|
|
|
MIRA INFORM REPORT
|
Report No. : |
499515 |
|
Report Date : |
21.03.2018 |
IDENTIFICATION DETAILS
|
Name : |
MYLAN LABORATORIES LIMITED (w.e.f. 05.10.2011) |
|
|
|
|
Formerly Known
As : |
MATRIX LABORATORIES LIMITTED (w.e.f. 21.03.2001) HERREN DRUGS AND PHARMACEUTICALS LIMITED (w.e.f. 27.06.1994) HERREN DRUGS LIMITED (w.e.f. 19.10.1992) HERREN DRUGS PRIVATE LIMITED |
|
|
|
|
Registered
Office : |
House No. 8-2-293/82/J III, Plot No. 564/A/22, Road No.92, Jubilee Hills,
Hyderabad – 500033, Telangana |
|
Tel. No.: |
91-40-30496666 |
|
|
|
|
Country : |
India |
|
|
|
|
Financials (as
on) : |
31.03.2016 |
|
|
|
|
Date of
Incorporation : |
29.11.1984 |
|
|
|
|
Com. Reg. No.: |
36-005146 |
|
|
|
|
Capital Investment
/ Paid-up Capital : |
INR 487.030 Million |
|
|
|
|
CIN No.: [Company Identification
No.] |
U24231TG1984PLC005146 |
|
|
|
|
IEC No.: [Import-Export Code No.] |
0988008858 |
|
|
|
|
GSTN : [Goods & Service Tax
Registration No.] |
36AADCM3491M1Z2 |
|
|
|
|
TIN No.: |
36370135109 |
|
|
|
|
TAN No.: [Tax Deduction &
Collection Account No.] |
Not Available |
|
|
|
|
PAN No.: [Permanent Account No.] |
AADCM3491M |
|
|
|
|
Legal Form : |
A Closely Held Public Limited Liability Company |
|
|
|
|
Line of Business
: |
Subject is engaged in the manufacture of Active
Pharmaceutical Ingredients, Finished Dosage Formulations, Injectable and
research and development activities. [Registered Activity] |
|
|
|
|
No. of Employees
: |
Information denied by the management |
RATING & COMMENTS
(Mira Inform has adopted New Rating mechanism w.e.f. 23rd
January 2017)
|
MIRA’s Rating : |
A+ |
|
Credit Rating |
Explanation |
Rating Comments |
|
A+ |
Low Risk |
Business dealings permissible with low
risk of default |
|
Status : |
Excellent |
|
|
|
|
Payment Behaviour : |
Regular |
|
|
|
|
Litigation : |
Exist |
|
|
|
|
Comments : |
Subject is a subsidiary of “MP Laboratories (Mauritius) Limited, Mauritius” and was incorporated in the year 1984. It is having excellent track records. As per registrar of companies the date of balance sheet (i.e. financial filed) is shown as 31.03.2017 but the documents related to the financial for the year 2017 are not available from any sources. For the financial year 2016, the company has increased its revenue from operation as compared to previous year but reported losses. However, rating takes into consideration strong financial profile of the company marked by healthy networth base and average debt balance sheet. Further, the company also derives strength from its strong holding company support, established market position along with presence in various therapeutic segments and geographical diversity. However, these rating strengths are partially offset by working capital-intensive operations and exposure to increasing regulatory scrutiny and competition in the global generics market. Trade relations are reported as fair. Payments are reported to be regular. In view of established market position, the company can be considered
good for normal business dealings at usual trade terms and conditions. |
NOTES:
Any query related to this report can be made
on e-mail: infodept@mirainform.com
while quoting report number, name and date.
ECGC Country Risk Classification List
|
Country Name |
Previous Rating (30.09.2017) |
Current Rating (31.12.2017) |
|
India |
A1 |
A1 |
|
Risk Category |
ECGC
Classification |
|
Insignificant |
A1 |
|
Low Risk |
A2 |
|
Moderately Low Risk |
B1 |
|
Moderate Risk |
B2 |
|
Moderately High Risk |
C1 |
|
High Risk |
C2 |
|
Very High Risk |
D |
EXTERNAL AGENCY RATING
|
Rating Agency Name |
CRISIL |
|
Rating |
Long Term Rating= AA- |
|
Rating Explanation |
High degree of safety and low credit risk. |
|
Date |
31.03.2017 |
RBI DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available RBI Defaulters’ list.
EPF (Employee Provident Fund) DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available EPF (Employee Provident Fund) Defaulters’ list as of
31-03-2016.
BIFR (Board for Industrial & Financial Reconstruction) LISTING STATUS
Subject’s name is not listed as a Sick Unit in
the publicly available BIFR (Board for Industrial & Financial
Reconstruction) list as of 21.03.2018
IBBI (Insolvency and Bankruptcy Board of India) LISTING STATUS
Subject’s name is not listed in the publicly
available IBBI (Insolvency and Bankruptcy Board of India) list as of report
date.
INFORMATION DENIED BY
|
Name : |
Not Divulged |
|
Designation : |
Accounts Department |
|
Contact No.: |
91-80-66278000 |
LOCATIONS
|
Registered Office : |
House No. 8-2-293/82/J III, Plot No.564/A/22, Road No.92, Jubilee
Hills, Hyderabad – 500033, Telangana, India |
|
Tel. No.: |
91-40-30496666 / 23550543 |
|
Fax No.: |
91-40-27700343 / 30866699 |
|
E-Mail : |
|
|
Website : |
|
|
|
|
|
Corporate Office : |
F-4 and F-12, MIDC Malegaon, Sinnar, Nashik – 422113,
Maharashtra, India |
|
|
|
|
Branch Office 1 : |
7th Floor, Tower 2B, Indiabulls Centre, 841, Senapati Bapat Marg, Elphinstone Road (West), Mumbai – 400021, Maharashtra, India |
|
|
|
|
Branch Office 2 : |
Nirmal, 20th Floor, Nariman Point, Mumbai – 400021, Maharashtra, India |
|
|
|
|
Factories : |
Located at : ·
Hyderabad (4 Factories) ·
Maharashtra
(2 Factories) ·
Aurangabad (1 Factory) ·
Indore (1 Factory) ·
Andhra Pradesh (2 Factories) |
DIRECTORS
AS ON 31.03.2017
|
Name : |
Mr. Rakesh Bamzai |
||||||||||||||||||||||||||||||||||||
|
Designation : |
Managing Director |
||||||||||||||||||||||||||||||||||||
|
Address : |
No.79/12, Tuisi Sunny Brooks, Sarjapur Road, Doddakannahalli,
Bangalore – 560035, Karnataka, India |
||||||||||||||||||||||||||||||||||||
|
Date of Birth/Age : |
01.10.1965 |
||||||||||||||||||||||||||||||||||||
|
Qualification : |
B.Sc (Tech) |
||||||||||||||||||||||||||||||||||||
|
Date of Appointment : |
01.11.2014 |
||||||||||||||||||||||||||||||||||||
|
PAN No.: |
AANPB2957J |
||||||||||||||||||||||||||||||||||||
|
DIN No.: |
06997113 |
||||||||||||||||||||||||||||||||||||
|
Other Directorship:
|
|||||||||||||||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||||||||||||||
|
Name : |
Ms. Sarada Kalyani Bhagawati |
||||||||||||||||||||||||||||||||||||
|
Designation : |
Whole-Time Director |
||||||||||||||||||||||||||||||||||||
|
Address : |
H No. 1-1-380 / 38, Ashok Nagar Extension, Hyderabad – 500020,
Telangana, India |
||||||||||||||||||||||||||||||||||||
|
Date of Birth/Age : |
29.09.1968 |
||||||||||||||||||||||||||||||||||||
|
Qualification : |
CA, ICWA |
||||||||||||||||||||||||||||||||||||
|
Date of Appointment : |
01.11.2014 |
||||||||||||||||||||||||||||||||||||
|
PAN No.: |
ACXPB3665R |
||||||||||||||||||||||||||||||||||||
|
DIN No.: |
02680074 |
||||||||||||||||||||||||||||||||||||
|
Other Directorship:
|
|||||||||||||||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||||||||||||||
|
Name : |
Mr. Rajiv Krishan Luthra |
||||||||||||||||||||||||||||||||||||
|
Designation : |
Director |
||||||||||||||||||||||||||||||||||||
|
Address : |
3/15, Shanti Niketan, New Delhi – 110021, India |
||||||||||||||||||||||||||||||||||||
|
Date of Birth/Age : |
01.08.1957 |
||||||||||||||||||||||||||||||||||||
|
Qualification : |
LL.M |
||||||||||||||||||||||||||||||||||||
|
Date of Appointment : |
14.08.2015 |
||||||||||||||||||||||||||||||||||||
|
PAN No.: |
AAAPL9458K |
||||||||||||||||||||||||||||||||||||
|
DIN No.: |
00022285 |
||||||||||||||||||||||||||||||||||||
|
Other Directorship:
|
|||||||||||||||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||||||||||||||
|
Name : |
Mr. Mohit Saraf |
||||||||||||||||||||||||||||||||||||
|
Designation : |
Director |
||||||||||||||||||||||||||||||||||||
|
Address : |
Farm No 2, Kapashera Village, Farm House, Delhi – 110037, India |
||||||||||||||||||||||||||||||||||||
|
Date of Birth/Age : |
19.11.1966 |
||||||||||||||||||||||||||||||||||||
|
Qualification : |
LL.M |
||||||||||||||||||||||||||||||||||||
|
Date of Appointment : |
14.08.2015 |
||||||||||||||||||||||||||||||||||||
|
PAN No.: |
AKAPS4446E |
||||||||||||||||||||||||||||||||||||
|
DIN No.: |
00057284 |
||||||||||||||||||||||||||||||||||||
|
Other Directorship:
|
|||||||||||||||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||||||||||||||
|
Name : |
Mr. Malik Rajiv |
||||||||||||||||||||||||||||||||||||
|
Designation : |
Director |
||||||||||||||||||||||||||||||||||||
|
Address : |
B-6B, Gangotri Enclave, Alaknanda, New Delhi – 110019, India |
||||||||||||||||||||||||||||||||||||
|
Date of Birth/Age : |
12.03.1961 |
||||||||||||||||||||||||||||||||||||
|
Qualification : |
M. Pharm (Degree in Pharmaceutical Industry) |
||||||||||||||||||||||||||||||||||||
|
Date of Appointment : |
01.07.2008 |
||||||||||||||||||||||||||||||||||||
|
PAN No.: |
ADHPM8833H |
||||||||||||||||||||||||||||||||||||
|
DIN No.: |
00120557 |
||||||||||||||||||||||||||||||||||||
KEY EXECUTIVES
|
Name : |
Mr. Sarada Kalyani Bhagawati |
|
Designation : |
Chief Financial Officer |
|
Address : |
H. No. 1-1-380/38, Ashok Nagar Extension, Hyderabad – 500020,
Telangana, India |
|
Date of Birth/Age : |
29.09.1968 |
|
Qualification : |
CA,ICWA |
|
Date of Appointment : |
01.11.2014 |
|
PAN No.: |
ACXPB3665R |
|
|
|
|
Name : |
Mr. Rakesh Bamzai |
|
Designation : |
Chief Executive Officer |
|
Address : |
No. 79/12, Tulsi Sunny Brooks, Sarjapur Road, Doddakannahalli, Bangalore – 560035, Karnataka, India |
|
Date of Birth/Age : |
01.10.1965 |
|
Qualification : |
B.Sc (Tech) |
|
Date of Appointment : |
01.11.2014 |
|
PAN No.: |
AANPB2957J |
|
|
|
|
Name : |
Mr. Nagaraj Goud Bodige |
|
Designation : |
Company Secretary |
|
Address : |
H. No. 5-14-88, Indiranagar Colony II, APHB Colony,
Movlali, Hyderabad – 500040, Telangana, India |
|
Date of Birth/Age : |
02.12.1977 |
|
Qualification : |
Company Secretary, LL.B |
|
Date of Appointment : |
25.07.2011 |
|
PAN No.: |
AGYPB3840P |
MAJOR SHAREHOLDERS
AS ON 31.03.2017
|
Names of Shareholders |
|
No. of Shares |
|
MP Laboratories (Mauritius) Limited, Mauritius |
|
157402332 |
|
Mylan Luxembourg 2 S.a.r.l |
|
28508547 |
|
Mylan Group B.V. |
|
57601744 |
|
B. Hari Babu |
|
2 |
|
Sarada Kalyani Bhagawati |
|
2 |
|
Yasir Rawjee jointly held with Mylan Luxembourg 2 S.a.r.l. |
|
2 |
|
Rajeev Mukundan |
|
2 |
|
Susanto Banerjee |
|
2 |
|
B. Nagaraj Goud |
|
2 |
|
|
|
|
|
Total |
|
243512635 |
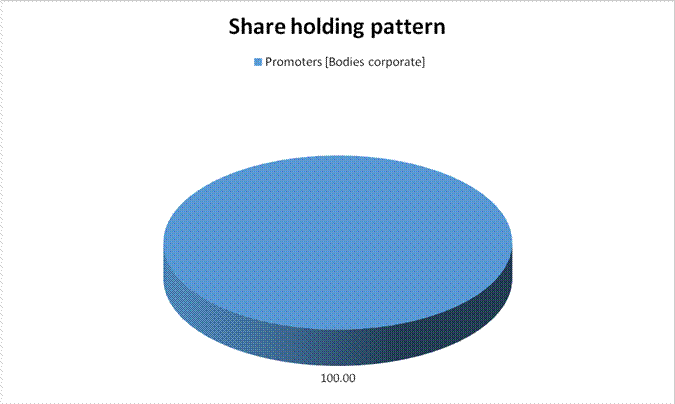
Equity Share Break up (Percentage of Total Equity)
AS ON 22.09.2017
|
Category |
Percentage |
|
Promoters [Bodies
corporate] |
100.00 |
|
Total |
100.00 |
BUSINESS DETAILS
|
Line of Business : |
Subject is engaged in the manufacture of Active
Pharmaceutical Ingredients, Finished Dosage Formulations, Injectable and
research and development activities. [Registered Activity] |
||||||||||||
|
|
|
||||||||||||
|
Products / Services
: |
|
||||||||||||
|
|
|
||||||||||||
|
Brand Names : |
Not Available |
||||||||||||
|
|
|
||||||||||||
|
Agencies Held : |
Not Available |
||||||||||||
|
|
|
||||||||||||
|
Exports : |
Not Divulged |
||||||||||||
|
|
|
||||||||||||
|
Imports : |
Not Divulged |
||||||||||||
|
|
|
||||||||||||
|
Terms : |
Not Divulged |
PRODUCTION STATUS – (NOT AVAILABLE)
GENERAL INFORMATION
|
Suppliers : |
|
||||||||||||||||||||||
|
|
|
||||||||||||||||||||||
|
Customers : |
|
||||||||||||||||||||||
|
|
|
||||||||||||||||||||||
|
No. of Employees : |
Information denied by the management |
||||||||||||||||||||||
|
|
|
||||||||||||||||||||||
|
Bankers : |
|
||||||||||||||||||||||
|
|
|
||||||||||||||||||||||
|
Facilities : |
|
|
Auditors : |
|
|
Name : |
Deloitte Haskins and Sells Chartered Accountants |
|
Address : |
Gowra Grand, III Floor, 1-8-1-384 and 385, SP Road, Secunderabad –
500003, Telangana, India |
|
PAN No.: |
AACFD3771D |
|
|
|
|
Memberships : |
Not Available |
|
|
|
|
Collaborators : |
Not Available |
|
|
|
|
Holding Company : |
· MP Laboratories (Mauritius) Limited |
|
|
|
|
Ultimate Holding Company: |
· Mylan Inc., USA (upto 26th Feb 2015) · Mylan N.V., Netherlands (w.e.f. 27th Feb 2015) |
|
|
|
|
Subsidiary Company : |
· Famy Care Europe Limited (w.e.f. 21st Nov 2015) |
|
|
|
|
Fellow Subsidiary Companies : |
· Mylan Pharmaceuticals Inc. · Mylan Pharmaceuticals ULC · Mylan Seiyaku Limited · Gerard Laboratories Limited · Alphapharm Pty. Limited · Mylan Pharmaceuticals Private Limited · Mylan Pharma UK Limited · Mylan Technologies, Inc. · Mylan S.A.S. · Mylan New Zealand Limited · Xixia Pharmaceuticals (Pty) Limited · Mylan Ireland Limited · Mylan S.p.A · Mylan Institutional Inc. · Mylan Laboratories Inc. · Mylan Luxembourg 2 S.a.r.l · Mylan Luxembourg 8 S.a.r.l · Agila Specialties Polska sp. Z.o.o · Farma Plus AS · Sagent Agila LLC · Agila Specialties Global Pte. Limited · Agila Farmaceutica Participacoes Ltda · Onco Laboratories Limited · Agila Specialties Inc. · Mylan Ireland Holdings Limited · Mylan Foundation, India · Mylan (Pty) Limited · Mylan Teoranta · Mylan Inc., USA (w.e.f 27th Feb 2015) · Agila Especialidades Farmaceutica Ltda. · Mylan Brasil Distribuidora · Mylan Group B.V. · Mylan Luxembourg S.A.R.L · Mylan Taiwan Limited · Mylan FZ LLC · Mylan B.V · Mylan Laboratorios Ltda. |
CAPITAL STRUCTURE
AS ON 22.09.2017
Authorised Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
350000000 |
Equity Shares |
INR 2/- each |
INR 700.000 Million |
|
|
|
|
|
Issued, Subscribed & Paid-up Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
243512635 |
Equity Shares |
INR 2/- each |
INR 487.025 Million |
|
|
|
|
|
FINANCIAL DATA
[all figures are
in INR Million]
ABRIDGED
BALANCE SHEET
|
SOURCES OF FUNDS |
31.03.2016 |
31.03.2015 |
31.03.2014 |
|
I.
EQUITY
AND LIABILITIES |
|
|
|
|
(1)Shareholders' Funds |
|
|
|
|
(a) Share Capital |
487.030 |
378.560 |
369.510 |
|
(b) Reserves and Surplus |
67766.300 |
45010.120 |
41421.410 |
|
(c) Money
received against share warrants |
0.000 |
0.000 |
0.000 |
|
|
|
|
|
|
(2) Share Application money pending
allotment |
0.000 |
1742.130 |
0.000 |
|
Total
Shareholders’ Funds (1) + (2) |
68253.330 |
47130.810 |
41790.920 |
|
|
|
|
|
|
(3)
Non-Current Liabilities |
|
|
|
|
(a) long-term borrowings |
101979.960 |
67782.740 |
60339.510 |
|
(b) Deferred tax liabilities (Net) |
1537.050 |
3429.760 |
4010.940 |
|
(c) Other long-term
liabilities |
5341.700 |
1745.640 |
0.000 |
|
(d) long-term
provisions |
427.540 |
354.440 |
221.290 |
|
Total Non-current
Liabilities (3) |
109286.250 |
73312.580 |
64571.740 |
|
|
|
|
|
|
(4)
Current Liabilities |
|
|
|
|
(a) Short-term
borrowings |
4405.640 |
1102.410 |
5399.010 |
|
(b) Trade
payables |
18153.910 |
16371.760 |
16111.710 |
|
(c) Other
current liabilities |
10501.290 |
9932.830 |
15591.660 |
|
(d) Short-term
provisions |
463.080 |
318.960 |
428.300 |
|
Total Current
Liabilities (4) |
33523.920 |
27725.960 |
37530.680 |
|
|
|
|
|
|
TOTAL |
211063.500 |
148169.350 |
143893.340 |
|
|
|
|
|
|
II.
ASSETS |
|
|
|
|
(1) Non-current
assets |
|
|
|
|
(a) Fixed
Assets |
|
|
|
|
(i)
Tangible assets |
34811.900 |
30990.220 |
26772.180 |
|
(ii)
Intangible Assets |
73278.470 |
29109.470 |
35426.620 |
|
(iii) Tangible assets
capital work-in-progress |
2654.710 |
1766.270 |
2562.500 |
|
(iv)
Intangible assets under development |
5392.000 |
7646.000 |
7646.000 |
|
(b) Non-current Investments |
2.330 |
2.250 |
24.880 |
|
(c) Deferred tax assets (net) |
0.000 |
0.000 |
0.000 |
|
(d)
Long-term
loans and advances |
6553.500 |
5536.540 |
3661.240 |
|
(e) Other
Non-current assets |
0.000 |
0.000 |
0.000 |
|
Total Non-Current
Assets |
122692.910 |
75050.750 |
76093.420 |
|
|
|
|
|
|
(2)
Current assets |
|
|
|
|
(a)
Current investments |
0.000 |
1627.900 |
1539.150 |
|
(b) Inventories |
37593.250 |
31571.150 |
24704.120 |
|
(c) Trade
receivables |
31263.340 |
21396.870 |
22549.750 |
|
(d) Cash and bank
balances |
3837.440 |
1139.230 |
3183.850 |
|
(e)
Short-term loans and advances |
9601.030 |
8306.380 |
5365.500 |
|
(f) Other current
assets |
6075.530 |
9077.070 |
10457.550 |
|
Total
Current Assets |
88370.590 |
73118.600 |
67799.920 |
|
|
|
|
|
|
TOTAL |
211063.500 |
148169.350 |
143893.340 |
PROFIT
& LOSS ACCOUNT
|
|
PARTICULARS |
31.03.2016 |
31.03.2015 |
31.03.2014 |
|
|
SALES |
|
|
|
|
|
Total
Revenue from operations |
94672.820 |
78571.570 |
69783.950 |
|
|
Other Income |
3499.840 |
4953.380 |
2495.730 |
|
|
TOTAL |
98172.660 |
83524.950 |
72279.680 |
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
Cost of Materials
Consumed |
51030.800 |
43855.570 |
39270.100 |
|
|
Purchases of
Stock-in-Trade |
506.140 |
574.120 |
972.540 |
|
|
Changes in inventories of
finished goods, work-in-progress and Stock-in-Trade |
(4122.210) |
(4586.190) |
(3185.570) |
|
|
Employee benefit expense |
8099.680 |
6452.050 |
4058.510 |
|
|
Other expenses |
26649.880 |
22350.570 |
16935.600 |
|
|
Exceptional items before tax |
0.000 |
0.000 |
(2983.770) |
|
|
TOTAL |
82164.290 |
68646.120 |
55067.410 |
|
|
|
|
|
|
|
|
PROFIT/
(LOSS) BEFORE INTEREST, TAX,
DEPRECIATION AND AMORTISATION |
16008.370 |
14878.830 |
17212.270 |
|
|
|
|
|
|
|
Less |
FINANCIAL
EXPENSES |
7164.590 |
5686.310 |
2484.620 |
|
|
|
|
|
|
|
|
PROFIT / (LOSS) BEFORE TAX, DEPRECIATION AND AMORTISATION |
8843.780 |
9192.520 |
14727.650 |
|
|
|
|
|
|
|
Less |
DEPRECIATION/
AMORTISATION |
11908.010 |
8518.950 |
3461.790 |
|
|
|
|
|
|
|
|
PROFIT/ (LOSS)
BEFORE TAX |
(3064.230) |
673.570 |
11265.860 |
|
|
|
|
|
|
|
Less |
TAX (H) |
(1716.720) |
(669.260) |
2816.780 |
|
|
|
|
|
|
|
|
PROFIT/ (LOSS)
AFTER TAX |
(1347.510) |
1342.830 |
8449.080 |
|
|
|
|
|
|
|
|
EARNINGS IN FOREIGN
CURRENCY |
|
|
|
|
|
F.O.B. Value of Exports |
79607.150 |
64964.530 |
55052.740 |
|
|
Other
Earning |
3673.490 |
4114.310 |
4301.790 |
|
|
TOTAL EARNINGS |
83280.640 |
69078.840 |
59354.530 |
|
|
|
|
|
|
|
|
IMPORTS |
|
|
|
|
|
Raw Materials |
25622.620 |
23807.860 |
18136.720 |
|
|
Chemicals/ Consumables |
864.490 |
651.200 |
694.650 |
|
|
Capital Goods |
2281.360 |
1257.760 |
1110.590 |
|
|
TOTAL IMPORTS |
28768.470 |
25716.820 |
19941.960 |
|
|
|
|
|
|
|
|
Earnings / (Loss) Per
Share (INR) |
(6.49) |
7.09 |
33.02 |
CURRENT MATURITIES
OF LONG TERM DEBT DETAILS
|
Particulars |
31.03.2016 |
31.03.2015 |
31.03.2014 |
|
Current Maturities of Long term debt |
3113.630 |
852.560 |
529.360 |
|
Net cash flows from (used in) operations |
2805.100 |
7449.420 |
6733.470 |
|
Net cash flows from (used in) operating activities |
2561.760 |
6791.590 |
3844.740 |
KEY
RATIOS
EFFICIENCY RATIOS
|
PARTICULARS |
31.03.2016 |
31.03.2015 |
31.03.2014 |
|
Average Collection Days (Sundry
Debtors / Income * 365 Days) |
120.53 |
99.40 |
117.94 |
|
|
|
|
|
|
Account Receivables Turnover (Income / Sundry Debtors) |
3.03 |
3.67 |
3.09 |
|
|
|
|
|
|
Average Payment Days (Sundry Creditors / Purchases * 365 Days) |
128.57 |
134.50 |
146.13 |
|
|
|
|
|
|
Inventory Turnover (Operating Income / Inventories) |
0.43 |
0.47 |
0.70 |
|
|
|
|
|
|
Asset Turnover (Operating Income / Net Fixed Assets) |
0.14 |
0.21 |
0.24 |
LEVERAGE RATIOS
|
PARTICULARS |
31.03.2016 |
31.03.2015 |
31.03.2014 |
|
Debt Ratio ((Borrowing
+ Current Liabilities) / Total Assets) |
0.66 |
0.65 |
0.68 |
|
|
|
|
|
|
Debt Equity Ratio (Total Liability / Networth) |
1.60 |
1.48 |
1.59 |
|
|
|
|
|
|
Current Liabilities to Networth (Current Liabilities / Net Worth) |
0.49 |
0.59 |
0.90 |
|
|
|
|
|
|
Fixed Assets to Networth (Net Fixed Assets / Networth) |
1.70 |
1.47 |
1.73 |
|
|
|
|
|
|
Interest Coverage Ratio (PBIT / Financial Charges) |
2.23 |
2.62 |
6.93 |
PROFITABILITY RATIOS
|
PARTICULARS |
|
31.03.2016 |
31.03.2015 |
31.03.2014 |
|
PAT to Sales ((PAT
/ Sales) * 100) |
% |
(1.42) |
1.71 |
12.11 |
|
|
|
|
|
|
|
Return on Total Assets ((PAT / Total Assets) * 100) |
% |
(0.64) |
0.91 |
5.87 |
|
|
|
|
|
|
|
Return on Investment (ROI) ((PAT / Networth) * 100) |
% |
(1.97) |
2.85 |
20.22 |
SOLVENCY RATIOS
|
PARTICULARS |
31.03.2016 |
31.03.2015 |
31.03.2014 |
|
Current Ratio (Current
Assets / Current Liabilities) |
2.64 |
2.64 |
1.81 |
|
|
|
|
|
|
Quick Ratio ((Current Assets – Inventories) / Current
Liabilities) |
1.51 |
1.50 |
1.15 |
|
|
|
|
|
|
G-Score Ratio Financial (Networth / Total Assets) |
0.32 |
0.32 |
0.29 |
|
|
|
|
|
|
G-Score Ratio Debt (Debts / Equity Capital) |
224.83 |
184.22 |
179.34 |
|
|
|
|
|
|
G-Score Ratio Liquidity (Total Current Assets / Total Current Liabilities) |
2.64 |
2.64 |
1.81 |
Total
Liability = Short-term Debt + Long-term Debt + Current Maturities of Long-term
debts
FINANCIAL ANALYSIS
[all figures are
in INR Million]
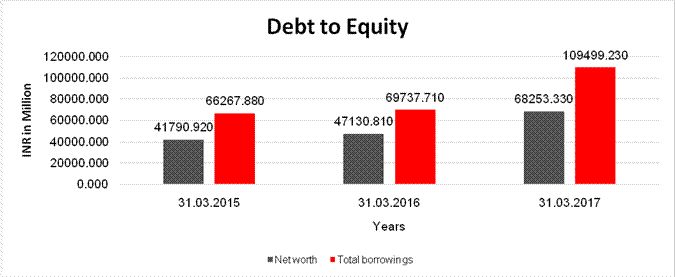
DEBT EQUITY RATIO
|
Particular |
31.03.2014 |
31.03.2015 |
31.03.2016 |
|
|
INR
In Million |
INR
In Million |
INR
In Million |
|
Share Capital |
369.510 |
378.560 |
487.030 |
|
Reserves & Surplus |
41421.410 |
45010.120 |
67766.300 |
|
Share Application money pending allotment |
0.000 |
1742.130 |
0.000 |
|
Net
worth |
41790.920 |
47130.810 |
68253.330 |
|
|
|
|
|
|
Long Term borrowings |
60339.510 |
67782.740 |
101979.960 |
|
Short Term borrowings |
5399.010 |
1102.410 |
4405.640 |
|
Current maturities of long term debt |
529.360 |
852.560 |
3113.630 |
|
Total
borrowings |
66267.880 |
69737.710 |
109499.230 |
|
Debt/Equity
ratio |
1.586 |
1.480 |
1.604 |
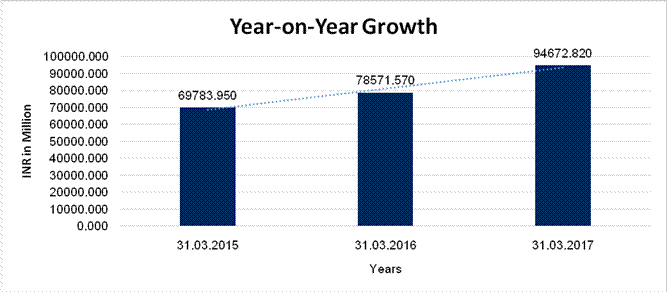
YEAR-ON-YEAR GROWTH
|
Year
on Year Growth |
31.03.2014 |
31.03.2015 |
31.03.2016 |
|
|
INR
In Million |
INR
In Million |
INR In
Million |
|
Sales
|
69783.950 |
78571.570 |
94672.820 |
|
|
|
12.593 |
20.492 |
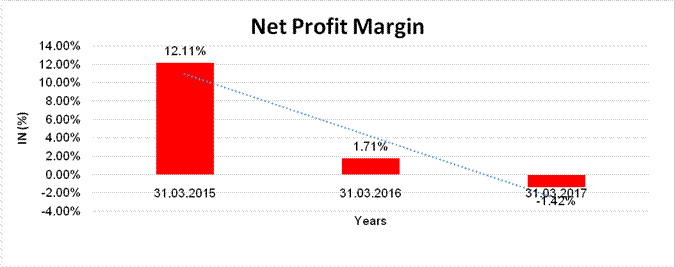
NET PROFIT MARGIN
|
Net
Profit Margin |
31.03.2014 |
31.03.2015 |
31.03.2016 |
|
|
INR
In Million |
INR
In Million |
INR
In Million |
|
Sales
|
69783.950 |
78571.570 |
94672.820 |
|
Profit/(Loss) |
8449.080 |
1342.830 |
(1347.510) |
|
|
12.11% |
1.71% |
(1.42%) |
LEGAL
CASES
|
HIGH COURT
HIGH COURT
HIGH COURT
HIGH COURT
HIGH COURT
HIGH COURT
|
LOCAL AGENCY FURTHER INFORMATION
|
Sr. No. |
Check list by
info agents |
Available in
Report (Yes/No) |
|
1 |
Year of establishment |
Yes |
|
2 |
Constitution of the entity -Incorporation
details |
Yes |
|
3 |
Locality of the entity |
Yes |
|
4 |
Premises details |
No |
|
5 |
Buyer visit details |
-- |
|
6 |
Contact numbers |
Yes |
|
7 |
Name of the person contacted |
No |
|
8 |
Designation of contact person |
Yes |
|
9 |
Promoter’s background |
Yes |
|
10 |
Date of Birth of Proprietor / Partners /
Directors |
Yes |
|
11 |
Pan Card No. of Proprietor / Partners |
Yes |
|
12 |
Voter Id Card No. of Proprietor / Partners |
No |
|
13 |
Type of business |
Yes |
|
14 |
Line of Business |
Yes |
|
15 |
Export/import details (if applicable) |
No |
|
16 |
No. of employees |
No |
|
17 |
Details of sister concerns |
Yes |
|
18 |
Major suppliers |
No |
|
19 |
Major customers |
No |
|
20 |
Banking Details |
Yes |
|
21 |
Banking facility details |
Yes |
|
22 |
Conduct of the banking account |
-- |
|
23 |
Financials, if provided |
Yes |
|
24 |
Capital in the business |
Yes |
|
25 |
Last accounts filed at ROC, if applicable |
No |
|
26 |
Turnover of firm for last three years |
Yes |
|
27 |
Reasons for variation <> 20% |
-- |
|
28 |
Estimation for coming financial year |
No |
|
29 |
Profitability for last three years |
Yes |
|
30 |
Major shareholders, if available |
Yes |
|
31 |
External Agency Rating, if available |
Yes |
|
32 |
Litigations that the firm/promoter
involved in |
-- |
|
33 |
Market information |
-- |
|
34 |
Payments terms |
No |
|
35 |
Negative Reporting by Auditors in the
Annual Report |
No |
NOTE: Registered office of the company has been shifted from 1-1-151/1,Sairam Towers,4th Floor, Alexander Road, Secunderabad – 500003, Telangana, India to the present address w.e.f. 15.03.2011
CORPORATE OVERVIEW
The company (formerly, Matrix Laboratories Limited), is engaged in the manufacture of Active Pharmaceutical Ingredients, Finished Dosage Formulations, Injectable and research and development activities. The Company is a subsidiary of MP Laboratories (Mauritius) Limited and the ultimate holding company is Mylan N.V., Netherlands. The Company has manufacturing locations spread across India.
REVIEW OF OPERATIONS
During the year, the turnover increased by 19% over the previous year. The increase in sales was mainly due to the increase in the sales of Active Pharmaceutical Ingredients (APIs) and Finished Dosage Form (FDF) products. The profit before interest, depreciation and tax for the year also showed a growth of 7.6 % over the previous year. The net loss was INR 1347.510 million as against the net profit of INR 1342.830 million over the previous year. The net loss was due to loss from injectable operations, amortization of goodwill / intangibles and interest on debentures.
During the year, the Company has raised funds through a mix of equity and debt by issue of 57601744 equity shares of INR 2/- each to Mylan Group B.V., and issue of 248034 Bonds, having a face value of INR 100000/- each to Mylan Luxembourg S.A.R.L., on private placement basis, for acquisition of the business of Jai Pharma Limited from Famy Care Limited.
The Company is in compliance with the regulations relating to downstream investment as laid down in the Foreign Exchange Management Regulations for the financial year 2015-2016 and the same is certified by the statutory auditors of the Company.
During the year there, the Company filed 12 US Drug Master Files (DMFs) and 7 EU DMFs / Certificates of Suitability to European Pharmacopoeia (CEPs) in Active pharmaceutical ingredient segment. With these filings, as on March 31, 2016, the cumulative number of DMFs filed by the Company, together with its subsidiaries and associates is 213 U.S. DMFs and 133 EU DMFs / CEPs.
During the year in Oral Solid Dosage segment, the Company has filed 10 ANDAs with the USFDA, 9 with European regulatory agencies, 2 with the WHO, 5 with Canada regulatory agencies, 9 with Japan PMDA, 7 with Australian regulatory agencies, 6 with New Zealand regulatory agencies and 13 with South Africa regulatory agencies. Aggregate filings covering Finished Dosage Forms during the year were 61 in number.
Cumulatively, the Company made the filings of 194 ANDAs with the USFDA, 133 regulatory filings with European regulatory agencies, 58 filings with the WHO, 77 with Canadian regulatory agencies, 71 with Australian regulatory agencies, 15 with Japan regulatory agencies, 47 with New Zealand, and 111 with South Africa regulatory agencies aggregating to 706 regulatory submissions. During the year their, the Company secured 14 approvals from the USA regulatory agencies, 3 approvals from the Australia regulatory agencies, 10 approvals from the Canada regulatory agencies, 12 approvals from the European regulatory agencies, 2 approvals from the Japan regulatory agencies, 1 approval from the New Zealand regulatory agencies, 3 approvals from the WHO regulatory agencies, and 1 from South Africa regulatory agencies.
In Injectable Segment, the Company has 90 approved ANDAs with the USFDA, 47 approved with Canada regulatory agencies and 18 with Africa regulatory agencies. Further, the Company has 115 pending for approval ANDAs with USFDA, 1 with Canada regulatory agencies and 63 with Africa regulatory agencies. Cumulatively, this segment has 334 regulatory submissions.
UNSECURED LOANS:
|
PARTICULARS |
31.03.2016 INR In Million |
31.03.2015 INR In Million |
|
LONG-TERM
BORROWINGS |
|
|
|
Term loans from others |
26896.490 |
0.000 |
|
Deferred payment liabilities |
13.170 |
16.540 |
|
Debentures |
0.000 |
50266.900 |
|
|
|
|
|
SHORT-TERM
BORROWINGS |
|
|
|
Loans repayable on demand |
1416.980 |
477.430 |
|
Other loans and advances |
2988.660 |
624.980 |
|
|
|
|
|
Total |
31315.300 |
51385.850 |
INDEX OF CHARGES:
|
S No |
SRN |
Charge Id |
Charge Holder Name |
Date of Creation |
Date of Modification |
Date of Satisfaction |
Amount |
Address |
|
1 |
C36138147 |
90128817 |
ANDHRA BANK |
12/04/2005 |
06/07/2007 |
11/12/2014 |
186000000.0 |
R P. ROADSECUNDERABADSECUNDERABADAP500003IN |
|
2 |
C15233083 |
10038930 |
HDFC BANK LIMITED |
19/02/2007 |
20/03/2009 |
08/08/2014 |
450000000.0 |
HDFC BANK HOUSESENAPATI BAPAT MARGLOWER PAREL WMUMBAIMH400013IN |
|
3 |
C14790752 |
10015348 |
ABN AMRO BANK |
09/07/2006 |
20/03/2009 |
06/08/2014 |
450000000.0 |
6-3-24/1/1/A, ROAD NO. 1BANJARA HILLSHYDERABADAP500034IN |
|
4 |
C14053219 |
10179307 |
INDUSIND BANK LTD. |
21/07/2009 |
- |
31/07/2014 |
300000000.0 |
2401 GEN THIMMAYYA ROADCONTONMENTPUNEMH411001IN |
|
5 |
C14362313 |
10073046 |
BANK OF NOVA SCOTIA |
21/09/2007 |
29/12/2008 |
30/07/2014 |
400000000.0 |
6-3-341/1, ROAD NO. 6BANJARA HILLSHYDERABADAP500034IN |
|
6 |
C14362636 |
10155499 |
THE BANK OF NOVA SCOTIA |
20/03/2009 |
- |
30/07/2014 |
400000000.0 |
6-3-346/1, ROAD NO. 1BANJARA HILSHYDERABADAP500034IN |
|
7 |
C13672852 |
90130616 |
HDFC BANK LIMITED |
15/01/2005 |
08/06/2007 |
23/07/2014 |
434000000.0 |
HDFC BANK HOUSESENAPATI BAPAT MARGLOWER PAREL WMUMBAIMH400013IN |
|
8 |
C13669296 |
90130372 |
HDFC BANK LIMITED |
01/12/2003 |
- |
23/07/2014 |
262500000.0 |
LAKDIKAPUL BRANCHSADEED PLAZAHYDERABADAPIN |
|
9 |
C12053500 |
10198487 |
THE ROYAL BANK OF SCOTLAND N. V. |
08/01/2010 |
29/07/2010 |
21/07/2014 |
470000000.0 |
74, SAKHAR BHAVAN,7TH FLOOR, NARIMAN POINT,MUMBAIMH400021IN |
|
10 |
C12007159 |
10268093 |
DBS BANK LTD |
27/01/2011 |
- |
18/07/2014 |
480000000.0 |
SALARPURIA WINDSORNO.3, (OLD NO.10), ULSOOR ROAD, WARD NO. 78BANGALOREKA560042IN |
CONTINGENT
LIABILITIES:
|
PARTICULARS |
31.03.2016 INR In Million |
31.03.2015 INR In Million |
|
Claims against company not acknowledged as debt |
6254.300 |
6173.420 |
|
Other money for which company is contingently liable |
165.820 |
151.570 |
|
|
|
|
|
Total |
6420.120 |
6324.990 |
FIXED ASSETS:
Tangible Assets:
· Freehold land
· Buildings
· Plant and equipment
· Furniture and fixtures
· Vehicles
· Office equipment
· Computer hardware
· Electrical equipment
· Lab equipment
Intangible Asset:
· Computer software
PRESS RELEASE / WEBSITE DETAILS
DELHI HIGH COURT
GIVES BIOCON, MYLAN GREEN LIGHT TO MARKET BLOCKBUSTER BIOSIMILAR FOR THREE
CANCERS
NEW DELHI: The Delhi High Court has allowed Biocon and Mylan to sell their copies of Swiss giant Roche's blockbuster cancer drug ‘trastuzumab’ to treat three types of cancers, giving the biotech majors greater access to a INR 3000.000 Million potential market.
According to an earlier court ruling, both companies could sell the drug to
treat only one type of cancer and could not use the data of Roche’s own
trastuzumab in package inserts for their brands ‘CANMab’ and ‘Hertraz’.
The court’s latest move, which allows the companies to use Roche’s product data
in their package inserts, is expected to allow Biocon and Mylan to grab more
revenue in the cancer therapy market.
A division bench comprising Justices Badar Durrez Ahmed and Sanjeev Sachdeva on
Friday passed an interim order permitting Biocon and Mylan to sell their
trastuzumab brands to treat metastatic breast cancer, early breast cancer and
metastatic gastric cancer. So far, Biocon and Mylan were marketing their
trastuzumab brands for only metastatic breast cancer.
The order supersedes all previous interim orders on the issue, said the judges. They are expected to hear the case next on March 30.
The court allowed Biocon and Mylan to market their products for the additional
cancers based on approvals granted by the Drug Controller General of India
(DCGI), India’s apex drug regulator, to manufacture, sell and market their
brands as ‘biosimilar’ to Roche’s own trastuzumab brands. Government’s counsel
Additional Solicitor General (ASG) Sanjay Jain told the court that DCGI had
further approved package inserts that would allow the companies to sell CANMab
and Hertraz to treat all three cancers.
A ‘biosimilar’ is a copy of a complex drug made of living cells.
Roche, which invented trastuzumab, sells it under the brand names ‘Herclon’ and
‘Biceltis’ in India and as Herceptin globally.
The judges also raised questions over Roche’s lawsuit against the companies and
DCGI.
The interim order was passed after counsels of Roche, Biocon and Mylan debated
the use of Roche’s clinical trial data on the safety and efficacy of
trastuzumab to treat early breast cancer and metastatic gastric cancer. Roche’s
counsels argued that the data were being used by Biocon and Mylan to pass their
own brands off as equally safe and effective without the companies doing their
own clinical tests.
Counsels for Biocon and Mylan, on the other hand, argued that the data used in
the companies’ latest package insert were publicly available, and not exclusive
to Roche. “The court has once again confirmed that there is no data exclusivity
in India,” Pratibha Singh, counsel for Biocon, told ET.
“This is a case that should have gone through the regulatory appellate board,
and I hope this (order) sets a precedent for future such issues,” Biocon
founder Kiran Mazumdar Shaw told ET.
"We see this as a significant positive development which will pave the way
fo greater access to affordable biosimilar trastuzumab for cancer patients in
India," stated a company spokesperson. CANMab has been developed as per
existing Indian biosimilar guidelines and was introduced in the market in 2014
after clinical studies and necessary regulatory approvals were obtained, the
spokesperson added.
Biocon’s share price on the Bombay Stock Exchange rose 3.76% to close at INR
1072.75 on Friday following the order.
“Today’s decision means critical information about the clinical studies of our
product Herceptin will be included on the packaging for these products, even
though there continues to be no evidence in the public domain that the
companies producing these products have conducted the studies required for
biosimilars,” stated Roche in an official release following the order. “We
believe it is important to ensure physicians and patients are not misled into
believing these medicines will provide the same benefit and safety experience
as our innovator medicine Herceptin.”
Roche added that it would continue to challenge companies that fail to present
data outlined in the Indian biosimilar framework.
Mylan did not respond to ET’s emailed queries by the time this report went for
publication.
Since 2014, Biocon and Mylan have been locked in a legal tussle against Roche
over their approvals to sell copies of the Swiss company’s trastuzumab
brands.
The biotech majors had approached the Delhi High Court last year to appeal an
April 25, 2016, order by Justice Manmohan Singh that ruled Biocon's CANMab and
Mylan's Hertraz could not be called "biosimilar" or ascribe any
biosimilarity to Herceptin, Herclon or Biceltis.
The order had restrained the biosimilar makers from using data relating to the
manufacturing process, safety, efficacy and tests until a final decision is
made on the issue of their biosimilarity.
The order had also called into question the processes followed by the Indian
drug controller to approve the drugs as biosimilars. The division bench stayed
this order on April 28, 2016.
The appeal proceedings are still ongoing at the Delhi court.
Apart from Biocon and Mylan, Reliance Life Sciences has also
approached the court to appeal a similar order against their trastuzumab drug.
This appeal is expected to be heard on March 30.
USFDA ACCEPTS MYLAN'S
APPLICATION FOR PROPOSED BIOSIMILAR TRASTUZUMAB
NEW DELHI: Biotechnology major Biocon and pharma firm Mylan today said that the US health regulator has accepted Mylan's biologics license application (BLA) for MYL- 1401O, a proposed biosimilar trastuzumab indicated for treatment of breast cancer.
The proposed biosimilar trastuzumab is one of the six biologic products
co-developed by Mylan and Biocon for the global marketplace, the two companies
said in a statement.
Commenting on the development, Mylan President Rajiv Malik said:
"The USFDA acceptance of our BLA for proposed biosimilar trastuzumab marks
an important step toward increasing access to this treatment option for
patients in the US."
The company is committed to bringing
this product to market and look forward to working
with FDA over the next months, he added.
This is Mylan and Biocon's "first US regulatory submission through the
351(k) pathway and reinforces the strength of our collaboration to increase
access to a broad portfolio of high -quality, affordable biosimilars
worldwide," Malik said.
Mylan has exclusive commercialisation rights for the proposed
biosimilar trastuzumab in the US, Canada, Japan, Australia, New Zealand and in
the European Union and European Free Trade
Association countries, the statement said.
Biocon has co-exclusive commercialisation rights with Mylan
for the product in the rest of the world, it added.
"It is a major milestone for the Mylan and Biocon collaboration since it
is the first US regulatory submission through our joint global biosimilars
program," Biocon CEO and Joint Managing Director Arun Chandavarkar
said.
This development positions Biocon and Mylan among the first companies to be
able to address the critical need of US patients for a high-quality biosimilar
to treat certain HER2- positive breast cancers, in the near future, he
added.
Mylan and Biocon's proposed biosimilar trastuzumab is also under review by
the European Medicines Agency (EMA), the statement said.
The product is a proposed biosimilar to branded trastuzumab, which is indicated
to treat certain HER2-positive breast cancers, the companies said.
Mylan and Biocon are exclusive partners on a broad portfolio of biosimilar and insulin
products.
USFDA ACCEPTS MYLAN'S
BLA FOR PROPOSED BIOSIMILAR PEGFILGRASTIM
NEW DELHI: US health regulator USFDA has accepted pharma major Mylan's biologics licence application (BLA) for MYL-1401H, a proposed biosimilar Pegfilgrastim, Biocon and Mylan have said.
The proposed biosimilar Pegfilgrastim is one of the six biologic products
co-developed by Mylan and Biocon for the global marketplace, the two companies
said in a statement.
Mylan President Rajiv Malik said: "This is the second BLA accepted
for review by FDA as part of the Mylan and Biocon partnership within the past
two months”.
The milestone builds on the acceptance of regulatory filings for proposed
biosimilar Pegfilgrastim in Europe, Australia and Canada, he added.
"Once approved, proposed biosimilar Pegfilgrastim will complement Mylan's
broad oncology portfolio focused on expanding access to more affordable
treatments for multiple types of cancer," Malik said.
The FDA goal date set under the Biosimilar User Fee Act (BsUFA) is October 9,
2017, the statement said.
"The FDA's acceptance for review of our second BLA for
a proposed biosimilar developed by Biocon and Mylan is an outcome of our strong
R&D and manufacturing capabilities," Biocon CEO and Joint MD Arun
Chandavarkar said.
Once approved, the proposed biosimilar Pegfilgrastim will provide a high
quality alternative to branded Pegfilgrastim (Neulasta) for cancer patients
during cytotoxic chemotherapy, he added.
Mylan has exclusive commercialisation rights for the proposed biosimilar
Pegfilgrastim in the US, Canada, Japan, Australia, New Zealand and in the
European Union and European Free Trade Association countries, the statement
said.
Biocon has co-exclusive commercialisation rights with
Mylan for the product in the rest of the world, it added.
The proposed biosimilar to Neulasta is used to reduce the duration of
neutropenia and the incidence of fever associated with neutropenia in adult
patients treated with chemotherapy in certain types of cancer.
The stock of Biocon was today trading at INR 1106.90 in the afternoon trade on
BSE, up 1.12 per cent from its previous close.
LUPIN GETS FINAL
USFDA NOD FOR MYLAN'S SKIN DISEASE CREAM
NEW DELHI: Pharma
major LupinBSE -0.45 % today said it has received final approval from
the US health regulator to market its generic version of
Mylan's triamcinolone acetonide cream used for treatment of skin
disease.
The approval is for multiple strengths of 0.025 per cent, 0.1 per cent and 0.5
per cent triamcinolone acetonide cream which is used for relief from
inflammatory and pruritic manifestations of corticosteroid responsive
dermatoses, the company said in a BSE filing.
"Lupin shall commence promoting the product shortly," it added.
US FDA ACCEPTS BIOCON-MYLAN
BIOSIMILAR APPLICATION FOR PROPOSED ANTI-CANCER BIO-SIMILAR
MUMBAI: Bangalore based Biocon announced that the U.S. Food and Drug Administration (FDA) has accepted Mylan’s (Biocon’s partner for biosimilar development) Biologics License Application (BLA) for MYL-1401H, a proposed biosimilar to Neulasta® (pegfilgrastim), an anti cancer drug.
The proposed biosimilar to Neulasta is used to reduce the duration of neutropenia (low count of neutrophils, a type of white blood cells) and the incidence of fever associated with neutropenia in adult patients treated with chemotherapy in certain types of cancer.
The FDA goal date set under the Biosimilar User Fee Act (BsUFA) is Oct. 9,
2017.
“We’re proud of the FDA acceptance of our BLA for proposed biosimilar
pegfilgrastim. This is the second BLA accepted for review by FDA as part of
the Mylan and Biocon partnership within the past two months”, said
Rajiv Malik President Mylan.
The milestone builds upon the acceptance of regulatory filings for proposed
biosimilar pegfilgrastim in Europe, Australia, and Canada, once approved,
proposed biosimilar pegfilgrastim will complement Mylan’s broad
oncology portfolio focused on expanding access to more affordable
treatments for multiple types of cancer, Malik added.
Biocon said that the FDA's acceptance for review of this
biosimilar is an outcome of the company’s strong R&D and manufacturing
capabilities.
CMT REPORT (Corruption, Money Laundering & Terrorism]
The Public Notice information has been collected from various sources
including but not limited to: The Courts,
1] INFORMATION ON
DESIGNATED PARTY
No records exist designating subject or any of its beneficial owners,
controlling shareholders or senior officers as terrorist or terrorist
organization or whom notice had been received that all financial transactions
involving their assets have been blocked or convicted, found guilty or against
whom a judgement or order had been entered in a proceedings for violating money-laundering,
anti-corruption or bribery or international economic or anti-terrorism sanction
laws or whose assets were seized, blocked, frozen or ordered forfeited for
violation of money laundering or international anti-terrorism laws.
2] Court Declaration :
No records exist to suggest that subject is
or was the subject of any formal or informal allegations, prosecutions or other
official proceeding for making any prohibited payments or other improper payments
to government officials for engaging in prohibited transactions or with
designated parties.
3] Asset Declaration :
No records exist to suggest that the property or assets of the subject
are derived from criminal conduct or a prohibited transaction.
4] Record on Financial
Crime :
Charges or conviction
registered against subject: None
5] Records on Violation of
Anti-Corruption Laws :
Charges or
investigation registered against subject: None
6] Records on Int’l
Anti-Money Laundering Laws/Standards :
Charges or
investigation registered against subject: None
7] Criminal Records
No
available information exist that suggest that subject or any of its principals
have been formally charged or convicted by a competent governmental authority
for any financial crime or under any formal investigation by a competent
government authority for any violation of anti-corruption laws or international
anti-money laundering laws or standard.
8] Affiliation with
Government :
No record exists
to suggest that any director or indirect owners, controlling shareholders,
director, officer or employee of the company is a government official or a
family member or close business associate of a Government official.
9] Compensation Package :
Our market
survey revealed that the amount of compensation sought by the subject is fair
and reasonable and comparable to compensation paid to others for similar
services.
10] Press Report :
No press reports / filings exists on
the subject.
CORPORATE GOVERNANCE
MIRA INFORM as part of its Due Diligence do provide comments on
Corporate Governance to identify management and governance. These factors often
have been predictive and in some cases have created vulnerabilities to credit
deterioration.
Our Governance Assessment focuses principally on the interactions
between a company’s management, its Board of Directors, Shareholders and other
financial stakeholders.
CONTRAVENTION
Subject is not known to have contravened any existing local laws, regulations
or policies that prohibit, restrict or otherwise affect the terms and
conditions that could be included in the agreement with the subject.
FOREIGN EXCHANGE RATES
|
Currency |
Unit
|
INR |
|
US Dollar |
1 |
INR 65.22 |
|
|
1 |
INR 91.33 |
|
Euro |
1 |
INR 79.96 |
INFORMATION DETAILS
|
Information
Gathered by : |
SVA |
|
|
|
|
Analysis Done by
: |
PSD |
|
|
|
|
Report Prepared
by : |
NKT |
SCORE FACTORS
|
DEMERIT POINTS |
|
|
|
--BANK CHARGES |
YES/NO |
YES |
|
--LITIGATION |
YES/NO |
YES |
|
--OTHER ADVERSE INFORMATION |
YES/NO |
NO |
|
MERIT POINTS |
|
|
|
--SOLE DISTRIBUTORSHIP |
YES/NO |
NO |
|
--EXPORT ACTIVITIES |
YES/NO |
NO |
|
--AFFILIATION |
YES/NO |
YES |
|
--LISTED |
YES/NO |
NO |
|
--OTHER MERIT FACTORS |
YES/NO |
YES |
RATING EXPLANATIONS
|
Credit Rating |
Explanation |
Rating Comments |
|
A++ |
Minimum Risk |
Business dealings permissible with minimum
risk of default |
|
A+ |
Low Risk |
Business dealings permissible with low
risk of default |
|
A |
Acceptable Risk |
Business dealings permissible with
moderate risk of default |
|
B |
Medium Risk |
Business dealings permissible on a regular
monitoring basis |
|
C |
Medium High Risk |
Business dealings permissible preferably
on secured basis |
|
D |
High Risk |
Business dealing not recommended or on
secured terms only |
|
NB |
New Business |
No recommendation can be done due to
business in infancy stage |
|
NT |
No Trace |
No recommendation can be done as the
business is not traceable |
NB is stated where there is insufficient information to facilitate rating. However, it is not to be considered as unfavourable.
This score serves as a reference to assess
SC’s credit risk and to set the amount of credit to be extended. It is
calculated from a composite of weighted scores obtained from each of the major
sections of this report. The assessed factors are as follows:
·
Financial
condition covering various ratios
·
Company
background and operations size
·
Promoters
/ Management background
·
Payment
record
·
Litigation
against the subject
·
Industry
scenario / competitor analysis
·
Supplier
/ Customer / Banker review (wherever available)
This report is issued at
your request without any risk and responsibility on the part of MIRA INFORM
PRIVATE LIMITED (MIPL) or its officials.

