|
|
|
MIRA INFORM REPORT
|
Report No. : |
486275 |
|
Report Date : |
13.01.2018 |
IDENTIFICATION DETAILS
|
Name : |
HOSPIRA HEALTHCARE INDIA PRIVATE LIMITED (w.e.f. 21.12.2009) |
|
|
|
|
Formerly Known
As : |
OJAS PHARMACEUTICALS INDIA PRIVATE LIMITED |
|
|
|
|
Registered
Office : |
Plot No. B3, SIPCOT Industrial Park, Irungattukottai, Sriperumbudur,
Kancheepuram – 602117, Tamilnadu |
|
Tel. No.: |
91-44-39370000 |
|
|
|
|
Country : |
India |
|
|
|
|
Financials (as
on) : |
31.03.2016 |
|
|
|
|
Date of
Incorporation : |
17.11.2009 |
|
|
|
|
Com. Reg. No.: |
18-073563 |
|
|
|
|
Capital
Investment / Paid-up Capital : |
INR 39.877 Million |
|
|
|
|
CIN No.: [Company Identification
No.] |
U24232TN2009PTC073563 |
|
|
|
|
IEC No.: |
0409039471 |
|
|
|
|
TAN No.: [Tax Deduction &
Collection Account No.] |
Not Available |
|
|
|
|
PAN No.: [Permanent Account No.] |
AABCO2190F |
|
|
|
|
Legal Form : |
Private Limited Liability Company |
|
|
|
|
Line of Business
: |
Manufacturer of Speciality Injectable Pharmaceuticals (SIP) and Active Pharmaceutical Ingredient (API). [Registered Activity] |
|
|
|
|
No. of
Employees: |
Not Available |
RATING & COMMENTS
(Mira Inform has adopted New Rating mechanism w.e.f. 23rd
January 2017)
|
MIRA’s Rating : |
A+ |
|
Credit Rating |
Explanation |
Rating Comments |
|
A+ |
Low Risk |
Business dealings permissible with low
risk of default |
|
Maximum Credit Limit : |
USD 68000000 |
|
|
|
|
Status : |
Good |
|
|
|
|
Payment Behaviour : |
Regular |
|
|
|
|
Litigation : |
Clear |
|
|
|
|
Comments : |
Subject is a subsidiary of “Hospira Inc”. It was incorporated in the year 2009 and is engaged in manufacturing of pharmaceuticals products and medical devices. It is an established company having fine track record. As per the Registrar of Companies date of balance sheet (i.e.
financial filed) is shown as 31.03.2017 but documents related to financial
for the year 31.03.2017 are not available from any sources. The company has reflects significant improvement in the operational profile along with healthy net profit against previous year net losses. The rating takes into consideration on company’s healthy financial profile
marked by adequate networth base and strong financial as well as managerial
support that company receives from its parent company. Trade relations are fair. Payments are regular and as per commitment. In view of strong parental support, the company can be considered for
business dealings at usual trade terms and conditions. |
NOTES :
Any query related to this report can be made
on e-mail : infodept@mirainform.com
while quoting report number, name and date.
ECGC Country Risk Classification List
|
Country Name |
Previous Rating (30.06.2017) |
Current Rating (30.09.2017) |
|
India |
A1 |
A1 |
|
Risk Category |
ECGC
Classification |
|
Insignificant |
A1 |
|
Low Risk |
A2 |
|
Moderately Low Risk |
B1 |
|
Moderate Risk |
B2 |
|
Moderately High Risk |
C1 |
|
High Risk |
C2 |
|
Very High Risk |
D |
EXTERNAL AGENCY RATING
NOT AVAILABLE
RBI DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available RBI Defaulters’ list.
EPF (Employee Provident Fund) DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available EPF (Employee Provident Fund) Defaulters’ list as of
31-03-2016.
BIFR (Board for Industrial & Financial Reconstruction) LISTING
STATUS
Subject’s name is not listed as a Sick Unit in
the publicly available BIFR (Board for Industrial & Financial
Reconstruction) list as of 13.01.2018
IBBI (Insolvency and Bankruptcy Board of India) LISTING STATUS
Subject’s name is not listed in the publicly
available IBBI (Insolvency and Bankruptcy Board of India) list as of report
date.
LOCATIONS
|
Registered Office/ Factory 1 : |
Plot No. B3, SIPCOT Industrial Park, Irungattukottai, Sriperumbudur,
Kancheepuram – 602117, Tamilnadu, India |
|
Tel. No.: |
91-44-27141111/39370000 |
|
Fax No.: |
91-44-27156816/28151116 |
|
E-Mail : |
|
|
Website: |
|
|
|
|
|
Factory 2 : |
City SEZ, Plot No. 116 to 119, Part 111 and 123 Jawaharial
Nehru Pharma City, Visakapatnam – 531019, Andhra Pradesh, India |
|
|
|
|
Corporate office/Factory 3 : |
Sri Nivas, New No. - 86 (Old No. 89) G. N. Chetty, Chennai – 600017, Tamilnadu, India |
|
|
|
|
Factory 4 : |
L-8(P), L-9, Gut No.:38 (P) HHIP Aura-API-1, Aurangabad-431136,
Maharashtra, India |
|
Tel No.: |
91-240-2567399 |
|
|
|
|
Research development office : |
129-140, Greams Road, Thousand Lights, 7th Floor, Prestige Palladium
Building, Chennai- 600006, Tamilnadu, India |
|
|
|
|
Factory 5 : |
476/14, Old Mahabalopuram Road, Sholinganallur, Chennai – 600119,
Tamilnadu, India |
DIRECTORS
AS ON 31.03.2017
|
Name : |
Mr. Balasubramanian Srinivasan |
||||||||
|
Designation : |
Managing Director |
||||||||
|
Address : |
Rainenweg 136 Reinach BL 4153 CH |
||||||||
|
Date of birth/age: |
26.05.1963 |
||||||||
|
Qualification: |
Master of Business Administartion (University of London); Bachelor of Pharmacy (University of Madras) |
||||||||
|
Date of Appointment : |
17.03.2015 |
||||||||
|
DIN No.: |
07121117 |
||||||||
|
Other Directorship:
|
|||||||||
|
|
|
||||||||
|
Name : |
Mr. Raviprakash Venkata Subbaraya Bhagavathula |
||||||||
|
Designation : |
Director |
||||||||
|
Address : |
Flat No. 1101, 11th Floor, Seasons Heights, Chapel Lane, Santacruz
West Mumbai – 400054, Maharashtra, India |
||||||||
|
Date of Appointment : |
23.12.2015 |
||||||||
|
DIN No.: |
07282100 |
||||||||
|
Other Directorship:
|
|||||||||
|
|
|
||||||||
|
Name : |
Mr. Niall Joseph Condon |
||||||||
|
Designation : |
Director |
||||||||
|
Address : |
11, Logging Road Katonah New York 10536 US |
||||||||
|
Date of Appointment : |
23.12.2015 |
||||||||
|
DIN No.: |
07413594 |
||||||||
|
|
|
||||||||
|
Name : |
Gordon Edward Loh |
||||||||
|
Designation : |
Director |
||||||||
|
Address : |
219-5-5, 235 East 42 Street, New York 10017 United States |
||||||||
|
Date of Appointment : |
29.08.2016 |
||||||||
|
DIN No.: |
07791140 |
||||||||
KEY EXECUTIVES
|
Name : |
Kavita Surana |
|
Designation : |
Company Secretary |
|
Address : |
71-73, Perambur Barracks Road, Vepery, Chennai – 600007,
Tamilnadu, India |
|
Date of Appointment : |
13.09.2016 |
|
PAN No.: |
CGVPS2778G |
MAJOR SHAREHOLDERS
AS ON 31.03.2017
|
Names of Shareholders |
|
No. of Shares |
|
Hospira Pte Limited, Singapore |
|
3987642 |
|
Hospira Bahamas (Ireland) Corp., Bahamas |
|
10 |
|
|
|
|
|
Total |
|
3987652 |
Equity Share Break up (Percentage of Total Equity)
AS ON 28.09.2016
|
Category |
Percentage |
|
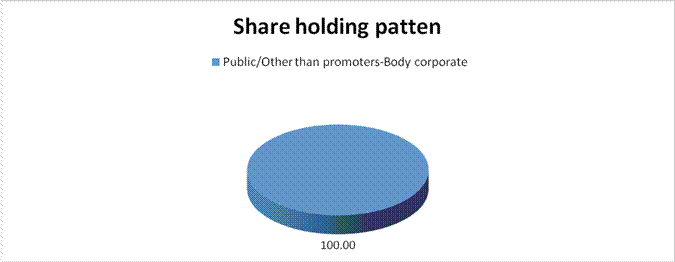
Public/Other than promoters-Body corporate |
100.00 |
|
Total |
100.00 |
BUSINESS DETAILS
|
Line of Business : |
Manufacturer of Speciality Injectable Pharmaceuticals
(SIP) and Active Pharmaceutical Ingredient (API). [Registered Activity] |
||||||
|
|
|
||||||
|
Products / Services
: |
|
||||||
|
|
|
||||||
|
Brand Names : |
Not Available |
||||||
|
|
|
||||||
|
Agencies Held : |
Not Available |
||||||
|
|
|
||||||
|
Exports : |
Not Available |
||||||
|
|
|
||||||
|
Imports : |
Not Available |
||||||
|
|
|
||||||
|
Terms : |
Not Available |
PRODUCTION STATUS – (NOT AVAILABLE)
GENERAL INFORMATION
|
Suppliers : |
|
||||||||||||||||||||||
|
|
|
||||||||||||||||||||||
|
Customers : |
|
||||||||||||||||||||||
|
|
|
||||||||||||||||||||||
|
No. of Employees
: |
Not Available |
||||||||||||||||||||||
|
|
|
||||||||||||||||||||||
|
Bankers : |
|
||||||||||||||||||||||
|
|
|
||||||||||||||||||||||
|
Facilities : |
(INR
In Million)
|
|
Auditors : |
|
|
Name : |
B S R and Company LLP Chartered Accountants |
|
Address : |
Building No.10, 8th Floor, Tower B, DLF Cyber City, Phase II, Gurugram-122002, Haryana, India |
|
PAN No.: |
AAAFB9852F |
|
|
|
|
Memberships : |
Not Available |
|
|
|
|
Collaborators : |
Not Available |
|
|
|
|
Holding Company (AS
ON 31.03.2016): |
|
|
|
|
|
Ultimate Holding
Company (AS ON 31.03.2016): |
|
|
|
|
|
Fellow Subsidiaries
- With whom transactions have taken place during the year (AS ON 31.03.2016): |
|
CAPITAL STRUCTURE
AS ON 28.09.2017
Authorised Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
50,00,000 |
Equity Shares |
INR 10/- each |
INR 50.000 Million |
|
|
|
|
|
Issued, Subscribed & Paid-up Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
39,87,652 |
Equity Shares |
INR 10/- each |
INR 39.877 Million |
|
|
|
|
|
FINANCIAL DATA
[all figures are
INR Million]
ABRIDGED
BALANCE SHEET
|
SOURCES OF FUNDS |
31.03.2016 |
31.03.2015 |
31.03.2014 |
|
I.
EQUITY
AND LIABILITIES |
|
|
|
|
(1)Shareholders' Funds |
|
|
|
|
(a) Share Capital |
39.877 |
39.877 |
28.531 |
|
(b) Reserves and Surplus |
23704.723 |
20347.023 |
15601.691 |
|
(c) Money
received against share warrants |
0.000 |
0.000 |
300.300 |
|
|
|
|
|
|
(2) Share Application money pending
allotment |
0.000 |
0.000 |
0.000 |
|
Total
Shareholders’ Funds (1) + (2) |
23744.600 |
20386.900 |
15930.522 |
|
|
|
|
|
|
(3)
Non-Current Liabilities |
|
|
|
|
(a) long-term borrowings |
35233.800 |
29398.900 |
15031.060 |
|
(b) Deferred tax liabilities (Net) |
0.000 |
0.000 |
0.000 |
|
(c) Other long-term
liabilities |
37.000 |
31.600 |
22.109 |
|
(d) long-term
provisions |
189.900 |
170.800 |
81.820 |
|
Total Non-current
Liabilities (3) |
35460.700 |
29601.300 |
15134.989 |
|
|
|
|
|
|
(4)
Current Liabilities |
|
|
|
|
(a) Short-term
borrowings |
0.000 |
0.000 |
1778.501 |
|
(b) Trade
payables |
2858.600 |
2871.500 |
1305.465 |
|
(c) Other
current liabilities |
7885.500 |
3014.500 |
1211.921 |
|
(d) Short-term
provisions |
27.600 |
41.400 |
9.029 |
|
Total Current
Liabilities (4) |
10771.700 |
5927.400 |
4304.916 |
|
|
|
|
|
|
TOTAL |
69977.000 |
55915.600 |
35370.427 |
|
|
|
|
|
|
II.
ASSETS |
|
|
|
|
(1)
Non-current assets |
|
|
|
|
(a) Fixed
Assets |
|
|
|
|
(i)
Tangible assets |
31342.400 |
21243.600 |
8485.306 |
|
(ii)
Intangible Assets |
4011.700 |
5136.800 |
3418.449 |
|
(iii) Tangible assets
capital work-in-progress |
9101.200 |
15058.600 |
12800.933 |
|
(iv)
Intangible assets under development |
226.500 |
226.500 |
0.000 |
|
(b) Non-current Investments |
0.400 |
0.400 |
0.429 |
|
(c) Deferred tax assets (net) |
0.000 |
0.000 |
0.000 |
|
(d)
Long-term
loans and advances |
452.100 |
995.900 |
1847.239 |
|
(e) Other Non-current
assets |
0.000 |
0.000 |
0.000 |
|
Total Non-Current
Assets |
45134.300 |
42661.800 |
26552.356 |
|
|
|
|
|
|
(2)
Current assets |
|
|
|
|
(a)
Current investments |
0.000 |
0.000 |
0.000 |
|
(b)
Inventories |
9119.400 |
6960.800 |
4724.888 |
|
(c) Trade
receivables |
7050.100 |
1801.200 |
1550.128 |
|
(d) Cash and bank
balances |
5130.000 |
2566.900 |
1476.864 |
|
(e)
Short-term loans and advances |
1716.300 |
1136.400 |
1066.191 |
|
(f) Other
current assets |
1826.900 |
788.500 |
0.000 |
|
Total
Current Assets |
24842.700 |
13253.800 |
8818.071 |
|
|
|
|
|
|
TOTAL |
69977.000 |
55915.600 |
35370.427 |
PROFIT
& LOSS ACCOUNT
|
|
PARTICULARS |
31.03.2016 |
31.03.2015 |
31.03.2014 |
|
|
SALES |
|
|
|
|
|
Income |
17731.200 |
12934.900 |
10141.129 |
|
|
Other Income |
5933.400 |
18.900 |
14.227 |
|
|
TOTAL |
23664.600 |
12953.800 |
10155.356 |
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
Cost of Materials
Consumed |
5294.600 |
9817.500 |
|
|
|
Changes in inventories of
finished goods, work-in-progress and Stock-in-Trade |
(538.000) |
(3060.700) |
|
|
|
Employee benefit expense |
4143.800 |
2336.200 |
|
|
|
Other expenses |
7172.100 |
6141.600 |
|
|
|
TOTAL |
16072.500 |
15234.600 |
11209.317 |
|
|
|
|
|
|
|
|
PROFIT/
(LOSS) BEFORE INTEREST, TAX,
DEPRECIATION AND AMORTISATION |
7592.100 |
(2280.800) |
(1053.961) |
|
|
|
|
|
|
|
Less |
FINANCIAL
EXPENSES |
617.400 |
227.400 |
932.636 |
|
|
|
|
|
|
|
|
PROFIT / (LOSS) BEFORE TAX, DEPRECIATION AND
AMORTISATION |
6974.700 |
(2508.200) |
(1986.597) |
|
|
|
|
|
|
|
Less/ Add |
DEPRECIATION/
AMORTISATION |
3617.000 |
4081.200 |
2835.153 |
|
|
|
|
|
|
|
|
PROFIT/ (LOSS) BEFORE
TAX |
3357.700 |
(6589.400) |
(4821.750) |
|
|
|
|
|
|
|
Less |
TAX |
0.000 |
0.000 |
0.000 |
|
|
|
|
|
|
|
|
PROFIT/ (LOSS)
AFTER TAX |
3357.700 |
(6589.400) |
(4821.750) |
|
|
|
|
|
|
|
|
EARNINGS
IN FOREIGN CURRENCY |
|
|
|
|
|
F.O.B. Value of Exports |
9656.400 |
11319.300 |
|
|
|
Earnings on other income |
13420.000 |
1014.700 |
|
|
|
TOTAL EARNINGS |
23076.400 |
12334.000 |
9394.318 |
|
|
|
|
|
|
|
|
IMPORTS |
|
|
|
|
|
Raw Materials |
2893.700 |
2010.100 |
58317.700 |
|
|
Components and Stores parts |
1743.800 |
2533.300 |
50829.500 |
|
|
Capital Goods |
623.700 |
760.300 |
0.000 |
|
|
TOTAL
IMPORTS |
5261.200 |
5303.700 |
109147.200 |
|
|
|
|
|
|
|
|
Earnings / (Loss) Per Share (INR) |
842.00 |
(1804.00) |
(1209.17) |
CURRENT MATURITIES
OF LONG TERM DEBT DETAILS
|
Particulars |
31.03.2016 |
31.03.2015 |
31.03.2014 |
|
Current Maturities of Long term debt |
6707.000 |
0.000 |
NA |
|
|
|
|
|
|
Cash generated from operations |
NA |
NA |
NA |
|
|
|
|
|
|
Net cash flows from (used in) operations |
(1319.800) |
(777.200) |
(1664.044) |
|
|
|
|
|
|
Net cash flows from (used in) operating activities |
(1321.100) |
(780.300) |
(1666.924) |
KEY
RATIOS
EFFICIENCY RATIOS
|
PARTICULARS |
31.03.2016 |
31.03.2015 |
31.03.2014 |
|
Average Collection Days (Sundry
Debtors / Income * 365 Days) |
145.13 |
50.83 |
55.79 |
|
|
|
|
|
|
Account Receivables Turnover (Income / Sundry Debtors) |
2.52 |
7.18 |
6.54 |
|
|
|
|
|
|
Average Payment Days (Sundry Creditors / Purchases * 365 Days) |
197.07 |
106.76 |
NA |
|
|
|
|
|
|
Inventory Turnover (Operating Income / Inventories) |
0.83 |
(0.33) |
(0.22) |
|
|
|
|
|
|
Asset Turnover (Operating Income / Net Fixed Assets) |
0.17 |
(0.05) |
(0.04) |
LEVERAGE RATIOS
|
PARTICULARS |
31.03.2016 |
31.03.2015 |
31.03.2014 |
|
Debt Ratio ((Borrowing
+ Current Liabilities) / Total Assets) |
0.75 |
0.63 |
0.55 |
|
|
|
|
|
|
Debt Equity Ratio (Total Liability / Networth) |
1.77 |
1.44 |
1.06 |
|
|
|
|
|
|
Current Liabilities to Networth (Current Liabilities / Net Worth) |
0.45 |
0.29 |
0.27 |
|
|
|
|
|
|
Fixed Assets to Networth (Net Fixed Assets / Networth) |
1.88 |
2.04 |
1.55 |
|
|
|
|
|
|
Interest Coverage Ratio (PBIT / Financial Charges) |
12.30 |
(10.03) |
(1.13) |
PROFITABILITY RATIOS
|
PARTICULARS |
|
31.03.2016 |
31.03.2015 |
31.03.2014 |
|
Net Profit Margin ((PAT / Sales) * 100) |
% |
18.94 |
(50.94) |
(47.55) |
|
|
|
|
|
|
|
Return on Total Assets ((PAT / Total Assets) * 100) |
% |
4.80 |
(11.78) |
(13.63) |
|
|
|
|
|
|
|
Return on Investment (ROI) ((PAT / Networth) * 100) |
% |
14.14 |
(32.32) |
(30.27) |
SOLVENCY RATIOS
|
PARTICULARS |
31.03.2016 |
31.03.2015 |
31.03.2014 |
|
Current Ratio (Current
Assets / Current Liabilities) |
2.31 |
2.24 |
2.05 |
|
|
|
|
|
|
Quick Ratio ((Current Assets – Inventories) / Current
Liabilities) |
1.46 |
1.06 |
0.95 |
|
|
|
|
|
|
G-Score Ratio Financial (Networth / Total Assets) |
0.34 |
0.36 |
0.45 |
|
|
|
|
|
|
G-Score Ratio Debt (Debts / Equity Capital) |
1051.75 |
737.24 |
589.17 |
|
|
|
|
|
|
G-Score Ratio Liquidity (Total Current Assets / Total Current Liabilities) |
2.31 |
2.24 |
2.05 |
Total
Liability = Short-term Debt + Long-term Debt + Current Maturities of Long-term
debts
FINANCIAL ANALYSIS
[all figures are
INR Million]
DEBT EQUITY RATIO
|
Particular |
31.03.2014 |
31.03.2015 |
31.03.2016 |
|
|
INR
In Million |
INR
In Million |
INR
In Million |
|
Share Capital |
28.531 |
39.877 |
39.877 |
|
Reserves & Surplus |
15601.691 |
20347.023 |
23704.723 |
|
Share Application money pending allotment |
0.000 |
0.000 |
0.000 |
|
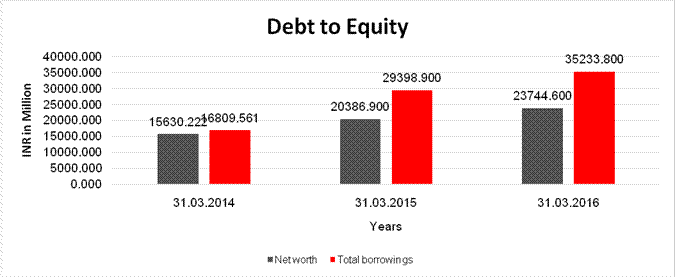
Net
worth |
15630.222 |
20386.900 |
23744.600 |
|
|
|
|
|
|
Long Term borrowings |
15031.060 |
29398.900 |
35233.800 |
|
Short Term borrowings |
1778.501 |
0.000 |
0.000 |
|
Total
borrowings |
16809.561 |
29398.900 |
35233.800 |
|
Debt/Equity
ratio |
1.075 |
1.442 |
1.484 |
YEAR-ON-YEAR GROWTH
|
Year
on Year Growth |
31.03.2014 |
31.03.2015 |
31.03.2016 |
|
|
INR
In Million |
INR
In Million |
INR
In Million |
|
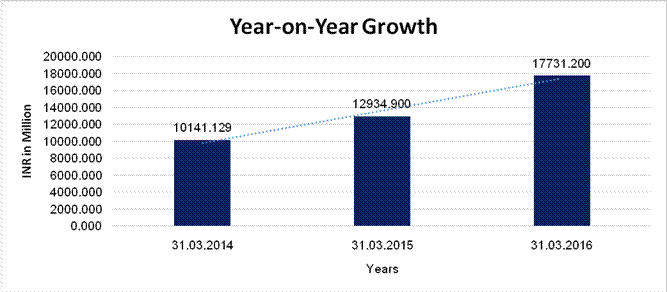
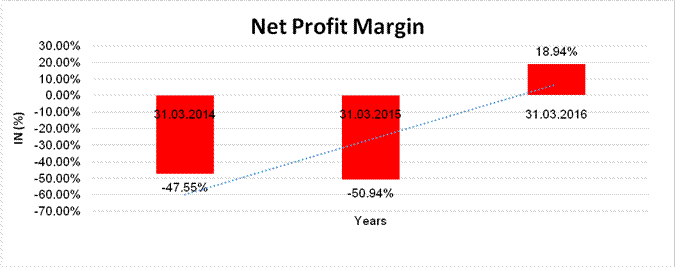
Sales
|
10141.129 |
12934.900 |
17731.200 |
|
|
|
27.549 |
37.080 |
NET PROFIT MARGIN
|
Net
Profit Margin |
31.03.2014 |
31.03.2015 |
31.03.2016 |
|
|
INR
In Million |
INR
In Million |
INR
In Million |
|
Sales
|
10141.129 |
12934.900 |
17731.200 |
|
Profit/(Loss) |
(4821.750) |
(6589.400) |
3357.700 |
|
|
(47.55%) |
(50.94%) |
18.94% |
LOCAL AGENCY FURTHER INFORMATION
|
Sr. No. |
Check list by
info agents |
Available in
Report (Yes/No) |
|
1 |
Year of establishment |
Yes |
|
2 |
Constitution of the entity -Incorporation
details |
Yes |
|
3 |
Locality of the entity |
Yes |
|
4 |
Premises details |
No |
|
5 |
Buyer visit details |
-- |
|
6 |
Contact numbers |
Yes |
|
7 |
Name of the person contacted |
No |
|
8 |
Designation of contact person |
No |
|
9 |
Promoter’s background |
Yes |
|
10 |
Date of Birth of Proprietor / Partners /
Directors |
No |
|
11 |
Pan Card No. of Proprietor / Partners |
No |
|
12 |
Voter Id Card No. of Proprietor / Partners |
No |
|
13 |
Type of business |
Yes |
|
14 |
Line of Business |
Yes |
|
15 |
Export/import details (if applicable) |
No |
|
16 |
No. of employees |
Yes |
|
17 |
Details of sister concerns |
Yes |
|
18 |
Major suppliers |
No |
|
19 |
Major customers |
No |
|
20 |
Banking Details |
No |
|
21 |
Banking facility details |
Yes |
|
22 |
Conduct of the banking account |
-- |
|
23 |
Financials, if provided |
Yes |
|
24 |
Capital in the business |
Yes |
|
25 |
Last accounts filed at ROC, if applicable |
No |
|
26 |
Turnover of firm for last three years |
Yes |
|
27 |
Reasons for variation <> 20% |
-- |
|
28 |
Estimation for coming financial year |
No |
|
29 |
Profitability for last three years |
Yes |
|
30 |
Major shareholders, if available |
Yes |
|
31 |
External Agency Rating, if available |
No |
|
32 |
Litigations that the firm/promoter
involved in |
-- |
|
33 |
Market information |
-- |
|
34 |
Payments terms |
No |
|
35 |
Negative Reporting by Auditors in the
Annual Report |
No |
INDEX OF CHARGE: NO CHARGES EXISTS FOR COMPANY
Note:
The Registered office of the company has been shifted Plot No. B3,
SIPCOT Industrial Park Irungattukottai, Sriperumbudur – 602105, Tamilnadu,
India to the present address w.e.f. 01.11.2013
BACKGROUND (AS ON 31.03.2016):
Subject a wholly
owned subsidiary of Hospira Inc., USA was incorporated in India on November 17,
2009. HHIPL acquired the generic injectable pharmaceuticals business (including
certain manufacturing and Research and Development (“R&D”) assets and drug
pipeline) of Orchid Chemicals and Pharmaceuticals Limited (“Orchid”) on March
30, 2010 pursuant to a Business Transfer Agreement dated December 15, 2009
between the parties. The Company is executing plans to expand its manufacturing
capacity at Irungattukottai and has also set up a new injectables manufacturing
facility at Vizag utilizing the site on the long term lease acquired from
Orchid in March 2010. The Company also acquired the Penem and Penicilin Active
Pharmaceutical Ingredient Business and Associated Research and Development
Facility of Orchid Chemicals and Pharmaceuticals Limited (“Orchid”) on July 04,
2014 pursuant to a Business Transfer Agreement dated August 19, 2012 between
the parties.
On February 05,
2015, the ultimate parent company, Hospira Inc., entered into an Agreement and
Plan of Merger (“Merger Agreement”) with Pfizer Inc. (“Pfizer”) and a wholly
owned Subsidiary of Pfizer, Perkins Holding Company (“Merger Subsidiary”).
Pursuant to the Merger Agreement, subject to the terms and conditions of the
Merger Agreement, at the closing, the Merger Subsidiary will merge with and
into Hospira
Inc., (the
“Merger”), with Hospira Inc., surviving the Merger as a wholly owned subsidiary
of Pfizer, and all of the issued and outstanding shares of Hospira Inc’s common
stock (other than certain excluded shares) will be converted into the right to
receive $90.00 in cash per share.
On September 3,
2015 (the acquisition date), Pfizer acquired Hospira, Inc. (Hospira) for
approximately $16.1 billion in cash ($15.7 billion, net of cash acquired).
Consequent to the acquisition, the ultimate holding company has been changed
from Hospira Inc, to Pfizer Inc., and all of Hospira’s commercial operations
are included within Global Established Pharma (GEP) business.
Overview of Indian Economic and Healthcare
Environment: (AS ON 31.03.2016):
Indias GDP grew by
7.6% in 2015-16, making it one of the fastest growing major economies in the
world. Inflation remained under control and fiscal and current account deficits
continued to remain moderate. The Government of Indias commitment to fiscal
targets, focus on infrastructure creation and attracting investments bolstered
the confidence of entrepreneurs and investors.
The growth in the
healthcare market in India has been buoyant on account of several advantages
and government initiatives on increased access through ongoing programs such as
the National Rural Health Mission (NHRM), expansion of the Universal
Immunization Program, Governmentand State funded health insurance schemes like
Arogyasri and the expansion of the Jan Aushadhi scheme to increase access to
affordable generic drugs.
Governments
commitment to improve competitiveness: Government initiatives like Make in
India, Smart Cities campaign, Pradhan Mantri Jan Dhan Yojana are boost for
urbanization and augur well for the countrys overall development and
competitiveness.
Manufacturing and Quality Operations: (AS ON
31.03.2016):
The Company’s IKKT
Manufacturing facility received Voluntary Action Indicated (VAI) status from
USFDA in June 2015. Visakhapatnam Manufacturing facility received Voluntary
Action Indicated (VAI) status from USFDA. During the year the regulatory
commitments made pursuant to the USFDA inspection at Visakhapatnam manufacturing
site were completed in time. The Company's Aurangabad manufacturing facility
passed through the inspection of CDSCO along with the State FDA in May 2015 and
received the GMP certificate after due submission and on-site verification of
compliance classifying site as Acceptable.
FIXED ASSETS:
Tangible assets
- Land
- Buildings
- Plant and equipment
- Furniture and fixtures
- Office equipment
- Leasehold improvements
- Vehicles
Intangible assets
- Goodwill
- Computer software
- Know-how
- Commercial contractual rights
CMT REPORT (Corruption, Money Laundering & Terrorism]
The Public Notice information has been collected from various sources
including but not limited to: The Courts,
1] INFORMATION ON
DESIGNATED PARTY
No exist designating subject or any of its beneficial owners,
controlling shareholders or senior officers as terrorist or terrorist
organization or whom notice had been received that all financial transactions
involving their assets have been blocked or convicted, found guilty or against
whom a judgement or order had been entered in a proceedings for violating
money-laundering, anti-corruption or bribery or international economic or
anti-terrorism sanction laws or whose assets were seized, blocked, frozen or
ordered forfeited for violation of money laundering or international
anti-terrorism laws.
2] Court Declaration :
No records exist to suggest that subject is
or was the subject of any formal or informal allegations, prosecutions or other
official proceeding for making any prohibited payments or other improper
payments to government officials for engaging in prohibited transactions or
with designated parties.
3] Asset Declaration :
No records exist to suggest that the property or assets of the subject
are derived from criminal conduct or a prohibited transaction.
4] Record on Financial
Crime :
Charges or conviction
registered against subject: None
5] Records on Violation of
Anti-Corruption Laws :
Charges or
investigation registered against subject: None
6] Records on Int’l
Anti-Money Laundering Laws/Standards :
Charges or
investigation registered against subject: None
7] Criminal Records
No
available information exist that suggest that subject or any of its principals have
been formally charged or convicted by a competent governmental authority for
any financial crime or under any formal investigation by a competent government
authority for any violation of anti-corruption laws or international anti-money
laundering laws or standard.
8] Affiliation with
Government :
No record
exists to suggest that any director or indirect owners, controlling
shareholders, director, officer or employee of the company is a government
official or a family member or close business associate of a Government
official.
9] Compensation Package :
Our market
survey revealed that the amount of compensation sought by the subject is fair
and reasonable and comparable to compensation paid to others for similar
services.
10] Press Report :
No press reports / filings exists on
the subject.
CORPORATE GOVERNANCE
MIRA INFORM as part of its Due Diligence do provide comments on
Corporate Governance to identify management and governance. These factors often
have been predictive and in some cases have created vulnerabilities to credit
deterioration.
Our Governance Assessment focuses principally on the interactions
between a company’s management, its Board of Directors, Shareholders and other
financial stakeholders.
CONTRAVENTION
Subject is not known to have contravened any existing local laws,
regulations or policies that prohibit, restrict or otherwise affect the terms
and conditions that could be included in the agreement with the subject.
FOREIGN EXCHANGE RATES
|
Currency |
Unit
|
INR |
|
US Dollar |
1 |
INR 63.53 |
|
|
1 |
INR 86.05 |
|
Euro |
1 |
INR 76.53 |
INFORMATION DETAILS
|
Analysis Done by
: |
NYT |
|
|
|
|
Report Prepared
by : |
KVT |
SCORE FACTORS
|
DEMERIT POINTS |
|
|
|
--BANK CHARGES |
YES/NO |
YES |
|
--LITIGATION |
YES/NO |
NO |
|
--OTHER ADVERSE INFORMATION |
YES/NO |
NO |
|
MERIT POINTS |
|
|
|
--SOLE DISTRIBUTORSHIP |
YES/NO |
NO |
|
--EXPORT ACTIVITIES |
YES/NO |
NO |
|
--AFFILIATION |
YES/NO |
YES |
|
--LISTED |
YES/NO |
NO |
|
--OTHER MERIT FACTORS |
YES/NO |
YES |
RATING EXPLANATIONS
|
Credit Rating |
Explanation |
Rating Comments |
|
A++ |
Minimum Risk |
Business dealings permissible with minimum
risk of default |
|
A+ |
Low Risk |
Business dealings permissible with low
risk of default |
|
A |
Acceptable Risk |
Business dealings permissible with
moderate risk of default |
|
B |
Medium Risk |
Business dealings permissible on a regular
monitoring basis |
|
C |
Medium High Risk |
Business dealings permissible preferably
on secured basis |
|
D |
High Risk |
Business dealing not recommended or on
secured terms only |
|
NB |
New Business |
No recommendation can be done due to
business in infancy stage |
|
NT |
No Trace |
No recommendation can be done as the
business is not traceable |
NB is stated where there is insufficient information to facilitate rating. However, it is not to be considered as unfavourable.
This score serves as a reference to assess
SC’s credit risk and to set the amount of credit to be extended. It is
calculated from a composite of weighted scores obtained from each of the major
sections of this report. The assessed factors are as follows:
·
Financial
condition covering various ratios
·
Company
background and operations size
·
Promoters
/ Management background
·
Payment
record
·
Litigation
against the subject
·
Industry
scenario / competitor analysis
·
Supplier
/ Customer / Banker review (wherever available)
This report is issued at
your request without any risk and responsibility on the part of MIRA INFORM
PRIVATE LIMITED (MIPL) or its officials.

