|
|
|
MIRA INFORM REPORT
|
Report No. : |
506245 |
|
Report Date : |
28.04.2018 |
IDENTIFICATION DETAILS
|
Name : |
ASTRAZENECA PHARMA INDIA LIMITED |
|
|
|
|
Registered
Office : |
Block N1, 12th Floor, Manyata Embassy Business Park, Rachenahalli, Outer Ring Road, Bangalore-560045, Karnataka |
|
Tel. No.: |
91-80-67748000 |
|
|
|
|
Country : |
India |
|
|
|
|
Financials (as
on) : |
31.03.2017 |
|
|
|
|
Date of
Incorporation : |
11.07.1979 |
|
|
|
|
Com. Reg. No.: |
08-003563 |
|
|
|
|
Capital
Investment / Paid-up Capital : |
INR 50.000 Million |
|
|
|
|
CIN No.: [Company Identification
No.] |
L24231KA1979PLC003563 |
|
|
|
|
IEC No.: [Import-Export Code No.] |
Not Divulged |
|
|
|
|
GSTN : [Goods & Service Tax
Registration No.] |
Not Divulged |
|
|
|
|
TIN No.: |
Not Divulged |
|
|
|
|
TAN No.: [Tax Deduction &
Collection Account No.] |
Not Available |
|
|
|
|
PAN No.: [Permanent Account No.] |
Not Divulged |
|
|
|
|
Legal Form : |
A Public Limited Liability Company. The Company’s Shares are Listed on
the Stock Exchanges. |
|
|
|
|
Line of Business
: |
Manufacturing, Distribution and Marketing of
Pharmaceutical Products. [Registered Activity] |
|
|
|
|
No. of Employees
: |
1141 (Approximately) |
RATING & COMMENTS
(Mira Inform has adopted New Rating mechanism w.e.f. 23rd
January 2017)
|
MIRA’s Rating : |
A++ |
|
Credit Rating |
Explanation |
Rating Comments |
|
A++ |
Minimum Risk |
Business dealings permissible with minimum
risk of default |
|
Maximum Credit Limit : |
USD 5155900 |
|
|
|
|
Status : |
Excellent |
|
|
|
|
Payment Behaviour : |
Regular |
|
|
|
|
Litigation : |
Clear |
|
|
|
|
Comments : |
Subject is subsidiary of Astrazeneca Pharmaceuticals AB, Sweden. The company was incorporated in the year 1979 and is based in Bangalore, Karnataka. It is an established company having excellent track record. For the financial year ended 2017, revenue of the company has marginally decline. However, it has witnessed a growth in its profit and has achieved fair profit margin at 4.53% (approx.). The company possesses strong financial profile marked by healthy net worth base along with debt free balance sheet and favourable gap between trade payables to its trade receivables.
|
NOTES :
Any query related to this report can be made
on e-mail : infodept@mirainform.com
while quoting report number, name and date.
ECGC Country Risk Classification List
|
Country Name |
Previous Rating (30.09.2017) |
Current Rating (31.12.2017) |
|
India |
A1 |
A1 |
|
Risk Category |
ECGC
Classification |
|
Insignificant |
A1 |
|
Low Risk |
A2 |
|
Moderately Low Risk |
B1 |
|
Moderate Risk |
B2 |
|
Moderately High Risk |
C1 |
|
High Risk |
C2 |
|
Very High Risk |
D |
EXTERNAL AGENCY RATING
NOT AVAILABLE
RBI DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available RBI Defaulters’ list.
EPF (Employee Provident Fund) DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available EPF (Employee Provident Fund) Defaulters’ list as of
31-03-2016.
BIFR (Board for Industrial & Financial Reconstruction) LISTING
STATUS
Subject’s name is not listed as a Sick Unit in
the publicly available BIFR (Board for Industrial & Financial
Reconstruction) list as of 28.04.2018.
IBBI (Insolvency and Bankruptcy Board of India) LISTING STATUS
Subject’s name is not listed in the publicly
available IBBI (Insolvency and Bankruptcy Board of India) list as of report
date.
INFORMATION DECLINED
MANAGEMENT NON-COOPERATIVE
[Contact No: 91-80-23622020]
LOCATIONS
|
Registered/ Corporate Office : |
Block N1, 12th Floor, Manyata Embassy Business Park, Rachenahalli, Outer Ring Road, Bangalore-560045, Karnataka, India |
|
Tel. No.: |
91-80-67748000/ 23622020 |
|
Fax No.: |
91-80-67748557 |
|
E-Mail : |
|
|
Website : |
|
|
|
|
|
Factory : |
12th Mile on Bellary Road, Kattigenahalli Village, Yelahanka, Bangalore-560063, Karnataka, India |
|
Tel. No.: |
91-80-67749000 |
|
Fax No.: |
91-80-28462208/ 67749628 |
|
|
|
|
Sales Outlets : |
Located at: · Ahmedabad · Bangalore* · Chennai · Cuttack · Dehradun · Delhi* · Guwahati · Hyderabad · Indore · Jaipur, Kochi · Kolkata · Lucknow · Mumbai · Patna, Ranchi · Chandigarh, · Panchkula · Zirakhpur Note: *Company outlets
(Bangalore and Delhi) |
DIRECTORS
AS ON: 31.03.2017
|
Name : |
Mr. Narayan Keelveedhi Seshadri |
|
Designation : |
Director |
|
Address : |
Flat No.10, 7th Floor, Skylark Chs Ltd, Little Gibbs Road,
Malabar Hill, Mumbai-400006, Maharashtra, India |
|
Date of Appointment : |
06.12.2012 |
|
DIN No.: |
00053563 |
|
|
|
|
Name : |
Mrs. Revathy Ashok |
|
Designation : |
Director |
|
Address : |
139/6-2 Domlur Layout Sharadamma Layout, Bangalore-560071,
Karnataka, India |
|
Date of Appointment : |
02.12.2016 |
|
DIN No.: |
00057539 |
|
|
|
|
Name : |
Mr. Ian John Parish |
|
Designation : |
Director |
|
Address : |
Sherbrooke House 5 Styal Road Wilmslow Cheshire Sk94ae Gb |
|
Date of Appointment : |
08.08.2017 |
|
DIN No.: |
00391534 |
|
|
|
|
Name : |
Mr. Rajesh Marwaha |
|
Designation : |
Wholetime Director |
|
Address : |
D-1102, Purva Venezia, Yelahanka New Town,
Bangalore-560064, Karnataka, India |
|
Profile : |
Mr. Rajesh Marwaha is a Chartered Accountant with over 24 years of Indian and international working experience, in varied roles in FMCG, Home Appliances, Pharmaceutical and Retail Mall sector. Prior to joining AstraZeneca, Mr. Rajesh Marwaha had worked with PepsiCo for seven years. He joined Groupe SEB SA, a French company in home appliances business in 1996. There, he worked in different functions; finance in India, Global internal audit at headquarters in France, Regional controller sales & commercial in Dubai. He came back to India to join MSD pharmaceuticals in India in 2008 and worked there for 6 years. In his previous work engagements, he has set up new subsidiaries in different countries which has developed a strong business enabling approach in him. |
|
Date of Appointment : |
02.12.2016 |
|
DIN No.: |
01458768 |
|
|
|
|
Name : |
Mrs. Kimsuka Narasimhan |
|
Designation : |
Director |
|
Address : |
309-B, Beverly Park – I, Gurgaon-122002, Haryana, India |
|
Date of Appointment : |
02.02.2017 |
|
DIN No.: |
02102783 |
|
|
|
|
Name : |
Mr. Gregory David Emil Mueller |
|
Designation : |
Director |
|
Address : |
5 Morland Terrace Brooklands Avenue Cambridge Cb28de Gb |
|
Date of Appointment : |
02.12.2016 |
|
DIN No.: |
07667074 |
|
|
|
|
Name : |
Mr. Gagan Singh Bedi |
|
Designation : |
Managing Director |
|
Address : |
Crown Aura, #5a, 5th Floor, Block 1b- 25 Jakkur Village,
Jakkur Planation, Yelaha Nka Hobli, Bengaluru-560064, Karnataka, India |
|
Profile : |
Mr. Gagan Singh Bedi (“Mr. Singh”) joined AstraZeneca Pharma India Limited in February 2006. He held various roles as a Business Director and later as Director Strategic Planning. He also held the position of Vice President-Sales & Marketing – Cardiac and Diabetes. In 2012, Mr. Singh was appointed as the Country President for AstraZeneca Philippines. Post a successful stint in this role, he moved to the United Kingdom as Global Head of Established Brands and Global Medical Lead – Gastrointestinal / Cardio Vascular. Prior to AstraZeneca, Mr. Singh had worked with Baxter India and Eli Lilly. Mr. Singh holds a degree in Economics from the University of Delhi. |
|
Date of Appointment : |
01.07.2017 |
|
DIN No.: |
07844333 |
KEY EXECUTIVES
|
Name : |
Mr. Pratap Rudra Bhuvanagiri |
|
Designation : |
Company Secretary |
|
Address : |
413, 5th M Main, 2nd Block, HRBR Layout, Bangalore-560043,
Karnataka, India |
|
Date of Appointment : |
02.02.2017 |
|
PAN No.: |
AWDPP9893R |
|
|
|
|
Name : |
Mr. Rajesh Marwaha |
|
Designation : |
Chief Financial Officer (KMP) |
|
Address : |
D-1102, Purva Venezia, Yelahanka New Town,
Bangalore-560064, Karnataka, India |
|
Date of Appointment : |
04.08.2014 |
|
PAN No.: |
AAAPM1413D |
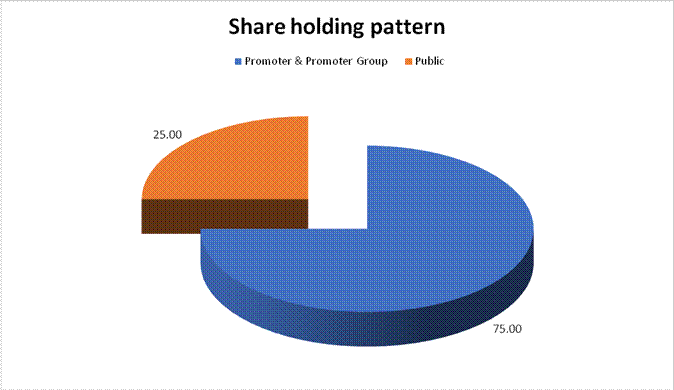
MAJOR SHAREHOLDERS / SHAREHOLDING PATTERN
AS ON 31.03.2018
|
Names of Shareholders |
No. of Shares |
Percentage of Holding |
|
Promoter & Promoter Group |
18750000 |
75.00 |
|
Public |
6250000 |
25.00 |
|
|
|
|
|
Total |
25000000 |
100.00 |
Statement showing
shareholding pattern of the Promoter and Promoter Group
|
Category
of shareholder |
No.
of fully paid up equity shares held |
Shareholding
as a % of total no. of shares (calculated as per SCRR, 1957)As a % of
(A+B+C2) |
|
|
A1) Indian |
0.00 |
|
|
|
A2) Foreign |
0.00 |
|
|
|
Any Other
(specify) |
1,87,50,000 |
75.00 |
|
|
ASTRAZENECA PHARMACEUTICALS AB |
1,87,50,000 |
75.00 |
|
|
Sub Total A2 |
1,87,50,000 |
75.00 |
|
|
A=A1+A2 |
1,87,50,000 |
75.00 |
|
Statement showing shareholding pattern of the Public shareholder
|
Category
& Name of the Shareholders |
No.
of fully paid up equity shares held |
Shareholding
% calculated as per SCRR, 1957 As a % of (A+B+C2) |
|
|
|||
|
|||
|
|||
|
B1) Institutions |
0 |
0.00 |
|
|
Mutual Funds/ |
1928461 |
7.71 |
|
|
ICICI PRUDENTIAL MUTUAL FUND (VARIOUS
SCHEMES) |
1645657 |
6.58 |
|
|
RELIANCE CAPITAL TRUSTEE CO. LTD-A/C
RELIANCESMALL CAP FUND |
282804 |
1.13 |
|
|
Foreign
Portfolio Investors |
31747 |
0.13 |
|
|
Financial
Institutions/ Banks |
18943 |
0.08 |
|
|
Sub Total B1 |
1979151 |
7.92 |
|
|
B2) Central
Government/ State Government(s)/ President of India |
0 |
0.00 |
|
|
B3) Non-Institutions |
0 |
0.00 |
|
|
Individual share
capital upto INR 0.200 Million |
2483809 |
9.94 |
|
|
Individual share
capital in excess of INR 0.200 Million |
1026791 |
4.11 |
|
|
SHIVANI TEJAS TRIVEDI |
427176 |
1.71 |
|
|
MINAXI BHALCHANDRA TRIVEDI |
428959 |
1.72 |
|
|
NBFCs registered
with RBI |
47 |
0.00 |
|
|
Any Other
(specify) |
760202 |
3.04 |
|
|
Unclaimed or Suspense or Escrow Account |
32440 |
0.13 |
|
|
Bodies Corporate |
612724 |
2.45 |
|
|
Clearing Members |
47713 |
0.19 |
|
|
Trusts |
25 |
0.00 |
|
|
IEPF |
67300 |
0.27 |
|
|
Sub Total B3 |
4270849 |
17.08 |
|
|
B=B1+B2+B3 |
6250000 |
25.00 |
|
BUSINESS DETAILS
|
Line of Business : |
Manufacturing, Distribution and Marketing of
Pharmaceutical Products. [Registered Activity] |
|
|
|
|
Products/ Services : |
Pharmaceutical Products |
|
|
|
|
Brand Names : |
Not Available |
|
|
|
|
Agencies Held : |
Not Available |
|
|
|
|
Exports : |
Not Divulged |
|
|
|
|
Imports : |
Not Divulged |
|
|
|
|
Terms : |
|
|
Selling : |
Not Divulged |
|
|
|
|
Purchasing : |
Not Divulged |
PRODUCTION STATUS: (NOT AVAILABLE)
GENERAL INFORMATION
|
Suppliers : |
|
||||||||||||||
|
|
|
||||||||||||||
|
Customers : |
|
||||||||||||||
|
|
|
||||||||||||||
|
No. of Employees : |
1141 (Approximately) |
||||||||||||||
|
|
|
||||||||||||||
|
Bankers : |
The Hongkong and Shanghai Banking Corporation Limited |
||||||||||||||
|
|
|
||||||||||||||
|
Facilities : |
--- |
|
|
|
|
Auditors : |
|
|
Name : |
Price Waterhouse and Company Chartered Accountants |
|
Address : |
5th Floor, Tower D, The Millenia, 1 & 2 Murphy Road,
Ulsoor, Bangalore-560008, Karnataka, India |
|
Tel. No.: |
91-80-40795000 |
|
Fax No.: |
91-80-40795222 |
|
|
|
|
Memberships : |
Not Available |
|
|
|
|
Collaborators : |
Not Available |
|
|
|
|
Holding Company : |
AstraZeneca Pharmaceuticals AB, Sweden |
|
|
|
|
Holding Company of AstraZeneca
Pharmaceuticals AB, Sweden : |
AstraZeneca AB, Sweden |
|
|
|
|
Holding Company of AstraZeneca AB, Sweden
: |
AstraZeneca Treasury Limited, United Kingdom |
|
|
|
|
Ultimate Holding Company : |
AstraZeneca Plc, United Kingdom |
|
|
|
|
Fellow Subsidiaries
: |
· AstraZeneca Singapore Pte Limited, Singapore · AstraZeneca Pharmaceuticals (Phils) Inc, Philippines · AstraZeneca India Private Limited, India · AstraZeneca Pty Ltd, Australia · AstraZeneca Pharmaceutical Co. Limited, China · AstraZeneca Pharmaceuticals LP, USA · IPR Pharmaceuticals Inc, Puerto Rico · AstraZeneca GmbH, Germany · AstraZeneca UK Limited, United Kingdom |
CAPITAL STRUCTURE
AS ON: 31.03.2017
Authorised Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
25000000 |
Equity Shares |
INR 2/- each |
INR 50.000 Million |
|
|
|
|
|
Issued, Subscribed & Paid-up Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
25000000 |
Equity Shares |
INR 2/- each |
INR 50.000 Million |
|
|
|
|
|
(a) Reconciliation of number of shares:
|
|
31.03.2017 |
|
|
|
No. |
INR in Million |
|
Balance at the beginning of the year |
25000000 |
50.000 |
|
Movement during the year |
--- |
--- |
|
Balance at the end
of the year |
25000000 |
50.000 |
(b) Rights,
preferences and restrictions attached to equity shares
The Company has only one class of share referred to as equity shares having par value of INR 2 each. Each holder of equity shares is entitled to one vote per share. The Company declares and pays dividends in Indian rupees.
In the event of liquidation of the Company, the holders of equity shares will be entitled to receive any of the remaining assets of the Company, after distribution of all preferential amounts, in proportion to their shareholding.
The dividend proposed by the Board of Directors is subject to the approval of the shareholders at the Annual General Meeting. During the current year, the amount of per share dividend recognised as proposed distributions to equity shareholders is ` Nil per share (2016: INR Nil per share).
(c) Shares held by Holding/Ultimate Holding Company and/or
their Subsidiaries / Associates:
|
Name of Shareholders |
31.03.2017 |
|
|
|
No. |
INR in Million |
|
Equity shares of INR 2 each, fully paid |
|
|
|
AstraZeneca Pharmaceuticals AB, Sweden (Holding Company) |
18750000 |
37.500 |
(d) Details
of Shareholders holding more than 5% shares in the Company:
|
Name of the
Shareholders |
31.03.2017 |
|
|
|
No. of shares |
% Holding |
|
Equity
shares of ` 2
each, fully paid |
|
|
|
AstraZeneca Pharmaceuticals AB, Sweden (Holding Company) |
18750000 |
75.00 |
|
ICICI Prudential Asset Management Company Limited |
1747500 |
6.99 |
(e) The Company has not allotted any fully paid-up equity shares by way of bonus shares, or in pursuant to contract without payment being received in cash nor has bought back equity shares during the period of five years immediately preceding the Balance Sheet date.
FINANCIAL DATA
[all figures are
in INR Million]
ABRIDGED
BALANCE SHEET
|
SOURCES OF FUNDS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
I.
EQUITY AND LIABILITIES |
|
|
|
|
(1)Shareholders' Funds |
|
|
|
|
(a) Share Capital |
50.000 |
50.000 |
50.000 |
|
(b) Reserves & Surplus |
1754.566 |
1510.143 |
1457.553 |
|
(c) Money received against share warrants |
0.000 |
0.000 |
0.000 |
|
|
|
|
|
|
(2) Share
Application money pending allotment |
0.000 |
0.000 |
0.000 |
|
Total
Shareholders’ Funds (1) + (2) |
1804.566 |
1560.143 |
1507.553 |
|
|
|
|
|
|
(3)
Non-Current Liabilities |
|
|
|
|
(a) Long-term borrowings |
0.000 |
0.000 |
0.000 |
|
(b) Deferred tax liabilities (Net) |
0.000 |
0.000 |
0.000 |
|
(c)
Other long term liabilities |
15.149 |
12.669 |
10.189 |
|
(d)
long-term provisions |
109.053 |
116.570 |
0.000 |
|
Total
Non-current Liabilities (3) |
124.202 |
129.239 |
10.189 |
|
|
|
|
|
|
(4) Current Liabilities |
|
|
|
|
(a)
Short term borrowings |
0.000 |
0.000 |
0.000 |
|
(b)
Trade payables |
357.704 |
946.284 |
1112.716 |
|
(c)
Other current liabilities |
1293.636 |
1210.848 |
1116.001 |
|
(d)
Short-term provisions |
197.340 |
183.325 |
369.469 |
|
Total
Current Liabilities (4) |
1848.680 |
2340.457 |
2598.186 |
|
|
|
|
|
|
TOTAL |
3777.448 |
4029.839 |
4115.928 |
|
|
|
|
|
|
II.
ASSETS |
|
|
|
|
(1) Non-current assets |
|
|
|
|
(a)
Fixed Assets |
|
|
|
|
(i)
Tangible assets |
868.363 |
973.560 |
1009.875 |
|
(ii)
Intangible Assets |
0.000 |
0.000 |
0.000 |
|
(iii)
Capital work-in-progress |
48.125 |
63.122 |
122.065 |
|
(iv) Intangible assets under development |
0.000 |
0.000 |
0.000 |
|
(b) Non-current
Investments |
0.000 |
0.005 |
0.005 |
|
(c) Deferred tax assets
(net) |
0.000 |
0.000 |
0.000 |
|
(d) Long-term Loan
and Advances |
327.215 |
316.381 |
345.524 |
|
(e)
Other Non-current assets |
6.055 |
6.055 |
5.853 |
|
Total
Non-Current Assets |
1249.758 |
1359.123 |
1483.322 |
|
|
|
|
|
|
(2) Current assets |
|
|
|
|
(a)
Current investments |
0.000 |
0.000 |
0.000 |
|
(b)
Inventories |
577.177 |
767.296 |
861.307 |
|
(c)
Trade receivables |
436.032 |
831.433 |
427.727 |
|
(d)
Cash and cash equivalents |
1149.619 |
768.317 |
250.844 |
|
(e)
Short-term loans and advances |
221.698 |
276.369 |
1074.607 |
|
(f)
Other current assets |
143.164 |
27.301 |
18.121 |
|
Total
Current Assets |
2527.690 |
2670.716 |
2632.606 |
|
|
|
|
|
|
TOTAL |
3777.448 |
4029.839 |
4115.928 |
PROFIT
& LOSS ACCOUNT
|
|
PARTICULARS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
|
|
SALES |
|
|
|
|
|
|
|
Revenue from Operations [Net] |
5395.701 |
5637.323 |
5170.694 |
|
|
|
Other Income |
196.429 |
67.727 |
58.483 |
|
|
|
TOTAL |
5592.130 |
5705.050 |
5229.177 |
|
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
|
|
Cost of Materials Consumed |
140.042 |
347.585 |
267.812 |
|
|
|
Purchases of Stock-in-Trade |
1421.553 |
1588.871 |
1672.775 |
|
|
|
Changes in inventories of finished goods, work-in-progress
and Stock-in-Trade |
175.087 |
54.152 |
60.076 |
|
|
|
Employees benefits expense |
1634.764 |
1693.453 |
1598.404 |
|
|
|
Other expenses |
1730.376 |
1789.089 |
1685.692 |
|
|
|
TOTAL |
5101.822 |
5473.150 |
5284.759 |
|
|
|
|
|
|
|
|
|
PROFIT
BEFORE INTEREST, TAX, DEPRECIATION AND AMORTISATION |
490.308 |
231.900 |
(55.582) |
|
|
|
|
|
|
|
|
|
Less |
FINANCIAL
EXPENSES |
0.000 |
0.000 |
0.000 |
|
|
|
|
|
|
|
|
|
|
PROFIT/
(LOSS) BEFORE TAX, DEPRECIATION AND AMORTISATION |
490.308 |
231.900 |
(55.582) |
|
|
|
|
|
|
|
|
|
Less/ Add |
DEPRECIATION/
AMORTISATION |
158.260 |
174.264 |
152.815 |
|
|
|
|
|
|
|
|
|
|
PROFIT/ (LOSS)
BEFORE TAX |
332.048 |
57.636 |
(208.397) |
|
|
|
|
|
|
|
|
|
Less |
TAX |
87.625 |
5.046 |
0.000 |
|
|
|
|
|
|
|
|
|
|
PROFIT/ (LOSS)
AFTER TAX |
244.423 |
52.590 |
(208.397) |
|
|
|
|
|
|
|
|
|
|
EARNINGS IN
FOREIGN CURRENCY |
|
|
|
|
|
|
|
F.O.B. value of exports |
0.000 |
0.000 |
23.472 |
|
|
|
Sale of services |
216.383 |
97.041 |
9.281 |
|
|
TOTAL EARNINGS |
216.383 |
97.041 |
32.753 |
|
|
|
|
|
|
|
|
|
|
IMPORTS |
|
|
|
|
|
|
|
Purchase of traded goods |
1092.516 |
978.486 |
0.000 |
|
|
|
Raw and packing materials (net of returns) |
168.412 |
162.005 |
102.623 |
|
|
|
Capital Goods |
0.000 |
1.470 |
5.435 |
|
|
TOTAL IMPORTS |
1260.928 |
1141.961 |
108.058 |
|
|
|
|
|
|
|
|
|
|
Earnings/ (Loss)
Per Share (INR) |
9.78 |
2.10 |
(8.34) |
|
CURRENT MATURITIES OF LONG TERM DEBT DETAILS
|
PARTICULARS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Current Maturities of Long term debt |
NA |
NA |
NA |
|
|
|
|
|
|
Cash generated from operations |
500.826 |
599.684 |
(448.332) |
|
|
|
|
|
|
Net cash generated from operating activities |
383.089 |
569.885 |
(457.005) |
QUARTERLY RESULTS
|
PARTICULARS |
30.06.2017 |
30.09.2017 |
31.12.2017 |
|
Type |
1st
Quarter |
2nd
Quarter |
3rd
Quarter |
|
Net Sales |
1284.660 |
1642.580 |
1285.390 |
|
Total Expenditure |
1222.490 |
1286.090 |
1393.850 |
|
PBIDT (Excl OI) |
62.170 |
356.490 |
(108.460) |
|
Other Income |
46.540 |
24.960 |
27.600 |
|
Operating Profit |
108.710 |
381.460 |
(80.860) |
|
Interest |
0.000 |
0.000 |
0.000 |
|
Exceptional Items |
0.000 |
0.000 |
0.000 |
|
PBDT |
108.710 |
381.460 |
(80.860) |
|
Depreciation |
37.080 |
37.490 |
37.300 |
|
Profit Before Tax |
71.630 |
343.970 |
(118.160) |
|
Tax |
13.660 |
70.120 |
(14.640) |
|
Provisions and contingencies |
0.000 |
0.000 |
0.000 |
|
Profit After Tax |
57.970 |
273.85 |
(103.520) |
|
Extraordinary Items |
0.000 |
0.000 |
0.000 |
|
Prior Period Expenses |
0.000 |
0.000 |
0.000 |
|
Other Adjustments |
0.000 |
0.000 |
0.000 |
|
Net Profit |
57.970 |
273.85 |
(103.520) |
KEY
RATIOS
EFFICIENCY RATIOS
|
PARTICULARS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Average Collection Days (Sundry
Debtors / Income * 365 Days) |
29.50 |
53.83 |
30.19 |
|
|
|
|
|
|
Account Receivables Turnover (Income / Sundry Debtors) |
12.37 |
6.78 |
12.09 |
|
|
|
|
|
|
Average Payment Days (Sundry Creditors / Purchases * 365 Days) |
83.61 |
178.36 |
209.29 |
|
|
|
|
|
|
Inventory Turnover (Operating Income / Inventories) |
0.85 |
0.30 |
-0.06 |
|
|
|
|
|
|
Asset Turnover (Operating Income / Net Fixed Assets) |
0.53 |
0.22 |
-0.05 |
LEVERAGE RATIOS
|
PARTICULARS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Debt Ratio ((Borrowing
+ Current Liabilities) / Total Assets) |
0.49 |
0.58 |
0.63 |
|
|
|
|
|
|
Debt Equity Ratio (Total Liability / Networth) |
0.00 |
0.00 |
0.00 |
|
|
|
|
|
|
Current Liabilities to Networth (Current Liabilities / Net Worth) |
1.02 |
1.50 |
1.72 |
|
|
|
|
|
|
Fixed Assets to Networth (Net Fixed Assets / Networth) |
0.51 |
0.66 |
0.75 |
|
|
|
|
|
|
Interest Coverage Ratio (PBIT / Financial Charges) |
0.00 |
0.00 |
0.00 |
PROFITABILITY RATIOS
|
PARTICULARS |
|
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Net Profit Margin ((PAT / Sales) * 100) |
% |
4.53 |
0.93 |
(4.03) |
|
|
|
|
|
|
|
Return on Total Assets ((PAT / Total Assets) * 100) |
% |
6.47 |
1.31 |
(5.06) |
|
|
|
|
|
|
|
Return on Investment (ROI) ((PAT / Networth) * 100) |
% |
13.54 |
3.37 |
(13.82) |
SOLVENCY RATIOS
|
PARTICULARS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Current Ratio (Current
Assets / Current Liabilities) |
1.37 |
1.14 |
1.01 |
|
|
|
|
|
|
Quick Ratio ((Current Assets – Inventories) / Current
Liabilities) |
1.06 |
0.81 |
0.68 |
|
|
|
|
|
|
G-Score Ratio Financial (Networth / Total Assets) |
0.48 |
0.39 |
0.37 |
|
|
|
|
|
|
G-Score Ratio Debt (Debts / Equity Capital) |
0.00 |
0.00 |
0.00 |
|
|
|
|
|
|
G-Score Ratio Liquidity (Total Current Assets / Total Current Liabilities) |
1.37 |
1.14 |
1.01 |
Total
Liability = Short-term Debt + Long-term Debt + Current Maturities of Long-term
debts
STOCK
PRICES
|
Face Value |
INR 2.00/- |
|
|
|
|
Market Value |
INR 1002.00/- |
FINANCIAL ANALYSIS
[all figures are
in INR Million]
DEBT EQUITY RATIO
|
Particular |
31.03.2015 |
31.03.2016 |
31.03.2017 |
|
|
INR
In Million |
INR
In Million |
INR In
Million |
|
Share Capital |
50.000 |
50.000 |
50.000 |
|
Reserves & Surplus |
1457.553 |
1510.143 |
1754.566 |
|
Money received against share
warrants |
0.000 |
0.000 |
0.000 |
|
Share Application money
pending allotment |
0.000 |
0.000 |
0.000 |
|
Net worth |
1507.553 |
1560.143 |
1804.566 |
|
|
|
|
|
|
long-term borrowings |
0.000 |
0.000 |
0.000 |
|
Short term borrowings |
0.000 |
0.000 |
0.000 |
|
Total
borrowings |
0.000 |
0.000 |
0.000 |
|
Debt/Equity
ratio |
0.000 |
0.000 |
0.000 |

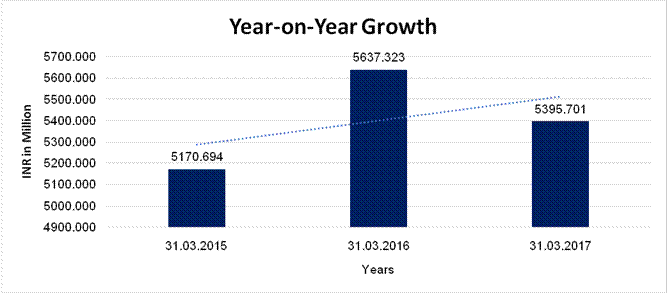
YEAR-ON-YEAR GROWTH
|
Year
on Year Growth |
31.03.2015 |
31.03.2016 |
31.03.2017 |
|
|
INR
In Million |
INR
In Million |
INR
In Million |
|
Sales |
5170.694 |
5637.323 |
5395.701 |
|
|
|
9.024 |
-4.286 |
NET PROFIT MARGIN
|
Net
Profit Margin |
31.03.2015 |
31.03.2016 |
31.03.2017 |
|
|
INR
In Million |
INR
In Million |
INR
In Million |
|
Sales |
5170.694 |
5637.323 |
5395.701 |
|
Profit/ (Loss) |
(208.397) |
52.590 |
244.423 |
|
|
(4.03%) |
0.93% |
4.53% |

LOCAL AGENCY FURTHER INFORMATION
|
Sr. No. |
Check list by
info agents |
Available in
Report (Yes/No) |
|
1 |
Year of establishment |
Yes |
|
2 |
Constitution of the entity -Incorporation
details |
Yes |
|
3 |
Locality of the entity |
Yes |
|
4 |
Premises details |
No |
|
5 |
Buyer visit details |
-- |
|
6 |
Contact numbers |
Yes |
|
7 |
Name of the person contacted |
No |
|
8 |
Designation of contact person |
No |
|
9 |
Promoter’s background |
Yes |
|
10 |
Date of Birth of Proprietor / Partners /
Directors |
Yes |
|
11 |
Pan Card No. of Proprietor / Partners |
No |
|
12 |
Voter Id Card No. of Proprietor / Partners |
No |
|
13 |
Type of business |
Yes |
|
14 |
Line of Business |
Yes |
|
15 |
Export/import details (if applicable) |
No |
|
16 |
No. of employees |
Yes |
|
17 |
Details of sister concerns |
Yes |
|
18 |
Major suppliers |
No |
|
19 |
Major customers |
No |
|
20 |
Banking Details |
Yes |
|
21 |
Banking facility details |
Yes |
|
22 |
Conduct of the banking account |
-- |
|
23 |
Financials, if provided |
Yes |
|
24 |
Capital in the business |
Yes |
|
25 |
Last accounts filed at ROC, if applicable |
Yes |
|
26 |
Turnover of firm for last three years |
Yes |
|
27 |
Reasons for variation <> 20% |
-- |
|
28 |
Estimation for coming financial year |
No |
|
29 |
Profitability for last three years |
Yes |
|
30 |
Major shareholders, if available |
Yes |
|
31 |
External Agency Rating, if available |
No |
|
32 |
Litigations that the firm/promoter
involved in |
-- |
|
33 |
Market information |
-- |
|
34 |
Payments terms |
No |
|
35 |
Negative Reporting by Auditors in the
Annual Report |
No |
INDEX OF CHARGES
|
SNO |
SRN |
CHARGE ID |
CHARGE HOLDER NAME |
DATE OF CREATION |
DATE OF MODIFICATION |
DATE OF SATISFACTION |
AMOUNT |
ADDRESS |
|
1 |
A55774020 |
10083046 |
IL & FS TRUST COMPANY LIMITED |
16/01/2008 |
- |
11/01/2009 |
625000000.0 |
IL & FS FINANCIAL CENTREPLOT NO C22 G BLOCK BANDRAKURLA COMPLEX BANDRA EASTMUMBAIMH400051IN |
GENERAL INFORMATION
AstraZeneca Pharma India Limited (‘the Company’) is a public limited company domiciled in India having its registered office in Bangalore. The Company’s equity shares are listed in National Stock Exchange of India Limited (NSE) and Bombay Stock Exchange Limited (BSE).
The Company is engaged in the business of manufacture, distribution and marketing of pharmaceutical products.
MANAGEMENT DISCUSSION
& ANALYSIS REPORT
INDIAN ECONOMY
Indian economy ranks 7th in the Gross Domestic Product (GDP) global rankings, with a GDP of $ 2.25 trillion and 3rd in terms of the Purchasing Power Parity (PPP) only behind China and the United States(1). Its GDP is projected to grow at 7.7% in the fiscal year 2017 and 7.6% in fiscal year 2018 as per the United Nations World Economic Situation and Prospects (WESP) report and will continue to be the world’s fastest growing major economy. Consumer Price Inflation (CPI) averaged at 4.9% during Apr-Dec 2016. The fiscal deficit of the centre currently stands at 3.5% of GDP (2).
Two recent developments - demonetisation and the Goods and Services Tax (GST) bill have been of significant consequence in India. While demonetisation caused a short-term disruption initially, it is expected to self-correct itself, expand the formal economy and widen the tax base. The GST, which was rolled out on July 1, 2017 has replaced at least 17 state and federal taxes and brought them under one unified tax structure. It is expected to create a common Indian market, improve tax compliance and governance, and boost investment and growth (2).
The Indian economy is on a high growth trajectory despite the challenging global macroeconomic factors like Brexit, US elections and weakening of the Yuan. Investment is expected to pick up helped by monetary easing and Government efforts towards infrastructure investments and publicprivate partnerships and the implementation of domestic reforms such as the introduction of the GST Bill (4). Even so, large non-performing assets, high leverage ratios for some companies and difficulty in passing key structural reforms are holding the economy back (3).
INDIA HEALTHCARE
ENVIRONMENT
The Healthcare sector comprises of hospital services, diagnostic services & products, medical devices, telemedicine, e-Health services, clinical trials, medical tourism and health insurance segments. It is valued at INR 7150 ($110) billion and is expected to grow to INR 18200 ($280) billion by 2020. Currently, approximately 80% of the hospital sector, which is the major sector in healthcare, is predominantly privatized6.
The market size of the diagnostic segment is also expected to grow at a Compounded Annual Growth Rate (CAGR) of 20% to INR 2080 ($ 32) billion in 2022 from INR 325 ($ 5) billion in 201221. With government focus on quality and affordable healthcare for everyone and numerous other private initiatives, telemedicine is another segment whose future looks promising and poised for growth. Growing at a CAGR of over 20%, it is expected to more than double to INR 2048 ($32) million by 2020 from the current INR 960 ($15) million according to a recent Assocham’s study17.
Investment in healthcare start-ups in areas such as Healthcare discovery, Pharmaceutical delivery, Information management systems and Home healthcare services is gaining momentum in India. The Healthcare sector received the 3rd highest Private Equity (PE) & Venture Capital (VC) investments in India, next only to the Information Technology (IT) & Banking, Financial services & Insurance (BFSI) sector15.
The National Health Policy (NHP 2017) (the first new health policy introduced by the Government in the last 15 years) addresses the current healthcare landscape and is expected to improve the access to and quality of healthcare and lower healthcare delivery costs.
Foreign Direct Investment (FDI) up to 100% is allowed in the hospital sector and in the manufacture of medical devices13. In the pharmaceutical sector, FDI is permitted up to 100 % in Greenfield projects and 74% in Brownfield projects under the automatic route14.
Total healthcare spend in India is 4.7% of GDP of which the Government spending is 30%. India’s public healthcare expenditure is at a dismal low of 1.4% against the world average of 6%9 though the NHP 2017 (passed in March 2017) envisages raising it progressively to 2.5% of the GDP by 2025. The Indian healthcare system, however, is still characterised with glaring gaps in infrastructure.
It has only 0.9 hospital beds per 1000 people, 0.7 doctors and 1.7 nurses per 1000 people - which are the lowest rates amongst the BRICS nations7 .
Gaps in public healthcare infrastructure has led to a rise in the out of pocket (OOP) expenditure and consequent impoverishment. The percentage of population having some degree of health insurance has increased marginally to 24% but the out-ofpocket spending remains as high as 62% of the total healthcare expense leading to increased burden on the patients10.
Concerns remain of low awareness among the beneficiaries about the entitlements, how and when to use the Rashtriya Swasthiya Bima Yojana (RSBY) card, denial of services by private hospitals for many categories of illnesses and fraudulent measures, including charging informal payments. Variability in the quality and rationality of care provided by the private players also remains an area of concern.
The rising occurrence of Non-Communicable diseases (NCDs) in India is a public health challenge with NCDs accounting for 60% of all deaths in India. The National Health Policy 2017 focusses on screening for Oral, Breast and Cervical cancer and for Chronic Obstructive Pulmonary Disease (COPD) in addition to Hypertension and Diabetes to deal with the growing NCD burden. There are also increased hospitalisations for the NCDs, growing at a CAGR of 18%, 16% and 19% for Cardiac, Oncology and Diabetes segments respectively6 .
INDIAN PHARMACEUTICAL
MARKET (IPM)
The INR 1143 ($17.6) billion Indian Pharmaceutical industry experienced a value growth of 9.1% in FY 2016-17 over FY 2015-16 as per March 2017 IMS Health database. Indian companies dominate the market with a 78.5% share of IPM and a value growth of 9.9%. The value growth for MNCs at 6.5% in FY 2016-17, is lower than the market growth rate as well as the FY 2015-16 value growth of 12.7%. There has been a sharp decline in volume (per unit) growth for both the MNCs and the Indian companies as well. While the Indian companies registered a volume growth of 3% in FY 2016-17 as against 6% in FY 2015-2016, the MNCs experienced a decline in volumes, -7.9% versus last year’s growth of 10.2%. This has been observed across the industry with most of the therapy areas growing less in FY 2016-17 vis-à-vis last year11.
PHARMACEUTICAL
BUSINESS ENVIRONMENT OUTLOOK
With increased thrust for affordable and quality healthcare coupled with economic expansion, insurance penetration, population growth and epidemiological trends, the demand for medicines is poised to grow in the coming years. The total pharmaceutical market is forecast to grow at a CAGR of 11.0% (±3.0%) over the period 2016-202110.
Revision of the National List of Essential Medicines (NLEM) along with appropriate price control mechanisms for drugs will remain a key strategy for government for decreasing costs of care, as is evident from the February, 2017, reduction of stent prices by up to 85% by the National Pharmaceutical Pricing Authority (NPPA)18. The legal uncertainty towards the ban of various fixed-dose combinations (FDCs) in India will continue to impact growth in the short term. Signs of a more flexible approach to drug registration is being witnessed and clinical trial requirements are being eased, allowing faster access for new drugs. Implementation of the long-awaited GST is set to improve the operating environment and simplify the supply chain, resulting in reduced production, distribution and warehousing costs.
Collaborations in the healthcare space to increase patient access are on the rise. Distribution deals are increasing as various companies are trying to expand their reach; a few of them being - AstraZeneca & Sun, in the cardiology space; AstraZeneca & DRL, in the diabetes space; Sun & MSD (Merck, Sharp & Dohme), in the diabetes space; Novartis & Lupin in the Asthma & COPD space. Major pharmaceutical companies also pursued divestments and swap deals to concentrate on their focus areas. Novartis sold its Animal Health division to Eli Lily and traded its vaccine business to GlaxoSmithKline (GSK). Similarly, AstraZeneca divested its small molecule antibiotics business to Pfizer, allowing it to focus on its growth platforms – Oncology, CardioVascular & Metabolics and Respiratory. The Pharmaceutical sector has witnessed a significant increase in Merger and Acquisition (M&A) activities over the years. In the last three years, Pharmaceuticals segment accounted for more than 70% of M&A deals in the Indian health care space6 .
With the increased incidence of chronic, NCDs, led by cardiovascular disease, chronic respiratory diseases and type 2 diabetes, the demand for private inpatient care will continue to grow. Shift from curative to preventive health care, online patient communities and e-pharmacies may also pick up rapidly. Detailing will remain central to promotional strategies. The implementation timing remains unclear for the Uniform Code of Pharmaceutical Marketing Practice (UCPMP) which is intended to curb unethical marketing practices. Existing marketing capabilities may have to go beyond classical pharmaceutical selling to include disease management, market creation capabilities and customized approaches with sharper segmentation and targeting of customers. Consequently the existing models of sales force coverage and sales force excellence may also require an overhaul. New promotional tools and capabilities may be required as accessing prescribers will become more difficult.
A new Intellectual Property Rights (IPR) Policy was released in May 2016, which acknowledged the importance of IP rights, but did not change either patentability definitions or compulsory licensing provisions contained in the 2005 Act. The Policy is in alignment with WTO's (World Trade Organization) agreement on TRIPS (Trade Related aspects of IPRs) and it aims to sustain entrepreneurship and boost Prime M
OUTLOOK
In FY 2017-18, AstraZeneca continues to prioritize investment in its growth platforms; Cardiovascular and Metabolic Diseases Therapy Area (TA), Respiratory TA and Oncology TA. It will also continue to take an opportunity driven approach toward the mature products. Accelerating new products is a key priority and the Company is committed to maintaining timelines of key regulatory milestones to align with the global pipeline, subject to conduct of clinical trials, regulatory approvals and reasonable commercial viability.
However, in FY 2017-2018, the Company will also need to manage the various risks and uncertainties in the pharmaceutical market and government policies; prominent among them being authorities to approve new molecules and competitive pricing pressure.
According to IMS Health data (March 2017), Antidiabetic Therapy valued at INR 100.6 billion, is the second largest contributor to IPM and at 17.6% value growth, is amongst the fastest growing therapy segments in the IPM. The Oral Antidiabetic segment at INR 74.4 billion constitutes 74% of this market and is growing at 16.4%. Needless to say, the competitive pressures in this market is also enormous with a number of companies marketing their branded generics and innovative medicines to the healthcare community and providing multiple choices to patients. In this crowded market, the Company will continue to focus on its antidiabetic portfolio consisting of the Dapaglifozin franchise as well as the Saxagliptin franchise. The products are differentiated and enjoy a good acceptance by the medical community as one of their solutions to diabetic patients. The Company’s innovator brand in Oral Anti Platelet (OAP) market, Brilinta® (Ticagrelor) continues to improve its value market share over the last one year. Barring unforeseen circumstances, the Company along with its Ticagrelor partner brand will endeveour to deliver the brand’s potential to reduce cardiovascular deaths through ongoing clinical studies and sustained focus on plans for market leadership.
With a growth rate of 9.78%, the domestic market for respiratory diseases is one amongst the fastest growing therapies in India. Increasing air pollution in our cities and rural areas is contributing to a high burden of respiratory disease. This may present an opportunity to AstraZeneca with Symbicort®, which is an ICS/LABA combination with an innovative delivery mechanism. The Company is building its expertise in this disease area and will need sustained efforts to build a good portfolio of offerings to patients.
Finally, cancer is becoming a major cause of mortality in India. Every year, nearly 1 million new patients are diagnosed with cancer. The Company has in its current Oncology Portfolio - products to provide treatment in the areas of women’s cancer and lung cancer. These cancers have a relatively high prevalence in India and the Company will hope to expand its access to more patient’s subject to scientific and clinical appropriateness.
The Company will continue to emphasize high standards of sales and marketing practices, maintaining a strong focus on patient safety and patient needs. The Company will remain committed to high product quality, which underpins the safety and efficacy of its medicines.
The Company will maintain a strong focus on cost optimization and controls. The Company is undertaking measures to reduce unproductive discretionary and non-customer facing spends. It also continues to develop simple and more efficient processes to encourage accountability and improve decision making and communication.
STANDALONE
UNAUDITED FINANCIAL RESULTS FOR THE QUARTER AND NINE MONTHS ENDED 31.12.2017
(INR IN MILLION)
|
Particulars |
3 months ended |
9 months ended |
|
|
|
31.12.2017 (Unaudited) |
30.09.2017 (Unaudited) |
31.12.2017 (Unaudited) |
|
INCOME FROM OPERATIONS |
|
|
|
|
Revenue from operations |
1285.386 |
1645.583 |
4212.627 |
|
Other Income |
27.597 |
24.957 |
99.094 |
|
Total
Income from Operations |
1312.983 |
1667.540 |
4311.721 |
|
|
|
|
|
|
EXPENSES |
|
|
|
|
Cost of Materials consumed |
77.878 |
31.240 |
163.894 |
|
Purchase of stock-in-trade |
732.639 |
477.819 |
1794.854 |
|
Changes in inventories of finished goods, work-in-progress and
stock-in-trade |
(426.083) |
25.476 |
(588.326) |
|
Excise Duty |
--- |
--- |
9.850 |
|
Employee benefits expense |
371.165 |
364.019 |
1084.572 |
|
Depreciation and Amortization expenses |
37.301 |
37.485 |
111.866 |
|
Selling, marketing and distribution expense |
245.681 |
152.649 |
516.921 |
|
Other Expenditure |
392.563 |
234.884 |
920.654 |
|
Finance Costs |
--- |
--- |
--- |
|
Total
Expenses |
1431.144 |
1323.572 |
4014.285 |
|
Profit/(Loss) before Exceptional and extraordinary Items
and tax |
(118.161) |
343.968 |
210.022 |
|
Exceptional Items |
--- |
--- |
--- |
|
Profit/(Loss) before Extraordinary Items and tax |
(118.161) |
343.968 |
210.022 |
|
Extraordinary Items |
--- |
--- |
--- |
|
Profit / (Loss) before Tax
|
(118.161) |
343.968 |
210.022 |
|
Tax Expenses |
|
|
|
|
Current Tax |
(14.639) |
69.877 |
70.499 |
|
Deferred Tax |
--- |
0.243 |
--- |
|
Total
|
(14.639) |
70.120 |
70.499 |
|
Profit/Loss
for the period |
(103.522) |
273.848 |
226.937 |
|
Other comprehensive
income, net of income tax |
|
|
|
|
Items that will not be reclassified to profit or loss |
24.327 |
(0.943) |
28.260 |
|
Items that will be reclassified to profit or loss |
--- |
--- |
--- |
|
Total comprehensive income (net of
income tax) |
24.327 |
(0.943) |
28.260 |
|
Total comprehensive income for the
period |
(79.195) |
272.905 |
255.197 |
|
Paid up equity share capital (Face value of INR 2/- per equity share) |
50.000 |
50.000 |
50.000 |
|
Earnings per equity
share of INR 2/- each (Basic and Diluted) |
(4.14) |
10.95 |
9.08 |
Notes:
1. The above statement of results has been reviewed by the Audit Committee and approved by the Board of Directors of the Company at their respective meetings held on 5 February 2018. The statutory auditors of the Company have carried out a limited review of this statement of results for the quarter ended 31 December 2017.
2. This Statement has been prepared in accordance with the Companies (Indian
Accounting Standards) Rules, 2015 (Ind AS) prescribed under Section 133 of the
Companies Act, 2013 and other recognised accounting practices and policies to
the extent applicable. Beginning 1 April 2017, the Company has for the first
time adopted Ind AS with a transition date of 1 April 2016.
3. The Company has identified 'Healthcare Segment' as its only reportable
segment in accordance with the requirements of Ind AS 108, 'Operating
Segments'. Accordingly no segment information has been provided.
4. As previously disclosed, by way of a letter dated 1 March 2014, AstraZeneca
Pharmaceuticals AB, the promoter of the Company had proposed a voluntary
delisting of the Company's equity shares from the National Stock Exchange and
the Bombay Stock Exchange. Such proposed delisting is subject to an on-going
inquiry with SEBI and that inquiry has not yet been resolved. In any event,
based on the passage of time, any potential future proposal for voluntary
delisting of the Company would need to be conducted de novo.
5. The Statement does not include Ind AS compliant statement of results for the
previous year ended 31 March 2017 as the same is not mandatory as per SEBI's
circular dated 5 July 2016.
6. Previous period's figures have been regrouped to conform with the current
period's presentation for the purpose of comparability.
7. The reconciliation of net profit reported in accordance with Indian GAAP to
total comprehensive income in accordance with Ind AS is given below:
|
Particulars |
3
months ended |
9 month ended |
|
|
31.12.2016 (Unaudited) |
31.12.2016 (Unaudited) |
|
Net Profits as per previous GAAP Indian GAAP |
159.629 |
273.176 |
|
Add/(Less): Adjustment for Ind AS |
|
|
|
Impact on provision of expected credit loss |
(3.327) |
(2.759) |
|
Reversal of rent equalization reserve |
0.633 |
1.898 |
|
Reclassification of net actuarial gain/loss on defined benefit plan to other Comprehensive Income |
5.859 |
21.313 |
|
Impact of employee stock compensation plan |
(1.940) |
(1.857) |
|
Others |
1.250 |
4.375 |
|
Net profit after tax as per Ind AS |
162.104 |
296.144 |
|
Other Comprehensive Income net of Income tax |
|
|
|
Actuarial loss on defined benefit plan |
(5.859) |
(21.313) |
|
Total Comprehensive Income under Ind AS |
156.245 |
274.831 |
CONTINGENT
LIABILITIES:
|
Particulars |
31.03.2017 (INR
In Million) |
31.03.2016 (INR
In Million) |
|
Claims against the company not
acknowledged as debts |
|
|
|
Excise and service tax matters* (net of payment under protest) |
15.316 |
14.941 |
|
Income tax matters # (net of payment under protest) |
80.034 |
137.585 |
|
Guarantees |
|
|
|
Bank guarantee issued to National Highways Authority of India |
13.704 |
13.704 |
Notes:
* The Company had received a service tax demand of INR 23.883 Million for the period April 2006 to March 2012, on the expenditure incurred in foreign currency for various expenses such as registration fee, transportation, accommodation for attending conferences / seminars, meetings and trainings. The Commissioner vide OIO 62/2014 confirmed the demand along with interest and penalties, against which the Company has filed an appeal with Customs Excise and Service Tax Appellate Tribunal ("CESTAT") on 8 January 2015 which is currently pending. Out of the above balance, the Company has provided INR 11.064 Million as a matter of prudent accounting practice and the balance amount of INR 12.819 Million is considered as a contingent liability.
* The Company had received a service tax demand of INR 5.875 Million for the period April 2010 to November 2012 vide OIO 138/2015 disallowing input tax credit on services such as sponsorship, insurance, event management, waste disposal services. The order has been passed by Commissioner confirming the demand along with interest and penalties, against which the Company has paid INR 0.441 Million under protest as on date and has filed an appeal with Commissioner of Central Excise (Appeals) on 18 February 2016 which is currently pending. Out of the above demand, the Company has provided INR 2.937 Million as a matter of prudent accounting practice and the balance amount of INR 2.497 Million is considered as a contingent liability.
# The Transfer Pricing Officer ("TPO") vide its Order for the period April 2008 to March 2009 made an adjustment to the clinical trial income of the Company by determining the arm's length margin at 43.73%. Moreover, the Assessing Officer ("AO") carried out adjustments relating to disallowance of provision for doubtful advances, difference between interest income as per books and TDS certificate and disallowance of expenses in respect of sample distribution, grants, sponsorship, medical donations and equipment donation. The Dispute Resolution Panel passed an unfavorable order against the Company on 19 November 2013 after which the AO confirmed the demand vide its Order dated 30 December 2013 amounting to INR 84.300 Million. The Company filed a submission before the Income Tax Appellate Tribunal ("ITAT") on 28 February 2014. 50% of the total demand has been deposited under protest as per the order of the AO amounting to INR 42.150 Million. The stay order on the balance tax demand expired on 4 August 2014, for which the Company filed an application with the ITAT. The Bench members heard both the parties and decided to club the stay matter alongwith the main hearing on technical merits and listed the matter for hearing on 9 July, 2015. The Department Representative informed the Bench that no coercive action will be initiated in the interim. The ITAT has passed an order on 27 December 2016 which was received by the Company on 10 January 2017. The ITAT has given relief on adjustments made with respect to arm's length margin for clinical trials income and Corporate Tax adjustments (Allowance for expenses in respect of sample distribution, grants, sponsorship). The Company is currently in the process of filing an application before the AO/ TPO along with the relevant submissions requesting them to give effect to the order of ITAT. The Transfer Pricing Officer ("TPO") vide its Order for the period April 2010 to March 2011 made an adjustment to the clinical trials income of the Company. Moreover, the AO carried out adjustments relating to disallowance of provision for doubtful advances, difference between interest income as per books and TDS certificate and disallowance of expenses in respect of sample distribution, grants, sponsorship, medical donations and equipment donation. The Company filed an appeal with the Dispute Resolution Panel ("DRP") on 27 March 2015, Post the hearing held during the year, the DRP on 20 January 2016 has passed the final order confirming the final liability at INR 10.397 Million. The Company has filed an appeal with ITAT on 17 March 2016 challenging the disallowances made by the DRP. The case is listed before the ITAT on 18 May 2017 for hearing.
The Transfer Pricing Officer ("TPO") vide its Order for the period April 2011 to March 2012 has charged a markup on the receipt of reimbursement of expenses by the Company from overseas group companies. Moreover, the Assessing Officer ("AO") carried out adjustments relating to disallowance of expenses incurred on health care professionals, payout made to DHS against price difference, sales returns not supported by evidence, cost of samples, additional depreciation claim, SAD refund, VRS expenses and 40(a)(ia) Disallowance. The Company has filed an appeal with the Dispute Resolution Panel ("DRP") on 18 March 2016 and DRP passed an order on 11 November 2016. Subsequently the final order was issued by the AO reducing the net refund due to the Company by INR 27.487 Million.
The Company is not carrying any provision for the cases other than those mentioned above in its books of account, as the Company is confident of successfully litigating the matters.
FIXED ASSETS:
Tangible Assets
· Leasehold Land
·
Buildings
·
Plant and Machinery
·
Furniture and Fixtures
·
Vehicles
·
Office Equipment
·
Roads and culverts
PRESS RELEASE :
ASTRAZENECA TO INVEST INR 6000.000 MILLION IN INDIA OVER NEXT 5 YEARS
Apr 17, 2018
The Investment
Commitment will Span Across Astrazeneca’s Diverse Footprint in India with an
Aim to Strengthen its Manufacturing, Clinical Operations, Patient Safety and
Regulatory Science, It Services and Commercial Operations
Anglo-Swedish drugmaker AstraZeneca on Tuesday said it will be investing around INR 6000.000 Million over the next 5 years in India.
The announcement was made following a meeting between the company’s Executive Vice President for International Region, Leon Wang, and Prime Minister Narendra Modi, during the latter’s visit to Sweden, where the two countries signed a joint declaration on innovation partnership for a sustainable future.
The investment commitment will span across AstraZeneca’s diverse footprint in India with an aim to strengthen its manufacturing, clinical operations, patient safety & regulatory science, IT services and commercial operations.
“Furthermore, this investment will support scientific data generation, quality manufacturing, science talent development and collaborative development of innovative solutions to improve the standard of care of non-communicable diseases in India. The commitment also aligns closely with the Indian government’s initiatives to create a stronger business, innovation, and healthcare environment such as Skill India, National Health Policy and Ayushman Bharat,” AstraZeneca said in a statement.
AstraZeneca’s strategy in India is to address the growing burden of non-communicable diseases. The company invests in many initiatives and programmes such as the Young Health Programme, Early Action in Diabetes & Healthy Lung for early action in awareness, prevention, detection and holistic management of non-communicable diseases.
At present, AstraZeneca has a workforce of over 4000 employees in India spanning manufacturing, sales and marketing, clinical operations, IT services, digital services and global medicines development.
The company recently announced expansion of its Global Technology Centre (GTC) in Chennai, now employing more than 2,000 employees. GTC is AstraZeneca’s first insourced IT facility. AstraZeneca’s manufacturing facility in Bengaluru has a sophisticated production facility designed to meet the most stringent international standards, conforming to World Health Organisation cGMP (current Good Manufacturing Practices) norms. AstraZeneca also has a sizeable clinical operation and is expanding its global medicines development unit that focuses on patient safety and regulatory science in India.
Shares of AstraZeneca rose 4.11 per cent to close at INR 994.55 on BSE, the benchmark Sensex gained 0.26 per cent to end at 34,395.06 points.
CMT REPORT (Corruption, Money Laundering & Terrorism]
The Public Notice information has been collected from various sources
including but not limited to: The Courts, India Prisons Service,
Interpol, etc.
1] INFORMATION ON
DESIGNATED PARTY
No exist designating subject or any of its beneficial owners,
controlling shareholders or senior officers as terrorist or terrorist
organization or whom notice had been received that all financial transactions
involving their assets have been blocked or convicted, found guilty or against
whom a judgement or order had been entered in a proceedings for violating
money-laundering, anti-corruption or bribery or international economic or
anti-terrorism sanction laws or whose assets were seized, blocked, frozen or
ordered forfeited for violation of money laundering or international
anti-terrorism laws.
2] Court Declaration :
No records exist to suggest that subject is
or was the subject of any formal or informal allegations, prosecutions or other
official proceeding for making any prohibited payments or other improper
payments to government officials for engaging in prohibited transactions or
with designated parties.
3] Asset Declaration :
No records exist to suggest that the property or assets of the subject
are derived from criminal conduct or a prohibited transaction.
4] Record on Financial
Crime :
Charges or conviction
registered against subject: None
5] Records on Violation of
Anti-Corruption Laws :
Charges or
investigation registered against subject: None
6] Records on Int’l
Anti-Money Laundering Laws/Standards :
Charges or
investigation registered against subject: None
7] Criminal Records
No
available information exist that suggest that subject or any of its principals
have been formally charged or convicted by a competent governmental authority
for any financial crime or under any formal investigation by a competent
government authority for any violation of anti-corruption laws or international
anti-money laundering laws or standard.
8] Affiliation with
Government :
No record
exists to suggest that any director or indirect owners, controlling
shareholders, director, officer or employee of the company is a government
official or a family member or close business associate of a Government
official.
9] Compensation Package :
Our market
survey revealed that the amount of compensation sought by the subject is fair
and reasonable and comparable to compensation paid to others for similar
services.
10] Press Report :
No press reports / filings exists on
the subject.
CORPORATE GOVERNANCE
MIRA INFORM as part of its Due Diligence do provide comments on
Corporate Governance to identify management and governance. These factors often
have been predictive and in some cases have created vulnerabilities to credit
deterioration.
Our Governance Assessment focuses principally on the interactions
between a company’s management, its Board of Directors, Shareholders and other
financial stakeholders.
CONTRAVENTION
Subject is not known to have contravened any existing local laws,
regulations or policies that prohibit, restrict or otherwise affect the terms
and conditions that could be included in the agreement with the subject.
FOREIGN EXCHANGE RATES
|
Currency |
Unit
|
INR |
|
US Dollar |
1 |
INR 66.78 |
|
UK Pound |
1 |
INR 92.89 |
|
Euro |
1 |
INR 80.74 |
INFORMATION DETAILS
|
Information
Gathered by : |
GYT |
|
|
|
|
Analysis Done by
: |
DIV |
|
|
|
|
Report Prepared
by : |
IND |
SCORE FACTORS
|
DEMERIT POINTS |
|
|
|
--BANK CHARGES |
YES/NO |
YES |
|
--LITIGATION |
YES/NO |
NO |
|
--OTHER ADVERSE INFORMATION |
YES/NO |
NO |
|
MERIT POINTS |
|
|
|
--SOLE DISTRIBUTORSHIP |
YES/NO |
NO |
|
--EXPORT ACTIVITIES |
YES/NO |
NO |
|
--AFFILIATION |
YES/NO |
YES |
|
--LISTED |
YES/NO |
YES |
|
--OTHER MERIT FACTORS |
YES/NO |
YES |
RATING EXPLANATIONS
|
Credit Rating |
Explanation |
Rating Comments |
|
A++ |
Minimum Risk |
Business dealings permissible with minimum
risk of default |
|
A+ |
Low Risk |
Business dealings permissible with low
risk of default |
|
A |
Acceptable Risk |
Business dealings permissible with
moderate risk of default |
|
B |
Medium Risk |
Business dealings permissible on a regular
monitoring basis |
|
C |
Medium High Risk |
Business dealings permissible preferably
on secured basis |
|
D |
High Risk |
Business dealing not recommended or on
secured terms only |
|
NB |
New Business |
No recommendation can be done due to
business in infancy stage |
|
NT |
No Trace |
No recommendation can be done as the
business is not traceable |
NB is stated where there is insufficient information to facilitate rating. However, it is not to be considered as unfavourable.
This score serves as a reference to assess
SC’s credit risk and to set the amount of credit to be extended. It is
calculated from a composite of weighted scores obtained from each of the major
sections of this report. The assessed factors are as follows:
·
Financial
condition covering various ratios
·
Company
background and operations size
·
Promoters
/ Management background
·
Payment
record
·
Litigation
against the subject
·
Industry
scenario / competitor analysis
·
Supplier
/ Customer / Banker review (wherever available)
This report is issued at
your request without any risk and responsibility on the part of MIRA INFORM
PRIVATE LIMITED (MIPL) or its officials.

