MIRA INFORM REPORT
|
Report No. : |
348325 |
|
Report Date : |
14.11.2015 |
IDENTIFICATION DETAILS
|
Name : |
NECTAR LIFE SCIENCES LIMITED [w.e.f.26.03.2004] |
|
|
|
|
Formerly Known
As : |
SURYA MEDICARE LIMITED |
|
|
|
|
Registered
Office : |
Village Saidpura, Tehsil Dera Bassi, District Patiala - 140507, Punjab |
|
Tel. No.: |
91-1762-308000/ 91-172-3047701 |
|
|
|
|
Country : |
|
|
|
|
|
Financials (as
on) : |
31.03.2015 |
|
|
|
|
Date of
Incorporation : |
27.06.1995 |
|
|
|
|
Com. Reg. No.: |
16-016664 |
|
|
|
|
Capital
Investment / Paid-up Capital : |
Rs. 224.260 Million |
|
|
|
|
CIN No.: [Company Identification
No.] |
L24232PB1995PLC016664 |
|
|
|
|
IEC No.: |
3095006365 |
|
|
|
|
TAN No.: [Tax Deduction &
Collection Account No.] |
PTLS10181D |
|
|
|
|
PAN No.: [Permanent Account No.] |
AABCS6468G |
|
|
|
|
Legal Form : |
A Public Limited Liability Company. The Company’s Shares are Listed on
the Stock Exchanges. |
|
|
|
|
Line of Business
: |
Manufacturer and Exporter of Pharmaceutical Products. |
|
|
|
|
No. of Employees
: |
2053 (Approximately) |
RATING & COMMENTS
|
MIRA’s Rating : |
A (60) |
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
56-70 |
A |
Financial & operational base are regarded healthy. General
unfavourable factors will not cause fatal effect. Satisfactory capability for
payment of interest and principal sums |
Fairly Large |
|
Maximum Credit Limit : |
USD 27000000 |
|
|
|
|
Status : |
Good |
|
|
|
|
Payment Behaviour : |
Regular |
|
|
|
|
Litigation : |
Exist |
|
|
|
|
Comments : |
Subject was incorporated in the year 1995 and it is a pharmaceutical
company. The company offers semi synthetic penicillin and cephalosporin-based
oral and sterile bulk drugs. It also offers contract manufacturing services
and contract research services which primarily comprise medicinal chemistry
and process research and development. For the financial year ended 2015, company reported stagnant growth in
its revenue profile and it has maintained satisfactory margins at 3.97%
during the year under a review. Rating rakes into consideration company’s healthy financial profile
marked by its adequate net worth base along with healthy debt coverage indicators
and favorable liquidity position. Rating also takes into account of company’s well established track
record of business operations and extensive industry experience of promoters
in pharmaceutical segment. However, rating strength partially offset by company’s working capital
intense nature of business operations and its profitability exposed to
volatility in foreign exchange fluctuations and adverse raw material price
movements. Trade relations are reported as fair. Payments are reported to be
regular and as per commitment. The company can be considered good for normal business dealings at
usual trade terms and conditions. |
NOTES:
Any query related to this report can be made
on e-mail: infodept@mirainform.com
while quoting report number, name and date.
ECGC Country Risk Classification List – March 31, 2015
|
Country Name |
Previous Rating (31.12.2014) |
Current Rating (31.03.2015) |
|
India |
A1 |
A1 |
|
Risk Category |
ECGC
Classification |
|
Insignificant |
A1 |
|
Low |
A2 |
|
Moderate |
B1 |
|
High |
B2 |
|
Very High |
C1 |
|
Restricted |
C2 |
|
Off-credit |
D |
EXTERNAL AGENCY RATING
|
Rating Agency Name |
FITCH |
|
Rating |
Long-Term Issuer Rating (Suspended) = A- |
|
Rating Explanation |
Adequate degree of safety and low credit
risk |
|
Date |
Absence of adequate information |
RBI DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available RBI Defaulters’ list.
EPF (Employee Provident Fund) DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available EPF (Employee Provident Fund) Defaulters’ list as of
31-03-2015.
INFORMATION DENIED BY
|
Name : |
Mr. Sandeep Goel |
|
Designation : |
Chief Executive Officer |
|
Contact No.: |
91-1762-308000/ 91-172-3047701 |
|
Date : |
04.11.2015 |
LOCATIONS
|
Registered Office : |
Village Saidpura, Tehsil Dera Bassi, District Patiala – 140507,
Punjab, India |
|
Tel. No.: |
91-1762-308000/ 91-172-3047701 |
|
Mobile No.: |
Not Available |
|
Fax No.: |
91-1762-308135 |
|
E-Mail : |
|
|
Website : |
|
|
|
|
|
Plant 1: |
Nectar Lifesciences Limited, Unit I Village: Saidpura,
Tehsil Derabassi, District Mohali, Punjab, India |
|
|
|
|
Plant 2: |
Nectar Lifesciences Limited unit II Village: Saidpura,
Tehsil Derabassi, District Mohali, Punjab, India |
|
|
|
|
Plant 3: |
Empty Hard Gelatin Capsule Unit Village Bhatoli Kalan,
Pargana Dharmpur, Tehsil Nalagarh, District Solan, Himachal Pradesh, India |
|
|
|
|
Plant 4: |
Formulation Unit, Village Bhatoli Kalan, Pargana Dharmpur,
Tehsil Nalagarh, District Solan, Himachal Pradesh, India |
|
|
|
|
Plant 5: |
Narbada Industries Plot No. 2, Lane No. 4, Phase II, SIDCO
Industrial Complex Bari Brahmana, Jammu (Jammu & Kashmir), India |
DIRECTORS
AS ON 31.03.2015
|
Name : |
Mr. Sanjiv Goyal |
||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Designation : |
Chairman and Managing Director |
||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Address : |
# 309, Sec 9-D, Chandigarh - 160009, Chandigarh, India |
||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Date of Appointment : |
01.06.2012 |
||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
DIN No.: |
00002841 |
||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Other Directorship :
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Name : |
Mr. Dinesh Dua |
|||||||||||||||||||||||||||
|
Designation : |
Chief Executive Officer and Director |
|||||||||||||||||||||||||||
|
Address : |
B - 107, Pushpanjali, Encl Pitam Pura, Delhi - 110034, Delhi, India |
|||||||||||||||||||||||||||
|
Date of Appointment : |
30.09.2014 |
|||||||||||||||||||||||||||
|
DIN No.: |
02436706 |
|||||||||||||||||||||||||||
|
Other Directorship :
|
||||||||||||||||||||||||||||
|
Name : |
Mr. Harprakash Gill |
|
Designation : |
President (Operation) and Directors |
|
Address : |
H.I.G. 202, Sector 71, S.A.S Nagar, Mohali - 160071, Punjab, India |
|
Date of Appointment : |
01.11.2015 |
|
DIN No.: |
06414839 |
|
Name : |
Mr. Ajay Swaroop |
|||||||||||||||||||||||||||||||||||||||||||||
|
Designation : |
Independent Director |
|||||||||||||||||||||||||||||||||||||||||||||
|
Address : |
House No 696, Sector 8-B, Chandigarh - 160008, Chandigarh, India |
|||||||||||||||||||||||||||||||||||||||||||||
|
Date of Appointment : |
30.09.2014 |
|||||||||||||||||||||||||||||||||||||||||||||
|
DIN No.: |
02291700 |
|||||||||||||||||||||||||||||||||||||||||||||
|
Other Directorship :
|
||||||||||||||||||||||||||||||||||||||||||||||
|
Name : |
Mr. Neena Singh |
|
Designation : |
Independent Director |
|
Name : |
Mr. Raman Kapur |
|
Designation : |
Independent Director |
|
Name : |
Dr. [Maj Gen] S.S.
Chauhan, VSM [Retd.] |
|
Designation : |
Independent Director |
|
Address : |
House No. 126-A, Sector 6, Panchkula - 134109, Haryana, India |
|
Date of Appointment : |
30.09.2014 |
|
DIN No.: |
02954776 |
|
Name : |
Mr. Vijay J Shah |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Designation : |
Independent Director |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Address : |
26/B, Jain Merchant Society, Nr. Mahalaxmi Cross Road, Paldi, Ahmedabad, 380007, Gujarat, India |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Date of Appointment : |
30.09.2014 |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
DIN No.: |
00747226 |
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Other Directorship :
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Name : |
Mr. Vivek Seth |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Designation : |
Director |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Address : |
1001 Marathon Heights, P. B. Marg, Worli, Mumbai - 400013, Maharashtra, India |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Date of Appointment : |
29.09.2010 |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
DIN No.: |
00031084 |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Other Directorship :
|
|||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
KEY EXECUTIVES
|
Name : |
Mr. Sunder Lal |
|
Designation : |
Vice President and Company Secretary |
|
Address : |
House No. 437, Sector 41-A, Chandigarh - 160036, Chandigarh, India |
|
PAN No.: |
AAJPL9162B |
|
|
|
|
Name : |
Mr. Sandeep Goel |
|
Designation : |
Chief Executive Officer |
|
Address : |
House No. 468, Sector 20-A, Chandigarh - 160020, Chandigarh, India |
|
PAN No.: |
ACSPG9220E |
|
|
|
|
Name : |
Mr. R. K. Aggarwal |
|
Designation : |
Vice President |
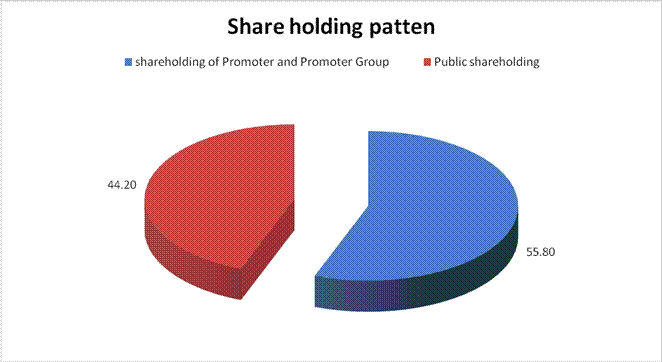
SHAREHOLDING PATTERN
AS ON 30.09.2015
|
Category of
Shareholder |
Total No. of Shares |
Total Shareholding as
a % of Total No. of Shares |
|
As a % of (A+B+C) |
||
|
(A) Shareholding of Promoter and Promoter Group |
||
|
|
|
|
|
|
99468000 |
44.35 |
|
|
99468000 |
44.35 |
|
|
|
|
|
Total shareholding of Promoter and Promoter Group (A) |
99468000 |
44.35 |
|
(B) Public Shareholding |
||
|
|
|
|
|
|
101587 |
0.05 |
|
|
2700000 |
1.20 |
|
|
3396237 |
1.51 |
|
|
26000000 |
11.59 |
|
|
26000000 |
11.59 |
|
|
32197824 |
14.36 |
|
|
|
|
|
|
15432226 |
6.88 |
|
|
|
|
|
|
24655466 |
10.99 |
|
|
4068209 |
1.81 |
|
|
2439245 |
1.09 |
|
|
2205791 |
0.98 |
|
|
193904 |
0.09 |
|
|
35550 |
0.02 |
|
|
4000 |
0.00 |
|
|
46595146 |
20.78 |
|
Total Public shareholding (B) |
78792970 |
35.13 |
|
Total (A)+(B) |
178260970 |
79.49 |
|
(C) Shares held by Custodians and against which Depository Receipts
have been issued |
0 |
0.00 |
|
|
0 |
0.00 |
|
|
46000000 |
20.51 |
|
|
46000000 |
20.51 |
|
Total (A)+(B)+(C) |
224260970 |
100.00 |
BUSINESS DETAILS
|
Line of Business : |
Manufacturer and Exporter of Pharmaceutical Products. |
||||||
|
|
|
||||||
|
Products : |
|
||||||
|
|
|
||||||
|
Brand Names : |
Not Available |
||||||
|
|
|
||||||
|
Agencies Held : |
Not Available |
||||||
|
|
|
||||||
|
Exports : |
Not Available |
||||||
|
|
|
||||||
|
Imports : |
Not Available |
||||||
|
|
|
||||||
|
Terms : |
Not Available |
PRODUCTION STATUS – NOT AVAILABLE
GENERAL INFORMATION
|
Suppliers : |
|
|||||||||||||||||||||||||||
|
|
|
|||||||||||||||||||||||||||
|
Customers : |
|
|||||||||||||||||||||||||||
|
|
|
|||||||||||||||||||||||||||
|
No. of Employees : |
2053 (Approximately) |
|||||||||||||||||||||||||||
|
|
|
|||||||||||||||||||||||||||
|
Bankers : |
· State Bank of India, Specialized Commercial Branch, Sector 17B, Chandigarh, India · State Bank of Bikaner and Jaipur, Delhi, India · Saraswat Bank, India · IFCI Limited, Chandigarh · Exim Bank, Mumbai, Maharashtra, India |
|||||||||||||||||||||||||||
|
|
|
|||||||||||||||||||||||||||
|
Facilities : |
|
|
Auditors : |
|
|
Name : |
Data and Singla and Company Chartered Accountants |
|
Address : |
SCO 2935-36, 1st floor, Sector-22-C, Chandigarh, Punjab, India |
|
|
|
|
Collaborators : |
Not Available |
|
|
|
|
Membership : |
Not Available |
|
|
|
|
Associates/Subsidiaries : |
|
|
|
|
|
Entities over which
key management personnel/their relatives are able to exercise significant
influence*: |
* With whom the company had transactions during the year. |
CAPITAL STRUCTURE
AS ON 31.03.2015
Authorised Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
350000000 |
Equity Shares |
Rs. 10/- each |
Rs. 350.000 Million |
|
|
|
|
|
Issued, Subscribed & Paid-up Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
224260970 |
Equity Shares |
Rs. 10/- each |
Rs. 224.260 Million |
|
|
|
|
|
FINANCIAL DATA
[all figures are
in Rupees Million]
ABRIDGED
BALANCE SHEET
|
SOURCES OF FUNDS |
31.03.2015 |
31.03.2014 |
31.03.2013 |
|
I.
EQUITY
AND LIABILITIES |
|
|
|
|
(1)Shareholders' Funds |
|
|
|
|
(a) Share Capital |
224.260 |
224.260 |
224.260 |
|
(b) Reserves & Surplus |
9355.220 |
9046.360 |
8451.800 |
|
(c) Money
received against share warrants |
0.000 |
0.000 |
0.000 |
|
|
|
|
|
|
(2) Share Application money pending
allotment |
0.000 |
0.000 |
0.000 |
|
Total
Shareholders’ Funds (1) + (2) |
9579.480 |
9270.620 |
8676.060 |
|
|
|
|
|
|
(3)
Non-Current Liabilities |
|
|
|
|
(a) long-term borrowings |
2655.290 |
2233.380 |
2609.610 |
|
(b) Deferred tax liabilities (Net) |
1101.930 |
1118.930 |
957.560 |
|
(c) Other long term
liabilities |
151.870 |
8.070 |
10.300 |
|
(d) long-term
provisions |
60.110 |
49.000 |
37.890 |
|
Total Non-current
Liabilities (3) |
3969.200 |
3409.380 |
3615.360 |
|
|
|
|
|
|
(4)
Current Liabilities |
|
|
|
|
(a) Short
term borrowings |
5952.980 |
5798.420 |
5091.110 |
|
(b) Trade
payables |
4555.770 |
4400.610 |
3825.190 |
|
(c) Other
current liabilities |
905.000 |
867.150 |
1312.760 |
|
(d) Short-term
provisions |
142.220 |
194.480 |
219.950 |
|
Total Current
Liabilities (4) |
11555.970 |
11260.660 |
10449.010 |
|
|
|
|
|
|
TOTAL |
25104.650 |
23940.660 |
22740.430 |
|
|
|
|
|
|
II.
ASSETS |
|
|
|
|
(1)
Non-current assets |
|
|
|
|
(a) Fixed
Assets |
|
|
|
|
(i) Tangible
assets |
9337.310 |
9475.430 |
9138.390 |
|
(ii)
Intangible Assets |
0.000 |
0.000 |
0.000 |
|
(iii)
Capital work-in-progress |
913.120 |
1222.640 |
1385.700 |
|
(iv)
Intangible assets under development |
0.000 |
0.000 |
0.000 |
|
(b) Non-current Investments |
0.500 |
0.500 |
0.500 |
|
(c) Deferred tax assets (net) |
0.000 |
0.000 |
0.000 |
|
(d) Long-term Loan and Advances |
804.430 |
868.670 |
704.860 |
|
(e) Other
Non-current assets |
359.120 |
0.070 |
0.150 |
|
Total Non-Current Assets |
11414.480 |
11567.310 |
11229.600 |
|
|
|
|
|
|
(2)
Current assets |
|
|
|
|
(a)
Current investments |
4.350 |
3.140 |
3.190 |
|
(b)
Inventories |
7909.750 |
7584.270 |
6760.000 |
|
(c) Trade
receivables |
4464.960 |
3766.500 |
3493.860 |
|
(d) Cash
and cash equivalents |
333.820 |
99.790 |
62.400 |
|
(e)
Short-term loans and advances |
814.320 |
658.300 |
868.430 |
|
(f) Other
current assets |
162.970 |
261.350 |
322.950 |
|
Total
Current Assets |
13690.170 |
12373.350 |
11510.830 |
|
|
|
|
|
|
TOTAL |
25104.650 |
23940.660 |
22740.430 |
PROFIT
& LOSS ACCOUNT
|
|
PARTICULARS |
31.03.2015 |
31.03.2014 |
31.03.2013 |
|
|
SALES |
|
|
|
|
|
Income |
16428.890 |
16377.690 |
16248.140 |
|
|
Operating Income |
7.530 |
5.430 |
14.370 |
|
|
Other Income |
199.990 |
116.490 |
160.400 |
|
|
TOTAL (A) |
16636.410 |
16499.610 |
16422.910 |
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
Cost of Materials
Consumed |
11299.040 |
11726.710 |
11465.220 |
|
|
Purchases of Stock-in-Trade |
396.060 |
398.230 |
599.980 |
|
|
Changes in inventories of
finished goods, work-in-progress and Stock-in-Trade |
(281.760) |
(578.110) |
(644.020) |
|
|
Employees benefits
expense |
627.070 |
513.290 |
486.110 |
|
|
Other expenses |
1726.940 |
1435.520 |
1454.420 |
|
|
TOTAL EXPENSES |
13767.350 |
13495.640 |
13361.710 |
|
|
|
|
|
|
|
Less |
PROFIT/ (LOSS) BEFORE INTEREST, TAX, DEPRECIATION AND
AMORTISATION |
2869.060 |
3003.970 |
3061.200 |
|
|
|
|
|
|
|
Less |
FINANCIAL EXPENSES |
1255.880 |
1379.870 |
1154.890 |
|
|
|
|
|
|
|
|
PROFIT / (LOSS) BEFORE
TAX, DEPRECIATION AND AMORTISATION |
1613.180 |
1624.100 |
1906.310 |
|
|
|
|
|
|
|
Less |
DEPRECIATION/
AMORTISATION |
765.640 |
841.860 |
759.560 |
|
|
|
|
|
|
|
|
PROFIT/ (LOSS) BEFORE TAX |
847.540 |
782.240 |
1146.750 |
|
|
|
|
|
|
|
Less |
TAX |
184.900 |
161.440 |
290.090 |
|
|
|
|
|
|
|
|
PROFIT/ (LOSS) AFTER TAX
|
662.640 |
620.800 |
856.660 |
|
|
|
|
|
|
|
|
EARNINGS / (LOSS) PER
SHARE (RS.) |
2.95 |
2.77 |
3.82 |
CURRENT MATURITIES
OF LONG TERM DEBT DETAILS
|
Particulars |
31.03.2015 |
31.03.2014 |
31.03.2013 |
|
Current Maturities of Long term debt |
635.400 |
548.170 |
1089.020 |
|
Cash generated from operations |
1,507.000 |
2,748.510 |
3626.030 |
|
Cash generated from (Used) operations |
NA |
NA |
NA |
|
Cash generated from Operating Activities |
1,444.100 |
2,548.730 |
3607.780 |
KEY
RATIOS
|
PARTICULARS |
|
31.03.2015 |
31.03.2014 |
31.03.2013 |
|
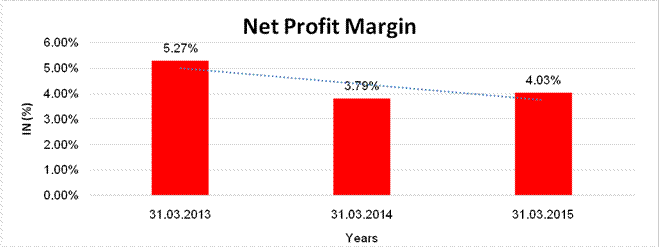
Net Profit Margin (PAT / Sales) |
(%) |
4.03 |
3.79 |
5.27 |
|
|
|
|
|
|
|
Operating Profit Margin (PBIDT/Sales) |
(%) |
17.46 |
18.34 |
18.84 |
|
|
|
|
|
|
|
Return on Total Assets (PBT/Total Assets} |
(%) |
3.50 |
3.44 |
5.37 |
|
|
|
|
|
|
|
Return on Investment (ROI) (PBT/Networth) |
|
0.09 |
0.08 |
0.13 |
|
|
|
|
|
|
|
Debt Equity Ratio (Total Debt /Networth) |
|
0.90 |
0.87 |
0.89 |
|
|
|
|
|
|
|
Current Ratio (Current Asset/Current Liability) |
|
1.18 |
1.10 |
1.10 |
STOCK
PRICES
|
Face Value |
Rs. 1.00/- |
|
Market Value |
Rs. 52.15/- |
FINANCIAL ANALYSIS
[all figures are
in Rupees Million]
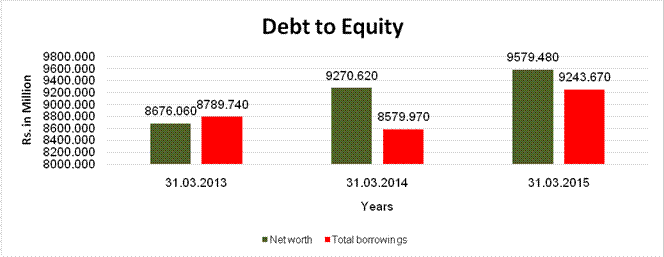
DEBT EQUITY RATIO
|
Particular |
31.03.2013 |
31.03.2014 |
31.03.2015 |
|
|
Rs.
In Million |
Rs. In
Million |
Rs.
In Million |
|
Share Capital |
224.260 |
224.260 |
224.260 |
|
Reserves & Surplus |
8451.800 |
9046.360 |
9355.220 |
|
Net
worth |
8676.060 |
9270.620 |
9579.480 |
|
|
|
|
|
|
long-term borrowings |
2609.610 |
2233.380 |
2655.290 |
|
Short term borrowings |
5091.110 |
5798.420 |
5952.980 |
|
Current Maturities of Long term debt |
1089.020 |
548.170 |
635.400 |
|
Total
borrowings |
8789.740 |
8579.970 |
9243.670 |
|
Debt/Equity
ratio |
1.013 |
0.926 |
0.965 |
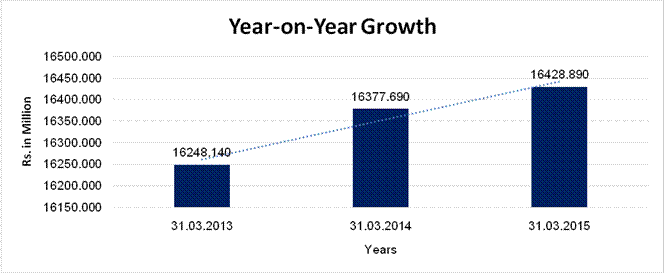
YEAR-ON-YEAR GROWTH
|
Year
on Year Growth |
31.03.2013 |
31.03.2014 |
31.03.2015 |
|
|
Rs.
In Million |
Rs.
In Million |
Rs.
In Million |
|
Sales |
16248.140 |
16377.690 |
16428.890 |
|
|
|
0.797 |
0.313 |
NET PROFIT MARGIN
|
Net
Profit Margin |
31.03.2013 |
31.03.2014 |
31.03.2015 |
|
|
Rs.
In Million |
Rs.
In Million |
Rs.
In Million |
|
Sales
|
16248.140 |
16377.690 |
16428.890 |
|
Profit |
856.660 |
620.800 |
662.640 |
|
|
5.27% |
3.79% |
4.03% |
LOCAL AGENCY FURTHER INFORMATION
|
Sr. No. |
Check list by
info agents |
Available in
Report (Yes/No) |
|
1 |
Year of establishment |
Yes |
|
2 |
Constitution of the entity -Incorporation
details |
Yes |
|
3 |
Locality of the entity |
Yes |
|
4 |
Premises details |
No |
|
5 |
Buyer visit details |
-- |
|
6 |
Contact numbers |
Yes |
|
7 |
Name of the person contacted |
Yes |
|
8 |
Designation of contact person |
Yes |
|
9 |
Promoter’s background |
Yes |
|
10 |
Date of Birth of Proprietor / Partners /
Directors |
No |
|
11 |
Pan Card No. of Proprietor / Partners |
No |
|
12 |
Voter Id Card No. of Proprietor / Partners |
No |
|
13 |
Type of business |
Yes |
|
14 |
Line of Business |
Yes |
|
15 |
Export/import details (if applicable) |
No |
|
16 |
No. of employees |
No |
|
17 |
Details of sister concerns |
Yes |
|
18 |
Major suppliers |
No |
|
19 |
Major customers |
No |
|
20 |
Banking Details |
Yes |
|
21 |
Banking facility details |
Yes |
|
22 |
Conduct of the banking account |
-- |
|
23 |
Financials, if provided |
Yes |
|
24 |
Capital in the business |
Yes |
|
25 |
Last accounts filed at ROC, if applicable |
Yes |
|
26 |
Turnover of firm for last three years |
Yes |
|
27 |
Reasons for variation <> 20% |
-- |
|
28 |
Estimation for coming financial year |
No |
|
29 |
Profitability for last three years |
Yes |
|
30 |
Major shareholders, if available |
Yes |
|
31 |
External Agency Rating, if available |
Yes |
|
32 |
Litigations that the firm/promoter
involved in |
Yes |
|
33 |
Market information |
-- |
|
34 |
Payments terms |
No |
|
35 |
Negative Reporting by Auditors in the
Annual Report |
No |
LITIGATION
DETAILS:
HIGH
COURT OF PUNJAB AND HARYANA
|
CASE DETAILS FOR CASE
CA-106-2012 |
|||
|
Diary Number |
791874 |
District |
OTHER-DISTRICT |
|
Category |
- |
Main Case Detail |
CP-25-2012 |
|
Party Detail |
M/S HETERO DRUGS LIMITED V/S M/S NECTAR LIFESCIENCES LIMITED |
||
|
Advocate Name |
ANAND CHHIBBAR |
List Type |
|
|
Status |
PENDING |
Next date |
|
|
|
|||
|
Case Listing Details |
|||
|
No Listing Data Available |
|||
|
Judgment Details For Case: CA-106-2012 |
|||
|
Order Date |
Order Type |
Bench |
Judgment Link |
|
No Judgment/Order Found. |
|||
GLOBAL PHARMACEUTICAL
INDUSTRY:
Industry Outlook Global spending on medicines is expected to
meet $1.17 trillion by 2017, according to a report by Pharmaceutical market
research firm, IMS Healthcare Informatics. Spending during this period will
likely be marked by greater cost pressures and a higher bar for product
innovation that reflects an increased demand for value from both regulators and
payers. The next 5 years also sees a continuing growth split between developed
and Pharmerging markets. IMS forecasts that the developed markets of North
America, Europe and Japan will see modest single-digit spending during the next
five years due to a combination of economic and healthcare austerity measures
and savings realized from the growing availability of low cost generic versions
of brands following patent expiry, and also on account of greatest impact from
implementation of the Affordable Care Act in the USA. The highpotential
“pharmerging” markets offer tremendous opportunities for pharmaceutical
manufacturers that face mounting pressures in the mature markets. As more
pharma companies target these new growth markets, they also face the challenge
of an increasingly complex, demanding and fast changing logistics and
regulatory environment. Traditional approaches to manufacturing and distribution
are being challenged by shorter market cycles, outsourced production, and
increasing government regulation. Globalization continues to have a profound
impact on the pharma supply chain. As a growing amount of R&D and
manufacturing migrates to Asia, the industry has to manage resources that are
much more widely dispersed and to cope with greater potential for pandemics.
Innovation, the ultimate engine of growth for the global provision of
medicines, will see revival of activity through 2017, with increases in the
number of global innovative launches since 2010.
PHARMERGING MARKETS:
Innovation and technology will be important differentiators
as pharmaceutical companies try to drive growth in emerging markets that face rapid
growth of various chronic diseases like diabetes, hypertension, heart disease,
and cancer. Emerging markets are predicted to account for a third of global
pharmaceutical spend by the end of next year and seen as critical for the
sustained growth of leading pharma companies. In India, for example, the
prevalence of diabetes and cancer is projected to rise by 25-40 per cent over
the next 10 years," the IMS report said. This shift gives pharma companies
an opportunity to market their global products in emerging markets, backed by
tested 'go-to-market' strategies and operating models.
Key barriers which need to be addressed in many emerging markets are the affordability and accessibility of medicines. Improvements in affordability will be driven by rising disposable incomes and increasing insurance coverage. Growth in accessibility will come from increases in government spending and medical infrastructure, and new business models for rural areas. The acceptability of medicines is also expected to rise, as a result of the growth in chronic conditions and the resulting increase in the self-administration of drugs. Pharmaceutical markets in developed countries may still be quite sizable, but past few years have been quite sluggish in terms of growth. Contrary to developed markets, the emerging economies have seen a significant amount of growth.
INDIAN PHARMACEUTICAL
INDUSTRY:
The Indian pharmaceutical industry which is presently
thriving at USD 16 Billion is set to reach approximately USD 55 Billion by 2020
in terms of business value.
- Presently Indian
pharmaceutical industry ranks 3 in the the world in terms of volume and 13
in the world in terms of business value.
- India is expected to be
the third-largest global generic active pharmaceutical
ingredient (API) merchant market by 2020, with
a 7.2 per cent market share.
- Indian pharmaceutical
industry bags about 72% of revenue from generic version of drugs.
- In terms of value, exports
of pharmaceutical products from India increased at a CAGR of 25 percent to
US$ 14.1 Billion during FY06–15.
- The country's
pharmaceuticals industry accounts for about 2.4% of the global Pharma
industry by value and 10% by volume.
- Industry revenues are
expected to expand at a CAGR of 12.1% during 2012-20 and reach USD 45
Billion.
- The healthcare sector in
India is expected to grow to USD 250 Billion by 2020 from USD 65 Billion
currently.
- The generics market is
expected to grow to USD 28.1 Billion by 2018 from USD 11.3 Billion in
2011.
ABOUT NLL:
Nectar Lifesciences Limited ('NLL' or 'NECLIFE') is an
integrated pharmaceutical organization incorporated in 1995 based in
Chandigarh, Punjab India. NLL has developed fully integrated sustainable
production systems to manufacture high quality Cephalosporin intermediates,
–APIs and Formulations to meet the diverse requirements of its customer base in
India and over 50 countries worldwide.
NECLIFE: A NICHE
PHARMACEUTICAL PLAYER:
Pharmaceuticals –APIs
With Global capacities of some of the Cephalosporin molecules
and strong tie ups Nectar has been experiencing a robust growth in this
segment. With an expertise in R&D, Nectar is also working on new generation
Cephalosporin molecules to be a globally dedicated Cephalosporin player.
Overall, API's witnessed an increase of 39% taking the turnover from Rs.
9966.000 Millions to Rs. 13851.000 Millions Our API facility has global
regulatory accreditations like USFDA, EUGMP INFARMED, KFDA, PMDA, MCC, ANVISA
Brazil and others. As a step up a strategic decision was made to optimize the
international sales wherein NLL registered a robust growth of 34% was observed
over the last year. Domestic sales also witnessed an increase by 42% as
compared to last fiscal. Neclife with robust capacity over a year certainly
leads Cephalosporins' in India & ROW markets.
Pharmaceuticals –
Finished Dosage formulations
Finished dosage Formulations business is marked by a long
gestation period in terms of registrations and its approval by various Health
Authorities across the world. Significant numbers of filings have been done
across the world in various geographies. The FDF facility in Baddi also has
been recognized & approved by regulatory bodies like INFARMED Portugal for
European Union, ANVISA Brazil, OGYI Hungary for European Union, Pharmacy &
Poison Board Kenya, NMRC Namibia, NDA Uganda, TFDA and others.
The Facility has already been inspected by USFDA and written
approval of the same is expected soon.
- NLL's formulations
business witnessed a growth of 73% in FY15 as compared to
the last year.
- ROW-FDF business grew by
more than 80% in FY15 as compared to the last fiscal, boosted by exports
to emerging markets.
Total Pharmaceutical
Business in FY 14–15
The pharmaceutical business of NLL registered a growth of
41% in FY 14–15 with a total turnover of Rs.14710.000 Cr. as compared to
previous year's Rs. 10460.000 Cr. As compared to FY-13, NLL pharma business
registered CAGR of 49%, fuelled by growth in API business in exports as well as
domestics and FDF business to emerging markets.
TOTAL PHARMACEUTICAL
BUSINESS IN FY 14–15:
The pharmaceutical business of NLL registered a growth of 41% in FY 14–15 with a total turnover of Rs.14710.000 Million as compared to previous year's Rs. 10460.000 Million. As compared to FY-13, NLL pharma business registered CAGR of 49%, fuelled by growth in API business in exports as well as domestics and FDF business to emerging markets.
Phytochemicals
(Menthol):
NLL entered the Menthol Business in 2006 and as in the past
concentrated on value added pharmaceutical menthol business and succeeded in
both domestic and international markets with good margins. Menthol business
overall sales is Rs. 1994.000 Million in financial year 2014-15. Menthol's
performance during FY 2014-15 witnessed a roller coaster ride which was
consciously attributed to a strategic shift in business option. This de-growth
in menthol was primarily owing to the following reasons:
- Company's decision to
cater to high value Pharmaceutical products.
- High Market fluctuations
led to lower natural menthol demands.
Other Non-API
businesses:
- NLL also has high quality
facilities for its other Non-API businesses like Empty Hard Gelatin
Capsules, Generics & Diagnostics.
- EHGC– Being the one of the
largest manufacturer, the EHGC facility in Baddi has a capacity to produce
approximately 4 Billion empty capsules per annum. The EHGC Facility has
approvals from ISO 9001:2008, KOSHER, HALAL, HACCAP & C-14.
- Generics India Business–
the Company has more than 300 Stock Keeping Units (SKUs) in India with Pan
India presence & about 1 Lac retailers are covered. This section is
growing at the rate of 36% YOY.
- Diagnostics: The business
unit is known as Necpath, manufacturing various Diagnostics like:
§ Rapid
tests
§ Pregnancy
cards
§ HIV/
Malaria/Bio reagents
§ In-Vitro
Diagnostic equipments etc.
COMPANY'S
PERFORMANCE:
Overall, Active Pharmaceutical Ingredients (APIs) witnessed
a tremendous increase in business. However, compared with steep pharmaceuticals
growth curve, Phytochemicals (Menthol) performance during FY 2014-15 witnessed
a roller coaster ride which was consciously attributed to a strategic shift in
business option. This de-growth in menthol was primarily owing to the following
reasons:
- Company's decision to
cater to high value Pharmaceutical products.
- High Market fluctuations
led to lower natural menthol demands.
Despite the, above, the Gross revenues increased to Rs. 17463.860 Million, a growth of around 1.51% against Rs. 17203.550 Million in the previous year. Profit before depreciation and taxation was Rs. 2869.060 Million against Rs. 3003.960 Million in the previous year. The Profit before Tax increased to Rs. 847.540 Million, a growth of around 8.35% against Rs. 782.230 Million in the previous year, due to effective cost management. The Profit after Tax increased to Rs. 662.640 Million, a growth of around 6.74% against Rs. 620.800 Million in the previous year. The financial results of the company for the quarter ended on June 30, 2015 are available on the website of the company (URL: www.neclife.com).
OPPORTUNITIES &
OUTLOOK- NLL'S PATH AHEAD:
The company expects a positive outlook for the next year.
NLL is expecting to continue its strategic entry in US and EU market with more
filings in its Cephalosporin's range during 2015-16. The U.S. is the single
largest generics market. Regardless of the intense competition & pricing
pressure the US segment will continue to be the single largest market, reaching
between $350 Billion to $380 Billion. Europe forms the world's 2nd largest
generic market, backed by Govt. reforms to curb healthcare cost and increased
demand from ageing population.
Japanese generic market: With a view to reduce burgeoning
healthcare costs and tackling rising cases of life-threatening diseases, the
Japanese government is taking various measures to increase the adoption of
generic drugs among people in the country. The Japanese generic drugs market,
evolving to become the world's next generic hub, offers a wide range of
opportunities to both domestic and international players. The patent expiry of
a large number of branded drugs and active pharmaceutical ingredients are the
major attractions for companies willing to enter the market. It is one of the
most lucrative destinations for pharmaceutical player mainly because of the
rapid ageing population & increasing incidence of western lifestyle
diseases. NLL expects future value growth from this market for Cephalosporin
products along with other regulated markets such as US and EU.
NLL also expects continued momentum from its domestic and
export markets. Next few years will see NLL's ability to discover new markets
& new opportunities gaining an invaluable advantage over competitors.
Future Outlook:
v
Enhance accessibility in the regulated generics
business.
v
Consolidate its presence in the API business by
focusing on regulated markets & pharmerging markets.
v
Expand relationships with marque clients with
additional new products.
v
Enhance market penetration with existing
products.
v
Focus on long-term partnerships with major
pharma companies.
v
Increase its operations in emerging markets.
v
Focus on Cephalosporin's R & D and work on
new cephalosporin molecules.
To unlock the true potential of huge investments made by NLL
in Fixed Assets and Intellectual Property, NLL is at an inflection point of
making strategic entry into highly regulated markets.
UNSECURED LOAN:
|
Particulars |
31.03.2015 Rs.
In Million |
31.03.2014 Rs.
In Million |
|
Long-term
Borrowings |
|
|
|
From Banks |
3.760 |
1.640 |
|
From Banks |
7.940 |
5.860 |
|
Total |
11.700 |
7.500 |
CONTINGENT
LIABILITIES:
(Rs. in millions)
|
PARTICULARS |
31.03.2015 |
|
Claims Not
Acknowledged By Debt |
|
|
Income Tax Matters |
5.970 |
|
Excise Matters |
112.950 |
|
Service Tax Matters |
16.220 |
|
Bank Guarantees |
11.750 |
|
Bill Discounted |
80.700 |
|
Letter Of Credit |
249.750 |
|
Other Money Which Company Is Contingency Liable |
19.110 |
|
Estimated Amounts Of Contract Remaingin To Be Executed On Capital Account And Not Provided For (Net Of Advance) |
7.800 |
|
|
504.250 |
STANDALONE
UNAUDITED FINANCIAL RESULTS FOR THE QUARTER ENDED 30TH JUNE 2015
(Rs. In Million)
|
Particulars |
3 Months Ended |
|
|
30.06.2015 |
|
|
(UNAUDITED) |
|
1.
Income from operations |
|
|
a) Net sales/ Income from operation (net of excise duty) |
40768.68 |
|
b) Other operating income |
1.52 |
|
Total
income from Operations(net) |
40770.20 |
|
2.Expenditure |
|
|
a) Cost of material consumed |
26469.33 |
|
b) Purchases of stock in trade |
854.58 |
|
c) Changes in inventories of finished goods,
work-in-progress and stock-in-trade |
290.36 |
|
d) Employees benefit expenses |
1686.87 |
|
e) Depreciation and amortization expenses |
2178.71 |
|
g) Other expenditure |
4413.68 |
|
Total expenses |
35893.53 |
|
3. Profit from operations before other income and
financial costs |
4876.67 |
|
4. Other income |
126.71 |
|
5. Profit from ordinary activities before finance costs |
5003.38 |
|
6. Finance costs |
2733.24 |
|
7. Net profit/(loss) from ordinary activities
after finance costs but before exceptional items |
2270.14 |
|
8. Exchange Currency Fluctuation Loss/ (Gain) |
0.00 |
|
9. Profit from ordinary activities before tax
Expense: |
2270.14 |
|
10.Tax expenses |
528.80 |
|
11.Net Profit / (Loss) from
ordinary activities after tax (9-10) |
1741.34 |
|
12.Extraordinary Items (net of tax expense) |
0.00 |
|
13.Net Profit / (Loss) for the period (11 -12) |
1741.34 |
|
14.Paid-up equity share capital (Nominal value Rs.10/- per share) |
2,242.61 |
|
15. Reserve excluding Revaluation Reserves as per
balance sheet of previous accounting year |
-- |
|
16.i) Earnings per share (before extraordinary
items) of Rs.10/- each) (not annualised): |
|
|
(a) Basic and diluted |
0.78 |
|
|
|
|
A. Particulars of shareholding |
|
|
1. Public Shareholding |
|
|
- Number of shares |
124,792,970 |
|
- Percentage of shareholding |
55.65 |
|
2. Promoters and Promoters group Shareholding- |
|
|
a) Pledged /Encumbered |
|
|
Number of shares |
55,555,600 |
|
Percentage of shares (as a % of total shareholding of the
promoter and promoter group) |
55.85 |
|
Percentage of shares (as a % of total share capital of the
company) |
24.77 |
|
|
|
|
b) Non Encumbered |
|
|
Number of shares |
43,912,400 |
|
Percentage of shares (as a % of total shareholding of the
promoter and promoter group) |
44.15 |
|
Percentage of shares (as a % of total share capital of the
company) |
19.58 |
|
|
|
|
B.
Investor Complaints |
|
|
Pending at the beginning of the quarter |
- |
|
Receiving during the quarter |
3 |
|
Disposed of during the quarter |
3 |
|
Remaining unreserved at the end of the quarter |
-- |
Notes:
- The above financial
results were reviewed by Audit Committee on 13.08.2015 and approved by the
Board in its meeting held on 14.08.2015 and further limited reviewed by the
Statutory Auditors of the Company.
- Previous year figures
have been regrouped and reclassified wherever necessary to make them
comparable with current period.
- The above financial
results are on stand alone basis.
- The company is
exclusively in the pharmaceutical business segment.
INDEX OF CHARGE:
|
Sr. No. |
Charge ID |
Date of Charge
Creation/Modification |
Charge amount
secured |
Charge Holder |
Address |
Service Request
Number (SRN) |
|
1 |
10589665 |
15/07/2015 |
2,600,000.00 |
HDFC BANK LIMITED |
HDFC BANK HOUSE, SENAPATI BAPAT MARG,
LOWER PAREL |
C63532527 |
|
2 |
10582045 |
09/07/2015 |
1,164,000.00 |
ICICI BANK LIMITED |
SCO 129-130, SECTOR 9-C, CHANDIGARH,
CHANDIGARH - |
C59865683 |
|
3 |
10578142 |
15/05/2015 |
900,000.00 |
HDFC BANK LIMITED |
HDFC BANK HOUSE, SENAPATI BAPAT MARG,
LOWER PAREL |
C58030784 |
|
4 |
10562363 |
18/04/2015 |
800,000,000.00 |
IFCI LIMITED |
IFCI LTD, IFCI TOWER, 61, NEHRU PLACE, NEW
DELHI, |
C50432814 |
|
5 |
10563385 |
27/03/2015 |
650,000.00 |
HDFC BANK LIMITED |
HDFC BANK HOUSE, SENAPATI BAPAT MARG,
LOWER PAREL |
C50863398 |
|
6 |
10555532 |
05/03/2015 |
150,000,000.00 |
The Saraswat Co-operative Bank Limited |
20-A, LAJPAT NAGAR - IV, RING ROAD, NEW
DELHI, DELHI - 110024, INDIA |
C46934600 |
|
7 |
10555534 |
05/03/2015 |
250,000,000.00 |
The Saraswat Co-operative Bank Limited |
20-A, LAJPAT NAGAR - IV, RING ROAD, NEW
DELHI, DELHI - 110024, INDIA |
C46935482 |
|
8 |
10550516 |
02/02/2015 |
470,000.00 |
HDFC BANK LIMITED |
HDFC BANK HOUSESENAPATI BAPAT MARG, LOWER
PAREL W |
C44303469 |
|
9 |
10537006 |
26/11/2014 |
500,000.00 |
HDFC BANK LIMITED |
HDFC BANK HOUSESENAPATI BAPAT MARG, LOWER
PAREL W |
C37160702 |
|
10 |
10537007 |
26/11/2014 |
500,000.00 |
HDFC BANK LIMITED |
HDFC BANK HOUSESENAPATI BAPAT MARG, LOWER
PAREL W |
C37161080 |
*Date of modification Charges
FIXED ASSETS:
·
Land
·
Building
·
Plant and Machinery
·
Computer
·
Vehicles
·
Furniture and Fixture
CMT REPORT (Corruption, Money Laundering & Terrorism]
The Public Notice information has been collected from various sources
including but not limited to: The Courts,
1] INFORMATION ON
DESIGNATED PARTY
No exist designating subject or any of its
beneficial owners, controlling shareholders or senior officers as terrorist or
terrorist organization or whom notice had been received that all financial
transactions involving their assets have been blocked or convicted, found
guilty or against whom a judgement or order had been entered in a proceedings
for violating money-laundering, anti-corruption or bribery or international
economic or anti-terrorism sanction laws or whose assets were seized, blocked,
frozen or ordered forfeited for violation of money laundering or international
anti-terrorism laws.
2] Court Declaration :
No records exist to suggest that subject is
or was the subject of any formal or informal allegations, prosecutions or other
official proceeding for making any prohibited payments or other improper
payments to government officials for engaging in prohibited transactions or
with designated parties.
3] Asset Declaration :
No records exist to suggest that the property
or assets of the subject are derived from criminal conduct or a prohibited
transaction.
4] Record on Financial
Crime :
Charges or conviction
registered against subject: None
5] Records on Violation of
Anti-Corruption Laws :
Charges or investigation
registered against subject: None
6] Records on Int’l
Anti-Money Laundering Laws/Standards :
Charges or
investigation registered against subject: None
7] Criminal Records
No
available information exist that suggest that subject or any of its principals
have been formally charged or convicted by a competent governmental authority
for any financial crime or under any formal investigation by a competent
government authority for any violation of anti-corruption laws or international
anti-money laundering laws or standard.
8] Affiliation with
Government :
No
record exists to suggest that any director or indirect owners, controlling
shareholders, director, officer or employee of the company is a government
official or a family member or close business associate of a Government
official.
9] Compensation Package :
Our
market survey revealed that the amount of compensation sought by the subject is
fair and reasonable and comparable to compensation paid to others for similar
services.
10] Press Report :
No press reports / filings exists on
the subject.
CORPORATE GOVERNANCE
MIRA INFORM as part of its Due Diligence do provide comments on
Corporate Governance to identify management and governance. These factors often
have been predictive and in some cases have created vulnerabilities to credit
deterioration.
Our Governance Assessment focuses principally on the interactions
between a company’s management, its Board of Directors, Shareholders and other
financial stakeholders.
CONTRAVENTION
Subject is not known to have contravened any existing local laws,
regulations or policies that prohibit, restrict or otherwise affect the terms
and conditions that could be included in the agreement with the subject.
FOREIGN EXCHANGE RATES
|
Currency |
Unit
|
Indian Rupees |
|
US Dollar |
1 |
Rs. 66.14 |
|
|
1 |
Rs. 100.55 |
|
Euro |
1 |
Rs. 71.26 |
INFORMATION DETAILS
|
Information
Gathered by : |
SUP |
|
|
|
|
Analysis Done by
: |
KRN |
|
|
|
|
Report Prepared
by : |
SYL |
SCORE & RATING EXPLANATIONS
|
SCORE FACTORS |
RANGE |
POINTS |
|
HISTORY |
1~10 |
7 |
|
PAID-UP CAPITAL |
1~10 |
6 |
|
OPERATING SCALE |
1~10 |
7 |
|
FINANCIAL
CONDITION |
|
|
|
--BUSINESS SCALE |
1~10 |
7 |
|
--PROFITABILITY |
1~10 |
6 |
|
--LIQUIDITY |
1~10 |
7 |
|
--LEVERAGE |
1~10 |
7 |
|
--RESERVES |
1~10 |
7 |
|
--CREDIT LINES |
1~10 |
6 |
|
--MARGINS |
-5~5 |
-- |
|
DEMERIT POINTS |
|
|
|
--BANK CHARGES |
YES/NO |
YES |
|
--LITIGATION |
YES/NO |
YES |
|
--OTHER ADVERSE INFORMATION |
YES/NO |
NO |
|
MERIT POINTS |
|
|
|
--SOLE DISTRIBUTORSHIP |
YES/NO |
NO |
|
--EXPORT ACTIVITIES |
YES/NO |
NO |
|
--AFFILIATION |
YES/NO |
YES |
|
--LISTED |
YES/NO |
YES |
|
--OTHER MERIT FACTORS |
YES/NO |
YES |
|
DEFAULTER
|
|
|
|
--RBI |
YES/NO |
NO |
|
--EPF |
YES/NO |
NO |
|
|
|
|
|
TOTAL |
|
60 |
This score serves as
a reference to assess SC’s credit risk and to set the amount of credit to be
extended. It is calculated from a composite of weighted scores obtained from each
of the major sections of this report. The assessed factors and their relative
weights (as indicated through %) are as follows:
Financial
condition (40%) Ownership
background (20%) Payment
record (10%)
Credit history
(10%) Market trend (10%) Operational size
(10%)
RATING EXPLANATIONS
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
>86 |
Aaa |
Possesses an extremely sound financial base with the strongest
capability for timely payment of interest and principal sums |
Unlimited |
|
71-85 |
Aa |
Possesses adequate working capital. No caution needed for credit
transaction. It has above average (strong) capability for payment of interest
and principal sums |
Large |
|
56-70 |
A |
Financial & operational base are regarded healthy. General
unfavourable factors will not cause fatal effect. Satisfactory capability for
payment of interest and principal sums |
Fairly Large |
|
41-55 |
Ba |
Overall operation is considered normal. Capable to meet normal
commitments. |
Satisfactory |
|
26-40 |
B |
Capability to overcome financial difficulties seems comparatively
below average. |
Small |
|
11-25 |
Ca |
Adverse factors are apparent. Repayment of interest and principal sums
in default or expected to be in default upon maturity |
Limited with
full security |
|
<10 |
C |
Absolute credit risk exists. Caution needed to be exercised |
Credit not
recommended |
|
NB |
NEW BUSINESS |
||
This report is issued at your request without any
risk and responsibility on the part of MIRA INFORM PRIVATE LIMITED (MIPL)
or its officials.