MIRA INFORM REPORT
|
Report No.: |
327475 |
|
Report Date : |
17.06.2015 |
IDENTIFICATION DETAILS
|
Name : |
CADILA HEALTHCARE LIMITED |
|
|
|
|
Registered Office
: |
‘Zydus Tower’, Satellite Cross Roads, Sarkhej –
Gandhinagar Highway, Ahmedabad – 380015, Gujarat |
|
Tel. No.: |
91-79-26868100 |
|
|
|
|
Country : |
India |
|
|
|
|
Financials (as
on) : |
31.03.2015 |
|
|
|
|
Date of
Incorporation : |
15.05.1995 |
|
|
|
|
Com. Reg. No.: |
04-025878 |
|
|
|
|
Capital
Investment / Paid-up Capital : |
Rs. 1023.700 Million |
|
|
|
|
CIN No.: [Company Identification
No.] |
L24230GJ1995PLC025878 |
|
|
|
|
IEC No.: |
Not Available |
|
|
|
|
TAN No.: [Tax Deduction & Collection
Account No.] |
AHMC00020G |
|
|
|
|
PAN No.: [Permanent Account No.] |
AAACC6253G |
|
|
|
|
Legal Form : |
A Public Limited Liability Company. The Company’s Shares are Listed on
the Stock Exchanges. |
|
|
|
|
Line of Business
: |
Development, Production, Marketing and Distribution of
Pharmaceutical Products. |
|
|
|
|
No. of Employees
: |
Information declined by the management |
RATING & COMMENTS
|
MIRA’s Rating : |
Aa (76) |
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
71-85 |
Aa |
Possesses adequate working capital. No caution needed for credit
transaction. It has above average (strong) capability for payment of interest
and principal sums |
Large |
|
Maximum Credit Limit : |
USD 129000000 |
|
|
|
|
Status : |
Excellent |
|
|
|
|
Payment Behaviour : |
Regular |
|
|
|
|
Litigation : |
Exist |
|
|
|
|
Comments : |
Subject is a well-established and reputed company having an excellent
track record. Financial position of the company is sound. Fundamentals of the company
are strong and healthy. Share price are quoted high on stock exchange (Face Value Rs 5 with
Share price Rs 1752) Directors are reported to be trustworthy. Business is active. Payments
are reported to be regular and as per commitment. The company can be considered good for business dealings at usual
trade terms and condition It can be regarded as a promising business partner in medium to long
run. |
NOTES :
Any query related to this report can be made
on e-mail : infodept@mirainform.com
while quoting report number, name and date.
ECGC Country Risk Classification List – March 31, 2015
|
Country Name |
Previous Rating (31.12.2014) |
Current Rating (31.03.2015) |
|
India |
A1 |
A1 |
|
Risk Category |
ECGC
Classification |
|
Insignificant |
A1 |
|
Low |
A2 |
|
Moderate |
B1 |
|
High |
B2 |
|
Very High |
C1 |
|
Restricted |
C2 |
|
Off-credit |
D |
EXTERNAL AGENCY RATING
|
Rating Agency Name |
Not Available |
|
Rating |
Not Available |
|
Rating Explanation |
Not Available |
|
Date |
Not Available |
RBI DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available RBI Defaulters’ list.
EPF (Employee Provident Fund) DEFAILTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available EPF (Employee Provident Fund) Defaulters’ list as of
31-03-2013.
INFORMATION DENIED
Management non co-operative (Tel. No.: 91-79-26868100)
LOCATIONS
|
Registered/ Corporate
Office : |
‘Zydus Tower’, Satellite Cross Roads, Sarkhej – Gandhinagar Highway, Ahmedabad – 380015, Gujarat, India |
|
Tel. No.: |
91-79-26770100 (EPBX) (20 Lines)/ 26868100/ 26868235 |
|
Fax No.: |
91-79-26732365/ 26732366/ 26862365 |
|
E-Mail : |
investor.grievance@zyduscadila.com
|
|
Website : |
|
|
|
|
|
Factories : |
Formulation Unit:
Ø S. No.417, 419 and 420, Village Moraiya, Taluka Sanand, District Ahmedabad, Gujarat, India Ø Kundaim Industrial Estate, Ponda – 403401, Goa, India Ø Village Saraj Mujra, P. O.– Baddi, Tehsil – Nalagarh, District – Solan, Himachal Pradesh, India |
|
|
|
|
Topical Plant: |
Plot No. 254/255, Behind Zyfine Chemicals, Sarkhej Bavla. N. H. No. 8A, Changodar Road, Taluka Sanand, District-Ahmedabad, Gujarat, India |
|
|
|
|
Zydus Biologics: |
Survey No. 40P, 23, 25P, 42, 37, Opposite Ramdev Masala, Sarkhej-Bavla N. H. No. 8A, Changodar, Ahmedabad, Gujarat, India |
|
|
|
|
API Units : |
Ø GIDC Estate, Ankleshwar, Gujarat, India Ø Dabhasa, Tal. Padra, District Vadodara, Gujarat, India Ø Block No. 162, Ekalbara Umraya Road, Village Dabhasa, Taluka Padra, District-Vadodara, Gujarat, India |
|
|
|
|
SEZ Unit: |
Plot No. 1/1A, and 2, PHARMEZ, Sarkhej-Bavla N. H. No. 8A, Village – Matoda, Taluka Sanad, District-Ahmedabad |
|
|
|
|
Zydus Research
Center : |
S. No. 396/403, Village Moraiya, Taluka Sanand, District Ahmedabad, Gujarat, India |
|
|
|
|
Registrar and Share Transfer
Agents : |
M/S. Sharepro Services (India) Private Limited, 416-420, 4th Floor, Devnandan Mall, Opp. Sanyas Ashram, Nr. M.J. Library, Ellisbridge, Ahmedabad - 380 009, Gujarat, India |
|
|
|
|
Zyfine and Topical Plant : |
Block No. 265/266, Village Changodar, Sarkhej Bavla N.H.No. 8a, Changodar Road, Taluka- Anand, District-Ahmedabad, Gujarat, India |
DIRECTORS
As on 31.03.2014
|
Name |
Mr. Ramanbhai B.
Patel |
|
Designation |
Founder |
|
Address |
16, Azad Society,
Ambawadi, Ahmedabad – 380015, Gujarat, India |
|
|
|
|
Name |
Mr. Pankaj R.
Patel |
|
Designation |
Chairman and
Managing Director |
|
|
|
|
Name |
Dr. Sharvil P.
Patel |
|
Designation |
Deputy Managing
Director |
|
Address |
16, Azad Society,
Ambawadi, Ahmedabad – 380015, Gujarat, India |
|
|
|
|
Name |
Mr. Humayun Dhanrajgiri |
|
Designation |
Director |
|
|
|
|
Name |
Mr. Mukesh M.
Patel |
|
Designation |
Director |
|
|
|
|
Name |
Mr. Nitin
Raojibhai Desai |
|
Designation |
Director |
|
|
|
|
Name |
Mr. Apurva S. Diwanji |
|
Designation |
Director |
|
|
|
|
Name |
Mrs. Dharmishta
N. Rawal |
|
Designation |
Director |
KEY EXECUTIVES
|
Name |
Mr. Upen H. Shah |
|
Designation |
Company Secretary |
|
|
|
|
Name |
Mr. Nitin D Parekh |
|
Designation |
Chief Financial Officer |
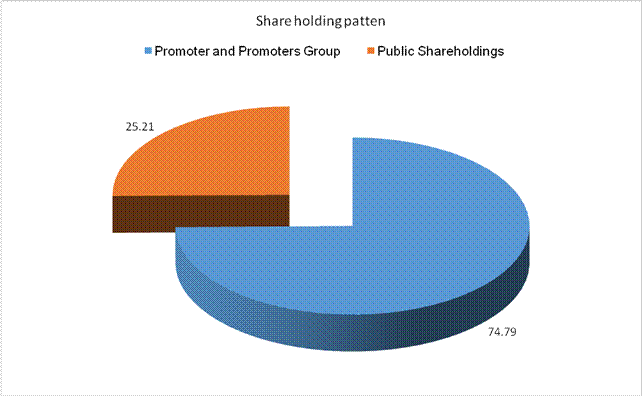
MAJOR SHAREHOLDERS / SHAREHOLDING PATTERN
AS ON 31.03.2015
|
Category of
Shareholder |
No. of Shares |
% of Holdings |
|
(A) Shareholding
of Promoter and Promoter Group |
||
|
|
|
|
|
|
153134446 |
74.79 |
|
|
3600 |
0.00 |
|
|
153138046 |
74.79 |
|
|
|
|
|
Total
shareholding of Promoter and Promoter Group (A) |
153138046 |
74.79 |
|
(B) Public
Shareholding |
||
|
|
|
|
|
|
8728746 |
4.26 |
|
|
368984 |
0.18 |
|
|
178295 |
0.09 |
|
|
7653925 |
3.74 |
|
|
13169657 |
6.43 |
|
|
48000 |
0.02 |
|
|
30147607 |
14.72 |
|
|
|
|
|
|
8738553 |
4.27 |
|
|
|
|
|
|
9185372 |
4.49 |
|
|
2690509 |
1.31 |
|
|
848433 |
0.41 |
|
|
848433 |
0.41 |
|
|
21462867 |
10.48 |
|
Total Public
shareholding (B) |
51610474 |
25.21 |
|
Total (A)+(B) |
204748520 |
100.00 |
|
(C) Shares held by
Custodians and against which Depository Receipts have been issued |
|
|
|
|
0 |
0.00 |
|
|
0 |
0.00 |
|
|
0 |
0.00 |
|
Total
(A)+(B)+(C) |
204748520 |
0.00 |
BUSINESS DETAILS
|
Line of Business : |
Development, Production, Marketing and Distribution of
Pharmaceutical Products. |
|
|
|
|
Products : |
Not Divulged |
|
|
|
|
Brand Names : |
Not Available |
|
|
|
|
Agencies Held : |
Not Available |
|
|
|
|
Exports : |
Not Divulged |
|
|
|
|
Imports : |
Not Divulged |
|
|
|
|
Terms : |
Not Divulged |
GENERAL INFORMATION
|
Suppliers : |
Not Divulged
|
|||||||||||||||||||||||||||||||||||||||||||||
|
|
|
|||||||||||||||||||||||||||||||||||||||||||||
|
Customers : |
Not Divulged
|
|||||||||||||||||||||||||||||||||||||||||||||
|
|
|
|||||||||||||||||||||||||||||||||||||||||||||
|
No. of Employees : |
Information declined by the management |
|||||||||||||||||||||||||||||||||||||||||||||
|
|
|
|||||||||||||||||||||||||||||||||||||||||||||
|
Bankers : |
·
Bank of Baroda ·
BNP Paribas ·
Credit Agricole Corporate and Investment Bank ·
Citibank N. A. ·
Exim Bank ·
HDFC Bank Limited ·
ICICI Bank Limited ·
IDBI Bank ·
State Bank of India ·
Standard Chartered Bank |
|||||||||||||||||||||||||||||||||||||||||||||
|
|
|
|||||||||||||||||||||||||||||||||||||||||||||
|
Facilities : |
|
|||||||||||||||||||||||||||||||||||||||||||||
|
Banking
Relations : |
|
|
|
|
|
Auditors : |
|
|
Name : |
Mukesh M. Shah and Company Chartered Accountants |
|
Address : |
3, H. K. House, Second Floor, Ashram Road, Ahmedabad – 380009, Gujarat, India |
|
|
|
|
Memberships : |
-- |
|
|
|
|
Collaborators : |
-- |
|
|
|
|
Subsidiary Companies/ Concerns: |
·
Dialforhealth India Limited ·
Zydus Pharmaceuticals (USA) Inc. [USA] ·
Dialforhealth Unity Limited ·
Nesher Pharmaceuticals (USA) LLC [USA] [Formerly
known as Zynesher Pharmaceuticals (USA) LLC] ·
Dialforhealth Greencross Limited ·
German Remedies Limited ·
Zydus Healthcare (USA) LLC [USA] ·
Zydus Pharmaceuticals Limited ·
Zydus Noveltech Inc. [USA] ·
Zydus Animal Health Limited ·
Zydus Healthcare S.A. (Pty) Limited [South
Africa] ·
Zydus Wellness Limited ·
Simayla Pharmaceuticals (Pty) Limited [South
Africa] ·
Zydus Wellness-Sikkim, a Partnership Firm Liva
Healthcare Limited ·
Script Management Services (Pty) Limited [South
Africa] ·
Zydus Nikkho Farmaceutica Ltda. [Brazil]
[Formerly known as Zydus Healthcare Brasil Ltda] ·
Zydus Technologies Limited ·
Biochem Pharmaceutical Industries Limited ·
Zydus Pharmaceuticals Mexico SA De CV [Mexico] ·
Finest Procuring Solutions Limited ·
Zydus Pharmaceuticals Mexico Services Company SA
De C.V.[Mexico] ·
Zydus Healthcare, a Partnership Firm Zydus
International Private Limited [Ireland] ·
ZAHL B.V. [the Netherlands] [Formerly known as
RFCL B.V.] ·
Zydus Netherlands B.V. [the Netherlands] ·
ZAHL Europe B.V. [the Netherlands] [Formerly
known as RFCL Europe B.V.] ·
Zydus France, SAS [France] ·
Etna Biotech S.R.L. [Italy] ·
Bremer Pharma GmbH [Germany] ·
Zydus Pharma Japan Company Limited [Japan] ·
Zydus Lanka (Private) Limited [Sri Lanka] ·
Laboratorios Combix S.L. [Spain] |
|
|
|
|
Joint Ventures : |
·
Zydus BSV Pharma Private Limited ·
Zydus Hospira Oncology Private Limited ·
Zydus Nycomed Healthcare Private Limited ·
Bayer Zydus Pharma Private Limited |
|
|
|
|
Enterprises significantly influenced by Directors and/ or their
relatives: |
·
Cadmach Machinery Company Private Limited ·
Western Ahmedabad Effluent Conveyance Company
Private Limited ·
Zydus Hospitals and Healthcare Research Private
Limited ·
Zydus Hospitals (Vadodra) Private Limited ·
Pripan Investment Private Limited ·
Zydus Hospitals (Rajkot) Private Limited ·
Zest Aviation Private Limited ·
MabS Biotech Private Limited ·
Zandra Infrastructure LLP ·
Zydus Infrastructure Private Limited ·
C. M. C. Machinery ·
Cadila Laboratories Private Limited ·
Cadam Enterprises |
CAPITAL STRUCTURE
As on 31.03.2014
Authorised Capital : Rs. 1725.000 Million
Issued, Subscribed & Paid-up Capital : Rs. 1023.743 Million
As on 31.03.2015
Authorised Capital :Not Available
Issued, Subscribed & Paid-up Capital : Rs. 1023.700
Million
FINANCIAL DATA
[all figures are
in Rupees Million]
ABRIDGED
BALANCE SHEET
|
SOURCES OF FUNDS |
31.03.2015 |
31.03.2014 |
31.03.2013 |
|
EQUITY AND
LIABILITIES |
|
|
|
|
(1)Shareholders' Funds |
|
|
|
|
(a) Share Capital |
1023.700 |
1024.000 |
1024.000 |
|
(b) Reserves & Surplus |
44229.500 |
35275.000 |
28091.000 |
|
(c) Money received against share warrants |
0.000 |
0.000 |
0.000 |
|
|
|
|
|
|
(2) Share Application money pending allotment |
0.000 |
0.000 |
0.000 |
|
Total
Shareholders’ Funds (1) + (2) |
45253.200 |
36299.000 |
29115.000 |
|
|
|
|
|
|
(3)
Non-Current Liabilities |
|
|
|
|
(a) long-term borrowings |
5835.600 |
8536.000 |
7941.000 |
|
(b) Deferred tax liabilities (Net) |
1236.400 |
1236.000 |
1236.000 |
|
(c) Other long term
liabilities |
239.100 |
206.000 |
272.000 |
|
(d) long-term
provisions |
805.000 |
468.000 |
348.000 |
|
Total Non-current
Liabilities (3) |
8116.100 |
10446.000 |
9797.000 |
|
|
|
|
|
|
(4) Current Liabilities |
|
|
|
|
(a) Short term borrowings |
6795.700 |
5576.000 |
8514.000 |
|
(b) Trade payables |
6713.200 |
4617.000 |
3607.000 |
|
(c) Other current liabilities |
3300.000 |
2986.000 |
2695.000 |
|
(d) Short-term
provisions |
3195.500 |
2507.000 |
1792.000 |
|
Total Current
Liabilities (4) |
20004.400 |
15686.000 |
16608.000 |
|
|
|
|
|
|
TOTAL |
73373.700 |
62431.000 |
55520.000 |
|
|
|
|
|
|
ASSETS |
|
|
|
|
(1) Non-current assets |
|
|
|
|
(a) Fixed Assets |
|
|
|
|
(i) Tangible assets |
21350.300 |
15392.000 |
14644.000 |
|
(ii) Intangible Assets |
0.000 |
249.000 |
327.000 |
|
(iii) Capital work-in-progress |
0.000 |
5306.000 |
4638.000 |
|
(iv)
Intangible assets under development |
0.000 |
0.000 |
0.000 |
|
(b) Non-current Investments |
17507.500 |
12653.000 |
10640.000 |
|
(c) Deferred tax assets (net) |
0.000 |
0.000 |
0.000 |
|
(d) Long-term Loan and Advances |
7542.600 |
8032.000 |
7187.000 |
|
(e) Other
Non-current assets |
0.000 |
0.000 |
0.000 |
|
Total Non-Current
Assets |
46400.400 |
41632.000 |
37436.000 |
|
|
|
|
|
|
(2) Current assets |
|
|
|
|
(a) Current investments |
4583.400 |
2924.000 |
2159.000 |
|
(b) Inventories |
8043.200 |
6635.000 |
5872.000 |
|
(c) Trade receivables |
10561.200 |
7220.000 |
6830.000 |
|
(d) Cash and cash equivalents |
1293.900 |
894.000 |
916.000 |
|
(e) Short-term loans and advances |
2018.500 |
2709.000 |
1910.000 |
|
(f) Other current assets |
473.100 |
417.000 |
397.000 |
|
Total Current
Assets |
26973.300 |
20799.000 |
18084.000 |
|
|
|
|
|
|
TOTAL |
73373.700 |
62431.000 |
55520.000 |
PROFIT
& LOSS ACCOUNT
|
|
PARTICULARS |
31.03.2015 |
31.03.2014 |
31.03.2013 |
|
|
|
SALES |
|
|
|
|
|
|
|
Revenue from operations |
14539.500 |
40421.000 |
35274.000 |
|
|
|
Other Income |
149.000 |
3086.000 |
529.000 |
|
|
|
TOTAL (A) |
14688.500 |
43507.000 |
35803.000 |
|
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
|
|
Cost of Materials Consumed |
2967.800 |
10681.000 |
9441.000 |
|
|
|
Purchases of Stock-in-Trade |
1266.600 |
3827.000 |
3324.000 |
|
|
|
Changes in Inventories of Finished goods, Work-in-progress and
Stock-in-Trade |
(53.600) |
(357.000) |
(90.000) |
|
|
|
Employee Benefits Expense |
1500.900 |
5251.000 |
4179.000 |
|
|
|
Other Expenses |
4228.200 |
12949.000 |
11357.000 |
|
|
|
TOTAL (B) |
9909.900 |
32351.000 |
28211.000 |
|
|
|
|
|
|
|
|
Less |
PROFIT
BEFORE INTEREST, TAX, DEPRECIATION AND AMORTISATION (A-B) (C) |
4778.600 |
11156.000 |
7592.000 |
|
|
|
|
|
|
|
|
|
Less |
FINANCIAL
EXPENSES (D) |
138.600 |
425.000 |
1110.000 |
|
|
|
|
|
|
|
|
|
|
PROFIT
BEFORE TAX, DEPRECIATION AND AMORTISATION (C-D) (E) |
4640.000 |
10731.000 |
6482.000 |
|
|
|
|
|
|
|
|
|
Less/ Add |
DEPRECIATION/
AMORTISATION (F) |
553.900 |
1274.000 |
1168.000 |
|
|
|
|
|
|
|
|
|
|
PROFIT BEFORE
TAX (E-F) (G) |
4086.100 |
9457.000 |
5314.000 |
|
|
|
|
|
|
|
|
|
Less |
TAX (H) |
615.500 |
421.000 |
328.000 |
|
|
|
|
|
|
|
|
|
|
PROFIT AFTER TAX
(G-H) (I) |
3470.600 |
9036.000 |
4986.000 |
|
|
|
|
|
|
|
|
|
Add |
PREVIOUS
YEARS’ BALANCE BROUGHT FORWARD |
17366.000 |
11896.000 |
10094.000 |
|
|
|
|
|
|
|
|
|
Less |
APPROPRIATIONS |
|
|
|
|
|
|
|
Transfer to General Reserve |
NA |
1000.000 |
1000.000 |
|
|
|
Transfer to Debenture Redemption Reserve |
NA |
450.000 |
450.000 |
|
|
|
Proposed Dividend |
NA |
1843.000 |
--- |
|
|
|
Interim Dividend |
NA |
---- |
1536.000 |
|
|
|
Corporate Dividend Tax on Proposed Dividend |
NA |
273.000 |
198.000 |
|
|
BALANCE CARRIED
TO THE B/S |
20836.600 |
17366.000 |
11896.000 |
|
|
|
|
|
|
|
|
|
|
EARNINGS IN
FOREIGN CURRENCY |
|
|
|
|
|
|
|
Export Earnings |
NA |
20038.000 |
15442.000 |
|
|
|
Royalty, Know-how, professional and consultation fees |
NA |
301.000 |
601.000 |
|
|
|
Other Earnings |
NA |
274.000 |
193.000 |
|
|
TOTAL EARNINGS |
NA |
20613.000 |
16236.000 |
|
|
|
|
|
|
|
|
|
|
IMPORTS |
|
|
|
|
|
|
|
Raw Materials |
NA |
2907.000 |
2360.000 |
|
|
|
Stores & Spares |
NA |
354.000 |
178.000 |
|
|
|
Capital Goods |
NA |
414.000 |
1112.000 |
|
|
|
Finished Goods |
NA |
303.000 |
223.000 |
|
|
|
Packing materials |
NA |
232.000 |
200.000 |
|
|
TOTAL IMPORTS |
NA |
4210.000 |
4073.000 |
|
|
|
|
|
|
|
|
|
|
Earnings Per
Share (Rs.) |
|
|
|
|
|
|
Basic
and Diluted EPS |
|
|
|
|
|
|
- Before
Exceptional Items |
16.95 |
44.13 |
24.35 |
|
|
|
- After
Exceptional Items |
16.95 |
44.13 |
24.35 |
|
CURRENT MATURITIES
OF LONG TERM DEBT DETAILS
|
Particulars |
31.03.2015 |
31.03.2014 |
31.03.2013 |
|
Current Maturities of Long term debt |
NA |
175.000 |
140.000 |
|
Cash generated from operations |
NA |
814.400 |
505.000 |
QUARTERLY RESULTS
|
PARTICULARS |
30.06.2014 (Unaudited) |
30.09.2014 (Unaudited) |
31.03.2014 (Unaudited) |
|
Net Sales |
11127.900 |
12528.100 |
14648.800 |
|
Total Expenditure |
8489.400 |
9288.800 |
9815.100 |
|
PBIDT (Excl OI) |
2638.500 |
3239.300 |
4833.700 |
|
Other Income |
163.400 |
1437.900 |
101.800 |
|
Operating Profit |
2801.900 |
4677.200 |
4935.500 |
|
Interest |
82.500 |
102.300 |
105.000 |
|
Exceptional Items |
0.000 |
0.000 |
0.000 |
|
PBDT |
2719.400 |
4574.900 |
4830.500 |
|
Depreciation |
529.500 |
522.400 |
512.700 |
|
Profit Before Tax |
2189.900 |
4052.500 |
4317.800 |
|
Tax |
217.000 |
396.400 |
706.200 |
|
Provisions and contingencies |
0.000 |
0.000 |
0.000 |
|
Profit After Tax |
1972.900 |
3565.100 |
3611.600 |
|
Extraordinary Items |
0.000 |
0.000 |
0.000 |
|
Prior Period Expenses |
0.000 |
0.000 |
0.000 |
|
Other Adjustments |
0.000 |
0.000 |
0.000 |
|
Net Profit |
1972.900 |
3565.100 |
3611.600 |
KEY
RATIOS
|
PARTICULARS |
|
31.03.2015 |
31.03.2014 |
31.03.2013 |
|
Net Profit Margin (PAT / Sales) |
(%) |
23.87 |
22.35 |
14.14 |
|
|
|
|
|
|
|
Operating Profit Margin (PBIDT/Sales) |
(%) |
32.87 |
27.60 |
21.52 |
|
|
|
|
|
|
|
Return on Total Assets (PBT/Total Assets} |
(%) |
7.31 |
21.27 |
13.21 |
|
|
|
|
|
|
|
Return on Investment (ROI) (PBT/Networth) |
|
0.09 |
0.26 |
0.18 |
|
|
|
|
|
|
|
Debt Equity Ratio (Total Debt /Networth) |
|
0.28 |
0.39 |
0.57 |
|
|
|
|
|
|
|
Current Ratio (Current Asset/Current Liability) |
|
1.35 |
1.33 |
1.09 |
STOCK
PRICES
|
Face Value |
Rs. 5.00/- |
|
Market Value |
Rs. 1752.95/- |
FINANCIAL ANALYSIS
[all figures are
in Rupees Million]
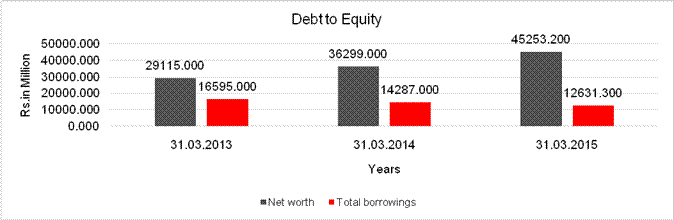
DEBT EQUITY RATIO
|
Particular |
31.03.2013 |
31.03.2014 |
31.03.2015 |
|
|
(Rs.
In Million) |
(Rs.
In Million) |
(Rs.
In Million) |
|
Share Capital |
1024.000 |
1024.000 |
1023.700 |
|
Reserves & Surplus |
28091.000 |
35275.000 |
44229.500 |
|
Share Application money
pending allotment |
0.000 |
0.000 |
0.000 |
|
Net
worth |
29115.000 |
36299.000 |
45253.200 |
|
|
|
|
|
|
long-term borrowings |
7941.000 |
8536.000 |
5835.600 |
|
Short term borrowings |
8514.000 |
5576.000 |
6795.700 |
|
Current Maturities of Long
Term Debt |
140.000 |
175.000 |
0.000 |
|
Total
borrowings |
16595.000 |
14287.000 |
12631.300 |
|
Debt/Equity
ratio |
0.570 |
0.394 |
0.279 |
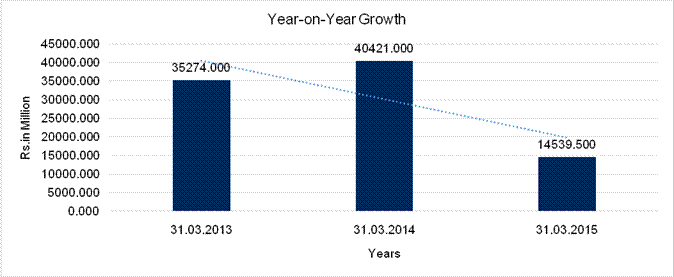
YEAR-ON-YEAR GROWTH
|
Year
on Year Growth |
31.03.2013 |
31.03.2014 |
31.03.2015 |
|
|
(Rs.
In Million) |
(Rs.
In Million) |
(Rs.
In Million) |
|
Sales |
35274.000 |
40421.000 |
14539.500 |
|
|
|
14.591 |
(64.030) |
NET PROFIT MARGIN
|
Net
Profit Margin |
31.03.2013 |
31.03.2014 |
31.03.2015 |
|
|
(Rs.
In Million) |
(Rs.
In Million) |
(Rs.
In Million) |
|
Sales |
35274.000 |
40421.000 |
14539.500 |
|
Profit |
4986.000 |
9036.000 |
3470.600 |
|
|
14.14% |
22.35% |
23.87% |
C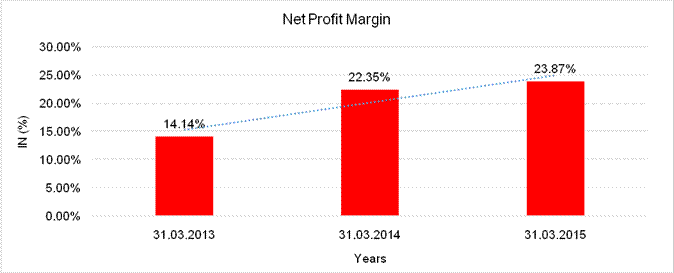
LOCAL AGENCY FURTHER INFORMATION
|
Sr. No. |
Check list by
info agents |
Available in
Report (Yes/No) |
|
1 |
Year of establishment |
Yes |
|
2 |
Constitution of the entity -Incorporation
details |
Yes |
|
3 |
Locality of the entity |
Yes |
|
4 |
Premises details |
No |
|
5 |
Buyer visit details |
------ |
|
6 |
Contact numbers |
Yes |
|
7 |
Name of the person contacted |
No |
|
8 |
Designation of contact person |
No |
|
9 |
Promoter’s background |
No |
|
10 |
Date of Birth of Proprietor / Partners /
Directors |
No |
|
11 |
Pan Card No. of Proprietor / Partners |
No |
|
12 |
Voter Id Card No. of Proprietor / Partners |
No |
|
13 |
Type of business |
Yes |
|
14 |
Line of Business |
Yes |
|
15 |
Export/import details (if applicable) |
No |
|
16 |
No. of employees |
No |
|
17 |
Details of sister concerns |
Yes |
|
18 |
Major suppliers |
No |
|
19 |
Major customers |
No |
|
20 |
Banking Details |
Yes |
|
21 |
Banking facility details |
Yes |
|
22 |
Conduct of the banking account |
------ |
|
23 |
Financials, if provided |
Yes |
|
24 |
Capital in the business |
Yes |
|
25 |
Last accounts filed at ROC, if applicable |
Yes |
|
26 |
Turnover of firm for last three years |
Yes |
|
27 |
Reasons for variation <> 20% |
------ |
|
28 |
Estimation for coming financial year |
No |
|
29 |
Profitability for last three years |
Yes |
|
30 |
Major shareholders, if available |
Yes |
|
31 |
External Agency Rating, if available |
Yes |
|
32 |
Litigations that the firm/promoter
involved in |
Yes |
|
33 |
Market information |
------ |
|
34 |
Payments terms |
No |
|
35 |
Negative Reporting by Auditors in the
Annual Report |
No |
LITIGATION DETAILS:
HIGH COURT OF
GUJARAT
|
|
TAX APPEAL No. 379 of 2014 |
|
Status : PENDING |
( Converted from
: O/ST/1050/2014 ) |
CCIN No :
001092201400379 |
|
|
|||||
|
|||||
|
|||||
|
S.NO. |
Name of the Petitioner |
Advocate On Record |
|
1 |
COMMISSIONER OF INCOME TAX-I |
MRS MAUNA M BHATT for: Appellant(s) |
|
S.NO. |
Name of the Respondant |
Advocate On Record |
|
1 |
CADILA HEALTHCARE LTD |
RULE SERVED for :Opponent(s) |
|
|
|||
|
Presented On |
: 21/04/2014 |
Registered On |
: 23/04/2014 |
|
Bench Category |
: - |
District |
: AHMEDABAD |
|
Case Originated From |
: THROUGH HIS/HER/THEIR ADVOCATE |
Listed |
: 141 times |
|
StageName |
: ADMISSION (FRESH MATTERS) |
||
|
Classification |
·
DB - OJ - TAX APPEAL - INCOME TAX ACT, 1961 - APPEAL
TO HIGH COURT - U/S 260 OF IT ACT - LONG TERM CAPITAL GAIN - U/S 54EA |
|
Act |
·
INCOME-TAX ACT, 1961 |
Office
Details
|
|
S.
No. |
Filing
Date |
Document
Name |
Advocate
Name |
Court
Fee on Document |
Document
Details |
|
1 |
21/04/2014 |
APPEARANCE NOTE |
MRS MAUNA M BHATT ADVOCATE |
0 |
MRS MAUNA M
BHATT(174) for P:1 |
|
2 |
21/04/2014 |
MEMO OF APPEAL/PETITION/SUIT |
MRS MAUNA M BHATT ADVOCATE |
20 |
MRS MAUNA M
BHATT(174), for P:1 |
|
3 |
21/04/2014 |
CERTIFIED COPY |
MRS MAUNA M BHATT ADVOCATE |
60 |
MRS MAUNA M
BHATT(174), for P:1 |
|
4 |
25/04/2014 |
VAKALATNAMA |
MR RK PATEL ADVOCATE |
5 |
MR RK
PATEL(1082) for R:1 |
Court
Proceedings
|
|||||
|
S.
No. |
Notified
Date |
CourtCode |
Board
Sr. No. |
Stage |
Action |
Coram |
|
1 |
28/04/2014 |
7 |
2- |
ADMISSION (FRESH MATTERS) |
RULE/ADMIT |
·
HONOURABLE MR.JUSTICE AKIL KURESHI ·
HONOURABLE MS JUSTICE SONIA GOKANI |
Available
Orders
|
||||||
|
S.
No. |
Case
Details |
Judge
Name |
Order
Date |
CAV |
Judgement |
Questions |
Transferred |
Download |
|
|
1 |
TAX APPEAL/379/2014 |
·
HONOURABLE MR.JUSTICE AKIL KURESHI ·
HONOURABLE MS JUSTICE SONIA GOKANI |
28/04/2014 |
N |
ORDER |
- |
Y |
Download |
|
Certified
Copy
|
|||||||||
|
S.
No. |
ApplicantName |
ApplicationType |
Application
Date |
UOL
Number |
Order
Date |
Notify
Date |
Delivery
Date |
Status |
Nature
Of Document |
|
1 |
MR RK PATEL |
ORDINARY |
29/04/2014 |
O/17809/2014 |
28/04/2014 |
01/05/2014 |
05/05/2014 |
Delivered |
ORDER |
|
2 |
MRS MAUNA M BHATT |
ORDINARY |
29/04/2014 |
O/17775/2014 |
28/04/2014 |
01/05/2014 |
02/05/2014 |
Delivered |
ORDER |
UNSECURED LOANS
|
PARTICULARS |
31.03.2014 |
31.03.2013 |
|
|
(Rs. In Millions) |
|
|
LONG TERM
BORROWINGS |
|
|
|
Term Loans from
Banks: |
|
|
|
External Commercial Borrowings in Foreign Currency [Unsecured] |
1199.000 |
1268.000 |
|
Deferred Payment
Liabilities [Unsecured]: |
|
|
|
Interest free deemed loan against deferment of sales tax: |
|
|
|
a) From a Financial Institution |
-- |
14.000 |
|
b) Deferred amount |
-- |
56.000 |
|
From Others [Unsecured] |
73.000 |
83.000 |
|
SHORT TERM
BORROWINGS |
|
|
|
Loans repayable on Demand: |
|
|
|
Working Capital Loans from Banks [Unsecured] [**] |
3956.000 |
4509.000 |
|
|
|
|
|
Total |
5228.000 |
5930.000 |
|
Terms of Repayment
for Unsecured Long Term Borrowings: Interest Free Deemed loan against deferment of sales tax from a financial institution is guaranteed personally by the Chairman and Managing Director of the Company and another loan is interest free sales tax deferred loan, repayable in six equal yearly installments starting from May 30, 2009. The outstanding amount as at March 31, 2014 is Rs. 71.000 [as at March 31, 2013: Rs. 141.000] Millions. Loan from Department of Science and Technology is repayable in ten yearly equal installments starting from November 1, 2012 along with interest @ 3% p.a. Interest accrued up to October 31, 2012 will be payable in 5 yearly installments along with repayment installment starting from November 1, 2012. The outstanding amount as at March 31, 2014 is Rs. 82.000 [as at March 31, 2013: Rs. 93.000] Millions. ECB of USD 10.000 Millions is repayable in three equal yearly installments starting from May 14, 2012 along with interest for the period. Interest rates are reset every six months at the rate of 6 months USD LIBOR plus 150 bps p.a. The outstanding amount as at March 31, 2014 is Rs. 200.000 [as at March 31, 2013: Rs. 363.000] Millions. ECB of USD 20.000 Millions is repayable in three yearly installments starting from December 26, 2016 along with interest for the period. The first installment of Rs. 360.000 Millions [USD 6 Millions] and the last two installments of Rs. 419.000 Millions [USD 7 Millions]. Interest rates are reset every month at the rate of 1 month USD LIBOR plus 223 bps p.a. The outstanding amount as at March 31, 2014 is Rs. 1199.000 [as at March 31, 2013: Rs. 1086.000] Millions. |
||
INDEX OF CHARGES
|
S.No. |
Charge ID |
Date of Charge Creation/Modification |
Charge amount secured |
Charge Holder |
Address |
Service Request Number (SRN) |
|
1 |
10516028 |
07/08/2014 |
930,000,000.00 |
CITI BANK N.A. |
CITIBANK BUILDING,
110, THOMPSON BOULEVARD,NASSU, |
C17406232 |
|
2 |
10504520 |
28/05/2014 |
1,204,040,000.00 |
THE BANK OF TOKYO-MITSUBISHI UFJ LIMITED |
9, RAFFLES PLACE,, # 01-01, REPUBLIC PLAZA,, SINGAPORE, - 048619, SINGAPORE |
C07604234 |
|
3 |
10465522 |
21/11/2013 |
945,000,000.00 |
CITI BANK N.A. |
CITIBANK BUILDING,110 THOMSON BOULEVARD, NASSU, - 0, BAHAMAS |
B91692251 |
|
4 |
10456406 |
10/10/2013 |
1,300,000,000.00 |
BARCLAYS BANK PLC |
1 CHURCHILL PLACE, E 14 5 HP, LONDON, - 1026167, UNITED KINGDOM |
B88096995 |
|
5 |
10430802 |
31/05/2013 |
1,124,860,000.00 |
THE BANK OF TOKYO-MITSUBISHI UFJ LTD |
9 RAFFLES PLACE, #01-01 REPUBLIC PLAZA, SINGAPORE - 048619, SINGAPORE |
B76984210 |
|
6 |
10396390 |
17/06/2014 * |
8,500,000,000.00 |
BANK OF BARODA |
ASHRAM RROAAD
BRANCH, VALLABH SADAN,, OPP. NATRAJ |
C08947178 |
|
7 |
10332648 |
24/01/2012 |
1,020,000,000.00 |
THE BANK OF NOVA SCOTIA ASIA LIMITED |
ONE REFFLES QUAY, #
20-01 NORTH TOWER, SINGAPORE, |
B30856926 |
|
8 |
10323573 |
12/12/2011 |
384,000,000.00 |
BANK OF AMERICA N.A. |
1ST FLOOR,DLF CENTRE, SANSAD MARG, NEW DELHI - 110001, DELHI, INDIA |
B27990894 |
|
9 |
10201696 |
19/02/2010 |
500,000,000.00 |
IDBI TRUSTEESHIP SERVICES LIMITED |
ASIAN BLDG., GROUND
FLOOR, 17, R.KAMANI MARG,, BA |
A79162178 |
ECONOMY AND
INDUSTRY OVERVIEW
GLOBAL ECONOMY AND
PHARMACEUTICAL INDUSTRY
The global economy appears to be on the path of revival with high income economies showing signs of firm recovery after years of low growth and/or recession. This recovery in the high-income economies is expected to be a major contributor to the projected acceleration in the global growth. In these countries, the drag on growth from fiscal consolidation and policy uncertainty is easing and private sector recoveries are gaining firmer footing. This likely acceleration in the global growth marks a significant shift from recent years when developing countries alone pushed the global economy forward. In addition to providing a sound basis for global growth, stronger high income growth and import demand are likely to be important drivers of developing countries’ exports. Economic activity and business sentiments improved in the developing countries from mid-2013 on the back of strengthening high-income demand and the rebound in China. This positive development though, was partially offset by tighter financial conditions and reduced capital flows. Increase in long-term interest rates in the United States in response to the expectations of the gradual withdrawal of quantitative easing was one of the key factors responsible for this.
The growth registered by the developing countries in 2013 was almost the same as the growth registered by them in 2012. However, some of the regions and/or countries of the developing world displayed significant variation in the growth rates registered in 2013 vis-à-vis 2012. While the countries of the developing Eastern Europe, Central Asia and Sub-Saharan Africa witnessed significant increase in the GDP growth in 2013 vis-à-vis 2012, countries of Middle East and North Africa registered significant decline in GDP growth over the same period.
The global pharmaceutical industry is estimated to have grown by approximately 3% in 2013 and reached a size of over US$ 975 bn. The growth in 2013 was lower compared to the growth of over 5% registered during the period 2007 to 2012. Healthcare spending in the US, the world's largest pharmaceutical market, as well as in Europe, declined marginally during 2013, while the Japanese pharmaceutical market grew by over 2.5% in 2013. On the other hand, developing markets of Asia, Africa, Australia and Latin America, which are termed as 'pharmerging' markets continued to grow by 10-14% in 2013, driving the overall growth of the global pharmaceutical industry.
Generic drugs continued to increase their share in the overall prescription for pharmaceutical drugs dispensed across the globe, though the growth in 2013 was impacted by relatively lower value of innovator drugs losing their patent protection compared to that in 2012. In the US, generic drugs now account for over 83% of the total prescriptions.
INDIAN ECONOMY AND
PHARMACEUTICAL INDUSTRY
The Indian economy registered a growth of 4.9% during the financial year 2013-14, marginally better than the growth of 4.5% registered during the financial year 2012-13. However, the growth rate was much lower than the recent growth rates. Among the three sectors of the economy viz. agriculture, industry and services, the industrial sector was the worst performer as it grew by only 0.7% during the year 2013-14, mainly due to the de-growth registered by the manufacturing and mining sectors.
Overall inflationary pressure on the economy eased during the year. The average Wholesale Price Index (WPI) inflation rate for 2013-14 was 5.93% as against 7.35% during 2012-13, although it moved from below 5% during the initial part of the year to a peak of 7.52% in November 2013, from which it started a downward trend and fell below 6% in March 2014.
The financial year 2013-14 saw a steep depreciation of the Indian Rupee vis-à-vis global currencies. The exchange rate between the Indian Rupee and the US Dollar, which was at around Rs. 54 in the beginning of the financial year started an upward movement and reached a level of Rs. 68-69 in the month of August. From the month of October, the Indian Rupee started appreciating against the US Dollar but the exchange rate remained more or less in the range of Rs. 60-63 during the rest of the financial year.
The year 2013-14 turned out to be one of the worst phases for the Indian pharmaceutical market in the recent past as it grew by a mere 6.1% and reached a size of approximately Rs. 75,600 crores after growing well in excess of 10% during the previous few years (Source: AIOCD-AWACS Report, MAT March 2014). The Government of India notified the new Drug Price Control Order (DPCO) 2013 whereby it expanded the scope of the National List of Essential Medicines (NLEM), covering a larger number of medicines under the list and notified revised prices, mostly downward, for the same. This adversely impacted a large number of companies in the Indian pharmaceutical market as they realised lower prices as notified by the new DPCO 2013 on an increased number of products. The new DPCO, 2013 also adversely affected the volume growth during the year for the products covered under NLEM. During the year, the products covered under the NLEM saw an overall sales decline of approximately 8% whereas those out of the purview of the list grew by around 8.5%.
HOME MARKETS
INDIA FORMULATIONS
The year 2013-14 was a challenging one for the Company’s formulations business in India, currently the largest contributor to the topline. The price reductions imposed by the Government of India on the products covered under NLEM impacted both the volume and the value growth of these products. Despite this, the growth registered by the Company in the India Formulations Business was a little higher than the overall market growth of 6%.
The Company retained its leading positions in the participated market segments of cardiovascular, gastro intestinal, women's healthcare and respiratory therapeutic areas. The Company also gained 2 positions in the dermatology segment, from 6th rank last year to the 4th rank this year. 16 of the Company’s brands feature among the top 300 pharmaceutical brands in India.
The Company continued its drive of adding new products to the existing portfolio and launched more than 75 new products, including line extensions in India during the year. Of these, 19 were first-time launches. This included the launch of Lipaglyn (Saroglitazar), the first New Chemical Entity (NCE), discovered and developed indigenously by an Indian pharmaceutical company.
Overall, the Company’s formulations business in India posted sales of Rs. 24,644 Millions. During the year, up by 6.1% from Rs. 23,232 Millions last year.
US FORMULATIONS
The US remains the world’s largest pharmaceutical market accounting for more than 30% of the global market. The Company’soperations in the US are spearheaded by its wholly-owned subsidiary Zydus Pharmaceuticals (USA) Inc.
The Company is now ranked 8th amongst the US generic companies based on scripts (Source: IMS), moving two positions from the 10th rank last year.
The Company’s business in the US completed yet another year of successful operations and posted sales of Rs. 21,704 Mio., up by 44%. The Company launched 7 new products in the US during the year.
The Company filed a record 50 ANDAs with the USFDA during the year, taking the cumulative ANDA filings to 227.
The Company’s continuous endeavour to improve service levels coupled with a focus on quality has helped achieve high levels of customer satisfaction. The Company received the ‘Service Level Excellence’ award from a large US wholesaler with an overall customer service level exceeding 99%.
Going forward, the Company’s focus will continue to be on launching complex, difficult-to-make oral solids and formulations of other dosage forms like injectables, nasals, creams and ointments to further strengthen its position in the US generic market.
BRAZIL
Brazil, which is the largest pharmaceutical market in Latin America and one of the promising ‘pharmerging’ markets, has been undergoing the process of consolidation in the recent past. Entry of large distributors, big retail chains buying the smaller ones and the increased competition have posed new challenges for the existing companies. In 2013, the Brazilian pharmaceutical market grew by approximately 3%.
The overall market scenario and the lack of new product approvals posed several challenges to the Company during the year.Despite this, the Company continued its focus on brand building initiatives with an aspiration to be a leading player in the women's healthcare, respiratory, cardiovascular, diabetes and neuropsychiatry therapy segments. During the year, the Company took several initiatives in the areas of optimising the cost of operations, inventory management and sales force effectiveness.
The Company posted sales of Rs. 2,353 Millions. in Brazil during the year
MEXICO
The Mexican pharmaceutical market, valued at around US$ 10 bn., is the second largest pharmaceutical market in Latin America after Brazil. The Company is present in the Mexican pharmaceutical market through its subsidiary Zydus Pharmaceuticals Mexico S.A. de C.V.
The Company commenced commercial operations in Mexico in June 2013 and launched 7 products. During the year, the Company posted sales of Rs. 109 Millions in Mexico.
Going forward, the Company intends to expand its focus on the cardiovascular and the primary care specialty area by aggressively launching new products in the Mexican market.
OTHER MARKETS AND
BUSINESSES
EUROPE
The Company is present in the generic market of France through its subsidiary, Zydus France SAS, while in the Spanish generic market, its presence is marked by its subsidiary, Laboratorios Combix S.L. During the year, the French generic market grew by about 6%, reflecting a fall in the Government-led initiatives to increase generic penetration towards the later part of the year. The French generic market is likely to remain challenging as the French government introduces price cuts to keep the healthcare costs down. The Spanish generic market, which is highly competitive, grew by 12% during the year. Going forward, this market is expected to continue strengthening as the fiscal crisis has started to ease.
During the year, the Company consolidated its business in France by restructuring the product portfolio and customer base which helped in improving overall profitability. The Company launched 8 new products in France, including two Day-1 launches. The Spanish business registered growth mostly in line with the market growth rate. The Company launched 12 new products, including one Day-1 launch in the Spanish market during the year. In both these markets, the focus now is on improving the profitability further by increasing the proportion of products supplied from India.
Overall, the Company’s business in Europe posted sales of Rs. 3,902 Mio. up by 6% during the year.
EMERGING MARKETS OF
ASIA PACIFIC, AFRICA AND THE MIDDLE EAST
During the year, the Company continued to strengthen its branded generics business in the key markets of Asia Pacific, Africa and the Middle East. The Company launched over 35 new products in these markets during the year, including several firsts in these markets.
The Company posted sales of Rs. 3,592 Mio. in these markets during the year with a growth of 15% over the previous year.
JAPAN
During the year, the Company decided to discontinue its existing business in Japan, which was operated through its subsidiary Zydus Pharma Japan Company Limited
APIS
The Company’s APIs and intermediates’ business continued to provide support to the formulations business by providing key input materials in a timely and cost efficient manner for the Indian as well as the developed and emerging markets across the world and also contributed to the launch of new formulations in these markets. The API business also fulfilled the requirements of the external customers by supplying them APIs and intermediates in a timely manner and at the most competitive prices. During the year, the Company filed 10 more DMFs with the USFDA, taking the cumulative filings to 117.
During the year, the Company’s API business grew by 13% and posted sales of Rs. 3,497 Millions.
CONSUMER WELLNESS
The Company is present in the consumer wellness space through its subsidiary, Zydus Wellness Limited, which has three established brands viz. Sugar Free, a low calorie sugar substitute, EverYuth, a range of skincare products and Nutralite, a cholesterol free table spread.
During the year 2013-14, Sugar Free maintained its leadership position in the low calorie sugar substitute market with a market share of more than 93%, which virtually represents the entire low calorie sugar substitute market in India (Source: AC Nielsen).
The skincare category witnessed a slowdown primarily due to reduced discretionary spending on personal care products by consumers driven by high inflation, which has impacted all the three categories in which EverYuth has a presence viz. face wash, scrub and peel-off. The Company continued its thrust on new product launches with the launch of several variants in the existing categories.
Despite strong competition from lower priced regional brands, Nutralite continued to maintain its leadership position in the margarine category due to the focus on superior quality, high standards of customer service and launch of a lower priced variant of Nutralite called ‘Nutralite Yummy’ in North India.
During the year, Zydus Wellness Limited. registered a sales of Rs. 4,296 Millions. up by 5% and a net profit of Rs. 965 Millions. Marginally down by 1%.
ANIMAL HEALTH
With a strong presence, the Company ranks amongst the leading companies in India in the animal healthcare space. The Company has the distinction of launching one of the highest number of 'first time in India' products. During the year, the Company introduced a range of specialty products for companion animals through a separate division 'Petz' to focus on growing pet care needs. During the year, the Company launched 11 new products in the animal health market in India. Bremer Pharma GmbH, Germany, a subsidiary of the Company runs the global animal health business, which has a presence in more than 50 markets around the world.
On a consolidated basis, the Company’s animal health business posted sales of Rs. 2,754 Millions with a growth of 17% during the year.
FIXED ASSETS
· Freehold Land
· Leasehold Land
· Buildings
· Plant and Machinery
· Furniture and Fixture
· Office Equipments
· Vehicles
· Trademarks, Patents and Designs
· Technical Know-how
·
Commercial Rights
PRESS RELEASE
ZYDUS CADILA’S NET PROFIT SOARS TO RS. 11510.000 MILLION
Announcing the results for the year ended 31st March 2015, Zydus Cadila registered a Net Profit of Rs. 11510.000 Million, up by 43% from Rs. 804 crores. During the year, the company registered gross sales of Rs. 86580.000 Million, up by 20% from Rs. 72080.000 Million in the corresponding period last year, on a consolidated basis.
For the fourth quarter ended 31st March 2015, the group registered gross sales of Rs. 22970.000 Million, up by 18% from Rs. 19520.000 Million in the corresponding period last year on a consolidated basis. Net Profit for the same period stood at Rs. 3500.000 Million. The Board of Directors of the Company in their meeting proposed a dividend of 240%.
During the year, the group launched Exemptia, the world’s first biosimilar for Adalimumab which is the largest selling therapy worldwide. This biosimilar is a part of the group’s robust biologics programme which has the largest number of monoclonal antibodies under development in India. Exemptia is marketed by Zydus Biovation and Zydus BioNext - new divisions launched exclusively to market this ground breaking therapy for inflammatory arthritis and Inflammatory Bowel Disease. During the year, the group also launched SoviHep, the breakthrough therapy for Hepatitis C in alliance with Gilead Sciences.
Advancing its discovery research programme, the group has been exploring newer treatment options for its breakthrough drug LipaglynTM (Saroglitazar) and initiated Phase III trials of the molecule for patients suffering from lipodystrophy and nonalcoholic steatohepatitis (NASH), during the year. The group initiated Phase I clinical trials in the United States for ZYDPLA1, a novel, orally active, small molecule DPP-4 inhibitor to treat Type-2 Diabetes. Phase-I clinical trials are also underway in Australia for its IND - ZYAN 1, a HIF-PH inhibitor for treating anemia.
Strengthening its regulatory pipeline, the group filed 38 ANDAs during the year with the US FDA, taking the cumulative filings to 260. The group received 8 ANDA approvals during the year taking the total to 99 product approvals.
CMT REPORT (Corruption, Money Laundering & Terrorism]
The Public Notice information has been collected from various sources including
but not limited to: The Courts, India Prisons Service, Interpol, etc.
1] INFORMATION ON
DESIGNATED PARTY
No records exist designating subject or any of its beneficial owners,
controlling shareholders or senior officers as terrorist or terrorist organization
or whom notice had been received that all financial transactions involving
their assets have been blocked or convicted, found guilty or against whom a
judgement or order had been entered in a proceedings for violating
money-laundering, anti-corruption or bribery or international economic or
anti-terrorism sanction laws or whose assets were seized, blocked, frozen or
ordered forfeited for violation of money laundering or international
anti-terrorism laws.
2] Court Declaration :
No records exist to suggest that subject is
or was the subject of any formal or informal allegations, prosecutions or other
official proceeding for making any prohibited payments or other improper
payments to government officials for engaging in prohibited transactions or
with designated parties.
3] Asset Declaration :
No records exist to suggest that the property or assets of the subject
are derived from criminal conduct or a prohibited transaction.
4] Record on Financial
Crime :
Charges or conviction registered
against subject: None
5] Records on Violation of
Anti-Corruption Laws :
Charges or
investigation registered against subject: None
6] Records on Int’l
Anti-Money Laundering Laws/Standards :
Charges or
investigation registered against subject: None
7] Criminal Records
No
available information exist that suggest that subject or any of its principals
have been formally charged or convicted by a competent governmental authority for
any financial crime or under any formal investigation by a competent government
authority for any violation of anti-corruption laws or international anti-money
laundering laws or standard.
8] Affiliation with
Government :
No record
exists to suggest that any director or indirect owners, controlling
shareholders, director, officer or employee of the company is a government
official or a family member or close business associate of a Government
official.
9] Compensation Package :
Our market
survey revealed that the amount of compensation sought by the subject is fair
and reasonable and comparable to compensation paid to others for similar
services.
10] Press Report :
No press reports / filings exists on
the subject.
CORPORATE GOVERNANCE
MIRA INFORM as part of its Due Diligence do provide comments on
Corporate Governance to identify management and governance. These factors often
have been predictive and in some cases have created vulnerabilities to credit
deterioration.
Our Governance Assessment focuses principally on the interactions
between a company’s management, its Board of Directors, Shareholders and other
financial stakeholders.
CONTRAVENTION
Subject is not known to have contravened any existing local laws,
regulations or policies that prohibit, restrict or otherwise affect the terms
and conditions that could be included in the agreement with the subject.
FOREIGN EXCHANGE RATES
|
Currency |
Unit
|
Indian Rupees |
|
US Dollar |
1 |
Rs. 64.15 |
|
UK Pound |
1 |
Rs. 100.04 |
|
Euro |
1 |
Rs. 72.27 |
INFORMATION DETAILS
|
Information
Gathered by : |
SUV |
|
|
|
|
Analysis Done by
: |
DIV |
|
|
|
|
Report Prepared
by : |
JYO |
SCORE & RATING EXPLANATIONS
|
SCORE FACTORS |
RANGE |
POINTS |
|
HISTORY |
1~10 |
9 |
|
PAID-UP CAPITAL |
1~10 |
8 |
|
OPERATING SCALE |
1~10 |
8 |
|
FINANCIAL
CONDITION |
|
|
|
--BUSINESS SCALE |
1~10 |
9 |
|
--PROFITABILITY |
1~10 |
9 |
|
--LIQUIDITY |
1~10 |
8 |
|
--LEVERAGE |
1~10 |
8 |
|
--RESERVES |
1~10 |
9 |
|
--CREDIT LINES |
1~10 |
8 |
|
--MARGINS |
-5~5 |
-- |
|
DEMERIT POINTS |
|
|
|
--BANK CHARGES |
YES/NO |
YES |
|
--LITIGATION |
YES/NO |
YES |
|
--OTHER ADVERSE INFORMATION |
YES/NO |
NO |
|
MERIT POINTS |
|
|
|
--SOLE DISTRIBUTORSHIP |
YES/NO |
NO |
|
--EXPORT ACTIVITIES |
YES/NO |
NO |
|
--AFFILIATION |
YES/NO |
YES |
|
--LISTED |
YES/NO |
YES |
|
--OTHER MERIT FACTORS |
YES/NO |
YES |
|
DEFAULTER |
|
|
|
--RBI |
YES/NO |
NO |
|
--EPF |
YES/NO |
NO |
|
TOTAL |
|
76 |
This score serves as a reference to assess SC’s
credit risk and to set the amount of credit to be extended. It is calculated
from a composite of weighted scores obtained from each of the major sections of
this report. The assessed factors and their relative weights (as indicated
through %) are as follows:
Financial
condition (40%) Ownership
background (20%) Payment
record (10%)
Credit history
(10%) Market trend (10%) Operational size
(10%)
RATING EXPLANATIONS
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
>86 |
Aaa |
Possesses an extremely sound financial base with the strongest
capability for timely payment of interest and principal sums |
Unlimited |
|
71-85 |
Aa |
Possesses adequate working capital. No caution needed for credit
transaction. It has above average (strong) capability for payment of interest
and principal sums |
Large |
|
56-70 |
A |
Financial & operational base are regarded healthy. General
unfavourable factors will not cause fatal effect. Satisfactory capability for
payment of interest and principal sums |
Fairly Large |
|
41-55 |
Ba |
Overall operation is considered normal. Capable to meet normal
commitments. |
Satisfactory |
|
26-40 |
B |
Capability to overcome financial difficulties seems comparatively below
average. |
Small |
|
11-25 |
Ca |
Adverse factors are apparent. Repayment of interest and principal sums
in default or expected to be in default upon maturity |
Limited with
full security |
|
<10 |
C |
Absolute credit risk exists. Caution needed to be exercised |
Credit not
recommended |
|
-- |
NB |
New Business |
-- |
This report is issued at your request without any
risk and responsibility on the part of MIRA INFORM PRIVATE LIMITED (MIPL) or
its officials.