|
|
|
MIRA
INFORM REPORT
|
Report No. : |
500113 |
|
Report Date : |
24.03.2018 |
IDENTIFICATION DETAILS
|
Name : |
GLENMARK PHARMACEUTICALS LIMITED |
|
|
|
|
Registered
Office : |
B/2, Mahalaxmi
Chambers, 22, Bhulabhai Desai Road, Mumbai – 400026, Maharashtra |
|
Tel. No.: |
91-22-24964893/
24964894 |
|
|
|
|
Country : |
India |
|
|
|
|
Financials (as
on) : |
31.03.2017 |
|
|
|
|
Date of
Incorporation : |
18.11.1977 |
|
|
|
|
Com. Reg. No.: |
11-019982 |
|
|
|
|
Capital
Investment / Paid-up Capital : |
INR 282.170 Million |
|
|
|
|
CIN No.: [Company Identification
No.] |
L24299MH1977PLC019982 |
|
|
|
|
TIN No.: |
27030000077 |
|
|
|
|
IEC No.: [Import-Export Code No.] |
0388014482 |
|
|
|
|
TAN No.: [Tax Deduction &
Collection Account No.] |
MUMG07883B |
|
|
|
|
GSTN : [Goods & Service Tax
Registration No.] |
Not Divulged |
|
|
|
|
PAN No.: [Permanent Account No.] |
AAACG2207L |
|
|
|
|
Legal Form : |
A Public Limited Liability Company. The Company’s Shares are Listed on
the Stock Exchanges. |
|
|
|
|
Line of Business
: |
The Company is primarily engaged in the business of development, manufacture and marketing of pharmaceutical products. The Company also markets active pharmaceutical ingredients. (Registered activity) |
|
|
|
|
No. of Employees
: |
12967 (Approximately) |
RATING & COMMENTS
(Mira Inform has adopted New Rating mechanism w.e.f. 23rd
January 2017)
|
MIRA’s Rating : |
A++ |
|
Credit Rating |
Explanation |
Rating Comments |
|
A++ |
Minimum Risk |
Business dealings permissible with minimum
risk of default |
|
Maximum Credit Limit : |
USD 270000000 |
|
|
|
|
Status : |
Excellent |
|
|
|
|
Payment Behaviour : |
Regular |
|
|
|
|
Litigation : |
Exists |
|
|
|
|
Comments : |
Subject was incorporated in the year 1977 and it manufactures and markets pharmaceutical formulation and active pharmaceutical ingredients, which it market in India and in the International market. It also undertakes research and development on new chemical entities and new biological entities. It manufacturing facilities are at Nashik, Sholapur and Pune (all in Maharashtra), Ankleshwar (Gujarat), Bardez (Goa) and Solan (Himachal Pradesh). As per financials of March 2017, the company has registered a growth of 30.51% in its revenue as compared to its previous year’s revenue and has reported good profit margin of 26.44% under review. Rating takes into consideration the company’s established track record of business operations marked by healthy net worth base along with low solvency indicators due to zero debt balance sheet and good liquidity position. Rating also takes into account the strong position in the fast-growing chronic-therapeutic segments in India and its increasing presence in the International generics market. Share are quoted very high on Stock Exchanges. (Share are traded at a price of INR 534.95 against its face value of INR 1.) However, rating strength is partially offset by its long working capital cycle and its exposure to intensifying competition in the US generics market. As per unaudited quarterly financials of December 2017, the company has achieved a revenue of INR 15,565.49 million and has reported decent profit margin of 12.47%. Payments seem to be regular as per commitments. In view of aforesaid, the company can be considered good for business dealings at normal trade terms and conditions. |
NOTES :
Any query related to this report can be made
on e-mail : infodept@mirainform.com
while quoting report number, name and date.
ECGC Country Risk Classification List
|
Country Name |
Previous Rating (30.09.2017) |
Current Rating (31.12.2017) |
|
India |
A1 |
A1 |
|
Risk Category |
ECGC Classification |
|
Insignificant |
A1 |
|
Low Risk |
A2 |
|
Moderately Low
Risk |
B1 |
|
Moderate Risk |
B2 |
|
Moderately High
Risk |
C1 |
|
High Risk |
C2 |
|
Very High Risk |
D |
EXTERNAL AGENCY RATING
|
Rating Agency Name |
INDIA RATING |
|
Rating |
Long Term Rating = AA- |
|
Rating Explanation |
High degree of safety and low credit risk. |
|
Date |
15.03.2017 |
|
Rating Agency Name |
INDIA RATING |
|
Rating |
Short Term Rating = A1+ |
|
Rating Explanation |
Very strong degree of safety and carry lowest credit risk. |
|
Date |
15.03.2017 |
RBI DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available RBI Defaulters’ list.
EPF (Employee Provident Fund) DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available EPF (Employee Provident Fund) Defaulters’ list as of
31-03-2016.
BIFR (Board for Industrial & Financial Reconstruction)
LISTING STATUS
Subject’s name is
not listed as a Sick Unit in the publicly available BIFR (Board for Industrial
& Financial Reconstruction) list as of 24.03.2018
IBBI (Insolvency and Bankruptcy Board of India) LISTING STATUS
Subject’s name is not listed in the publicly
available IBBI (Insolvency and Bankruptcy Board of India) list as of report
date.
INFORMATION DENIED
MANAGEMENT NON-COOPERATIVE: Tel. No.: 91-22-40189999
Tel. No.: 91-22-24964893 / 94 / 95 / 96 / 56549999 – Not Exist
LOCATIONS
|
Registered/ Administrative Office : |
B/2, Mahalaxmi Chambers, 22, Bhulabhai
Desai Road, Mumbai – 400026, Maharashtra, India |
|
Tel. No.: |
91-22-24964893/ 24964894/ 24964895/
24964896/ 56549999/ 55902491/ 92 |
|
Fax No.: |
91-22-24932648/ 23512177/ 23519652 |
|
E-Mail : |
|
|
Website : |
|
|
|
|
|
Corporate Office : |
Gelnmark House, HDO – Corporate Building
Wing A, B D Sawant Marg, Chakala, Off Western Express Highway, Andheri
(East), Mumbai – 400099, Maharashtra, India |
|
Tel. No.: |
91-22-40189999 |
|
Fax No.: |
91-22-40189986/88 / 4018 9990 |
|
Email : |
|
|
|
|
|
Manufacturing Facilities : |
Formulations · E – 37, MIDC Industrial Area, D-Road, Satpur, Nasik – 422007, Maharashtra, India · Plot No. 7 and 9, Colvale Industrial Estate, Bardez – 403 115, Goa, India · Unit – I, Village-Kishanpura, Baddi Nalagarh Road, Tehsil Nalagarh, District Solan, Baddi – 174 101, Himachal Pradesh, India · Business Unit II, Village Bhattanwala, PO Rajpura, Nalagarh District Solan, Himachal Pradesh, India · Unit - III, Village Kishanpura, Baddi-Nalagarh Road, Dist. - Solan – 174101, Himachal Pradesh, India · Plot No. 2, Phase-II, Pharma Zone, Special Economic Zone Area, Pitampur, Indore – 454 775, Madhya Pradesh, India · Rua Frei Liberato De Gries, 548, Jardim Arpoadar, CEP: 05572-210, Sao Paulo, Brazil · Glenmark Pharmaceuticals S.R.O., Fibichova 143, 56617, Vysoke Myto, Czech Republic · Calle 9 Ing Meyer Oks N 593, Parque Industrial Pilar, B1629MX Buenos Aires, Argentina · Growth Centre, Samlik-Marchak, District – East Sikkim, Sikkim. · Plot No. B-25, Five Star MIDC, Shendra, District - Aurangabad, Maharashtra · 4147 Goldmine Road, Monroe, NC 28110, USA · Fibichova 143, 56617, Vysoke Myto, Czech Republic · Chemin de la Combeta 5, 2300 La Chaux-de-fonds, Switzerland API ·
3109-C, GIDC Industrial Estate, Ankleshwar,
District Bharuch - 393 002, · Plot No. B-25, Five Star MIDC, Shendra, Dist. - Aurangabad, Maharashtra, India ·
Plot No. 163-165/170-172, Chandramouli Industrial
Estate, Mohal Bazarpeth, ·
Plot No. A80, MIDC Area, Kurkumbh, Daund, Pune
– 413 802, ·
Z-103 I, Dahej SEZ, Dahej District, Bharuch,
Gujarat, Indi |
|
|
|
|
R and D Centers : |
·
Plot
No. A-607, TTC Industrial Area, MIDC, Mahape, Vashi, Navi Mumbai - 400 705, ·
Chemindela
Combeta 5, 2300 La Chaux-de-fonds, Switzerland ·
Plot
No. C 152, MIDC Sinnar Industrial Area, ·
Plot
No. M4, Taloja Industrial Area, MIDC Taloja, Takula Panvel – 410 208,
District – Raigad, |
|
|
|
|
Clinical
Research Centre : |
·
Plot
No. D 508, TTC Industrial Estate, MIDC, Turbhe, Navi Mumbai – 400705,
Maharashtra, India ·
Building
2, Croxley Green Business Park, Merlins Meadow, Watford, Hertfordshire, UK |
DIRECTORS
As on 31.03.2017
|
Name : |
Blanche Elizabeth Saldanha |
|
Designation : |
Director |
|
Address : |
Windmere Co-Operative Housing Society Limited,
Flat No. 2, 236 A, B. J. Road, Bandra (West), Mumbai - 400050, Maharashtra,
India |
|
Qualification : |
Graduated in B.Sc., B.Ed., from Bombay University |
|
Date of Appointment : |
14.08.2009 |
|
DIN No.: |
00007671 |
|
|
|
|
Name : |
Sridhar Gorthi |
|
Designation : |
Director |
|
Address : |
1002, 10th Floor, June Blossoms, Manuel
Gonsalves Road, Bandra (West), Mumbai-400050, Maharashtra, India |
|
Qualification : |
B.A., L.L.B. (Hons.) |
|
Date of Appointment : |
26.04.2005 |
|
DIN No.: |
00035824 |
|
|
|
|
Name : |
Julio Francis Ribeiro |
|
Designation : |
Director |
|
Address : |
5 Floor Room 51 Sagar Tarang Building, 15A
K A G Khan Road, Mumbai - 400030, Maharashtra, India |
|
Date of Appointment : |
26.08.1999 |
|
DIN No.: |
00047630 |
|
|
|
|
Name : |
Glenn Mario Saldanha |
|
Designation : |
Managing Director |
|
Address : |
Flat No.91, 9th Floor, Ritu Apartment, 208
B J Road, Bandra West, Mumbai - 400050, Maharashtra, India |
|
Qualification : |
MBA from New York University’s Leonard N. Stern School of Business (US). |
|
Date of Appointment : |
12.10.1998 |
|
DIN No.: |
00050607 |
|
|
|
|
Name : |
Rajesh Vasudeo Desai |
|
Designation : |
Whole-Time Director |
|
Address : |
102-D Wing, Shrinath Bhuvan, Nicholus Wadi,
Yashwant Tawade Path, Dahisar (West), Mumbai - 400068, Maharashtra, India |
|
Qualification : |
Science graduate from Bombay University and a Chartered Accountant from Institute of Chartered Accountants of India |
|
Date of Appointment : |
09.11.2011 |
|
DIN No.: |
00050838 |
|
|
|
|
Name : |
Brian William Tempest |
|
Designation : |
Director |
|
Address : |
South Park Farm, Grayswood Road, Haslemere,
Surrey, GU272DJ, United Kingdom |
|
Qualification : |
PHD in Chemistry from Lancaster University |
|
Date of Appointment : |
30.01.2012 |
|
DIN No.: |
00101235 |
|
|
|
|
Name : |
Milind Shripad Sarwate |
|
Designation : |
Additional Director |
|
Address : |
E-201/202, Sita Vihar, Near Damani Estate,
L.B.S. Marg, Naupada, Thane-400602, Maharashtra, India |
|
Qualification : |
Chartered Accountant |
|
Date of Appointment : |
29.10.2015 |
|
DIN No.: |
00109854 |
|
|
|
|
Name : |
Cherylann Maria Pinto |
|
Designation : |
Whole-Time Director |
|
Address : |
Flat No 6 Windemere Co-Operative Housing Society,
236 A Byramji Jeejibhoy Road Bandra West, Mumbai-400050, Maharashtra, India |
|
Qualification : |
Graduate in Pharmacy from the University Department of Chemical Technology in Mumbai. |
|
Date of Appointment : |
06.10.1999 |
|
DIN No.: |
00111844 |
|
|
|
|
Name : |
Devendra Raj Mehta |
|
Designation : |
Director |
|
Address : |
B - 5 Mahavir Udyan Marg, Bajaj Nagar,
Jaipur-302015, Rajasthan, India |
|
Qualification : |
Graduated in Arts and Law from Rajasthan University. |
|
Date of Appointment : |
14.08.2009 |
|
DIN No.: |
01067895 |
|
|
|
|
Name : |
Bernard Hubert Munos |
|
Designation : |
Director |
|
Address : |
301, Kessler Boulevard West Drive,
Indianapolis, Indiana, Na, United States of America |
|
Qualification : |
MBA from Stanford University |
|
Date of Appointment : |
30.01.2012 |
|
DIN No.: |
05198283 |
|
|
|
|
Name : |
Murali Neelakantan |
|
Designation : |
Wholetime Director |
|
Address : |
3702, B Wing, Lodha Bellissimo, Apollo Hills Compound, N M
Joshi Marg, M Ahalaxmi, Mumbai – 400011, Maharashtra, India |
|
Date of Appointment : |
11.05.2017 |
|
DIN No.: |
02453014 |
KEY EXECUTIVES
|
Name : |
Mr. Subramani Vaidyanathan |
|
Designation : |
Chief Finance Officer |
|
Address : |
6/107 Mamta Garodia Nagar, Ghatkopar, Mumbai – 400077, Maharashtra, India |
|
Date of Appointment : |
16.11.2017 |
|
PAN No.: |
AAGPM5727N |
|
|
|
|
Name : |
Mr. Harish Vinayak Kuber |
|
Designation : |
Company Secretary |
|
Address : |
F 314 Parmar Co-Operative Housing Society Limited,
Paranjape B Scheme Subhash Road, Vile Parle, Mumbai 400057, Maharashtra, India |
|
Date of Appointment : |
02.02.2017 |
|
PAN No.: |
ABRPK8790R |
MAJOR SHAREHOLDERS / SHAREHOLDING PATTERN
As on December 2017
|
Category
of shareholder |
No.
of fully paid up equity shares held |
Shareholding
as a % of total no. of shares |
|
|
(A) Promoter & Promoter Group |
131308786 |
46.54 |
|
|
(B) Public |
150859370 |
53.46 |
|
|
Grand
Total |
282168156 |
100.00 |

Statement showing
shareholding pattern of the Promoter and Promoter Group
|
Category of
shareholder |
No. of fully
paid up equity shares held |
Shareholding as
a % of total no. of shares |
|
|
A1) Indian |
0.00 |
||
|
Individuals/Hindu
undivided Family |
131308786 |
46.54 |
|
|
CHERYLANN MARIA PINTO |
719305 |
0.25 |
|
|
ROBIN JOSEPH PINTO |
476000 |
0.17 |
|
|
SALDANHA FAMILY TRUST |
128241936 |
45.45 |
|
|
BLANCHE ELIZABETH SALDANHA |
1035122 |
0.37 |
|
|
NEHA SALDANHA |
6000 |
0.00 |
|
|
GLENN MARIO SALDANHA |
830423 |
0.29 |
|
|
Sub
Total A1 |
131308786 |
46.54 |
|
|
A2) Foreign |
0.00 |
||
|
A=A1+A2 |
131308786 |
46.54 |
Statement showing
shareholding pattern of the Public shareholder
|
Category &
Name of the Shareholders |
No. of fully
paid up equity shares held |
Shareholding %
calculated as per SCRR, 1957 As a % of (A+B+C2) |
|
|
B1)
Institutions |
0 |
0.00 |
|
|
Mutual
Funds/ |
9018112 |
3.20 |
|
|
Alternate
Investment Funds |
120000 |
0.04 |
|
|
Foreign
Portfolio Investors |
87543416 |
31.03 |
|
|
HSBC POOLED INVESTMENT FUND -
HSBC POOLED ASIA |
6985548 |
2.48 |
|
|
FRANKLIN TEMPLETON INVESTMENT
FUND |
5148698 |
1.82 |
|
|
NEW HORIZON OPPORTUNITIES
MASTER FUND |
4000000 |
1.42 |
|
|
ARANDA INVESTMENTS (MAURITIUS)
PTE LTD |
11261010 |
3.99 |
|
|
Financial
Institutions/ Banks |
7864151 |
2.79 |
|
|
LIC OF INDIA |
3688999 |
1.31 |
|
|
Any
Other (specify) |
42973 |
0.02 |
|
|
Sub
Total B1 |
104588652 |
37.07 |
|
|
B2)
Central Government/ State Government(s)/ President of India |
0 |
0.00 |
|
|
B3)
Non-Institutions |
0 |
0.00 |
|
|
Individual
share capital upto INR 0.200 Million |
26692007 |
9.46 |
|
|
Individual
share capital in excess of INR 0.200 Million |
6135443 |
2.17 |
|
|
NBFCs
registered with RBI |
20695 |
0.01 |
|
|
Any
Other (specify) |
13422573 |
4.76 |
|
|
Trusts |
1804316 |
0.64 |
|
|
NRI |
1603949 |
0.57 |
|
|
Clearing Members |
998819 |
0.35 |
|
|
NRI – Non- Repat |
376756 |
0.13 |
|
|
Bodies Corporate |
7833315 |
2.78 |
|
|
IEPF |
805418 |
0.29 |
|
|
Sub
Total B3 |
46270718 |
16.40 |
|
|
B=B1+B2+B3 |
150859370 |
53.46 |
BUSINESS DETAILS
|
Line of Business : |
The Company is primarily engaged in the business of development, manufacture and marketing of pharmaceutical products. The Company also markets active pharmaceutical ingredients. (Registered activity) |
||||
|
|
|
||||
|
Products : |
|
||||
|
|
|
||||
|
Brand Names : |
Not Divulged |
||||
|
|
|
||||
|
Agencies Held : |
Not Divulged |
||||
|
|
|
||||
|
Exports : |
Not Divulged |
||||
|
|
|
||||
|
Imports : |
Not Divulged |
||||
|
|
|
||||
|
Terms : |
Not Divulged |
PRODUCTION STATUS NOT AVAILABLE
GENERAL INFORMATION
|
Suppliers : |
|
||||||||||||||||||||||
|
|
|
||||||||||||||||||||||
|
Customers : |
|
||||||||||||||||||||||
|
|
|
||||||||||||||||||||||
|
No. of Employees : |
12967 (Approximately) |
||||||||||||||||||||||
|
|
|
||||||||||||||||||||||
|
Bankers : |
|
||||||||||||||||||||||
|
|
|
||||||||||||||||||||||
|
Facilities : |
Note: Short-term
borrowings Working Capital Facilities are secured by hypothecation of stocks of raw materials, packing materials, finished goods, work-in-process, receivables and equitable mortgage on fixed assets at certain locations. The Company has not defaulted on repayment of loan and interest during the year. The Company has taken working capital facility / term loans from banks at interest rates ranging between 0.60 % to 9.70 % p.a. |
|
Auditors : |
|
|
Name : |
Walker, Chandiok and Company LLP Chartered Accountants |
|
Address : |
21st Floor, DLF Square, Jacaranda Marg, DLF Phase II,
Goregaon, Mumbai - 122002, Maharashtra, India |
|
Tel. No.: |
91-124-4628000 |
|
Fax No.: |
91-124-4628001 |
|
|
|
|
Cost Auditors: |
|
|
Name : |
Sevekari, Khare and Associates, Cost Accountants |
|
Address : |
Mumbai, Maharashtra, India |
|
|
|
|
Solicitor: |
|
|
Name : |
Trilegal |
|
Address : |
Mumbai, Maharashtra, India |
|
|
|
|
Memberships : |
Not Available |
|
|
|
|
Collaborators : |
Not Available |
|
|
|
|
Subsidiary companies: |
|
|
|
|
|
Enterprise over
which key managerial personnel excercise significant influence : |
|
|
|
|
|
Related party
relationships where transactions have taken place during the year Subsidiary
Companies / Enterprise over which key managerial personnel exercise
significant influence |
|
CAPITAL STRUCTURE
After 29.09.2017
Authorised Capital : INR 2770.000
Million
Issued, Subscribed & Paid-up Capital : INR 282.168 Million
As on 31.03.2017
Authorised Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
2370000000 |
Equity Shares |
Re.1/- each |
INR 2370.000 Million |
|
4000000 |
Cumulative Redeemable Non-Convertible Preference Shares |
INR100/- each |
INR 400.000 Million |
|
|
Total |
|
INR 2770.000
Million |
Issued, Subscribed & Paid-up Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
282158156 |
Equity Shares |
INR 1/- each |
INR 282.160
Million |
|
10000 |
Add: Issued during the year - Under the employee Stock Option Scheme,
2003 (ESOS) |
INR 1/- each |
INR 0.010
Million |
|
|
Total |
|
INR 282.170 Million |
|
|
As at 31 March
2016 |
|
|
List of
shareholders holding more than 5% shares |
% of Holding |
of Shares |
|
Saldanha Family Trust |
45.45 |
128,241,936 |
As at 31 March 2017, pursuant to Employee Stock Option Scheme 2003, 47,000 options were outstanding, which upon exercise are convertible into equivalent number of equity shares . Pursuant to Employee Stock Options Scheme 2016, 619,757 options were outstanding, which upon exercise are convertible into equivalent number of equity shares.
Right, Preference and
restriction on shares
The Company presently has only one class of ordinary equity shares. For all matters submitted to vote in the shareholders meeting, every holder of ordinary equity shares, as reflected in the records of the Company on the date of the shareholders’ meeting, has one vote in respect of each share held. All shares are equally eligible to receive dividends and the repayment of capital in the event of liquidation of the Company.
In the period of five years immediately preceding 31 March 2017, the Company has not allotted any shares as fully paid up pursuant to contracts without payment being received in cash. Further, the Company has neither issued bonus shares nor bought back any shares during the aforementioned period.
Employee Stock Option
Scheme, 2003 and 2016 (ESOS)
The Company has formulated an Employee Stock Option Scheme 2003 and Employee Stock Option Scheme 2016 (‘ESOS’) namely ESOS 2003 and ESOS 2016 under which it has made grants on various dates from time to time. Each grant has a vesting period which varies from 1 - 2 years and up to 4 - 6 years from the date of grant depending on the terms of the grant. The grants are made at the market price of the equity shares of the Company on either the date of the grant or the closing price of the date prior to the day of the grant or the price decided by the Nomination & Remuneration Committee of the Board. As at 31 March 2017, pursuant to ESOS 2003, 47,000 options were outstanding, which upon exercise are convertible into upon exercise are convertible into equivalent number of equity shares.
The aggregate share
options and weighted average exercise price under the above mentioned plan are
as follows:
|
|
2017 |
|
|
|
Number* |
Weighted average Price in Rupees |
|
Outstanding at the beginning of the year |
84500 |
279.99 |
|
Granted during the year |
640695 |
800.00 |
|
Forfeited during the year |
(48438) |
515.79 |
|
Exercised during the year * |
(10000) |
263.89 |
|
Outstanding at the end of the year |
666757 |
762.78 |
All share based employee payments would be settled in equity. The Company has no legal or constructive obligation to repurchase or settle the options.
The fair value of
options granted are determined using the Black-Scholes valuation model.
Significant inputs into the calculation are:
|
Particulars |
31 March 2017 |
|
Share price (INR)* |
215.85 – 800.00 |
|
Exercise price (INR)* |
215.85 – 800.00 |
|
Weighted average
volatility rate |
30% - 60% |
|
Dividend payout |
200% |
|
Risk free rate |
7.70%-9.00% |
|
Average
remaining life |
1-52 months |
*All figures have been accordingly adjusted for
- Split of face value from INR 10 to INR 2 in October 2003
- 1:1 bonus issue in April 2005 and split of face value from INR 2 to INR 1 in September 2007.
The underlying expected volatility was determined by reference to historical data, adjusted for unusual share price movements. No special features inherent to the options granted were incorporated into the measurement of fair value.
FINANCIAL DATA
[all figures are
in INR Million]
ABRIDGED
BALANCE SHEET
|
SOURCES
OF FUNDS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
|
|
|
|
|
I.
EQUITY AND LIABILITIES |
|
|
|
|
(1)Shareholders' Funds |
|
|
|
|
(a) Share Capital |
282.170 |
282.160 |
271.290 |
|
(b) Merger consideration,
pending allotment |
0.000 |
0.000 |
0.020 |
|
(c) Reserves & Surplus |
94084.020 |
73377.460 |
49249.220 |
|
(d) Money received against
share warrants |
0.000 |
0.000 |
0.000 |
|
|
|
|
|
|
(2) Share Application money
pending allotment |
0.000 |
0.000 |
0.000 |
|
Total
Shareholders’ Funds (1) + (2) |
94366.190 |
73659.620 |
49520.530 |
|
|
|
|
|
|
(3) Non-Current Liabilities |
|
|
|
|
(a) long-term borrowings |
25893.460 |
0.000 |
0.000 |
|
(b) Deferred tax liabilities
(Net) |
0.000 |
0.000 |
1323.970 |
|
(c) Other long term
liabilities |
24.050 |
46.950 |
1219.210 |
|
(d) long-term provisions |
0.000 |
0.000 |
0.000 |
|
Total
Non-current Liabilities (3) |
25917.510 |
46.950 |
2543.180 |
|
|
|
|
|
|
(4) Current Liabilities |
|
|
|
|
(a) Short term borrowings |
1871.890 |
7874.180 |
3475.990 |
|
(b) Trade payables |
14670.900 |
16259.530 |
15667.860 |
|
(c) Other current liabilities |
2765.230 |
2997.770 |
3786.990 |
|
(d) Short-term provisions |
413.740 |
292.050 |
1513.800 |
|
Total
Current Liabilities (4) |
19721.760 |
27423.530 |
24444.640 |
|
|
|
|
|
|
TOTAL |
140005.460 |
101130.100 |
76508.350 |
|
|
|
|
|
|
II.
ASSETS |
|
|
|
|
(1) Non-current assets |
|
|
|
|
(a) Fixed Assets |
|
|
|
|
(i) Tangible assets |
14704.960 |
13219.240 |
11524.840 |
|
(ii) Intangible Assets |
1258.740 |
1160.800 |
781.940 |
|
(iii) Capital work-in-progress |
2351.350 |
2609.320 |
2864.770 |
|
(iv) Intangible assets under
development |
355.240 |
151.310 |
77.120 |
|
(b) Non-current Investments |
18666.990 |
18584.250 |
16595.180 |
|
(c) Deferred tax assets (net) |
5940.640 |
4011.580 |
0.000 |
|
(d) Long-term Loan and Advances |
36426.840 |
14473.240 |
8546.310 |
|
(e) Other Non-current assets |
792.400 |
641.050 |
138.630 |
|
Total
Non-Current Assets |
80497.160 |
54850.790 |
40528.790 |
|
|
|
|
|
|
(2) Current assets |
|
|
|
|
(a) Current investments |
0.000 |
0.000 |
0.000 |
|
(b) Inventories |
11450.550 |
9680.010 |
7366.320 |
|
(c) Trade receivables |
38794.040 |
30576.550 |
24408.310 |
|
(d) Cash and cash equivalents |
2508.820 |
742.430 |
485.470 |
|
(e) Short-term loans and
advances |
0.000 |
0.000 |
3511.090 |
|
(f) Other current assets |
6754.890 |
5280.320 |
208.370 |
|
Total
Current Assets |
59508.300 |
46279.310 |
35979.560 |
|
|
|
|
|
|
TOTAL |
140005.460 |
101130.100 |
76508.350 |
PROFIT
& LOSS ACCOUNT
|
|
PARTICULARS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
|
SALES |
|
|
|
|
|
Income |
80955.000 |
62030.810 |
50856.020 |
|
|
Other Income |
1482.390 |
1172.830 |
849.410 |
|
|
TOTAL
|
82437.390 |
63203.640 |
51705.430 |
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
Cost of Materials Consumed |
22420.130 |
17922.430 |
15640.260 |
|
|
Purchases of Stock-in-Trade |
2669.960 |
2199.750 |
1739.540 |
|
|
Changes in inventories of
finished goods, work-in-progress and Stock-in-Trade |
(835.170) |
(735.510) |
(696.530) |
|
|
Employees benefits expense |
9144.710 |
7662.540 |
6622.540 |
|
|
Exceptional items |
2364.510 |
0.000 |
1687.370 |
|
|
Other expenses |
18568.950 |
17328.810 |
12900.310 |
|
|
TOTAL |
54333.090 |
44378.020 |
37893.490 |
|
|
|
|
|
|
|
|
PROFIT/
(LOSS) BEFORE INTEREST, TAX,
DEPRECIATION AND AMORTISATION |
28104.300 |
18825.620 |
13811.940 |
|
|
|
|
|
|
|
Less |
FINANCIAL
EXPENSES |
1526.020 |
362.240 |
301.890 |
|
|
|
|
|
|
|
|
PROFIT
/ (LOSS) BEFORE TAX, DEPRECIATION AND AMORTISATION |
26578.280 |
18463.380 |
13510.050 |
|
|
|
|
|
|
|
Less/
Add |
DEPRECIATION/
AMORTISATION |
1049.320 |
998.100 |
1194.600 |
|
|
|
|
|
|
|
|
PROFIT/
(LOSS) BEFORE TAX |
25528.960 |
17465.280 |
12315.450 |
|
|
|
|
|
|
|
Less |
TAX |
4122.880 |
2622.610 |
2240.200 |
|
|
|
|
|
|
|
|
PROFIT/
(LOSS) AFTER TAX |
21406.080 |
14842.670 |
10075.250 |
|
|
|
|
|
|
|
|
EARNINGS
IN FOREIGN CURRENCY |
|
|
|
|
|
Export of goods calculated on
FOB basis |
|
|
29720.470 |
|
|
Sale of Service |
|
|
634.050 |
|
|
Guarantee commission |
|
|
329.660 |
|
|
Interest on loan to
subsidiaries |
|
|
263.440 |
|
|
Royalty Income |
|
6.630 |
|
|
|
Other operating income |
|
|
344.950 |
|
|
Miscellaneous receipt |
|
0.000 |
|
|
|
TOTAL
EARNINGS |
56151.570 |
36944.730 |
31299.200 |
|
|
|
|
|
|
|
|
IMPORTS |
|
|
|
|
|
Capital goods |
NA |
NA |
710.960 |
|
|
Materials |
NA |
NA |
3969.300 |
|
|
TOTAL
IMPORTS |
NA |
NA |
4680.260 |
|
|
|
|
|
|
|
|
Earnings
/ (Loss) Per Share (INR) |
75.86 |
52.87 |
37.14 |
CURRENT MATURITIES OF LONG TERM DEBT DETAILS
|
Particulars |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Current Maturities of Long term debt |
NA |
NA |
NA |
|
Cash generated from operations |
15242.340 |
7302.600 |
3855.780 |
|
Net cash flow from operating activity |
8777.240 |
3547.540 |
1451.080 |
QUARTERLY RESULTS
|
Particulars |
30.06.2017 |
30.09.2017 |
31.12.2017 |
|
Audited / Unaudited |
Unaudited |
Unaudited |
Unaudited |
|
|
1ST Quarter |
2nd Quarter |
3rd Quarter |
|
Net Sales |
15477.780 |
16598.120 |
15565.490 |
|
Total Expenditure |
11948.360 |
12623.560 |
12897.530 |
|
PBIDT (Excl OI) |
3529.420 |
3974.560 |
2667.960 |
|
Other Income |
425.880 |
563.040 |
372.320 |
|
Operating Profit |
3955.300 |
4537.600 |
3040.280 |
|
Interest |
457.370 |
474.410 |
469.990 |
|
Exceptional Items |
NA |
NA |
NA |
|
PBDT |
3497.930 |
4063.190 |
2570.290 |
|
Depreciation |
299.410 |
288.930 |
295.530 |
|
Profit Before Tax |
3198.520 |
3774.260 |
2274.760 |
|
Tax |
490.14 |
495.470 |
333.600 |
|
Provisions and contingencies |
NA |
NA |
NA |
|
Profit After Tax |
2708.380 |
3278.790 |
1941.160 |
|
Extraordinary Items |
NA |
NA |
NA |
|
Prior Period Expenses |
NA |
NA |
NA |
|
Other Adjustments |
NA |
NA |
NA |
|
Net Profit |
2708.380 |
3278.790 |
1941.160 |
KEY
RATIOS
EFFICIENCY RATIOS
|
PARTICULARS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Average Collection Days (Sundry Debtors / Income * 365 Days) |
174.91 |
179.92 |
175.18 |
|
|
|
|
|
|
Account Receivables Turnover (Income / Sundry
Debtors) |
2.09 |
2.03 |
2.08 |
|
|
|
|
|
|
Average Payment Days (Sundry Creditors
/ Purchases * 365 Days) |
213.43 |
294.93 |
329.05 |
|
|
|
|
|
|
Inventory Turnover (Operating Income
/ Inventories) |
2.45 |
1.94 |
1.88 |
|
|
|
|
|
|
Asset Turnover (Operating Income
/ Net Fixed Assets) |
1.51 |
1.10 |
0.91 |
LEVERAGE RATIOS
|
PARTICULARS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Debt Ratio ((Borrowing + Current Liabilities) / Total
Assets) |
0.33 |
0.27 |
0.32 |
|
|
|
|
|
|
Debt Equity Ratio (Total Liability
/ Networth) |
0.29 |
0.11 |
0.07 |
|
|
|
|
|
|
Current Liabilities to Networth (Current
Liabilities / Net Worth) |
0.21 |
0.37 |
0.49 |
|
|
|
|
|
|
Fixed Assets to Networth (Net Fixed Assets
/ Networth) |
0.20 |
0.23 |
0.31 |
|
|
|
|
|
|
Interest Coverage Ratio (PBIT / Financial
Charges) |
18.42 |
51.97 |
45.75 |
PROFITABILITY RATIOS
|
PARTICULARS |
|
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Net Profit Margin ((PAT / Sales) *
100) |
% |
26.44 |
23.93 |
19.81 |
|
|
|
|
|
|
|
Return on Total Assets ((PAT / Total
Assets) * 100) |
% |
15.29 |
14.68 |
13.17 |
|
|
|
|
|
|
|
Return on Investment (ROI) ((PAT / Networth)
* 100) |
% |
22.68 |
20.15 |
20.35 |
SOLVENCY RATIOS
|
PARTICULARS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Current Ratio (Current Assets / Current Liabilities) |
3.02 |
1.69 |
1.47 |
|
|
|
|
|
|
Quick Ratio ((Current Assets
– Inventories) / Current Liabilities) |
2.44 |
1.33 |
1.17 |
|
|
|
|
|
|
G-Score Ratio Financial (Networth / Total
Assets) |
0.67 |
0.73 |
0.65 |
|
|
|
|
|
|
G-Score Ratio Debt (Debts / Equity
Capital) |
98.40 |
27.91 |
12.81 |
|
|
|
|
|
|
G-Score Ratio Liquidity (Total Current
Assets / Total Current Liabilities) |
3.02 |
1.69 |
1.47 |
Total Liability = Short-term Debt + Long-term
Debt + Current Maturities of Long-term debts
STOCK PRICES
|
Face Value |
INR 1.00/- |
|
Market Value |
INR 535.90/- |
FINANCIAL ANALYSIS
[all figures are
INR Million]
DEBT EQUITY RATIO
|
Particular |
31.03.2015 |
31.03.2016 |
31.03.2017 |
|
|
INR
In Million |
INR
In Million |
INR
In Million |
|
Share Capital |
271.290 |
282.160 |
282.170 |
|
Reserves & Surplus |
49249.220 |
73377.460 |
94084.020 |
|
Merger consideration, pending allotment |
0.020 |
0.000 |
0.000 |
|
Net
worth |
49520.530 |
73659.620 |
94366.190 |
|
|
|
|
|
|
Long Term borrowings |
0.000 |
0.000 |
25893.460 |
|
Short Term borrowings |
3475.990 |
7874.180 |
1871.890 |
|
Total
borrowings |
3475.990 |
7874.180 |
27765.350 |
|
Debt/Equity
ratio |
0.070 |
0.107 |
0.294 |

YEAR-ON-YEAR GROWTH
|
Year
on Year Growth |
31.03.2015 |
31.03.2016 |
31.03.2017 |
|
|
INR
In Million |
INR
In Million |
INR
In Million |
|
Sales
|
50856.020 |
62030.810 |
80955.000 |
|
|
|
21.973 |
30.508 |

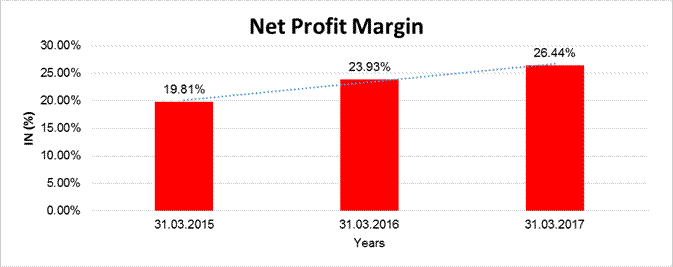
NET PROFIT MARGIN
|
Net
Profit Margin |
31.03.2015 |
31.03.2016 |
31.03.2017 |
|
|
INR
In Million |
INR
In Million |
INR
In Million |
|
Sales
|
50856.020 |
62030.810 |
80955.000 |
|
Profit |
10075.250 |
14842.670 |
21406.080 |
|
|
19.81% |
23.93% |
26.44% |
ABRIDGED
BALANCE SHEET – (CONSOLIDATED)
|
SOURCES
OF FUNDS |
|
31.03.2017 |
31.03.2016 |
|
|
|
|
|
|
I.
EQUITY AND LIABILITIES |
|
|
|
|
(1)Shareholders' Funds |
|
|
|
|
(a) Share Capital |
|
282.170 |
282.160 |
|
(b) Reserves & Surplus |
|
44643.080 |
36014.220 |
|
(c) Money received against
share warrants |
|
0.000 |
0.000 |
|
|
|
|
|
|
(2) Non-controlling interests |
|
(4.230) |
(3.010) |
|
Total
Shareholders’ Funds (1) + (2) |
|
44921.020 |
36293.370 |
|
|
|
|
|
|
(3) Non-Current Liabilities |
|
|
|
|
(a) long-term borrowings |
|
45363.390 |
24872.970 |
|
(b) Deferred tax liabilities
(Net) |
|
0.000 |
0.000 |
|
(c) Other long term
liabilities |
|
327.430 |
769.900 |
|
(d) long-term provisions |
|
0.000 |
0.000 |
|
Total
Non-current Liabilities (3) |
|
45690.820 |
25642.870 |
|
|
|
|
|
|
(4) Current Liabilities |
|
|
|
|
(a) Short term borrowings |
|
1871.890 |
7874.180 |
|
(b) Trade payables |
|
19035.220 |
19407.930 |
|
(c) Other current liabilities |
|
769.930 |
632.640 |
|
(d) Short-term provisions |
|
5349.790 |
12077.730 |
|
Total
Current Liabilities (4) |
|
27026.830 |
39992.480 |
|
|
|
|
|
|
TOTAL |
|
117638.670 |
101928.720 |
|
|
|
|
|
|
II.
ASSETS |
|
|
|
|
(1) Non-current assets |
|
|
|
|
(a) Fixed Assets |
|
|
|
|
(i) Tangible assets |
|
17836.970 |
16437.270 |
|
(ii) Intangible Assets |
|
9235.010 |
8923.400 |
|
(iii) Goodwill |
|
478.920 |
574.800 |
|
(iv) Capital work-in-progress |
|
6295.500 |
4978.290 |
|
(v) Intangible assets under
development |
|
785.620 |
449.660 |
|
(b) Non-current Investments |
|
156.940 |
171.950 |
|
(c) Deferred tax assets (net) |
|
13112.690 |
10648.840 |
|
(d) Long-term Loan and Advances |
|
0.000 |
0.000 |
|
(e) Other Non-current assets |
|
990.630 |
701.510 |
|
Total
Non-Current Assets |
|
48892.280 |
42885.720 |
|
|
|
|
|
|
(2) Current assets |
|
|
|
|
(a) Current investments |
|
0.000 |
0.000 |
|
(b) Inventories |
|
21390.500 |
15677.600 |
|
(c) Trade receivables |
|
24043.200 |
24926.460 |
|
(d) Cash and cash equivalents |
|
10563.640 |
8571.210 |
|
(e) Short-term loans and
advances |
|
0.000 |
0.000 |
|
(f) Other current assets |
|
12749.050 |
9867.730 |
|
Total
Current Assets |
|
68746.390 |
59043.000 |
|
|
|
|
|
|
TOTAL |
|
117638.670 |
101928.720 |
PROFIT
& LOSS ACCOUNT– (CONSOLIDATED)
|
|
PARTICULARS |
|
31.03.2017 |
31.03.2016 |
|
|
SALES |
|
|
|
|
|
Income |
|
91856.810 |
76495.830 |
|
|
Other Income |
|
373.650 |
200.000 |
|
|
TOTAL
|
|
92230.460 |
76695.830 |
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
Cost of Materials Consumed |
|
23548.130 |
19287.470 |
|
|
Purchases of Stock-in-Trade |
|
7191.200 |
5139.970 |
|
|
Changes in inventories of
finished goods, work-in-progress and Stock-in-Trade |
|
(4596.070) |
(1401.600) |
|
|
Employees benefits expense |
|
16408.060 |
13781.950 |
|
|
Exceptional items |
|
809.490 |
0.000 |
|
|
Other expenses |
|
28938.490 |
25316.520 |
|
|
TOTAL |
|
72299.300 |
62124.310 |
|
|
|
|
|
|
|
|
PROFIT/
(LOSS) BEFORE INTEREST, TAX,
DEPRECIATION AND AMORTISATION |
|
19931.160 |
14571.520 |
|
|
|
|
|
|
|
Less |
FINANCIAL
EXPENSES |
|
2373.180 |
1788.850 |
|
|
|
|
|
|
|
|
PROFIT
/ (LOSS) BEFORE TAX, DEPRECIATION AND AMORTISATION |
|
17557.980 |
12782.670 |
|
|
|
|
|
|
|
Less/
Add |
DEPRECIATION/
AMORTISATION |
|
2643.680 |
2342.840 |
|
|
|
|
|
|
|
|
PROFIT/
(LOSS) BEFORE TAX |
|
14914.300 |
10439.830 |
|
|
|
|
|
|
|
Less |
TAX |
|
3826.770 |
3009.380 |
|
|
|
|
|
|
|
|
PROFIT/
(LOSS) AFTER TAX |
|
11087.530 |
7430.450 |
|
|
|
|
|
|
|
|
Earnings
/ (Loss) Per Share (INR) |
|
39.29 |
26.47 |
LEGAL CASE
DETAILS FILE ATTACHED
LOCAL AGENCY FURTHER INFORMATION
|
Sr. No. |
Check list by
info agents |
Available in
Report (Yes/No) |
|
1 |
Year of establishment |
Yes |
|
2 |
Constitution of the entity -Incorporation
details |
Yes |
|
3 |
Locality of the entity |
Yes |
|
4 |
Premises details |
No |
|
5 |
Buyer visit details |
-- |
|
6 |
Contact numbers |
Yes |
|
7 |
Name of the person contacted |
Yes |
|
8 |
Designation of contact person |
Yes |
|
9 |
Promoter’s background |
Yes |
|
10 |
Date of Birth of Proprietor / Partners /
Directors |
Yes |
|
11 |
Pan Card No. of Proprietor / Partners |
No |
|
12 |
Voter Id Card No. of Proprietor / Partners |
No |
|
13 |
Type of business |
Yes |
|
14 |
Line of Business |
Yes |
|
15 |
Export/import details (if applicable) |
No |
|
16 |
No. of employees |
Yes |
|
17 |
Details of sister concerns |
Yes |
|
18 |
Major suppliers |
No |
|
19 |
Major customers |
No |
|
20 |
Banking Details |
Yes |
|
21 |
Banking facility details |
Yes |
|
22 |
Conduct of the banking account |
-- |
|
23 |
Financials, if provided |
Yes |
|
24 |
Capital in the business |
Yes |
|
25 |
Last accounts filed at ROC, if applicable |
Yes |
|
26 |
Turnover of firm for last three years |
Yes |
|
27 |
Reasons for variation <> 20% |
-- |
|
28 |
Estimation for coming financial year |
No |
|
29 |
Profitability for last three years |
Yes |
|
30 |
Major shareholders, if available |
Yes |
|
31 |
External Agency Rating, if available |
Yes |
|
32 |
Litigations that the firm/promoter
involved in |
Yes |
|
33 |
Market information |
-- |
|
34 |
Payments terms |
No |
|
35 |
Negative Reporting by Auditors in the
Annual Report |
No |
COMPANY INFORMATION
Subject (the “Company”) is a public limited company incorporated in Mumbai, India. The registered office of the Company is at B/2, Mahalaxmi Chambers, 22 Bhulabhai Desai Road, Mumbai – 400026, India.
The Company is primarily engaged in the business of development, manufacture and marketing of pharmaceutical products. The Company also markets active pharmaceutical ingredients. The Companies research and development facilities are located at Mahape, Sinnar, Turbhe and Taloja in India and manufacturing facilities are located at Nasik, Colvale, Baddi, Nalagarh, Ankleshwar, Mohol, Kurkumbh, Sikkim, Indore, Dahej and Aurangabad.
The Company’s shares are listed on the BSE Limited (“BSE”) and the National Stock Exchange of India (“NSE”).
RESULTS OF OPERATIONS
Indian Accounting
Standards (Ind AS)
Financial statements have been prepared in accordance with the Indian Accounting Standards (hereinafter referred to as the ‘Ind AS’) as notified by the Ministry of Corporate Affairs pursuant to Section 133 of the Companies Act, 2013 read with the Companies (Indian Accounting Standards) Rules, 2015 as amended and other relevant provisions of the Act. These financial statements for the year ended 31 March 2017 are the first financials with comparatives, prepared under Ind AS. For all previous periods including the year ended 31 March 2016, the Company had prepared its financial statements in accordance with the accounting standards notified under Companies (Accounting Standard) Rule, 2006 (as amended) and other relevant provisions of the Act (hereinafter referred to as ‘Previous GAAP’) used for its statutory reporting requirement in India.
The Company has also voluntarily presented the consolidated results in accordance with the recognition and measurement principles as per International Financial Reporting Standards (IFRS).
On Standalone basis the Company achieved gross revenue of INR 82437.390 million and the Standalone operating profit before finance costs, depreciation and tax was INR 30468.810 million as compared to INR 18825.620 million in the previous year.
MANAGEMENT DISCUSSION
AND ANALYSIS
Global Environment
The World Economic Outlook (WEO) recently raised its projection for global growth to 3.5% for 2017 compared to 3.4% in 2016 due to pick up in overall demand. Stronger activity and expectations of more robust global demand, coupled with agreed restrictions on oil supply, have helped commodity prices recover in early 2016. However, the long term potential growth rates remain subdued across the globe, especially in advanced economies with protectionism attitude towards its trade policy. The activity is projected to pick up in emerging markets and developing economies because the strained conditions are gradually expected to improve, supported by the partial recovery in commodity prices, while growth is projected to remain strong in China and many other developing nations. In advanced economies, the pickup is primarily driven by higher projected growth in the United States, where activity was held back in 2016. Hence, the projected pickup in growth in the next two years primarily reflect forecasts of a gradual improvement of growth rates in countries currently in economic distress, notably Brazil, Russia, and some countries in the Middle East, though the growth momentum is still modest and downside risks continue to dominate, with heightened policy uncertainty and persistent structural headwinds could be frustrated by new economic or political shocks.
India remains the fastest growing economy in the world, despite the demonetisation, which temporarily disrupted the economy in the latter half of the FY 2017. The economy is set to grow at 7.4% in the current fiscal year 2017-18 against 7.1% in the previous year, on the back of pick-up in consumption demand and higher public investment. However, India imports nearly 80% of its fossil fuel needs, a rapid increase in the price of oil could undermine the country’s fiscal position, effect inflation badly and swell the current account deficit, which may pose a potential risk for the Indian economy.
Global Pharma
Scenario
Reports project global health care expenditures to reach USD 8.7 trillion by 2020, from USD 7 trillion in 2015, driven by improved treatments in therapeutic areas coupled with rising labor costs and increased life expectancy. Health care spending as a percentage of gross domestic product (GDP) is expected to also rise slightly, from an estimated 10.4% in 2015 to 10.5% in 2020. Government health care expenditures as a percentage of GDP are projected to rise more quickly in low-income countries than other income groups. Life expectancy is projected to increase by one year by 2020, which will increase the aging population (over 65 years old) by 8%, from 559 million in 2015 to 604 million in 2020. While the developed markets would continue to use branded and specialty medicines the pharmerging markets would use more non-original brands, generics and over the counter products. Furthermore, the adoption of newer medicines will remain higher in developed markets than in pharmerging markets.
The spending on medicines across all regions is projected to increase. The US is the largest pharmaceutical market in the world accounting for approximately 35% of the global share. Analysts expect that the lower price advantage associated with generic drugs may be partially offset by increasing industry consolidation. Single-digit spending growth is forecast for the US market. The US market is expected to fall and projected to grow 6% to 9% through 2021.
The US will account for 53% of forecasted growth over the next five years while China will continue as the second largest market, a position it has held since 2012, contributing 12% of the growth. Developed market spending growth will be driven by original brands while pharmerging markets will continue to be fueled by non-original products that make up an average 91% of pharmerging market volume and 78% of spending.
European market is expected to maintain tight constraints on drug budgets. Forecasted low pre-rebate and discount growth of 1% to 4% in the EU5 countries (France, Germany, Italy, Spain, the United Kingdom). The impact of BREXIT on the UK pharmaceutical market is expected to be modest, driving at most a 1.5% slower growth rate.
The growth in spending on medicines in pharmerging markets by 2020 would be about USD 125 billion primarily driven by wider use of medicines. The per capita increase in volume and spending is expected to result from the strong commitment by government to widen the access to healthcare and the expanded private insurance in these markets.
Business Environment
India Formulations
During the year, the India Formulations (IF) business performed well registering revenue of INR 23,037.77 mn (USD 344 mn) as against INR 21, 092.74 mn (USD 322.91 mn) recording growth of 9.22%.
As per IMS MAT March 2017, Glenmark’s India business improved its rank to 15th, compared to 18th MAT March 2016. Glenmark increased its market share by 0.20%, exhibiting value growth of 14% vis-à-vis IPM growth of 9%. This growth has been driven by strong performance of leading brands resulting in market share improvement across therapeutic categories.
Growth across
therapeutic categories
The India business strengthened itself in the following therapeutic areas with considerable growth in market share from IMS MAT March 2016 to MAT March 2017 respectively:
> Derma therapy market share improved from 8.6% to 9.2%
> Respiratory therapy market share rose from 4.1% to 4.5%
> Cardiac therapy market share increased from 3.9% to 4%
Brands in IPM Top –
300
> Glenmark’s brand Telma (Telmisartan) secured its position among the Top 50 brands in IPM and is currently ranked 48th
> Telma-H (Telmisartan Hydrochloride) ranked 64 in IPM closing a value growth of 18.4% over the last year
> Glenmark’s brand Ascoril+ (IPM rank 114), Candid (IPM rank 118), Candid-B (IPM rank 120), Telma- AM (IPM rank 186), Alex (IPM rank 192) and Ascoril –LS (IPM rank 197) are some of the other brands among the Top 200 brands in IPM 300 brands league
In respiratory
therapy, Glenmark launched
> Digihaler - India’s first Digital Dose Inhaler (DDI) pan India. This next-gen inhaler provides accurate digital dose counter along with low dose warning indicator to enable Asthma and chronic obstructive pulmonary disease (COPD) patients track adherence to their therapy
> NebZmart – an advanced nebulizer with active vibrating mesh technology that can nebulise both solutions and suspensions efficiently. This Zmart choice is expected to liberate Asthma and COPD patients (requiring nebulisation) from conventional, bulky, noisy and difficult to use nebulisers dependent on electricity
> Airz which is India’s first glycopyrronium DPI monotherapy used as a bronchodilator therapy to relieve symptoms in adult patients with COPD. Glenmark Airz provides compliance to the COPD patients (moderate to severe COPD) as only one capsule once daily is to be taken by the patient
In the dermatology
therapy, Glenmark launched
> Lulican Cream/ Lotion, and Luligee – Luliconazole brands and Fintop AF (amorolfine) cream for anti-fungal topical treatment
> Canditz (itraconazole), an oral antifungal specially targeted for tinea infections
> Ecziderm, an emollient for treatment of eczema
> Fenza (Fenticonazole) for treatment vulvovaginal candidiasis
> Onabet SD (Sertaconazole) lotion for the treatment of seborrheic dermatitis
> Sabiglow ML, which is formulated with scientifically validated nutrients targeted to address hyper-pigmentation concerns
In the Chronic
therapy, Glenmark launched
> XMET-Trio for treatment of diabetes
> Razel CV for treatment of heart conditions
> Dubinor was launched as adjuvant therapy for people suffering from chronic disorders
> Glenmark also launched FONYL (Fosfomycin) an anti-infective in the critical care segment for treatment of urinary tract infection
Active Pharmaceutical
Ingredients (API)
Revenue from sale of API to regulated and semi-regulated markets globally was INR 8,094.10 mn (USD 120.86 mn) during the year as against INR 6,682.88 mn (USD 102.31 mn) for the previous year, recording 21.12% increase.
The good growth of the business was due to the successful launch of Olmesartan. In the fourth quarter, Glenmark received an EIR from the USFDA for its Ankleshwar facility.
APIs are the principal ingredients for finished dosages and are also known as bulk actives or bulk drugs. APIs become formulations when the dosage is administered by using additional inactive ingredients either in oral forms such as tablets, capsules, dry syrups or liquid orals or in sterile forms like injectable dry powder vials or liquid injectables. The Company also markets and supplies its API products to leading generic manufacturers in the US, Europe and Japan, in addition to fulfilling captive API requirements. Glenmark’s API product portfolio comprises Lercanidipine, Amiodarone, Rosuvastatin, Perindopril, Adapalene and Atovaquone, among others. As on 31 March 2017, the Company has filed over 320 Global DMFs in various markets including 94 USDMFs, 23 CEP’s, 39 EU-DMF’s, 22 Canadian DMF’s, 12 Japan DMF’s, 13 Australian DMF’s and other DMF’s in various Glenmark Brazil team ROW countries.
OUTLOOK
Despite the challenging economic situation in most emerging markets including the volatile currencies, Glenmark continues to remain positive on the long-term growth prospects in key emerging markets. The focus in emerging markets will be to continuously invest in product pipeline namely in the areas of dermatology, respiratory and the oncology therapy. While Glenmark will contain its new investments in emerging markets it will continuously focus on building the product pipeline in these therapy areas. The US remains the most important market for Glenmark and the organisation continues to invest significantly in this market. All the incremental R&D resources are being invested in the US market and this region will be a key driver for growth in the future. On the generics front, Glenmark will continuously file products in the area of dermatology and injectables including complex injectables. On the discovery front, the pipeline is progressing well with several molecules in clinical or pre-clinical development.
The Company will also continue with its approach of out-licensing its molecules. Going ahead, the organisation will continue to lay equal emphasis on small molecules as well as biologics and will continue to focus on discovering primarily firstin- class molecules globally for unmet medical needs.
Their primary objective has always been to facilitate the Company’s evolution from a generics organisation to a fully integrated, globally commercialised pharmaceutical company with innovative products. Glenmark has always been focused on a long-term growth strategy while meeting the short-term growth objectives. Today, INR have a strong pipeline of products in the US which primarily consists of differentiated products. They have built a robust India business and have set up a strong foundation for their future growth. In markets like Europe, they anticipate to grow in double digit over the next 3-4 years. The emerging markets (ex-India) though a small portion of the overall revenue will also continue to grow and will be led primarily by Russia business, which is growing at 25%.
Further, with seven novel molecule and three specialty products in their research and development pipeline and with their end-to-end capabilities from R&D to full-scale manufacturing (both in small molecules and novel biologics), the Company enjoys a strong position in IP leadership and global footprint for rapid market penetration. Their complex generic portfolio will also play a significant role in Glenmark’s growth strategy in various markets in which they operate and they continue to have complex generic products in their filed pipeline, and they would continue to develop more products in-house.
Their strategy is to leverage both inhouse and external capabilities to develop complex generic products portfolio to differentiate ourselves from the competitors.
UNSECURED LOAN
|
Unsecured Loan |
31.03.2017 (INR
in Million) |
31.03.2016 (INR
in Million) |
|
Long-term
Borrowings |
|
|
|
Foreign currency convertible bonds (FCCB) |
13178.950 |
0.000 |
|
Senior notes |
12714.510 |
0.000 |
|
|
|
|
|
Short-term
borrowings |
|
|
|
From banks |
1845.950 |
7718.920 |
|
|
|
|
|
Total |
27739.410 |
7718.920 |
|
Note: Long-term
Borrowings During the year, the Company issued U.S. $ 200,000,000 2.00% Resettable Onward Starting Equity-linked Securities (Bonds) and U.S.$ 200,000,000 4.5% Senior Notes (Notes), the brief description of the same is provided herein below U.S. $ 200,000,000,
2.00% Resettable Onward Starting Equity-linked Securities (Bonds): The Company issued Bonds on 28 June 2016. The Bonds will be convertible at the option of the holders’ of the Bonds (the “Bondholders”) at any time on or after 1 December 2017 and upto the close of business on 18 June 2022 into equity shares. Each Bond will be convertible at the option of the holder thereof into fully paid equity share at an initial conversion price to be determined on 30 November 2017: Unless previously converted, redeemed or purchased and cancelled, the Bonds will be redeemed on 28 June 2022 (Maturity Date) at 126.42% of their principal amount, together with accrued interest (if any), calculated upto but excluding the Maturity Date. The Company may, at its own discretion, redeem the Bonds in whole, but not in part, subject to satisfaction of certain conditions. Each Bondholder has the right to require the Company to redeem in whole or in part, such Bondholder’s Bonds, on 28 July 2021, at a price equal to 121.78% of its outstanding principal amount of Bonds, together with interest (if any) accrued but unpaid on 28 July 2021. The Bonds are listed on the Singapore Stock Exchange. U.S. $ 200,000,000, 4.5% Senior Notes (Notes) : The Company issued Notes on 1 August 2016. The Notes will mature on 2 August 2021. The interest on Notes will be payable semi-annually in arrears on 1 February and 1 August each year. The final interest payment and the payment of principal will occur on 2 August 2021. The Notes are Redeemable at any time on or after 2 August 2019, all or part of the Notes by paying the redemption price, subject to fulfilment of certain conditions. The Company, at its discretion, may redeem all or a portion of the Notes at a redemption price equal to 100% of the principal amount, plus the applicable redemption premium, and accrued and unpaid interest and additional amounts, if any |
||
INDEX OF CHARGES:
|
SNo |
SRN |
Charge Id |
Charge Holder Name |
Date of Creation |
Date of
Modification |
Date of
Satisfaction |
Amount |
Address |
|
1 |
A54985908 |
10114486 |
Bank of India |
09/06/2008 |
19/12/2008 |
- |
1700000000.0 |
Mahalaxmi Branch8A-Bhulabhai Desai Road, Mumbai - 400026IN |
|
2 |
Z00144350 |
80033325 |
State Bank of India |
23/09/2004 |
- |
- |
100000000.0 |
Atlanta Building, Nariman PointMumbaiMH400001IN |
|
3 |
G01792555 |
80020353 |
Bank of India |
21/05/1985 |
29/03/2016 |
- |
7800000000.0 |
8-A, Bhulabhai Desai RoadMahalaxmiMumbaiMH400026IN |
|
4 |
C44435386 |
10141439 |
CENTRAL BANK OF INDIA |
16/01/2009 |
20/10/2009 |
04/02/2015 |
1500000000.0 |
CORPORATE FINANCE BRANCH, IST FLOOR ,M.M.O. BUILDING, M.G.ROAD,FORT, MUMBAIMH400023IN |
|
5 |
B75549402 |
10197827 |
The Hongkong and Shanghai Banking Corporation Limited |
12/09/2007 |
07/04/2008 |
22/05/2013 |
2000000000.0 |
52/60, MAHATMA GANDHI ROAD, FORT, MUMBAIMH400001IN |
|
6 |
B23541352 |
10161795 |
Axis Bank Limited |
18/06/2009 |
15/04/2011 |
20/10/2011 |
750000000.0 |
AXIS HOUSE, 2ND FLOOR, E-WING, BOMBAY DYEING MILLS COMPOUND PANDURANG BUDHKAR MARG WORLI, MUMBAIMH400025IN |
|
7 |
B04595377 |
10172979 |
Bank of India |
29/07/2009 |
- |
27/01/2011 |
600000000.0 |
Mahalaxmi Branch, 8-A, Bhulabhai Desai Road, Near Mahalaxmi Temple, Mumbai - 400026IN |
|
8 |
A69823748 |
10068660 |
THE HONGKONG AND SHANGHAI BANKING CORPORATION LIMITED |
12/09/2007 |
07/04/2008 |
07/09/2009 |
2000000000.0 |
52/60, MAHATMA GANDHI ROAD, FORT, MUMBAI - 400001IN |
|
9 |
A18517789 |
80033323 |
State Bank Patiala |
25/11/2004 |
- |
22/06/2007 |
250000000.0 |
Atlanta Building, Nariman Point, Mumbai 400001IN |
|
10 |
A18511683 |
80033867 |
STATE BANK OF PATIALA |
23/09/2004 |
- |
22/06/2007 |
100000000.0 |
ATLANTA BUILDING, NARIMAN POINT, MUMBAI - 400001IN |
STATEMENT OF
STANDALONE UNAUDITED FINANCIAL RESULTS FOR THE QUARTER AND NINE MONTHS ENDED 31TH
DECEMBER 2017
|
|
|
Particulars |
quarter ended |
quarter ended |
Nine months ended |
|
|
|
|
31.12.2017 |
30.09.2017 |
31.12.2017 |
|
1 |
|
Income from
Operations |
|
|
|
|
|
|
Sales/Income from Operations (Gross) |
15565.490 |
16598.120 |
47641.390 |
|
|
|
b) Other Operating Income |
372.320 |
563.040 |
1361.240 |
|
|
Total Income from
Operations (Net) |
15937.810 |
17161.160 |
49002.630 |
|
|
2 |
Expenses |
|
|
|
|
|
|
a) |
Cost of Materials consumed |
5224700 |
4848.220 |
5073.130 |
|
|
b) |
Purchase of Stock-in-trade |
891.980 |
664.070 |
2249.080 |
|
|
c) |
Changes in inventories of finished goods, work-in-progress and
stock-in-trade |
(304.490) |
584.970 |
171.140 |
|
|
d) |
Employee benefit expenses |
2553.980 |
3264.260 |
7895.910 |
|
|
e) |
Finance Costs |
469.990 |
474.410 |
1401.760 |
|
|
f) |
Depreciation and amortization expense |
295.530 |
288.930 |
883.860 |
|
|
G) |
Other expenses |
4531.390 |
3262.040 |
12080.190 |
|
|
Total Expenses |
13663.050 |
13386.900 |
39755.070 |
|
|
|
|
|
|
|
|
|
|
Profit /(Loss) from
ordinary activities after finance costs but before exceptional items |
2274.760 |
3774.260 |
9247.560 |
|
|
|
Exceptional Items |
-- |
-- |
-- |
|
|
|
Profit /(Loss) from
ordinary activities before tax |
2274.760 |
3774.260 |
9247.560 |
|
|
|
Tax Expense |
|
|
|
|
|
|
Current Tax |
489.590 |
801.350 |
1973.250 |
|
|
|
Deferred Tax |
(155.990) |
(305.880) |
(654.040) |
|
|
|
Net Profit /(Loss)
from ordinary activities after tax |
1941.160 |
3278.790 |
7928.350 |
|
|
|
|
|
|
|
|
|
|
Other Comprehensive
Income: |
|
|
|
|
|
|
Other Comprehensive
Income: |
|
|
|
|
|
|
A. Items that will not be reclassified to profit or loss |
(11.920) |
(26.090) |
(46.610) |
|
|
|
Income tax relating to items that will be reclassified to profit or loss |
4.120 |
9.020 |
16.130 |
|
|
|
B. Items that will be reclassified to profit or loss |
-- |
-- |
-- |
|
|
|
Income tax relating to items that will be reclassified to profit or loss |
-- |
-- |
-- |
|
|
|
Total Other
Comprehensive Income for the period |
1933.360 |
3261.720 |
7897.870 |
|
|
|
|
|
|
|
|
|
12 |
Paid up equity share capital (Eq. shares of INR 10/- each) |
-- |
-- |
-- |
|
|
14 |
|
Earnings per share (before/after extraordinary items) of Rs.10/- each |
|
|
|
|
|
|
Basic & Diluted |
6.88 |
11.62 |
28.10 |
CONTINGENT
LIABILITIES:
(INR in million)
|
PARTICULARS |
31.03.2017 |
31.03.2016 |
|
Claims against the
Company not acknowledged as debts: |
|
|
|
Labour dispute |
15.770 |
12.400 |
|
Disputed taxes and duties |
243.620 |
259.620 |
|
(a) In January 2014, the National Pharmaceutical Pricing Authority (NPPA) issued a demand notice of INR 122.30 as overcharging liability of product “Doxovent 400 mg tab” for the period February 2010 to May 2013. The notice also envisaged a payment of INR 33.30 towards interest @15% p.a. on the overcharged amount up to 31 January 2014. The Company has filed a petition under Article 32 with the Hon’ble Supreme Court of India (Hon’ble Court), challenging the issue of the above mentioned demand notice on various grounds. This petition has been tagged along with other petition/s filed by other pharmaceutical companies as well, pending before Supreme Court relating to the inclusion criteria of certain drugs including “Theophylline” in the schedule of the DPCO, 1995. The matters are sub-judice before the Supreme Court. The Hon’ble Court passed an ad-interim order stating that no coercive steps be taken against the Company towards the said demand. The Hon’ble Court has constituted a Special bench to hear the petition (along with other petitions filed in this regard) and the matter is expected to be listed in due course. The company based on legal advice, has an arguable case on merits as well as with regard to mitigation of the demand. (b) On 10 March 2016 Ministry of Health and Family Welfare issued notifications prohibiting manufacture for sale, sale and distribution for human use of several Fixed Dose Combination (“FDC”) with immediate effect. Several products of the Company are also covered in the notified prohibited “FDC’s”. The Company has filed five writ petitions in Hon’ble Delhi High Court challenging the notifications issued. The Hon’ble Delhi High Court has granted interim relief to the Company by staying the notifications banning the FDC’s. The company based on legal advise, has an arguable case on merits though the liability in this case cannot be computed. In an adverse scenario, the Company would be restricted from manufacturing, selling and marketing the impacted FDC’s. The company has revised the composition of the FDC’s and market the revised product. The matter is now clubbed with other petition with other companies before the supreme court. |
||
FIXED ASSETS
- Freehold Land
- Leasehold land
- Factory Building
- Plant and equipment
- Furniture and Fixture
- Office equipment
- Vehicles
CMT REPORT (Corruption, Money Laundering & Terrorism]
The Public Notice information has been collected from various sources
including but not limited to: The Courts,
1] INFORMATION ON
DESIGNATED PARTY
No records exist designating subject or any of its beneficial owners,
controlling shareholders or senior officers as terrorist or terrorist
organization or whom notice had been received that all financial transactions
involving their assets have been blocked or convicted, found guilty or against
whom a judgement or order had been entered in a proceedings for violating money-laundering,
anti-corruption or bribery or international economic or anti-terrorism sanction
laws or whose assets were seized, blocked, frozen or ordered forfeited for
violation of money laundering or international anti-terrorism laws.
2] Court Declaration :
No records exist to suggest that subject is
or was the subject of any formal or informal allegations, prosecutions or other
official proceeding for making any prohibited payments or other improper
payments to government officials for engaging in prohibited transactions or
with designated parties.
3] Asset Declaration :
No records exist to suggest that the property or assets of the subject
are derived from criminal conduct or a prohibited transaction.
4] Record on Financial
Crime :
Charges or conviction
registered against subject: None
5] Records on Violation of
Anti-Corruption Laws :
Charges or
investigation registered against subject: None
6] Records on Int’l
Anti-Money Laundering Laws/Standards :
Charges or
investigation registered against subject: None
7] Criminal Records
No
available information exist that suggest that subject or any of its principals
have been formally charged or convicted by a competent governmental authority
for any financial crime or under any formal investigation by a competent
government authority for any violation of anti-corruption laws or international
anti-money laundering laws or standard.
8] Affiliation with
Government :
No record
exists to suggest that any director or indirect owners, controlling shareholders,
director, officer or employee of the company is a government official or a
family member or close business associate of a Government official.
9] Compensation Package :
Our market
survey revealed that the amount of compensation sought by the subject is fair
and reasonable and comparable to compensation paid to others for similar
services.
10] Press Report :
No press reports / filings exists on
the subject.
CORPORATE GOVERNANCE
MIRA INFORM as part of its Due Diligence do provide comments on
Corporate Governance to identify management and governance. These factors often
have been predictive and in some cases have created vulnerabilities to credit
deterioration.
Our Governance Assessment focuses principally on the interactions
between a company’s management, its Board of Directors, Shareholders and other
financial stakeholders.
CONTRAVENTION
Subject is not known to have contravened any existing local laws,
regulations or policies that prohibit, restrict or otherwise affect the terms
and conditions that could be included in the agreement with the subject.
FOREIGN EXCHANGE RATES
|
Currency |
Unit
|
INR |
|
US Dollar |
1 |
INR 65.06 |
|
|
1 |
INR 92.06 |
|
Euro |
1 |
INR 80.37 |
INFORMATION DETAILS
|
Information
Gathered by : |
SVT |
|
|
|
|
Analysis Done by
: |
VIV |
|
|
|
|
Report Prepared
by : |
SUJ |
SCORE FACTORS
|
DEMERIT POINTS |
|
|
|
--BANK CHARGES |
YES/NO |
YES |
|
--LITIGATION |
YES/NO |
YES |
|
--OTHER ADVERSE INFORMATION |
YES/NO |
NO |
|
MERIT POINTS |
|
|
|
--SOLE DISTRIBUTORSHIP |
YES/NO |
NO |
|
--EXPORT ACTIVITIES |
YES/NO |
NO |
|
--AFFILIATION |
YES/NO |
YES |
|
--LISTED |
YES/NO |
YES |
|
--OTHER MERIT FACTORS |
YES/NO |
YES |
RATING EXPLANATIONS
|
Credit Rating |
Explanation |
Rating Comments |
|
A++ |
Minimum Risk |
Business dealings permissible with minimum
risk of default |
|
A+ |
Low Risk |
Business dealings permissible with low risk
of default |
|
A |
Acceptable Risk |
Business dealings permissible with
moderate risk of default |
|
B |
Medium Risk |
Business dealings permissible on a regular
monitoring basis |
|
C |
Medium High Risk |
Business dealings permissible preferably
on secured basis |
|
D |
High Risk |
Business dealing not recommended or on
secured terms only |
|
NB |
New Business |
No recommendation can be done due to
business in infancy stage |
|
NT |
No Trace |
No recommendation can be done as the
business is not traceable |
NB is stated where there is insufficient information to facilitate rating. However, it is not to be considered as unfavourable.
This score serves as a reference to assess
SC’s credit risk and to set the amount of credit to be extended. It is
calculated from a composite of weighted scores obtained from each of the major
sections of this report. The assessed factors are as follows:
·
Financial
condition covering various ratios
·
Company
background and operations size
·
Promoters
/ Management background
·
Payment
record
·
Litigation
against the subject
·
Industry
scenario / competitor analysis
·
Supplier
/ Customer / Banker review (wherever available)
This report is issued at
your request without any risk and responsibility on the part of MIRA INFORM
PRIVATE LIMITED (MIPL) or its officials.

