MIRA INFORM REPORT
|
Report No. : |
306479 |
|
Report Date : |
05.02.2015 |
IDENTIFICATION DETAILS
|
Name : |
JUBILANT LIFE SCIENCES LIMITED (w.e.f.01.10.2010) |
|
|
|
|
Formerly Known
As : |
JUBILANT ORGANOSYS LIMITED |
|
|
|
|
Registered
Office : |
Bhartiagram, Gajraula, Jyotiba Phoolay Nagar – 244223, Uttar Pradesh |
|
|
|
|
Country : |
India |
|
|
|
|
Financials (as
on) : |
31.03.2014 |
|
|
|
|
Date of
Incorporation : |
21.06.1978 |
|
|
|
|
Com. Reg. No.: |
20-004624 |
|
|
|
|
Capital Investment
/ Paid-up Capital : |
Rs. 154.460 Millions |
|
|
|
|
CIN No.: [Company Identification
No.] |
L24116UP1978PLC004624 |
|
|
|
|
IEC No.: |
Not Available |
|
|
|
|
TAN No.: [Tax Deduction &
Collection Account No.] |
MRTJ00275C |
|
|
|
|
PAN No.: [Permanent Account No.] |
AABCV0200H |
|
|
|
|
Legal Form : |
A Public Limited Liability Company. The Company’s Shares are Listed on
the Stock Exchanges. |
|
|
|
|
Line of Business
: |
Subject is a global
Pharmaceutical and Life Sciences player engaged in manufacture and supply of
Generics (Including Active Pharmaceuticals Ingredients (APIs) and Solid
Dosage Formulations) and Life Science Ingredients (Including Proprietary
Products and Exclusive Synthesis, Nutrition Ingredients and Life Sciences
Chemicals). |
|
|
|
|
No. of Employees
: |
623 (Approximately) |
RATING & COMMENTS
|
MIRA’s Rating : |
Ba (54) |
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
41-55 |
Ba |
Overall operation is considered normal. Capable to meet normal
commitments. |
Satisfactory |
|
Maximum Credit Limit : |
USD 50000000 |
|
|
|
|
Status : |
Satisfactory |
|
|
|
|
Payment Behaviour : |
Regular |
|
|
|
|
Litigation : |
Clear |
|
|
|
|
Comments : |
Subject is a well-established integrated pharmaceutical and life
sciences company with presence in India, North America, Europe and China
having satisfactory track. The management has reported a consecutive loss for past two years as
on Financial Year 2013, due to which the position of the reserves has seen a
slight deterioration, whereas the subject has reported a minimal net
profitability during Financial Year 2014. However, the company possesses a healthy business profile, supported
by its strong domestic market position in custom research and manufacturing
services. The ratings also take into consideration adequate liquidity position
maintaining sufficient cash balance during 2014. Trade relations are fair. Business is active. Payment terms are
reported as regular and as per commitment. In view of leadership position in niche products, the subject can be
considered normal for business dealings at usual trade terms and conditions. |
NOTES :
Any query related to this report can be made
on e-mail : infodept@mirainform.com
while quoting report number, name and date.
ECGC Country Risk Classification List – December 31, 2014
|
Country Name |
Previous Rating (30.09.2014) |
Current Rating (31.12.2014) |
|
India |
A1 |
A1 |
|
Risk Category |
ECGC
Classification |
|
Insignificant |
A1 |
|
Low |
A2 |
|
Moderate |
B1 |
|
High |
B2 |
|
Very High |
C1 |
|
Restricted |
C2 |
|
Off-credit |
D |
EXTERNAL AGENCY RATING
|
Rating Agency Name |
INDIA RATINGS AND RESEARCH |
|
Rating |
Long Term Rating=A+ |
|
Rating Explanation |
Adequate degree of safety and low credit
risk. |
|
Date |
20.01.2014 |
|
Rating Agency Name |
INDIA RATINGS AND RESEARCH |
|
Rating |
Short Term Rating=A1+ |
|
Rating Explanation |
Very strong degree of safety and lowest
credit risk. |
|
Date |
20.01.2014 |
RBI DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available RBI Defaulters’ list.
EPF (Employee Provident Fund) DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available EPF (Employee Provident Fund) Defaulters’ list as of
31-03-2014.
LOCATIONS
|
Registered Office/ Factory 1 : |
Bhartiagram, Gajraula, Jyotiba Phoolay Nagar – 244223, Uttar Pradesh,
India |
|
Tel. No.: |
91-5924-252351-60 |
|
Fax No.: |
91-5924-252352 |
|
E-Mail : |
|
|
Website : |
|
|
|
|
|
Corporate Office : |
Plot No.1A, Sector – 16-A, Noida–201301, Uttar Pradesh, India. |
|
Tel. No.: |
91-120-4361000 |
|
Fax No.: |
91-120-4234881/ 84/ 85/ 87/ 95/ 96 |
|
E-Mail : |
|
|
|
|
|
Mumbai Office: |
|
|
|
|
|
Factory 2 : |
Village
Nimbut, Near Nira Railway Station, District Pune, |
|
Tel. No.: |
91-2112-269155-57 |
|
Fax No.: |
91-2112-269154 |
|
|
|
|
Factory 3 : |
Block
No. 133, P. O. Samlaya, Savli Jarod Road, Taluka Savli, Vadodara–391520,
Gujarat, India |
|
Tel. No.: |
91-2667-251361/
251563-4 |
|
Fax No.: |
91-2667-251305 |
|
|
|
|
Factory 4 : |
Village
Sikanderpur Bhainswal Bhagwanpur, Roorkee, District Haridwar, Uttrakhand,
India |
|
Tel. No.: |
91-332-235161-66 |
|
Fax No.: |
91-332-235169 |
|
|
|
|
Factory 5 : |
N-34,
MIDC, Anand Nagar, Ambernath - 421506, Maharashtra, India |
|
Tel. No.: |
91-251-2620437/
438 |
|
Fax No.: |
91-251-2620439 |
|
|
|
|
Factory 6 : |
SEZ,
Bharuch Plot No.5, Vilayat GIDC, Taluka Vagra, Bharuch – 392012,
Gujarat, India |
|
Tel. No.: |
91-2641-281500 (30 Lines) |
|
Fax No.: |
91-2641-281515 |
|
|
|
|
International Manufacturing
Facilities : |
Add : Jubilant DraxImage, Inc 16751, Tel. : 91-514- 630 7030 Fax : 91-514 -694 9295 Add : Jubilant HollisterStier LLC 3525, N. Regal, Tel. : 91- 509 -489 5656 Fax : 91- 509 -484 4320 Add : Jubilant Cadista Pharmaceuticals Tel. : 91- 410- 860 8500 Fax : 91- 410- 860 8719
|
|
|
|
|
R and D
Facilities : |
Located at:
UTTAR PRADESH (CENTRAL R&D) · C-26, Sector 59, Noida - 201 301, India · D-12, Sector 59, Noida - 201 301, India · C- 46, Sector 62, Noida - 201 301, India UTTAR PRADESH (GAJRAULA R&D) Bhartiagram, Gajraula - 244 223, District Jyotiba Phoolay Nagar, India KARNATAKA (NANJANGUD R&D) 51-56, KIADB Industrial Area, Nanjangud, District Mysore - 571 302, India GUJARAT (SAVLI R&D) Block 133, Village Samalaya, Taluka Savli, District Vadodara - 391 520, India |
|
|
|
|
Branch Offices :
|
Located at: ·
Lucknow · Chennai · Hyderabad · Bangalore · Mumbai · Pune ·
Ahmedabad ·
Kolkata |
DIRECTORS
As on 31.03.2014
|
Name : |
Mr. Shyam S Bhartia |
|
Designation : |
Chairman and Managing Director |
|
|
|
|
Name : |
Mr. Hari S Bhartia |
|
Designation : |
Co-Chairman and Managing Director |
|
|
|
|
Name : |
Mr. Shyamsundar Bang |
|
Designation : |
Executive Director (Manufacturing and Supply Chain Operations) |
|
Qualification : |
M. Tech (Chem Engg.) |
|
|
|
|
Name : |
Mr. Abhay Havaldar |
|
Designation : |
Director |
|
|
|
|
Name : |
Mr. Shardul S Shroff |
|
Designation : |
Director |
|
|
|
|
Name : |
Dr. Inder Mohan Verma |
|
Designation : |
Director |
|
|
|
|
Name : |
Mr. Suresh Kumar |
|
Designation : |
Director |
|
|
|
|
Name : |
Mr. S Sridhar |
|
Designation : |
Director |
|
|
|
|
Name : |
Ms. Sudha Pillai |
|
Designation : |
Director |
KEY EXECUTIVES
|
Name : |
Mr. Lalit Jain |
|
Designation : |
Company Secretary |
|
|
|
|
Name : |
Mr. Pramod Yadav |
|
Designation : |
CEO [Advance Intermediates and Nutritional Products] |
|
|
|
|
Name : |
Mr. Rajesh Srivastava |
|
Designation : |
CEO [Fine Chemicals and CRAMS] |
|
|
|
|
Name : |
Mr. Neeraj Agrawal |
|
Designation : |
CEO [Generics] |
|
|
|
|
Name : |
Mr. Marcelo Morales |
|
Designation : |
CEO [Contract Manufacturing and Services, Jubilant HollisterStier] |
|
|
|
|
Name : |
Mr. Scott Delaney |
|
Designation : |
CEO [Jubilant Cadista] |
|
|
|
|
Name : |
Mr. Chandan Singh |
|
Designation : |
President [Acetyls and Ethanol] |
|
|
|
|
Name : |
Mr. Martyn Coombs |
|
Designation : |
President [Jubilant DraxImage] |
|
|
|
|
Name : |
Mr. Kevin Garrity |
|
Designation : |
President [Allergy Business] |
|
|
|
|
Name : |
Dr. Vijayesh Kumar Gupta |
|
Designation : |
President [Branded Generics – India] |
|
|
|
|
Name : |
Dr. Subir Kumar Basak |
|
Designation : |
President [Jubilant Drug Discovery Services (Jubilant Biosys and
Jubilant Chemsys)] |
|
|
|
|
Name : |
Mr. Nayan Nanavati |
|
Designation : |
CEO [Jubilant Clinsys] |
|
|
|
|
Name : |
Dr. Ashutosh Agarwal |
|
Designation : |
Chief Scientific Officer [Chemicals and Life Science Ingredients] |
|
|
|
|
Name : |
Dr. Goutam Muhuri |
|
Designation : |
President [RandD - Dosage Forms] |
MAJOR SHAREHOLDERS / SHAREHOLDING PATTERN
As on 31.12.2014
|
Category of
Shareholder |
Total No. of Shares |
As a % |
|
(A) Shareholding of Promoter and Promoter Group |
|
|
|
|
|
|
|
|
1903435 |
1.20 |
|
|
78577176 |
49.33 |
|
|
80480611 |
50.53 |
|
|
|
|
|
|
5570445 |
3.50 |
|
|
5570445 |
3.50 |
|
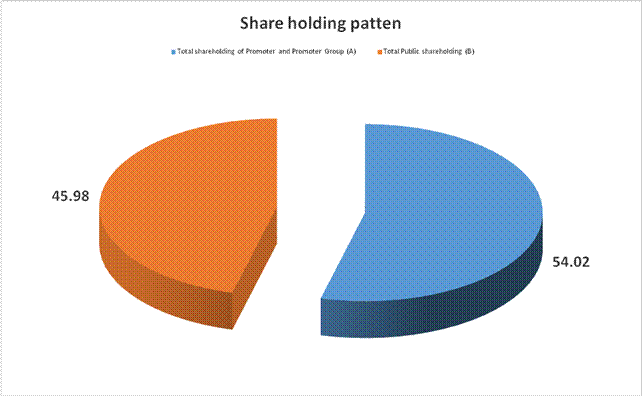
Total shareholding of Promoter and Promoter Group (A) |
86051056 |
54.02 |
|
(B) Public Shareholding |
|
|
|
|
|
|
|
|
102428 |
0.06 |
|
|
1131237 |
0.71 |
|
|
24619389 |
15.46 |
|
|
11380339 |
7.14 |
|
|
11380339 |
7.14 |
|
|
37233393 |
23.38 |
|
|
|
|
|
|
10453290 |
6.56 |
|
|
|
|
|
|
13501934 |
8.48 |
|
|
383540 |
0.24 |
|
|
468782 |
0.29 |
|
|
11189144 |
7.02 |
|
|
1137395 |
0.71 |
|
|
5098545 |
3.20 |
|
|
4953204 |
3.11 |
|
|
35996690 |
22.60 |
|
Total Public shareholding (B) |
73230083 |
45.98 |
|
Total (A)+(B) |
159281139 |
100.00 |
|
(C) Shares held by Custodians and against which Depository
Receipts have been issued |
0 |
0.00 |
|
|
0 |
0.00 |
|
|
0 |
0.00 |
|
|
0 |
0.00 |
|
Total (A)+(B)+(C) |
159281139 |
0.00 |
BUSINESS DETAILS
|
Line of Business : |
Subject is a
global Pharmaceutical and Life Sciences player engaged in manufacture and
supply of Generics (Including Active Pharmaceuticals Ingredients (APIs) and
Solid Dosage Formulations) and Life Science Ingredients (Including
Proprietary Products and Exclusive Synthesis, Nutrition Ingredients and Life
Sciences Chemicals). |
|
|
|
|
Products : |
Not Available |
|
|
|
|
Brand Names : |
Not Available |
|
|
|
|
Agencies Held : |
Not Available |
|
|
|
|
Exports : |
Not Divulged |
|
|
|
|
Imports : |
Not Divulged |
|
|
|
|
Terms : |
Not Divulged |
GENERAL INFORMATION
|
Suppliers : |
Not Divulged |
|||||||||||||||||||||||||||||||||
|
|
|
|||||||||||||||||||||||||||||||||
|
Customers : |
Not Divulged |
|||||||||||||||||||||||||||||||||
|
|
|
|||||||||||||||||||||||||||||||||
|
No. of Employees : |
623 (Approximately) |
|||||||||||||||||||||||||||||||||
|
|
|
|||||||||||||||||||||||||||||||||
|
Bankers : |
· Central Bank of India · Corporation Bank · Export Import Bank of India · ICICI Bank Limited · ING Vysya Bank Limited · Punjab National Bank · State Bank of India · The Hong Kong and Shanghai Bank Corporation Limited
|
|||||||||||||||||||||||||||||||||
|
|
|
|||||||||||||||||||||||||||||||||
|
Facilities : |
(Rs.
In Millions)
LONG-TERM BORROWINGS Nature of security of long term borrowings and other terms
of repayment
SHORT TERM BORROWINGS Nature of
security of short term borrowings and other terms of repayment:
|
|
Statutory Auditors : |
|
|
Name : |
K. N. Gutgutia and Company Chartered Accountants |
|
Address : |
11K, Gopala Tower, 25, Rajendra Place, New Delhi - 110008, India |
|
|
|
|
IFRS Auditors : |
|
|
Name : |
KPMG |
|
Address : |
Building No. 10, 8th Floor, Tower B, DLF Cyber City, Phase-II, Gurgaon
- 122002 Haryana, India |
|
|
|
|
Cost Auditors : |
|
|
Name : |
J K Kabra and Company Cost Accountants |
|
Address : |
552/1B, Arjun Street, Main Vishwas Road, Vishwas Nagar, Delhi -
110032, India |
|
|
|
|
Internal Auditors : |
|
|
Name : |
Ernst and Young Private Limited Chartered Accountants |
|
Address : |
Hindustan Times Building, 6th Floor, 18-20, Kasturba Gandhi Marg, New
Delhi - 110001, India |
|
|
|
|
Memberships : |
Not Available |
|
|
|
|
Collaborators : |
Not Available |
|
|
|
|
Subsidiaries including Step-down subsidiaries: |
·
Jubilant Pharma Pte Limited ·
Draximage Limited Cyprus ·
Draximage Limited, Ireland ·
Draximage LLC ·
Jubilant DraxImage (USA) Inc. ·
Deprenyl Inc., USA ·
Jubilant DraxImage Inc. ·
6963196 Canada Inc. ·
6981364 Canada Inc ·
DAHI Animal Health (UK) Limited ·
Draximage (UK) Limited ·
Jubilant Life Sciences Holdings Inc. ·
Jubilant Clinsys Inc. ·
Cadista Holdings Inc. ·
Jubilant Cadista Pharmaceuticals Inc. ·
Jubilant Life Sciences International Pte. Limited ·
HSL Holdings Inc. ·
Jubilant HollisterStier LLC ·
Jubilant Life Sciences (Shanghai) Limited ·
Jubilant Pharma NV ·
Jubilant Pharmaceuticals NV ·
PSI Supply NV ·
Jubilant Life Sciences (USA) Inc. ·
Jubilant Life Sciences (BVI) Limited ·
Jubilant Biosys (BVI) Limited ·
Jubilant Biosys (Singapore) Pte. Limited ·
Jubilant Biosys Limited, ·
Jubilant Discovery Services, Inc. ·
Jubilant Drug Development Pte. Limited ·
Jubilant Chemsys Limited ·
Jubilant Clinsys Limited ·
Jubilant Infrastructure Limited ·
Jubilant First Trust Healthcare Limited ·
Asia Healthcare Development Limited ·
Jubilant Innovation (BVI) Limited ·
Jubilant Innovation Pte. Limited ·
Jubilant DraxImage Limited India ·
Jubilant Innovation (India) Limited ·
Jubilant Innovation (USA) Inc ·
Jubilant HollisterStier Inc. ·
Draxis Pharma LLC ·
Generic Pharmaceuticals Holdings, Inc. ·
Jubilant Life Sciences (Switzerland) AG ·
First Trust Medicare Private Limited ·
Jubilant Drug Discovery and Development Services
Inc. ·
Vanthys Pharmaceutical Development Private
Limited |
|
|
|
|
Other Entities: |
·
Jubilant HollisterStier General Partnership Canada,
·
Draximage General Partnership Canada (controlled
through subsidiaries/step down subsidiaries). |
|
|
|
|
Enterprise over which certain key management personnel have
significant influence: |
·
Jubilant Enpro Private Limited ·
Jubilant Oil and Gas Private Limited ·
Jubilant FoodWorks Limited ·
Tower Promoters Private Limited ·
B&M Hot Breads Private Limited ·
Jubilant Industries Limited ·
Jubilant Agri and Consumer Products Limited ·
Jubilant Motors Private Limited, ·
Jubilant Aeronautics Private Limited ·
Sankur Chalets Private Limited |
|
|
|
|
Others: |
·
Vam Employees Provident Fund Trust ·
Jubilant Employee Welfare Trust ·
Jubilant Bhartia Foundation ·
Vam Officers Superannuation Fund |
CAPITAL STRUCTURE
As on 02.09.2014
Authorised Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
655,000,000 |
Equity Shares |
Re. 1/- each |
Rs. 655.000 Millions |
|
|
|
|
|
Paid-up ,Issued and Subscribed Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
159,281,139 |
Equity Shares |
Re. 1/- each |
Rs. 159.281
Millions |
|
|
|
|
|
As on 31.03.2014
Authorised Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
655,000,000 |
Equity Shares |
Re. 1/- each |
Rs. 655.000 Millions |
|
|
|
|
|
Issued and Subscribed Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
159,313,139 |
Equity Shares |
Re. 1/- each |
Rs. 159.310
Millions |
|
|
|
|
|
Paid-up Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
159,281,139 |
Equity Shares |
Re. 1/- each |
Rs. 159.280
Millions |
|
|
Add: Equity shares forfeited (paid up) |
|
Rs. 0.020
Million |
|
|
Less: Shares held in trust for employees under ESOP Scheme |
|
Rs. 4.840
Millions |
|
|
Total |
|
Rs. 154.460 Millions |
Paid
up capital includes, 501,364, equity shares of Rs. 1 each allotted and issued
pursuant to the Scheme of Amalgamation and Demerger, to the shareholders of
erstwhile Pace Marketing Specialties Limited for consideration other than cash
during the year ended 31 March 2011
The
Company has only one class of shares referred to as equity shares having par
value of Rs. 1 each. Holder of each equity share is entitled to one vote per
share. In the event of liquidation of the Company, the holders of equity shares
will be entitled to receive any of the remaining assets of the Company, after
distribution of all preferential amounts. However, no such preferential amounts
exist currently. The distribution will be in proportion to the number of equity
shares held by the shareholders.
The details of shareholders holding more than 5% shares is
set out below:
|
Name of the
shareholder |
31.03.2014 |
|
|
|
No of shares |
% held |
|
Jubilant Stock Holding Private Limited |
29676992 |
18.63% |
|
SSB Consultants and Management Services Private Limited |
21007665 |
13.19% |
|
HSB Corporate Consultants Private Limited |
18698979 |
11.74% |
|
GA Global Investments Limited |
11707200 |
7.35% |
The reconciliation of the shares outstanding at the beginning
and at the end of the year:
|
Particulars |
31.03.2014 |
|
|
|
No of shares |
Rs. in Millions |
|
At the beginning and end of the year |
159281139 |
159.280 |
a)
114,835 (Previous year 114,835), equity shares of Rs. 1 each allotted on exercise
of the vested stock options in accordance with the terms of exercise under the
“Jubilant Employees Stock Option Plan, 2005”.
b)
Under the Jubilant Employees Stock Option 2005 Plan, as at 31 March 2014 -
132,684 (Previous year 145,443) options are outstanding convertible into
663,420 (Previous year 727,215) shares.
c)
Under the Jubilant Employees Stock Option 2011 Plan, as at 31 March 2014 -
1,428,939 (Previous year 1,585,055) options are outstanding convertible into
1,428,939 (Previous year 1,585,055) shares.
The Company declares and pays dividends in Indian rupees. The dividend
proposed by the Board of Directors is subject to the approval of the
shareholders in the ensuing Annual General Meeting.
FINANCIAL DATA
[all figures are in
Rupees Millions]
ABRIDGED BALANCE
SHEET
|
SOURCES OF FUNDS |
31.03.2014 |
31.03.2013 |
31.03.2012 |
|
I.
EQUITY
AND LIABILITIES |
|
|
|
|
(1)Shareholders' Funds |
|
|
|
|
(a) Share Capital |
154.460 |
159.300 |
159.300 |
|
(b) Reserves & Surplus |
17173.080 |
18276.580 |
19011.680 |
|
(c) Money
received against share warrants |
0.000 |
0.000 |
0.000 |
|
|
|
|
|
|
(2) Share Application money pending
allotment |
0.000 |
0.000 |
0.000 |
|
Total
Shareholders’ Funds (1) + (2) |
17327.540 |
18435.880 |
19170.980 |
|
|
|
|
|
|
(3)
Non-Current Liabilities |
|
|
|
|
(a) long-term borrowings |
11410.500 |
20512.530 |
24222.120 |
|
(b) Deferred tax liabilities (Net) |
1734.200 |
2601.600 |
2114.130 |
|
(c) Other long term
liabilities |
104.060 |
50.470 |
0.000 |
|
(d) long-term
provisions |
2104.130 |
2273.500 |
1568.850 |
|
Total Non-current
Liabilities (3) |
15352.890 |
25438.100 |
27905.100 |
|
|
|
|
|
|
(4)
Current Liabilities |
|
|
|
|
(a) Short term
borrowings |
10971.260 |
10426.990 |
4847.320 |
|
(b) Trade
payables |
5992.450 |
5152.220 |
7067.000 |
|
(c) Other
current liabilities |
14682.490 |
4652.980 |
1867.860 |
|
(d) Short-term
provisions |
2146.010 |
810.820 |
883.540 |
|
Total Current Liabilities
(4) |
33792.210 |
21043.010 |
14665.720 |
|
|
|
|
|
|
TOTAL |
66472.640 |
64916.990 |
61741.800 |
|
|
|
|
|
|
II.
ASSETS |
|
|
|
|
(1)
Non-current assets |
|
|
|
|
(a) Fixed
Assets |
|
|
|
|
(i)
Tangible assets |
20458.890 |
20901.220 |
17663.100 |
|
(ii)
Intangible Assets |
1052.470 |
953.620 |
737.580 |
|
(iii)
Capital work-in-progress |
211.620 |
257.600 |
2520.780 |
|
(iv)
Intangible assets under development |
2207.800 |
1875.970 |
1668.800 |
|
(b) Non-current Investments |
20056.920 |
19785.060 |
19380.240 |
|
(c) Deferred tax assets (net) |
0.000 |
0.000 |
0.000 |
|
(d) Long-term Loan and Advances |
2989.760 |
3438.370 |
4632.240 |
|
(e) Other
Non-current assets |
4.990 |
2.180 |
3.700 |
|
Total Non-Current
Assets |
46982.450 |
47214.020 |
46606.440 |
|
|
|
|
|
|
(2)
Current assets |
|
|
|
|
(a)
Current investments |
3.750 |
0.000 |
217.000 |
|
(b)
Inventories |
7335.180 |
6124.800 |
5933.260 |
|
(c) Trade
receivables |
5295.970 |
3932.510 |
4038.250 |
|
(d) Cash
and cash equivalents |
1787.100 |
2558.850 |
2028.270 |
|
(e)
Short-term loans and advances |
5050.780 |
4493.760 |
2749.490 |
|
(f) Other
current assets |
17.410 |
593.050 |
169.090 |
|
Total
Current Assets |
19490.190 |
17702.970 |
15135.360 |
|
|
|
|
|
|
TOTAL |
66472.640 |
64916.990 |
61741.800 |
PROFIT & LOSS
ACCOUNT
|
|
PARTICULARS |
31.03.2014 |
31.03.2013 |
31.03.2012 |
|
|
|
SALES |
|
|
|
|
|
|
|
Income |
36727.920 |
31463.030 |
26410.670 |
|
|
|
Other Income |
298.810 |
349.720 |
89.410 |
|
|
|
TOTAL |
37026.730 |
31812.750 |
26500.080 |
|
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
|
|
Cost of Materials Consumed |
18330.490 |
15285.770 |
12399.520 |
|
|
|
Purchases of Stock-in-Trade |
1740.180 |
1917.380 |
2436.790 |
|
|
|
Changes in inventories of
finished goods, work-in-progress and Stock-in-Trade |
(212.070) |
(241.750) |
(932.610) |
|
|
|
Employees benefits expense |
2982.320 |
2490.170 |
2072.320 |
|
|
|
Other expenses |
8069.980 |
7170.490 |
1700.320 |
|
|
|
Exceptional Items |
2268.630 |
1237.320 |
1800.840 |
|
|
|
Other manufacturing expenses |
0.000 |
0.000 |
4618.120 |
|
|
|
TOTAL (B) |
33179.530 |
27859.380 |
24095.300 |
|
|
|
|
|
|
|
|
Less |
PROFIT
BEFORE INTEREST, TAX, DEPRECIATION AND AMORTISATION |
3847.200 |
3953.370 |
2404.780 |
|
|
|
|
|
|
|
|
|
Less |
FINANCIAL
EXPENSES |
2775.910 |
2559.790 |
1544.210 |
|
|
|
|
|
|
|
|
|
|
PROFIT
BEFORE TAX, DEPRECIATION AND AMORTISATION |
1071.290 |
1393.580 |
860.570 |
|
|
|
|
|
|
|
|
|
Less/ Add |
DEPRECIATION/
AMORTISATION |
1752.870 |
1521.670 |
1320.030 |
|
|
|
|
|
|
|
|
|
|
PROFIT BEFORE
TAX |
(681.580) |
(128.090) |
(459.460) |
|
|
|
|
|
|
|
|
|
Less |
TAX |
(689.820) |
305.140 |
349.690 |
|
|
|
|
|
|
|
|
|
|
PROFIT AFTER TAX
|
8.240 |
(433.230) |
(809.150) |
|
|
|
|
|
|
|
|
|
Add |
PREVIOUS
YEARS’ BALANCE BROUGHT FORWARD |
6870.620 |
7862.850 |
9227.000 |
|
|
|
|
|
|
|
|
|
Less |
APPROPRIATIONS |
|
|
|
|
|
|
|
Dividend |
463.000 |
478.000 |
478.000 |
|
|
|
Tax on Dividend |
81.000 |
81.000 |
77.000 |
|
|
|
Total |
544.000 |
559.000 |
555.000 |
|
|
|
|
|
|
|
|
|
BALANCE CARRIED
TO THE B/S |
6334.860 |
6870.620 |
7862.850 |
|
|
|
|
|
|
|
|
|
|
EARNINGS IN
FOREIGN CURRENCY |
|
|
|
|
|
|
|
Export Earnings |
21932.650 |
18008.460 |
13576.020 |
|
|
|
Other Operating Income |
71.410 |
77.600 |
103.130 |
|
|
|
Interest Income |
0.040 |
0.030 |
0.210 |
|
|
|
TOTAL
EARNINGS |
22004.100 |
18086.090 |
13679.360 |
|
|
|
|
|
|
|
|
|
IMPORTS |
|
|
|
|
|
|
|
Raw Materials |
8980.930 |
5649.480 |
4761.490 |
|
|
|
Stores, spares, chemicals and
packing material |
541.470 |
335.100 |
1729.340 |
|
|
|
Capital Goods |
44.580 |
99.610 |
227.100 |
|
|
|
TOTAL
IMPORTS |
9566.980 |
6084.190 |
6717.930 |
|
|
|
|
|
|
|
|
|
Earnings Per
Share (Rs.) |
0.05 |
(2.72) |
(5.08) |
|
KEY RATIOS
|
PARTICULARS |
|
31.03.2014 |
31.03.2013 |
31.03.2012 |
|
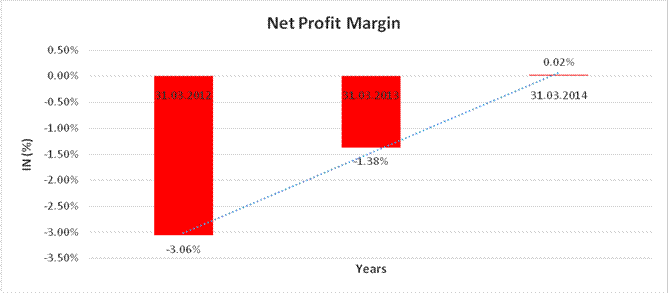
Net Profit Margin (PAT/Sales) |
(%) |
0.02 |
(1.38) |
(3.06) |
|
|
|
|
|
|
|
Operating Profit Margin (PBDIT / Sales) |
(%) |
10.47 |
12.57 |
9.11 |
|
|
|
|
|
|
|
Return on Total Assets (PBT/Total Assets} |
(%) |
(1.55) |
(0.30) |
(1.20) |
|
|
|
|
|
|
|
Return on Investment (ROI) (PBT/Networth) |
|
(0.04) |
(0.01) |
(0.02) |
|
|
|
|
|
|
|
Debt Equity Ratio (Total Debt /Networth) |
|
1.29 |
1.68 |
1.52 |
|
|
|
|
|
|
|
Current Ratio (Current Asset/Current Liability) |
|
0.58 |
0.84 |
1.03 |
FINANCIAL ANALYSIS
[all figures are
in Rupees Millions]
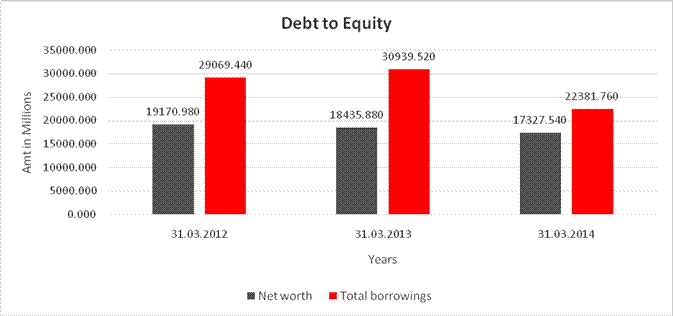
DEBT EQUITY RATIO
|
Particular |
31.03.2012 |
31.03.2013 |
31.03.2014 |
|
|
(Rs.
In Millions) |
(Rs.
In Millions) |
(Rs.
In Millions) |
|
Share Capital |
159.300 |
159.300 |
154.460 |
|
Reserves & Surplus |
19011.680 |
18276.580 |
17173.080 |
|
Net
worth |
19170.980 |
18435.880 |
17327.540 |
|
|
|
|
|
|
long-term borrowings |
24222.120 |
20512.530 |
11410.500 |
|
Short term borrowings |
4847.320 |
10426.990 |
10971.260 |
|
Total
borrowings |
29069.440 |
30939.520 |
22381.760 |
|
Debt/Equity
ratio |
1.516 |
1.678 |
1.292 |
YEAR-ON-YEAR GROWTH
|
Year
on Year Growth |
31.03.2012 |
31.03.2013 |
31.03.2014 |
|
|
(Rs.
In Millions) |
(Rs.
In Millions) |
(Rs.
In Millions) |
|
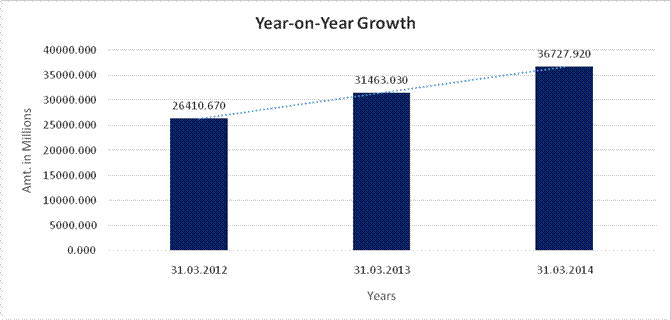
Sales |
26410.670 |
31463.030 |
36727.920 |
|
|
|
19.130 |
16.734 |
NET PROFIT MARGIN
|
Net
Profit Margin |
31.03.2012 |
31.03.2013 |
31.03.2014 |
|
|
(Rs.
In Millions) |
(Rs.
In Millions) |
(Rs.
In Millions) |
|
Sales |
26410.670 |
31463.030 |
36727.920 |
|
Profit |
(809.150) |
(433.230) |
8.240 |
|
|
(3.06)% |
(1.38)% |
0.02% |
LOCAL AGENCY FURTHER INFORMATION
|
Sr. No. |
Check List by Info
Agents |
Available in Report (Yes / No) |
|
1] |
Year of Establishment |
Yes |
|
2] |
Locality of the firm |
Yes |
|
3] |
Constitutions of the firm |
Yes |
|
4] |
Premises details |
No |
|
5] |
Type of Business |
Yes |
|
6] |
Line of Business |
Yes |
|
7] |
Promoter's background |
No |
|
8] |
No. of employees |
Yes |
|
9] |
Name of person contacted |
Yes |
|
10] |
Designation of contact person |
Yes |
|
11] |
Turnover of firm for last three years |
Yes |
|
12] |
Profitability for last three years |
Yes |
|
13] |
Reasons for variation <> 20% |
---------------------- |
|
14] |
Estimation for coming financial year |
No |
|
15] |
Capital in the business |
Yes |
|
16] |
Details of sister concerns |
Yes |
|
17] |
Major suppliers |
No |
|
18] |
Major customers |
No |
|
19] |
Payments terms |
No |
|
20] |
Export / Import details (if applicable) |
No |
|
21] |
Market information |
---------------------- |
|
22] |
Litigations that the firm / promoter involved in |
---------------------- |
|
23] |
Banking Details |
Yes |
|
24] |
Banking facility details |
Yes |
|
25] |
Conduct of the banking account |
---------------------- |
|
26] |
Buyer visit details |
---------------------- |
|
27] |
Financials, if provided |
Yes |
|
28] |
Incorporation details, if applicable |
Yes |
|
29] |
Last accounts filed at ROC |
Yes |
|
30] |
Major Shareholders, if available |
Yes |
|
31] |
Date of Birth of Proprietor/Partner/Director, if available |
No |
|
32] |
PAN of Proprietor/Partner/Director, if available |
No |
|
33] |
Voter ID No of Proprietor/Partner/Director, if available |
No |
|
34] |
External Agency Rating, if available |
Yes |
UNSECURED LOAN
(Rs.
In Million)
|
Particulars |
As
on 31.03.2014 |
As
on 31.03.2013 |
|
Long Term
Borrowing |
|
|
|
India Rupee Loans from Subsidiaries |
1015.900 |
428.700 |
|
|
|
|
|
Short Term
Borrowing |
|
|
|
Loan Repayable on Demand From Banks |
4930.850 |
3784.270 |
|
Loans from related parties |
70.000 |
93.500 |
|
Total |
6016.750 |
4306.470 |
INDEX OF CHARGES
|
S.NO. |
CHARGE ID |
DATE OF CHARGE CREATION/MODIFICATION |
CHARGE AMOUNT SECURED |
CHARGE HOLDER |
ADDRESS |
SERVICE REQUEST NUMBER (SRN) |
|
1 |
10424562 |
25/04/2013 * |
1,600,000,000.00 |
INDIAN BANK |
47-48 PRAGATI
HOUSE, NEHRU PLACE, NEW DELHI - 110019, INDIA |
B74962739 |
|
2 |
10343200 |
25/04/2013 * |
2,504,900,000.00 |
DBS BANK LIMITED
|
ACTING ON BEHALF
OF DBS BANK LIMITED, SINGAPORE, CAPITOL POINT, BABA KHARAK SINGH MARG,
NEW DELHI - 110001, INDIA |
B74167784 |
|
3 |
10303869 |
20/03/2012 * |
2,400,000,000.00 |
CENTRAL BANK OF
INDIA |
PRESS AREA BRANCH,
LINK HOUSE, 3 BAHADUR SHAH MARG, NEW DELHI - 110002, INDIA |
B35922640 |
|
4 |
10274305 |
20/03/2012 * |
3,000,000,000.00 |
AXIS BANK
LIMITED |
B-2 & B-3,
SECTOR-16, NOIDA, NOIDA, UTTAR PRADESH - 201301, INDIA |
B35468982 |
|
5 |
10271462 |
20/03/2012 * |
1,000,000,000.00 |
CORPORATION BANK
INDUSTRIAL FINANCE BRANCH |
H.T. HOUSE, 10TH
FLOOR, K.G.MARG, CONNAUGHT PLACE, NEW DELHI - 110001, INDIA |
B35922079 |
|
6 |
10271469 |
20/03/2012 * |
2,700,000,000.00 |
CORPORATION BANK
INDUSTRIAL FINANCE BRANCH |
H.T. HOUSE, 10TH
FLOOR, K.G.MARG, CONNAUGHT PLACE, NEW DELHI - 110001, INDIA |
B35921576 |
|
7 |
10229540 |
12/07/2010 |
4,884,000,000.00 |
HOUSING
DEVELOPMENT FINANCE CORPORATION LIMITED |
RAMON HOUSE 169BACKBAY
RECLAMATION, H T PAREKH MARG, MUMBAI - 400020, MAHARASHTRA, INDIA |
A90177528 |
|
8 |
10217801 |
20/03/2012 * |
2,300,000,000.00 |
EXPORT-IMPORT
BANK OF INDIA |
CENTRE ONE
BUILDING, FLOOR 21, WORD TRADE CENTRE COMPLEX, CUFFE PARADE, MUMBAI - 400005,
MAHARASHTRA, INDIA |
B35908292 |
|
9 |
10173318 |
12/07/2010 * |
4,884,000,000.00 |
HOUSING
DEVELOPMENT FINANCE CORPORATION LIMITED |
RAMON HOUSE
169BACKBAY RECLAMATION, H T PAREKH MARG, MUMBAI - 400020, MAHARASHTRA, INDIA |
A89713697 |
|
10 |
80046092 |
20/09/2013 * |
15,000,000,000.00 |
ICICI BANK
LIMITED |
LANDMARK RACE
COURCE CIRCLE, ALKAPURI, VADODARA - 390007, GUJARAT, INDIA |
B87010690 |
* Date of charge modification
OVERVIEW
Jubilant
Life Sciences Limited (‘the Company’ or ‘Jubliant’) is a global Pharmaceutical
and Life Sciences Company engaged in manufacture and supply of Active
Pharmaceuticals Ingredients, Solid Dosage Formulations, Radiopharmaceuticals,
Allergy Therapy Products and Life Sciences Ingredients. It also provides
services in Contract Manufacturing of Sterile Injectables and Drug Discovery
and Development. The Company’s strength lies in its unique offerings of
Pharmaceutical and Life Sciences products and services across the value chain.
With 10 world-class manufacturing facilities in India, US and Canada and a
teamof about 6,200 multicultural people across the globe, the Company is
committed to deliver value to its customers spread across over 100 countries.
The Company is well recognized as a “Partner of Choice” by leading
pharmaceuticals and life sciences companies globally.
PERFORMANCE
REVIEW
The
year FY 2014 saw Income from Operations of Rs. 58,034.000 million, a growth of
12% year on year (YoY). Earnings before Interest, Taxes and Depreciation and
Amortization (EBITDA) stood at Rs. 10,267.000 million with EBITDA margins of
17.7%. Profits before Exceptional Items, Tax and Minority Interest were at Rs.
4,218 million. The Profit after Tax (PAT) was reported at Rs. 1,090 million
whereas the Normalized PAT after adjusting for exceptional items was at Rs.
3,235 million.
they
continue to focus on expanding their business globally. Their products and
services now reach outto customers in over 100 countries of the world. International
revenues account for 75% of the revenue mix at Rs. 43,261 million with revenues
primarily from North America, Europe and Japan.
In FY
2014, the Pharmaceuticals segment witnessed a revenue increase of 2% at Rs.
27,277.000 million, contributing 47% to the overall Income from Operations.
This progression is driven by new product introduction and price improvements
in Radiopharmaceuticals. In Solid Dosage Formulations business, they have 46
commercial products and a pipeline of 455 filings pending approval including 37
ANDAs pending in the US, 7 in Canada and 3 in Europe. During the year, they
made 121 filings including 7 in the US, 1 in Canada and 2 in Europe. In Active
Pharmaceutical Ingredients business, they have 30 commercial APIs and a pipeline
of 522 filings pending approval including 46 in the US, 16 in Canada and 9
CEPs. During the year, they made 33 filings including 9 in the US, 2 in Canada
and 9 CEPs.
Regarding
the warning letters at their CMO operations - the issue in Montreal has been
resolved and in Spokane, they have responded to all of the FDA observations.
They are confident of bringing back growth in CMO of Sterile Injectables
business on the back of strong order book and better compliance.
During
the year, they sold their Hospital business to Narayana Health. The business
has been transferred on a going concern basis as a slump sale, which will
enable us to focus on their core businesses in Pharmaceuticals and Life
Sciences.
In FY
2014, Life Science Ingredients segment delivered revenue growth of 23% to Rs.
30,757 million thus contributing to 53% to the overall Income from Operations.
Volume growth along with the price hikes that took in some of their key
products aided their performance in this segment.
As of
March 31, 2014, Net Debt stood at Rs. 39,130.000 million. It comprises of Rs.
18,324.000 million in Net Long-Term Debt and Rs. 20,806 million in Working
Capital Debt. Post adjustment for exchange rates, when compared with March 31,
2013, Net Debt was down by Rs. 2,604.000 million at Rs. 36,526 million.
AWARDS AND ACCOLADES
During
the year, various awards and accolades were received by the Company/its
management. These are:
•
‘14th Annual Greentech Environment Award 2013’ won byGajraula plant, India;
• ‘Certificate
from CocaCola for compliance to Global Food Safety Initiative as per TCCC norms
for the year 2013’ presented to Gajraula plant, India;
•
‘Amity Global Business School - CSR Award 2013’ won by the Company in
appreciation of outstanding work in the CSR domain;
•
‘Shri Janeshwar Mishra Export Award’ - Excellence Award for the Best
Performance in the field of Export and First Award for performance in the field
of Export under the category of Chemicals, Pharmaceuticals & Cosmetic
Products;
• ‘Annual
Greentech Safety Award 2013’ - Silver Award under chemical sector won by
Gajraula plant, India;
•
‘Express, Logistics & Supply Chain Leadership Award’ under the category
Excellence in Manufacturing Supply Chain in Chemical;
• ‘CII
– National Award for Excellence in Energy Management 2013’ won by Gajraula
plant as Energy Efficient Unit; and
•
‘Lifetime Achievement Award 2010-11’ won by Mr. Shyam S. Bhartia, presented by
Mr. Anand Sharma, Minister of Commerce and Industry, Govt. of India at
CHEMEXCIL’s Export Award
BUSINESS OBJECTIVES
Jubilant
Life Sciences today is a global integrated pharmaceutical and life sciences
player with over 35 years of proven track record. They have a well-diversified
portfolio across segments of Pharmaceuticals and Life Science Ingredients.
Strategically, they have pursued a cohesive business model across the entire
Pharmaceutical ecosystem and have positioned ourselves as a one-stop-shop for
the global Pharmaceutical and Life Sciences industry.
They
continue to deliver meaningful innovations in health care and are also building
a more agile company to seize evolving market opportunities. their competitive
strength lies in their integrated low cost production and their innovative
product portfolio backed by efficient R&D capabilities and manufacturing
capabilities.
OUTLOOK
They
expect strong growth momentum in both the segments of their businesses. The
Pharmaceuticals business is expected to deliver on account of resolution of Warning
Letter in Montreal, focused attention to resolve USFDA issues in Spokane,
consolidation of global quality system for compliance, new product launches in
Generics business and better price realisations and expected launch of
Ruby-fill in Radiopharmaceuticals
Growth
in Life Science Ingredients business is to be led by higher capacity
utilisation, better pricing and entry into new geographies. They will continue
to be prudent in any incremental capital expenditure spending and look forward
to improving their cash balances and reducing debt levels.
They
wish to convey their earnest gratitude to all their esteemed stakeholders
including customers, suppliers, vendors, bankers and shareholders for reposing
faith in us and being supportive through all their endeavours. They would like
to welcome Mrs. Sudha Pillai, who joined us as an Independent Director on their
Board. they would also like to express a note of thanks to their employees,
globally, for assisting us in the delivery of their broader agenda of profitable
growth.
MANAGEMENT DISCUSSION AND ANALYSIS
BUSINESS
EXCELLENCE
Business
Excellence function focuses on new improvement strategies to enhance the
competitive advantage of the Company. These strategies focus on all key aspect
of the Company viz Customer, Process and People and encompasses all facets of
the business including Manufacturing, Sales and Marketing, R&D, Supply
Chain,
Corporate
HR and Projects etc.
Business
Excellence has adopted various improvement methodologies in-line with organisation
priorities like Six Sigma, Lean, and Design for Six Sigma (DFSS), World-Class
Manufacturing (WCM), Total Productive Maintenance (TPM), Supply Chain
Re-engineering (SCOR), Theory of Constraints (TOC), Project Management (EPM),
Operation Research (OR), Business Intelligence (BI), Maynard Operation
Sequencing Technique (MOST) etc.
Business
Excellence helped in aiding in Supply Chain re-engineering solutions through
planning and sourcing and also providing business intelligence solutions in the
contract manufacturing businesses aiding critical cost and quality improvement.
Capacity debottlenecking projects through application of Lean Six Sigma and
process simulation in APIs, Solid Dosage Formulations and Fine Ingredients
business were also undertaken. More than 175 green belts are undergoing and 55
green belts have completed their requirements for certification by undergoing
the Lean Six Sigma training and completing at least one Improvement project
sponsored by their functional head.
Along
with improvement in existing processes, a need was felt for a systematic
mechanism for new product development and capital projects execution. In this
framework, the Stage Gate approach was adopted for initialisation, execution
& governance throughout the life cycle of the project. Quality by Design
(QBD) has also been very well-integrated with new product development.
The
Company won award and recognition from Confederation of Indian Industry (CII),
Kaizen Institute and Frost and Sullivan for various accomplishments in
manufacturing excellence. With respect to bringing about improvement in the
Company, knowledge based newsletters were shared across all businesses, yellow
and green belt trainings for corporate functions were undertaken covering many
employees and a Kaizen scheme was launched to generate number of ideas on cost
reduction, capacity enhancement and quality improvement.
FIXED ASSETS
Tangible assets:
· Freehold Land
· Leasehold Land
· Buildings
· Plant and equipment
· Furniture and fixtures
· Vehicles
· Vehicles-leased
· Office equipments
·
Railway sidings
Intangible assets:
· Internally generated
· Product registration/market authorization
· Rights
·
Software
STATEMENT OF
UNAUDITED STANDALONE RESULTS FOR THE QUARTER AND HALF YEAR ENDED 30 SEPTEMBER
2014
(Rs.
In Millions)
|
Particulars |
|
||
|
Quarter Ended |
Half Year Ended |
||
|
30.09.2014 |
30.06.2014 |
30.09.2014 |
|
|
(Unaudited) |
(Unaudited) |
(Unaudited) |
|
|
PART
I |
|
|
|
|
Income
from operations |
|
|
|
|
(a) Net sales/Income from operations (Net
of excise duty) |
6959.500 |
10260.100 |
17219.600 |
|
(b) Other Operating Income |
50.300 |
102.300 |
152.600 |
|
Total
income from operations (net) |
7009.800 |
10362.400 |
17372.200 |
|
Expenses |
|
|
|
|
a)
Cost of materials consumed |
3985.800 |
5303.400 |
9289.200 |
|
b)
Purchase of traded goods |
340.500 |
563.300 |
903.800 |
|
c)
Change in inventories of finished goods, work-in progress and traded
goods |
62.700 |
103.200 |
165.900 |
|
d)
Power & fuel |
793.000 |
925.300 |
1718.300 |
|
e)
Employee benefits expenses |
533.800 |
797.000 |
1330.800 |
|
f)
Depreciation and amortization expenses |
207.100 |
412.800 |
619.900 |
|
g)
Other expenses |
840.100 |
1324.400 |
2164.500 |
|
Total
Expenses |
6763.000 |
9429.400 |
16192.400 |
|
Profit/(Loss) from operations before other
income, finance cost and exceptional items |
246.800 |
933.000 |
1179.800 |
|
Other Income |
520.500 |
76.700 |
597.200 |
|
Profit/(Loss) from ordinary activities
before finance costs & exceptional items |
767.300 |
1009.700 |
1777.000 |
|
Finance costs |
612.000 |
631.400 |
1243.400 |
|
Profit/(Loss) from ordinary activities
after finance costs but before exceptional items |
155.300 |
378.300 |
533.600 |
|
Exceptional items |
(2711.900) |
(1529.400) |
(4241.300) |
|
Profit/(Loss) from ordinary activities
before tax |
2867.200 |
1907.700 |
4774.900 |
|
Tax expense (Net) |
699.600 |
(129.500) |
570.100 |
|
Net
Profit/(Loss) from ordinary activities after tax |
2167.600 |
2037.200 |
4204.800 |
|
Extraordinary items (net of tax expenses) |
0.000 |
0.000 |
0.000 |
|
Net
Profit/(Loss) for the period |
2167.600 |
2037.200 |
420.800 |
|
Paid-up equity share capital (Face value per
share Re.1) |
159.300 |
159.300 |
159.300 |
|
Reserves (excluding revaluation reserve) |
|
|
|
|
Earnings per share before and after
extraordinary items (Not annualized) |
|
|
|
|
Basic
(Rs.) |
13.61 |
12.79 |
26.40 |
|
Diluted
(Rs.) |
13.61 |
12.79 |
26.40 |
|
PART
II |
|
|
|
|
PARTICULARS
OF SHAREHOLDING |
|
|
|
|
Public
shareholding |
|
|
|
|
-
Number of shares (Re. 1 each) |
73230083 |
73230083 |
73230083 |
|
-
Percentage of shareholding |
45.98 |
45.98 |
45.98 |
|
Promoters
and promoter group shareholding |
|
|
|
|
a) Pledged/Encumbered |
|
|
|
|
-
Number of shares |
10715000 |
9700000 |
10715000 |
|
-
Percentage of shares (as a % of the total shareholding of promoter and
promoter group) |
12.45 |
11.27 |
12.45 |
|
- Percentage
of shares (as a % of the total share capital of the company) |
6.72 |
6.09 |
6.72 |
|
b) Non-Encumbered |
|
|
|
|
-
Number of shares |
75336056 |
76351056 |
75336056 |
|
- Percentage
of shares (as a % of the total shareholding of promoter and promoter group) |
87.55 |
88.73 |
87.55 |
|
-
Percentage of shares (as a % of the total share capital of the
company) |
47.30 |
47.93 |
47.30 |
|
Particulars
|
31.03.2014 |
|
Investors
Complaints (Nos.) |
|
|
Pending at the beginning of the quarter |
Nil |
|
Received during the quarter |
5 |
|
Disposed of during the quarter |
5 |
|
Remaining unresolved at the end of the quarter |
Nil |
NOTE: 1 UNAUDITED STANDALONE SEGMENT WISE REVENUE, RESULTS AND CAPITAL
EMPLOYED FOR THE QUARTER AND HALF YEAR ENDED 30 SEPTEMBER 2014
|
Particulars |
|
||
|
Quarter Ended |
Half Year Ended |
||
|
30.09.2014 |
30.06.2014 |
30.09.2014 |
|
|
(Unaudited) |
(Unaudited) |
(Unaudited) |
|
|
|
|
|
|
a. Pharmaceuticals |
21.900 |
2020.000 |
2041.900 |
|
b. Life Sciences Ingredients |
6987.900 |
8351.600 |
15339.500 |
|
Total |
7009.800 |
10371.600 |
17381.400 |
|
Less: Inter segment revenue |
0.000 |
9.200 |
9.200 |
|
Net
Sales/Income from operations |
7009.800 |
10342.400 |
17372.200 |
|
a. Pharmaceuticals |
21.900 |
2020.000 |
2041.900 |
|
b. Life Sciences Ingredients |
6987.900 |
5342.400 |
15330.300 |
|
Total |
7009.800 |
10362.400 |
17372.200 |
|
|
|
|
|
a. Pharmaceuticals |
(78.900) |
157.500 |
1014.600 |
|
b.
Life Sciences Ingredients |
456.300 |
1026.000 |
1585.200 |
|
Total |
377.400 |
1183.500 |
2599.800 |
|
Less: i. Interest (Finance costs) |
612.000 |
631.400 |
1401.700 |
|
ii. Other un-allocable expenditure
(including exceptional items) |
157.000 |
371.900 |
3174.000 |
|
iii. Un-allocable Income (including
exceptional items) |
(3258.800) |
(1727.500) |
(150.700) |
|
Total
Profit/(Loss) before tax |
2867.200 |
1907.700 |
(1825.200) |
|
|
|
|
|
a. Pharmaceuticals |
(15.300) |
10294.700 |
11117.300 |
|
b. Life Sciences Ingredients |
18786.600 |
18235.400 |
20180.300 |
|
Total
capital employed in segments |
18771.300 |
28530.100 |
31297.600 |
|
Add: Un-allocable corporate assets less
liabilities |
27313.600 |
22459.500 |
21608.100 |
|
Total
capital employed in the Company |
46084.900 |
50989.600 |
52905.700 |
NOTE: 2 STATEMENT OF STANDALONE ASSETS AND
LIABILITIES
(Rs. In Millions)
|
SOURCES OF FUNDS |
30.09.2014 (Unaudited) |
|
I.
EQUITY
AND LIABILITIES |
|
|
(1)Shareholders' Funds |
|
|
(a) Share Capital |
154.500 |
|
(b) Reserves & Surplus |
21528.700 |
|
Total
Shareholders’ Funds |
21683.200 |
|
|
|
|
(2)
Non-Current Liabilities |
|
|
(a) long-term borrowings |
12005.800 |
|
(b) Deferred tax liabilities (Net) |
1548.600 |
|
(c) Other long term
liabilities |
0.000 |
|
(d) long-term
provisions |
1890.900 |
|
Total Non-current
Liabilities (3) |
15445.300 |
|
|
|
|
(3)
Current Liabilities |
|
|
(a) Short term
borrowings |
6556.800 |
|
(b) Trade
payables |
4291.800 |
|
(c) Other
current liabilities |
6379.700 |
|
(d) Short-term
provisions |
2532.900 |
|
Total Current
Liabilities (4) |
19761.200 |
|
|
|
|
TOTAL |
56889.700 |
|
|
|
|
II.
ASSETS |
|
|
(1)
Non-current assets |
|
|
(a) Fixed
Assets |
15286.700 |
|
(b) Non-current Investments |
17556.400 |
|
(c) Deferred tax assets (net) |
0.000 |
|
(d) Long-term Loan and Advances |
3010.900 |
|
(e) Other
Non-current assets |
4.500 |
|
Total Non-Current
Assets |
35858.500 |
|
|
|
|
(2)
Current assets |
|
|
(a)
Current investments |
100.000 |
|
(b)
Inventories |
6152.400 |
|
(c) Trade
receivables |
3769.000 |
|
(d) Cash
and cash equivalents |
324.500 |
|
(e)
Short-term loans and advances |
10675.400 |
|
(f) Other
current assets |
9.900 |
|
Total
Current Assets |
21031.200 |
|
|
|
|
TOTAL |
56889.700 |
3.In order to
complete the Pharma consolidation as approved by the Board, under its wholly owned
subsidiary Jubilant Pharma Limited, Singapore (JPL), the Company has
transferred, with effect from 1 July
2014, its Active Pharmaceutical Ingredients and Dosage Forms business to
Jubilant Generics limited (JGL), a wholly owned Subsidiary of JPL, by way of a
slump sale on going concern basis for a lump sum consideration of Rs. 9293.000
Millions (net of debts of Rs. 3923.000 millions} and the profit on sale of such
businesses amounting to Rs. 2754.300 millions has been classified under
exceptional items. As reported earlier the Company had already transferred
shares held by it in Jubilant Pharma Holdings Inc, USA and Jubilant Pharma NV,
Belgium to JGL, and the profit on the sale of such share amounting to Rs.
1650.800 millions was also classified as exceptional item in previous quarter.
The Active
Pharmaceutical Ingredients and Dosage Forms business of the Company has been
treated as discontinuing operations till the period up to 30 June 2014 in the
financial results. The required relevant information for these discontinued
operations for all periods presented is as below:
(Rs. In Million)
|
Particulars |
|
||
|
Quarter Ended |
Half Year Ended |
||
|
30.09.2014 |
30.06.2014 |
30.09.2014 |
|
|
(Unaudited) |
(Unaudited) |
(Unaudited) |
|
|
|
|
|
|
|
Total revenue |
-- |
2007.100 |
2007.100 |
|
Total expenditure |
-- |
1913.400 |
1913.400 |
|
Profit before tax |
-- |
93.700 |
93.700 |
|
Tax expense |
-- |
22.900 |
22.900 |
|
Profit after tax |
-- |
70.800 |
70.800 |
4. Pursuant to the
Companies Act, 2013 ('the Act'} being effective from 1 April 2014, the Company
has revised depreciation rates on fixed assets as per the useful life specified
in Part 'C' of Schedule II of the Act.
As a result of this change, the depreciation charge for the quarters ended 30
September 2014, 30 June 2014 and half year ended 30 September 2014 is lower by
Rs. 82.500 Millions, Rs. 366 Millions, and Rs. 1191.100 Million respectively.
Further, based on the transitional provision provided in Note 7(b) of the Schedule
II an amount of Rs.101.500 Million (after adjustment for related tax impact)
has been debited to opening balance of
retained earnings in respect of the fixed assets where life has expired as per
the said Schedule as on 31 March 2014.
5.Finance costs
includes exchange difference arising from foreign currency short-term
borrowings regarded as an adjustment to interest costs as per AS
16-"Borrowing Costs ", and is gross of credit on the swap contracts
as under.
|
Particulars |
|
||
|
Quarter Ended |
Half Year Ended |
||
|
30.09.2014 |
30.06.2014 |
30.09.2014 |
|
|
(Unaudited) |
(Unaudited) |
(Unaudited) |
|
|
- Finance Cost net of credit on swap contracts |
395.100 |
486.600 |
881.700 |
|
Add : Foreign Exchange differences and credit on swap contracts |
216.900 |
144.800 |
361.700 |
|
- Gross Finance Cost |
612.00 |
631.400 |
1243.400 |
6. Exceptional
items for each period presented includes:
i) Amortization of debit balance in Foreign Currency
Monetary Items Translation Difference Account (FCMITDA) of Rs. 77.800 millions,
Rs. 243.500 millions, Rs. 387.400 million; Rs. 321.300 million, Rs. 6425 lacs
and Rs.1000.200 millions for the quarters ended 30 September 2014, 30 June
2014, 30 September 2013; half year ended 30 September 2014, 30 September 2013
and year ended 31 March 2014; respectively, representing exchange difference on long-term foreign currency
monetary liabilities which has been used for the purpose other than acquiring
fixed assets.
ii) The remaining amount of exceptional items,
other than profit on sale of business/ investments as mentioned in note 3, for
all periods presented primarily represents foreign exchange difference for
the period (excluding portion included
in finance cost} and mark to market gain/ loss (net of related contractual
recoveries) in respect of forward contracts, currency and interest rate swap
contracts.
7. Previous period figures have been
reclassified to conform to the current period's classification. Further~ the
figures for the current period are not comparable to the previous periods due
to transfer of certain businesses as explained in Note 3.
8. The above unaudited results were, subjected
to limited review by the Statutory Auditors of the Company, reviewed by the
Audit Committee and approved by the Board of Directors at its meeting held on
28 October 2014. The review report of the Statutory Auditors is being filed
with the Bombay Stock Exchange and National Stock Exchange and is also available
on the Company's website at www.jubl.com.
PRESS RELASES
Ind-Ra
Assigns Jubilant Life Sciences’ Proposed NCDs ‘IND A+(exp)’; Affirms other
Ratings
Ind-Ra-New
Delhi-22 January 2015: India Ratings & Research (Ind-Ra) has assigned
Jubilant Life Sciences Limited’s (JLL) proposed INR4,000m non-convertible
debenture (NCD) an ‘IND A+(exp)’ rating. The agency has also affirmed JLL’s
Long-Term Issuer Rating at ‘IND A+’. The Outlook is Stable. A full list of
rating actions is at the end of this commentary.
The proceeds from the proposed NCD are to be used by JLL for general corporate
purposes including repayments of existing debt. The assignment of the final
rating is contingent upon the receipt of final documents conforming to the
information already received.
KEY RATING DRIVERS
Presence Across Pharmaceutical Value Chain: JLL has established operations across
the entire pharmaceutical value chain - right from the inception of a drug
concept (through drug discovery and development solutions business), walking past
the active pharmaceutical ingredients (API) business and culminating into the
final dosage or injectable products. Moreover, the company also provides
contract manufacturing at various levels which helps it derive business from
major pharmaceutical players globally.
Vertically Integrated Operations: JLL
has a high degree of vertical integration with the solid dosage formulation
business leveraging upon the in-house API unit. The company is also the largest
supplier for pyridine and is exploring stabilisation of commercial volumes for
Symtet, another pyridine integrated product thus moving further in the pyridine
value chain. This level of vertical integration helps the company achieve cost
competitiveness and also helps in stabilisation of operation by having a
consistent raw material supply.
Well-diversified Market: JLL
supplies to a global market. The company derives 25% of its revenue from
domestic sales while a substantial 58% is earned by supplying to the regulated
markets of US and EU/Japan. Another 9% is derived from sales in the Chinese
market while the rest 8% is from supplying to the rest of the world. The
company has been growing its presence in Russia, Brazil and South Africa by
both licensing out and developing own distribution networks.
Adequate Liquidity; High Financial Leverage: An aggressive inorganic growth
strategy coupled with volatile profitability resulted in high financial
leverage in the past. For FY14, JLL had interest coverage (operating
EBITDA/gross interest expense) of 3.10x (FY13: 4.30x) and financial leverage
(net debt/operating EBITDA) of 3.91x (FY13: 3.80x). The company however has
comfortable liquidity with cash balance of INR4,603m at FYE14. Also, its
average fund-based working capital use is around 84%.
Revenue Growth, Volatile Margins: JLL’s
consolidated revenue grew 12.4% yoy in FY14, driven by 23% yoy growth in the
life sciences ingredients business. There was muted growth in the
pharmaceutical segment due to delays in approvals and warning letters at the
Montreal and Spokane CMO facilities. The company remains exposed to raw
material commodity cycles and finished product price fluctuations leading to
volatile EBITDA margin.
Lower-than-expected Half-yearly Overall Performance due to CMO Business: For 1HFY15, JLL reported revenue of
INR28.44bn (1.7% yoy). The EBIDTA margin remained low at 9% primarily due to
the impact of warning letter on the contract manufacturing (CMO) business at
Spokane. The issues mentioned in the warning letter to the Montreal facility
have already been resolved and lifted. The company took a voluntary shutdown at
the Spokane facility in 1QFY15 for update and to implement corrective measures,
which has also impacted the EBIDTA. However, Ind-Ra believes resumption of
operations at the facility and no loss of business contracts shall have a
stabilising impact on the revenue and EBIDTA margins in 2HFY15.
Business Reorganisation: The
company has undergone a business reorganisation whereby the entire
pharmaceutical business has been consolidated under JLL’s 100% subsidiary,
Jubilant Pharma Limited (JPL), Singapore. JPL has tied-up funding of USD200m
from International Finance Corporation for the business reorganisation
transaction. Of this USD147.5m has been disbursed to JPL and transferred to Jubilant
Generics Limited, a 100% subsidiary of JPL for the Indian pharmaceutical
business, by a mix of equity and compulsorily convertible debt. This
integration of higher margin pharmaceutical business provides JLL with the
opportunity to unlock a higher multiple at an opportune moment.
Foreign Exchange, Exceptional Losses: JLL
incurred forex losses in both FY14 and FY13 due to the foreign currency
denominated debt being restated at appreciated US dollar levels on the
consolidated balance sheet. However, Ind-Ra believes that JLL will have a more
stable foreign currency profile with the realignment of foreign currency debt
profiles with the foreign exchange earning entities and stable fundamentals for
Indian economy.
RATING SENSTIVITIES
Negative: A negative
rating action could result from debt-led acquisitions/capex leading to the
financial leverage being sustained above 4.5x.
Positive: A positive
rating action could result from a significant improvement in the consolidated
profitability leading to the financial leverage being sustained below 2.5x.
COMPANY PROFILE
JLL is an integrated pharmaceutical and life sciences company. It is India’s
largest custom research and manufacturing services player and a leading drug
discovery and development solution provider. The company has its presence in
India, North America, Europe and China. In FY14, the company reported
consolidated revenue of INR58.03bn, up 12.4% yoy and an EBITDA of INR10.08bn
resulting in an EBITDA margin of 17.4%.
JLL’s ratings are as follows:
·
Long-Term Issuer Rating: affirmed at ‘IND A+’; Outlook Stable
·
INR4,550m long-term bank loan (reduced from INR10,700m):
affirmed at ‘IND A+’
·
INR7,000m fund-based working capital limits: affirmed at ‘IND
A+’/‘IND A1+’
·
INR10,750m non-fund based working capital limits: affirmed at ‘IND
A1+’
·
INR3,000m short-term debt/commercial paper programme (carved out
of fund-based working capital limits): ‘IND
·
A1+’ rating withdrawn, as the instrument has been paid in full
·
Proposed INR4,000m non-convertible debenture programme: assigned
Long-Term ‘IND A+(exp)’
NEW DELHI:
Jubilant Life Sciences has completed tender offer to acquire all outstanding shares of Cadista HoldingsInc, thereby making the US-based firm as its wholly-owned subsidiary. Jubilant Generics Inc, a subsidiary of the company has successfully completed its previously announced tender offer for all of the outstanding shares of Cadista Holdings Inc, Jubilant Life Sciences said in a statement.
"Accordingly, Cadista Holdings has now become a wholly-owned subsidiary of Jubilant Life Sciences Limited," it added.
As of the expiration of the tender offer, a total of 17,018,378 shares were validly tendered and not withdrawn in the offer, representing around 82 per cent of Cadista Holdings' currently outstanding shares not already owned by Jubilant Generics and its affiliates, it said.
Jubilant Generics has accepted for payment of all validly tendered shares, it added.
"As a result of the merger, all shares of Cadista Holdings held by the remaining minority stockholders of Cadista Holdings have been cancelled and, subject to appraisal rights under Delaware Law, converted into the right to receive a $1.60 per share in cash," it said.
Jubilant Generics Inc had commenced the open offer to acquire all outstanding shares of the US-based Cadista Holdings Inc in November 2014.
Jubilant shares were trading 1.61 per cent up at Rs 126 apiece during afternoon session on the BSE
CMT REPORT (Corruption, Money Laundering & Terrorism]
The Public Notice information has been collected from various sources
including but not limited to: The Courts,
1] INFORMATION ON
DESIGNATED PARTY
No records exist designating subject or any of its beneficial owners, controlling
shareholders or senior officers as terrorist or terrorist organization or whom
notice had been received that all financial transactions involving their assets
have been blocked or convicted, found guilty or against whom a judgement or
order had been entered in a proceedings for violating money-laundering,
anti-corruption or bribery or international economic or anti-terrorism sanction
laws or whose assets were seized, blocked, frozen or ordered forfeited for
violation of money laundering or international anti-terrorism laws.
2] Court Declaration :
No records exist to suggest that subject is
or was the subject of any formal or informal allegations, prosecutions or other
official proceeding for making any prohibited payments or oher improper payments
to government officials for engaging in prohibited transactions or with
designated parties.
3] Asset Declaration :
No records exist to suggest that the property or assets of the subject
are derived from criminal conduct or a prohibited transaction.
4] Record on Financial
Crime :
Charges or conviction
registered against subject: None
5] Records on Violation of
Anti-Corruption Laws :
Charges or
investigation registered against subject: None
6] Records on Int’l
Anti-Money Laundering Laws/Standards :
Charges or
investigation registered against subject: None
7] Criminal Records
No
available information exist that suggest that subject or any of its principals
have been formally charged or convicted by a competent governmental authority for
any financial crime or under any formal investigation by a competent government
authority for any violation of anti-corruption laws or international anti-money
laundering laws or standard.
8] Affiliation with
Government :
No record
exists to suggest that any director or indirect owners, controlling
shareholders, director, officer or employee of the company is a government
official or a family member or close business associate of a Government
official.
9] Compensation Package :
Our market
survey revealed that the amount of compensation sought by the subject is fair
and reasonable and comparable to compensation paid to others for similar
services.
10] Press Report :
No press reports / filings exists on
the subject.
CORPORATE GOVERNANCE
MIRA INFORM as part of its Due Diligence do provide comments on
Corporate Governance to identify management and governance. These factors often
have been predictive and in some cases have created vulnerabilities to credit
deterioration.
Our Governance Assessment focuses principally on the interactions
between a company’s management, its Board of Directors, Shareholders and other
financial stakeholders.
CONTRAVENTION
Subject is not known to have contravened any existing local laws,
regulations or policies that prohibit, restrict or otherwise affect the terms
and conditions that could be included in the agreement with the subject.
FOREIGN EXCHANGE RATES
|
Currency |
Unit
|
Indian Rupees |
|
US Dollar |
1 |
Rs. 61.68 |
|
|
1 |
Rs. 93.53 |
|
Euro |
1 |
Rs. 70.75 |
INFORMATION DETAILS
|
Analysis Done by
: |
SUB |
|
|
|
|
Report Prepared
by : |
JYTK |
SCORE & RATING EXPLANATIONS
|
SCORE FACTORS |
RANGE |
POINTS |
|
HISTORY |
1~10 |
6 |
|
PAID-UP CAPITAL |
1~10 |
6 |
|
OPERATING SCALE |
1~10 |
6 |
|
FINANCIAL
CONDITION |
|
|
|
--BUSINESS SCALE |
1~10 |
6 |
|
--PROFITABILIRY |
1~10 |
6 |
|
--LIQUIDITY |
1~10 |
6 |
|
--LEVERAGE |
1~10 |
6 |
|
--RESERVES |
1~10 |
6 |
|
--CREDIT LINES |
1~10 |
6 |
|
--MARGINS |
-5~5 |
-- |
|
DEMERIT POINTS |
|
|
|
--BANK CHARGES |
YES/NO |
YES |
|
--LITIGATION |
YES/NO |
NO |
|
--OTHER ADVERSE INFORMATION |
YES/NO |
NO |
|
MERIT POINTS |
|
|
|
--SOLE DISTRIBUTORSHIP |
YES/NO |
NO |
|
--EXPORT ACTIVITIES |
YES/NO |
NO |
|
--AFFILIATION |
YES/NO |
YES |
|
--LISTED |
YES/NO |
YES |
|
--OTHER MERIT FACTORS |
YES/NO |
YES |
|
DEFAULTER
|
|
|
|
--RBI |
YES/NO |
NO |
|
--EPF |
YES/NO |
NO |
|
TOTAL |
|
54 |
This score serves as a reference to assess
SC’s credit risk and to set the amount of credit to be extended. It is calculated
from a composite of weighted scores obtained from each of the major sections of
this report. The assessed factors and their relative weights (as indicated
through %) are as follows:
Financial
condition (40%) Ownership
background (20%) Payment record
(10%)
Credit history
(10%) Market trend (10%) Operational size
(10%)
RATING EXPLANATIONS
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
>86 |
Aaa |
Possesses an extremely sound financial base with the strongest capability
for timely payment of interest and principal sums |
Unlimited |
|
71-85 |
Aa |
Possesses adequate working capital. No caution needed for credit
transaction. It has above average (strong) capability for payment of interest
and principal sums |
Large |
|
56-70 |
A |
Financial & operational base are regarded healthy. General
unfavourable factors will not cause fatal effect. Satisfactory capability for
payment of interest and principal sums |
Fairly Large |
|
41-55 |
Ba |
Overall operation is considered normal. Capable to meet normal
commitments. |
Satisfactory |
|
26-40 |
B |
Capability to overcome financial difficulties seems comparatively
below average. |
Small |
|
11-25 |
Ca |
Adverse factors are apparent. Repayment of interest and principal sums
in default or expected to be in default upon maturity |
Limited with
full security |
|
<10 |
C |
Absolute credit risk exists. Caution needed to be exercised |
Credit not
recommended |
|
-- |
NB |
New Business |
-- |
This report is issued at your request without any
risk and responsibility on the part of MIRA INFORM PRIVATE LIMITED (MIPL)
or its officials.