MIRA INFORM REPORT
[]
|
Report No. : |
305432.2 |
|
Report Date : |
09.02.2015 |
IDENTIFICATION DETAILS
|
Name : |
CIPLA LIMITED |
|
|
|
|
Registered
Office : |
Cipla House, |
|
|
|
|
Country : |
|
|
|
|
|
Financials (as
on) : |
31.03.2014 |
|
|
|
|
Date of
Incorporation : |
17.08.1935 |
|
|
|
|
Com. Reg. No.: |
11-002380 |
|
|
|
|
Capital
Investment / Paid-up Capital : |
Rs.1605.800 Millions |
|
|
|
|
CIN No.: [Company Identification
No.] |
L24239MH1935PLC002380 |
|
|
|
|
TAN No.: [Tax Deduction &
Collection Account No.] |
MUMC00352C |
|
|
|
|
PAN: |
Not Available |
|
|
|
|
Legal Form : |
A Public Limited Liability Company. The company’s Shares are Listed on
the Stock Exchanges. |
|
|
|
|
Line of Business
: |
Manufacturer and Distributor of Drugs and Healthcare Products. |
|
|
|
|
No. of Employees
: |
Information
denied by management |
RATING & COMMENTS
|
MIRA’s Rating : |
Aa (80) |
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
71-85 |
Aa |
Possesses adequate working capital. No caution needed for credit
transaction. It has above average (strong) capability for payment of interest
and principal sums |
Large |
|
Status : |
Excellent |
|
|
|
|
Payment Behaviour : |
Regular |
|
|
|
|
Litigation : |
Exist |
|
|
|
|
Comments : |
Subject is a well-established and reputed company having excellent
track record. The rating reflect Cipla’s reputed brand having global presence
supported by its well established and experienced promoters, dominant market
position in domestic pharmaceutical industry, robust operational performance
and strong financial risk profile. Trade relations are reported as fair. Business is active. Payments are
reported to be regular and as per commitments. The company can be considered for business dealings at usual trade
terms and conditions. |
NOTES :
Any query related to this report can be made
on e-mail : infodept@mirainform.com
while quoting report number, name and date.
EXTERNAL AGENCY RATING
|
Rating Agency Name |
CARE |
|
Rating |
Long Term Bank Facilities: “AAA” |
|
Rating Explanation |
Highest degree of safety and lowest credit
risk. |
|
Date |
January 06, 2015 |
|
Rating Agency Name |
CARE |
|
Rating |
Short Term Bank Facilities: “A1+” |
|
Rating Explanation |
Very strong degree of safety and lowest
credit risk. |
|
Date |
January 06, 2015 |
RBI DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available RBI Defaulters’ list.
EPF (Employee Provident Fund) DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available EPF (Employee Provident Fund) Defaulters’ list as of
31-03-2014.
INFORMATION DENIED
Reception (91-22-24826000)
LOCATIONS
|
Registered Office : |
Cipla House, Peninsula Business Park, Ganpatrao Kadam Marg, Lower Parel, Mumbai – 400013, Maharashtra, India |
|
Tel. No.: |
91-22-23095521 / 24826701 / 24826300/24826000 |
|
Fax No.: |
91-22-24826893/24826120 |
|
E-Mail : |
|
|
Website : |
http://www.cipla.com |
|
|
|
|
Corporate Office/ Head Office : |
289, Opposite Sahil Hotel, Near Citi Center Mall, Bellasis
Road, Mumbai Central, Mumbai – 400008, Maharashtra, India |
|
Tel. No.: |
91-22-23095521/ 23082891/ 23023272/ 23025272 |
|
Fax No.: |
91-22-23070013/ 23070393/ 85/ 23008101 |
|
|
|
|
Branch Office: |
C/4, Raj Plaza, LBS Marg, Vikhroli (W), Mumbai-400083, Maharashtra, India |
|
|
|
|
Factory 1: |
Virgonagar, Old Madras Road, Bangalore – 560049, Karnataka, India |
|
|
|
|
Factory 2: |
Bommasandra-Jigani Link Road, Industrial Area, KIADB 4th Phase, Bangalore - 560099, Karnataka, India |
|
|
|
|
Factory 3: |
MIDC, Patalganga, District Raigad – 410220, Maharashtra, India |
|
|
|
|
Factory 4: |
MIDC Industrial Area, Kurkumbh, Daund District Pune - 413802, Maharashtra, India |
|
|
|
|
Factory 5: |
Verna Industrial Estate, Verna, Salcette, Panaji – 403722, Goa, India |
|
|
|
|
Factory 6: |
Village Malpur Upper, P.O. Bhud, Nalagarh, Baddi, District Solan - 173205, Himachal Pradesh, India |
|
|
|
|
Factory 7: |
Village Kumrek, P.O. Rangpoo-737132, East District Sikkim |
|
|
|
|
Factory 8: |
Indore SEZ, Phase ll, Sector III, Pharma Zone, P.O. Pithampur, District Dhar -454774, Madhya Pradesh, India |
DIRECTORS
As on 31.03.2014
|
Name : |
Dr. Y.K. Hamied |
|
Designation : |
Chairman |
|
|
|
|
Name : |
Mr. M.K. Hamied |
|
Designation : |
Vice-Chairman |
|
|
|
|
Name : |
Mr. Subhanu Saxena |
|
Designation : |
Managing Director and Global Chief Executive Officer |
|
|
|
|
Name : |
Mr. S. Radhakrishnan |
|
Designation : |
Whole-time
Director |
|
|
|
|
Name : |
Mr. Rajesh Garg |
|
Designation : |
Executive Director and Global Chief Financial Officer |
|
|
|
|
Name : |
Dr. H.R. Manchanda |
|
Designation : |
Non-Executive
Directors |
|
|
|
|
Name : |
Mr. V.C. Kotwal |
|
Designation : |
Non-Executive
Directors |
|
|
|
|
Name : |
Mr. Pankaj Patel |
|
Designation : |
Non-Executive
Directors |
|
|
|
|
Name : |
Mr. Ashok Sinha |
|
Designation : |
Non-Executive
Directors |
|
|
|
|
Name : |
Dr. Peter Mugyenyi |
|
Designation : |
Non-Executive
Directors |
|
|
|
|
Name : |
Mr. Adil Zainulbhai |
|
Designation : |
Non-Executive
Directors |
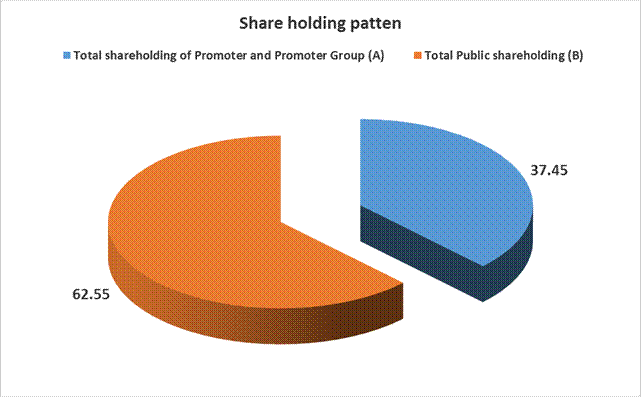
SHAREHOLDING PATTERN
As on 31.12.2014
|
Category of
Shareholder |
No.
of Shares |
Percentage
of Holding |
|
(A) Shareholding of Promoter
and Promoter Group |
|
|
|
|
|
|
|
|
111781000 |
14.17 |
|
|
6022791 |
0.76 |
|
|
117803791 |
14.93 |
|
|
|
|
|
|
177682187 |
22.52 |
|
|
177682187 |
22.52 |
|
Total shareholding of Promoter
and Promoter Group (A) |
295485978 |
37.45 |
|
(B) Public Shareholding |
|
|
|
|
|
|
|
|
33821517 |
4.29 |
|
|
1936258 |
0.25 |
|
|
55655339 |
7.05 |
|
|
176769128 |
22.40 |
|
|
15305249 |
1.94 |
|
|
15305249 |
1.94 |
|
|
283487491 |
35.93 |
|
|
|
|
|
|
33928372 |
4.30 |
|
|
|
|
|
|
51644289 |
6.55 |
|
|
95181347 |
12.06 |
|
|
29271372 |
3.71 |
|
|
1392260 |
0.18 |
|
|
134458 |
0.02 |
|
|
27411894 |
3.47 |
|
|
332385 |
0.04 |
|
|
375 |
0.00 |
|
|
210025380 |
26.62 |
|
Total Public shareholding (B) |
493512871 |
62.55 |
|
Total (A)+(B) |
788998849 |
100.00 |
|
(C) Shares held by Custodians
and against which Depository Receipts have been issued |
0 |
0.00 |
|
|
0 |
0.00 |
|
|
13922508 |
0.00 |
|
|
13922508 |
0.00 |
|
Total (A)+(B)+(C) |
802921357 |
0.00 |
Shareholding of securities (including shares, warrants,
convertible securities) of persons belonging to the category Promoter and
Promoter Group:
|
Sl.No. |
Name of the
Shareholder |
Details of Shares held |
|
|
No. of Shares
held |
As a % |
||
|
1 |
Dr. Y. K. Hamied |
116929500 |
14.56 |
|
2 |
Dr. Y. K. Hamied |
7770750 |
0.97 |
|
3 |
Dr. Y. K. Hamied |
127500 |
0.02 |
|
4 |
M. K. Hamied |
32854500 |
4.09 |
|
5 |
M. K. Hamied |
6835500 |
0.85 |
|
6 |
Farida Hamied |
41914937 |
5.22 |
|
7 |
Sophie Ahmed |
15471000 |
1.93 |
|
8 |
Sophie Ahmed |
15471000 |
1.93 |
|
9 |
Sophie Ahmed |
11322250 |
1.41 |
|
10 |
Sophie Ahmed |
3717750 |
0.46 |
|
11 |
Shirin Hamied |
6363000 |
0.79 |
|
12 |
Kamil Hamied |
6354000 |
0.79 |
|
13 |
Kamil Hamied |
4495500 |
0.56 |
|
14 |
Kamil Hamied |
90000 |
0.01 |
|
15 |
Samina Hamied |
5481000 |
0.68 |
|
16 |
Samina Hamied |
4378500 |
0.55 |
|
17 |
Rumana Hamied |
5472000 |
0.68 |
|
18 |
Rumana Hamied |
4414500 |
0.55 |
|
19 |
M. N. Rajkumar Garments LLP |
2601852 |
0.32 |
|
20 |
Shree Riddhi Chemicals LLP |
2434970 |
0.30 |
|
21 |
Alps Remedies Private Limited. |
492985 |
0.06 |
|
22 |
Hamsons Laboratories LLP |
492602 |
0.06 |
|
23 |
Neo Research Labs Private
Limited |
382 |
0.00 |
|
|
Total |
295485978 |
36.80 |
Shareholding of securities (including shares, warrants,
convertible securities) of persons belonging to the category Public and holding
more than 1% of the total number of shares:
|
Sl. No. |
Name of the
Shareholder |
No. of Shares
held |
Shares as % of
Total No. of Shares |
|
|
1 |
Life Insurance Corporation of
India |
53587684 |
6.67 |
|
|
2 |
Oppenheimer Developing Markets
Fund |
22160864 |
2.76 |
|
|
3 |
Virtus Emerging Markets
Opportunities Fund |
13185787 |
1.64 |
|
|
4 |
Vontobel India Fund |
8596897 |
1.07 |
|
|
5 |
Vontobel Fund A/C Vontobel Fund
Emerging |
8511275 |
1.06 |
|
|
|
Total |
106042507 |
13.21 |
Shareholding of securities (including shares, warrants,
convertible securities) of persons (together with PAC) belonging to the
category “Public” and holding more than 5% of the total number of shares of the
company:
|
Sl. No. |
Name(s) of the shareholder(s)
and the Persons Acting in Concert (PAC) with them |
No. of Shares |
Total shares
(including underlying shares assuming full conversion of warrants and
convertible securities) as a % of diluted share capital |
|
|
1 |
Life Insurance Corporation of India |
53587684 |
6.67 |
|
|
|
Total |
53587684 |
6.67 |
Details of Depository Receipts (DRs)
|
Sl. No. |
Type of
Outstanding DR (ADRs, GDRs, SDRs, etc.) |
No. of
Outstanding DRs |
Shares
Underlying Outstanding DRs as % of Total No. of Shares |
|
1 |
GDR |
13922508 |
1.73 |
|
|
Total |
13922508 |
1.73 |
BUSINESS DETAILS
|
Line of Business : |
Manufacturer and Distributor of Drugs and Healthcare Products. |
|
|
|
|
Products : |
·
Drugs ·
Healthcare Products |
|
|
|
|
Brand Names : |
Not Available |
|
|
|
|
Agencies Held : |
Not Available |
|
|
|
|
Exports : |
Not Applicable |
|
|
|
|
Imports : |
Not Applicable |
|
|
|
|
Terms : |
|
|
Selling : |
Not Divulged |
|
|
|
|
Purchasing : |
Not Divulged |
GENERAL INFORMATION
|
Suppliers : |
|
||||||||||||
|
|
|
||||||||||||
|
Customers : |
|
||||||||||||
|
|
|
||||||||||||
|
No. of Employees : |
Information denied by management |
||||||||||||
|
|
|
||||||||||||
|
Bankers : |
· Bank of Baroda · Canara Bank · Corporation Bank · Indian Overseas Bank · Standard Chartered Bank · The Hongkong and Shanghai Banking Corporation Limited · Union Bank of India |
||||||||||||
|
|
|
||||||||||||
|
Facilities : |
|
|
Auditors : |
|
|
Name 1 : |
V. Sankar Aiyar and Company Chartered Accountants |
|
|
|
|
Name 2 : |
R.G.N. Price and Company Chartered Accountants |
|
|
|
|
Subsidiaries (held directly): |
· Cipla FZE · Goldencross Pharma Private Limited · Cipla (Mauritius) Limited · Meditab Specialities Private Limited · Cipla Medpro South Africa (Pty) Limited (formerly Cipla Medpro South Africa Limited)# · Cipla Holding B.V.@ |
|
|
|
|
Subsidiaries
(held indirectly) : |
· Cipla (UK) Limited · Cipla Australia Pty. Limited · Cipla (EU) Limited · Medispray Laboratories Private Limited · Sitec Labs Private Limited · Four M Propack Private Limited · Meditab Holdings Limited · Meditab Pharmaceuticals South Africa (Pty) Limited · Meditab Specialities New Zealand Limited · Cipla Ilaç Ticaret Anonim Sirketi · Cipla USA Inc. · Cipla Kenya Limited · Cipla Malaysia Sdn. Bhd. · Cipla Europe NV* · Cipla Quality Chemical Industries Limited (formerly Quality Chemical Industries Limited) ** · Celeris d.o.o. *** · Cipla Medpro Manufacturing Proprietary Limited# · Galilee Marketing Proprietary Limited# · Inyanga Trading 386 Proprietary Limited# · Xeragen Laboratories Proprietary Limited# · Cipla Medpro Holdings Proprietary Limited# · Cape to Cairo Exports Proprietary Limited# · Cipla Agrimed Proprietary Limited# · Cipla Dibcare Proprietary Limited# · Cipla Health Care Proprietary Limited# · Cipla Life Sciences Proprietary Limited# · Cipla-Medpro Proprietary Limited# · Cipla-Medpro Distribution Centre Proprietary Limited# · Cipla Medpro ARV Proprietary Limited# · Cipla Medpro Botswana Proprietary Limited# · Cipla Medpro Cardio Respiratory Proprietary Limited# · Cipla Medpro Research and Development Proprietary Limited# · Cipla Nutrition Proprietary Limited# · Cipla Personal Care Proprietary Limited# · Cipla Vet Proprietary Limited# · Gardian Cipla Proprietary Limited# · Medpro Gen Proprietary Limited# · Medpro Holdings Proprietary Limited# · Medpro Pharmaceutica Proprietary Limited# · Medpro Pharmaceutica Africa Proprietary Limited# · Medpro-On-Line Proprietary Limited# · Med Man Care Proprietary Limited# · Smith and Couzin Proprietary Limited# |
|
|
|
|
Associates : |
· Quality Chemical Industries Limited# # · Stempeutics Research Private Limited · Mabpharm Private Limited |
|
|
|
|
Joint Venture :
|
·
Aspen-Cipla
Australia Pty Limited |
|
|
|
|
Entities over which
Key Management Personnel are able to exercise significant influence: |
· Okasa Private Limited · Okasa Pharma Private Limited · Cipla Cancer and AIDS Foundation · Hamied Foundation (earlier known as Dr. K. A. Hamied Foundation) · Cipla Foundation |
@ With effect from 28/08/2013
* With effect from 30/09/2013
** With effect from 20/11/2013
*** With effect from 04/12/2013
# With effect from 15/07/2013
# # from 01/10/2010 upto 19/11/2013
CAPITAL STRUCTURE
As on 31.03.2014
Authorised Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
875000000 |
Equity Shares |
Rs.2/- each |
Rs.1750.000 Millions |
Issued Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
803924752 |
Equity Shares |
Rs.2/- each |
Rs.1607.800
Millions |
Subscribed & Paid-up Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
802921357 |
Equity Shares |
Rs.2/- each |
Rs.1605.800
Millions |
There is no change in the shares outstanding
at the beginning and at the end of the reporting date and immediately preceding
reporting date
·
Details of Shareholders holding more than 5 percent
shares in the Company
|
Name of
Shareholder |
Number
of Shares |
% holding |
|
Y.K. Hamied |
12,48,27,750 |
15.55 |
|
Farida Hamied |
4,19,14,937 |
5.22 |
|
Sophie Ahmed |
4,59,82,000 |
5.73 |
|
Life Insurance Corporation of India |
5,24,97,490 |
6.54 |
·
Terms and
Rights attached to Equity Shares
The Company has only one class of
equity shares having a par value of Rs.2 per share. Each holder of equity share
is entitled to one vote per share. The Company declares and pays dividends in
Indian Rupees. The dividend proposed by the Board of Directors is subject to
the approval of the shareholders in the ensuing Annual General Meeting. In the
event of liquidation of the Company, the holders of equity shares will be
entitled to receive remaining assets of the Company, after distribution of all
preferential amounts. The distribution will be in proportion to the number of
equity shares held by the shareholder.
FINANCIAL DATA
[all figures are
in Rupees Millions]
ABRIDGED BALANCE
SHEET
|
SOURCES OF FUNDS |
31.03.2014 |
31.03.2013 |
31.03.2012 |
|
I.
EQUITY
AND LIABILITIES |
|
|
|
|
(1)Shareholders' Funds |
|
|
|
|
(a) Share Capital |
1605.800 |
1605.800 |
1605.800 |
|
(b) Reserves & Surplus |
99310.600 |
87089.400 |
73897.000 |
|
(c) Money
received against share warrants |
0.000 |
0.000 |
0.000 |
|
|
|
|
|
|
(2) Share Application money pending allotment |
0.000 |
0.000 |
0.000 |
|
Total
Shareholders’ Funds (1) + (2) |
100916.400 |
88695.200 |
75502.800 |
|
|
|
|
|
|
(3)
Non-Current Liabilities |
|
|
|
|
(a) long-term borrowings |
4.300 |
5.500 |
22.000 |
|
(b) Deferred tax liabilities (Net) |
3112.000 |
2812.000 |
2324.500 |
|
(c) Other long term liabilities |
300.000 |
300.000 |
300.000 |
|
(d) long-term provisions |
739.900 |
473.400 |
291.200 |
|
Total Non-current Liabilities (3) |
4156.200 |
3590.900 |
2937.700 |
|
|
|
|
|
|
(4) Current Liabilities |
|
|
|
|
(a) Short term borrowings |
8769.100 |
9652.600 |
100.000 |
|
(b) Trade payables |
9625.600 |
8270.900 |
6858.500 |
|
(c) Other current
liabilities |
3332.000 |
2426.200 |
2327.900 |
|
(d) Short-term provisions |
2440.700 |
2296.300 |
2206.400 |
|
Total Current Liabilities (4) |
24167.400 |
22646.000 |
11492.800 |
|
|
|
|
|
|
TOTAL |
129240.000 |
114932.100 |
89933.300 |
|
|
|
|
|
|
II.
ASSETS |
|
|
|
|
(1) Non-current assets |
|
|
|
|
(a) Fixed Assets |
|
|
|
|
(i) Tangible assets |
35195.900 |
34182.900 |
30026.600 |
|
(ii) Intangible Assets |
46.000 |
0.000 |
0.000 |
|
(iii) Capital
work-in-progress |
3196.400 |
3399.900 |
3434.500 |
|
(iv)
Intangible assets under development |
570.500 |
103.500 |
0.000 |
|
(b) Non-current Investments |
33282.800 |
5143.600 |
4618.300 |
|
(c) Deferred tax assets (net) |
0.000 |
0.000 |
0.000 |
|
(d) Long-term Loan and Advances |
5353.000 |
3737.200 |
3935.000 |
|
(e) Other Non-current assets |
615.700 |
3.100 |
2.400 |
|
Total Non-Current Assets |
78260.300 |
46570.200 |
42016.800 |
|
|
|
|
|
|
(2) Current assets |
|
|
|
|
(a) Current investments |
2588.500 |
20874.600 |
5733.200 |
|
(b) Inventories |
25111.600 |
23433.700 |
18245.000 |
|
(c) Trade receivables |
17281.000 |
16452.200 |
15193.100 |
|
(d) Cash and cash
equivalents |
460.400 |
1050.700 |
550.600 |
|
(e) Short-term loans and
advances |
5155.600 |
6527.900 |
7654.800 |
|
(f) Other current assets |
382.600 |
22.800 |
539.800 |
|
Total Current Assets |
50979.700 |
68361.900 |
47916.500 |
|
|
|
|
|
|
TOTAL |
129240.000 |
114932.100 |
89933.300 |
PROFIT & LOSS
ACCOUNT
|
|
PARTICULARS |
31.03.2014 |
31.03.2013 |
31.03.2012 |
|
|
|
SALES |
|
|
|
|
|
|
|
Income |
93,802.900 |
82,024.200 |
69,775.000 |
|
|
|
Other Income |
2,802.800 |
2,291.300 |
1,483.000 |
|
|
|
TOTAL (A) |
96,605.700 |
84,315.500 |
71,258.000 |
|
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
|
|
Cost of Materials Consumed |
31,453.400 |
26,468.300 |
23,008.500 |
|
|
|
Purchase of Traded Goods |
7,734.000 |
7,068.900 |
5,555.500 |
|
|
|
Changes in Inventories of Finished Goods, Work-in-Process
and Traded Goods |
(1,581.200) |
(2,907.500) |
112.400 |
|
|
|
Employee Benefits Expense |
12,847.500 |
9,692.800 |
7,282.100 |
|
|
|
Other Expenses |
23,453.900 |
20,510.300 |
17,997.900 |
|
|
|
TOTAL (B) |
73,907.600 |
60,832.800 |
53,956.400 |
|
|
|
|
|
|
|
|
Less |
PROFIT
BEFORE INTEREST, TAX, DEPRECIATION AND AMORTISATION (A-B) (C) |
22,698.100 |
23,482.700 |
17,301.600 |
|
|
|
|
|
|
|
|
|
Less |
FINANCIAL
EXPENSES (D) |
1,278.600 |
333.800 |
266.300 |
|
|
|
|
|
|
|
|
|
|
PROFIT
BEFORE TAX, DEPRECIATION AND AMORTISATION (C-D) (E) |
21,419.500 |
23,148.900 |
17,035.300 |
|
|
|
|
|
|
|
|
|
Less/ Add |
DEPRECIATION/
AMORTISATION (F) |
3,236.100 |
3,030.300 |
2,820.700 |
|
|
|
|
|
|
|
|
|
|
PROFIT BEFORE
TAX (E-F) (G) |
18,183.400 |
20,118.600 |
14,214.600 |
|
|
|
|
|
|
|
|
|
Less |
TAX (H) |
4,300.000 |
5,047.500 |
2,975.000 |
|
|
|
|
|
|
|
|
|
|
PROFIT AFTER TAX
(G-H) (I) |
13,883.400 |
15,071.100 |
11,239.600 |
|
|
|
|
|
|
|
|
|
Add |
PREVIOUS
YEARS’ BALANCE BROUGHT FORWARD |
42,701.300 |
31,108.900 |
22,979.300 |
|
|
|
|
|
|
|
|
|
Less |
APPROPRIATIONS |
|
|
|
|
|
|
|
Proposed Dividend |
1,605.800 |
1,605.800 |
1,600.000 |
|
|
|
Tax on Dividend |
272.900 |
272.900 |
260.000 |
|
|
|
Transfer to General Reserve |
1,400.000 |
1,600.000 |
1,250.000 |
|
|
BALANCE CARRIED
TO THE B/S |
53,306.000 |
42,701.300 |
31,108.900 |
|
|
|
|
|
|
|
|
|
|
EARNINGS IN
FOREIGN CURRENCY |
|
|
|
|
|
|
|
Export Earnings |
49479.600 |
44261.600 |
36920.300 |
|
|
|
Technical Know-how/ Fees |
1873.900 |
604.300 |
295.500 |
|
|
|
Others |
145.300 |
81.300 |
65.900 |
|
|
TOTAL EARNINGS |
51498.800 |
44947.200 |
37281.7 |
|
|
|
|
|
|
|
|
|
|
IMPORTS |
|
|
|
|
|
|
|
Raw Materials / Packing Materials |
13306.400 |
12067.800 |
9543.400 |
|
|
|
Components and Spare Parts |
307.000 |
262.300 |
324.200 |
|
|
|
Capital Goods |
647.100 |
832.100 |
1465.000 |
|
|
TOTAL IMPORTS |
14260.500 |
13162.200 |
11332.600 |
|
|
|
|
|
|
|
|
|
|
Earnings Per
Share (Rs.) |
17.29 |
18.77 |
14.00 |
|
QUARTERLY RESULTS
|
Particulars |
|
30.06.2014 (Unaudited) Quarter 1 |
30.09.2014 (Unaudited) Quarter 2 |
|
Net Sales / Interest Earned /
Operating Income |
|
24896.300 |
24744.600 |
|
Other Income |
|
232.400 |
372.800 |
|
Expenditure |
|
(19357.800) |
(19419.300) |
|
Interest |
|
(318.400) |
(251.500) |
|
Profit Before Depreciation and
Tax |
|
5452.500 |
5446.600 |
|
Depreciation |
|
(1097.700) |
(1142.400) |
|
Profit before Tax |
|
4354.800 |
4304.200 |
|
Tax |
|
(994.100) |
(982.500) |
|
Net Profit |
|
3360.700 |
3321.700 |
KEY RATIOS
|
PARTICULARS |
|
31.03.2014 |
31.03.2013 |
31.03.2012 |
|
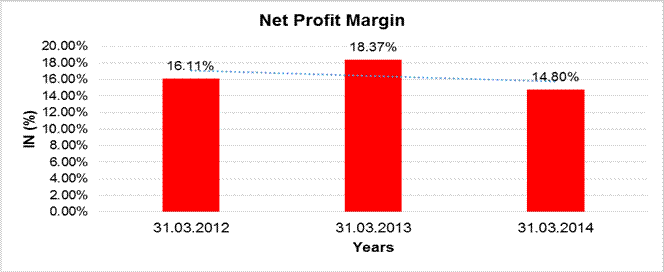
Net Profit Margin (PAT / Sales) |
(%) |
14.80 |
18.37 |
16.11 |
|
|
|
|
|
|
|
Operating Profit Margin (PBIDT/Sales) |
(%) |
24.20 |
28.63 |
24.80 |
|
|
|
|
|
|
|
Return on Total Assets (PBT/Total Assets} |
(%) |
19.72 |
18.93 |
18.67 |
|
|
|
|
|
|
|
Return on Investment (ROI) (PBT/Networth) |
|
0.18 |
0.23 |
0.19 |
|
|
|
|
|
|
|
Debt Equity Ratio (Total Debt /Networth) |
|
0.09 |
0.11 |
0.00 |
|
|
|
|
|
|
|
Current Ratio (Current Asset/Current Liability) |
|
2.11 |
3.02 |
3.85 |
FINANCIAL ANALYSIS
[All figures are
in Rupees Millions]
DEBT EQUITY RATIO
|
Particular |
31.03.2012 |
31.03.2013 |
31.03.2014 |
|
|
(Rs. In Millions) |
(Rs. In Millions) |
(Rs. In Millions) |
|
Share Capital |
1605.800 |
1605.800 |
1605.800 |
|
Reserves & Surplus |
73897.000 |
87089.400 |
99310.600 |
|
Net worth |
75502.800 |
88695.200 |
100916.400 |
|
|
|
|
|
|
long-term borrowings |
22.000 |
5.500 |
4.300 |
|
Short term borrowings |
100.000 |
9652.600 |
8769.100 |
|
Total borrowings |
122.000 |
9658.100 |
8773.400 |
|
Debt/Equity ratio |
0.002 |
0.109 |
0.087 |

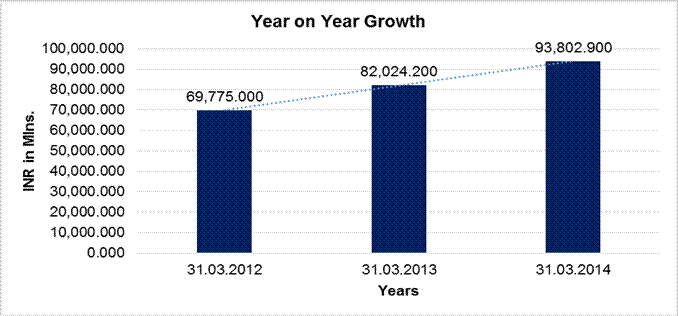
YEAR-ON-YEAR GROWTH
|
Year
on Year Growth |
31.03.2012 |
31.03.2013 |
31.03.2014 |
|
|
Rs.
In Millions |
Rs.
In Millions |
Rs.
In Millions |
|
Sales |
69,775.000 |
82,024.200 |
93,802.900 |
|
|
|
17.555 |
14.360 |
NET PROFIT MARGIN
|
Net
Profit Margin |
31.03.2012 |
31.03.2013 |
31.03.2014 |
|
|
Rs.
In Millions |
Rs.
In Millions |
Rs.
In Millions |
|
Sales
|
69,775.000 |
82,024.200 |
93,802.900 |
|
Profit |
11,239.600 |
15,071.100 |
13,883.400 |
|
|
16.11% |
18.37% |
14.80% |
LOCAL AGENCY FURTHER INFORMATION
CURRENT MATURITIES
OF LONG-TERM DEBT DETAILS:
|
Particulars |
31.03.201 |
31.03.2013 |
31.03.2012 |
|
|
(Rs. In Millions) |
||
|
Current maturities of long-term debt |
0.600 |
1.400 |
7.200
|
|
|
|
|
|
|
Total |
0.600 |
1.400 |
7.200
|
|
Sr. No. |
Check List by Info Agents |
Available in
Report (Yes / No) |
|
1] |
Year of Establishment |
Yes |
|
2] |
Locality of the firm |
Yes |
|
3] |
Constitutions of the firm |
Yes |
|
4] |
Premises details |
No |
|
5] |
Type of Business |
Yes |
|
6] |
Line of Business |
Yes |
|
7] |
Promoter's background |
Yes |
|
8] |
No. of employees |
No |
|
9] |
Name of person contacted |
No |
|
10] |
Designation of contact
person |
No |
|
11] |
Turnover of firm for last
three years |
Yes |
|
12] |
Profitability for last
three years |
Yes |
|
13] |
Reasons for variation
<> 20% |
----------- |
|
14] |
Estimation for coming
financial year |
No |
|
15] |
Capital in the business |
Yes |
|
16] |
Details of sister
concerns |
Yes |
|
17] |
Major suppliers |
No |
|
18] |
Major customers |
No |
|
19] |
Payments terms |
No |
|
20] |
Export / Import details
(if applicable) |
No |
|
21] |
Market information |
---------- |
|
22] |
Litigations that the firm
/ promoter involved in |
Yes |
|
23] |
Banking Details |
Yes |
|
24] |
Banking facility details |
Yes |
|
25] |
Conduct of the banking
account |
---------- |
|
26] |
Buyer visit details |
---------- |
|
27] |
Financials, if provided |
Yes |
|
28] |
Incorporation details, if
applicable |
Yes |
|
29] |
Last accounts filed at
ROC |
Yes |
|
30] |
Major Shareholders, if
available |
Yes |
|
31] |
Date of Birth of
Proprietor/Partner/Director, if available |
No |
|
32] |
PAN of
Proprietor/Partner/Director, if available |
No |
|
33] |
Voter ID No of Proprietor/Partner/Director,
if available |
No |
|
34] |
External Agency Rating,
if available |
Yes |
Note: The Registered
Office of the Company was shifted from above address to present address w.e.f.
01.04.2014.
LITIGATION
DETAILS:
|
Case Details |
|||||||
|
Bench:-Bombay |
|||||||
|
|
|||||||
|
Lodging No.:- |
ITXAL/2141/2014 |
Filing Date:- |
26.12.2014 |
||||
|
|
|||||||
|
Petitioner:- |
COMMISSIONER OF INCOME TAX, |
Respondent:- |
CIPLA LIMITED |
||||
|
Petn. Adv.:- |
Suresh Kumar (I2100) |
Resp. Adv.:- |
0 (0) |
||||
|
District:- |
MUMBAI |
||||||
|
|
|||||||
|
Bench:- |
DIVISION |
||||||
|
|
|||||||
|
Status:- |
Pre-Admission |
Category:- |
TAX APPEALS |
||||
|
|
|||||||
|
Last Date: |
05.01.2015 |
Stage: |
- |
||||
|
Last Coram: |
REGISTRAR (OS) PROTHONOTATRY & SR.
MASTER |
||||||
|
Act: |
Income Tax Act, 1961 Under Section: - 260A |
||||||
UNSECURED LOAN:
|
Particulars |
31.03.2014 Rs.
In Millions |
31.03.2014 Rs.
In Millions |
|
LONG TERM
BORROWINGS |
|
|
|
Deferred Payment Liability - Sales Tax Deferral Loan (Sales tax deferral loan is interest free and repayable in 3 equal instalments from the 10th year.) |
4.300 |
5.500 |
|
SHORT TERM
BORROWINGS |
|
|
|
Packing Credit
from Banks |
8769.100 |
9557.700 |
|
Total |
8773.400 |
9563.200 |
GLOBAL BUSINESS
REVIEW
With a footprint across five continents, subject is moving fast towards its goal of making affordable healthcare available to all. The Company’s revenue from operations on a consolidated basis during the financial year 2013-14 amounted to Rs.102180.000 Millions against Rs.83880.000 Millions in the previous year, recording a growth of 21.8%. The income from operations for domestic business increased by 14.7%, from Rs.35690.000 Millions in the previous financial year to Rs.40940.000 Millions in the financial year. Total exports increased by 25.0% during the year to Rs.56590.000 Millions. During the year, operating margin reduced by 5.4%. This was primarily due to the change in product mix, higher investments in R and D and talent acquisition. As a result, profit for the year reduced by 10.1% to Rs.13890.000 Millions from Rs.15450.000 Millions in the previous financial year. India Ratings and Research Private Limited, a Fitch Group Company, assigned a Long-Term Issuer Rating of ‘IND AAA’ with a stable outlook to the Company. The rating is the highest assigned in Fitch’s rating scale and indicates the highest degree of safety regarding timely servicing of financial obligations and lowest credit risk.
STATEMENT OF STANDALONE UNAUDITED RESULTS FOR THE QUARTER ENDED 30TH
SEPTEMBER 2014
(Rs. In Millions)
|
|
|
Quarter Ended Year |
Half Ended
Year |
|
|
|
Particulars |
30.09.2014 |
30.06.2014 |
30.09.2014 |
|
|
|
Unaudited |
||
|
1 |
Income from operations |
|
|
|
|
|
(a) Net sales/income from operations (net of excise duty) |
23492.800 |
24015.200 |
47508.000 |
|
|
(b) Other operating income |
1403.500 |
729.400 |
2132.900 |
|
|
Total income from operations (net) |
24896.300 |
24744.600 |
49640.900 |
|
2 |
Expenses |
|
|
|
|
|
a) Cost of materials consumed |
8561.800 |
6846.600 |
15408.400 |
|
|
b) Purchases of stock-in-trade |
2568.600 |
1570.300 |
4138.900 |
|
|
c) Changes in inventories of finished goods, work-in- process and
stock-in-trade |
(1916.600) |
1462.100 |
(454.500) |
|
|
d) Employee benefits expense |
3592.800 |
3828.100 |
7420.900 |
|
|
e) Depreciation and amortisation expense |
1097.700 |
1142.400 |
2240.100 |
|
|
g) Other expenses |
6551.200 |
5712.200 |
12263.400 |
|
|
Total expenses |
20455.500 |
20561.700 |
41017.200 |
|
|
|
|
|
|
|
3 |
Profit from operations before other income, finance costs and
exceptional items (1-2) |
4440.800 |
4182.900 |
8623.700 |
|
4 |
Other income |
232.400 |
372.800 |
605.200 |
|
5 |
Profit before finance costs and exceptional items (3+4) |
4673.200 |
4555.700 |
9228.900 |
|
6 |
Finance costs |
318.400 |
251.500 |
569.900 |
|
7 |
Profit / (Loss) from Ordinary Activities after finance costs but
before tax (5-6) |
4354.800 |
4304.200 |
8659.000 |
|
8 |
Exceptional Item |
0.000 |
0.000 |
0.000 |
|
9 |
Net Profit / (Loss) from Ordinary Activities after tax (7-8) |
4354.800 |
4304.200 |
8659.000 |
|
10 |
Tax expense |
994.100 |
982.500 |
1976.600 |
|
11 |
Net Profit (+)/Loss
(-) after tax (9-10) |
3360.700 |
3321.700 |
6682.400 |
|
12 |
Paid-up equity share capital (Face value-7l per equity share) |
1605.800 |
1605.800 |
1605.800 |
|
13 |
Reserves (excluding revaluation reserves) as per Balance Sheet of previous accounting year |
|
|
|
|
14 |
Earnings per share |
|
|
|
|
|
-Basic |
*4.18 |
*4.14 |
*8.32 |
|
|
-Diluted |
*4.17 |
*4.13 |
*8.30 |
|
|
|
|
|
|
|
A |
Particulars of Shareholding |
|
|
|
|
1 |
Public Shareholding |
|
|
|
|
|
- Number of shares |
498206108 |
498510398 |
498206108 |
|
|
- Percentage of shareholding |
62.05 |
62.09% |
62.05 |
|
2 |
Promoters and Promoter group Shareholding a) Pledged/encumbered |
|
|
|
|
|
-Number of shares |
Nil |
Nil |
Nil |
|
|
-Percentage of shares (as a % of the total shareholding of promoter and
promoter group) |
Nil |
Nil |
Nil |
|
|
-Percentage of shares (as a % of the total share capital of the
Company) |
Nil |
Nil |
Nil |
|
|
b) Non-encumbered |
|
|
|
|
|
-Number of shares |
295485978 |
295485978 |
29585978 |
|
|
-Percentage of shares (as a % of the total shareholding of promoter
and promoter group) |
100.00% |
100.00% |
100.00% |
|
|
Percentage of shares (as a % of the total share capital of |
36.80% |
36.80% |
36.80% |
|
Particulars |
30.09.2014 |
|
Investment Complaints |
|
|
Pending at the beginning of the quarter |
Nil |
|
Received during the quarter |
10 |
|
Disposed of during the quarter |
10 |
|
Remaining unresolved at the end of the quarter |
Nil |
· The Company is essentially in the pharmaceutical business segment
· In 2003, the Company received notice of demand from the National Pharmaceutical Pricing Authority, Government of India on account of alleged overcharging in respect of certain drugs under the Drug s (Price Control) Order, 1995. This was contested before the jurisdictional High Courts in Mumbai, Karnataka and Allahabad wherein it was held in favor of the Company. The orders of Hon'ble High Court of Allahabad and Bombay were challenged before the Hon'ble Supreme Court of India by the Government. Although in the challenge to the decision of the Hon'ble Bombay High Court, the Hon'ble Supreme Court of India restored the matter to the Hon'ble Bombay High Court in August 2003 for interpreting the Drug Policy on the basis of directions and principles laid down by them and the same was pending, in the challenge to the Hon'ble High Court of Allahabad's order, in February 2013, the Hon'ble Supreme Court of India transferred the Bombay High Court petition also before itself for a final hearing on both the matters. In an earlier order, the Hon'ble Supreme Court has already restrained the Government from taking any coercive action against the Company. The Company has been legally advised that on the basis of these orders there is no probability of demand crystallizing. Hence no provision is considered necessary in respect of notice of demand received by the company up to date aggregating to Rs.17685.100 Millions.
· During the quarter ended 30th September, 2014, in accordance with Employee Stock Option Scheme dated 31st October, 2013 (“ESOS 2013 -A”), the Company has granted 95,248 stock options to employees at an exercise price equal to face value of the equity share. These options would vest not earlier than 1 year and not later than 2 years from the date of grant of options.
· The figures of the previous year/period have been regrouped/ recast to render them comparable with the figures of the current period
STANDALONE STATEMENT OF ASSETS AND LIABILITIES
|
Particulars |
As at 30.09.2014 |
|
|
|
Particulars |
|
|
A |
EQUITY AND
LIABILITIES |
|
|
1 |
Shareholder’s Funds |
|
|
|
a) Share Capital |
1605.800 |
|
|
b) Reserves & Surplus |
105803.400 |
|
|
c) Money received against share warrants |
0.000 |
|
|
Sub Total-
Shareholders funds |
107409.200 |
|
2 |
Share application
money pending allotment |
|
|
3 |
Minority Interest |
|
|
4 |
Non-current
liabilities |
|
|
|
(a) Long term borrowings |
4.100 |
|
|
(b) Other long term liabilities |
300.000 |
|
|
(c) Differed Tax |
3066.700 |
|
|
(d) Long Term Borrowing |
874.000 |
|
|
Sub Total- Non
Current Liabilities |
4244.800 |
|
5 |
Current liabilities |
|
|
|
(a) Short term borrowings |
16131.900 |
|
|
(b) Trade Payables |
12077.400 |
|
|
(c) Other current liabilities |
3529.400 |
|
|
(d) Short term provisions |
735.600 |
|
|
Sub Total- Current
Liabilities |
32474.300 |
|
|
TOTAL-EQUITY AND
LIABILITIES |
144128.300 |
|
B |
ASSETS |
|
|
1 |
Non-current assets |
|
|
|
(a) Fixed assets |
38126.800 |
|
|
(b) Long term loans and advances |
5874.900 |
|
|
(c)Non-Current Investment |
34541.900 |
|
|
(d)Other
Non-current Assets |
615.600 |
|
|
Sub-Total-
Noncurrent assets |
79159.200 |
|
2 |
Current assets |
|
|
|
a) Current Investments |
13867.800 |
|
|
b) Inventories |
26441.600 |
|
|
c) Trade Receivables |
17059.500 |
|
|
d) Cash and cash equivalents |
530.400 |
|
|
(e) Short term loans and advances |
6355.100 |
|
|
(f) Other current assets |
714.700 |
|
|
Sub-Total- current
assets |
64969.100 |
|
|
TOTAL ASSETS |
144128.300 |
· The above standalone financial results have been subjected to Limited Review by the Statutory Auditors, reviewed by the Audit Committee and approved by the Board of Directors at its meeting held on 13th November, 2014.
INDEX OF CHARGE:
|
Sr. No. |
Charge ID |
Date of Charge
Creation/Modification |
Charge amount
secured |
Charge Holder |
Address |
Service Request
Number (SRN) |
|
1 |
80066651 |
17/03/1998 * |
685,000,000.00 |
The Hongkong and Shanghai Banking Corporation Limited (Lead Charge Holder) |
52/60, Mahatma Gandhi Road, Bombay, Bombay, Maharashtra - 400023, India |
- |
FIXED ASSETS:
·
Land
·
Building
·
Plant and Machinery
·
Computer
·
Vehicles
·
Furniture and Fixture
·
PRESS RELEASE:
MMV ANNOUNCES COLLABORATIONS WITH CIPLA AND STRIDES
COLLABORATIONS SET TO ADVANCE LIFE-SAVING PRE-REFERRAL TREATMENT FOR
SEVERE MALARIA
Switzerland and India, 27 November 2014. Medicines for Malaria Venture signs collaboration agreements with two Indian pharmaceutical companies, Cipla Limited and Strides Arcolab Limited for the development of rectal articulate for pre-referral treatment of children with severe malaria.
The collaborations, established under the MMV-led "Improving Severe Malaria Outcomes" project funded by UNITAID1, aim to develop a rectal articulate product for submission to WHO prequalification. Cipla and Strides will each develop a product building on the clinical studies led by TDR, the Special Programmed for Research and Training in Tropical Diseases hosted by the World Health Organization.
The goal is to achieve WHO-prequalification of a rectal articulate product by 2016.
An estimated 5.6 million cases of severe malaria each year result in around 627,000 deaths, mostly of children under 5.2 Severe malaria is the result of inadequately treated uncomplicated malaria and can quickly lead to death.
A WHO-TDR trial conducted in 17,000 patients in Bangladesh, Ghana and Tanzania demonstrated that "if patients with severe malaria cannot be treated orally and access to injections will take several hours, a single inexpensive articulate suppository at the time of referral substantially reduces the risk of death or permanent disability."3
Although the WHO guidelines for the treatment of malaria recommend use of rectal articulate, 4 there is currently no product with WHO prequalification or marketing authorization by a stringent regulatory authority. This prevents international donors from funding the procurement of this potentially life-saving treatment.
“This is a significant milestone in our project to improve severe malaria outcomes,” said Dr. David Reddy, MMV’s CEO. “they have agreed a clear pathway with these two manufacturers to bring this product to an internationally acceptable quality standard. This will help make the product available to more patients, buying more time and saving more lives. They look forward to working closely with Cipla and Strides and thank UNITAID for supporting these efforts.”
Mr Subhanu Saxena, MD & Global CEO, The company said: “They are happy to join hands with MMV that shares our ongoing commitment to fight against malaria. Cipla is one of the major suppliers of antimalarial products worldwide and They have designed many innovative medicines for children in developing countries. Through this collaboration, They will endeavor to make rectal articulate accessible to each and every child with severe malaria in a pre-referral setting in malaria-endemic countries.”
“They are excited to be part of this initiative, as it will expand access to quality, affordable medicines for a wider population and save more lives,” said Mohan Kumar, CEO, Strides Arcolab Limited.
Notes for editors:
About the topic
An estimated 5.6 million cases of severe malaria each year result in around 627,000 deaths, mostly of children under 5.2 Severe malaria is the result of inadequately y treated uncomplicated malaria and can quickly lead to death.
A WHO-TDR trial conducted in 17,000 patients in Bangladesh, Ghana and Tanzania demonstrated that "if patients with severe malaria cannot be treated orally and access to injections will take several hours, a single inexpensive articulate suppository at the time of referral substantially reduces the risk of death or permanent disability."
Although the WHO guidelines for the treatment of malaria recommend use of rectal artesunate, 4 there is currently no product with WHO prequalification or marketing authorization by a stringent regulatory authority. This prevents international donors from funding the procurement of this potentially life - saving treatment.
About Cipla Limited:
Cipla is a global pharmaceutical company which uses cutting edge technology and innovation to meet the everyday needs of all patients. For more than 70 years, Cipla has emerged as one of the most respected pharmaceutical names in India as well as across more than 150 countries. Cipla’s portfolio includes over 1500 products in various therapeutic categories with one quality standard globally. Cipla’s turnover in 2013/14 was 1.7 billion USD.
Whilst delivering a long- term sustainable business, Cipla recognizes its duty to provide affordable medicines. Cipla’s emphasis on access for patients was recognized globally for the pioneering role played in HIV/AIDS treatment as the first pharmaceutical company to provide a triple combination anti - retroviral (ARV) in Africa at less than one dollar a day and thereby treating many millions of patients since 2001.
Cipla’s research and development focuses on developing innovative products and drug delivery systems and has given India and the world many ‘firsts’ for instance Triomune. In a tightly regulated environment, the company’s manufacturing facilities have approvals from all the main regulators including US FDA, UKMHRA, WHO, MCC, ANVISA, and PMDA which means the company provides one universal standard both domestically and internationally
About Strides Arcolab
Limited
Strides Arcolab Limited, listed on the Bombay Stock Exchange Limited (532531) and National Stock Exchange of India Limited (STAR), is a global pharmaceutical Company headquartered in Bangalore, India that develops and manufactures a wide range of IP-led niche pharmaceutical products.
The Company has 8 manufacturing facilities presence in more than 75 countries in developed and emerging markets.
About Medicines for
Malaria Venture (MMV):
MMV is a leading product development partnership (PDP) in the field of antimalarial drug research and development. Its mission is to reduce the burden of malaria in disease-endemic countries by discovering, developing and facilitating delivery of new, effective and affordable antimalarial drugs.
Since its foundation in 1999, MMV has developed and brought to registration four new medicines with its partners: Pyramax®, (pyronaridine-artesunate) co-developed with Shin Poong; Eurartesim® (dihydroartemisinin-piperaquine) with Sigma-Tau; Guilin’s artesunate injection for the treatment of severe malaria, Artesun®; and Coartem® Dispersible(artemether-lumefantrine), a child-friendly formulation developed with Novartis. Since 2009, over 250 million courses of Coartem Dispersible treatment have been supplied to 50 malaria-endemic countries; and since prequalification in 2010, an estimated 25 million vials of artesunate injection have been delivered, saving 165,000 additional lives.
Managing the largest portfolio of antimalarial R&D projects ever assembled, of over 65 projects, MMV has seven new drugs in clinical development addressing unmet medical needs in malaria, including medicines for children, pregnant women and relapsing malaria, and drugs that could support the elimination/eradication agenda. MMV’s success in research and access & product management comes from its extensive partnership network of over 300 pharmaceutical, academic and endemic-country partners in 50 countries.
MMV’s vision is a world in which innovative medicines will cure and protect the vulnerable and under-served populations at risk of malaria, and ultimately help to eradicate this terrible disease.
MMV Disclaimer:
This document contains certain forward-looking statements that may be identified by words such as ‘believes,‘ expects’, ‘anticipates’, ‘projects’, ‘intends’, ‘should’, ‘seeks’, ‘estimates’, ‘future’ or similar expressions, or by discussion of, among other things, vision, strategy, goals, plans, or intentions. It contains hypothetical future product target profiles, development timelines and approval/launch dates, positioning statements, claims and actions for which the relevant data may still have to be established. Stated or implied strategies and action items may be implemented only upon receipt of approvals including, but not limited to, local institutional review board approvals, local regulatory approvals, and following local laws and regulations. Thus, actual results, performances or events may differ from those expressed or implied by such statements.
They ask you not rely unduly on these statements. Such forward looking statements reflect the current views of Medicines for Malaria Venture (MMV) and its partner(s) regarding future events, and involve known and unknown risks and uncertainties MMV accepts no liability for the information presented here, nor for the consequences of any actions taken on the basis of this information. Furthermore, MMV accepts no liability for the decisions made by its pharmaceutical partner(s), the impact of any of their decisions, their earnings and their financial status.
CMT REPORT (Corruption, Money Laundering & Terrorism]
The Public Notice information has been collected from various sources including
but not limited to: The Courts, India Prisons Service, Interpol, etc.
1] INFORMATION ON
DESIGNATED PARTY
No exist designating subject or any of its beneficial owners,
controlling shareholders or senior officers as terrorist or terrorist organization
or whom notice had been received that all financial transactions involving
their assets have been blocked or convicted, found guilty or against whom a
judgement or order had been entered in a proceedings for violating
money-laundering, anti-corruption or bribery or international economic or
anti-terrorism sanction laws or whose assets were seized, blocked, frozen or
ordered forfeited for violation of money laundering or international
anti-terrorism laws.
2] Court Declaration :
No records exist to suggest that subject is
or was the subject of any formal or informal allegations, prosecutions or other
official proceeding for making any prohibited payments or other improper
payments to government officials for engaging in prohibited transactions or
with designated parties.
3] Asset Declaration :
No records exist to suggest that the property or assets of the subject
are derived from criminal conduct or a prohibited transaction.
4] Record on Financial
Crime :
Charges or conviction
registered against subject: None
5] Records on Violation of
Anti-Corruption Laws :
Charges or
investigation registered against subject: None
6] Records on Int’l
Anti-Money Laundering Laws/Standards :
Charges or
investigation registered against subject: None
7] Criminal Records
No
available information exist that suggest that subject or any of its principals
have been formally charged or convicted by a competent governmental authority
for any financial crime or under any formal investigation by a competent government
authority for any violation of anti-corruption laws or international anti-money
laundering laws or standard.
8] Affiliation with
Government :
No record
exists to suggest that any director or indirect owners, controlling
shareholders, director, officer or employee of the company is a government
official or a family member or close business associate of a Government
official.
9] Compensation Package :
Our market
survey revealed that the amount of compensation sought by the subject is fair
and reasonable and comparable to compensation paid to others for similar
services.
10] Press Report :
No press reports / filings exists on
the subject.
CORPORATE GOVERNANCE
MIRA INFORM as part of its Due Diligence do provide comments on Corporate
Governance to identify management and governance. These factors often have been
predictive and in some cases have created vulnerabilities to credit
deterioration.
Our Governance Assessment focuses principally on the interactions
between a company’s management, its Board of Directors, Shareholders and other
financial stakeholders.
CONTRAVENTION
Subject is not known to have contravened any existing local laws,
regulations or policies that prohibit, restrict or otherwise affect the terms
and conditions that could be included in the agreement with the subject.
FOREIGN EXCHANGE RATES
|
Currency |
Unit
|
Indian Rupees |
|
US Dollar |
1 |
Rs.61.74 |
|
UK Pound |
1 |
Rs.94.65 |
|
Euro |
1 |
Rs.70.79 |
INFORMATION DETAILS
|
Information Gathered
by : |
SUM |
|
|
|
|
Analysis Done by
: |
KRN |
|
|
|
|
Report Prepared
by : |
MTN |
SCORE & RATING EXPLANATIONS
|
SCORE FACTORS |
RANGE |
POINTS |
|
HISTORY |
1~10 |
9 |
|
PAID-UP CAPITAL |
1~10 |
8 |
|
OPERATING SCALE |
1~10 |
9 |
|
FINANCIAL CONDITION |
|
|
|
--BUSINESS SCALE |
1~10 |
9 |
|
--PROFITABILIRY |
1~10 |
9 |
|
--LIQUIDITY |
1~10 |
9 |
|
--LEVERAGE |
1~10 |
9 |
|
--RESERVES |
1~10 |
9 |
|
--CREDIT LINES |
1~10 |
9 |
|
--MARGINS |
-5~5 |
- |
|
DEMERIT POINTS |
|
|
|
--BANK CHARGES |
YES/NO |
YES |
|
--LITIGATION |
YES/NO |
YES |
|
--OTHER ADVERSE INFORMATION |
YES/NO |
NO |
|
MERIT POINTS |
|
|
|
--SOLE DISTRIBUTORSHIP |
YES/NO |
NO |
|
--EXPORT ACTIVITIES |
YES/NO |
NO |
|
--AFFILIATION |
YES/NO |
YES |
|
--LISTED |
YES/NO |
YES |
|
--OTHER MERIT FACTORS |
YES/NO |
YES |
|
DEFAULTER |
|
|
|
--RBI |
YES/NO |
NO |
|
--EPF |
YES/NO |
NO |
|
|
|
|
|
TOTAL |
|
80 |
This score serves as a reference to assess SC’s credit risk
and to set the amount of credit to be extended. It is calculated from a
composite of weighted scores obtained from each of the major sections of this
report. The assessed factors and their relative weights (as indicated through
%) are as follows:
Financial
condition (40%) Ownership
background (20%) Payment
record (10%)
Credit history
(10%) Market trend
(10%) Operational
size (10%)
RATING EXPLANATIONS
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
>86 |
Aaa |
Possesses an extremely sound financial base with the strongest
capability for timely payment of interest and principal sums |
Unlimited |
|
71-85 |
Aa |
Possesses adequate working capital. No caution needed for credit transaction.
It has above average (strong) capability for payment of interest and
principal sums |
Large |
|
56-70 |
A |
Financial & operational base are regarded healthy. General
unfavourable factors will not cause fatal effect. Satisfactory capability for
payment of interest and principal sums |
Fairly Large |
|
41-55 |
Ba |
Overall operation is considered normal. Capable to meet normal
commitments. |
Satisfactory |
|
26-40 |
B |
Capability to overcome financial difficulties seems comparatively below
average. |
Small |
|
11-25 |
Ca |
Adverse factors are apparent. Repayment of interest and principal sums
in default or expected to be in default upon maturity |
Limited with
full security |
|
<10 |
C |
Absolute credit risk exists. Caution needed to be exercised |
Credit not
recommended |
|
-- |
NB |
New Business |
-- |
This report is issued at your request without any
risk and responsibility on the part of MIRA INFORM PRIVATE LIMITED (MIPL) or
its officials.