MIRA INFORM REPORT
|
Report No. : |
303714 |
|
Report Date : |
16.01.2015 |
IDENTIFICATION DETAILS
|
Name : |
RANBAXY LABORATORIES LIMITED |
|
|
|
|
Registered
Office : |
A-41, Industrial Area Phase VIII-A, Sahibzada Ajit Singh Nagar, Mohali
– 160071, Punjab |
|
|
|
|
Country : |
India |
|
|
|
|
Financials (as
on) : |
31.03.2014 |
|
|
|
|
Date of
Incorporation : |
16.06.1961 |
|
|
|
|
Com. Reg. No.: |
16-003747 |
|
|
|
|
Capital
Investment / Paid-up Capital : |
Rs.2116.600
Millions |
|
|
|
|
CIN No.: [Company Identification
No.] |
L24231PB1961PLC003747 |
|
|
|
|
IEC No.: |
Not Available |
|
|
|
|
TAN No.: [Tax Deduction &
Collection Account No.] |
PTLR10986D PTLR11862E |
|
|
|
|
PAN No.: [Permanent Account No.] |
AAACR0127N |
|
|
|
|
Legal Form : |
A Public Limited Liability Company. The Company’s Shares are Listed on
the Stock Exchanges. |
|
|
|
|
Line of Business
: |
Manufacturing and
Trading of formulations, active pharmaceuticals ingredients (API) and intermediate,
generics, drug discovery and consumer health care products and also engaged
in rendering of financial services. |
|
|
|
|
No. of Employees
: |
Not Available |
RATING & COMMENTS
|
MIRA’s Rating : |
A (60) |
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
56-70 |
A |
Financial & operational base are regarded healthy. General
unfavourable factors will not cause fatal effect. Satisfactory capability for
payment of interest and principal sums |
Fairly Large |
|
Maximum Credit Limit : |
USD 31000000 |
|
|
|
|
Status : |
Good |
|
|
|
|
Payment Behaviour : |
Regular |
|
|
|
|
Litigation : |
Exist |
|
|
|
|
Comments : |
Subject is a well-established company having fine track record. The company has reported consecutive losses from its operational
activities. However, the rating also takes into consideration Ranbaxy’s adequate
business and liquidity profile characterised by its established position in
the U.S. generics segment, its leading market presence in the Indian branded
formulations market and a diversified presence among emerging markets. Trade relations are reported as fair. Business is active. Payments are
reported to be regular and as per commitment. The company can be considered good for normal business dealings at
usual trade terms and conditions. |
NOTES :
Any query related to this report can be made
on e-mail : infodept@mirainform.com
while quoting report number, name and date.
ECGC Country Risk Classification List – September 30, 2014
|
Country Name |
Previous Rating (30.06.2014) |
Current Rating (30.09.2014) |
|
India |
A1 |
A1 |
|
Risk Category |
ECGC
Classification |
|
Insignificant |
A1 |
|
Low |
A2 |
|
Moderate |
B1 |
|
High |
B2 |
|
Very High |
C1 |
|
Restricted |
C2 |
|
Off-credit |
D |
EXTERNAL AGENCY RATING
|
Rating Agency Name |
CARE |
|
Rating |
Long Term bank Facilities = AA |
|
Rating Explanation |
High degree of safety and very low credit risk. |
|
Date |
October 14. 2014 |
|
Rating Agency Name |
CARE |
|
Rating |
Short Term Bank Facilities = A1+ |
|
Rating Explanation |
Very strong degree of safety and lowest credit risk. |
|
Date |
October 14. 2014 |
RBI DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available RBI Defaulters’ list.
EPF (Employee Provident Fund) DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available EPF (Employee Provident Fund) Defaulters’ list as of
31-03-2014.
LOCATIONS
|
Registered Office/ Factory 1 : |
A-41, Industrial Area Phase VIII-A, Sahibzada Ajit Singh Nagar, Mohali
– 160071, Punjab, India |
|
Tel. No.: |
91-172-2271450/ 5013655/ 6678666 |
|
Fax No.: |
91-172-2226925/ 5013376 |
|
E-Mail : |
corporate.communications@ranbaxy.com
|
|
Website : |
|
|
|
|
|
Corporate Office : |
Plot No.90, Sector 32, Gurgaon – 122001, Haryana, India |
|
Tel. No.: |
91-124-4135000 |
|
Fax No.: |
91-124-4135001/ 4106490 |
|
E-Mail : |
|
|
|
|
|
Research and |
Plot No.20, Sector - 18, Udyog Vihar Industrial Area, Gurgaon –
122001, Haryana, India |
|
Tel. No.: |
91-124 2342001-10 |
|
Fax No.: |
91-124-2343545 |
|
E-Mail : |
|
|
|
|
|
Factory 2 : |
Village Toansa,
P.O. Railmajra, District Nawansahar – 144533, Punjab, India |
|
|
|
|
Factory 3 : |
Industrial Area 3,
A.B. Road, Dewas – 450 001, Madhya Pradesh, India |
|
|
|
|
Factory 4 : |
Village and P.O.
Ganguwala, Tehsil Paonta Sahib, District Sirmour – 173025, Himachal Pradesh,
India |
|
|
|
|
Factory 5 : |
Village Batamandi,
Tehsil Paonta Sahib, District Sirmour – 173025, Himachal Pradesh, India |
|
|
|
|
Factory 6 : |
Plot No.B-2,
Madkaim Industrial Estate, Ponda, Goa, India |
|
|
|
|
Factory 7 : |
K-5, 6, 7, Ghirongi,
Malanpur, District Bhind – 477116, Madhya Pradesh, India |
|
|
|
|
Factory 8 : |
Plot No.1341 and
1342, EPIP-1, Hill Top Industrial Area, Village-Bhatolikalan (Barotiwala),
Baddi – 174103, Himachal Pradesh, India |
DIRECTORS
As on 31.03.2014
|
Name : |
Dr. Tsutomu Une |
|
Designation : |
Chairman |
|
|
|
|
Name : |
Mr. Akihiro Watanabe |
|
Designation : |
Director |
|
|
|
|
Name : |
Dr. Anthony H. Wild |
|
Designation : |
Director |
|
|
|
|
Name : |
Dr. Kazunori Hirokawa |
|
Designation : |
Director |
|
|
|
|
Name : |
Mr. Percy K. Shroff |
|
Designation : |
Director |
|
|
|
|
Name : |
Mr. Rajesh V. Shah |
|
Designation : |
Director |
|
|
|
|
Name : |
Mr. Takashi Shoda |
|
Designation : |
Director |
KEY EXECUTIVES
|
Name : |
Mr. Arun Sawhney |
|
Designation : |
Chief Executive Officer |
|
|
|
|
Name : |
Mr. Sushil K. Patawari |
|
Designation : |
Company Secretary |
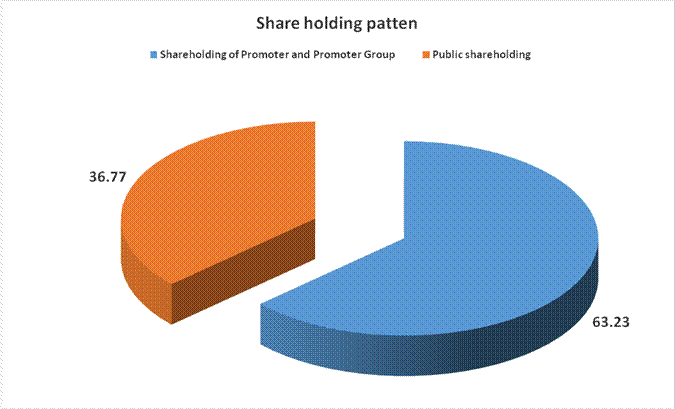
MAJOR SHAREHOLDERS / SHAREHOLDING PATTERN
As on 31.12.2014
|
Category
of Shareholders |
No. of Shares |
Percentage of holding |
|
(A) Shareholding of Promoter and Promoter Group |
|
|
|
(1) Indian |
|
|
|
|
|
|
|
|
268711323 |
63.23 |
|
|
268711323 |
63.23 |
|
Total shareholding of Promoter and Promoter Group (A) |
268711323 |
63.23 |
|
(B) Public Shareholding |
|
|
|
|
|
|
|
|
7942681 |
1.87 |
|
|
225640 |
0.05 |
|
|
29854254 |
7.02 |
|
|
51926151 |
12.22 |
|
|
89948726 |
21.16 |
|
|
|
|
|
|
15547929 |
3.66 |
|
|
|
|
|
Individual shareholders holding nominal share capital up
to Rs.0.100 Million |
39975617 |
9.41 |
|
Individual shareholders holding nominal share capital in
excess of Rs.0.100 Million |
3873704 |
0.91 |
|
|
6943873 |
1.63 |
|
|
2115688 |
0.50 |
|
|
946540 |
0.22 |
|
|
69237 |
0.02 |
|
|
3812408 |
0.90 |
|
|
66341123 |
15.61 |
|
Total Public shareholding (B) |
156289849 |
36.77 |
|
Total (A)+(B) |
425001172 |
100.00 |
|
(C) Shares held by Custodians and against which Depository
Receipts have been issued |
0 |
0.00 |
|
|
0 |
0.00 |
|
|
21728 |
0.00 |
|
|
21728 |
0.00 |
|
Total (A)+(B)+(C) |
425022900 |
100.00 |
BUSINESS DETAILS
|
Line of Business : |
Manufacturing and
Trading of formulations, active pharmaceuticals ingredients (API) and intermediate,
generics, drug discovery and consumer health care products and also engaged
in rendering of financial services. |
||||||||
|
|
|
||||||||
|
Products : |
|
||||||||
|
|
|
||||||||
|
Brand Names : |
Not Available |
||||||||
|
|
|
||||||||
|
Agencies Held : |
Not Available |
||||||||
|
|
|
||||||||
|
Exports : |
Not Available |
||||||||
|
|
|
||||||||
|
Imports : |
Not Available |
||||||||
|
|
|
||||||||
|
Terms : |
Not Available |
PRODUCTION STATUS – NOT AVAILABLE
GENERAL INFORMATION
|
Suppliers : |
|
|||||||||||||||||||||||||||
|
|
|
|||||||||||||||||||||||||||
|
Customers : |
|
|||||||||||||||||||||||||||
|
|
|
|||||||||||||||||||||||||||
|
No. of Employees : |
Not Available |
|||||||||||||||||||||||||||
|
|
|
|||||||||||||||||||||||||||
|
Bankers : |
· Credit Agricole CIB · Royal Bank of Scotland NV · Citibank NA · Deutsche Bank AG · Hong Kong and Shanghai Banking Corporation · Punjab National Bank ·
Standard Chartered Bank |
|||||||||||||||||||||||||||
|
|
|
|||||||||||||||||||||||||||
|
Facilities : |
|
|
Auditors : |
|
|
Name : |
BSR and Company Chartered Accountants |
|
Address : |
Building No.10,
8th Floor, Tower-B, DLF Cyber City, Phase – II, Gurgaon – 122002,
Haryana, India |
|
|
|
|
Holding company (also being the ultimate holding company) |
Daiichi Sankyo Company Limited, Japan |
|
|
|
|
Associate company : |
·
Zenotech Laboratories Limited, India ·
Daiichi Sankyo (Thailand) Limited,
Thailand [formerly known as Ranbaxy Unichem Co. Limited, Thailand, (an
associate w.e.f. 1 October 2013)] $ |
|
|
|
|
Subsidiaries including step down subsidiaries : |
·
Vidyut Investments Limited, India ·
Ranbaxy Signature LLC, USA ·
Be-Tabs Investments (Proprietary)
Limited, South Africa ·
S.C. Terapia Distributie S.R.L.,
Romania (Merged with S.C. Terapia S.A., Romania w.e.f. 7 February 2012) ·
Office Pharmaceutique Industriel et
Hospitalier SARL, France ·
Ranbaxy Holdings (UK) Limited.,
United Kingdom (‘U.K.’) ·
Ranbaxy Do Brazil Ltda., Brazil
(Liquidated on 7 November 2012) ·
Ranbaxy Pharma AB, Sweden (Liquidated
on 2 January 2014) ·
Ranbaxy GmbH, Germany (from 9
November 2012) ·
Ranbaxy Laboratories Inc., USA ·
Ranbaxy (Thailand) Co., Limited (from
20 February 2013) |
|
|
|
|
Fellow subsidiaries : |
·
Daiichi Sankyo India Pharma Private
Limited, India ·
Daiichi Sankyo Development Limited,
U.K. ·
Daiichi Sankyo Propharma Co.,
Limited., Japan ·
Daiichi Sankyo Espha Co. Limited.,
Japan ·
Daiichi Sankyo, Inc., USA ·
Daiichi Sankyo Venezuela S.A.,
Venezuela ·
Daiichi Sankyo Chemical Pharma Co.,
Limited., Japan ·
Ranbaxy Mexico S.A.de C.V., Mexico ·
Daiichi Sankyo Europe GmbH, Germany |
|
|
|
|
Subsidiaries including step down subsidiaries /
partnership firms : |
·
Ranbaxy Drugs and Chemicals Company,
India (Company with unlimited liability) # ·
Solus Pharmaceuticals Limited, India
# ·
Ranbaxy SEZ Limited, India # ·
Rexcel Pharmaceuticals Limited, India
# ·
Ranbaxy Life Sciences Research
Limited, India # ·
Gufic Pharma Limited, India ·
Ranbaxy Drugs Limited, India ·
Solrex Pharmaceuticals Company, India
(a Partnership firm) ·
Ranbaxy (Hong Kong) Limited, Hong
Kong @ ·
Ranbaxy Inc., USA ·
Ranbaxy USA, Inc., USA ·
Ranbaxy Egypt (L.L.C.), Egypt ·
Ranbaxy Farmaceutica Ltda., Brazil ·
Ranbaxy PRP (Peru) SAC, Peru ·
Ranbaxy Australia Proprietary
Limited., Australia ·
Daiichi Sankyo (Thailand) Limited,
Thailand [formerly known as Ranbaxy Unichem Co. Limited., Thailand ·
(subsidiary upto 30 September 2013)]
$ ·
Ranbaxy Italia S.p.A, Italy ·
Ranbaxy Malaysia Sdn. Bhd., Malaysia ·
Ranbaxy Poland S.P. Zoo, Poland ·
Ranbaxy Nigeria Limited, Nigeria ·
Ranbaxy Europe Limited, U.K. ·
Ranbaxy (UK) Limited, U.K. ·
Basics GmbH, Germany ·
ZAO Ranbaxy, Russia ·
S.C. Terapia S.A., Romania ·
Ranbaxy Pharmaceuticals, Inc., USA ·
Ohm Laboratories, Inc., USA ·
Ranbaxy Ireland Limited, Ireland ·
Ranbaxy South Africa Proprietary
Limited, South Africa ·
Laboratorios Ranbaxy S.L., Spain ·
Ranbaxy Pharmacie Generiques SAS,
France ·
Ranbaxy Pharmaceuticals Canada Inc.,
Canada ·
Sonke Pharmaceuticals (Proprietary)
Limited, South Africa ·
Ranbaxy Portugal - Com E Desenvolv
DeProd Farmaceuticos Unipessoal Lda, Portugal ·
Ranbaxy Belgium N.V., Belgium ·
Be-Tabs Pharmaceuticals (Proprietary)
Limited, South Africa ·
Rexcel Egypt (L.L.C.), Egypt ·
Ranbaxy Morocco LLC, Morocco ·
Ranbaxy Pharmaceuticals Ukraine LLC,
Ukraine (from 13 June 2012) ·
Ranbaxy (Netherlands) B.V., The
Netherlands #
Refer to note 14 for details on merger of these subsidiaries with RDL @
Refer to note 18 for details of liquidation of the entity during the current
period $
Refer to note 14 for details on business integration with Daiichi Sankyo
(Thailand) Limited. |
CAPITAL STRUCTURE
As on 31.03.2014
Authorised Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
598000000 |
Equity Shares |
Rs.5/- each |
Rs.2990.000 Millions |
|
100000 |
Cumulative Preference Shares |
Rs.100/- each |
Rs.10.000 Millions |
|
|
|
|
|
|
|
Total |
|
Rs.3000.000
Millions |
Issued, Subscribed & Paid-up Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
423779063 |
Equity Shares |
Rs.5/- each |
Rs.2118.900 Millions |
|
459602 |
Equity Shares |
Rs.5/- each |
Rs.2.300 Millions |
|
|
|
|
|
|
|
Total |
|
Rs.2116.600
Millions |
a.
Rights,
preferences and restrictions attached to shares
As per the
Memorandum of Association, the Company’s authorised share capital consists of
equity shares and preference shares.
All equity shares
rank equally with regard to dividends and share in the Company’s residual
assets. The equity shares are entitled to receive dividend as declared from
time to time. The voting rights of an equity shareholder on show of hand or
through proxy shall be in proportion to his share of the paid-up equity capital
of the Company. On winding up of the Company, the holders of equity shares will
be entitled to receive the residual assets of the Company, remaining after
distribution of all preferential amounts in proportion to the number of equity shares
held.
Preference shares
shall be entitled for such rate of dividend as may be decided by the Directors
of the Company at the time of issue of such shares and shall rank in priority
to the equity shares including arrears, if any, in the event of the winding up
of the Company, but shall not be entitled to any further participation in the
profits or surplus assets of the Company. Preference shares are entitled to one
vote per share at meetings of the Company only in respect of resolutions
directly affecting their rights. However, a cumulative preference shareholder
acquires voting rights on par with an equity shareholder if the dividend on
preference shares has remained unpaid for a period of not less than two years.
b.
Reconciliation of equity shares outstanding at
the commencement and at the end of the period
|
Equity Shares |
Number
of Shares |
Rs.
In Millions |
|
At the commencement of the period |
422913803 |
2114.570 |
|
Add: Shares issued
on exercise of employee stock options by the Company/ ESOP Trust |
405658 |
2.030 |
|
Add: Shares
issued to the Trust under Employees Stock Option Plan 2011 - (‘ESOP 2011’) |
-- |
-- |
|
At the end of
the period |
423319461 |
2116.600 |
c.
Equity shares held by holding/ ultimate holding
company
|
Equity Shares |
Number
of Shares |
Rs.
In Millions |
|
Daiichi Sankyo
Company Limited, Japan (Daiichi Sankyo), the holding company, also being the
ultimate holding company |
268711323 |
1343.560 |
d.
Particulars of shareholders holding more than 5%
shares of issued, subscribed and paid-up capital of equity shares
|
Name of Shareholder |
Number
of Shares |
% holding |
|
Daiichi Sankyo |
268711323 |
63.41 |
|
Life Insurance Corporation of India, India |
25494745 |
6.02 |
e.
Equity shares reserved for issue under employee
stock options:
Number of stock
options against which equity shares to be issued by the Company upon vesting
and exercise of those stock options by the option holders as per the relevant
scheme.
f.
During the current period, the Company has issued
600,000 (previous year 440,000) equity shares of Rs. 5 (previous year Rs. 5)
each issued for cash at par to the ESOP Trust, set up to administer the ESOP -
2011. Out of the total equity shares issued to the ESOP Trust, 666,636
(previous year 238,762) equity shares have been allotted by the ESOP Trust to
the respective employees upon exercise of stock options from time to time under
ESOP - 2011. As at 31 March 2014, 459,602 (previous year 526,238) equity shares
are pending to be allotted to the employees upon exercise of stock options
Pursuant to change
in accounting policy (Refer to note 2 k), as at 31 March 2014, the Company has
shown share capital net of 459,602 equity shares amounting to Rs. 2.30 held by
the ESOP Trust. The Company has not restated the comparative figures. The
movement of shares held by the ESOP trust is as follows:-
|
Equity Shares |
Number
of Shares |
Rs.
In Millions |
|
At the commencement of the period |
526238 |
2.63 |
|
Add: Shares allotted to the ESOP Trust |
600000 |
3.00 |
|
Less: Shares issued on exercise of employee stock options by the ESOP
Trust |
666636 |
3.33 |
|
At the end of the period |
459602 |
2.30 |
g.
During the five years immediately preceding the
current period and previous year, neither any bonus shares or shares issued for
consideration other than cash that have been issued nor any shares that have
been bought back.
h.
Issued, subscribed and paid-up share capital
includes 6,294,081 (previous year 8,963,108) Global Depository Shares (GDSs)
representing 6,294,081 (previous year 8,963,108) equity shares of Rs. 5
(previous year Rs. 5) each constituting 1.49% (previous year 2.12%) of the
issued, subscribed and paid-up share capital of the Company.
FINANCIAL DATA
[all figures are
in Rupees Millions]
ABRIDGED BALANCE
SHEET
|
SOURCES OF FUNDS |
31.03.2014 (15 Months) |
31.12.2012 (12 Months) |
31.12.2011 (12 Months) |
|
|
|
|
|
(1)Shareholders' Funds |
|
|
|
|
(a) Share Capital |
2116.600 |
2114.570 |
2110.000 |
|
(b) Reserves & Surplus |
8848.270 |
17095.100 |
17131.640 |
|
(c) Money received against share warrants |
0.000 |
0.000 |
0.000 |
|
|
|
|
|
|
(2) Share Application money pending allotment |
5.030 |
11.100 |
6.660 |
|
Total
Shareholders’ Funds (1) + (2) |
10969.900 |
19220.770 |
19248.300 |
|
|
|
|
|
|
(3) Non-Current Liabilities |
|
|
|
|
(a) long-term borrowings |
24721.880 |
19568.100 |
9524.110 |
|
(b) Deferred tax liabilities (Net) |
0.000 |
0.000 |
0.000 |
|
(c) Other long term liabilities |
3719.440 |
10363.480 |
15977.190 |
|
(d) long-term provisions |
1314.540 |
2739.040 |
2297.910 |
|
Total Non-current
Liabilities (3) |
29755.860 |
32670.620 |
27799.210 |
|
|
|
|
|
|
(4) Current Liabilities |
|
|
|
|
(a) Short term borrowings |
35188.770 |
28067.950 |
29310.020 |
|
(b) Trade payables |
9751.860 |
8588.110 |
9856.370 |
|
(c) Other current liabilities |
43495.900 |
13320.780 |
30004.520 |
|
(d) Short-term provisions |
1888.080 |
27831.110 |
26990.830 |
|
Total Current
Liabilities (4) |
90324.610 |
77807.950 |
96161.740 |
|
|
|
|
|
|
TOTAL |
131050.370 |
129699.340 |
143209.250 |
|
|
|
|
|
|
|
|
|
|
(1) Non-current assets |
|
|
|
|
(a) Fixed Assets |
|
|
|
|
(i) Tangible assets |
19460.460 |
19308.430 |
17882.550 |
|
(ii) Intangible Assets |
660.880 |
626.850 |
787.420 |
|
(iii) Capital work-in-progress |
1791.800 |
1465.370 |
2004.930 |
|
(iv) Intangible assets under development |
44.530 |
130.590 |
86.310 |
|
(b) Non-current Investments |
40789.890 |
31281.370 |
34081.470 |
|
(c) Deferred tax assets (net) |
0.000 |
0.000 |
0.000 |
|
(d) Long-term Loan and Advances |
10779.890 |
10107.120 |
9412.340 |
|
(e) Other Non-current assets |
1741.140 |
215.700 |
0.860 |
|
Total Non-Current
Assets |
75268.590 |
63135.430 |
64255.880 |
|
|
|
|
|
|
(2) Current assets |
|
|
|
|
(a) Current investments |
18.950 |
30.320 |
26.460 |
|
(b) Inventories |
16951.440 |
17318.390 |
16552.310 |
|
(c) Trade receivables |
12374.650 |
14358.880 |
36828.190 |
|
(d) Cash and cash equivalents |
7905.720 |
28347.730 |
19379.530 |
|
(e) Short-term loans and advances |
12732.860 |
5041.480 |
3399.750 |
|
(f) Other current assets |
5798.160 |
1467.110 |
2767.130 |
|
Total Current Assets |
55781.780 |
66563.910 |
78953.370 |
|
|
|
|
|
|
TOTAL |
131050.370 |
129699.340 |
143209.250 |
PROFIT & LOSS
ACCOUNT
|
|
PARTICULARS |
31.03.2014 (15 Months) |
31.12.2012 (12 Months) |
31.12.2011 (12 Months) |
|
|
|
SALES |
|
|
|
|
|
|
|
Revenue from operations |
68649.370 |
63035.440 |
77990.570 |
|
|
|
Other Income |
7848.200 |
2571.630 |
2226.550 |
|
|
|
TOTAL (A) |
76497.570 |
65607.070 |
80217.120 |
|
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
|
|
Cost of materials consumed |
20653.280 |
15286.610 |
17849.130 |
|
|
|
Purchases of stock-in-trade |
9826.430 |
8090.010 |
6367.310 |
|
|
|
Change in
inventories of finished goods, work-in-progress and stock-in-trade |
(1751.220) |
(492.450) |
(1357.220) |
|
|
|
Employee
benefits expense |
12747.280 |
10195.890 |
8607.110 |
|
|
|
Other expenses |
30418.440 |
25526.160 |
35783.820 |
|
|
|
Exceptional items: |
|
|
|
|
|
|
Profit on sale of intellectual property rights |
(4327.690) |
0.000 |
0.000 |
|
|
|
Settlement provision reversal |
(1458.050) |
0.000 |
26480.000 |
|
|
|
Provision in respect of non-current investment in a
subsidiary |
3050.960 |
1030.000 |
0.000 |
|
|
|
Provision
for other-than-temporary diminution in value of noncurrent investment in an
associate |
713.110 |
0.000 |
0.000 |
|
|
|
Inventory
provision/ write off and other costs |
3557.920 |
|
|
|
|
|
Loss
on foreign currency option derivatives, net (other than on loans) |
3279.160 |
412.050 |
11242.850 |
|
|
|
Product
recall expenses |
0.000 |
2370.200 |
0.000 |
|
|
|
TOTAL (B) |
76709.620 |
62418.470 |
104973.000 |
|
|
|
|
|
|
|
|
Less |
PROFIT/
(LOSS) BEFORE INTEREST, TAX, DEPRECIATION AND AMORTISATION (A-B) (C) |
(212.050) |
3188.600 |
(24755.880) |
|
|
|
|
|
|
|
|
|
Less |
FINANCIAL
EXPENSES (D) |
5470.480 |
2969.820 |
2989.990 |
|
|
|
|
|
|
|
|
|
|
PROFIT/
(LOSS) BEFORE TAX, DEPRECIATION AND AMORTISATION (C-D) (E) |
(5682.530) |
218.780 |
(27745.870) |
|
|
|
|
|
|
|
|
|
Less |
DEPRECIATION/
AMORTISATION (F) |
2801.720 |
1861.610 |
2740.830 |
|
|
|
|
|
|
|
|
|
|
PROFIT/ (LOSS)
BEFORE TAX (E-F) (G) |
(8484.250) |
(1642.830) |
(30486.700) |
|
|
|
|
|
|
|
|
|
Less |
TAX (H) |
305.700 |
(19.440) |
33.790 |
|
|
|
|
|
|
|
|
|
|
PROFIT/ (LOSS)
AFTER TAX (G-H) (I) |
(8789.950) |
(1623.390) |
(30520.490) |
|
|
|
|
|
|
|
|
|
Add |
PREVIOUS
YEARS’ BALANCE BROUGHT FORWARD |
(25312.700) |
(23689.310) |
6828.680 |
|
|
|
|
|
|
|
|
|
Less |
APPROPRIATIONS |
|
|
|
|
|
|
|
Proposed dividend |
-- |
-- |
0.650 |
|
|
|
Tax on proposed dividend |
-- |
-- |
(3.150) |
|
|
BALANCE CARRIED TO
THE B/S |
(34102.650) |
(25312.700) |
(23689.310) |
|
|
|
|
|
|
|
|
|
|
EARNINGS IN
FOREIGN CURRENCY |
|
|
|
|
|
|
|
Exports on F.O.B. basis (excluding sales made to customers located in
Nepal) |
36621.780 |
37856.870 |
54114.790 |
|
|
|
Royalty, milestone, technical know-how and product development |
503.390 |
538.170 |
613.160 |
|
|
|
Interest |
177.990 |
104.540 |
131.180 |
|
|
|
Dividend |
6121.320 |
10.040 |
11.830 |
|
|
|
Others (freight/
insurance recoveries and other operating revenues) |
1728.700 |
1006.100 |
944.200 |
|
|
|
Profit on sale
of intellectual property rights |
4327.690 |
0.000 |
0.000 |
|
|
|
Settlement
provision reversal |
1458.050 |
0.000 |
0.000 |
|
|
TOTAL EARNINGS |
50938.920 |
39515.720 |
55815.160 |
|
|
|
|
|
|
|
|
|
|
IMPORTS |
|
|
|
|
|
|
|
Raw Materials (including packing materials) |
8567.040 |
7179.750 |
7592.690 |
|
|
|
Components, stores and spare parts |
123.400 |
145.750 |
134.290 |
|
|
|
Capital Goods |
457.250 |
472.490 |
560.780 |
|
|
TOTAL IMPORTS |
9147.690 |
7797.990 |
8287.760 |
|
|
|
|
|
|
|
|
|
|
Earnings/ (Loss)
Per Share (Rs.) |
(20.79) |
(3.85) |
(72.42) |
|
QUARTERLY RESULTS
(Rs.
In Millions)
|
Particulars |
|
30.06.2014 (Unaudited) |
30.09.2014 (Unaudited) |
|
|
|
1st
Quarter |
2nd Quarter |
|
Net sales |
|
21786.300 |
13189.100 |
|
Total Expenditure |
|
13212.800 |
15392.500 |
|
PBIDT (Excluding Other Income) |
|
8573.500 |
(2203.300) |
|
Other income |
|
257.600 |
200.200 |
|
Operating Profit |
|
8831.100 |
(2003.200) |
|
Interest |
|
728.800 |
721.600 |
|
Exceptional Items |
|
197.700 |
(356.300) |
|
PBDT |
|
8300.100 |
(3081.100) |
|
Depreciation |
|
656.500 |
601.500 |
|
Profit Before Tax |
|
7643.600 |
(3682.600) |
|
Tax |
|
92.400 |
0.000 |
|
Profit after tax |
|
7551.200 |
(3682.600) |
KEY RATIOS
|
PARTICULARS |
|
31.03.2014 (15 Months) |
31.12.2012 (12 Months) |
31.12.2011 (12 Months) |
|
Net Profit Margin (PAT / Sales) |
(%) |
(12.80) |
(2.58) |
(39.13) |
|
|
|
|
|
|
|
Operating Profit Margin (PBDIT/Sales) |
(%) |
(0.31) |
5.06 |
(31.74) |
|
|
|
|
|
|
|
Return on Total Assets (PBT/Total Assets} |
(%) |
(9.59) |
(1.70) |
(28.48) |
|
|
|
|
|
|
|
Return on Investment (ROI) (PBT/Networth) |
|
(0.77) |
(0.09) |
(1.58) |
|
|
|
|
|
|
|
Debt Equity Ratio (Total Debt /Networth) |
|
5.46 |
2.48 |
2.02 |
|
|
|
|
|
|
|
Current Ratio (Current Asset/Current Liability) |
|
0.62 |
0.86 |
0.82 |
FINANCIAL ANALYSIS
[all figures are
in Rupees Millions]
DEBT EQUITY RATIO
|
Particular |
31.12.2011
(12
Months) |
31.12.2012
(12
Months) |
31.03.2014
(15
Months) |
|
|
Rs. In
Millions |
Rs.
In Millions |
Rs.
In Millions |
|
Share Capital |
2110.000 |
2114.570 |
2116.600 |
|
Reserves & Surplus |
17131.640 |
17095.100 |
8848.270 |
|
Share Application money
pending allotment |
6.660 |
11.100 |
5.030 |
|
Net
worth |
19248.300 |
19220.770 |
10969.900 |
|
|
|
|
|
|
long-term borrowings |
9524.110 |
19568.100 |
24721.880 |
|
Short term borrowings |
29310.020 |
28067.950 |
35188.770 |
|
Total
borrowings |
38834.130 |
47636.050 |
59910.650 |
|
Debt/Equity
ratio |
2.018 |
2.478 |
5.461 |

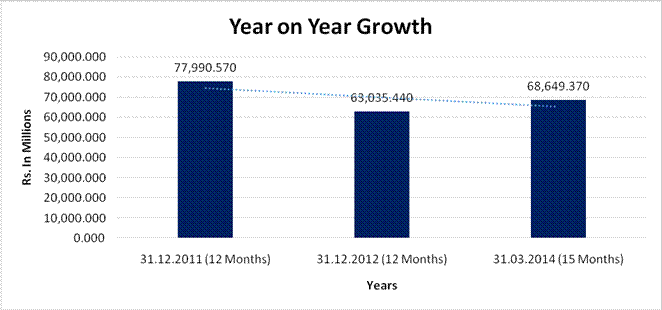
YEAR-ON-YEAR GROWTH
|
Year
on Year Growth |
31.12.2011
(12
Months) |
31.12.2012
(12
Months) |
31.03.2014
(15
Months) |
|
|
Rs.
In Millions |
Rs.
In Millions |
Rs. In
Millions |
|
Sales |
77,990.570 |
63,035.440 |
68,649.370 |
|
|
|
(19.176) |
8.906 |
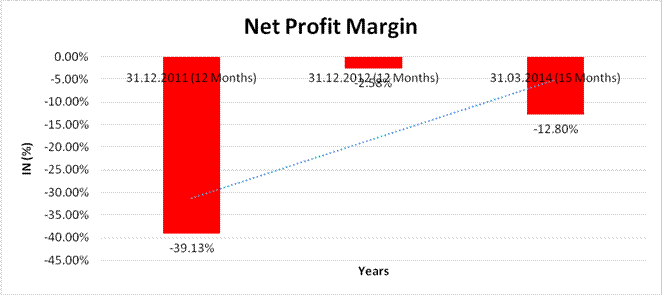
NET PROFIT MARGIN
|
Net
Profit Margin |
31.12.2011
(12
Months) |
31.12.2012
(12
Months) |
31.03.2014
(15
Months) |
|
|
Rs.
In Millions |
Rs.
In Millions |
Rs.
In Millions |
|
Sales
|
77,990.570 |
63,035.440 |
68,649.370 |
|
Profit |
(30,520.490) |
(1,623.390) |
(8,789.950) |
|
|
(39.13%) |
(2.58%) |
(12.80%) |
LOCAL AGENCY FURTHER INFORMATION
|
Sr. No. |
Check List by Info Agents |
Available in
Report (Yes / No) |
|
1] |
Year of Establishment |
Yes |
|
2] |
Locality of the firm |
Yes |
|
3] |
Constitutions of the firm |
Yes |
|
4] |
Premises details |
No |
|
5] |
Type of Business |
Yes |
|
6] |
Line of Business |
Yes |
|
7] |
Promoter's background |
No |
|
8] |
No. of employees |
Yes |
|
9] |
Name of person contacted |
No |
|
10] |
Designation of contact
person |
No |
|
11] |
Turnover of firm for last
three years |
Yes |
|
12] |
Profitability for last
three years |
Yes |
|
13] |
Reasons for variation
<> 20% |
----------- |
|
14] |
Estimation for coming
financial year |
No |
|
15] |
Capital in the business |
Yes |
|
16] |
Details of sister
concerns |
Yes |
|
17] |
Major suppliers |
No |
|
18] |
Major customers |
No |
|
19] |
Payments terms |
No |
|
20] |
Export / Import details
(if applicable) |
No |
|
21] |
Market information |
---------- |
|
22] |
Litigations that the firm
/ promoter involved in |
Yes |
|
23] |
Banking Details |
Yes |
|
24] |
Banking facility details |
Yes |
|
25] |
Conduct of the banking
account |
---------- |
|
26] |
Buyer visit details |
---------- |
|
27] |
Financials, if provided |
Yes |
|
28] |
Incorporation details, if
applicable |
Yes |
|
29] |
Last accounts filed at
ROC |
Yes |
|
30] |
Major Shareholders, if
available |
Yes |
|
31] |
Date of Birth of
Proprietor/Partner/Director, if available |
No |
|
32] |
PAN of
Proprietor/Partner/Director, if available |
No |
|
33] |
Voter ID No of
Proprietor/Partner/Director, if available |
No |
|
34] |
External Agency Rating,
if available |
Yes |
LITIGATION
DETAILS:
|
High Court of Punjab and Haryana Chandigarh |
||||||
|
Case Details For
Case CA-117-2011 |
||||||
|
Diary Number |
609725 |
District |
Other-Districts |
|||
|
Category |
- |
Main Case Detail |
CP-30-2011 |
|||
|
Party Detail |
M/S SUPRIYA PHARMACEUTICALS LIMITED
V/S M/S RANBAXY LABORATORIES LIMITED |
|||||
|
Advocate Name |
AASHISH CHOPRA |
List Type |
ORDINARY |
|||
|
Status |
PENDING |
|||||
|
Case Listing
Details |
||||||
|
No Listing Data Available |
||||||
|
Judgment Details
For Case: CA-117-2011 Party Detail: M/S SUPRIYA PHARMACEUTICALS
LIMITED V/S M/S RANBAXY LABORATORIES LIMITED |
||||||
|
Order Date |
Order Type |
Bench |
Judgment Link |
|||
|
No Judgment/Order Found. |
||||||
|
Designed and Developed by National Informatics
Centre Contents
Published and Managed by Punjab and Haryana High Court, Chandigarh. Disclaimer |
||||||
UNSECURED LOAN:
|
Particulars |
31.03.2014 (15
Months) Rs.
In Millions |
31.12.2012 (12
Months) Rs.
In Millions |
|
Long Term
Borrowings |
|
|
|
External commercial borrowings |
17210.880 |
12046.100 |
|
Other |
2500.000 |
2500.000 |
|
From other party |
11.000 |
22.000 |
|
|
|
|
|
Short Term
Borrowings |
|
|
|
From Banks |
1022.700 |
0.000 |
|
Other loans advances from banks |
21298.540 |
20626.150 |
|
Commercial Paper |
7750.000 |
3000.000 |
|
|
|
|
|
Total |
49793.120 |
38194.250 |
INDEX OF CHARGE:
|
Sr. No. |
Charge ID |
Date of Charge
Creation/Modification |
Charge amount
secured |
Charge Holder |
Address |
Service Request Number
(SRN) |
|
1 |
10406999 |
18/02/2013 |
5,000,000,000.00 |
AXIS
TRUSTEE SERVICES LIMITED |
AXIS
HOUSE, 2ND FLR, BOMBAY DYEING MILLS COMPOUND, PANDURANG BUDHKAR MARG, WORLI,,
MUMBAI, MAHARASHTRA - 400025, INDIA |
B68476811 |
|
2 |
80052154 |
30/12/2005 |
728,500,000.00 |
CITI
BANK N.A. |
JEEVAN
VIHAR BUILDING PARLIAMENT STREET, NEW DELHI - 110001, INDIA |
- |
|
3 |
90172173 |
24/10/2003 |
3,000,000,000.00 |
STATE
BANK OF INDIA |
JEEVAN
VYAPAR BHAVAN, 11 TH & 12TH FLOOR 1 TOLSTOY MARG, NEW DELHI- 110001,
INDIA |
- |
|
4 |
90169791 |
09/04/2003 |
364,000,000.00 |
CREDIT
LYONNAIS |
6TH
FLOOR 15 KASTURBA MARG, NEW DELHI, DELHI - 110001, INDIA |
- |
|
5 |
90169771 |
17/02/2003 |
301,700,000.00 |
DEUTCHE
BANK |
BRANCH
OFFICE15-17 TOLSTOY, HOUSE TOLSTOY MARG, |
- |
|
6 |
90169680 |
03/07/2002 |
484,000,000.00 |
STANDARD
CHARTERED BANK |
PARLIAMENT
STREET, NEW DELHI, DELHI, INDIA |
- |
|
7 |
80052153 |
24/10/2001 |
484,000,000.00 |
THE HONGKONG
AND SHANGHAI BANKING CORPORATION LTD |
ECE
HOUSE K G MARG, NEW DELHI, DELHI - 110001, INDIA |
- |
|
8 |
90169551 |
12/03/2003
* |
728,500,000.00 |
ABN
AMRO BNAK N V |
DLF
CENTRE, PARLIAMENT STREET, NEW DELHI, DELHI - |
- |
|
9 |
90169517 |
30/03/2001 |
2,122,500,000.00 |
PUNJAB
NATIONAL BAN K |
7
BHIKAJI CAMA PLACE AFRICA, AVENUE, NEW DELHI, DELHI - 110066, INDIA |
- |
CHANGE
IN FINANCIAL YEAR
The
Board of Directors of the Company approved change in the financial year of the
Company from January-December to April-March effective April 1, 2014. In view
of this, the current financial year is for a period of 15 months i.e. January
1, 2013 to March 31, 2014.
OPERATIONS
The
Company continued to be among the top pharmaceutical companies from India with
consolidated global sales of Rs.130403.240 million for the period of fifteen
months ended March 31, 2014. Profit before exceptional items, tax, share in
loss of associates (net) and minority interest stood at Rs.1225.750 million.
However, the Company incurred a loss of Rs.10852.520 million primarily due to
provision for diminution in the value of investments, impairment of goodwill,
stock provision/write off due to inclusion of Mohali and Toansa plants to
certain terms of the Consent Decree by the US FDA and loss on foreign currency
option derivatives.
During
the period, in terms of the settlement with the US DOJ, the Group paid the
settlement amount of US$ 515.40 million (including interest expense and other
related cost) towards resolution of civil and criminal allegations. During the
period, US FDA issued import alerts for the Company’s plants at Mohali and
Toansa and advised that both these plants will be subject to certain terms of
the Consent Decree earlier entered into by the Company. The Company
proactively, temporarily stopped API supplies from Toansa and Dewas facilities
to the rest of the world pending further internal review. This voluntary
decision was taken as a precautionary measure and out of abundant caution to
better allow the Company to assess and review the processes and controls at
these sites.
In
March 2014, US DOJ, United States Attorney’s Office for the District of New
Jersey has issued an administrative subpoena seeking information primarily related
to the Company’s API manufacturing facility at Toansa. The Company is fully
cooperating with this information request.
The
Company is continuing its focus for improving margins through innovative
product development, better product mix, emphasis on branded products and
control on cost. Significant measures have been taken for simplification of
processes and structures which will result in improvement in productivity and
efficiency across the organisation.
SCHEME
OF ARRANGEMENT BETWEEN THE COMPANY AND SUN PHARMACEUTICAL INDUSTRIES LIMITED
The
Board of Directors at its meeting held on April 6, 2014 approved the Scheme of
Arrangement for merger of the Company with Sun Pharmaceutical Industries Ltd.
(SPIL) with an Appointed Date of April 1, 2014 at a Share Exchange Ratio of 4
Equity Shares of SPIL of Re.1 each fully paid-up for every 5 Equity Shares of
the Company of Rs.5 each fully paid-up subject to requisite regulatory
approvals in India and overseas as well as the approval of shareholders, creditors
and the Courts in India. The transaction will be beneficial to all the
stakeholders of the Company. Post-merger, the combined entity is expected to
have a leadership position in the Indian Pharmaceutical Market with about 9.2%
market share and No.1 Indian Pharma Company in the USA market, with more than
$2 billion in sales. The combined entity will have operations in 65 countries
and 47 manufacturing facilities across the globe.
MANAGEMENT DISCUSSION AND ANALYSIS
REPORT
GLOBAL
INDUSTRY STRUCTURE AND DEVELOPMENTS
The
global expenditure on medicines is expected to exceed US$ 1 trillion for the
first time in 2014 and reach almost US$ 1.2 trillion in 2017, up from US$ 956
billion in 2011. The market is forecasted to grow at a compounded annual growth
rate (CAGR) of 3-6% over 2013-17. Of this increase, over 70% is expected to
come from Pharmerging1 markets, which are expected to grow at 12-15%, while the
rest of the growth is expected from the Developed3 markets, which could grow at
1-4% per annum. Sales in the largest pharmaceutical market, i.e., the United
States of America (USA), is expected to be US$ 350-380 billion by 2017, with
growth in the range of 1-4% per annum. Sales in Japan, the second largest
pharmaceutical market is expected to be in the range of US$ 105-110 billion by
2017, reflecting a CAGR of 2-5% during the period 2013-17. The top 5 European
markets are expected to grow at a CAGR of 0-3% for the period 2013-17, as
compared to 2% CAGR for 2008-12, to achieve sales in the range of US$ 145-160
billion. Pharmerging market sales, with their higher rate of growth, are
expected to match or slightly exceed those in the USA pharmaceutical market by
2017, in value terms.
The
global pharmaceutical industry for patented products continues to remain fragmented
and fiercely competitive as it faces increased genericisation. The generics
industry, on the other hand, has the opportunity to capitalise on the products
going off-patent in the coming years. In its attempt to cope with these
challenges, the industry has witnessed consolidation; this may happen across
the global market, especially in the generics space. The larger markets2 of the
USA, Germany, France, Italy, the UK, Spain, Japan and China are expected to
have a share of 67% of the world pharmaceuticals market in 2017 and to
contribute 59% of the global growth in the 5 year period to 2017.
The
Pharmerging markets are expected to grow at a significantly higher rate than
the rest of the world and are expected to account for over 30% of the global
pharmaceutical spending by 2017.
UNAUDITED FINANCIAL RESULTS FOR QUARTER ENDED 30.09.2014
(Rs. In Millions)
|
Particulars |
Quarter Ended (Unaudited) |
Half Year Ended (Unaudited) |
||
|
|
30.09.2014 |
30.06.2014 |
30.09.2014 |
|
|
1. Income
from operations |
|
|
|
|
|
a) Net sales/ Income from operation (net of excise duty) |
1272.687 |
21169.800 |
3389.667 |
|
|
b) Other operating income |
46.225 |
616.450 |
97.642 |
|
|
Total
income from Operations(net) |
1318.912 |
21786.250 |
3487.309 |
|
|
2.Expenditure |
|
|
|
|
|
a) Cost of material consumed |
335.938 |
2807.330 |
616.671 |
|
|
b) Purchases of stock in trade |
227.006 |
1842.680 |
411.274 |
|
|
c) Changes in inventories of finished goods,
work-in-progress and stock-in-trade |
101.169 |
1027.390 |
203.908 |
|
|
d) Employees benefit expenses |
279.668 |
2547.380 |
534.406 |
|
|
e) Depreciation and amortization expenses |
60.152 |
656.450 |
125.797 |
|
|
f) Other expenditure |
478.536 |
4273.180 |
895.626 |
|
|
g) Foreign exchange loss/(Gain) |
52.264 |
24.440 |
54.708 |
|
|
Total expenses |
1534.733 |
13178.850 |
2842.390 |
|
|
3. Profit from operations before other income and
financial costs |
(215.821) |
8607.400 |
644.949 |
|
|
4. Other income |
20.019 |
257.640 |
45.783 |
|
|
5. Profit from ordinary activities before finance costs |
(195.802) |
8865.040 |
690.702 |
|
|
6. Finance costs |
136.823 |
1419.140 |
278.737 |
|
|
7. Net profit/(loss) from ordinary activities
after finance costs but before exceptional items |
(332.625) |
7445.900 |
411.965 |
|
|
8.
Exceptional item |
(35.632) |
197.700 |
(15.862) |
|
|
9. Profit from ordinary activities before tax
Expense: |
(368.257) |
7643.600 |
396.103 |
|
|
10.Tax expenses |
-- |
92.370 |
9.237 |
|
|
11.Net
Profit / (Loss) from ordinary activities after tax (9-10) |
(368.257) |
7551.230 |
386.866 |
|
|
12.Extraordinary Items (net of tax expense) |
0.000 |
0.000 |
0.000 |
|
|
13.Net Profit / (Loss) for the period (11 -12) |
(368.257) |
7551.230 |
386.866 |
|
|
14.Paid-up
equity share capital (Nominal value Rs.10/- per share) |
211.949 |
2117.750 |
211.949 |
|
|
15. Reserve excluding Revaluation
Reserves as per balance sheet of previous accounting year |
|
|
|
|
|
16.i) Earnings per share (before extraordinary
items) of Rs.10/- each) (not annualised): |
|
|
|
|
|
(a) Basic and diluted |
(8.69) |
17.83 |
9.13 |
|
|
ii) Earnings per share (after extraordinary items) |
|
|
|
|
|
(a) Basic and diluted |
(8.69) |
17.80 |
9.11 |
|
|
Debt Service Coverage Ratio |
|
|
0.40 |
|
|
Interest Service Coverage Ratio |
|
|
2.42 |
|
|
|
|
|
|
|
|
A. Particulars of shareholding |
|
|
|
|
|
1. Public Shareholding |
|
|
|
|
|
- Number of shares |
149313910 |
144852564 |
149313910 |
|
|
- Percentage of shareholding |
35.22 |
34.18 |
35.22 |
|
|
2. Promoters and Promoters group Shareholding- |
|
|
|
|
|
a) Pledged /Encumbered |
|
|
|
|
|
Number of shares |
-- |
-- |
-- |
|
|
Percentage of shares (as a % of total shareholding of the
promoter and promoter group) |
-- |
-- |
-- |
|
|
Percentage of shares (as a % of total share capital of the
company) |
-- |
-- |
-- |
|
|
|
|
|
|
|
|
b) Non Encumbered |
|
|
|
|
|
Number of shares |
268711323 |
268711323 |
268711323 |
|
|
Percentage of shares (as a % of total shareholding of the
promoter and promoter group) |
100.00 |
100.00 |
100.00 |
|
|
Percentage of shares (as a % of total share capital of the
company) |
63.37 |
63.40 |
63.51 |
|
|
|
|
|
|
|
|
|
|
|
||
|
B.
Investor Complaints |
|
|
|
|
|
Pending at the beginning of the quarter |
Nil |
|
|
|
|
Receiving during the quarter |
6 |
|
|
|
|
Disposed of during the quarter |
6 |
|
|
|
|
Remaining unreserved at the end of the quarter |
Nil |
|
|
|
STANDALONE
STATEMENT OF ASSTES AND LIABILITIES AS ON 30.09.2014
Rs. In Millions
|
SOURCES OF FUNDS |
|
|
30.09.2014 (Unaudited) |
|
I.
EQUITY
AND LIABILITIES |
|
|
|
|
(1)Shareholders' Funds |
|
|
|
|
(a) Share Capital |
|
|
211.939 |
|
(b) Reserves & Surplus |
|
|
1272.799 |
|
Sub-total Shareholders’ |
|
|
1484.738 |
|
|
|
|
|
|
(2)Share application money pending allotment |
|
|
11.789 |
|
|
|
|
|
|
(2) Non-Current Liabilities |
|
|
|
|
(a) long-term borrowings |
|
|
1970.382 |
|
(b) Deferred tax liabilities (Net) |
|
|
0.000 |
|
(c) Other long term liabilities |
|
|
104.547 |
|
(d) long-term provisions |
|
|
128.878 |
|
Sub-total of
Non-Current liabilities |
|
|
2203.807 |
|
|
|
|
|
|
(3)
Current liabilities |
|
|
|
|
(a) Short
term borrowings |
|
|
3742.231 |
|
(b) Trade
payables |
|
|
859.442 |
|
(c) Other
current liabilities |
|
|
4697.970 |
|
(d) Short-term
provisions |
|
|
205.406 |
|
Sub-total of Current liabilities |
|
|
9505.049 |
|
|
|
|
|
|
TOTAL |
|
|
13205.393 |
|
|
|
|
|
|
II.
ASSETS |
|
|
|
|
(1)
Non-current assets |
|
|
|
|
(a) Fixed
Assets |
|
|
2113.510 |
|
(b)
Non-Current investments |
|
|
4078.989 |
|
(c) Deferred
tax assets (net) |
|
|
0.000 |
|
(d) Long
term loans and advances |
|
|
1082.808 |
|
(e) Other non-current
assets |
|
|
118.018 |
|
Sub-total of
Non-Current Assets |
|
|
7393.325 |
|
|
|
|
|
|
(2)
Current assets |
|
|
|
|
(a)
Current investments |
|
|
4.305 |
|
(b)
Inventories |
|
|
1402.533 |
|
(c) Trade
receivables |
|
|
2404.863 |
|
(d) Cash
and cash equivalents |
|
|
281.577 |
|
(e)
Short-term loans and advances |
|
|
1223.043 |
|
(f) Other
current assets |
|
|
495.747 |
|
Sub-total of
Current Assets |
|
|
5812.068 |
|
|
|
|
|
|
TOTAL |
|
|
13205.393 |
NOTES:
1.
Sales outside India also include sales
relating to First-To-File (FTF) products in the United States of America
("USA") in certain periods. Further, pursuant to the accounting
policy followed by the Company, sales outside India for certain periods
presented include transfer pricing adjustments with its subsidiaries for
materials already supplied to them (including supplied in earlier periods),
determined on the basis of significant judgment and estimates.
2.
(a) The Company had, on 16th September 2013, received an
‘import alert’ from the Food and Drug Administration of the USA ('US FDA') on
its manufacturing facility located in Mohali. Further, on 23rd January 2014,
the US FDA also prohibited the Company from manufacturing and distributing
active pharmaceutical ingredients (’APIs') from its Toansa manufacturing
facility and finished drug products containing APIs manufactured at this
facility into the US regulated market. Consequentially, both the above
facilities are subject to certain terms of the Consent Decree of permanent
injunction entered into by the Company in January 2012 (‘Consent Decree’).
Considering the above matters, provisions/ write-off
(primarily relating to inventories, trade commitments, sales return etc.), amounting
to Rs. 695.14 and Rs. 3,557.92 had been recognised in the financial results for
the quarter and six months ended 30th September 2013 and fifteen months ended
31st March 2014 respectively, based on the best information and estimates
available with the management.
(b) The Department of Justice of the USA (’US DOJ'), United
States Attorney’s Office for the District of New Jersey had issued an
administrative subpoena dated 13th March 2014 to the Company seeking
information primarily related to the Company’s API Toansa manufacturing
facility in India for which a Form 483 containing findings of the US FDA was
issued in January 2014. The Company is fully cooperating with this information
request and is in dialogue with the US DOJ for submission of the requisite
information.
3.
During the fifteen months ended 31st
March 2014, the Company had negotiated and settled with the US DOJ for
resolution of civil and criminal allegations on 13th May 2013 as per the decree
of the court of Maryland. The Company had recorded a provision of Rs. 26,480
(USD 500 million) in the year ended 31st December 2011, to cover all civil and
criminal liabilities. The settlement of this liability (along with related
interest and other cost) in compliance with the terms of settlement is subject
to regulatory/ statutory provisions. The above mentioned decrial amount of
liability (along with related interest and other cost) had been paid by the
Company's US subsidiaries including Ranbaxy Pharmaceuticals Inc. ('RPI'), USA,
a limited risk distributor. Under the said agreement of distribution, RPI had
invoked indemnity for itself and inter alia its affiliates. The settlement
amount had, accordingly, been apportioned between the Company and its US
subsidiaries. The resultant accounting adjustment for reversal of earlier
provision to the extent of apportionment to the US subsidiaries amounting to
Rs. 1,458.05 (USD 26.1 million) had been disclosed as an exceptional item in
the financial results for the fifteen months ended 31 March 2014.
4.
The research and development expenses
are classified under respective heads of total expenses according to the nature
of expense.
5.
The amount represents foreign exchange
gain/ (loss), net, on foreign currency option derivatives taken during previous
years (other than on option derivatives relating to loans) which are accounted
in accordance with Accounting Standard 30, "Financial Instruments:
Recognition and Measurement".
6.
Due to issuance of debentures in the
year ended 31st December 2012, certain required ratios have been presented. The
ratios have been computed as below:
- Debt service coverage ratio = Earnings before Interest and Tax/ (Interest
expense for the period t- Principal Repayment for all the loan funds during the
period)
- Interest service coverage ratio = Earnings before Interest
and Tax / Interest expense for the period.
- Earning before Interest and Tax = Earnings from ordinary activity before lax
(serial no. 9) f Finance cost (serial no. 6)
7.
On exercise of Employees Stock Options,
559,561 equity shares have been allotted on 11st October 2014. The total number
of Employees Stock Options outstanding as at 30th September 2014 were 4,163,481
out of which 3,064,057 have vested.
8.
Other income for the fifteen months
ended 31 March 2014 include dividend received from Ranbaxy (Netherlands) B.V.,
The Netherlands, a subsidiary of the Company, amounting to Rs. 6,113.97.
9.
During the fifteen months ended 31st
March 2014, the Company had transferred all significant risks and rewards of
ownership of the Intellectual Property of its branded generic product ‘Ketanov’
(including technology/ know-how, brand, marketing, authorisations, dossiers
etc.) to its subsidiary in Romania. The sales consideration of Rs. 4,327.69 was
determined by the management on the basis of a valuation report by an expert,
using the best estimate. Pursuant to this transaction, the Company had
recognised a gain of Rs. 4,327.69 which was disclosed as an exceptional item in
the financial results for the fifteen months ended 31st March 2014.
10.
The Company's business activity falls
within a single primary business segment viz. 'Pharmaceutical'.
11.
Finance costs include exchange
differences arising from foreign currency borrowings to the extent that they
are regarded as an adjustment to interest costs.
12.
In accordance with an opinion of the
Expert Advisory Committee ('EAC') of The Institute of Chartered Accountants of
India, the shares issued to an Employee Stock Option Plan ('ESOP') trust but
yet to be allotted to employees are required to be shown as a deduction from
the share capital. Accordingly, as at 30 September 2014, 30th June 2014 and
31st March 2014, the paid-up equity share capital has been disclosed after
deduction of Rs. 0.58, Rs. 1.28 and Rs. 2.30 in respect of 115,308, 255,818 and
459,602 such equity shares respectively, with a corresponding adjustment to the
loan receivable from the ESOP trust. To conform to this presentation, the
paid-up share capital as at 30th September 2013 has also been disclosed after
deduction of Rs. 0.44 in respect of 87,471 such equity shares.
13.
With regard to the Scheme of
Arrangement ('Scheme') providing inter-alia reduction of capital and merger of
the Company with M/s. Sun Pharmaceutical Industries Limited ('SPIL') with
effect from the appointed date of 01st April 2014, the National Stock Exchange
of India Limited CNSE') and the Bombay Stock Exchange Limited ('BSE') have,
based on the observations of the Securities and Exchange Board of India
('SEBI') with regard to the Scheme, conveyed their 'No Objection' in July 2014.
Subsequently, pursuant to the orders of the Hon’ble High Court of Punjab &
Haryana, Court convened meeting of Equity Shareholders was held on 19th
September 2014 where Equity Shareholders of the Company had approved the said
Scheme with requisite majority. The Company is in the process of obtaining
necessary approvals from the concerned authorities in this regard.
14.
Pursuant to Companies Act, 2013 (’the
Act') being effective from 01st April 2014, the Company has revised depreciation
rate on certain fixed assets as per the useful life specified in Part ‘C’ of
Schedule II of the Act or as per the management’s estimate based on an internal
evaluation. As a result of this change, the depreciation charge for the quarter
and six months ended 30th September 2014 is higher by Rs. 129.22 and 319.05
respectively. In respect of assets whose useful life is already exhausted as on
01st April 2014, depreciation of Rs. 179.39 (net of deferred tax impact of Rs.
92.37) has been adjusted in Reserves and Surplus in accordance with the
requirements of Schedule II of the Act.
15.
In view of the relevant provisions of
the Act, the Company, during the quarter and six months ended 30th September
2014, has not accrued any remuneration for its CEO and Managing Director,
except for certain perquisites and amortization of deferred employees stock
options compensation aggregating to Rs 1.22 and Rs 3.02 for the respective
periods. The Company has filed an application with the Central Government to
obtain the necessary approvals required under the provisions of the Act.
16.
During the fifteen months ended 31st
March 2014, the Company had changed its financial year from January-December to
April-March effective 01st April 2014. In view of this, the previous financial
year was for a period of 15 months i.e. 01st January 2013 to 31st March 2014.
Accordingly in these results, quarter and six months ended 30 September 2013
are disclosed as corresponding quarter and corresponding six months of the
previous period. The figures for the corresponding six months are the balancing
figures between unaudited published figures in respect of the nine months ended
30th September 2013 and unaudited published figures for the quarter ended 31st
March 2013.
17.
On 15th October 2014, the Company has
notified the Bank of New York Mellon ("The Bank") to terminate the
Deposit Agreements dated 07th July 1994 [under which, the Rule 144A Global
Depositary Shares ('GDS')/ Regulation S Global Depositary Shares have been
issued] and underlying Global Depositary Receipts ('GDR') program. The Bank has
issued notice of termination of GDR Program to the GDS holders and accordingly,
the Bank will complete the process of termination of GDR program as per the
terms of such notice of termination.
18.
Figures pertaining to previous periods
have been reclassified to conform to current period’s classification.
19.
The above results were reviewed by the
Audit Committee at their meeting held on 27th October 2014 and approved by the
Board of Directors at their meeting held on 28th October 2014 and have
undergone a "Limited Review" by the Statutory Auditors of the
Company. The review report of the Statutory Auditors is being filed with the
NSE and the BSE and is also available on the Company’s website at www.ranbaxy.com
FIXED ASSETS:
Tangible assets
· Freehold Land
· Leasehold Land
· Buildings
· Plant and Machinery
· Furniture and Fixtures
· Office Equipments
· Vehicles
Intangible assets
· Product development
· Patent rights, trade marks, designs and Licenses
· Computer software
· Non-compete fee
PRESS RELEASE
CCI
APPROVES RANBAXY ACQUISITION BY SUN PHARMA
Mumbai and Gurgaon, India:
Sun Pharmaceutical Industries Ltd. (Reuters:SUN.BO, Bloomberg: SUNP IN, NSE:
SUNPHARMA, BSE: 524715) and Ranbaxy Laboratories Ltd(Reuters: RANB.BO,
Bloomberg: RBXY IN, NSE: RANBAXY, BSE: 500359) today announced that theyhave
received the order of the Competition Commission of India (“CCI”) dated
December 5, 2014(“Order”) by which CCI has approved the acquisition of Ranbaxy
by Sun Pharma, subject to compliance with certain conditions.
Sun Pharma and Ranbaxy had
filed the notice with the CCI on 6 th May 2014 and sought its approval with
respect to the deal. Over the past few months, the CCI has sought information
and detailed clarifications for the purposes of making its assessment.
One of the preconditions
of the Order is that parties procure the divestment of seven products. These
products constitute less than 1% of the combined entity's revenues in India.
Dilip Shanghvi, Managing
Director of Sun Pharma said, “The Order of the CCI approving the deal isan
important milestone for the transaction. It revalidates our view that the Sun
Pharma and Ranbaxy businesses complement each other with limited product
overlap, and will offer a comprehensive product basket to enable future growth.
We are pleased with the open and transparent manner in which the matter has
progressed.
”Arun Sawhney, CEO &
Managing Director of Ranbaxy said, “The approval by CCI is a significant step
forward. We are confident that post closure, the combined entity will enable
sustainable long term growth and deliver immense value for all stakeholders.
“Sun Pharma and Ranbaxy are looking forward to progressing towards the completion
of the transaction and will comply with the conditions laid down by the CCI
within the specified time.
About Sun Pharma (CIN – L24230GJ1993PLC019050)
Established in 1983,
listed since 1994 and headquartered in India, Sun Pharmaceutical Industries Ltd.
(Reuters:SUN.BO, Bloomberg: SUNP IN, NSE: SUNPHARMA, BSE: 524715) is an
international specialty pharmaceutical company with over 75% sales from global
markets. It manufactures and markets a large basket of pharmaceutical
formulations as branded generics as well as generics in US, India and several
other markets across the world. For the year ending March 2014, overall
revenues were at US$2.7 billion, of which US contributed US$1.6 billion. In
India, the company is a leader in niche therapy areas of psychiatry, neurology,
cardiology, nephrology, gastroenterology, orthopedics and ophthalmology. The
company has strong skills in product development, process chemistry, and
manufacturing of complex dosage forms. More information about the company can
be found at www.sunpharma.com.
About Ranbaxy (CIN – L24231PB1961PLC003747)
Ranbaxy Limited is an
integrated, research based, international pharmaceutical company producing a
wide range of quality, affordable generic medicines, trusted by healthcare
professionals and patients across geographies. Ranbaxy’s continued focus on
R&D has resulted in several approvals in developed and emerging markets,
many of which incorporate proprietary Novel Drug Delivery Systems and
technologies developed at its own labs. The company has further strengthened
its focus on generics research and is increasingly working on more complex and
specialty areas. Ranbaxy serves its customers in over 150 countries and has an
expanding international portfolio of affiliates, joint ventures and alliances,
ground operations in 43 countries and manufacturing operations in 8 countries.
For more information, please visit www.ranbaxy.com.
RANBAXY Q1 FY 2015
(APR’14-JUN’14) SALES RS.23.7 BN
Growth registered in Base business
profitability
Gurgaon, India: The Board of Directors of
Ranbaxy Laboratories Limited (RLL, NSE: RANBAXY, BSE: 500359), at its meeting
held today, took on record the unaudited results for the Quarter ended June 30,
2014 (“Apr-Jun 2014”) under Indian GAAP.
Key Financial Highlights
Financial Performance for the
quarter ended June 30, 2014 (Apr-Jun 2014)
·
Consolidated Sales were Rs.23.7 Bn
[Apr-June 2013: Sales Rs.25.8 Bn].
·
Earnings before Interest, Tax,
Depreciation & Amortization (EBITDA) was Rs.2.4 Bn.
Commenting on the business results for the Quarter, Arun
Sawhney, CEO & Managing Director, Ranbaxy, said, “We continue to work
towards growing our base business with focus on emerging markets, while at the
same time, restoring the business on growth trajectory in our traditional
markets such as USA and Europe.”
Key Highlights/ Developments
Business and Financial
· On June 26, 2014, the Company received approval from the U.S. Food and Drug Administration (US FDA) to manufacture and market Valsartan 40 mg, 80 mg, 160 mg, and 320 mg tablets on an exclusive basis. Valsartan is indicated for the treatment of high blood pressure and heart failure. Total annual market sales for Diovan® were $2.19 billion (IMS – MAT: April 2014).
·
The India business recorded 12% growth
as against the Indian Pharma Market (IPM) growth of 10%. The Company expects to
continue the momentum in the months ahead. Base business EBITDA for the quarter
ending June 2014 continued to grow over the corresponding period.
·
Ranbaxy maintained its strong market
share in Absorica™, isotretenoin NDA in the USA. As of June 27, 2014 market
share was ~20%.
Regulatory, Research &
Development and Manufacturing
·
A joint inspection by multiple European
Agencies including UK, Ireland, Germany, Switzerland and TGA Australia was
successfully completed at the Toansa API facility during March 2014 with no
critical observations. The inspection team concluded that there was no evidence
that the products manufactured at the Toansa API facility have any product
quality or patient safety issues. As a consequence on June 5, 2014, the EU
authorities reinstated the EU GMP certificate for the Toansa facility.
·
The Company has received communications
from the National Stock Exchange (NSE) and Bombay Stock Exchange (BSE)
conveying their No Objection based on the observations of the Securities and
Exchange Board of India (SEBI) in regard to the Scheme of Arrangement for
merger of the Company with Sun Pharmaceutical Industries Ltd.
·
The Company has filed a petition with
the Honourable High Court of Punjab & Haryana to convene meetings of
shareholders etc.
Global Sales
·
Consolidated sales for the Quarter were
Rs.23.7 Bn as compared to Rs.25.8 Bn during the corresponding quarter.
·
Branded and OTC category contributed
Rs.13.7 Bn accounting for 58% of total sales during the Quarter. Generics and
others category recorded Rs.10 Bn of sales for the Company during the Quarter.
·
North America: Sales for the Quarter
were Rs.7.6 Bn.
·
In the USA, sales for the Quarter were
Rs.7.0 Bn primarily driven by AbsoricaTM with a market share of 20% (IMS
June’14).
·
India: In the domestic market, sales
for the Quarter were Rs.6.1 Bn, a growth of 12% over the corresponding period.
·
OTC business (Consumer Healthcare)
contributed Rs.0.9 Bn
·
East Europe & CIS: The region
recorded sales of Rs.3.4 Bn. Despite the challenges in the Ukrainian belt,
Ranbaxy continues to work effectively.
·
West Europe: Sales for the Quarter were
Rs.2.4 Bn, a growth of 10% over the corresponding quarter. Growth was led by
all round performance in UK, Germany and Spain.
·
Africa and Middle East: Sales for the
Quarter were Rs.2.0 Bn.
·
Asia Pacific and LATAM (including Sri
Lanka): Sales for the Quarter were Rs.1.6 Bn.
·
In LATAM sales for the Quarter were
Rs.0.4 Bn
·
API business and others contributed
sales of Rs.0.5 Bn. Business was impacted by the voluntary suspension of
shipments from its Toansa and Dewas facilities.
CMT REPORT (Corruption, Money Laundering & Terrorism]
The Public Notice information has been collected from various sources
including but not limited to: The Courts,
1] INFORMATION ON
DESIGNATED PARTY
No exist designating subject or any of its beneficial owners,
controlling shareholders or senior officers as terrorist or terrorist
organization or whom notice had been received that all financial transactions
involving their assets have been blocked or convicted, found guilty or against
whom a judgement or order had been entered in a proceedings for violating
money-laundering, anti-corruption or bribery or international economic or
anti-terrorism sanction laws or whose assets were seized, blocked, frozen or
ordered forfeited for violation of money laundering or international
anti-terrorism laws.
2] Court Declaration :
No exist to suggest that subject is or was
the subject of any formal or informal allegations, prosecutions or other
official proceeding for making any prohibited payments or other improper
payments to government officials for engaging in prohibited transactions or
with designated parties.
3] Asset Declaration :
No records exist to suggest that the property or assets of the subject
are derived from criminal conduct or a prohibited transaction.
4] Record on Financial
Crime :
Charges or conviction
registered against subject: None
5] Records on Violation of
Anti-Corruption Laws :
Charges or
investigation registered against subject: None
6] Records on Int’l
Anti-Money Laundering Laws/Standards :
Charges or
investigation registered against subject: None
7] Criminal Records
No
available information exist that suggest that subject or any of its principals have
been formally charged or convicted by a competent governmental authority for
any financial crime or under any formal investigation by a competent government
authority for any violation of anti-corruption laws or international anti-money
laundering laws or standard.
8] Affiliation with
Government :
No record
exists to suggest that any director or indirect owners, controlling
shareholders, director, officer or employee of the company is a government
official or a family member or close business associate of a Government
official.
9] Compensation Package :
Our market
survey revealed that the amount of compensation sought by the subject is fair
and reasonable and comparable to compensation paid to others for similar
services.
10] Press Report :
No press reports / filings exists on
the subject.
CORPORATE GOVERNANCE
MIRA INFORM as part of its Due Diligence do provide comments on
Corporate Governance to identify management and governance. These factors often
have been predictive and in some cases have created vulnerabilities to credit
deterioration.
Our Governance Assessment focuses principally on the interactions
between a company’s management, its Board of Directors, Shareholders and other
financial stakeholders.
CONTRAVENTION
Subject is not known to have contravened any existing local laws,
regulations or policies that prohibit, restrict or otherwise affect the terms
and conditions that could be included in the agreement with the subject.
FOREIGN EXCHANGE RATES
|
Currency |
Unit
|
Indian Rupees |
|
US Dollar |
1 |
Rs.61.76 |
|
|
1 |
Rs.94.01 |
|
Euro |
1 |
Rs.72.63 |
INFORMATION DETAILS
|
Analysis Done by
: |
KAR |
|
|
|
|
Report Prepared
by : |
KLS |
SCORE & RATING EXPLANATIONS
|
SCORE FACTORS |
RANGE |
POINTS |
|
HISTORY |
1~10 |
8 |
|
PAID-UP CAPITAL |
1~10 |
7 |
|
OPERATING SCALE |
1~10 |
8 |
|
FINANCIAL
CONDITION |
|
|
|
--BUSINESS SCALE |
1~10 |
8 |
|
--PROFITABILITY |
1~10 |
- |
|
--LIQUIDITY |
1~10 |
7 |
|
--LEVERAGE |
1~10 |
7 |
|
--RESERVES |
1~10 |
7 |
|
--CREDIT LINES |
1~10 |
8 |
|
--MARGINS |
-5~5 |
-- |
|
DEMERIT POINTS |
|
|
|
--BANK CHARGES |
YES/NO |
YES |
|
--LITIGATION |
YES/NO |
YES |
|
--OTHER ADVERSE INFORMATION |
YES/NO |
NO |
|
MERIT POINTS |
|
|
|
--SOLE DISTRIBUTORSHIP |
YES/NO |
NO |
|
--EXPORT ACTIVITIES |
YES/NO |
NO |
|
--AFFILIATION |
YES/NO |
YES |
|
--LISTED |
YES/NO |
YES |
|
--OTHER MERIT FACTORS |
YES/NO |
YES |
|
DEFAULTER
|
|
|
|
--RBI |
YES/NO |
NO |
|
--EPF |
YES/NO |
NO |
|
TOTAL |
|
60 |
This score serves as a reference to assess
SC’s credit risk and to set the amount of credit to be extended. It is
calculated from a composite of weighted scores obtained from each of the major
sections of this report. The assessed factors and their relative weights (as
indicated through %) are as follows:
Financial
condition (40%) Ownership
background (20%) Payment
record (10%)
Credit history
(10%) Market trend (10%) Operational size
(10%)
RATING EXPLANATIONS
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
>86 |
Aaa |
Possesses an extremely sound financial base with the strongest
capability for timely payment of interest and principal sums |
Unlimited |
|
71-85 |
Aa |
Possesses adequate working capital. No caution needed for credit
transaction. It has above average (strong) capability for payment of interest
and principal sums |
Large |
|
56-70 |
A |
Financial & operational base are regarded healthy. General
unfavourable factors will not cause fatal effect. Satisfactory capability for
payment of interest and principal sums |
Fairly Large |
|
41-55 |
Ba |
Overall operation is considered normal. Capable to meet normal
commitments. |
Satisfactory |
|
26-40 |
B |
Capability to overcome financial difficulties seems comparatively
below average. |
Small |
|
11-25 |
Ca |
Adverse factors are apparent. Repayment of interest and principal sums
in default or expected to be in default upon maturity |
Limited with
full security |
|
<10 |
C |
Absolute credit risk exists. Caution needed to be exercised |
Credit not
recommended |
|
-- |
NB |
New Business |
-- |
This report is issued at your request without any
risk and responsibility on the part of MIRA INFORM PRIVATE LIMITED (MIPL) or
its officials.