|
|
|
MIRA INFORM REPORT
|
Report No. : |
500236 |
|
Report Date : |
24.03.2018 |
IDENTIFICATION DETAILS
|
Name : |
TORRENT PHARMACEUTICALS LIMITED |
|
|
|
|
Registered
Office : |
Torrent House, Off. Ashram Road, Navarangpura 9, Ahmedabad – 380009, |
|
Tel. No.: |
91-79-26583060 / 26585090 |
|
|
|
|
Country : |
|
|
|
|
|
Financials (as
on) : |
31.03.2017 |
|
|
|
|
Date of
Incorporation : |
15.07.1972 |
|
|
|
|
Com. Reg. No.: |
04-002126 |
|
|
|
|
Capital Investment
/ Paid-up Capital : |
INR 846.200 Million |
|
|
|
|
CIN No.: [Company Identification
No.] |
L24230GJ1972PLC002126 |
|
|
|
|
IEC No.: [Import-Export Code No.] |
0888038241 |
|
|
|
|
GSTN : [Goods & Service Tax
Registration No.] |
Not Divulged |
|
|
|
|
TAN No.: [Tax Deduction &
Collection Account No.] |
Not Available |
|
|
|
|
PAN No.: [Permanent Account No.] |
AAACT5456A |
|
|
|
|
Legal Form : |
A Public Limited Liability Company. The Company’s Shares are Listed on
the Stock Exchanges. |
|
|
|
|
Line of Business
: |
The company engaged in research, development, manufacturing and marketing of generic pharmaceutical formulations. [Registered Activity] |
|
|
|
|
No. of Employees
: |
11781 (Approximately) |
|
MIRA’s Rating : |
A++ |
|
Credit Rating |
Explanation |
Rating Comments |
|
A++ |
Minimum Risk |
Business dealings permissible with minimum
risk of default |
|
Maximum Credit Limit : |
USD 130000000 |
|
|
|
|
Status : |
Excellent |
|
|
|
|
Payment Behaviour : |
Regular |
|
|
|
|
Litigation : |
Clear |
|
|
|
|
Comments : |
Subject is a flagship company of Torrent Group in India based in Ahmedabad,
Gujarat and strong player of pharmaceutical industry. The company is one of the leading manufacturers and trader of branded as well as unbranded generic pharmaceutical products and also has significant presence in fast growing, life style related therapeutic segments like Cardiovascular (CVS) and Central nervous system (CNS). For the financial year 2017, the company has witnessed slight decline in its revenue compared to previous year along with good profitability margin of 19.42%. Ratings
take into consideration healthy financial profile on back of low financial
leverage with favourable capital structure and comfortable liquidity
position. Rating
also takes into consideration the company’s established track record and
enjoys strong market position in domestic formulation market with focus on
CVS, CNS and GI therapeutic areas. On 8th May 2017, the company has acquired two brands i.e. Regestrone and Pregachieve for India from global Pharma player Novartis AG, Switzerland. This recent acquisition provides company a strong foothold in women’s healthcare segment. Further, the company is listed on BSE and NSE. Price quoted in BSE is held at INR 1250.90 against its value of INR 5. As per quarterly results of December 2017, the company has achieved decent revenue of INR 11,220 million and has clocked profit margin of 7.54%. However,
ratings strengths are partially offset by exposure to intense competition in
pharmaceutical industry. Payments
are reported to be regular. In view
of aforesaid, the company can be considered for business dealings at usual
trade terms and conditions. |
NOTES:
Any query related to this report can be made
on e-mail: infodept@mirainform.com
while quoting report number, name and date.
ECGC Country Risk Classification List
|
Country Name |
Previous Rating (30.09.2017) |
Current Rating (31.12.2017) |
|
India |
A1 |
A1 |
|
Risk Category |
ECGC
Classification |
|
Insignificant |
A1 |
|
Low Risk |
A2 |
|
Moderately Low Risk |
B1 |
|
Moderate Risk |
B2 |
|
Moderately High Risk |
C1 |
|
High Risk |
C2 |
|
Very High Risk |
D |
EXTERNAL AGENCY RATING
|
Rating Agency Name |
ICRA |
|
Rating |
Commercial Paper = A1+ |
|
Rating Explanation |
Very strong degree of safety and carry
lowest credit risk. |
|
Date |
24.01.2018 |
|
Rating Agency Name |
ICRA |
|
Rating |
Non-convertible Debenture Programme = AA |
|
Rating Explanation |
High degree of safety and very low credit
risk. |
|
Date |
24.01.2018 |
RBI DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available RBI Defaulters’ list.
EPF (Employee Provident Fund) DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available EPF (Employee Provident Fund) Defaulters’ list as of
31-03-2016.
BIFR (Board for Industrial & Financial Reconstruction) LISTING
STATUS
Subject’s name is not listed as a Sick Unit in
the publicly available BIFR (Board for Industrial & Financial
Reconstruction) list as of 24.03.2018
IBBI (Insolvency and Bankruptcy Board of India) LISTING STATUS
Subject’s name is not listed in the publicly
available IBBI (Insolvency and Bankruptcy Board of India) list as of report
date.
INFORMATION DENIED
Management Non-Cooperative (91-79-26583060)
LOCATIONS
|
Registered /
Corporate Office : |
Torrent House, Off. Ashram Road, Navarangpura 9, Ahmedabad – 380009, Gujarat, India |
|
Tel. No.: |
91-79-26583060 /26585090 |
|
Fax No.: |
91-79-26582100 |
|
E-mail : |
|
|
Website : |
|
|
|
|
|
Factory 1 : |
Indrad Plant: Village Indrad,
Taluka Kadi, District: Mehsana – 382721, Gujarat, India |
|
Tel. No.: |
91-2764-233671-75,
233678-80 |
|
Fax No.: |
91-2764-233676 |
|
E-Mail : |
|
|
|
|
|
Factory 2 : |
Baddi Plant : Village Bhud and
Makhnu Majra, Baddi, Tehsil Nalagarh, District: Solan, |
|
Tel. No.: |
91-1795-246821 |
|
Fax No.: |
91-1795-247159 |
|
|
|
|
Factory 3 : |
32 No. Middle Camp, NH-31A, East District, Gangtok, Sikkim,
Himalaya, India |
|
|
|
|
Factory 4 : |
Plot
No. 810, Sector III, Industrial Area, Pithampura, District: Dhar, Madhya Pradesh, India |
|
|
|
|
Factory 5 : |
Plot
No.77, J N Pharma City, Thanam Village, Parawada Mandal, Vizag, Andhra
Pradesh, India |
|
|
|
|
Factory 6 : |
Plot No. Z104-106,
Dahej SEZ Phase II, Taluka Vagra, District: Bharuch, Gujarat, India |
|
|
|
|
Project Site : |
Bileshwarpura, Taluka Kalol, District: Gandhinagar,
Gujarat, India |
|
|
|
|
Research and
Development Facility : |
Torrent Research Centre,
Near Kanoria Hospital, Village Bhat, District: Gandhinagar – 382428, Gujarat,
India |
|
Tel. No.: |
91-79-23269124/23969100 |
|
Fax No.: |
91-79-23269135/23969135/
23969124-34 |
|
E-Mail : |
|
|
|
|
|
Mumbai Office
: |
215, Atrium, 'B' 3rd Floor, Beside Courtyard Marriot Hotel, Andheri - Kurla Road, Andheri (East), Mumbai – 400093, Maharashtra, India |
DIRECTORS
AS ON 31.03.2017
|
Name : |
Mr. Sudhir Uttamlal Mehta |
|
Designation : |
Chairman Emeritus and Director |
|
Address : |
Akalpya, Opposite Jain Temple, Sarkhej
Gandhinagar Highway, Ahmedabad – 380058, Gujarat, India |
|
Date of Birth /Age : |
43 years |
|
Qualification : |
CA, CWA |
|
Date of Appointment : |
23.06.1982 |
|
DIN No.: |
00061871 |
|
|
|
|
Name : |
Mr. Samir Uttamlal Mehta |
|
Designation : |
Executive Chairman |
|
Address : |
Akalpya, Opposite Jain Temple, Sarkhej
Gandhinagar Highway, Ahmedabad – 380058, Gujarat, India |
|
Date of Birth /Age : |
52 years |
|
Qualification : |
B.Com., MBA |
|
Date of Appointment : |
01.08.2010 |
|
DIN No.: |
00061903 |
|
|
|
|
Name : |
Dr. Chaitanya Mavinkurve Dutt |
|
Designation : |
Whole-time Director (Research and Development) |
|
Address : |
Arvind Colony, Shahibaug, Ahmedabad –
380004, Gujarat, India |
|
Date of Birth /Age : |
65 years |
|
Qualification : |
M.D. (Medicine) |
|
Date of Appointment : |
01.01.2012 |
|
DIN No.: |
00110312 |
|
|
|
|
Name : |
Mr. Haigreve Khaitan |
|
Designation : |
Director |
|
Address : |
1104, Sterling Seaface, Dr. Annie Besant
Road, Worli, Mumbai – 400018, Maharashtra, India |
|
Date of Appointment : |
23.07.2012 |
|
DIN No.: |
00005290 |
|
|
|
|
Name : |
Mr. Shailesh Vishnubhai Haribhakti |
|
Designation : |
Director |
|
Address : |
10 and 11 Sahil Apartment, S K Barodawala
Road, 14 Altamount Road, Cumballa Hill, Mumbai – 400026, Maharashtra, India |
|
Date of Appointment : |
30.07.2011 |
|
DIN No.: |
00007347 |
|
|
|
|
Name : |
Mr. Markand Induprasad Bhatt |
|
Designation : |
Director |
|
Address : |
2 Panchsheel Enclave, Near Sundervan
Satellite Road, Ahmedabad – 380015, Gujarat, India |
|
Date of Appointment : |
19.10.2000 |
|
DIN No.: |
00061955 |
|
|
|
|
Name : |
Mr. Pradeep Kumar Bhargava |
|
Designation : |
Director |
|
Address : |
Flat No. 19, Cozy Retreat No. 3 Sindh
Housing Society, Aundh, Pune – 411007, Maharashtra, India |
|
Date of Appointment : |
26.07.2013 |
|
DIN No.: |
00525234 |
|
|
|
|
Name : |
Renu Challu |
|
Designation : |
Director |
|
Address : |
A 34/1, Afochs, Sainikpuri, Secunderabad – 500094,
Telangana, India |
|
Date of Appointment : |
27.07.2015 |
|
DIN No.: |
00157204 |
KEY EXECUTIVES
|
Name : |
Mr. Ashok Saligram Modi |
|
Designation : |
Executive Director and Chief Financial Officer |
|
Address : |
C-42, Akash Apartments, Near Judges
Bunglow, Premchand Nagar Road, Ahmedabad – 380015, Gujarat, India |
|
Date of Birth /Age : |
53 years |
|
Qualification : |
CA |
|
Date of Appointment : |
09.05.2014 |
|
PAN No.: |
AAPPM4742C |
|
|
|
|
Name : |
Mr. Mahesh Dwarkaprasad Agrawal |
|
Designation : |
VP (Legal) and Company Secretary |
|
Address : |
B-3,Surdhara Apartment, Mehta Park Society,
Near Hira Baug Crossing, Ambawadi, Ahmedabad – 380006, Gujarat, India |
|
Date of Appointment : |
28.11.2003 |
|
PAN No.: |
AAYPA3793N |
|
|
|
|
Audit and Risk
Management Committee : |
· Mr. Shailesh Haribhakti - Chairman · Mr. Haigreve Khaitan · Mr. Pradeep Bhargava · Mrs. Renu Challu |
|
|
|
|
Securities transfer and stakeholders
Relationship committee : |
· Mrs. Renu Challu – Chairperson · Mr. Shailesh Haribhakti · Mr. Haigreve Khaitan |
|
|
|
|
Nomination and
remuneration committee : |
· Mr. Ashish Nanda - Chairman · Mr. Markand Bhatt · Mr. Pradeep Bhargava |
|
|
|
|
Corporate social
responsibility committee : |
· Mr. Pradeep Bhargava – Chairman · Mrs. Renu Challu · Dr. Chaitanya Dutt |
SHAREHOLDING PATTERN
AS ON December 2017
|
Category of
Shareholder |
No.
of Shares |
Percentage
of Holding |
|
(A) Promoter
& Promoter Group |
120564720 |
71.25 |
|
(B) Public |
48658000 |
28.75 |
|
Grand Total |
169222720 |
100.00 |
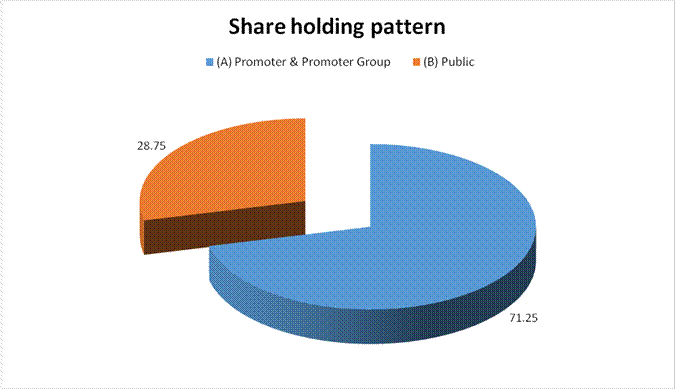
Statement showing shareholding pattern of the Promoter
and Promoter Group
|
Category of
Shareholder |
No.
of Shares |
Percentage
of Holding |
|
A1) Indian |
|
0.00 |
|
Individuals/Hindu
undivided Family |
900 |
0.00 |
|
Sudhir Uttamlal
Mehta (HUF) |
100 |
0.00 |
|
Samir Uttamlal
Mehta (HUF) |
100 |
0.00 |
|
Sudhir Uttamlal
Mehta |
100 |
0.00 |
|
Samir Uttamlal
Mehta |
100 |
0.00 |
|
Anita Sudhir
Mehta |
100 |
0.00 |
|
Sapna Samir Mehta |
100 |
0.00 |
|
Jinal Sudhir
Mehta |
100 |
0.00 |
|
Varun Sudhir
Mehta |
100 |
0.00 |
|
Aman Mehta |
100 |
0.00 |
|
Any Other
(specify) |
120563720 |
71.25 |
|
Torrent Private
Limited |
120563720 |
71.25 |
|
Sub Total A1 |
120564620 |
71.25 |
|
A2) Foreign |
|
0.00 |
|
Individuals
(NonResident Individuals/ Foreign Individuals) |
100 |
0.00 |
|
Shaan Mehta |
100 |
0.00 |
|
Sub Total A2 |
100 |
0.00 |
|
A=A1+A2 |
120564720 |
71.25 |
Statement showing shareholding pattern of the Public shareholder
|
Category of
Shareholder |
No.
of Shares |
Percentage
of Holding |
|
B1) Institutions |
0 |
0.00 |
|
Mutual Funds/ |
15249865 |
9.01 |
|
Aditya Birla Sun
Life Trustee Private Limited A/C Aditya Birla Sun Life Balanced 95 Fund |
2047847 |
1.21 |
|
HDFC Trustee Co
Limited- HDFC Multiple Yield Fund- Plan 2005 |
3099615 |
1.83 |
|
Franklin India
Pension Plan |
2374853 |
1.40 |
|
UTI-Mahila Unit
Scheme |
2056853 |
1.22 |
|
Alternate
Investment Funds |
381992 |
0.23 |
|
Foreign Portfolio
Investors |
16635986 |
9.83 |
|
Financial
Institutions/ Banks |
116471 |
0.07 |
|
Insurance
Companies |
36763 |
0.02 |
|
Sub Total B1 |
32421077 |
19.16 |
|
B2) Central
Government/ State Government(s)/ President of India |
0 |
0.00 |
|
B3)
Non-Institutions |
0 |
0.00 |
|
Individual share
capital upto INR 0.200 million |
7369257 |
4.35 |
|
Individual share
capital in excess of INR 0.200 million |
4914875 |
2.90 |
|
NBFCs registered
with RBI |
3435 |
0.00 |
|
Any Other
(specify) |
3949356 |
2.33 |
|
Trusts |
1400 |
0.00 |
|
NRI |
239853 |
0.14 |
|
Clearing Members |
44243 |
0.03 |
|
Bodies Corporate |
3422974 |
2.02 |
|
NRI – Non- Repat |
189983 |
0.11 |
|
IEPF |
50903 |
0.03 |
|
Sub Total B3 |
16236923 |
9.60 |
|
B=B1+B2+B3 |
48658000 |
28.75 |
BUSINESS DETAILS
|
Line of Business : |
The company engaged in research, development, manufacturing and marketing of generic pharmaceutical formulations. [Registered Activity] |
||||
|
|
|
||||
|
Products / Services
: |
|
||||
|
|
|
||||
|
Brand Names : |
Not Divulged |
||||
|
|
|
||||
|
Agencies Held : |
Not Divulged |
||||
|
|
|
||||
|
Exports : |
Not Divulged |
||||
|
|
|
||||
|
Imports : |
Not Divulged |
||||
|
|
|
||||
|
Terms : |
Not Divulged |
PRODUCTION STATUS – NOT AVAILABLE
GENERAL INFORMATION
|
Suppliers : |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Customers : |
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
No. of Employees : |
11781 (Approximately) |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Bankers : |
·
Axis Bank Limited, 2nd Floor, 3rd
Eye One, Near Panchvati Circle, C. G. Road, Ahmedabad – 380009, Gujarat,
India ·
The Hongkong and Shanghai Banking Corporation
Limited, 52/60, Mahatma Gandhi Road, Fort, Mumbai – 400001, Maharashtra,
India |
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
Facilities : |
NOTES: LONG TERM
BORROWINGS (i)
Term Loans from banks referred above to the
extent of : (a)
INR 3761.400 million (Previous year INR 5495.900
million) are secured by first pari-passu mortgage/ charge on immovable as
well as tangible movable assets, present and future, located at village
Indrad (Manufacturing facility on identified land), Bhat (Research facility),
Corporate office, Ahmedabad, all in Gujarat, and village Baddi (Manufacturing
facility) in Himachal Pradesh. (b)
INR 324.200 million (Previous year Nil) will be
secured by first pari-passu mortgage/ charge on immovable as well as tangible
movable assets, present and future, located at village Indrad (Manufacturing facility
on identified land), Bhat (Research facility), Corporate office, Ahmedabad,
all in Gujarat, and village Baddi (Manufacturing facility) in Himachal
Pradesh. The Company is in the process of creating charge. (c)
INR 2126.800 million (Previous year INR 9995.000
million) are secured by first pari-passu mortgage/ charge on immovable and
tangible movable assets, present and future located at Dahej (SEZ) in Gujarat
(Manufacturing facility) and Gangtok in Sikkim (Manufacturing facility) as
well as on certain identified trademarks of the Company including its future
line extensions. (d)
Nil (Previous year INR 2000.000 million) is
secured by first pari-passu mortgage/ charge on freehold land located at
village Rakanpur and freehold land and buildings located at Delhi, to the
extent of Company’s share in such properties, as well as on certain
identified trademarks of the Company. (e)
INR 3242.000 million (Previous year Nil) from
bank is secured by first charge on certain identified trademarks of the
Company including its future line extensions. (ii)
(a)
Non-convertible debentures referred above to the
extent of INR 4878.200 million (Previous year INR 4870.600 million) are
secured by first pari-passu mortgage/ charge on immovable and tangible
movable assets, present and future located at Dahej (SEZ) in Gujarat
(Manufacturing facility) and Gangtok in Sikkim (Manufacturing facility) as
well as on certain identified trademarks of the Company including its future
line extensions. (b)
Non-convertible debentures referred above to the
extent of INR 9986.200 million (Previous year Nil) will be secured by first
pari-passu mortgage/ charge on immovable and tangible movable assets, present
and future located at Dahej (SEZ) in Gujarat (Manufacturing facility) and
Gangtok in Sikkim (Manufacturing facility) as well as on certain identified
trademarks of the Company including its future line extensions. The Company
is in the process of creating charge. (iii) The terms of repayment of loan obligations on principal amount repayable in yearly installments, for the secured and unsecured long-term loans are as under: [INR in million]
(iv)
Maturity profile and rate of interest of non-convertible debentures are set
out as below: [INR in million]
|
|
Auditors : |
|
|
Name : |
Deloitte Haskins and Sells Chartered Accountants |
|
Address : |
19th Floor, Shapath-V, S.G. Highway, Ahmedabad – 380015, Gujarat, India |
|
Tel No.: |
91-79-66827300 |
|
Fax No.: |
91-79-66827400 |
|
Membership No.: |
33590 |
|
|
|
|
Memberships : |
Not Available |
|
|
|
|
Collaborators : |
Not Available |
|
|
|
|
Subsidiaries [having
100% proportion of ownership interest] : |
· Zao Torrent Pharma, Russia · Torrent Do Brasil Ltda., Brazil · Torrent Pharma Gmbh (TPG), Germany · Torrent Pharma Inc., USA · Torrent Pharma Philippines Inc., Philippines · Laboratorios Torrent, S.A. de C.V., Mexico · Torrent Australasia Pty Limited, Australia · Torrent Pharma (Thailand) Co., Limited, Thailand · Torrent Pharma S.R.L., Romania · Torrent Pharma (UK) Limited (TPUK), United Kingdom · Laboratories Torrent (Malaysia) SDN.BHD., Malaysia · Torrent Pharma France S.A.S., France |
|
|
|
|
Step-down
subsidiaries of TPG [having 100% proportion of ownership interest] : |
· Heumann Pharma Gmbh and Co. Generica KG, Germany · Heunet Pharma Gmbh, Germany · Norispharm Gmbh, Germany |
|
|
|
|
Step-down subsidiaries
of TPUK [having 100% proportion of ownership interest] : |
· Aptil Pharma Limited, United Kingdom |
|
|
|
|
Partnership Firm
[having 97% proportion of ownership interest] : |
· Torrent Pharmaceuticals (Sikkim), India |
CAPITAL STRUCTURE
AS ON 31.03.2017
Authorised Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
200000000 |
Equity Shares |
INR 5/- each |
INR 1000.000 Million |
|
2500000 |
Preference Shares |
INR 100/- each |
INR 250.000 Million |
|
|
|
|
|
|
|
TOTAL |
|
INR 1250.000
Million |
Issued Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
169236720 |
Equity Shares |
INR 5/- each |
INR 846.200 Million |
|
|
|
|
|
Subscribed & Paid-up Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
169222720 |
Equity Shares |
INR 5/- each |
INR 846.200 Million |
|
|
Amount originally paid up on 14,000 Equity shares of INR 5 each forfeited * Amount INR 35,000 |
|
* |
|
|
|
|
|
|
|
Total |
|
INR 846.200
Million |
i) Reconciliation of equity shares outstanding
at the beginning and at the end of the reporting year:
|
Particulars |
As on 31.03.2017 |
|
|
Numbers |
INR In Million |
|
|
As at beginning of the year |
169222720 |
846.200 |
|
Outstanding at the end of the year |
169222720 |
846.200 |
ii) Details
of shares allotted for consideration other than cash, bonus shares and shares
bought back in previous five financial years is as under:
The Company allotted 84,611,360 equity shares as fully paid up bonus shares of INR 5 each, pursuant to the shareholders' resolution passed on 12th July, 2013.
iii) Torrent Private Limited, the holding Company, holds 120,563,720 (previous year 86,115,472) equity shares of INR 5 each, equivalent to 71.25% (previous year 50.89%) of the total number of subscribed and paid up equity shares, which is the only shareholder holding more than 5% of total equity shares.
iv) The Company has one class of equity shares having par value of INR 5 each. Each shareholder is eligible for one vote per share held. The final dividend proposed by the Board of Directors is subject to the approval of the shareholders in the ensuing Annual General Meeting. In the event of liquidation, the equity shareholders are eligible to receive the remaining assets of the Company after distribution of all preferential amount, in proportion to shareholding.
FINANCIAL DATA
[all figures are
INR Million]
ABRIDGED
BALANCE SHEET (STANDALONE)
|
SOURCES OF FUNDS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
I.
EQUITY
AND LIABILITIES |
|
|
|
|
(1)Shareholders' Funds |
|
|
|
|
(a) Share Capital |
846.200 |
846.200 |
846.200 |
|
(b) Reserves & Surplus |
43690.900 |
36226.900 |
26209.700 |
|
(c) Money
received against share warrants |
0.000 |
0.000 |
0.000 |
|
|
|
|
|
|
(2) Share Application money pending
allotment |
0.000 |
0.000 |
0.000 |
|
Total
Shareholders’ Funds (1) + (2) |
44537.100 |
37073.100 |
27055.900 |
|
|
|
|
|
|
(3)
Non-Current Liabilities |
|
|
|
|
(a) long-term borrowings |
22087.400 |
17842.700 |
21852.200 |
|
(b) Deferred tax liabilities (Net) |
1007.300 |
1713.000 |
1888.400 |
|
(c) Other long term
liabilities |
68.800 |
90.200 |
69.400 |
|
(d) long-term provisions |
1263.400 |
1146.900 |
971.200 |
|
Total Non-current
Liabilities (3) |
24426.900 |
20792.800 |
24781.200 |
|
|
|
|
|
|
(4)
Current Liabilities |
|
|
|
|
(a) Short
term borrowings |
0.000 |
0.000 |
1000.000 |
|
(b) Trade
payables |
5950.600 |
6651.700 |
6302.300 |
|
(c) Other
current liabilities |
5336.500 |
7898.300 |
4137.100 |
|
(d) Short-term
provisions |
554.000 |
469.500 |
1692.600 |
|
Total Current
Liabilities (4) |
11841.100 |
15019.500 |
13132.000 |
|
|
|
|
|
|
TOTAL |
80805.100 |
72885.400 |
64969.100 |
|
|
|
|
|
|
II.
ASSETS |
|
|
|
|
(1)
Non-current assets |
|
|
|
|
(a) Fixed
Assets |
|
|
|
|
(i)
Tangible assets |
18328.700 |
9437.600 |
9053.100 |
|
(ii)
Intangible Assets |
15469.900 |
16286.200 |
18581.900 |
|
(iii)
Capital work-in-progress |
4730.700 |
9836.900 |
6277.800 |
|
(iv)
Intangible assets under development |
0.000 |
0.000 |
0.000 |
|
(v) Goodwill |
1436.300 |
1090.100 |
0.000 |
|
(b) Non-current Investments |
1484.100 |
1539.100 |
1503.500 |
|
(c) Deferred tax assets (net) |
0.000 |
0.000 |
0.000 |
|
(d) Long-term Loan and Advances |
1088.200 |
1517.100 |
1860.500 |
|
(e) Other
Non-current assets |
2101.700 |
1800.400 |
502.800 |
|
Total Non-Current
Assets |
44639.600 |
41507.400 |
37779.600 |
|
|
|
|
|
|
(2)
Current assets |
|
|
|
|
(a)
Current investments |
7874.300 |
7642.400 |
2834.800 |
|
(b)
Inventories |
10322.900 |
9701.300 |
7811.500 |
|
(c) Trade
receivables |
10184.700 |
9115.000 |
11813.200 |
|
(d) Cash
and cash equivalents |
782.200 |
823.700 |
696.800 |
|
(e)
Short-term loans and advances |
426.900 |
401.900 |
1034.100 |
|
(f) Other
current assets |
6574.500 |
3693.700 |
2999.100 |
|
Total
Current Assets |
36165.500 |
31378.000 |
27189.500 |
|
|
|
|
|
|
TOTAL |
80805.100 |
72885.400 |
64969.100 |
PROFIT
& LOSS ACCOUNT (STANDALONE)
|
|
PARTICULARS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
||
|
|
SALES |
|
|
|
||
|
|
|
Income |
45926.800 |
54394.800 |
34755.500 |
|
|
|
|
Other Income |
2996.000 |
2931.400 |
3056.700 |
|
|
|
|
TOTAL |
48922.800 |
57326.200 |
37812.200 |
|
|
|
|
|
|
|
||
|
Less |
EXPENSES |
|
|
|
||
|
|
|
Cost of Materials Consumed |
11764.000 |
10409.700 |
9682.000 |
|
|
|
|
Purchase of Stock-in-trade |
2209.900 |
1921.500 |
2007.800 |
|
|
|
|
Changes in Inventories of finished goods, work-in-progress and
stock-in-trade |
(109.500) |
(503.600) |
(855.700) |
|
|
|
|
Employee Benefits Expenses |
6990.400 |
5721.000 |
5074.100 |
|
|
|
|
Exceptional items |
0.000 |
1928.200 |
0.000 |
|
|
|
|
Other Expenses |
13800.500 |
10433.500 |
10413.800 |
|
|
|
|
TOTAL |
34655.300 |
29910.300 |
26322.000 |
|
|
|
|
|
|
|
||
|
|
PROFIT/(LOSS)
BEFORE INTEREST, TAX, DEPRECIATION AND AMORTISATION |
14267.500 |
27415.900 |
11490.200 |
||
|
|
|
|
|
|
||
|
Less |
FINANCIAL
EXPENSES |
2023.800 |
1798.300 |
1727.200 |
||
|
|
|
|
|
|
||
|
|
PROFIT/(LOSS)
BEFORE TAX, DEPRECIATION AND AMORTISATION |
12243.700 |
25617.600 |
9763.000 |
||
|
|
|
|
|
|
||
|
Less |
DEPRECIATION/
AMORTISATION |
2693.600 |
2134.100 |
1802.200 |
||
|
|
|
|
|
|
||
|
|
PROFIT/(LOSS)
BEFORE TAX |
9550.100 |
23483.500 |
7960.800 |
||
|
|
|
|
|
|
||
|
Less |
TAX |
1007.900 |
6053.600 |
1729.000 |
||
|
|
|
|
|
|
||
|
|
PROFIT/(LOSS)
AFTER TAX |
8542.200 |
17429.900 |
6231.800 |
||
|
|
|
|
|
|
||
|
|
EARNINGS IN
FOREIGN CURRENCY |
|
|
|
||
|
|
|
F.O.B. Value of Exports |
NA |
28934.300 |
14135.900 |
|
|
|
|
Interest |
NA |
134.300 |
8.700 |
|
|
|
|
Other income [Product registration dossiers & others] |
NA |
116.800 |
120.100 |
|
|
|
TOTAL EARNINGS |
NA |
29185.400 |
14264.700 |
||
|
|
|
|
|
|
||
|
|
IMPORTS |
|
|
|
||
|
|
|
Raw Materials and Packing
Material |
NA |
3197.900 |
2259.500 |
|
|
|
|
Components and Spares
Parts |
NA |
265.800 |
146.200 |
|
|
|
|
Capital Goods |
NA |
1120.300 |
394.900 |
|
|
|
TOTAL IMPORTS |
NA |
4584.000 |
2800.600 |
||
|
|
|
|
|
|
||
|
|
Earnings / (Loss)
Per Share (INR) |
50.48 |
102.99 |
36.83 |
||
CURRENT MATURITIES OF LONG TERM DEBT DETAILS
|
Particulars |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Current Maturities of Long term debt |
2368.900 |
4651.100 |
2360.600 |
|
Cash generated from operations |
9309.100 |
28927.700 |
10989.400 |
|
Net cash from operating activities |
6757.300 |
24030.000 |
10651.600 |
QUARTERLY
RESULTS
|
Particulars |
30.06.2017 (Unaudited) |
30.09.2017 (Unaudited) |
31.12.2017 (Unaudited) |
|
|
1st
Quarter |
2nd
Quarter |
3rd
Quarter |
|
Net sales |
8470.000 |
10260.000 |
10410.000 |
|
Total Expenditure |
7260.000 |
7900.000 |
7820.000 |
|
PBIDT (Excluding Other Income) |
1210.000 |
2360.000 |
2590.000 |
|
Other income |
1090.000 |
910.000 |
810.000 |
|
Operating Profit |
2300.000 |
3270.000 |
3400.000 |
|
Interest |
550.000 |
510.000 |
710.000 |
|
Exceptional Items |
NA |
NA |
NA |
|
PBDT |
1750.000 |
2760.000 |
2690.000 |
|
Depreciation |
740.000 |
780.000 |
890.000 |
|
Profit Before Tax |
1010.000 |
1980.000 |
1800.000 |
|
Tax |
170.000 |
400.000 |
1600.000 |
|
Provisions and
contingencies |
NA |
NA |
NA |
|
Profit after tax |
840.000 |
1580.000 |
200.000 |
|
Extraordinary Items |
NA |
NA |
NA |
|
Prior Period Expenses |
NA |
NA |
NA |
|
Other Adjustments |
NA |
NA |
NA |
|
Net Profit |
840.000 |
1580.000 |
200.000 |
KEY
RATIOS
EFFICIENCY RATIOS
|
PARTICULARS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Average Collection Days (Sundry
Debtors / Income * 365 Days) |
80.94 |
61.16 |
124.06 |
|
|
|
|
|
|
Account Receivables Turnover (Income / Sundry Debtors) |
4.51 |
5.97 |
2.94 |
|
|
|
|
|
|
Average Payment Days (Sundry Creditors / Purchases * 365 Days) |
155.43 |
196.89 |
196.78 |
|
|
|
|
|
|
Inventory Turnover (Operating Income / Inventories) |
1.38 |
2.83 |
1.47 |
|
|
|
|
|
|
Asset Turnover (Operating Income / Net Fixed Assets) |
0.37 |
0.77 |
0.34 |
LEVERAGE RATIOS
|
PARTICULARS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Debt Ratio ((Borrowing
+ Current Liabilities) / Total Assets) |
0.45 |
0.51 |
0.57 |
|
|
|
|
|
|
Debt Equity Ratio (Total Liability / Networth) |
0.55 |
0.61 |
0.93 |
|
|
|
|
|
|
Current Liabilities to Networth (Current Liabilities / Net Worth) |
0.27 |
0.41 |
0.49 |
|
|
|
|
|
|
Fixed Assets to Networth (Net Fixed Assets / Networth) |
0.87 |
0.96 |
1.25 |
|
|
|
|
|
|
Interest Coverage Ratio (PBIT / Financial Charges) |
7.05 |
15.25 |
6.65 |
PROFITABILITY RATIOS
|
PARTICULARS |
|
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
PAT to Sales ((PAT
/ Sales) * 100) |
% |
18.60 |
32.04 |
17.93 |
|
|
|
|
|
|
|
Return on Total Assets ((PAT / Total Assets) * 100) |
% |
10.57 |
23.91 |
9.59 |
|
|
|
|
|
|
|
Return on Investment (ROI) ((PAT / Networth) * 100) |
% |
19.18 |
47.01 |
23.03 |
SOLVENCY RATIOS
|
PARTICULARS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Current Ratio (Current
Assets / Current Liabilities) |
3.05 |
2.09 |
2.07 |
|
|
|
|
|
|
Quick Ratio ((Current Assets – Inventories) / Current Liabilities) |
2.18 |
1.44 |
1.48 |
|
|
|
|
|
|
G-Score Ratio Financial (Networth / Total Assets) |
0.55 |
0.51 |
0.42 |
|
|
|
|
|
|
G-Score Ratio Debt (Debts / Equity Capital) |
28.90 |
26.58 |
29.80 |
|
|
|
|
|
|
G-Score Ratio Liquidity (Total Current Assets / Total Current Liabilities) |
3.05 |
2.09 |
2.07 |
Total
Liability = Short-term Debt + Long-term Debt + Current Maturities of Long-term
debts
STOCK
PRICES
|
Face Value |
INR 5.00/- |
|
Market Value |
INR 1250.90/- |
FINANCIAL ANALYSIS
[all figures are
INR Million]
DEBT EQUITY RATIO
|
Particular |
31.03.2015 |
31.03.2016 |
31.03.2017 |
|
|
INR
In Million |
INR
In Million |
INR
In Million |
|
Share Capital |
846.200 |
846.200 |
846.200 |
|
Reserves & Surplus |
26209.700 |
36226.900 |
43690.900 |
|
Net
worth |
27055.900 |
37073.100 |
44537.100 |
|
|
|
|
|
|
Long-Term Borrowings |
21852.200 |
17842.700 |
22087.400 |
|
Short Term Borrowings |
1000.000 |
0.000 |
0.000 |
|
Current Maturities of Long term debt |
2360.600 |
4651.100 |
2368.900 |
|
Total
borrowings |
25212.800 |
22493.800 |
24456.300 |
|
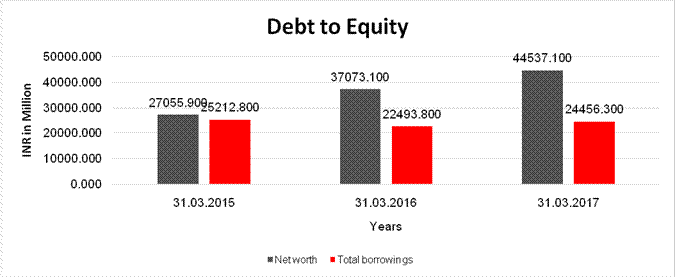
Debt/Equity
ratio |
0.932 |
0.607 |
0.549 |
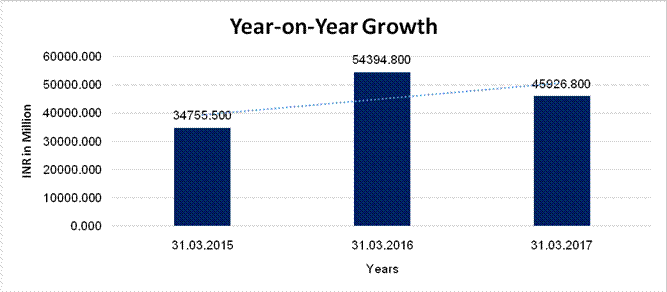
YEAR-ON-YEAR GROWTH
|
Year
on Year Growth |
31.03.2015 |
31.03.2016 |
31.03.2017 |
|
|
INR
In Million |
INR
In Million |
INR
In Million |
|
Sales |
34755.500 |
54394.800 |
45926.800 |
|
|
|
56.507 |
(15.568) |
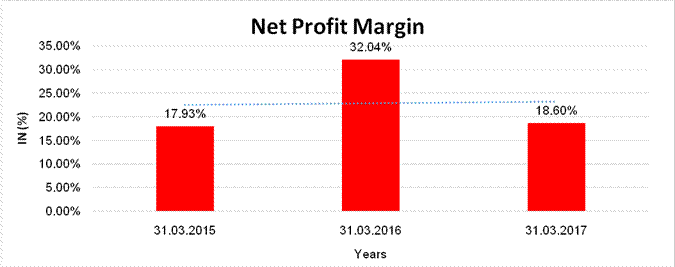
NET PROFIT MARGIN
|
Net
Profit Margin |
31.03.2015 |
31.03.2016 |
31.03.2017 |
|
|
INR
In Million |
INR
In Million |
INR
In Million |
|
Sales
|
34755.500 |
54394.800 |
45926.800 |
|
Profit/(Loss) |
6231.800 |
17429.900 |
8542.200 |
|
|
17.93% |
32.04% |
18.60% |
ABRIDGED
BALANCE SHEET (CONSOLIDATED)
|
SOURCES OF FUNDS |
31.03.2017 |
31.03.2016 |
|
I.
EQUITY AND LIABILITIES |
|
|
|
(1)Shareholders' Funds |
|
|
|
(a) Share Capital |
846.200 |
846.200 |
|
(b) Reserves &
Surplus |
42655.100 |
34094.800 |
|
(c) Money received
against share warrants |
0.000 |
0.000 |
|
|
|
|
|
(2) Non-controlling
interests |
4.900 |
4.600 |
|
Total Shareholders’ Funds
(1) + (2) |
43506.200 |
34945.600 |
|
|
|
|
|
(3) Non-Current
Liabilities |
|
|
|
(a) long-term borrowings |
22408.300 |
18500.800 |
|
(b) Deferred tax
liabilities (Net) |
1014.100 |
1717.500 |
|
(c) Other long term
liabilities |
79.300 |
96.500 |
|
(d) long-term provisions |
2630.400 |
2463.600 |
|
Total Non-current
Liabilities (3) |
26132.100 |
22778.400 |
|
|
|
|
|
(4) Current Liabilities |
|
|
|
(a) Short term borrowings |
0.000 |
33.200 |
|
(b) Trade payables |
22978.700 |
22602.200 |
|
(c) Other current
liabilities |
7326.000 |
8945.200 |
|
(d) Short-term provisions |
1306.900 |
1274.200 |
|
Total Current Liabilities
(4) |
31611.600 |
32854.800 |
|
|
|
|
|
TOTAL |
101249.900 |
90578.800 |
|
|
|
|
|
II.
ASSETS |
|
|
|
(1) Non-current assets |
|
|
|
(a) Fixed Assets |
|
|
|
(i) Tangible assets |
18714.100 |
9751.500 |
|
(ii) Intangible Assets |
16575.300 |
17590.500 |
|
(iii) Capital
work-in-progress |
5194.900 |
10415.800 |
|
(iv) Intangible assets
under development |
0.000 |
0.000 |
|
(v) Goodwill |
1595.000 |
1248.800 |
|
(b) Non-current
Investments |
0.900 |
0.900 |
|
(c) Deferred tax assets
(net) |
3098.800 |
2831.900 |
|
(d) Long-term Loan and Advances |
21.600 |
11.300 |
|
(e) Other Non-current
assets |
2207.400 |
1837.800 |
|
Total Non-Current Assets |
47408.000 |
43688.500 |
|
|
|
|
|
(2) Current assets |
|
|
|
(a) Current investments |
8035.500 |
7795.900 |
|
(b) Inventories |
15591.500 |
13579.800 |
|
(c) Trade receivables |
13441.900 |
14450.900 |
|
(d) Cash and cash
equivalents |
8937.200 |
6470.300 |
|
(e) Short-term loans and
advances |
21.500 |
16.900 |
|
(f) Other current assets |
7814.300 |
4576.500 |
|
Total Current Assets |
53841.900 |
46890.300 |
|
|
|
|
|
TOTAL |
101249.900 |
90578.800 |
PROFIT
& LOSS ACCOUNT (CONSOLIDATED)
|
|
PARTICULARS |
31.03.2017 |
31.03.2016 |
|
|
SALES |
|
|
|
|
Income |
58569.200 |
66869.300 |
|
|
Other Income |
2233.000 |
2255.700 |
|
|
TOTAL |
60802.200 |
69125.000 |
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
Cost of Materials
Consumed |
11803.300 |
10465.500 |
|
|
Purchases of
Stock-in-Trade |
7510.600 |
6784.900 |
|
|
Changes in inventories of
finished goods, work-in-progress and Stock-in-Trade |
(1397.300) |
(1477.700) |
|
|
Employees benefits
expense |
9934.100 |
8423.500 |
|
|
Exceptional items |
0.000 |
1936.300 |
|
|
Other expenses |
16945.800 |
15342.800 |
|
|
TOTAL |
44796.500 |
41475.300 |
|
|
|
|
|
|
|
PROFIT/ (LOSS) BEFORE INTEREST, TAX, DEPRECIATION AND AMORTISATION
|
16005.700 |
27649.700 |
|
|
|
|
|
|
Less |
FINANCIAL EXPENSES |
2055.600 |
1839.800 |
|
|
|
|
|
|
|
PROFIT / (LOSS) BEFORE
TAX, DEPRECIATION AND AMORTISATION |
13950.100 |
25809.900 |
|
|
|
|
|
|
Less |
DEPRECIATION/ AMORTISATION
|
3069.200 |
2375.500 |
|
|
|
|
|
|
|
PROFIT/ (LOSS) BEFORE TAX |
10880.900 |
23434.400 |
|
|
|
|
|
|
Less |
TAX |
1545.100 |
6101.700 |
|
|
|
|
|
|
|
PROFIT/ (LOSS) AFTER TAX
|
9335.800 |
17332.700 |
|
|
|
|
|
|
|
Earnings /
(Loss) Per Share (INR) |
55.17 |
102.42 |
LOCAL AGENCY FURTHER INFORMATION
|
Sr. No. |
Check list by
info agents |
Available in
Report (Yes/No) |
|
1 |
Year of establishment |
Yes |
|
2 |
Constitution of the entity -Incorporation
details |
Yes |
|
3 |
Locality of the entity |
Yes |
|
4 |
Premises details |
No |
|
5 |
Buyer visit details |
-- |
|
6 |
Contact numbers |
Yes |
|
7 |
Name of the person contacted |
No |
|
8 |
Designation of contact person |
No |
|
9 |
Promoter’s background |
Yes |
|
10 |
Date of Birth of Proprietor / Partners /
Directors |
Yes |
|
11 |
Pan Card No. of Proprietor / Partners |
No |
|
12 |
Voter Id Card No. of Proprietor / Partners |
No |
|
13 |
Type of business |
Yes |
|
14 |
Line of Business |
Yes |
|
15 |
Export/import details (if applicable) |
No |
|
16 |
No. of employees |
Yes |
|
17 |
Details of sister concerns |
Yes |
|
18 |
Major suppliers |
No |
|
19 |
Major customers |
No |
|
20 |
Banking Details |
Yes |
|
21 |
Banking facility details |
Yes |
|
22 |
Conduct of the banking account |
-- |
|
23 |
Financials, if provided |
Yes |
|
24 |
Capital in the business |
Yes |
|
25 |
Last accounts filed at ROC, if applicable |
Yes |
|
26 |
Turnover of firm for last three years |
Yes |
|
27 |
Reasons for variation <> 20% |
-- |
|
28 |
Estimation for coming financial year |
No |
|
29 |
Profitability for last three years |
Yes |
|
30 |
Major shareholders, if available |
Yes |
|
31 |
Litigations that the firm/promoter
involved in |
-- |
|
32 |
Market information |
-- |
|
33 |
Payments terms |
No |
|
34 |
Negative Reporting by Auditors in the
Annual Report |
No |
CORPORATE INFORMATION
The company is a public limited company incorporated and domiciled in India. The address of its registered office is Torrent House, Off Ashram Road, Ahmedabad – 380 009, Gujarat, India. The Company is one of the leading Indian Pharmaceutical Company engaged in research, development, manufacturing and marketing of generic pharmaceutical formulations. The Company’s research and development facility is located in the state of Gujarat, India, and its manufacturing facilities are located in the states of Gujarat, Himachal Pradesh, Madhya Pradesh, Andhra Pradesh and Sikkim.
MANAGEMENT DISCUSSION
AND ANALYSIS
GLOBAL PHARMACEUTICALS MARKET
Global Economy:
Global economic growth in 2016 is estimated at 3.1% down from 3.4% in 2015 with advanced economies decline from 2.1% in 2015 to 1.7% in 2016 and emerging economies from 4.2% in 2015 to 4.1% in 2016. However, from second half of 2016, global economy gained some momentum especially in advanced economies due to recovery in investment, manufacturing and trade. These include resumption in global demand, strengthening of commodity prices lifting global inflation, reducing deflationary pressures and positive developments in financial markets. Stronger than expected pickup in growth in advanced economies in latter part of 2016 reflects projected cyclical recovery in global manufacturing and uptick in business confidence especially after the United States elections. In the United Kingdom too, the growth remained strong where spending proved resilient in the aftermath of June, 2016 referendum in favor of leaving the European Union (BREXIT). Activity surprised on the upside in Japan due to strong net exports, as well as in euro market countries, such as Germany and Spain, as a result of strong domestic demand. Economic performance across emerging markets and developing economies has remained mixed. Whereas China’s growth remained strong, reflecting continued policy support, economic activity has slowed in India because of the impact of demonetization as well as in Brazil, which has been mired in deep recession. With the rebound in activity in advanced economies, stabilization of commodity prices and sustainable pattern of growth in developing economies, the overall global economy is projected to grow at 3.5% in 2017.
Global Pharma Market:
Although the life sciences sector operates around the issues relating to cost and pricing, clinical and operational innovation, customer and consumer engagement and regulatory compliance, it is expected to have sales growth for the next several years due to favorable demographic trends and significant unmet medical needs. The global pharmaceutical sales grew by around 7% in the year 2016 compared to the year 2015 and key growth drivers continue to be shift towards use of generic medicines accompanied by patent expiries mainly in the regulated market and higher growth in Pharmerging markets. During 2016, share of United States, Europe and Emerging markets in global pharmaceutical sales remained relatively static compared to 2015.
The largest pharmaceutical market, US is estimated to be approximately US$ 462 Bn in 2016 registering a growth of around 6 to 7% (from 12% in 2015). The removal of historic impact of breakthrough cures for Hepatitis C treatment, along with the increased level of patent expiry impact have combined to reduce the growth rate by half. The market is expected to grow at a CAGR of 7 to 8% through 2021 due to fewer patent expiries and launches of more innovative medicines which is a reflection of a shift in the balance of the “innovation cycle” —the amount of new medicines being launched and utilized compared to the value of branded medicines that are facing new generic competition.
The European market is estimated to be approximately US$ 152 Bn with a negligible growth of around 1% in 2016. Relatively weak economic growth in the region, budget concerns arising from adopting and paying for innovative medicines and mechanisms to control healthcare spending will lead to a slower CAGR of around 1 to 4% through 2021. The Pharmerging markets mainly led by China, Brazil, India and Russia are estimated to be US$ 243 Bn and expected to grow slow at a CAGR of 6 to 9% through 2021, as compared to CAGR of around 10% of last five years. Pharmerging markets will grow more slowly than in last five years as China, the largest Pharmerging market slows to 5 to 8% growth from an average of 12% in last five years. Slowing macroeconomic growth along with delay in healthcare access expansion programs is impacting medicine usage in pharmerging markets. Over 90% of the medicines used in Pharmerging markets are off patent generic products. Volume usage growth is projected to slow to 3 to 6% from 2017-2021 compared to 6.6% from 2012-2016.
The global spending on medicines is forecasted to reach close to US$ 1.5 trillion by 2021, an increase of about 33% over the 2016 level, growing at 4 to 7% only slightly slower than 6.2% growth over prior five years. Developed market spending growth will be driven by original brands and innovation in specialty medicines while Pharmerging markets will continue to be driven by off patent generic products. Innovation in specialty medicines will continue lifting the share of global spending from 30% in 2016 to 35% in 2021 driven by the adoption of new breakthrough medicines.
The volumes are projected to increase by 3% CAGR globally in the next five years compared to 6% from 2006-2011 and 3% from 2011-2016. The global volume increased from nearly 2.5 trillion doses of medicines (standard units) in 2006 to almost 4 trillion doses in 2016 with ¾ of that growth from pharmerging markets. People in pharmerging countries will consume more than half of the medicines used globally, consistent with the more than half of the world’s population who live there.
Future of Generics:
For several years now, the generic market has grown at a faster rate than the total pharmaceutical market. This recent growth was driven by the “Patent cliff,” whereby many blockbuster molecules lost patent protection, support for generics from governments, new complex generics coming into the market and industry consolidation. This has compelled generic drug manufacturers to strengthen the new product pipeline by increasing research and development spend and partnering with contract research and manufacturing organizations (CROs and CMOs). Global generic drug market accounted for around USD 200 billion in 2015 and is expected to reach approximately USD 381 billion by 2021, with a CAGR of around 10.8% between 2016 and 2021.
New emerging markets of developing countries and low cost of generic drugs are majorly responsible for thickening the growth of generic drugs market. In these markets where affordability and access to medicines are key influencers, legacy generic medicines for treatment of infection, pain, respiratory, cardiovascular and mental diseases continue to offer growth. Whereas in developed markets, specialty therapies such as oncology, autoimmune and hepatitis therapies show a continued focus. Generics market is affected by high competition within the globe. In future, new developing and uncovered markets may be responsible for generation of new opportunities.
US represents the world’s largest market for generic drugs by revenue. With 90% medicines dispensed being generics, the transformation of US generic drug industry has been catalyzed by continuous expiration of drug patents, rising ageing population, increasing prevalence of chronic diseases, continuous efforts by Government and health care service providers to control their healthcare expenditures. In the US, the number of generic drug approvals hit a record high in 2015, due to a new program that aimed to speed up approvals. Japan is also becoming a significant user of generic medicines and the Government is targeting 80% generic penetration by 2021. Emerging economies including Brazil, Russia, India, China and Mexico are expected to exhibit a significant increase in generic volume growth. This will be driven by expansion of universal healthcare and growing incidence of lifestyle diseases such as obesity and diabetes, brought about by increases in disposable income and the expansion of the middle class.
Opportunities for generics therefore exist across the globe with an increasing demand for affordable, safe, and effective medicines. Each region has specific needs and opportunities that will reflect the particular, and sometimes peculiar requirements at a country level.
MERGERS AND ACQUISITIONS (M&A):
M&A continues to be one of the life sciences companies principal growth strategies. Most transactions focus on factors such as consolidating in the face of pricing pressure, strengthening existing product portfolios, replenishing pipelines depleted by patent expiry, deepening capabilities in priority areas, entering new and/or emerging markets and acquiring innovative technologies to leverage current assets or generate cost saving synergies. The sentiment and outlook for M & A activity remains quite favorable.
Companies are venturing abroad for targets sensing bargains and opportunities to expand their global footprints. Pressure to reduce costs and boost shareholder value is prompting companies to divest low growth assets and presenting other companies with opportunities to invest in high-value niche product lines. Additionally, the ever-changing market is ripe for sector convergence with technology as well as consolidation within certain sectors.
UNSECURED LOANS:
|
PARTICULARS |
31.03.2017 INR
In Million |
31.03.2016 INR
In Million |
|
LONG TERM
BORROWINGS |
|
|
|
Unsecured term loans from others |
112.600 |
106.200 |
|
|
|
|
|
Total |
112.600 |
106.200 |
STANDALONE
UNAUDITED FINANCIAL RESULTS FOR THE QUARTER AND NINE MONTHS ENDED 31ST
DECEMBER 2017
(INR In Million)
|
Particulars |
Quarter ended |
Nine Months ended |
|
|
|
31.12.2017 |
30.09.2017 |
31.12.2017 |
|
|
Unaudited |
||
|
Revenue |
|
|
|
|
Net Sales (including excise duty) |
10120.000 |
10020.000 |
28480.000 |
|
Other Operating Income |
290.000 |
240.000 |
660.000 |
|
Revenue
from Operations |
10410.000 |
10260.000 |
29140.000 |
|
Other Income |
810.000 |
910.000 |
2810.000 |
|
Total Revenue |
11220.0000 |
11170.000 |
31950.000 |
|
EXPENSES |
|
|
|
|
Cost of materials consumed |
2570.0000 |
1770.000 |
6730.000 |
|
Purchase of Stock in Trade |
1290.000 |
410.000 |
2400.000 |
|
Changes in inventories of finished goods and
work-in-progress |
(1390.000) |
620.000 |
(1650.000) |
|
Employee benefits expense |
2010.000 |
2070.000 |
5950.000 |
|
Finance costs |
710.000 |
510.000 |
1770.000 |
|
Depreciation and Amortization expenses |
890.000 |
780.000 |
2410.000 |
|
Other Expenditure |
3340.000 |
3030.000 |
9550.000 |
|
Total
Expenses |
9420.000 |
9190.000 |
27160.000 |
|
Profit from ordinary activities before tax |
1800.000 |
1980.000 |
4790.000 |
|
Tax
Expense |
|
|
|
|
-
Current tax |
390.000 |
420.000 |
1030.000 |
|
-
Deferred tax |
1210.000 |
(20.000) |
1140.000 |
|
Total
Tax Expense |
1600.000 |
400.000 |
2170.000 |
|
Profit
/ (Loss) after Tax |
200.000 |
1580.000 |
2620.000 |
|
Other
Comprehensive Income |
|
|
|
|
- Items that will not be reclassified to profit or loss |
(20.000) |
(30.000) |
(80.000) |
|
- Income tax elating to items that will not be reclassified to profit or loss |
10.000 |
10.000 |
30.000 |
|
- Items that will be reclassified to profit or loss |
210.000 |
(750.000) |
(1210.000) |
|
- Income tax elating to items that will be reclassified to profit or loss |
(70.000) |
260.000 |
420.000 |
|
Total other Comprehensive Income |
130.000 |
(510.000) |
(840.000) |
|
Total
Comprehensive Income |
330.000 |
1070.000 |
1780.000 |
|
Paid-up Equity Share Capital (Face value INR 5/- per
share) |
84.62 |
84.62 |
84.62 |
|
Other Equity excluding Revaluation Reserves |
|
|
|
|
Earnings
per Share (EPS) - INR |
1.25 |
9.31 |
15.53 |
NOTES:
1. The above results were reviewed by the Audit and Risk Management Committee and approved by the Board of Directors in their respective meetings held on 08-Feb-2018. The auditor have carried out an audit of the above said results. There is no qualification in the Auditors report on this statement of financial results.
2. The Company operates in a single segment i.e Generic Formulation Business.
3. The Company has completed acquisition of branded business of Unichem Laboratories Limited (Acquisition) for India and Nepal on a going concern basis by way of slump sale on 14-Dec-2017. The Company is in the process of making a final determination of fair value and the same is expected to be completed by 31-Mar-2018. Pending this the amortization of intangible assets in the books of accounts has been taken based on management estimates.
4. The deferred tax expense during the quarter and nine months ended 31-Dec-2017 includes an amount of INR 990.000 million pertaining to the Acquisition.
5. Results for the year ended 31-Mar-2017 and quarter and nine months ended 31-Dec-2016 include exceptional revenues and profits primarily on account of launch of a new product in the USA which had limited competition.
6.
(a) The listed Non-Convertible Debentures of the company aggregating INR 14900.000 million as on 31-Dec-2017 (previous year ended INR 4900.000 million) are secured by way of first pari passu charge created through mortgage on certain specified immovable & movable assets and hypothecation of identified trademarks of the Company and the asset cover thereof exceeds hundred percent of the principal amount of the said debentures.
(b) The listed Non-Convertible Debentures of the company aggregating INR 10000.000 million as on 31-Dec-2017 (previous year ended INR 10000.000 million) to be secured by way of first pari passu charge on certain specified immovable & movable assets and identified trademarks of the Company.
7. The Board of Directors in their meeting held on 08-Feb-2018, declared an interim equity dividend of INR 9.00 per equity share of INR 5.00 each fully paid up for the year 2017-18. The aggregate amount of interim equity dividend proposed to be distributed is INR 1833.100 million including dividend distribution tax of INR 310.000 million.
8. The figures for the corresponding previous period have been restated/regrouped wherever necessary, to make them comparable.
INDEX OF CHARGES:
|
S No |
SRN |
Charge Id |
Charge Holder Name |
Date of Creation |
Date of Modification |
Date of Satisfaction |
Amount |
Address |
|
1 |
G45704533 |
100104568 |
IDBI TRUSTEESHIP SERVICES LIMITED |
12/05/2017 |
- |
- |
5000000000.0 |
ASIAN BUILDING, GROUND FLOOR, 17,R. KAMANI MARG, BALLARD ESTATEMUMBAIMH400001IN |
|
2 |
G33438094 |
100072246 |
CITI BANK N.A. |
20/12/2016 |
- |
- |
3316000000.0 |
14TH FLOOR, FIFC, PLOT NO C - 54 & C - 55, G BLOCKBANDRA KURLA COMPLEX, BANDRA (EAST) MUMBAI MH400051IN |
|
3 |
G12588687 |
10607195 |
IDBI TRUSTEESHIP SERVICES LIMITED |
09/11/2015 |
14/09/2016 |
- |
2500000000.0 |
ASIAN BUILDING, GROUND FLOOR, 17,R. KAMANI MARG, BALLARD ESTATE MUMBAI MH400001IN |
|
4 |
G12589263 |
10607198 |
IDBI TRUSTEESHIP SERVICES LIMITED |
09/11/2015 |
14/09/2016 |
- |
2400000000.0 |
ASIAN BUILDING, GROUND FLOOR, 17,R. KAMANI MARG, BALLARD ESTATE MUMBAI MH400001IN |
|
5 |
G05080767 |
10525000 |
AXIS BANK LIMITED |
20/09/2014 |
10/03/2016 |
- |
3000000000.0 |
2ND FLOOR, 3RD EYE ONE, NEAR PANCHVATI CIRCLE,C. G. ROAD, AHMEDABAD GJ380009IN |
|
6 |
G12589750 |
10500496 |
IDBI TRUSTEESHIP SERVICES LIMITED |
07/06/2014 |
14/09/2016 |
- |
14000000000.0 |
ASIAN BUILDING, GROUND FLOOR, 17,R. KAMANI MARG, BALLARD ESTATE MUMBAI MH400001IN |
|
7 |
G21364526 |
10447179 |
HSBC BANK (MAURITIUS) LIMITED |
13/08/2013 |
13/10/2016 |
- |
1100000000.0 |
HSBC CENTRE18 CYBERCITY, EBENE MAURITIUS NA000MU |
|
8 |
B73713752 |
10421600 |
BNP PARIBAS |
05/04/2013 |
- |
- |
500000000.0 |
203, SAKAR-II, ELLISBRIDGE, AHMEDABAD GJ380006IN |
|
9 |
C50587385 |
10399912 |
THE HONGKONG AND SHANGHAI BANKING CORPORATION LIMITED |
31/12/2012 |
23/03/2015 |
- |
500000000.0 |
52/60, MAHATMA GANDHI ROAD FORT MUMBAI MH400001IN |
|
10 |
A02291342 |
10010037 |
STANDARD CHARTERED BANK |
04/07/2006 |
- |
- |
448000000.0 |
ABHIJEET II, GROUND FLOOR, MITHAKHALI SIX ROADS AHMEDABAD GJ380006IN |
CONTINGENT
LIABILITIES:
|
PARTICULARS |
31.03.2017 INR
In Million |
31.03.2016 INR
In Million |
|
Claims against the Company not acknowledged as debts: |
|
|
|
Disputed demand of income tax for which appeals have been preferred |
33.900 |
52.100 |
|
Disputed employee state insurance contribution liability under E.S.I. Act, 1948 |
104.400 |
92.900 |
|
Disputed demand of excise and service tax |
900.500 |
749.800 |
|
Disputed demand of local sales tax and C.S.T. |
9.900 |
7.500 |
|
Disputed cases at labour court / industrial court |
39.000 |
47.900 |
|
Disputed bonus liability under Payment of Bonus (Amendment) Act, 2015 |
4.000 |
4.000 |
|
Total |
1091.700 |
954.200 |
|
Against the claims not acknowledged as debts, the Company has paid INR 2.300 million (previous year INR 1.300 million). The expected outflow will be determined at the time of final outcome in respect of the concerned matter. No amount is expected to be reimbursed. |
||
FIXED ASSETS:
Tangible Assets
· Office buildings
· Factory buildings
· Plant and equipments
· Laboratory equipment
· Electrical equipment
· Furniture and Fixtures
· Office equipment
· Computer equipment
Intangible Assets
· Software
· Product license
· Brands
· Non-compete fees
· Drug master files
PRESS RELEASE:
TORRENT PHARMA
ACQUIRES BRANDS FROM NOVARTIS AG
May 8th
2017
Ahmedabad based Torrent Pharma, today announced that it has completed the acquisition of Regestrone and Pregachieve brands for India from global Pharma player Novartis AG, Switzerland. These brands are widely prescribed by Gynaecologists for the management of Abnormal Uterine bleeding, Peri & Post- menopausal symptoms and Infertility.
Women Healthcare is an important therapy area for Torrent Pharma, which it intends to focus in the future. With acquisition of key brands of Elder like Shelcal and Deviry, impetus on this therapy area has also increased. Regestrone and Pregachieve will further fortify the Hormone Segment. The use of Hormones amongst the Gynaecologists is on an increase because of lifestyle changes.
This acquisition would reaffirm Torrent’s commitment towards the key important therapy of women healthcare.
About Torrent Pharma
Torrent Pharma, with annual revenues of more than Rs. 6600 crores is the flagship Company of the Rs. 18,300 crores Torrent Group.
Torrent Pharma continues to be at the forefront of the Indian pharmaceutical industry with many of its products ranking amongst the top 500 brands (AIOCD Dataset) in India. Its widespread international presence also includes several markets where Torrent is amongst the leading pharmaceutical companies in the respective countries.
Torrent has a fully equipped Research Centre, employing almost 900 scientists, to support the Company's operations and product pipeline for both Domestic and Overseas markets. The Company's manufacturing plants located at Indrad, Baddi, Sikkim, Dahej, Pithampur and Vizag have facilities to produce Formulations and Bulk drugs. The plants are approved by authorities from various regulated and semi regulated markets like US, UK, Brazil, Germany, Australia and South Africa.
TORRENT PHARMA MAY
RAISE INR 15000.000 MILLION IN BID FOR SANOFI GENERICS BUSINESS
March 14 2018
The proposed fundraising will help Torrent Pharma strengthen its balance
sheet as it prepares a bid for Sanofi’s generics unit Zentiva
Mumbai: Torrent Pharma Ltd
is likely to raise at least INR 15000.000
million by selling
shares to institutional investors, as it prepares a bid for the generic drugs
unit of France’s Sanofi, two people aware of the development said.
Torrent Pharma is readying a €2 billion (INR
16000.000 million) binding bid for Sanofi’s unit Zentiva NV, Mint reported on 7 March. It has tied
up funding from several banks for the bid, the deadline for which ends on 28
March, the report added.
“Torrent has appointed JP Morgan and JM
Financial to help it raise INR 15000.000
million to support
its acquisition efforts. The firm has already had a couple of rounds of
meetings with probable investors for the purpose of the fund-raising,” one of
the people cited above said, requesting anonymity, as he is not authorized to
speak to reporters.
The proposed fundraising will help the pharma
company strengthen its balance sheet as it looks to absorb yet another major
asset, he added.
On 1 March, Torrent told exchanges that its
shareholders had approved raising as much as INR
50000.000 million of equity
capital through a qualified institutional placement (QIP) and other routes.
According to the second person cited above,
Torrent has a strong track record of growing through acquisitions and therefore
it should not see much difficulty in convincing investors to participate in its
QIP. He too requested anonymity. In January, the company announced the
acquisition of US-based generic pharmaceuticals company, Biopharm Inc (BPI).
BPI is a maker of oral solutions, suspensions and suppositories.
In November last year, it acquired the
domestic business of Unichem Laboratories for close to INR
36000.000 million. In 2014, Torrent acquired the branded domestic
formulations business of Elder Pharma for INR
20000.000 million and
acquired select brands of Novartis, and manufacturing plants of Zyg Pharma and
Glochem Industries in 2015.
According to analysts, Torrent has a track
record of turning around acquired assets and controlling its debt post
acquisition and that should comfort investors. “Post the Unichem acquisition,
net debt increased to INR 40000.000
million (as
against INR 23500.000 million in September 2017). Torrent Pharma’s track
record of successful turnaround of the acquired Elder business and debt reduction
post the acquisition in FY14, provides comfort,” Spark Capital said in a
research note last month.
Emails sent on Friday to Torrent Pharma and
JM Financial did not elicit any response. JP Morgan declined to comment.
Torrent’s acquisition of Zentiva, if it
happens, will be the biggest outbound transaction by an Indian drug maker. As
on date, the record for the largest outbound acquisition stands in the name of
Lupin Limited, which in 2015 acquired Gavis Pharmaceuticals LLC. and Novel
Laboratories Inc. for $880 million.
Sanofi had acquired Zentiva in 2009 for close
to $2.6 billion. It is currently the third-largest generics company in Europe
and sells medicines for cardiovascular, gastrointestinal, anti-inflammatory,
pain management, metabolic and blood disorders, among others.
Incorporated in 1971, Torrent Pharma has a
presence in markets such as the US, UK, Germany, Brazil and Mexico. On Monday,
its shares fell 0.38%, or INR 4.95, to INR
1305.65
while the benchmark Sensex shed 0.18%, or 61.16 points, to end the day at
33,856.78.
CMT REPORT (Corruption, Money Laundering & Terrorism]
The Public Notice information has been collected from various sources
including but not limited to: The Courts,
1] INFORMATION ON
DESIGNATED PARTY
No records exist designating subject or any of its beneficial owners,
controlling shareholders or senior officers as terrorist or terrorist
organization or whom notice had been received that all financial transactions involving
their assets have been blocked or convicted, found guilty or against whom a
judgement or order had been entered in a proceedings for violating
money-laundering, anti-corruption or bribery or international economic or
anti-terrorism sanction laws or whose assets were seized, blocked, frozen or
ordered forfeited for violation of money laundering or international
anti-terrorism laws.
2] Court Declaration :
No records exist to suggest that subject is
or was the subject of any formal or informal allegations, prosecutions or other
official proceeding for making any prohibited payments or other improper
payments to government officials for engaging in prohibited transactions or
with designated parties.
3] Asset Declaration :
No records exist to suggest that the property or assets of the subject
are derived from criminal conduct or a prohibited transaction.
4] Record on Financial
Crime :
Charges or conviction
registered against subject: None
5] Records on Violation of
Anti-Corruption Laws :
Charges or
investigation registered against subject: None
6] Records on Int’l
Anti-Money Laundering Laws/Standards :
Charges or
investigation registered against subject: None
7] Criminal Records
No
available information exist that suggest that subject or any of its principals
have been formally charged or convicted by a competent governmental authority
for any financial crime or under any formal investigation by a competent
government authority for any violation of anti-corruption laws or international
anti-money laundering laws or standard.
8] Affiliation with
Government :
No record
exists to suggest that any director or indirect owners, controlling
shareholders, director, officer or employee of the company is a government
official or a family member or close business associate of a Government
official.
9] Compensation Package :
Our market
survey revealed that the amount of compensation sought by the subject is fair
and reasonable and comparable to compensation paid to others for similar
services.
10] Press Report :
No press reports / filings exists on
the subject.
CORPORATE GOVERNANCE
MIRA INFORM as part of its Due Diligence do provide comments on
Corporate Governance to identify management and governance. These factors often
have been predictive and in some cases have created vulnerabilities to credit
deterioration.
Our Governance Assessment focuses principally on the interactions
between a company’s management, its Board of Directors, Shareholders and other
financial stakeholders.
CONTRAVENTION
Subject is not known to have contravened any existing local laws,
regulations or policies that prohibit, restrict or otherwise affect the terms
and conditions that could be included in the agreement with the subject.
FOREIGN EXCHANGE RATES
|
Currency |
Unit
|
INR |
|
US Dollar |
1 |
INR 65.06 |
|
|
1 |
INR 92.06 |
|
Euro |
1 |
INR 80.37 |
INFORMATION DETAILS
|
Information
Gathered by : |
GYT |
|
|
|
|
Analysis Done by
: |
NIS |
|
|
|
|
Report Prepared
by : |
NKT |
SCORE FACTORS
|
DEMERIT POINTS |
|
|
|
--BANK CHARGES |
YES/NO |
YES |
|
--LITIGATION |
YES/NO |
NO |
|
--OTHER ADVERSE INFORMATION |
YES/NO |
NO |
|
MERIT POINTS |
|
|
|
--SOLE DISTRIBUTORSHIP |
YES/NO |
NO |
|
--EXPORT ACTIVITIES |
YES/NO |
NO |
|
--AFFILIATION |
YES/NO |
YES |
|
--LISTED |
YES/NO |
YES |
|
--OTHER MERIT FACTORS |
YES/NO |
YES |
RATING EXPLANATIONS
|
Credit Rating |
Explanation |
Rating Comments |
|
A++ |
Minimum Risk |
Business dealings permissible with minimum
risk of default |
|
A+ |
Low Risk |
Business dealings permissible with low
risk of default |
|
A |
Acceptable Risk |
Business dealings permissible with
moderate risk of default |
|
B |
Medium Risk |
Business dealings permissible on a regular
monitoring basis |
|
C |
Medium High Risk |
Business dealings permissible preferably
on secured basis |
|
D |
High Risk |
Business dealing not recommended or on
secured terms only |
|
NB |
New Business |
No recommendation can be done due to
business in infancy stage |
|
NT |
No Trace |
No recommendation can be done as the
business is not traceable |
NB is stated where there is insufficient information to facilitate rating. However, it is not to be considered as unfavourable.
This score serves as a reference to assess
SC’s credit risk and to set the amount of credit to be extended. It is
calculated from a composite of weighted scores obtained from each of the major
sections of this report. The assessed factors are as follows:
·
Financial
condition covering various ratios
·
Company
background and operations size
·
Promoters
/ Management background
·
Payment
record
·
Litigation
against the subject
·
Industry
scenario / competitor analysis
·
Supplier
/ Customer / Banker review (wherever available)
This report is issued at
your request without any risk and responsibility on the part of MIRA INFORM
PRIVATE LIMITED (MIPL) or its officials.

