MIRA INFORM REPORT
|
Report No. : |
330665 |
|
Report Date : |
08.07.2015 |
IDENTIFICATION DETAILS
|
Name : |
GRANULES INDIA LIMITED |
|
|
|
|
Registered
Office : |
Second Floor, 3rd Block, My Home Hub, Madhapur,
Cyberabad, Hyderabad – 500 081, Telangana |
|
Tel. No.: |
91-40-66760000 |
|
|
|
|
Country : |
India |
|
|
|
|
Financials (as
on) : |
31.03.2015 |
|
|
|
|
Date of
Incorporation : |
18.03.1991 |
|
|
|
|
Com. Reg. No.: |
36-012471 |
|
|
|
|
Capital
Investment / Paid-up Capital : |
Rs.204.761 Million |
|
|
|
|
CIN No.: [Company Identification
No.] |
L24110TG1991PLC012471 |
|
|
|
|
TAN No.: [Tax Deduction &
Collection Account No.] |
HYDG00432F |
|
|
|
|
PAN No.: [Permanent Account No.] |
AAACG7369K |
|
|
|
|
Legal Form : |
A Public Limited Liability Company. The Company’s Shares are Listed on
the Stock Exchanges. |
|
|
|
|
Line of Business
: |
Manufacture Export of Paper board – based printed packaging material |
|
|
|
|
No. of Employees
: |
1500 (Approximately) |
RATING & COMMENTS
|
MIRA’s Rating : |
Ba (50) |
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
41-55 |
Ba |
Overall operation is considered normal. Capable to meet normal
commitments. |
Satisfactory |
|
Maximum Credit Limit : |
USD 12000000 |
|
|
|
|
Status : |
Satisfactory |
|
|
|
|
Payment Behaviour : |
Usually correct |
|
|
|
|
Litigation : |
Exist |
|
|
|
|
Comments : |
Subject is an established company having satisfactory track record. Financial position of the company is good. Rating takes into
consideration, healthy liquidity position and good profitability of the
company. Trade relations are reported as fair. Business is active. Payment
terms are reported to be usually correct. In the view of long track record of operations and extensive
experience of promoters, the Company can be considered normal for business
dealings at usual trade terms and conditions. |
NOTES :
Any query related to this report can be made
on e-mail : infodept@mirainform.com
while quoting report number, name and date.
ECGC Country Risk Classification List – March 31, 2015
|
Country Name |
Previous Rating (31.12.2014) |
Current Rating (31.03.2015) |
|
India |
A1 |
A1 |
|
Risk Category |
ECGC
Classification |
|
Insignificant |
A1 |
|
Low |
A2 |
|
Moderate |
B1 |
|
High |
B2 |
|
Very High |
C1 |
|
Restricted |
C2 |
|
Off-credit |
D |
EXTERNAL AGENCY RATING
|
Rating Agency Name |
CARE |
|
Rating |
Long Term Bank Facilities : A- Carry |
|
Rating Explanation |
Adequate degree of safety and low credit
risk. |
|
Date |
24.07.2014 |
|
Rating Agency Name |
CARE |
|
Rating |
Short Term Bank Facilities: A2 + Carry |
|
Rating Explanation |
Strong degree of safety and low credit risk.
|
|
Date |
24.07.2014 |
RBI DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available RBI Defaulters’ list.
EPF (Employee Provident Fund) DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available EPF (Employee Provident Fund) Defaulters’ list as of
31-03-2013.
INFORMATION PARTED BY (GENERAL DETAILS)
|
Name : |
Mr. B. Chandrashekhar |
|
Designation : |
General Manager Finance |
|
Contact No.: |
91-9963027747 |
|
Date : |
|
LOCATIONS
|
Registered Office/ Corporate Office : |
Second Floor, 3rd Block, My Home Hub, Madhapur,
Cyberabad, Hyderabad – 500 081, Telangana, India |
|
Tel. No.: |
91-40-66760000 |
|
Mobile No.: |
91-9963027747 (Mr. B. Chandrashekhar) |
|
Fax No.: |
91-40-23115145 |
|
E-Mail : |
|
|
Website: |
|
|
Location : |
Rented |
|
Locality : |
Industrial |
|
|
|
|
Factory 1 : |
Plot No.15/A/1,
Phase-III, I.D.A. Jeedimetla, Hyderabad – 500055, Telangana, India |
|
|
|
|
Factory 2 : |
Temple Road, Bonthapally, Medak District – 502313, Telangana, India |
|
|
|
|
Factory 3 : |
Plot No.160/A and 161/E, Gagillapur Village, Qutubullapur Mandal, R.R. District – 500043, Andhra Pradesh, India |
|
|
|
|
Factory : |
Gat No.258, Shreeram Building, Lawale Phata, Pirangut, Taluka Mulshi,
District Pune – 412 108, Maharashtra, India |
|
|
|
|
R&D Centre 1 : |
Formulations Gagillapur, Qutubullapur Mandal, Ranga Reddy District – 500043, Andhra Pradesh, India |
|
|
|
|
R&D Centre 2 : |
API Plot No.15/A/1, Phase-III, I.D.A. Jeedimetla, Hyderabad – 500055, Andhra Pradesh, India |
|
|
|
|
API &
Formulations : |
Plot no. 56, Road no. 5, ALEAP, Hyderabad, Telangana, India |
|
|
|
|
Overseas Offices : |
Located at: · Europe · U.S. · Canada ·
· Asia · Middle East · Africa |
DIRECTORS
As on 31.03.2014
|
Name : |
Mr. C. Krishna Prasad |
|
Designation : |
Managing Director – Non-Independent |
|
Date of Birth/Age : |
02.10.1954 |
|
Qualification : |
B. Sc. |
|
Date of Appointment : |
31.08.1994 |
|
|
|
|
Name : |
Mr. L.S. Sarma |
|
Designation : |
Director – Non-Executive, Independent |
|
|
|
|
Name : |
Mr. A.P. Kurian |
|
Designation : |
Director – Non-Executive, Independent |
|
|
|
|
Name : |
Mr. C. Parthasarathy |
|
Designation : |
Director – Non-Executive, Independent |
|
Date of Birth/Age : |
07.07.1955 |
|
Qualification : |
B. Sc., LLB, FCA, FCS |
|
Date of Appointment : |
27.05.2009 |
|
|
|
|
Name : |
Dr. Krishna Murthy Ella |
|
Designation : |
Director – Non-Executive, Independent |
|
|
|
|
Name : |
Mr. Arun Rao Akinepally |
|
Designation : |
Director – Non-Executive, Independent |
|
|
|
|
Name : |
Ms. Harsha Chigurupati |
|
Designation : |
Executive Director – Non-Independent |
|
|
|
|
Name : |
Mrs. Uma Chigurupati |
|
Designation : |
Director |
|
|
|
|
Name : |
Mr. K.B. Sankara rao |
|
Designation : |
Director |
KEY EXECUTIVES
|
Name : |
Mr. B. Chandrashekhar |
|
Designation : |
General Manager Finance |
MAJOR SHAREHOLDERS / SHAREHOLDING PATTERN
As on 31.03.2015
|
Category of Shareholder |
Total No. of Shares |
Total Shareholding as a % of Total No. of Shares |
|
|
||
|
(A) Shareholding of
Promoter and Promoter Group |
||
|
|
|
|
|
|
86386620 |
42.29 |
|
|
7491270 |
3.67 |
|
|
93877890 |
45.96 |
|
|
|
|
|
|
5369310 |
2.63 |
|
|
5369310 |
2.63 |
|
Total shareholding of
Promoter and Promoter Group (A) |
99247200 |
48.59 |
|
|
|
|
|
(B) Public Shareholding |
||
|
|
|
|
|
|
152056 |
0.07 |
|
|
8243996 |
4.04 |
|
|
8396052 |
4.11 |
|
|
|
|
|
|
9315594 |
4.56 |
|
|
|
|
|
Individual shareholders
holding nominal share capital up to Rs. 0.100 Million |
35794440 |
17.52 |
|
Individual shareholders holding
nominal share capital in excess of Rs. 0.100 Million |
14487449 |
7.09 |
|
|
37010805 |
18.12 |
|
|
31983350 |
15.66 |
|
|
4116778 |
2.02 |
|
|
910677 |
0.45 |
|
|
96608288 |
47.30 |
|
Total Public shareholding
(B) |
105004340 |
51.41 |
|
|
|
|
|
Total (A)+(B) |
204251540 |
100.00 |
|
|
|
|
|
(C) Shares held by Custodians
and against which Depository Receipts have been issued |
|
|
|
|
0 |
0.00 |
|
|
0 |
0.00 |
|
|
0 |
0.00 |
|
|
|
|
|
Total (A)+(B)+(C) |
204251540 |
100.00 |

BUSINESS DETAILS
|
Line of Business : |
Manufacture Export of Paper board – based printed packaging material |
||||
|
|
|
||||
|
Products : (As on 31.03.2014) |
|
||||
|
|
|
||||
|
Brand Names : |
Not Divulged |
||||
|
|
|
||||
|
Agencies Held : |
Not Divulged |
||||
|
|
|
||||
|
Exports : |
Not Divulged |
||||
|
|
|
||||
|
Imports : |
Not Divulged |
||||
|
|
|
||||
|
Terms : |
|
||||
|
Selling : |
Cash, Cheque and Credit |
||||
|
|
|
||||
|
Purchasing : |
Cash, Cheque and Credit |
PRODUCTION STATUS NOT AVAILABLE
GENERAL INFORMATION
|
Suppliers : |
|
||||||||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||||||||
|
Customers : |
Retailers, End Users and OEM’s
|
||||||||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||||||||
|
No. of Employees : |
1500 (Approximately) |
||||||||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||||||||
|
Bankers : |
|
||||||||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||||||||
|
Facilities : |
|
||||||||||||||||||||||||||||||
![]()
|
Financial Institution : (As on
31.03.2014) |
International Finance Corporation |
|
|
|
|
Statutory Auditors : (As on
31.03.2014) |
|
|
Name : |
Kumar and Giri Chartered Accountants |
|
Address : |
D.No.1-11-126/D, Opposite Aeroview Towers, Begumpet,
Hyderabad – 500 016, Telangana, India |
|
|
|
|
Internal Auditors : (As on
31.03.2014) |
|
|
Name : |
Dhanunjaya and Haranath Chartered Accountants |
|
Address : |
302, Wings, 8-3-960/6/2,
Srinagar Colony, Hyderabad – 500073, Telangana,
India |
|
|
|
|
Wholly Owned Subsidiary Companies : (As on
31.03.2014) |
·
Granules USA Inc. ·
GIL Life Sciences Private Limited ·
Granules Singapore Pte Limited ·
Auctus Pharma Limited |
|
|
|
|
Joint Venture : (As on
31.03.2014) |
·
Granules-Biocause Pharmaceutical Co. Limited ·
Granules Omnichem Private Limited |
|
|
|
|
Directors
Interest : (As on
31.03.2014) |
Karvy Computershare Private Limited |
CAPITAL STRUCTURE
As on 31.03.2015
Authorised Capital : Not Available
Issued, Subscribed & Paid-up Capital : Rs.204.761
Million
As on 31.03.2014
Authorised Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
30000000 |
Equity Shares |
Rs.10/- each |
Rs.300.000 Million |
|
|
|
|
|
Issued, Subscribed & Paid-up Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
20281154 |
Equity Shares |
Rs.10/- each
|
Rs.202.812
Million |
|
|
|
|
|
Reconciliation of the
number of shares outstanding and the amount of share capital as at March 31,
2013
|
Particulars |
As at 31st March, 2014 |
|
|
No. of Shares |
Amount (Rs. in Million) |
|
|
Number of shares
at the beginning of the year |
20126154 |
201.262 |
|
Add: Shares
issued on exercise of employee stock options |
155000 |
1.550 |
|
Number of shares
at the end of the year |
20281154 |
202.812 |
Terms/Rights
attached to equity shares:
The Company has only
one class of equity shares having a par value of Rs.10/- . Each holder of equity shares is entitled to one vote per
share. The Company declares and pays dividends in Indian rupees. The dividend
proposed by the Board of Directors is subject to the approval of the
shareholders in the ensuing Annual General Meeting.
During the year
end 31-Mar-2014, the amount of per share dividend recognized as distribution to
equity shareholders was Rs.3.50/-
(31-Mar-2013: Rs.2.00/-)
In the event of liquidation
of the Company, the holders of equity shares will be entitled to receive
remaining assets of the Company after distribution of all preferential amounts.
The distribution will be in proportion to the number of equity shares held by
the shareholders.
Details of
shareholders holding more than 5% shares in the Company
|
PARTICULARS |
As at 31st March, 2014 |
|
|
No. of Shares |
% |
|
|
C. Krishna Prasad |
7473538 |
36.85 |
|
Investco Management LLC |
2211200 |
10.90 |
|
Ridgeback Capital Asia Limited |
1144135 |
5.64 |
|
International Finance Corporation |
---- |
---- |
FINANCIAL DATA
[all figures are
in Rupees Million]
ABRIDGED
BALANCE SHEET
|
SOURCES
OF FUNDS |
31.03.2015 |
31.03.2014 |
31.03.2013 |
|
|
|
|
|
|
I.
EQUITY AND LIABILITIES |
|
|
|
|
(1)Shareholders' Funds |
|
|
|
|
(a) Share Capital |
204.252 |
202.812 |
201.262 |
|
(b) Reserves & Surplus |
4049.841 |
3240.315 |
2503.668 |
|
(c) Money received against
share warrants |
0.000 |
0.000 |
0.000 |
|
|
|
|
|
|
(2) Share Application money
pending allotment |
0.000 |
2.548 |
3.150 |
|
Total
Shareholders’ Funds (1) + (2) |
4254.093 |
3445.675 |
2708.080 |
|
|
|
|
|
|
(3) Non-Current Liabilities |
|
|
|
|
(a) long-term borrowings |
2446.973 |
2220.918 |
1407.237 |
|
(b) Deferred tax liabilities
(Net) |
532.810 |
358.081 |
250.986 |
|
(c) Other long term
liabilities |
0.000 |
0.000 |
0.000 |
|
(d) long-term provisions |
51.274 |
36.990 |
21.238 |
|
Total
Non-current Liabilities (3) |
3031.057 |
2615.989 |
1679.461 |
|
|
|
|
|
|
(4) Current Liabilities |
|
|
|
|
(a) Short term borrowings |
1148.695 |
774.862 |
767.148 |
|
(b) Trade payables |
1838.435 |
1262.48 |
838.475 |
|
(c) Other current liabilities |
616.326 |
376.049 |
129.266 |
|
(d) Short-term provisions |
122.916 |
83.048 |
47.093 |
|
Total
Current Liabilities (4) |
3726.372 |
2496.439 |
1781.982 |
|
|
|
|
|
|
TOTAL |
11011.522 |
8558.103 |
6169.523 |
|
|
|
|
|
|
II.
ASSETS |
|
|
|
|
(1) Non-current assets |
|
|
|
|
(a) Fixed Assets |
|
|
|
|
(i) Tangible assets |
4352.754 |
3264.286 |
2324.008 |
|
(ii) Intangible Assets |
868.928 |
92.443 |
124.338 |
|
(iii) Capital work-in-progress |
184.997 |
391.421 |
916.024 |
|
(iv) Intangible assets under
development |
0.000 |
0.000 |
0.000 |
|
(b) Non-current Investments |
724.719 |
1454.460 |
412.960 |
|
(c) Deferred tax assets (net) |
0.000 |
0.000 |
0.000 |
|
(d) Long-term Loan and Advances |
186.965 |
31.466 |
125.276 |
|
(e) Other Non-current assets |
18.360 |
25.558 |
35.764 |
|
Total
Non-Current Assets |
6336.723 |
5259.634 |
3938.370 |
|
|
|
|
|
|
(2) Current assets |
|
|
|
|
(a) Current investments |
0.000 |
0.000 |
94.592 |
|
(b) Inventories |
1793.963 |
1202.873 |
1084.907 |
|
(c) Trade receivables |
1898.094 |
1218.197 |
680.966 |
|
(d) Cash and cash equivalents |
522.309 |
219.258 |
61.597 |
|
(e) Short-term loans and
advances |
38.645 |
31.434 |
56.479 |
|
(f) Other current assets |
421.788 |
626.707 |
252.612 |
|
Total
Current Assets |
4674.799 |
3298.469 |
2231.153 |
|
|
|
|
|
|
TOTAL |
11011.522 |
8558.103 |
6169.523 |
PROFIT
& LOSS ACCOUNT
|
|
PARTICULARS |
31.03.2015 |
31.03.2014 |
31.03.2013 |
|
|
SALES |
|
|
|
|
|
Income from Operations |
12133.113 |
10016.799 |
6797.97 |
|
|
Other Income |
29.632 |
36.475 |
17.181 |
|
|
TOTAL
(A) |
12162.745 |
10053.274 |
6815.151 |
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
Cost of Materials Consumed |
7177.712 |
5873.565 |
4252.584 |
|
|
Purchases of Stock-in-Trade |
0.000 |
0.000 |
0.000 |
|
|
Changes in inventories of
finished goods, work-in-progress and Stock-in-Trade |
(105.618) |
(23.762) |
(74.062) |
|
|
Employees benefits expense |
892.321 |
692.738 |
504.205 |
|
|
Manufacturing Expenses |
752.640 |
0.000 |
0.000 |
|
|
Freight outward and clearing
charges |
499.325 |
0.000 |
0.000 |
|
|
R and D Expenses |
84.039 |
0.000 |
0.000 |
|
|
Other expenses |
734.850 |
1844.176 |
1324.668 |
|
|
TOTAL
(B) |
10035.269 |
8386.717 |
6007.395 |
|
|
|
|
|
|
|
PROFIT/
(LOSS) BEFORE INTEREST, TAX,
DEPRECIATION AND AMORTISATION (C) |
2127.476 |
1666.557 |
807.756 |
|
|
|
|
|
|
|
|
Less |
FINANCIAL
EXPENSES (D) |
309.294 |
189.270 |
164.949 |
|
|
|
|
|
|
|
|
PROFIT
/ (LOSS) BEFORE TAX, DEPRECIATION AND AMORTISATION (C-D) (E) |
1818.182 |
1477.287 |
642.807 |
|
|
|
|
|
|
|
Less/
Add |
DEPRECIATION/
AMORTISATION (F) |
495.587 |
263.979 |
202.089 |
|
|
|
|
|
|
|
|
PROFIT/ (LOSS)
BEFORE TAX (E-F) (G) |
1322.595 |
1213.308 |
440.718 |
|
|
|
|
|
|
|
Less |
TAX (H) |
370.331 |
403.194 |
138.965 |
|
|
|
|
|
|
|
|
PROFIT/ (LOSS)
AFTER TAX (G-H) (I) |
952.264 |
810.114 |
301.753 |
|
|
|
|
|
|
|
|
EARNINGS
IN FOREIGN CURRENCY |
|
|
|
|
|
Export of goods calculated on
F.O.B. basis |
NA |
8239.59 |
5457.763 |
|
|
TOTAL
EARNINGS |
NA |
8239.590 |
5457.763 |
|
|
|
|
|
|
|
|
IMPORTS |
|
|
|
|
|
Raw Materials |
NA |
3569.567 |
2759.834 |
|
|
Components and Stores parts |
NA |
20.906 |
12.277 |
|
|
Capital Goods |
NA |
95.356 |
150.829 |
|
|
TOTAL
IMPORTS |
NA |
3685.829 |
2922.940 |
|
|
|
|
|
|
|
|
Earnings
/ (Loss) Per Share (Rs.) |
4.67 |
40.06 |
15.02 |
CURRENT MATURITIES OF LONG TERM DEBT DETAILS
|
Particulars |
31.03.2015 |
31.03.2014 |
31.03.2013 |
|
Current Maturities of Long term debt |
NA |
302.613 |
95.214 |
|
Cash generated from operations |
NA |
1317.301 |
1023.853 |
|
Net cash flow from operating activity |
NA |
1079.484 |
911.831 |
KEY
RATIOS
|
PARTICULARS |
|
31.03.2015 |
31.03.2014 |
31.03.2013 |
|
Net Profit Margin (PAT / Sales) |
(%) |
7.85 |
8.09 |
4.44 |
|
|
|
|
|
|
|
Operating Profit Margin (PBIDT/Sales) |
(%) |
17.53 |
16.64 |
11.88 |
|
|
|
|
|
|
|
Return on Total Assets (PBT/Total Assets} |
(%) |
13.09 |
18.08 |
9.10 |
|
|
|
|
|
|
|
Return on Investment (ROI) (PBT/Networth) |
|
0.31 |
0.35 |
0.16 |
|
|
|
|
|
|
|
Debt Equity Ratio (Total Debt /Networth) |
|
0.85 |
0.96 |
0.84 |
|
|
|
|
|
|
|
Current Ratio (Current Asset/Current Liability) |
|
1.25 |
1.32 |
1.25 |
STOCK
PRICES
|
Face Value |
Rs.1.00 |
|
Market Value |
Rs.82.30 |
FINANCIAL ANALYSIS
[all figures are
in Rupees Million]
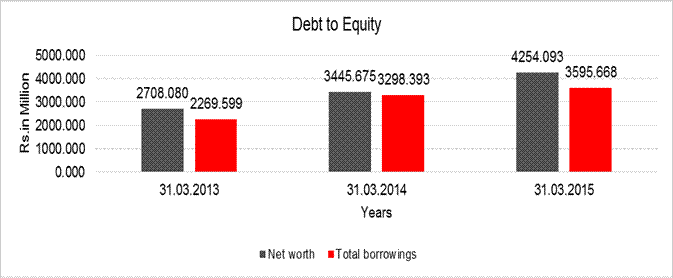
DEBT EQUITY RATIO
|
Particular |
31.03.2013 |
31.03.2014 |
31.03.2015 |
|
|
(Rs.
In Million) |
(Rs.
In Million) |
(Rs.
In Million) |
|
Share Capital |
201.262 |
202.812 |
204.252 |
|
Reserves & Surplus |
2503.668 |
3240.315 |
4049.841 |
|
Share Application money pending allotment |
3.150 |
2.548 |
0.000 |
|
Net
worth |
2708.080 |
3445.675 |
4254.093 |
|
|
|
|
|
|
long-term borrowings |
1407.237 |
2220.918 |
2446.973 |
|
Short term borrowings |
767.148 |
774.862 |
1148.695 |
|
Current Maturities of
Long-Term Debts |
95.214 |
302.613 |
0.000 |
|
Total
borrowings |
2269.599 |
3298.393 |
3595.668 |
|
Debt/Equity
ratio |
0.838 |
0.957 |
0.845 |
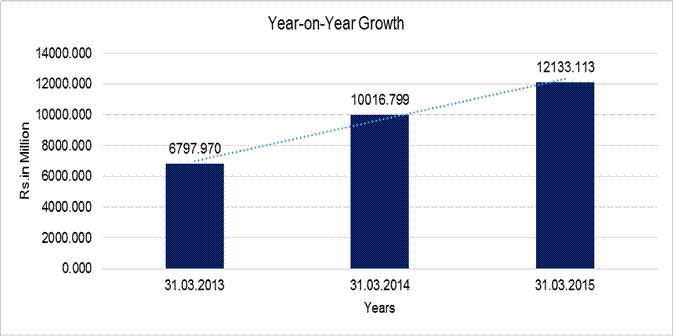
YEAR-ON-YEAR GROWTH
|
Year
on Year Growth |
31.03.2013 |
31.03.2014 |
31.03.2015 |
|
|
(Rs.
In Million) |
(Rs.
In Million) |
(Rs.
In Million) |
|
Sales |
6797.970 |
10016.799 |
12133.113 |
|
|
|
47.350 |
21.128 |
NET PROFIT MARGIN
|
Net
Profit Margin |
31.03.2013 |
31.03.2014 |
31.03.2015 |
|
|
(Rs.
In Million) |
(Rs.
In Million) |
(Rs.
In Million) |
|
Sales |
6797.970 |
10016.799 |
12133.113 |
|
Profit |
301.753 |
810.114 |
952.264 |
|
|
4.44% |
8.09% |
7.85% |

LOCAL AGENCY FURTHER INFORMATION
|
Sr. No. |
Check list by
info agents |
Available in
Report (Yes/No) |
|
1 |
Year of establishment |
Yes |
|
2 |
Constitution of the entity -Incorporation
details |
Yes |
|
3 |
Locality of the entity |
Yes |
|
4 |
Premises details |
No |
|
5 |
Buyer visit details |
-- |
|
6 |
Contact numbers |
Yes |
|
7 |
Name of the person contacted |
Yes |
|
8 |
Designation of contact person |
Yes |
|
9 |
Promoter’s background |
Yes |
|
10 |
Date of Birth of Proprietor / Partners /
Directors |
Yes |
|
11 |
Pan Card No. of Proprietor / Partners |
No |
|
12 |
Voter Id Card No. of Proprietor / Partners |
No |
|
13 |
Type of business |
Yes |
|
14 |
Line of Business |
Yes |
|
15 |
Export/import details (if applicable) |
No |
|
16 |
No. of employees |
Yes |
|
17 |
Details of sister concerns |
Yes |
|
18 |
Major suppliers |
No |
|
19 |
Major customers |
No |
|
20 |
Banking Details |
Yes |
|
21 |
Banking facility details |
Yes |
|
22 |
Conduct of the banking account |
-- |
|
23 |
Financials, if provided |
Yes |
|
24 |
Capital in the business |
Yes |
|
25 |
Last accounts filed at ROC, if applicable |
Yes |
|
26 |
Turnover of firm for last three years |
Yes |
|
27 |
Reasons for variation <> 20% |
-- |
|
28 |
Estimation for coming financial year |
No |
|
29 |
Profitability for last three years |
Yes |
|
30 |
Major shareholders, if available |
Yes |
|
31 |
External Agency Rating, if available |
Yes |
|
32 |
Litigations that the firm/promoter
involved in |
-- |
|
33 |
Market information |
-- |
|
34 |
Payments terms |
Yes |
|
35 |
Negative Reporting by Auditors in the
Annual Report |
No |
LITIGATION DETAILS
|
CASE STATUS INFORMATION SYSTEM
|
UNSECURED LOAN
|
Particulars |
31.03.2014 (Rs.
in Million) |
31.03.2013 (Rs.
in Million) |
|
LONG TERM
BORROWINGS |
|
|
|
Deferred sales
tax loan (Deferred sales tax
loan is interest free and payable in 14 yearly installments commencing from
June 2013 onwards.) |
6.389 |
6.473 |
|
|
|
|
|
Creditors for capital goods |
61.331 |
0.000 |
|
Total
|
67.720 |
6.473 |
CORPORATE INFORMATION:
(As on 31.03.2014)
The Company is a public domiciled in India and incorporated under the Companies Act, 1956. Its shares are listed on two stock exchanges in India. The Company is engaged in the manufacturing and selling of Active Pharmaceutical Ingredients (APIs), Pharmaceutical Formulation Intermediates (PFIs) and Finished Dosages (FDs). The Company caters to both domestic and international markets.
REVIEW OF OPERATIONS (As on 31.03.2014)
The Company posted
very good results during the financial year FY 14. The Company reported record
sales of Rs.10016.799 Million in the FY 14 compared to net sales of Rs.6797.970
Million in FY 13, registering growth of 47.35% in the current financial year.
The Company’s growth was driven by strong performance across all manufacturing
facilities. The primary growth driver in FY 14 was led by the Company’s
commercialization of its PFI and Finished Dosage expansion at its Gagillapur
facility. The Company improved utilization in the newly expanded facility throughout
the year and expects to continue improving utilization in the future. In
addition, the API facilities continued to increase production which contributed
to strong sales. On a standalone basis, the Finished Dosage business
contributed the largest share of revenue at 35% while PFI and API contributed
33% and 32%, respectively. This is compared to 31%, 32% and 37% for Finished
Dosages, PFI and API, respectively in FY 13.
The EBITDA stood
at Rs.1666.557 Million in FY 14 compared to
Rs.807.756 Million in FY 13, registering a
growth of 106.32%. The profit after tax for FY 14 stood at Rs.810.114 Million compared to Rs.301.753 Million in FY 13, registering a growth of 168.47%
in the current financial year. The profitability outpaced revenue growth due to
several reasons. First, the Company improved its economy of scale by increasing
production utilization in its newly expanded PFI and Finished Dosage capacity
at the Gagillapur facility. Due to the increased utilization, the product mix
shifted more towards Finished Dosages which bolstered profitability. Also, due
to the Company’s focus on operational excellence, the API units also increased
capacity through de-bottlenecking which drove profitability. The Company
believes the profitability margins from the standalone operations are
sustainable.
The Company was
able to increase sales due to relentless focus on delivering high-quality
material at a cost-effective price. The Company believes that its product
portfolio offers compelling opportunities and will continue to strengthen its
leadership position. In addition to growing its core business, it has been
looking at opportunities to diversify its sales by leveraging its core
competency of efficient manufacturing. In FY 14, Company made its first
acquisition by purchasing Auctus Pharma Limited (‘Auctus’). The acquisition of
Auctus fits into Company’s strategy of being a fully integrated manufacturer
while diversifying its product portfolio by adding high-value products with
significant market demand. In the shortto- mid term, Company will focus on
selling APIs from the Auctus portfolio to customers in the regulated markets.
Over the long-term, it will add value by offering Finished Dosages from select
APIs in the same portfolio. The Company will continue to strengthen its model
and build systems that are sustainable as it continue to scale-up.
EXPANSIONS (As on 31.03.2014)
During FY 14, the Company commercialized its expansion at the
Gagillapur facility. The expansion involved a capacity expansion in the PFI and
Finished Dosage facilities. The expansion mainly focused on efficient design
and output in terms of material transfer and automation. During the year, the
Company received customer approvals for both expansions and capacity
utilization increased accordingly. The Company also continued construction of a
new central warehouse at its Gagillapur plant in FY 14.
SUBSIDIARY
COMPANIES (As on 31.03.2014)
GRANULES USA INC
Granules USA Inc, a wholly–owned subsidiary company Company achieved a turnover of Rs.1107.999 Million against the turnover of Rs.751.000 Million of FY 13 and the profit after tax is Rs.8.640 Million against Rs.27.096 Million of FY 13. The Company achieved a turnover of Rs.1107.999 Million against the turnover of Rs.751.000 Million of FY 13 and the profit after tax is Rs.8.640 Million against Rs.27.096 Million of FY 13.
GIL LIFESCIENCES
PRIVATE LIMITED (As on 31.03.2014)
The Company has not commenced any activity so far. As on 31st March 2014 the Authorized Share Capital of the Company is Rs.35.000 Million divided into 3.500 Million equity shares of Rs.10/– each and the Paid Up Share Capital of the Company is Rs.29.462 Million divided into 2.946 Million equity shares of Rs.10 each.
GRANULES SINGAPORE
PTE LIMITED (As on 31.03.2014)
During the year ended 31st March 2014 Granules Singapore Pte. Limited (Singapore) was liquidated due to no operations.
AUCTUS PHARMA LIMITED
(As on 31.03.2014)
Auctus Pharma Limited is a public limited and unlisted company incorporated on 25th April 2000 located at Hyderabad (Telangana). Auctus Pharma Limited is the manufacturer and seller of bulk drugs and drug intermediates. The Company acquired Auctus Pharma Limited by purchasing its total 3,900,000 (thirty nine lakhs) equity shares on 14th February 2014 on payment of Rs.1020.000 Million as purchase consideration to the shareholders of the Auctus Pharma Limited. The Share Capital of the Company as on 31st March 2014 is Rs.39.000 Million. The net sales of the Company in FY 14 stood at Rs.108.800 Million compared to the net sales of Rs.114.212 Million in FY 13, registering a reduction of 4.74% in the current financial year. The EBITDA stood at Rs.3.490 Million in FY 14 compared to Rs.21.260 Million in FY 13, registering a reduction of 116.42%. The loss after tax for FY 14 stood at Rs.64.489 Million compared to Rs.40.518 Million in FY 13, registering a increase of 59.16% in the current financial year. The Company is working on increasing the productivity.
JOINT VENTURE
COMPANIES (As on 31.03.2014)
GRANULES–BIOCAUSE
PHARMACEUTICAL CO. LIMITED
The Share Capital of the Company as on 31st March 2014 is Rs.181.903 Million. During FY 14, the Company achieved a turnover of Rs.2639.889 Million of which Granules India Limited reports 50% in its consolidated revenue. Granules–Biocause successfully completed a U.S. FDA inspection in FY 14 with zero 483 observations.
GRANULES OMNICHEM
PRIVATE LIMITED (As on 31.03.2014)
The Share Capital of the Company as on 31st March 2014 is Rs.414.000 Million. The Company has not yet commenced its commercial activity during the period hence there was no income during FY 14. However the Company incurred a loss of Rs.13.817 Million. Granules Omni Chem Private Limited is a 50:50 joint venture that will manufacture pharmaceutical intermediates and APIs in a green field facility in Visakhapatnam (AP) and mainly focus on high–value, low–volume APIs for the regulated markets.
The joint venture Company is setting up 100% export oriented unit at Jawaharlal Nehru Pharma city, Parwada Mandal, Visakhapatnam (AP) under the APIIC special economic zone to manufacture active pharmaceutical ingredients. The Company plans to obtain all necessary regulatory approvals from the U.S. Food and Drug Administration ("FDA") and other regulatory authorities by 31st March 2016.
AMALGAMATION OF
AUCTUS PHARMA LIMITED (As on 31.03.2014)
The Board of Directors of the Company at its meeting dated 20th February 2014 have approved the Scheme of Arrangement for amalgamation of Auctus Pharma Limited with the Company with effect from the "Appointed Date" i.e 1st April 2013 or such other date as may be fixed by the Hon'ble High Court of Judicature at Hyderabad for the State of Telangana and the State of Andhra Pradesh. Since both Companies are dealing in the similar line of business, the amalgamation will reap the following benefits for the Company:
- Focused strategic leadership and integrated business synergies.
- Channelized synergies; optimum utilization of the available resources and broadened customer base.
- Higher long–term financial returns and inculcation of greater financial strength and flexibility.
- Diversified product portfolio which would enable to manage risks in a better way.
- Manufacturing of value added products for fetching higher returns.
- Greater rationalization and reduction of duplication of systems and processes.
- Efficient and more cost effective management system in view of consolidation of operations.
- Pooling of financial, managerial and technical resources, personnel capabilities, skills, expertise and technologies of the merging companies leading to increased competitive strength.
The financial position of Granules India Limited will continue to remain strong and in light of the benefits flowing from the amalgamation and shall be strengthened by the vesting of the assets of the Auctus Pharma Limited under the Scheme of Arrangement. Hence, the rights and interests of the shareholders and creditors of Granules India Limited will not be prejudicially affected by the Scheme. The Scheme of Arrangement has been drawn up in consonance with the provisions of securities laws or the stock exchange requirements including the SEBI Act, 1992, the Securities Contracts (Regulation) Act, 1956, requirements under SEBI Circular No. CIR/CFD/DIL/5/2013 dated February 4, 2013 read with Circular No. CIR/CFD/ DIL/8/2013 dated 21st May 2013, the Depositories Act, 1996 and the provisions of the Companies Act, 1956, which are administered by SEBI and the rules, regulations, guidelines etc. made under these Acts and the Listing Agreement. The National Stock Exchange of India Limited (NSE) and the Bombay Stock Exchange Limited (BSE) have respectively conveyed their 'No Objection' as required under clause 24(f) of the Listing Agreement vide their Observation Letter NSE/LIST/239123–K dated 19th May 2014 and Observation Letter DCS/AMAL/ LP/24(f)/047/2014–15 dated 16 May 2014 for filing the Scheme of Arrangement with the Hon'ble High Court.
The Company Auctus Pharma Limited, being the applicant has filed the petition for amalgamation with High Court of Judicature of Andhra Pradesh and Telangana at Hyderabad under Sections 391 and 394 of the Companies Act, 1956 and other applicable provisions of the Act, rules and regulations and sanction of the High Court is still awaited.
MANAGEMENT DISCUSSION
AND ANALYSIS (As on 31.03.2014)
PRODUCT OVERVIEW
Granules’ focus is primarily on pharmaceutical products with high API and/or finished dosage volume requirements. In many high-volume pharmaceutical products, there are dozens of suppliers leading to oversupply. While there are large surpluses, high quality material for the regulated markets is in short supply and can only be serviced by a handful of suppliers.
Customers in the regulated markets and an increasingly growing number of customers in the semi-regulated markets require high-quality supplies. Due to increased accountability and consumer pressure, countries in the semi-regulated markets are implementing tighter controls and demanding increased stringent quality parameters.
Quality conscious customers value supply security and quality over pricing. Customers generally work closely with their suppliers’ regulatory and quality control departments. Once they select a supplier, the customer usually stays with the supplier for years and only periodically reviews alternatives.
In addition, branded formulation and branded generics manufacturers typically grow faster than the overall market since they are gaining market share at the expense of lesser known suppliers. As new consumers gain access to pharmaceutical products, they become likely to purchase the brand of a well-known manufacturer whether it is an innovator or a generics manufacturer.
PRODUCT BASKET (As on
31.03.2014)
ACTIVE PHARMACEUTICAL INGREDIENT (API)
The Company’s API portfolio went through a tremendous shift in FY14 due to the acquisition of Auctus Pharma. The acquisition will introduce more than a dozen API s across several therapeutic sectors including anti-histamine, antihypertensive, anti-fungal as well as others. The portfolio includes drugs such as Cetrizine, Clopidogrel Bisulphate, Fluconazole, Losartan Potassium, Rifaximin and Valsartan. The APIs will be made in a separate API facility in Vizag. These are higher in value than Granules’ existing portfolio and offer tremendous opportunity for the Company.
The Company’s core API portfolio continued to report double-digit growth despite the markets for most of these products reporting single-digit growth. Granules entered this segment in 1984 through the manufacture of Paracetamol APIs and expanded into other products such as Ibuprofen and Metformin.
Over the decades, the Company emerged among premier global API manufacturers due to a combination of scale, quality, compliance and reliability. Going forward, more of the APIs from the core portfolio will be used for captive consumption whether it is for PFIs or Finished Dosages. The API vertical will continue to be critical to the Company’s success since it will continue to represent the starting point of the Company’s PFI and FD verticals.
PHARMACEUTICAL
FORMULATION INTERMEDIATES (PFI) (As on 31.03.2014)
Granules
pioneered the concept of commercializing PFIs, saving customers the need to
manufacture their own PFIs and leaving them free to focus on finished dosage
manufacturing and marketing.
Granules
entered this business segment in the early- 90s following an insight that most
Finished Dosage manufacturers were not efficiently producing PFIs.
Manufacturers were granulating but not able to derive operational efficiencies
because they focused on dozens of products or only created the product in
limited runs. This pulled down the overall return from their investments,
especially because PFI manufacture accounts for 80% of the total cost of a
finished dosage.
The Company,
through its PFI facilities at Gagillapur and Jeedimetla, is a leader in
manufacturing PFIs and has the world’s largest capacity. The manufacturing facility
uses high-shear and fluid-bed granulation processes with a 6 ton batch size,
the largest in the industry. The capacity at the Gagillapur facility was
expanded in FY14 and continues to be de-bottlenecked.
FINISHED DOSAGE (FD)
(As on 31.03.2014)
At Granules, the manufacture of finished dosages represents the apex of the value chain. The Company entered this business in FY09 with an installed capacity of six billion tablets and expanded it to eighteen billion tablets in FY14. The business accounts for 35% of the Company’s standalone revenue.
Granules’ finished dosage facility in Gagillapur comprises automated processes, robust infrastructure and superior quality systems that efficiently produce finished dosages. Granules offers multiple finished dosage forms comprising tablets, caplets and press fit capsules in bulk, blister packs and bottles.
INDEX
OF CHARGES
|
S.No. |
Charge ID |
Date of Charge Creation/Modification |
Charge amount secured |
Charge Holder |
Address |
Service Request Number (SRN) |
|
1 |
10540765 |
30/12/2014 |
200,000,000.00 |
ANDHRA BANK |
PB 34,H.No.6-3-352/2,Astral Heights, Upper GroundF, Road No.1, Banjara
Hills, Hyderabad, Telangana |
C39301502 |
|
2 |
10476418 |
08/02/2014 |
1,645,000,000.00 |
ANDHRA BANK |
Somajiguda Branch, 6-3-352/2, Astral Heights,, Road No. 1, Banjara
Hills, Hyderabad, Andhra Pradesh |
B95849865 |
|
3 |
10389714 |
30/11/2012 |
527,500,000.00 |
DEG - DEUTSCHE INVESTITIONS- UND ENTWICKLUNGSGESEL |
KAMMERGASSE 22, COLOGNE, - 50676, GERMANY |
B63236012 |
|
4 |
10389717 |
30/11/2012 |
527,500,000.00 |
INTERNATIONAL FINANCE CORPORATION |
2121 PENNSYLVANIA AVENUE, N.W., WASHINGTON DC, WASHINGTON DC, - 20433,
UNITED STATES OF AMERICA |
B63236624 |
|
5 |
10319592 |
22/11/2011 |
49,150,000.00 |
INTERNATIONAL FINANCE CORPORATION |
2121 PENNSYLVANIA AVENUE, N.W., WASHINGTON DC, 20433, WASHINGTON DC, -
110021, UNITED STATES OF AMERICA |
B26012047 |
|
6 |
10082831 |
22/12/2007 |
360,000,000.00 |
International Finance Coporation |
IFC's South Asia Department, Gate No.3, Niti Mag, 50-M,
Shanthipath, Chanakyapuri,, New Delhi - 110021, INDIA |
A30260624 |
|
7 |
90139379 |
24/03/2014 * |
4,710,100,000.00 |
ANDHRA BANK |
Somajiguda Branch, 6-3-352/2, Astral Heights,, Road No. 1, Banjara
Hills, Hyderabad, Andhra Pradesh |
C03962818 |
|
8 |
90132933 |
22/06/2001 |
5,000,000.00 |
THE A.P.VARDHAMAN(MAHILA) CO-OP URBAN BANK |
3-5-141/F/7; GROUND FLOOR, GEETANJALI COMPLEX, HYDERABAD, Andhra
Pradesh, INDIA |
- |
|
9 |
90131139 |
11/01/1995 |
861,250.00 |
A.P.STATE FINANCIAL CORPORATION |
SANGAREDDY BRANCH, SANGAREDDY, Andhra Pradesh, INDIA |
- |
FIXED ASSETS
Tangible Assets
·
Land
·
Buildings
·
Plant and Machinery
·
Computers
·
Office Equipment
·
R&D Equipment
·
Furniture and Fixtures
·
Vehicles
Intangible Assets
·
Technical Knowhow
CMT REPORT (Corruption, Money Laundering & Terrorism]
The Public Notice information has been collected from various sources
including but not limited to: The Courts,
1] INFORMATION ON
DESIGNATED PARTY
No exist designating subject or any of its beneficial owners,
controlling shareholders or senior officers as terrorist or terrorist
organization or whom notice had been received that all financial transactions
involving their assets have been blocked or convicted, found guilty or against
whom a judgement or order had been entered in a proceedings for violating
money-laundering, anti-corruption or bribery or international economic or
anti-terrorism sanction laws or whose assets were seized, blocked, frozen or
ordered forfeited for violation of money laundering or international
anti-terrorism laws.
2] Court Declaration :
No exist to suggest that subject is or was
the subject of any formal or informal allegations, prosecutions or other
official proceeding for making any prohibited payments or other improper
payments to government officials for engaging in prohibited transactions or
with designated parties.
3] Asset Declaration :
No records exist to suggest that the property or assets of the subject
are derived from criminal conduct or a prohibited transaction.
4] Record on Financial
Crime :
Charges or conviction
registered against subject: None
5] Records on Violation of
Anti-Corruption Laws :
Charges or investigation
registered against subject: None
6] Records on Int’l
Anti-Money Laundering Laws/Standards :
Charges or
investigation registered against subject: None
7] Criminal Records
No available
information exist that suggest that subject or any of its principals have been
formally charged or convicted by a competent governmental authority for any
financial crime or under any formal investigation by a competent government
authority for any violation of anti-corruption laws or international anti-money
laundering laws or standard.
8] Affiliation with
Government :
No record
exists to suggest that any director or indirect owners, controlling
shareholders, director, officer or employee of the company is a government
official or a family member or close business associate of a Government
official.
9] Compensation Package :
Our market
survey revealed that the amount of compensation sought by the subject is fair
and reasonable and comparable to compensation paid to others for similar
services.
10] Press Report :
No press reports / filings exists on
the subject.
CORPORATE GOVERNANCE
MIRA INFORM as part of its Due Diligence do provide comments on Corporate
Governance to identify management and governance. These factors often have been
predictive and in some cases have created vulnerabilities to credit
deterioration.
Our Governance Assessment focuses principally on the interactions
between a company’s management, its Board of Directors, Shareholders and other
financial stakeholders.
CONTRAVENTION
Subject is not known to have contravened any existing local laws,
regulations or policies that prohibit, restrict or otherwise affect the terms and
conditions that could be included in the agreement with the subject.
FOREIGN EXCHANGE RATES
|
Currency |
Unit
|
Indian Rupees |
|
US Dollar |
1 |
Rs.63.57 |
|
|
1 |
Rs.99.10 |
|
Euro |
1 |
Rs.70.29 |
INFORMATION DETAILS
|
Information Gathered
by : |
MNJ |
|
|
|
|
Analysis Done by
: |
SAN |
|
|
|
|
Report Prepared
by : |
SUJ |
SCORE & RATING EXPLANATIONS
|
SCORE FACTORS |
RANGE |
POINTS |
|
HISTORY |
1~10 |
6 |
|
PAID-UP CAPITAL |
1~10 |
6 |
|
OPERATING SCALE |
1~10 |
6 |
|
FINANCIAL CONDITION |
|
|
|
--BUSINESS SCALE |
1~10 |
6 |
|
--PROFITABILITY |
1~10 |
6 |
|
--LIQUIDITY |
1~10 |
5 |
|
--LEVERAGE |
1~10 |
5 |
|
--RESERVES |
1~10 |
5 |
|
--CREDIT LINES |
1~10 |
5 |
|
--MARGINS |
-5~5 |
-- |
|
DEMERIT POINTS |
|
|
|
--BANK CHARGES |
YES/NO |
YES |
|
--LITIGATION |
YES/NO |
YES |
|
--OTHER ADVERSE INFORMATION |
YES/NO |
NO |
|
MERIT POINTS |
|
|
|
--SOLE DISTRIBUTORSHIP |
YES/NO |
NO |
|
--EXPORT ACTIVITIES |
YES/NO |
YES |
|
--AFFILIATION |
YES/NO |
YES |
|
--LISTED |
YES/NO |
YES |
|
--OTHER MERIT FACTORS |
YES/NO |
YES |
|
DEFAULTER |
|
|
|
--RBI |
YES/NO |
NO |
|
--EPF |
YES/NO |
NO |
|
TOTAL |
|
50 |
This score serves as a reference to assess
SC’s credit risk and to set the amount of credit to be extended. It is calculated
from a composite of weighted scores obtained from each of the major sections of
this report. The assessed factors and their relative weights (as indicated
through %) are as follows:
Financial
condition (40%) Ownership
background (20%) Payment record
(10%)
Credit history
(10%) Market trend (10%) Operational size
(10%)
RATING EXPLANATIONS
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
>86 |
Aaa |
Possesses an extremely sound financial base with the strongest capability
for timely payment of interest and principal sums |
Unlimited |
|
71-85 |
Aa |
Possesses adequate working capital. No caution needed for credit
transaction. It has above average (strong) capability for payment of interest
and principal sums |
Large |
|
56-70 |
A |
Financial & operational base are regarded healthy. General
unfavourable factors will not cause fatal effect. Satisfactory capability for
payment of interest and principal sums |
Fairly Large |
|
41-55 |
Ba |
Overall operation is considered normal. Capable to meet normal
commitments. |
Satisfactory |
|
26-40 |
B |
Capability to overcome financial difficulties seems comparatively
below average. |
Small |
|
11-25 |
Ca |
Adverse factors are apparent. Repayment of interest and principal sums
in default or expected to be in default upon maturity |
Limited with
full security |
|
<10 |
C |
Absolute credit risk exists. Caution needed to be exercised |
Credit not
recommended |
|
-- |
NB |
New Business |
-- |
This report is issued at your request without any
risk and responsibility on the part of MIRA INFORM PRIVATE LIMITED (MIPL)
or its officials.