|
|
|
MIRA
INFORM REPORT
|
Report No. : |
490337 |
|
Report Date : |
06.02.2018 |
IDENTIFICATION DETAILS
|
Name : |
STATE PHARMACEUTICALS MANUFACTURING CORPORATION |
|
|
|
|
Registered Office : |
11, Sir John Kotelawala Mawatha, Kandawala Estate, Ratmalana |
|
|
|
|
Country : |
Sri Lanka |
|
|
|
|
Financials (as on) : |
31.12.2015 |
|
|
|
|
Date of Incorporation : |
04.06.1987 |
|
|
|
|
Legal Form : |
Corporation (State
Owned) |
|
|
|
|
Line of Business : |
Manufacturers of
Medical Drugs for Distribution at Affordable Prices (Tablets, Capsules & Syrups) |
|
|
|
|
No. of Employees : |
300+ Approximately |
RATING & COMMENTS
(Mira Inform has adopted New Rating mechanism w.e.f. 23rd
January 2017)
|
MIRA’s Rating : |
A |
|
Credit Rating |
Explanation |
Rating Comments |
|
A |
Acceptable Risk |
Business dealings permissible with
moderate risk of default |
|
Maximum Credit Limit : |
2,230,404.370 USD |
|
|
|
|
Status : |
Good |
|
|
|
|
Payment Behaviour : |
No Complaints |
|
|
|
|
Litigation : |
Clear |
NOTES :
Any query related to this report can be made
on e-mail : infodept@mirainform.com
while quoting report number, name and date.
ECGC Country Risk Classification List
|
Country Name |
Previous Rating (30.06.2017) |
Current Rating (30.09.2017) |
|
Sir Lanka |
A2 |
A2 |
|
Risk Category |
ECGC
Classification |
|
Insignificant |
A1 |
|
Low Risk |
A2 |
|
Moderately Low Risk |
B1 |
|
Moderate Risk |
B2 |
|
Moderately High Risk |
C1 |
|
High Risk |
C2 |
|
Very High Risk |
D |
SRI LANKA - ECONOMIC OVERVIEW
Sri Lanka is attempting to sustain economic growth while maintaining
macroeconomic stability under an IMF program. The government's high debt
payments and bloated civil service cadre, which have contributed to
historically high budget deficits and low tax revenues, remain a concern.
Government debt is almost 80% of GDP and remains among the highest of the
emerging markets.
The new government in 2015 drastically increased wages for public sector
employees, which boosted demand for consumer goods but hurt the overall balance
of payments and reduced foreign exchange reserves.
Tourism has experienced strong growth during recent years following the
resolution of the government's 26-year conflict with the Liberation Tigers of
Tamil Eelam. The government has been pursuing large-scale reconstruction and
development projects in its efforts to spur growth.
|
Source
: CIA |
Company profile
|
Name of Subject of Inquiry |
STATE
PHARMACEUTICALS MANUFACTURING CORPORATION |
|
Legal Form & Status |
Corporation (State
Owned) |
|
Chairman |
Dr Sayuru G
Samaradundera |
|
Primary Line of Business |
Manufacturers of
Medical drugs for distribution at affordable prices (Tablets, Capsules & Syrups) |
|
Principal Place of Business |
No 11, Sir John
Kothalawala Mawatha, Kandawala Estate Ratmalana Sri Lanka. Tel. No: (00 94 11) 2635353 Fax. No: (00 94
11) 2634771 E-Mail:
chairman@spmclanka.lk / chairmanspmc@sltnet.lk Web Site:
www.spmclanka.com |
|
Employees |
300+ Approximately |
Company details
|
Registration No |
Established in 1987 under the State Industrial Corporations Act No. 49
of 1957 as a state owned business
Undertaking. |
|
|
Date of Registration |
04th June 1987 |
|
|
Re-registration
No |
NA |
|
|
Registered
Office |
11, Sir John Kotelawala Mawatha, Kandawala Estate, Ratmalana, Sri
Lanka. |
|
|
Board of
Directors As At 31.12.2017 |
Dr Sayuru G Samaradundera |
Chairman |
|
Mr P Suntharalingam |
Managing Director |
|
|
Dr Susi Perera |
Director |
|
|
Dr D Venkatheswaran |
Director |
|
|
Mrs N Godakanda |
Director |
|
|
Mr Safeec Rajabdeen |
Director |
|
|
Mr Miuru Bhashitha Liyanage |
Director |
|
|
Mrs S W Jayasundara |
General Manager |
|
|
Stated Share
Capital |
LKR 690,079,000.00 |
|
|
No. of Shares |
NA |
|
|
Preferential
Shares |
NA |
|
|
Nominal Value of
Share |
NA |
|
|
Auditors |
Auditor General’s Department – Government of Sri Lanka |
|
|
Company
Secretaries |
NA |
|
|
Main Bank/s |
Bank of Ceylon, Peoples Bank of Sri Lanka, National Savings Bank |
|
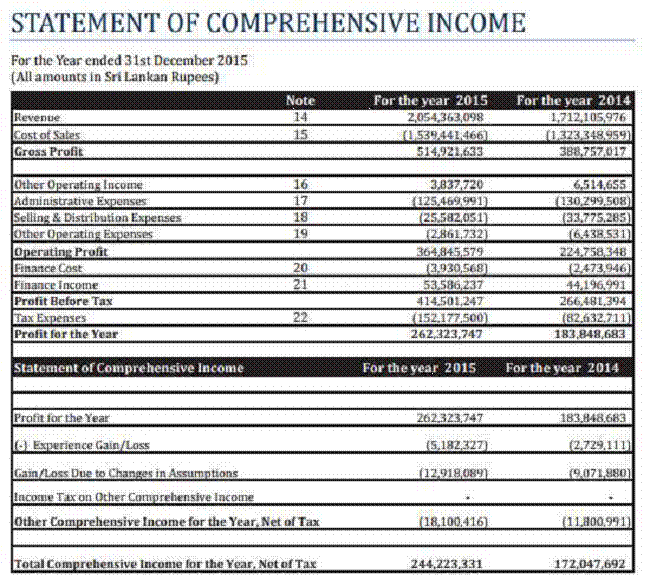
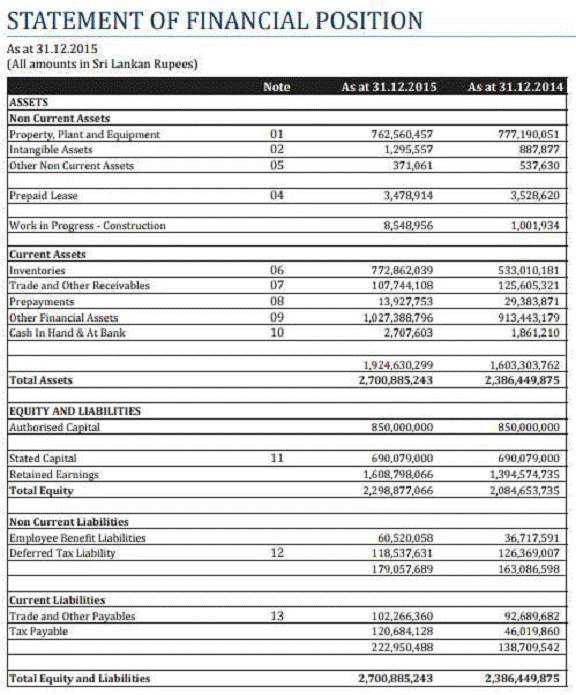
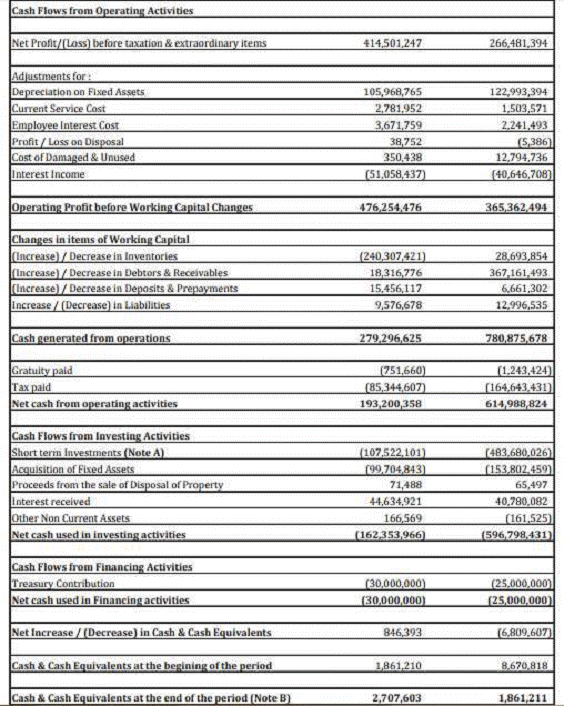
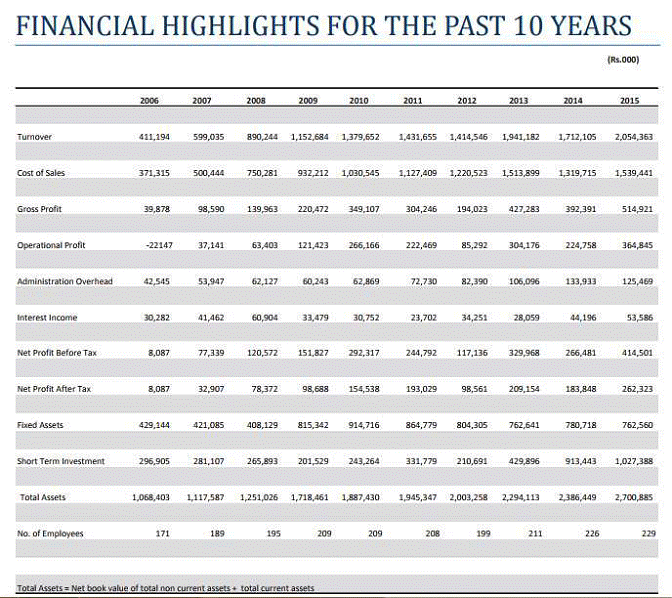
FINANCIAL DETAILS
Most recent
available Financial Information
Mrs.
Asanthi Aturaliya
DGM-Finance
Office
Direct : 011 - 2626630
Office
: 0112637574 , 2635353 , Ext. 311
E-Mail
- aturaliya.agds@spmclanka.lk
Current investigation

COMPANY LOGO&
LISTING
History & Operational
Details
The SPMC was established in 1987 under the Industrial Corporation Act
No. 49 of 1957, based on a grant aid received from the Japanese government
through JICA (Japan International Cooperation Agency) with a commitment to
manufacture quality, effective and safe medicinal drugs. This far-sighted
decision was first proposed in the Bibile - Wickramasinghe Report published in
March 1971.
The main functions
of the SPMC as mandated by the Industrial Corporation Act No 49 are:
·
Manufacturing, processing, stocking, packing and
re-packing of drugs
·
Provision of technical assistance for the
manufacture and processing of drugs
·
Pharmacological and pharmaceutical research and the
standardization of drugs
·
Marketing of Pharmaceuticals.
SPMC's current product range consist of 46 items of Tablets and
Capsules. SPMC is the only state sector corporation engaged in manufacturing
pharmaceuticals in Sri Lanka. All products released to the market meet with the
British Pharmacopeia (BP) / United State Pharmacopeia (USP) requirements.
Implementation of cGMP (current Good Manufacturing Practices) regulations and
procedures are regularly monitored through internal quality auditing and
factory inspection. The pharmaceuticals manufacturing facility of the SPMC is equipped
with modern Japanese, German & Korean machines. The manufacturing facility has
an ultra-clean atmosphere and adheres to the requirement of cGMP as laid down
by the World Health Organization (WHO).
Products
·
Paracetamol Tablets BP 500mg (Blister)
·
Paracetamol Tablets BP 500mg
·
Indometacin Capsules BP 25mg
·
Diclofenac Sodium Tablets USP 50mg
·
Aluminium Hydroxide Tablets BP 500mg
·
Cimetidine Tablets BP 200 mg (Film Coated)
·
Famotidine Tablets USP 20mg (Blister)
·
Salbutamol Tablets BP 2mg
·
Salbutamol Tablets BP 4mg
·
Theophylline (Extended Release) Tablets SPMC 125mg
·
Rifampicin Capsules BP 150mg
·
Amoxiciilin Capsules BP 250mg
·
Amoxiciilin Capsules BP 250mg ( Blister )
·
Amoxiciilin Capsules BP 500mg
·
Amoxiciilin Capsules BP 500mg ( Blister )
·
Amoxiciilin TabletsUSP 125mg
·
Cloxacillin Capsules BP 250mg
·
Cloxacillin Capsules BP 500mg
·
Phenoxymethyl penicillin Tablets BP 125mg
·
Phenoxymethyl penicillin Tablets BP 250mg
·
Chloramphenicol Capsuls BP 250mg
·
Erythromycin Stearate Tablets BP 250mg(Film Coated)
·
Co-Trimoxazole Tablets BP 480mg ( Adult )
·
Co-Trimoxazole Tablets BP 120mg (Paediatric)
·
Carbamazepine Tablets BP 200mg
·
Gliclazide Tablets BP 80mg
·
Metformin Tablets BP 500mg (Film Coated)
·
Diethylcarbamazin Tablets BP 50mg
·
Enalapril Maleate Tablets USP 5mg
·
Verapamil Tablets BP 40mg ( Film Coated )
·
Diltiazim Hydrochloride Tablets 60mg USP
·
Propranolol Tablets BP 10mg
·
Propranolol Tablets BP 40mg
·
Atenolol Tablets BP 50mg (Film Coated)
·
Atenolol Tablets BP 50mg (Blister)
·
Chloroquine Phosphate Tablets BP 250mg
·
Primaquine Phosphate Tablets 7.5mg ( Film Coated )
·
Benzhexol Tablets BP 2mg
·
Trifluoperazine Tablets BP 5mg ( Film Coated )
·
Mebendazole Tablets USP 500mg
·
Mebendazole Tablets USP 500mg( Sachet )
·
Mebendazole Tablets USP 100mg
·
Prednisolone Tablets BP 5mg
·
Furosemide Tablets BP 40mg( Frusemide Tablets BP
40mg)
·
Bisacodyl Tablets BP 5mg ( Enteric Film Coated )
·
Ascorbic Acid Tablets BP 100mg
·
Folic Acid Tablets BP 1mg
·
Vitamin B Complex Tablets ( Vitamin B1 , B2 &
Nicotinamide)
·
Vitamin B Compound Strong Tablets BPC( Vitamin B1 ,
B2 , B6 & Nicotinamide)
Employees
300+ Approximately
Export Countries
Egypt, Myanmar,
Philippines, FIJI Islands, Bangladesh & Papua New Guinea and other
international markets
through the WHO & JICA recommendation
Associate Organizations
State Pharmaceutical
Corporation
Ministry
of Health
INFORMATION DENIED
Management non-co-operative (0094-11-2635353)
FOREIGN EXCHANGE RATES
|
Currency |
Unit
|
Indian Rupees |
|
US Dollar |
1 |
INR 64.03 |
|
|
1 |
INR 90.40 |
|
Euro |
1 |
INR 79.73 |
|
LKR |
1 |
INR 0.42 |
Note :
Above are approximate rates obtained from sources believed to be correct
INFORMATION DETAILS
|
Information
Gathered by : |
SPA |
|
|
|
|
Analysis Done by
: |
PRI |
|
|
|
|
Report Prepared
by : |
TPT |
RATING EXPLANATIONS
|
Credit Rating |
Explanation |
Rating Comments |
|
A++ |
Minimum Risk |
Business dealings permissible with minimum
risk of default |
|
A+ |
Low Risk |
Business dealings permissible with low
risk of default |
|
A |
Acceptable Risk |
Business dealings permissible with
moderate risk of default |
|
B |
Medium Risk |
Business dealings permissible on a regular
monitoring basis |
|
C |
Medium High Risk |
Business dealings permissible preferably
on secured basis |
|
D |
High Risk |
Business dealing not recommended or on
secured terms only |
|
NB |
New Business |
No recommendation can be done due to business
in infancy stage |
|
NT |
No Trace |
No recommendation can be done as the
business is not traceable |
NB is stated where there is insufficient information to facilitate rating. However, it is not to be considered as unfavourable.
This score serves as a reference to assess
SC’s credit risk and to set the amount of credit to be extended. It is
calculated from a composite of weighted scores obtained from each of the major
sections of this report. The assessed factors are as follows:
·
Financial
condition covering various ratios
·
Company
background and operations size
·
Promoters
/ Management background
·
Payment
record
·
Litigation
against the subject
·
Industry
scenario / competitor analysis
·
Supplier
/ Customer / Banker review (wherever available)
This report is issued at
your request without any risk and responsibility on the part of MIRA INFORM
PRIVATE LIMITED (MIPL) or its officials.

