|
|
|
MIRA INFORM REPORT
|
Report No. : |
499184 |
|
Report Date : |
21.03.2018 |
IDENTIFICATION DETAILS
|
Name
: |
SUN PHARMACEUTICAL INDUSTRIES LIMITED |
|
|
|
|
Registered
Office : |
Sun Pharma Advanced Research Centre (SPARC), Tandalja,
Vadodara-390020, Gujarat |
|
Tel.
No.: |
91-265-6615500 |
|
|
|
|
Country
: |
India |
|
|
|
|
Financials
(as on) : |
31.03.2017 |
|
|
|
|
Date
of Incorporation : |
01.03.1993 |
|
|
|
|
Com.
Reg. No.: |
04-019050 |
|
|
|
|
Capital
Investment / Paid-up Capital : |
INR 2399.300 Million |
|
|
|
|
CIN
No.: [Company
Identification No.] |
L24230GJ1993PLC019050 |
|
|
|
|
GSTN
: [Goods &
Service Tax Registration No.] |
24AADCS3124K1ZJ |
|
|
|
|
TIN
No: |
24250900383 |
|
|
|
|
IEC
No.: [Import-Export
Code No.] |
0392072823 |
|
|
|
|
TAN
No.: [Tax Deduction
& Collection Account No.] |
Not Available |
|
|
|
|
PAN
No.: [Permanent
Account No.] |
AADCS3124K |
|
|
|
|
Legal
Form : |
A Public Limited Liability Company. The Company’s Shares
are Listed on the Stock Exchanges. |
|
|
|
|
Line
of Business : |
Manufacturing, Producing,
Developing and Marketing a wide range of branded and generic formulations and
Active Pharmaceutical Ingredients (APIs). [Registered Activity] |
|
|
|
|
No.
of Employees : |
17516 (Approximately) |
RATING & COMMENTS
(Mira Inform has adopted New Rating mechanism w.e.f. 23rd
January 2017)
|
MIRA’s Rating : |
A+ |
|
Credit
Rating |
Explanation |
Rating
Comments |
|
A+ |
Low Risk |
Business dealings permissible
with low risk of default |
|
Status : |
Excellent |
|
|
|
|
Payment Behaviour : |
Regular |
|
|
|
|
Litigation : |
Exists |
|
|
|
|
Comments : |
Sun Pharma is one of the fastest-growing Indian
pharmaceutical companies, with a leading position in the high-value
cardiovascular and neuro/central nervous system segments. Incorporated in the
year 1993 and is based in Vadodara Gujarat. It is well established and
reputed company having excellent track records. The company has been continuously incurring losses from
its operational activities. However, general financial of the company is
sound. Fundamentals of the company are strong and healthy. Share price are quoted high on stock exchange. (Face value
INR 1 with Share price INR 510). The rating also takes into consideration Sun Pharma’s
leading and well established position in the domestic formulations segment,
strong presence in the regulated generics market, and increasing reach in the
emerging markets and over-the- counter (OTC) segment, following Ranbaxy’s
acquisition. Directors are reported to be experienced and
knowledgeable, resourceful and respectable businessmen. Trade relations are reported as trustworthy. Business is
active. Payments are reported as regular and as per commitment. In view of long track record and extensive experience of
its promoters the company can be considered good for normal business dealings
at usual trade terms and conditions. |
NOTES :
Any query related to this report
can be made on e-mail : infodept@mirainform.com
while quoting report number, name and date.
EXTERNAL AGENCY RATING
|
Rating Agency Name |
CRISIL |
|
Rating |
Long Term Rating= (AAA) |
|
Rating Explanation |
Highest degree of safety and
carry lowest credit risk. |
|
Date |
19.03.2018 |
|
Rating Agency Name |
CRISIL |
|
Rating |
Short Term Rating=(A1+) |
|
Rating Explanation |
Very strong degree of safety and
carry lowest credit risk. |
|
Date |
19.03.2018 |
RBI DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as
a defaulter in the publicly available RBI Defaulters’ list.
RBI DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as
a defaulter in the publicly available RBI Defaulters’ list.
BIFR (Board for Industrial & Financial Reconstruction)
LISTING STATUS
Subject’s name is not listed as a Sick
Unit in the publicly available BIFR (Board for Industrial & Financial
Reconstruction) list as of 21.03.2018.
EPF (Employee Provident Fund) DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as
a defaulter in the publicly available EPF (Employee Provident Fund) Defaulters’
list as of 31-03-2016.
INFORMATION DENIED
91-265-6615500/ 6001700/ 91-2243244324 (Management non
cooperative)
91-265-6615500
(Incorrect number)
LOCATIONS
|
Registered Office : |
Sun Pharma Advanced Research Centre (SPARC), Tandalja,
Vadodara-390020, Gujarat, India |
|
Tel. No.: |
91-265-6615500/ 600/ 700 |
|
Fax No.: |
91-265-2354897 |
|
E-Mail : |
|
|
Website : |
|
|
Area
: |
10000 sq. ft. |
|
Location
: |
Owned |
|
|
|
|
Head
Office: |
ACME Plaza, Andheri Kurla
Road, Andheri (East), Mumbai – 400059, Maharashtra, India |
|
Tel. No.: |
91-22-66969696/ 42244156 |
|
Fax No.: |
91-22-28212010 |
|
|
|
|
Corporate Office : |
Sun House, CTS No. 201 B/1,
Western Express Highway, Goregaon (East), Mumbai-400063, Maharashtra,
India |
|
Tel. No.: |
91-22-43244324 |
|
Fax No.: |
91-22-43244343 |
|
|
|
|
Research Centre 1 : |
Sun Pharma Advanced Research Centre (SPARC), Akota Road,
Akota, Vadodara – 390 020, Gujarat, India |
|
|
|
|
Research Centre 2 : |
F.P.27, Part Survey No. 27, C.S. No. 1050, TPS No. 24,
Village Tandalja, District Vadodara - 390020, Gujarat, India |
|
|
|
|
Research Centre 3 : |
17-B, Mahal Industrial Estate, Mahakali Caves Road, Andheri
(East), Mumbai - 400093, Maharashtra, India |
|
|
|
|
Research Centre 4 : |
Chemistry and Discovery Research Israel, 14 Hakitor
Street, P.O. Box 10347 Haifa Bay 2624761, Israel |
|
|
|
|
Research Centre 5 : |
Village Sarhaul, Sector-18, Gurugram
– 122015, Haryana, India |
|
|
|
|
Research Centre 6 : |
Taro Pharmaceuticals Inc., 130
East Drive, Brampton, Ontario L6T 1C1, Canada |
|
|
|
|
Research Centre 7 : |
Ohm Laboratories Inc., Terminal
Road, New Brunswick, New Jersey 08901 USA |
|
|
|
|
Plants : |
|
|
|
|
|
Overseas Plant: |
|
DIRECTORS
AS ON 31.03.2017
|
Name : |
Mr. Dilip Shantilal Shanghvi |
|
Designation : |
Managing Director |
|
Address : |
801, Abhay Building, 8th Floor,
NS 9th Road,Nutan Laxmi Co-Operative Society JVPD Scheme, Vile Parle (West), Mumbai
– 400049, Maharashtra, India |
|
Date of Birth/Age : |
61 Years |
|
Date of Appointment : |
01.04.2008 |
|
Qualification : |
Mr. Dilip S. Shanghvi is a
graduate in commerce from the Kolkata University. He is the Managing Director
of the Company and Chairman & Managing Director of Sun Pharma Advanced
Research Company Limited. He is the founding partner of Sun Pharmaceutical
Industries, a firm which was later converted into Sun Pharmaceutical
Industries Limited (SPIL) in 1993. Under his leadership, SPIL has recorded an
all-round growth in business. He has extensive experience in the
pharmaceutical industry. As the promoter of SPIL, he has been actively
involved in international pharmaceutical markets, business strategy, business
development and research and development functions in the Company. |
|
Profile : |
Mr. Shanghvi was conferred with
the prestigious ‘Padma Shri’ award by the Hon’ble President of India in the year
2016. He is recipient of several awards as listed under ‘Recognition or
Awards’ in point 3 of Clause II of ‘Statement of Information provided under
Schedule V’ in the Explanatory Statement |
|
DIN No.: |
00005588 |
|
|
|
|
Name : |
Mr. Sailesh Trambaklal Desai |
|
Designation : |
Whole-time Director |
|
Address : |
1003, Building A, Shri Aditya
CHS, MHB Colony, Samarth Ramdas Road, Vile Parle (West), Opposite Juhu
Suprema SH Centre, Mumbai – 400049, Maharashtra, India |
|
Date of Birth/Age : |
62 Years |
|
Qualification : |
Mr. Sailesh T. Desai is a
science graduate from Kolkata University and is a successful entrepreneur
with more than three decades of wide industrial experience including more
than two decades in the pharmaceutical industry. |
|
Date of Appointment : |
20.09.2006 |
|
DIN No.: |
00005443 |
|
|
|
|
Name : |
Mr. Sudhir Vrundavandas Valia |
|
Designation : |
Whole-time Director |
|
Address : |
801, Alaap Building, 8th Floor 173,
Sir Balchandra Road, Dadar (East), Mumbai – 400014, Maharashtra, India |
|
Date of Appointment : |
31.01.1994 |
|
DIN No.: |
00005561 |
|
|
|
|
Name : |
Keki Minoo Mistry |
|
Designation : |
Director |
|
Address : |
Flat No. 2603, 26th Floor Vivarea,
B-Wing, S G Marg, Mahalaxmi (East), Mumbai – 400011, Maharashtra, India |
|
Date of Appointment : |
28.08.2002 |
|
DIN No.: |
00008886 |
|
|
|
|
Name : |
Mr. Ashwin Suryakant Dani |
|
Designation : |
Director |
|
Address : |
Home Villa 48, Krishna Sanghi
Path, Mumbai – 400007, Maharashtra, India |
|
Date of Appointment : |
28.01.2004 |
|
DIN No.: |
00009126 |
|
|
|
|
Name : |
Subhagmal Mohanchand Dadha |
|
Designation : |
Director |
|
Address : |
New No.250/ Old No 268, Lloyds Road,
Royapettah, Chennai – 600014, Tamilnadu, India |
|
Date of Appointment : |
29.05.1997 |
|
DIN No.: |
00087414 |
|
|
|
|
Name : |
Mr. Vivek Chaand Sehgal |
|
Designation : |
Director |
|
Address : |
Villa No. 40, Dubai Creek Villas
Dubai Creek Golf and Yacht Club, Port Saeed Deira Dubai UAE |
|
Date of Appointment : |
14.11.2017 |
|
DIN No.: |
00291126 |
|
|
|
|
Name : |
Israel Makov |
|
Designation : |
Director |
|
Address : |
20, Hanarkis Street, Karne Yosef
99797, Israel 99797 Il |
|
Date of Birth/Age : |
78 Years |
|
Qualification : |
Mr. Israel Makov holds a B.Sc.
in Agriculture and M. Sc. in Economics from the Hebrew University, Jerusalem. |
|
Date of Appointment : |
29.05.2012 |
|
DIN No.: |
05299764 |
|
|
|
|
Name : |
Ms. Rekha Sethi |
|
Designation : |
Director |
|
Address : |
30/19, East Patel Nagar, New
Delhi – 110008, India |
|
Date of Appointment : |
27.09.2014 |
|
Date of Birth/Age : |
52 Years |
|
DIN No.: |
06809515 |
|
|
|
|
Name : |
Mr. Kalyanasundaram Iyer Natesan
Subramanian |
|
Designation : |
Additional Director |
|
Address : |
25, Hill Park, Dr. A. G. Bell
Road, Malabar Hill, Mumbai – 400006, Maharashtra, India |
|
Date of Appointment : |
14.02.2017 |
|
Qualification : |
Mr. Kal is a Chemistry graduate and
a Chartered Accountant from India with 37 years of experience of which some
30 years in the pharmaceutical industry |
|
DIN No.: |
00179072 |
KEY EXECUTIVES
|
Name : |
Mr. Sunil Roshanlal Ajmera |
|
Designation : |
Company Secretary |
|
Address : |
802, Orchid, Vasant Valley,
Flimcity Road, Malad (East), Mumbai – 400097, Maharashtra, India |
|
Date of Appointment : |
01.04.2011 |
|
PAN No.: |
AACPA0366P |
|
|
|
|
Name : |
Mr. Chinnadharavaram Sundaresan
Muralidharan |
|
Designation : |
Chief Financial Officer (w.e.f.
June 19, 2017) |
|
Address : |
302, B Wing, Avalon Building,
Hiranandani Garden, Powai, Mumbai – 400076, Maharashtra, India |
|
Date of Appointment : |
19.06.2017 |
|
PAN No.: |
AEVPM8378J |
|
|
|
|
Name : |
Mr. Uday V. Baldota |
|
Designation : |
Chief Financial Officer (Upto
June 19, 2017) |
|
Address : |
X-601,Callalily Nahar Amrit
Shakhti, Chandivali, Mumbai – 400072, Maharashtra, India |
|
Date of Appointment : |
12.08.2014 |
|
PAN No.: |
ABAPB4075D |
MAJOR SHAREHOLDERS / SHAREHOLDING PATTERN
As on DECEMBER 2017
|
Category
of shareholder |
No. of fully paid up equity shares
held |
Shareholding as a % of total no. of
shares (calculated as per SCRR, 1957)As a % of |
|
|
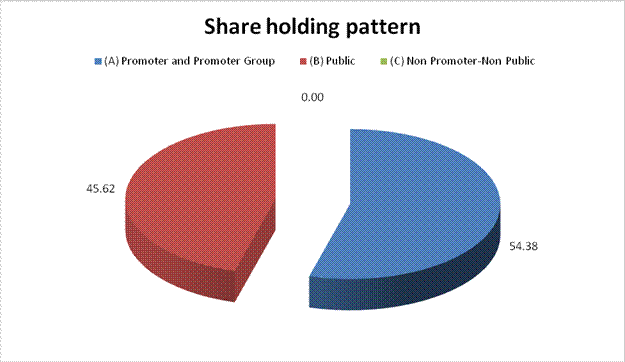
(A) Promoter and Promoter Group |
1304855381 |
54.38 |
|
|
(B) Public |
1094453965 |
45.62 |
|
|
Grand
Total |
2399309346 |
100.00 |
Statement showing shareholding pattern of the Promoter and
Promoter Group
|
Category
of shareholder |
No.
of fully paid up equity shares held |
Shareholding
as a % of total no. of shares (calculated as per SCRR, 1957)As a % of |
|
|
A1) Indian |
0.00 |
||
|
Individuals/Hindu
undivided Family |
259370161 |
10.81 |
|
|
Dilip Shantilal Shanghvi |
230285690 |
9.60 |
|
|
Sudhir Valia |
14345019 |
0.60 |
|
|
Vibha Dilip Shanghvi |
8840280 |
0.37 |
|
|
Aalok Dilip Shanghvi |
2877280 |
0.12 |
|
|
Vidhi Dilip Shanghvi |
2822427 |
0.12 |
|
|
Kumud S. Shanghvi |
199465 |
0.01 |
|
|
Any Other (specify) |
1045485220 |
43.57 |
|
|
Shanghvi Family and
Friends Benefit Trust |
1276774 |
0.05 |
|
|
Aditya Medisales Limited |
40153960 |
1.67 |
|
|
Raksha S. Valia |
33830352 |
1.41 |
|
|
Unimed Investments
Limited |
10400850 |
0.43 |
|
|
Viditi Investment Private
Limited |
200846362 |
8.37 |
|
|
Tejaskiran Pharmachem
Industries Private Limited |
194820971 |
8.12 |
|
|
Family Investment Private
Limited |
182437880 |
7.60 |
|
|
Quality Investments
Private Limited |
182379237 |
7.60 |
|
|
Virtuous Finance Private
Limited |
96851821 |
4.04 |
|
|
Virtuous Share
Investments Private Limited |
83751259 |
3.49 |
|
|
Sholapur Organics Private
Limited |
15992110 |
0.67 |
|
|
Jeevanrekha Investrade
Private Limited |
1458806 |
0.06 |
|
|
Package Investrade
Private Limited |
895039 |
0.04 |
|
|
Shanghvi Finance Private
Limited |
282603 |
0.01 |
|
|
Asawari Investment And Finance
Private Limited |
49468 |
0.00 |
|
|
Flamboyawer Finance
Private Limited |
20865 |
0.00 |
|
|
Sanghvi Properties
Private Limited |
15479 |
0.00 |
|
|
Gujarat Sun
Pharmaceutical Industries Private Limited |
14362 |
0.00 |
|
|
Nirmit Exports Private
Limited |
7022 |
0.00 |
|
|
Sub Total A1 |
1304855381 |
54.38 |
|
|
A2) Foreign |
0.00 |
||
|
A=A1+A2 |
1304855381 |
54.38 |
Statement showing shareholding pattern of the Public
shareholder
|
Category & Name of the Shareholders |
No. of fully paid up equity shares held |
Shareholding % calculated as per SCRR, 1957 As a % of (A+B+C2) |
|
|
B1) Institutions |
0 |
0.00 |
|
|
Mutual Funds/ |
175402036 |
7.31 |
|
|
ICICI Prudential Value
Discovery Fund |
63519616 |
2.65 |
|
|
Alternate Investment Funds |
1806005 |
0.08 |
|
|
Foreign Portfolio Investors |
412686542 |
17.20 |
|
|
Government of Singapore |
30037965 |
1.25 |
|
|
Financial Institutions/ Banks |
22047855 |
0.92 |
|
|
Insurance Companies |
156281920 |
6.51 |
|
|
Life Insurance Corporation of
India |
132627785 |
5.53 |
|
|
Any Other (specify) |
2254600 |
0.09 |
|
|
Foreign Bank |
25422 |
0.00 |
|
|
UTI |
2229178 |
0.09 |
|
|
Sub Total B1 |
770478958 |
32.11 |
|
|
B2) Central Government/ State
Government(s)/ President of India |
0 |
0.00 |
|
|
Central Government/ State Government(s)/
President of India |
469252 |
0.02 |
|
|
Sub Total B2 |
469252 |
0.02 |
|
|
B3) Non-Institutions |
0 |
0.00 |
|
|
Individual share capital
upto INR 0.200 Million |
153900491 |
6.41 |
|
|
Individual share capital
in excess of INR 0.200 Million |
26338232 |
1.10 |
|
|
Any Other (specify) |
143267032 |
5.97 |
|
|
Trusts |
15346588 |
0.64 |
|
|
Foreign Individuals |
20865 |
0.00 |
|
|
HUF |
4468644 |
0.19 |
|
|
Foreign Companies |
671865 |
0.03 |
|
|
NRI – Non- Repat |
2642156 |
0.11 |
|
|
Director or Director's
Relatives |
3782726 |
0.16 |
|
|
NRI – Repat |
5160842 |
0.22 |
|
|
Overseas corporate bodies |
46000 |
0.00 |
|
|
Clearing Members |
2134157 |
0.09 |
|
|
Bodies Corporate |
108993189 |
4.54 |
|
|
Lakshdeep Investments &
Finance (P) Ltd. |
35124907 |
1.46 |
|
|
Sub Total B3 |
323505755 |
13.48 |
|
|
B=B1+B2+B3 |
1094453965 |
45.62 |
BUSINESS DETAILS
|
Line
of Business : |
Manufacturing, Producing,
Developing and Marketing a wide range of branded and generic formulations and
Active Pharmaceutical Ingredients (APIs). [Registered Activity] |
||||
|
|
|
||||
|
Products : |
|
||||
|
|
|
||||
|
Brand Names : |
·
Pantocid ·
Gemer ·
Susten ·
Levipil ·
Pantocid-D ·
Aztor ·
Glucored Group ·
Istamet ·
Rozavel ·
Montek-LC |
||||
|
|
|
||||
|
Agencies Held : |
Not Available |
||||
|
|
|
||||
|
Exports : |
Not Divulged |
||||
|
|
|
||||
|
Imports : |
Not Divulged |
||||
|
|
|
||||
|
Terms : |
Not Divulged |
GENERAL INFORMATION
|
Suppliers : |
|
||||||||||||||||||
|
|
|
||||||||||||||||||
|
Customers : |
|
||||||||||||||||||
|
|
|
||||||||||||||||||
|
No. of Employees : |
17516 (Approximately) |
||||||||||||||||||
|
|
|
||||||||||||||||||
|
Bankers : |
|
||||||||||||||||||
|
|
|
||||||||||||||||||
|
Facilities
: |
|
|
Auditors 1: |
|
|
Name
: |
Deloitte Haskins and Sells Chartered Accountants |
|
Address
: |
Indiabulls Finance Centre, Tower 3, 27th -32nd
Floor, Senapati Bapat Marg, Elphinstone Road (West), Mumbai-400013,
Maharashtra, India |
|
Tel.
No.: |
91-22-61854000 |
|
Fax
No.: |
91-22-67854501/ 4601 |
|
|
|
|
Auditors 2: |
|
|
Name
: |
SRBC and Co. LLP Chartered Accountants |
|
Address
: |
14th Floor, The Ruby 29, Senapati Bapat Marg,
Dadar (East), Mumbai – 400028, Maharashtra, India |
|
Tel.
No.: |
91-22-61920000 |
|
Fax
No.: |
91-22-61921000 |
|
|
|
|
Memberships : |
Not Available |
|
|
|
|
Collaborators : |
Not Available |
|
|
|
|
Subsidiaries |
|
|
|
|
|
Step down Subsidiaries |
|
|
|
|
|
Joint Ventures |
S and I Ophthalmic LLC United
States of America |
|
|
|
|
Associates |
|
|
|
|
|
Enterprise under control of Key Managerial Personnel or their
relatives |
Makov Associates Limited |
|
|
|
|
Enterprise under significant Influence of Key Managerial Personnel or their relatives |
|
CAPITAL STRUCTURE
AS ON
31.03.2017
Authorised Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
5990000000 |
Equity Shares |
INR 1/- each |
INR 5990.000 Million |
|
100000 |
Cumulative Preference Shares |
INR 100/- each |
INR 10.000 Million |
|
|
Total |
|
INR 6000.000 Million |
Issued, Subscribed & Paid-up Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
2399260815 |
Equity Shares |
INR 1/- each |
INR 2399.300 Million |
|
|
|
|
|
b) Reconciliation of the number of shares outstanding at the
beginning and the end of the reporting period:
|
|
31.03.2017 |
|
|
|
No.
of Shares held |
INR
in Million |
|
Opening balance |
2406605118 |
2406.600 |
|
Add : shares allotted to
employees on exercise of employee stock option (excluding shares held by ESOP
trust) |
155697 |
0.200 |
|
Less : buy-back of shares |
(7500000) |
(7.500) |
|
Outstanding at the end of the period |
2399260815 |
2399.300 |
c) The movement of equity shares issued to ESOP Trust at
face value is as follows: (As at April 01, 2015, excluding share suspense
account):
|
|
31.03.2017 |
|
|
|
No.
of Shares held |
INR
in Million |
|
Opening balance |
123381 |
0.100 |
|
Add : shares allotted pursuant
to the scheme of amalgamation |
|
|
|
Less : shares allotted by ESOP
Trust on exercise of employee stock option |
(93015) |
(0.100) |
|
Outstanding at the end of the
period |
30366 |
0.00 |
d) Shares in the company held by each shareholder holding
more than 5 percent shares specifying the number of shares held:
|
Name
of Shareholders |
31.03.2017 |
|
|
|
No.
of Shares held |
% of
Holding |
|
Dilip Shantilal Shanghvi |
230,285,690 |
9.60 |
|
Viditi Investment Private
Limited |
200,846,362 |
8.40 |
|
Tejaskiran Pharmachem Industries
Private Limited |
194,820,971 |
8.10 |
|
Family Investment Private
Limited |
182,437,880 |
7.60 |
|
Quality Investments Private
Limited |
182,379,237 |
7.60 |
(i)
1,035,581,955 (upto
March 31, 2016: 1,035,581,955; upto April 01, 2015: 1,035,581,955) equity
shares of INR 1 each have been allotted as fully paid up bonus shares during
the period of five years immediately preceding the date at which the Balance
Sheet is prepared.
(ii)
334,956,764 (upto
March 31, 2016: 334,956,764; upto April 01, 2015: Nil) equity shares of INR 1
each have been allotted, pursuant to scheme of amalgamation, without payment
being received in cash during the period of five years immediately preceding
the date at which the Balance Sheet is prepared.
(iii)
7,500,000 (upto March
31, 2016: Nil, upto April 01, 2015: Nil) equity shares of INR 1 each have been
bought back during the period of five years immediately preceding the date at
which the Balance Sheet is prepared. The shares bought back in the current year
were cancelled immediately.
(iv)
Rights, Preference and
Restrictions attached to equity shares: The Equity Shares of the Company,
having par value of INR 1 per share, rank pari passu in all respects including
voting rights and entitlement to dividend.
(v)
50 for number of
employee stock options against which equity shares are to be issued by the
Company / ESOP Trust upon vesting and exercise of those stock options.
FINANCIAL DATA
[all
figures are in INR Million]
ABRIDGED BALANCE SHEET (STANDALONE)
|
SOURCES OF FUNDS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
|
|
|
|
|
I.
EQUITY AND LIABILITIES |
|
|
|
|
(1)Shareholders' Funds |
|
|
|
|
(a) Share Capital |
2399.300 |
2406.600 |
2071.200 |
|
(b) Reserves & Surplus |
206315.800 |
212424.300 |
68719.500 |
|
(c) Money
received against share warrants |
0.000 |
0.000 |
0.000 |
|
|
|
|
|
|
(2) Share Application
money pending allotment |
0.000 |
6.700 |
483.800 |
|
Total Shareholders’ Funds
(1) + (2) |
208715.100 |
214837.600 |
71274.500 |
|
|
|
|
|
|
(3) Non-Current
Liabilities |
|
|
|
|
(a) long-term borrowings |
7606.400 |
19292.700 |
11703.200 |
|
(b) Deferred tax
liabilities (Net) |
0.000 |
0.000 |
0.000 |
|
(c) Other long term
liabilities |
6.800 |
135.700 |
143.700 |
|
(d) long-term provisions |
11328.300 |
19245.500 |
24225.100 |
|
Total Non-current
Liabilities (3) |
18941.500 |
38673.900 |
36072.000 |
|
|
|
|
|
|
(4) Current Liabilities |
|
|
|
|
(a) Short term borrowings |
40540.400 |
37337.200 |
42528.100 |
|
(b) Trade payables |
20942.000 |
17724.400 |
14849.700 |
|
(c) Other current
liabilities |
29873.400 |
19068.300 |
32475.500 |
|
(d) Short-term provisions |
18469.900 |
14257.900 |
11977.700 |
|
Total Current Liabilities
(4) |
109825.700 |
88387.800 |
101831.000 |
|
|
|
|
|
|
TOTAL |
337482.300 |
341899.300 |
209177.500 |
|
|
|
|
|
|
II.
ASSETS |
|
|
|
|
(1) Non-current assets |
|
|
|
|
(a) Fixed Assets |
|
|
|
|
(i) Tangible assets |
38319.400 |
35129.000 |
42758.600 |
|
(ii) Intangible Assets |
484.600 |
552.700 |
0.000 |
|
(iii) Capital
work-in-progress |
10533.200 |
7677.300 |
0.000 |
|
(iv) Intangible assets
under development |
453.900 |
0.000 |
0.000 |
|
(b) Non-current
Investments |
193510.200 |
222831.100 |
93666.200 |
|
(c) Deferred tax assets
(net) |
7517.000 |
0.000 |
0.000 |
|
(d) Long-term Loan and Advances |
48.500 |
21654.700 |
18952.300 |
|
(e) Other Non-current
assets |
22916.900 |
617.500 |
419.500 |
|
Total Non-Current Assets |
273783.700 |
288462.300 |
155796.600 |
|
|
|
|
|
|
(2) Current assets |
|
|
|
|
(a) Current investments |
400.100 |
824.900 |
939.300 |
|
(b) Inventories |
22866.200 |
21321.600 |
21892.500 |
|
(c) Trade receivables |
27256.700 |
20168.100 |
18028.200 |
|
(d) Cash and cash
equivalents |
1638.100 |
1693.900 |
3042.700 |
|
(e) Short-term loans and
advances |
138.200 |
7450.300 |
6966.900 |
|
(f) Other current assets |
11399.300 |
1978.200 |
2511.300 |
|
Total Current Assets |
63698.600 |
53437.000 |
53380.900 |
|
|
|
|
|
|
TOTAL |
337482.300 |
341899.300 |
209177.500 |
PROFIT & LOSS ACCOUNT (STANDALONE)
|
|
PARTICULARS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
|
SALES |
|
|
|
|
|
Income |
78067.000 |
76144.600 |
77309.300 |
|
|
Other Income |
5144.100 |
4318.200 |
3924.700 |
|
|
TOTAL |
83211.100 |
80462.800 |
81234.000 |
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
Cost of Materials
Consumed |
22845.200 |
20198.500 |
22611.100 |
|
|
Purchases of
Stock-in-Trade |
12365.000 |
11700.000 |
9342.200 |
|
|
Changes in inventories of
finished goods, work-in-progress and Stock-in-Trade |
(1627.900) |
(684.800) |
3181.000 |
|
|
Employees benefits
expense |
14861.700 |
14805.100 |
14866.900 |
|
|
Other expenses |
28670.300 |
34475.600 |
34689.500 |
|
|
Exceptional Items |
0.000 |
701.300 |
0.000 |
|
|
TOTAL |
77114.300 |
81195.700 |
84690.700 |
|
|
|
|
|
|
|
Less |
PROFIT/ (LOSS) BEFORE INTEREST, TAX, DEPRECIATION AND
AMORTISATION |
6096.800 |
(732.900) |
(3456.700) |
|
|
|
|
|
|
|
Less |
FINANCIAL EXPENSES |
2235.600 |
5306.400 |
5512.500 |
|
|
|
|
|
|
|
|
PROFIT / (LOSS) BEFORE TAX,
DEPRECIATION AND AMORTISATION |
3861.200 |
(6039.300) |
(8969.200) |
|
|
|
|
|
|
|
Less/ Add |
DEPRECIATION/
AMORTISATION |
4185.600 |
4639.800 |
6606.800 |
|
|
|
|
|
|
|
|
PROFIT/ (LOSS) BEFORE TAX |
(324.400) |
(10679.100) |
(15576.000) |
|
|
|
|
|
|
|
Less |
TAX |
25.100 |
54.500 |
(848.400) |
|
|
|
|
|
|
|
|
PROFIT/ (LOSS) AFTER TAX |
(349.500) |
(10733.600) |
(14727.600) |
|
|
|
|
|
|
|
|
Earnings / (Loss) Per
Share (INR) |
(0.10) |
(4.50) |
6.12 |
CURRENT MATURITIES OF LONG TERM
DEBT DETAILS
|
Particulars |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Current Maturities of Long term debt |
13319.300 |
1722.800 |
13838.700 |
|
Cash generated from operations |
(8450.600) |
(8917.200) |
3420.700 |
|
Net cash flow from operating activity |
(16239.500) |
(12444.500) |
1485.900 |
QUARTERLY RESULTS
|
Particulars |
30.06.2017 |
30.09.2017 |
31.12.2017 |
|
|
Unaudited |
||
|
|
1st Quarter |
2nd Quarter |
3rd Quarter |
|
Net Sales |
18025.200 |
18238.400 |
20452.300 |
|
Total Expenditure |
19753.300 |
19073.700 |
18669.100 |
|
PBIDT (Excl OI) |
(1728.100) |
(835.300) |
1783.200 |
|
Other Income |
257.900 |
8918.200 |
334.700 |
|
Operating Profit |
(1470.200) |
8082.900 |
2117.900 |
|
Interest |
834.400 |
1295.800 |
647.000 |
|
Exceptional Items |
(9505.000) |
NA |
NA |
|
PBDT |
(11809.600) |
6787.100 |
1470.900 |
|
Depreciation |
1087.700 |
1050.800 |
1060.500 |
|
Profit Before Tax |
(12897.300) |
5736.300 |
410.400 |
|
Tax |
12.600 |
(34.200) |
5.400 |
|
Provisions and contingencies |
NA |
NA |
NA |
|
Profit After Tax |
(12909.900) |
5770.500 |
405.000 |
|
Extraordinary Items |
NA |
NA |
NA |
|
Prior Period Expenses |
NA |
NA |
NA |
|
Other Adjustments |
NA |
NA |
NA |
|
Net Profit |
(12909.900) |
5770.500 |
405.000 |
KEY RATIOS
EFFICIENCY RATIOS
|
PARTICULARS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Average Collection Days (Sundry Debtors / Income * 365 Days) |
127.44 |
96.68 |
85.12 |
|
|
|
|
|
|
Account Receivables Turnover ( Income / Sundry Debtors) |
2.86 |
3.78 |
4.29 |
|
|
|
|
|
|
Average Payment Days (Sundry Creditors / Purchases * 365
Days) |
217.09 |
202.81 |
169.63 |
|
|
|
|
|
|
Inventory Turnover (Operating Income / Inventories) |
0.27 |
(0.03) |
(0.16) |
|
|
|
|
|
|
Asset Turnover (Operating Income / Net Fixed Assets) |
0.12 |
(0.02) |
(0.08) |
LEVERAGE RATIOS
|
PARTICULARS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Debt Ratio ((Borrowing + Current Liabilities) / Total Assets) |
0.39 |
0.32 |
0.61 |
|
|
|
|
|
|
Debt Equity Ratio (Total Liability / Networth) |
0.29 |
0.27 |
0.96 |
|
|
|
|
|
|
Current Liabilities to Networth (Current Liabilities / Net Worth) |
0.53 |
0.41 |
1.43 |
|
|
|
|
|
|
Fixed Assets to Networth (Net Fixed Assets / Networth) |
0.24 |
0.20 |
0.60 |
|
|
|
|
|
|
Interest Coverage Ratio (PBIT / Financial Charges) |
2.73 |
(0.14) |
(0.63) |
PROFITABILITY RATIOS
|
PARTICULARS |
|
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Net Profit Margin [(PAT / Sales)
* 100] |
% |
(0.45) |
(14.10) |
(19.05) |
|
|
|
|
|
|
|
Return on Total Assets ((PAT / Total Assets) * 100) |
% |
(0.10) |
(3.14) |
(7.04) |
|
|
|
|
|
|
|
Return on Investment (ROI) ((PAT / Networth) * 100) |
% |
(0.17) |
(5.00) |
(20.66) |
SOLVENCY RATIOS
|
PARTICULARS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Current Ratio (Current Assets / Current Liabilities) |
0.58 |
0.60 |
0.52 |
|
|
|
|
|
|
Quick Ratio ((Current Assets – Inventories) /
Current Liabilities) |
0.37 |
0.36 |
0.31 |
|
|
|
|
|
|
G-Score Ratio Financial (Networth / Total Assets) |
0.62 |
0.63 |
0.34 |
|
|
|
|
|
|
G-Score Ratio Debt (Debts / Equity Capital) |
25.62 |
24.25 |
32.87 |
|
|
|
|
|
|
G-Score Ratio Liquidity (Total Current Assets / Total Current
Liabilities) |
0.58 |
0.60 |
0.52 |
Total Liability = Short-term Debt + Long-term Debt + Current
Maturities of Long-term debts
STOCK PRICES
|
Face Value |
INR 1.00/- |
|
Market Value |
INR 510.00/- |
FINANCIAL ANALYSIS
[all
figures are in INR Million]
DEBT EQUITY RATIO
|
Particular |
31.03.2015 |
31.03.2016 |
31.03.2017 |
|
|
INR
In Million |
INR
In Million |
INR
In Million |
|
Share Capital |
2071.200 |
2406.600 |
2399.300 |
|
Share Suspense Account |
68719.500 |
212424.300 |
206315.800 |
|
Money received against share
warrants |
0.000 |
0.000 |
0.000 |
|
Share Application money
pending allotment |
483.800 |
6.700 |
0.000 |
|
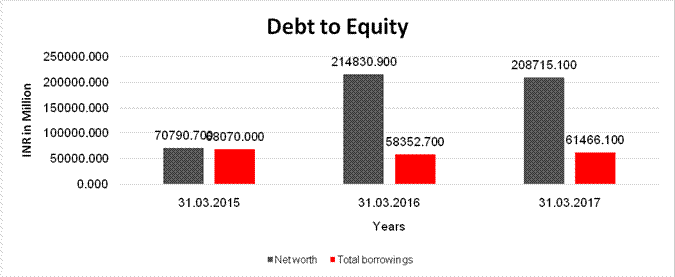
Net
worth |
70790.700 |
214830.900 |
208715.100 |
|
|
|
|
|
|
long-term borrowings |
11703.200 |
19292.700 |
7606.400 |
|
Short term borrowings |
42528.100 |
37337.200 |
40540.400 |
|
Current Maturities of Long
term debt |
13838.700 |
1722.800 |
13319.300 |
|
Total
borrowings |
68070.000 |
58352.700 |
61466.100 |
|
Debt/Equity
ratio |
0.962 |
0.272 |
0.294 |
YEAR-ON-YEAR GROWTH
|
Year on Year Growth |
31.03.2015 |
31.03.2016 |
31.03.2017 |
|
|
INR
In Million |
INR
In Million |
INR
In Million |
|
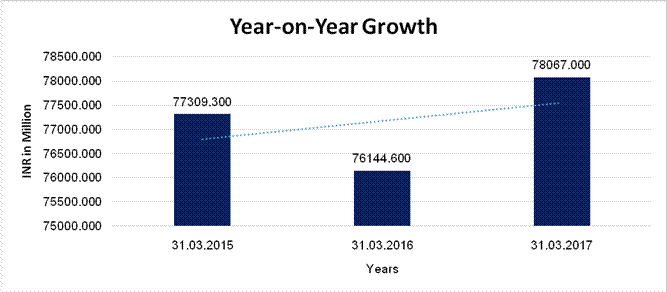
Sales |
77309.300 |
76144.600 |
78067.000 |
|
|
|
(1.507) |
2.525 |
NET PROFIT MARGIN
|
Net Profit Margin |
31.03.2015 |
31.03.2016 |
31.03.2017 |
|
|
INR
In Million |
INR
In Million |
INR
In Million |
|
Sales |
77309.300 |
76144.600 |
78067.000 |
|
Profit/ (Loss) |
(14727.600) |
(10733.600) |
(349.500) |
|
|
(19.05%) |
(14.10%) |
(0.45%) |

ABRIDGED BALANCE SHEET - CONSOLIDATED
|
SOURCES OF FUNDS |
|
31.03.2017 |
31.03.2016 |
|
|
|
|
|
|
I.
EQUITY AND LIABILITIES |
|
|
|
|
(1)Shareholders' Funds |
|
|
|
|
(a) Share Capital |
|
2399.300 |
2406.600 |
|
(b)
Share Suspense Account |
|
0.000 |
0.000 |
|
(c)
Reserves & Surplus |
|
363997.400 |
311635.600 |
|
(d)
Money received against share warrants |
|
0.000 |
0.000 |
|
|
|
|
|
|
(2) Share Application
money pending allotment |
|
0.000 |
6.700 |
|
(3)
Minority Interest |
|
37908.600 |
40852.500 |
|
Total Shareholders’ Funds
(1) + (2) |
|
404305.300 |
354901.400 |
|
|
|
|
|
|
(3) Non-Current
Liabilities |
|
|
|
|
(a) long-term borrowings |
|
14360.800 |
31167.300 |
|
(b) Deferred tax
liabilities (Net) |
|
3147.900 |
616.100 |
|
(c) Other long term
liabilities |
|
1307.100 |
2237.100 |
|
(d) long-term provisions |
|
12111.100 |
20797.000 |
|
Total Non-current Liabilities
(3) |
|
30926.900 |
54817.500 |
|
|
|
|
|
|
(4) Current Liabilities |
|
|
|
|
(a) Short term borrowings |
|
66549.200 |
52213.700 |
|
(b) Trade payables |
|
43953.900 |
34895.800 |
|
(c) Other current
liabilities |
|
28208.000 |
11192.400 |
|
(d) Short-term provisions |
|
40159.100 |
34174.700 |
|
Total Current Liabilities
(4) |
|
178870.200 |
132476.600 |
|
|
|
|
|
|
TOTAL |
|
614102.400 |
542195.500 |
|
|
|
|
|
|
II.
ASSETS |
|
|
|
|
(1) Non-current assets |
|
|
|
|
(a) Fixed Assets |
|
|
|
|
(i) Tangible assets |
|
84952.900 |
75559.200 |
|
(ii) Intangible Assets |
|
36436.600 |
40708.500 |
|
(iii) Capital
work-in-progress |
|
15647.600 |
12034.600 |
|
(iv) Intangible assets under
development |
|
12366.200 |
5303.700 |
|
(b) Goodwill of consolidated |
|
55362.200 |
41811.100 |
|
(c) Non-current
Investments |
|
9610.000 |
5933.200 |
|
(d) Deferred tax assets
(net) |
|
24928.200 |
21875.200 |
|
(e) Long-term Loan and Advances |
|
698.100 |
29360.300 |
|
(f) Other Non-current
assets |
|
44564.100 |
964.000 |
|
Total Non-Current Assets |
|
284565.900 |
233549.800 |
|
|
|
|
|
|
(2) Current assets |
|
|
|
|
(a) Current investments |
|
2308.800 |
7152.800 |
|
(b) Inventories |
|
68328.100 |
64236.300 |
|
(c) Trade receivables |
|
72026.100 |
67958.900 |
|
(d) Cash and cash
equivalents |
|
151408.400 |
139892.500 |
|
(e) Short-term loans and
advances |
|
10190.800 |
26403.900 |
|
(f) Other current assets |
|
25274.300 |
3001.300 |
|
Total Current Assets |
|
329536.500 |
308645.700 |
|
|
|
|
|
|
TOTAL |
|
614102.400 |
542195.500 |
PROFIT & LOSS ACCOUNT - CONSOLIDATED
|
|
PARTICULARS |
|
31.03.2017 |
31.03.2016 |
|
|
SALES |
|
|
|
|
|
Income |
|
315784.400 |
282697.100 |
|
|
Other Income |
|
6231.500 |
6169.700 |
|
|
TOTAL |
|
322015.900 |
288866.800 |
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
Cost of Materials
Consumed |
|
51246.100 |
43881.300 |
|
|
Purchases of
Stock-in-Trade |
|
32777.600 |
25429.200 |
|
|
Changes in inventories of
finished goods, work-in-progress and Stock-in-Trade |
|
(2716.300) |
(4478.600) |
|
|
Employees benefits
expense |
|
49023.000 |
47971.300 |
|
|
Other expenses |
|
84561.300 |
86655.000 |
|
|
Exceptional Items |
|
0.000 |
6851.700 |
|
|
TOTAL |
|
214891.700 |
206309.900 |
|
|
|
|
|
|
|
Less |
PROFIT/ (LOSS) BEFORE INTEREST, TAX, DEPRECIATION AND
AMORTISATION |
|
107124.200 |
82556.900 |
|
|
|
|
|
|
|
Less |
FINANCIAL EXPENSES |
|
3998.000 |
4768.900 |
|
|
|
|
|
|
|
|
PROFIT / (LOSS) BEFORE
TAX, DEPRECIATION AND AMORTISATION |
|
103126.200 |
77788.000 |
|
|
|
|
|
|
|
Less |
DEPRECIATION/
AMORTISATION |
|
12647.500 |
10135.200 |
|
|
|
|
|
|
|
|
PROFIT/ (LOSS) BEFORE TAX |
|
90478.700 |
67652.800 |
|
|
|
|
|
|
|
Less |
TAX |
|
12115.700 |
9349.000 |
|
|
|
|
|
|
|
|
PROFIT/ (LOSS) AFTER TAX |
|
78363.000 |
58303.800 |
|
|
|
|
|
|
|
Less |
SHARE IN LOSS OF ASSOCIATES |
|
99.300 |
18.700 |
|
|
|
|
|
|
|
|
PROFIT AFTER TAX BEFORE ADJUSTMENT FOR MINORITY INTEREST |
|
78263.700 |
58285.100 |
|
|
|
|
|
|
|
Less |
SHARE OF PROFIT ATTRIBUTABLE TO MINORITY INTEREST |
|
8818.600 |
11126.000 |
|
|
|
|
|
|
|
|
Profit for the Year attributable to the Shareholders of
the Company |
|
69445.100 |
47159.100 |
|
|
|
|
|
|
|
|
Earnings / (Loss) Per
Share (INR) |
|
29.00 |
19.60 |
LEGALS
FILE
NOTE: LEGAL CASE FILES DETAILS ATTACHED
LOCAL AGENCY FURTHER INFORMATION
|
Sr.
No. |
Check
list by info agents |
Available
in Report (Yes/No) |
|
1 |
Year of establishment |
Yes |
|
2 |
Constitution of the entity
-Incorporation details |
Yes |
|
3 |
Locality of the entity |
Yes |
|
4 |
Premises details |
Yes |
|
5 |
Buyer visit details |
- |
|
6 |
Contact numbers |
Yes |
|
7 |
Name of the person contacted |
Yes |
|
8 |
Designation of contact person |
Yes |
|
9 |
Promoter’s background |
Yes |
|
10 |
Date of Birth of Proprietor /
Partners / Directors |
Yes |
|
11 |
Pan Card No. of Proprietor /
Partners |
No |
|
12 |
Voter Id Card No. of
Proprietor / Partners |
No |
|
13 |
Type of business |
Yes |
|
14 |
Line of Business |
Yes |
|
15 |
Export/import details (if
applicable) |
No |
|
16 |
No. of employees |
Yes |
|
17 |
Details of sister concerns |
Yes |
|
18 |
Major suppliers |
No |
|
19 |
Major customers |
No |
|
20 |
Banking Details |
Yes |
|
21 |
Banking facility details |
Yes |
|
22 |
Conduct of the banking account |
-- |
|
23 |
Financials, if provided |
Yes |
|
24 |
Capital in the business |
Yes |
|
25 |
Last accounts filed at ROC, if
applicable |
Yes |
|
26 |
Turnover of firm for last
three years |
Yes |
|
27 |
Reasons for variation <>
20% |
-- |
|
28 |
Estimation for coming
financial year |
No |
|
29 |
Profitability for last three
years |
Yes |
|
30 |
Major shareholders, if
available |
Yes |
|
31 |
External Agency Rating, if
available |
Yes |
|
32 |
Litigations that the firm/promoter
involved in |
Yes |
|
33 |
Market information |
--- |
|
34 |
Payments terms |
No |
|
35 |
Negative Reporting by Auditors
in the Annual Report |
No |
MANAGEMENT DISCUSSION AND ANALYSIS
GLOBAL PHARMACEUTICAL INDUSTRY
The global spending on medicines
is expected to reach nearly US$ 1.5 Trillion by 2021. This is an increase of
nearly US$ 370 Billion from the 2016 estimated spending level, representing a
CAGR of 4-7%. The two main drivers of this growth will be introduction of new
innovative products in the developed markets and increased volumes of branded
generics in the emerging markets.
The growth of a country’s
pharmaceutical industry closely mirrors its general economic progress. As
economies of the world demonstrate widely divergent growth patterns, industry
growth is also different. However, taking a macro perspective, global
pharmaceutical growth depends on worldwide economic momentum, government
healthcare programmes and spending patterns. While R&D efforts will drive
the introduction of new products in the market, challenges remain. For
countries grappling with sluggish economies and limited resources, funding
access to these medicines remains an uphill task.
Each country in the world is
facing these challenges and addressing them in its own way. Overall, generic
products will continue to be an integral part of these efforts, targeted at
striking a balance between access to healthcare and ability to fund it.
The key trends for the next five years:
The US will continue as the
world’s largest pharmaceutical market.
New innovative products will drive
the growth in pharmaceutical spending in developed markets, but will be partly
offset by patent expires. Growth will be driven primarily by oncology,
autoimmune and diabetes treatments.
Pharmerging markets will grow
faster than developed markets, driven mainly by rising income levels, increased
healthcare awareness, government policies directed at achieving universal
healthcare and increasing incidence of chronic ailments.
Innovation in specialty medicines
will drive the share of global specialty spending from 30% in 2016 to 35% in
2021. This increase will be driven by the acceptance of new breakthrough
medicines.
The specialty segment will be a
key focus area for payers and they are likely to focus on lowering healthcare
costs and the therapeutic value offered by such specialty medicines. The US and
Western Europe will be the key drivers of specialty medicines.
GLOBAL GENERICS
The global generics market consists
of both non-branded and branded generics. Branded generics in emerging markets
will be the key drivers of growth for the overall generics market. This growth
will be driven by many macroeconomic factors like rising per capita incomes,
growing healthcare awareness, increasing medical insurance penetration and
higher incidence of chronic ailments. The efforts of governments in emerging
markets to achieve universal healthcare are also expected to drive the growth
of branded generics.
The global demand for non-branded
generic drugs will continue to grow as governments, payors and consumers pursue
avenues to reduce healthcare costs, mainly in the developed economies.
GROWTH DRIVERS OF GLOBAL PHARMACEUTICAL INDUSTRY
Changing demographic pattern Ageing
population and growing life expectancy will remain a longterm growth driver for
global pharmaceutical consumption. The combination of population ageing and
increased life expectancy — up from an estimated 72.3 years in 2014 to 73.3
years in 2019 — will take the number of people aged 65-plus to over 604
Million, or 10.8% of the total global population. This number is anticipated to
be even higher in Western Europe (nearly 21%) and Japan (28%).
Factors that have contributed to
enhanced life expectancy are declining infant mortality, enhanced living
conditions, improved sanitation, better prevention of communicable diseases,
and growing access to healthcare. Increased life expectancy, coupled with other
macroeconomic factors (rising per capital incomes, growing healthcare
awareness, enhanced medical insurance penetration) will remain key growth
drivers for the pharmaceutical industry.
OUTLOOK
The global spending on medicines
is estimated to grow at 4-7% CAGR between 2016 and 2021, to reach approximately
US$ 1.5 Trillion. Pharmaceutical spending growth in developed markets, will be
driven by oncology, autoimmune and diabetes treatments. Developed market
spending growth will be driven by original brands but will be partly
constrained by patent expiries and the cost and access controls instituted by
payors. Growth in pharmerging markets will continue to be fuelled by
branded-generic and pure generic products.
INDIAN PHARMACEUTICAL MARKET8
India’s pharmaceutical market
ranks third in the world in terms of volume and 11th in terms of value. At US$
17.4 Billion, the market in India accounted for 1.6% share of the global market
in 2016. It is expected to grow at a CAGR of 10-13% to US$ 26-30 Billion by
2021.
The overall penetration of modern
medicines is quite low in India. The per capita spending on pharmaceuticals in
India is one of the lowest among emerging markets. Compared to the emerging
market average per capita spend of about US$ 117 per year, the spending in
India is approximately US$ 15-25 per year. Affordability, access and awareness
are the prime factors, which determine demand for pharmaceutical products in
the Indian market.
Other factors like rising per
capita income, improving access to healthcare facilities, and higher government
spending on healthcare drive market demand. Moreover, increasing insurance
penetration, more healthcare awareness and enhanced investments for treating
chronic ailments serve as key growth drivers.
GENERAL INFORMATION
Subject the Company is a public limited
company incorporated and domiciled in India and has its listing on the BSE
Limited and National Stock Exchange of India Limited. The addresses of its
registered office and principal place of business are disclosed in the
introduction to the annual report. The Company is in the business of
manufacturing, producing, developing and marketing a wide range of branded and
generic formulations and Active Pharmaceutical Ingredients (APIs). The Company
has various manufacturing locations spread across the country with trading v
and other incidental and related activities extending to the global markets.
UNSECURED
LOAN
|
PARTICULARS
|
31.03.2017 (INR in Million) |
31.03.2016 (INR in Million) |
|
Long-term
Borrowings |
|
|
|
Term Loans from Banks |
7498.200 |
19215.400 |
|
Short-term
borrowings |
|
|
|
From Banks |
25021.500 |
26756.800 |
|
Loans and Advances from a
related party |
0.000 |
8019.900 |
|
Other Loans and Advances
Commercial Paper |
15320.800 |
0.000 |
|
Total |
47840.500 |
53992.100 |
CONTINGENT
LIABILITIES:
(INR
in million)
|
PARTICULARS |
31.03.2017 |
|
Claims against the Company not
acknowledged as debts |
355.0000 |
|
Liabilities Disputed - Appeals
filed with respect to : |
|
|
Income Tax on account of Disallowances
/ Additions |
45998.300 |
|
Sales Tax on account of Rebate /
Classification |
45.700 |
|
Excise Duty on account of
Valuation / Cenvat Credit |
1102.200 |
|
ESIC Contribution on account of
applicability |
132.800 |
|
Service tax on certain services
performed outside India under reverse charge basis |
0.000 |
|
Drug Price Equalisation Account
[DPEA] on account of demand towards unintended benefit, enjoyed by
the Company |
3488.200 |
|
Demand by JDGFT import duty with
respect to import alleged to be in excess of entitlement as per the Advanced
License Scheme |
16.700 |
|
Fine imposed for
anti-competitive settlement agreement by European Commission |
715.400 |
|
Octroi demand on account of rate
difference |
171.000 |
|
Other matters - employee /
worker related cases, State electricity board, Punjab Land Preservation Act
related matters etc. |
67.500 |
INDEX
OF CAHREGS:
|
SNo |
SRN |
Charge Id |
Charge Holder Name |
Date of Creation |
Date of Modification |
Date of Satisfaction |
Amount |
Address |
|
1 |
G05259965 |
10351563 |
BIOTECHNOLOGY INDUSTRY RESEARCH
ASSISTANCE COUNCIL |
31/03/2012 |
29/02/2016 |
- |
178140000.0 |
MTNL Building, 1st Floor,9 CGO
Complex, Lodi RoadNew DelhiDe110003IN |
|
2 |
Y10182237 |
90095637 |
NVFC FINANCE PRIVATE LTD |
26/05/1999 |
27/04/2000 |
- |
7500000.0 |
1843; 6TH CROSS 20TH MAINJ.P.
NAGAR; II PHASEBANGLOREKA560078IN |
|
3 |
Y10181712 |
90095112 |
RISK CAPITAL AND TECHNOLOGY
FINANCE CORPORATION LTD |
29/04/1999 |
05/08/2004 |
- |
15000000.0 |
E-216; 3RD FLOOREAST KAILASHNEW
DELHIDL110065IN |
|
4 |
Y10181683 |
90095083 |
INDUSTRIAL DEVELOPMENT BANK OF
INDIA |
17/11/1998 |
09/07/2005 |
- |
32500000.0 |
IDBI TOWERCOLABAMUMBAIMH400005IN |
|
5 |
Y10181537 |
90094937 |
INDUSTRIAL DEVELOPMENT BANK OF
INDIA |
13/09/1996 |
28/11/1997 |
- |
92000000.0 |
IDBI TOWERCOLABAMUMBAIMH400005IN |
|
6 |
Y10191137 |
90101445 |
GUJARAT INDUSTRIAL INVESTMENT
CORPORATION LTD. |
11/10/1993 |
11/10/1993 |
- |
13600000.0 |
CHUNIOBHAI CHAMBERSASHRAM
ROADAHMEDABADGJ380009IN |
|
7 |
G74082454 |
90098193 |
ICICI BANK LIMITED |
03/07/1995 |
26/08/2013 |
19/01/2018 |
1260000000.0 |
ICICI BANK TOWERSBANDRA KURLA
COMPLEX, BANDRA (EAST),MUMBAIMH400051IN |
|
8 |
G74058710 |
90097707 |
BANK OF BARODA |
06/04/1993 |
06/04/1993 |
17/01/2018 |
87831000.0 |
INDUSTRIAL ESTATE BRANCHVAPIGJIN |
|
9 |
G74064015 |
90101587 |
BANK OF BARODA |
03/07/1995 |
03/10/2001 |
17/01/2018 |
252500000.0 |
ALKAPURI BRANCH; OPP. PETROL PUMPLAKAPURIBARODAGJ338159IN |
|
10 |
G74062829 |
90098061 |
BANK OF BARODA |
28/01/1995 |
- |
17/01/2018 |
178000000.0 |
ALKAPURI BRANCHR. C. DUTT
ROADBARODAGJIN |
FIXED ASSETS
- Freehold Land
- Leasehold Land
- Buildings
- Buildings –
Leased
- Plant and
Equipment
- Plant and
Equipment – Leased
- Vehicles
- Office
Equipment
- Furniture and
Fixtures
- Computer
Software
UNAUDITED FINANCIAL RESULTS FOR THE QUARTER
ENDED AND Nine MONTHS 31.12.2017
(INR In Million)
|
PARTICULARS |
3 Months |
9 Months |
|
|
|
31.12.2017 |
30.09.2017 |
31.03.2017 |
|
|
[Unaudited] |
[Unaudited] |
[Unaudited] |
|
1. Income from Operations |
|
|
|
|
Net Sales/income from
operations |
20452.300 |
18238.400 |
56644.200 |
|
Other Operating Income |
334.700 |
8918.200 |
9511.500 |
|
Total income from operations (net) |
20787.000 |
27156.600 |
66155.700 |
|
|
|
|
|
|
Expenses |
|
|
|
|
Cost of materials consumed |
2057.500 |
3385.000 |
11398.200 |
|
Purchases of stock-in trade |
2955.600 |
1954.800 |
7102.800 |
|
Changes in inventories of finished goods. work-in-progress and stock
in trade |
4051.700 |
3660.200 |
7689.600 |
|
Employee benefits expense |
4040.700 |
3929.600 |
12111.100 |
|
Depreciation and Amortization Expenses |
1060.500 |
1050.800 |
3207.600 |
|
Other Expenses |
5563.600 |
6144.100 |
19074.500 |
|
Finance Costs |
647.000 |
1295.800 |
2777.200 |
|
Total expenses |
20376.600 |
21420.300 |
63361.000 |
|
Profit/ (Loss) from ordinary activities after finance cost but before
exceptional items |
410.400 |
5736.300 |
2794.700 |
|
Exceptional items |
0.000 |
0.000 |
9505.000 |
|
Profit/ (Loss) from ordinary activities before tax |
410.400 |
5736.300 |
(6710.300) |
|
Tax expenses |
5.400 |
(34.200) |
(1.800) |
|
Net Profit / (Loss) from ordinary activities after tax |
405.000 |
5770.500 |
(6708.500) |
|
Extraordinary item (net of tax expense) |
0.000 |
0.000 |
0.000 |
|
Net Profit / (Loss) for the period |
405.000 |
5770.500 |
(6708.500) |
|
Comprehensive Income |
(184.500) |
(165.900) |
(504.000) |
|
Net Profit/ (Loss) after taxes, minority interest and share of
profit/(loss) of associates |
220.500 |
5604.600 |
(7212.500) |
|
|
|
|
|
|
Paid up equity share capital (Face Value of INR 1/-each) |
2399.300 |
2399.300 |
2399.300 |
|
Reserve excluding Revaluation Reserve as per Balance Sheet of previous
accounting year |
- |
- |
- |
|
Earnings per share (before extraordinary items) of INR 1/- each (not
annualized): |
- |
- |
- |
|
(a) Basic |
0.20 |
2.40 |
-2.80 |
|
(b) Diluted |
0.20 |
2.40 |
-2.80 |
Notes:
1) The above standalone unaudited
financial results of the Company have been reviewed by the Audit Committee and
approved by the Board of Directors at their respective meetings held on February
14, 2018. The statutory auditors of the Company have conducted a "Limited
Review" of the above standalone unaudited financial results.
2) The Company has adopted Indian Accounting Standards (Ind AS) prescribed
under section 133 of the Companies Act, 2013, read with relevant rules issued
thereunder, and accordingly, these standalone unaudited financial results have
been prepared in accordance with the recognition and measurement principles
laid down in Ind AS 34 Interim financial reporting, and other accounting
principles generally accepted in India.
3) During the quarter ended December 31, 2017, 1,260 equity shares of INR 1
each have been allotted under Employee Stock Option Scheme of the
Company.
4) Post implementation of Goods and Service Tax ("GST") with effect
from July 01, 2017, revenue from operations is disclosed net of GST. Revenue
from operations for the earlier periods included excise duty which is now
subsumed in GST. Revenue from operations for the nine months ended December 31,
2017 includes excise duty for the period ended June 30, 2017. Accordingly,
revenue from operations for quarter and nine months ended December 31, 2017 are
not comparable with those of the previous periods presented.
5) The Board of Directors of the Company at their meeting held on November 10,
2016 and the shareholders and unsecured creditors of the Company at their
respective meetings held on June 20, 2017 approved the proposed scheme of
arrangement u/s 230 to 232 of the Companies Act, 2013 for amalgamation of Sun
Pharma Medisales Private Limited, Ranbaxy Drugs Limited, Gufic Pharma Limited
and Vidyut Investments Limited into the Company with effect from April 01,
2017, the appointed date ("the Proposed Scheme"). The Company
received the amalgamation order effective September 06, 2017. The results for
previous periods have been restated to give effect to the merger.
6) In respect of an antitrust litigation, relating to a product Modafinil, the
Company and one of its wholly-owned subsidiaries entered into settlements with
certain plaintiffs (Apotex Corporation and Retailer Purchasers), whereby the
Company agreed to pay an aggregate amount of USD 147 Million. The equivalent
Indian rupee liability of INR 9505.000 Million has been provided in nine months
ended December 31, 2017 and disclosed as an exceptional item.
7) The Company has only one reportable segment namely 'Pharmaceuticals'.
WEBSITE DETAILS
PRESS RELEASES/ NEWS
SUN PHARMA POISED TO CLEAR US FDA'S WARNING
ON HALOL: ANALYSTS
The US FDA conducted Good
Manufacturing Practices (GMP) inspection of Sun Pharma's Halol facility from
February 12 - 23.
Mar 06, 2018: Sun Pharma,
India’s largest drug maker, is well poised to resolve the US FDA’s warning
letter on its Halol site in Gujarat – as the three Form 483 observations the
company received from the agency turned out to be minor in nature, according to
analysts Moneycontrol spoke to.
US FDA inspectors issue
observations on Form 483 at the end of the site inspection outlining any
deviations of current good manufacturing practices. The company then will have
to file its response in 15 days providing corrective and preventive action plan
referred as CAPA. Any failure to respond to the satisfaction of the agency may
escalate to warning letter and in some worst cases even an import ban. If the
company responds to the satisfaction the US FDA issues an establishment
inspection report (EIR) indicating closure of the inspection.
The US FDA conducted Good
Manufacturing Practices (GMP) inspection of Sun Pharma's Halol facility from
February 12 - 23.
Moneycontrol on February 23 had
reported about Sun Pharma’s founder and managing director Dilip Shanghvi
expressing confidence about resolving the three observations
pointed out by the American drug regulator.
The three observations that were
made public by US FDA relates to certain deficiencies in preventing of
contamination and lack of sufficient written procedures for cleaning and
maintenance of equipment, as well as sampling, testing, approval and rejection
of drug product containers
Below are the three observations:
Observation 1: Separate or
defined areas to prevent contamination or mix-ups are deficient regarding
operations related to aseptic processing of drug products.
Observation 2: Written
procedures for cleaning and maintenance fail to include description in
sufficient detail of the methods of disassembling and reassembling equipment as
necessary to assure proper cleaning and maintenance.
Observation 3: Written
procedures are lacking which describe in sufficient detail the sampling,
testing, approval and rejection of drug product containers and closures. (Click Here: Halol 483)
“The warning letter on the Halol
facility is expected to be cleared in next 6-8 months,” said Amey Chalke,
Research Analyst at HDFC Securities point out to the minor observations.
Chalke said the development is
already priced in the stock price.
“They view these to be minor
observations, meaning no data integrity issue and no observations in the
technical areas,” said brokerage house IIFL.
“Also USFDA has not asked to
provide any sort of studies, which indicates that the plant may not require
re-inspection if Sun Pharma’s response is found adequate by the USFDA,” IIFL
added.
Halol is critical for Sun Pharma
as most of the complex pipeline that includes injectables, topical solutions
and inhalation products are filed from this facility. Sun Pharma is banking on
complex products to beat the pricing pressure it faces in the US market.
The current inspection was
triggered after US FDA re-inspected the Halol facility from November 17, 2016
through December 1, 2016 and issued nine Form 483 observations.
The observations were related to
data reporting standards, testing procedures, and lab controls.
Halol is an important plant for
the company and contributed 10-15 percent to its US sales before the factory
received a warning letter from the US FDA for violation of manufacturing norms
in December 2015.
The warning letter blocked new
product approval from that facility impacting the company’s US business.
CLSA forecasted that incremental
revenue from the clearance of Halol is expected to be at least USD 100 million
and USD 150 million in years ending March 2019 and 2020, respectively, and even
possibly go up to USD 200 million and USD 300 million, increasing its earnings
per share by 4 and 10 percent.
“While a possible Halol resolution
is positive for the company, but Sun Pharma’s concerns run much deep,” Chalke
of HDFC Securities said.
“The declining sales at its US
subsidiary Taro, the pipeline that doesn’t look too promising to compensate for
the erosion of base business and investments of specialty that may take some
more years to deliver are some of the concerns to even maintain a USD 1.6
billion topline,” Chalke said.
Shares of Sun Pharma declined 0.93
percent and were trading at INR 542.60 on BSE at 12.10 pm, while the benchmark
Sensex gained 0.26 percent to 33,836.12 points.
SUN PHARMA FALLS 3% POST HALOL OBSERVATIONS;
NOMURA REMAINS NEUTRAL
The US FDA conducted Good Manufacturing
Practices (GMP) inspection of Sun Pharma's Halol facility from February 12 -
23.
Feb 26, 2018: Shares
of Sun Pharma fell
around 3 percent intraday on Monday as investors turned wary of observations
issued to its Halol plant by the US drug regulator.
US FDA issued three Form 483
observations for its Halol site in Gujarat.
The company didn't disclose the
nature of these observations.
A Form 483 is issued by the US FDA
inspectors at the end of the inspection outlining any deviations from GMP.
The US FDA conducted Good
Manufacturing Practices (GMP) inspection of Sun Pharma's Halol facility from
February 12 - 23.
"The company is preparing the
response to the observations, which will be submitted to the US FDA within 15
business days. The company is committed to addressing these observations
promptly," said Sun Pharma in a statement to stock exchanges.
"The company remains
committed to working closely with the US FDA and continues to enhance its GMP
compliance on an ongoing basis," the statement added.
The company's founder and managing
director Dilip Shanghvi expressed confidence about resolving the three issues
pointed out by the American drug regulator.
Brokerage houses are neutral on
the stock citing closure time for the observations and EIR issuance.
Brokerage: Nomura | Rating: Neutral | Target At INR 479
Nomura said that it is awaiting
483 observation details to assess potential time to resolution. Further, it
expects closure of inspection and EIR issuance to take about six months. It
expects slight slippage from assumption of early FY19 resolution.
Brokerage: Jefferies | Rating: Hold | Target: INR 520
The global research firm said that
it is building a resolution for Halol in the current quarter. Further, the resolution
remains a key with valuations at 21.5 times FY20 PE. Earnings recovery will be
gradual as investment in specialty to continue.
ANALYSTS EXPECT SUN PHARMA TO FALL UP TO 25%
POST Q3 EARNINGS, AWAIT HALOL RESOLUTION
All brokerage houses barring Motilal Oswal
highlighted in this article are having Reduce to Hold rating and expect the
stock to fall up to 25 percent.
Feb 15, 2018: Brokerage houses are
not positive on Sun Pharmaceutical
Industriesafter its disappointing Q3 earnings and expect the stock
to fall up to 25 percent over next 12 months. The stock fell as much as 1.6
percent in early trade, but then gradually recovered to gain up to 1.8 percent
before closing up 0.5 percent.
The pharma major's profit fell
sharply by 75.2 percent year-on-year to INR 3654.000 million, dragged by
exceptional tax expenses.
"Tax expenses (exceptional)
for the quarter represented estimated impact of INR 5130.200 million on account
of re-measurement of the group's deferred tax assets as a result of the Tax Cut
and Jobs Act enacted in the US on December 2017," the pharma major said in
its filing.
Consolidated revenue from
operations also slipped 16 percent year-on-year to INR 66532.300 million in Q3
due to decline in the US business. Consolidated operating profit degrowth of
INR 40.8 percent year-on-year INR 14534.000 million and margin decline of 910
basis points at 21.8 percent for December quarter 2017 were lower than
CNBC-TV18 poll estimates of INR 14348.000 million and 21.1 percent,
respectively.
All brokerage houses barring
Motilal Oswal highlighted in this article are having Reduce to Hold rating and
expect the stock to fall up to 25 percent.
Brokerage - Edelweiss | Rating - Hold | Target - INR 550
Challenging macro environment,
regulatory woes and endeavour to create a US specialty business are exerting
significant pressure on the business.
They believe two things can
reverse the tide in the short term: 1) clearance of Halol, for which
re-inspection is underway; and 2) approval for and launch of MK-3222 and
Seciera, respectively, in FY19.
The stock has run up 10 percent
over the past few days on the Halol re-inspection news. It is now trading at
around 20 percent premium to the sector at 23x FY20E earnings of INR 25, i.e.,
70 percent growth on FY18 earnings estimates assuming all the upsides from the
re-inspection and the successful launch of the specialty pipeline.
They believe, the risk-reward is
unfavourable, and perceive limited upside from the current level. Therefore,
they downgrade to Hold with revised target price of INR 550.
Brokerage - Motilal Oswal | Rating - Buy | Target - INR 675
US business declined around 36
percent YoY to USD 328 million due to inclusion of sales from Imatinib (six
months’ exclusivity) in the base quarter. Sequential improvement in the US
business can be attributed to re-coup of deferred sales from Q2FY18 and some
expansion in market share.
They expect the stock to remain
under pressure in the near term due to challenges related to growth and
margins. They maintain Buy rating with a target price of INR 675. They cut
FY18/19 EPS estimates by 6/5 percent, building in a slower recovery in the US
business and margin improvement.
Brokerage - Prabhudas Lilladher | Rating - Reduce | Target
- INR 447
US generics are impacted with a)
strong competition in Derma portfolio of Taro, b) competitive intensity in
older generics, and c) supply disruption from Halol. These have resulted in
impacting its core generic sales.
There is no visibility of generic
Glumetza launch in FY18 as management remained cautious of any launch that
invokes recall of products in US. Nevertheless, there are array of competitive
issues those impact sales growth while build-up costs for investments in
generic pipeline and front-end for specialty/NDA products in US need to be
incurred upfront.
The return on specialty and NDA
products are back-ended with expected break-even in FY20. They believe Sun will
be facing higher challenges even meeting their guidance due to likely increase
in overhead costs going forward. There is a chance of further reduction in Taro
and in Lipodox revenue and margins (with launch of DRL generics).
They maintain recommendation to
Reduce and increase target price to INR 447 (from INR 359).
Brokerage - ICICIdirect | Rating - Hold | Target - INR 530
Broadly, the challenges persist on
the generics pricing front in the base business. Unlike other generic players,
the approval momentum is slightly slow in Sun’s case, mainly due to pending
Halol resolution.
However, progress in the
speciality portfolio is promising, which is the key differentiator vis-a-vis
peers. They maintain Hold rating with a new target price of INR 530 based on
22x FY20E EPS of INR 22.4 and INR 38 NPV for Tildrakizumab.
Brokerage - CLSA | Rating - Sell | Target - INR 430
Risks to specialty ramp-up/delay
in Halol clearance is not priced in. Key earnings driver going forward will be
improvement in US revenue.
They retain Sell call with a
target price of INR 430.
Brokerage - Nomura | Rating - Neutral | Target - INR 479
Nomura maintains Neutral call on
the stock with a target price at INR 479 per share. Halol resolution is a key
as it’s factored into stock price.
The approval from USFDA to
Tildrakizumab by March 2018 is also critical.
Brokerage - Morgan Stanley | Rating - Underweight | Target
- INR 448
Morgan Stanley has Underweight call
on the stock with a target price of INR 448. They believe earnings are
bottoming.
Brokerage - Jefferies | Rating - Hold | Target - INR 520
Jefferies has Hold call on Sun
Pharma with increased target price at INR 520 from INR 475 per share.
They build a Halol resolution by
FY18-end & see strong growth in FY19. Company guided for Q4 revenue is
similar to Q3, implying over 15 percent decline in FY18 versus earlier target.
Investment in specialty can limit
near-term earnings recovery. Valuations & Halol re-inspection leave little
room for upside risk.
CMT REPORT (Corruption, Money Laundering & Terrorism]
The Public Notice information has been collected from
various sources including but not limited to: The Courts, India Prisons
Service, Interpol, etc.
1] INFORMATION
ON DESIGNATED PARTY
No records exist designating subject or any of its
beneficial owners, controlling shareholders or senior officers as terrorist or
terrorist organization or whom notice had been received that all financial transactions
involving their assets have been blocked or convicted, found guilty or against
whom a judgement or order had been entered in a proceedings for violating
money-laundering, anti-corruption or bribery or international economic or
anti-terrorism sanction laws or whose assets were seized, blocked, frozen or
ordered forfeited for violation of money laundering or international
anti-terrorism laws.
2] Court
Declaration :
No records exist to suggest that
subject is or was the subject of any formal or informal allegations,
prosecutions or other official proceeding for making any prohibited payments or
other improper payments to government officials for engaging in prohibited
transactions or with designated parties.
3] Asset
Declaration :
No records exist to suggest that the property or assets of
the subject are derived from criminal conduct or a prohibited transaction.
4] Record
on Financial Crime :
Charges or
conviction registered against subject: None
5] Records
on Violation of Anti-Corruption Laws :
Charges or
investigation registered against subject: None
6] Records
on Int’l Anti-Money Laundering Laws/Standards :
Charges or
investigation registered against subject: None
7] Criminal
Records
No
available information exist that suggest that subject or any of its principals
have been formally charged or convicted by a competent governmental authority
for any financial crime or under any formal investigation by a competent
government authority for any violation of anti-corruption laws or international
anti-money laundering laws or standard.
8] Affiliation
with Government :
No
record exists to suggest that any director or indirect owners, controlling
shareholders, director, officer or employee of the company is a government
official or a family member or close business associate of a Government
official.
9] Compensation
Package :
Our
market survey revealed that the amount of compensation sought by the subject is
fair and reasonable and comparable to compensation paid to others for similar
services.
10] Press Report
:
No press reports / filings exists on
the subject.
CORPORATE GOVERNANCE
MIRA INFORM as part of its Due Diligence do provide comments
on Corporate Governance to identify management and governance. These factors
often have been predictive and in some cases have created vulnerabilities to
credit deterioration.
Our Governance Assessment focuses principally on the
interactions between a company’s management, its Board of Directors,
Shareholders and other financial stakeholders.
CONTRAVENTION
Subject is not known to have contravened any existing local
laws, regulations or policies that prohibit, restrict or otherwise affect the
terms and conditions that could be included in the agreement with the subject.
FOREIGN EXCHANGE RATES
|
Currency |
Unit
|
INR |
|
US Dollar |
1 |
INR 65.20 |
|
UK Pound |
1 |
INR 91.49 |
|
Euro |
1 |
INR 80.46 |
INFORMATION DETAILS
|
Information
Gathered by : |
AKY |
|
|
|
|
Analysis
Done by : |
DIV |
|
|
|
|
Report
Prepared by : |
MTN |
SCORE FACTORS
|
DEMERIT POINTS |
|
|
|
--BANK CHARGES |
YES/NO |
YES |
|
--LITIGATION |
YES/NO |
YES |
|
--OTHER ADVERSE INFORMATION |
YES/NO |
NO |
|
MERIT POINTS |
|
|
|
--SOLE DISTRIBUTORSHIP |
YES/NO |
NO |
|
--EXPORT ACTIVITIES |
YES/NO |
NO |
|
--AFFILIATION |
YES/NO |
YES |
|
--LISTED |
YES/NO |
YES |
|
--OTHER MERIT FACTORS |
YES/NO |
YES |
RATING EXPLANATIONS
|
Credit
Rating |
Explanation |
Rating
Comments |
|
A++ |
Minimum Risk |
Business dealings permissible
with minimum risk of default |
|
A+ |
Low Risk |
Business dealings permissible
with low risk of default |
|
A |
Acceptable Risk |
Business dealings permissible
with moderate risk of default |
|
B |
Medium Risk |
Business dealings permissible
on a regular monitoring basis |
|
C |
Medium High Risk |
Business dealings permissible
preferably on secured basis |
|
D |
High Risk |
Business dealing not
recommended or on secured terms only |
|
NB |
New Business |
No recommendation can be done
due to business in infancy stage |
|
NT |
No Trace |
No recommendation can be done as
the business is not traceable |
NB is stated where there is
insufficient information to facilitate rating. However, it is not to be considered
as unfavourable.
This score serves as a reference
to assess SC’s credit risk and to set the amount of credit to be extended. It
is calculated from a composite of weighted scores obtained from each of the
major sections of this report. The assessed factors are as follows:
·
Financial condition covering various ratios
·
Company background and operations size
·
Promoters / Management background
·
Payment record
·
Litigation against the subject
·
Industry scenario / competitor analysis
·
Supplier / Customer / Banker review (wherever available)
This report is issued at your
request without any risk and responsibility on the part of MIRA INFORM
PRIVATE LIMITED (MIPL) or its officials.

