|
|
|
MIRA
INFORM REPORT
|
Report No. : |
497283 |
|
Report Date : |
10.03.2018 |
IDENTIFICATION DETAILS
|
Name : |
ALEMBIC PHARMACEUTICALS LIMITED (w.e.f. 12.03.2011) |
|
|
|
|
Formerly Known
As : |
ALEMBIC PHARMA LIMITED |
|
|
|
|
Registered
Office : |
Alembic Road, Gorwa, Vadodara - 390003, Gujarat |
|
Tel. No.: |
91-265-2280550/ 2280880/ 3007000/ 2282506 |
|
|
|
|
Country : |
India |
|
|
|
|
Financials (as
on) : |
31.03.2017 |
|
|
|
|
Date of
Incorporation : |
16.06.2010 |
|
|
|
|
Com. Reg. No.: |
04-061123 |
|
|
|
|
Capital
Investment / Paid-up Capital : |
INR 377.032 Million |
|
|
|
|
CIN No.: [Company Identification
No.] |
L24230GJ2010PLC061123 |
|
|
|
|
IEC No.: [Import-Export Code No.] |
3410006061 |
|
|
|
|
TIN No.: |
11985550713 |
|
|
|
|
TAN No.: [Tax Deduction &
Collection Account No.] |
Not Available |
|
|
|
|
PAN No.: [Permanent Account No.] |
AAICA5591M |
|
|
|
|
GSTN : [Goods & Service Tax
Registration No.] |
24AAICA5591M1Z9 |
|
|
|
|
Legal Form : |
A Public Limited Liability Company. The Company’s Shares are Listed on
the Stock Exchanges. |
|
|
|
|
Line of Business
: |
Manufacturing and Selling of Pharmaceuticals Products i.e. Active Pharmaceutical Ingredients and Formulations. [Registered Activity] |
|
|
|
|
No. of Employees
: |
8825 (Approximately) |
RATING & COMMENTS
(Mira Inform has adopted New Rating mechanism w.e.f. 23rd
January 2017)
|
MIRA’s Rating : |
A+ |
|
Credit Rating |
Explanation |
Rating Comments |
|
A+ |
Low Risk |
Business dealings permissible with low
risk of default |
|
Maximum Credit Limit : |
USD 53000000 |
|
|
|
|
Status : |
Excellent |
|
|
|
|
Payment Behaviour : |
Regular |
|
|
|
|
Litigation : |
Clear |
|
|
|
|
Comments : |
Subject was incorporated in the year 2010 and is engaged in manufacturing of pharmaceuticals. Its products include active pharmaceuticals ingredients, bulk pharma chemicals, formulations, herbal nutraceuticals, and veterinary and collaborated research services. For the financial year 2017, there has been slight decline in its revenue by 0.77% but has managed to maintain profit margin of 14.42%. The robust financial profile of the company is marked by healthy networth base along with strong debt coverage indicators due to debt free balance sheet profile. Rating reflects the company's strong position in the domestic formulations market marked by increasing presence in the international generics market along with its established track record of business operations and promoters extensive industry experience. The company has its share price trading at around INR 533.65 against the Face Value (FV) of INR 02 on BSE as on 9th March, 2018. Business is active. Payments seems to be regular. In view of aforesaid, the company can be considered good for normal business dealings at usual trade terms and conditions. |
NOTES :
Any query related to this report can be made
on e-mail : infodept@mirainform.com
while quoting report number, name and date.
ECGC Country Risk Classification List
|
Country Name |
Previous Rating (30.09.2017) |
Current Rating (31.12.2017) |
|
India |
A1 |
A1 |
|
Risk Category |
ECGC
Classification |
|
Insignificant |
A1 |
|
Low Risk |
A2 |
|
Moderately Low Risk |
B1 |
|
Moderate Risk |
B2 |
|
Moderately High Risk |
C1 |
|
High Risk |
C2 |
|
Very High Risk |
D |
EXTERNAL AGENCY RATING
|
Rating Agency Name |
CRISIL |
|
Rating |
Long Term = AA |
|
Rating Explanation |
High degree of safety and very low credit
risk |
|
Date |
10.01.2018 |
|
Rating Agency Name |
CRISIL |
|
Rating |
Short Term = A1+ |
|
Rating Explanation |
Very strong degree of safety and carry
lowest credit risk |
|
Date |
10.01.2018 |
RBI DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available RBI Defaulters’ list.
EPF (Employee Provident Fund) DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available EPF (Employee Provident Fund) Defaulters’ list as of
31-03-2016.
BIFR (Board for Industrial & Financial
Reconstruction) LISTING STATUS
Subject’s name is
not listed as a Sick Unit in the publicly available BIFR (Board for Industrial
& Financial Reconstruction) list as of 10.03.2018
IBBI (Insolvency and Bankruptcy Board of India) LISTING STATUS
Subject’s name is not listed in the publicly
available IBBI (Insolvency and Bankruptcy Board of India) list as of report date.
INFORMATION DENIED BY
|
Name : |
Mr. Milind Nagode |
|
Designation : |
Not Divulged |
|
Contact No.: |
91-9833933029 |
|
Date : |
09.03.2018 |
MANAGEMENT
NON-COOPERATIVE (TEL No.: 91-265-2280550 / 3007000)
LOCATIONS
|
Registered / Corporate Office/ Factory 1 : |
Alembic Road, Gorwa, Vadodara - 390003, Gujarat, India |
|
Tel. No.: |
91-265-2280550/ 2280880/ 3007000/ 2282506 |
|
Mobile No.: |
91-9004085531 (Mr. Warade) 91-9177909974 (Mr. Praveen Gupta) 91-9833933029 (Mr. Milind Nagode) |
|
Fax No.: |
91-265 -2281508/ 2284728/ 2282506 |
|
E-Mail : |
|
|
Website : |
|
|
|
|
|
Corporate office : |
2nd Floor, Prime Corporate Park, Behind ITC Grand Maratha
Sheraton, Sahar Road, Andheri (East), Mumbai - 400099, Maharashtra, India |
|
Tel. No.: |
91-22-30611666 |
|
|
|
|
Factory 2 : |
Panelav, Taluka Halol, District Panchmahal-389350, Gujarat, India |
|
|
|
|
Factory 3 : |
Plot No. 21/22, EPIP, Phase-I, Jhamajri, Baddi, Tehsil – Nalagarh,
Solan – 173205, Himachal Pradesh, India |
|
|
|
|
Factory 4 : |
Village Karakhadi, Taluka Padra, District Vadodara, Gujarat, India |
|
|
|
|
Factory 5 : |
Samardung Basti, Karek Block, Namthang, South Sikkim, India |
|
|
|
|
Research Centre : |
Division of Alembic Limited, Alembic Campus, Vadodara – 390003, Gujarat, India |
|
Tel No.: |
91-265-3007989/ 3007874/ 3007652/ 3007300 |
|
|
|
|
Research and Development Centre : |
Lab Suites 111,112,114,123,124, Block-1, MN Park, Genome Valley, Shameerpet Mandal, Turkapally Village, Hyderabad - 500078, Telangana, India |
|
Tel. No.: |
91-40-30573036 |
DIRECTORS
As on 31.03.2017
|
Name : |
Mr. Chirayu Ramanbhai Amin |
|
Designation : |
Wholetime Director |
|
Address : |
F 10/195, Race Course Circle, Gorti Road, Vadodara - 390007, Gujarat, India |
|
Date of Birth/Age : |
04.12.1946 |
|
Qualification : |
M.B.A |
|
Experience : |
34 Years |
|
Date of Appointment : |
16.06.2010 |
|
PAN No.: |
ABMPA5729P |
|
DIN No.: |
00242549 |
|
|
|
|
Name : |
Mr. Pranav Chirayu Amin |
|
Designation : |
Managing Director |
|
Date of Birth/Age : |
42 Years |
|
Qualification : |
M.B.A |
|
Experience : |
17 Years |
|
Date of Appointment : |
31.03.2011 |
|
DIN No.: |
00245099 |
|
|
|
|
Name : |
Dr. Archana Niranjan Hingorani |
|
Address : |
10, Jeevan Dhara, Dr. Ambedkar Road, Bandra (West), Mumbai – 400050, Maharashtra, India |
|
Designation : |
Independent
Director |
|
Date of Birth/Age : |
70
Years |
|
Qualification : |
M.B.A |
|
Experience : |
34
Years |
|
Date of Appointment : |
04.02.2015 |
|
DIN No.: |
00028037 |
|
|
|
|
Name : |
Mr. Paresh Manilal Saraiya |
|
Address : |
18, Charotar Society, Old Padra Road, Vadodara - 390020, Gujarat, India |
|
Designation : |
Independent
Director |
|
Date of Appointment : |
31.03.2011 |
|
DIN No.: |
00063971 |
|
|
|
|
Name : |
Mr. Krishnapuram Ramanathan |
|
Address : |
Srinidhi Bunglow, No. 1, Octroi Naka, Gotri Road, Vadodara – 390021, Gujarat, India |
|
Designation : |
Director |
|
Date of Appointment : |
31.03.2016 |
|
DIN No.: |
00243928 |
|
|
|
|
Name : |
Mr. Shaunak Chirayu Amin |
|
Address : |
F10/195, Race Course Circle, Gotri Road, Vadodara - 390007, Gujarat, India |
|
Designation : |
Managing Director |
|
Date of Birth/Age : |
39 Years |
|
Qualification : |
B.A |
|
Experience : |
14 Years |
|
Date of Appointment : |
02.05.2013 |
|
DIN No.: |
00245523 |
|
|
|
|
Name : |
Mr. Pranav Natverlal Parikh |
|
Address : |
F10/195, Race Course Circle, Gotri Road, Vadodara – 390007, Gujarat, India |
|
Designation : |
Managing Director |
|
Date of Birth/Age : |
18.02.1943 |
|
Date of Appointment : |
31.03.2011 |
|
DIN No.: |
00318726 |
|
|
|
|
Name : |
Mr. Milin Kaimas Mehta |
|
Address : |
204 Meghdhanush Appartment, Racecourse Circle, Vadodara – 390007, Gujarat, India |
|
Designation : |
Independent
Director |
|
Date of Appointment : |
31.03.2011 |
|
DIN No.: |
01297508 |
|
|
|
|
Name : |
Mr. Rajkumar Shreeram Baheti |
|
Address : |
B-2-302, Orcadia, Nautilus Complex, Off Vasna Road, Vadodara – 390007, Gujarat, India |
|
Designation : |
Wholetime Director |
|
Date of Appointment : |
31.03.2011 |
|
DIN No.: |
00332079 |
KEY EXECUTIVES
|
Name : |
Mr. Rajkumar Shreeram Baheti |
|
Designation : |
Chief Finance Officer |
|
Address : |
B-2-302, Orcadia, Nautilus Complex, Off Vasna Road,
Vadodara – 390007, Gujarat, India |
|
Date of Appointment : |
28.07.2014 |
|
PAN No.: |
ABXPB2161K |
|
|
|
|
Name : |
Mr. Ajay Kumar Desai |
|
Designation : |
Company Secretary |
|
Address : |
C-504, Lakshachandi Heights, Gokuldham, Goragaon (East), Mumbai – 400063, Maharashtra, India |
|
Date of Appointment : |
28.07.2014 |
|
PAN No.: |
ABWPD3196M |
|
|
|
|
Name : |
Mr. Chirayu Ramanbhai Amin |
|
Designation : |
Chairman
and Chief Executive Officer |
|
Address : |
F 10/195, Race Course Circle, Gorti Road, Vadodara - 390007, Gujarat, India |
|
Date of Appointment : |
27.04.2016 |
|
PAN No.: |
ABMPA5729P |
|
|
|
|
Name : |
Mr. Praveen Gupta |
|
Designation : |
Assistant General Manager (Research and Development) |
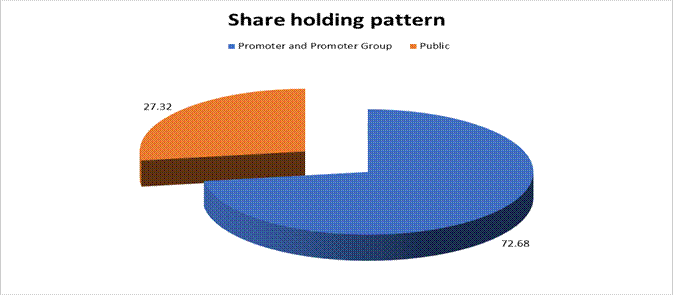
MAJOR SHAREHOLDERS / SHAREHOLDING PATTERN
As on 31.12.2017
|
Category of shareholder |
No. of fully paid up equity shares held |
Shareholding as a % of total no. of
shares |
|
|
Promoter and Promoter Group |
137007545 |
72.68 |
|
|
Public |
51508369 |
27.32 |
|
|
Grand Total |
188515914 |
100.00 |
STATEMENT SHOWING SHAREHOLDING PATTERN OF THE PROMOTER AND PROMOTER
GROUP
|
Category of shareholder |
No. of fully paid up equity shares held |
Shareholding as a % of total no. of
shares |
|
|
A1) Indian |
0.00 |
||
|
Individuals/Hindu undivided Family |
10670955 |
5.66 |
|
|
Chirayu Ramanbhai Amin |
4521465 |
2.40 |
|
|
Malika Chirayu Amin |
3005730 |
1.59 |
|
|
Pranav Chirayu Amin |
1009800 |
0.54 |
|
|
Shaunak Chirayu Amin |
1006980 |
0.53 |
|
|
Udit Chirayu Amin |
1006980 |
0.53 |
|
|
Inaaya Shaunak Amin |
30000 |
0.02 |
|
|
Naintara Shaunak Amin |
30000 |
0.02 |
|
|
Ranvir Pranav Amin |
30000 |
0.02 |
|
|
Samira Pranav Amin |
30000 |
0.02 |
|
|
Any Other (specify) |
126336590 |
67.02 |
|
|
Alembic Limited |
55000000 |
29.18 |
|
|
Shreno Ltd |
20698780 |
10.98 |
|
|
Whitefield Chemtech Private Limited |
18285230 |
9.70 |
|
|
Sierra Investments Private Limited. |
17337670 |
9.20 |
|
|
Nirayu Pvt Limited |
13713755 |
7.27 |
|
|
Paushak Limited |
855 |
0.00 |
|
|
Vidyanidhi Trust |
809550 |
0.43 |
|
|
Arogyavardhini Society |
280950 |
0.15 |
|
|
Utkarsh Vidyakendra |
146250 |
0.08 |
|
|
Ujjwal Vidyalaya |
62250 |
0.03 |
|
|
Laburnum Family Trust |
200 |
0.00 |
|
|
Virsad Family Trust |
200 |
0.00 |
|
|
Viramya Packlight LLP |
900 |
0.00 |
|
|
Sub Total A1 |
137007545 |
72.68 |
|
|
A2) Foreign |
0.00 |
||
|
A=A1+A2 |
137007545 |
72.68 |
STATEMENT SHOWING SHAREHOLDING PATTERN OF THE PUBLIC SHAREHOLDER
|
Category of shareholder |
No. of fully paid up equity shares held |
Shareholding as a % of total no. of
shares |
|
|
B1) Institutions |
0 |
0.00 |
|
|
Mutual Funds/ |
5507452 |
2.92 |
|
|
Foreign Portfolio Investors |
20290665 |
10.76 |
|
|
Elara India Opportunities Fund Limited |
2555079 |
1.36 |
|
|
Matthews India Fund |
2640862 |
1.40 |
|
|
Financial Institutions/ Banks |
599065 |
0.32 |
|
|
Sub Total B1 |
26397182 |
14.00 |
|
|
B2) Central Government/ State Government(s)/ President of India |
0 |
0.00 |
|
|
B3) Non-Institutions |
0 |
0.00 |
|
|
Individual share capital upto INR 0.200 Million |
16383439 |
8.69 |
|
|
Individual share capital in excess of INR 0.200 Million |
4558180 |
2.42 |
|
|
Any Other (specify) |
4169568 |
2.21 |
|
|
Trusts |
11292 |
0.01 |
|
|
HUF |
482814 |
0.26 |
|
|
NRI – Non- Repat |
391839 |
0.21 |
|
|
NRI – Repat |
222076 |
0.12 |
|
|
Foreign Individuals |
100 |
0.00 |
|
|
Unclaimed or Suspense or Escrow Account |
782208 |
0.41 |
|
|
Clearing Members |
113417 |
0.06 |
|
|
Bodies Corporate |
2165822 |
1.15 |
|
|
Sub Total B3 |
25111187 |
13.32 |
|
|
B=B1+B2+B3 |
51508369 |
27.32 |
BUSINESS DETAILS
|
Line of Business : |
Manufacturing and Selling of Pharmaceuticals Products i.e. Active Pharmaceutical Ingredients and Formulations. [Registered Activity] |
||||
|
|
|
||||
|
Products : |
|
||||
|
|
|
||||
|
Brand Names : |
Not Divulged |
||||
|
|
|
||||
|
Agencies Held : |
Not Divulged |
||||
|
|
|
||||
|
Exports : |
Not Divulged |
||||
|
|
|
||||
|
Imports : |
Not Divulged |
||||
|
|
|
||||
|
Terms : |
Not Divulged |
PRODUCTION STATUS NOT AVAILABLE
GENERAL INFORMATION
|
Suppliers : |
|
||||||||||||||||||||||
|
|
|
||||||||||||||||||||||
|
Customers : |
|
||||||||||||||||||||||
|
|
|
||||||||||||||||||||||
|
No. of Employees : |
8825 (Approximately) |
||||||||||||||||||||||
|
|
|
||||||||||||||||||||||
|
Bankers : |
|
|
Statutory Auditors
: |
|
|
Name : |
K. S. Aiyar and Company Chartered Accountants |
|
Address : |
# F-7 Laxmi Mills, Shakti Mills Lane (Off Dr. E Moses Road),
Mahalaxmi, Mumbai - 400011, Maharashtra, India |
|
Tel. No.: |
91-22-24932502/ 66551770 |
|
Mobile No.: |
91-22-66551774 |
|
E-Mail : |
|
|
Website : |
|
|
|
|
|
Secretarial
Auditors: |
|
|
Name : |
Samdani Shah and Kabra Chartered Accountants |
|
|
|
|
Secretarial
Auditors: |
|
|
Name : |
H. R. Kapadia Chartered Accountants |
|
|
|
|
Internal Auditors: |
|
|
Name : |
Ernst and Young LLP Chartered Accountants |
|
|
|
|
Memberships : |
Not Available |
|
|
|
|
Collaborators : |
Not Available |
|
|
|
|
Subsidiary and Fellow Subsidiary: |
·
Alembic Global Holding SA. (Subsidiary of Alembic Pharmaceuticals
Limited) ·
Alembic Pharmaceuticals Australia Pty Limited (Subsidiary of Alembic Global Holding SA.) ·
Alembic Pharmaceuticals Europe Limited (Subsidiary of Alembic Global Holding SA.) ·
Alnova Pharmaceuticals SA (Subsidiary of Alembic Global Holding SA.) ·
Alembic Pharmaceuticals Inc. (Subsidiary of Alembic Global Holding SA.) ·
Alembic Pharmaceuticals Canada Limited
(Subsidiary of Alembic Global Holding SA.) ·
AG Research Private Limited, (Subsidiary of Alembic Pharmaceuticals
Limited.) U85120TG2014PTC093661 ·
Genius LLC, (Subsidiary of Alembic Global Holding SA.) · Aleor Dermaceuticals Limited (Subsidiary of Alembic Pharmaceuticals Limited) U24302GJ2016PLC092131 |
|
|
|
|
Associate Companies: |
·
Alembic Limited ·
Whitefield Chemtech Private Limited ·
Nirayu Private Limited ·
Quick Flight Limited ·
Shreno Limited ·
Paushak Limited ·
Sierra Investments Limited ·
Viramya Packlight Limited ·
Incozen Therapeutics Private Limited U24239TG2008PTC059967 ·
Rhizen Pharmaceuticals SA (Associate of Alembic Global Holding SA) ·
Dahlia Therapeutics (Subsidiary of
Rhizen Pharmaceuticals SA) ·
Rhizen Pharmaceuticals Inc (Subsidiary of Rhizen Pharmaceuticals SA.) |
|
|
|
|
Joint Venture |
·
Alembic Mami SPA (Joint venture of Alembic Global
Holding SA) |
CAPITAL STRUCTURE
As on 31.03.2017
Authorised Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
200000000 |
Equity Shares |
INR 2/- each |
INR 400.000 Million |
|
|
|
|
|
Issued, Subscribed & Paid-up Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
188515914 |
Equity Shares |
INR 2/- each |
INR 377.032 Million |
|
|
|
|
|
Reconciliation of the
number of shares outstanding at the beginning and at the end of the reporting
period
|
As at |
31st March, 2017 |
|
|
|
Numbers |
INR in Million |
|
At the beginning of the year |
188515914 |
377.032 |
|
Outstanding at the end of the year |
188515914 |
377.032 |
The rights, preferences and restrictions including restrictions on the distribution of dividends and the repayment of capital
The company is having only one class of shares i.e Equity carrying a nominal value of INR 2/- per share
Every holder of the equity share of the Company is entitled to one vote per share held.
In the event of liquidation of the Company, the equity shareholders will be entitled to receive remaining assets of the Company after the distribution / repayment of all creditors. The distribution to the equity shareholders will be in proportion of the number of shares held by each shareholder.
The Company declares and pays dividend on the equity shares in Indian Rupees. Dividend proposed by the Board of Directors is subject to approval of the shareholders at the ensuing Annual General Meeting.
Shares in the company
held by each shareholder holding more than 5 percent shares specifying the
number of shares held
|
As at |
31st March, 2017 |
|
|
|
Numbers |
% held |
|
Alembic Limited |
55000000 |
29.18 |
|
Shreno Limited |
20698780 |
10.98 |
|
Whitefield Chemtech Private Limited |
18285230 |
9.70 |
|
Sierra Investments Private Limited |
17337670 |
9.20 |
|
Nirayu Private Limited |
13713755 |
7.27 |
FINANCIAL DATA
[all figures are
in INR Million]
ABRIDGED
BALANCE SHEET (STANDALONE)
|
SOURCES OF FUNDS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
|
|
|
|
|
I.
EQUITY AND LIABILITIES |
|
|
|
|
(1)Shareholders' Funds |
|
|
|
|
(a) Share Capital |
377.000 |
377.000 |
377.032 |
|
(b) Reserves &
Surplus |
18296.200 |
14930.300 |
7958.620 |
|
(c) Money received
against share warrants |
0.000 |
0.000 |
0.000 |
|
|
|
|
|
|
(2) Share Application money
pending allotment |
0.000 |
0.000 |
0.000 |
|
Total Shareholders’ Funds
(1) + (2) |
18673.200 |
15307.300 |
8335.652 |
|
|
|
|
|
|
(3) Non-Current
Liabilities |
|
|
|
|
(a) long-term borrowings |
0.000 |
0.000 |
188.000 |
|
(b) Deferred tax
liabilities (Net) |
437.800 |
426.300 |
314.334 |
|
(c) Other long term
liabilities |
125.700 |
130.500 |
128.437 |
|
(d) long-term provisions |
572.200 |
282.300 |
93.615 |
|
Total Non-current
Liabilities (3) |
1135.700 |
839.100 |
724.386 |
|
|
|
|
|
|
(4) Current Liabilities |
|
|
|
|
(a) Short term borrowings |
0.000 |
0.000 |
1498.079 |
|
(b) Trade payables |
4658.200 |
4997.600 |
3164.520 |
|
(c) Other current
liabilities |
487.700 |
605.800 |
442.927 |
|
(d) Short-term provisions |
122.900 |
67.400 |
946.009 |
|
Total Current Liabilities
(4) |
5268.800 |
5670.800 |
6051.535 |
|
|
|
|
|
|
TOTAL |
25077.700 |
21817.200 |
15111.573 |
|
|
|
|
|
|
II.
ASSETS |
|
|
|
|
(1) Non-current assets |
|
|
|
|
(a) Fixed Assets |
|
|
|
|
(i) Tangible assets |
7990.100 |
6972.500 |
4886.799 |
|
(ii) Intangible Assets |
0.000 |
0.000 |
0.000 |
|
(iii) Capital
work-in-progress |
3477.500 |
923.400 |
831.403 |
|
(iv) Intangible assets
under development |
0.000 |
0.000 |
0.000 |
|
(b) Non-current
Investments |
1030.400 |
374.600 |
333.285 |
|
(c) Deferred tax assets
(net) |
0.000 |
0.000 |
0.000 |
|
(d) Long-term Loan and Advances |
0.000 |
0.000 |
650.273 |
|
(e) Other Non-current
assets |
365.200 |
299.600 |
0.000 |
|
Total Non-Current Assets |
12863.200 |
8570.100 |
6701.760 |
|
|
|
|
|
|
(2) Current assets |
|
|
|
|
(a) Current investments |
0.000 |
0.000 |
0.000 |
|
(b) Inventories |
5746.200 |
5337.200 |
3824.829 |
|
(c) Trade receivables |
4660.600 |
3715.000 |
3379.400 |
|
(d) Cash and cash
equivalents |
72.100 |
2848.100 |
44.050 |
|
(e) Short-term loans and
advances |
0.000 |
0.000 |
0.000 |
|
(f) Other current assets |
1735.600 |
1346.800 |
1161.534 |
|
Total Current Assets |
12214.500 |
13247.100 |
8409.813 |
|
|
|
|
|
|
TOTAL |
25077.700 |
21817.200 |
15111.573 |
PROFIT
& LOSS ACCOUNT (STANDALONE)
|
|
PARTICULARS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
|
SALES |
|
|
|
|
|
Income |
29859.000 |
30090.200 |
20189.960 |
|
|
Other Income |
24.200 |
68.200 |
5.723 |
|
|
TOTAL |
29883.200 |
30158.400 |
20195.683 |
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
Cost of Materials
Consumed |
7038.700 |
6525.200 |
5404.940 |
|
|
Purchases of
Stock-in-Trade |
2303.100 |
2551.300 |
2466.194 |
|
|
Changes in inventories of
finished goods, work-in-progress and Stock-in-Trade |
(479.000) |
(959.400) |
(728.890) |
|
|
Employees benefits
expense |
5274.600 |
4533.200 |
2965.671 |
|
|
Research and Development
Expenses |
0.000 |
0.000 |
1090.308 |
|
|
Other expenses |
9388.800 |
7612.500 |
4913.596 |
|
|
TOTAL |
23526.200 |
20262.800 |
16111.819 |
|
|
|
|
|
|
|
|
PROFIT/ (LOSS) BEFORE INTEREST, TAX, DEPRECIATION AND
AMORTISATION |
6357.000 |
9895.600 |
4083.864 |
|
|
|
|
|
|
|
Less |
FINANCIAL EXPENSES |
30.900 |
36.800 |
11.088 |
|
|
|
|
|
|
|
|
PROFIT / (LOSS) BEFORE
TAX, DEPRECIATION AND AMORTISATION |
6326.100 |
9858.800 |
4072.776 |
|
|
|
|
|
|
|
Less/ Add |
DEPRECIATION/
AMORTISATION |
829.000 |
718.600 |
444.127 |
|
|
|
|
|
|
|
|
PROFIT/ (LOSS) BEFORE TAX |
5497.100 |
9140.200 |
3628.649 |
|
|
|
|
|
|
|
Less |
TAX |
1190.800 |
2152.800 |
762.500 |
|
|
|
|
|
|
|
|
PROFIT/ (LOSS) AFTER TAX
|
4306.300 |
6987.400 |
2866.149 |
|
|
|
|
|
|
|
Less |
APPROPRIATIONS |
|
|
|
|
|
Transfer to General
Reserve |
3000.000 |
3000.000 |
1000.000 |
|
|
Dividend paid on Equity
Shares during the year |
754.100 |
659.900 |
659.800 |
|
|
Corporate Dividend tax
paid during the year |
153.500 |
134.300 |
134.300 |
|
|
Total (M) |
3907.600 |
3794.200 |
1794.100 |
|
|
|
|
|
|
|
|
EARNINGS IN FOREIGN
CURRENCY |
|
|
|
|
|
F.O.B. Value of Exports |
15625.800 |
17550.800 |
8084.547 |
|
|
Royalty |
179.700 |
259.700 |
199.739 |
|
|
TOTAL EARNINGS |
15805.500 |
17810.500 |
8284.286 |
|
|
|
|
|
|
|
|
IMPORTS |
|
|
|
|
|
Raw Materials |
3178.400 |
3283.000 |
2049.724 |
|
|
Packing Material,
Components and Spare Parts |
28.200 |
48.100 |
376.150 |
|
|
Stores and Spare Parts |
34.300 |
23.500 |
27.345 |
|
|
Professional and
Consultancy fees |
508.300 |
423.500 |
136.492 |
|
|
Interest |
7.500 |
10.900 |
12.389 |
|
|
Others |
1531.900 |
1529.100 |
883.641 |
|
|
TOTAL IMPORTS |
5288.600 |
5318.100 |
3485.741 |
|
|
|
|
|
|
|
|
Earnings / (Loss) Per
Share (INR) |
22.84 |
37.07 |
15.2 |
CURRENT MATURITIES OF LONG TERM DEBT DETAILS
|
Particulars |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Current Maturities of Long term debt |
-- |
-- |
-- |
|
Cash generated from operations |
4385.800 |
10436.000 |
2491.800 |
|
Net cash flow from operating activity |
3206.100 |
8475.200 |
1800.800 |
QUARTERLY RESULTS
|
Particulars |
30.06.2017 |
30.09.2017 |
30.12.2017 |
|
Audited / Unaudited |
Unaudited |
Unaudited |
Unaudited |
|
|
1ST Quarter |
2nd Quarter |
3rd Quarter |
|
Net Sales |
5901.10 |
7620.100 |
7549.100 |
|
Total Expenditure |
5032.70 |
5853.500 |
5944.400 |
|
PBIDT (Excl OI) |
868.40 |
1766.600 |
1604.700 |
|
Other Income |
2.30 |
76.100 |
3.300 |
|
Operating Profit |
870.70 |
1842.700 |
1607.900 |
|
Interest |
8.30 |
1.200 |
6.400 |
|
Exceptional Items |
NA |
NA |
NA |
|
PBDT |
862.40 |
1841.600 |
1601.500 |
|
Depreciation |
218.20 |
256.300 |
263.800 |
|
Profit Before Tax |
644.20 |
1585.300 |
1337.700 |
|
Tax |
137.50 |
364.300 |
287.300 |
|
Provisions and contingencies |
NA |
NA |
NA |
|
Profit After Tax |
506.70 |
1221.000 |
1050.400 |
|
Extraordinary Items |
NA |
NA |
NA |
|
Prior Period Expenses |
NA |
NA |
NA |
|
Other Adjustments |
NA |
NA |
NA |
|
Net Profit |
506.70 |
1221.000 |
1050.400 |
KEY
RATIOS
EFFICIENCY RATIOS
|
PARTICULARS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Average Collection Days (Sundry Debtors / Income * 365 Days) |
56.97 |
45.06 |
61.09 |
|
|
|
|
|
|
Account Receivables Turnover (Income / Sundry
Debtors) |
6.41 |
8.10 |
5.97 |
|
|
|
|
|
|
Average Payment Days (Sundry Creditors
/ Purchases * 365 Days) |
182.00 |
200.97 |
146.75 |
|
|
|
|
|
|
Inventory Turnover (Operating Income
/ Inventories) |
1.11 |
1.85 |
1.07 |
|
|
|
|
|
|
Asset Turnover (Operating Income
/ Net Fixed Assets) |
0.55 |
1.25 |
0.71 |
LEVERAGE RATIOS
|
PARTICULARS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Debt Ratio ((Borrowing + Current Liabilities) / Total
Assets) |
0.21 |
0.26 |
0.41 |
|
|
|
|
|
|
Debt Equity Ratio (Total Liability
/ Networth) |
0.00 |
0.00 |
0.20 |
|
|
|
|
|
|
Current Liabilities to Networth (Current Liabilities
/ Net Worth) |
0.28 |
0.37 |
0.73 |
|
|
|
|
|
|
Fixed Assets to Networth (Net Fixed Assets
/ Networth) |
0.61 |
0.52 |
0.69 |
|
|
|
|
|
|
Interest Coverage Ratio (PBIT / Financial
Charges) |
205.73 |
268.90 |
368.31 |
PROFITABILITY RATIOS
|
PARTICULARS |
|
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
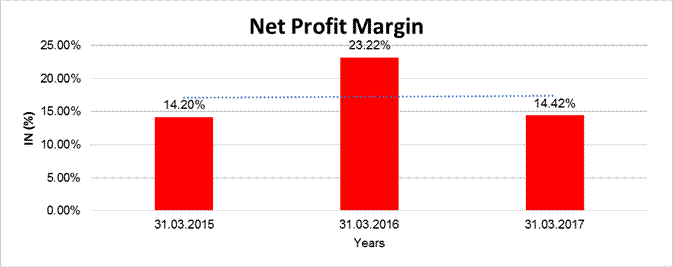
Net Profit Margin [(PAT / Sales) * 100] |
% |
14.42 |
23.22 |
14.20 |
|
|
|
|
|
|
|
Return on Total Assets ((PAT / Total
Assets) * 100) |
% |
17.17 |
32.03 |
18.97 |
|
|
|
|
|
|
|
Return on Investment (ROI) ((PAT / Networth)
* 100) |
% |
23.06 |
45.65 |
34.38 |
SOLVENCY RATIOS
|
PARTICULARS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Current Ratio (Current Assets / Current Liabilities) |
2.32 |
2.34 |
1.39 |
|
|
|
|
|
|
Quick Ratio ((Current Assets
– Inventories) / Current Liabilities) |
1.23 |
1.39 |
0.76 |
|
|
|
|
|
|
G-Score Ratio Financial (Networth / Total
Assets) |
0.74 |
0.70 |
0.55 |
|
|
|
|
|
|
G-Score Ratio Debt (Debts / Equity
Capital) |
0.00 |
0.00 |
4.47 |
|
|
|
|
|
|
G-Score Ratio Liquidity (Total Current
Assets / Total Current Liabilities) |
2.32 |
2.34 |
1.39 |
Total Liability = Short-term Debt + Long-term
Debt + Current Maturities of Long-term debts
STOCK
PRICES
|
Face Value |
INR 2.00/- each |
|
Market Value |
INR 553.60/- each |
FINANCIAL ANALYSIS
[all figures are
INR Million]
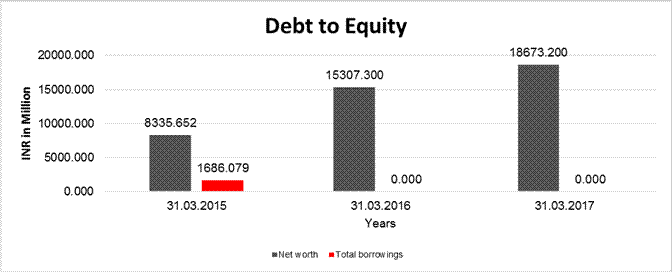
DEBT EQUITY RATIO
|
Particular |
31.03.2015 |
31.03.2016 |
31.03.2017 |
|
|
INR
In Million |
INR
In Million |
INR
In Million |
|
Share Capital |
377.032 |
377.000 |
377.000 |
|
Reserves & Surplus |
7958.620 |
14930.300 |
18296.200 |
|
Net
worth |
8335.652 |
15307.300 |
18673.200 |
|
|
|
|
|
|
Long Term borrowings |
188.000 |
0.000 |
0.000 |
|
Short Term borrowings |
1498.079 |
0.000 |
0.000 |
|
Total
borrowings |
1686.079 |
0.000 |
0.000 |
|
Debt/Equity
ratio |
0.202 |
0.000 |
0.000 |
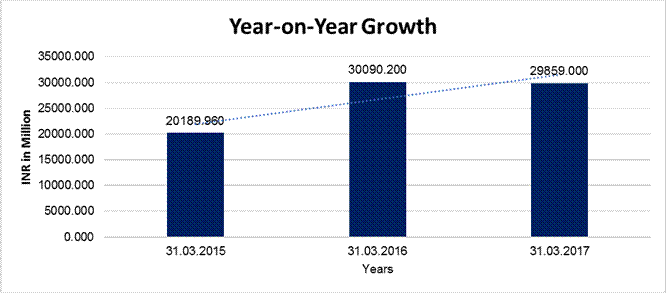
YEAR-ON-YEAR GROWTH
|
Year
on Year Growth |
31.03.2015 |
31.03.2016 |
31.03.2017 |
|
|
INR
In Million |
INR
In Million |
INR
In Million |
|
Sales
|
20189.960 |
30090.200 |
29859.000 |
|
|
|
49.035 |
(0.768) |
NET PROFIT MARGIN
|
Net
Profit Margin |
31.03.2015 |
31.03.2016 |
31.03.2017 |
|
|
INR
In Million |
INR
In Million |
INR
In Million |
|
Sales
|
20189.960 |
30090.200 |
29859.000 |
|
Profit |
2866.149 |
6987.400 |
4306.300 |
|
|
14.20% |
23.22% |
14.42% |
ABRIDGED
BALANCE SHEET – (CONSOLIDATED)
|
SOURCES OF FUNDS |
|
31.03.2017 |
31.03.2016 |
|
|
|
|
|
|
I.
EQUITY AND LIABILITIES |
|
|
|
|
(1)Shareholders' Funds |
|
|
|
|
(a) Share Capital |
|
377.000 |
377.000 |
|
(b) Reserves &
Surplus |
|
18634.000 |
15596.600 |
|
(c) Money received
against share warrants |
|
0.000 |
0.000 |
|
|
|
|
|
|
(2) Share Application
money pending allotment |
|
0.000 |
0.000 |
|
(3) Non-Controlling
Interest |
|
1.000 |
0.000 |
|
Total Shareholders’ Funds
(1) + (2) |
|
19012.000 |
15973.600 |
|
|
|
|
|
|
(3) Non-Current
Liabilities |
|
|
|
|
(a) long-term borrowings |
|
0.000 |
0.000 |
|
(b) Deferred tax
liabilities (Net) |
|
369.300 |
427.400 |
|
(c) Other long term
liabilities |
|
125.700 |
130.500 |
|
(d) long-term provisions |
|
572.200 |
282.300 |
|
Total Non-current
Liabilities (3) |
|
1067.200 |
840.200 |
|
|
|
|
|
|
(4) Current Liabilities |
|
|
|
|
(a) Short term borrowings |
|
802.400 |
1137.100 |
|
(b) Trade payables |
|
5232.200 |
5656.500 |
|
(c) Other current
liabilities |
|
629.400 |
610.600 |
|
(d) Short-term provisions |
|
123.600 |
67.700 |
|
Total Current Liabilities
(4) |
|
6787.600 |
7471.900 |
|
|
|
|
|
|
TOTAL |
|
26866.800 |
24285.700 |
|
|
|
|
|
|
II.
ASSETS |
|
|
|
|
(1) Non-current assets |
|
|
|
|
(a) Fixed Assets |
|
|
|
|
(i) Tangible assets |
|
7994.000 |
7080.900 |
|
(ii) Intangible Assets |
|
0.000 |
0.000 |
|
(iii) Capital
work-in-progress |
|
3692.800 |
925.200 |
|
(iv) Intangible assets
under development |
|
270.000 |
0.000 |
|
(b) Non-current
Investments |
|
501.600 |
534.400 |
|
(c) Deferred tax assets
(net) |
|
0.000 |
0.000 |
|
(d) Long-term Loan and Advances |
|
0.000 |
0.000 |
|
(e) Other Non-current
assets |
|
1197.800 |
632.600 |
|
Total Non-Current Assets |
|
13656.200 |
9173.100 |
|
|
|
|
|
|
(2) Current assets |
|
|
|
|
(a) Current investments |
|
0.000 |
0.000 |
|
(b) Inventories |
|
6327.500 |
5697.900 |
|
(c) Trade receivables |
|
3374.700 |
3504.800 |
|
(d) Cash and cash
equivalents |
|
1596.000 |
4508.300 |
|
(e) Short-term loans and
advances |
|
0.000 |
0.000 |
|
(f) Other current assets |
|
1912.400 |
1401.600 |
|
Total Current Assets |
|
13210.600 |
15112.600 |
|
|
|
|
|
|
TOTAL |
|
26866.800 |
24285.700 |
PROFIT
& LOSS ACCOUNT– (CONSOLIDATED)
|
|
PARTICULARS |
|
31.03.2017 |
31.03.2016 |
|
|
SALES |
|
|
|
|
|
Income |
|
31346.100 |
31659.900 |
|
|
Other Income |
|
24.700 |
72.700 |
|
|
TOTAL |
|
31370.800 |
31732.600 |
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
Cost of Materials
Consumed |
|
7038.700 |
6539.400 |
|
|
Purchases of
Stock-in-Trade |
|
2021.300 |
2231.300 |
|
|
Changes in inventories of
finished goods, work-in-progress and Stock-in-Trade |
|
(479.000) |
(959.400) |
|
|
Employees benefits
expense |
|
5588.300 |
4728.500 |
|
|
Other expenses |
|
11029.600 |
9053.200 |
|
|
TOTAL |
|
25198.900 |
21593.000 |
|
|
|
|
|
|
|
|
PROFIT/ (LOSS) BEFORE INTEREST, TAX, DEPRECIATION AND
AMORTISATION |
|
6171.900 |
10139.600 |
|
|
|
|
|
|
|
Less |
FINANCIAL EXPENSES |
|
51.200 |
54.300 |
|
|
|
|
|
|
|
|
PROFIT / (LOSS) BEFORE
TAX, DEPRECIATION AND AMORTISATION |
|
6120.700 |
10085.300 |
|
|
|
|
|
|
|
Less/ Add |
DEPRECIATION/
AMORTISATION |
|
829.700 |
722.100 |
|
|
|
|
|
|
|
|
Share of profit/(loss) of
an associate and a joint venture |
|
38.300 |
1.600 |
|
|
|
|
|
|
|
|
Share of Non-controlling
Interest |
|
0.900 |
0.000 |
|
|
|
|
|
|
|
|
PROFIT/ (LOSS) BEFORE TAX |
|
5253.600 |
9361.600 |
|
|
|
|
|
|
|
Less |
TAX |
|
1221.900 |
2161.300 |
|
|
|
|
|
|
|
|
PROFIT/ (LOSS) AFTER TAX
|
|
4031.700 |
7200.300 |
|
|
|
|
|
|
|
|
Earnings / (Loss) Per
Share (INR) |
|
21.39 |
38.19 |
LOCAL AGENCY FURTHER INFORMATION
|
Sr. No. |
Check list by
info agents |
Available in
Report (Yes/No) |
|
1 |
Year of establishment |
Yes |
|
2 |
Constitution of the entity -Incorporation
details |
Yes |
|
3 |
Locality of the entity |
Yes |
|
4 |
Premises details |
No |
|
5 |
Buyer visit details |
-- |
|
6 |
Contact numbers |
Yes |
|
7 |
Name of the person contacted |
Yes |
|
8 |
Designation of contact person |
No |
|
9 |
Promoter’s background |
Yes |
|
10 |
Date of Birth of Proprietor / Partners /
Directors |
Yes |
|
11 |
Pan Card No. of Proprietor / Partners |
Yes |
|
12 |
Voter Id Card No. of Proprietor / Partners |
No |
|
13 |
Type of business |
Yes |
|
14 |
Line of Business |
Yes |
|
15 |
Export/import details (if applicable) |
No |
|
16 |
No. of employees |
Yes |
|
17 |
Details of sister concerns |
Yes |
|
18 |
Major suppliers |
No |
|
19 |
Major customers |
No |
|
20 |
Banking Details |
Yes |
|
21 |
Banking facility details |
Yes |
|
22 |
Conduct of the banking account |
-- |
|
23 |
Financials, if provided |
Yes |
|
24 |
Capital in the business |
Yes |
|
25 |
Last accounts filed at ROC, if applicable |
Yes |
|
26 |
Turnover of firm for last three years |
Yes |
|
27 |
Reasons for variation <> 20% |
-- |
|
28 |
Estimation for coming financial year |
No |
|
29 |
Profitability for last three years |
Yes |
|
30 |
Major shareholders, if available |
Yes |
|
31 |
External Agency Rating, if available |
Yes |
|
32 |
Litigations that the firm/promoter
involved in |
-- |
|
33 |
Market information |
-- |
|
34 |
Payments terms |
Yes |
|
35 |
Negative Reporting by Auditors in the
Annual Report |
No |
DESCRIPTION OF
BUSINESS
Subject is principally engaged in the manufacturing and selling of Pharmaceuticals products i.e. Active Pharmaceutical Ingredients and Formulations. The Company is the public limited Company domiciled in India and is incorporated under the provision of the Companies Act applicable in India. Its shares are listed on the two recognised Stock Exchanges in India. The registered office of the Company is located at Alembic Road, Vadodara – 390 003, India.
The Financial Statements are approved by the Company’s Board of Directors on 3rd May, 2017.
OPERATIONAL OVERVIEW
GLOBAL ECONOMIC
OVERVIEW
Better performance from emerging countries, subsiding geopolitical uncertainties, rising oil prices and a resurgent Chinese economy helped the global economy clock a growth rate of ~3% in 2016.
Global economic
outlook:
Economic activity in advanced economies and EMDEs is forecast to accelerate in 2017 and 2018, with global growth projected at 3.5% and 3.6%, respectively. There is an optimism related to the prospects in the US, assuming a fiscal stimulus that could lead to a growth of 2.3% in 2017 and 2.5% in 2018.
Indian economic overview
With a GDP at US$ 2 trillion, India’s economy ranks as the seventh largest in the world.
Outlook: Economy experts and opinion makers are optimistic of India’s economic resurgence and expect the nation’s GDP growth to rebound in the range of 6.75-7.5% in 2017-18 catalysed by two important triggers – the GST rollout and thrust on infrastructure creation through a large allocation in Union Budget 2017-18. The IMF forecasts India’s GDP growth at 7.2% for 2017-18 and 7.7% for the fiscal thereafter. According to this institution, medium-term growth prospects appear favourable, with growth to rise to about 8% over the medium term following the implementation of key reforms, loosening of supplyside bottlenecks, and appropriate fiscal and monetary policies.
Indian pharma players
in the US
In a nearly US$400-billion US pharmaceutical market, generic drugs make up for just about US$70-80billion, though generic drugs account for 88% of prescriptions dispensed in the US. India supplies one-third of the generic medications consumed in the US. The Indian pharma industry has contributed significantly to moderate healthcare costs in the US by supplying quality, yet affordable, generic drugs. India-made generics sometimes cost just a tenth of branded drugs sold in the US; between 2005 and 2014, generic drugs saved the US healthcare system US$1.7 trillion. Indian generic drug companies are facing significant pricing pressure in the US because of consolidation of distribution channels and rising competition.
Global overview
The growth of the life sciences sector of a country correlates with its general economic strength and healthcare spending levels; both of these vary widely around the globe. While spending growth is expected to pick up, the pressure to reduce costs, increase efficiency, and demonstrate superior value remains intense. Because of these contradictory trends, global healthcare spending is expected to increase in low single digits. Demand for generic drugs should continue to rise as payers pursue avenues to reduce costs. In the United States, generic drugs already comprise about 70% of the pharma market by volume. Pricing pressures in the United States and unstable economic conditions in Brazil, Russia, and China, which collectively drive 50% of global pharma revenue, have led to a slowdown in the pharma segment. As a result, pharmaceutical companies are adapting to market dynamics and positioning themselves for growth through portfolio transformation, targeted deal-making, cost-cutting measures, and sharpened focus on high-performing therapeutic area and geographic markets.
Outlook through 2021: The total volume of medicines consumed globally could increase by about 3% annually through 2021. Global medicine spending could reach nearly US$1.5 trillion by 2021 on an invoice price basis, up nearly US$370 billion from the 2016 estimated spending level. Importantly spending growth is slowing in 2016, declining from nearly 9% growth in 2014 and 2015 to just 4–7% CAGR over the next five years. Most global spending growth, particularly in developed markets, will be driven by oncology, autoimmune and diabetes treatments where significant innovations are expected.
Domestic overview
The Indian pharmaceutical market accounts for ~2.4% of the global pharmaceutical industry in value terms and 10% in volume terms. India is the third-largest global generic API market, with a 7.2% market share and the largest exporter of formulations in terms of volume, with 14% market share and 12th in terms of export value. Despite a strong global position, the domestic market is significantly underpenetrated. India’s healthcare expenditure is among the lowest among the top 15 pharma markets with the per capita spend being pegged at US$60-64 per annum (between 2010 and 2014) as per World Bank. In India, public spending on health stood at 1.1% of GDP much lower than the global benchmark. These realities showcase a significant opportunity for Indian pharmaceutical players.
GROWTH DRIVERS
Demand drivers
· Increasing incidence of fatal diseases
· Improved accessibility to drugs
· Increasing penetration of health insurance
· Growing number of stress-related diseases
· Better diagnostic facilities
Supply drivers
- Cost advantage
- Skilled manpower
Policy support
National Health Policy 2015, focuses on increasing public healthcare expenditure
- Reduction in approval time for new facilities
- Plans to set up pharmaceutical education and research institutes
- Exemptions to drugs manufactured through indigenous R&D from price control under NPPP-2012
BUSINESS DIVISIONS
Revenue vertical 1
International
generics
This is the flagship vertical for the Company and contributes ~40% to the Company’s revenues. Revenues declined by 15.46% – from H1,462 crore in 2015-16 to H1,236 crore in 2016-17. Under this business segment, the Company markets generics primarily in regulated geographies in the US, Europe, Canada, South Africa and Australia. Even as the US market catalyses the growth of the international generics space, other geographies will continue to register healthy annual revenue growth.
The US front-end
The Company has established a front-end marketing office managed by a nine-member team. Having secured all required licenses from the regulatory authority, the Company created a basket of 29 products (product launches and transfers from partners to own label sales). In the first full year of operations, this outfit generated more than US$50 million in revenues.
ROW
The Company continued to launch and file new products in Europe, Canada and Australia – in line with the Company’s flanking growth strategy.
The Algerian presence
In 2014, Alembic entered into a joint manufacturing venture with Adwiya Mami SARL, one of the largest pharmaceutical distributors in Algeria which possesses an oral drug formulation factory. This inorganic initiative enabled the Company to establish a footprint in the US$3.4 billion Algerian pharmaceutical market (of which generics account for 70%). In 2016-17, the Company received approvals for 11 filings and launched these products towards the close of the year. The Company filed for 13 products during the year under review, taking its pipeline to 16 filings (March 31, 2017) at various stages of receiving the required approvals.
Competitive advantages
Front-end presence:
Alembic established a visible presence in the world’s largest and highly- competitive pharmaceutical market, thereby gaining respect in the global pharmaceutical space.
Wide footprint:
Other than its US operations, the Company enjoys a meaningful presence in other regulated markets like Europe, Australia and Canada.
Compliant infrastructure: The Company cleared USFDA audits for its API, formulations and R&D units, thereby showcasing the institutionalisation of globally-best systems and processes. Focused R&D: >90% of the R&D resources are allocated towards developing products for the US markets which, in turn, can be leveraged for strengthening its presence in other regulated markets.
Robust pipeline: The Company has >43 ANDAs pending approval, of which 40% comprise complex and challenging developments as well as >36 filings in other regulated markets at various stages of approval.
Highlights, 2016-17
• Launched three new products in US
• Added 18 products (transfer from partners) to widen our basket to 20 products on own label in US
• Signed 28 master service agreements with distributors and established business relations with 80+ customers
Priorities, 2017-18
• Successful launch of 8-10 products in the US
• Build a nimble supply chain to cater to market opportunities
• Unrelenting focus on product quality and regulatory compliance
• Timely commissioning of new oral, injectable and dermaceutical facilities
• Continued focus on improving R&D capabilities and increasing ANDA filings
Revenue vertical 2
APIS
Alembic’s API business represents critical backward integration that makes it possible to formulate niche products that find global acceptability.
The Company has three API manufacturing facilities (two at Panelav and one at Karakhadi), which cumulatively manufacture more than 100 APIs that find acceptance with customers globally.
Competitive
advantages Capability :
Ability to manufacture products involving complex chemistry, long processes and difficult-to-handle molecules, representing its competitive advantage.
Compliance:
Successfully cleared the USFDA audits for its API units, which showcase the institutionalization of global best practices in business systems and processes.
Capacity:
Recent investments in creating and unlocking capacity have created significant capacity headspace to capitalize on global growth opportunities.
Customer:
Increase in in-house production of intermediates and APIs, improving business profitability.
Highlights, 2016-17
• Revenues increased by 22% from H525 crore in 2015-16 to H640 crore in 2016-17.
• Received EIRs from the FDA (on successful audit clearance) for API-1 and API- 2 facilities.
• Filed 7 DMFs (the US market) and 3 CEPs (Europe market), taking the cumulative filing count to 123 (84 DMFs and 39 CEPs) as on March 31, 2017.
• Commissioned a state-ofthe- art two new API Blocks at Karkhadi and Panelav respectively.
INDEX OF CHARGES:
|
SNo |
SRN |
Charge Id |
Charge Holder Name |
Date of Creation |
Date of Modification |
Date of Satisfaction |
Amount |
Address |
|
1 |
C35754985 |
10534523 |
Bank of Baroda |
08/05/2014 |
- |
- |
13630734.0 |
CFS Branch, Bank of Baroda BuildingOpp. Alkapuri Petrol Pump, R. C. Dutt RoadVadodaraGJ390007IN |
|
2 |
G34911255 |
10296745 |
Bank of Baroda and Others |
30/06/2011 |
27/01/2017 |
- |
3500000000.0 |
First Floor, Bank of Baroda BuildingR C Dutt Road,VadodaraGJ390007IN |
|
3 |
G00923813 |
10323112 |
DBS BANK LIMITED |
02/12/2011 |
- |
21/03/2016 |
780000000.0 |
UPPER GROUND FLOR, BIRLA TOWER,25 BARAKHAMBA ROADNEW DELHIDL110001IN |
|
4 |
B59521583 |
10291650 |
IDBI TRUSTEESHIP SERVICES LIMITED |
01/04/2011 |
- |
11/10/2012 |
250000000.0 |
Asian Bldg., Ground Floor, 17, R.Kamani Marg,Ballard Estate,MUMBAIMH400001IN |
|
5 |
B56674856 |
10291649 |
IDBI TRUSTEESHIP SERVICES LIMITED |
01/04/2011 |
- |
29/08/2012 |
250000000.0 |
Asian Bldg., Ground Floor, 17, R.Kamani Marg,Ballard Estate,MUMBAIMH400001IN |
|
6 |
B43132604 |
10291646 |
IDBI TRUSTEESHIP SERVICES LIMITED |
01/04/2011 |
- |
04/07/2012 |
750000000.0 |
Asian Bldg., Ground Floor, 17, R.Kamani Marg,Ballard Estate,MUMBAIMH400001IN |
CONTINGENT
LIABILITIES:
(INR in million)
|
PARTICULARS |
31.03.2017 |
31.03.2016 |
|
Estimated amount of contracts net of advances remaining to be executed on capital accounts |
2271.800 |
1977.800 |
|
Contingent
liabilities not provided for |
|
|
|
Letter of credit, Guarantees and counter guarantees |
1579.400 |
2080.500 |
|
Liabilities
Disputed in appeals |
|
|
|
Excise duty |
59.900 |
56.400 |
|
Sales Tax |
29.100 |
56.900 |
|
Income Tax |
0.300 |
0.800 |
|
Claims against the company not acknowledged as debt |
3.700 |
34.300 |
|
Disputed liability in respect of Ministry of Industry, Department of Chemicals and Petrochemicals in respect of price of Rifampicin allowed in formulations and landed cost of import. |
3.500 |
3.500 |
|
Total |
96.500 |
151.900 |
FIXED ASSETS
- Building
- Plant and Equipment
- Furniture and Fixtures
- Vehicles
- Office Equipment
UNAUDITED FINANCIAL RESULTS FOR THE QUARTER AND HALF YEAR ENDED SEPTEMBER, 2017
(INR
in Million)
|
|
|
Particulars |
Quarter ended 30.12.2017 |
Quarter ended 30.09.2017 |
Nine months ended 30.12.2017 |
|
1 |
|
Income from
Operations |
|
|
|
|
|
|
Sales/Income from Operations (Gross) |
7549.100 |
7620.100 |
21070.300 |
|
|
|
b) Other Operating Income |
3.300 |
76.100 |
81.700 |
|
|
Total Income from
Operations (Net) |
7552.400 |
7696.200 |
21152.000 |
|
|
2 |
Expenses |
|
|
|
|
|
|
a) |
Cost of Materials consumed |
2189.600 |
1611.600 |
5053.700 |
|
|
b) |
Purchase of Stock-in-trade |
565.500 |
462.500 |
1585.600 |
|
|
c) |
Changes in inventories of finished goods, work-in-progress and
stock-in-trade |
(514.200) |
278.200 |
(367.500) |
|
|
d) |
Employee benefit expenses |
1391.200 |
1525.100 |
4429.400 |
|
|
e) |
Finance Costs |
6.400 |
1.200 |
15.800 |
|
|
f) |
Depreciation and amortization expense |
263.800 |
256.300 |
738.400 |
|
|
g) |
Other expenses |
2312.300 |
1976.100 |
6126.600 |
|
|
Total Expenses |
6214.600 |
6111.000 |
17585.000 |
|
|
|
|
|
|
|
|
|
3 |
Profit before Tax |
1337.700 |
1585.300 |
3567.100 |
|
|
4 |
Tax Expense |
287.300 |
364.300 |
789.100 |
|
|
5 |
Net Profit for the
period after tax |
1050.400 |
1221.000 |
2778.000 |
|
|
|
|
|
|
|
|
|
6 |
Other Comprehensive Income |
|
|
|
|
|
|
Items that will not be classified of profit or loss |
4.900 |
(15.000) |
(27.000) |
|
|
|
Income tax related to items no above |
(1.000) |
3.200 |
5.800 |
|
|
|
|
|
|
|
|
|
|
Total
Comprehensive Income |
1054.300 |
1209.200 |
2756.800 |
|
|
|
|
|
|
|
|
|
|
Earnings / (Loss) Per
Share (INR) |
5.57 |
64.80 |
14.74 |
|
NOTE :
1. The above standalone results, have been reviewed by Statutory Auditors, recommended by the Audit Committee and approved by the Board of Directors of the Company.
2. As per Ind AS 18, Revenue for the quarter ended September 30, 2017 & December 31, 2017 is reported net of GST. Sales of periods up to June 30, 2017 are reported inclusive of excise duty which now is subsumed in GST. The nine months period up to December 31, 2017 includes excise duty up to June 30, 2017, Accordingly, revenue from operations for the quarter and nine months ended December 31,2017 are not comparable with those of the previous periods.
3. As additional information to investors, the Research and Development Expenses are provided here under :
(INR in
Million)
|
Particulars |
Quarter ended 30.12.2017 |
Quarter ended 30.09.2017 |
Nine months ended 30.12.2017 |
|
Research and Development |
952.400 |
936.800 |
2780.500 |
|
|
|
|
|
|
Total |
952.400 |
936.800 |
2780.500 |
4. The Company is engaged in Pharmaceuticals business only and therefore, there is only one reportable segment.
5. The previous quarter's / year's figures have been regrouped / rearranged wherever necessary to make it comparable with the current quarter / period.
CMT REPORT (Corruption, Money Laundering & Terrorism]
The Public Notice information has been collected from various sources
including but not limited to: The Courts, India Prisons Service,
Interpol, etc.
1] INFORMATION ON
DESIGNATED PARTY
No records exist designating subject or any of its beneficial owners,
controlling shareholders or senior officers as terrorist or terrorist
organization or whom notice had been received that all financial transactions
involving their assets have been blocked or convicted, found guilty or against
whom a judgement or order had been entered in a proceedings for violating
money-laundering, anti-corruption or bribery or international economic or
anti-terrorism sanction laws or whose assets were seized, blocked, frozen or
ordered forfeited for violation of money laundering or international
anti-terrorism laws.
2] Court Declaration :
No records exist to suggest that subject is
or was the subject of any formal or informal allegations, prosecutions or other
official proceeding for making any prohibited payments or other improper
payments to government officials for engaging in prohibited transactions or
with designated parties.
3] Asset Declaration :
No records exist to suggest that the property or assets of the subject
are derived from criminal conduct or a prohibited transaction.
4] Record on Financial
Crime :
Charges or conviction
registered against subject: None
5] Records on Violation of
Anti-Corruption Laws :
Charges or
investigation registered against subject: None
6] Records on Int’l
Anti-Money Laundering Laws/Standards :
Charges or
investigation registered against subject: None
7] Criminal Records
No
available information exist that suggest that subject or any of its principals
have been formally charged or convicted by a competent governmental authority
for any financial crime or under any formal investigation by a competent
government authority for any violation of anti-corruption laws or international
anti-money laundering laws or standard.
8] Affiliation with
Government :
No record
exists to suggest that any director or indirect owners, controlling
shareholders, director, officer or employee of the company is a government
official or a family member or close business associate of a Government
official.
9] Compensation Package :
Our market
survey revealed that the amount of compensation sought by the subject is fair
and reasonable and comparable to compensation paid to others for similar
services.
10] Press Report :
No press reports / filings exists on
the subject.
CORPORATE GOVERNANCE
MIRA INFORM as part of its Due Diligence do provide comments on
Corporate Governance to identify management and governance. These factors often
have been predictive and in some cases have created vulnerabilities to credit
deterioration.
Our Governance Assessment focuses principally on the interactions
between a company’s management, its Board of Directors, Shareholders and other
financial stakeholders.
CONTRAVENTION
Subject is not known to have contravened any existing local laws,
regulations or policies that prohibit, restrict or otherwise affect the terms
and conditions that could be included in the agreement with the subject.
FOREIGN EXCHANGE RATES
|
Currency |
Unit
|
INR |
|
US Dollar |
1 |
INR 65.08 |
|
|
1 |
INR 89.85 |
|
Euro |
1 |
INR 80.16 |
INFORMATION DETAILS
|
Information
Gathered by : |
SUP |
|
|
|
|
Analysis Done by
: |
VAR |
|
|
|
|
Report Prepared
by : |
KJL |
SCORE FACTORS
|
DEMERIT POINTS |
|
|
|
--BANK CHARGES |
YES/NO |
YES |
|
--LITIGATION |
YES/NO |
NO |
|
--OTHER ADVERSE INFORMATION |
YES/NO |
NO |
|
MERIT POINTS |
|
|
|
--SOLE DISTRIBUTORSHIP |
YES/NO |
NO |
|
--EXPORT ACTIVITIES |
YES/NO |
NO |
|
--AFFILIATION |
YES/NO |
YES |
|
--LISTED |
YES/NO |
YES |
|
--OTHER MERIT FACTORS |
YES/NO |
YES |
RATING EXPLANATIONS
|
Credit Rating |
Explanation |
Rating Comments |
|
A++ |
Minimum Risk |
Business dealings permissible with minimum
risk of default |
|
A+ |
Low Risk |
Business dealings permissible with low
risk of default |
|
A |
Acceptable Risk |
Business dealings permissible with
moderate risk of default |
|
B |
Medium Risk |
Business dealings permissible on a regular
monitoring basis |
|
C |
Medium High Risk |
Business dealings permissible preferably
on secured basis |
|
D |
High Risk |
Business dealing not recommended or on
secured terms only |
|
NB |
New Business |
No recommendation can be done due to
business in infancy stage |
|
NT |
No Trace |
No recommendation can be done as the
business is not traceable |
NB is stated where there is insufficient information to facilitate rating. However, it is not to be considered as unfavourable.
This score serves as a reference to assess
SC’s credit risk and to set the amount of credit to be extended. It is
calculated from a composite of weighted scores obtained from each of the major
sections of this report. The assessed factors are as follows:
·
Financial
condition covering various ratios
·
Company
background and operations size
·
Promoters
/ Management background
·
Payment
record
·
Litigation
against the subject
·
Industry
scenario / competitor analysis
·
Supplier
/ Customer / Banker review (wherever available)
This report is issued at
your request without any risk and responsibility on the part of MIRA INFORM
PRIVATE LIMITED (MIPL) or its officials.

