MIRA INFORM REPORT
|
Report No. : |
324859 |
|
Report Date : |
29.05.2015 |
IDENTIFICATION DETAILS
|
Name : |
NEULAND LABORATORIES LIMITED |
|
|
|
|
Registered
Office : |
Sanali Info Park, ‘A’ Block, Ground Floor, 8-2-120/113, Road No. 2, Banjara
Hills, Hyderabad – 500034, Telangana |
|
Tel. No.: |
91-40-30211600/ 23551081 |
|
|
|
|
Country : |
India |
|
|
|
|
Financials (as
on) : |
31.03.2015 |
|
|
|
|
Date of
Incorporation : |
07.01.1984 |
|
|
|
|
Com. Reg. No.: |
01-004393 |
|
|
|
|
Capital
Investment / Paid-up Capital : |
Rs. 89.539 Million |
|
|
|
|
CIN No.: [Company Identification
No.] |
L85195TG1984PLC004393 |
|
|
|
|
IEC No.: |
0988005077 |
|
|
|
|
TAN No.: [Tax Deduction &
Collection Account No.] |
HYDN00013G HYDN00131F |
|
|
|
|
PAN No.: [Permanent Account No.] |
AAACN9531E |
|
|
|
|
Legal Form : |
A Public Limited Liability Company. The Company’s Shares are Listed on
the Stock Exchanges. |
|
|
|
|
Line of Business
: |
Manufacturer, Exporter and Importer of Bulk Drugs. |
|
|
|
|
No. of Employees
: |
Information declined by the management |
RATING & COMMENTS
|
MIRA’s Rating : |
Ba (42) |
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
41-55 |
Ba |
Overall operation is considered normal. Capable to meet normal
commitments. |
Satisfactory |
|
Maximum Credit Limit : |
USD 4600000 |
|
|
|
|
Status : |
Satisfactory |
|
|
|
|
Payment Behaviour : |
Usually correct |
|
|
|
|
Litigation : |
Exist |
|
|
|
|
Comments : |
Subject is an established company having satisfactory track record. There seems some dip in the profits of the company during FY 2015
however, reserves position of the company is healthy.General financial
position of the company is normal and acceptable for business dealing. Trade relations are reported as fair. Business is active. Payment
terms are reported to be usually correct. The company can be considered normal for business dealings at usual
trade terms and conditions. |
NOTES :
Any query related to this report can be made
on e-mail : infodept@mirainform.com
while quoting report number, name and date.
ECGC Country Risk Classification List – March 31, 2015
|
Country Name |
Previous Rating (31.12.2014) |
Current Rating (31.03.2015) |
|
India |
A1 |
A1 |
|
Risk Category |
ECGC
Classification |
|
Insignificant |
A1 |
|
Low |
A2 |
|
Moderate |
B1 |
|
High |
B2 |
|
Very High |
C1 |
|
Restricted |
C2 |
|
Off-credit |
D |
EXTERNAL AGENCY RATING
|
Rating Agency Name |
CARE |
|
Rating |
Long-term Bank Facilities BBB- |
|
Rating Explanation |
Moderate degree of safety and carry moderate
credit risk. |
|
Date |
July 04, 2014 |
|
Rating Agency Name |
CARE |
|
Rating |
Short-term Bank Facilities A3 |
|
Rating Explanation |
Moderate degree of safety and carry higher
credit risk. |
|
Date |
July 04, 2014 |
RBI DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available RBI Defaulters’ list.
EPF (Employee Provident Fund) DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available EPF (Employee Provident Fund) Defaulters’ list as of
31-03-2014.
INFORMATION DECLINED
Management non cooperative (91-40-30211600)
LOCATIONS
|
Registered Office / Corporate Office : |
Sanali Info Park, ‘A’ Block, Ground Floor, 8-2-120/113, Road No. 2, Banjara Hills, Hyderabad – 500034, Telangana, India |
|
Tel. No.: |
91-40-30211600/ 23551081 |
|
Fax No.: |
91-40-30211602 |
|
E-Mail : |
|
|
Website : |
|
|
Area : |
6000 Sq.ft. |
|
Location : |
Rented |
|
|
|
|
Manufacturing Facilities : |
Unit 1: Village: Bonthapally Mandal: Jinnaram District: Medak,
Andhra Pradesh Unit 2 IDA, Pashamylaram, Isnapur, Patancheru (M) Medak, Dist – 502319, Andhra Pradesh, India
Unit 3 Plot No. 92-94, 257-259
Industrial Development Area, Village: Pashamylaram,
Mandal: Patancheru, District: Medak, |
|
|
|
|
Overseas Office : |
US Office 2500, Regency
Parkway, Tel No: +1 (919) 654 6833 Fax No: +1 (919) 654 6834 E-Mail: johnpinna@neulandabs.com 2F Maruishi
Building Bekkan (Annex), 1-10-1 Kajicho, Chiyoda-ku, Tel No: 81-3-3526-5171 Fax No: 81-3-3526-5172 E-Mail: ykizawa@neulandlabs.com |
|
|
|
|
Research and
Development: |
Bonthapalli (V), Veerabhadraswamy Temple Road, Jinnaram (M), Medak, District – 502313, Andhra Pradesh, India |
DIRECTORS
As on 31.03.2014
|
Name : |
Dr. Davuluri Rama Mohan Rao |
|
Designation : |
Chairman and Managing Director |
|
Date of Birth/ Age : |
69 Years |
|
Qualification : |
Masters in Science from Andhra University, Post Graduate Diploma in Technology from IIT Kharagpur and a PhD in Organic Chemistry from the University of Notre Dame |
|
Expertise in
specific functional areas : |
General Management |
|
Date of Appointment : |
07.01.1984 |
|
|
|
|
Name : |
Mr. Davuluri Sucheth Rao |
|
Designation : |
Whole-time Director and Chief Executive Officer |
|
Qualification : |
Mechanical Engineer by profession and has a MBA in Corporate Finance and Operations Management from University of Notre Dame, USA |
|
|
|
|
Name : |
Mr. Davuluri Saharsh Rao |
|
Designation : |
Whole-time Director and President-Contract Research |
|
Qualification : |
Engineering Graduate and obtained his Masters in MIS from Weatherhhead School of Management, Cleveland, Ohio, USA. |
|
|
|
|
Name : |
Mr. G. V. K. Rama Rao |
|
Designation : |
Non-Executive Director |
|
Date of Birth/ Age : |
82 Years |
|
Qualification : |
LLB |
|
Expertise in
specific functional areas : |
Civil Litigation |
|
Date of Appointment : |
07.01.1984 |
|
|
|
|
Name : |
Mr. Humayun Dhanrajgir |
|
Designation : |
Independent Director |
|
Date of Birth/ Age : |
77 Years |
|
Qualification : |
Tech, MI, CHEM (E) |
|
Expertise in
specific functional areas : |
General Management and Pharmaceutical industry |
|
Date of Appointment : |
23.08.1994 |
|
|
|
|
Name : |
Mr. Shashi Bhushan Budhiraja* |
|
Designation : |
Independent |
|
Qualification : |
B. Tech |
|
|
|
|
Name : |
Mr. Parampally Vasudeva Maiya |
|
Designation : |
Independent Director |
|
Date of Birth/ Age : |
75 Years |
|
Qualification : |
Master of Arts |
|
Expertise in
specific functional areas : |
General Management and Finance |
|
Date of Appointment : |
24.07.1999 |
|
|
|
|
Name : |
Dr. William Gordon Mitchell |
|
Designation : |
Independent Director |
|
Date of Birth/ Age : |
61 Years |
|
Qualification : |
PhD from the School of Business Administration of the University of California, Berkeley |
|
Expertise in
specific functional areas : |
Corporate Strategy and International Business |
|
Date of Appointment : |
23.05.2008 |
|
|
|
|
Name : |
Dr. Christopher M. Cimarusti |
|
Designation : |
Non-Executive Director |
|
Qualification : |
PhD in Organic Chemistry |
|
|
|
|
Name : |
Mr. Nadeem Askari Panjetan |
|
Designation : |
Nominee Director |
|
Qualification : |
Commerce Graduate and has completed his Post-Graduation in Master of Arts. |
|
|
|
|
Name : |
Mrs. Bharati Rao # |
|
Designation : |
Independent Director |
|
Date of Birth/ Age : |
65 |
|
Qualification : |
M.A[Econ], CAIIB |
|
Expertise in specific
functional areas : |
General management and Finance |
|
Date of Appointment : |
09.05.2014 |
NOTE
# Appointed as a Director w.e.f. from May 9, 2014
* Ceased to be a Director w.e.f. from May 9, 2014
KEY EXECUTIVES
|
Name : |
Mr. N.S. Viswanathan |
|
Designation : |
Chief Financial Officer |
|
|
|
|
Name : |
Ms. Sarada Bhamidipati |
|
Designation : |
Company Secretary and Compliance officer |
|
|
|
|
Name : |
Mr. Satyanarayan Rao |
|
Designation : |
Senior Manager Accounts |
|
|
|
|
AUDIT COMMITTEE : |
· Mr. Parampally Vasudeva Maiya · Mr. Humayun Dhanrajgir · Mr. Shashi Bhushan Budhiraja* · Mr. Nadeem Panjetan · Mr. D. Sucheth Rao · Mrs. Bharati Rao# # Appointed w.e.f. from May 9, 2014 * Ceased to be a Member w.e.f. from May 9, 2014 |
|
|
|
|
NOMINATION AND
REMUNERATION COMMITTEE : |
Mr. Parampally Vasudeva Maiya Mr. Humayun Dhanrajgir Mr. Shashi Bhushan Budhiraja* Mrs. Bharati Rao# # Appointed w.e.f. from May 9, 2014 * Ceased to be a Member w.e.f. from May 9, 2014 |
|
|
|
|
STAKEHOLDERS RELATIONSHIP
COMMITTEE : |
· Mr. Parampally Vasudeva Maiya · Dr. Davuluri Rama Mohan Rao · Mr. D. Sucheth Rao |
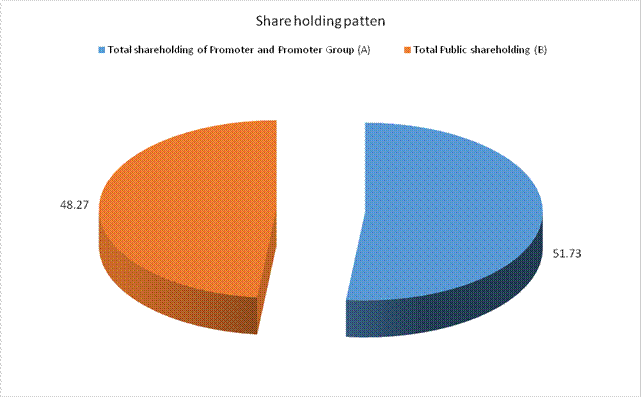
MAJOR SHAREHOLDERS / SHAREHOLDING PATTERN
As on 31.03.2015
|
Names of Shareholders |
No. of Shares |
Percentage of
Holding |
|
(A) Shareholding of Promoter and Promoter Group |
|
|
|
|
|
|
|
|
4819 |
0.05 |
|
|
4590608 |
51.68 |
|
|
4595427 |
51.73 |
|
|
|
|
|
|
200 |
0.00 |
|
|
200 |
0.00 |
|
Total shareholding of Promoter and Promoter Group (A) |
4595627 |
51.73 |
|
(B) Public Shareholding |
|
|
|
|
|
|
|
|
88685 |
1.00 |
|
|
2698 |
0.03 |
|
|
132996 |
1.50 |
|
|
224379 |
2.53 |
|
|
|
|
|
|
529869 |
5.96 |
|
|
|
|
|
|
1754035 |
19.75 |
|
|
953373 |
10.73 |
|
|
825971 |
9.30 |
|
|
40801 |
0.46 |
|
|
600000 |
6.75 |
|
|
185170 |
2.08 |
|
|
4063248 |
45.74 |
|
Total Public shareholding (B) |
4287627 |
48.27 |
|
Total (A)+(B) |
8883254 |
100.00 |
|
(C) Shares held by Custodians and against which Depository
Receipts have been issued |
0 |
0.00 |
|
|
0 |
0.00 |
|
|
0 |
0.00 |
|
|
0 |
0.00 |
|
Total (A)+(B)+(C) |
8883254 |
0.00 |
BUSINESS DETAILS
|
Line of Business : |
Manufacturing and Selling of Bulk Drugs. |
|
|
|
|
Products : |
Not Available |
|
|
|
|
Brand Names : |
Not Available |
|
|
|
|
Agencies Held : |
Not Available |
|
|
|
|
Exports : |
Not Divulged |
|
|
|
|
Imports : |
Not Divulged |
|
|
|
|
Terms : |
|
|
Selling : |
Not Divulged |
|
|
|
|
Purchasing : |
Not Divulged |
PRODUCTION STATUS NOT AVAILABLE
GENERAL INFORMATION
|
Suppliers : |
|
||||||||||||||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||||||||||||||
|
Customers : |
|
||||||||||||||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||||||||||||||
|
No. of Employees : |
Information declined by the management |
||||||||||||||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||||||||||||||
|
Bankers : |
· Export Import Bank of India, Khairatabad, Hyderabad, Andhra Pradesh, India · State Bank of India, Overseas Branch, Jubilee Hills, Hyderabad, Andhra Pradesh, India · Bank Of India, Mid Corporate Branch, Hyderabad, Andhra Pradesh, India · Indian Overseas Bank, Basheerbagh Branch, Hyderabad, Andhra Pradesh, India · SBI Global Factors Limited, Mumbai, Maharashtra, India · Consortium Bank |
||||||||||||||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||||||||||||||
|
Facilities : |
Fund and
Non-Fund based Limits : Rs. 1874.000 Million (From Consortium
Bank)
|
|
Banking
Relations : |
-- |
|
|
|
|
Statutory Auditors : |
|
|
Name : |
K. S. Aiyar and Company Chartered Accountants |
|
Address : |
No. F-7 Laxmi Mills, Shakti Mills Lane, (Off Dr. E. Moses Road),
Mahalaxmi, Mumbai - 400011, Maharashtra, India |
|
|
|
|
Internal Auditors : |
|
|
Name : |
Ernst and Young LLP Chartered Accountants |
|
Address : |
7th Floor, Block III, The Oval Office, 18 I-Labs Center, Madhapur, Hyderabad – 500081, Andhra Pradesh, India |
|
|
|
|
Holding Company
w.e.f. December 5, 2012 and Enterprise owned or significantly influenced by
Key Management Personnel till such date: (As on 31.03.2014) |
Neuland Health Sciences Private Limited |
|
|
|
|
|
|
|
Fellow Subsidiary : (As on 31.03.2014) |
Neuland Pharma Research Private Limited |
|
|
|
|
Wholly Owned
Subsidiary : (As on 31.03.2014) |
· Neuland Laboratories Inc., USA · Neuland Laboratories K.K., Japan |
|
|
|
|
Partly Owned
Subsidiary : (As on 31.03.2014) |
CATO Research Neuland (India) Private Limited |
CAPITAL STRUCTURE
As on 31.03.2015
Authorised Capital : Not Available
Issued, Subscribed & Paid-up Capital : Rs.89.539
Million
As on 24.06.2014
Authorised Capital : Rs.160.000 Million
Issued, Subscribed & Paid-up Capital : Rs.88.832
Million
As on 31.03.2014
Authorised Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
10000000 |
Equity Shares |
Rs.10/- each |
Rs.100.00 Million |
|
300000 |
Cumulative Redeemable Preference Shares |
Rs.100/- each |
Rs.30.00 Million |
|
300000 |
Cumulative or Non-cumulative and Redeemable Preference Shares |
Rs.100/- each |
Rs.30.00 Million |
|
|
|
|
|
|
|
Total |
|
Rs. 160.000
Million |
Issued, Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
7849223 |
Equity Shares |
Rs.10/- each |
Rs. 78.492 Million |
|
|
|
|
|
Subscribed Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
7758954 |
Equity Shares |
Rs.10/- each |
Rs. 77.590 Million |
|
|
|
|
|
Paid-up Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
7655678 |
Equity Shares |
Rs.10/- each |
Rs.76.557 Million |
|
|
Add: Forfeited Shares |
|
Rs.0.706 Million |
|
|
|
|
|
|
|
Total |
|
Rs. 77.263
Million |
NOTE
a. Reconciliation of
number of equity shares outstanding at the beginning and at the end of the year
|
Particulars |
As on March 31, 2014 |
|
Number of equity shares outstanding at the beginning of the year |
7644978 |
|
Add: Issued during the year (Refer Note 3(b)) |
10700 |
|
Number of equity shares outstanding at the end of the year |
7655678 |
b. (i) During the current year, the Company had on January 21, 2014, issued and allotted 10,700 fully paid up equity shares of a face value of Rs. 10 each, to eligible employees pursuant to exercise of stock options granted under Employee Stock Option Scheme, 2008.
(ii) During the previous year, the Company had on April 27, 2012, allotted 22,48,523 equity shares of a face value
of Rs. 10 each for cash at a price of Rs. 45 per equity share, including a Share Premium of Rs. 35 per equity share, aggregating to Rs. 101.184 Million to the existing equity shareholders of the Company on a rights basis in the ratio of 5 shares for every 12 shares held. Expenses incurred by the Company in relation to Rights Issue activity aggregating to Rs. 5.808 Million were adjusted to the securities premium account.
c. During the year, the Board of Directors of the Company at its meeting held on February 5, 2014 approved the issue of Equity Shares on rights basis up to Rs. 250.000 Million and the Company has filed the Draft Letter of Offer dated March 26, 2014 with the Securities and Exchange Board of India (SEBI) on March 27, 2014. Subsequently, the Company had received in-principle approval from The National Stock Exchange of India Ltd. and BSE Ltd. in respect of the proposed Rights Issue of the Company.
Proceeds of the proposed Rights issue will be utilized (i) to meet incremental working capital requirement, and (ii)
to meet issue expenses.
The Company has incurred expenses aggregating Rs. 0.636 Million in relation to the proposed Rights Issue which have been disclosed as “Rights Issue Expenses” under “Other Assets”. These expenses will be charged to the securities premium account proposed to be received from the Rights Issue of the equity shares of the Company.
d. Terms / Rights
attached to equity shares
The Company has only one class of equity shares having par value of Rs.10 per share. Each shareholder of equity shares is entitled to one vote per share. The Company declares and pays dividend in Indian Rupees. The dividend proposed by the Board of Directors is subject to prior consent from the Banks and approval of the shareholders in the ensuing Annual General Meeting.
In the event of liquidation of the Company, the holders of the equity shares will be entitled to receive remaining assets of the Company, after distribution of all preferential amounts. The distribution will be in proportion to the number of equity shares held by the shareholder.
e. Details of
Shareholders holding more than 5% shares in the Company
|
Name of the Share
holder |
As on March 31, 2014 |
|
|
No. Of shares |
% Of holding |
|
|
Equity Shares of
Rs. 10 each fully paid up |
|
|
|
1 Neuland Health Sciences Private Limited |
38,31,679 |
50.05 |
|
2 Unipharm Limited |
6,00,000 |
7.84 |
f. Employee Stock
Option Scheme – 2008
Pursuant to the resolution passed by the Board of directors on July 20, 2007 and members of the Company at the
Annual General Meeting held on July 20, 2007, the Company had introduced Employee Stock Option Scheme (“the scheme”) for permanent employees and directors of the Company and of its subsidiaries, as may be decided by the Compensation Committee/Board. The scheme provides that the total number of options granted there under will be not more than 3% of the paid up capital. Each option, on exercise, is convertible into one equity share of the Company having face value of Rs. 10. Pursuant to a resolution passed by the Remuneration and Compensation Committee vide Circular Resolution dated November 17, 2008, 34,500 options have been granted at an exercise price of Rs. 104, which is the market price as on the date of the grant. Accordingly, the Company has not recognized any expense on account of grant of stock options.
Stock options
activity under the scheme is as follows:
|
Description |
For the Year
ended March 31, 2014 |
|
Option Outstanding at the beginning of the year |
19,000 |
|
Options Granted |
-- |
|
Options Exercised |
10,700 |
|
Options Lapsed |
1,000 |
|
Options Outstanding at the year end |
7,300 |
Pursuant to the Rights Issue during the previous year, the Company revised the exercise price of each stock option to Rs. 95.34 from Rs. 104.00 (pre-rights) under the existing scheme.
FINANCIAL DATA
[all figures are
in Rupees Million]
ABRIDGED BALANCE SHEET
|
SOURCES OF FUNDS |
31.03.2015 |
31.03.2014 |
31.03.2013 |
|
I.
EQUITY
AND LIABILITIES |
|
|
|
|
(1)Shareholders'
Funds |
|
|
|
|
(a) Share Capital |
89.539 |
77.263 |
77.156 |
|
(b) Reserves & Surplus |
1508.714 |
1148.719 |
908.675 |
|
(c) Money
received against share warrants |
0.000 |
0.000 |
0.000 |
|
|
|
|
|
|
(2)
Share Application money pending allotment |
0.000 |
0.000 |
0.000 |
|
Total
Shareholders’ Funds (1) + (2) |
1598.253 |
1225.982 |
985.831 |
|
|
|
|
|
|
(3) Non-Current
Liabilities |
|
|
|
|
(a) long-term borrowings |
332.215 |
338.700 |
511.103 |
|
(b) Deferred tax liabilities (Net) |
122.455 |
102.635 |
27.074 |
|
(c) Other long term
liabilities |
27.300 |
29.300 |
33.300 |
|
(d) long-term
provisions |
63.848 |
51.388 |
47.050 |
|
Total Non-current
Liabilities (3) |
545.818 |
522.023 |
618.527 |
|
|
|
|
|
|
(4) Current Liabilities |
|
|
|
|
(a) Short
term borrowings |
1457.981 |
1396.162 |
1324.657 |
|
(b) Trade
payables |
1139.660 |
796.081 |
814.867 |
|
(c) Other
current liabilities |
309.549 |
590.791 |
426.241 |
|
(d) Short-term
provisions |
60.431 |
153.711 |
76.519 |
|
Total Current
Liabilities (4) |
2967.621 |
2936.745 |
2642.284 |
|
|
|
|
|
|
TOTAL |
5111.692 |
4684.750 |
4246.642 |
|
|
|
|
|
|
II.
ASSETS |
|
|
|
|
(1) Non-current assets |
|
|
|
|
(a) Fixed
Assets |
|
|
|
|
(i)
Tangible assets |
1645.428 |
1302.879 |
1351.308 |
|
(ii)
Intangible Assets |
0.000 |
5.656 |
3.926 |
|
(iii)
Capital work-in-progress |
0.000 |
345.616 |
356.888 |
|
(iv)
Intangible assets under development |
0.000 |
0.000 |
0.000 |
|
(b) Non-current Investments |
76.680 |
76.441 |
76.408 |
|
(c) Deferred tax assets (net) |
0.000 |
0.000 |
0.000 |
|
(d) Long-term Loan and Advances |
238.291 |
213.788 |
111.573 |
|
(e) Other
Non-current assets |
0.000 |
74.571 |
80.944 |
|
Total Non-Current
Assets |
1960.399 |
2018.951 |
1981.047 |
|
|
|
|
|
|
(2) Current assets |
|
|
|
|
(a)
Current investments |
0.000 |
0.000 |
0.000 |
|
(b)
Inventories |
1134.040 |
934.174 |
921.685 |
|
(c) Trade
receivables |
1288.483 |
1165.932 |
984.950 |
|
(d) Cash
and cash equivalents |
64.866 |
2.886 |
2.847 |
|
(e)
Short-term loans and advances |
488.664 |
350.421 |
257.981 |
|
(f) Other
current assets |
175.240 |
212.386 |
98.132 |
|
Total
Current Assets |
3151.293 |
2665.799 |
2265.595 |
|
|
|
|
|
|
TOTAL |
5111.692 |
4684.750 |
4246.642 |
PROFIT & LOSS
ACCOUNT
|
|
PARTICULARS |
31.03.2015 |
31.03.2014 |
31.03.2013 |
|
|
|
SALES |
|
|
|
|
|
|
|
Income |
4450.638 |
4656.933 |
4605.865 |
|
|
|
Other Income |
248.684 |
34.148 |
33.188 |
|
|
|
TOTAL (A) |
4699.322 |
4691.081 |
4639.053 |
|
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
|
|
Cost of Raw Materials Consumed |
2664.178 |
2464.422 |
2726.062 |
|
|
|
(Increase) / Decrease in Inventories of Work in Process
and Finished Goods |
(224.289) |
1.280 |
(34.496) |
|
|
|
Employee Benefits Expense |
489.661 |
362.551 |
348.827 |
|
|
|
Manufacturing Expenses |
556.322 |
484.224 |
458.440 |
|
|
|
Other Expenses |
541.995 |
650.606 |
525.736 |
|
|
|
TOTAL (B) |
4027.867 |
3963.083 |
4024.569 |
|
|
|
|
|
|
|
|
|
PROFIT/
(LOSS) BEFORE INTEREST, TAX,
DEPRECIATION AND AMORTISATION (A-B) (C) |
671.455 |
727.998 |
614.484 |
|
|
|
|
|
|
|
|
|
Less |
FINANCIAL
EXPENSES (D) |
273.486 |
236.376 |
313.300 |
|
|
|
|
|
|
|
|
|
|
PROFIT
/ (LOSS) BEFORE TAX, DEPRECIATION AND AMORTISATION (C-D) (E) |
397.969 |
491.622 |
301.184 |
|
|
|
|
|
|
|
|
|
Less/ Add |
DEPRECIATION/
AMORTISATION (F) |
153.198 |
149.386 |
146.229 |
|
|
|
|
|
|
|
|
|
|
PROFIT/
(LOSS) BEFORE TAX (E-F)
(G) |
244.771 |
342.236 |
154.955 |
|
|
|
|
|
|
|
|
|
Less |
TAX (H) |
86.958 |
75.561 |
18.555 |
|
|
|
|
|
|
|
|
|
|
PROFIT/ (LOSS) AFTER TAX (G-H) (I) |
157.813 |
266.675 |
136.400 |
|
|
|
|
|
|
|
|
|
Add |
PREVIOUS
YEARS’ BALANCE BROUGHT FORWARD |
432.596 |
219.282 |
82.162 |
|
|
|
|
|
|
|
|
|
Add |
ADJUSTMENTS TO
OPENING RESERVES |
NA |
0.000 |
14.791 |
|
|
|
|
|
|
|
|
|
Less |
APPROPRIATIONS |
|
|
|
|
|
|
|
Proposed Equity Dividend |
NA |
22.967 |
9.174 |
|
|
|
Tax on Proposed Equity Dividend |
NA |
3.726 |
1.488 |
|
|
|
Transfer to General Reserve |
NA |
26.668 |
3.409 |
|
|
BALANCE CARRIED
TO THE B/S |
NA |
432.596 |
219.282 |
|
|
|
|
|
|
|
|
|
|
EARNINGS IN
FOREIGN CURRENCY |
|
|
|
|
|
|
|
Export of Goods on FOB basis |
NA |
3193.308 |
3331.435 |
|
|
TOTAL EARNINGS |
NA |
3193.308 |
3331.435 |
|
|
|
|
|
|
|
|
|
|
IMPORTS |
|
|
|
|
|
|
|
Raw Materials |
NA |
1096.403 |
1148.950 |
|
|
|
Capital Goods |
NA |
0.000 |
0.000 |
|
|
TOTAL IMPORTS |
NA |
1096.403 |
1148.950 |
|
|
|
|
|
|
|
|
|
|
Earnings /
(Loss) Per Share (Rs.) |
|
|
|
|
|
|
Basic |
18.47 |
34.87 |
17.84 |
|
|
|
Diluted |
18.47 |
34.86 |
17.82 |
|
CURRENT MATURITIES
OF LONG TERM DEBT DETAILS
|
Particulars |
31.03.2015 |
31.03.2014 |
31.03.2013 |
|
Current Maturities of Long term debt |
NA |
166.105 |
269.031 |
|
Cash generated from operations |
NA |
NA |
NA |
KEY RATIOS
|
PARTICULARS |
|
31.03.2015 |
31.03.2014 |
31.03.2013 |
|
Net Profit Margin (PAT / Sales) |
(%) |
3.55 |
5.73 |
2.96 |
|
|
|
|
|
|
|
Operating Profit Margin |
(%) |
15.09 |
7.35 |
3.36 |
|
|
|
|
|
|
|
Return on Total Assets (PBT/Total Assets} |
(%) |
4.86 |
8.03 |
4.06 |
|
|
|
|
|
|
|
Return on Investment (ROI) (PBT/Networth) |
|
0.15 |
0.28 |
0.16 |
|
|
|
|
|
|
|
Debt Equity Ratio (Total Debt/Networth) |
|
1.12 |
1.42 |
1.86 |
|
|
|
|
|
|
|
Current Ratio (Current Asset/Current Liability) |
|
1.06 |
0.91 |
0.86 |
FINANCIAL ANALYSIS
[all figures are
in Rupees Million]
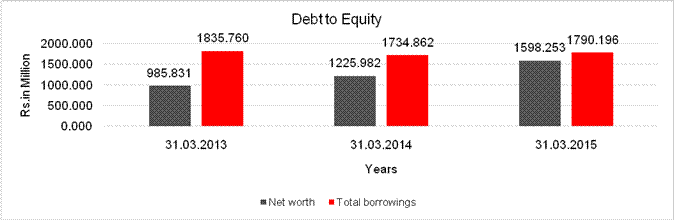
DEBT EQUITY RATIO
|
Particular |
31.03.2013 |
31.03.2014 |
31.03.2015 |
|
|
(Rs.
In Million) |
(Rs.
In Million) |
(Rs.
In Million) |
|
Share Capital |
77.156 |
77.263 |
89.539 |
|
Reserves & Surplus |
908.675 |
1148.719 |
1508.714 |
|
Net
worth |
985.831 |
1225.982 |
1598.253 |
|
|
|
|
|
|
long-term borrowings |
511.103 |
338.700 |
332.215 |
|
Short term borrowings |
1324.657 |
1396.162 |
1457.981 |
|
Total
borrowings |
1835.760 |
1734.862 |
1790.196 |
|
Debt/Equity
ratio |
1.862 |
1.415 |
1.120 |
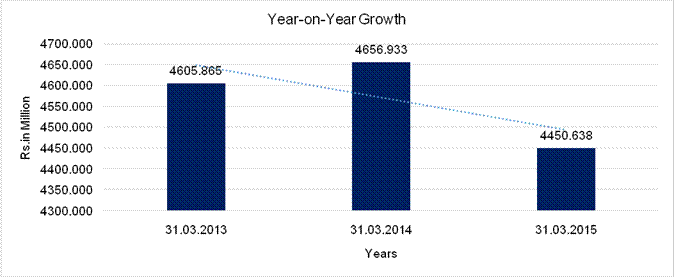
YEAR-ON-YEAR GROWTH
|
Year
on Year Growth |
31.03.2013 |
31.03.2014 |
31.03.2015 |
|
|
(Rs.
In Million) |
(Rs.
In Million) |
(Rs.
In Million) |
|
Sales |
4605.865 |
4656.933 |
4450.638 |
|
|
|
1.109 |
(4.430) |
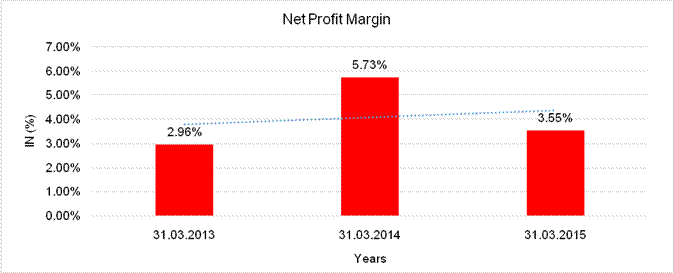
NET PROFIT MARGIN
|
Net
Profit Margin |
31.03.2013 |
31.03.2014 |
31.03.2015 |
|
|
(Rs.
In Million) |
(Rs.
In Million) |
(Rs.
In Million) |
|
Sales |
4605.865 |
4656.933 |
4450.638 |
|
Profit |
136.400 |
266.675 |
157.813 |
|
|
2.96% |
5.73% |
3.55% |
LOCAL AGENCY FURTHER INFORMATION
|
Sr. No. |
Check List by Info Agents |
Available in
Report (Yes / No) |
|
1] |
Year of establishment |
Yes |
|
2] |
Constitution of the entity -Incorporation
details |
Yes |
|
3] |
Locality of the entity |
Yes |
|
4] |
Premises details |
No |
|
5] |
Buyer visit details |
-- |
|
6] |
Contact numbers |
Yes |
|
7] |
Name of the person contacted |
No |
|
8] |
Designation of contact person |
No |
|
9] |
Promoter’s background |
Yes |
|
10] |
Date of Birth of Proprietor / Partners /
Directors |
Yes |
|
11] |
Pan Card No. of Proprietor / Partners |
No |
|
12] |
Voter Id Card No. of Proprietor / Partners |
No |
|
13] |
Type of business |
Yes |
|
14] |
Line of Business |
Yes |
|
15] |
Export/import details (if applicable) |
No |
|
16] |
No. of employees |
No |
|
17] |
Details of sister concerns |
Yes |
|
18] |
Major suppliers |
No |
|
19] |
Major customers |
No |
|
20] |
Banking Details |
Yes |
|
21] |
Banking facility details |
Yes |
|
22] |
Conduct of the banking account |
-- |
|
23] |
Financials, if provided |
Yes |
|
24] |
Capital in the business |
Yes |
|
25] |
Last accounts filed at ROC, if applicable |
Yes |
|
26] |
Turnover of firm for last three years |
Yes |
|
27] |
Reasons for variation <> 20% |
-- |
|
28] |
Estimation for coming financial year |
No |
|
29] |
Profitability for last three years |
Yes |
|
30] |
Major shareholders, if available |
Yes |
|
31] |
External Agency Rating, if available |
Yes |
|
32] |
Litigations that the firm/promoter
involved in |
-- |
|
33] |
Market information |
-- |
|
34] |
Payments terms |
No |
|
35] |
Negative Reporting by Auditors in the
Annual Report |
No |
LITIGATION DETAILS
|
ITTA 89 / 2012 |
ITTASR 3200 / 2008 |
CASE IS:PENDING |
|
||||||||||||
|
||||||||||||
|
INDEX OF CHARGES
|
S.No. |
Charge ID |
Date of Charge Creation/Modification |
Charge amount secured |
Charge Holder |
Address |
Service Request Number (SRN) |
|
1 |
10528730 |
29/10/2014 |
200,000,000.00 |
THE RATNAKAR BANK LIMITED |
SHAHUPURI,KOLHAPUR, KOLHAPUR, Maharashtra - 416001, INDIA |
C32009565 |
|
2 |
10529935 |
23/09/2014 |
150,000,000.00 |
KOTAK MAHINDRA BANK LIMITED |
27BKC, C 27, G Block, Bandra Kurla Complex, Bandra (E), Mumbai,
Maharashtra - 400051, INDIA |
C32801011 |
|
3 |
10507513 |
27/06/2014 |
200,000,000.00 |
Bank of India |
Hyderabad Mid corporate branch, 10-1-1199/2, 1st Floor, P.T.I
Building, A.C.Guards, Hyderabad, Telangana - 500004, INDIA |
C10168599 |
|
4 |
10317280 |
30/06/2014 * |
1,874,000,000.00 |
State Bank of India |
Overseas Branch, Plot no. 241/A, Rajala Centre,, Road No.36, Jubilee
Hills, Hyderabad, Telangana - 500033, INDIA |
C10194207 |
|
5 |
10312190 |
08/07/2013 * |
100,000,000.00 |
EXPORT-IMPORT BANK OF INDIA |
Floor 21, Center One Building,, World Trade Center, Cuffee Parade,
Mumbai, Maharashtra - 400005, INDIA |
B81105207 |
|
6 |
10243563 |
07/01/2014 * |
50,000,000.00 |
SBI GLOBAL FACTORS LIMITED |
6TH FLOOR, METROPOLITAN BUILDING, BANDRA-KURLA, C |
B95077830 |
|
7 |
10199917 |
23/01/2010 |
220,000,000.00 |
State Bank of India |
Overseas Branch, 5-9-300, Abids, Hyderabad, Andhra Pradesh - 500001,
INDIA |
A78226974 |
|
8 |
10082478 |
08/07/2013 * |
300,000,000.00 |
EXPORT-IMPORT BANK OF INDIA |
Floor 21, Center One Building,, World Trade Center, Cuffee Parade,
Mumbai, Maharashtra - 400005, INDIA |
B81101784 |
|
9 |
90135025 |
07/01/2014 * |
1,509,000,000.00 |
State Bank of India |
Overseas Branch, Plot no. 241/A, Rajala Towers,, |
B94788759 |
* Date of charge modification
UNSECURED LOANS
|
PARTICULAR |
31.03.2015 (Rs.
In Million) |
31.03.2014 (Rs.
In Million) |
|
LONG TERM
BORROWINGS |
|
|
|
Inter corporate deposit |
NA |
250.000 |
|
|
|
|
|
SHORT TERM
BORROWINGS |
|
|
|
Inter corporate deposit from related party |
NA |
30.000 |
|
Total |
NA |
280.000 |
CORPORATE INFORMATION
(As on 31.03.2014)
The Company is a public company domiciled in India and incorporated under the provisions of the Companies Act, 1956. Its shares are listed on two stock exchanges in India. The Company is engaged in the manufacturing and selling of bulk drugs. The Company caters to both domestic and international markets.
FINANCIAL RESULTS
For the financial year ended March 31, 2014, while there was a marginal growth in revenues, the EBIDTA grew by 17.16% to reach Rs. 728.000 Million and Profit after tax (PAT) increased to Rs. 266.700 Million from Rs. 136.400 Million in the previous financial year, recording a growth of 95.51%.
BUSINESS REVIEW
The Company continued to take key initiatives on product mix changes, scaling up new processes and improving
manufacturing processes, which enabled the Company to improve the profitability.
The Company has completed all necessary modifications to enable manufacturing and sales as envisaged in the Joint Business arrangement with API Corporation, Japan, which is part of the Mitsubishi Chemical for manufacturing and selling of APIs and Intermediates to Japan, it is anticipated that actual sales would commence by end of Q1 FY2015.
The Company is in the process of taking further systemic improvements, like product mix changes, cost optimization and process improvements, which are expected to have a positive impact on the profit margins, efficiencies and overall profitability. The management is working with a focus to build a sustainable business model which would add value to all stakeholders over the years to come.
During the year under review, the Company also successfully completed inspections by various regulatory authorities including USFDA, EMA (Europe), ANVISA (Brazil) and Cofepris (Mexico).
OUTLOOK
The Company is in a competitive environment, however its portfolio mix provides it with opportunities for growth and the Company is building on that potential by scaling up a further 9 products in the next year which will add to the pipeline and could boost growth prospects.
The Company’s quest for growth has been further aided by the significant custom manufacturing opportunities received by the Company. The research and development work done in the past has helped the Company to become a significant competitor in the custom manufacturing market as the Company has gained a better understanding of its customers’ needs. The Company’s forays in this segment have yielded considerable success in the form of securing contracts for manufacturing commercial compounds as well as clinical development candidates.
The Company has the requisite capabilities to build on its success so far and use the momentum for driving further growth.
MANAGEMENT DISCUSSION
AND ANALYSIS
ECONOMIC OVERVIEW
The Global efforts to revive the economy have started showing results in some countries. As per IMF WEO Outlook 2014, Global economy would grow at 3.6% in 2014 and 3.9% in 2015 up from 3.3% in 2013. Advanced economies are expected to grow at ~2.2 % with United States leading the lot. Emerging Economies are expected to grow at 5% in 2014 and a little above 5% in the next year from a 4.7% growth in 2013. The trends suggest that the emerging economies will draw impetus to grow from external demand only mostly from the Developed World. The biggest challenge, therefore for the emerging economies is to maintain the supply – demand ratio. The growth is expected to be modest based on the financial vulnerability of these countries to the developed World.
Indian Economy, alike other Emerging economies, is also faced with the challenges like currency devaluation, inflation and external pressures to demand. India’s GDP grew at a moderate 4.4 % in 2013 which is the lowest growth in the decade for the country. The growth of the country has been majorly affected by the external developments as for other emerging countries apart from lesser domestic demand. Internal issues like pressing Inflation rates and domestic policies have been rendering negative effect on the country’s growth. Delayed investment approvals and implementations, supply bottlenecks, external policies etc all add to the hindrances. As per IMF projections, the growth of ~5.5% can be expected if conditions like stronger global growth, improving export competitiveness, a positive result of recent policy changes come into play.
PHARMACEUTICAL
INDUSTRY LANDSCAPE
The Global Pharmaceutical industry is volatile and is undergoing changes at the moment due to aging populations
and expanding market growth. IMS Health, in its latest report, “The Global use of Medicines: Outlook through 2017”, predicts global spending on medicines to reach almost $1.2 trillion in 2017. With medical awareness, emphasis on universal medical coverage, ensured access to medicines and healthcare, the industry is expected to grow over the years to come. IMS health divides market into two parts- Developed market and ‘Pharmerging’. Developed markets include North America, USA and Japan while the Pharmerging market is composed of 7 emerging countries - Brazil, India, Turkey, Mexico, Russia, South Korea, and China. The report also emphasizes a modest growth in the developed markets in the next 5 years, due to economic and healthcare austerity measures. With patent expiry also approaching, availability of reasonably priced generics would also contribute to some realization. The emerging markets on the other hand are predicted to be seeing much higher double digit growth due to impetus on economic growth, demographic changes, and improved urban and socio-economic facilities. In light of innovations in new and conducive medicines, this is viewed as the future of the Pharmaceutical industry.
The Indian pharmaceutical industry is one of the fastest growing the world, and is third in terms of volume in the world. The Indian Pharmaceutical sector is expected to record revenues of $30 Bn in 2015 of which around $10 Bn will be from exports. Over the last decade, the industry has emerged as a major player in generic pharmaceutical production and now supplies around 20% of the global market for generic medicines.
They have seen the pharmaceutical industry across the world go through another phase of consolidation, acquisitions aimed at consolidation of both capabilities as well as markets. Another aspect in which they have seen substantial change is the approach of various national regulatory authorities. Regulations have become tighter and the expectations from manufacturers are higher. Another key trend which has been noted earlier but is worth mentioning is the movement of manufacturing facilities from the developed markets to India and other Pharmerging markets.
The Ministry of Commerce, Government of India, estimates investments of about US$ 6.3 billion will be made in the Indian pharmaceutical industry by 2015. The Indian pharmaceutical industry remains an attractive destination
for foreign investors, usually in the form of acquisition of Indian companies.
It is estimated that in 2015, the trade surplus from pharmaceuticals alone may move up to US$12.8 billion. It is likely that by 2015, the Indian elementary drugs share out of the aggregate supplies from Asia-Pacific may soar to 10%, only behind Japan and China.
COMPANY OVERVIEW
Neuland Laboratories has completed 30 years of existence as an API manufacturer. Neuland has consistently grown its API manufacturing business gaining expertise in different therapy segments. As a company they have been focused on delivering quality on time to their customers, which has seen us perform consistently in a very competitive environment. The Company has a rich basket of products marketed to customers across 85 countries. The Company also offers integrated and versatile manufacturing capabilities capable of handling complex reactions tuned to ensure seamless transfer from small-scale through validation to commercial manufacturing. This integration helps customers to expedite the discovery-to-market timelines.
The Company has two manufacturing facilities located close to Hyderabad which are compliant with health and regulatory agencies cGMP certification standards. The sites have been audited and certified by regulatory authorities time to time from FDA (USA), TGA (Australia), AIFA (Italy), ANSM (France), PMDA (Japan), ANVISA (Brazil), and COFEPRIS (Mexico) among others.
The Company’s strengths are in synthetic chemistry, process development and controlled supply chain coupled with a project management approach built into all its operations and product development programs.
· Neuland has made a successful foray into Custom Manufacturing Solutions building on its strengths of proven expertise in manufacturing at varied scales
· A deep understanding of complex chemistry
· Manufacturing facilities that are compliant with the guidelines of the leading regulatory authorities such as USFDA, EMA, COFEPRIS and ANVISA.
STRATEGIC REVIEW
During the past year the Company has taken a lot of steps to build on its momentum to ensure sustainable growth while enhancing profitability, strengthening the balance sheet and enhancing its credit rating. Through the last year they have taken several strategic steps to ensure that profitability is enhanced, while also ensuring top-line growth in the nearterm and as well as long-term.
Towards this end, the Company has strengthened its Marketing and Business Development capabilities by expanding the team to enable them to serve more markets as well focus better on their customers.
They have also further strengthened their product selection process such that they have a rich pipeline of products under development which will enable growth as well as sustainability in the future.
During the past year they have continued with the emphasis on changing the portfolio mix towards more profitable, lowervolume products. They have actively pursued new customers over the last year and have had orders from over 100 new customers.
They have gone further in their journey of partnership with API Corporation, by getting the infrastructure ready during this fiscal year. This partnership while translating into long-term business gains for both companies will also open more such avenues for the Company.
OUTLOOK
Buoyed by a robust financial performance in FY 2013-14, the Company believes that it can sustain the growth momentum in FY 2014-15. Its strategy to focus on niche, high-margin products with continuous expansion of its product portfolio gives it the ability to yield revenue and profitability growth over coming years. Its foray into contract manufacturing is likely to not only add to its revenues and profitability but flag-off its evolution as a custom manufacturer for many other pharmaceuticals companies. Its alliance with Mitsubishi Chemicals is expected to strengthen the Company’s revenue growth over many years.
As a result of steps taken over the past few years, they believe that Neuland is now on the threshold of a very exciting journey. Considering the steps taken to strengthen the Company in the last year and the continuous evolution of the Company’s portfolio-mix, customers and pipeline, they are positioning ourselves well to take advantage. They believe that this is an opportune time for them to leverage the opportunities opening up to them by strategically investing in their capabilities. As they continue on this trajectory of growth, all these factors in conjunction with the vision of the Company to become a leading player in API market worldwide. They believe that as they continue on their path of growth, the future is Neuland’s to claim.
CONTINGENT
LIABILITIES (AS ON 31.03.2014):
(a) Claims against the Company not acknowledged as debts
(i) Andhra Pradesh Gas Power Corporation Limited and its shareholders (including Neuland) have filed writ petition before the Division Bench of High Court of A.P, which has been admitted and favourable interim orders have been granted. The Company has been advised that it has a strong case to succeed in the pending appeal.
(ii) Certain disputes, for unascertained amounts, are pending in the Labour Courts, A.P. As the chances of appellants succeeding in their claims being remote, the Company expects no liability on this account.
(iii) Income Tax department has filed a writ petition before the Hon’ble High Court of Andhra Pradesh to set aside the Income Tax Appellate Tribunal orders for the Assessment Year 2001-2002 and 2002-2003 against the claim on deduction U/sec 80HHC. The Hon’ble High Court of Andhra Pradesh has admitted the appeal and the matter is pending. If there is an adverse ruling against our Company, the estimated financial impact on the Company would be Rs. 0.342 Million and Rs. 1.415 Million respectively.
(iv) Income Tax department has filed a writ petition before the Hon’ble High Court of Andhra Pradesh to set aside the Income Tax Appellate Tribunal order bearing TA No. 842/H/06 dated May 5, 2008 for the Assessment Year 2003-2004 against the allowability of Employee’s contribution towards PF, ESI. The Hon’ble High Court of Andhra Pradesh has admitted the appeal on June 20, 2012 and the matter is pending. If there is an adverse ruling against
our Company, the estimated financial impact on the Company would be Rs. 0.144 Million.
(v) The Company has filed an appeal before Income Tax Appellate Tribunal against the order of the Commissioner of Income Tax (Appeals) for the Assessment Year 1998-1999 against the disallowance of Commission paid to Nonresident agents for not deducted at source u/s 40(a)(i) of Income Tax Act, 1961 and the matter is pending. If there is an adverse ruling against our Company, the estimated financial impact on the Company would be Rs. 0.634 Million.
(vi) The Company has filed an appeal before Income Tax Appellate Tribunal against the order of the Commissioner of Income Tax (Appeals) for the Assessment Year 2008-2009 and 2009-2010 against the disallowance of Commission paid to Non-resident agents and other payments to Non-residents for not deducted at source u/s 40(a)(i) of Income Tax Act, 1961 and the matter is pending. If there is an adverse ruling against our Company, the estimated financial impact on the Company would be Rs. 1.917 Million and Rs. 1.682 Million respectively.
(b) Unexpired Letters of Credit opened on behalf of the Company by Bank for the raw material amounting to Rs. 350.147 Million (March 31, 2013: Rs. 352.130 Million).
(c) Bank Guarantees given by the Company to Central Excise and Customs and other Government authorities amounting to Rs. 8.000 Million (March 31, 2013: Rs. 9.500 Million).
FIXED ASSETS
TANGIBLE ASSETS
· Land
· Buildings
· Plant and Equipment
· R&D Equipment
· Data Processing Machines
· Furniture and Fixtures
· Vehicles
INTANGIBLE ASSETS
· Process Development Cost
· Computer Software - SAP
CMT REPORT (Corruption, Money Laundering & Terrorism]
The Public Notice information has been collected from various sources
including but not limited to: The Courts,
1] INFORMATION ON
DESIGNATED PARTY
No exist designating subject or any of its beneficial owners,
controlling shareholders or senior officers as terrorist or terrorist
organization or whom notice had been received that all financial transactions
involving their assets have been blocked or convicted, found guilty or against
whom a judgement or order had been entered in a proceedings for violating money-laundering,
anti-corruption or bribery or international economic or anti-terrorism sanction
laws or whose assets were seized, blocked, frozen or ordered forfeited for
violation of money laundering or international anti-terrorism laws.
2] Court Declaration :
No exist to suggest that subject is or was
the subject of any formal or informal allegations, prosecutions or other
official proceeding for making any prohibited payments or other improper
payments to government officials for engaging in prohibited transactions or
with designated parties.
3] Asset Declaration :
No records exist to suggest that the property or assets of the subject
are derived from criminal conduct or a prohibited transaction.
4] Record on Financial
Crime :
Charges or conviction
registered against subject: None
5] Records on Violation of
Anti-Corruption Laws :
Charges or
investigation registered against subject: None
6] Records on Int’l
Anti-Money Laundering Laws/Standards :
Charges or investigation
registered against subject: None
7] Criminal Records
No
available information exist that suggest that subject or any of its principals
have been formally charged or convicted by a competent governmental authority
for any financial crime or under any formal investigation by a competent
government authority for any violation of anti-corruption laws or international
anti-money laundering laws or standard.
8] Affiliation with
Government :
No record
exists to suggest that any director or indirect owners, controlling
shareholders, director, officer or employee of the company is a government
official or a family member or close business associate of a Government
official.
9] Compensation Package :
Our market
survey revealed that the amount of compensation sought by the subject is fair
and reasonable and comparable to compensation paid to others for similar
services.
10] Press Report :
No press reports / filings exists on
the subject.
CORPORATE GOVERNANCE
MIRA INFORM as part of its Due Diligence do provide comments on
Corporate Governance to identify management and governance. These factors often
have been predictive and in some cases have created vulnerabilities to credit
deterioration.
Our Governance Assessment focuses principally on the interactions
between a company’s management, its Board of Directors, Shareholders and other
financial stakeholders.
CONTRAVENTION
Subject is not known to have contravened any existing local laws,
regulations or policies that prohibit, restrict or otherwise affect the terms
and conditions that could be included in the agreement with the subject.
FOREIGN EXCHANGE RATES
|
Currency |
Unit
|
Indian Rupees |
|
US Dollar |
1 |
Rs.63.95 |
|
|
1 |
Rs.98.64 |
|
Euro |
1 |
Rs.69.86 |
INFORMATION DETAILS
|
Information
Gathered by : |
GAY |
|
|
|
|
Analysis Done by
: |
DIV |
|
|
|
|
Report Prepared
by : |
JYTK |
SCORE & RATING EXPLANATIONS
|
SCORE FACTORS |
RANGE |
POINTS |
|
HISTORY |
1~10 |
5 |
|
PAID-UP CAPITAL |
1~10 |
5 |
|
OPERATING SCALE |
1~10 |
4 |
|
FINANCIAL
CONDITION |
|
|
|
--BUSINESS SCALE |
1~10 |
5 |
|
--PROFITABILIRY |
1~10 |
4 |
|
--LIQUIDITY |
1~10 |
5 |
|
--LEVERAGE |
1~10 |
5 |
|
--RESERVES |
1~10 |
5 |
|
--CREDIT LINES |
1~10 |
4 |
|
--MARGINS |
-5~5 |
-- |
|
DEMERIT POINTS |
|
|
|
--BANK CHARGES |
YES/NO |
YES |
|
--LITIGATION |
YES/NO |
YES |
|
--OTHER ADVERSE INFORMATION |
YES/NO |
NO |
|
MERIT POINTS |
|
|
|
--SOLE DISTRIBUTORSHIP |
YES/NO |
NO |
|
--EXPORT ACTIVITIES |
YES/NO |
NO |
|
--AFFILIATION |
YES/NO |
YES |
|
--LISTED |
YES/NO |
YES |
|
--OTHER MERIT FACTORS |
YES/NO |
YES |
|
DEFAULTER |
|
|
|
--RBI |
YES/NO |
NO |
|
--EPF |
YES/NO |
NO |
|
TOTAL |
|
42 |
This score serves as a reference to assess SC’s
credit risk and to set the amount of credit to be extended. It is calculated
from a composite of weighted scores obtained from each of the major sections of
this report. The assessed factors and their relative weights (as indicated
through %) are as follows:
Financial
condition (40%) Ownership
background (20%) Payment
record (10%)
Credit history
(10%) Market trend (10%) Operational size
(10%)
RATING EXPLANATIONS
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
>86 |
Aaa |
Possesses an extremely sound financial base with the strongest
capability for timely payment of interest and principal sums |
Unlimited |
|
71-85 |
Aa |
Possesses adequate working capital. No caution needed for credit transaction.
It has above average (strong) capability for payment of interest and
principal sums |
Large |
|
56-70 |
A |
Financial & operational base are regarded healthy. General
unfavourable factors will not cause fatal effect. Satisfactory capability for
payment of interest and principal sums |
Fairly Large |
|
41-55 |
Ba |
Overall operation is considered normal. Capable to meet normal
commitments. |
Satisfactory |
|
26-40 |
B |
Capability to overcome financial difficulties seems comparatively below
average. |
Small |
|
11-25 |
Ca |
Adverse factors are apparent. Repayment of interest and principal sums
in default or expected to be in default upon maturity |
Limited with
full security |
|
<10 |
C |
Absolute credit risk exists. Caution needed to be exercised |
Credit not
recommended |
|
-- |
NB |
New Business |
-- |
This report is issued at your request without any
risk and responsibility on the part of MIRA INFORM PRIVATE LIMITED (MIPL) or
its officials.