MIRA INFORM REPORT
|
Report No. : |
320762 |
|
Report Date : |
06.05.2015 |
IDENTIFICATION DETAILS
|
Name : |
AUROBINDO PHARMA LIMITED |
|
|
|
|
Registered
Office : |
Plot No. 2, Maithri Vihar, Behind Maithri Vanam, Ameerpet, Hyderabad – 500 038, Telangana |
|
|
|
|
Country : |
India |
|
|
|
|
Financials (as
on) : |
31.03.2014 |
|
|
|
|
Date of
Incorporation : |
26.12.1986 |
|
|
|
|
Com. Reg. No.: |
015190 |
|
|
|
|
Capital
Investment / Paid-up Capital : |
Rs. 291.465 Million |
|
|
|
|
CIN No.: [Company Identification
No.] |
L24239TG1986PLC015190 |
|
|
|
|
IEC No.: |
Not Available |
|
|
|
|
TAN No.: [Tax Deduction &
Collection Account No.] |
HYDA01477A |
|
|
|
|
Legal Form : |
A Public Limited Liability Company. The Company’s Shares are Listed on
the Stock Exchanges. |
|
|
|
|
Line of Business
: |
Manufacturing and Marketing Bulk Drugs, Formulations, Tablets and
Capsules, Syrups and Injectiables. |
|
|
|
|
No. of Employees
: |
9500 (Approximately) |
RATING & COMMENTS
|
MIRA’s Rating : |
Ba (53) |
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
41-55 |
Ba |
Overall operation is considered normal. Capable to meet normal
commitments. |
Satisfactory |
|
Maximum Credit Limit : |
USD 110000000 |
|
|
|
|
Status : |
Good |
|
|
|
|
Payment Behaviour : |
Regular |
|
|
|
|
Litigation : |
Exist |
|
|
|
|
Comments : |
Subject is a well-established company having a fine track record. Financial position of the company seems to be sound. Trade relations are fair. Business is active. Payment terms are
reported to be regular and as per commitments. The company can be considered good for business dealings at usual
trade terms and conditions. |
NOTES :
Any query related to this report can be made
on e-mail : infodept@mirainform.com
while quoting report number, name and date.
ECGC Country Risk Classification List – December 31, 2014
|
Country Name |
Previous Rating (30.09.2014) |
Current Rating (31.12.2014) |
|
India |
A1 |
A1 |
|
Risk Category |
ECGC
Classification |
|
Insignificant |
A1 |
|
Low |
A2 |
|
Moderate |
B1 |
|
High |
B2 |
|
Very High |
C1 |
|
Restricted |
C2 |
|
Off-credit |
D |
EXTERNAL AGENCY RATING
Not Available
RBI DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available RBI Defaulters’ list.
EPF (Employee Provident Fund) DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available EPF (Employee Provident Fund) Defaulters’ list as of
31-03-2014.
LOCATIONS
|
Registered Office/
Corporate Office : |
Plot No. 2, Maithri Vihar, Behind Maithri Vanam, Ameerpet, Hyderabad – 500 038, Telangana, India |
|
Tel. No.: |
91-40-23741083 / 23741084 / 23744919 / 66725000 / 66725401 / 23736370 |
|
Fax No.: |
91-40-23746833 / 23741080 / 23748112 / 23747340 |
|
E-Mail : |
|
|
Website : |
|
|
|
|
|
Corporate Office : |
Water Mark Building, Plot No.11, Survey No.9, Kondapur, Hitech City, Hyderabad - 500 084, Telangana, India. |
|
Tel. No.: |
91-40-66725000 |
|
Fax No.: |
91-40-23741080 / 23746833 |
|
|
|
|
Factory 1 : |
Survey No.379,385,386,388 to 396 and 269, Borpatla, Hatnoor Mandal, Medak District, 502 296, Telangana, India |
|
|
|
|
Factory 2 : |
Plot No.103/A and 104/A, SVCIE, Industrial Development Area, Bollaram, Jinnaram (Mandal) Medak District, 500 092, Telangana, India |
|
|
|
|
Factory 3 : |
Survey No.313 and 314 Bachupally, Quthubullapur Mandal, Range Reddy District, 500 090, Telangana, India |
|
|
|
|
Factory 4 : |
Plot No.4 in Survey No.151 and Plot Nos.34 to 48 in Survey No. part of 146, 150, 151, 152, 153 and 154 situated in Phase-III, SPIIC, EPIP, IDA, Pashamylaram, Patancheru Mandal, Medak District, 502 307, Telangana, India |
|
|
|
|
Factory 5 : |
Plot No.68 to 70, 73 to 91, 95, 96, 260 and 261 Industrial Development Area, Chemical Zone, Pashamylaram, Patancheru Mandal, Medak District, 502 307, Telangana, India |
|
|
|
|
Factory 6 : |
Survey No. 329/39 and 329/47, Chitkul Village, Patancheru Mandal, Medak District, 502 307, Telangana, India |
|
|
|
|
Factory 7 : |
Survey No. 411/P, 425/P, 434/P, 435/P and 458/P, Plot No.S1(Part), Special Economic Zone (Pharma), APIIC, Green Industrial Park, Polepally Village, Jedcherla Mandal, Mahaboob Nagar, 509 302, Telangana, India |
|
|
|
|
Factory 8 : |
Survey No.10 and 13, Gaddapothram, Industrial Development Area - Kazipally Industrial Area, Jinnaram Mandal, Medak District, 502 319, Telangana, India |
|
|
|
|
Factory 9 : |
Survey No.369, 370 371 and 374, Gundlamachanoor, Hatnoora Mandal, Medak District, 502 296, Telangana, India |
|
|
|
|
Factory 10 : |
Survey No.61-66, Industrial Development Area, Pydibhimavaram, Ranasthalam Mandal, Srikakulam, 532 409, Telangana, India |
|
|
|
|
Factory 11 : |
Survey No.314, Bachupally, Quthubullapur Mandal, Range Reddy District, 500 090, Telangana, India |
|
|
|
|
Factory 12 : |
JN Pharma City, Road No.10,11 and 19, 20, E Bonangi Village, Parawada, Visakhapatnam District, 531 021, Telangana, India |
|
|
|
|
Factory 13 : |
1128, RIICO
Phase-III, Bhiwadi, 301 019, Rajasthan, India (Sub-leased to Auronext Pharma
Private Limited, a subsidiary of the Company) |
|
|
|
|
APLRC – I |
Survey No.313 and 314 Bachupally, Quthubullapur Mandal, Ranga Reddy District - 500 090, Telangana, India |
|
|
|
|
APLRC - II |
Survey No.71 and 72, 502203, Telangana, India |
DIRECTORS
AS ON 31.03.2014
|
Name : |
Mr. K. Ragunathan |
|
Designation : |
Non-executive Director |
|
Date of Birth/Age : |
51 Years |
|
Experience : |
29 years |
|
|
|
|
Name : |
Mr. K. Nityananda Reddy |
|
Designation : |
Managing Director |
|
Date of Birth/Age : |
56 Years |
|
Qualification : |
Masters Degree in Science (Organic Chemistry) |
|
|
|
|
Name : |
Mr. N. Govindarajan, |
|
Designation : |
Managing Director |
|
Date of Birth/Age : |
46 Years |
|
Experience : |
21 years |
|
Qualification : |
B.E. (Mechanical) |
|
|
|
|
Name : |
Dr. M. Sivakumaran |
|
Designation : |
Whole-Time Director |
|
Date of Birth/Age : |
71 Years |
|
Qualification : |
Masters Degree in Science |
|
Experience : |
41 years |
|
|
|
|
Name : |
Mr. M. Madan Mohan Reddy |
|
Designation : |
Whole-Time Director |
|
Date of Birth/Age : |
54 Years |
|
Qualification : |
Masters Degree in Science (Organic
Chemistry) |
|
|
|
|
Name : |
Mr. P.V. Ramprasad Reddy |
|
Designation : |
Non-Executive Director and Promoter pf the Company |
|
Date of Birth/Age : |
56 Years |
|
Qualification : |
Post-Graduate |
|
|
|
|
Name : |
Mr. P. Sarath Chandra
Reddy |
|
Designation : |
Non-Executive Director |
|
Date of Birth/Age : |
29 Yeas |
|
Qualification : |
Graduate in Business Administration |
|
|
|
|
Name : |
Mr. M. Sitarama Murthy |
|
Designation : |
Non-Executive Director |
|
Date of Birth/Age : |
71 Years |
|
Qualification : |
Masters in Electronics |
|
|
|
|
Name : |
Dr. D. Rajagopala Reddy |
|
Designation : |
Non-Executive Director |
|
Date of Birth/Age : |
55 Years |
|
Qualification : |
Master's Degree in Science |
|
|
|
|
Name : |
Dr. C. Channa Reddy |
|
Designation : |
Non-Executive Director |
|
Date of Birth/Age : |
70 Years |
KEY EXECUTIVES
|
Name : |
Mr. Sudhir B Singhi (upto June 30, 2014) |
|
Designation : |
Chief Financial Officer |
|
|
|
|
Name : |
Mr. Subramanian Santhanam (From July 1, 2014) |
|
Designation : |
Chief Financial Officer |
|
|
|
|
Name : |
Mr. A. Mohan Rami Reddy |
|
Designation : |
Company Secretary |
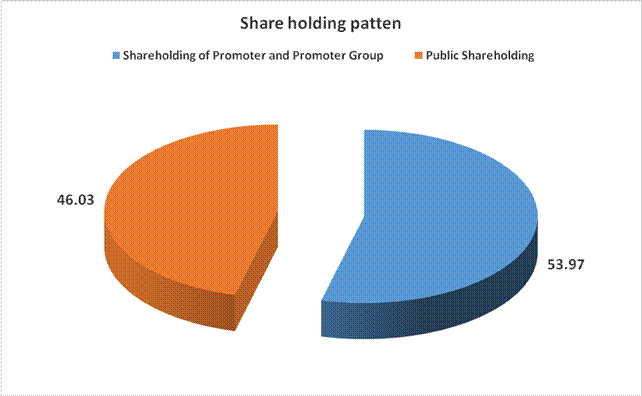
SHAREHOLDING PATTERN
As on 31.03.2015
|
Category
of Shareholder |
Total No.
of Shares |
Total
Shareholding as a % of Total No. of Shares |
|
As a % of (A+B) |
||
|
(A) Shareholding of Promoter
and Promoter Group |
||
|
|
|
|
|
|
139883428 |
47.91 |
|
|
8692358 |
2.98 |
|
|
148575786 |
50.89 |
|
|
|
|
|
|
9000000 |
3.08 |
|
|
9000000 |
3.08 |
|
Total shareholding of Promoter
and Promoter Group (A) |
157575786 |
53.97 |
|
(B) Public Shareholding |
||
|
|
|
|
|
|
17988239 |
6.16 |
|
|
181669 |
0.06 |
|
|
52951 |
0.02 |
|
|
86367982 |
29.58 |
|
|
104590841 |
35.82 |
|
|
|
|
|
|
4510378 |
1.54 |
|
|
|
|
|
|
16783102 |
5.75 |
|
|
6915881 |
2.37 |
|
|
1606287 |
0.55 |
|
|
1287804 |
0.44 |
|
|
311186 |
0.11 |
|
|
7297 |
0.00 |
|
|
29815648 |
10.21 |
|
Total Public shareholding (B) |
134406489 |
46.03 |
|
Total (A)+(B) |
291982275 |
100.00 |
|
(C) Shares held by Custodians and
against which Depository Receipts have been issued |
|
|
|
|
0 |
0.00 |
|
|
0 |
0.00 |
|
|
0 |
0.00 |
|
Total (A)+(B)+(C) |
291982275 |
100.00 |
BUSINESS DETAILS
|
Line of Business : |
Manufacturing and Marketing Bulk Drugs, Formulations, Tablets and
Capsules, Syrups and Injectiables. |
|
|
|
|
|
|
Products : |
Not Available |
|
|
|
|
|
|
Brand Names : |
Not Available |
|
|
|
|
|
|
Agencies Held : |
Not Available |
|
|
|
|
|
|
Exports : |
Not Available |
|
|
|
|
|
|
Imports : |
Not Available |
|
|
|
|
|
|
Terms : |
Not Available |
|
PRODUCTION STATUS: NOT AVAILABLE
GENERAL INFORMATION
|
Suppliers : |
Not Available |
|||||||||||||||||||||||||||||||||
|
|
|
|||||||||||||||||||||||||||||||||
|
Customers : |
Not Available |
|||||||||||||||||||||||||||||||||
|
|
|
|||||||||||||||||||||||||||||||||
|
No. of Employees : |
9500 (Approximately) |
|||||||||||||||||||||||||||||||||
|
|
|
|||||||||||||||||||||||||||||||||
|
Bankers : |
|
|||||||||||||||||||||||||||||||||
|
|
|
|||||||||||||||||||||||||||||||||
|
Facilities : |
LONG TERM
BORROWINGS:
|
|
|
|
|
Auditors : |
|
|
Statutory
Auditors : |
|
|
Name : |
S R Batliboi and Company Chartered Accountants |
|
Address : |
Oval Office, 18 iLabs Centre, Hi-tech City, Madhapur, Hyderabad – 500081, Andhra Pradesh, India |
|
Internal
Auditors : |
|
|
Name : |
KPMG Chartered Accountants |
|
Address : |
1st Floor, Lodha Excelus, Apollo Mills Compound, N M Joshi
Marg, Mahalakshmi, Mumbai – 400 011, |
|
|
|
|
Memberships : |
Not Divulged |
|
|
|
|
Collaborators : |
Not Divulged |
|
|
|
|
Subsidiaries |
|
|
|
|
|
Joint ventures |
|
|
|
|
|
Enterprises over
which key management personnel or their relatives exercise significant
influence |
|
CAPITAL STRUCTURE
AS ON 27.08.2014
Authorised Capital : Rs.
760.000 Million
Issued, Subscribed & Paid-up Capital : Rs. 291.796 Million
AS ON 31.03.2014
Authorised Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
660000000 |
Equity Shares |
Rs.1/- each |
Rs.660.000 Million |
|
1000000 |
Preference Shares |
Rs.100/- each |
Rs.100.000 Million |
|
|
Total |
|
Rs. 760.000
Million |
Issued, Subscribed & Paid-up Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
291457021 |
Equity Shares |
Rs.1/- each |
Rs.291.457 Million |
|
|
|
|
|
Reconciliation of the
equity shares outstanding at the beginning and at the end of the year
|
|
As at March 31, 2014 |
|
|
Numbers |
Value |
|
|
Equity Shares |
|
|
|
At the beginning of the year |
291211290 |
291.200 |
|
Issued during the year under Employee Stock Option Plan |
245731 |
0.300 |
|
Outstanding at the
end of the year |
291457021 |
291.500 |
Terms/rights attached
to equity shares
The Company has only one class of equity shares having a par values of `1 per share. Each holder of equity shares is entitled to one vote per share.
The Company declares and pays dividends in Indian rupees. The dividend proposed by the Board of Directors is subject to the approval of shareholders in the ensuing Annual General Meeting.
During the year ended March 31, 2014, the amount of dividend per share recognized as distributions to equity shareholders was Rs. 3 (March 31, 2013: Rs. 1.5) including interim dividend of Rs. 3 (March 31, 2013: Rs.1).
In the event of liquidation of the Company, the holders of equity shares will be entitled to receive remaining assets of the Company, after distribution of all preferential amounts. However, no such preferential amounts exist currently. The distribution will be in proportion to the number of equity shares held by the shareholders.
Details of
shareholders holding more than 5% equity shares in the Company
|
|
As at Mach 31, 2014 |
|
|
Numbers |
% holding |
|
|
Mr. P.V. Ramprasad Reddy |
19481440 |
6.68 |
|
Mrs. P. Suneela Rani |
90830550 |
31.16 |
|
Total |
110311990 |
|
As per of the Company, including its register of shareholders/members and other declarations received from shareholders regarding beneficial interest, the above shareholding represents both legal and beneficial ownerships of shares.
FINANCIAL DATA
[all figures are
in Rupees Million]
ABRIDGED BALANCE
SHEET
|
SOURCES OF FUNDS |
31.03.2014 |
31.03.2013 |
31.03.2012 |
|
|
|
|
|
|
I.
EQUITY AND LIABILITIES |
|
|
|
|
(1)Shareholders' Funds |
|
|
|
|
(a) Share Capital |
291.500 |
291.200 |
291.100 |
|
(b) Reserves &
Surplus |
39832.400 |
29099.900 |
24640.600 |
|
(c) Money received against
share warrants |
0.000 |
0.000 |
0.000 |
|
|
|
|
|
|
(2) Share Application
money pending allotment |
0.000 |
0.000 |
0.000 |
|
Total Shareholders’ Funds
(1) + (2) |
40123.900 |
29391.100 |
24931.700 |
|
|
|
|
|
|
(3) Non-Current Liabilities |
|
|
|
|
(a) long-term borrowings |
10323.100 |
10410.600 |
8337.400 |
|
(b) Deferred tax
liabilities (Net) |
2052.500 |
679.400 |
37.900 |
|
(c) Other long term
liabilities |
0.000 |
0.000 |
0.000 |
|
(d) long-term provisions |
83.000 |
85.000 |
41.200 |
|
Total Non-current
Liabilities (3) |
12458.600 |
11175.000 |
8416.500 |
|
|
|
|
|
|
(4) Current Liabilities |
|
|
|
|
(a) Short term borrowings |
17825.500 |
17339.000 |
16082.100 |
|
(b) Trade payables |
12361.600 |
9012.700 |
5848.500 |
|
(c) Other current
liabilities |
1923.900 |
680.400 |
4057.800 |
|
(d) Short-term provisions |
1061.800 |
647.500 |
575.500 |
|
Total Current Liabilities
(4) |
33172.800 |
27679.600 |
26563.900 |
|
|
|
|
|
|
TOTAL |
85755.300 |
68245.700 |
59912.100 |
|
|
|
|
|
|
II.
ASSETS |
|
|
|
|
(1) Non-current assets |
|
|
|
|
(a) Fixed Assets |
|
|
|
|
(i) Tangible assets |
19379.400 |
20119.100 |
16266.500 |
|
(ii) Intangible Assets |
0.000 |
0.000 |
0.900 |
|
(iii) Capital
work-in-progress |
2038.900 |
1663.400 |
5580.800 |
|
(iv) Intangible assets
under development |
0.000 |
0.000 |
0.000 |
|
(b) Non-current
Investments |
8725.900 |
7079.400 |
6103.200 |
|
(c) Deferred tax assets
(net) |
0.000 |
0.000 |
0.000 |
|
(d) Long-term Loan and Advances |
4636.000 |
2891.000 |
1784.900 |
|
(e) Other Non-current
assets |
163.300 |
185.800 |
1.200 |
|
Total Non-Current Assets |
34943.500 |
31938.700 |
29737.500 |
|
|
|
|
|
|
(2) Current assets |
|
|
|
|
(a) Current investments |
0.300 |
0.400 |
186.800 |
|
(b) Inventories |
17118.100 |
14317.300 |
12192.600 |
|
(c) Trade receivables |
29701.200 |
17305.900 |
14262.800 |
|
(d) Cash and cash
equivalents |
97.200 |
1145.700 |
140.100 |
|
(e) Short-term loans and
advances |
3147.300 |
2759.800 |
2624.800 |
|
(f) Other current assets |
747.700 |
777.900 |
767.500 |
|
Total Current Assets |
50811.800 |
36307.000 |
30174.600 |
|
|
|
|
|
|
TOTAL |
85755.300 |
68245.700 |
59912.100 |
PROFIT & LOSS
ACCOUNT
|
|
PARTICULARS |
31.03.2014 |
31.03.2013 |
31.03.2012 |
|
|
SALES |
|
|
|
|
|
Income |
71107.100 |
54251.000 |
42814.500 |
|
|
Other Income |
748.000 |
265.100 |
190.600 |
|
|
TOTAL (A) |
71855.100 |
54516.100 |
43005.100 |
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
Cost of Materials
Consumed |
34223.400 |
30536.400 |
23932.900 |
|
|
Purchases of
Stock-in-Trade |
968.900 |
780.900 |
355.200 |
|
|
Changes in inventories of
finished goods, work-in-progress and Stock-in-Trade |
(357.500) |
(1210.800) |
898.700 |
|
|
Employees benefits
expense |
5142.100 |
4314.200 |
3641.000 |
|
|
Other expenses |
11935.500 |
10249.700 |
8251.700 |
|
|
Exceptional Items |
|
|
3198.600 |
|
|
TOTAL (B) |
51912.400 |
44670.400 |
40278.100 |
|
|
|
|
|
|
|
Less |
PROFIT/ (LOSS) BEFORE INTEREST, TAX, DEPRECIATION AND
AMORTISATION (C) |
19942.700 |
9845.700 |
2727.000 |
|
|
|
|
|
|
|
Less |
FINANCIAL EXPENSES (D) |
2888.400 |
2500.600 |
2675.800 |
|
|
|
|
|
|
|
|
PROFIT / (LOSS) BEFORE
TAX, DEPRECIATION AND AMORTISATION (C-D) (E) |
17054.300 |
7345.100 |
51.200 |
|
|
|
|
|
|
|
Less/ Add |
DEPRECIATION/
AMORTISATION (F) |
1859.700 |
1713.900 |
1429.400 |
|
|
|
|
|
|
|
|
PROFIT/ (LOSS) BEFORE TAX (E-F) (G) |
15194.600 |
5631.200 |
(1378.200) |
|
|
|
|
|
|
|
Less |
TAX (H) |
3473.700 |
671.300 |
(952.100) |
|
|
|
|
|
|
|
|
PROFIT/ (LOSS) AFTER TAX
(G-H) (I) |
11720.900 |
4959.900 |
(426.100) |
|
|
|
|
|
|
|
Add |
PREVIOUS YEARS’ BALANCE
BROUGHT FORWARD (J) |
18752.200 |
14797.100 |
15561.500 |
|
|
|
|
|
|
|
Less |
APPROPRIATIONS |
|
|
|
|
|
Transfer to General
Reserve |
1172.100 |
496.000 |
0.000 |
|
|
Dividend |
874.100 |
436.800 |
291.100 |
|
|
Tax on Dividend |
148.500 |
72.000 |
47.200 |
|
|
Total (K) |
2194.700 |
1004.800 |
338.300 |
|
|
|
|
|
|
|
|
Balance Carried to the
B/S (I+J-K) |
28278.400 |
18752.200 |
14797.100 |
|
|
|
|
|
|
|
|
EARNINGS IN FOREIGN CURRENCY |
|
|
|
|
|
F.O.B. Value of Exports |
53269.000 |
38710.100 |
29239.900 |
|
|
Others |
150.700 |
346.000 |
540.100 |
|
|
TOTAL EARNINGS |
53419.700 |
39056.100 |
29780.000 |
|
|
|
|
|
|
|
|
IMPORTS |
|
|
|
|
|
Raw Materials |
20877.900 |
17845.800 |
13845.100 |
|
|
Stores and spares, lab
chemicals and other consumables |
340.800 |
84.400 |
123.400 |
|
|
Capital Goods |
388.200 |
349.100 |
730.400 |
|
|
TOTAL IMPORTS |
21606.900 |
18279.300 |
14698.900 |
|
|
|
|
|
|
|
|
Earnings / (Loss) Per
Share (Rs.) |
|||
|
|
- Basic |
40.24 |
17.04 |
(1.46) |
|
|
- Diluted |
40.20 |
17.02 |
(1.46) |
QUARTERLY /
SUMMARISED RESULTS
|
Particulars |
Jun 2014 |
Sep 2014 |
Dec 2014 |
|
Audited / Un
Audited |
Un Audited |
Un Audited |
Un Audited |
|
Net Sales |
1,9510.000 |
2,0077.000 |
2,0979.900 |
|
Total Expenditure |
1,3630.100 |
1,4532.800 |
1,5454.600 |
|
PBIDT (Excl OI) |
5879.900 |
5544.200 |
5525.300 |
|
Other Income |
121.600 |
186.500 |
117.100 |
|
Operating Profit |
6001.500 |
5730.700 |
5642.400 |
|
Interest |
138.500 |
137.900 |
148.500 |
|
Exceptional Items |
0.000 |
0.000 |
0.000 |
|
PBDT |
5863.000 |
5592.800 |
5493.900 |
|
Depreciation |
634.000 |
595.400 |
595.500 |
|
Profit Before Tax |
5229.000 |
4997.400 |
4898.400 |
|
Tax |
1295.000 |
1080.800 |
955.700 |
|
Provisions and contingencies |
0.000 |
0.000 |
0.000 |
|
Profit After Tax |
3934.000 |
3916.600 |
3942.700 |
|
Extraordinary Items |
0.000 |
0.000 |
0.000 |
|
Prior Period Expenses |
0.000 |
0.000 |
0.000 |
|
Other Adjustments |
0.000 |
0.000 |
0.000 |
|
Net Profit |
3934.000 |
3916.600 |
3942.700 |
KEY RATIOS
|
PARTICULARS |
|
31.03.2014 |
31.03.2013 |
31.03.2012 |
|
|
|
|
|
|
|
PAT / Total Income |
(%) |
16.31 |
9.10 |
-0.99 |
|
|
|
|
|
|
|
Net Profit Margin (PBT/Sales) |
(%) |
21.37 |
10.38 |
-3.22 |
|
|
|
|
|
|
|
Return on Total Assets (PBT/Total Assets} |
(%) |
20.26 |
9.46 |
-2.86 |
|
|
|
|
|
|
|
Return on Investment (ROI) (PBT/Networth) |
|
0.38 |
0.19 |
-0.06 |
|
|
|
|
|
|
|
Debt Equity Ratio (Total Debt /Networth) |
|
0.70 |
0.94 |
0.98 |
|
|
|
|
|
|
|
Current Ratio (Current Asset/Current Liability) |
|
1.53 |
1.31 |
1.14 |
FINANCIAL ANALYSIS
[all figures are
in Rupees Million]
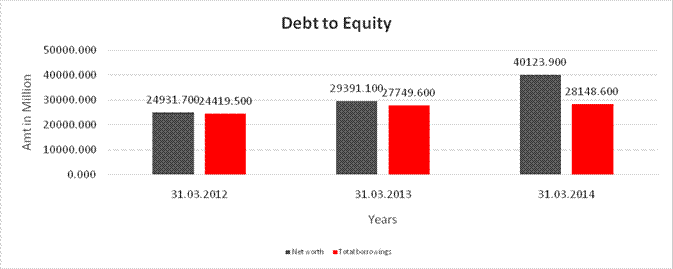
DEBT EQUITY RATIO
|
Particular |
31.03.2012 |
31.03.2013 |
31.03.2014 |
|
|
(Rs. In Million) |
(Rs. In Million) |
(Rs. In Million) |
|
Share Capital |
291.100 |
291.200 |
291.500 |
|
Reserves & Surplus |
24640.600 |
29099.900 |
39832.400 |
|
Net worth |
24931.700 |
29391.100 |
40123.900 |
|
|
|
|
|
|
long-term borrowings |
8337.400 |
10410.600 |
10323.100 |
|
Short term borrowings |
16082.100 |
17339.000 |
17825.500 |
|
Total borrowings |
24419.500 |
27749.600 |
28148.600 |
|
Debt/Equity ratio |
0.979 |
0.944 |
0.702 |
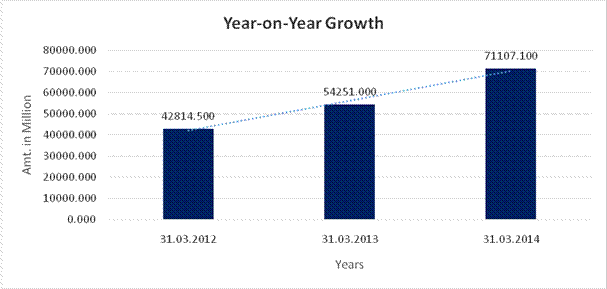
YEAR-ON-YEAR GROWTH
|
Year on Year Growth |
31.03.2012 |
31.03.2013 |
31.03.2014 |
|
|
(Rs. In Million) |
(Rs. In Million) |
(Rs. In Million) |
|
Sales |
42814.500 |
54251.000 |
71107.100 |
|
|
|
26.712 |
31.071 |
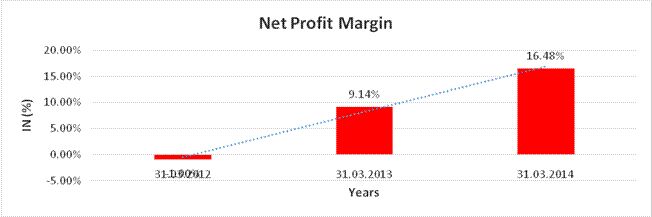
NET PROFIT MARGIN
|
Net Profit Margin |
31.03.2012 |
31.03.2013 |
31.03.2014 |
|
|
(Rs. In Million) |
(Rs. In Million) |
(Rs. In Million) |
|
Sales |
42814.500 |
54251.000 |
71107.100 |
|
Profit |
(426.100) |
4959.900 |
11720.900 |
|
|
(1.00%) |
9.14% |
16.48% |
LOCAL AGENCY FURTHER INFORMATION
|
Sr. No. |
Check List by Info Agents |
Available in
Report (Yes / No) |
|
1] |
Year of Establishment |
Yes |
|
2] |
Locality of the firm |
Yes |
|
3] |
Constitutions of the firm |
Yes |
|
4] |
Premises details |
No |
|
5] |
Type of Business |
Yes |
|
6] |
Line of Business |
Yes |
|
7] |
Promoter's background |
Yes |
|
8] |
No. of employees |
Yes |
|
9] |
Name of person contacted |
No |
|
10] |
Designation of contact
person |
No |
|
11] |
Turnover of firm for last
three years |
Yes |
|
12] |
Profitability for last
three years |
Yes |
|
13] |
Reasons for variation
<> 20% |
--------------------- |
|
14] |
Estimation for coming
financial year |
No |
|
15] |
Capital in the business |
Yes |
|
16] |
Details of sister
concerns |
Yes |
|
17] |
Major suppliers |
No |
|
18] |
Major customers |
No |
|
19] |
Payments terms |
No |
|
20] |
Export / Import details
(if applicable) |
No |
|
21] |
Market information |
---------------------- |
|
22] |
Litigations that the firm
/ promoter involved in |
Yes |
|
23] |
Banking Details |
Yes |
|
24] |
Banking facility details |
Yes |
|
25] |
Conduct of the banking
account |
---------------------- |
|
26] |
Buyer visit details |
Yes |
|
27] |
Financials, if provided |
Yes |
|
28] |
Incorporation details, if
applicable |
Yes |
|
29] |
Last accounts filed at
ROC |
Yes |
|
30] |
Major Shareholders, if
available |
Yes |
|
31] |
Date of Birth of
Proprietor/Partner/Director, if available |
Yes |
|
32] |
PAN of
Proprietor/Partner/Director, if available |
No |
|
33] |
Voter ID No of
Proprietor/Partner/Director, if available |
No |
|
34] |
External Agency Rating,
if available |
No |
LITIGATION
DETAILS:
CASE
STATUS INFORMATION
|
ARBAPPL 33 / 2014 |
ARBAPPLSR 2501 /
2014 |
CASE IS:PENDING |
|
PETITIONER |
RESPONDENT |
|
|
M/ B. RAMA RAO AND COMPANY, HYD |
VS |
M/S. AUROBINDO PHARMA LIMITED, HYD |
|
PET.ADV. : SUDARSHAN |
RESP.ADV. : VIKRAM POOSERLA |
|
|
SUBJECT: ARBITRATION APLICATION |
DISTRICT: HYDERABAD |
|
FILING DATE: 09.04.2014 |
POSTING STAGE : FOR ADMISSION |
|
|
REG. DATE : 10.04.2014 |
LISTING DATE : 10.10.2014 |
STATUS : NOTICES |
|
HON'BLE JUDGE(S): |
HON'BLE THE CHIEF JUSTICE |
|
ECONOMIC BACKGROUND
The Central Statistics Office had estimated India's real GDP to have grown by 4.6% during April-December 2013.
It had projected the GDP growth for the entire fiscal year 2013- 14 at 4.9%, thus implicitly estimating the growth for the last quarter at 5.5%.
The country's real GDP growth (growth in gross domestic product at factor cost at 2004-05 constant prices) was expected to pick up in the last quarter of 2013-14. The early data releases - IIP and merchandise exports - indicate that the recovery has been prolonged. As per preliminary estimates, the economy has grown at a rate of 4.7% or even lower in 2013-14.
The growth is likely to have been subdued because of poor performance of the mining, manufacturing, construction and trade, hotels, transport, storage & communication services sectors. While the size of the mining sector has shrunk on supply constraints, the others have suffered due to poor domestic demand, both consumption and investment. The agriculture, financial, insurance, real estate and business services and community, social & personal services sectors are believed to have grown well in 2013-14. The growth in India's real GDP is expected to improve to 5.5% in 2014-15 from 4.7% in 2013-14.
The agriculture sector that did well in 2013- 14, owing to a good monsoon, is expected to witness a sharp deceleration in growth to 1.8% in 2014-15 from 3.8% in 2013-14.Unlike last year, the monsoon is expected to be weak in 2014, as El Nino conditions are expected to develop. The deficient rainfall and high base of last year, is expected to pull down the growth of the agricultural sector in 2014-15.
On the other hand, given the upbeat business confidence, industrial and services sectors are expected to show acceleration in growth in 2014-15. Industrial sector is expected to grow by 3.3%, faster than a low 0.6% growth estimated for 2013-14. The mining sector is expected to return to growth provided no natural calamities hamper production. The sector will grow by 2.8%, after shrinking for three successive years.
Investment demand in India is expected to pick up gradually in 2014-15, as the Cabinet Committee on Investments (CCI) has been clearing several projects. The land acquisition process has become easy post implementation of the new land acquisition act. Fast tracking of projects is expected to boost the construction activity in India, generate new employment and create fresh demand for items like cement, steel and machinery.
High inflation and firm interest rates had eaten into discretionary spending and savings of the middle and lower class in 2013-14. But, inflation is unlikely to rise in 2014-15 which, in turn, can boost the growth of the manufacturing sector. Exports, which account for 19% of the sales of the manufacturing sector, are also expected to contribute to the industrial growth in 2014-15. Export earnings are expected to rise by 9.1% in US dollar terms on weak rupee and pick-up in global economy.
The manufacturing sector is expected see a turnaround in 2014-15, registering a 2.8% growth, as against a 0.5% fall estimated for 2013-14. The electricity sector is expected to maintain its growth rate at around 6% in 2014-15.
The services sector, which accounts for 60% of the real GDP, is also expected to show an improvement in growth to 7.3% in 2014-15 from 6.8% in 2013-14. The acceleration in growth is expected to come from the trade, hotels, transport, storage and communication services sector. An improvement in the growth of mining, manufacturing and construction sectors is expected to have a cascading effect on the performance of these services. The growth of the sector is expected to accelerate to 5.7% in 2014-15 from 4.4% in 2013-14.
It is estimated that the finance, real estate and insurance services sector and the community, social & personal services sector will maintain the growth rate at around 11% and 5.5%, respectively in 2014-15.
The HSBC Trade Confidence Index, the largest trade confidence survey in the world, has positioned India at the top with 142 points. The increasing demand due to its population makes the country a good market for consumption goods, according to the report.
With a newly elected government in place, Indian economy is expected to improve in 2014-15. The recovery will be calibrated, although several sectors might see a jump start. On the whole, there is improvement in business confidence across the country and good days for the economy are expected in the near future.
INDUSTRY PERSPECTIVE
India has over 10,500 manufacturing units and over 3,000 pharma companies and exports all forms of pharmaceuticals from APIs to formulations, both in modern medicine and traditional Indian medicines. Globally, India ranks among the top exporters of formulations by volume.
The country's exports of generics have been growing at a rate of nearly 24% annually over the past four years. As per 'Pharma Vision 2020', the Government of India aims to make India a global leader in end-to-end drug manufacturing epicenter, leveraging on the fact that cost of production in India is approximately 35% to 40% lower than in the developed countries. The enormous opportunity can be best illustrated from the projected human resource requirement of the Indian pharma sector, estimated to be about 2.15 million by 2020.
According to an estimate, India accounts for 35.7%, about 3,000 of the 8,374 Drug Master Files filed with the USA, which is the highest by any country outside of USA. Higher commitment of resources and continuing efforts of the industry participants in conjunction with product patent cliff has made India a major destination for generic drug manufacturing. According to a recent study, India has already been accredited with 907 CEPs, 845 TGA and 513 sites registered with the US FDA.
India is the third-largest exporter of drugs to the United States by volume. In 2013-14, at USD 14.84 billion (approximately Rs. 898 billion), the growth rate of India's pharmaceutical exports slowed sharply to just 1.2%. The near stagnation in growth is because of import alerts and bans by US regulators, a slowdown in the European Union and increased competition.
Seen on a global perspective, compared to other industries, over the past two years, the pharmaceutical industry across the world remained less impacted by the global economic uncertainty in certain parts of the world; yet, it would be appropriate to say that the industry is facing pressure from escalating costs and overwhelmed health systems across the world. An overview of recent sector performance shows that it is favorably positioned to achieve success in 2014 and beyond.
Among the drivers for growth are an aging population, rising incidence of chronic diseases, technological advancements and product innovation, and certain anticipated impact from health care reform provisions including increases in government funding and insurance coverage. Opportunities in emerging markets could continue to see traction, although many companies are looking more cautiously at these markets due to slowing growth and other pressures.
Factors such as India's low cost of production and strong R&D growth are the driving factors in attracting global pharmaceutical companies to India and at the same time, the comparative cost advantage enhances pharma exports. In fact, the rising global demand for generic drugs is also playing an important role in development of India as a hub for generic drug manufacturing.
India holds over 10% share in the global pharma production with over 60,000 generic brands across 60 therapeutic categories and manufacturing over 400 different active pharmaceutical ingredients (APIs). There is no doubt on the growth potential of the Indian pharma industry. In fact, a recent Deloitte report added that Indian companies can be expected to garner USD 40 billion in sales as close to 46 US drug patents will expire by 2015.
Globally, pharmaceuticals generated total revenue of USD 959 billion in 2012, growing 2.4 percent from 2011 (considerably below the 5.3% increase posted the year prior). Oncology is the leading therapeutic class; other focus areas include pain management, hypertension, diabetes, mental health, and respiratory ailments.
Recently, Deloitte Touche Tohmatsu examined common elements of the current wave of global reform, national differences, and how life sciences companies are reacting. Among their key survey findings are:
- Reducing costs, enhancing innovation and improving market access are the defining goals of health care reform. Some countries are adopting valuebased pricing structures for life sciences products, while others are combining cost containment with assistance for companies investing in R&D;
- Main impact of reform eventually will be on innovation and sales models;
- Specific elements of reform vary by country, requiring companies to have national approaches. Policy changes are predominantly shaped by specific national contexts with elements that are unique to their national systems;
- Leading companies are remodeling their innovation and sales activities in the face of reforms. Companies are working to change their innovation processes and sales models to benefit from opportunities arising from reforms.
Overall, cost containment is a common reform objective in both developed and developing markets; however, strategies vary. Most national health care systems have been encouraging greater use of generic drugs; in the U.S., for example, the proportion of prescriptions filled by generics has risen from around 50 to 80% over the last decade. Brazil is making branded generics and proprietary drugs of greater interest to pharmaceutical companies, and in China, recent reforms have put intense pressure on the prices of all drugs, including generic and over-the-counter (OTC) medicines.
In another cost-containment approach, Germany and several other countries have turned to value-based pricing for new drugs, which allows a price differential from existing offerings - including generics - based on a new product's demonstrated superiority.
Pharma exports from India will be more than the size of the domestic sales by FY15, according a recent report by India Ratings and Research. While revising its outlook for the sector for next fiscal to positive from stable because of increased exports, the firm said that the domestic pharma market is expected to see high single digit revenue growth and profit margins are expected to improve because of increasing utilization of manufacturing facilities.
As stated earlier, India is emerging as the manufacturing hub of the global pharmaceutical industry driven by large labour force, skills and education, to become a potent competitor to the developed countries. According to PriceWaterhouse Coopers, finished generics supplied from India account for about 20% of the global generic market by volumes. More than 90% of WHO pre-qualified Active Pharmaceutical Ingredients (ARV, Anti-tubercular and antimalarials) are sourced from India.
55% of India's total pharma exports amounting to USD 14.84 billion is shipped to the highly regulated markets of the world, including the US and EU countries. Japan too holds a big potential for Indian pharma sector.
OUTLOOK
Pharmaceutical industry is faced with major challenges but Aurobindo is part of product segments that display growth. In a world that is striving to achieve lower drug costs at every level, production costs will continue to remain a key measure. Aurobindo has a good foundation of reliable sourcing and cost effective manufacturing systems and is exploring further ways of reducing costs and strengthening competitiveness.
Sales are being ramped up across all the geographies. This trend, as in the past, is expected to continue with several new launches as well as improving the existing business. A further thrust is being given to the Company's presence in Europe while adding to the market share of the injectable side of the business. In the case of the APIs, the emphasis is to grow the high value products, gain momentum in developed markets and taper-off non-competitive products.
Capacity utilization is improving at all production units. Indeed, keeping the likely requirement for growth in the generics market, balancing equipment is being added in API manufacturing facilities. The Company has an enviable product basket with a large portfolio of regulatory approvals. The focus will be to continue to step up the volumes of high value products, improve the reach in the market while taking care to reduce overall costs.
The Company will capitalize on its inherent strengths, some of which are iterated below:
- Cost effective vertically integrated manufacturing systems;
- Current Good Manufacturing Practices (cGMP) and regulatory compliant facilities producing high-quality APIs and finished dosage formulations;
- Best-in class, best-in-cost large manufacturing capacity;
- High visibility in API and generics;
- Strong financial position with ability to scale up;
- Highly skilled professionals with regulatory expertise and competent to deliver on development, product processes and regulatory standards;
- Access to new technologies.
The corporate objectives are structured to achieve enhanced shareholder value while delivering what the customers want. An increased thrust on combination drugs, inlicensing initiatives, alliances with MNCs and other measures such as enhanced focus on injectables, OTCs, institutional segment as well as focus on reaching direct to the customers are some of the drivers for gaining traction in enhancing revenues, EBITDA margin and Return on Investment higher than the industry average. The target is to stay cash flow positive, lower the leverage, reduce interest outgo and strive to expand earnings.
OVERVIEW
Aurobindo Pharma, an integrated global pharmaceutical company engaged in API and formulations business segments, faces various business risks just like any other business. Such business risks are broadly categorized into strategic, operational, financial and compliance risks. Aurobindo believes that there can be no growth or creation of value in the Company without risk-taking, while risks not properly managed can affect the Company's ability to achieve its objectives. Risk management system plays a key role in directing the Company's activities within the desired parameters.
The Company defines risks as events that have the potential to negatively impact achievement of objectives and anything that would prevent the Company from achieving its business objectives, including both internally and externally driven, or due to either action or inaction on the part of Company.
Aurobindo has embedded and aligned risk management system with every part of critical business processes in order to systematically ensure that processes are designed to achieve strategic objectives and the business risks are identified across the organisation in a holistic manner rather than in silos.
PERFORMANCE REVIEW
The Company has delivered satisfactory results despite several challenges including rising costs, severe competitive pressures and sluggishness in customer countries. They are pleased to report that market conditions for The Company's products were better exploited with focused investments in the markets and products supplemented by significant firstto- launch advantages. The Company continued to invest to add to its market presence for existing products, widen the geographical reach both within US and Europe, position new products by adding to shelf space, convert product approvals into invoices by reducing the time-to-market and rationalize on low value offers.
Team Aurobindo focused on continued growth within boundaries of its business plan. Initiatives were carefully planned in new products and investments were made in to prioritized growth markets. The year's performance demonstrates Aurobindo's strength in the injectables segment, successful introduction of new products in the developed markets, emphasis on cost competitiveness benefiting from the structured integrated business model, and the ability to seize the opportunities in a highly competitive market. There was a more positive momentum in a number of high value products and the teams are presently striving to ensure sustained growth quarter-on-quarter.
The consolidated revenue (net) from operations was higher over the previous year by 38.3% at Rs.80997.900 million in the year as against Rs.58553.200 million in Aurobindo Annual Report 2013-14 / 38 the previous year. The formulation and API ratio during the year was 65:35. Consolidated net profit is Rs.11728.500 million, a significant growth over Rs. 2938.600 million reported in the previous year. The Company delivered earnings per share of `40.2 as against `10.1 in the previous year.
Gross revenue from formulation during the year was Rs. 53785.000 million, 58.8% higher on a yearon- year basis as compared to Rs. 33872.000 million reported in 2012-13. The Company strived to increase its share of high value products and special efforts were made to build relationships in the developed markets. API revenues for the year were Rs. 28642.000 million, a growth of 12.9% over the previous year, on account of favorable demand scenario as well as focused efforts at enhancing product realizations.
EBITDA at the consolidated level for the year was Rs. 21552.100 million, which is 26.6% of consolidated revenue (net), and has gone up by 142.3% over 2012-13. Profitability during the year has improved due to better sales and business mix which had favorable impact on material consumption to net sales. Cost of materials for the year was 44.5% of consolidated revenue (net) in comparison to 51.1% in the previous year.
As far as foreign exchange is concerned, the closing rupee dollar rate was Rs. 59.915 on March 31, 2014 while it was Rs. 54.285 on March 31, 2013. The rupee has been highly volatile through the year and has depreciated by 10.4% during the financial year. This has resulted in a net exchange loss of Rs. 2030.500 million during the year which includes an amount of Rs. 2022.200 million on borrowings adjusted to finance charges as per revised Schedule VI of the Companies Act, 1956.
Europe and the rest of the world geographies recorded a sale of Rs. 11355.000 million, thereby growing at 28.4% over the previous year, and in ARV formulation sales by 12% to Rs. 8402.000 million. As in the previous year, strategic action was taken to be selective in building products and markets that contribute to the bottom line.
In terms of segmental contribution to the formulations revenue, the share of US was 63.2% against 51.7% in the previous year. Similarly, European as well as the rest of the world was 21.2% against 26.2% and ARV was 15.6% against 22.1% in the previous year. The segmental shift in both API and formulations is reflective of The Company's efforts to improve margins and this trend is expected to continue.
In generic markets of US, UK, Germany, Spain, France and Netherlands, The Company is progressing well. Additional thrust to raise the marketing presence and gain margin is ongoing in countries such as Japan, Portugal and Italy. Focused efforts were made during the year to improve bottomline, even as progress was made to expand markets.
In the formulation business, The Company identifies and secures success by market adapted product development together with quick and effective commercializing of new launches. Aurobindo has had a significant success in its new launches, especially in the US. The priority has been to optimize the portfolio and capitalize on the opportunities for their product offering. Efforts were made to increase market share and leverage existing relationships. Aggressive positions were taken in preparations for new launches. Aurobindo today has a balanced portfolio withvisibility for clearly defined plan to climb the value chain.
The Company has completed acquisition of certain commercial operations in Western Europe from Actavis plc. Aurobindo acquired personnel, commercial infrastructure, products, marketing authorizations and dossier licence rights in seven European countries. Actavis and The Company have also entered into a mutually beneficial long-term commercial and supply arrangement which envisages collaboration with Actavis to ensure business continuity and a smooth transition.
Following receipt of clearances from competent authorities, The Company intends to combine the strength of both enterprises (including its vertically integrated platform and existing commercial infrastructure) in these markets and to identify and maximize all opportunities to improve the Company's performance. The Company with its inherent cost competitiveness and group structure would build on Actavis' strong market position in the West European countries and strive to become a significant generics player in Europe.
The Company will position itself as one of the leading Indian pharmaceutical companies in Europe and strive to achieve a critical mass in Western Europe with a top 10 position in several key markets. The objective is to expand the front-end operations into five segments (generics, prescription products, over-thecounter products, hospital products and generics tenders) with approximately 1,250 dossiers and an additional pipeline of over 200 products.The efforts are on to achieve a rapid and successful integration.
Aurobindo has a clear commitment to creating value for all its stakeholders. The Company has the strategies and core strengths required to expand the market, scale and efficiencies to leverage product portfolio globally and enhance the profitability.
OUTLOOK
Aurobindo is building momentum on its way to become one of the world's leading generic pharmaceutical companies. The efforts of the past in setting up a formidable foundation with several drivers of growth have started to pay off. The Company has carefully crafted architecture for sustained growth with a robust structure of manufacturing systems, large regulatory approved product basket, an enviable geographic and marketing spread created by a reservoir of talented and experienced managers and employees focused on piloting the Company's staircase of growth.
The global pharmaceutical market has several driving factors, which primarily include demand for cost effective drugs to meet the needs of growing population, gradual increase in life expectancy, and a shift towards generics for a range of drugs with a greater focus on lifestyle diseases. Aurobindo anticipated and prepared itself for the paradigm over the years.
The Company has technologies for collecting and synthesizing complex chemistry to face industry challenges of patent cliff,more efficient and compliance conscious processes to offer cost effective products that answer the needs of markets coping with spiraling healthcare cost. Actions to improve the operational efficiencies, especially in the area of supply chain are expected to support profitability and cash flow going forward.
Every effort now is to sustain the momentum to become a stake holder friendly company that meets customer expectations, grows to be a preferred employer, and expands earnings while it enhances shareholder value
UNSECURED LOANS
|
PARTICULAR |
31.03.2014 (Rs.
in Million) |
31.03.2013 (Rs.
in Million) |
|
LONG TERM
BORROWINGS |
|
|
|
Others |
|
|
|
Deferred sales tax loan |
537.000 |
639.300 |
|
SHORT TERM
BORROWINGS |
|
|
|
Buyers Credit |
25.100 |
1152.400 |
|
Packing credit loans |
7884.500 |
6115.000 |
|
Short –term loans from Banks |
0.000 |
1085.700 |
|
Total |
8446.600 |
8992.400 |
INDEX OF CHARGES
|
S. No. |
Charge ID |
Date of Charge Creation/Modification |
Charge amount secured |
Charge Holder |
Address |
Service Request Number (SRN) |
|
1 |
10402714 |
08/01/2013 |
1,300,000,000.00 |
DBS Bank Ltd |
Salarpuria Windsor,
No.3, Ulsoor Road, Bengaluru, |
B67978148 |
|
2 |
10359358 |
25/05/2012 |
1,300,000,000.00 |
IFCI Limited |
IFCI Tower, 5-9-13,
Taramandal Complex, Saifabad, |
B41106543 |
|
3 |
10359357 |
25/05/2012 |
1,560,000,000.00 |
IFCI Limited |
IFCI Tower, 5-9-13,
Taramandal Complex, Saifabad, |
B41106147 |
|
4 |
10269583 |
22/09/2011 * |
379,750,000.00 |
Axis Bank Limited |
TRISHUL 3RD FLOOR OPP SAMARTHESHWAR TEMPLE, LAW GARDEN ELLISBRIDGE, AHMEDABAD, Gujarat - 380006, INDIA |
B22085377 |
|
5 |
10266490 |
28/12/2010 |
1,645,000,000.00 |
IFCI Limited |
IFCI TOWER, 5-9-13, Taramandal Complex, Safifabad, Hyderabad, Andhra Pradesh - 500004, INDIA |
B05139555 |
|
6 |
10265641 |
28/12/2010 |
1,856,000,000.00 |
ING Bank N V |
Singapore Branch, 9 Raffles Place,#19-02 Republic Plaza, Singapore, - 048619, SINGAPORE |
B03586161 |
|
7 |
10262723 |
28/12/2010 |
2,250,000,000.00 |
IFCI Limited |
IFCI TOWER, 5-9-13, Taramandal Complex, Safifabad, Hyderabad, Andhra Pradesh - 500004, INDIA |
B03928439 |
|
8 |
10215595 |
19/03/2014 * |
2,050,000,000.00 |
ICICI Bank Limited |
ICICI Bank Tower, Plot No.12, Nanakram Guda, 6th Floor, Tower II, North Wing, Hyderabad, Andhra Pradesh - 500032, INDIA |
C03655834 |
|
9 |
10044936 |
25/08/2014 * |
1,640,000,000.00 |
HDFC BANK LIMITED |
HDFC BANK HOUSESENAPATI BAPAT MARG, LOWER PAREL W, MUMBAI, Maharashtra - 400013, INDIA |
C21711395 |
|
10 |
10030551 |
22/10/2013 * |
2,800,000,000.00 |
IDBI Bank Limited |
5-9-89/1 & 2,
Chapel Road, PB No.370, Hyderabad, |
B89389597 |
* Date of charge modification
STATEMENT OF STAND ALONE UNAUDITED RESULTS FOR THE QUARTER AND NINE
YEAR ENDED 31.12.2014
(Rs.
In Million)
|
Particulars |
Three Months Ended |
Nine Months Ended |
||
|
31.12.2014 |
30.09.2014 |
31.12.2014 |
||
|
Unaudited |
Unaudited |
Unaudited |
||
|
1 |
Income from Operations |
|
|
|
|
|
(a) Net sates/income from operations (Net
of excise duty) |
20743.600 |
19886.600 |
59975.800 |
|
|
(b) Other Operating Income |
236.300 |
190.400 |
591.100 |
|
|
Total
income from operations (net) |
20979.900 |
20077.000 |
60566.900 |
|
2 |
Expenses |
|
|
|
|
|
(a) Cost of materials consumed |
9836.500 |
9825.000 |
28800.000 |
|
|
(b) Purchases of stock-in trade |
17.900 |
73.500 |
146.200 |
|
|
(c) Changes in inventories of finished goods.
work-in-progress and stock in trade |
121.600 |
(614.000) |
(552.700) |
|
|
(d) Employee benefits expense |
1726.200 |
1596.200 |
4802.400 |
|
|
(e) Depreciation and Anmortisation Expenses |
595.500 |
595.400 |
1824.900 |
|
|
(f) Provision for decline in the value of long-term investment |
-- |
-- |
-- |
|
|
(g) Other Expenses |
3643.800 |
3210.500 |
9871.400 |
|
|
Total
expenses |
15941.500 |
14686.600 |
44892.200 |
|
3 |
Profit/ (Loss) from operations before other
Income, finance costs, foreign exchange (gain)/Loss and exceptional Items (1-2) |
5038.400 |
5390.400 |
15674.700 |
|
4 |
Other Income |
117.100 |
186.500 |
360.500 |
|
5 |
Profit/ (Loss) from operations before other
income, finance costs, foreign exchange (gain)/Loss and exceptional items (3+4) |
5155.500 |
5576.900 |
16035.200 |
|
6 |
Finance Costs |
148.500 |
137.900 |
424.900 |
|
7 |
Foreign exchange (Gain)/Loss |
108.600 |
441.600 |
485.500 |
|
8 |
Profit/ (Loss) from ordinary activities
after finance cost but before exceptional items (5-6) |
4898.400 |
4997.400 |
15124.800 |
|
9 |
Exceptional items |
0.000 |
0.000 |
0.000 |
|
10 |
Profit/ (Loss) from ordinary activities
before tax (7+8) |
4898.400 |
4997.400 |
15124.800 |
|
11 |
Tax expenses |
955.700 |
1080.800 |
3331.500 |
|
12 |
Net Profit / (Loss) for the period (11-12) |
3942.700 |
3916.600 |
11793.300 |
|
13 |
Minority Interest |
|
|
|
|
14 |
Net Profit/ (Loss) after taxes, minority interest
(12-13) |
3942.700 |
3916.600 |
11793.300 |
|
15 |
Paid up equity share capital (Face Value of
Rs10/- each) |
291.500 |
291.500 |
291.500 |
|
16 |
Reserve excluding Revaluation Reserve as
per Balance Sheet of previous accounting year |
|
|
|
|
17 |
Earnings per share of Re.1/- each (not annualised) |
|
|
|
|
|
(a) Basic |
13.52 |
13.44 |
40.46 |
|
|
(b) Diluted |
13.52 |
13.42 |
40.46 |
|
A |
PARTICULARS OF SHAREHOLDING |
|
|
|
|
1 |
Public Shareholding |
|
|
|
|
|
- Number of shares |
133888935 |
133771235 |
133888935 |
|
|
- Percentage of shareholding |
45.94 |
45.90 |
45.94 |
|
2 |
Promoters and Promoter group shareholding |
|
|
|
|
|
a) Pledged / Encumbered |
|
|
|
|
|
- Number of shares |
13490000 |
14400000 |
13490000 |
|
|
- Percentage of shares (as a % of the total
shareholding of Promoter & Promoter group) |
8.56 |
9.13 |
8.56 |
|
|
- Percentage of shares (as a % of the total
Share Capital of the Company) |
4.63 |
4.94 |
4.63 |
|
|
b) Non Encumbered |
|
|
|
|
|
- Number of shares |
144085786 |
143285786 |
144085786 |
|
|
- Percentage of shares (as a % of the total
shareholding of Promoter & Promoter group) |
91.44 |
90.87 |
91.44 |
|
|
- Percentage of shares (as a % of the total
Share Capital of the Company) |
49.43 |
49.16 |
49.43 |
|
|
Particulars |
Nine Months Ended |
|
|
|
31.12.2014 |
|
B |
INVESTOR
COMPLAINTS |
|
|
|
|
|
|
|
Pending at the beginning of the quarter |
1 |
|
|
Received during the quarter |
39 |
|
|
Disposed off during the quarter |
40 |
|
|
Remaining unresolved at the end of the
quarter |
Nil |
Note:
1. The above unaudited financial results for the quarter and nine months ended December 31, 2014 as reviewed by the Audit Committee have been approved by the Board at its meeting held on February 04, 2015. A Limited Review of the standalone financial results for the quarter ended December 31, 2014 has been carried out by the Statutory Auditors.
2. The consolidated financial results have been prepared in accordance with AS - 21 on ‘Consolidated Financial Statement' and AS-27 ' Financial Reporting of Interests in Joint Ventures' and includes financial results of all Subsidiaries and a Joint Venture.
3. The Company's operations fall within a single primary business segment viz. 'Pharmaceutical Products'.
4. Sales of standalone for the current quarter include exports Rs. 16753.600 Million (Quarter ended December 31, 2013: Rs. 14368.200 Million).
5. Net sales for the current quarter include dossier Income
in standalone of Rs. 6.4 Million (December 31, 2013: Rs. 9.800 Million) and in
consolidated of Rs. 20.800 Million (December 31, 2013: Rs. 16.700 Million).
6. Foreign exchange (gain)/loss for standalone and consolidated includes
exchange difference of Rs. 4,63.500 Million, 665.700 Million, Rs. 519.600
Million, for three months ended December 31, 2014, September 30, 2014, and
December 31, 2013 respectively and Rs. 1234.100 Million, Rs. 1516.400 Million
for the nine months ended December 31, 2014 and December 31, 2013 respectively,
and Rs. 2022.200 Million for the year ended March 31,2014 arising from foreign
currency borrowings to the extent that they are regarded as an adjustment to
finance cost as per para 4(e) of "AS 16" on Borrowing costs.
7. The Board of Directors at their meeting held on September 13, 2013 decided
to transfer its injectable unit of the Company on a going concern basis
comprising assets and liabilities pertaining to the said unit to its wholly
owned subsidiary Curepro Parenterals Limited w.e.f. April 1, 2014. The same is
subject to requisite consent, approval or permission of the statutory or
regulatory authorities. Pending such approvals, no effect of this scheme has
been given in the above results.
8. Pursuant to the requirements of Schedule II to the Companies Act, 2013, the
management of the Company had reassessed the useful lives of fixed assets held
as at April 1, 2014, Based on such internal technical reassessment the Company
has accounted for additional depreciation amounting to Rs. 103.500 Million and
Rs.115.800 Million for three months ended December 31, 2014 and September 30,
2014 respectively and Rs. 391.600 Million for the nine months ended December
31, 2014 in standalone and Rs. 122.500 Million and Rs.124.500 Million for three
months ended December 31,2014 and September 30, 2014 respectively and Rs.
418.300 Million for the nine months ended December 31, 2014 in consolidated
results. Further based on transitional provisions, an amount of Rs.185.700
Million (net of deferred tax) has been adjusted with standalone and
consolidated opening retained earnings.
9. During the quarter, (i) Aurex B.V., The Netherlands, a step down subsidiary
of the Company has been incorporated (ii) Natrol LLC, USA has become a step
down subsidiary of the Company, (iii) Aurovitas SL, Spain was ceased to be a
step down subsidiary of the Company, (iv) Aurobindo Pharma France SARL, France,
a step down subsidiary of the Company was merged with Arrow Generiques SAS,
France another step down subsidiary of the Company, (v) The ownership of APL
Swift Services (Malta) Limited, a step down subdiary of the Company was
transferred from APL Holdings (Jersey) Limited, to Aurobindo Pharma (Malta)
Limited another step down subsidiary of the Company.
10. During the current year, on April 1, 2014, Company’s European subsidiary
Agile Pharma B.V., Netherlands, has acquired select Western European business
of Actavis. The above Consolidated results for the quarter and nine months
ended December 31, 2014 and quarter ended September 30, 2014 includes the
financial results of operations of the above business of Actavis from April 01,
2014. The corresponding figures of the previous periods are not comparable.
11. During the current year, on December 4, 2014, Company's USA subsidiary Aurobindo Pharma USA Inc. has acquired Natrol LLC, USA. The above Consolidated results for the quarter and nine months ended December 31, 2014 includes financial results of operations of Natrol LLC, USA from December 4, 2014. The corresponding figures of the previous periods are not comparable.
12. On 17th January, 2015 the Company has made allotment of 331,200 equity shares of Re. 1/- each at a premium of Rs. 90.50 per equity share to the employees under Employee Stock Option Plan 2005. Consequent to the allotment the paid up share capital of the Company has increased to 291,795,921 equity shares of Re. 1/- each.
13. The Board has approved a second interim dividend @200% on the equity share capital of the Company i.e. Rs. 2/- per share. This is in addition to interim dividend @150% on equity share capital of the Company i.e.Rs. 1.50 per share paid in September, 2014 for the year 2014-15.
14. Previous period/year figures have been regrouped/rearranged wherever considered necessary to conform to the current period presentation.
CONTINGENT
LIABILITIES:
|
PARTICULARS |
31.03.2014 (Rs.
In Million) |
31.03.2013 (Rs.
In Million) |
|
Outstanding bank guarantees |
771.800 |
486.300 |
|
Claims arising from disputes not acknowledged as debts |
|
|
|
- indirect taxes (excise duty and service tax)* |
223.300 |
196.300 |
|
Claims arising from disputes not acknowledged as debts - direct taxes* |
105.000 |
105.000 |
|
Claims against the Company not acknowledged as debts* |
150.300 |
493.100 |
|
Bills discounted with banks |
1060.600 |
3252.900 |
FIXED ASSETS
·
·
·
Leasehold buildings
·
Freehold buildings
·
Plant and Machinery
·
Furniture and Fittings
·
Vehicles
·
Office Equipment
PRESS RELEASE:
15th April 2015 Aurobindo Pharma receives USFDA Approval for Cefixime for Oral Suspension USP Aurobindo Pharma Limited is pleased to announce that the company has received final approvals from the US Food & Drug Administration (USFDA) to manufacture and market Cefixime for Oral Suspension USP, 100mg/5mL and 200mg/5mL (ANDA 204835). The product is ready for launch.
The approved ANDAs are bioequivalent and therapeutically equivalent to the reference listed drug product (RLD) Suprax® Oral Suspension USP 100mg/5mL and 200mg/5mL respectively of Lupin Pharmaceuticals Inc.
Cefixime for Oral Suspension is indicated for the treatment of adults and pediatric patients six months of age or older, with infections caused by susceptible strains of the designated organisms in urinary tract infections, otitis media, acute exacerbations of chronic bronchitis, uncomplicated gonorrhea (cervical/urethral), pharyngitis and tonsillitis
The product has an estimated market size of US$ 123 Million for the twelve months ending February 2015 according to IMS.
Aurobindo now has 11 ANDAs (represented by 7 product classes) approved out of Unit VI formulation facility in Hyderabad, India for manufacturing Oral Cephalosporin products.
About Aurobindo
Pharma Limited:
Aurobindo Pharma Limited (www.aurobindo.com), headquartered at Hyderabad, India, manufactures generic pharmaceuticals and active pharmaceutical ingredients. The company’s manufacturing facilities are approved by several leading regulatory agencies like US FDA, UK MHRA, Japan PMDA, WHO, Health Canada, MCC South Africa, ANVISA Brazil. The company’s robust product portfolio is spread over 6 major therapeutic/product areas encompassing Antibiotics, Anti-Retrovirals, CVS, CNS, Gastroenterologicals, and Anti-Allergics, supported by an outstanding R&D set-up. The Company is marketing these products globally, in over 125 countries.
CMT REPORT (Corruption, Money Laundering & Terrorism]
The Public Notice information has been collected from various sources
including but not limited to: The Courts,
1] INFORMATION ON
DESIGNATED PARTY
No exist designating subject or any of its beneficial owners,
controlling shareholders or senior officers as terrorist or terrorist organization
or whom notice had been received that all financial transactions involving
their assets have been blocked or convicted, found guilty or against whom a
judgement or order had been entered in a proceedings for violating
money-laundering, anti-corruption or bribery or international economic or
anti-terrorism sanction laws or whose assets were seized, blocked, frozen or
ordered forfeited for violation of money laundering or international
anti-terrorism laws.
2] Court Declaration :
No records exist to suggest that subject is
or was the subject of any formal or informal allegations, prosecutions or other
official proceeding for making any prohibited payments or other improper
payments to government officials for engaging in prohibited transactions or
with designated parties.
3] Asset Declaration :
No records exist to suggest that the property or assets of the subject
are derived from criminal conduct or a prohibited transaction.
4] Record on Financial
Crime :
Charges or conviction registered
against subject: None
5] Records on Violation of
Anti-Corruption Laws :
Charges or
investigation registered against subject: None
6] Records on Int’l
Anti-Money Laundering Laws/Standards :
Charges or
investigation registered against subject: None
7] Criminal Records
No
available information exist that suggest that subject or any of its principals
have been formally charged or convicted by a competent governmental authority
for any financial crime or under any formal investigation by a competent
government authority for any violation of anti-corruption laws or international
anti-money laundering laws or standard.
8] Affiliation with
Government :
No record
exists to suggest that any director or indirect owners, controlling shareholders,
director, officer or employee of the company is a government official or a
family member or close business associate of a Government official.
9] Compensation Package :
Our market
survey revealed that the amount of compensation sought by the subject is fair
and reasonable and comparable to compensation paid to others for similar
services.
10] Press Report :
No press reports / filings exists on
the subject.
CORPORATE GOVERNANCE
MIRA INFORM as part of its Due Diligence do provide comments on Corporate
Governance to identify management and governance. These factors often have been
predictive and in some cases have created vulnerabilities to credit
deterioration.
Our Governance Assessment focuses principally on the interactions
between a company’s management, its Board of Directors, Shareholders and other
financial stakeholders.
CONTRAVENTION
Subject is not known to have contravened any existing local laws,
regulations or policies that prohibit, restrict or otherwise affect the terms
and conditions that could be included in the agreement with the subject.
FOREIGN EXCHANGE RATES
|
Currency |
Unit
|
Indian Rupees |
|
US Dollar |
1 |
Rs. 63.52 |
|
|
1 |
Rs. 95.96 |
|
Euro |
1 |
Rs. 70.54 |
INFORMATION DETAILS
|
Analysis Done by
: |
RAS |
|
|
|
|
Report Prepared
by : |
TRU |
SCORE & RATING EXPLANATIONS
|
SCORE FACTORS |
RANGE |
POINTS |
|
HISTORY |
1~10 |
6 |
|
PAID-UP CAPITAL |
1~10 |
5 |
|
OPERATING SCALE |
1~10 |
6 |
|
FINANCIAL
CONDITION |
|
|
|
--BUSINESS SCALE |
1~10 |
6 |
|
--PROFITABILITY |
1~10 |
6 |
|
--LIQUIDITY |
1~10 |
6 |
|
--LEVERAGE |
1~10 |
6 |
|
--RESERVES |
1~10 |
6 |
|
--CREDIT LINES |
1~10 |
6 |
|
--MARGINS |
-5~5 |
-- |
|
DEMERIT POINTS |
|
|
|
--BANK CHARGES |
YES/NO |
YES |
|
--LITIGATION |
YES/NO |
YES |
|
--OTHER ADVERSE INFORMATION |
YES/NO |
NO |
|
MERIT POINTS |
|
|
|
--SOLE DISTRIBUTORSHIP |
YES/NO |
NO |
|
--EXPORT ACTIVITIES |
YES/NO |
NO |
|
--AFFILIATION |
YES/NO |
YES |
|
--LISTED |
YES/NO |
YES |
|
--OTHER MERIT FACTORS |
YES/NO |
YES |
|
DEFAULTER |
|
|
|
--RBI |
YES/NO |
NO |
|
--EPF |
YES/NO |
NO |
|
TOTAL |
|
53 |
This score serves as a reference to assess
SC’s credit risk and to set the amount of credit to be extended. It is calculated
from a composite of weighted scores obtained from each of the major sections of
this report. The assessed factors and their relative weights (as indicated
through %) are as follows:
Financial
condition (40%) Ownership
background (20%) Payment record
(10%)
Credit history
(10%) Market trend (10%) Operational size
(10%)
RATING EXPLANATIONS
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
>86 |
Aaa |
Possesses an extremely sound financial base with the strongest capability
for timely payment of interest and principal sums |
Unlimited |
|
71-85 |
Aa |
Possesses adequate working capital. No caution needed for credit
transaction. It has above average (strong) capability for payment of interest
and principal sums |
Large |
|
56-70 |
A |
Financial & operational base are regarded healthy. General
unfavourable factors will not cause fatal effect. Satisfactory capability for
payment of interest and principal sums |
Fairly Large |
|
41-55 |
Ba |
Overall operation is considered normal. Capable to meet normal
commitments. |
Satisfactory |
|
26-40 |
B |
Capability to overcome financial difficulties seems comparatively
below average. |
Small |
|
11-25 |
Ca |
Adverse factors are apparent. Repayment of interest and principal sums
in default or expected to be in default upon maturity |
Limited with
full security |
|
<10 |
C |
Absolute credit risk exists. Caution needed to be exercised |
Credit not
recommended |
|
-- |
NB |
New Business |
-- |
This report is issued at your request without any
risk and responsibility on the part of MIRA INFORM PRIVATE LIMITED (MIPL)
or its officials.