for any
financial crime or under any formal investigation by a competent government
authority for any violation of anti-corruption laws or international anti-money
laundering laws or standard.
8] Affiliation with
Government :
No record
exists to suggest that any director or indirect owners, controlling
shareholders, director, officer or employee of the company is a government
official or a family member or close business associate of a Government
official.
9] Compensation Package :
Our market
survey revealed that the amount of compensation sought by the subject is fair
and reasonable and comparable to compensation paid to others for similar
services.
10] Press Report :
No press reports / filings exists on
the subject.
CORPORATE GOVERNANCE
MIRA INFORM as part of its Due Diligence do provide comments on
Corporate Governance to identify management and governance. These factors often
have been predictive and in some cases have created vulnerabilities to credit
deterioration.
Our Governance Assessment focuses principally on the interactions
between a company’s management, its Board of Directors, Shareholders and other
financial stakeholders.
CONTRAVENTION
Subject is not known to have contravened any existing local laws,
regulations or policies that prohibit, restrict or otherwise affect the terms
and conditions that could be included in the agreement with the subject.
FOREIGN EXCHANGE RATES
|
Currency |
Unit
|
Indian Rupees |
|
US Dollar |
1 |
Rs.66.20 |
|
|
1 |
Rs.98.29 |
|
Euro |
1 |
Rs.72.41 |
INFORMATION DETAILS
|
Information
Gathered by : |
GYT |
|
|
|
|
Analysis Done by
: |
TRI |
|
|
|
|
Report Prepared
by : |
SUJ |
SCORE & RATING EXPLANATIONS
|
SCORE FACTORS |
RANGE |
POINTS |
|
HISTORY |
1~10 |
10 |
|
PAID-UP CAPITAL |
1~10 |
9 |
|
OPERATING SCALE |
1~10 |
10 |
|
FINANCIAL
CONDITION |
|
|
|
--BUSINESS SCALE |
1~10 |
10 |
|
--PROFITABILITY |
1~10 |
9 |
|
--LIQUIDITY |
1~10 |
9 |
|
--LEVERAGE |
1~10 |
9 |
|
--RESERVES |
1~10 |
9 |
|
--CREDIT LINES |
1~10 |
10 |
|
--MARGINS |
-5~5 |
-- |
|
DEMERIT POINTS |
|
|
|
--BANK CHARGES |
YES/NO |
YES |
|
--LITIGATION |
YES/NO |
NO |
|
--OTHER ADVERSE INFORMATION |
YES/NO |
NO |
|
MERIT POINTS |
|
|
|
--SOLE DISTRIBUTORSHIP |
YES/NO |
NO |
|
--EXPORT ACTIVITIES |
YES/NO |
NO |
|
--AFFILIATION |
YES/NO |
YES |
|
--LISTED |
YES/NO |
NO |
|
--OTHER MERIT FACTORS |
YES/NO |
YES |
|
DEFAULTER |
|
|
|
--RBI |
YES/NO |
NO |
|
--EPF |
YES/NO |
NO |
|
TOTAL |
|
85 |
This score serves as a reference to assess
SC’s credit risk and to set the amount of credit to be extended. It is
calculated from a composite of weighted scores obtained from each of the major
sections of this report. The assessed factors and their relative weights (as
indicated through %) are as follows:
Financial
condition (40%) Ownership
background (20%) Payment
record (10%)
Credit history
(10%) Market trend (10%) Operational size
(10%)
RATING EXPLANATIONS
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
>86 |
Aaa |
Possesses an extremely sound financial base with the strongest
capability for timely payment of interest and principal sums |
Unlimited |
|
71-85 |
Aa |
Possesses adequate working capital. No caution needed for credit
transaction. It has above average (strong) capability for payment of interest
and principal sums |
Large |
|
56-70 |
A |
Financial & operational base are regarded healthy. General unfavourable
factors will not cause fatal effect. Satisfactory capability for payment of
interest and principal sums |
Fairly Large |
|
41-55 |
Ba |
Overall operation is considered normal. Capable to meet normal
commitments. |
Satisfactory |
|
26-40 |
B |
Capability to overcome financial difficulties seems comparatively
below average. |
Small |
|
11-25 |
Ca |
Adverse factors are apparent. Repayment of interest and principal sums
in default or expected to be in default upon maturity |
Limited with full
security |
|
<10 |
C |
Absolute credit risk exists. Caution needed to be exercised |
Credit not
recommended |
|
-- |
NB |
New Business |
-- |
This report is issued at your request without any
risk and responsibility on the part of MIRA INFORM PRIVATE LIMITED (MIPL) or
its officials.