|
|
|
MIRA INFORM REPORT
|
Report No. : |
485179 |
|
Report Date : |
11.01.2018 |
IDENTIFICATION DETAILS
|
Name : |
CLARIS LIFESCIENCES LIMITED (w.e.f. 2010) |
|
|
|
|
Formerly Known
As : |
CORE LABORATORIES
LIMITED (w.e.f. 28.07.1994) ORACLE
LABORATORIES LIMITED |
|
|
|
|
Registered
Office : |
Claris Corporate
Headquarters, Near Parimal Crossing, Ellisbridge, Ahmedabad – 380006, |
|
Tel. No.: |
91-79-26563331/ 66309330 |
|
|
|
|
Country : |
India |
|
|
|
|
Financials (as
on) : |
31.03.2017 |
|
|
|
|
Date of
Incorporation : |
19.07.1994 |
|
|
|
|
Com. Reg. No.: |
04-022543 |
|
|
|
|
Capital
Investment / Paid-up Capital : |
INR 545.678 Million |
|
|
|
|
CIN No.: [Company Identification
No.] |
L85110GJ1994PLC022543 |
|
|
|
|
IEC No.: |
Not Divulged |
|
|
|
|
GSTIN : |
Not Divulged |
|
|
|
|
TAN No.: [Tax Deduction &
Collection Account No.] |
Not Available |
|
|
|
|
PAN No.: [Permanent Account No.] |
Not Available |
|
|
|
|
Legal Form : |
A Public Limited Liability Company. The Company’s Shares are Listed on
the Stock Exchanges. |
|
|
|
|
Line of Business
: |
Subject is engaged into Manufacturer and Wholesaler of Drugs
and Pharmaceuticals. [Registered Activity] |
|
|
|
|
No. of Employees
: |
143 (Approximately) Total manpower of the Group is 2662 nos comprising of 1640 employee
and 1022 contact workers. |
RATING & COMMENTS
(Mira Inform has adopted New Rating mechanism w.e.f. 23rd January
2017)
|
MIRA’s Rating : |
A |
|
Credit Rating |
Explanation |
Rating Comments |
|
A |
Acceptable Risk |
Business dealings permissible with
moderate risk of default |
|
Maximum Credit Limit : |
USD 21150000 |
|
|
|
|
Status : |
Good |
|
|
|
|
Payment Behaviour : |
Regular |
|
|
|
|
Litigation : |
Clear |
|
|
|
|
Comments : |
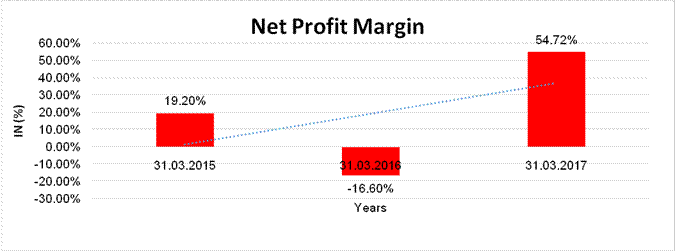
Subject was incorporated in the year 1994 and it is engaged in wholesale of pharmaceutical and medical products. ‘Athanas Enterprise Private Limited’ holds the maximum stake of 50.13% in the company. In February 2017, the company has transferred the ownership of its subsidiary company i.e. Claris Injectables Limited to Baxter International INC (Baxter Group) based in USA. As per financials of March 2017, the company has registered sharp decline in its revenue as compared to its previous year revenue and has reported a good profit margin of 54.72% under review. Rating takes into consideration the company’s established track record of business operations marked by sound net worth along with low solvency indicators and good liquidity position. Share are quoted high on Stock Exchanges. (Share are traded at a price of INR 356.45 against its face value of INR 10 on BSE.) However, rating strength is partially offset by product concentration risk as its branded formulations basket has only three large segment anaesthesia, anti-infective and renal therapy along with its dependence on selective brands, exposing it to brand concentration risk and limited diversification of product portfolio. As per quarterly financials of September 2017, the company has achieved a revenue of INR 27.3 million and reported decent profit margin of 6.21%. Payments are reported to be regular. In view of aforesaid, the company can be considered for business dealings at usual trade terms and conditions. |
NOTES : Any query related to this
report can be made on e-mail : infodept@mirainform.com
while quoting report number, name and date.
ECGC Country Risk Classification List
|
Country Name |
Previous Rating (30.06.2017) |
Current Rating (30.09.2017) |
|
India |
A1 |
A1 |
|
Risk Category |
ECGC
Classification |
|
Insignificant |
A1 |
|
Low Risk |
A2 |
|
Moderately Low Risk |
B1 |
|
Moderate Risk |
B2 |
|
Moderately High Risk |
C1 |
|
High Risk |
C2 |
|
Very High Risk |
D |
RBI DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available RBI Defaulters’ list.
EPF (Employee Provident Fund) DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available EPF (Employee Provident Fund) Defaulters’ list as of
31-03-2016.
BIFR (Board for Industrial & Financial Reconstruction) LISTING
STATUS
Subject’s name is not listed as a Sick Unit in
the publicly available BIFR (Board for Industrial & Financial
Reconstruction) list as of 11.01.2018
IBBI (Insolvency and Bankruptcy Board of India) LISTING STATUS
Subject’s name is not listed in the publicly
available IBBI (Insolvency and Bankruptcy Board of India) list as of report
date.
LOCATIONS
|
Registered/ Corporate Office : |
Claris Corporate
Headquarters, Near Parimal Crossing, Ellisbridge, Ahmedabad – 380006,
Gujarat, India |
|
Tel. No.: |
91-79-26563331/
66309330/ 66309339 |
|
Mobile No.: |
Not Available |
|
Fax No.: |
91-79-26408053 |
|
E-Mail : |
|
|
Website : |
|
|
|
|
|
Factory : |
Sarkhej-Bavla Road, Chacharvadi-Vasna, Ahmedabad - 382213, Gujarat, India |
|
Tel. No.: |
91-2717-661999 |
DIRECTORS
As on 31.03.2017
|
Name : |
Mr. Arjun Sushilkumar Handa |
|
Designation : |
Chairman and Managing Director |
|
Address : |
Sharanya, Judges Bunglows Road, Vastrapur, Bodakdev, Ahmedabad – 380054, Gujarat, India |
|
Date of Appointment : |
26.09.2011 |
|
DIN No.: |
00159413 |
|
|
|
|
Name : |
Mr. Chandrasingh Shivrambhai Purohit |
|
Designation : |
Whole time Director |
|
Address : |
17, Rajvi Emeralds, Near Gala Gymkhana, Bopal, Ahmedabad – 380054, Gujarat, India |
|
Date of Appointment : |
03.07.2009 |
|
DIN No.: |
00199651 |
|
|
|
|
Name : |
Mr. Shyamsunder Sharma |
|
Designation : |
Whole Time Director (Additional Director) |
|
Address : |
A-302, Sukirti Park, Near Kanchanbhumi Apartment, 100 Ft. Ring Road, Satelite Ahmedabad – 380015, Gujarat, India |
|
Date of Appointment : |
20.05.2017 |
|
DIN No : |
07563680 |
|
|
|
|
Name : |
Mr. Surrinder Lal Kapur |
|
Designation : |
Chairman and Non Executive Director |
|
Address : |
161 A/1 Western Avenue, Sainik Farms, New Delhi – 110062, India |
|
Date of Appointment : |
26.09.2008 |
|
|
|
|
Name : |
Mr. Tiruponitura Venkatraman Ananthanarayanan |
|
Designation : |
Non-Executive and Director |
|
Address : |
41/135 (Old), New 19/165 Thangamalai Road, Kotagiri – 643217, Tamilnadu, India |
|
Date of Appointment : |
28.01.2008 |
|
DIN No.: |
00109197 |
|
|
|
|
Name : |
Mr. Anup Pravin Shah |
|
Designation : |
Director |
|
Address : |
Suite 8, Jal Kiran Cuffe Parade, Mumbai - 400005, Maharashtra, India |
|
Date of Appointment : |
23.02.2013 |
|
DIN No.: |
00293207 |
|
|
|
|
Name : |
Mr. Aditya Sushilkumar Handa |
|
Designation : |
Director |
|
Address : |
Sharanya, Judges Bunglows Road Opposite Ruchir Bunglows, Bodakdev
Ahmedabad - 380054, Gujarat, India |
|
Date of Appointment : |
13.06.2006 |
|
DIN No.: |
00308513 |
|
|
|
|
Name : |
Ms. Milina Purnendu Bose |
|
Designation : |
Director |
|
Address : |
E 504, Sarjan Towers, Off. Gurukul Road, Memnagar, Ahmedabad – 380052,
Gujarat, India |
|
Date of Appointment : |
11.08.2014 |
|
DIN No.: |
02204540 |
|
|
|
|
Name : |
Mr. Amish Vyas |
|
Designation : |
Non-Executive and Non-Independent Director |
KEY EXECUTIVES
|
Name : |
Mr. Chandrasingh Shivrambhai Purohit |
|
Designation : |
Chief Finance Officer |
|
Address : |
17, Rajvi Emeralds, Near Gala Gymkhana, Bopal, Ahmedabad – 380054, Gujarat, India |
|
Date of Appointment : |
09.05.2014 |
|
PAN No.: |
AFKPP3128B |
|
|
|
|
Name : |
Mr. Kiritkumar Himatlal Kanjaria |
|
Designation : |
Company Secretary and Compliance Officer |
|
Address : |
Ashirwad-III, Bunglow No.4 Opposite Auda Garden, Prahladnagar, Ahmedabad -380051, Gujarat, India |
|
Date of Appointment : |
13.04.2013 |
|
PAN No.: |
ADPPK8970R |
|
|
|
|
Audit Committee : |
|
|
|
|
|
Stakeholders
Relationship Committee : |
|
|
|
|
|
Nomination and
Remuneration Committee : |
|
|
|
|
|
Corporate Social
Responsibility Committee : |
|
|
|
|
|
Committee of
Directors : |
|
MAJOR SHAREHOLDERS
As on 30.09.2017
|
Category of shareholder |
Total nos. shares held |
Shareholding as a % of total no. of shares (calculated as per SCRR,
1957)As a % of |
|
|
(A) Promoter &
Promoter Group |
27355964 |
50.13 |
|
|
(B) Public |
27211801 |
49.87 |
|
|
Grand Total |
54567765 |
100.00 |
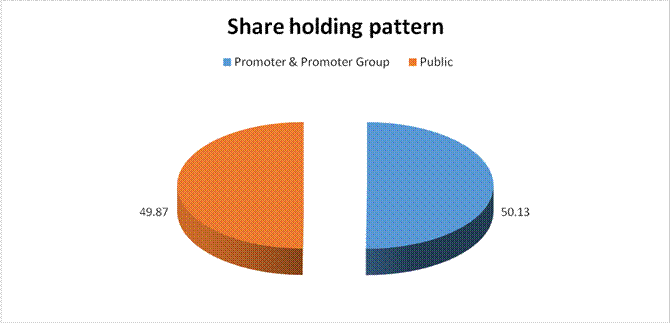
"Statement
showing shareholding pattern of the Promoter and Promoter Group"
|
Category of
shareholder |
Total nos. shares
held |
Shareholding as a %
of total no. of shares (calculated as per SCRR, 1957)As a % of |
|
|
A1) Indian |
0.00 |
||
|
Any Other (specify) |
27355964 |
50.13 |
|
|
Athanas Enterprise Private Limited |
27353580 |
50.13 |
|
|
Abellon Energy Limited |
2384 |
0.00 |
|
|
Sub Total A1 |
27355964 |
50.13 |
|
|
A2) Foreign |
0.00 |
||
|
A=A1+A2 |
27355964 |
50.13 |
Statement showing shareholding pattern of the Public shareholder
|
Category & Name
of the Shareholders |
Total no. shares
held |
Shareholding % calculated
as per SCRR, 1957 As a % of |
|
|
B1) Institutions |
0.00 |
||
|
Mutual Funds/ |
1054255 |
1.93 |
|
|
Reliance Capital Trustee Co. Ltd-A/C Reliancesmall Cap Fund |
1054255 |
1.93 |
|
|
Foreign Portfolio Investors |
13982586 |
25.62 |
|
|
East Bridge Capital Master Fund Limited |
2422780 |
4.44 |
|
|
Government Pension Fund Global |
1623970 |
2.98 |
|
|
Cobra India (Mauritius) Limited |
1495411 |
2.74 |
|
|
Orbimed Partners Master Mauritius Limited |
1345345 |
2.47 |
|
|
Signet Healthcare Partners QP Partnership III LP |
1216982 |
2.23 |
|
|
Ashoka Pte Ltd |
1178962 |
2.16 |
|
|
Morgan Stanley Asia (Singapore) Pte. |
1119792 |
2.05 |
|
|
HBM Healthcare Investments (Cayman) Ltd |
1092358 |
2.00 |
|
|
Orbimed Partners II Mauritius Limited |
1039551 |
1.91 |
|
|
Sub Total B1 |
15036841 |
27.56 |
|
|
B2) Central Government/ State Government(s)/ President of India |
0.00 |
||
|
B3) Non-Institutions |
0.00 |
||
|
Individual share capital upto Rs. 2 Lacs |
4781460 |
8.76 |
|
|
Individual share capital in excess of Rs. 2 Lacs |
2259273 |
4.14 |
|
|
Any Other (specify) |
5134227 |
9.41 |
|
|
HUF |
442305 |
0.81 |
|
|
NRI – Non- Repat |
325921 |
0.60 |
|
|
NRI – Repat |
164913 |
0.30 |
|
|
Clearing Members |
494035 |
0.91 |
|
|
Bodies Corporate |
3707053 |
6.79 |
|
|
Sub Total B3 |
12174960 |
22.31 |
|
|
B=B1+B2+B3 |
27211801 |
49.87 |
BUSINESS DETAILS
|
Line of Business : |
Subject is engaged into Manufacturer and Wholesaler of Drugs
and Pharmaceuticals. [Registered Activity] |
||||
|
|
|
||||
|
Products / Services
: |
|
||||
|
|
|
||||
|
Brand Names : |
Not Available |
||||
|
|
|
||||
|
Agencies Held : |
Not Available |
||||
|
|
|
||||
|
Exports : |
Not Divulged |
||||
|
|
|
||||
|
Imports : |
Not Divulged |
||||
|
|
|
||||
|
Terms : |
Not Divulged |
PRODUCTION STATUS – (NOT AVAILABLE)
GENERAL INFORMATION
|
Suppliers : |
|
||||||||||||||||||||||
|
|
|
||||||||||||||||||||||
|
Customers : |
|
||||||||||||||||||||||
|
|
|
||||||||||||||||||||||
|
No. of Employees : |
143 (Approximately) |
||||||||||||||||||||||
|
|
|
||||||||||||||||||||||
|
Bankers : |
|
||||||||||||||||||||||
|
|
|
||||||||||||||||||||||
|
Facilities : |
Notes:
b. The term loan as at March 31, 2016 is secured by first and exclusive charge over the immovable and movable assets of Solar Plant located at Modasa. c. The term loan as at March 31, 2017 is secured by equitable mortgage on the one of the Company's immovable property. |
|
Financial Institution: |
Tata Capital Financial Services Limited |
|
Auditors : |
|
|
Name : |
Shah and Shah Associates Chartered Accountants |
|
Address : |
Ahmedabad, Gujarat, India |
|
|
|
|
Internal Auditors |
|
|
Name : |
KPMG Chartered Accountants |
|
Address : |
Ahmedabad, Gujarat, India |
|
|
|
|
Memberships : |
Not Available |
|
|
|
|
Collaborators : |
Not Available |
|
|
|
|
Holding Company: |
Athanas Enterprise Private Limited [U51909GJ2013PTC075961] |
|
|
|
|
Subsidiary Companies
(including step-down subsidiaries) : |
|
|
|
|
|
Associate Company : |
(Formerly known as Claris Otsuka
Private Limited) |
|
|
|
|
Companies over
which Key Management Personnel and their relatives are able to exercise
significant influence : |
|
CAPITAL STRUCTURE
As on 31.03.2017
Authorised Capital:
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
120510000 |
Equity Shares |
INR 10/- each |
INR 1205.100 Million |
|
|
|
|
|
Issued, Subscribed & Paid-up Capital:
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
54567765 |
Equity Shares |
INR 10/- each |
INR 545.678
Million |
|
|
|
|
|
a) Reconciliation of the Shares Outstanding at
the beginning and at the end of the reporting period
|
PARTICULARS |
Year ended 31.03.2017 |
|
Equity Share |
Number of shares |
Value (in Million) |
|
At the beginning of
the period |
54567765 |
INR 545.678
Million |
|
Issued during the period - Bonus issue |
-- |
-- |
|
Bought back during the year |
-- |
-- |
|
Outstanding at the
end of the period |
54567765 |
INR 545.678 Million |
b) Rights, Preferences and Restrictions
attached to equity shares
The Company has only one class of equity shares having a face value of Rs.10 per share. Each shareholder is eligible for one vote per equity share held. In the event of liquidation, the equity shareholders are eligible to receive the remaining assets of the Company after distribution of all the preferential amounts, in the proportion of their shareholding.
|
Name of the
shareholders |
As at March
31, 2017 |
|
|
|
|
|
|
Equity Shares held by
Holding Company |
No. of
Shares |
% of Shareholding |
|
Athanas Enterprise Private Limited |
27353580 |
50.13% |
|
Shareholders
holding more than 5% of total equity shares |
|
|
|
Athanas Enterprise Private Limited |
27353580 |
50.13% |
|
First Carlyle Ventures III |
- |
- |
|
Abellon Energy Limited |
- |
- |
c) During the period ended on March 31, 2015, the company had bought back 92,50,000 equity shares of the face value of Rs. 10 each (representing 14.49 % of the total equity share capital of the Company) at the price of Rs.250 per equity share aggregating to Rs.23,125 Lacs which is less than 25% of the aggregate of equity share capital and free reserves of the Company as per audited financial statements of the Company for the financial year ended December 31, 2012 through “Tender Offer” route as prescribed under the Securities and Exchange Board of India (Buy-Back of Securities) Regulations, 1998.
FINANCIAL DATA
[all figures are INR
Million]
ABRIDGED
BALANCE SHEET (STANDALONE)
|
SOURCES
OF FUNDS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
|
|
|
|
|
I.
EQUITY AND LIABILITIES |
|
|
|
|
(1)Shareholders' Funds |
|
|
|
|
(a) Share Capital |
545.678 |
545.678 |
545.678 |
|
(b) Reserves & Surplus |
6857.099 |
6876.887 |
8030.036 |
|
(c) Money received against
share warrants |
0.000 |
0.000 |
0.000 |
|
|
|
|
|
|
(2) Share Application money
pending allotment |
0.000 |
0.000 |
0.000 |
|
Total
Shareholders’ Funds |
7402.777 |
7422.565 |
8575.714 |
|
|
|
|
|
|
(3) Non-Current Liabilities |
|
|
|
|
(a) long-term borrowings |
165.835 |
116.737 |
143.168 |
|
(b) Deferred tax liabilities
(Net) |
0.000 |
0.000 |
27.257 |
|
(c) Other long term
liabilities |
0.000 |
0.000 |
0.000 |
|
(d) long-term provisions |
50.877 |
37.716 |
25.061 |
|
Total
Non-current Liabilities |
216.712 |
154.453 |
195.486 |
|
|
|
|
|
|
(4) Current Liabilities |
|
|
|
|
(a) Short term borrowings |
0.000 |
0.000 |
226.576 |
|
(b) Trade payables |
607.675 |
1202.478 |
956.433 |
|
(c) Other current liabilities |
1149.785 |
1238.475 |
2687.654 |
|
(d) Short-term provisions |
3.084 |
2.457 |
46.084 |
|
Total
Current Liabilities |
1760.544 |
2443.410 |
3916.747 |
|
|
|
|
|
|
TOTAL |
9380.033 |
10020.428 |
12687.947 |
|
|
|
|
|
|
II.
ASSETS |
|
|
|
|
(1) Non-current assets |
|
|
|
|
(a) Fixed Assets |
|
|
|
|
(i) Tangible assets |
493.456 |
324.285 |
368.636 |
|
(ii) Intangible Assets |
6.670 |
10.668 |
14.666 |
|
(iii) Capital work-in-progress |
24.019 |
60.622 |
17.723 |
|
(iv) Intangible assets under
development |
0.000 |
0.000 |
0.000 |
|
(b) Non-current Investments |
17.670 |
319.321 |
192.696 |
|
(c) Deferred tax assets (net) |
235.571 |
189.207 |
0.000 |
|
(d) Long-term Loan and Advances |
430.717 |
0.000 |
88.773 |
|
(e) Other Non-current assets |
177.734 |
172.566 |
0.000 |
|
Total
Non-Current Assets |
1385.837 |
1076.669 |
682.494 |
|
|
|
|
|
|
(2) Current assets |
|
|
|
|
(a) Current investments |
1872.289 |
1910.035 |
3316.125 |
|
(b) Inventories |
0.000 |
0.000 |
0.000 |
|
(c) Trade receivables |
229.618 |
782.310 |
1518.498 |
|
(d) Cash and cash equivalents |
222.262 |
452.611 |
442.412 |
|
(e) Short-term loans and
advances |
4957.220 |
5049.385 |
1107.017 |
|
(f) Other current assets |
712.307 |
749.418 |
5621.401 |
|
Total
Current Assets |
7993.696 |
8943.759 |
12005.453 |
|
|
|
|
|
|
Assets
classified as held for sale |
0.500 |
0.000 |
0.000 |
|
|
|
|
|
|
TOTAL |
9380.033 |
10020.428 |
12687.947 |
PROFIT
& LOSS ACCOUNT (STANDALONE)
|
|
PARTICULARS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
|
SALES |
|
|
|
|
|
Income |
211.162 |
1220.538 |
7222.841 |
|
|
Other Income |
480.258 |
425.184 |
762.552 |
|
|
TOTAL
|
691.420 |
1645.722 |
7985.393 |
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
Cost of Materials Consumed |
0.000 |
0.000 |
761.628 |
|
|
Purchases of Stock-in-Trade |
82.160 |
968.377 |
3726.794 |
|
|
Changes in inventories of
finished goods, work-in-progress and Stock-in-Trade |
0.000 |
0.000 |
(42.769) |
|
|
Employees benefits expense |
277.396 |
184.517 |
666.150 |
|
|
Other expenses |
441.969 |
851.397 |
1247.507 |
|
|
Exceptional Items |
0.000 |
0.000 |
(52.303) |
|
|
TOTAL |
801.525 |
2004.291 |
6307.007 |
|
|
|
|
|
|
|
|
PROFIT/
(LOSS) BEFORE INTEREST, TAX,
DEPRECIATION AND AMORTISATION |
(110.105) |
(358.569) |
1678.386 |
|
|
|
|
|
|
|
Less |
FINANCIAL
EXPENSES |
37.318 |
39.104 |
310.440 |
|
|
|
|
|
|
|
|
PROFIT
/ (LOSS) BEFORE TAX, DEPRECIATION AND AMORTISATION |
(147.423) |
(397.673) |
1367.946 |
|
|
|
|
|
|
|
Less |
DEPRECIATION/
AMORTISATION |
33.241 |
35.595 |
295.756 |
|
|
|
|
|
|
|
|
PROFIT/
(LOSS) BEFORE TAX |
(180.664) |
(433.268) |
1072.190 |
|
|
|
|
|
|
|
Less |
TAX |
(296.220) |
(230.643) |
(314.706) |
|
|
|
|
|
|
|
|
PROFIT/
(LOSS) AFTER TAX |
115.556 |
(202.625) |
1386.896 |
|
|
|
|
|
|
|
|
EARNINGS
IN FOREIGN CURRENCY |
|
|
|
|
|
F.O.B. Value of Exports |
0.000 |
0.000 |
6045.380 |
|
|
Freight |
0.000 |
0.000 |
115.768 |
|
|
TOTAL
EARNINGS |
0.000 |
0.000 |
6161.148 |
|
|
|
|
|
|
|
|
IMPORTS |
|
|
|
|
|
Raw Materials |
0.000 |
0.000 |
66.287 |
|
|
Components and Stores parts |
0.000 |
0.000 |
314.042 |
|
|
Capital Goods |
0.000 |
0.000 |
261.264 |
|
|
Plant & Machinery |
0.000 |
0.000 |
651.158 |
|
|
Stores and Spares |
0.000 |
0.000 |
7.772 |
|
|
TOTAL
IMPORTS |
0.000 |
0.000 |
641.593 |
|
|
|
|
|
|
|
|
Earnings
/ (Loss) Per Share (INR) |
2.12 |
(3.71) |
7.20 |
CURRENT MATURITIES
OF LONG TERM DEBT DETAILS
|
Particulars |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Current Maturities of Long term debt |
|
|
|
|
Term loans from Bank |
37.800 |
13.591 |
- |
|
Vehicle loans |
11.936 |
7.380 |
- |
|
Total |
49.736 |
20.971 |
19.014 |
|
Cash generated from operations |
2596.690 |
536.927 |
911.997 |
|
Net cash flows from (used in) operating activity |
1793.547 |
204.588 |
485.750 |
QUARTERLY
RESULTS
|
Particulars |
|
30.06.2017 Unaudited |
30.09.2017 Unaudited |
|
|
|
|
|
|
Net Sales |
|
15.230 |
27.270 |
|
Total Expenditure |
|
153.930 |
128.430 |
|
PBIDT (Excl OI) |
|
(138.700) |
(101.160) |
|
Other Income |
|
101.220 |
438.340 |
|
Operating Profit |
|
(37.480) |
337.180 |
|
Interest |
|
8.920 |
7.550 |
|
Exceptional Items |
|
NA |
NA |
|
PBDT |
|
(46.400) |
329.630 |
|
Depreciation |
|
10.420 |
10.920 |
|
Profit Before Tax |
|
(56.820) |
318.710 |
|
Tax |
|
(22.420) |
115.580 |
|
Provisions and
contingencies |
|
NA |
NA |
|
Profit After Tax |
|
(34.400) |
203.130 |
|
Extraordinary Items |
|
NA |
NA |
|
Prior Period Expenses |
|
NA |
NA |
|
Other Adjustments |
|
NA |
NA |
|
Net Profit |
|
0.400 |
16936.730 |
KEY
RATIOS
EFFICIENCY RATIOS
|
PARTICULARS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Average Collection Days (Sundry Debtors / Income * 365 Days) |
396.90 |
233.95 |
76.74 |
|
|
|
|
|
|
Account Receivables Turnover (Income / Sundry
Debtors) |
0.92 |
1.56 |
4.76 |
|
|
|
|
|
|
Average Payment Days (Sundry Creditors
/ Purchases * 365 Days) |
2699.63 |
453.24 |
77.78 |
|
|
|
|
|
|
Inventory Turnover (Operating Income
/ Inventories) |
0.00 |
0.00 |
0.00 |
|
|
|
|
|
|
Asset Turnover (Operating Income
/ Net Fixed Assets) |
(0.21) |
(0.91) |
4.19 |
LEVERAGE RATIOS
|
PARTICULARS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Debt Ratio ((Borrowing + Current Liabilities) / Total
Assets) |
0.21 |
0.26 |
0.32 |
|
|
|
|
|
|
Debt Equity Ratio (Total Liability
/ Networth) |
0.03 |
0.02 |
0.05 |
|
|
|
|
|
|
Current Liabilities to Networth (Current Liabilities
/ Net Worth) |
0.24 |
0.33 |
0.46 |
|
|
|
|
|
|
Fixed Assets to Networth (Net Fixed Assets
/ Networth) |
0.07 |
0.05 |
0.05 |
|
|
|
|
|
|
Interest Coverage Ratio (PBIT / Financial
Charges) |
-2.95 |
-9.17 |
5.41 |
PROFITABILITY RATIOS
|
PARTICULARS |
|
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Net Profit Margin ((PAT / Sales) *
100) |
% |
54.72 |
(16.60) |
19.20 |
|
|
|
|
|
|
|
Return on Total Assets ((PAT / Total
Assets) * 100) |
% |
1.23 |
(2.02) |
10.93 |
|
|
|
|
|
|
|
Return on Investment (ROI) ((PAT / Networth)
* 100) |
% |
1.56 |
(2.73) |
16.17 |
SOLVENCY RATIOS
|
PARTICULARS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Current Ratio (Current Assets / Current Liabilities) |
4.54 |
3.66 |
3.07 |
|
|
|
|
|
|
Quick Ratio ((Current Assets
– Inventories) / Current Liabilities) |
4.54 |
3.66 |
3.07 |
|
|
|
|
|
|
G-Score Ratio Financial (Networth / Total
Assets) |
0.79 |
0.74 |
0.68 |
|
|
|
|
|
|
G-Score Ratio Debt (Debts / Equity
Capital) |
0.40 |
0.25 |
0.71 |
|
|
|
|
|
|
G-Score Ratio Liquidity (Total Current
Assets / Total Current Liabilities) |
4.54 |
3.66 |
3.07 |
Total Liability = Short-term Debt + Long-term
Debt + Current Maturities of Long-term debts
STOCK
PRICES
|
Face Value |
INR 10.00/- |
|
|
|
|
Market Value |
INR 356.45/- [BSE] |
FINANCIAL ANALYSIS
[all figures are
in INR Million]
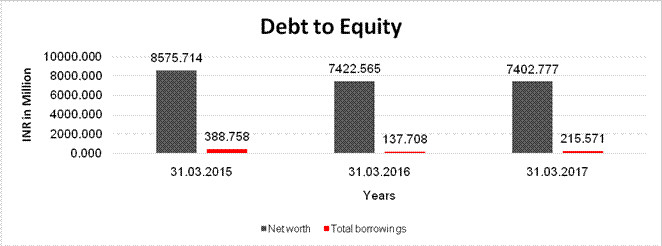
DEBT EQUITY RATIO
|
Particular |
31.03.2015 |
31.03.2016 |
31.03.2017 |
|
|
INR
In Million |
INR
In Million |
INR
In Million |
|
Share Capital |
545.678 |
545.678 |
545.678 |
|
Reserves & Surplus |
8030.036 |
6876.887 |
6857.099 |
|
Money received against share
warrants |
0.000 |
0.000 |
0.000 |
|
Share Application money pending allotment |
0.000 |
0.000 |
0.000 |
|
Net
worth |
8575.714 |
7422.565 |
7402.777 |
|
|
|
|
|
|
long-term borrowings |
143.168 |
116.737 |
165.835 |
|
Short term borrowings |
226.576 |
0.000 |
0.000 |
|
Current maturities of
long-term debts |
19.014 |
20.971 |
49.736 |
|
Total
borrowings |
388.758 |
137.708 |
215.571 |
|
Debt/Equity
ratio |
0.045 |
0.019 |
0.029 |
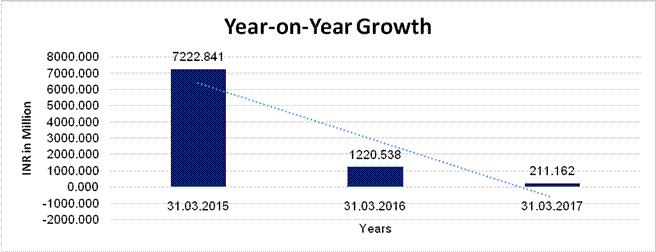
YEAR-ON-YEAR GROWTH
|
Year
on Year Growth |
31.03.2015 |
31.03.2016 |
31.03.2017 |
|
|
INR
In Million |
INR
In Million |
INR
In Million |
|
Sales |
7222.841 |
1220.538 |
211.162 |
|
|
|
(83.102) |
(82.699) |
NET PROFIT MARGIN
|
Net
Profit Margin |
31.03.2015 |
31.03.2016 |
31.03.2017 |
|
|
INR
In Million |
INR
In Million |
INR
In Million |
|
Sales |
7222.841 |
1220.538 |
211.162 |
|
Profit / (Loss) |
1386.896 |
(202.625) |
115.556 |
|
|
19.20% |
(16.60%) |
54.72% |
ABRIDGED
BALANCE SHEET (CONSOLIDATED)
|
SOURCES OF FUNDS |
|
31.03.2017 |
31.03.2016 |
|
I.
EQUITY AND LIABILITIES |
|
|
|
|
(1)Shareholders' Funds |
|
|
|
|
(a) Share Capital |
|
545.678 |
545.678 |
|
(b) Reserves & Surplus |
|
9653.681 |
8649.377 |
|
(c) Money received against share warrants |
|
0.000 |
0.000 |
|
|
|
|
|
|
(2) Share
Application money pending allotment |
|
0.000 |
0.000 |
|
Total Shareholders’
Funds |
|
10199.359 |
9195.055 |
|
|
|
|
|
|
(3)
Non-Current Liabilities |
|
|
|
|
(a) Long-term borrowings |
|
165.835 |
3516.704 |
|
(b) Deferred tax liabilities (Net) |
|
185.257 |
247.190 |
|
(c)
Other long term liabilities |
|
0.000 |
42.150 |
|
(d)
long-term provisions |
|
69.384 |
141.171 |
|
Total
Non-current Liabilities |
|
420.476 |
3947.215 |
|
|
|
|
|
|
(4) Current Liabilities |
|
|
|
|
(a)
Short term borrowings |
|
0.000 |
1709.992 |
|
(b)
Trade payables |
|
467.654 |
1455.611 |
|
(c)
Other current liabilities |
|
237.574 |
1400.011 |
|
(d)
Short-term provisions |
|
7.826 |
12.281 |
|
Total
Current Liabilities |
|
713.054 |
4577.895 |
|
|
|
|
|
|
LIABILITIES
ASSOCIATED WITH ASSETS HELD FOR SALE |
|
6673.976 |
0.000 |
|
|
|
|
|
|
TOTAL |
|
18006.865 |
17720.165 |
|
|
|
|
|
|
II.
ASSETS |
|
|
|
|
(1) Non-current assets |
|
|
|
|
(a)
Fixed Assets |
|
|
|
|
(i)
Tangible assets |
|
557.061 |
8650.676 |
|
(ii)
Intangible Assets |
|
6.990 |
10.988 |
|
(iii)
Capital work-in-progress |
|
24.019 |
104.216 |
|
(iv) Intangible assets under development |
|
0.000 |
0.000 |
|
(b) Non-current
Investments |
|
1035.302 |
1172.806 |
|
(c) Deferred tax assets
(net) |
|
246.610 |
352.890 |
|
(d) Long-term Loan
and Advances |
|
0.000 |
0.000 |
|
(e)
Other Non-current assets |
|
194.484 |
322.394 |
|
|
|
|
|
|
Total
Non-Current Assets |
|
2064.466 |
10613.970 |
|
|
|
|
|
|
(2) Current assets |
|
|
|
|
(a)
Current investments |
|
1877.286 |
1910.038 |
|
(b)
Inventories |
|
0.000 |
946.807 |
|
(c)
Trade receivables |
|
181.312 |
1674.198 |
|
(d)
Cash and cash equivalents |
|
306.103 |
1861.906 |
|
(e)
Short-term loans and advances |
|
95.141 |
172.757 |
|
(f)
Other current assets |
|
500.722 |
540.489 |
|
Total
Current Assets |
|
2960.564 |
7106.195 |
|
|
|
|
|
|
ASSETS CLASSIFICATION AS HELD FOR SALE |
|
12981.835 |
0.000 |
|
|
|
|
|
|
TOTAL |
|
18006.865 |
17720.165 |
PROFIT
& LOSS ACCOUNT (CONSOLIDATED)
|
|
PARTICULARS |
|
31.03.2017 |
31.03.2016 |
|
|
|
SALES |
|
|
|
|
|
|
|
Revenue from Operations |
|
255.287 |
1331.625 |
|
|
|
Other Income |
|
374.588 |
458.475 |
|
|
|
TOTAL |
|
629.875 |
1790.100 |
|
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
|
|
Purchases of Stock-in-Trade |
|
82.160 |
1023.967 |
|
|
|
Employees benefits expense |
|
326.246 |
223.694 |
|
|
|
Other expenses |
|
478.000 |
863.544 |
|
|
|
Share in Profit/ (Loss) of
Associates |
|
260.927 |
921.210 |
|
|
|
TOTAL |
|
1147.333 |
3032.415 |
|
|
|
|
|
|
|
|
|
PROFIT
BEFORE INTEREST, TAX, DEPRECIATION AND AMORTISATION |
|
(517.458) |
(1242.315) |
|
|
|
|
|
|
|
|
|
Less |
FINANCIAL
EXPENSES |
|
39.832 |
54.792 |
|
|
|
|
|
|
|
|
|
|
PROFIT
BEFORE TAX, DEPRECIATION AND AMORTISATION |
|
(557.290) |
(1297.107) |
|
|
|
|
|
|
|
|
|
Less |
DEPRECIATION/
AMORTISATION |
|
39.684 |
38.573 |
|
|
|
|
|
|
|
|
|
|
PROFIT BEFORE
TAX |
|
(596.974) |
(1335.680) |
|
|
|
|
|
|
|
|
|
Less |
TAX |
|
(1696.224) |
(941.330) |
|
|
|
|
|
|
|
|
|
|
PROFIT AFTER TAX
|
|
1099.250 |
(394.350) |
|
LOCAL AGENCY FURTHER INFORMATION
|
Sr. No. |
Check List by Info Agents |
Available in
Report (Yes / No) |
|
1] |
Year of establishment |
Yes |
|
2] |
Constitution of the entity Incorporation
details |
Yes |
|
3] |
Locality of the entity |
Yes |
|
4] |
Premises details |
Yes |
|
5] |
Buyer visit details |
-- |
|
6] |
Contact numbers |
No |
|
7] |
Name of the person contacted |
No |
|
8] |
Designation of contact person |
No |
|
9] |
Promoter’s background |
Yes |
|
10] |
Date of Birth of Proprietor / Partners / Directors |
Yes |
|
11] |
Pan Card No. of Proprietor / Partners |
No |
|
12] |
Voter Id Card No. of Proprietor / Partners |
No |
|
13] |
Type of business |
Yes |
|
14] |
Line of Business |
Yes |
|
15] |
Export/import details (if applicable) |
No |
|
16] |
No. of employees |
Yes |
|
17] |
Details of sister concerns |
Yes |
|
18] |
Major suppliers |
No |
|
19] |
Major customers |
No |
|
20] |
Banking Details |
Yes |
|
21] |
Banking facility details |
No |
|
22] |
Conduct of the banking account |
-- |
|
23] |
Financials, if provided |
Yes |
|
24] |
Capital in the business |
Yes |
|
25] |
Last accounts filed at ROC, if applicable |
Yes |
|
26] |
Turnover of firm for last three years |
Yes |
|
27] |
Reasons for variation <> 20% |
-- |
|
28] |
Estimation for coming financial year |
No |
|
29] |
Profitability for last three years |
Yes |
|
30] |
Major shareholders, if available |
Yes |
|
31] |
Litigations that the firm/promoter
involved in |
-- |
|
32] |
Market information |
-- |
|
33] |
Payments terms |
No |
|
34] |
Negative Reporting by Auditors in the
Annual Report |
No |
CORPORATE INFORMATION
The Company is a public company domiciled in India and is incorporated under the provisions of the Companies Act applicable in India. The Company's shares are listed on BSE, a recognised stock exchange, in India.
The registered office of the company is located at Claris
Corporate Headquarters, Near Parimal Crossing, Ellisbridge, Ahmedabad - 380
006, India. The Company is engaged in the business of selling injectable
products and providing management consulting towards various operational and
strategic activities and certain other shared services to some of its
subsidiaries. A significant majority of injectables products are generic drugs,
capable of being directly injected into the human body, predominantly used in
the treatment of critical illnesses. The standalone financial statements were
authorised for issue in accordance with a resolution of the directors on May
20, 2017.
MANAGEMENT DISCUSSION
& ANALYSIS
Global Injectables
Outlook
The global injectable drug delivery market, in terms of value, is projected to reach USD 624.50 Billion by 2021 from USD 362.38 Billion in 2016, at a CAGR of 11.5% during the forecast period. US Generic Injectable market expected to grow at 10% CAGR 2015-2020.
The injectable drug delivery market is segmented based on type, formulation packaging, therapeutic application, usage pattern, site of administration, distribution channel, facility of use, and regions.
As per ICRA research, during the 2015-19 period Injectables drugs worth ~US$16 billion are expected to go off-patent in the US alone. While significant price erosion is normal once the drugs go off-patent, the injectable segment still holds a sizeable opportunity to gain meaningful revenue growth for Indian generic companies because the upcoming patent expiries. In addition, increase in manufacturing costs, required as part of complying with USFDA cGMP norms and drug shortages, has led to price increases in the past and the trend is expected to continue forward - contributing to the overall market growth. Supported by the patent expiries and favourable pricing environment, the US generic injectable market is expected to grow at a CAGR of 10% over the next five years. Stringent USFDA cGMP regulations have led to supply issues for various injectable players and focus on maintaining quality remains critical.
Drug shortages are led by supply disruptions because of USFDA cGMP violations as well as consolidation of in-house manufacturing facilities. Several warning letters had been issued by USFDA in the past to generic injectable players, though sorted out over period.
USA remains a challenging market due to high entry barriers and established incumbents. The generic injectable market is characterised by high capital investments and operational costs coupled with relatively higher compliance requirement owing to the sterile nature of the products. These factors have resulted in high barriers to entry and, thus, a limited number of competitors relative to other segments. These have further reduced due to mergers and acquisition (“M&A”) activities within the sector over the last few years. For the US Generic Injectable market, 70% of the market (by value) has three or less than three companies compared to five or more players for the oral solids generics corroborating high level of entry barriers owing to high upfront capital investments, compliance issues and economies of scale requirements. In 2015, the top five generic companies controlled ~52% of the market share by value and ~73% of the market by volume (source: Industry). Any new entrant is required to have a relatively large basket of products and cost advantage to gain traction with customers.
Over the past few years, manufacturing units of several large players operating in the US generic injectable market have faced regulatory interruptions due to non-compliance to cGMP guidelines. Injectables, being sterile products, require stringent manufacturing processes across development, formulation, packaging, storage and transportation phases and attract greater scrutiny from regulatory agencies. Approximately 65% of the drug shortages in the US are because of quality manufacturing/ delays or capacity constraints and thereby making them one of the critical success factors to build sustainable business.
INDIA OUTLOOK
Stable Profitability Supports Stable Outlook: It is expected that a stable outlook on the pharmaceutical sector for FY18 will be maintained, on the expectation that the sector’s profitability will remain stable. Domestic market growth to remain brisk and compensate for weak export growth.
Exports continue to Support Profitability: Though export revenue growth remained weak, the profitability of companies exporting to regulated markets improved during FY16 and FY17. Indian companies received close to 200 abbreviated new drug application (ANDA) approvals each in 2015 and 2016 (2014: 122) from US Food and Drug Administration (USFDA). A higher proportion of revenue from new products during this period aided in margin expansion. The intensity of new actions by the USFDA was lower in 2016. Eight warning letters and three import alerts (2015: 17 and 12, respectively) were issued and the number of facilities under import alert has now increased to 45. Increased regulatory actions by the USFDA has resulted in export revenue going down to 6.6% CAGR for the three years ended FY16 as compared to the 13.7% CAGR for the six years ended FY16.
M&A can delay deleveraging: It is anticipated that the pharmaceutical companies may engage in M&A to overcome regulatory and competition headwinds. The high intensity of competition among peers to acquire ‘attractive’ assets can lead to an overheated market. We also expect companies with strong balance sheets to take advantage of favorable credit market conditions and use debt to fund acquisitions, which can delay deleveraging. A sustained improvement in export growth of the sector and/ or an increase in the proportion of revenue from differentiated generic products resulting in a sustained improvement in operating profitability can be positive for the sector.
Sale of Global
Generic Injectables Business to Baxter International Inc.
The injectable generics sector has been through a sustained period of consolidation over the last few years as the leading companies have increased their presence through acquisition. Interest in this specialist sector has come from both established generic manufacturers and major pharmaceutical companies.
They had announced on December 15, 2016, that we have entered into definitive agreements with Baxter International Inc. (Baxter) for the sale of their global generic Speciality Injectables business, carried on by them through their subsidiary Claris Injectables Limited (CIL) and their other identified indirect subsidiaries, at an enterprise value of US$ 625 million on a cash free debt free basis.
CIL manufactures and/or markets products across multiple delivery systems, markets, and therapeutic segments including anesthesia, anti-infectives, critical care and nephrology. A significant majority of these products are generic drugs, capable of being directly injected into the human body, predominantly used in the treatment of critical illnesses. The customer base primarily includes government & private hospitals, aid agencies, and Group Purchasing Organizations. With emphasis on quality, technology, and innovation, theyoffer a range of niche technology-driven injectable products across delivery systems such as glass bottles, vials & ampoules, and non-PVC/PVC bags.
The Injectables business, has been growing rapidly over the last few years and has been attracting significant interest. The business encompasses several specialty high entry barrier products and it has capabilities and technologies that enables an integrated business model from research to marketing with an advantage of lower cost of production in India as compared to the other developed countries. We believe that this strategic partnership with Baxter will create a very promising future roadmap for the business including for the team and the stakeholders. Over the last 5 years, the management and team at the Company have scaled up and created significant value across both their businesses – the earlier infusions business and now the Speciality Injectables.
Baxter International Inc. is a US$ 10 billion (FY2015) revenue company, employing around 50,000 employees worldwide with manufacturing presence in more than 27countries world-wide. Baxter provides a broad portfolio of essential renal and hospital products, including home, acute and in-center dialysis; sterile IV solutions; infusion systems and devices; parenteral nutrition; bio-surgery products and anesthetics; and pharmacy automation, software and services. The company’s global footprint and the critical nature of its products and services play a key role in expanding access to healthcare in emerging and developed countries. Baxter’s employees worldwide are building upon the company’s rich heritage of medical breakthroughs to advance the next generation of healthcare innovations that enable patient care.
Baxter is a company with a very deep and long history and roots in the hospital business with a focused strategy to grow in Injectables market and is perfectly suited to take their business forward. It has the requisite expertise and the resources along with its global reach to leverage the business as a strategic platform and to catapult its R&D, manufacturing and people capabilities and build it into a world class Injectables story. After achieving this milestone of signing definitive agreement, we along with Baxter are working towards closing the transaction and make it fully effective.
The Board of Directors in their meeting held on December 15,
2016, has approved sale and transfer of the ‘Injectables Business’ carried on
by them in India and overseas, through their subsidiary Claris Injectables
Limited and their other identified indirect subsidiaries, through one or more
transactions involving the transfer of ownership of the subsidiary(ies) to the
Baxter Group at an aggregate enterprise value of USD 625 million for the said
transaction relating to the sale of Injectables business, subject to agreed
adjustments, permitted under applicable law, including for repayment of lenders
debt, certain inter-group transactions, and other closing adjustments, which
may be substantial. We have taken the approval of their Company’s shareholders
through the postal ballot on February 17, 2017. Accordingly, the Injectables
business is considered as Discontinued Operations in terms of Ind-AS 105 in
their standalone and consolidated financials. We intend to repatriate a
significant majority of the net cash proceeds (post taxes and expenses) to
their Company’s shareholders.
FINANCIAL PERFORMANCE
The financial statements have been prepared in accordance with Indian Accounting Standards (Ind AS) notified under the Companies (Indian Accounting Standards) Rules, 2015.
For all the periods,
up to and including the year ended March 31, 2016, we prepared their financial
statements in accordance with the accounting standards notified under the
section 133 of the Companies Act 2013, read together with paragraph 7 of the
Companies (Accounts) Rules, 2014 (Indian GAAP). We have adopted IND AS for the
year ended on March 31, 2017 with transition date of April 1, 2015.
Accordingly, these financial statements are their first financial statements
prepared in accordance with Ind AS. The financial statements for the year ended
on March 31, 2016 are restated in accordance with IND AS.
INDEX
OF CHARGES
|
SNo |
SRN |
Charge Id |
Charge Holder Name |
Date of Creation |
Date of
Modification |
Date of
Satisfaction |
Amount |
Address |
|
1 |
G10004240 |
100046036 |
ICICI BANK LIMITED |
12/08/2016 |
- |
- |
2300000.0 |
LANDMARKRACE COURCE CIRCLEALKAPURIBARODAGu390015IN |
|
2 |
G05463526 |
100032156 |
ICICI BANK LIMITED |
24/05/2016 |
- |
- |
2054000.0 |
LANDMARKRACE COURCE CIRCLEALKAPURIBARODAGu390015IN |
|
3 |
G05463971 |
100032158 |
ICICI BANK LIMITED |
24/05/2016 |
- |
- |
3750000.0 |
LANDMARKRACE COURCE CIRCLEALKAPURIBARODAGu390015IN |
|
4 |
G05463112 |
100032154 |
ICICI BANK LIMITED |
24/05/2016 |
- |
- |
3500000.0 |
LANDMARKRACE COURCE CIRCLEALKAPURIBARODAGu390015IN |
|
5 |
G52120540 |
100053478 |
Punjab National Bank |
30/09/2016 |
- |
24/08/2017 |
200000000.0 |
Large Corporate Branch, Neelkamal BuildingAshram RoadAhmedabadGJ380009IN |
|
6 |
G05862065 |
10459609 |
TATA CAPITAL FINANCIAL SERVICES LIMITED |
24/10/2013 |
- |
08/06/2016 |
150000000.0 |
One Forbes,Dr. V. B. Gandhi Marg, FortMumbaiMH400001IN |
|
7 |
G04747044 |
10551917 |
Deutsche Bank AG |
02/02/2015 |
- |
25/05/2016 |
742080000.0 |
ECE House, 28 Kasturba Gandhi Marg,New DelhiDL110001IN |
|
8 |
C31931579 |
10168330 |
Canara Bank |
23/07/2009 |
31/07/2013 |
31/10/2014 |
3354100000.0 |
Prime Corporate Branch, 104, Jyoti Commercial CompNear Shyamal cross road, Satellite, VejalpurAhmedabadGJ380015IN |
|
9 |
C31929482 |
10129270 |
Canara Bank |
17/11/2008 |
31/07/2013 |
31/10/2014 |
3354100000.0 |
Prime Corporate Branch, 104, Jyoti Commercial CompNear Shyamal cross road, Satellite, VejalpurAhmedabadGJ380015IN |
|
10 |
B80640923 |
10250995 |
Central of Bank India |
03/11/2010 |
- |
31/07/2013 |
470400000.0 |
Gulbai Tekra Branch,101,Sears Tower,EllisbridgeGJ380006IN |
CONTINGENT
LIABILITIES:
(INR in million)
|
PARTICULARS |
31.03.2017 |
31.03.2016 |
|
Claim against the company not acknowledge as debts |
249.121 |
87.209 |
|
Disputed demand under : |
|
|
|
35.420 |
38.220 |
|
4.771 |
1.785 |
|
9.219 |
9.219 |
|
1040.000 |
1040.000 |
|
- |
0.726 |
|
Bills discounted |
14.786 |
102.755 |
|
Guarantees given by the Company |
5048.474 |
8642.377 |
STATEMENT OF
STANDALONE UNAUDITED FINANCIAL RESULTS FOR THE HALF YEAR AND QUARTER ENDED
30.09.2017
(INR
In Million)
|
PARTICULARS |
QUARTER ENDED |
HALF YEAR ENDED 30.09.2017 |
|
|
30.09.2017 Unaudited |
30.06.2017 Unaudited |
||
|
|
|
|
|
|
|
|
|
|
|
REVENUE FROM OPERATIONS |
|
|
|
|
Revenue from operations |
27.274 |
15.233 |
42.507 |
|
Other Income |
438.339 |
101.233 |
539.562 |
|
Total
Income |
465.613 |
116.466 |
582.069 |
|
EXPENSES |
|
|
|
|
Employee benefits expense |
75.091 |
110.247 |
185.338 |
|
Finance Costs |
7.545 |
8.923 |
16.468 |
|
Depreciation and Amortization expenses |
10.919 |
10.419 |
21.339 |
|
Other Expenditure |
53.348 |
43.684 |
97.031 |
|
Total
Expenses |
146.903 |
173.273 |
320.176 |
|
Profit
/ (Loss) before share of profit or loss of associates, exceptional item and
tax from continuing operation |
318.710 |
(56.817) |
261.893 |
|
Share in profit/ loss of Associate |
-- |
- |
- |
|
Profit
/ (Loss) before exceptional item and tax from continuing operation |
318.710 |
(56.817) |
261.893 |
|
Exceptional item |
-- |
- |
- |
|
Profit
/ (Loss) before tax from continuing operation |
318.710 |
(56.817) |
261.893 |
|
Tax Expense |
|
|
|
|
Current Tax |
- |
- |
- |
|
Deferred Tax Liability |
115.582 |
(22.420) |
93.162 |
|
Tax
Expense |
115.582 |
(22.420) |
93.162 |
|
Profit
/ (Loss) for the period from continuing operation |
203.128 |
(34.397) |
168.731 |
|
Profit / (Loss) for the period from discontinuing
operation |
2289.173 |
53.214 |
22942.387 |
|
Tax Expense discontinuing operation |
6155.567 |
18.416 |
6173.984 |
|
Profit
/ (Loss) for the period from continuing operation |
16733.606 |
34.798 |
16768.403 |
|
Profit
/ (Loss) for the period |
16936.734 |
0.401 |
16937.134 |
|
Other comprehensive
income/expenses |
|
|
|
|
Gain/ loss of on FVTOCI debt instrument |
(21.966) |
2.728 |
(19.238) |
|
income tax after effect |
7.602 |
(0.944) |
6.658 |
|
Items that will not be reclassified to profit and loss |
|
|
|
|
Re- measurment gains / losses on defied benefit plans |
(0.346) |
(2.377) |
(2.723) |
|
income tax after effect |
0.120 |
0.823 |
0.942 |
|
comprehensive income for the period |
(14.590) |
0.230 |
(14.361) |
|
Total comprehensive
income for the period |
16922.144 |
0.631 |
16922.773 |
|
Paid-up equity share capital |
545.678 |
545.678 |
545.678 |
|
Basic and Diluted EPS - continuing operation |
3.72 |
(0.63) |
3.09 |
|
Basic and Diluted EPS - Discontinuing operation |
306.66 |
0.64 |
307.30 |
|
Basic and Diluted
EPS - Continuing and Discontinuing
operation |
310.38 |
0.01 |
310.39 |
STATEMENT OF UNAUDITED STANDALONE ASSETS AND LIABILITIES AS ON
30.09.2017
(INR in
Million)
|
SOURCES
OF FUNDS |
|
30.09.2017 |
|
|
Unaudited |
|
|
I.
EQUITY AND LIABILITIES |
|
|
|
(1)
Shareholders' Funds |
|
|
|
(a) Share Capital |
|
545.678 |
|
(b) Reserves & Surplus |
|
23648.520 |
|
(c) Money received against
share warrants |
|
0.000 |
|
|
|
|
|
(d)
Share Application Money Pending Allotment |
|
0.000 |
|
Total
Shareholders’ Funds |
|
24194.198 |
|
|
|
|
|
(3)
Non-Current Liabilities |
|
|
|
(a) long-term borrowings |
|
10.000 |
|
(b) Deferred tax liabilities
(Net) |
|
0.000 |
|
(c) Other long term
liabilities |
|
0.000 |
|
(d) long-term provisions |
|
69.355 |
|
Total
Non-current Liabilities |
|
79.355 |
|
|
|
|
|
(4) Current Liabilities |
|
|
|
(a) Short term borrowings |
|
0.000 |
|
(b) Trade payables |
|
536.856 |
|
(c) Other current liabilities |
|
9932.524 |
|
(d) Short-term provisions |
|
4.305 |
|
Total
Current Liabilities |
|
10473.685 |
|
|
|
|
|
TOTAL |
|
34747.238 |
|
|
|
|
|
II.
ASSETS |
|
|
|
(1) Non-current assets |
|
|
|
(a) Fixed Assets |
|
|
|
(i) Tangible assets |
|
475.702 |
|
(ii) Intangible Assets |
|
4.666 |
|
(iii) Capital work-in-progress |
|
46.161 |
|
(iv) Intangible assets under
development |
|
0.000 |
|
(b) Non-current Investments |
|
428.173 |
|
(c) Deferred tax assets (net) |
|
156.466 |
|
(d) Long-term Loan and
Advances |
|
0.000 |
|
(e) Other Non-current assets |
|
101.391 |
|
Total
Non-Current Assets |
|
1212.559 |
|
|
|
|
|
(2) Current assets |
|
|
|
(a) Current investments |
|
2947.110 |
|
(b) Inventories |
|
0.000 |
|
(c) Trade receivables |
|
333.560 |
|
(d) Cash and cash equivalents |
|
3354.159 |
|
(e) Short-term loans and advances |
|
153.908 |
|
(f) Other current assets |
|
222.942 |
|
Total Current Assets |
|
33534.679 |
|
|
|
|
|
TOTAL |
|
34747.238 |
Notes :
- The above financial results have been reviewed by the Audit
Committee and approved to issue by the Board of Directors at their meeting
held on November 14, 2017.
- On July 27,
2017, post receipt of all the regulatory approvals as may be required ,
Company has received sale consideration thereof related to transfer of
Injectable Business’ carried on by the Company in India and Overseas, though
its Subsidiary Claries Injectables Limited and other identified indirect
subsidiaries of the Company, through one or more transactions involving
the transfer of ownership of the subsidiary(ies) to the Baxter Group and
transferred the Injectable Business to Baxter on such date.
On September 21, 2017, post receipt of all the regulatory approvals. Company received sale consideration thereof for transfer of its 20% stake in erstwhile associate, Otsuka Pharmaceutical India Private Limited (formerly known as Clans Otsuka Private Limited). Accordingly, the said stake stands transferred on such date.
In line with
Ind-AS 105, Injectable business and holding in Otsuka Phannaceutical India
Private Limited, were classified as Discontinuing operations for the year ended
March 31, 2017 and quarter ended June 30, 2017 respectively, hence, profit on
transfer of such operations, net off the expenses to sale and tax thereon has
been treated as profit from discontinuing operations.
- Tax
expenses include current tax, deferred tax and adjustment of taxes for the
earlier years/period.
- Following
the completion of sale of Injectable business composing of the Group's
Drug & Pharmaceuticals segment, Clans Lifesciences is evaluating
various sectors to invest proceeds received on sale of its injectable
business, m a way that results in long term value creation for
shareholders. The operating segment of the company continues to be
identified to be "Drug and Phannaceuticals".
- Previous
period's figures have been regrouped/rearranged wherever considered
necessary, to confirm to the current period s classification.
FIXED ASSETS
Tangible Asset
- Buildings
- Plant and Equipment
- Electrical Installation
- Furniture and Fixtures
- Office Equipment
- Vehicles
Intangible Asset
- Computer Software
CMT REPORT (Corruption, Money Laundering & Terrorism]
The Public Notice information has been collected from various sources
including but not limited to: The Courts,
1] INFORMATION ON
DESIGNATED PARTY
No exist designating subject or any of its beneficial owners,
controlling shareholders or senior officers as terrorist or terrorist
organization or whom notice had been received that all financial transactions
involving their assets have been blocked or convicted, found guilty or against
whom a judgement or order had been entered in a proceedings for violating
money-laundering, anti-corruption or bribery or international economic or
anti-terrorism sanction laws or whose assets were seized, blocked, frozen or
ordered forfeited for violation of money laundering or international
anti-terrorism laws.
2] Court Declaration :
No records exist to suggest that subject is
or was the subject of any formal or informal allegations, prosecutions or other
official proceeding for making any prohibited payments or other improper
payments to government officials for engaging in prohibited transactions or
with designated parties.
3] Asset Declaration :
No records exist to suggest that the property or assets of the subject
are derived from criminal conduct or a prohibited transaction.
4] Record on Financial
Crime :
Charges or conviction
registered against subject: None
5] Records on Violation of
Anti-Corruption Laws :
Charges or
investigation registered against subject: None
6] Records on Int’l
Anti-Money Laundering Laws/Standards :
Charges or
investigation registered against subject: None
7] Criminal Records
No
available information exist that suggest that subject or any of its principals
have been formally charged or convicted by a competent governmental authority
for any financial crime or under any formal investigation by a competent
government authority for any violation of anti-corruption laws or international
anti-money laundering laws or standard.
8] Affiliation with
Government :
No record
exists to suggest that any director or indirect owners, controlling
shareholders, director, officer or employee of the company is a government
official or a family member or close business associate of a Government
official.
9] Compensation Package :
Our market
survey revealed that the amount of compensation sought by the subject is fair
and reasonable and comparable to compensation paid to others for similar
services.
10] Press Report :
No press reports / filings exists on
the subject.
CORPORATE GOVERNANCE
MIRA INFORM as part of its Due Diligence do provide comments on
Corporate Governance to identify management and governance. These factors often
have been predictive and in some cases have created vulnerabilities to credit
deterioration.
Our Governance Assessment focuses principally on the interactions
between a company’s management, its Board of Directors, Shareholders and other
financial stakeholders.
CONTRAVENTION
Subject is not known to have contravened any existing local laws,
regulations or policies that prohibit, restrict or otherwise affect the terms
and conditions that could be included in the agreement with the subject.
FOREIGN EXCHANGE RATES
|
Currency |
Unit
|
INR |
|
US Dollar |
1 |
INR 63.82 |
|
|
1 |
INR 86.28 |
|
Euro |
1 |
INR 76.13 |
INFORMATION DETAILS
|
Analysis Done by
: |
VIK |
|
|
|
|
Report Prepared
by : |
TRUP |
SCORE FACTORS
|
DEMERIT POINTS |
|
|
|
--BANK CHARGES |
YES/NO |
YES |
|
--LITIGATION |
YES/NO |
NO |
|
--OTHER ADVERSE INFORMATION |
YES/NO |
NO |
|
MERIT POINTS |
|
|
|
--SOLE DISTRIBUTORSHIP |
YES/NO |
NO |
|
--EXPORT ACTIVITIES |
YES/NO |
NO |
|
--AFFILIATION |
YES/NO |
YES |
|
--LISTED |
YES/NO |
NO |
|
--OTHER MERIT FACTORS |
YES/NO |
YES |
RATING EXPLANATIONS
|
Credit Rating |
Explanation |
Rating Comments |
|
A++ |
Minimum Risk |
Business dealings permissible with minimum
risk of default |
|
A+ |
Low Risk |
Business dealings permissible with low
risk of default |
|
A |
Acceptable Risk |
Business dealings permissible with
moderate risk of default |
|
B |
Medium Risk |
Business dealings permissible on a regular
monitoring basis |
|
C |
Medium High Risk |
Business dealings permissible preferably
on secured basis |
|
D |
High Risk |
Business dealing not recommended or on
secured terms only |
|
NB |
New Business |
No recommendation can be done due to
business in infancy stage |
|
NT |
No Trace |
No recommendation can be done as the
business is not traceable |
NB is stated where there is insufficient information to facilitate rating. However, it is not to be considered as unfavourable.
This score serves as a reference to assess
SC’s credit risk and to set the amount of credit to be extended. It is
calculated from a composite of weighted scores obtained from each of the major
sections of this report. The assessed factors are as follows:
·
Financial
condition covering various ratios
·
Company
background and operations size
·
Promoters
/ Management background
·
Payment
record
·
Litigation
against the subject
·
Industry
scenario / competitor analysis
·
Supplier
/ Customer / Banker review (wherever available)
This report is issued at
your request without any risk and responsibility on the part of MIRA INFORM
PRIVATE LIMITED (MIPL) or its officials.

