MIRA INFORM REPORT
|
Report No. : |
308616 |
|
Report Date : |
20.02.2015 |
IDENTIFICATION DETAILS
|
Name : |
SUN PHARMA ADVANCED RESEARCH COMPANY LIMITED |
|
|
|
|
Registered
Office : |
Akota Road, Akota, Vadodara 390 020, Gujarat |
|
|
|
|
Country : |
India |
|
|
|
|
Financials (as
on) : |
31.03.2014 |
|
|
|
|
Date of
Incorporation : |
01.03.2006 |
|
|
|
|
Com. Reg. No.: |
04-047837 |
|
|
|
|
Capital
Investment / Paid-up Capital : |
Rs. 236.680 Millions |
|
|
|
|
CIN No.: [Company Identification
No.] |
L73100GJ2006PLC047837 |
|
|
|
|
IEC No.: |
Not Available |
|
|
|
|
TAN No.: [Tax Deduction &
Collection Account No.] |
Not Available |
|
|
|
|
PAN No.: [Permanent Account No.] |
Not Available |
|
|
|
|
Legal Form : |
A Public Limited Liability Company. The Company’s Shares are Listed on
the Stock Exchanges. |
|
|
|
|
Line of Business
: |
Pharmaceuticals Research and Development |
|
|
|
|
No. of Employees
: |
Information declined by the management. |
RATING & COMMENTS
|
MIRA’s Rating : |
Ba (51) |
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
41-55 |
Ba |
Overall operation is considered normal. Capable to meet normal
commitments. |
Satisfactory |
|
Maximum Credit Limit : |
USD 3900000 |
|
|
|
|
Status : |
Satisfactory |
|
|
|
|
Payment Behaviour : |
Usually correct |
|
|
|
|
Litigation : |
Clear |
|
|
|
|
Comments : |
Subject is a subsidiary of ‘Sun Pharmaceutical Industries Limited’. It
is an established company having a satisfactory track record. Trade relations are reported as fair. Business is active. Payment
terms are usually correct. In view of strong holding company, the subject can be considered for
business dealing at usual trade terms and conditions. S |
NOTES :
Any query related to this report can be made
on e-mail : infodept@mirainform.com
while quoting report number, name and date.
ECGC Country Risk Classification List – December 31, 2014
|
Country Name |
Previous Rating (30.09.2014) |
Current Rating (31.12.2014) |
|
India |
A1 |
A1 |
|
Risk Category |
ECGC
Classification |
|
Insignificant |
A1 |
|
Low |
A2 |
|
Moderate |
B1 |
|
High |
B2 |
|
Very High |
C1 |
|
Restricted |
C2 |
|
Off-credit |
D |
EXTERNAL AGENCY RATING
Not Available
RBI DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available RBI Defaulters’ list.
EPF (Employee Provident Fund) DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available EPF (Employee Provident Fund) Defaulters’ list as of
31-03-2014.
INFORMATION DENIED
Management Non Cooperative (91-22-28212010)
LOCATIONS
|
Registered Office : |
Akota Road, Akota, Vadodara 390 020, Gujarat, India |
|
Tel. No.: |
91-265-2330815 |
|
Fax No.: |
91-265-2354897 |
|
E-Mail : |
|
|
Website : |
|
|
|
|
|
Mumbai Office / Research
Centres 1 : |
17-B, Mahal Industrial Estate, Mahakali Caves Road, Andheri (East), Mumbai – 400 093, Maharashtra, India |
|
|
|
|
Research Centres 2 : |
|
DIRECTORS
AS ON 31.03.2014
|
Name : |
Mr. Dilip S. Shanghvi |
|
Designation : |
Chairman and Managing Director |
|
|
|
|
Name : |
Dr. T. Rajamannar |
|
Designation : |
Whole time Director and Executive Vice President, R&D |
|
|
|
|
Name : |
Mr. Sudhir V. Valia |
|
Designation : |
Director |
|
|
|
|
Name : |
Prof. Dr. Andrea Vasella |
|
Designation : |
Director |
|
|
|
|
Name : |
Prof. Dr. Goverdhan Mehta |
|
Designation : |
Director |
|
|
|
|
Name : |
Mr. S. Mohanchand Dadha |
|
Designation : |
Director |
KEY EXECUTIVES
|
Name : |
Ms. Meetal Sampat |
|
Designation : |
Company Secretary |
|
|
|
|
Name : |
Mr. Anil Reghavan |
|
Designation : |
Chief Executive Officer |
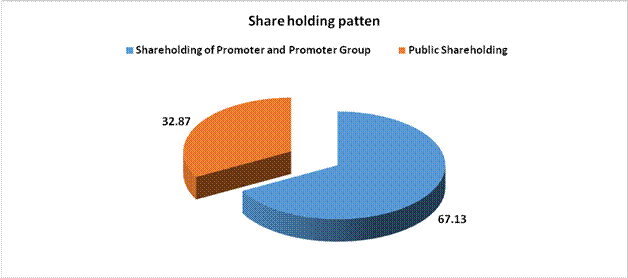
SHAREHOLDING PATTERN
As on 31.12.2014
|
Category
of Shareholder |
Total No.
of Shares |
Total
Shareholding as a % of Total No. of Shares |
|
As a % of (A+B) |
||
|
(A) Shareholding of Promoter
and Promoter Group |
||
|
|
|
|
|
|
35470143 |
14.98 |
|
|
123275262 |
52.08 |
|
|
147791 |
0.06 |
|
|
147791 |
0.06 |
|
|
158893196 |
67.13 |
|
|
|
|
|
Total shareholding of Promoter
and Promoter Group (A) |
158893196 |
67.13 |
|
(B) Public Shareholding |
||
|
|
|
|
|
|
2771373 |
1.17 |
|
|
210515 |
0.09 |
|
|
229144 |
0.10 |
|
|
11002518 |
4.65 |
|
|
14213550 |
6.00 |
|
|
|
|
|
|
13489071 |
5.70 |
|
|
|
|
|
|
30575170 |
12.92 |
|
|
16961420 |
7.17 |
|
|
2572040 |
1.09 |
|
|
1141449 |
0.48 |
|
|
786055 |
0.33 |
|
|
3593 |
0.00 |
|
|
9600 |
0.00 |
|
|
620843 |
0.26 |
|
|
10500 |
0.00 |
|
|
63597701 |
26.87 |
|
Total Public shareholding (B) |
77811251 |
32.87 |
|
Total (A)+(B) |
236704447 |
100.00 |
|
(C) Shares held by Custodians and
against which Depository Receipts have been issued |
|
|
|
|
0 |
0.00 |
|
|
0 |
0.00 |
|
|
0 |
0.00 |
|
Total (A)+(B)+(C) |
236704447 |
100.00 |
BUSINESS DETAILS
|
Line of Business : |
Pharmaceuticals Research and Development |
|
|
|
|
Products : |
Not Divulged |
|
|
|
|
Brand Names : |
Not Available |
|
|
|
|
Agencies Held : |
Not Available |
|
|
|
|
Exports : |
Not Divulged |
|
|
|
|
Imports : |
Not Divulged |
|
|
|
|
Terms : |
|
|
Selling : |
Not Divulged |
|
|
|
|
Purchasing : |
Not Divulged |
PRODUCTION STATUS : Not Available
GENERAL INFORMATION
|
Suppliers : |
|
|||||||||||||||
|
|
|
|||||||||||||||
|
Customers : |
|
|||||||||||||||
|
|
|
|||||||||||||||
|
No. of Employees : |
Information declined by the management. |
|||||||||||||||
|
|
|
|||||||||||||||
|
Bankers : |
|
|||||||||||||||
|
|
|
|||||||||||||||
|
Facilities : |
|
|
|
|
|
Banking
Relations : |
-- |
|
|
|
|
Auditors : |
|
|
Name : |
Deloitte Haskins and Sells Chartered Accountants |
|
Address : |
Mumbai, Maharashtra, India |
|
|
|
|
Memberships : |
Not Available |
|
|
|
|
Collaborators : |
Not Available |
|
|
|
|
Enterprise under
significant Influence of Key Management Personnel : |
|
CAPITAL STRUCTURE
As on 31.03.2014
Authorised Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
266500000 |
Equity Shares |
Rs.1/- each |
Rs.266.500 Millions |
|
|
|
|
|
Issued, Subscribed & Paid-up Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
236704447 |
Equity Shares |
Rs.1/- each |
Rs. 236.704Millions |
|
|
Less : Calls unpaid |
|
Rs. 0.024 Million |
|
|
Total |
|
Rs. 236.680
Millions |
FINANCIAL DATA
[all figures are
in Rupees Millions]
ABRIDGED BALANCE
SHEET
|
SOURCES OF FUNDS |
31.03.2014 |
31.03.2013 |
31.03.2012 |
|
|
|
|
|
|
I.
EQUITY AND LIABILITIES |
|
|
|
|
(1)Shareholders' Funds |
|
|
|
|
(a) Share Capital |
236.680 |
236.599 |
207.116 |
|
(b) Reserves &
Surplus |
1156.307 |
847.707 |
(873.202) |
|
(c) Money received against
share warrants |
0.000 |
0.000 |
0.000 |
|
|
|
|
|
|
(2) Share Application
money pending allotment |
0.000 |
0.000 |
0.000 |
|
Total Shareholders’ Funds
(1) + (2) |
1392.987 |
1084.306 |
-666.086 |
|
|
|
|
|
|
(3) Non-Current
Liabilities |
|
|
|
|
(a) long-term borrowings |
38.166 |
43.618 |
57.420 |
|
(b) Deferred tax
liabilities (Net) |
0.000 |
0.000 |
0.000 |
|
(c) Other long term
liabilities |
1.572 |
2.358 |
2.505 |
|
(d) long-term provisions |
14.768 |
17.255 |
12.380 |
|
Total Non-current
Liabilities (3) |
54.506 |
63.231 |
72.305 |
|
|
|
|
|
|
(4) Current Liabilities |
|
|
|
|
(a) Short term borrowings |
8.237 |
759.655 |
619.419 |
|
(b) Trade payables |
297.463 |
127.932 |
162.350 |
|
(c) Other current liabilities |
75.762 |
37.929 |
660.645 |
|
(d) Short-term provisions |
6.435 |
16.872 |
7.865 |
|
Total Current Liabilities
(4) |
387.897 |
942.388 |
1450.279 |
|
|
|
|
|
|
TOTAL |
1835.390 |
2089.925 |
856.498 |
|
|
|
|
|
|
II.
ASSETS |
|
|
|
|
(1) Non-current assets |
|
|
|
|
(a) Fixed Assets |
|
|
|
|
(i) Tangible assets |
644.882 |
655.492 |
639.299 |
|
(ii) Intangible Assets |
0.000 |
0.000 |
0.000 |
|
(iii) Capital
work-in-progress |
7.610 |
1.631 |
11.398 |
|
(iv) Intangible assets
under development |
0.000 |
0.000 |
0.000 |
|
(b) Non-current
Investments |
0.000 |
0.000 |
0.000 |
|
(c) Deferred tax assets
(net) |
0.000 |
0.000 |
0.000 |
|
(d) Long-term Loan and Advances |
75.999 |
34.160 |
18.187 |
|
(e) Other Non-current
assets |
5.734 |
8.507 |
5.203 |
|
Total Non-Current Assets |
734.225 |
699.790 |
674.087 |
|
|
|
|
|
|
(2) Current assets |
|
|
|
|
(a) Current investments |
65.002 |
969.059 |
0.000 |
|
(b) Inventories |
0.000 |
0.000 |
0.000 |
|
(c) Trade receivables |
279.989 |
250.564 |
42.642 |
|
(d) Cash and cash
equivalents |
65.551 |
67.237 |
65.050 |
|
(e) Short-term loans and
advances |
649.391 |
100.489 |
73.634 |
|
(f) Other current assets |
41.232 |
2.786 |
1.085 |
|
Total Current Assets |
1101.165 |
1390.135 |
182.411 |
|
|
|
|
|
|
TOTAL |
1835.390 |
2089.925 |
856.498 |
PROFIT & LOSS
ACCOUNT
|
|
PARTICULARS |
31.03.2014 |
31.03.2013 |
31.03.2012 |
|
|
SALES |
|
|
|
|
|
Income |
1670.297 |
872.790 |
289.765 |
|
|
Other Income |
100.030 |
16.169 |
11.457 |
|
|
TOTAL (A) |
1770.327 |
888.959 |
301.222 |
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
Cost of Materials
Consumed |
80.079 |
93.054 |
74.155 |
|
|
Employees benefits
expense |
403.972 |
365.537 |
304.279 |
|
|
Other expenses |
851.338 |
581.854 |
610.737 |
|
|
TOTAL (B) |
1335.389 |
1040.445 |
989.171 |
|
|
|
|
|
|
|
Less |
PROFIT/ (LOSS) BEFORE INTEREST, TAX, DEPRECIATION AND
AMORTISATION (C) |
434.938 |
(151.486) |
(687.949) |
|
|
|
|
|
|
|
Less |
FINANCIAL EXPENSES (D) |
55.973 |
39.506 |
2.749 |
|
|
|
|
|
|
|
|
PROFIT / (LOSS) BEFORE
TAX, DEPRECIATION AND AMORTISATION (C-D) (E) |
378.965 |
(190.992) |
(690.698) |
|
|
|
|
|
|
|
Less/ Add |
DEPRECIATION/
AMORTISATION (F) |
35.723 |
33.955 |
31.623 |
|
|
|
|
|
|
|
|
PROFIT/ (LOSS) BEFORE TAX (E-F) (G) |
343.242 |
(224.947) |
(722.321) |
|
|
|
|
|
|
|
Less |
TAX (H) |
40.000 |
0.000 |
0.000 |
|
|
|
|
|
|
|
|
PROFIT/ (LOSS) AFTER TAX
(G-H) (I) |
303.242 |
(224.947) |
(722.321) |
|
|
|
|
|
|
|
Add |
PREVIOUS YEARS’ BALANCE
BROUGHT FORWARD (J) |
(1437.915) |
(1212.968) |
(490.647) |
|
|
|
|
|
|
|
|
Balance Carried to the
B/S (I+J) |
(1134.673) |
(1437.915) |
(1212.968) |
|
|
|
|
|
|
|
|
EARNINGS IN FOREIGN
CURRENCY |
|
|
|
|
|
Sale
of Products - Technology / Know-how |
262.257 |
699.080 |
160.444 |
|
|
Sale of Services -
License Fees / Royalty on Technology |
1032.715 |
42.043 |
0.000 |
|
|
Insurance |
|
|
|
|
|
TOTAL EARNINGS |
1294.972 |
741.123 |
160.444 |
|
|
|
|
|
|
|
|
IMPORTS |
|
|
|
|
|
R & D Material
Consumed |
23.936 |
20.759 |
21.349 |
|
|
Capital Goods |
7.737 |
19.442 |
26.724 |
|
|
Spare and components |
22.063 |
7.113 |
7.808 |
|
|
TOTAL IMPORTS |
53.736 |
47.314 |
55.881 |
|
|
|
|
|
|
|
|
Earnings / (Loss) Per
Share (Rs.) |
1.28 |
(1.03) |
(3.43) |
KEY RATIOS
|
PARTICULARS |
|
31.03.2014 |
31.03.2013 |
31.03.2012 |
|
|
|
|
|
|
|
Net Profit Margin (PAT / Sales) |
(%) |
18.15 |
(25.77) |
(249.28) |
|
|
|
|
|
|
|
Operating Profit Margin (PBDIT/Sales) |
(%) |
26.04 |
(17.36) |
(237.42) |
|
|
|
|
|
|
|
Return on Total Assets (PBT/Total Assets} |
(%) |
18.78 |
(10.77) |
(85.47) |
|
|
|
|
|
|
|
Return on Investment (ROI) (PBT/Networth) |
|
0.25 |
(0.21) |
1.08 |
|
|
|
|
|
|
|
Debt Equity Ratio (Total Debt /Networth) |
|
0.03 |
0.74 |
(1.02) |
|
|
|
|
|
|
|
Current Ratio (Current Asset/Current Liability) |
|
2.84 |
1.48 |
0.13 |
FINANCIAL ANALYSIS
[all figures are
in Rupees Millions]
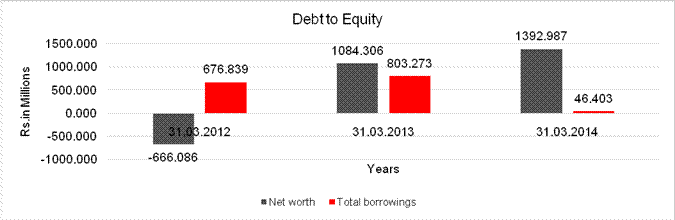
DEBT EQUITY RATIO
|
Particular |
31.03.2012 |
31.03.2013 |
31.03.2014 |
|
|
(Rs. In Millions) |
(Rs. In Millions) |
(Rs. In Millions) |
|
Share Capital |
207.116 |
236.599 |
236.680 |
|
Reserves & Surplus |
(873.202) |
847.707 |
1156.307 |
|
Net worth |
(666.086) |
1084.306 |
1392.987 |
|
|
|
|
|
|
long-term borrowings |
57.420 |
43.618 |
38.166 |
|
Short term borrowings |
619.419 |
759.655 |
8.237 |
|
Total borrowings |
676.839 |
803.273 |
46.403 |
|
Debt/Equity ratio |
(1.016) |
0.741 |
0.033 |
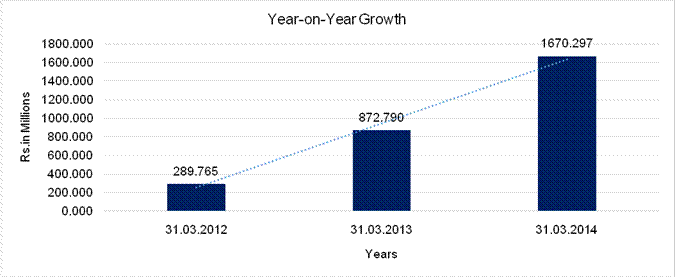
YEAR-ON-YEAR GROWTH
|
Year on Year Growth |
31.03.2012 |
31.03.2013 |
31.03.2014 |
|
|
(Rs. In Millions) |
(Rs. In Millions) |
(Rs. In Millions) |
|
Sales |
289.765 |
872.790 |
1670.297 |
|
|
|
201.206 |
91.374 |
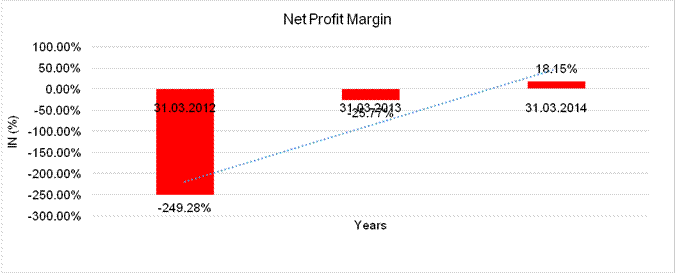
NET PROFIT MARGIN
|
Net Profit Margin |
31.03.2012 |
31.03.2013 |
31.03.2014 |
|
|
(Rs. In Millions) |
(Rs. In Millions) |
(Rs. In Millions) |
|
Sales |
289.765 |
872.790 |
1670.297 |
|
Profit |
(722.321) |
(224.947) |
303.242 |
|
|
(249.28%) |
(25.77%) |
18.15% |
LOCAL AGENCY FURTHER INFORMATION
|
Sr. No. |
Check List by Info Agents |
Available in
Report (Yes / No) |
|
1] |
Year of Establishment |
Yes |
|
2] |
Locality of the firm |
Yes |
|
3] |
Constitutions of the firm |
Yes |
|
4] |
Premises details |
No |
|
5] |
Type of Business |
Yes |
|
6] |
Line of Business |
Yes |
|
7] |
Promoter's background |
No |
|
8] |
No. of employees |
No |
|
9] |
Name of person contacted |
No |
|
10] |
Designation of contact
person |
No |
|
11] |
Turnover of firm for last
three years |
Yes |
|
12] |
Profitability for last
three years |
Yes |
|
13] |
Reasons for variation
<> 20% |
------------------------ |
|
14] |
Estimation for coming
financial year |
No |
|
15] |
Capital in the business |
Yes |
|
16] |
Details of sister
concerns |
Yes |
|
17] |
Major suppliers |
No |
|
18] |
Major customers |
No |
|
19] |
Payments terms |
No |
|
20] |
Export / Import details
(if applicable) |
No |
|
21] |
Market information |
---------------------- |
|
22] |
Litigations that the firm
/ promoter involved in |
---------------------- |
|
23] |
Banking Details |
Yes |
|
24] |
Banking facility details |
Yes |
|
25] |
Conduct of the banking
account |
---------------------- |
|
26] |
Buyer visit details |
---------------------- |
|
27] |
Financials, if provided |
Yes |
|
28] |
Incorporation details, if
applicable |
Yes |
|
29] |
Last accounts filed at
ROC |
Yes |
|
30] |
Major Shareholders, if
available |
Yes |
|
31] |
Date of Birth of Proprietor/Partner/Director,
if available |
No |
|
32] |
PAN of
Proprietor/Partner/Director, if available |
No |
|
33] |
Voter ID No of
Proprietor/Partner/Director, if available |
No |
|
34] |
External Agency Rating,
if available |
No |
OUTLOOK
Being an R&D company, it is imperative for SPARC to strike a reasonable balance between risks and rewards that such a business necessitates. Over the past few years, SPARC has attempted to balance out between medium-term and long-term R&D projects. Broadly, their New Drug Delivery System (NDDS) projects are directed at potential commercialization in the medium-term while their New Chemical Entity (NCE) projects will potentially get commercialized in the long-term.
As they take their NCE and NDDS projects ahead on the research pathway, they are learning about how to manage in a changing regulatory environment, handle the technical demands of innovation, and balance the requirements of projects that have short term, medium term and long term timeframes. While they are satisfied with the progress on their projects so far, THEY recognize that they have quite some distance to go before they reach market, though some NDDS projects are closer to market than they were previously.
VIEW INDEX OF
CHARGES: No Charges Exist for Company
UNSECURED LOANS
|
PARTICULAR |
31.03.2014 (Rs.
in Millions) |
31.03.2013 (Rs.
in Millions) |
|
LONG TERM
BORROWINGS |
|
|
|
Term Loan from Department of Science and Technology (DST), Government of India under the “Drug and Pharmaceutical Research Program” (Unsecured) [Repayable in 8 (Previous Year 9) Annual Installments of Rs. 5.452 Millions each. Last installment is due on 1st September, 2021]. For the current maturities of long term borrowings refer Note 9 Other Current Liabilities. |
38.166 |
43.618 |
|
|
|
|
|
SHORT TERM
BORROWINGS |
|
|
|
Loans Repayable on Demand - From Banks Bank Overdraft Facility |
5.994 |
16.925 |
|
Loans from a Related Party |
0.000 |
738.410 |
|
Total |
44.160 |
798.953 |
CONTINGENT
LIABILITIES:
|
PARTICULARS |
31.03.2014 (Rs.
In Millions) |
31.03.2013 (Rs.
In Millions) |
|
Guarantees given by the bankers against Advance License Scheme |
43.020 |
52.651 |
FIXED ASSETS
- Buildings
- Plant and Equipment
- Furniture and Fixtures
- Vehicles
UNAUDITED
FINANCIAL RESULTS FOR THE QUARTER AND NINE MONTHS ENDED DECEMBER 31, 2014
|
Particulars |
Quarter ended |
Nine Months ended |
||
|
31.12.2014 |
30.09.2014 |
31.12.2014 |
||
|
Unaudited |
Unaudited |
Unaudited |
||
|
1 |
Income from Operations |
|
|
|
|
|
Income from operations |
312.800 |
459.100 |
1130.400 |
|
|
Total income |
312.800 |
459.100 |
1130.400 |
|
2 |
Expenses |
|
|
|
|
|
Cost of materials consumed |
32.500 |
19.500 |
70.400 |
|
|
Employee benefits expense |
115.000 |
112.600 |
338.900 |
|
|
Clinical Trials and Professional Charges |
288.000 |
268.300 |
828.600 |
|
|
Depreciation Expenses |
18.000 |
20.600 |
54.600 |
|
|
License expenses |
1.300 |
3.500 |
7.000 |
|
|
Other Expenses |
55.800 |
55.600 |
156.400 |
|
|
Total expenses |
510.600 |
480.100 |
1455.900 |
|
3 |
Profit/ (Loss) from operations before other Income, finance costs and
exceptional Items |
(197.800) |
(21.000) |
(325.500) |
|
4 |
Other Income |
9.300 |
6.800 |
26.000 |
|
5 |
Profit/ (Loss) from operations before other income, finance costs and
exceptional items |
(188.500) |
(14.200) |
(299.500) |
|
6 |
Finance Costs |
0.600 |
0.500 |
1.700 |
|
7 |
Profit / (Loss) before tax |
(189.100) |
(14.700) |
(301.200) |
|
8 |
Tax expenses |
0.000 |
0.000 |
0.000 |
|
9 |
Profit / (Loss) for the period / year |
(189.100) |
(14.700) |
(301.200) |
|
10 |
Paid up equity share capital (Face Value of Rs1/-each) |
236.700 |
236.700 |
236.700 |
|
11 |
Reserve excluding Revaluation Reserve |
|
|
|
|
12 |
Earnings per share Rs. 1/- each
Basic Diluted |
(0.80) |
(0.06) |
(1.27) |
|
|
|
|
|
|
|
A |
PARTICULARS OF SHAREHOLDING |
|
|
|
|
1 |
Public Shareholding |
|
|
|
|
|
- Number of shares |
77811251 |
77811251 |
77811251 |
|
|
- Percentage of shareholding |
32.87 |
32.87 |
32.87 |
|
2 |
Promoters and Promoter group shareholding |
|
|
|
|
|
a) Pledged / Encumbered |
|
|
|
|
|
- Number of shares |
675000 |
675000 |
675000 |
|
|
- Percentage of shares (as a % of the total shareholding of Promoter
& Promoter group) |
0.42 |
0.42 |
0.42 |
|
|
- Percentage of shares (as a % of the total Share Capital of the
Company) |
0.29 |
0.29 |
0.29 |
|
|
b) Non Encumbered |
|
|
|
|
|
- Number of shares |
158218196 |
158218196 |
158218196 |
|
|
- Percentage of shares (as a % of the total shareholding of Promoter
& Promoter group) |
99.58 |
99.58 |
99.58 |
|
|
- Percentage of shares (as a % of the total Share Capital of the
Company) |
66.84 |
66.84 |
66.84 |
|
B |
INVESTOR
COMPLAINTS |
|
|
|
Pending at the beginning of the quarter Received during the quarter Disposed of during the quarter Remaining unresolved at the end of the quarter |
-- -- -- -- |
Note:
1. The above financial results of the Company have been reviewed by the Audit Committee and approved by the Board of Directors at their respective meetings held on January 30, 2015 and have been subjected to a limited review by the Statutory Auditors of the Company.
2. The Company has only one reportable business segment namely 'Pharmaceutical
Research & Development'.
3. Status of Utilisation of rights issue proceeds:
|
Particular |
Total Projected
Uitlisation |
Projected
Utilisation upto |
Actual Utilisation
upto |
Actual utilization
up to |
|
|
|
31.03.2014 |
31.03.2014 |
31.012.2014 |
|
|
|
|
|
|
|
Any Research and Development activities / expenses |
1031.600 |
516.100 |
514.300 |
950.500 |
|
Repayment of identified loans availed from group Entities |
610.000 |
610.000 |
610.000 |
610.000 |
|
General corporate purposes |
325.600 |
325.600 |
325.600 |
325.600 |
|
Issue expenses |
15.200 |
15.200 |
15.200 |
15.200 |
|
Funds utilized |
1982.400 |
|
1465.100 |
1901.300 |
|
Un-utilised rights issue proceeds |
-- |
|
515.700 |
79.700 |
|
|
1982.400 |
|
1980.800 |
1981.000 |
|
Calls unpaid |
-- |
|
1.600 |
1.400 |
|
Total |
1982.400 |
1466.900 |
1982.400 |
1982.400 |
Revised from ‘Pharmaceutical research and development activities – Clinical trials’ in terms of solution passed by shareholders at the Annual General Meeting held on July 30, 2013. Given the highly unpredictable nature of the company’s business of Pharmaceutical Research and Development, the actual utilization of the funds varies from the projections.
4. The Company has adopted the useful lives of fixed assets as indicated in
Part C of Schedule II of the Companies Act, 2013 and amendment thereto vide notification
dated August 29, 2014 issued by Ministry of Corporate Affairs. Consequently,
the depreciation charge for the quarter and nine months ended December 31, 2014
is higher by Rs. 7.500 Millions and Rs. 25.300 Millions respectively.
5. Figures for the previous periods / year have been regrouped / reclassified,
wherever considered necessary.
PRESS RELEASE
Sun Pharma Advanced
Research zooms 60% in one week
The stock rallied 13%
to Rs 316, extending its past four day's over 40% rally on BSE on back of heavy
volumes.
SI
Reporter | Mumbai January 23, 2015 Last Updated at 10:30
IST
Shares of Sun Pharma Advanced Research Company (SPARC) have rallied
13% to Rs 316, extending its past four day’s over 40% rally on BSE, on back of
heavy volumes.
The stock opened at Rs 281 and touched a record high of Rs 324 on BSE. The
trading volumes on the counter more than doubled with a combined 8.71 million
shares changed hands on the counter till 1015 hours on BSE and NSE.
In past one week, the stock of pharmaceutical company has rallied 60% from Rs
198 on January 16, 2015, since the company announced that a meeting of the
board of directors of the company will be held on January 30, 2015, to consider
and take on record the un-audited financial results of the company for the
third quarter ended December 31, 2014 (Q3). The benchmark S&P BSE Sensex
has gained 4% during the same period.
Meanwhile, in clarification on sudden increase in volume, SPARC has clarified to BSE that "there is no event
which will have a bearing on the performance / operations of the company nor
any price sensitive information of the company which are reportable under
clause 36 or any other clause of the listing agreement with the exchanges which
are not shared with the Exchanges”.
SPARC had sought US Food and Drug Administration (USFDA) approval for Latanoprost BAK-free to sale in the US market.
On December 1, 2014, the company announced that the USFDA had issued a Complete Response letter to its
New Drug Application (NDA) for
Latanoprost BAK-free eyedrops.
CMT REPORT (Corruption, Money Laundering & Terrorism]
The Public Notice information has been collected from various sources
including but not limited to: The Courts,
1] INFORMATION ON
DESIGNATED PARTY
No exist designating subject or any of its beneficial owners, controlling
shareholders or senior officers as terrorist or terrorist organization or whom
notice had been received that all financial transactions involving their assets
have been blocked or convicted, found guilty or against whom a judgement or
order had been entered in a proceedings for violating money-laundering,
anti-corruption or bribery or international economic or anti-terrorism sanction
laws or whose assets were seized, blocked, frozen or ordered forfeited for
violation of money laundering or international anti-terrorism laws.
2] Court Declaration :
No exist to suggest that subject is or was
the subject of any formal or informal allegations, prosecutions or other
official proceeding for making any prohibited payments or other improper payments
to government officials for engaging in prohibited transactions or with
designated parties.
3] Asset Declaration :
No records exist to suggest that the property or assets of the subject
are derived from criminal conduct or a prohibited transaction.
4] Record on Financial
Crime :
Charges or conviction
registered against subject: None
5] Records on Violation of
Anti-Corruption Laws :
Charges or
investigation registered against subject: None
6] Records on Int’l
Anti-Money Laundering Laws/Standards :
Charges or
investigation registered against subject: None
7] Criminal Records
No
available information exist that suggest that subject or any of its principals
have been formally charged or convicted by a competent governmental authority
for any financial crime or under any formal investigation by a competent
government authority for any violation of anti-corruption laws or international
anti-money laundering laws or standard.
8] Affiliation with
Government :
No record
exists to suggest that any director or indirect owners, controlling
shareholders, director, officer or employee of the company is a government
official or a family member or close business associate of a Government
official.
9] Compensation Package :
Our market
survey revealed that the amount of compensation sought by the subject is fair
and reasonable and comparable to compensation paid to others for similar
services.
10] Press Report :
No press reports / filings exists on
the subject.
CORPORATE GOVERNANCE
MIRA INFORM as part of its Due Diligence do provide comments on
Corporate Governance to identify management and governance. These factors often
have been predictive and in some cases have created vulnerabilities to credit
deterioration.
Our Governance Assessment focuses principally on the interactions
between a company’s management, its Board of Directors, Shareholders and other
financial stakeholders.
CONTRAVENTION
Subject is not known to have contravened any existing local laws,
regulations or policies that prohibit, restrict or otherwise affect the terms
and conditions that could be included in the agreement with the subject.
FOREIGN EXCHANGE RATES
|
Currency |
Unit
|
Indian Rupees |
|
US Dollar |
1 |
Rs. 62.25 |
|
|
1 |
Rs. 95.57 |
|
Euro |
1 |
Rs. 71.01 |
INFORMATION DETAILS
|
Information
Gathered by : |
SVA |
|
|
|
|
Analysis Done by
: |
SUB |
|
|
|
|
Report Prepared
by : |
TRU |
SCORE & RATING EXPLANATIONS
|
SCORE FACTORS |
RANGE |
POINTS |
|
HISTORY |
1~10 |
7 |
|
PAID-UP CAPITAL |
1~10 |
6 |
|
OPERATING SCALE |
1~10 |
5 |
|
FINANCIAL
CONDITION |
|
|
|
--BUSINESS SCALE |
1~10 |
6 |
|
--PROFITABILITY |
1~10 |
5 |
|
--LIQUIDITY |
1~10 |
6 |
|
--LEVERAGE |
1~10 |
6 |
|
--RESERVES |
1~10 |
5 |
|
--CREDIT LINES |
1~10 |
5 |
|
--MARGINS |
-5~5 |
-- |
|
DEMERIT POINTS |
|
|
|
--BANK CHARGES |
YES/NO |
NO |
|
--LITIGATION |
YES/NO |
NO |
|
--OTHER ADVERSE INFORMATION |
YES/NO |
NO |
|
MERIT POINTS |
|
|
|
--SOLE DISTRIBUTORSHIP |
YES/NO |
NO |
|
--EXPORT ACTIVITIES |
YES/NO |
NO |
|
--AFFILIATION |
YES/NO |
YES |
|
--LISTED |
YES/NO |
YES |
|
--OTHER MERIT FACTORS |
YES/NO |
YES |
|
DEFAULTER |
|
|
|
--RBI |
YES/NO |
NO |
|
--EPF |
YES/NO |
NO |
|
TOTAL |
|
51 |
This score serves as a reference to assess SC’s
credit risk and to set the amount of credit to be extended. It is calculated
from a composite of weighted scores obtained from each of the major sections of
this report. The assessed factors and their relative weights (as indicated
through %) are as follows:
Financial
condition (40%) Ownership
background (20%) Payment
record (10%)
Credit history
(10%) Market trend (10%) Operational size
(10%)
RATING EXPLANATIONS
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
>86 |
Aaa |
Possesses an extremely sound financial base with the strongest
capability for timely payment of interest and principal sums |
Unlimited |
|
71-85 |
Aa |
Possesses adequate working capital. No caution needed for credit transaction.
It has above average (strong) capability for payment of interest and
principal sums |
Large |
|
56-70 |
A |
Financial & operational base are regarded healthy. General
unfavourable factors will not cause fatal effect. Satisfactory capability for
payment of interest and principal sums |
Fairly Large |
|
41-55 |
Ba |
Overall operation is considered normal. Capable to meet normal
commitments. |
Satisfactory |
|
26-40 |
B |
Capability to overcome financial difficulties seems comparatively below
average. |
Small |
|
11-25 |
Ca |
Adverse factors are apparent. Repayment of interest and principal sums
in default or expected to be in default upon maturity |
Limited with
full security |
|
<10 |
C |
Absolute credit risk exists. Caution needed to be exercised |
Credit not
recommended |
|
-- |
NB |
New Business |
-- |
This report is issued at your request without any
risk and responsibility on the part of MIRA INFORM PRIVATE LIMITED (MIPL) or
its officials.