MIRA INFORM REPORT
|
Report No. : |
307208 |
|
Report Date : |
11.02.2015 |
IDENTIFICATION DETAILS
|
Name : |
LUPIN LIMITED |
|
|
|
|
Registered
Office : |
159, C.S.T. Road, Kalina, Santacruz (East), Mumbai – 400098,
Maharashtra |
|
|
|
|
Country : |
India |
|
|
|
|
Financials (as
on) : |
31.03.2014 |
|
|
|
|
Date of
Incorporation : |
01.03.1983 |
|
|
|
|
Com. Reg. No.: |
11-029442 |
|
|
|
|
Capital
Investment / Paid-up Capital : |
Rs. 896.800 Millions |
|
|
|
|
CIN No.: [Company Identification
No.] |
L24100MH1983PLC029442 |
|
|
|
|
IEC No.: |
Not Available |
|
|
|
|
TAN No.: [Tax Deduction &
Collection Account No.] |
PNEL03519F MUML04496C |
|
|
|
|
PAN No.: [Permanent Account No.] |
AAACL1069K |
|
|
|
|
Legal Form : |
A Public Limited Liability company. The company’s Shares are Listed on
the Stock Exchanges. |
|
|
|
|
Line of Business
: |
Manufacturers of Bulk Drugs and
Formulations. |
|
|
|
|
No. of Employees
: |
4484 (Approximately) |
RATING & COMMENTS
|
MIRA’s Rating : |
Aa (72) |
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
71-85 |
Aa |
Possesses adequate working capital. No caution needed for credit
transaction. It has above average (strong) capability for payment of interest
and principal sums |
Large |
|
Status : |
Good |
|
|
|
|
Payment Behaviour : |
Regular |
|
|
|
|
Litigation : |
Exist |
|
|
|
|
Comments : |
Subject is a well-established company having fine track record. The rating reflects company’s healthy financial risk profile marked by
adequate liquidity position and decent profitability levels of the company. Trade relations are reported as fair. Business is active. Payments are
reported to be regular and as per commitments. The company can be considered good for normal business dealings at
usual trade terms and conditions. |
NOTES :
Any query related to this report can be made
on e-mail : infodept@mirainform.com
while quoting report number, name and date.
ECGC Country Risk Classification List – September 30, 2014
|
Country Name |
Previous Rating (30.06.2014) |
Current Rating (30.09.2014) |
|
India |
A1 |
A1 |
|
Risk Category |
ECGC
Classification |
|
Insignificant |
A1 |
|
Low |
A2 |
|
Moderate |
B1 |
|
High |
B2 |
|
Very High |
C1 |
|
Restricted |
C2 |
|
Off-credit |
D |
EXTERNAL AGENCY RATING
|
Rating Agency Name |
ICRA |
|
Rating |
Long Term Fund Facilities: AA+ |
|
Rating Explanation |
High degree of safety and very low credit
risk |
|
Date |
September, 2013 |
|
Rating Agency Name |
ICRA |
|
Rating |
Short Term Non Fund Facilities: A1+ |
|
Rating Explanation |
Very strong degree of safety and lowest
credit risk |
|
Date |
September, 2013 |
RBI DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available RBI Defaulters’ list.
EPF (Employee Provident Fund) DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available EPF (Employee Provident Fund) Defaulters’ list as of
31-03-2014.
INFORMATION DECLINED BY
|
Name : |
Mr. C.V. Venkatraman |
|
Designation : |
-- |
|
Contact No.: |
91-22-66402323 |
LOCATIONS
|
Registered Office : |
159, C.S.T. Road, Kalina, Santacruz (East), Mumbai – 400098,
Maharashtra, India |
|
Tel. No.: |
91-22-26931001 / 26526391 / 26528311 /
56402222 / 66402323 |
|
Fax No.: |
91-22-26540484 / 26114008 / 56402299 /
26528806 / 66402051 |
|
E-Mail : |
rajvardhansatam@lupinpharma.com |
|
Website : |
|
|
|
|
|
Corporate Office : |
B/4 |
|
Tel. No.: |
91-22-66402222 |
|
Fax No.: |
91-22-66402130 |
|
Website : |
|
|
|
|
|
Factory 1 : |
T-142 MIDC Industrial Estate, Tarapur Industrial Area, Boisar,
District Thane - 401506, Maharashtra, India |
|
|
|
|
Factory 2 : |
Plot No. 2 and M-1, Special Economic Zone, Misc. Zone, Apparel Park,
Pithampur, District Dhar - 454775, Madhya Pradesh, India |
|
|
|
|
Factory 3 : |
Plot No. 124 and 2201/2202, GIDC Industrial Estate, Ankleshwar –
393002, Gujarat, India |
|
|
|
|
Factory 4 : |
A28/1, MIDC Area, Chikalthana, Aurangabad – 431001, Maharashtra, India |
|
|
|
|
Factory 5 : |
B-15 Phase I-A Verna Industiral Area, Verma Salcette, Goa – 403722, |
|
|
|
|
Factory 6 : |
Export promotion Industrial Park, SIDCO Industrial Complex, Kartholi,
Bari Brahmana, Jammu – 181133, Jammu Kashmir, India |
|
|
|
|
Factory 7 : |
Gate No. 1156, Village Ghotawade, Taluka – Mulshi, District - Pune, |
|
|
|
|
Factory 8 : |
Block 21, Dabhasa Padra, Taluka - Vadodara – 391440, Gujarat, India |
|
|
|
|
Factory 9 : |
198-202, New Industrial Area II, Mandideep, District Raisen - 462046,
Madhya Pradesh |
|
|
|
|
Factory 10 : |
Kyowa Pharmaceutical Industry Company Limited, 11-1 |
|
|
|
|
Factory 11: |
Plot 6A, Sector-17, Special Economic Zone, Mihan Notified Area, Nagpur - 441108, Maharashtra, India |
|
|
|
|
Factory 12: |
I’rom Pharmaceutical
Company Limited Post code 243-0014 4-18-29, Asahi-cho, Atsugi city, Kanagawa prefecture Japan |
DIRECTORS
As on 31.03.2014
|
Name : |
Dr. Desh Bandhu Gupta |
|
Designation : |
Chairman |
|
|
|
|
Name : |
Dr. Kamal K. Sharma |
|
Designation : |
Vice Chairman |
|
|
|
|
Name : |
Mrs. M. D. Gupta |
|
Designation : |
Executive Director |
|
|
|
|
Name : |
Ms. Vinita Gupta |
|
Designation : |
Director and Chief Executive Officer |
|
|
|
|
Name : |
Mr. Nilesh Gupta |
|
Designation : |
Managing Director |
|
|
|
|
Name : |
Dr. Vijay Kelkar |
|
Designation : |
Director |
|
|
|
|
Name : |
Mr. Richard Zahn |
|
Designation : |
Director |
|
|
|
|
Name : |
Mr. R. A. Shah |
|
Designation : |
Director |
|
|
|
|
Name : |
Dr. K. U. Mada |
|
Designation : |
Director |
|
|
|
|
Name : |
Mr. Dileep C. Choksi, |
|
Designation : |
Additional Director |
|
Date of Appointment : |
23.10.2012 |
KEY EXECUTIVES
|
Name : |
Mr. R. V. Satam |
|
Designation : |
Company Secretary and Compliance Officer |
|
|
|
|
AUDIT COMMITTEE |
· Dr. K. U. Mada, Chairman · Dr. Kamal K. Sharma · Mr. Dileep C. Choksi |
|
|
|
|
INVESTORS’
GRIEVANCES COMMITTEE |
· Dr. Vijay Kelkar, Chairman · Dr. K. U. Mada |
|
|
|
|
REMUNERATION
COMMITTEE |
· Dr. K. U. Mada (Chairman) · Mr. R. A. Shah |
|
|
|
|
SENIOR MANAGEMENT
TEAM |
|
|
Name : |
Dr. Desh Bandhu Gupta |
|
Designation : |
Chairman |
|
|
|
|
Name : |
Dr. Kamal K. Sharma |
|
Designation : |
Vice Chairman |
|
|
|
|
Name : |
Ms. Vinita Gupta |
|
Designation : |
Chief Executive Officer |
|
|
|
|
Name : |
Mr. Nilesh Gupta |
|
Designation : |
Managing Director |
|
|
|
|
Name : |
Mr. Shakti Chakraborty |
|
Designation : |
Group President - India Region Formulations |
|
|
|
|
Name : |
Mr. Vinod Dhawan |
|
Designation : |
Group President - AAMLA and Business Development |
|
|
|
|
Name : |
Dr. Rajender Kamboj |
|
Designation : |
President - Novel Drug Discovery and Development |
|
|
|
|
Name : |
Mr. Ramesh Swaminathan |
|
Designation : |
Chief Financial Officer |
|
|
|
|
Name : |
Mr. Naresh Gupta |
|
Designation : |
President - API and Global TB |
|
|
|
|
Name : |
Mr. Divakar Kaza |
|
Designation : |
President - Human Resources |
|
|
|
|
Name : |
Mr. Alok Ghosh |
|
Designation : |
President - Technical Operations |
|
|
|
|
Name : |
Dr. Cyrus Karkaria |
|
Designation : |
President – Biotechnology |
|
|
|
|
Name : |
Mr. Paul McGarty |
|
Designation : |
President - Lupin Pharmaceuticals Inc., USA |
|
|
|
|
Name : |
Dr. Sofia Mumtaz |
|
Designation : |
Head - Pipeline Management and Legal |
|
|
|
|
Name : |
Mr. Sunil Makharia |
|
Designation : |
President - Finance |
|
|
|
|
Name : |
Mr. Debabrata Chakravorty |
|
Designation : |
President - Global Sourcing |
|
|
|
|
Name : |
Dr. Maurice Chagnaud |
|
Designation : |
President - Europe and Head of Inhalation Strategy |
|
|
|
|
Name : |
Ms. Theresa Stevens |
|
Designation : |
Chief Corporate Development Officer |
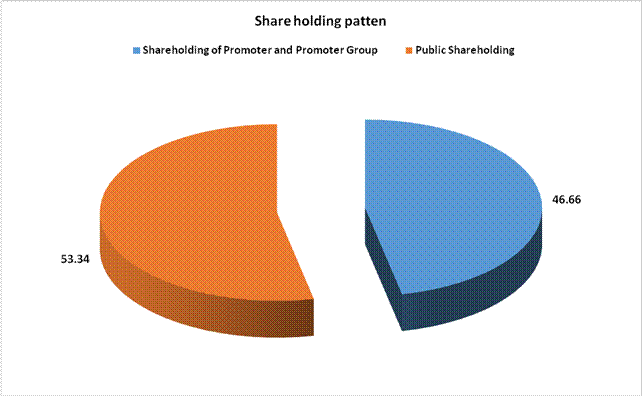
MAJOR SHAREHOLDERS / SHAREHOLDING PATTERN
As on 31.12.2014
|
Category
of Shareholder |
Total No.
of Shares |
Total
Shareholding as a % of Total No. of Shares |
|
As a % of (A+B) |
||
|
(A) Shareholding of Promoter
and Promoter Group |
||
|
|
|
|
|
|
6957870 |
1.55 |
|
|
201840570 |
44.93 |
|
|
208798440 |
46.48 |
|
|
|
|
|
|
803800 |
0.18 |
|
|
803800 |
0.18 |
|
Total shareholding of Promoter
and Promoter Group (A) |
209602240 |
46.66 |
|
(B) Public Shareholding |
||
|
|
|
|
|
|
23867391 |
5.31 |
|
|
490021 |
0.11 |
|
|
23861192 |
5.31 |
|
|
142732262 |
31.77 |
|
|
7079 |
0.00 |
|
|
7079 |
0.00 |
|
|
190957945 |
42.51 |
|
|
|
|
|
|
9146565 |
2.04 |
|
|
|
|
|
|
23277167 |
5.18 |
|
|
13652163 |
3.04 |
|
|
2575743 |
0.57 |
|
|
153562 |
0.03 |
|
|
732586 |
0.16 |
|
|
1644648 |
0.37 |
|
|
44947 |
0.01 |
|
|
48651638 |
10.83 |
|
Total Public shareholding (B) |
239609583 |
53.34 |
|
Total (A)+(B) |
449211823 |
100.00 |
|
(C) Shares held by Custodians and
against which Depository Receipts have been issued |
|
|
|
|
0 |
0.00 |
|
|
0 |
0.00 |
|
|
0 |
0.00 |
|
Total (A)+(B)+(C) |
449211823 |
100.00 |
BUSINESS DETAILS
|
Line of Business : |
Manufacturers of Bulk Drugs and
Formulations. |
|
|
|
|
Products : |
Not Divulged |
|
|
|
|
Brand Names : |
Not Divulged |
|
|
|
|
Agencies Held : |
Not Divulged |
|
|
|
|
Exporter : |
Not Divulged |
|
|
|
|
Importer : |
Not Divulged |
|
|
|
|
Terms : |
Not Divulged |
GENERAL INFORMATION
|
Suppliers : |
|
|||||||||||||||
|
|
|
|||||||||||||||
|
Customers : |
|
|||||||||||||||
|
|
|
|||||||||||||||
|
No. of Employees : |
4484 (Approximately) |
|||||||||||||||
|
|
|
|||||||||||||||
|
Bankers : |
· Central Bank of India · Bank of Baroda · State Bank of India · Citibank N.A. · The Hong Kong and Shanghai · Banking Corporation Limited · Standard Chartered Bank · ICICI Bank Limited · Kotak Mahindra Bank Limited ·
JP Morgan Chase Bank, N.A. |
|||||||||||||||
|
|
|
|||||||||||||||
|
Facilities : |
|
|||||||||||||||
|
|
|
|
Auditors : |
|
|
Name : |
Deloitte Haskins and Sells Chartered Accountants |
|
Address : |
Indiabulls Finance centre Tower 3, 27th – 32nd Floor Senapati Bapat Marg, Elphinstone Road (West), Mumbai – 400013, Maharashtra, India |
|
Tel No.: |
91-22-61854000 |
|
Fax No.: |
91-22-61854501/4601 |
|
|
|
|
Internal Auditors: |
|
|
|
Ernst and Young LLP Chartered Accountants |
|
|
|
|
Subsidiaries: |
·
Lupin Pharmaceuticals, Inc., USA ·
Kyowa Pharmaceutical Industry Company, Limited,
Japan ·
Lupin Australia Pty Limited, Australia ·
Lupin Holdings B.V., Netherlands ·
Pharma Dynamics (Proprietary) Limited, South
Africa ·
Hormosan Pharma GmbH, Germany ·
Multicare Pharmaceuticals Philippines Inc.,
Philippines ·
Lupin Atlantis Holdings SA, Switzerland ·
Lupin (Europe) Limited, UK ·
Amel Touhoku, Japan (up to 28th February 2013) ·
Lupin Pharma Canada Limited, Canada ·
Lupin Mexico SA de CV, Mexico ·
Generic Health Pty Limited, Australia ·
Bellwether Pharma Pty Limited, Australia ·
Generic Health Inc., USA (from 27th September
2010) (upto 4th October 2011) ·
Max Pharma Pty Limited, Australia ·
Lupin Philippines Inc., Philippines ·
Lupin Healthcare Limited, India ·
Generic Health SDN. BHD., Malaysia (from 18th May
2011) ·
I’rom Pharmaceutical Company, Limited, Japan
(from 30th November 2011) ·
Lupin Middle East FZ-LLC, UAE (from 13th June
2012) · Lupin GmbH, Switzerland (from 15 August 2013) · Lupin Inc., USA (from 27 June 2013) · Farma World Importacao e Exportacao De Medicamentos LTDA - EPP, Brazil (from 17 December 2013) ·
Nanomi B.V., Netherlands (from 30 January 2014) |
|
|
|
|
Relatives of Key Management Personnel and Entities in which the Key Management
Personnel have control or significant influence |
·
Enzal Chemicals (India) Limited (upto 31st March
2012) ·
Lupin Human Welfare and Research Foundation ·
Lupin International Private Limited ·
Lupin Investments Private Limited ·
Lupin Marketing Private Limited ·
Matashree Gomati Devi Jana Seva Nidhi ·
Novamed Investments Private Limited (formerly
Novamed Pharmaceuticals Private Limited) ·
Polynova Industries Limited ·
Rahas Investments Private Limited ·
Synchem Investments Private Limited (formerly
Synchem Chemicals (I) Private Limited) ·
Visiomed (India) Private Limited ·
Zyma Laboratories Limited |
CAPITAL STRUCTURE
As on 30.07.2014
Authorised Capital : Rs. 1000.000 Millions
Issued, Subscribed & Paid-up Capital : Rs. 898.664
Millions
As on 31.03.2014
Authorised Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
500000000 |
Equity Shares |
Rs.2/- each |
Rs. 1000.000 Millions |
|
|
|
|
|
Issued, Subscribed & Paid-up Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
448375804 |
Equity Shares |
Rs.2/- each |
Rs. 896.800
Millions |
|
|
|
|
|
Reconciliation of the number of shares and amount outstanding at the
beginning and at the end of the reporting period
|
Particulars |
As at 31 March 2014 |
|
|
No. of Shares |
Amount (Rs in Millions) |
|
|
Equity Shares outstanding at the beginning of the year |
447529493 |
895.100 |
|
Equity Shares issued during the year in the form of ESOPs |
846311 |
1.700 |
|
Equity Shares outstanding at the end of the year |
448375804 |
896.800 |
Rights attached to Equity Share
The Company has only one class of equity shares with voting rights having a par value of Rs. 2 per share. The Company declares and pays dividends in Indian Rupees. The dividend proposed by the Board of Directors is subject to the approval of the shareholders at the ensuing Annual General Meeting, except in case of interim dividend.
During the year ended 31 March 2014, the Company declared an interim dividend of Rs. 3 per equity share. A final dividend of Rs. 3 per equity share has been recommended by the Board of Directors at its meeting held on 7 May 2014. On approval by the shareholders of the final dividend of Rs. 3 per equity share, the total dividend for the year would be Rs. 6 (previous year Rs. 4) per equity share.
In the event of liquidation of the Company, the shareholders of equity shares will be entitled to receive remaining assets of the Company after distribution of all preferential amounts. The distribution will be in proportion to the number of equity shares held by the shareholders.
Details of shares
held by each shareholder holding more than 5% equity shares
|
Name of Shareholder |
As at 31 March 2014 |
|
|
No. of Shares |
% of Holding |
|
|
Zyma Laboratories Limited |
54960490 |
12.26 |
|
Rahas Investments Private Limited |
45699510 |
10.19 |
|
Visiomed Investments Private Limited [formerly Visiomed (India) Private Limited] |
43514660 |
9.70 |
|
Lupin Holdings Pvt. Limited [formerly Lupin Marketing Private Limited] |
40401000 |
9.01 |
Shares reserved for
issuance under Stock Option Plans of the Company
|
Particulars |
As at 31 March 2014 |
|
No. of Shares |
|
|
Lupin Employees Stock Options Plan 2003 |
687134 |
|
Lupin Employees Stock Options Plan 2005 |
532479 |
|
Lupin Employees Stock Options Plan 2011 |
3297482 |
|
Lupin Subsidiary Companies Employees Stock Options Plan 2005 |
371103 |
|
Lupin Subsidiary Companies Employees Stock Options Plan 2011 |
895113 |
Aggregate number of shares issued during last five years pursuant
to Stock Option Plans of the Company
|
Particulars |
Aggregate No. of Shares |
|
Equity Shares: |
|
|
Issued under various Stock Option Plans of the Company |
5194344 |
No shares have been allotted without payment being received in cash or by way of bonus shares during the period of five years immediately preceding the Balance Sheet date.
FINANCIAL DATA
[all figures are
in Rupees Millions]
ABRIDGED BALANCE
SHEET
|
SOURCES
OF FUNDS |
31.03.2014 |
31.03.2013 |
31.03.2012 |
|
I.
EQUITY
AND LIABILITIES |
|
|
|
|
(1)Shareholders' Funds |
|
|
|
|
(a) Share Capital |
896.800 |
895.100 |
893.300 |
|
(b) Reserves & Surplus |
68893.600 |
47572.000 |
36450.800 |
|
(c) Money received
against share warrants |
0.000 |
0.000 |
0.000 |
|
|
|
|
|
|
(2) Share
Application money pending allotment |
0.000 |
0.000 |
0.000 |
|
Total Shareholders’ Funds (1) + (2) |
69790.400 |
48467.100 |
37344.100 |
|
|
|
|
|
|
(3) Non-Current
Liabilities |
|
|
|
|
(a) long-term
borrowings |
242.400 |
292.000 |
1349.200 |
|
(b) Deferred tax liabilities (Net) |
2479.300 |
2329.200 |
1905.000 |
|
(c) Other long term
liabilities |
95.100 |
114.000 |
376.100 |
|
(d) long-term
provisions |
769.200 |
683.500 |
346.100 |
|
Total Non-current
Liabilities (3) |
3586.000 |
3418.700 |
3976.400 |
|
|
|
|
|
|
(4) Current Liabilities |
|
|
|
|
(a) Short
term borrowings |
1151.600 |
5260.900 |
8577.300 |
|
(b) Trade
payables |
9838.500 |
8694.200 |
6984.200 |
|
(c) Other
current liabilities |
1265.100 |
2189.300 |
2502.800 |
|
(d) Short-term
provisions |
2358.000 |
2427.100 |
2202.300 |
|
Total Current
Liabilities (4) |
14613.200 |
18571.500 |
20266.600 |
|
|
|
|
|
|
TOTAL |
87989.600 |
70457.300 |
61587.100 |
|
|
|
|
|
|
II.
ASSETS |
|
|
|
|
(1) Non-current assets |
|
|
|
|
(a) Fixed
Assets |
|
|
|
|
(i)
Tangible assets |
21709.100 |
20006.300 |
16972.300 |
|
(ii)
Intangible Assets |
85.500 |
130.200 |
102.500 |
|
(iii)
Capital work-in-progress |
2670.500 |
2401.200 |
3573.300 |
|
(iv) Intangible assets under development |
0.000 |
0.000 |
0.000 |
|
(b) Non-current
Investments |
9890.500 |
6880.400 |
6872.900 |
|
(c) Deferred tax
assets (net) |
0.000 |
0.000 |
0.000 |
|
(d) Long-term Loan and Advances |
3197.000 |
3620.300 |
3835.600 |
|
(e) Other
Non-current assets |
0.000 |
0.000 |
0.000 |
|
Total Non-Current
Assets |
37552.600 |
33038.400 |
31356.600 |
|
|
|
|
|
|
(2) Current assets |
|
|
|
|
(a)
Current investments |
1746.100 |
0.000 |
0.000 |
|
(b)
Inventories |
13722.400 |
13308.300 |
11235.600 |
|
(c) Trade
receivables |
28599.200 |
18742.700 |
14908.000 |
|
(d) Cash
and cash equivalents |
1462.800 |
201.200 |
192.000 |
|
(e)
Short-term loans and advances |
2594.400 |
2840.100 |
2684.400 |
|
(f) Other
current assets |
2312.100 |
2326.600 |
1210.500 |
|
Total
Current Assets |
50437.000 |
37418.900 |
30230.500 |
|
|
|
|
|
|
TOTAL |
87989.600 |
70457.300 |
61587.100 |
PROFIT & LOSS
ACCOUNT
|
|
PARTICULARS |
31.03.2014 |
31.03.2013 |
31.03.2012 |
|
|
SALES |
|
|
|
|
|
Income |
89393.800 |
71225.100 |
53848.300 |
|
|
Other Income |
4153.800 |
233.100 |
34.900 |
|
|
TOTAL (A) |
93547.600 |
71458.200 |
53883.200 |
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
Cost of Raw and Packing Materials Consumed |
21320.000 |
19272.100 |
15921.700 |
|
|
Purchases of
Stock-in-Trade |
8291.900 |
7760.300 |
5992.700 |
|
|
Changes in inventories of
finished goods, work-in-progress and Stock-in-Trade |
(762.100) |
(1824.400) |
(1325.300) |
|
|
Employees benefits
expense |
8443.200 |
7264.400 |
5812.200 |
|
|
Other expenses |
22977.500 |
19913.500 |
15828.400 |
|
|
TOTAL (B) |
60270.500 |
52385.900 |
42229.700 |
|
|
|
|
|
|
|
Less |
PROFIT/ (LOSS) BEFORE INTEREST, TAX, DEPRECIATION AND
AMORTISATION (C) |
33277.100 |
19072.300 |
11653.500 |
|
|
|
|
|
|
|
Less |
FINANCIAL EXPENSES (D) |
209.900 |
332.800 |
286.800 |
|
|
|
|
|
|
|
|
PROFIT / (LOSS) BEFORE TAX,
DEPRECIATION AND AMORTISATION (C-D) (E) |
33067.200 |
18739.500 |
11366.700 |
|
|
|
|
|
|
|
Less/ Add |
DEPRECIATION/
AMORTISATION (F) |
1676.300 |
1501.400 |
1319.600 |
|
|
|
|
|
|
|
|
PROFIT/ (LOSS) BEFORE TAX (E-F) (G) |
31390.900 |
17238.100 |
10047.100 |
|
|
|
|
|
|
|
Less |
TAX (I) |
8148.700 |
4633.800 |
2003.400 |
|
|
|
|
|
|
|
|
PROFIT/ (LOSS) AFTER TAX (G-I) (J) |
23242.200 |
12604.300 |
8043.700 |
|
|
|
|
|
|
|
Add |
PREVIOUS YEARS’ BALANCE
BROUGHT FORWARD (K) |
28539.200 |
19530.200 |
14647.900 |
|
|
|
|
|
|
|
Less |
APPROPRIATIONS |
|
|
|
|
|
Transfer
to General Reserve |
2500.000 |
1500.000 |
1500.000 |
|
|
Dividend |
1345.100 |
1790.100 |
1429.200 |
|
|
Interim Dividend on
Equity Shares |
1345.000 |
0.000 |
0.000 |
|
|
Dividend
on Equity Shares issued after the previous year end |
1.100 |
0.900 |
0.300 |
|
|
Reversal of Corporate Tax
on Final Dividend for previous years |
(304.200) |
0.000 |
0.000 |
|
|
Corporate
Tax on Dividend |
247.800 |
304.300 |
231.900 |
|
|
BALANCE CARRIED TO THE
B/S |
46646.600 |
28539.200 |
19530.200 |
|
|
|
|
|
|
|
|
EARNINGS IN FOREIGN
CURRENCY |
|
|
|
|
|
FOB value of exports |
57177.000 |
43156.500 |
30329.200 |
|
|
Deemed exports |
0.000 |
0.000 |
372.300 |
|
|
Sale-Research Services |
2302.800 |
1066.300 |
1416.300 |
|
|
Reimbursement of freight and
insurance on Exports |
292.800 |
281.200 |
237.400 |
|
|
Compensation & Settlement
Income |
200.300 |
186.000 |
77.200 |
|
|
Dividend Income |
3020.200 |
0.000 |
0.000 |
|
|
Others |
83.000 |
59.200 |
11.600 |
|
|
TOTAL EARNINGS |
63076.100 |
44749.200 |
32444.000 |
|
|
|
|
|
|
|
|
IMPORTS |
|
|
|
|
|
Raw
Materials |
7206.300 |
6615.600 |
5905.300 |
|
|
Packing
Materials |
405.200 |
284.700 |
327.800 |
|
|
Capital
Goods |
992.900 |
728.400 |
788.200 |
|
|
Purchase
of Traded Goods |
488.400 |
388.300 |
280.200 |
|
|
Consumable,
Stores and Spares |
560.700 |
374.000 |
306.800 |
|
|
TOTAL IMPORTS |
9653.500 |
8391.000 |
7608.300 |
|
|
|
|
|
|
|
Earnings
Per Share (Rs.) |
||||
|
Basic |
51.88 |
28.19 |
18.02 |
|
|
Diluted |
51.62 |
28.07 |
17.94 |
KEY RATIOS
|
PARTICULARS |
|
31.03.2014 |
31.03.2013 |
31.03.2012 |
|
Net Profit Margin (PAT/Sales) |
(%) |
26.00 |
17.70 |
14.94 |
|
|
|
|
|
|
|
Operating Profit Margin (PBITD/Sales) |
(%) |
37.23 |
26.78 |
21.64 |
|
|
|
|
|
|
|
Return on Total Assets (PBT/Total Assets} |
(%) |
41.62 |
28.18 |
19.65 |
|
|
|
|
|
|
|
Return on Investment (ROI) (PBT/Networth) |
|
0.45 |
0.36 |
0.27 |
|
|
|
|
|
|
|
Debt Equity Ratio (Total Debt/Networth) |
|
0.02 |
0.11 |
0.27 |
|
|
|
|
|
|
|
Current Ratio (Current Asset/Current Liability) |
|
3.45 |
2.01 |
1.49 |
FINANCIAL ANALYSIS
[all figures are
in Rupees Millions]
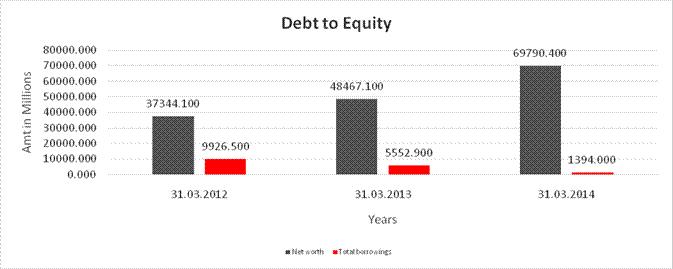
DEBT EQUITY RATIO
|
Particular |
31.03.2012 |
31.03.2013 |
31.03.2014 |
|
|
(Rs. In Millions) |
(Rs. In Millions) |
(Rs. In Millions) |
|
Share Capital |
893.300 |
895.100 |
896.800 |
|
Reserves & Surplus |
36450.800 |
47572.000 |
68893.600 |
|
Net worth |
37344.100 |
48467.100 |
69790.400 |
|
|
|
|
|
|
long-term borrowings |
1349.200 |
292.000 |
242.400 |
|
Short term borrowings |
8577.300 |
5260.900 |
1151.600 |
|
Total borrowings |
9926.500 |
5552.900 |
1394.000 |
|
Debt/Equity ratio |
0.266 |
0.115 |
0.020 |
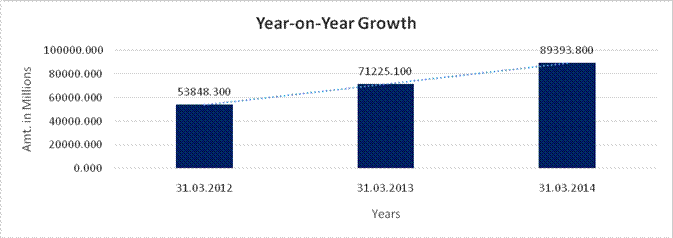
YEAR-ON-YEAR GROWTH
|
Year on Year Growth |
31.03.2012 |
31.03.2013 |
31.03.2014 |
|
|
(Rs. In Millions) |
(Rs. In Millions) |
(Rs. In Millions) |
|
Sales |
53848.300 |
71225.100 |
89393.800 |
|
|
|
32.270 |
25.509 |
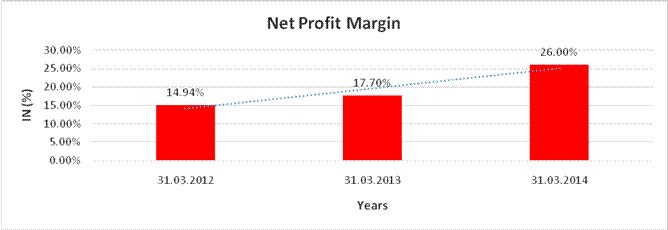
NET PROFIT MARGIN
|
Net Profit Margin |
31.03.2012 |
31.03.2013 |
31.03.2014 |
|
|
(Rs. In Millions) |
(Rs. In Millions) |
(Rs. In Millions) |
|
Sales |
53848.300 |
71225.100 |
89393.800 |
|
Profit |
8043.700 |
12604.300 |
23242.200 |
|
|
14.94% |
17.70% |
26.00% |
LOCAL AGENCY FURTHER INFORMATION
|
Sr. No. |
Check List by Info
Agents |
Available in Report
(Yes / No) |
|
1] |
Year of Establishment |
Yes |
|
2] |
Locality of the firm |
Yes |
|
3] |
Constitutions of the firm |
Yes |
|
4] |
Premises details |
No |
|
5] |
Type of Business |
Yes |
|
6] |
Line of Business |
Yes |
|
7] |
Promoter's background |
No |
|
8] |
No. of employees |
Yes |
|
9] |
Name of person contacted |
Yes |
|
10] |
Designation of contact person |
No |
|
11] |
Turnover of firm for last three years |
Yes |
|
12] |
Profitability for last three years |
Yes |
|
13] |
Reasons for variation <> 20% |
-- |
|
14] |
Estimation for coming financial year |
No |
|
15] |
Capital in the business |
Yes |
|
16] |
Details of sister concerns |
Yes |
|
17] |
Major suppliers |
No |
|
18] |
Major customers |
No |
|
19] |
Payments terms |
No |
|
20] |
Export / Import details (if applicable) |
No |
|
21] |
Market information |
-- |
|
22] |
Litigations that the firm / promoter involved in |
Yes |
|
23] |
Banking Details |
Yes |
|
24] |
Banking facility details |
Yes |
|
25] |
Conduct of the banking account |
-- |
|
26] |
Buyer visit details |
-- |
|
27] |
Financials, if provided |
Yes |
|
28] |
Incorporation details, if applicable |
Yes |
|
29] |
Last accounts filed at ROC |
Yes |
|
30] |
Major Shareholders, if available |
Yes |
|
31] |
Date of Birth of Proprietor/Partner/Director, if available |
No |
|
32] |
PAN of Proprietor/Partner/Director, if available |
No |
|
33] |
Voter ID No of Proprietor/Partner/Director, if available |
No |
|
34] |
External Agency Rating, if available |
Yes |
LITIGATION DETAILS:
HIGH
COURT OF BOMBAY
|
Bench:- Bombay |
|||||||
|
Presentation Date:- 20/01/2014 |
|||||||
|
Lodging No:- |
ARBPL/175/2014 |
Filing Date:- |
20/01/2014 |
Reg. No.:- |
ARBP/1066/2014 |
Reg. Date:- |
11/08/2014 |
|
Petitioner:- |
M/S ANKUR CONSTRUCTION COMPANY |
Respondent:- |
LUPIN LIMITED, A PUBLIC LIMITED COMPANY |
||||
|
Petn. Adv.: |
VIJAY KUMAR AGARWAL (I1475) |
Resp. Adv:- |
PS LEGAL (RESPONDENT) (I5209) |
||||
|
District:- |
OUTSIDE MAHARASHTRA |
||||||
|
Bench:- |
SINGLE |
||||||
|
Status:- |
Pre-Admission |
Category:- |
ARBITRATION ACT. |
||||
|
Last Date:- |
12/01/2015 |
Stage:- |
ARBP FOR ADMISSION U/S 34 FRESH |
||||
|
Last Coram:- |
PROVISIONAL BOARD |
|
|
||||
|
Act:- |
Arbitration and Conciliation Act 1996 |
Under section:- |
34 |
||||
INDEX OF CHARGES
|
S. No. |
Charge ID |
Date of Charge Creation/Modification |
Charge amount secured |
Charge Holder |
Address |
Service Request Number (SRN) |
|
1 |
90226754 |
10/01/2013 * |
15,000,000,000.00 |
Central Bank of India |
Andheri (East) Branch, Vastu Darshan, 'B' Wing, 1st Floor, Azad Road,
Andheri (East), Mumbai, Maharashtra - 400069, INDIA |
B65839037 |
* Date of charge modification
UNSECURED LOAN
|
PARTICULARS |
31.03.2014 (Rs.
In Millions) |
31.03.2013 (Rs.
In Millions) |
|
LONG TERM
BORROWINGS |
|
|
|
Term Loans - from
other parties |
|
|
|
Deferred Sales Tax Loan from Government of Maharashtra |
46.200 |
54.600 |
|
Term Loans from Council for Scientific and Industrial Research (CSIR) |
154.700 |
185.600 |
|
Term Loans from Department of Science and Technology (DST) |
41.500 |
51.800 |
|
|
|
|
|
SHORT TERM
BORROWINGS |
|
|
|
Loans from Banks |
651.600 |
1147.900 |
|
Total |
894.000 |
1439.900 |
|
Note:
· Deferred Sales Tax Loan is interest free and payable in 5 equal annual installments after expiry of initial 10 years moratorium period from each such year of deferral period from 1998-99 to 2009-10. · Term Loans from CSIR carry interest of 3% p.a. and is payable in 6 annual installments of Rs. 30.900 millions each along with interest. · Term Loans from DST carry interest of 3% p.a. and is payable in 5 annual installments of Rs. 10.400 millions each along with interest. · The Company has not defaulted on repayment of loans and interest during the year. SHORT TERM BORROWINGS · Unsecured loans comprise of Short-Term Loans, Packing Credit, Post Shipment Credit, Bills Discounted and Overseas Import Credit. It includes foreign currency loans of Rs. 608.100 million (previous year Rs. 1085.700 million). · Foreign Currency loans carry interest rate in the range of 0.28% to 0.54% p.a. and those in Indian Rupees carries interest rate in the range of 10.00% to 12.75% p.a. ·
The Company has not defaulted on repayment of
loans and interest during the year. |
||
OVERVIEW:
The Company was incorporated in 1983 as Lupin Chemicals Private Limited. Lupin Laboratories Limited which was incorporated in 1972 was amalgamated with the Company w.e.f. 01.04.2000, pursuant to an Order passed by the Mumbai High Court. The Company is an innovation led Transnational Pharmaceutical Company producing, developing and marketing a wide range of branded and generic formulations and APIs. The Company along with its subsidiaries has manufacturing locations spread across India and Japan with trading and other incidental and related activities extending to the world markets.
PERFORMANCE REVIEW
During the year, the Company’s turnover crossed Rs. 111 billion, with Consolidated Gross Sales clocking Rs. 111671.200 million as against Rs. 95235.300 million of the previous year, higher by 17%. International markets accounted for 74% of sales. Profit before interest, depreciation and tax increased by 36% at Rs. 31192.700 million as against Rs. 22977.400 million in the previous year. Profit before tax was Rs. 28316.500 million, higher by 47% over the previous year. After providing for taxes and minority interest, net profit was Rs. 18363.700 million, higher by 40% over the previous year. Earnings per share was Rs. 40.99.
ACQUISITIONS
In its strategy to pursue inorganic growth for further accelerating its progress and expanding its presence in select geographies, your Company made the following acquisitions through Lupin Atlantis Holdings SA, Switzerland, wholly-owned subsidiary: -
a) the entire shareholding of Laboratorios Grin, S.A. DE C.V., Mexico, (‘Lab Grin’). Lab Grin, a 60 year old company specializing in Opthalmics. It is ranked 4th in the Opthalmics segment in the Mexican market, commanding 11% market share with a CAGR of 10%. Lab Grin has superior brand recognition and an outstanding reputation with the medical community. It has state-of-the-art manufacturing facility, a highly regarded
management team and attractive profitability margins. The Company believes that the acquisition would add value in the opthalmic pharmaceutical and opthalmic vision care segments. The deal is subject to compliance to certain conditions which were yet to be fulfilled as on date.
(b) the entire shareholding of Nanomi B.V., the Netherlands, (‘Nanomi’) to make significant in‐roads into the niche area of complex injectables. Nanomi has patented technology platforms to develop complex injectable products and a rich talent pool of scientists who would be backed by the Company’s global R&D and manufacturing teams. Lupin Holdings B.V., Netherlands, wholly-owned subsidiary of the Company, acquired the entire shareholding of Farma World Importacao e Exportacao de Medicamentos LTDA - EPP, Brazil (‘Farma World’). The objective of Farma World is to import and export finished dosages, API, R&D consulting and participate in investments in Brazil and abroad.
CONTINGENT LIABILITIES:
|
PARTICULAR |
31.03.2014 (Rs.
In Millions) |
31.03.2013 (Rs.
In Millions) |
|
a) Income tax demands / matters on account of deductions / disallowances in earlier years, pending in appeals [RS. 49.7 million (previous year 49.7 million) consequent to department preferring appeals against the orders of the Appellate Authorities passed in favour of the Company]. Amount paid there against and included under note 14 “Long-Term Loans and Advances” H 26.3 million (previous year H 23.5 million). |
173.200 |
82.300 |
|
b) Excise duty, Service tax and Sales tax demands for input tax credit disallowances and demand for additional Entry Tax arising from dispute on applicable rate are in appeals and pending decisions. Amount paid there against and included under note 19 “Short-Term Loans and Advances” H 30.4 million (previous year H 31.7 million). |
355.500 |
424.400 |
|
c) Claims against the Company not acknowledged as debts [excluding interest (amount unascertained) in respect of a claim] for transfer charges of land, octroi duty, local body tax, employee claims, power, trade marks, pricing, indemnity and stamp duty. Amount paid there against without admitting liability and included “Short-Term Loans and Advances” H 12.6 million (previous year H 12.6 million). |
830.800 |
419.900 |
|
d) Counter guarantee given to GIDC in connection with repayment of loan sanctioned by a financial institution to a company, jointly promoted by an Association of Industries (of which, the Company is a member) and GIDC. |
7.500 |
7.500 |
|
e) Letter of comfort issued by the Company towards the credit facilities sanctioned by the bankers of subsidiary companies aggregating H 133.5 million (previous year H 133.0 million). |
26.700 |
39.900 |
|
f) Corporate guarantee given in respect of credit facility sanctioned by bankers of subsidiary companies aggregating H 2264.2 million (previous year H 2738.9 million). |
2124.100 |
2645.800 |
Future cash outflows in respect of the above, if any, is determinable only on receipt of judgement / decisions pending with the relevant authorities. The Company does not expect the outcome of the matters stated above to have a material adverse impact on the Company’s financial condition, results of operations or cash flows.
The Company does not envisage any likely reimbursements in respect of the above.
The Company is involved in various legal proceedings, including product liability related claims, employment claims and other regulatory matters relating to conduct of its business. The Company carries product liability insurance policy with an amount it believes is sufficient for its needs. In respect of other claims, the Company believes, these claims do not constitute material litigation matters and with its meritorious defenses the ultimate disposition of these matters will not have material adverse effect on its Financial Statements.
STATEMENT OF STANDALONE
UNAUDITED FINANCIAL RESULTS FOR THE QUARTER AND NINE MONTH ENDED 31st
DECEMBER, 2014
(Rs. In Millions)
|
Sr. No. |
Particular |
3
months ended 31.12.2014 |
3
months ended 30.09.2014 |
9
months ended 31.12.2014 |
|
|
|
Unaudited |
Unaudited |
Unaudited |
|
1. |
Income from Operation |
|
|
|
|
|
a. Net Sales/
Income from Operations |
23025.200 |
22074.800 |
74658.400 |
|
|
b. Other
Operating Income |
252.200 |
498.100 |
1214.100 |
|
|
Total Income from Operations |
23277.400 |
22572.900 |
75872.500 |
|
2. |
Expenses |
|
|
|
|
|
Consumption
of materials consumed |
5464.700 |
5862.700 |
16802.300 |
|
|
Purchase
of stock in trade |
2224.100 |
2536.700 |
7223.100 |
|
|
Change
in inventories of Finished Goods and stock in process |
(204.600) |
(281.000) |
(804.600) |
|
|
Employee
benefits expenses |
2424.100 |
2715.600 |
7556.900 |
|
|
Depreciation
and amortization expenses |
845.300 |
858.100 |
2574.900 |
|
|
Other
expenses |
6002.400 |
5740.300 |
17161.800 |
|
|
Total Expenses |
16756.000 |
17432.400 |
50514.400 |
|
|
|
|
|
|
|
3. |
Profit from Operations before Other Income, Finance Costs & Exceptional Items (1-2) |
6521.400 |
5140.500 |
25356.100 |
|
4. |
Other Income (net) |
782.900 |
905.400 |
1923.600 |
|
5. |
Profit from ordinary activities Before Finance Costs & Exceptional Items (3+4) |
7304.300 |
6045.900 |
27279.700 |
|
6. |
Finance Costs |
10.800 |
10.200 |
33.200 |
|
7. |
Profit from ordinary activities after finance costs but before exceptional items (5-6) |
7293.500 |
6035.700 |
27246.500 |
|
8. |
Exceptional Items |
-- |
--- |
--- |
|
9. |
Profit from ordinary activities before Tax (7-8) |
7293.500 |
6035.700 |
27246.500 |
|
10. |
Tax Expense |
2160.500 |
1.439.500 |
7350.000 |
|
11. |
Net Profit from ordinary activities after tax (9-10) |
5133.000 |
4.596.200 |
19896.500 |
|
12. |
Extraordinary Items (net of tax expense) |
-- |
-- |
--- |
|
13. |
Net Profit for the period |
5133.000 |
4.596.200 |
19896.500 |
|
14. |
Paid up Equity Share Capital (Face Value of Rs.2/- per share) |
898.400 |
897.900 |
898.400 |
|
15. |
Reserves excluding revaluation reserve as per balance sheet of the previous accounting year |
|
|
|
|
16. |
Earnings Per Share (of Rs. 2/- each) (not annualised) |
|
|
|
|
|
a)
Basic |
11.44 |
10.25 |
44.34 |
|
|
b)
Diluted |
11.38 |
10.19 |
44.09 |
|
|
|
|
|
|
|
15. |
Public Shareholding |
|
|
|
|
|
-Number
of Shares |
239609583 |
239324005 |
239609583 |
|
|
-
Percentage of Shareholding |
53.34 |
53.31 |
53.34 |
|
|
|
|
|
|
|
19. |
Promoters and Promoter Group
Shareholding |
|
|
|
|
|
a) Pledged/Encumbered |
|
|
|
|
|
-
Number of Shares |
--- |
--- |
--- |
|
|
-
Percentage of Shares (as a % of the Total Shareholding of promoter and
promoter group) |
--- |
--- |
--- |
|
|
- Percentage
of Shares (as a % of the Total Share Capital of the Company) |
--- |
--- |
--- |
|
|
|
|
|
|
|
|
b) Non Encumbered |
|
|
|
|
|
-
Number of Shares |
209602240 |
209602240 |
209602240 |
|
|
- Percentage
of Shares (as a % of the Total Shareholding of Promoter and Promoter Group) |
100.00 |
100.00 |
100.00 |
|
|
-
Percentage of Shares (as a % of the Total Share Capital of the Company) |
46.66 |
46.69 |
46.66 |
|
Particulars |
3 Months Ended 31.12.2014 |
|
B. INVESTOR COMPLAINTS (Nos.) |
|
|
Pending at the beginning of the quarter |
-- |
|
Received during the quarter |
13 |
|
Disposed of during the quarter |
13 |
|
Remaining unresolved at the end of the
quarter |
-- |
The above Standalone Financial Results were reviewed by the
Audit Committee and thereafter approved and taken on record by the Board of
Directors at their meeting held on February 03, 2015. The Statutory Auditors of
the Company have carried out limited review of the above standalone results
pursuant to clause 41 of the Listing Agreements.
During the quarter, 285,578 (year-to-date 836,019) equity shares of Rs. 2/-
each, fully paid-up, were allotted upon exercise of the vested stock options
pursuant to the Lupin Employees Stock Option Plans, resulting in an increase in
the paid-up share capital by Rs. 5 lakhs (year-to-date Rs. 1.600 Millions) and
securities premium account by Rs. 110.400 Millions (year-to-date Rs. 313.700
Millions).
The aggregate amount of revenue expenditure incurred on Research and Development as reflected under the respective heads of account is as under :
Rs. In Millions
|
3 Months
Ended |
3 Months
Ended |
3 Months
Ended |
9 Months
Ended |
9 Months
Ended |
Accounting Year Ended |
|
31.12.2014 |
30.09.2014 |
31.12.2013 |
31.12.2014 |
31.12.2013 |
31.03.2014 |
|
1957.200 |
2199.800 |
2294.400 |
6094.500 |
6093.200 |
8112.000 |
The Company operates in one reportable business segment i.e.
"Pharmaceuticals".
In terms of Schedule II to the Companies Act, 2013 read together with
Accounting Standard 6 (AS - 6) "Depreciation Accounting", the
management of the Company has, based on technical evaluation, reassessed the
remaining useful lives of fixed assets and undertaken the componentization of
major items of fixed assets with effect from April 01, 2014. In terms of these
evaluations, the useful lives of certain assets required changes from their
previous estimates. As a result, the charge for depreciation in respect of
fixed assets held as at April 01, 2014 is higher by Rs. 391.500 Millions for
the quarter ended December 31, 2014 (year-to-date Rs. 12,84.400 Millions).
Figures for the previous periods have been regrouped, wherever necessary, to
correspond with the figures of the current period.
FIXED ASSETS
Tangible Assets
· Freehold Land
· Leasehold Land
· Buildings
· Plant and Equipment
· Furniture and Fixtures
· Vehicles
· Office Equipment
Intangible Assets
· Goodwill
· Computer Software
· Trademarks and Licences
CMT REPORT (Corruption, Money Laundering & Terrorism]
The Public Notice information has been collected from various sources
including but not limited to: The Courts,
1] INFORMATION ON
DESIGNATED PARTY
No exist designating subject or any of its beneficial owners,
controlling shareholders or senior officers as terrorist or terrorist
organization or whom notice had been received that all financial transactions
involving their assets have been blocked or convicted, found guilty or against
whom a judgement or order had been entered in a proceedings for violating
money-laundering, anti-corruption or bribery or international economic or
anti-terrorism sanction laws or whose assets were seized, blocked, frozen or
ordered forfeited for violation of money laundering or international
anti-terrorism laws.
2] Court Declaration :
No exist to suggest that subject is or was
the subject of any formal or informal allegations, prosecutions or other
official proceeding for making any prohibited payments or other improper
payments to government officials for engaging in prohibited transactions or
with designated parties.
3] Asset Declaration :
No records exist to suggest that the property or assets of the subject
are derived from criminal conduct or a prohibited transaction.
4] Record on Financial
Crime :
Charges or conviction
registered against subject: None
5] Records on Violation of
Anti-Corruption Laws :
Charges or
investigation registered against subject: None
6] Records on Int’l
Anti-Money Laundering Laws/Standards :
Charges or
investigation registered against subject: None
7] Criminal Records
No
available information exist that suggest that subject or any of its principals
have been formally charged or convicted by a competent governmental authority
for any financial crime or under any formal investigation by a competent
government authority for any violation of anti-corruption laws or international
anti-money laundering laws or standard.
8] Affiliation with
Government :
No record
exists to suggest that any director or indirect owners, controlling
shareholders, director, officer or employee of the company is a government
official or a family member or close business associate of a Government
official.
9] Compensation Package :
Our market
survey revealed that the amount of compensation sought by the subject is fair
and reasonable and comparable to compensation paid to others for similar
services.
10] Press Report :
No press reports / filings exists on
the subject.
CORPORATE GOVERNANCE
MIRA INFORM as part of its Due Diligence do provide comments on
Corporate Governance to identify management and governance. These factors often
have been predictive and in some cases have created vulnerabilities to credit
deterioration.
Our Governance Assessment focuses principally on the interactions
between a company’s management, its Board of Directors, Shareholders and other
financial stakeholders.
CONTRAVENTION
Subject is not known to have contravened any existing local laws,
regulations or policies that prohibit, restrict or otherwise affect the terms
and conditions that could be included in the agreement with the subject.
FOREIGN EXCHANGE RATES
|
Currency |
Unit
|
Indian Rupees |
|
US Dollar |
1 |
Rs. 62.14 |
|
|
1 |
Rs. 94.79 |
|
Euro |
1 |
Rs. 70.38 |
INFORMATION DETAILS
|
Information
Gathered by : |
HET |
|
|
|
|
Analysis Done by
: |
KAR |
|
|
|
|
Report Prepared
by : |
TRU |
SCORE & RATING EXPLANATIONS
|
SCORE FACTORS |
RANGE |
POINTS |
|
HISTORY |
1~10 |
8 |
|
PAID-UP CAPITAL |
1~10 |
8 |
|
OPERATING SCALE |
1~10 |
8 |
|
FINANCIAL
CONDITION |
|
|
|
--BUSINESS SCALE |
1~10 |
8 |
|
--PROFITABILITY |
1~10 |
8 |
|
--LIQUIDITY |
1~10 |
8 |
|
--LEVERAGE |
1~10 |
8 |
|
--RESERVES |
1~10 |
8 |
|
--CREDIT LINES |
1~10 |
8 |
|
--MARGINS |
-5~5 |
-- |
|
DEMERIT POINTS |
|
|
|
--BANK CHARGES |
YES/NO |
YES |
|
--LITIGATION |
YES/NO |
YES |
|
--OTHER ADVERSE INFORMATION |
YES/NO |
NO |
|
MERIT POINTS |
|
|
|
--SOLE DISTRIBUTORSHIP |
YES/NO |
NO |
|
--EXPORT ACTIVITIES |
YES/NO |
NO |
|
--AFFILIATION |
YES/NO |
YES |
|
--LISTED |
YES/NO |
YES |
|
--OTHER MERIT FACTORS |
YES/NO |
YES |
|
DEFAULTER
|
|
|
|
--RBI |
YES/NO |
NO |
|
--EPF |
YES/NO |
NO |
|
TOTAL |
|
72 |
This score serves as a reference to assess
SC’s credit risk and to set the amount of credit to be extended. It is
calculated from a composite of weighted scores obtained from each of the major
sections of this report. The assessed factors and their relative weights (as
indicated through %) are as follows:
Financial
condition (40%) Ownership
background (20%) Payment
record (10%)
Credit history
(10%) Market trend (10%) Operational size
(10%)
RATING EXPLANATIONS
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
>86 |
Aaa |
Possesses an extremely
sound financial base with the strongest capability for timely payment of
interest and principal sums |
Unlimited |
|
71-85 |
Aa |
Possesses adequate working
capital. No caution needed for credit transaction. It has above average
(strong) capability for payment of interest and principal sums |
Large |
|
56-70 |
A |
Financial &
operational base are regarded healthy. General unfavourable factors will not cause
fatal effect. Satisfactory capability for payment of interest and principal
sums |
Fairly
Large |
|
41-55 |
Ba |
Overall operation is
considered normal. Capable to meet normal commitments. |
Satisfactory |
|
26-40 |
B |
Capability to overcome financial
difficulties seems comparatively below average. |
Small |
|
11-25 |
Ca |
Adverse factors are
apparent. Repayment of interest and principal sums in default or expected to
be in default upon maturity |
Limited
with full security |
|
<10 |
C |
Absolute credit risk
exists. Caution needed to be exercised |
Credit
not recommended |
|
-- |
NB |
New
Business |
-- |
This report is issued at your request without any
risk and responsibility on the part of MIRA INFORM PRIVATE LIMITED (MIPL) or
its officials.