|
|
|
MIRA INFORM REPORT
|
Report No. : |
494149 |
|
Report Date : |
28.02.2018 |
IDENTIFICATION DETAILS
|
Name : |
AKUMS DRUGS AND PHARMACEUTICALS LIMITED |
|
|
|
|
Registered
Office : |
304, Mohan Place, LSC, Saraswati Vihar, Delhi – 110034 |
|
Tel. No.: |
91-11-47511000 |
|
|
|
|
Country : |
India |
|
|
|
|
Financials (as
on) : |
31.03.2016 |
|
|
|
|
Date of Incorporation
: |
19.04.2004 |
|
|
|
|
Com. Reg. No.: |
55-125888 |
|
|
|
|
Capital
Investment / Paid-up Capital : |
INR 11.750 Million |
|
|
|
|
CIN No.: [Company Identification
No.] |
U24239DL2004PLC125888 |
|
|
|
|
IEC No.: [Import-Export Code No.] |
0505004429 |
|
|
|
|
GSTN : [Goods & Service Tax
Registration No.] |
07AAECA7090B1ZY |
|
|
|
|
TAN No.: [Tax Deduction &
Collection Account No.] |
Not Available |
|
|
|
|
PAN No.: [Permanent Account No.] |
AAECA7090B |
|
|
|
|
Legal Form : |
A Closely Held Public Limited Liability Company |
|
|
|
|
Line of Business
: |
Manufacturer
of Pharmaceuticals Formulation medicinal and Chemicals products. [Registered
Activity] |
|
|
|
|
No. of Employees
: |
Information denied by the management |
RATING & COMMENTS
(Mira Inform has adopted New Rating mechanism w.e.f. 23rd
January 2017)
|
MIRA’s Rating : |
A+ |
|
Credit Rating |
Explanation |
Rating Comments |
|
A+ |
Low Risk |
Business dealings permissible with low
risk of default |
|
Maximum Credit Limit : |
USD 19030000 |
|
|
|
|
Status : |
Good |
|
|
|
|
Payment Behaviour : |
Usually correct |
|
|
|
|
Litigation : |
Clear |
|
|
|
|
Comments : |
Subject was incorporated in the year 2004. It is a manufacturer and trader
of all kind of Pharmaceuticals products and Formulations. For the financial year ended 2016, the revenue of the company has
decreased by 41.63% but has maintain decent profit margin of 7.79%. The strong financial profile of the company marked by healthy net
worth base along with strong debt coverage indicators due to low debt balance
sheet profile. The company has favorable Earning per Share (EPS) of INR 374.16
against a face value (FV) of INR 10. However, rating strength are partially offset by unfavourable gap
between trade payables and trade receivables. Business is active. Payments seems to be usually correct. In view of aforesaid, the company can be considered good business
dealings at usual trade terms and conditions. Note: As per the Registrar of Companies date of balance sheet (i.e.
financial filed) is shown as 31.03.2017 but documents related to financial
for the year 31.03.2017 are not available from any sources. |
NOTES :
Any query related to this report can be made
on e-mail : infodept@mirainform.com
while quoting report number, name and date.
ECGC Country Risk Classification List
|
Country Name |
Previous Rating (30.09.2017) |
Current Rating (31.12.2017) |
|
India |
A1 |
A1 |
|
Risk Category |
ECGC
Classification |
|
Insignificant |
A1 |
|
Low Risk |
A2 |
|
Moderately Low Risk |
B1 |
|
Moderate Risk |
B2 |
|
Moderately High Risk |
C1 |
|
High Risk |
C2 |
|
Very High Risk |
D |
EXTERNAL AGENCY RATING
|
Rating Agency Name |
CARE |
|
Rating |
Cash Credit facilities = A+ |
|
Rating Explanation |
Adequate degree of safety and low credit
risk |
|
Date |
20.02.2017 |
|
|
|
|
Rating Agency Name |
CARE |
|
Rating |
Unallocated = A1+ |
|
Rating Explanation |
Very strong degree of safety and carry
lowest credit risk |
|
Date |
20.02.2017 |
RBI DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available RBI Defaulters’ list.
EPF (Employee Provident Fund) DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available EPF (Employee Provident Fund) Defaulters’ list as of
31-03-2016.
BIFR (Board for Industrial & Financial Reconstruction) LISTING STATUS
Subject’s name is not listed as a Sick Unit in
the publicly available BIFR (Board for Industrial & Financial
Reconstruction) list as of 28.02.2018.
IBBI (Insolvency and Bankruptcy Board of India) LISTING STATUS
Subject’s name is not listed in the publicly
available IBBI (Insolvency and Bankruptcy Board of India) list as of report
date.
INFORMATION DENIED BY
Tel. No. 91-11-47511000
(Office executive)
LOCATIONS
|
Registered / Corporate Office : |
304, Mohan Place, LSC, Saraswati Vihar, Delhi – 110034, India |
|
Tel. No.: |
91-11-47511000 |
|
Fax No.: |
91-11-27023256 |
|
E-Mail : |
|
|
Website : |
|
|
|
|
|
Unit-1 (Oral Solid Dosage Forms) : |
19, 20 and 21, Sector - 6A, I.I.E, SIDCUL, Ranipur, Haridwar – 249403, Uttarakhand, India |
|
|
|
|
Unit-2 (Liquid Orals) : |
22, Sector - 6A, I.I.E, SIDCUL, Ranipur, Haridwar -
249403, Uttarakhand, India |
|
|
|
|
Unit-3 (Injectables) : |
2, 3, 4 and 5, Sector – 6B, I.I.E, SIDCUL, Ranipur, Haridwar -
249403, Uttarakhand, India |
|
|
|
|
Unit-4 (Hormonal Preparations) : |
48, Sector – 6A, SIDCUL, Haridwar – 249403, Uttarakhand, India |
|
|
|
|
Unit-5 (Ointments and Cosmetics) : |
47, Sector-6A, SIDCUL, I.I.E., Ranipur, Haridwar – 249403, Uttarakhand, India |
|
|
|
|
Unit-6 (Ayurvedic, Food supplement, Nutraceuticals) : |
13, Sector 6A, SIDCUL, Haridwar – 249403, Uttarakhand, India |
|
|
|
|
Unit-7 (Oral solid dosages, injections, ointment and cosmetics) : |
Pure and Cure Healthcare Private Limited 26A, 27, 28, 29 and 30, Sector – 8A, IIE, Ranipur, Haridwar – 249403,
India |
|
|
|
|
Unit-8 (Cephalosporin Unit) : |
Malik Lifesciences Private Limited 16, Vardhaman Industrial Estate, Village Bahadarpur Saini, Roorkee,
Haridwar, India |
|
|
|
|
Unit-9 (Penicillin Unit) : |
Malik Lifesciences Private Limited 16, Vardhaman Industrial Estate, Village Bahadarpur Saini, Roorkee,
Haridwar, India |
|
|
|
|
Unit-10 (Under Construction) : |
Village-Tezpur, Mouza, Rampur, District - Kamrup, Guwahati (Assam),
India |
|
|
|
|
Centralized R and D Centre : |
Plot No. 6, Sector 6B, SIDCUL, Haridwar – 249403, India |
|
|
|
|
Mumbai Branch Office : |
503/504, Hubtown Solaris, Professor
NS Phadke Marg, Andheri (East), Mumbai – 400069, Maharashtra, India |
|
|
|
|
Ahmedabad Branch Office : |
405, Pushpak Tower, Ahmedabad – 380015, Gujarat, India |
|
|
|
|
Bangalore Branch Office : |
43/33, 59th A-Cross, 4th N Block, Rajaji Nagar,
Bangalore – 560010, Karnataka, India |
DIRECTORS
AS ON 31.03.2017
|
Name : |
Mr. Sanjeev Jain |
||||||||||||||||
|
Designation : |
Whole-time Director |
||||||||||||||||
|
Address : |
E - 1052, Saraswati Vihar, Delhi – 110034, India |
||||||||||||||||
|
Date of Birth/Age : |
11.04.1966 |
||||||||||||||||
|
Qualification : |
Master Degree |
||||||||||||||||
|
Date of Appointment : |
19.04.2004 |
||||||||||||||||
|
PAN No.: |
AAAPJ1897J |
||||||||||||||||
|
DIN No.: |
00323433 |
||||||||||||||||
|
Other Directorship:
|
|||||||||||||||||
|
|
|
||||||||||||||||
|
Name : |
Mr. Sandeep Jain |
||||||||||||||||
|
Designation : |
Whole-time Director |
||||||||||||||||
|
Address : |
E - 1052, Saraswati Vihar, Delhi – 110034, India |
||||||||||||||||
|
Date of Birth/Age : |
05.08.1967 |
||||||||||||||||
|
Qualification : |
Master Degree |
||||||||||||||||
|
Date of Appointment : |
19.04.2004 |
||||||||||||||||
|
PAN No.: |
AAAPJ1891Q |
||||||||||||||||
|
DIN No.: |
00323476 |
||||||||||||||||
|
Other Directorship:
|
|||||||||||||||||
|
|
|
||||||||||||||||
|
Name : |
Mr. Vijayapal Singh Rawat |
||||||||||||||||
|
Designation : |
Whole-time Director |
||||||||||||||||
|
Address : |
19-21, Sector-6A, Ranipur,
Haridwar – 249403, Uttarkhand, India |
||||||||||||||||
|
Date of Birth/Age : |
13.09.1961 |
||||||||||||||||
|
Qualification : |
Master in Science |
||||||||||||||||
|
Date of Appointment : |
27.09.2015 |
||||||||||||||||
|
PAN No.: |
AENPR5657K |
||||||||||||||||
|
DIN No.: |
06744017 |
||||||||||||||||
|
|
|
||||||||||||||||
|
Name : |
Mr. Narendra Dev Sachdeva |
||||||||||||||||
|
Designation : |
Director |
||||||||||||||||
|
Address : |
40/22, 3rd Floor, C R Park, Delhi – 110019, India |
||||||||||||||||
|
Date of Birth/Age : |
15.10.1950 |
||||||||||||||||
|
Qualification : |
B. Pharma |
||||||||||||||||
|
Date of Appointment : |
27.08.2004 |
||||||||||||||||
|
PAN No.: |
AYZPS4234M |
||||||||||||||||
|
DIN No.: |
00330005 |
||||||||||||||||
|
|
|
||||||||||||||||
|
Name : |
Mr. Nand Lal Kalra |
||||||||||||||||
|
Designation : |
Director |
||||||||||||||||
|
Address : |
4 BA 11, Jawahar Nagar, Jaipur – 302004, Rajasthan, India |
||||||||||||||||
|
Date of Birth/Age : |
05.02.1950 |
||||||||||||||||
|
Qualification : |
Masters In Science |
||||||||||||||||
|
Date of Appointment : |
01.06.2012 |
||||||||||||||||
|
PAN No.: |
AFTPK6497D |
||||||||||||||||
|
DIN No.: |
05268554 |
||||||||||||||||
|
|
|
||||||||||||||||
|
Name : |
Ms. Neena Vivek |
||||||||||||||||
|
Designation : |
Director |
||||||||||||||||
|
Address : |
3rd Floor, H-1/135-136, Sector - 16, Rohini, Delhi –
110085, India |
||||||||||||||||
|
Date of Appointment : |
13.03.2015 |
||||||||||||||||
|
DIN No.: |
07118115 |
||||||||||||||||
|
Other Directorship:
|
|||||||||||||||||
KEY EXECUTIVES
|
Name : |
Mr. Dharamvir Malik |
|
Designation : |
Company Secretary |
|
Address : |
RZ/A-1/20, 2nd Floor, Mahavir Enclave, Near Bank of
Baroda, New Delhi -110045, India |
|
Date of Birth/Age : |
21.06.1983 |
|
Date of Appointment : |
01.04.2014 |
|
PAN No.: |
AQPPM7439Q |
MAJOR SHAREHOLDERS
AS ON 31.03.2017
|
Names of Shareholders |
No. of Shares |
|
Sanjeev Jain |
587075 |
|
Lata Jain |
75 |
|
Sanjeev Jain J/w Arushi Jain |
300 |
|
Lata Jain J/w Umang Jain |
50 |
|
Sandeep Jain |
587150 |
|
Sandeep Jain J/w Kanishk Jain |
300 |
|
Manan Jain U/g Sandeep Jain |
50 |
|
Total |
1175000 |
AS ON 24.09.2017
|
Equity Share
Break up |
Percentage |
|
Category |
|
|
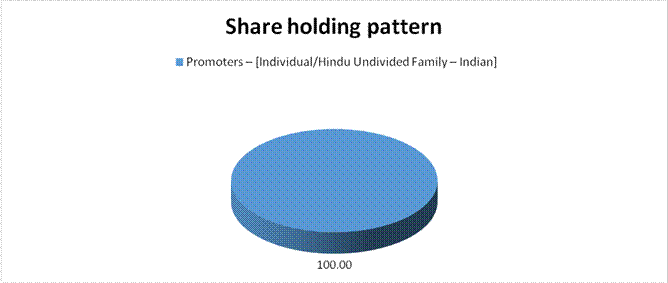
Promoters – [Individual/Hindu Undivided Family – Indian] |
100.00 |
|
Total |
100.00 |
BUSINESS DETAILS
|
Line of Business : |
Manufacturer
of Pharmaceuticals Formulation medicinal and Chemicals products. [Registered
Activity] |
||||||||
|
|
|
||||||||
|
Products / Services
: |
|
||||||||
|
|
|
||||||||
|
Brand Names : |
Not Available |
||||||||
|
|
|
||||||||
|
Agencies Held : |
Not Available |
||||||||
|
|
|
||||||||
|
Exports : |
Not Divulged |
||||||||
|
|
|
||||||||
|
Imports : |
Not Divulged |
||||||||
|
|
|
||||||||
|
Terms : |
Not Divulged |
PRODUCTION STATUS – (NOT AVAILABLE)
GENERAL INFORMATION
|
Suppliers : |
|
||||||||||||||||||||||
|
|
|
||||||||||||||||||||||
|
Customers : |
|
||||||||||||||||||||||
|
|
|
||||||||||||||||||||||
|
No. of Employees : |
Information denied by the management |
||||||||||||||||||||||
|
|
|
||||||||||||||||||||||
|
Bankers : |
|
||||||||||||||||||||||
|
|
|
||||||||||||||||||||||
|
Facilities : |
|
|
Auditors : |
|
|
Name : |
Anil Jain D.D and Company Chartered Accountants |
|
Address : |
611, |
|
PAN No.: |
AARFA8414D |
|
|
|
|
Memberships : |
Not Available |
|
|
|
|
Collaborators : |
Not Available |
|
|
|
|
Subsidiary Companies : (As on 2017) |
[CIN No.U99999DL1998PLC095297]
[CIN No.U24100DL2013PLC286876]
[CIN No.U63021DL2004PTC299408]
[CIN No.U74899DL1994PLC057323]
[CIN No.U15400DL2006PLC145272]
[CIN No.U24232DL2009PLC186841]
[CIN No.U24232DL2008PLC185686
[CIN No.U24232DL2005PTC266385]
[CIN No.U24233DL2013PLC262364] |
|
|
|
|
Subsidiary Companies : (As on 31.03.2016) |
[CIN No. U72900DL2010PLC206414]
[CIN No.U99999DL1998PLC095297]
[CIN No.U24230DL2006PLC147642]
[CIN No.U24100DL2013PLC286876]
[CIN No.U24297DL2012PLC243745]
[CIN No.U74899DL1994PLC057323]
[CIN No.U24232DL2008PLC185686
[CIN No.U63021DL2004PTC299408]
[CIN No.U15400DL2006PLC145272]
[CIN No.U24232DL2009PLC186841]
[CIN No.U24232DL2008PLC185686
[CIN No.U24232DL2005PTC266385]
[CIN No.U24233DL2013PLC262364 |
|
Enterprises which are
owned, or have significant influence of or are partners with Key Management personnel and their relatives: (As on 31.03.2016) |
|
CAPITAL STRUCTURE
AS ON 24.09.2017
Authorised Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
1200000 |
Equity shares |
INR 10/- each |
INR 12.000 Million |
|
|
|
|
|
Issued, Subscribed & Paid-up Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
1175000 |
Equity Shares |
INR 10/- each |
INR 11.750 Million |
|
|
|
|
|
FINANCIAL DATA
[all figures are
INR Million]
ABRIDGED
BALANCE SHEET (STANDLONE)
|
SOURCES OF FUNDS |
31.03.2016 |
31.03.2015 |
31.03.2014 |
|
|
|
|
|
|
I.
EQUITY AND LIABILITIES |
|
|
|
|
(1)Shareholders' Funds |
|
|
|
|
(a) Share Capital |
11.750 |
11.750 |
11.750 |
|
(b) Reserves &
Surplus |
6648.830 |
6206.186 |
5359.833 |
|
(c) Money received
against share warrants |
0.000 |
0.000 |
0.000 |
|
|
|
|
|
|
(2) Share Application
money pending allotment |
0.000 |
0.000 |
0.000 |
|
Total Shareholders’ Funds
(1) + (2) |
6660.580 |
6217.936 |
5371.583 |
|
|
|
|
|
|
(3) Non-Current
Liabilities |
|
|
|
|
(a) long-term borrowings |
120.000 |
240.000 |
15.122 |
|
(b) Deferred tax
liabilities (Net) |
225.862 |
220.293 |
234.976 |
|
(c) Other long term
liabilities |
20.600 |
21.252 |
22.671 |
|
(d) long-term provisions |
211.014 |
202.086 |
201.850 |
|
Total Non-current
Liabilities (3) |
577.476 |
683.631 |
474.619 |
|
|
|
|
|
|
(4) Current Liabilities |
|
|
|
|
(a) Short term borrowings |
379.388 |
701.496 |
1245.165 |
|
(b) Trade payables |
410.443 |
572.230 |
1110.557 |
|
(c) Other current
liabilities |
344.042 |
232.620 |
258.372 |
|
(d) Short-term provisions |
1.424 |
141.253 |
52.544 |
|
Total Current Liabilities
(4) |
1135.297 |
1647.599 |
2666.638 |
|
|
|
|
|
|
TOTAL |
8373.353 |
8549.166 |
8512.840 |
|
|
|
|
|
|
II.
ASSETS |
|
|
|
|
(1) Non-current assets |
|
|
|
|
(a) Fixed Assets |
|
|
|
|
(i) Tangible assets |
2283.231 |
2332.779 |
2554.277 |
|
(ii) Intangible Assets |
31.013 |
40.346 |
42.450 |
|
(iii) Capital
work-in-progress |
0.000 |
0.000 |
0.000 |
|
(iv) Intangible assets
under development |
0.000 |
0.000 |
0.000 |
|
(b) Non-current
Investments |
217.838 |
39.351 |
44.397 |
|
(c) Deferred tax assets
(net) |
0.000 |
0.000 |
0.000 |
|
(d) Long-term Loan and Advances |
3291.249 |
3632.717 |
2031.161 |
|
(e) Other Non-current
assets |
0.000 |
0.000 |
0.000 |
|
Total Non-Current Assets |
5823.331 |
6045.193 |
4672.285 |
|
|
|
|
|
|
(2) Current assets |
|
|
|
|
(a) Current investments |
0.000 |
0.000 |
0.000 |
|
(b) Inventories |
1200.226 |
918.212 |
1608.615 |
|
(c) Trade receivables |
1163.151 |
1425.133 |
1911.317 |
|
(d) Cash and cash
equivalents |
20.805 |
20.150 |
211.548 |
|
(e) Short-term loans and
advances |
165.840 |
140.478 |
109.075 |
|
(f) Other current assets |
0.000 |
0.000 |
0.000 |
|
Total Current Assets |
2550.022 |
2503.973 |
3840.555 |
|
|
|
|
|
|
TOTAL |
8373.353 |
8549.166 |
8512.840 |
PROFIT
& LOSS ACCOUNT (STANDLONE)
|
|
PARTICULARS |
31.03.2016 |
31.03.2015 |
31.03.2014 |
|
|
SALES |
|
|
|
|
|
Income |
5643.047 |
9667.950 |
8966.357 |
|
|
Other Income |
440.399 |
377.494 |
212.866 |
|
|
TOTAL |
6083.446 |
10045.444 |
9179.223 |
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
Cost of Materials
Consumed |
3802.236 |
6629.618 |
6121.779 |
|
|
Changes in inventories of
finished goods, work-in-progress and Stock-in-Trade |
(236.035) |
106.057 |
(33.781) |
|
|
Employees benefits
expense |
1020.444 |
1101.802 |
916.684 |
|
|
Exceptional Item |
30.000 |
0.000 |
0.000 |
|
|
Other expenses |
629.540 |
637.590 |
548.499 |
|
|
TOTAL |
5246.185 |
8475.067 |
7553.181 |
|
|
|
|
|
|
|
|
PROFIT/ (LOSS) BEFORE INTEREST, TAX, DEPRECIATION AND
AMORTISATION |
837.261 |
1570.377 |
1626.042 |
|
|
|
|
|
|
|
Less |
FINANCIAL EXPENSES |
77.499 |
138.495 |
76.518 |
|
|
|
|
|
|
|
|
PROFIT / (LOSS) BEFORE TAX,
DEPRECIATION AND AMORTISATION |
759.762 |
1431.882 |
1549.524 |
|
|
|
|
|
|
|
Less |
DEPRECIATION/
AMORTISATION |
221.271 |
237.063 |
143.542 |
|
|
|
|
|
|
|
|
PROFIT/ (LOSS) BEFORE TAX |
538.491 |
1194.819 |
1405.982 |
|
|
|
|
|
|
|
Less |
TAX |
98.847 |
348.466 |
349.034 |
|
|
|
|
|
|
|
|
PROFIT/ (LOSS) AFTER TAX
|
439.644 |
846.353 |
1056.948 |
|
|
|
|
|
|
|
|
Earnings / (Loss) Per
Share (INR) |
374.16 |
720.30 |
899.53 |
CURRENT MATURITIES
OF LONG TERM DEBT DETAILS
|
Particulars |
31.03.2016 |
31.03.2015 |
31.03.2014 |
|
Current Maturities of Long term debt |
120.000 |
21.252 |
60.490 |
|
Net cash flow from (used in) operations |
315.294 |
1841.094 |
753.489 |
|
Net cash flow from operating activities |
34.397 |
1680.833 |
1530.302 |
KEY
RATIOS
EFFICIENCY RATIOS
|
PARTICULARS |
31.03.2016 |
31.03.2015 |
31.03.2014 |
|
Average Collection Days (Sundry Debtors / Income * 365 Days) |
75.23 |
53.80 |
77.81 |
|
|
|
|
|
|
Account Receivables Turnover (Income / Sundry
Debtors) |
4.85 |
6.78 |
4.69 |
|
|
|
|
|
|
Average Payment Days (Sundry Creditors
/ Purchases * 365 Days) |
39.40 |
31.50 |
66.21 |
|
|
|
|
|
|
Inventory Turnover (Operating Income
/ Inventories) |
0.70 |
1.71 |
1.01 |
|
|
|
|
|
|
Asset Turnover (Operating Income
/ Net Fixed Assets) |
0.36 |
0.66 |
0.63 |
LEVERAGE RATIOS
|
PARTICULARS |
31.03.2016 |
31.03.2015 |
31.03.2014 |
|
Debt Ratio ((Borrowing + Current Liabilities) / Total
Assets) |
0.16 |
0.22 |
0.32 |
|
|
|
|
|
|
Debt Equity Ratio (Total Liability
/ Networth) |
0.09 |
0.15 |
0.25 |
|
|
|
|
|
|
Current Liabilities to Networth (Current
Liabilities / Net Worth) |
0.17 |
0.26 |
0.50 |
|
|
|
|
|
|
Fixed Assets to Networth (Net Fixed Assets
/ Networth) |
0.35 |
0.38 |
0.48 |
|
|
|
|
|
|
Interest Coverage Ratio (PBIT / Financial
Charges) |
10.80 |
11.34 |
21.25 |
PROFITABILITY RATIOS
|
PARTICULARS |
|
31.03.2016 |
31.03.2015 |
31.03.2014 |
|
Net Profit Margin [(PAT / Sales)
* 100] |
% |
7.79 |
8.75 |
11.79 |
|
|
|
|
|
|
|
Return on Total Assets ((PAT / Total Assets) * 100) |
% |
5.25 |
9.90 |
12.42 |
|
|
|
|
|
|
|
Return on Investment (ROI) ((PAT / Networth) * 100) |
% |
6.60 |
13.61 |
19.68 |
SOLVENCY RATIOS
|
PARTICULARS |
31.03.2016 |
31.03.2015 |
31.03.2014 |
|
Current Ratio (Current Assets / Current Liabilities) |
2.25 |
1.52 |
1.44 |
|
|
|
|
|
|
Quick Ratio ((Current Assets –
Inventories) / Current Liabilities) |
1.19 |
0.96 |
0.84 |
|
|
|
|
|
|
G-Score Ratio Financial (Networth / Total
Assets) |
0.80 |
0.73 |
0.63 |
|
|
|
|
|
|
G-Score Ratio Debt (Debts / Equity
Capital) |
52.71 |
81.94 |
112.41 |
|
|
|
|
|
|
G-Score Ratio Liquidity (Total Current
Assets / Total Current Liabilities) |
2.25 |
1.52 |
1.44 |
Total Liability = Short-term Debt + Long-term
Debt + Current Maturities of Long-term debts
FINANCIAL ANALYSIS
[all figures are
INR Million]
DEBT EQUITY RATIO
|
Particular |
31.03.2014 |
31.03.2015 |
31.03.2016 |
|
|
INR
In Million |
INR
In Million |
INR
In Million |
|
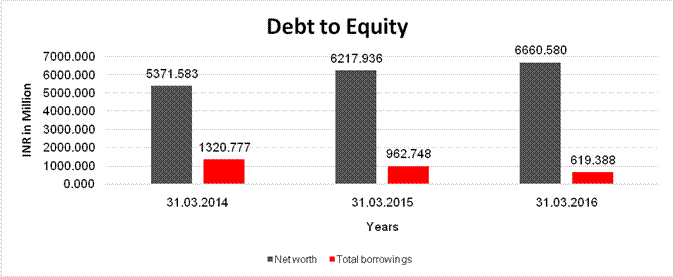
Share Capital |
11.750 |
11.750 |
11.750 |
|
Reserves & Surplus |
5359.833 |
6206.186 |
6648.830 |
|
Share Application money pending allotment |
0.000 |
0.000 |
0.000 |
|
Net
worth |
5371.583 |
6217.936 |
6660.580 |
|
|
|
|
|
|
Long Term borrowings |
15.122 |
240.000 |
120.000 |
|
Short Term borrowings |
1245.165 |
701.496 |
379.388 |
|
Current Maturities of Long term debt |
60.490 |
21.252 |
120.000 |
|
Total
borrowings |
1320.777 |
962.748 |
619.388 |
|
Debt/Equity
ratio |
0.246 |
0.155 |
0.093 |
YEAR-ON-YEAR GROWTH
|
Year
on Year Growth |
31.03.2014 |
31.03.2015 |
31.03.2016 |
|
|
INR
In Million |
INR
In Million |
INR
In Million |
|
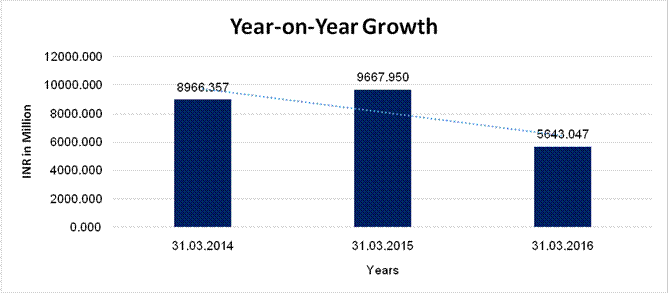
Sales
|
8966.357 |
9667.950 |
5643.047 |
|
|
|
7.825 |
(41.631) |
NET PROFIT MARGIN
|
Net
Profit Margin |
31.03.2014 |
31.03.2015 |
31.03.2016 |
|
|
INR
In Million |
INR
In Million |
INR
In Million |
|
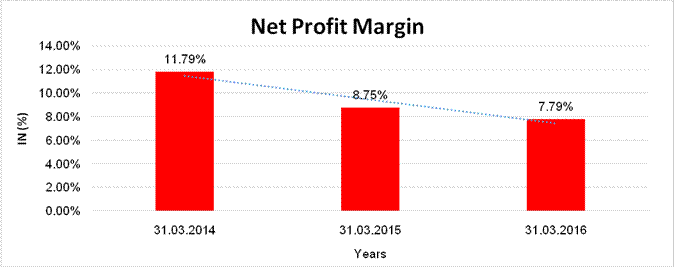
Sales
|
8966.357 |
9667.950 |
5643.047 |
|
Profit |
1056.948 |
846.353 |
439.644 |
|
|
11.79% |
8.75% |
7.79% |
FINANCIAL DATA
[all figures are
INR Million]
ABRIDGED
BALANCE SHEET (CONSOLIDATED)
|
SOURCES OF FUNDS |
|
31.03.2016 |
31.03.2015 |
|
|
|
|
|
|
I.
EQUITY AND LIABILITIES |
|
|
|
|
(1)Shareholders' Funds |
|
|
|
|
(a) Share Capital |
|
11.750 |
11.750 |
|
(b) Reserves &
Surplus |
|
6281.880 |
4928.448 |
|
(c) Money received
against share warrants |
|
0.000 |
0.000 |
|
|
|
|
|
|
(2) Share Application
money pending allotment |
|
0.000 |
0.000 |
|
(3) Minority interest |
|
121.111 |
26.253 |
|
Total Shareholders’ Funds
(1) + (2) |
|
6414.741 |
4966.451 |
|
|
|
|
|
|
(3) Non-Current
Liabilities |
|
|
|
|
(a) long-term borrowings |
|
516.842 |
778.750 |
|
(b) Deferred tax
liabilities (Net) |
|
394.062 |
254.026 |
|
(c) Other long term
liabilities |
|
60.070 |
63.345 |
|
(d) long-term provisions |
|
258.664 |
233.970 |
|
Total Non-current
Liabilities (3) |
|
1229.638 |
1330.091 |
|
|
|
|
|
|
(4) Current Liabilities |
|
|
|
|
(a) Short term borrowings |
|
1355.055 |
1562.530 |
|
(b) Trade payables |
|
1506.644 |
1532.371 |
|
(c) Other current
liabilities |
|
1035.379 |
829.678 |
|
(d) Short-term provisions |
|
65.381 |
197.446 |
|
Total Current Liabilities
(4) |
|
3962.459 |
4122.025 |
|
|
|
|
|
|
TOTAL |
|
11606.838 |
10418.567 |
|
|
|
|
|
|
II.
ASSETS |
|
|
|
|
(1) Non-current assets |
|
|
|
|
(a) Fixed Assets |
|
|
|
|
(i) Tangible assets |
|
4901.223 |
4390.005 |
|
(ii) Intangible Assets |
|
103.958 |
53.043 |
|
(iii) Capital
work-in-progress |
|
67.623 |
0.000 |
|
(iv) Intangible assets
under development |
|
37.557 |
0.000 |
|
(b) Non-current
Investments |
|
178.988 |
0.000 |
|
(c) Deferred tax assets
(net) |
|
0.000 |
0.000 |
|
(d) Long-term Loan and Advances |
|
512.533 |
366.448 |
|
(e) Other Non-current
assets |
|
|
|
|
Total Non-Current Assets |
|
5801.882 |
4809.496 |
|
|
|
|
|
|
(2) Current assets |
|
|
|
|
(a) Current investments |
|
0.000 |
0.000 |
|
(b) Inventories |
|
2887.218 |
2608.462 |
|
(c) Trade receivables |
|
2591.228 |
2727.598 |
|
(d) Cash and cash
equivalents |
|
136.813 |
118.703 |
|
(e) Short-term loans and
advances |
|
128.690 |
95.569 |
|
(f) Other current assets |
|
61.007 |
58.739 |
|
Total Current Assets |
|
5804.956 |
5609.071 |
|
|
|
|
|
|
TOTAL |
|
11606.838 |
10418.567 |
PROFIT
& LOSS ACCOUNT (CONSOLIDATED)
|
|
PARTICULARS |
31.03.2016 |
31.03.2015 |
|
|
|
SALES |
|
|
|
|
|
Income |
|
16509.143 |
14642.695 |
|
|
Other Income |
|
56.301 |
68.501 |
|
|
TOTAL |
|
16565.444 |
14711.196 |
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
Cost of Materials
Consumed |
|
8129.449 |
7686.360 |
|
|
Purchases of
Stock-in-Trade |
|
1302.363 |
957.580 |
|
|
Changes in inventories of
finished goods, work-in-progress and Stock-in-Trade |
|
(266.825) |
(301.241) |
|
|
Employees benefits
expense |
|
3146.435 |
2681.894 |
|
|
Other expenses |
|
2766.960 |
2383.283 |
|
|
TOTAL |
|
15078.382 |
13407.876 |
|
|
|
|
|
|
|
|
PROFIT/ (LOSS) BEFORE INTEREST, TAX, DEPRECIATION AND
AMORTISATION |
|
1487.062 |
1303.320 |
|
|
|
|
|
|
|
Less |
FINANCIAL EXPENSES |
|
215.227 |
225.654 |
|
|
|
|
|
|
|
|
PROFIT / (LOSS) BEFORE
TAX, DEPRECIATION AND AMORTISATION |
|
1271.835 |
1077.666 |
|
|
|
|
|
|
|
Less |
DEPRECIATION/
AMORTISATION |
|
384.011 |
310.323 |
|
|
|
|
|
|
|
|
PROFIT/ (LOSS) BEFORE TAX |
|
887.824 |
767.343 |
|
|
|
|
|
|
|
Less |
TAX |
|
442.472 |
384.878 |
|
|
|
|
|
|
|
|
PROFIT/ (LOSS) AFTER TAX
|
|
445.352 |
382.465 |
|
|
|
|
|
|
|
|
Profit
(Loss) of minority interest |
|
169.408 |
52.834 |
|
|
|
|
|
|
|
|
TOTAL PROFIT (LOSS) FOR
PERIOD |
|
614.761 |
430.653 |
|
|
|
|
|
|
|
|
Earnings / (Loss) Per
Share (INR) |
|
379.02 |
325.5 |
LOCAL AGENCY FURTHER INFORMATION
|
Sr. No. |
Check list by
info agents |
Available in
Report (Yes/No) |
|
1 |
Year of establishment |
Yes |
|
2 |
Constitution of the entity -Incorporation
details |
Yes |
|
3 |
Locality of the entity |
Yes |
|
4 |
Premises details |
Yes |
|
5 |
Buyer visit details |
-- |
|
6 |
Contact numbers |
Yes |
|
7 |
Name of the person contacted |
No |
|
8 |
Designation of contact person |
Yes |
|
9 |
Promoter’s background |
Yes |
|
10 |
Date of Birth of Proprietor / Partners /
Directors |
Yes |
|
11 |
Pan Card No. of Proprietor / Partners |
Yes |
|
12 |
Voter Id Card No. of Proprietor / Partners |
No |
|
13 |
Type of business |
Yes |
|
14 |
Line of Business |
Yes |
|
15 |
Export/import details (if applicable) |
No |
|
16 |
No. of employees |
No |
|
17 |
Details of sister concerns |
Yes |
|
18 |
Major suppliers |
No |
|
19 |
Major customers |
No |
|
20 |
Banking Details |
Yes |
|
21 |
Banking facility details |
Yes |
|
22 |
Conduct of the banking account |
-- |
|
23 |
Financials, if provided |
Yes |
|
24 |
Capital in the business |
Yes |
|
25 |
Last accounts filed at ROC, if applicable |
No |
|
26 |
Turnover of firm for last three years |
Yes |
|
27 |
Reasons for variation <> 20% |
-- |
|
28 |
Estimation for coming financial year |
No |
|
29 |
Profitability for last three years |
Yes |
|
30 |
Major shareholders, if available |
Yes |
|
31 |
External Agency Rating, if available |
Yes |
|
32 |
Litigations that the firm/promoter
involved in |
-- |
|
33 |
Market information |
-- |
|
34 |
Payments terms |
No |
|
35 |
Negative Reporting by Auditors in the
Annual Report |
No |
OPERATING RESULTS
The Standalone revenue (Sales and other incomes) of Akums Drugs and Pharmaceuticals Limited (Akums) has been INR 6083.446 million (Previous Year INR 10045.444 million). Sales have been to INR 5643.047 million (Previous Year INR 9667.950 million). Other Income increased to INR 440.399 million from INR 377.494 million.
The EBIDTA had been INR 837.261 million (Previous year 1570.377 million). The Profit before tax had been INR 538.491 million (Previous year 1194.819 million). The net profits after tax (including capital subsidy) had been INR 439.644 million (Previous Year INR 846.353 Million).
The financials for Financial Year 2015-16 clearly reflect the impact of expiry of excise exemption limit of Plant-1 of the Company, which has been the biggest manufacturing and revenue generating unit of the Company and was contributing approximately 70% revenue of the Company.
Therefore, financial of the current year are not strictly comparable to the financial numbers for Financial Year 2014-15. The Company would upgrade its Plant-I for export purpose.
STATE OF AFFAIRS OF
THE COMPANY
Akums undertakes contract manufacturing of pharmaceuticals formulations in almost all therapeutic segments and in the form of tablets, Capsules, Soft gelatin, dry syrup, liquid orals, injections dry and liquids, pre-filled syringes and eye & ear drops, hormones, ointments, cosmetics, Ayurveda’s, herbals, nutraceuticals and other similar products for domestic as well as multi-national Pharmaceutical Companies in India and abroad.
MANAGEMENT DISCUSSION
& ANALYSIS REPORT
SECTOR STRUCTURE AND
DEVELOPMENT
GLOBAL PHARMACEUTICAL
SECTOR
Standing at the midpoint of 2016, the pharmaceutical sector looks in very good shape, the market for medicines could be worth nearly $1.6 trillion by 2020, indeed, it could be worth even more. Demand for pharma’s products is rising dramatically, as the global population increasing same time it being more sedentary. In 2010, there were an estimated 6.9 billion people. By 2020, there will be more than 7.6 billion, and, if present lifestyle would continue, people would be more health conscious.
More than 20% will be overweight or obese; and more than 13% will be 60 or older. Thus number of people reaching old age is mounting, and the prevalence of a chronic diseases doubles every five years after the age of 65.
In short, there are more people – and more sick or elderly people – in the world today than ever before. More people have access to affordable healthcare than ever before, and, in coming years, access to healthcare may well be regarded everywhere as a basic human right.
CHALLENGES FOR THE
GLOBAL PHARMACEUTICAL SECTOR
Some of the challenges the pharmaceutical sector faces include research and development failures, changes in regulatory aspects, patent, litigation, patent, and foreign currency movements.
However, the major challenge is the drug pricing, which has been a major global debate. As the population over age 65 is increasing. A higher incidence of lifestyle-related diseases is further fueling governments’ healthcare expenditure worldwide.
The governments across the world are trying to curb these healthcare costs either by drug price revisions, asking for higher discounts and rebates to the manufacturers, or by promoting generic drugs.
However, with the launch of innovative drugs, pharmaceutical companies can bring in sales growth. The life-saving abilities of the drugs fetch higher prices and wider margins.
INDIAN PHARMACEUTICAL
SECTOR
Indian pharmaceutical sector accounts for about 2.4 per cent of the global pharmaceutical sector in value terms and 10 per cent in volume terms and is expected to expand at a Compound Annual Growth Rate (CAGR) of 15.92 per cent to US$ 42 billion by 2020 from US$ 20 billion in 2015.
With 71 per cent market share, generic drugs form the largest segment of the Indian pharmaceutical sector. The country accounts for the second largest number of Abbreviated New Drug Applications (ANDAs) and is the world’s leader in Drug Master Files (DMFs) applications with the US.
Indian drugs are exported to more than 200 countries in the world, with the US as the key market. Generic drugs account for 20 per cent of global exports in terms of volume, making the country the largest provider of generic medicines globally and expected to expand even further in coming years. Pharmaceuticals Exports Promotion Council (Pharmexcil) expects pharmaceutical exports to reach US$ 26 billion in 2016.
The Indian pharma sector, which is expected to grow over 15 per cent per annum between 2015 and 2020, will outperform the global pharma sector, which is set to grow at an annual rate of 5 per cent between the same periods.
India enjoys an important position in the global pharmaceuticals sector. The country also has a large pool of scientists and engineers who have the potential to steer the industry ahead to an even higher level.
ROAD AHEAD
The Indian pharmaceutical market size is expected to grow to US$ 100 billion by 2025, driven by increasing consumer spending, rapid urbanization, and raising healthcare insurance among others.
Going forward, better growth in domestic sales would also depend on the ability of companies to align their product portfolio towards chronic therapies for diseases such as such as cardiovascular, anti-diabetes and anti-depressants that are on the rise. In 2016, growth is expected to continue to be healthy, as the momentum continues and improving economic growth allows individuals more legroom to spend on health.
The Indian government has taken many steps to reduce costs and bring down healthcare expenses. Speedy introduction of generic drugs into the market has remained in focus and is expected to benefit the Indian pharmaceutical companies.
In addition, the thrust on rural health programmers and lifesaving drugs and preventive vaccines also augurs well for the pharmaceutical companies.
Yet pharma also faces some enormous obstacles. Innovation has declined, the regulations are becoming more onerous and market conditions are getting harsher, as healthcare costs everywhere keep rising.
The regulatory environment is simultaneously getting more rigorous.
INDUSTRY STRUCTURE
AND DEVELOPMENT
CONTRACT
MANUFACTURING IN PHARMA INDUSTRY
The pharmaceutical companies are looking for various opportunities to reduce cost, improve efficiencies, improve pipeline and reduce the time-to-market. To reduce cost, the pharmaceutical companies are adopting different strategies like outsourcing (in areas like manufacturing and product developments and other functions). The marketing companies have specialized themselves in marketing and exports and left the activity of product development and manufacturing to contract manufacturing, who have developed their expertise in those areas.
DRIVERS FOR CONTRACT
MANUFACTURING
The key objectives and drivers for outsourcing the manufacturing functions for pharma marketing companies are as follows:
- Improving cost
- Better capacity management with flexibility to handle business needs
- Effectively utilizing internal core expertise and other resources (including financial) and more opportunity to focus on core competencies of marketing
- Quick time-to-market
- Leveraging external expertise (and addressing the challenge of non-availability of internal resources and capabilities)
- Investing less capital (and leveraging financial resource in other core activities)
- Leveraging vendor’s innovative, state-of-the-art process and production technologies to support the rapid technical transfer of products from R and D to full scale commercial manufacturing.
The pharmaceutical contract manufacturing (PCM) industry and Contract Research and Manufacturing Service (CRAMS) continues to grow at a steady pace, driven by cost pressures, stringent regulatory requirements and patent expires.
Contract manufacturing approach is adopted for improving cost, process and capabilities as Contract Manufacturing Organizations (CMO) can leverage their skills and competencies to improve process, avail volume discounts on raw material on account of economies of scale. The knowledge and experience of CMO helps marketing companies to get the best practices and leverage experiences of contract manufacturing companies for their products.
Apart from the above CMO provides a range of services like product development, obtaining DCGI approvals, lesser costs due to manufacturing in excise free zones and other related services. CMO employs latest manufacturing facilities and techniques and has the capability to understand and deploy multiple regulatory requirements. CMO brings maturity while integrating with customer process and system. To provide efficient and
Integrated services, CMO integrates with supply chain activities of the pharmaceutical companies.
OUTSOURCING STRATEGY
DECISION FRAMEWORK
Their decision to outsource the marketing companies base on the following factors:
- Company internal capabilities and capacity
- Product development related to capabilities of manufacture & ability to take risks
- Strategic importance; and availability of pharmaceutical formulations at lower price because of manufacturing in tax free zones.
- Cost of internal operations; being higher than contract manufacturer.
- Availability of internal resources (including Financial)
- Stage of drug lifecycle
PREVAILING TREND:
The cost of drug development has increased in last few years forcing pharmaceutical companies to adopt new approaches towards drug discovery and delivery. As part of various core and non-core operations, companies are increasingly deploying outsourcing strategies to increase revenues through faster and cost effective drug development. In addition to outsourcing other non-core and secondary functions, most of the pharmaceutical companies outsourcing manufacturing functions. Consequently, cost is controlled with flexible model, without making any large capital investment and also allows companies to invest and focus resources on their core competencies to generate more value, through their expertise in marketing.
Most pharma companies evaluate its manufacturing strategy based on new options, challenges, internal capabilities and constraints.
Recently, a large pharmaceutical companies announced that they will outsource all the manufacturing functions of the company within the next 10 years as they do not consider manufacturing as their “core” activity, but “innovation and brand-building in marketing” as core activity.
The Global Pharmaceuticals Contract Manufacturing market is worth about USD 60 billion. This market is highly fragmented.
Contract manufacturing in India is forecast to expand at a CAGR of 20%. Big Pharma Companies are increasingly outsourcing manufacturing to low cost destinations due to cost and margin pressures, India is one of the best market for Pharma Contract Manufacturing, which offer skilled manpower and a robust manufacturing infrastructure.
AKUMS POSITION IN
PHARMA CONTRACT MANUFACTURING INDUSTRY
Akums is India’s largest contract manufacturing company engaged in manufacturing of pharmaceuticals formulations, Ayurveda / herbal preparations, hormones, nutraceuticals and more, in all dosage forms like tablets, capsules, injectable, Ear and Eye drops, hormonal preparations, liquid orals, creams & ointments, dry syrup etc. Akums manufacturers over 10% of India’s total medicine consumptions.
Quality, innovation and customer satisfaction are virtues at Akums. The approach is technology driven to serve the emerging needs in the ever expanding market. The 10 (Ten) state of art manufacturing facilities dedicated to separate dosage forms across therapeutic segments are equipped with sophisticated and ultra-modern equipments & machines, with huge manufacturing capacities in all sections Akums has an acumen to innovate and dedication to provide quality products.
Akums carry out various steps for its customers like research, testing, regulatory approvals, manufacturing, customization packaging & logistics support etc. which prove true the concept of “one stop shop”. Akums has state of the art technology, equipment, automation and robust Health, Safety & Environment procedures to ensure the wellbeing of all employees and protection of the environment; and to provide best quality medicines to the customers.
In their continuous pursuit of a healthier world, we are continuously working toward redesigning global healthcare while setting new goals for ourselves.
AKUMS’ BUSINESS MODEL
Akums focus rests on specialized products with limited reliance on commodity products. The company’s strengths lies in developing new formulations, conducting Stability studies, undertaking Bio-equivalence studies, clinical trials, obtaining Drug Controller General of India’s (DCGI) approval for manufacturing and marketing new Fixed Dose Combinations (FDCs) and formulations; and thereafter offering new formulations with Akums own technology to its customers under marketing Company’s brand names.
Business Model Includes –
a. Contract Research and Manufacturing Services (CRAMS) in Medicines.
b. Research and Development of New Formulations and obtaining DCGI approval
c. Technology Development and Transfer to others
d. Technical Collaboration and Joint Ventures
e. Offering tailor made Specialized products
f. Arranging Clinical Trials and Bio-equivalence Studies
g. Research and Development of formulations based on N.D.D.S
h. Patenting of Innovative Drugs
i. Development in Process and Packaging
j. Sustained Improvement in Product Quality.
k. Loan License
l. Institutional Business
Akums manufacture Pharmaceutical Formulations in most of the Therapeutic Segments and in almost all dosage forms, covering about 4000 formulations. It is a research based company manufacturing products on contract basis mostly with its own technology.
INVESTING FOR FUTURE:
Akums continue to strengthen and build its leadership position in pharma contract manufacturing business segments. The Company has large 10 manufacturing facilities at Haridwar, out of which 9 facilities are eligible for excise exemption which differ from unit to unit. As a part of the focus towards continues serve its customers for speedy delivery of finished products at best possible competitive rates, the Company is under process to establish a large manufacturing facility at Guwahati (Assam) in order to continuously provide excise benefits to its valuable clientele.
OUTLOOK
An analysis by IMS shows that the Indian Pharmaceuticals Market will likely grow between 10% and 11% year on year to more than INR 0.100 million by 2018. This growth will be driven by increasing affordability, increasing access particularly as private providers expand to lower tier towns and cities and favorable demographic factors – an overall increase in Indian population as well as increased prevalence of chronic diseases.
INDEX OF CHARGE:
|
SNo |
SRN |
Charge Id |
Charge Holder Name |
Date of Creation |
Date of Modification |
Date of Satisfaction |
Amount |
Address |
|
1 |
C63041800 |
10556644 |
STANDARD CHARTERED INVESTMENTS AND LOANS (INDIA) LIMITED |
24/03/2015 |
01/09/2015 |
- |
300000000.0 |
Floor No. 6, Crescenzo Building,
C-38/39,"G" Block, Bandra Kurla Complex, Bandra (East), Mumbai
- 400051, Maharashtra, India |
|
2 |
C77442853 |
10217825 |
Standard Chartered Bank |
27/04/2010 |
28/01/2016 |
- |
150000000.0 |
Credit Risk Controlnarain Manzil, 23
Barakhamba Road, New Delhi - 110001, India |
|
3 |
G32268138 |
10019466 |
State Bank of India |
25/08/2006 |
29/12/2016 |
- |
600000000.0 |
Industrial Finance Branch, 14th Floor, Jawahar Vyapar Bhawan, 1, Tolstoy Marg, New Delhi – 110001, India |
|
4 |
C53455101 |
10217291 |
Standard Chartered Bank |
26/03/2010 |
21/12/2010 |
12/05/2015 |
250000000.0 |
Credit Risk Controlnarain Manzil, 23
Barakhamba Road, New Delhi - l110001, India |
|
5 |
B83390161 |
10120161 |
ICICI BANK LIMITED |
21/08/2008 |
- |
29/08/2013 |
200000000.0 |
Landmarkrace Cource Circle, Alkapuri, Vadodara-390015, Gujarat, India |
|
6 |
B35066018 |
10032291 |
ICICI BANK LIMITED |
29/11/2006 |
- |
29/02/2012 |
200000000.0 |
Landmarkrace Cource Circle, Alkapuri, Vadodara-390015, Gujarat, India |
|
7 |
B18495754 |
10050600 |
State Bank of India |
25/08/2006 |
27/01/2011 |
08/08/2011 |
92100000.0 |
Commercial Branch M-47, Connaught Circus, New Delhi-110001, India |
|
8 |
A59661512 |
10107784 |
STANDARD CHARTERED BANK |
23/06/2008 |
- |
01/04/2009 |
150000000.0 |
Credit Risk Control Narain Manzil, 23 Barakhamba Road, New Delhi-110001, India |
|
9 |
A04341079 |
90039296 |
STATE BANK OF INDIA |
30/08/2004 |
- |
25/08/2006 |
132000000.0 |
Wazirpur Industrial Area, Delhi-110052, India |
|
10 |
A04340576 |
80011044 |
STATE BANK OF INDIA |
30/08/2004 |
- |
25/08/2006 |
52000000.0 |
Wazirpur Industrial Area, Delhi-110052, India |
FIXED ASSETS:
Tangible Assets
- Land
- Building
- Plant and
Machinery
- Furniture and
Fixture
- Computer
- Vehicles
- Office equipment
Intangible assets
- Brands and trade marks
- Licenses and franchise
CMT REPORT (Corruption, Money Laundering & Terrorism]
The Public Notice information has been collected from various sources
including but not limited to: The Courts, India Prisons Service,
Interpol, etc.
1] INFORMATION ON
DESIGNATED PARTY
No exist designating subject or any of its beneficial owners,
controlling shareholders or senior officers as terrorist or terrorist
organization or whom notice had been received that all financial transactions
involving their assets have been blocked or convicted, found guilty or against
whom a judgement or order had been entered in a proceedings for violating
money-laundering, anti-corruption or bribery or international economic or
anti-terrorism sanction laws or whose assets were seized, blocked, frozen or
ordered forfeited for violation of money laundering or international
anti-terrorism laws.
2] Court Declaration :
No records exist to suggest that subject is
or was the subject of any formal or informal allegations, prosecutions or other
official proceeding for making any prohibited payments or other improper
payments to government officials for engaging in prohibited transactions or
with designated parties.
3] Asset Declaration :
No records exist to suggest that the property or assets of the subject
are derived from criminal conduct or a prohibited transaction.
4] Record on Financial
Crime :
Charges or conviction
registered against subject: None
5] Records on Violation of
Anti-Corruption Laws :
Charges or
investigation registered against subject: None
6] Records on Int’l
Anti-Money Laundering Laws/Standards :
Charges or
investigation registered against subject: None
7] Criminal Records
No
available information exist that suggest that subject or any of its principals
have been formally charged or convicted by a competent governmental authority
for any financial crime or under any formal investigation by a competent
government authority for any violation of anti-corruption laws or international
anti-money laundering laws or standard.
8] Affiliation with
Government :
No record
exists to suggest that any director or indirect owners, controlling
shareholders, director, officer or employee of the company is a government
official or a family member or close business associate of a Government
official.
9] Compensation Package :
Our market
survey revealed that the amount of compensation sought by the subject is fair
and reasonable and comparable to compensation paid to others for similar
services.
10] Press Report :
No press reports / filings exists on
the subject.
CORPORATE GOVERNANCE
MIRA INFORM as part of its Due Diligence do provide comments on
Corporate Governance to identify management and governance. These factors often
have been predictive and in some cases have created vulnerabilities to credit
deterioration.
Our Governance Assessment focuses principally on the interactions
between a company’s management, its Board of Directors, Shareholders and other
financial stakeholders.
CONTRAVENTION
Subject is not known to have contravened any existing local laws,
regulations or policies that prohibit, restrict or otherwise affect the terms
and conditions that could be included in the agreement with the subject.
FOREIGN EXCHANGE RATES
|
Currency |
Unit
|
INR |
|
US Dollar |
1 |
INR 64.85 |
|
UK Pound |
1 |
INR 90.58 |
|
Euro |
1 |
INR 79.97 |
INFORMATION DETAILS
|
Information
Gathered by : |
PON |
|
|
|
|
Analysis Done by
: |
VAR |
|
|
|
|
Report Prepared
by : |
BHG |
SCORE FACTORS
|
DEMERIT POINTS |
|
|
|
--BANK CHARGES |
YES/NO |
YES |
|
--LITIGATION |
YES/NO |
NO |
|
--OTHER ADVERSE INFORMATION |
YES/NO |
NO |
|
MERIT POINTS |
|
|
|
--SOLE DISTRIBUTORSHIP |
YES/NO |
NO |
|
--EXPORT ACTIVITIES |
YES/NO |
NO |
|
--AFFILIATION |
YES/NO |
YES |
|
--LISTED |
YES/NO |
NO |
|
--OTHER MERIT FACTORS |
YES/NO |
YES |
RATING EXPLANATIONS
|
Credit Rating |
Explanation |
Rating Comments |
|
A++ |
Minimum Risk |
Business dealings permissible with minimum
risk of default |
|
A+ |
Low Risk |
Business dealings permissible with low
risk of default |
|
A |
Acceptable Risk |
Business dealings permissible with
moderate risk of default |
|
B |
Medium Risk |
Business dealings permissible on a regular
monitoring basis |
|
C |
Medium High Risk |
Business dealings permissible preferably
on secured basis |
|
D |
High Risk |
Business dealing not recommended or on
secured terms only |
|
NB |
New Business |
No recommendation can be done due to
business in infancy stage |
|
NT |
No Trace |
No recommendation can be done as the
business is not traceable |
NB is stated where there is insufficient information to facilitate rating. However, it is not to be considered as unfavourable.
This score serves as a reference to assess
SC’s credit risk and to set the amount of credit to be extended. It is
calculated from a composite of weighted scores obtained from each of the major
sections of this report. The assessed factors are as follows:
·
Financial
condition covering various ratios
·
Company
background and operations size
·
Promoters
/ Management background
·
Payment
record
·
Litigation
against the subject
·
Industry
scenario / competitor analysis
·
Supplier
/ Customer / Banker review (wherever available)
This report is issued at
your request without any risk and responsibility on the part of MIRA INFORM
PRIVATE LIMITED (MIPL) or its officials.

