|
|
|
MIRA
INFORM REPORT
|
Report No. : |
498235 |
|
Report Date : |
17.03.2018 |
IDENTIFICATION DETAILS
|
Name : |
CIPLA LIMITED [w.e.f.20.07.1984] |
|
|
|
|
Formerly Known
as : |
THE CHEMICAL INDUSTRIAL AND PHARMACEUTICAL
LABORATORIES LIMITED |
|
|
|
|
Registered
Office : |
Cipla House, Peninsula Business Park, Ganpatrao Kadam Marg, Lower Parel, Mumbai – 400013, Maharashtra |
|
Tel. No.: |
91-22-24826000 |
|
|
|
|
Country : |
India |
|
|
|
|
Financials (as
on) : |
31.03.2017 |
|
|
|
|
Date of
Incorporation : |
17.08.1935 |
|
|
|
|
Com. Reg. No.: |
11-002380 |
|
|
|
|
Capital
Investment / Paid-up Capital : |
INR 1609.000 Million |
|
|
|
|
CIN No.: [Company Identification
No.] |
L24239MH1935PLC002380 |
|
|
|
|
IEC No.: |
0388003791 |
|
|
|
|
TAN No.: [Tax Deduction &
Collection Account No.] |
Not Available |
|
|
|
|
PAN No.: [Permanent Account No.] |
AAACC1450B |
|
|
|
|
Legal Form : |
A Public Limited Liability Company. The Company’s Shares are Listed on
the Stock Exchanges. |
|
|
|
|
Line of Business
: |
Manufacturer of Active Pharmaceutical Ingredients (APIs) and
Formulations. [Registered Activity] |
|
|
|
|
No. of Employees
: |
23043 (Approximately) |
RATING & COMMENTS
(Mira Inform has adopted New Rating mechanism w.e.f. 23rd
January 2017)
|
MIRA’s Rating : |
A++ |
|
Credit Rating |
Explanation |
Rating Comments |
|
A++ |
Minimum Risk |
Business dealings permissible with minimum
risk of default |
|
Maximum Credit Limit : |
USD 365728800 |
|
|
|
|
Status : |
Excellent |
|
|
|
|
Payment Behaviour : |
Regular |
|
|
|
|
Litigation : |
Exist |
|
|
|
|
Comments : |
Subject was incorporated 1935 and has a diverse range of more than
1,000 products and 180 global partners across 120 countries. Cipla has a
presence in Africa, Middle East, Latin America, Asia Pacific and China. For the financial year ended 2017, the company has slightly decreased
its operational performance as compared to previous year and but maintained
average profit margin of 9.17%. Rating continues to derive strength from the company’s reputed brand
having global presence and dominant market position of Cipla in the domestic
pharmaceutical industry backed by experienced promoters and synergies derived
from various acquisitions and strong product pipeline. The company has strong and diversified product portfolio with
integrated and diversified operations across various geographies. Rating also takes into account of strong financial profile along with
adequate net worth position and strong debt coverage indicators. Trade relations are reported as fair. Payments are reported to be
regular and as per commitment. In view of aforesaid, the company can be considered good for normal
business dealings at usual trade terms and conditions. |
NOTES :
Any query related to this report can be made
on e-mail : infodept@mirainform.com
while quoting report number, name and date.
ECGC Country Risk Classification List
|
Country Name |
Previous Rating (30.09.2017) |
Current Rating (31.12.2017) |
|
India |
A1 |
A1 |
|
Risk Category |
ECGC
Classification |
|
Insignificant |
A1 |
|
Low Risk |
A2 |
|
Moderately Low Risk |
B1 |
|
Moderate Risk |
B2 |
|
Moderately High Risk |
C1 |
|
High Risk |
C2 |
|
Very High Risk |
D |
EXTERNAL AGENCY RATING
|
Rating Agency Name |
INDIA RATINGS |
|
Rating |
Long Term Rating=AAA |
|
Rating Explanation |
Highest degree of safety and carry lowest
credit risk |
|
Date |
July, 2017 |
|
|
|
|
Rating Agency Name |
INDIA RATINGS |
|
Rating |
Short-term non-fund based bank facilities=A1+ |
|
Rating Explanation |
Very Strong degree of safety and carry lowest
credit risk |
|
Date |
July, 2017 |
RBI DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available RBI Defaulters’ list.
EPF (Employee Provident Fund) DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available EPF (Employee Provident Fund) Defaulters’ list as of
31-03-2016.
BIFR (Board for Industrial & Financial Reconstruction) LISTING
STATUS
Subject’s name is not listed as a Sick Unit in
the publicly available BIFR (Board for Industrial & Financial
Reconstruction) list as of 17.03.2018.
IBBI (Insolvency and Bankruptcy Board of India) LISTING STATUS
Subject’s name is not listed in the publicly
available IBBI (Insolvency and Bankruptcy Board of India) list as of report
date.
INFORMATION DECLINED
MANAGEMENT NON-COOPERATIVE
(Contact No: 91-22-24826300/ 25781791)
[91-22-24826951/ 24826701/ 25781014/ 25761900 does not exists]
LOCATIONS
|
Registered Office : |
Cipla House, Peninsula Business Park, Ganpatrao Kadam Marg, Lower Parel, Mumbai – 400013, Maharashtra, India |
|
Tel. No.: |
91-22-24826000/ 24826300/ 25781791 |
|
Fax No.: |
91-22-24826120 |
|
E-Mail : |
|
|
Website : |
http://www.cipla.com |
|
|
|
|
Active
Pharmaceutical Ingredients (API) Manufacturing
Facility |
|
|
Factory 1: |
Virgonagar, Old Madras Road, Bangalore – 560049, Karnataka, India |
|
|
|
|
Factory 2: |
Bommasandra-Jigani Link Road, Industrial Area, KIADB 4th Phase, Bangalore - 560099, Karnataka, India |
|
|
|
|
Active
Pharmaceutical Ingredients (API) and Formulations Manufacturing
Facility |
|
|
Factory 3: |
MIDC, Patalganga, District Raigad – 410220, Maharashtra, India |
|
|
|
|
Factory 4: |
MIDC Industrial Area, Kurkumbh, Daund, District Pune - 413802, Maharashtra, India |
|
|
|
|
Formulations
Manufacturing Facility |
|
|
Factory 5: |
Verna Industrial Estate, Verna, Salcette, Panaji – 403722, Goa, India |
|
|
|
|
Factory 6: |
Village Malpur Upper, P.O. Bhud, Nalagarh, Baddi, District Solan - 173205, Himachal Pradesh, India |
|
|
|
|
Factory 7: |
Village Kumrek, P.O. Rangpoo-737132, East District Sikkim, India |
|
|
|
|
Factory 8: |
Indore SEZ, Phase ll, Sector III, Pharma Zone, P.O. Pithampur, District Dhar -454774, Madhya Pradesh, India |
|
|
|
|
Factory 9: |
Taza Block, Amba Tareythang Illaka, Rorathang- 737133, Sikkim, India |
DIRECTORS
AS ON 31.03.2017
|
Name : |
Mrs. Naina Kidwai Lal |
|
Designation : |
Director |
|
Address : |
Mustail-29, Killa No 13, Opp Dig Farm Village Jaunapur,
Mehrauli, New Delhi-110047, India |
|
Date of Appointment : |
06.11.2015 |
|
DIN No.: |
00017806 |
|
|
|
|
Name : |
Mrs. Samina Vaziralli |
|
Designation : |
Wholetime Director |
|
Address : |
Flat no 401/402, R.K. Sadan, 4th floor 63 Sir Pochkhanwala
Road, Worli, Mumbai-400025, Maharashtra, India |
|
Date of Appointment : |
27.08.2015 |
|
DIN No.: |
00027923 |
|
|
|
|
Name : |
Mr. Yusuf Khwaja Hamied |
|
Designation : |
Director |
|
Address : |
Windsor Villa, 2nd Floor, Westfiled Estate, Bhulabhai
Desai Road, Mumbai-400026, Maharashtra, India |
|
Date of Appointment : |
21.07.1972 |
|
DIN No.: |
00029049 |
|
|
|
|
Name : |
Mr. Mustafa Khwaja Hamied |
|
Designation : |
Director |
|
Address : |
Flat No. 1c, Somerset Place Bhulabhai Desai Road,
Mumbai-400026, Maharashtra, India |
|
Date of Appointment : |
16.08.1977 |
|
DIN No.: |
00029084 |
|
|
|
|
Name : |
Mr. Ashok Sinha |
|
Designation : |
Director |
|
Address : |
Flat No 18, 4th Floor, Kailash 50 Pedder Road,
Mumbai-400026, Maharashtra, India |
|
Date of Appointment : |
16.07.2013 |
|
DIN No.: |
00070477 |
|
|
|
|
Name : |
Mr. Umang Vohra |
|
Designation : |
Managing Director |
|
Address : |
4604, 46th Floor, Imperial Towers B. B. Nakashe Marg,
Tardeo, Mumbai- 400034, Maharashtra, India |
|
Date of Appointment : |
01.09.2016 |
|
DIN No.: |
02296740 |
|
|
|
|
Name : |
Mr. Radhakrishnan Sethuraman |
|
Designation : |
Wholetime Director |
|
Address : |
Flat No.701, Emerald Heights, Cts - 155, Union Park,
Chembur, Mumbai-400071, Maharashtra, India |
|
Date of Appointment : |
25.08.2011 |
|
DIN No.: |
02313000 |
|
|
|
|
Name : |
Mrs. Punita Lal |
|
Designation : |
Director |
|
Address : |
909b, The Aralias Dlf Golf Links, Gurgaon 122009, Haryana,
India |
|
Date of Appointment : |
13.11.2014 |
|
DIN No.: |
03412604 |
|
|
|
|
Name : |
Mrs. Ireena Vittal |
|
Designation : |
Additional Director |
|
Address : |
4 Alhambra Carmichael Road, Mumbai-400026, Maharashtra,
India |
|
Date of Appointment : |
01.12.2016 |
|
DIN No.: |
05195656 |
|
|
|
|
Name : |
Mr. Adil Zainulbhai |
|
Designation : |
Director |
|
Address : |
The Imperial Apt, Flat No 4701 B B Nakashe Marg, Tardeo,
Mumbai-400034, Maharashtra, India |
|
Date of Appointment : |
23/07/2014 |
|
DIN No.: |
06646490 |
|
|
|
|
Name : |
Mr. Peter Ndimbirwe Mugyenyi |
|
Designation : |
Director |
|
Address : |
Kironde Road Zone, Kayonza Zone, Muyenga-Kabalagala
Kampala NA UG |
|
Date of Appointment : |
12.02.2014 |
|
DIN No.: |
06799942 |
|
|
|
|
Name : |
Mr. Peter Andrew Lankau |
|
Designation : |
Additional Director |
|
Address : |
876 Penn Oak Rd Lower Gwynedd PA 19002 US |
|
Date of Appointment : |
10.01.2017 |
|
DIN No.: |
07688110 |
KEY EXECUTIVES
|
Name : |
Mr. Rajendra Kumar Chopra |
|
Designation : |
Company Secretary |
|
Address : |
H No. C - 69 South City 1, Gurugram-122007, Haryana, India
|
|
Date of Appointment : |
09.02.2017 |
|
PAN No.: |
AACPC7408A |
|
|
|
|
Name : |
Mr. Kedar Narayan Upadhye |
|
Designation : |
Chief Financial Officer (KMP) |
|
Address : |
Flat 503, Tower 2B, Sumer Trinity Near Saamna Press,
Prabhadevi, Mumbai- 400025, Maharashtra, India |
|
Date of Appointment : |
01.08.2016 |
|
PAN No.: |
AAIPU1917C |
MAJOR SHAREHOLDERS / SHAREHOLDING PATTERN
AS ON 31.12.2017
|
Category Of Shareholder |
Total Nos. Shares Held |
Shareholding As A % Of Total No. Of Shares (Calculated As Per Scrr,
1957)As A % Of (A+B+C2) |
|
|
(A) Promoter &
Promoter Group |
295485596 |
36.71 |
|
|
(B) Public |
498004224 |
61.87 |
|
|
(C1) Shares
Underlying Drs |
11454870 |
1.42 |
|
|
|
|
|
|
|
Grand Total |
804944690 |
100.00 |
|

Statement showing
shareholding pattern of the Promoter and Promoter Group
|
Category
of shareholder |
No.
of fully paid up equity shares held |
Shareholding
as a % of total no. of shares (calculated as per SCRR, 1957)As a % of
(A+B+C2) |
|
|
A1) Indian |
0.00 |
|
|
|
Individuals/Hindu
undivided Family |
11,17,81,000 |
14.09 |
|
|
Mustafa Khwaja Hamied |
3,16,40,000 |
3.99 |
|
|
Sophie Ahmed |
1,54,71,000 |
1.95 |
|
|
Sophie Ahmed |
1,54,71,000 |
1.95 |
|
|
Sophie Ahmed |
1,13,22,250 |
1.43 |
|
|
Sophie Ahmed |
37,17,750 |
0.47 |
|
|
Rumana Hamied |
54,72,000 |
0.69 |
|
|
Rumana Hamied |
44,14,500 |
0.56 |
|
|
Samina Vaziralli |
1,35,31,000 |
1.71 |
|
|
Samina Vaziralli |
43,78,500 |
0.55 |
|
|
Shirin Hamied |
63,63,000 |
0.80 |
|
|
Any Other
(specify) |
60,22,409 |
0.76 |
|
|
MN Rajkumar Garments LLP |
26,01,852 |
0.33 |
|
|
Shree Riddhi Chemicals LLP |
24,34,970 |
0.31 |
|
|
Alps Remedies Pvt Ltd |
4,92,985 |
0.06 |
|
|
Hamsons Laboratories LLP |
4,92,602 |
0.06 |
|
|
Neo Research Labs Private Limited |
0.00 |
|
|
|
Sub Total A1 |
11,78,03,409 |
14.85 |
|
|
A2) Foreign |
0.00 |
|
|
|
Individuals
(NonResident Individuals/ Foreign Individuals) |
17,76,82,187 |
22.39 |
|
|
Yusuf Khwaja Hamied |
16,67,42,687 |
21.01 |
|
|
Kamil Hamied |
63,54,000 |
0.80 |
|
|
Kamil Hamied |
44,95,500 |
0.57 |
|
|
Kamil Hamied |
90,000 |
0.01 |
|
|
Farida Hamied |
0.00 |
|
|
|
Sub Total A2 |
17,76,82,187 |
22.39 |
|
|
A=A1+A2 |
29,54,85,596 |
37.24 |
|
Statement showing
shareholding pattern of the Public shareholder
|
Category
& Name of the Shareholders |
No.
of fully paid up equity shares held |
Shareholding
% calculated as per SCRR, 1957 As a % of (A+B+C2) |
|
|
|||
|
|||
|
|||
|
B1) Institutions |
0 |
0.00 |
|
|
Mutual Funds/ |
73308763 |
9.24 |
|
|
ICICI Prudential Mutual Fund |
29464194 |
3.71 |
|
|
HDFC Mutual Fund |
19797093 |
2.49 |
|
|
Reliance Capital Mutual Fund |
11212712 |
1.41 |
|
|
Foreign
Portfolio Investors |
185580848 |
23.39 |
|
|
First State Investments ICVC-Stewart
Investors Asia Pacific Leaders Fund |
22132075 |
2.79 |
|
|
Government Pension Fund Global |
15175186 |
1.91 |
|
|
First State Investments ICVC-Stewart
Investors Global Emerging Market Leaders Fund |
11193580 |
1.41 |
|
|
Financial
Institutions/ Banks |
2276172 |
0.29 |
|
|
Insurance
Companies |
36665195 |
4.62 |
|
|
Life Insurance Corporation of India |
34688152 |
4.37 |
|
|
Any Other
(specify) |
2454633 |
0.31 |
|
|
Foreign Institutional Investors / Foreign
Institutional Investors - DR |
2454633 |
0.31 |
|
|
Sub Total B1 |
300285611 |
37.84 |
|
|
B2) Central
Government/ State Government(s)/ President of India |
0 |
0.00 |
|
|
B3)
Non-Institutions |
0 |
0.00 |
|
|
Individual share
capital upto INR 0.200 Million |
62436930 |
7.87 |
|
|
Individual share
capital in excess of INR 0.200 Million |
72275926 |
9.11 |
|
|
NBFCs registered
with RBI |
54984 |
0.01 |
|
|
Any Other
(specify) |
62950773 |
7.93 |
|
|
Non-Resident Indians/Foreign
Nationals/Foreign Bodies |
28223836 |
3.56 |
|
|
Trusts |
4484144 |
0.57 |
|
|
Overseas corporate bodies |
375 |
0.00 |
|
|
Clearing Members |
927943 |
0.12 |
|
|
Bodies Corporate |
25360821 |
3.20 |
|
|
Foreign Bodies - DR |
160525 |
0.02 |
|
|
Investor Education and Protection Fund
Authority Ministry of Corporate Affairs |
3793129 |
0.48 |
|
|
Sub Total B3 |
197718613 |
24.92 |
|
|
B=B1+B2+B3 |
498004224 |
62.76 |
|
Statement showing shareholding pattern of the Non
Promoter- Non Public shareholder
|
Category
& Name of the Shareholders(I) |
Nos.
of shares underlying Depository Receipts(VI) |
Shareholding
% calculated as per SCRR, 1957 As a % of (A+B+C2)(VIII) |
|
|
|||
|
C1) Custodian/DR
Holder |
0.00 |
|
|
|
Custodian/DR
Holder |
1,14,54,870 |
0.00 |
|
|
The Bank of New York Mellon |
1,14,54,870 |
0.00 |
|
|
Sub Total C1 |
1,14,54,870 |
0.00 |
|
|
C2) Employee
Benefit Trust |
0.00 |
|
|
|
C= C1+C2 |
1,14,54,870 |
0.00 |
|
BUSINESS DETAILS
|
Line of Business : |
Manufacturer of Active Pharmaceutical Ingredients (APIs) and
Formulations. [Registered Activity] |
|
|
|
|
Brand Names : |
“CIPLA” |
|
|
|
|
Agencies Held : |
Not Available |
|
|
|
|
Exports : |
Not Divulged |
|
|
|
|
Imports : |
Not Divulged |
|
|
|
|
Terms : |
|
|
Selling : |
Not Divulged |
|
|
|
|
Purchasing : |
Not Divulged |
PRODUCTION STATUS: NOT AVAILABLE
GENERAL INFORMATION
|
Suppliers : |
|
||||||||||||||
|
|
|
||||||||||||||
|
Customers : |
|
||||||||||||||
|
|
|
||||||||||||||
|
No. of Employees : |
23043 (Approximately) |
||||||||||||||
|
|
|
||||||||||||||
|
Bankers : |
· The Hongkong and Shanghai Banking Corporation Limited 52/60, Mahatma Gandhi Road, Mumbai-400023, Maharashtra, India |
||||||||||||||
|
|
|
||||||||||||||
|
Facilities : |
--- |
|
Auditors : |
|
|
Name : |
Walker Chandiok and Company LLP Chartered Accountants |
|
Address
: |
16th Floor, Tower II, Indiabulls Finance Centre, S B Marg,
Elphinstone (West), Mumbai-400013, Maharashtra, India |
|
Tel No: |
91-22-66262600 |
|
Fax No: |
91-22-66262601 |
|
|
|
|
Cost Auditor: |
|
|
Name : |
Mr. D. H. Zaveri Cost Accountant |
|
|
|
|
Secretarial Auditor: |
|
|
Name : |
BNP and Associates Chartered Accountants |
|
|
|
|
Chief Internal
Auditor: |
|
|
Name : |
Mr. Deepak Viegas |
|
|
|
|
Memberships : |
Not Available |
|
|
|
|
Collaborators : |
Not Available |
|
|
|
|
Subsidiaries (held directly): |
· Cipla FZE · Goldencross Pharma Private Limited · Cipla (Mauritius) Limited · Meditab Specialities Private Limited · Cipla Medpro South Africa Proprietary Limited · Cipla Holding B.V. · Cipla Biotec Private Limited · Cipla (EU) Limited · Saba Investment Limited · Jay Precision Pharmaceuticals Private Limited · Cipla Health Limited |
|
|
|
|
Subsidiaries (held indirectly) : |
· Four M Propack Private Limited** · Cipla (UK) Limited · Cipla Australia Pty. Limited · Medispray Laboratories Private Limited · Sitec Labs Private Limited · Meditab Holdings Limited · Meditab Specialities New Zealand Limited · Cipla İlaç Ticaret Anonim Şirketi · Cipla Kenya Limited · Cipla Malaysia Sdn. Bhd. · Cipla Europe NV · Cipla Quality Chemical Industries Limited · Cipla Croatia d.o.o. · Galilee Marketing Proprietary Limited · Inyanga Trading 386 Proprietary Limited · Xeragen Laboratories Proprietary Limited · Cipla Medpro Holdings Proprietary Limited · Cape to Cairo Exports Proprietary Limited · Cipla Agrimed Proprietary Limited · Cipla Dibcare Proprietary Limited · Cipla Life Sciences Proprietary Limited · Cipla-Medpro Proprietary Limited · Cipla-Medpro Distribution Centre Proprietary Ltd. · Cipla Medpro Botswana Proprietary Limited · Cipla Medpro Research and Development Proprietary Ltd.* · Cipla Nutrition Proprietary Limited · Cipla Vet Proprietary Limited · Medpro Pharmaceutica Proprietary Limited · Med Man Care Proprietary Limited · Breathe Free Lanka (Private) Limited · Cipla Canada Inc. *** · Medica Pharmaceutical Industries Company Limited · Al Jabal For Drugs And Medical Appliances Company Limited · Cipla Pharma Lanka (Private) Limited · Cipla Pharma Nigeria Limited · Cipla Brasil Importadora E Distribuidora De Medicamentos Ltda. · Cipla Maroc SA · Cipla Middle East Pharmaceuticals FZ-LLC · Quality Chemicals Limited · Cipla Philippines Inc. · Cipla USA Inc. · InvaGen Pharmaceuticals Inc. · Exelan Pharmaceuticals Inc. · Cipla BioTec South Africa Proprietary Limited ## ·
CIPLA Algérie # |
|
|
|
|
Associates : |
Stempeutics Research Private Limited |
|
|
|
|
Trusts over which company has control/ significance influence : |
· Cipla Employees Stock Option Trust · Cipla Health Employees Stock Option Trust |
|
|
|
|
Entity over which key Management Personnel are able to excise
significant influence : |
· Cipla Foundation · Hamied Foundation (w.e.f. 3rd February, 2016) · Cipla Cancer & AIDS Foundation |
|
Notes: * De-registered on 6th December, 2016 ** Ceased to be subsidiary w.e.f. 1st February, 2017 *** D-registered on 1st March, 2017 ## w.e.f 10th June, 2016 # w.e.f. 6th June, 2016 |
|
CAPITAL STRUCTURE
AFTER 11.08.2017
Authorised Capital : INR
1750.000 Million
Issued, Subscribed & Paid-up Capital : INR 1610.198 Million
AS ON 31.03.2017
Authorised Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
875000000 |
Equity Shares |
INR 2/- each |
INR 1750.000 Million |
|
|
|
|
|
Issued Capital
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
805513469 |
Equity Shares |
INR 2/- each |
INR 1611.000
Million |
|
|
|
|
|
Subscribed & Paid-up Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
804510074 |
Equity Shares |
INR 2/- each |
INR 1609.000
Million |
|
|
|
|
|
Reconciliation of the
number of shares and amount outstanding at the beginning and at the end of the
reporting period
|
Particulars |
31.03.2017 |
|
|
|
Number of shares |
Amount |
|
Equity Shares at the beginning of the year |
803384282 |
1606.800 |
|
Add: Equity Shares issued on exercise of employee stock options |
1125792 |
2.200 |
|
Equity Shares at the end of the year |
804510074 |
1609.000 |
Details of Shareholders
holding more than 5 percent shares in the Company
|
Name of
Shareholders |
31.03.2017 |
|
|
Number of shares |
% of Holding |
|
|
Dr. Y. K. Hamied |
166742687 |
20.73% |
|
ICICI Prudential Mutual Fund |
41190092 |
5.12% |
|
Sophie Ahmed |
45982000 |
5.72% |
|
Life Insurance Corporation of India |
45325137 |
5.63% |
Terms and Rights attached to Equity Shares
The Company has only one class of equity shares having a par value of INR 2 per share. Each holder of equity share is entitled to one vote per share. The Company declares and pays dividends in Indian Rupees. The dividend proposed by the Board of Directors is subject to the approval of the shareholders in the ensuing Annual General Meeting.
In the event of liquidation of the Company, the holders of equity shares will be entitled to receive remaining assets of the Company, after distribution of all preferential amounts. The distribution will be in proportion to the number of equity shares held by the shareholder.
FINANCIAL DATA
[all figures are
in INR Million]
ABRIDGED
BALANCE SHEET [STANDALONE]
|
SOURCES OF FUNDS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
I.
EQUITY AND LIABILITIES |
|
|
|
|
(1)Shareholders' Funds |
|
|
|
|
(a) Share Capital |
1609.000 |
1606.800 |
1605.900 |
|
(b) Reserves & Surplus |
126396.100 |
118252.000 |
105191.200 |
|
(c) Money received against share warrants |
0.000 |
0.000 |
0.000 |
|
|
|
|
|
|
(2) Share
Application money pending allotment |
0.000 |
0.000 |
0.000 |
|
Total
Shareholders’ Funds (1) + (2) |
128005.100 |
119858.800 |
106797.100 |
|
|
|
|
|
|
(3)
Non-Current Liabilities |
|
|
|
|
(a) Long-term borrowings |
0.700 |
1.300 |
4.100 |
|
(b) Deferred tax liabilities (Net) |
0.000 |
358.500 |
1145.300 |
|
(c)
Other long term liabilities |
1252.000 |
1307.200 |
1572.500 |
|
(d)
long-term provisions |
1256.100 |
1320.000 |
1019.300 |
|
Total
Non-current Liabilities (3) |
2508.800 |
2987.000 |
3741.200 |
|
|
|
|
|
|
(4) Current Liabilities |
|
|
|
|
(a)
Short term borrowings |
3242.600 |
11316.800 |
13802.000 |
|
(b)
Trade payables |
12982.100 |
9908.400 |
13822.300 |
|
(c)
Other current liabilities |
6854.300 |
5827.300 |
5435.500 |
|
(d)
Short-term provisions |
2479.300 |
2492.200 |
1453.700 |
|
Total
Current Liabilities (4) |
25558.300 |
29544.700 |
34513.500 |
|
|
|
|
|
|
TOTAL |
156072.200 |
152390.500 |
145051.800 |
|
|
|
|
|
|
II.
ASSETS |
|
|
|
|
(1) Non-current assets |
|
|
|
|
(a)
Fixed Assets |
|
|
|
|
(i)
Tangible assets |
40951.600 |
37022.800 |
34498.100 |
|
(ii)
Intangible Assets |
1401.000 |
1238.300 |
1255.800 |
|
(iii)
Capital work-in-progress |
5405.200 |
5128.100 |
3390.000 |
|
(iv) Intangible assets under development |
152.500 |
379.100 |
217.100 |
|
(b) Non-current
Investments |
36480.300 |
37165.900 |
34357.600 |
|
(c) Deferred tax assets
(net) |
595.400 |
0.000 |
0.000 |
|
(d) Long-term Loan
and Advances |
2157.500 |
2193.700 |
2107.400 |
|
(e)
Other Non-current assets |
5475.300 |
4588.900 |
3809.000 |
|
Total
Non-Current Assets |
92618.800 |
87716.800 |
79635.000 |
|
|
|
|
|
|
(2) Current assets |
|
|
|
|
(a)
Current investments |
6381.800 |
5395.200 |
3844.600 |
|
(b)
Inventories |
26535.000 |
29184.700 |
32892.000 |
|
(c)
Trade receivables |
19387.900 |
18964.100 |
20198.900 |
|
(d)
Cash and cash equivalents |
584.600 |
530.100 |
827.600 |
|
(e)
Short-term loans and advances |
95.300 |
109.200 |
123.200 |
|
(f)
Other current assets |
10468.800 |
10490.400 |
7530.500 |
|
Total
Current Assets |
63453.400 |
64673.700 |
65416.800 |
|
|
|
|
|
|
TOTAL |
156072.200 |
152390.500 |
145051.800 |
PROFIT
& LOSS ACCOUNT
|
|
PARTICULARS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
|
|
SALES |
|
|
|
|
|
|
|
Revenue from Operations |
109745.800 |
121177.200 |
101317.800 |
|
|
|
Other Income |
1298.500 |
2803.000 |
1479.100 |
|
|
|
TOTAL (A) |
111044.300 |
123980.200 |
102796.900 |
|
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
|
|
Cost of Materials Consumed |
29560.400 |
36333.400 |
34267.400 |
|
|
|
Purchases of Stock-in-Trade |
11289.900 |
10375.600 |
9034.100 |
|
|
|
Changes in inventories of
finished goods, work-in-progress and Stock-in-Trade |
562.700 |
2283.500 |
(3490.500) |
|
|
|
Employees benefits expense |
17289.700 |
17785.600 |
14996.300 |
|
|
|
Other expenses |
32566.400 |
33864.800 |
26897.400 |
|
|
|
Impairment of Investments |
2514.100 |
0.000 |
0.000 |
|
|
|
TOTAL
(B) |
93783.200 |
100642.900 |
81704.700 |
|
|
|
|
|
|
|
|
|
PROFIT/(LOSS)
BEFORE INTEREST, TAX, DEPRECIATION AND AMORTISATION (A-B) (C) |
17261.100 |
23337.300 |
21092.200 |
|
|
|
|
|
|
|
|
|
Less |
FINANCIAL
EXPENSES (D) |
392.000 |
1470.700 |
1360.500 |
|
|
|
|
|
|
|
|
|
|
PROFIT/(LOSS) BEFORE TAX, DEPRECIATION AND AMORTISATION
(C-D) (E) |
16869.100 |
21866.600 |
19731.700 |
|
|
|
|
|
|
|
|
|
Less/ Add |
DEPRECIATION/
AMORTISATION (F) |
4999.700 |
4426.900 |
4332.000 |
|
|
|
|
|
|
|
|
|
|
PROFIT/(LOSS) BEFORE TAX (E-F) (G) |
11869.400 |
17439.700 |
15399.700 |
|
|
|
|
|
|
|
|
|
Less |
TAX (H) |
2120.000 |
2816.700 |
3588.800 |
|
|
|
|
|
|
|
|
|
|
PROFIT/(LOSS) AFTER TAX (G-H) (I) |
9749.400 |
14623.000 |
11810.900 |
|
|
|
|
|
|
|
|
|
|
EARNINGS IN
FOREIGN CURRENCY |
|
|
|
|
|
|
|
FOB value of export |
50049.700 |
66836.700 |
47363.300 |
|
|
|
Technical Know-how and Licensing Fees |
459.400 |
384.300 |
1568.200 |
|
|
|
Other Service Fees etc. |
557.200 |
240.300 |
172.500 |
|
|
TOTAL EARNINGS |
51066.300 |
67461.300 |
49104.000 |
|
|
|
|
|
|
|
|
|
|
IMPORTS |
|
|
|
|
|
|
|
Raw Materials and Packing Materials |
9389.400 |
14530.100 |
15154.300 |
|
|
|
Components and Spare Parts |
226.700 |
270.000 |
237.300 |
|
|
|
Capital Goods |
2586.700 |
2470.000 |
1237.500 |
|
|
TOTAL IMPORTS |
12202.800 |
17270.100 |
16629.100 |
|
|
|
|
|
|
|
|
|
|
Earnings/(Loss) Per Share (INR) |
|
|
|
|
|
|
Basic |
12.13 |
18.21 |
14.71 |
|
|
|
Diluted
|
12.11 |
18.16 |
14.66 |
|
CURRENT MATURITIES
OF LONG TERM DEBT DETAILS
|
Particulars |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Current Maturities of Long term debt – Sales Tax Deferral Loan |
0.700 |
0.700 |
0.100 |
|
|
|
|
|
|
Cash Generated from Operations |
25368.300 |
21190.300 |
13998.200 |
|
|
|
|
|
|
Net Cash Flow from Operating Activities |
22055.600 |
17284.000 |
10514.100 |
KEY
RATIOS
EFFICIENCY RATIOS
|
PARTICULARS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Average Collection Days (Sundry
Debtors / Income * 365 Days) |
64.48 |
57.12 |
72.77 |
|
|
|
|
|
|
Account Receivables Turnover (Income / Sundry Debtors) |
5.66 |
6.39 |
5.02 |
|
|
|
|
|
|
Average Payment Days (Sundry Creditors / Purchases * 365 Days) |
116.00 |
77.43 |
116.51 |
|
|
|
|
|
|
Inventory Turnover (Operating Income / Inventories) |
0.65 |
0.80 |
0.64 |
|
|
|
|
|
|
Asset Turnover (Operating Income / Net Fixed Assets) |
0.36 |
0.53 |
0.54 |
LEVERAGE RATIOS
|
PARTICULARS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Debt Ratio ((Borrowing
+ Current Liabilities) / Total Assets) |
0.16 |
0.19 |
0.24 |
|
|
|
|
|
|
Debt Equity Ratio (Total Liability / Networth) |
0.03 |
0.09 |
0.13 |
|
|
|
|
|
|
Current Liabilities to Networth (Current Liabilities / Net Worth) |
0.20 |
0.25 |
0.32 |
|
|
|
|
|
|
Fixed Assets to Networth (Net Fixed Assets / Networth) |
0.37 |
0.37 |
0.37 |
|
|
|
|
|
|
Interest Coverage Ratio (PBIT / Financial Charges) |
44.03 |
15.87 |
15.50 |
PROFITABILITY RATIOS
|
PARTICULARS |
|
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Net Profit Margin ((PAT / Sales) * 100) |
% |
8.88 |
12.07 |
11.66 |
|
|
|
|
|
|
|
Return on Total Assets ((PAT / Total Assets) * 100) |
% |
6.25 |
9.60 |
8.14 |
|
|
|
|
|
|
|
Return on Investment (ROI) ((PAT / Networth) * 100) |
% |
7.62 |
12.20 |
11.06 |
SOLVENCY RATIOS
|
PARTICULARS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Current Ratio (Current
Assets / Current Liabilities) |
2.48 |
2.19 |
1.90 |
|
|
|
|
|
|
Quick Ratio ((Current Assets – Inventories) / Current
Liabilities) |
1.44 |
1.20 |
0.94 |
|
|
|
|
|
|
G-Score Ratio Financial (Networth / Total Assets) |
0.82 |
0.79 |
0.74 |
|
|
|
|
|
|
G-Score Ratio Debt (Debts / Equity Capital) |
2.02 |
7.04 |
8.60 |
|
|
|
|
|
|
G-Score Ratio Liquidity (Total Current Assets / Total Current Liabilities) |
2.48 |
2.19 |
1.90 |
Total
Liability = Short-term Debt + Long-term Debt + Current Maturities of Long-term
debts
STOCK
PRICES
|
Face Value |
INR 2.00/- |
|
|
|
|
Market Value |
INR 561.00/- |
FINANCIAL DATA
[all figures are
in INR Million]
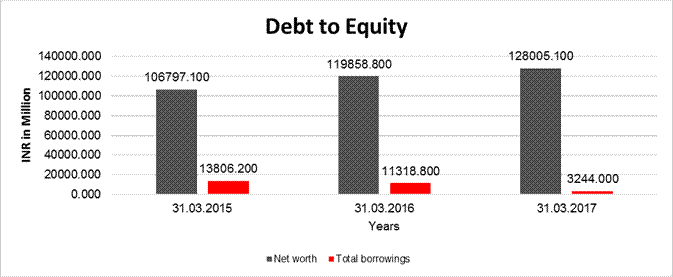
DEBT EQUITY RATIO
|
Particular |
31.03.2015 |
31.03.2016 |
31.03.2017 |
|
|
INR
In Million |
INR
In Million |
INR
In Million |
|
Share Capital |
1605.900 |
1606.800 |
1609.000 |
|
Reserves & Surplus |
105191.200 |
118252.000 |
126396.100 |
|
Money received against share
warrants |
0.000 |
0.000 |
0.000 |
|
Share Application money pending
allotment |
0.000 |
0.000 |
0.000 |
|
Net
worth |
106797.100 |
119858.800 |
128005.100 |
|
|
|
|
|
|
long-term borrowings |
4.100 |
1.300 |
0.700 |
|
Short term borrowings |
13802.000 |
11316.800 |
3242.600 |
|
Current Maturities of Long
term debt |
0.100 |
0.700 |
0.700 |
|
Total
borrowings |
13806.200 |
11318.800 |
3244.000 |
|
Debt/Equity
ratio |
0.129 |
0.094 |
0.025 |
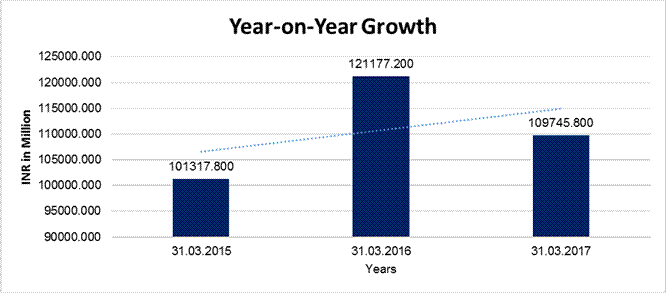
YEAR-ON-YEAR GROWTH
|
Year
on Year Growth |
31.03.2015 |
31.03.2016 |
31.03.2017 |
|
|
INR
In Million |
INR
In Million |
INR
In Million |
|
Sales |
101317.800 |
121177.200 |
109745.800 |
|
|
|
19.601 |
(9.434) |
NET PROFIT MARGIN
|
Net
Profit Margin |
31.03.2015 |
31.03.2016 |
31.03.2017 |
|
|
INR
In Million |
INR
In Million |
INR
In Million |
|
Sales |
101317.800 |
121177.200 |
109745.800 |
|
Profit |
11810.900 |
14623.000 |
9749.400 |
|
|
11.66% |
12.07% |
8.88% |

ABRIDGED
BALANCE SHEET [CONSOLIDATED]
|
SOURCES OF FUNDS |
|
31.03.2017 |
31.03.2016 |
|
I.
EQUITY AND LIABILITIES |
|
|
|
|
(1)Shareholders' Funds |
|
|
|
|
(a) Share Capital |
|
1609.000 |
1606.800 |
|
(b) Reserves & Surplus |
|
123645.200 |
113555.400 |
|
(c) Money received against share warrants |
|
0.000 |
0.000 |
|
|
|
|
|
|
(2) Share
Application money pending allotment |
|
0.000 |
0.000 |
|
(3) Non Controlling Interest |
|
4382.300 |
3500.900 |
|
Total
Shareholders’ Funds (1) + (2) |
|
129636.500 |
118663.100 |
|
|
|
|
|
|
(3)
Non-Current Liabilities |
|
|
|
|
(a) Long-term borrowings |
|
36453.600 |
2218.800 |
|
(b) Deferred tax liabilities (Net) |
|
7568.900 |
9757.300 |
|
(c)
Other long term liabilities |
|
1387.100 |
1435.300 |
|
(d)
long-term provisions |
|
1405.200 |
1446.800 |
|
Total
Non-current Liabilities (3) |
|
46814.800 |
14858.200 |
|
|
|
|
|
|
(4) Current Liabilities |
|
|
|
|
(a)
Short term borrowings |
|
4672.300 |
49696.700 |
|
(b)
Trade payables |
|
15711.400 |
14758.200 |
|
(c)
Other current liabilities |
|
9478.000 |
10197.100 |
|
(d)
Short-term provisions |
|
3219.000 |
3108.500 |
|
Total
Current Liabilities (4) |
|
33080.700 |
77760.500 |
|
|
|
|
|
|
TOTAL |
|
209532.000 |
211281.800 |
|
|
|
|
|
|
II.
ASSETS |
|
|
|
|
(1) Non-current assets |
|
|
|
|
(a)
Fixed Assets |
|
|
|
|
(i)
Tangible assets |
|
50086.900 |
46048.500 |
|
(ii)
Intangible Assets |
|
17848.800 |
20579.000 |
|
(iii)
Capital work-in-progress |
|
7192.300 |
7410.100 |
|
(iv) Intangible assets under development |
|
9637.500 |
13198.600 |
|
(b) Non-current
Investments |
|
1373.600 |
1763.500 |
|
(c) Deferred tax assets
(net) |
|
1681.300 |
786.900 |
|
(d) Long-term Loan
and Advances |
|
394.800 |
418.400 |
|
(e)
Other Non-current assets |
|
6471.800 |
5608.800 |
|
(f)Goodwill
on Consolidated |
|
26784.300 |
27055.700 |
|
Total
Non-Current Assets |
|
121471.300 |
122869.500 |
|
|
|
|
|
|
(2) Current assets |
|
|
|
|
(a)
Current investments |
|
8373.900 |
5823.400 |
|
(b)
Inventories |
|
34852.800 |
38080.500 |
|
(c)
Trade receivables |
|
24974.200 |
23562.700 |
|
(d)
Cash and cash equivalents |
|
6242.100 |
8714.000 |
|
(e)
Short-term loans and advances |
|
95.300 |
109.200 |
|
(f)
Other current assets |
|
12831.800 |
12122.500 |
|
(g) Assets classified as held for sale (net) |
|
690.600 |
0.000 |
|
Total
Current Assets |
|
88060.700 |
88412.300 |
|
|
|
|
|
|
TOTAL |
|
209532.000 |
211281.800 |
PROFIT
& LOSS ACCOUNT
|
|
PARTICULARS |
|
31.03.2017 |
31.03.2016 |
|
|
|
SALES |
|
|
|
|
|
|
|
Revenue from Operations |
|
146302.400 |
137901.000 |
|
|
|
Other Income |
|
2286.900 |
2082.100 |
|
|
|
TOTAL |
|
148589.300 |
139983.100 |
|
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
|
|
Cost of Materials Consumed |
|
42726.600 |
41781.200 |
|
|
|
Purchases of Stock-in-Trade |
|
9335.000 |
8481.900 |
|
|
|
Changes in inventories of
finished goods, work-in-progress and Stock-in-Trade |
|
1109.600 |
635.500 |
|
|
|
Employees benefits expense |
|
26338.200 |
24340.100 |
|
|
|
Other expenses |
|
42035.100 |
37865.600 |
|
|
|
TOTAL
|
|
121544.500 |
113104.300 |
|
|
|
|
|
|
|
|
|
PROFIT/(LOSS)
BEFORE INTEREST, TAX, DEPRECIATION AND AMORTISATION |
|
27044.800 |
26878.800 |
|
|
|
|
|
|
|
|
|
Less |
FINANCIAL
EXPENSES |
|
1593.800 |
2066.300 |
|
|
|
|
|
|
|
|
|
|
PROFIT/(LOSS) BEFORE TAX, DEPRECIATION AND AMORTISATION |
|
25451.000 |
24812.500 |
|
|
|
|
|
|
|
|
|
Less/ Add |
DEPRECIATION/
AMORTISATION |
|
13229.300 |
7542.200 |
|
|
|
|
|
|
|
|
|
|
PROFIT/(LOSS) BEFORE TAX |
|
12221.700 |
17270.300 |
|
|
|
|
|
|
|
|
|
Less |
TAX |
|
1797.600 |
3315.900 |
|
|
|
|
|
|
|
|
|
|
PROFIT/ (LOSS)
AFTER TAX FOR THE YEAR BEFORE
SHARE OF PROFIT/ (LOSS) FROM ASSOCIATES AND INTEREST |
|
10424.100 |
13954.400 |
|
|
|
|
|
|
|
|
|
Less |
SHARE
OF LOSS FROM ASSOCIATES |
|
69.900 |
120.200 |
|
|
|
|
|
|
|
|
|
|
PROFIT FOR THE YEAR |
|
10354.200 |
13834.200 |
|
|
|
|
|
|
|
|
|
|
Earnings/(Loss) Per Share (INR) |
|
|
|
|
|
|
Basic |
|
12.52 |
16.93 |
|
|
|
Diluted
|
|
12.50 |
16.89 |
|
LEGAL CASES
HIGH COURT
|
AJANTA PHARMA LTD. VS CIPLA LTD. AND ANR |
|
high court-Mumbai |
|
Case no:965. NMS/1067/2012 |
|
Case status:Pending |
|
Judge:HON'BLE SHRI JUSTICE N.M. JAMDAR |
|
Date:2013-02-13T00:00:00Z |
HIGH COURT
|
THE COMMISSIONER OF CENTR AL EXCISEPUNEIII VS MS. CIPLA LTD. |
|
high court-Mumbai |
|
Case no:903. NMA/1305/2012 |
|
Case status:Pending |
|
Judge:HON'BLE SHRI JUSTICE A. M. THIPSAY |
|
Date:2012-06-26T00:00:00Z |
HIGH COURT
|
C.I.T.BOMBAY CITYV. VS MS. CIPLA LIMITED. |
|
high court-Mumbai |
|
Case no:136. ITR/201/1996 |
|
Case status:Pending |
|
Judge:REGISTRAR(OS)/PROTHONOTARY & SR. MASTER |
|
Date:2009-04-28T00:00:00Z |
HIGH COURT
|
SYNTEX PHARM AG SWITZERL AND VS CIPLA LTD. AND ANR |
|
high court-Mumbai |
|
Case no:17. S/2997/2008 |
|
Case status:Pending |
|
Judge:OFFICIAL ASSIGNEE |
|
Date:2011-05-01T00:00:00Z |
HIGH COURT
|
THE COMMISSIONER OF CENTR AL EXCISE MUMBAI III VS MS CIPLA LTD. |
|
high court-Mumbai |
|
Case no:953. CEXA/211/2006 |
|
Case status:Pending |
|
Judge:HON'BLE SHRI JUSTICE A. R. JOSHI |
|
Date:2011-12-17T00:00:00Z |
HIGH COURT
|
THE COMMISSIONER OF INCOM E TAX CENTRAL I MUMBAI VS MS. CIPLA LTD . |
|
high court-Mumbai |
|
Case no:104. ITXA/1129/2010 |
|
Case status:Pending |
|
Judge:REGISTRAR (JUDICIAL - I) |
|
Date:2011-02-28T00:00:00Z |
HIGH COURT
|
mrs sendra leveena lasrad o VS cipla ltd and anr |
|
high court-Mumbai |
|
Case no:72. COAPP/13/2007 |
|
Case status:Pending |
|
Judge:MASTER & ASSTT. PROTHONOTARY(JUDL) |
|
Date:2009-11-20T00:00:00Z |
HIGH COURT
|
mrs irene louis lasrado VS cipla ltd and anr |
|
high court-Mumbai |
|
Case no:72. COAPP/12/2007 |
|
Case status:Pending |
|
Judge:MASTER & ASSTT. PROTHONOTARY(JUDL) |
|
Date:2009-11-20T00:00:00Z |
HIGH COURT
|
SHRAMIK MAHASANGH VS Ms. CIPLA LIMITED |
|
high court-Mumbai |
|
Case no:13. WP/1906/2012 |
|
Case status:Pending |
|
Judge:BEFORE MRS. A. A. UPADHYE,MASTER & REGISTRAR IN EQUITY AND |
|
Date:2012-09-26T00:00:00Z |
HIGH COURT
|
THE COMMISSIONER OF CENTR AL EXCISEPUNEIII VS MS. CIPLA LTD. |
|
high court-Mumbai |
|
Case no:22. CEXAL/69/2012 |
|
Case status:Pending |
|
Judge:BEFORE MRS. A. A. UPADHYE,MASTER & REGISTRAR IN EQUITY AND |
|
Date:2012-09-26T00:00:00Z |
HIGH COURT
|
THE COMMISSIONER OF CENTR AL EXCISEPUNEIII VS MS. CIPLA LTD. |
|
high court-Mumbai |
|
Case no:22. CEXAL/68/2012 |
|
Case status:Pending |
|
Judge:BEFORE MRS. A. A. UPADHYE,MASTER & REGISTRAR IN EQUITY AND |
|
Date:2012-09-26T00:00:00Z |
LOCAL AGENCY FURTHER INFORMATION
|
Sr. No. |
Check list by
info agents |
Available in
Report (Yes/No) |
|
1 |
Year of establishment |
Yes |
|
2 |
Constitution of the entity -Incorporation
details |
Yes |
|
3 |
Locality of the entity |
Yes |
|
4 |
Premises details |
No |
|
5 |
Buyer visit details |
-- |
|
6 |
Contact numbers |
Yes |
|
7 |
Name of the person contacted |
No |
|
8 |
Designation of contact person |
No |
|
9 |
Promoter’s background |
Yes |
|
10 |
Date of Birth of Proprietor / Partners /
Directors |
No |
|
11 |
Pan Card No. of Proprietor / Partners |
No |
|
12 |
Voter Id Card No. of Proprietor / Partners |
No |
|
13 |
Type of business |
Yes |
|
14 |
Line of Business |
Yes |
|
15 |
Export/import details (if applicable) |
No |
|
16 |
No. of employees |
Yes |
|
17 |
Details of sister concerns |
Yes |
|
18 |
Major suppliers |
No |
|
19 |
Major customers |
No |
|
20 |
Banking Details |
Yes |
|
21 |
Banking facility details |
Yes |
|
22 |
Conduct of the banking account |
-- |
|
23 |
Financials, if provided |
Yes |
|
24 |
Capital in the business |
Yes |
|
25 |
Last accounts filed at ROC, if applicable |
Yes |
|
26 |
Turnover of firm for last three years |
Yes |
|
27 |
Reasons for variation <> 20% |
-- |
|
28 |
Estimation for coming financial year |
No |
|
29 |
Profitability for last three years |
Yes |
|
30 |
Major shareholders, if available |
Yes |
|
31 |
External Agency Rating, if available |
Yes |
|
32 |
Litigations that the firm/promoter
involved in |
Yes |
|
33 |
Market information |
-- |
|
34 |
Payments terms |
No |
|
35 |
Negative Reporting by Auditors in the
Annual Report |
No |
INDEX OF CHARGES
|
SNo |
SRN |
Charge Id |
Charge Holder Name |
Date of Creation |
Date of Modification |
Date of Satisfaction |
Amount |
Address |
|
1 |
G00928911 |
80066651 |
The Hongkong and Shanghai Banking
Corporation Limited |
17/04/1986 |
17/03/1998 |
17/03/2016 |
685000000.0 |
52/60, Mahatma
Gandhi RoadBombayBombayMH400023IN |
UNSECURED LOANS
|
PARTICULARS |
31.03.2017 (INR
In Million) |
31.03.2016 (INR
In Million) |
|
LONG-TERM BORROWINGS |
|
|
|
Deferred Payment Liability -Sales Tax Deferral Loan* |
0.700 |
1.300 |
|
|
|
|
|
SHORT TERM BORROWINGS |
|
|
|
Packing Credit from Banks |
3242.500 |
9176.300 |
|
Buyers' Credit |
0.000 |
2050.000 |
|
Bank overdraft |
0.100 |
90.500 |
|
|
|
|
|
Total |
3243.300 |
11318.100 |
|
Notes: LONG-TERM BORROWINGS *Sales tax deferral loan is interest free and repayable by 2018-19. |
||
CORPORATE
INFORMATION
Cipla Ltd.
(“the Company”) is a public company domiciled in India and is incorporated
under the provisions of the
Companies Act
applicable in India. Cipla Ltd. is a global pharmaceutical company which uses
cutting edge technology and innovation to meet the everyday needs of all
patients. The Company has its wide network of operations in local as well
foreign markets.
MANAGEMENT DISCUSSION AND ANALYSIS
GLOBAL PHARMACEUTICAL MARKET AND
MACROECONOMICS
As per a recent
IMS# report, the global medicine spending is expected to reach nearly USD 1.5
trillion by 2021. This represents a 4-7% CAGR over the next five years. Increased
spending in Oncology, Autoimmune and Diabetology treatments is expected to
drive a large part of the spending growth. the US will continue to remain the
largest pharmaceutical market with spending growth driven by originator brands.
Increased focus on developing Specialty medicines by both innovators and
generic players is expected to drive increase in the Specialty share of global
spending from 30% in 2016 to 35% in 2021.
In the US,
increasing penetration of generics and channel consolidation will lead to
significant decline in patient out-of-pocket costs despite rising costs of
brand prescriptions. With rising income levels, penetration of healthcare
services and expansion of healthcare insurance schemes, the Indian
Pharmaceutical Market is expected to grow in double digits. However, an
uncertain regulatory environment can have a considerable impact on the growth
prospects. While other emerging markets continue to grapple with challenges of
slowed economic growth and weakening currencies, Government support in
expanding healthcare infrastructure will be critical in driving volume growth.
Given the volatility in economic growth in these markets, countries are
adjusting their healthcare policies leading to significant volatility in demand.
CIPLA
Over the past
81 Years of Caring for Life, Cipla has bolstered its leadership position in the
Indian Pharmaceutical industry. The Company today is the 3rd* largest
pharmaceutical player in India. It has 43 manufacturing facilities and presence
in over 80 countries. After several steps towards a determined strategic
transformation in the past years, Cipla is now augmenting its global focus by
consolidating and deepening its presence in the priority markets of India,
North America, South Africa and key countries within the emerging markets territories.
The Company is focusing on improving the quality of revenues and earnings
through a number of initiatives. To drive sustainable growth in the future, the
Company is also investing towards building a well-balanced portfolio with focus
on differentiated and complex products. In the year ahead, the Company aims to
maintain its focus to further streamline its operations to obtain
cost-efficiencies. In Cipla’s mission to provide affordable lifesaving drugs to
the world, the Company has laid a strong foundation of a future-ready Cipla on
the back of a strong management team driven by the OneCipla Credo.
GROWTH MOMENTUM
AT CIPLA
Cipla operates
through its two business segments- Pharmaceuticals and New Ventures. During the
financial year:
The Company demonstrated
strong business momentum in India, growing by ~10%. Cipla maintained its
leadership position across key therapy areas including Respiratory, Urology and
Pediatrics. The Company continued to maintain a strong position in
Anti-infectives and Cardiology as well.
With InvaGen
and Exelan successfully integrated, the Company showcased strong commercial
execution capabilities in the US. Cipla is now ranked No.9 in TRx amongst all
generic companies as per IMS MAT March 2017. The Company is currently the
market leader (#1) in 10/45 (22%) marketed products as per IMS 2017.
Cipla continued
its dominance in South Africa by growing ~17% in local currency terms. The
Company today is the fourth largest pharmaceutical company in South Africa with
significant market share in key therapeutic areas like Respiratory, Central
Nervous System (CNS), Oncology and Musculoskeletal System. As per IMS, Cipla’s
growth in the private market for FY 2016-17 was 13.3% as compared to the overall
market growth of 7.8%.
Despite geopolitical
and currency volatilities, the Company continued to maintain its leadership position
in various emerging markets including Yemen, Sri Lanka, Morocco, Nepal and
Myanmar. The Company also launched its direct-to-market (DTM) operations in
Malaysia and Columbia during the year.
The Company
implemented a successful business turnaround in Europe from a direct-to-market
(DTM) to a partnership-based model bringing the business back on a profitable
trajectory. The Company also launched its flagship product,
Salmeterol-Fluticasone MDI (Brand Name: SerefloTM) in UK during the year. On
the regulatory front, the Company had a very successful year with EIRs received
from the US FDA for its Indore, Goa and InvaGen plants.
Cipla invested
significantly in staying ahead of the curve for its quality and R&D systems
with multiple automation and process improvement related projects. Also, during
the year, the Company expanded capacities at Sikkim, Goa, Indore and Bangalore
API facilities.
Laid the
foundation for future growth with 32 ANDA filings in the US.
During the
financial year, Cipla divested its 16.7% stake in Chase Pharmaceuticals to
Allergan in the US along with other shareholders. The Company will continue to
invest in building an in-house Specialty franchise along with exploring
partnership opportunities. Cipla launched its first innovator Breath Actuated Inhaler
(BAI) SynchroBreathe in South Africa. The product has been extremely
well-received gaining 31% market share within a short period of its launch. During
the financial year, the Company also commenced commercial production at its
newly built formulation unit at Sikkim.
FY 2017-18
OUTLOOK
The Company
achieved significant progress on its key priorities in FY 2016-17 despite a
number of challenges around an uncertain regulatory environment, volatility in emerging
markets and pricing pressure across the globe. For FY 2017-18, Cipla will
remain focused on its agenda of superior revenue growth, cost consciousness and
improving the overall margin profile of the Company. The Company expects to
taper down capital investments and continue its focus on operational
efficiencies to drive strong cash flows. The Company is aiming to ramp-up investments
in R&D and maintain the filing momentum of 20-25 ANDAs in the US. Given the
challenges related to
GST and certain
key product launches in the US market, the Company expects higher growth in the
second half of the next fiscal. Cipla will continue its focus on investing towards
building its Specialty franchise through a mix of in-house development and
in-organic opportunities.
INDIA
As one of
India’s leading pharmaceutical companies, Cipla’s commitment is to drive better
patient outcomes. For Cipla, the India business contributes 38% of the Company’s
global revenues. In FY 2016-17, the business had another year of strong
performance. Despite pricing challenges and the impact of demonetisation
Cipla’s India business grew at 10%.
Growth of
prescription business has been at par with the market. The prescription business
accounts for around 81% of the India business. Cipla enjoys a market share of
5.2% as of March 2017. As per IMS, Cipla surpassed market growth rate in
Respiratory (10.5% vs 9.8%), Urology (19.4% vs 13.7%) and GastrointestinaI
(10.4% vs 8.5%). FY 2016-17 saw development and implementation of various
innovative marketing campaigns and sales
execution
strategies.
COMPANY OVERVIEW
Cipla is a
leading global pharmaceutical company which uses cutting-edge technology and
innovation to meet the everyday needs of all patients. For over eight decades, the
Company has emerged as one of the most respected pharmaceutical companies in
India as well as in over 80 countries. The Company’s portfolio includes over
1500 products across a wide range of therapeutic categories with one uniform
global standard of quality.
Whilst
delivering a long-term sustainable business, the Company recognises its duty to
provide affordable medicines. The Company’s pioneering role in HIV/ AIDS
treatment in 2001 was recognised globally when it became the first
pharmaceutical company to offer a triple combination anti-retroviral (ARV)
therapy in Africa at less than a dollar a day, thereby ensuring access to
lifesaving medicines for millions of patients. The Company’s R&D focuses on
developing innovative products and drug delivery systems.
STANDALONE
UNAUDITED FINANCIAL RESULTS FOR THE QUARTER AND NINE MONTHS ENDED 31.12.2017
(INR IN MILLION)
|
|
Particulars |
Quarter ended |
Nine months ended |
|
|
|
|
31.12.2017 (Unaudited) |
30.09.2017 (Unaudited) |
31.12.2017 (Unaudited) |
|
1. |
Income
from operations |
|
|
|
|
|
a)
Net Sales/Income from Operations |
28924.400 |
29759.400 |
84735.400 |
|
|
b)
Other Operating Income |
775.100 |
893.300 |
2459.300 |
|
|
Total Income from
Operations |
29699.500 |
30652.700 |
87194.700 |
|
|
|
|
|
|
|
2. |
Other Income |
409.400 |
1804.200 |
2720.000 |
|
|
|
|
|
|
|
3. |
Total Income
(1+2) |
30108.900 |
32456.900 |
89914.700 |
|
|
|
|
|
|
|
4. |
Expenses |
|
|
|
|
|
a)
Cost of materials consumed |
9087.800 |
8862.600 |
25411.200 |
|
|
b)
Purchase of traded goods |
2458.500 |
2771.700 |
7945.000 |
|
|
c)
Changes in inventories of finished goods, work-in-progress and traded goods |
(1120.700) |
(238.100) |
(1765.800) |
|
|
d)
Employee benefits expense |
4360.000 |
4435.800 |
13226.700 |
|
|
e)
Finance costs |
19.000 |
51.700 |
78.300 |
|
|
f)
Depreciation, impairment and amortisation expense |
1378.800 |
1325.400 |
3966.400 |
|
|
g)
Other expenses |
8119.900 |
8066.600 |
24655.600 |
|
|
h)
Impairment of Investment |
--- |
--- |
--- |
|
|
Total Expenses |
24303.300 |
25275.700 |
73517.400 |
|
|
|
|
|
|
|
5. |
Profit
before tax (3-4) |
5805.600 |
7181.200 |
16397.300 |
|
6. |
Tax
expense |
|
|
|
|
|
a)
Current tax |
1571.400 |
1399.900 |
3685.700 |
|
|
b)
Deferred tax |
48.800 |
119.700 |
274.500 |
|
|
Total Tax expense |
1620.200 |
1519.600 |
3960.200 |
|
|
|
|
|
|
|
7. |
Net Profit after tax (5-6) |
4185.400 |
5661.600 |
12437.100 |
|
8. |
Other
Comprehensive Income/loss for the period |
|
|
|
|
|
i)
Items that will not be reclassified to statement of profit and loss |
83.200 |
34.700 |
38.300 |
|
|
ii)
Income tax on items that will not be reclassified to statement of profit and
loss |
(28.900) |
(12.000) |
(13.300) |
|
|
i)
Items that will be reclassified to statement of profit and loss |
76.400 |
20.300 |
96.700 |
|
|
ii)
Income tax on items that will be reclassified to statement of profit and loss |
(26.500) |
(7.000) |
(33.500) |
|
|
Other Comprehensive Income/loss for the
period |
104.200 |
36.000 |
88.200 |
|
|
|
|
|
|
|
9. |
Total
Comprehensive Income/loss for the period |
4289.600 |
5697.600 |
12525.300 |
|
10. |
Paid
- up Equity Share Capital (Face
value of INR 2/- each) |
1609.900 |
1609.400 |
1609.900 |
|
11. |
Other equity |
--- |
--- |
--- |
|
12. |
Earnings Per share (Face
value of INR 2/- each) |
|
|
|
|
|
a)
Basic |
*5.20 |
*7.03 |
*15.46 |
|
|
b)
Diluted |
*5.19 |
*7.02 |
*15.43 |
|
|
*Not
Annualised |
|
|
|
NOTES:
1. The result have been prepared in accordance with Indian
Accounting Standards ('Ind AS') prescribed under section 133 of the Companies
Act, 2013 read with relevant rules thereunder and In terms of Regulation 33 of
the SEBI (Listing Obligations and Disclosure Requirements) Regulations, 2015
and SEBI circular dated 5th July 2016.
2. The Government of India introduced the Goods and Service Tax (GST) with
effect from 1st July 2017 which subsumes excise duty and various other indirect
taxes. As required under Ind AS 18, revenue for the quarter ended 31st December
2017 and 30th September 2017 are reported net of GST. Sales of periods up to
quarter ending 30th June 2017 are reported inclusive of excise duty. The
revenue for nine months ended 31st December 2017 Includes excise duty up to
30th June 2017. Accordingly, income from operations for the quarter and nine
months ended 31st December 2017 are not comparable with corresponding previous
periods.
3. The Company had received notices of demand from the National Pharmaceutical
Pricing Authority (NPPA), Government of India, on account of alleged
overcharging in respect of certain drugs under the Drugs (Prices Control)
Order, 1995 ("DPCO, 1995"). These notices have been subject to challenge
by the Company on the question of fixation of retail prices without adhering to
the formula/process laid down in DPCO, 1995 and also if some of the specified
drugs be subjected to price control, based on the parameters contained in the
Drug Policy, 1994. The Company challenged these notices in the Hon’ble Bombay
High Court on the ground that bulk drugs contained in the said formulations are
not amenable to price control, based on the parameters contained in the Drug
Policy, 1994 on which the DPCO, 1995 is based and in the Hon'ble Allahabad High
Court on process followed for fixation of pricing norms. These Petitions were
decided in favour of the Company and the matters were carried in Appeal by the
Government to the Hon'ble Supreme Court of India. The Hon'ble Supreme Court of
India in August 2003 remanded the question of inclusion of certain drugs under
price control to the Hon'ble Bombay High Court, after Interpreting some of the
criteria laid down in the Drug policy for inclusion/exclusion of drugs under
price control.
In February 2013, the Hon'ble Supreme Court of India transferred the Hon'ble
Bombay High Court Petitions, also before itself for a final hearing on both the
matters. These Petitions were thereafter transferred back to Hon'ble Bombay
High Court vide Order dated 20th July 2016, along with directions that 50% of
the demands raised as mentioned in its earlier Order dated August 2003 be
deposited by the Petitioners in the Bombay Petitions, within six (6) weeks.
Accordingly, the Company deposited a sum of INR 1750.800 Million on 22nd August
2016.
The Hon'ble Supreme Court of India vide its Order and Judgment dated 21st
October 2016, allowed the Appeals filed by the Government against the Judgment
and Order of the Hon'ble Allahabad High Court regarding fixation of retail
prices. Further, the said order was specific to fixation of retail prices
without adhering to the formula/process laid down in DPCO, 1995. The grounds
relating to inclusion of certain drugs within the span of price control continues
to be sub-judice with the Hon'ble Bombay High Court. The Company has been
legally advised that it has a substantially strong case on the merits of the
matter, especially under the guidelines/principles of interpretation of the
Drug Policy enunciated by the Hon'ble Supreme Court of India. Although, the
recent decision of Hon'ble Supreme Court dated 21st October 2016 referred above
was in favour of the Government, basis the facts and legal advice on the matter
sub-judice with the Hon'ble Bombay High Court, no provision is considered
necessary in respect of the notices of demand received till date aggregating to
INR 17877.500 Million. It may be noted that NPPA in its public disclosure has
stated the total demand amount against the Company to be INR 25504.700 Million,
however, the Company has not received any further notices beyond an aggregate
amount of INR 17877.500 Million.
4. The paid-up equity share capital stands increased to INR 1609.900 Million
(804,944,690 equity shares of INR 2 each) upon allotment of 2,51,869 equity
shares of INR 2 each pursuant to “ESOS 2013-A" during the quarter ended
31st December 2017.
5. The figures of the previous year/period have been regrouped/recast to render
them comparable with the figures of the current period.
6. The above results have been subjected to Limited Review by the Statutory
Auditors, who Issued an unmodified review report, reviewed by the Audit
Committee and approved by the Board of Directors at its meeting held on 7th
February, 2018.
CONTINGENT LIABILITIES:
|
PARTICULARS |
31.03.2017 (INR
In Million) |
31.03.2016 (INR
In Million) |
|
Claims against the Company not acknowledge as debt |
3153.000 |
173.600 |
|
Guarantees* |
39567.400 |
37546.000 |
|
Letters of Credit |
267.000 |
433.700 |
|
Income Tax on account of disallowances/additions |
1002.900 |
1376.900 |
|
Excise Duty/Services Tax on account of valuation/cenvat credit |
1168.900 |
1617.400 |
|
Sales Tax on account of credit/classification |
24.300 |
23.400 |
|
|
|
|
|
TOTAL |
45183.500 |
41171.000 |
Note:
*The Company has given guarantees in favour of various banks for INR 38057.200 Million (31st March, 2016 INR 36440.300 Million, 1st April, 2015 INR Nil) against the loan granted to Cipla (EU) Ltd. and InvaGen Pharmaceuticals Inc.
FIXED ASSETS:
Tangible Assets
·
Freehold Land
·
Leasehold Land
·
Buildings and Flat
·
Plant and Machinery
·
Office Equipments
·
Furniture and Fixtures
·
Vehicles
·
Software
·
Technical Know-how
·
Trademarks
·
Brands
CMT REPORT (Corruption, Money Laundering & Terrorism]
The Public Notice information has been collected from various sources
including but not limited to: The Courts,
1] INFORMATION ON
DESIGNATED PARTY
No records exist designating subject or any of its beneficial owners,
controlling shareholders or senior officers as terrorist or terrorist
organization or whom notice had been received that all financial transactions
involving their assets have been blocked or convicted, found guilty or against
whom a judgement or order had been entered in a proceedings for violating
money-laundering, anti-corruption or bribery or international economic or
anti-terrorism sanction laws or whose assets were seized, blocked, frozen or
ordered forfeited for violation of money laundering or international
anti-terrorism laws.
2] Court Declaration :
No records exist to suggest that subject is
or was the subject of any formal or informal allegations, prosecutions or other
official proceeding for making any prohibited payments or other improper
payments to government officials for engaging in prohibited transactions or
with designated parties.
3] Asset Declaration :
No records exist to suggest that the property or assets of the subject
are derived from criminal conduct or a prohibited transaction.
4] Record on Financial
Crime :
Charges or conviction
registered against subject: None
5] Records on Violation of
Anti-Corruption Laws :
Charges or
investigation registered against subject: None
6] Records on Int’l
Anti-Money Laundering Laws/Standards :
Charges or
investigation registered against subject: None
7] Criminal Records
No
available information exist that suggest that subject or any of its principals
have been formally charged or convicted by a competent governmental authority
for any financial crime or under any formal investigation by a competent
government authority for any violation of anti-corruption laws or international
anti-money laundering laws or standard.
8] Affiliation with
Government :
No record
exists to suggest that any director or indirect owners, controlling
shareholders, director, officer or employee of the company is a government
official or a family member or close business associate of a Government
official.
9] Compensation Package :
Our market
survey revealed that the amount of compensation sought by the subject is fair
and reasonable and comparable to compensation paid to others for similar
services.
10] Press Report :
No press reports / filings exists on
the subject.
CORPORATE GOVERNANCE
MIRA INFORM as part of its Due Diligence do provide comments on
Corporate Governance to identify management and governance. These factors often
have been predictive and in some cases have created vulnerabilities to credit
deterioration.
Our Governance Assessment focuses principally on the interactions between
a company’s management, its Board of Directors, Shareholders and other
financial stakeholders.
CONTRAVENTION
Subject is not known to have contravened any existing local laws,
regulations or policies that prohibit, restrict or otherwise affect the terms
and conditions that could be included in the agreement with the subject.
FOREIGN EXCHANGE RATES
|
Currency |
Unit
|
INR |
|
US Dollar |
1 |
INR 64.87 |
|
|
1 |
INR 90.49 |
|
Euro |
1 |
INR 79.91 |
INFORMATION DETAILS
|
Information
Gathered by : |
AKY |
|
|
|
|
Analysis Done by
: |
PRS |
|
|
|
|
Report Prepared
by : |
IND |
SCORE FACTORS
|
DEMERIT POINTS |
|
|
|
--BANK CHARGES |
YES/NO |
YES |
|
--LITIGATION |
YES/NO |
YES |
|
--OTHER ADVERSE INFORMATION |
YES/NO |
NO |
|
MERIT POINTS |
|
|
|
--SOLE DISTRIBUTORSHIP |
YES/NO |
NO |
|
--EXPORT ACTIVITIES |
YES/NO |
NO |
|
--AFFILIATION |
YES/NO |
YES |
|
--LISTED |
YES/NO |
YES |
|
--OTHER MERIT FACTORS |
YES/NO |
YES |
RATING EXPLANATIONS
|
Credit Rating |
Explanation |
Rating Comments |
|
A++ |
Minimum Risk |
Business dealings permissible with minimum
risk of default |
|
A+ |
Low Risk |
Business dealings permissible with low
risk of default |
|
A |
Acceptable Risk |
Business dealings permissible with
moderate risk of default |
|
B |
Medium Risk |
Business dealings permissible on a regular
monitoring basis |
|
C |
Medium High Risk |
Business dealings permissible preferably
on secured basis |
|
D |
High Risk |
Business dealing not recommended or on
secured terms only |
|
NB |
New Business |
No recommendation can be done due to
business in infancy stage |
|
NT |
No Trace |
No recommendation can be done as the
business is not traceable |
NB is stated where there is insufficient information to facilitate rating. However, it is not to be considered as unfavourable.
This score serves as a reference to assess
SC’s credit risk and to set the amount of credit to be extended. It is
calculated from a composite of weighted scores obtained from each of the major
sections of this report. The assessed factors are as follows:
·
Financial
condition covering various ratios
·
Company
background and operations size
·
Promoters
/ Management background
·
Payment
record
·
Litigation
against the subject
·
Industry
scenario / competitor analysis
·
Supplier
/ Customer / Banker review (wherever available)
This report is issued at
your request without any risk and responsibility on the part of MIRA INFORM
PRIVATE LIMITED (MIPL) or its officials.

