MIRA INFORM REPORT
|
Report No. : |
343672 |
|
Report Date : |
06.10.2015 |
IDENTIFICATION DETAILS
|
Name : |
DR. REDDY’S LABORATORIES LIMITED |
|
|
|
|
Registered
Office : |
8-2-337, Road No. 3, Banjara Hills, Hyderabad - 500034, Telangana |
|
Tel. No.: |
91-40-49002900 |
|
|
|
|
Country : |
India |
|
|
|
|
Financials (as
on) : |
31.03.2015 |
|
|
|
|
Date of
Incorporation : |
24.02.1984 |
|
|
|
|
Com. Reg. No.: |
36-004507 |
|
|
|
|
Capital
Investment / Paid-up Capital : |
Rs. 852.000 Million |
|
|
|
|
CIN No.: [Company Identification
No.] |
L85195TG1984PLC004507 |
|
|
|
|
IEC No.: |
Not Available |
|
|
|
|
TAN No.: [Tax Deduction &
Collection Account No.] |
HYDD00080D |
|
|
|
|
PAN No.: [Permanent Account No.] |
Not Available |
|
|
|
|
Legal Form : |
A Public Limited Liability Company. The Company’s Shares are Listed on
the Stock Exchanges. |
|
|
|
|
Line of Business
: |
Subject operates
as an integrated pharmaceutical company. It operates in three segments: Global Generics, Pharmaceutical Services and Active
Ingredients (PSAI), and Proprietary Products: ·
The Global
Generics segment produces and markets finished pharmaceutical products as
branded formulations or generic finished dosages. This segment also engages
in biologics business. ·
The PSAI
segment develops active pharmaceutical ingredients (APIs) and intermediaries,
which are used as principal ingredients for finished pharmaceutical products.
This segment also offers contract research services to other companies; and
manufactures and sells APIs and steroids in accordance with the specific
customer requirements. · The Proprietary Products segment focuses on the discovery, development, and commercialization of novel small molecule agents in therapeutic areas, such as anti-infective, metabolic disorders, and pain and inflammation. It also provides differentiated formulations for unmet medical needs; and a portfolio of in-licensed patented dermatology products |
|
|
|
|
No. of Employees
: |
19857 (Approximately) |
RATING & COMMENTS
|
MIRA’s Rating : |
Aa (81) |
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
71-85 |
Aa |
Possesses adequate working capital. No caution needed for credit transaction.
It has above average (strong) capability for payment of interest and
principal sums |
Large |
|
Maximum Credit Limit : |
USD 303892000 |
|
|
|
|
Status : |
Excellent |
|
|
|
|
Payment Behavior : |
Regular |
|
|
|
|
Litigation : |
Clear |
|
|
|
|
Comments : |
Subject is an old, well established and a reputed company incorporated in the year 1984 having excellent track record. It is engaged in manufacturing and marketing of pharmaceuticals. The company manufacturers wide range of pharmaceuticals products in India and Overseas. Financial position of the company is sound. Fundamentals are strong and healthy. Directors are reported to be experienced, respectable and resourceful businessmen. Share price are quoted high on exchange (face value Rs.5 with share
price Rs.4230) Trade relations are fair. Business is active. Payments are reported to be regular and as per commitments. The company can be considered good for normal business
dealings under usual trade terms and conditions. |
NOTES :
Any query related to this report can be made
on e-mail : infodept@mirainform.com
while quoting report number, name and date.
ECGC Country Risk Classification List – March 31, 2015
|
Country Name |
Previous Rating (31.12.2014) |
Current Rating (31.03.2015) |
|
India |
A1 |
A1 |
|
Risk Category |
ECGC
Classification |
|
Insignificant |
A1 |
|
Low |
A2 |
|
Moderate |
B1 |
|
High |
B2 |
|
Very High |
C1 |
|
Restricted |
C2 |
|
Off-credit |
D |
EXTERNAL AGENCY RATING
NOT AVAILABLE
RBI DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available RBI Defaulters’ list.
EPF (Employee Provident Fund) DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available EPF (Employee Provident Fund) Defaulters’ list as of
31-03-2015.
LOCATIONS
|
Registered Office/ Corporate
Office : |
8-2-337, Road No. 3, Banjara Hills, Hyderabad - 500034, Telangana,
India |
|
Tel. No.: |
91-40-49002900 |
|
Fax No.: |
91-40-49002999 |
|
E-Mail : |
ssrinivasan@drreddys.com |
|
Website : |
|
|
|
|
|
API
Manufacturing Facilities : |
Plot No. 137, 138, IDA Bollaram, Jinnaram Mandal Medak District -
502325, Telangana, India
Plot No. 75B, 105, 110 and 111, IDA Bollaram, Jinnaram Mandal, Medak
District - 502325, Telangana, India
Plot No. 116, 116A and 126C and SY No. 157, IDA Bollaram, Jinnaram
Mandal, Medak District - 502325, Telangana, India
Plot No. 9/A, 9/B, 22A, 22B and 22C, Phase – III, IDA Jeedimetla,
Ranga Reddy District – 502325, Telangana, India
Peddadevulapally, Tripuraram Mandal, Nalgonda District – 502325, Telangana, India
IDA, Pydibheemavaram Ransthal Mandal, Srikakulam, District - 502325, Andhra
Pradesh,
India
Sector No. 28 and 34 Devunipalavalasa
Village, Ranastalam Mandal, Srikakulam District - 502325, Andhra Pradesh,
India |
|
|
|
|
Formulation
Manufacturing Facilities : |
Plot No. 146, IDA Bollaram, Jinnaram Mandal, Medak District – 502325, Telangana, India
S Y No. 42, 45, 46 and 54 Bachupally, Qutubullapur Mandal, Ranga Reddy
District – 502325, Telangana, India
S Y No. 41 Bachupally, Qutubullapur Mandal, Ranga Reddy – 502325, Telangana, India
Ward-F, Block-4, Adavipolam Yanam, Pondicherry – 533464, India
Khol, Nalagarh Solan, Nalagarh Road, Baddi – 173205, Himachal Pradesh,
India
Plot No P1-P9, Phase III Duvvada, VSEZ, Visakapatanam – 530046, Andhra
Pradesh, India
Village Mauja Thana, Nalagarh Baddi Road, Baddi, District Solan –
173205, Himachal Pradesh, India
Sector No. 9-13 and 17-20 Devunipalavalasa Village, Ranastalam Mandal,
Srikakulam District – 532409, Andhra Pradesh, India
Survey No.47, Bachupally
Quthbullapur Mandal, Ranga Reddy District – 500043, Telangana, India |
|
|
|
|
Research and Development
Facility : |
Bachupally Village, Qutubullapur Mandal, Ranga Reddy District -
500123, Andhra
Pradesh,
India
Bollaram Road, Miyapur Hyderabad - 500049, Telangana, India
Bollaram Road, Miyapur Hyderabad - 500049, Telangana, India
Plot 31A, IDA, Jeedimetla Hyderabad -
500050, Telangana, India |
|
|
|
|
Facility
Locations Outside India : |
No. 258, Huang Pu Jiang (M) Road, Kunshan Development Zone, Jiangsu
Province P. R. China 215300
Carretera Federal Cuernavaca-Cuautla KM 4.5 CIVAC, Jiutepec, Morelos
Mexico 62578
6, Riverview Road, Beverly, East Yorkshire HU 17 OLD, United Kingdom
Steanard Lane, Mirfield, West Yorkshire, WF 14, 8HZ, United Kingdom
410 Cambridge Science Park Milton Road, Cambridge CB4 0PE, United
Kingdom
8800 Line Avenue, Shreveport, Louisiana 71106. USA
P.O. Box 9002, 201 Industrial Drive Bristol, TN 37621- 9002, USA
1974 Route 145, Middleburgh, New York 12122, USA
Zernikedreef 12, 2333 CL Leiden, The
Netherlands |
DIRECTORS
AS ON 31.03.2015
|
Name : |
Mr. Omkar Goswami |
|
Designation : |
Independent Director |
|
Address : |
E-121, Masjid Moth, First Floor, Greater Kailash-III, New Delhi - 110048, India |
|
Qualification: |
D.Phil. (Ph.D.) Economics |
|
Date of Appointment : |
31.07.2014 |
|
DIN No.: |
00004258 |
|
|
|
|
Name : |
Dr. Ashok Sekhar Ganguly |
|
Designation : |
Director |
|
Address : |
N6 Pemino, Altamount Road, Mumbai - 400026, Maharashtra, India |
|
Date of Appointment : |
31.07.2014 |
|
DIN No.: |
00010812 |
|
|
|
|
Name : |
Ms. Kalpana Jaisingh Morparia |
|
Designation : |
Director |
|
Address : |
B-92, Ocean Gold CHS, Twin Tower Lane, Prabhadevi, Mumbai - 400025, Maharashtra, India |
|
Qualification: |
Science and Law |
|
Date of Appointment : |
31.07.2014 |
|
DIN No.: |
00046081 |
|
|
|
|
Name : |
Mr. Gunupati Venkateswara Prasad |
|
Designation : |
Managing Director |
|
Address : |
Plot
No.32, Road No.8, Banjara Hills, Hyderabad - 500034, Telangana,
India |
|
Date of Birth/Age : |
54 Years |
|
Qualification: |
B.Sc. (Chem. |
|
Date of Appointment : |
13.05.2014 |
|
DIN No.: |
00057433 |
|
|
|
|
Name : |
Mr. Satish Kallam Reddy |
|
Designation : |
Whole-Time Director |
|
Address : |
8-2-576/1, Road No. 7, Banjara Hills, Hyderabad - 500034, Telangana, India |
|
Date of Birth/Age : |
47 Years |
|
Qualification
: |
B. Tech., M.S. (Medicinal Chemistry) |
|
Date of Appointment : |
13.05.2014 |
|
DIN No.: |
00129701 |
|
|
|
|
Name : |
Mr. Ravi Bhoothalingam |
|
Designation : |
Independent Director |
|
Address : |
308 B, The Laburnum, Block A, Shushant Lok, Gurgaon - 122002, Haryana, India |
|
Qualification
: |
Master’s degree in Experimental Psychology |
|
Date of Appointment : |
31.07.2014 |
|
DIN No.: |
00194530 |
|
|
|
|
Name : |
Mr. Anupam Pradip Puri |
|
Designation : |
Director |
|
Address : |
17 East 16th Street, Ny, New York, Ny - 10003, United States of America |
|
Qualification
: |
B.A. in Economics |
|
Date of Appointment : |
31.07.2014 |
|
DIN No.: |
00209113 |
|
|
|
|
Name : |
Mr. Sridar Arvamudhan Iyengar |
|
Designation : |
Director |
|
Address : |
85, Fair Oaks Lane, Atherton, Ca - 94027, United States of America |
|
Qualification
: |
B.Com ( Chartered Accountants) |
|
Date of Appointment : |
31.07.2014 |
|
DIN No.: |
00278512 |
|
|
|
|
Name : |
Mr. Bruce Leonard Andrews Carter |
|
Designation : |
Director |
|
Address : |
3915 NE Surber Dr, Seattle Wa - 98105, United States of America |
|
Qualification
: |
Ph.D. in Microbiology |
|
Date of Appointment : |
31.07.2014 |
|
DIN No.: |
02331774 |
KEY EXECUTIVES
|
Name : |
Gunupati Venkateswara Prasad |
|
Designation : |
Chief Executive Officer |
|
Address : |
Plot No.32, Road No.8, Banjara Hills, Hyderabad - 500034, Telangana , India |
|
PAN No : |
AAHPP9553C |
|
Date of Appointment : |
13.05.2014 |
|
|
|
|
Name : |
Saumen Chakraborty |
|
Designation : |
Chief Finance Officer |
|
Address : |
Survey No. 1009/P, Block 2B, F 900 Beverly Hills, Lodha Belleza, KPHB Colony, Hyderabad - 500016, Telangana, India |
|
PAN No : |
AASPC7909A |
|
Date of Appointment : |
13.05.2014 |
|
|
|
|
Name : |
Sandeep Poddar |
|
Designation : |
Secretary |
|
Address : |
Flat No. 301, 1-11-220, Chakkilam Residency, Street No. 4, Begumpet, Hyderabad - 500016, Telangana, India |
|
PAN No : |
AFPPP6507B |
|
Date of Appointment : |
22.07.2010 |
|
|
|
|
Name : |
Dr. S Chandrasekhar |
|
Designation : |
President And Head, Human Resources |
|
|
|
|
Name : |
Alok Sonig |
|
Designation : |
Senior Vice President and Head, India Business (Generics) |
|
|
|
|
Name : |
Samiran Das |
|
Designation : |
Executive Vice President and Head, FTO and Ggpm |
|
|
|
|
Name : |
M V Ramana |
|
Designation : |
Executive Vice-President and Head, Emerging Markets, Global Generics |
|
|
|
|
Name : |
Dr. Amit Biswas |
|
Designation : |
Executive Vice-President, Integrated Product Development |
|
|
|
|
Name : |
Abhijit Mukherjee |
|
Designation : |
Chief Operating Officer |
SHAREHOLDING PATTERN
AS ON 30.06.2015
|
Category of
Shareholder |
Total No. of Shares |
% of Total No. of
Shares |
|
|
||
|
(A) Shareholding of
Promoter and Promoter Group |
||
|
|
|
|
|
|
3688528 |
2.61 |
|
|
39762934 |
28.10 |
|
|
43451462 |
30.71 |
|
|
|
|
|
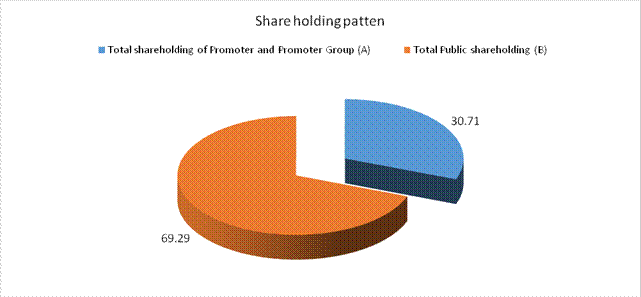
Total shareholding of
Promoter and Promoter Group (A) |
43451462 |
30.71 |
|
(B) Public Shareholding |
||
|
|
|
|
|
|
6691651 |
4.73 |
|
|
190737 |
0.13 |
|
|
3214613 |
2.27 |
|
|
64388951 |
45.51 |
|
|
74485952 |
52.64 |
|
|
|
|
|
|
7120577 |
5.03 |
|
|
|
|
|
|
11865535 |
8.39 |
|
|
810343 |
0.57 |
|
|
3754238 |
2.65 |
|
|
126244 |
0.09 |
|
|
2130108 |
1.51 |
|
|
430390 |
0.30 |
|
|
1059749 |
0.75 |
|
|
7747 |
0.01 |
|
|
23550693 |
16.64 |
|
Total Public shareholding
(B) |
98036645 |
69.29 |
|
Total (A)+(B) |
141488107 |
100.00 |
|
(C) Shares held by Custodians
and against which Depository Receipts have been issued |
0 |
0.00 |
|
|
0 |
0.00 |
|
|
29069815 |
0.00 |
|
|
29069815 |
0.00 |
|
Total (A)+(B)+(C) |
170557922 |
0.00 |
BUSINESS DETAILS
|
Line of Business : |
Subject operates
as an integrated pharmaceutical company. It operates in three segments: Global Generics, Pharmaceutical Services and Active
Ingredients (PSAI), and Proprietary Products: ·
The Global
Generics segment produces and markets finished pharmaceutical products as
branded formulations or generic finished dosages. This segment also engages
in biologics business. ·
The PSAI
segment develops active pharmaceutical ingredients (APIs) and intermediaries,
which are used as principal ingredients for finished pharmaceutical products.
This segment also offers contract research services to other companies; and
manufactures and sells APIs and steroids in accordance with the specific
customer requirements. · The Proprietary Products segment focuses on the discovery, development, and commercialization of novel small molecule agents in therapeutic areas, such as anti-infective, metabolic disorders, and pain and inflammation. It also provides differentiated formulations for unmet medical needs; and a portfolio of in-licensed patented dermatology products |
|
|
|
|
Products : |
|
|
|
|
|
Brand Names : |
Not Available |
|
|
|
|
Agencies Held : |
Not Available |
|
|
|
|
Exports : |
Not Available |
|
|
|
|
Imports : |
Not Available |
|
|
|
|
Terms : |
Not Available |
PRODUCTION STATUS: NOT AVAILABLE
GENERAL INFORMATION
|
Suppliers : |
|
||||||||||||||||||||||
|
|
|
||||||||||||||||||||||
|
Customers : |
|
||||||||||||||||||||||
|
|
|
||||||||||||||||||||||
|
No. of Employees : |
19857 (Approximately) |
||||||||||||||||||||||
|
|
|
||||||||||||||||||||||
|
Bankers : |
|
||||||||||||||||||||||
|
|
|
||||||||||||||||||||||
|
Facilities : |
Note: LONG TERM
BORROWINGS Finance lease obligations are towards lease rentals
payable for the vehicles leased by the Company. Lease rentals are paid in
monthly instalment, with the last instalment due in April 2017. |
|
Auditors : |
|
|
Name : |
BSR and Company LLP Chartered Accountants |
|
Address : |
8-2-618/2, Reliance Humsafar, 4th Floor, Road No. 11,
Banjara Hills, Hyderabad – 500034, Telangana, India |
|
Tel No: |
91-40-30465000 |
|
Fax No: |
91-40-30465299 |
|
|
|
|
Memberships : |
Not Available |
|
|
|
|
Collaborators : |
Not Available |
|
|
|
|
Subsidiaries
including step down subsidiaries: |
|
|
|
|
|
Joint Ventures : |
|
|
|
|
|
List of other
related parties with whom transactions have taken place during the current and/or
previous year : |
|
CAPITAL STRUCTURE
AS ON 31.03.2015
Authorised Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
240000000 |
Equity Shares |
Rs.5/- each |
Rs. 1200.000 Million |
|
|
|
|
|
Issued, Subscribed & Paid-up Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
170381374 |
Equity Shares |
Rs.10/- each |
Rs. 852.000 Million |
|
|
|
|
|
(a) Reconciliation of
the equity shares outstanding is set out below:
|
Equity Shares |
Number
of Shares |
Rs. In Million |
|
Number of shares outstanding at the beginning of the year |
170108868 |
851.000 |
|
Add : Equity shares issued pursuant to employee stock option plan |
272306 |
1.000 |
|
Number of shares outstanding at the end of the year |
170381174 |
852.000 |
(b) Terms / rights
attached to equity shares
The Company has only one class of equity shares having a par value of Rs. 5 per share. Each holder of equity shares is entitled to one vote per share. The Company declares and pays dividends in Indian rupees. During the year ended 31 March 2015, the amount of per share dividend recognized as distributions to equity shareholders is Rs. 20. The dividend proposed by the Board of Directors is subject to the approval of the shareholders in the ensuing Annual General Meeting. In the event of liquidation, the equity shareholders are eligible to receive the remaining assets of the Company after distribution of all preferential amounts, in proportion to their shareholding.
(c) Details of
shareholders holding more than 5% shares in the Company
|
Name of
Shareholder |
Number
of Shares |
% holding |
|
Dr. Reddy’s Holdings Limited |
39729284 |
23.32 |
|
First state investments management (UK) Limited, First state
investments International Limited and their associates |
14389390 |
8.45 |
(d) 585,454 (previous year: 651,674) stock options are outstanding and are to be issued by the Company upon exercise of the same in accordance with the terms of exercise under the “Dr. Reddy’s Employees Stock Option Plan 2002” and 98,350 (previous year: 97,463) stock options are outstanding and are to be issued by the Company upon exercise of the same in accordance with the terms of exercise under the “Dr. Reddy’s Employees ADR Stock Option Plan 2007”.
(e) Represents 200 equity shares of Rs. 5/- each, amount paid-up Rs. 500/- (rounded off in millions in the note above) forfeited due to non-payment of allotment money.
FINANCIAL DATA
[All figures are
in Rupees Million]
ABRIDGED
BALANCE SHEET
|
SOURCES OF FUNDS |
31.03.2015 |
31.03.2014 |
31.03.2013 |
|
|
|
|
|
|
I.
EQUITY AND LIABILITIES |
|
|
|
|
(1)Shareholders' Funds |
|
|
|
|
(a) Share Capital |
852.000 |
851.000 |
849.000 |
|
(b) Reserves &
Surplus |
105488.000 |
92439.000 |
76985.000 |
|
(c) Money received against
share warrants |
0.000 |
0.000 |
0.000 |
|
|
|
|
|
|
(2) Share Application
money pending allotment |
0.000 |
0.000 |
0.000 |
|
Total Shareholders’ Funds
(1) + (2) |
106340.000 |
93290.000 |
77834.000 |
|
|
|
|
|
|
(3) Non-Current Liabilities |
|
|
|
|
(a) long-term borrowings |
9391.000 |
9015.000 |
63.000 |
|
(b) Deferred tax
liabilities (Net) |
1290.000 |
1252.000 |
937.000 |
|
(c) Other long term
liabilities |
255.000 |
47.000 |
28.000 |
|
(d) long-term provisions |
498.000 |
335.000 |
298.000 |
|
Total Non-current
Liabilities (3) |
11434.000 |
10649.000 |
1326.000 |
|
|
|
|
|
|
(4) Current Liabilities |
|
|
|
|
(a) Short term borrowings |
21857.000 |
17630.000 |
15828.000 |
|
(b) Trade payables |
7160.000 |
8423.000 |
7678.000 |
|
(c) Other current
liabilities |
12374.000 |
10294.000 |
13011.000 |
|
(d) Short-term provisions |
5395.000 |
4795.000 |
4214.000 |
|
Total Current Liabilities
(4) |
46786.000 |
41142.000 |
40731.000 |
|
|
|
|
|
|
TOTAL |
164560.000 |
145081.000 |
119891.000 |
|
|
|
|
|
|
II.
ASSETS |
|
|
|
|
(1) Non-current assets |
|
|
|
|
(a) Fixed Assets |
|
|
|
|
(i) Tangible assets |
31294.000 |
26937.000 |
23355.000 |
|
(ii) Intangible Assets |
1200.000 |
546.000 |
515.000 |
|
(iii) Capital
work-in-progress |
4883.000 |
5761.000 |
4232.000 |
|
(iv) Intangible assets
under development |
0.000 |
0.000 |
0.000 |
|
(b) Non-current
Investments |
17601.000 |
17401.000 |
21826.000 |
|
(c) Deferred tax assets
(net) |
0.000 |
0.000 |
0.000 |
|
(d) Long-term Loan and Advances |
5538.000 |
5358.000 |
3752.000 |
|
(e) Other Non-current
assets |
11.000 |
0.000 |
209.000 |
|
Total Non-Current Assets |
60527.000 |
56003.000 |
53889.000 |
|
|
|
|
|
|
(2) Current assets |
|
|
|
|
(a) Current investments |
21022.000 |
10664.000 |
1966.000 |
|
(b) Inventories |
17233.000 |
15921.000 |
15265.000 |
|
(c) Trade receivables |
47117.000 |
45615.000 |
29639.000 |
|
(d) Cash and cash
equivalents |
9014.000 |
6651.000 |
9191.000 |
|
(e) Short-term loans and
advances |
8657.000 |
8287.000 |
8634.000 |
|
(f) Other current assets |
990.000 |
1940.000 |
1307.000 |
|
Total Current Assets |
104033.000 |
89078.000 |
66002.000 |
|
|
|
|
|
|
TOTAL |
164560.000 |
145081.000 |
119891.000 |
PROFIT
& LOSS ACCOUNT
|
|
PARTICULARS |
31.03.2015 |
31.03.2014 |
31.03.2013 |
|
|
SALES |
|
|
|
|
|
Income |
100110.000 |
97280.000 |
84340.000 |
|
|
Other Income |
2228.000 |
1515.000 |
1417.000 |
|
|
TOTAL |
102338.000 |
98795.000 |
85757.000 |
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
Cost of Materials
Consumed |
22484.000 |
21918.000 |
22773.000 |
|
|
Purchases of
Stock-in-Trade |
5261.000 |
4690.000 |
3931.000 |
|
|
Conversion charges |
924.000 |
785.000 |
592.000 |
|
|
Changes in inventories of
finished goods, work-in-progress and Stock-in-Trade |
(289.000) |
(1706.000) |
(1006.000) |
|
|
Employees benefits
expense |
14909.000 |
14199.000 |
11381.000 |
|
|
Other expenses |
32910.000 |
29777.000 |
19667.000 |
|
|
Excise duty |
0.000 |
0.000 |
636.000 |
|
|
Research and Development
Expenses |
0.000 |
0.000 |
6509.000 |
|
|
TOTAL |
76199.000 |
69663.000 |
64483.000 |
|
|
|
|
|
|
|
Less |
PROFIT/ (LOSS) BEFORE INTEREST, TAX, DEPRECIATION AND AMORTISATION
|
26139.000 |
29132.000 |
21274.000 |
|
|
|
|
|
|
|
Less |
FINANCIAL EXPENSES |
638.000 |
783.000 |
614.000 |
|
|
|
|
|
|
|
|
PROFIT / (LOSS) BEFORE
TAX, DEPRECIATION AND AMORTISATION |
25501.000 |
28349.000 |
20660.000 |
|
|
|
|
|
|
|
Less/ Add |
DEPRECIATION/
AMORTISATION |
4902.000 |
3805.000 |
3128.000 |
|
|
|
|
|
|
|
|
PROFIT/ (LOSS) BEFORE TAX |
20599.000 |
24544.000 |
17532.000 |
|
|
|
|
|
|
|
Less |
TAX |
3805.000 |
5216.000 |
4877.000 |
|
|
|
|
|
|
|
|
PROFIT/ (LOSS) AFTER TAX |
16794.000 |
19328.000 |
12655.000 |
|
|
|
|
|
|
|
Add |
PREVIOUS YEARS’ BALANCE
BROUGHT FORWARD |
56599.000 |
43614 |
36049 |
|
|
|
|
|
|
|
Less |
APPROPRIATIONS |
|
|
|
|
|
Proposed dividend on equity
shares |
3408.000 |
3062.000 |
2548.000 |
|
|
Tax on proposed dividend |
694.000 |
520.000 |
433.000 |
|
|
Credit of dividend distribution
tax |
(13.000) |
(2.000) |
(4.000) |
|
|
Dividend of previous years
(including tax) |
6.000 |
3.000 |
3.000 |
|
|
Debenture Redemption Reserve |
---- |
827.000 |
845.000 |
|
|
Transfer to General Reserve |
1679.000 |
1933.000 |
1265.000 |
|
|
Total |
5774.000 |
6343.000 |
5090.000 |
|
|
|
|
|
|
|
|
Balance Carried to the
B/S |
67619.000 |
56599.000 |
43614.000 |
|
|
|
|
|
|
|
|
EARNINGS IN FOREIGN
CURRENCY |
|
|
|
|
|
F.O.B.
Value of Exports |
74146.000 |
74059.000 |
61004.000 |
|
|
Interest
on loan to subsidiaries |
187.000 |
90.000 |
181.000 |
|
|
Service
income and license fees |
390.000 |
1495.000 |
1684.000 |
|
|
Royalty
income |
0.000 |
0.000 |
0.000 |
|
|
Guarantee
commission |
132.000 |
176.000 |
177.000 |
|
|
Litigation
settlement |
0.000 |
162.000 |
1220.000 |
|
|
Others |
15.000 |
25.000 |
24.000 |
|
|
TOTAL EARNINGS |
74870.000 |
76007.000 |
64290.000 |
|
|
|
|
|
|
|
|
IMPORTS |
|
|
|
|
|
Raw Materials |
11512.000 |
11031.000 |
8837.000 |
|
|
Capital equipment (including spares and components) |
1973.000 |
1931.000 |
2650.000 |
|
|
TOTAL IMPORTS |
13485.000 |
12962.000 |
11487.000 |
|
|
|
|
|
|
|
|
Earnings / (Loss) Per
Share (Rs.) |
|
|
|
|
|
Basic |
98.60 |
113.67 |
74.54 |
|
|
Diluted |
98.18 |
113.13 |
74.17 |
CURRENT MATURITIES
OF LONG TERM DEBT DETAILS
|
Particulars |
31.03.2015 |
31.03.2014 |
31.03.2013 |
|
Current
Maturities of Long term borrowing |
|
|
|
|
Finance lease obligations |
6.000 |
4.000 |
12.000 |
|
Sales tax deferment loan from the Government of Telangana (interest
free) |
7.000 |
13.000 |
19.000 |
|
Cash generated from operations |
30699.000 |
26840.000 |
6646.000 |
|
Net cash flow from (used in) operations |
17861.000 |
9055.000 |
2888.000 |
KEY
RATIOS
|
PARTICULARS |
|
31.03.2015 |
31.03.2014 |
31.03.2013 |
|
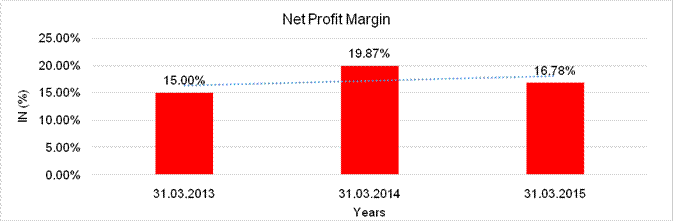
Net Profit Margin (PAT / Sales) |
(%) |
16.78 |
19.87 |
15.00 |
|
|
|
|
|
|
|
Operating Profit Margin (PBIDT/Sales) |
(%) |
26.11 |
29.95 |
25.22 |
|
|
|
|
|
|
|
Return on Total Assets (PBT/Total Assets} |
(%) |
14.50 |
20.13 |
18.68 |
|
|
|
|
|
|
|
Return on Investment (ROI) (PBT/Networth) |
|
0.19 |
0.26 |
0.23 |
|
|
|
|
|
|
|
Debt Equity Ratio (Total Debt /Networth) |
|
0.29 |
0.29 |
0.20 |
|
|
|
|
|
|
|
Current Ratio (Current Asset/Current Liability) |
|
2.22 |
2.17 |
1.62 |
QUARTERLY RESULTS
|
PARTICULARS |
|
|
30.06.2015 |
|
Unaudited |
|
|
1rd
Quarter |
|
Net Sales/Income from operations |
|
|
25128.900 |
|
Other Operating Income |
|
|
130.400 |
|
Total Income From Operations |
|
|
25259.300 |
|
EXPENDITURE |
|
|
|
|
Consumption of Raw Materials |
|
|
5011.900 |
|
Purchase of Traded Goods |
|
|
1543.000 |
|
Increase/Decrease in Stocks |
|
|
(380.700) |
|
Employees Cost |
|
|
4087.700 |
|
Depreciation |
|
|
1413.900 |
|
Admin. And Selling Expenses |
|
|
2622.900 |
|
Other Expenses |
|
|
5760.300 |
|
P/L Before Other Inc., Int., Except. Items and Tax |
|
|
5200.300 |
|
Other Income |
|
|
909.700 |
|
P/L Before Int., Except. Items and Tax |
|
|
6110.000 |
|
Interest |
|
|
213.200 |
|
P/L Before Exceptional Items and Tax |
|
|
5896.800 |
|
P/L Before Tax |
|
|
5896.800 |
|
Tax |
|
|
1138.300 |
|
P/L After Tax from Ordinary Activities |
|
|
4758.500 |
STOCK
PRICES
|
Face Value |
Rs. 5.00/- |
|
Market Value |
Rs. 4230.00/- |
FINANCIAL ANALYSIS
[All figures are
in Rupees Million]
DEBT EQUITY RATIO
|
Particular |
31.03.2013 |
31.03.2014 |
31.03.2015 |
|
|
(Rs. In Million) |
(Rs. In Million) |
(Rs. In Million) |
|
Share Capital |
849.000 |
851.000 |
852.000 |
|
Reserves & Surplus |
76985.000 |
92439.000 |
105488.000 |
|
Share Application money pending allotment |
0.000 |
0.000 |
0.000 |
|
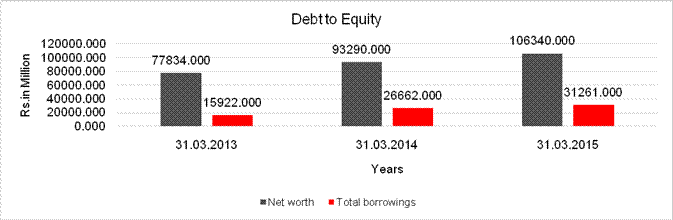
Net worth |
77834.000 |
93290.000 |
106340.000 |
|
|
|
|
|
|
long-term borrowings |
63.000 |
9015.000 |
9391.000 |
|
Short term borrowings |
15828.000 |
17630.000 |
21857.000 |
|
Current maturities of
long-term borrowing |
31.000 |
17.000 |
13.000 |
|
Total borrowings |
15922.000 |
26662.000 |
31261.000 |
|
Debt/Equity ratio |
0.205 |
0.286 |
0.294 |
YEAR-ON-YEAR GROWTH
|
Year on Year Growth |
31.03.2013 |
31.03.2014 |
31.03.2015 |
|
|
(Rs. In Million) |
(Rs. In Million) |
(Rs. In Million) |
|
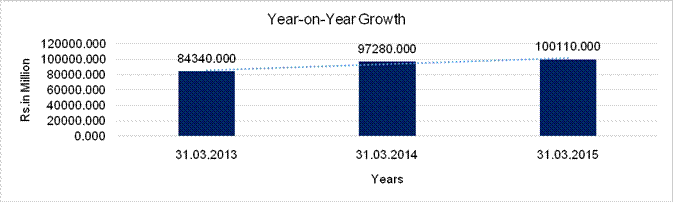
Sales |
84340.000 |
97280.000 |
100110.000 |
|
|
|
15.343 |
2.909 |
NET PROFIT MARGIN
|
Net Profit Margin |
31.03.2013 |
31.03.2014 |
31.03.2015 |
|
|
(Rs. In Million) |
(Rs. In Million) |
(Rs. In Million) |
|
Sales |
84340.000 |
97280.000 |
100110.000 |
|
Profit |
12655.000 |
19328.000 |
16794.000 |
|
|
15.00% |
19.87% |
16.78% |
LOCAL AGENCY FURTHER INFORMATION
|
Sr. No. |
Check list by
info agents |
Available in
Report (Yes/No) |
|
1 |
Year of establishment |
Yes |
|
2 |
Constitution of the entity -Incorporation
details |
Yes |
|
3 |
Locality of the entity |
Yes |
|
4 |
Premises details |
No |
|
5 |
Buyer visit details |
------------ |
|
6 |
Contact numbers |
Yes |
|
7 |
Name of the person contacted |
No |
|
8 |
Designation of contact person |
No |
|
9 |
Promoter’s background |
Yes |
|
10 |
Date of Birth of Proprietor / Partners /
Directors |
Yes |
|
11 |
Pan Card No. of Proprietor / Partners |
No |
|
12 |
Voter Id Card No. of Proprietor / Partners |
No |
|
13 |
Type of business |
Yes |
|
14 |
Line of Business |
Yes |
|
15 |
Export/import details (if applicable) |
No |
|
16 |
No. of employees |
Yes |
|
17 |
Details of sister concerns |
Yes |
|
18 |
Major suppliers |
No |
|
19 |
Major customers |
No |
|
20 |
Banking Details |
Yes |
|
21 |
Banking facility details |
Yes |
|
22 |
Conduct of the banking account |
-- |
|
23 |
Financials, if provided |
Yes |
|
24 |
Capital in the business |
Yes |
|
25 |
Last accounts filed at ROC, if applicable |
Yes |
|
26 |
Turnover of firm for last three years |
Yes |
|
27 |
Reasons for variation <> 20% |
-- |
|
28 |
Estimation for coming financial year |
No |
|
29 |
Profitability for last three years |
Yes |
|
30 |
Major shareholders, if available |
Yes |
|
31 |
External Agency Rating, if available |
No |
|
32 |
Litigations that the firm/promoter
involved in |
--- |
|
33 |
Market information |
--- |
|
34 |
Payments terms |
No |
|
35 |
Negative Reporting by Auditors in the
Annual Report |
No |
UNSECURED LOAN
|
PARTICULARS |
31.03.2015 (Rs.
in Million) |
31.03.2014 (Rs.
in Million) |
|
Long Term
Borrowing |
|
|
|
Sales tax deferment loan from the government of Telangana (interest free) |
8.000 |
15.000 |
|
Long term loans from banks |
9375.000 |
8987.000 |
|
Short Term
Borrowing |
|
|
|
From banks |
|
|
|
Packing credit loans |
20857.000 |
17630.000 |
|
Other short term borrowing |
1000.000 |
------ |
|
Total |
31240.000 |
26632.000 |
LONG TERM
BORROWING NOTES:
Sales tax deferment loan is repayable in 5 instalments, with the last instalment due on 31 March 2019.
External Commercial Borrowings of USD 150 million carrying interest rate of LIBOR plus 125 bps and is repayable in five equal quarterly instalments ending in February 2019. As part of the loan arrangement, the Company is required to comply with certain financial covenants and the Company was in compliance with such covenants as at 31 March 2015 and 31 March 2014.
Packing credit loans for the current year comprised of USD and EUR denominated loans carrying interest rates of LIBOR plus 7.5 - 40 bps and RUB denominated loans carrying fixed interest rate of 9.80% - 22.30%, and are repayable within 3 to 12 months from the date of drawdown. Packing Credit loans for the previous year comprised of USD and EUR denominated loans carrying interest rates of LIBOR plus 20 - 85 bps, RUB denominated loans carrying interest rate of Moscow Prime Offered Rate plus 60 bps, RUB denominated loans carrying fixed interest rate of 7.20% - 7.75% per annum and INR denominated loans carrying fixed interest rate of 9.50% - 10%, and are repayable within 1 to 6 months from the date of drawdown.
Other short term borrowing comprises of INR denominated loan
carrying fixed interest rate of 10.00% and is repayable in April 2015
MANAGEMENT DISCUSSION
AND ANALYSIS:
Global Generics (GG) includes branded and unbranded prescription and Over-The-Counter (OTC) drug products business. It also includes the operations of our Biologics business.
Pharmaceutical Services and Active Ingredients (PSAI) consists of Active Pharmaceutical Ingredients (API) and Custom Pharmaceutical Services (CPS) businesses.
Proprietary Products comprises Differentiated Formulations business, New Chemical Entities (NCEs) business and our dermatology focused specialty business.
We have a strong presence in highly regulated markets such as the United States of America, the United Kingdom and Germany, as well as in other key markets such as India, Russia, Venezuela, Romania, South Africa and certain countries of the former Soviet Union.
Given below is a brief outline of the businesses. Further details are to be found in subsequent sections of this chapter.
GLOBAL GENERICS
Revenues from Global Generics stood at Rs. 120560.000 Million registering a year-on-year growth of 15% — driven largely by North America, Venezuela and India.
Revenues from North America stood at Rs. 64720.000 Million registering a year-on-year growth of 17%. Growth was largely driven by ‘complex to make’ injectable products launched during FY2014, market share gains in some of the key molecules and contribution from new products. During the year, Dr. Reddy’s launched 12 products with Valganciclovir and Sirolimus being the major products. FY2015 also saw 13 product fi lings in the USA. Cumulatively, 68 Abbreviated New Drug Applications (ANDAs) are pending for approval from the US Food and Drug Administration (USFDA). of these, 43 are Para IVs — out of which Dr. Reddy’s believes 13 to have ‘First to File’ status.
Revenues from
Emerging Markets stood at Rs. 30770.000 Million registering a year-on-year
growth of 13%. Within Emerging Markets: Revenue from Russia and other CIS
markets declined by 11% to Rs. 17710.000 Million, primarily due to the sharp
depreciation of the rouble. Revenue from the Rest of the World (RoW)
territories was Rs. 13060.000 Million and grew by 77%
PHARMACEUTICAL
SERVICES AND ACTIVE INGREDIENTS (PSAI)
Revenues from PSAI stood at Rs. 25460.000 Million registering a year-on-year growth of 6%. Despite moderate sales growth, the margins improved significantly due to a better product mix.
77 Drug Master Files (DMFs) were fi led globally, including
12 in the US and 16 in Europe. The cumulative number of DMF as on 31 March 2015
was 735.
OUTLOOK
The Company believes that its focus on profitable growth and targeting a leadership position in Global Generics and PSAI will create significant value in the near term.
In GG, improving depth through portfolio expansion, cost leadership, consistent delivery of limited competition products and supply chain excellence should lead us to leadership positions in key markets. In PSAI, the objective is to be the partner of choice by creating compelling value information that is included in this report. This write-up includes some forward-looking statements, as defined in the US Private Securities Litigation Reform Act of 1995. The management has based these forward-looking statements on its current expectations and projections about future events. Such statements involve known and unknown risks, uncertainties and other factors that may cause actual results to differ materially. These factors include, but are not limited to, changes in local and global economic conditions, the Company’s ability to successfully implement its strategy, the market’s acceptance of and demand for its products, growth and expansion, technological change and exposure to market risks. By their nature, these expectations and projections are only estimates and could be materially different from actual results in the future.
For customers through leveraging IP, technology and cost leadership. In Proprietary Products, the aim is to create a viable business by calibrating investments to produce a self-sustainable model.
The Company has a positive outlook for the next year. The largest increment of growth is expected to be contributed by the North America generics and Emerging Markets businesses.
In a dynamic environment, Dr. Reddy’s base business model is exposed to volatility, both upwards and downwards. While the upsides create non-linear value for the organization, there is a conscious attempt to protect it against the downsides.
STATEMENT OF UNAUDITED STANDALONE
FINANCIAL RESULTS FOR THE QUARTER ENDED 30 JUNE 2015.
(Rs. in Million)
|
Income from Operations |
30.06.2015 (Unaudited) |
|
(a) Net sates/income from operations (Net of excise duty) |
25083.300 |
|
(b) license fees and service income |
45.600 |
|
(c) Other Operating Income |
130.400 |
|
Total income from
operations (net) |
25259.300 |
|
Expenses |
|
|
(a) Cost of materials consumed |
5011.900 |
|
(b) Purchases of stock-in trade |
1543.000 |
|
(c) Changes in inventories of finished goods. work-in-progress and
stock in trade |
(380.700) |
|
(d) Employee benefits expense |
4087.700 |
|
(e) selling expense |
2622.900 |
|
(e) Depreciation and Amortization Expenses |
1413.900 |
|
(f) Other Expenses |
5760.300 |
|
(g) Power and Fuel |
|
|
(h) Value of Wind Power |
|
|
Total expenses |
20059.000 |
|
Profit/ (Loss) from operations before other Income, finance costs and
exceptional Items (1-2) |
5200.300 |
|
Other Income |
909.700 |
|
Profit/ (Loss) from operations before other income, finance costs and
exceptional items (3+4) |
6110.000 |
|
Finance Costs |
213.200 |
|
Profit/ (Loss) from ordinary activities after finance cost but before
exceptional items (5-6) |
5896.800 |
|
Exceptional items |
0.000 |
|
Profit/ (Loss) from ordinary activities before tax (7+8) |
5896.800 |
|
Tax expenses |
1138.300 |
|
Net Profit / (Loss) from ordinary activities after tax (9-10) |
4758.500 |
|
Extraordinary item (net of tax expense) |
|
|
Net Profit / (Loss) for the period (11-12) |
4758.500 |
|
Share of profit' (loss) of associates |
|
|
Minority Interest |
|
|
Net Profit/ (Loss) after taxes, minority interest and share of
profit/(loss) of associates (13+14+15) |
|
|
Paid up equity share capital (Face Value of Rs 10/-each) |
852.8 |
|
Reserve excluding Revaluation Reserve as per Balance Sheet of previous
accounting year |
|
|
Earnings per share (before extraordinary items) of Rs.10/- each (not
annualized): |
|
|
(a) Basic |
27.92 |
|
(b) Diluted |
27.81 |
|
PARTICULARS |
30.06.2015 Quarter
ended |
|
|
|
|
PARTICULARS OF SHAREHOLDING |
|
|
A. Public Shareholding |
|
|
- Number of shares |
98036645 |
|
- Percentage of shareholding |
57.48 |
|
Promoters and Promoter group shareholding |
|
|
a) Pledged / Encumbered |
|
|
- Number of shares |
|
|
- Percentage of shares (as a % of the total shareholding of Promoter
& Promoter group) |
|
|
- Percentage of shares (as a % of the total Share Capital of the
Company) |
|
|
b) Non Encumbered |
|
|
- Number of shares |
43451462 |
|
- Percentage of shares (as a % of the total shareholding of Promoter
& Promoter group) |
100 |
|
- Percentage of shares (as a % of the total Share Capital of the
Company) |
25.48 |
|
Investor
Complaints |
|
|
Pending at the beginning
of the quarter |
Nil |
|
Received during the
quarter |
8 |
|
Disposed of during the
quarter |
8 |
|
Remaining unresolved at
the end of the quarter |
Nil |
SEGMENTWISE
REVENUE, RESULTS AND CAPITAL EMPLOYED
(Rs. In Million)
|
Particulars |
Quarter
Ended |
|
|
30.06.2015 [Unaudited] |
|
1.
Segment Revenue |
|
|
Pharmaceutical services and
active ingredients |
5777.900 |
|
Global generics |
20667.800 |
|
Proprietary products |
------ |
|
Total |
26445.700 |
|
Less : Inter Segment Revenue |
1186.400 |
|
Add: Other unallocable income |
---- |
|
Net
Sales |
25259.300 |
|
|
|
|
2.
Segment Result |
|
|
Profit before tax and interest from each
segment |
|
|
Pharmaceutical services and
active ingredients |
276.400 |
|
Global generics |
7569.400 |
|
Proprietary products |
(718.800) |
|
Total |
7127.000 |
|
Less : Interest |
213.200 |
|
Exceptional
Item - Foreign Exchange Gain/(Loss) |
1017.000 |
|
Profit
from ordinary activities before tax |
5896.800 |
|
|
|
|
3. Capital Employed (Segment Assets-Segment
Liabilities) |
|
|
Pharmaceutical services and
active ingredients |
30069.600 |
|
Global generics |
71036.400 |
|
Proprietary products |
(837.600) |
|
Unallocated |
10936.200 |
|
Total |
111204.600 |
Notes:
1. The above financial results are prepared in accordance with Indian generally accepted accounting principles (GAAP) under the historical cost convention on the accrual basis except for certain financial instrument which are measured at fair value. GAAP comprises accounting standards specified under section 133 of the companies act, 2013 read with rule 7 of companies (accounts) rules, 2014, guidelines issued by securities and exchange board of India and other accounting principles generally accepted in India.
2. On 1 April 2015, the company entered into a definitive agreement to acquire a select portfolio of established products’ business of UCB in the territories of India, Nepal, Sri Lanka, and Maldives for a total consideration of Rs. 8000.000 Million on 16 June 2015, the company completed the acquisition and recorded Rs. 200.000 Million, Rs. 7480.000 Million and Rs. 320.000 Million towards various current and fixed assets, intangible assets, and goodwill respectively. The acquisition pertains to company’s global generics segment.
3. The audited results have been reviewed by the committee of the board and approved by the board of directors of the company at their meeting held on 30 July 2015.
4. The results for the quarter ended 30 June 2015 have been subjected to a “limited review” by the statutory auditors of the company. An unqualified report has been issued by them thereon.
5. The figures for the previous periods have been re-grouped/ re-classified, whenever necessary, to conform to the current period’s classification.
FIXED ASSETS
Tangible assets
· Land
· Building
· Plant and machinery
· Electrical equipment
· Laboratory equipment
· Furniture and fixture
· Office equipment
· Vehicles
Intangible assets
· Technical know how
· Product related intangible
· Copyrights and patents
·
Customer contracts
WEBSITE DETAILS
PRESS RELEASES /
NEWS:
DR
REDDY'S LABS TO BUY IP RIGHTS OF FONDAPARINUX
September
25, 2015
Australian drug discovery and development company Alchemia Limited has entered
into an agreement for sale of exclusive intellectual property rights of
fondaparinux sodium to Dr Reddy's Laboratories (DRL), for USD 17.5 million
(about Rs 1156.000 Million). Fondaparinux is a generic version of the
anticoagulant drug Arixtra which is indicated for the treatment of deep vein
thrombosis (DVT) and pulmonary embolism (PE). It is also indicated for
prevention of DVT after major surgery, such as knee and hip replacement
worldwide.
According to a statement issued by Alchemia, under the binding terms of the sale, Alchemia will receive USD 17.5 Million in cash from Dr Reddy's upon the closing of the transaction. In 2007, Alchemia granted Dr Reddy's non-exclusive rights to manufacture fondaparinux sodium in API (Active Pharmaceutical Ingredient) form, and exclusive rights to market fondaparinux in the North America. In exchange for these manufacturing and marketing rights, Alchemia is entitled to receive 50 per cent of the net profit generated from sales of fondaparinux in North America.
Dr Reddy's is responsible for finishing of the product, all regulatory filings
and market launch. "Should the transaction be approved by shareholders and
the transaction subsequently closed, the company will not be entitled to
receive any further profit share revenues from fondaparinux sales. “The sale is
conditional on shareholder approval, as the sale constitutes a disposal of the
company's main undertaking as per the ASX Listing Rules," Alchemia said.
Dr Reddys Labs stock
price
On October 01, 2015, Dr Roddys Laboratories closed at Rs 4230.05, up Rs 74.60,
or 1.80 percent. The 52-week high of the share was Rs 4337.00 and the 52-week
low was Rs 2883.40.
The company's trailing 12-month (TTM) EPS was at Rs 91.01 per share as per the quarter ended June 2015. The stock's price-to-earnings (P/E) ratio was 46.91. The latest book value of the company is Rs 623.38 per share. At current value, the price-to-book value of the company is 6.85.
DR REDDY'S SIGNS
COMMERCIALISATION DEAL WITH HATCHTECH
September 14, 2015
Drugmaker Dr Reddy's Laboratories said it has signed a commercialization deal
with Australia's Hatchtech for the latter's prescription head lice product
Xeglyze Lotion.
As part of the agreement, Dr Reddy's will pay Hatchtech an upfront USD 10 million, followed by up to USD 50 million based on pre-commercialization milestones and an undisclosed amount based on post-commercialisation milestones, linked to achievement of annual net sales targets.
"The exclusive rights for this product are applicable to the territories of the United States, Canada, India, Russia and the CIS, Australia, New Zealand and Venezuela," Dr Reddy's Laboratories said in a BSE filing.
"Additionally, Hatchtech announced it will be filing its New Drug Application for Xeglyze with the US Food and Drug Administration (FDA) today. If approved, the product will be marketed in the United States by Promius Pharma, a wholly-owned specialty company of Dr Reddy's Laboratories.
"Dr Reddy's said Hatchtech in September 2014 had announced positive results from its two pivotal phase three clinical studies, evaluating Xeglyze Lotion as a potential treatment for head lice infestation.
"The studies involved 704 people across 14 clinical trial sites in the US and resulted in an 81.5 percent success rate, following the observation period of 14 days after a single 10 minute application," it added.
Xeglyze Lotion, a topical formulation containing abametapir, an inhibitor of metalloproteases, has demonstrated both ovicidal and lousicidal activity and offers potential for a more effective treatment using only a single application, the company said.
The active drug substance was developed in collaboration with Dr Reddy's Custom Pharmaceutical Services (CPS) business unit. The stock of Dr Reddy's Laboratories was trading at Rs 4,006.20, up 0.02 percent, on BSE.
Dr Roddys Labs stock price
On October 06, 2015, Dr Reddys Laboratories was quoting at Rs 4230.05, up Rs
74.60, or 1.80 percent. The 52-week high of the share was Rs 4337.00 and the
52-week low was Rs 2883.40.
The company's trailing 12-month (TTM) EPS was at Rs 91.01 per share as per the quarter ended June 2015. The stock's price-to-earnings (P/E) ratio was 46.48. The latest book value of the company is Rs 623.38 per share. At current value, the price-to-book value of the company is 6.85.
CIPLA ROPES IN DR
REDDY'S UMANG VOHRA AS GLOBAL CFO
September 02, 2015
In a management rejig, drug major Cipla Limited appointed former head of
Dr Reddy's North American business Umang Vohra as its Global Chief Financial
and Strategy Officer.
While, former HR Head of Reliance Industries Prabir Jha is appointed as Global Chief People Officer. These changes will be effective from October 1, the company said in a BSE filing.
Further the company said that Jha, in addition to providing leadership to the human resources function globally will lead the corporate communications and administration functions as well.
Commenting about the appointment of Vohra, Cipla Limited MD and Global CEO
Subhanu Saxena said, "He brings deep insights about the global generics
industry as well as an understanding of value creation through his background
in leading business and finance roles."
Prior to Dr Reddy's, Umang Vohra worked with PepsiCo and Eicher Motors. Samina Vaziralli, Executive Director who has been interim Head of HR said: "We truly believe focusing and building on human capital is one of the ultimate sources of sustainable, competitive advantage."
Cipla stock price
On October 06, 2015, Cipla closed at Rs 656.65, up Rs 635.20, down Rs 2.6, or 0.41 percent. The 52-week high of the share was Rs 752.45 and the 52-week low was Rs 571.05.
The company's trailing 12-month (TTM) EPS was at Rs 19.16 per share as per the quarter ended June 2015. The stock's price-to-earnings (P/E) ratio was 33.15. The latest book value of the company is Rs 137.98 per share. At current value, the price-to-book value of the company is 4.60.
CMT REPORT (Corruption, Money Laundering & Terrorism]
The Public Notice information has been collected from various sources
including but not limited to: The Courts,
1] INFORMATION ON
DESIGNATED PARTY
No exist designating subject or any of its beneficial owners,
controlling shareholders or senior officers as terrorist or terrorist
organization or whom notice had been received that all financial transactions
involving their assets have been blocked or convicted, found guilty or against
whom a judgement or order had been entered in a proceedings for violating
money-laundering, anti-corruption or bribery or international economic or
anti-terrorism sanction laws or whose assets were seized, blocked, frozen or
ordered forfeited for violation of money laundering or international
anti-terrorism laws.
2] Court Declaration :
No exist to suggest that subject is or was
the subject of any formal or informal allegations, prosecutions or other
official proceeding for making any prohibited payments or other improper
payments to government officials for engaging in prohibited transactions or
with designated parties.
3] Asset Declaration :
No records exist to suggest that the property or assets of the subject
are derived from criminal conduct or a prohibited transaction.
4] Record on Financial
Crime :
Charges or conviction
registered against subject: None
5] Records on Violation of
Anti-Corruption Laws :
Charges or investigation
registered against subject: None
6] Records on Int’l
Anti-Money Laundering Laws/Standards :
Charges or
investigation registered against subject: None
7] Criminal Records
No
available information exist that suggest that subject or any of its principals
have been formally charged or convicted by a competent governmental authority
for any financial crime or under any formal investigation by a competent
government authority for any violation of anti-corruption laws or international
anti-money laundering laws or standard.
8] Affiliation with
Government :
No record
exists to suggest that any director or indirect owners, controlling
shareholders, director, officer or employee of the company is a government
official or a family member or close business associate of a Government
official.
9] Compensation Package :
Our market
survey revealed that the amount of compensation sought by the subject is fair
and reasonable and comparable to compensation paid to others for similar
services.
10] Press Report : No press reports / filings exists on the
subject.
CORPORATE GOVERNANCE
MIRA INFORM as part of its Due Diligence do provide comments on
Corporate Governance to identify management and governance. These factors often
have been predictive and in some cases have created vulnerabilities to credit
deterioration.
Our Governance Assessment focuses principally on the interactions
between a company’s management, its Board of Directors, Shareholders and other
financial stakeholders.
CONTRAVENTION
Subject is not known to have contravened any existing local laws,
regulations or policies that prohibit, restrict or otherwise affect the terms
and conditions that could be included in the agreement with the subject.
FOREIGN EXCHANGE RATES
|
Currency |
Unit
|
Indian Rupees |
|
US Dollar |
1 |
Rs. 65.29 |
|
|
1 |
Rs. 99.31 |
|
Euro |
1 |
Rs. 73.39 |
INFORMATION DETAILS
|
Analysis Done by
: |
DIV |
|
|
|
|
Report Prepared
by : |
SANS |
SCORE & RATING EXPLANATIONS
|
SCORE FACTORS |
RANGE |
POINTS |
|
HISTORY |
1~10 |
9 |
|
PAID-UP CAPITAL |
1~10 |
9 |
|
OPERATING SCALE |
1~10 |
9 |
|
FINANCIAL
CONDITION |
|
|
|
--BUSINESS SCALE |
1~10 |
9 |
|
--PROFITABILITY |
1~10 |
9 |
|
--LIQUIDITY |
1~10 |
9 |
|
--LEVERAGE |
1~10 |
9 |
|
--RESERVES |
1~10 |
9 |
|
--CREDIT LINES |
1~10 |
9 |
|
--MARGINS |
-5~5 |
---- |
|
DEMERIT POINTS |
|
|
|
--BANK CHARGES |
YES/NO |
YES |
|
--LITIGATION |
YES/NO |
NO |
|
--OTHER ADVERSE INFORMATION |
YES/NO |
NO |
|
MERIT POINTS |
|
|
|
--SOLE DISTRIBUTORSHIP |
YES/NO |
NO |
|
--EXPORT ACTIVITIES |
YES/NO |
NO |
|
--AFFILIATION |
YES/NO |
YES |
|
--LISTED |
YES/NO |
YES |
|
--OTHER MERIT FACTORS |
YES/NO |
YES |
|
DEFAULTER |
|
|
|
--RBI |
YES/NO |
NO |
|
--EPF |
YES/NO |
NO |
|
TOTAL |
|
81 |
This score serves as a reference to assess
SC’s credit risk and to set the amount of credit to be extended. It is
calculated from a composite of weighted scores obtained from each of the major
sections of this report. The assessed factors and their relative weights (as
indicated through %) are as follows:
Financial
condition (40%) Ownership
background (20%) Payment
record (10%)
Credit history
(10%) Market trend (10%) Operational size
(10%)
RATING EXPLANATIONS
|
RATING |
STATUS |
PROPOSED CREDIT LINE |
|
|
>86 |
Aaa |
Possesses an extremely sound financial base with the strongest
capability for timely payment of interest and principal sums |
Unlimited |
|
71-85 |
Aa |
Possesses adequate working capital. No caution needed for credit transaction.
It has above average (strong) capability for payment of interest and
principal sums |
Large |
|
56-70 |
A |
Financial & operational base are regarded healthy. General
unfavorable factors will not cause fatal effect. Satisfactory capability for
payment of interest and principal sums |
Fairly Large |
|
41-55 |
Ba |
Overall operation is considered normal. Capable to meet normal
commitments. |
Satisfactory |
|
26-40 |
B |
Capability to overcome financial difficulties seems comparatively
below average. |
Small |
|
11-25 |
Ca |
Adverse factors are apparent. Repayment of interest and principal sums
in default or expected to be in default upon maturity |
Limited with full
security |
|
<10 |
C |
Absolute credit risk exists. Caution needed to be exercised |
Credit not
recommended |
|
-- |
NB |
New Business |
-- |
This report is issued at your request without any
risk and responsibility on the part of MIRA INFORM PRIVATE LIMITED (MIPL) or
its officials.