|
|
|
MIRA INFORM REPORT
|
Report No. : |
504948 |
|
Report Date : |
21.04.2018 |
IDENTIFICATION DETAILS
|
Name : |
SUN PHARMA ADVANCED RESEARCH COMPANY LIMITED |
|
|
|
|
Registered
Office : |
Akota Road, Akota, Vadodara – 390020, Gujarat |
|
Tel. No.: |
91-265-2330815/ 2339257 |
|
|
|
|
Country : |
India |
|
|
|
|
Financials (as
on) : |
31.03.2017 |
|
|
|
|
Date of
Incorporation : |
01.03.2006 |
|
|
|
|
Com. Reg. No.: |
04-047837 |
|
|
|
|
Capital
Investment / Paid-up Capital : |
INR 246.904 Million |
|
|
|
|
CIN No.: [Company Identification
No.] |
L73100GJ2006PLC047837 |
|
|
|
|
IEC No.: [Import-Export Code No.] |
Not Divulged |
|
|
|
|
TAN No.: [Tax Deduction &
Collection Account No.] |
Not Available |
|
|
|
|
PAN No.: [Permanent Account No.] |
Not Available |
|
|
|
|
GSTN : [Goods & Service Tax
Registration No.] |
Not Divulged |
|
|
|
|
Legal Form : |
A Public Limited Liability Company. The Company’s Shares are Listed on
the Stock Exchanges. |
|
|
|
|
Line of Business
: |
Subject is engaged in Research and experimental development on natural
sciences and engineering. (Registered Activity) |
|
|
|
|
No. of Employees
: |
398 (Approximately) |
RATING & COMMENTS
(Mira Inform has adopted New Rating mechanism w.e.f. 23rd January
2017)
|
MIRA’s Rating : |
A+ |
|
Credit Rating |
Explanation |
Rating Comments |
|
A+ |
Low Risk |
Business dealings permissible with low
risk of default |
|
Status : |
Excellent |
|
|
|
|
Payment Behaviour : |
Regular |
|
|
|
|
Litigation : |
Clear |
|
|
|
|
Comments : |
Subject was incorporated in the year 2006. For the financial year 2017, the revenue of the company has increased by 12.24% but has incurred operational loss. The sound financial profile of the company is marked by healthy networth base along with negligible debt level. The company has its share price trading at around INR 416.40 against the Face Value (FV) of INR 01 on BSE as on April 19, 2018. As per the unaudited quarterly financials of December 2017, the company has achieved revenue of INR 194.2 million but has incurred operational loss. However, rating strength is partially offset by continue loss incurred by the company Business is active. Payment seems to be regular. In view of aforesaid, the company can be considered for good business dealings at usual trade terms and conditions. NOTE: As per information gathered from other sources the scheme of arrangement of demerger, sanctioned by the High Court of Gujarat through its order dated March 01, 2007, issued on March 28, 2007 and as per supplementary Trust Deed dated may 18, 2007, the FCCB's holders of Sun Pharma Industries Limited, are entitled for one equity share of INR 1 each of the company for every equity share of INR 5 each of SPIL. |
NOTES :
Any query related to this report can be made
on e-mail : infodept@mirainform.com
while quoting report number, name and date.
EXTERNAL AGENCY RATING
|
Rating Agency Name |
Not Available |
|
Rating |
Not Available |
|
Rating Explanation |
Not Available |
|
Date |
Not Available |
RBI DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available RBI Defaulters’ list.
EPF (Employee Provident Fund) DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available EPF (Employee Provident Fund) Defaulters’ list as of
31-03-2018.
BIFR (Board for Industrial & Financial Reconstruction) LISTING
STATUS
Subject’s name is not listed as a Sick Unit in
the publicly available BIFR (Board for Industrial & Financial
Reconstruction) list as of 21.04.2018
IBBI (Insolvency and Bankruptcy Board of India) LISTING STATUS
Subject’s name is not listed in the publicly
available IBBI (Insolvency and Bankruptcy Board of India) list as of report
date.
INFORMATION DENIED
MANAGEMENT NON-COOPERATIVE (Tel. No.: 91-22-66455645)
(Tel. No.: 91-265-5515500 – Not working)
LOCATIONS
|
Registered Office : |
Akota Road, Akota, Vadodara – 390020, Gujarat, India |
|
Tel. No.: |
91-265-2330815/ 2339257 |
|
Fax No.: |
91-265-2354897 |
|
E-Mail : |
|
|
Website : |
|
|
|
|
|
Mumbai Office / Research Centre 1 : |
17-B, Mahal Industrial Estate, Mahakali Caves Road, Andheri (East), Mumbai - 400 093, Maharashtra, India |
|
Tel. No.: |
91-22-66455645 |
|
Fax No.: |
91-22-66455685 |
|
|
|
|
Research Centre 2 : |
F.P. 27 Part Survey No. 27, C.S. No. 1050, T.P.S No. 24, SPARC, Village Tandalja, District: Vadodara -390 020, Gujarat, India |
|
|
|
|
Research Centre 3 : |
907/4, GIDC, Makarpura, Vadodara - 390 010, Gujarat, India |
DIRECTORS
As on 31.03.2017
|
Name : |
Mr. Dilip Shantilal Shanghvi |
|
Designation : |
Managing Director |
|
Address : |
801, Abhay Building, 8th Floor, Ns 9th Road, Nutan, Laxmi Co-Op Society JVPD Scheme, Vile Parle (West), Mumbai - 400049, Maharashtra, India |
|
Date of Appointment : |
01.03.2012 |
|
DIN No.: |
00005588 |
|
|
|
|
Name : |
Mr. Sudhir Vrundavandas Valia |
|
Designation : |
Director |
|
Address : |
801, Alaap Building, 8th Floor, 173, Sir Balchandra Road, Dadar
(East), Mumbai - 400014, Maharashtra, India |
|
Date of Birth/Age : |
26.07.1956 |
|
Date of Appointment : |
31.07.2014 |
|
DIN No.: |
00005561 |
|
|
|
|
Name : |
Mr. Ferzaan Nariman Engineer |
|
Designation : |
Director |
|
Address : |
11/1, Regency Place Apartment, 7, Richmind Road, Bangalore – 560025, Karnataka, India |
|
Date of Appointment : |
05.05.2017 |
|
DIN No.: |
00025758 |
|
|
|
|
Name : |
Ms. Bhavna Gautam Doshi |
|
Designation : |
Director |
|
Address : |
Flat C - 191, Grand Paradi, August Kranti Road, Kemps Corner, Mumbai – 400036, Maharashtra, India |
|
Date of Birth/Age : |
26.06.1953 |
|
Date of Appointment : |
01.08.2015 |
|
DIN No.: |
00400508 |
|
|
|
|
Name : |
Dr. Rajamannar Thennati |
|
Designation : |
Director |
|
Address : |
G-114, Avishkar Complex, Old Padra Road, Vadodara - 390015, Gujarat, India |
|
Date of Appointment : |
24.04.2014 |
|
DIN No.: |
01415412 |
|
|
|
|
Name : |
Mark Jonathan Simon |
|
Designation : |
Director |
|
Address : |
641, Shunpike Road, Chatham 07928 US |
|
Date of Appointment : |
05.05.2017 |
|
DIN No.: |
07807378 |
|
|
|
|
Name : |
Prof. Dr. Andrea Vasella |
|
Designation : |
Independent Director |
|
|
|
|
Name : |
Prof. Dr. Goverdhan Mehta |
|
Designation : |
Independent Director |
|
|
|
|
Name : |
Mr. S. Mohanchand Dadha |
|
Designation : |
Independent Director |
KEY EXECUTIVES
|
Name : |
Mr. Chetan Manharlal Rajpara |
|
Designation : |
Chief Financial Officer |
|
Address : |
F-702, Sterling Court, Orkay lane, M.I.D.C. Andheri(E) MUMBAI 400093 MH IN |
|
Date of Appointment : |
01.02.2017 |
|
PAN No.: |
ABBPR1748N |
|
|
|
|
Name : |
Mr. Debashis Dey |
|
Designation : |
Company Secretary |
|
Address : |
F-14, Self Help Co-Operative Housing Society, St.Francis Limited, Vile Parle (West), Mumbai – 400056, Maharashtra, India |
|
Date of Appointment : |
06.05.2015 |
|
PAN No.: |
AIGPD5976L |
|
|
|
|
Name : |
Mr. Anil Raghavan |
|
Designation : |
Chief Executive Officer |
MAJOR SHAREHOLDERS / SHAREHOLDING PATTERN
As on 31.03.2018
|
Category of
shareholder |
No. of fully
paid up equity shares held |
Shareholding as a
% of total no. of shares (calculated as per SCRR, 1957)As a % of |
|
|
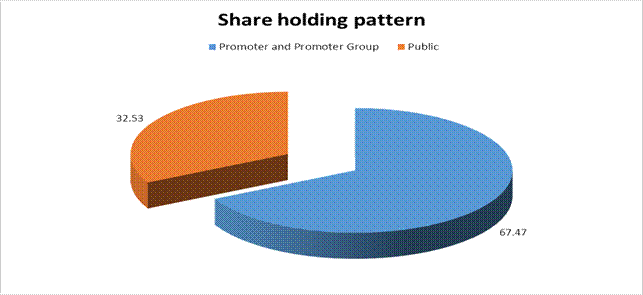
(A) Promoter and Promoter
Group |
169296119 |
67.47 |
|
|
(B) Public |
81640276 |
32.53 |
|
|
Grand
Total |
250936395 |
100.00 |
Statement showing
shareholding pattern of the Promoter and Promoter Group
|
Category of
shareholder |
No. of fully
paid up equity shares held |
Shareholding as
a % of total no. of shares (calculated as per SCRR, 1957)As a % of |
|
|
A1) Indian |
0.00 |
||
|
Individuals/Hindu
undivided Family |
31733020 |
12.65 |
|
|
Dilip.S.Shanghvi |
28102795 |
11.20 |
|
|
Sudhir Vrundavandas Valia |
1833951 |
0.73 |
|
|
Vibha D. Shanghvi |
1063236 |
0.42 |
|
|
Aalok Dilip Shanghvi |
350162 |
0.14 |
|
|
Vidhi Dilip Shanghvi |
344081 |
0.14 |
|
|
Kumud Shantilal Shanghvi |
38795 |
0.02 |
|
|
Any Other (specify) |
137563099 |
54.82 |
|
|
Shanghvi Family &
Friends Benefit Trust |
154922 |
0.06 |
|
|
Quality Investments
Private Limited |
25449040 |
10.14 |
|
|
Viditi Investment Private
Limited |
24691877 |
9.84 |
|
|
Tejaskiran Pharmachem
Industries Private Limited |
24238132 |
9.66 |
|
|
Family Investment Private
Limited |
23668141 |
9.43 |
|
|
Virtuous Finance Private
Limited |
13422122 |
5.35 |
|
|
Virtuous Share
Investments Private Limited |
13353555 |
5.32 |
|
|
Sholapur Organics Private
Limited |
1949601 |
0.78 |
|
|
Jeevanrekha Investrade
Private Limited. |
177842 |
0.07 |
|
|
Package Investrade Private
Limited |
109114 |
0.04 |
|
|
Shanghvi Finance Private
Limited |
34453 |
0.01 |
|
|
Asawari Investment And
Finance Private Limited |
5942 |
0.00 |
|
|
Flamboyawer Finance
Private Limited |
2543 |
0.00 |
|
|
Sanghvi Properties
Private Limited |
1887 |
0.00 |
|
|
Gujarat Sun
Pharmaceutical Industries Private Limited |
1751 |
0.00 |
|
|
Nirmit Exports Private
Limited |
857 |
0.00 |
|
|
Aditya Medisales Limited |
4888126 |
1.95 |
|
|
Raksha S.Valia |
4145231 |
1.65 |
|
|
Unimed Investments
Limited |
1267963 |
0.51 |
|
|
Sub Total A1 |
169296119 |
67.47 |
|
|
A2) Foreign |
0.00 |
||
|
A=A1+A2 |
169296119 |
67.47 |
Statement showing
shareholding pattern of the Public shareholder
|
Category &
Name of the Shareholders |
No. of fully
paid up equity shares held |
Shareholding % calculated
as per SCRR, 1957 As a % of |
|
|
B1) Institutions |
0 |
0.00 |
|
|
Mutual Funds/ |
4912391 |
1.96 |
|
|
Foreign Portfolio
Investors |
17515455 |
6.98 |
|
|
Seafarer Overseas Growth
And Income Fund |
8625000 |
3.44 |
|
|
Financial Institutions/ Banks |
125679 |
0.05 |
|
|
Sub Total B1 |
22553525 |
8.99 |
|
|
B2) Central Government/
State Government(s)/ President of India |
0 |
0.00 |
|
|
B3) Non-Institutions |
0 |
0.00 |
|
|
Individual share capital
upto INR 0.200 Million |
24124620 |
9.61 |
|
|
Individual share capital
in excess of INR 0.200 Million |
11176664 |
4.45 |
|
|
Any Other (specify) |
23785467 |
9.48 |
|
|
Trusts |
10319170 |
4.11 |
|
|
Sun Pharmaceutical
Industries Key Employees Benefit Trust |
10317377 |
4.11 |
|
|
HUF |
1491895 |
0.59 |
|
|
Non-Resident Indian (NRI) |
385641 |
0.15 |
|
|
Others |
611714 |
0.24 |
|
|
Overseas Corporate Bodies |
9600 |
0.00 |
|
|
Clearing Members |
221935 |
0.09 |
|
|
Bodies Corporate |
10745512 |
4.28 |
|
|
Lakshdeep Investments
& Finance (P) Limited |
4080142 |
1.63 |
|
|
Sub Total B3 |
59086751 |
23.55 |
|
|
B=B1+B2+B3 |
81640276 |
32.53 |
BUSINESS DETAILS
|
Line of Business : |
Subject is engaged in Research and experimental development on natural
sciences and engineering. (Registered Activity) |
||||
|
|
|
||||
|
Products/ Services : |
|
||||
|
|
|
||||
|
Brand Names : |
Not Available |
||||
|
|
|
||||
|
Agencies Held : |
Not Available |
||||
|
|
|
||||
|
Exports : |
Not Divulged |
||||
|
|
|
||||
|
Imports : |
Not Divulged |
||||
|
|
|
||||
|
Terms : |
Not Divulged |
PRODUCTION STATUS NOT AVAILABLE
GENERAL INFORMATION
|
Suppliers : |
|
||||||||||||||||||||||
|
|
|
||||||||||||||||||||||
|
Customers : |
|
||||||||||||||||||||||
|
|
|
||||||||||||||||||||||
|
No. of Employees : |
398 (Approximately) |
||||||||||||||||||||||
|
|
|
||||||||||||||||||||||
|
Bankers : |
|
||||||||||||||||||||||
|
|
|
||||||||||||||||||||||
|
Facilities : |
|
|
Auditors : |
|
|
Name : |
Deloitte Haskins and Sells LLP Chartered Accountants |
|
Address : |
Indiabulls Finance Centre, Tower 3, 27th - 32nd Floor,
Senapati Bapat Marg, Elphinstone Road (West), Mumbai - 400013, Maharashtra,
India |
|
Tel. No.: |
91-022-61854000 |
|
Fax No.: |
91-22-61854501/ 4601 |
|
|
|
|
Memberships : |
Not Available |
|
|
|
|
Collaborators : |
Not Available |
|
|
|
|
Enterprise under
significant Influence of Key Management Personnel (with whom transactions are
entered) |
|
CAPITAL STRUCTURE
As on 05.08.2017
Authorised Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
266500000 |
Equity Shares |
INR 1/- each |
INR 266.500 Million |
|
|
|
|
|
Issued, Subscribed & Paid-up Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
250936395 |
Equity Shares |
INR 1/- each |
INR 250.936 Million |
|
|
|
|
|
As on 31.03.2017
Authorised Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
266500000 |
Equity Shares |
INR 1/- each |
INR 266.500 Million |
|
|
|
|
|
Issued, Subscribed & Paid-up Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
236687354 |
Equity Shares |
INR 1/- each |
INR 236.697
Million |
|
|
Add : Annulment of forfeited shares |
|
INR 0.003
Million |
|
|
Add : Shares issued on Right basis |
|
INR 10.204
Million |
|
|
Total |
|
INR 246.904 Million |
FINANCIAL DATA
[all figures are
in INR Million]
ABRIDGED
BALANCE SHEET
|
SOURCES OF FUNDS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
I.
EQUITY
AND LIABILITIES |
|
|
|
|
(1)Shareholders' Funds |
|
|
|
|
(a) Share Capital |
246.904 |
236.697 |
236.683 |
|
(b) Reserves & Surplus |
1349.574 |
62.330 |
761.303 |
|
(c) Money
received against share warrants |
0.000 |
0.000 |
0.000 |
|
|
|
|
|
|
(2) Share Application money pending
allotment |
0.000 |
18.997 |
0.000 |
|
Total
Shareholders’ Funds (1) + (2) |
1596.478 |
318.024 |
997.986 |
|
|
|
|
|
|
(3)
Non-Current Liabilities |
|
|
|
|
(a) Long-term borrowings |
21.809 |
27.261 |
32.714 |
|
(b) Deferred tax liabilities (Net) |
0.000 |
0.000 |
0.786 |
|
(c) Other long term
liabilities |
0.000 |
0.000 |
0.000 |
|
(d) Long-term
provisions |
41.819 |
32.960 |
23.733 |
|
Total Non-current
Liabilities (3) |
63.628 |
60.221 |
57.233 |
|
|
|
|
|
|
(4)
Current Liabilities |
|
|
|
|
(a) Short
term borrowings |
4.110 |
524.762 |
3.955 |
|
(b) Trade
payables |
944.529 |
581.511 |
270.107 |
|
(c) Other
current liabilities |
98.134 |
120.855 |
89.751 |
|
(d) Short-term
provisions |
33.494 |
21.228 |
7.665 |
|
Total Current
Liabilities (4) |
1080.267 |
1248.356 |
371.478 |
|
|
|
|
|
|
TOTAL |
2740.373 |
1626.601 |
1426.697 |
|
|
|
|
|
|
II.
ASSETS |
|
|
|
|
(1)
Non-current assets |
|
|
|
|
(a) Fixed
Assets |
|
|
|
|
(i)
Tangible assets |
691.587 |
699.564 |
667.911 |
|
(ii)
Intangible Assets |
0.000 |
0.000 |
0.000 |
|
(iii)
Capital work-in-progress |
14.668 |
8.730 |
0.241 |
|
(iv)
Intangible assets under development |
0.000 |
0.000 |
0.000 |
|
(b) Non-current Investments |
0.000 |
0.000 |
0.000 |
|
(c) Deferred tax assets (net) |
0.000 |
0.000 |
0.000 |
|
(d)
Long-term Loan and Advances |
436.168 |
240.365 |
126.230 |
|
(e) Other
Non-current assets |
12.147 |
12.032 |
2.968 |
|
Total Non-Current
Assets |
1154.570 |
960.691 |
797.350 |
|
|
|
|
|
|
(2)
Current assets |
|
|
|
|
(a)
Current investments |
95.539 |
0.000 |
0.000 |
|
(b)
Inventories |
0.000 |
0.000 |
0.000 |
|
(c) Trade
receivables |
389.002 |
91.596 |
198.868 |
|
(d) Cash
and cash equivalents |
703.063 |
127.485 |
173.441 |
|
(e)
Short-term loans and advances |
388.347 |
440.910 |
254.252 |
|
(f) Other
current assets |
9.852 |
5.919 |
2.786 |
|
Total
Current Assets |
1585.803 |
665.910 |
629.347 |
|
|
|
|
|
|
TOTAL |
2740.373 |
1626.601 |
1426.697 |
PROFIT
& LOSS ACCOUNT
|
|
PARTICULARS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
|
SALES |
|
|
|
|
|
Income |
1810.248 |
1612.824 |
1557.375 |
|
|
Other Income |
136.257 |
29.367 |
30.340 |
|
|
TOTAL |
1946.505 |
1642.191 |
1587.715 |
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
Cost of Materials
Consumed |
251.835 |
188.681 |
100.965 |
|
|
Purchases of
Stock-in-Trade |
0.000 |
0.000 |
0.000 |
|
|
Employees benefits
expense |
844.319 |
566.032 |
457.927 |
|
|
Clinical Trials and Professional
Charges |
1458.556 |
1195.450 |
1110.468 |
|
|
Other expenses |
493.873 |
294.593 |
238.976 |
|
|
TOTAL |
3048.583 |
2244.756 |
1908.336 |
|
|
|
|
|
|
|
|
PROFIT/ (LOSS) BEFORE INTEREST, TAX, DEPRECIATION AND
AMORTISATION |
(1102.078) |
(602.565) |
(320.621) |
|
|
|
|
|
|
|
Less |
FINANCIAL EXPENSES |
18.198 |
21.569 |
2.183 |
|
|
|
|
|
|
|
|
PROFIT / (LOSS) BEFORE
TAX, DEPRECIATION AND AMORTISATION |
(1120.276) |
(624.134) |
(322.804) |
|
|
|
|
|
|
|
Less/ Add |
DEPRECIATION/
AMORTISATION |
82.397 |
75.786 |
72.396 |
|
|
|
|
|
|
|
|
PROFIT/ (LOSS) BEFORE TAX |
(1202.673) |
(699.920) |
(395.200) |
|
|
|
|
|
|
|
Less |
TAX |
0.000 |
0.000 |
0.000 |
|
|
|
|
|
|
|
|
PROFIT/ (LOSS) AFTER TAX
|
(1202.673) |
(699.920) |
(395.200) |
|
|
|
|
|
|
|
Add |
PREVIOUS YEARS’ BALANCE
BROUGHT FORWARD |
(2229.793) |
(1529.873) |
(1134.673) |
|
|
|
|
|
|
|
|
Balance Carried to the
B/S |
(3432.466) |
(2229.793) |
(1529.873) |
|
|
|
|
|
|
|
|
EARNINGS IN FOREIGN
CURRENCY |
1422.535 |
1185.291 |
1247.408 |
|
|
|
|
|
|
|
|
Earnings / (Loss) Per
Share (INR) |
(4.87) |
(2.96) |
(1.67) |
CURRENT MATURITIES OF LONG TERM DEBT DETAILS
|
Particulars |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Current Maturities of Long term debt |
5.452 |
5.452 |
5.452 |
|
Cash generated from operations |
1073.153 |
303.392 |
385.297 |
|
Net cash flow from operating activity |
1280.378 |
442.116 |
411.682 |
QUARTERLY RESULTS
|
Particulars |
30.06.2017 |
30.09.2017 |
31.12.2017 |
|
Unaudited |
1st Quarter |
2nd Quarter |
3rd Quarter |
|
Net Sales |
233.900 |
153.600 |
194.200 |
|
Total Expenditure |
1013.300 |
768.200 |
775.900 |
|
PBIDT (Excl OI) |
(779.400) |
(614.600) |
(581.700) |
|
Other Income |
12.300 |
9.900 |
21.400 |
|
Operating Profit |
(767.100) |
(604.700) |
(560.300) |
|
Interest |
0.700 |
0.200 |
0.200 |
|
Exceptional Items |
NA |
NA |
NA |
|
PBDT |
(767.800) |
(604.900) |
(560.500) |
|
Depreciation |
20.600 |
20.800 |
20.200 |
|
Profit Before Tax |
(788.400) |
(625.700) |
(580.700) |
|
Tax |
NA |
NA |
NA |
|
Provisions and contingencies |
NA |
NA |
NA |
|
Profit After Tax |
(788.400) |
(625.700) |
(580.700) |
|
Extraordinary Items |
NA |
NA |
NA |
|
Prior Period Expenses |
NA |
NA |
NA |
|
Other Adjustments |
NA |
NA |
NA |
|
Net Profit |
(788.400) |
(625.700) |
(580.700) |
KEY
RATIOS
EFFICIENCY RATIOS
|
PARTICULARS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Average Collection Days (Sundry
Debtors / Income * 365 Days) |
78.43 |
20.73 |
46.61 |
|
|
|
|
|
|
Account Receivables Turnover ( Income / Sundry Debtors) |
4.65 |
17.61 |
7.83 |
|
|
|
|
|
|
Average Payment Days (Sundry Creditors / Purchases * 365 Days) |
1368.96 |
1124.92 |
976.47 |
|
|
|
|
|
|
Inventory Turnover (Operating Income / Inventories) |
0.00 |
0.00 |
0.00 |
|
|
|
|
|
|
Asset Turnover (Operating Income / Net Fixed Assets) |
(1.56) |
(0.85) |
(0.48) |
LEVERAGE RATIOS
|
PARTICULARS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Debt Ratio ((Borrowing
+ Current Liabilities) / Total Assets) |
0.40 |
0.79 |
0.29 |
|
|
|
|
|
|
Debt Equity Ratio (Total Liability / Networth) |
0.02 |
1.75 |
0.04 |
|
|
|
|
|
|
Current Liabilities to Networth (Current Liabilities / Net Worth) |
0.68 |
3.93 |
0.37 |
|
|
|
|
|
|
Fixed Assets to Networth (Net Fixed Assets / Networth) |
0.44 |
2.23 |
0.67 |
|
|
|
|
|
|
Interest Coverage Ratio (PBIT / Financial Charges) |
(60.56) |
(27.94) |
(146.87) |
PROFITABILITY RATIOS
|
PARTICULARS |
|
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Net Profit Margin [(PAT / Sales)
* 100] |
% |
(66.44) |
(43.40) |
(25.38) |
|
|
|
|
|
|
|
Return on Total Assets ((PAT / Total Assets) * 100) |
% |
(43.89) |
(43.03) |
(27.70) |
|
|
|
|
|
|
|
Return on Investment (ROI) ((PAT / Networth) * 100) |
% |
(75.33) |
(220.08) |
(39.60) |
SOLVENCY RATIOS
|
PARTICULARS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Current Ratio (Current
Assets / Current Liabilities) |
1.47 |
0.53 |
1.69 |
|
|
|
|
|
|
Quick Ratio ((Current Assets – Inventories) / Current
Liabilities) |
1.47 |
0.53 |
1.69 |
|
|
|
|
|
|
G-Score Ratio Financial (Networth / Total Assets) |
0.58 |
0.20 |
0.70 |
|
|
|
|
|
|
G-Score Ratio Debt (Debts / Equity Capital) |
0.13 |
2.36 |
0.18 |
|
|
|
|
|
|
G-Score Ratio Liquidity (Total Current Assets / Total Current Liabilities) |
1.47 |
0.53 |
1.69 |
Total
Liability = Short-term Debt + Long-term Debt + Current Maturities of Long-term
debts
STOCK
PRICES
|
Face Value |
INR 1.00/- |
|
Market Value |
INR 416.40/- |
FINANCIAL ANALYSIS
[all figures are
in INR Million]
DEBT EQUITY RATIO
|
Particular |
31.03.2015 |
31.03.2016 |
31.03.2017 |
|
|
INR In Million |
INR In Million |
INR In Million |
|
Share Capital |
236.683 |
236.697 |
246.904 |
|
Reserves & Surplus |
761.303 |
62.330 |
1349.574 |
|
Money received against
share warrants |
0.000 |
0.000 |
0.000 |
|
Share Application money
pending allotment |
0.000 |
18.997 |
0.000 |
|
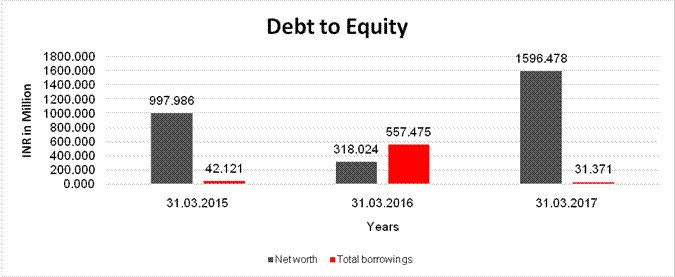
Net worth |
997.986 |
318.024 |
1596.478 |
|
|
|
|
|
|
long-term borrowings |
32.714 |
27.261 |
21.809 |
|
Short term borrowings |
3.955 |
524.762 |
4.110 |
|
Current Maturities of
Long term debt |
5.452 |
5.452 |
5.452 |
|
Total borrowings |
42.121 |
557.475 |
31.371 |
|
Debt/Equity ratio |
0.042 |
1.753 |
0.020 |
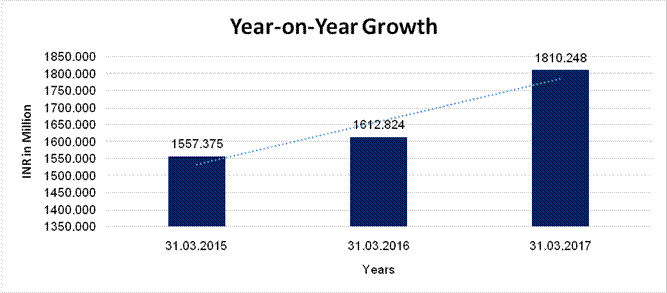
YEAR-ON-YEAR GROWTH
|
Year on Year Growth |
31.03.2015 |
31.03.2016 |
31.03.2017 |
|
|
INR In Million |
INR In Million |
INR In Million |
|
Sales |
1557.375 |
1612.824 |
1810.248 |
|
|
|
3.560 |
12.241 |
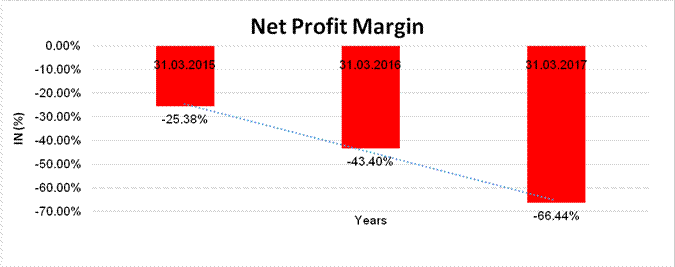
NET PROFIT MARGIN
|
Net Profit Margin |
31.03.2015 |
31.03.2016 |
31.03.2017 |
|
|
INR In Million |
INR In Million |
INR In Million |
|
Sales |
1557.375 |
1612.824 |
1810.248 |
|
Profit/(Loss) |
(395.200) |
(699.920) |
(1202.673) |
|
|
(25.38%) |
(43.40%) |
(66.44%) |
LOCAL AGENCY FURTHER INFORMATION
|
Sr. No. |
Check list by
info agents |
Available in
Report (Yes/No) |
|
1 |
Year of establishment |
Yes |
|
2 |
Constitution of the entity -Incorporation
details |
Yes |
|
3 |
Locality of the entity |
Yes |
|
4 |
Premises details |
No |
|
5 |
Buyer visit details |
-- |
|
6 |
Contact numbers |
Yes |
|
7 |
Name of the person contacted |
No |
|
8 |
Designation of contact person |
No |
|
9 |
Promoter’s background |
Yes |
|
10 |
Date of Birth of Proprietor / Partners /
Directors |
No |
|
11 |
Pan Card No. of Proprietor / Partners |
No |
|
12 |
Voter Id Card No. of Proprietor / Partners |
No |
|
13 |
Type of business |
Yes |
|
14 |
Line of Business |
Yes |
|
15 |
Export/import details (if applicable) |
No |
|
16 |
No. of employees |
Yes |
|
17 |
Details of sister concerns |
Yes |
|
18 |
Major suppliers |
No |
|
19 |
Major customers |
No |
|
20 |
Banking Details |
Yes |
|
21 |
Banking facility details |
Yes |
|
22 |
Conduct of the banking account |
-- |
|
23 |
Financials, if provided |
Yes |
|
24 |
Capital in the business |
Yes |
|
25 |
Last accounts filed at ROC, if applicable |
Yes |
|
26 |
Turnover of firm for last three years |
Yes |
|
27 |
Reasons for variation <> 20% |
-- |
|
28 |
Estimation for coming financial year |
No |
|
29 |
Profitability for last three years |
Yes |
|
30 |
Major shareholders, if available |
Yes |
|
31 |
External Agency Rating, if available |
No |
|
32 |
Litigations that the firm/promoter
involved in |
-- |
|
33 |
Market information |
-- |
|
34 |
Payments terms |
No |
|
35 |
Negative Reporting by Auditors in the
Annual Report |
No |
MANAGEMENT DISCUSSION
AND ANALYSIS
INDUSTRY OUTLOOK
The year 2017 is expected to be an exciting yet challenging one for the worldwide pharmaceutical industry.
Even as aging population and proliferating chronic diseases drive increased provider and patient demands for more and better therapies, reform-driven drug price controls and government cost-containment measures continue to challenge the life sciences sector. Besides, the shift from volume to value developments of new therapies and proliferation of orphan drug approvals, may spawn debates regarding prices and access to medicine. With public and private payers making increased use of value-based pricing and real-world evidence to provide clinical and commercial insights into the value of products, life sciences companies should balance the pursuit of clinical innovation with improving operational and cost efficiencies.
The industry would be required to win back public and political trust relating to drug pricing, exploitation of market monopolies, and promotional practices. The focus of the industry will be required to effectively shift towards patient-centricity.
OPERATIONS REVIEW
The Company earned net revenues of INR 1946.500 Million. Their source of revenue largely includes royalty and milestone income for the products previously licensed to partner(s).
They did not receive approval of XelprosTM and ElepsiaTM XR due to GMP compliance issues at their partner’s manufacturing site.
Their milestone and royalty income linked to approval and commercialisation of these products is deferred as a result of delay in approvals.
Since their products are not yet commercialized in major markets like US, they do not have a steady stream of royalty income. Their income therefore is irregular and depends on the milestone payments that they might receive at the time of licensing. They expect this trend to continue in the next year as well.
The Company reported net loss of INR 1202.700 Million. The major expenses were attributed to the conduct of clinical trials associated with their R&D programmes and towards employee costs. During the year, they initiated several clinical trials and also accelerated patient enrolment in ongoing clinical trials. This resulted in significant increase in clinical trial expenses compared to previous year.
In the year ahead, they anticipate increase in their clinical trials spending and investment in building critical developmental capabilities. They have initiated pivotal and Proof of Concept clinical trials for several of their programmes. They are also building their capabilities and increasing their efforts on hiring key resources to assist us in taking the organisation to the next level.
Drug discovery research is a high risk business. The Company has identified potential risks associated with the business and has developed a risk management plan to insulate the organisation from the potential impact of such risks.
OUTLOOK
While they progress through the challenging times of uncertainties for approval of 2 products filed with USFDA, their other clinical programmes are rapidly advancing, resulting in a visible increase in cash burn. The silver line is that they have completed patient enrolment in pivotal studies of Baclofen GRS and DPI and have initiated a pivotal study for TaclantisTM. If the outcome of these clinical trials are positive, they would have additional cash flow opportunities in the next 12 to 24 months.
They are aggressively replenishing their early stage pipeline as some of their promising early stage assets would transition to the clinical stage. Their early stage portfolio is now a balanced mix of delivery system innovations with medium term timelines for development and new chemical entities addressing the next generation of opportunities in known and validated biological pathways and a small set of opportunities addressing novel targets. Oncology, Neurodegeneration, Inflammation and Pain remain their therapeutic areas of focus.
UNSECURED LOAN
|
PARTICULARS |
31.03.2017 (INR
in Million) |
31.03.2016 (INR
in Million) |
|
Long-term
Borrowings |
|
|
|
Term Loan from Department of Science and Technology (DST), Government of India under the "Drug and Pharmaceutical Research Program" [Repayable in 5 (Previous Year 6) Annual Installments of INR 5.452 Million each. Last instalment is due on 1st September, 2021]. For the current maturities of long term debt. |
21.809 |
27.261 |
|
Short-term
borrowings |
|
|
|
Loans Repayable on Demand |
|
|
|
From Banks |
|
|
|
Bank Overdraft Facility |
4.110 |
22.011 |
|
From Others |
0.000 |
490.000 |
|
Total |
25.919 |
539.272 |
INDEX
OF CHARGES
No Charges Exists for Company
UNAUDITED FINANCIAL
RESULTS FOR THE QUARTER AND NINE MONTHS ENDED 31.12.2017
(INR In Million)
|
|
PARTICULARS |
Quarter Ended |
Nine Months Ended |
|
|
|
31.12.2017 (Unaudited) |
30.09.2017 (Unaudited) |
31.12.2017 (Unaudited) |
|
|
|
Revenue from Operations |
194.200 |
153.600 |
581.700 |
|
|
Other Income |
21.400 |
9.900 |
43.600 |
|
|
Total Income |
215.600 |
163.500 |
625.300 |
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
Cost of Materials
Consumed |
52.400 |
9.700 |
216.000 |
|
|
Employees benefits
expense |
246.800 |
265.200 |
760.000 |
|
|
Clinical Trials and Professional charges |
380.600 |
397.100 |
1253.500 |
|
|
Finance Costs |
0.200 |
0.200 |
1.100 |
|
|
Depreciation Expenses |
20.200 |
20.800 |
61.600 |
|
|
Other expenses |
96.100 |
96.200 |
327.900 |
|
|
Total Expenses |
796.300 |
789.200 |
2620.100 |
|
|
|
|
|
|
|
|
Profit/
(Loss) before tax |
(580.700) |
(625.700) |
(1994.800) |
|
|
Tax Expenses |
0.000 |
0.000 |
0.000 |
|
|
Profit/
(Loss) for the period |
(580.700) |
(625.700) |
(1994.800) |
|
|
Other
Comprehensive Income (OCI) (net of tax) |
|
|
|
|
|
Items that will not be reclassified to profit and
loss (Net actuarial loss on employee defined benefit obligation) |
(1.300) |
(1.300) |
(3.900) |
|
|
Total
Comprehensive Income |
(582.000) |
(627.000) |
(1998.700) |
|
|
Paid-up Equity Share Capital – Face Value (INR 1 Each) |
250.900 |
246.900 |
250.900 |
|
|
Earnings
per share of INR 1 each in (Basic and Diluted) |
(2.34) |
(2.53) |
(8.07) |
Note:
- The above unaudited financial results are as per the Regulation 33 of the SEBI (Listing Obligations and Disclosure Requirements) Regulations, 2015 and have been taken on record by the Board of Directors at its meeting held on January 29, 2018 after being reviewed by the Audit Committee and have been subjected to a limited review by Statutory Auditors of the Company.
- The Company adopted Indian Accounting Standard (`Ind AS`) from April 1, 2017 and accordingly these financial results have been prepared in accordance with the recognition and measurement principles laid down in the Ind AS 34 Interim Financial Reporting prescribed under Section 133 of the Companies Act, 2013 read with the relevant rules issued thereunder. The date of transition is April 1, 2016. The impact of transition has been accounted for in opening reserves and the comparative period results have been restated accordingly. The opening balance sheet as at April 1, 2016 and the results for the subsequent periods would get finalized along with the annual financial statements for the year ended March 31, 2018.
- The Company has only one reportable business segment namely 'Pharmaceutical Research and Development'.
- The Securities Allotment Committee of the Company in its meeting held on 15th December 2017, has allotted 4,040,404 fully paid up equity shares of FV INR 1 each of the Company pursuant to conversion of equivalent number of warrants held by the respective warrant holders upon exercise of ‘conversion option’ by the respective concerned warrant holders.
- Consequent to the above, the paid-up capital of the Company has increased from INR 246895991 divided into 246895991 equity shares of FV INR 1 each to INR 250936395 divided into 250936395 equity shares of FV INR 1 each, during the quarter. As on 31st December 2017, the Company had 11111111 outstanding warrants, which are yet to be converted into equity shares of the Company.
CONTINGENT
LIABILITIES:
(INR in million)
|
PARTICULARS |
31.03.2017 |
31.03.2016 |
|
Guarantees given by the bankers against custom licenses |
16.871 |
18.799 |
|
Disputed demands by Income Tax Authorities * |
509.906 |
181.200 |
|
Disputed demands by Sales Tax Authorities * |
-- |
2.984 |
|
* Amount paid under protest is classified under Short Term Loans & Advances |
246.200 |
91.359 |
Note: Future cash outflows in respect of the above matters are determinable only on receipt of judgements/decisions pending at various forums/authorities. The Company does not expect the outcome of the matters stated above to have material adverse impact on the Company’s financial condition, results of operation or cash flows. The Company doesn’t envisage any likely reimbursement in respect of the above.
FIXED ASSETS
·
Buildings
·
Plant and Equipment
·
Furniture and Fixtures
·
Vehicles
WEBSITE DETAILS
NEWS/ PRESS RELEASES
SUN PHARMA RESEARCH
ARM SPARC GETS INCOME TAX DEMAND NOTICE FOR INR 328.700 MILLION
24.02.2017
Sun Pharma Advanced Research Company says it is contesting the demand, which is for the assessment year 2013-14
Sun Pharma Advanced Research Company, or SPARC, was demerged from Sun Pharma as a pharma research and drug discovery company in 2007.
New Delhi: Sun Pharma Advanced Research Company (SPARC) on Friday said it has received a demand notice from income tax authorities for a sum of INR 328.700 Million.
The company is contesting the demand, which is for the assessment year 2013-14, SPARC said in a regulatory filing.
“The company is in receipt of a ‘Demand Notice’ under the Section 156 of the Income Tax, 1961, demanding the payment by the company of a sum of INR 328.700 Million for the assessment year 2013-14,” it said.
The company is contesting the demand and will file an appropriate appeal against this under the applicable provisions of the Income Tax Act, 1961, within the stipulated period, it added.
SPARC was demerged from Sun Pharma as a pharma research and drug discovery company in 2007.
CMT REPORT (Corruption, Money Laundering & Terrorism]
The Public Notice information has been collected from various sources
including but not limited to: The Courts,
1] INFORMATION ON
DESIGNATED PARTY
No exist designating subject or any of its beneficial owners,
controlling shareholders or senior officers as terrorist or terrorist
organization or whom notice had been received that all financial transactions
involving their assets have been blocked or convicted, found guilty or against
whom a judgement or order had been entered in a proceedings for violating
money-laundering, anti-corruption or bribery or international economic or
anti-terrorism sanction laws or whose assets were seized, blocked, frozen or
ordered forfeited for violation of money laundering or international
anti-terrorism laws.
2] Court Declaration :
No records exist to suggest that subject is
or was the subject of any formal or informal allegations, prosecutions or other
official proceeding for making any prohibited payments or other improper
payments to government officials for engaging in prohibited transactions or
with designated parties.
3] Asset Declaration :
No records exist to suggest that the property or assets of the subject
are derived from criminal conduct or a prohibited transaction.
4] Record on Financial
Crime :
Charges or conviction
registered against subject: None
5] Records on Violation of
Anti-Corruption Laws :
Charges or
investigation registered against subject: None
6] Records on Int’l
Anti-Money Laundering Laws/Standards :
Charges or
investigation registered against subject: None
7] Criminal Records
No available
information exist that suggest that subject or any of its principals have been
formally charged or convicted by a competent governmental authority for any
financial crime or under any formal investigation by a competent government
authority for any violation of anti-corruption laws or international anti-money
laundering laws or standard.
8] Affiliation with
Government :
No record
exists to suggest that any director or indirect owners, controlling
shareholders, director, officer or employee of the company is a government
official or a family member or close business associate of a Government
official.
9] Compensation Package :
Our market
survey revealed that the amount of compensation sought by the subject is fair
and reasonable and comparable to compensation paid to others for similar
services.
10] Press Report :
No press reports / filings exists on
the subject.
CORPORATE GOVERNANCE
MIRA INFORM as part of its Due Diligence do provide comments on
Corporate Governance to identify management and governance. These factors often
have been predictive and in some cases have created vulnerabilities to credit
deterioration.
Our Governance Assessment focuses principally on the interactions
between a company’s management, its Board of Directors, Shareholders and other
financial stakeholders.
CONTRAVENTION
Subject is not known to have contravened any existing local laws,
regulations or policies that prohibit, restrict or otherwise affect the terms and
conditions that could be included in the agreement with the subject.
FOREIGN EXCHANGE RATES
|
Currency |
Unit
|
INR |
|
US Dollar |
1 |
INR 66.01 |
|
|
1 |
INR 92.72 |
|
Euro |
1 |
INR 81.45 |
INFORMATION DETAILS
|
Information Gathered
by : |
ARC |
|
|
|
|
Analysis Done by
: |
VAR |
|
|
|
|
Report Prepared
by : |
SUD |
SCORE FACTORS
|
DEMERIT POINTS |
|
|
|
--BANK CHARGES |
YES/NO |
YES |
|
--LITIGATION |
YES/NO |
NO |
|
--OTHER ADVERSE INFORMATION |
YES/NO |
NO |
|
MERIT POINTS |
|
|
|
--SOLE DISTRIBUTORSHIP |
YES/NO |
NO |
|
--EXPORT ACTIVITIES |
YES/NO |
NO |
|
--AFFILIATION |
YES/NO |
YES |
|
--LISTED |
YES/NO |
YES |
|
--OTHER MERIT FACTORS |
YES/NO |
YES |
RATING EXPLANATIONS
|
Credit Rating |
Explanation |
Rating Comments |
|
A++ |
Minimum Risk |
Business dealings permissible with minimum
risk of default |
|
A+ |
Low Risk |
Business dealings permissible with low
risk of default |
|
A |
Acceptable Risk |
Business dealings permissible with moderate
risk of default |
|
B |
Medium Risk |
Business dealings permissible on a regular
monitoring basis |
|
C |
Medium High Risk |
Business dealings permissible preferably
on secured basis |
|
D |
High Risk |
Business dealing not recommended or on secured
terms only |
|
NB |
New Business |
No recommendation can be done due to
business in infancy stage |
|
NT |
No Trace |
No recommendation can be done as the
business is not traceable |
NB is stated where there is insufficient information to facilitate rating. However, it is not to be considered as unfavourable.
This score serves as a reference to assess
SC’s credit risk and to set the amount of credit to be extended. It is
calculated from a composite of weighted scores obtained from each of the major
sections of this report. The assessed factors are as follows:
·
Financial
condition covering various ratios
·
Company
background and operations size
·
Promoters
/ Management background
·
Payment
record
·
Litigation
against the subject
·
Industry
scenario / competitor analysis
·
Supplier
/ Customer / Banker review (wherever available)
This report is issued at
your request without any risk and responsibility on the part of MIRA INFORM
PRIVATE LIMITED (MIPL) or its officials.

