|
|
|
MIRA INFORM REPORT
|
Report No. : |
492800 |
|
Report Date : |
22.02.2018 |
IDENTIFICATION DETAILS
|
Name : |
EVERTOGEN LIFE SCIENCES LIMITED (w.e.f. 30.01.2016) |
|
|
|
|
Formerly Known
As : |
OPTIMUS
GENERICS LIMITED |
|
|
|
|
Registered
Office : |
G V Chambers, 7-2-C8 and C8/2, IE Sanath Nagar, Hyderabad-500018,
Telangana |
|
Tel. No.: |
91-40-39219900 |
|
|
|
|
Country : |
India |
|
|
|
|
Financials (as
on) : |
31.03.2017 |
|
|
|
|
Date of
Incorporation : |
11.01.2013 |
|
|
|
|
Com. Reg. No.: |
36-085349 |
|
|
|
|
Capital
Investment / Paid-up Capital : |
INR 661.998 Million |
|
|
|
|
CIN No.: [Company Identification
No.] |
U24232TG2013PLC085349 |
|
|
|
|
IEC No.: [Import-Export Code No.] |
Not Available |
|
|
|
|
GSTN : [Goods & Service Tax
Registration No.] |
36AABCO7786R1ZE |
|
|
|
|
TAN No.: [Tax Deduction &
Collection Account No.] |
Not Available |
|
|
|
|
PAN No.: [Permanent Account No.] |
AABCO7786R |
|
|
|
|
Legal Form : |
A Closely Held Public Limited Liability Company |
|
|
|
|
Line of Business
: |
[Registered Activity] |
|
|
|
|
No. of Employees
: |
Not Available |
RATING & COMMENTS
(Mira Inform has adopted New Rating mechanism w.e.f. 23rd
January 2017)
|
MIRA’s Rating : |
A |
|
Credit Rating |
Explanation |
Rating Comments |
|
A |
Acceptable Risk |
Business dealings permissible with
moderate risk of default |
|
Maximum Credit Limit : |
USD 1780000 |
|
|
|
|
Status : |
Satisfactory |
|
|
|
|
Payment Behaviour : |
Slow but correct |
|
|
|
|
Litigation : |
Clear |
|
|
|
|
Comments : |
Subject is a joint venture between Gil Agrotech (India) Private
Limited and Oragnosyn Life Science Private Limited. It was incorporated in the year 2013 and engaged in the business of
product development, contract development, contract manufacturing and supply
of finished dosages. As per financials of March 2017, the company has reported growth of
149.77% in its revenue as compared to its previous year’s revenue and has
reported decent profit margin of 5.94%. Rating takes into consideration the company’s healthy capital base
along with comfortable debt coverage indicators and decent liquidity
position. Rating also takes into account the extensive of promoters and stable
customer base. However, the rating strength is partially offset by negative reserves
and working capital intensive nature of operations. Payment seems to be slow but correct. In view of aforesaid, the company can be considered for business
dealings at usual trade terms and conditions. |
NOTES :
Any query related to this report can be made
on e-mail : infodept@mirainform.com
while quoting report number, name and date.
ECGC Country Risk Classification List
|
Country Name |
Previous Rating (30.09.2017) |
Current Rating (31.12.2017) |
|
India |
A1 |
A1 |
|
Risk Category |
ECGC
Classification |
|
Insignificant |
A1 |
|
Low Risk |
A2 |
|
Moderately Low Risk |
B1 |
|
Moderate Risk |
B2 |
|
Moderately High Risk |
C1 |
|
High Risk |
C2 |
|
Very High Risk |
D |
EXTERNAL AGENCY RATING
|
Rating Agency Name |
CRISIL |
|
Rating |
Long term rating = BBB- |
|
Rating Explanation |
Moderate degree of safety and moderate
credit risk |
|
Date |
18.09.2017 |
|
|
|
|
Rating Agency Name |
CRISIL |
|
Rating |
Short term rating = A3 |
|
Rating Explanation |
Moderate degree of safety and higher credit
risk |
|
Date |
18.09.2017 |
RBI DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available RBI Defaulters’ list.
EPF (Employee Provident Fund) DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available EPF (Employee Provident Fund) Defaulters’ list as of
31-03-2016.
BIFR (Board for Industrial & Financial Reconstruction) LISTING
STATUS
Subject’s name is not listed as a Sick Unit in
the publicly available BIFR (Board for Industrial & Financial
Reconstruction) list as of 22.02.2018.
IBBI (Insolvency and Bankruptcy Board of India) LISTING STATUS
Subject’s name is not listed in the publicly
available IBBI (Insolvency and Bankruptcy Board of India) list as of report
date.
LOCATIONS
|
Registered Office : |
G V Chambers, 7-2-C8 and C8/2, IE Sanath Nagar, Hyderabad-500018,
Telangana, India |
|
Tel. No.: |
91-40-39219900 |
|
Fax No.: |
Not Available |
|
E-Mail : |
|
|
Website : |
|
|
|
|
|
Plant : |
Plot No.S-8, S-9, S-13 (Partly) S-14 (Partly), TSIIC, Pharma SEZ, Polepally [V], Jadcherla [M], Mahbubnagar-509301, Telangana, India |
|
Tel. No.: |
91-8542-300400 |
DIRECTORS
AS ON 31.03.2017
|
Name : |
Mr. Subbarao Kattamuri |
||||||||||||||||||||||||||||
|
Designation : |
Managing Director |
||||||||||||||||||||||||||||
|
Address : |
Plot No. 48, Nava Nirman Nagar Road No.71, Jubilee Hills, Hyderabad-500033, Telangana, India |
||||||||||||||||||||||||||||
|
Date of Birth/Age : |
10.03.1953 |
||||||||||||||||||||||||||||
|
Qualification : |
Chartered Accountant |
||||||||||||||||||||||||||||
|
Date of Appointment : |
11.01.2013 |
||||||||||||||||||||||||||||
|
PAN No.: |
AEDPK4078F |
||||||||||||||||||||||||||||
|
DIN No.: |
00107121 |
||||||||||||||||||||||||||||
|
Other Directorship:
|
|||||||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||||||
|
Name : |
Mr. Arun Prakash |
||||||||||||||||||||||||||||
|
Designation : |
Director |
||||||||||||||||||||||||||||
|
Address : |
C-88, Kirti Nagar, New Delhi-110015, India |
||||||||||||||||||||||||||||
|
Date of Birth/Age : |
09.03.1962 |
||||||||||||||||||||||||||||
|
Qualification : |
Master of Sciences, MSC |
||||||||||||||||||||||||||||
|
Date of Appointment : |
13.02.2015 |
||||||||||||||||||||||||||||
|
PAN No.: |
AFUPP9783Q |
||||||||||||||||||||||||||||
|
DIN No.: |
00287125 |
||||||||||||||||||||||||||||
|
Other Directorship:
|
|||||||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||||||
|
Name : |
Mr. Ashok Reddy Bodanapu |
||||||||||||||||||||||||||||
|
Designation : |
Director |
||||||||||||||||||||||||||||
|
Address : |
8-2-293/82/A/347/1, Road No.23, Jubilee Hills, Hyderabad-500033, Telangana, India |
||||||||||||||||||||||||||||
|
Date of Birth/Age : |
01.07.1958 |
||||||||||||||||||||||||||||
|
Qualification : |
B.com, LLB |
||||||||||||||||||||||||||||
|
Date of Appointment : |
13.02.2015 |
||||||||||||||||||||||||||||
|
PAN No.: |
ABRPB6267A |
||||||||||||||||||||||||||||
|
DIN No.: |
01848553 |
||||||||||||||||||||||||||||
|
Other Directorship:
|
|||||||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||||||
|
Name : |
Mr. Kalyan Nagururu |
||||||||||||||||||||||||||||
|
Designation : |
Director |
||||||||||||||||||||||||||||
|
Address : |
Polaris - 1702, Meenakshi Skylounge, Khanamet Hitex Charminar, Hyderabad- 500084, Telangana, India |
||||||||||||||||||||||||||||
|
Date of Birth/Age : |
08.04.1979 |
||||||||||||||||||||||||||||
|
Qualification : |
MBA |
||||||||||||||||||||||||||||
|
Date of Appointment : |
11.01.2013 |
||||||||||||||||||||||||||||
|
PAN No.: |
ABWPN9299F |
||||||||||||||||||||||||||||
|
DIN No.: |
02652051 |
||||||||||||||||||||||||||||
|
Other Directorship:
|
|||||||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||||||
|
Name : |
Mr. Manav Jassal |
||||||||||||||||||||||||||||
|
Designation : |
Director |
||||||||||||||||||||||||||||
|
Address : |
213-G, Randhir Singh Nagar, Ludhiana-141402, Punjab, India |
||||||||||||||||||||||||||||
|
Date of Birth/Age : |
04.11.1970 |
||||||||||||||||||||||||||||
|
Qualification : |
Post Graduate in Pharmaceutical Science |
||||||||||||||||||||||||||||
|
Date of Appointment : |
11.01.2013 |
||||||||||||||||||||||||||||
|
PAN No.: |
AJBPJ3289N |
||||||||||||||||||||||||||||
|
DIN No.: |
03059931 |
||||||||||||||||||||||||||||
|
Other Directorship:
|
|||||||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||||||
|
Name : |
Mr. Sundeep Kattamuri |
||||||||||||||||||||||||||||
|
Designation : |
Director |
||||||||||||||||||||||||||||
|
Address : |
Plot No.48, Navanirman Nagar, Jubilee Hills, Road No.71, Hyderabad-500033, Telangana, India |
||||||||||||||||||||||||||||
|
Date of Birth/Age : |
17.11.1983 |
||||||||||||||||||||||||||||
|
Qualification : |
Engineering Graduate and Post Graduate in Chemical Engineering |
||||||||||||||||||||||||||||
|
Date of Appointment : |
13.02.2015 |
||||||||||||||||||||||||||||
|
PAN No.: |
BRMPK2131G |
||||||||||||||||||||||||||||
|
DIN No.: |
03128742 |
||||||||||||||||||||||||||||
|
Other Directorship:
|
|||||||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||||||
|
Name : |
Mr. Sanjay Kumar Nayak |
||||||||||||||||||||||||||||
|
Designation : |
Director |
||||||||||||||||||||||||||||
|
Address : |
Qr. No. Mig/115, Kalinga Vihar, Patrapada, Bhubaneswar-751019, Odisha, India |
||||||||||||||||||||||||||||
|
Date of Birth/Age : |
15.06.1971 |
||||||||||||||||||||||||||||
|
Qualification : |
Graduation and MBA |
||||||||||||||||||||||||||||
|
Date of Appointment : |
13.02.2015 |
||||||||||||||||||||||||||||
|
PAN No.: |
AFSPN2806G |
||||||||||||||||||||||||||||
|
DIN No.: |
06466666 |
||||||||||||||||||||||||||||
|
Other Directorship:
|
|||||||||||||||||||||||||||||
KEY EXECUTIVES
|
Name : |
Mrs. Saritha Rani Premraj |
|
Designation : |
Company Secretary |
|
Address : |
H.No:230, Lal-Bazar, Trimulgherry, Hyderabad-500015, Telangana, India |
|
Date of Birth/Age : |
21.08.1987 |
|
Date of Appointment : |
01.02.2016 |
|
PAN No.: |
BBNPP2906D |
MAJOR SHAREHOLDERS
AS ON 31.03.2017
|
Names of Shareholders |
|
No. of Shares |
|
Subbarao Kattamuri |
|
94 |
|
Manav Jassal |
|
1 |
|
Sundeep Kattamuri |
|
1 |
|
Kalyan Nagururu |
|
1 |
|
Sushma Kattamuri |
|
1 |
|
Parvati Kattamuri |
|
1 |
|
Arun Prakash |
|
1 |
|
Oragnosyn Life Science Private Limited (Allotted 541126 shares) |
|
31675126 |
|
Gil Agrotech (India) Private Limited |
|
32399553 |
|
Glochem Industries Limited |
|
2125000 |
|
Total |
|
66199779 |
AS ON 28.09.2017
|
Equity Share
Break up |
Percentage |
|
Category |
|
|
Promoters - Bodies corporate |
100.00 |
|
Total |
100.00 |
BUSINESS DETAILS
|
Line of Business : |
[Registered Activity] |
||||||
|
|
|
||||||
|
Products / Services
: |
|
||||||
|
|
|
||||||
|
Brand Names : |
Not Available |
||||||
|
|
|
||||||
|
Agencies Held : |
Not Available |
||||||
|
|
|
||||||
|
Exports : |
Not Available |
||||||
|
|
|
||||||
|
Imports : |
Not Available |
||||||
|
|
|
||||||
|
Terms : |
Not Available |
PRODUCTION STATUS – (NOT AVAILABLE)
GENERAL INFORMATION
|
Suppliers : |
|
|||||||||||||||||||||||||||
|
|
|
|||||||||||||||||||||||||||
|
Customers : |
|
|||||||||||||||||||||||||||
|
|
|
|||||||||||||||||||||||||||
|
No. of Employees : |
Not Available |
|||||||||||||||||||||||||||
|
|
|
|||||||||||||||||||||||||||
|
Bankers : |
|
|||||||||||||||||||||||||||
|
|
|
|||||||||||||||||||||||||||
|
Facilities : |
|
|
Auditors : |
|
|
Name : |
Walker Chandiok and Company LLP Chartered Accountants |
|
Address : |
7th Floor, Block III, White House, Kundan Bagh, Begumpet, Hyderabad, Telangana, India |
|
PAN No.: |
AAAFW4298E |
|
|
|
|
Memberships : |
Not Available |
|
|
|
|
Collaborators : |
Not Available |
|
|
|
|
Holding Company : |
Gil Agrotech (India) Private Limited, India CIN No.: U01119TG2010PTC069551 |
|
|
|
|
Enterprises which
are owned, or have significant influence of or are partners with Key
management personnel and their relatives : |
CIN No.: U24100DL2011PTC223635
CIN No.: U24110TG1995PTC022303 |
CAPITAL STRUCTURE
AFTER 28.09.2017
Authorised Capital : INR 800.000 Million
Issued, Subscribed & Paid-up Capital : INR 677.003
Million
AS ON 28.09.2017
Authorised Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
70000000 |
Equity Shares |
INR 10/- each |
INR 700.000 Million |
|
|
|
|
|
Issued, Subscribed & Paid-up Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
66199779 |
Equity Shares |
INR 10/- each |
INR 661.998 Million |
|
|
|
|
|
FINANCIAL DATA
[all figures are
INR Million]
ABRIDGED
BALANCE SHEET
|
SOURCES OF FUNDS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
I.
EQUITY
AND LIABILITIES |
|
|
|
|
(1)Shareholders' Funds |
|
|
|
|
(a) Share Capital |
661.998 |
635.337 |
635.337 |
|
(b) Reserves and Surplus |
(52.641) |
(127.252) |
(30.845) |
|
(c) Money
received against share warrants |
0.000 |
0.000 |
0.000 |
|
|
|
|
|
|
(2) Share Application money pending
allotment |
14.000 |
0.000 |
0.000 |
|
Total
Shareholders’ Funds (1) + (2) |
623.357 |
508.085 |
604.492 |
|
|
|
|
|
|
(3)
Non-Current Liabilities |
|
|
|
|
(a) long-term borrowings |
242.104 |
281.384 |
240.929 |
|
(b) Deferred tax liabilities (Net) |
58.269 |
43.275 |
18.237 |
|
(c) Other long-term
liabilities |
0.000 |
0.000 |
0.000 |
|
(d) long-term
provisions |
2.197 |
2.262 |
1.652 |
|
Total Non-current
Liabilities (3) |
302.570 |
326.921 |
260.818 |
|
|
|
|
|
|
(4)
Current Liabilities |
|
|
|
|
(a) Short-term
borrowings |
225.156 |
117.209 |
21.051 |
|
(b) Trade
payables |
207.941 |
187.096 |
48.140 |
|
(c) Other
current liabilities |
113.833 |
156.440 |
93.341 |
|
(d) Short-term
provisions |
0.068 |
0.092 |
0.098 |
|
Total Current
Liabilities (4) |
546.998 |
460.837 |
162.630 |
|
|
|
|
|
|
TOTAL |
1472.925 |
1295.843 |
1027.940 |
|
|
|
|
|
|
II.
ASSETS |
|
|
|
|
(1)
Non-current assets |
|
|
|
|
(a) Fixed
Assets |
|
|
|
|
(i)
Tangible assets |
835.545 |
830.763 |
784.233 |
|
(ii)
Intangible Assets |
37.712 |
19.610 |
2.904 |
|
(iii) Tangible assets
capital work-in-progress |
0.343 |
0.024 |
5.720 |
|
(iv)
Intangible assets under development |
119.151 |
128.513 |
107.142 |
|
(b) Non-current Investments |
0.000 |
0.000 |
0.000 |
|
(c) Deferred tax assets (net) |
0.000 |
0.000 |
0.000 |
|
(d)
Long-term loans and advances |
18.729 |
10.964 |
11.779 |
|
(e) Other
Non-current assets |
1.670 |
2.021 |
2.373 |
|
Total Non-Current
Assets |
1013.150 |
991.895 |
914.151 |
|
|
|
|
|
|
(2)
Current assets |
|
|
|
|
(a)
Current investments |
0.000 |
0.000 |
0.000 |
|
(b)
Inventories |
196.143 |
239.139 |
88.574 |
|
(c) Trade
receivables |
212.492 |
27.970 |
15.059 |
|
(d) Cash and
bank balances |
8.441 |
10.192 |
0.709 |
|
(e) Short-term
loans and advances |
10.776 |
11.374 |
5.635 |
|
(f) Other
current assets |
31.923 |
15.273 |
3.812 |
|
Total
Current Assets |
459.775 |
303.948 |
113.789 |
|
|
|
|
|
|
TOTAL |
1472.925 |
1295.843 |
1027.940 |
PROFIT
& LOSS ACCOUNT
|
|
PARTICULARS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
|
SALES |
|
|
|
|
|
Total
Revenue from operations |
935.174 |
374.414 |
71.445 |
|
|
Other Income |
54.072 |
8.652 |
4.466 |
|
|
TOTAL |
989.246 |
383.066 |
75.911 |
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
Cost of Materials
Consumed |
435.045 |
213.436 |
37.189 |
|
|
Purchases of
Stock-in-Trade |
67.372 |
0.000 |
0.000 |
|
|
Changes in inventories of
finished goods, work-in-progress and Stock-in-Trade |
16.625 |
(56.618) |
(56.335) |
|
|
Employee benefit expense |
87.518 |
70.401 |
29.003 |
|
|
Other expenses |
186.137 |
120.274 |
45.700 |
|
|
TOTAL |
792.697 |
347.493 |
55.557 |
|
|
|
|
|
|
|
|
PROFIT/
(LOSS) BEFORE INTEREST, TAX, DEPRECIATION
AND AMORTISATION |
196.549 |
35.573 |
20.354 |
|
|
|
|
|
|
|
Less |
FINANCIAL
EXPENSES |
58.767 |
51.596 |
11.150 |
|
|
|
|
|
|
|
|
PROFIT
/ (LOSS) BEFORE TAX, DEPRECIATION AND AMORTISATION |
137.782 |
(16.023) |
9.204 |
|
|
|
|
|
|
|
Less |
DEPRECIATION/
AMORTISATION |
67.215 |
55.234 |
25.235 |
|
|
|
|
|
|
|
|
PROFIT/ (LOSS)
BEFORE TAX |
70.567 |
(71.257) |
(16.031) |
|
|
|
|
|
|
|
Less |
TAX |
14.995 |
25.150 |
18.235 |
|
|
|
|
|
|
|
|
PROFIT/ (LOSS)
AFTER TAX |
55.572 |
(96.407) |
(34.266) |
|
|
|
|
|
|
|
Add |
PREVIOUS YEARS’ BALANCE BROUGHT
FORWARD |
(127.252) |
(30.845) |
3.421 |
|
|
|
|
|
|
|
|
Balance
Carried to the B/S |
(71.680) |
(127.252) |
(30.845) |
|
|
|
|
|
|
|
|
Earnings / (Loss) Per
Share (INR) |
0.86 |
(1.52) |
(0.61) |
CURRENT MATURITIES
OF LONG TERM DEBT DETAILS
|
Particulars |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Current Maturities of Long term debt |
48.504 |
29.004 |
2.250 |
|
Net cash flows from (used in) operations |
19.968 |
39.304 |
72.271 |
|
Net cash flows from (used in) operating activities |
19.679 |
37.963 |
69.744 |
KEY
RATIOS
EFFICIENCY RATIOS
|
PARTICULARS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Average Collection Days (Sundry
Debtors / Income * 365) |
82.94 |
27.27 |
76.93 |
|
|
|
|
|
|
Account Receivables Turnover (Income /
Sunday Debtors) |
4.40 |
13.39 |
4.74 |
|
|
|
|
|
|
Average Payment Days (Sundry Creditors / Purchases * 365 Days) |
151.07 |
319.96 |
472.48 |
|
|
|
|
|
|
Inventory Turnover (Operating Income / Inventories) |
1.00 |
0.15 |
0.23 |
|
|
|
|
|
|
Asset Turnover (Operating Income / Net Fixed Assets) |
0.20 |
0.04 |
0.02 |
LEVERAGE RATIOS
|
PARTICULARS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Debt Ratio ((Borrowing
+ Current Liabilities) / Total Assets) |
0.57 |
0.60 |
0.39 |
|
|
|
|
|
|
Debt Equity Ratio (Total Liability / Networth) |
0.83 |
0.84 |
0.44 |
|
|
|
|
|
|
Current Liabilities to Networth (Current Liabilities / Net Worth) |
0.88 |
0.91 |
0.27 |
|
|
|
|
|
|
Fixed Assets to Networth (Net Fixed Assets / Networth) |
1.59 |
1.93 |
1.49 |
|
|
|
|
|
|
Interest Coverage Ratio (PBIT / Financial Charges) |
3.34 |
0.69 |
1.83 |
PROFITABILITY RATIOS
|
PARTICULARS |
|
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Net Profit Margin [(PAT
/ Sales) * 100] |
% |
5.94 |
(25.75) |
(47.96) |
|
|
|
|
|
|
|
Return on Total Assets ((PAT / Total Assets) * 100) |
% |
3.77 |
(7.44) |
(3.33) |
|
|
|
|
|
|
|
Return on Investment (ROI) ((PAT / Networth) * 100) |
% |
8.91 |
(18.97) |
(5.67) |
SOLVENCY RATIOS
|
PARTICULARS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Current Ratio (Current
Assets / Current Liabilities) |
0.84 |
0.66 |
0.70 |
|
|
|
|
|
|
Quick Ratio ((Current Assets – Inventories) / Current
Liabilities) |
0.48 |
0.14 |
0.16 |
|
|
|
|
|
|
G-Score Ratio Financial (Networth / Total Assets) |
0.42 |
0.39 |
0.59 |
|
|
|
|
|
|
G-Score Ratio Debt (Debts / Equity Capital) |
0.78 |
0.67 |
0.42 |
|
|
|
|
|
|
G-Score Ratio Liquidity (Total Current Assets / Total Current Liabilities) |
0.84 |
0.66 |
0.70 |
Total
Liability = Short-term Debt + Long-term Debt + Current Maturities of Long-term
debts
FINANCIAL ANALYSIS
[all figures are
INR Million]
DEBT EQUITY RATIO
|
Particular |
31.03.2015 |
31.03.2016 |
31.03.2017 |
|
|
(INR
In Million) |
(INR
In Million) |
(INR
In Million) |
|
Share Capital |
635.337 |
635.337 |
661.998 |
|
Reserves & Surplus |
(30.845) |
(127.252) |
(52.641) |
|
Money received against share
warrants |
0.000 |
0.000 |
0.000 |
|
Share Application money
pending allotment |
0.000 |
0.000 |
14.000 |
|
Net
worth |
604.492 |
508.085 |
623.357 |
|
|
|
|
|
|
Long-term borrowings |
240.929 |
281.384 |
242.104 |
|
Short term borrowings |
21.051 |
117.209 |
225.156 |
|
Current maturities of
long-term debts |
2.250 |
29.004 |
48.504 |
|
Total
borrowings |
264.230 |
427.597 |
515.764 |
|
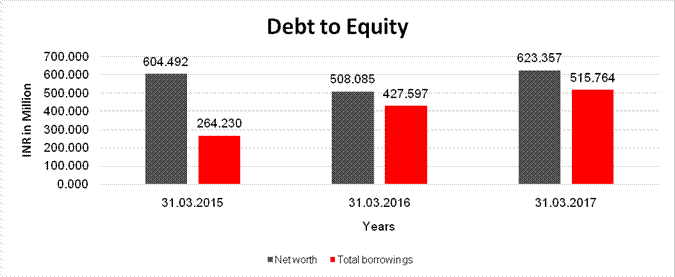
Debt/Equity
ratio |
0.437 |
0.842 |
0.827 |
YEAR-ON-YEAR GROWTH
|
Year
on Year Growth |
31.03.2015 |
31.03.2016 |
31.03.2017 |
|
|
(INR
In Million) |
(INR
In Million) |
(INR
In Million) |
|
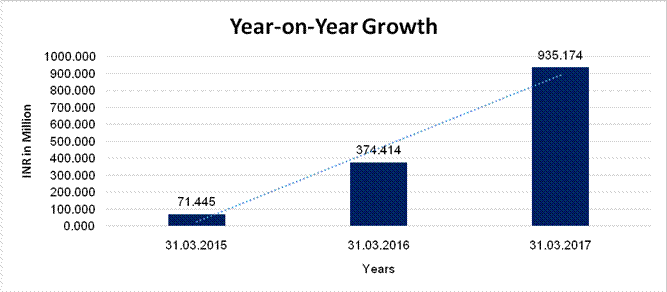
Sales |
71.445 |
374.414 |
935.174 |
|
|
|
424.059 |
149.770 |
NET PROFIT MARGIN
|
Net
Profit Margin |
31.03.2015 |
31.03.2016 |
31.03.2017 |
|
|
(INR
In Million) |
(INR
In Million) |
(INR
In Million) |
|
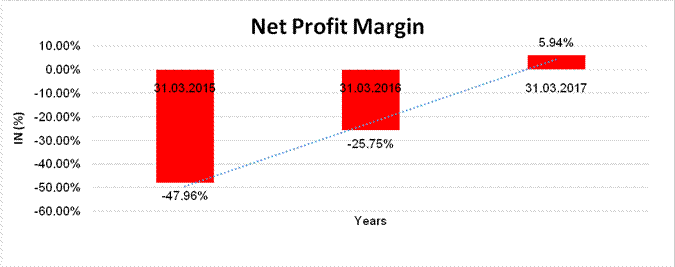
Sales |
71.445 |
374.414 |
935.174 |
|
Profit/ (Loss) |
(34.266) |
(96.407) |
55.572 |
|
|
(47.96%) |
(25.75%) |
5.94% |
LOCAL AGENCY FURTHER INFORMATION
|
Sr. No. |
Check list by
info agents |
Available in
Report (Yes/No) |
|
1 |
Year of establishment |
Yes |
|
2 |
Constitution of the entity -Incorporation
details |
Yes |
|
3 |
Locality of the entity |
Yes |
|
4 |
Premises details |
No |
|
5 |
Buyer visit details |
-- |
|
6 |
Contact numbers |
Yes |
|
7 |
Name of the person contacted |
No |
|
8 |
Designation of contact person |
No |
|
9 |
Promoter’s background |
Yes |
|
10 |
Date of Birth of Proprietor / Partners /
Directors |
Yes |
|
11 |
Pan Card No. of Proprietor / Partners |
Yes |
|
12 |
Voter Id Card No. of Proprietor / Partners |
No |
|
13 |
Type of business |
Yes |
|
14 |
Line of Business |
Yes |
|
15 |
Export/import details (if applicable) |
No |
|
16 |
No. of employees |
No |
|
17 |
Details of sister concerns |
Yes |
|
18 |
Major suppliers |
No |
|
19 |
Major customers |
No |
|
20 |
Banking Details |
Yes |
|
21 |
Banking facility details |
Yes |
|
22 |
Conduct of the banking account |
-- |
|
23 |
Financials, if provided |
Yes |
|
24 |
Capital in the business |
Yes |
|
25 |
Last accounts filed at ROC, if applicable |
Yes |
|
26 |
Turnover of firm for last three years |
Yes |
|
27 |
Reasons for variation <> 20% |
-- |
|
28 |
Estimation for coming financial year |
No |
|
29 |
Profitability for last three years |
Yes |
|
30 |
Major shareholders, if available |
Yes |
|
31 |
External Agency Rating, if available |
Yes |
|
32 |
Litigations that the firm/promoter
involved in |
-- |
|
33 |
Market information |
-- |
|
34 |
Payments terms |
No |
|
35 |
Negative Reporting by Auditors in the
Annual Report |
No |
COMPANY OVERVIEW
The Company is a Company domiciled in India and registered under the provisions of erstwhile the Companies Act, 1956 (the Act). The Company was a subsidiary of GIL Agrotech (India) Private Limited (GILAgro). Pursuant to the rights issues made in the current year on 27 September 2016, GIL Agro ceases to be the holding company of Evertogen Life Sciences Limited with effect from 27 September 2016. The Company is primarily engaged in manufacturing and selling of finished dosage formulations.
FINANCIAL PERFORMANCE
AND OPERATIONS REVIEW:
During the year, the Company had earned a Profit of INR 55.572 million against the loss of INR 96.407 million for the previous financial year. The Company had achieved a Turnover of INR 989.246 million (Previous Year: INR 383.066 million) and EBIDTA of INR 196.548 million (Previous year: INR 35.573 million) mainly arising from supply of goods under Licensing of dossiers, Contract manufacturing and service income from Licensing of Dossiers and providing Contract Manufacturing/development services to the customers. During the year the Company has successfully completed and obtained the regulatory approval from MHRA, WHO GMP and LOCAL GMP.
The Companys R&D trials and product development on various dossiers is progressing well and they are happy to inform that the Company has received approval for four dossiers during the year. The Companys investment into product development activities is significant for the growth of the Company and the Company has incurred INR 15.632 million (Previous Year: INR 40.532 million) towards dossier development cost in 2016-17. The Company has capitalized the Product development expenditure. According to the accounting policy of intangibles on Product development, the cost of development of dossiers will be amortized over the period of 5 years from the date on which the product is commercialized.
DEVELOPMENTS IN THE
INDUSTRY:
(i). GST Act (Tax reform)-
The introduction of GST would improve the operational efficiency by rationalizing the supply chain that could alone add 2 percent to the countrys Pharmaceutical industry. The biggest advantage for the companies would be the reduction in the overall transaction costs with the withdrawal of CST (Central Sales Tax). GST is also expected to lower the manufacturing cost. One more benefit likely to accrue due to GST is the reduction in the overall cost of technology. The import duty which is levied is not allowed as a tax credit under the present tax regulations. However, with GST this scenario will change. Under GST, duty charged on the import of such equipment and machinery would be allowed as a credit.
(ii). Policy support
Government unveiled Pharma Vision 2020 aimed at making India a global leader in end-to end drug manufacture, following are the highlights: Reduced approval time for new facilities to boost investments In this sector, 100 per cent FDI is allowed under automatic route Make in India initiative to promote manufacturing in India.
(iii). Trump Governance Effect (US Market)
Replacing Obamacare by new law (US Market): In March 2017, the US House of Representatives passed the 'American Health Care Act' to replace the PPACA (The Patient Protection and Affordable Care Act, nicknamed Obamacare). President Donald Trump had repeatedly made statements over reducing drug prices and increasing drug manufacturing in US. There is a talk of imposition of BAT, tax on imported drugs, to encourage domestic production in US. This is expected to affect the pricing of Indian drugs in US, when Trump administration is contemplating steps towards price reduction, any hike in price, even due to the tax, may affect the prospect of the Indian companies.
(iv). Increasing trend of manufacturing site audits by the USFDA
USFDA has focused on the quality aspect and in the last few months some Indian companies have received warning letters from USFDA for not following manufacturing standards at the production units. In a USFDA blog, Mary Lou Valdez, Association Commissioner for International Program, USFDA, has said that quality is a challenge for Indian pharma sector and has initiated actions accordingly. Out of the total 42 Good Manufacturing Practices (GMP) related warning letters issued by USFDA last year nine were addressed to Indian companies. It is probably the result of doubling of FDA inspections of pharma company sites since 2012. Edelweiss securities in its report apprehends that the number may go up even further as FDA would inspect 190 facilities that it had not in earlier five years. India has 572 USFDA compliant plants, the highest outside US.
(v). Brexit (Europe Market)
Brexit exclude one of the continents biggest buyer from the EUs single market from 2018 and consequently oblige companies to start obtaining separate certifications from agencies/authorities outside UK. It will also remove the expertise of UK government officials (with their traditionally industry friendly attitudes) from EU deliberations on pharmaceutical policy. And at the practical level, it will oblige EMA to quit its London headquarters and relocate elsewhere in Europe a prospect that is an administrative nightmare, but also, as its bosses openly admit, that it is already damaging staff motivation and impeding recruitment, with obvious implication for operational efficiency.
OUTLOOK FOR FUTURE:
The Company continues to focus and grow in each of the business segments, namely –
Out licensing of dossiers and supply of products
Contract manufacturing
Contract Development
Regulatory approvals are expected to be received during the year for some more products for which dossiers have been developed and the applications have been filed for approval during the previous years. Similarly, site variation approvals are expected to be received during the year for some more products, which have been validated and filed during the previous years. Company has also received and expected to receive regulatory approvals for some more products developed by the company during the previous year.
Commercial sales of the products are starting month after month for more and more products as and when the few more approvals are received and commercial orders are received. The sales revenue from the products are thus going up continuously month after month and the company is confident of achieving a turnover of over INR 1200.000 million in the current year.
Company continues to sign new contracts under each of the 3 business segments and the revenues/ incomes are expected to grow under the service income significantly during the year as well.
(i). Market Expansion in New Territories
Besides existing presence in regulated markets like EU, Canada, Australia and ongoing projects for US market, the company has started working on other semi-regulated markets like South East Asia/Asia Pacific and CIS/Russia with focused approach. Company has entered into business arrangement for New Zealand and Philippines markets through Licensing and supply agreement with reputed companies. Company has worked on market evaluation and entry strategies for Russia, Indonesia and Vietnam and is in discussion with few potential partners for commercialization.
(ii). Product Development
The company continues to lay the emphasis on developing new products with a view to launch the generic products immediately after the patent expiry and gain a significant/reasonable market share. Developing more and more new products and addition to its pipeline of products will help the Company in increasing the own products business segment which gives good margins/Profits.
Further, Company has screened about 30 molecules for development and is in the process of finalization to take up for development through market intelligence and evaluation of different data sources like Newport and Evaluate Pharma. Company has plans to subscribe IMS data for further due-diligence. Company has plans to further strengthen its portfolio for Anti-Diabetic (including Gliflozins and DPP-IV inhibitors), Cardio-vascular segment, Pain management and Non-Cytotoxic Oncology and Neurology.
INDEX OF CHARGE:
|
SNo |
SRN |
Charge Id |
Charge Holder Name |
Date of Creation |
Date of Modification |
Amount |
Address |
|
1 |
G73747297 |
10477914 |
Axis Bank Limited |
07/02/2014 |
29/12/2017 |
898000000.0 |
No. 6-3-879-B, G Pulla Reddy Building, Greenlands, Begumpet, Hyderabad-500016, Telangana, India |
FIXED ASSETS:
Tangible Asset:
- Land
- Building
- Plant and equipment
- Furniture and Fixture
- Office equipment
- Computer
- Vehicles
Intangible Asset:
· Computer software
CMT REPORT (Corruption, Money Laundering & Terrorism]
The Public Notice information has been collected from various sources
including but not limited to: The Courts,
1] INFORMATION ON
DESIGNATED PARTY
No records exist designating subject or any of its beneficial owners,
controlling shareholders or senior officers as terrorist or terrorist
organization or whom notice had been received that all financial transactions
involving their assets have been blocked or convicted, found guilty or against
whom a judgement or order had been entered in a proceedings for violating
money-laundering, anti-corruption or bribery or international economic or
anti-terrorism sanction laws or whose assets were seized, blocked, frozen or
ordered forfeited for violation of money laundering or international
anti-terrorism laws.
2] Court Declaration :
No records exist to suggest that subject is
or was the subject of any formal or informal allegations, prosecutions or other
official proceeding for making any prohibited payments or other improper
payments to government officials for engaging in prohibited transactions or
with designated parties.
3] Asset Declaration :
No records exist to suggest that the property or assets of the subject
are derived from criminal conduct or a prohibited transaction.
4] Record on Financial
Crime :
Charges or conviction
registered against subject: None
5] Records on Violation of
Anti-Corruption Laws :
Charges or investigation
registered against subject: None
6] Records on Int’l
Anti-Money Laundering Laws/Standards :
Charges or
investigation registered against subject: None
7] Criminal Records
No available
information exist that suggest that subject or any of its principals have been
formally charged or convicted by a competent governmental authority for any
financial crime or under any formal investigation by a competent government
authority for any violation of anti-corruption laws or international anti-money
laundering laws or standard.
8] Affiliation with
Government :
No record
exists to suggest that any director or indirect owners, controlling
shareholders, director, officer or employee of the company is a government
official or a family member or close business associate of a Government
official.
9] Compensation Package :
Our market
survey revealed that the amount of compensation sought by the subject is fair
and reasonable and comparable to compensation paid to others for similar
services.
10] Press Report :
No press reports / filings exists on
the subject.
CORPORATE GOVERNANCE
MIRA INFORM as part of its Due Diligence do provide comments on
Corporate Governance to identify management and governance. These factors often
have been predictive and in some cases have created vulnerabilities to credit
deterioration.
Our Governance Assessment focuses principally on the interactions
between a company’s management, its Board of Directors, Shareholders and other
financial stakeholders.
CONTRAVENTION
Subject is not known to have contravened any existing local laws,
regulations or policies that prohibit, restrict or otherwise affect the terms
and conditions that could be included in the agreement with the subject.
FOREIGN EXCHANGE RATES
|
Currency |
Unit
|
INR |
|
US Dollar |
1 |
INR 64.53 |
|
|
1 |
INR 90.13 |
|
Euro |
1 |
INR 79.86 |
INFORMATION DETAILS
|
Analysis Done by
: |
VIV |
|
|
|
|
Report Prepared
by : |
BHG |
SCORE FACTORS
|
DEMERIT POINTS |
|
|
|
--BANK CHARGES |
YES/NO |
YES |
|
--LITIGATION |
YES/NO |
NO |
|
--OTHER ADVERSE INFORMATION |
YES/NO |
NO |
|
MERIT POINTS |
|
|
|
--SOLE DISTRIBUTORSHIP |
YES/NO |
NO |
|
--EXPORT ACTIVITIES |
YES/NO |
NO |
|
--AFFILIATION |
YES/NO |
YES |
|
--LISTED |
YES/NO |
NO |
|
--OTHER MERIT FACTORS |
YES/NO |
YES |
RATING EXPLANATIONS
|
Credit Rating |
Explanation |
Rating Comments |
|
A++ |
Minimum Risk |
Business dealings permissible with minimum
risk of default |
|
A+ |
Low Risk |
Business dealings permissible with low
risk of default |
|
A |
Acceptable Risk |
Business dealings permissible with
moderate risk of default |
|
B |
Medium Risk |
Business dealings permissible on a regular
monitoring basis |
|
C |
Medium High Risk |
Business dealings permissible preferably
on secured basis |
|
D |
High Risk |
Business dealing not recommended or on
secured terms only |
|
NB |
New Business |
No recommendation can be done due to
business in infancy stage |
|
NT |
No Trace |
No recommendation can be done as the
business is not traceable |
NB is stated where there is insufficient information to facilitate rating. However, it is not to be considered as unfavourable.
This score serves as a reference to assess
SC’s credit risk and to set the amount of credit to be extended. It is
calculated from a composite of weighted scores obtained from each of the major
sections of this report. The assessed factors are as follows:
·
Financial
condition covering various ratios
·
Company
background and operations size
·
Promoters
/ Management background
·
Payment
record
·
Litigation
against the subject
·
Industry
scenario / competitor analysis
·
Supplier
/ Customer / Banker review (wherever available)
This report is issued at
your request without any risk and responsibility on the part of MIRA INFORM
PRIVATE LIMITED (MIPL) or its officials.

