|
|
|
MIRA INFORM REPORT
|
Report No. : |
496438 |
|
Report Date : |
27.03.2018 |
IDENTIFICATION DETAILS
|
Name : |
AUROBINDO PHARMA LIMITED AUROBINDO BIOLOGICS UNIT OF AUROBINDO PHARMA LIMITED |
|
|
|
|
Registered
Office : |
Plot No. 2, Maithri Vihar, Behind Maithri Vanam, Ameerpet,
Hyderabad – 500 038, Telangana |
|
Tel. No.: |
91-40-23741083 |
|
|
|
|
Country : |
India |
|
|
|
|
Financials (as
on) : |
31.03.2017 |
|
|
|
|
Date of
Incorporation : |
26.12.1986 |
|
|
|
|
Com. Reg. No.: |
36-015190 |
|
|
|
|
Capital
Investment / Paid-up Capital : |
INR 585.900 Million |
|
|
|
|
CIN No.: [Company Identification
No.] |
L24239TG1986PLC015190 |
|
|
|
|
IEC No.: [Import-Export Code No.] |
2588000011 |
|
|
|
|
TIN: |
36840211594 |
|
|
|
|
TAN No.: [Tax Deduction &
Collection Account No.] |
HYDA01477A |
|
|
|
|
GSTIN : [Goods & Service Tax
Registration No.] |
36AABCA7366H1ZL |
|
|
|
|
PAN No.: [Permanent Account No.] |
AABCA7366H |
|
|
|
|
Legal Form : |
A Public Limited Liability Company. The Company’s Shares are Listed on
the Stock Exchanges. |
|
|
|
|
Line of Business : |
Manufacturing of generic pharmaceuticals and
active pharmaceutical ingredients. (Registered Activity) |
|
|
|
|
No. of Employees
: |
13982 (Approximately) |
RATING & COMMENTS
(Mira Inform has adopted New Rating mechanism w.e.f. 23rd
January 2017)
|
MIRA’s Rating : |
A+ |
|
Credit Rating |
Explanation |
Rating Comments |
|
A+ |
Low Risk |
Business dealings permissible with low risk
of default |
|
Maximum Credit Limit : |
USD 240000000 |
|
|
|
|
Status : |
Excellent |
|
|
|
|
Payment Behaviour : |
Regular |
|
|
|
|
Litigation : |
Clear |
|
|
|
|
Comments : |
Subject was incorporated in the year 1986. It is a manufacturer of Generic Pharmaceuticals and active
pharmaceuticals Ingredients. For the financial year 2017, the revenue of the company has increased
by 4.92% along with profit margin of 17.45%. The robust financial profile of the company marked by healthy networth
base along with strong debt coverage indicators due to low debt balance sheet
profile. The company has favourable Earning Per Share (EPS) of INR 29.16
against a face value (FV) of INR 01. Rating also derives strength from company’s established track record
of business. The company has its share price trading at around INR 557.35
against the Face Value (FV) of INR 01
on BSE as on 24rd March, 2018. As per the unaudited quarterly financials of December 2017, the
company has achieved revenue of INR 26862.800 million and has reported profit
margin of 16.54%. However, these rating strengths are partially offset by unfavourable
gap between trade payables and trade receivables. Business is active. Payment seems to be regular. In view of aforesaid, the company can be considered for business
dealings at usual trade terms and conditions.
Note: Ms. Vijaya (Office Executive) has claimed that “Aurobindo
Biologics” is a unit of “AUROBINDO PHARMA LIMITED”. |
NOTES :
Any query related to this report can be made
on e-mail : infodept@mirainform.com
while quoting report number, name and date.
ECGC Country Risk Classification List
|
Country Name |
Previous Rating (30.09.2017) |
Current Rating (31.12.2017) |
|
India |
A1 |
A1 |
|
Risk Category |
ECGC
Classification |
|
Insignificant |
A1 |
|
Low Risk |
A2 |
|
Moderately Low Risk |
B1 |
|
Moderate Risk |
B2 |
|
Moderately High Risk |
C1 |
|
High Risk |
C2 |
|
Very High Risk |
D |
EXTERNAL AGENCY RATING
|
Rating Agency Name |
India Rating |
|
Rating |
Fund Based Working Capital (AA+) |
|
Rating Explanation |
High degree of safety and very low credit
risk |
|
Date |
12.05.2017 |
|
Rating Agency Name |
India Rating |
|
Rating |
Commercial Paper (A1+) |
|
Rating Explanation |
Very strong degree of safety and carry
lowest credit risk |
|
Date |
12.05.2017 |
RBI DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available RBI Defaulters’ list.
BIFR (Board for Industrial & Financial Reconstruction) LISTING
STATUS
Subject’s name is not listed as a Sick Unit in
the publicly available BIFR (Board for Industrial & Financial
Reconstruction) list as of 23.03.2018.
IBBI (Insolvency and Bankruptcy Board of India) LISTING STATUS
Subject’s name is not listed in the publicly
available IBBI (Insolvency and Bankruptcy Board of India) list as of report
date.
EPF (Employee Provident Fund) DEFAULTERS’ LIST STATUS
Subject’s name is not enlisted as a defaulter
in the publicly available EPF (Employee Provident Fund) Defaulters’ list as of
31-03-2016.
INFORMATION DENIED BY
|
Name : |
Ms. Vijaya |
|
Designation : |
Office Executive |
|
Contact No.: |
91-40-23040261 |
|
Date : |
27.03.2018 |
91-40-6672500 (Ringing)
LOCATIONS
|
Registered Office/
Corporate Office : |
Plot No. 2, Maithri Vihar, Behind Maithri Vanam, Ameerpet, Hyderabad – 500 038, Telangana, India |
|
Tel. No.: |
91-40-23741083 / 23741084 / 23744919 / 66725000 / 66725401 / 23736370 |
|
Fax No.: |
91-40-23746833 / 23741080 / 23748112 / 23747340 |
|
E-Mail : |
|
|
Website : |
|
|
|
|
|
Corporate Office : |
The Water Mark Building, Plot No.11, Survey No.9, Kondapur, Hitech City, Hyderabad - 500 084, Telangana, India. |
|
Tel. No.: |
91-40-66725000/ 1200 |
|
Fax No.: |
91-40-67074044/4059 |
|
|
|
|
Factory 1 : |
Survey No.379, 385, 386, 388 to 396 and 269, Borpatla, Hatnoor Mandal, Medak District- 502 296, Telangana, India |
|
|
|
|
Factory 2 : |
Plot No.103/A and 104/A, SVCIE, Industrial Development Area, Bollaram, Jinnaram (Mandal) Medak District - 500 092, Telangana, India |
|
|
|
|
Factory 3 : |
Survey No.313 and 314 Bachupally, Quthubullapur Mandal, Range Reddy District - 500 090, Telangana, India |
|
|
|
|
Factory 4 : |
Plot No.4 in Survey No.151 and Plot Nos.34 to 48 in Survey No. part of 146, 150, 151, 152, 153 and 154 situated in Phase-III, SPIIC, EPIP, IDA, Pashamylaram, Patancheru Mandal, Medak District - 502 307, Telangana, India |
|
|
|
|
Factory 5 : |
Plot No.68 to 70, 73 to 91, 95, 96, 260 and 261 Industrial Development Area, Chemical Zone, Pashamylaram, Patancheru Mandal, Medak District - 502 307, Telangana, India |
|
|
|
|
Factory 6 : |
Survey No. 329/39 and 329/47, Chitkul Village, Patancheru Mandal, Medak District - 502 307, Telangana, India |
|
|
|
|
Factory 7 : |
Survey No. 411/P, 425/P, 434/P, 435/P and 458/P, Plot No.S1(Part), Special Economic Zone (Pharma), APIIC, Green Industrial Park, Polepally Village, Jedcherla Mandal, Mahaboob Nagar - 509 302, Telangana, India |
|
|
|
|
Factory 8 : |
Survey No.10 and 13, Gaddapothram, Industrial Development Area - Kazipally Industrial Area, Jinnaram Mandal, Medak District - 502 319, Telangana, India |
|
|
|
|
Factory 9 : |
Survey No.369, 370 371 and 374, Gundlamachanoor, Hatnoora Mandal, Medak District - 502 296, Telangana, India |
|
|
|
|
Factory 10 : |
Plot No 16, APIIC, Multiproduct SEZ at Survey No.3(P) to 6(P) and 413(P) and 416(P) Palchur Village and 113 Part of Palepalem Village, Naidupeta Mandal, PSR Nellore District - 524 126,, Andhra Pradesh, India |
|
|
|
|
Factory 11 : |
Survey No.61-69, Industrial Development Area, Pydibhimavaram, Ranasthalam Mandal, Srikakulam District - 532 409, Telangana, India |
|
|
|
|
Factory 12 : |
Survey No.314, Bachupally, Quthubullapur Mandal, Range Reddy District - 500 090, Telangana, India |
|
|
|
|
Factory 13 : |
JN Pharma City, Plot No. 17, Road No.10, 11 & 19, 20, E Bonangi Village, Parawada, Visakhapatnam District - 531 021, Andhra Pradesh, India |
|
|
|
|
Factory 14 : |
JN Pharma City, Plot No. 17A, Road No.10,11 & 19,20, E Bonangi Village, Parawada, Visakhapatnam District - 531 021, Andhra Pradesh |
|
|
|
|
Factory 15 : |
Plot No.S-5/B, S-6 and S-7, Survey No.408 to 412, 418 to 435, 437 to 445, 452 to 459, TSIIC, SEZ, Polepally Village, Jedcherla Mandal, Mahaboobnagar District - 509302, Telangana, India |
|
|
|
|
Factory 16 : |
Survey No.77 and 78, Indrakaran Village, Kandi Mandal, Sangareddy District - 502 203, Telangana, India |
|
|
|
|
Factory 17 : |
Survey No.69, 70, 71 and 72, Indrakaran Village, Kandi Mandal, Sangareddy District - 502 203, Telangana, India |
|
|
|
|
APLRC – I |
Survey No.313 and 314 Bachupally, Quthubullapur Mandal, Ranga Reddy District - 500 090, Telangana, India |
|
|
|
|
APLRC - II |
Survey No.71 and 72, 502203, Telangana, India |
|
|
|
|
Bhiwadi Unit |
1128, RIICO Phase-III, Bhiwadi - 301 019, Rajasthan, india |
|
|
|
|
Branch Office : |
202, 2nd Floor, Inizio, Cardinal Gracious Road, Chakala, Andheri East, Mumbai – 400099, Maharashtra, Indias |
DIRECTORS
As on: 2017
|
Name : |
Mr. Govindarajan Narayanan |
|
Designation : |
Managing Director |
|
Address : |
8-2-293/82/Be/26, Plot No.26 Navanirman Nagar Colony, Jubilee Hills, Hyderabad-500033, Telangana, India |
|
Date of Birth/Age : |
47 Years |
|
Experience : |
22 years |
|
Qualification : |
B.E. (Mechanical) |
|
Date of Appointment : |
01.06.2012 |
|
DIN No.: |
00050482 |
|
|
|
|
Name : |
Kambam Nityananda Reddy |
|
Designation : |
Wholetime Director |
|
Address : |
Plot No.11, 8-2-269/N/11, Road No.2 Banjarahills, Navodaya Colony, Hyderabad-500034, Telangana, India |
|
Date of Birth/Age : |
57 Years |
|
Qualification : |
Master’s Degree in Science (Organic Chemistry) |
|
Date of Appointment : |
01.06.2012 |
|
DIN No.: |
01284195 |
|
|
|
|
Name : |
Mettu Madan Mohan Reddy |
|
Designation : |
Wholetime Director |
|
Address : |
H-101, Madhura Nagar, Ameerpet, Hyderabad-500038, Telangana, India |
|
Date of Birth/Age : |
55 Years |
|
Qualification : |
Master’s Degree in Science (Organic
Chemistry) |
|
Date of Appointment : |
01.01.2013 |
|
DIN No.: |
01284266 |
|
|
|
|
Name : |
Mr. Sivakumaran Meenakshisunderam |
|
Designation : |
Wholetime Director |
|
Address : |
Plot No.83, Lumbini Springs Kothaguda, Gachibowli Main Road, Hyderabad-500032, Telangana, India |
|
Date of Birth/Age : |
72 Years |
|
Qualification : |
Master’s Degree in Science |
|
Experience : |
42 years |
|
Date of Appointment : |
01.09.2009 |
|
DIN No.: |
01284320 |
|
|
|
|
Name : |
Penaka Sarath Chandra Reddy |
|
Designation : |
Whole time Director |
|
Address : |
Plot No. 46/8-3-169, Siddartha Nagar, Behind Vengalrao Nagar, Hyderabad-500038, Telangana, India |
|
Date of Birth/Age : |
30 Years |
|
Qualification : |
Graduate in Business Administration |
|
Date of Appointment : |
30.05.2016 |
|
DIN No.: |
01628013 |
|
|
|
|
Name : |
Mrs. Kannan Ragunathan |
|
Designation : |
Director |
|
Date of Birth/Age : |
52 Years |
|
Experience : |
30 years |
|
Address : |
P No 15b, 6-3-1099/1100, Somajiguda, Hyderabad-500082, Telangana, India |
|
Date of Appointment : |
01.06.2012 |
|
DIN No.: |
00523576 |
|
|
|
|
Name : |
Penaka Venkata Ramprasad Reddy |
|
Designation : |
Director |
|
Address : |
Plot No.46, Behind Vengalrao Nagar Sidhartha Nagar Hyderabad-500038, Telangana, India |
|
Date of Birth/Age : |
57 Years |
|
Qualification : |
Post-Graduate |
|
Date of Appointment : |
01.12.2012 |
|
DIN No.: |
01284132 |
|
|
|
|
Name : |
Mr. Avnit Bimal Singh |
|
Designation : |
Director |
|
Address : |
Plot No.108 Miyapur, JPN Colony, Hyderabad-500050, Telangana, India |
|
Date of Appointment : |
25.03.2015 |
|
DIN No.: |
01316166 |
|
|
|
|
Name : |
Mr. Sitarama Murthy Mandavilli |
|
Designation : |
Director |
|
Address : |
F.No.401, Siva Sai Apts Road No.-9, West Marredpally, Secunderabad-500003, Andhra Pradesh, India |
|
Date of Birth/Age : |
72 Years |
|
Qualification : |
Masters in Electronics |
|
Date of Appointment : |
27.09.2007 |
|
DIN No.: |
01694236 |
|
|
|
|
Name : |
Mrs. Savita Mahajan |
|
Designation : |
Director |
|
Address : |
89, Villa Greens Gandipet (Narsinghi-PS), Hyderabad-500075, Telangana, India |
|
Date of Appointment : |
16.12.2017 |
|
DIN No.: |
06492679 |
KEY EXECUTIVES
|
Name : |
Mr. Subramanian Santhanam |
|
Designation : |
Chief Financial Officer |
|
Address : |
Flat No.622, Block 3 Manasarovar Heights, Phase III, Hasmatpe T, Secunderabad-500009, Andhra Pradesh, India |
|
Date of Appointment : |
01.07.2014 |
|
PAN No: |
ARTPS2748D |
|
|
|
|
Name : |
Mr. Adi Reddy Baddigam |
|
Designation : |
Company Secretary (from 01.06.2016) |
|
Address : |
Flat No.204, E Block, Vertex Sadguru Apartments, Nizampet Road, Kukatpally Hyderabad-500072, Telangana, India |
|
Date of Appointment : |
01.06.2016 |
|
PAN No: |
ADXPB2434M |
|
|
|
|
Name : |
Mr. A. Mohan Rami Reddy |
|
Designation : |
VP (Legal) and Company Secretary (up to 31.05.2016) |
|
|
|
|
Name : |
Mr. Krishna |
|
Designation : |
Investor Relations Department |
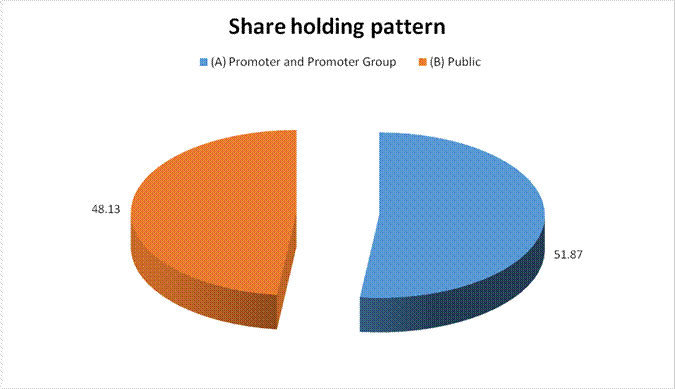
MAJOR SHAREHOLDERS / SHAREHOLDING PATTERN
AS ON: DECEMBER 2017
|
Category of
shareholder |
No. of fully paid up
equity shares held |
Shareholding as a %
of total no. of shares (calculated as per SCRR, 1957)As a % of (A+B+C2) |
|
|
(A) Promoter and Promoter Group |
303925934 |
51.87 |
|
|
(B) Public |
281956475 |
48.13 |
|
|
Grand Total |
585882409 |
100.00 |
Statement showing
shareholding pattern of the Promoter and Promoter Group
|
Category of
shareholder |
Total nos.
shares held |
Shareholding as a
% of total no. of shares (calculated as per SCRR, 1957)As a % of (A+B+C2) |
|
|
A1) Indian |
0.00 |
||
|
Individuals/Hindu undivided Family |
72164968 |
12.32 |
|
|
M SIVAKUMARAN |
14491360 |
2.47 |
|
|
K RAJESWARI |
1975500 |
0.34 |
|
|
PENAKA NEHA REDDY |
130000 |
0.02 |
|
|
K NITYANANDA REDDY |
25359572 |
4.33 |
|
|
KAMBAM PRASAD REDDY |
301156 |
0.05 |
|
|
K SURYAPRAKASH REDDY |
7380 |
0.00 |
|
|
M SUMANTH KUMAR REDDY |
1600000 |
0.27 |
|
|
KAMBAM KIRTHI REDDY |
20700000 |
3.53 |
|
|
KAMBAM SPOORTHI |
76,00000 |
1.30 |
|
|
Any Other (specify) |
213760966 |
36.49 |
|
|
RPR SONS ADVISORS PRIVATE LMITED, MRS.P.SUNEELA RANI
(JOINTLY HOLDING) |
196376250 |
33.52 |
|
|
AXIS CLINICALS LIMITED |
658000 |
0.11 |
|
|
AXIS CLINICALS LIMITED, TRIDENT CHEMPHAR LIMITED, RPR SONS
ADVISORS PRIVATE LMITED(JOINTLY HOLDING) |
16726716 |
2.85 |
|
|
Sub Total A1 |
285925934 |
48.80 |
|
|
A2) Foreign |
0.00 |
||
|
Individuals (NonResident Individuals/ Foreign Individuals) |
18000000 |
3.07 |
|
|
VENKATA RAMPRASAD REDDY PENAKA |
18000000 |
3.07 |
|
|
Sub Total A2 |
18000000 |
3.07 |
|
|
A=A1+A2 |
303925934 |
51.87 |
Statement showing
shareholding pattern of the Public shareholder
|
Category &
Name of the Shareholders |
No. of fully paid
up equity shares held |
Shareholding %
calculated as per SCRR, 1957 As a % of (A+B+C2) |
|
|
B1) Institutions |
0 |
0.00 |
|
|
Mutual Funds/ |
79808825 |
13.62 |
|
|
BIRLA SUN LIFE TRUSTEE COMPANY PRIVATE LIMITED AC BIRLA SUN
LIFE BALANCED 95 FUND |
10868403 |
1.86 |
|
|
HDFC TRUSTEE COMPANY LIMITED A/C HDFC GROWTH FUND |
31783784 |
5.42 |
|
|
RELIANCE CAPITAL TRUSTEE COMPANY LIMITED A/C RELIANCE
VISION FUND. |
11099560 |
1.89 |
|
|
SBI ARBITRAGE OPPORTUNITIES FUND |
13523081 |
2.31 |
|
|
Alternate Investment Funds |
40751 |
0.01 |
|
|
Foreign Portfolio Investors |
114972358 |
19.62 |
|
|
Financial Institutions/ Banks |
1484256 |
0.25 |
|
|
Insurance Companies |
1773158 |
0.30 |
|
|
Sub Total B1 |
198079348 |
33.81 |
|
|
B2) Central Government/ State Government(s)/ President of
India |
0 |
0.00 |
|
|
B3) Non-Institutions |
0 |
0.00 |
|
|
Individual share capital upto INR 0.200 million |
44684004 |
7.63 |
|
|
Individual share capital in excess of INR 0.200 Million |
13970726 |
2.38 |
|
|
JHUNJHUNWALA RAKESH RADHESHYAM |
6550000 |
1.12 |
|
|
NBFCs registered with RBI |
68576 |
0.01 |
|
|
Any Other (specify) |
25153821 |
4.29 |
|
|
Trusts |
2697063 |
0.46 |
|
|
NRI – Repat |
2407714 |
0.41 |
|
|
Clearing Members |
1555791 |
0.27 |
|
|
NRI – Non- Repat |
612634 |
0.10 |
|
|
Bodies Corporate |
17879437 |
3.05 |
|
|
Foreign Individuals |
1182 |
0.00 |
|
|
Sub Total B3 |
83877127 |
14.32 |
|
|
B=B1+B2+B3 |
281956475 |
48.13 |
BUSINESS DETAILS
|
Line of Business : |
Manufacturing of generic pharmaceuticals and active
pharmaceutical ingredients. (Registered Activity) |
||||
|
|
|
||||
|
Products : |
|
||||
|
|
|
||||
|
Brand Names : |
Not Available |
||||
|
|
|
||||
|
Agencies Held : |
Not Available |
||||
|
|
|
||||
|
Exports : |
Not Divulged |
||||
|
|
|
||||
|
Imports : |
Not Divulged |
||||
|
|
|
||||
|
Terms : |
Not Divulged |
PRODUCTION STATUS NOT AVAILABLE
GENERAL INFORMATION
|
Suppliers : |
|
||||||||||||||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||||||||||||||
|
Customers : |
|
||||||||||||||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||||||||||||||
|
No. of Employees : |
13982 (Approximately) |
||||||||||||||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||||||||||||||
|
Bankers : |
|
||||||||||||||||||||||||||||||||||||
|
|
|
||||||||||||||||||||||||||||||||||||
|
Facilities : |
Terms of borrowings
|
|
Auditors 1: |
|
|
Name : |
BSR and Associates LLP Chartered Accountants |
|
Address : |
Salarpuria Khowledge City, Orwell, 6th Floor, Unit – 3,
Sy.No.83/1, Plot No.2, Raidurg, Hyderabad – 500081, Telangana, India |
|
Tel No: |
91-40-71822000 |
|
Fax. No: |
91-40-71822399 |
|
|
|
|
Auditors 2: |
|
|
Name : |
S.R. Batliboi and Associates LLP Chartered Accountants |
|
Address : |
Oval Office, 18 Ilabs Centre, Hi-Tech City, Madhapur, Hyderabad-500 081, Telangana, India |
|
Tel No: |
91-40-67362000 |
|
Fax. No: |
91-40-67362200 |
|
|
|
|
Internal Auditors : |
|
|
Name : |
KPMG Chartered Accountants |
|
Address : |
1st Floor, Lodha Excellus, Apollo Mills Compound, N.M. Joshi Marg, Mahalaxmi, Mumbai-400 011, Maharashtra, India |
|
|
|
|
Memberships : |
--- |
|
|
|
|
Collaborators : |
-- |
|
|
|
|
Subsidiaries |
|
|
|
|
|
Joint ventures |
|
|
|
|
|
Enterprises over which
key management personnel or their relatives exercise significant influence |
|
CAPITAL STRUCTURE
AS ON 31.03.3017
Authorised Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
660000000 |
Equity Shares |
INR 1/- each |
INR 660.000 Million |
|
1000000 |
Preference Shares |
INR 100/- each |
INR 100.000 Million |
|
|
Total |
|
INR 760.000 Million |
Issued, Subscribed & Paid-up Capital :
|
No. of Shares |
Type |
Value |
Amount |
|
|
|
|
|
|
585882409 |
Equity Shares |
INR 1/- each |
INR 585.900
Million |
|
|
|
|
|
B) Terms/rights
attached to equity shares
The Company has only one class of equity shares having a par values of INR 1 per share. Each holder of equity shares is entitled to one vote per share. The Company declares and pays dividends in Indian rupees. The dividend proposed by the Board of Directors is subject to the approval of shareholders in the ensuing Annual General Meeting. For the year ended 31 March 2017, the amount of dividend per share declared as distributions to equity shareholders was INR 2.500 million (March 31, 2016: INR 2.500 million). Refer Note 14(c) for details of dividend declared/recognized in financial statements. In the event of liquidation of the Company, the holders of equity shares will be entitled to receive remaining assets of the Company, after distribution of all preferential amounts. However, no such preferential amounts exist currently. The distribution will be in proportion to the number of equity shares held by the shareholders.
C) Details of
shareholders holding more than 5% equity shares in the Company
|
|
As at Mach 31, 2017 |
|
|
Numbers |
% holding |
|
|
RPR Sons Advisors Private Limited and Mrs. P. Suneela Rani (Joint holders) |
196376250 |
33.52 |
|
Total |
196376250 |
33.52 |
As per records of the Company, including its register of shareholders/members and other declarations received from shareholders, the above shareholding represents legal ownership of shares and beneficial ownership is with RPR Enterprises, a partnership firm.
d) Aggregate number
of bonus shares issued, shares issued for consideration other than cash during
the period of five years immediately preceeding the reporting date:
|
|
As at Mach 31, 2017 |
|
Numbers |
|
|
Equity shares alloted as fully paid bonus shares by capitalization of securities premium |
291982275 |
FINANCIAL DATA
[all figures are
in Rupees Million]
ABRIDGED
BALANCE SHEET (STANDALONE)
|
SOURCES OF FUNDS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
I.
EQUITY
AND LIABILITIES |
|
|
|
|
(1)Shareholders' Funds |
|
|
|
|
(a) Share Capital |
585.900 |
585.200 |
292.000 |
|
(b) Reserves & Surplus |
83775.900 |
68073.300 |
53663.400 |
|
(c) Money
received against share warrants |
0.000 |
0.000 |
0.000 |
|
|
|
|
|
|
(2) Share Application money pending
allotment |
0.000 |
0.000 |
0.000 |
|
Total Shareholders’
Funds (1) + (2) |
84361.800 |
68658.500 |
53955.400 |
|
|
|
|
|
|
(3)
Non-Current Liabilities |
|
|
|
|
(a) long-term borrowings |
1189.000 |
3347.600 |
6871.600 |
|
(b) Deferred tax liabilities (Net) |
0.000 |
0.000 |
0.000 |
|
(c) Other long term
liabilities |
0.000 |
0.000 |
0.000 |
|
(d) long-term
provisions |
208.700 |
212.600 |
226.500 |
|
Total Non-current
Liabilities (3) |
1397.700 |
3560.200 |
7098.100 |
|
|
|
|
|
|
(4)
Current Liabilities |
|
|
|
|
(a) Short term
borrowings |
27512.700 |
34171.500 |
23142.200 |
|
(b) Trade
payables |
12677.000 |
12786.700 |
11460.400 |
|
(c) Other
current liabilities |
3665.000 |
5979.400 |
5186.000 |
|
(d) Short-term
provisions |
615.000 |
499.300 |
370.400 |
|
Total Current Liabilities
(4) |
44469.700 |
53436.900 |
40159.000 |
|
|
|
|
|
|
TOTAL |
130229.200 |
125655.600 |
101212.500 |
|
|
|
|
|
|
II.
ASSETS |
|
|
|
|
(1)
Non-current assets |
|
|
|
|
(a) Fixed
Assets |
|
|
|
|
(i)
Tangible assets |
32116.200 |
25681.300 |
21900.100 |
|
(ii)
Intangible Assets |
0.000 |
0.000 |
0.000 |
|
(iii)
Capital work-in-progress |
8783.100 |
7421.400 |
2271.900 |
|
(iv)
Intangible assets under development |
286.700 |
0.000 |
0.000 |
|
(b) Non-current Investments |
16819.300 |
11833.300 |
9939.500 |
|
(c) Deferred tax assets (net) |
0.800 |
68.600 |
506.700 |
|
(d) Long-term Loan and Advances |
463.600 |
296.000 |
82.400 |
|
(e) Other
Non-current assets |
2770.800 |
2610.000 |
1782.600 |
|
Total Non-Current
Assets |
61240.500 |
47910.600 |
36483.200 |
|
|
|
|
|
|
(2)
Current assets |
|
|
|
|
(a)
Current investments |
0.200 |
0.200 |
196.400 |
|
(b)
Inventories |
24336.200 |
24316.000 |
21450.500 |
|
(c) Trade
receivables |
37736.300 |
43809.700 |
38137.900 |
|
(d) Cash
and cash equivalents |
336.400 |
3308.100 |
111.300 |
|
(e)
Short-term loans and advances |
102.500 |
94.800 |
88.900 |
|
(f) Other
current assets |
6477.100 |
6216.200 |
4744.300 |
|
Total
Current Assets |
68988.700 |
77745.000 |
64729.300 |
|
|
|
|
|
|
TOTAL |
130229.200 |
125655.600 |
101212.500 |
PROFIT
& LOSS ACCOUNT (STANDALONE)
|
|
PARTICULARS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
|
SALES |
|
|
|
|
|
Revenue from Operations |
97812.100 |
93227.600 |
80951.000 |
|
|
Other Income |
1359.500 |
1883.600 |
672.200 |
|
|
TOTAL |
99171.600 |
95111.200 |
81623.200 |
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
Cost of Materials
Consumed |
46041.900 |
44258.000 |
39203.300 |
|
|
Purchases of
Stock-in-Trade |
112.100 |
63.100 |
157.600 |
|
|
Changes in inventories of
finished goods, work-in-progress and Stock-in-Trade |
313.200 |
(1415.900) |
(1590.900) |
|
|
Employees benefits
expense |
9273.000 |
8070.500 |
6687.500 |
|
|
Other expenses |
18332.400 |
17734.000 |
13989.300 |
|
|
TOTAL |
74072.600 |
68709.700 |
58446.800 |
|
|
|
|
|
|
|
Less |
PROFIT/ (LOSS) BEFORE INTEREST, TAX, DEPRECIATION AND
AMORTISATION |
25099.000 |
26401.500 |
23176.400 |
|
|
|
|
|
|
|
Less |
FINANCIAL EXPENSES |
451.600 |
2293.100 |
1321.400 |
|
|
|
|
|
|
|
|
PROFIT / (LOSS) BEFORE
TAX, DEPRECIATION AND AMORTISATION |
24647.400 |
24108.400 |
21855.000 |
|
|
|
|
|
|
|
Less/ Add |
DEPRECIATION/
AMORTISATION |
2861.700 |
2630.000 |
2451.500 |
|
|
|
|
|
|
|
|
PROFIT/ (LOSS) BEFORE TAX |
21785.700 |
21478.400 |
19403.500 |
|
|
|
|
|
|
|
Less |
TAX |
4718.100 |
5211.400 |
4240.000 |
|
|
|
|
|
|
|
|
PROFIT/ (LOSS) AFTER TAX |
17067.600 |
16267.000 |
15163.500 |
|
|
|
|
|
|
|
|
EARNINGS IN FOREIGN
CURRENCY |
|
|
|
|
|
F.O.B. Value of Exports |
75760.700 |
70926.800 |
62514.100 |
|
|
Others |
77.000 |
88.300 |
99.000 |
|
|
TOTAL EARNINGS |
75837.700 |
71015.100 |
62613.100 |
|
|
|
|
|
|
|
|
IMPORTS |
|
|
|
|
|
Raw Materials |
NA |
22688.200 |
21210.500 |
|
|
Stores and spares, lab
chemicals and other consumables |
NA |
245.000 |
185.100 |
|
|
Capital Goods |
NA |
2418.500 |
1210.800 |
|
|
TOTAL IMPORTS |
NA |
25351.700 |
22606.400 |
|
|
|
|
|
|
|
|
Earnings / (Loss) Per
Share (INR) |
|
|
|
|
Basic |
29.16 |
27.85 |
26.00 |
|
|
|
Diluted |
29.16 |
27.84 |
25.98 |
CURRENT MATURITIES
OF LONG TERM DEBT DETAILS
|
Particulars |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Current Maturities of Long term debt |
1837.400 |
4135.200 |
3882.000 |
|
Cash generated from operations |
29394.900 |
16693.800 |
10163.200 |
|
Net cash flows from operating activities |
24572.000 |
11851.800 |
5904.100 |
QUARTERLY
RESULTS
|
Particulars |
30.06.2017 (Unaudited) |
30.09.2017 (Unaudited) |
31.12.2017 (Unaudited) |
|
|
1st Quarter |
2nd
Quarter |
3rd
Quarter |
|
Net Sales |
21882.900 |
30738.600 |
26862.800 |
|
Total Expenditure |
17185.300 |
20346.200 |
20219.800 |
|
PBIDT (Excl OI) |
4697.600 |
10392.400 |
6643.000 |
|
Other Income |
305.000 |
288.300 |
337.900 |
|
Operating Profit |
5002.600 |
10680.700 |
6980.900 |
|
Interest |
102.500 |
120.200 |
132.100 |
|
Exceptional Items |
NA |
NA |
NA |
|
PBDT |
4900.100 |
10560.500 |
6848.800 |
|
Depreciation |
833.900 |
896.100 |
902.800 |
|
Profit Before Tax |
4066.200 |
9664.400 |
5946.000 |
|
Tax |
992.900 |
2136.100 |
1502.800 |
|
Provisions and
contingencies |
NA |
NA |
NA |
|
Profit After Tax |
3073.300 |
7528.300 |
4443.200 |
|
Extraordinary Items |
NA |
NA |
NA |
|
Prior Period Expenses |
NA |
NA |
NA |
|
Other Adjustments |
NA |
NA |
NA |
|
Net Profit |
3073.300 |
7528.300 |
4443.200 |
KEY
RATIOS
EFFICIENCY RATIOS
|
PARTICULARS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Average Collection Days (Sundry Debtors / Income * 365 Days) |
140.82 |
171.52 |
171.96 |
|
|
|
|
|
|
Account Receivables Turnover (Income / Sundry Debtors) |
2.59 |
2.13 |
2.12 |
|
|
|
|
|
|
Average Payment Days (Sundry Creditors / Purchases * 365 Days) |
100.25 |
105.30 |
106.27 |
|
|
|
|
|
|
Inventory Turnover (Operating Income / Inventories) |
1.03 |
1.09 |
1.08 |
|
|
|
|
|
|
Asset Turnover (Operating Income / Net Fixed Assets) |
0.61 |
0.80 |
0.96 |
LEVERAGE RATIOS
|
PARTICULARS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Debt Ratio ((Borrowing + Current Liabilities) / Total Assets) |
0.36 |
0.48 |
0.50 |
|
|
|
|
|
|
Debt Equity Ratio (Total Liability / Networth) |
0.36 |
0.61 |
0.63 |
|
|
|
|
|
|
Current Liabilities to Networth (Current Liabilities / Net Worth) |
0.53 |
0.78 |
0.74 |
|
|
|
|
|
|
Fixed Assets to Networth (Net Fixed Assets / Networth) |
0.49 |
0.48 |
0.45 |
|
|
|
|
|
|
Interest Coverage Ratio (PBIT / Financial Charges) |
55.58 |
11.51 |
17.54 |
PROFITABILITY RATIOS
|
PARTICULARS |
|
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Net Profit Margin (PAT / Sales) * 100) |
% |
17.45 |
17.45 |
18.73 |
|
|
|
|
|
|
|
Return on Total Assets ((PAT / Total Assets) * 100) |
% |
13.11 |
12.95 |
14.98 |
|
|
|
|
|
|
|
Return on Investment (ROI) ((PAT / Networth) * 100) |
% |
20.23 |
23.69 |
28.10 |
SOLVENCY RATIOS
|
PARTICULARS |
31.03.2017 |
31.03.2016 |
31.03.2015 |
|
Current Ratio (Current Assets / Current Liabilities) |
1.55 |
1.45 |
1.61 |
|
|
|
|
|
|
Quick Ratio ((Current Assets – Inventories) / Current Liabilities) |
1.00 |
1.00 |
1.08 |
|
|
|
|
|
|
G-Score Ratio Financial (Networth / Total Assets) |
0.65 |
0.55 |
0.53 |
|
|
|
|
|
|
G-Score Ratio Debt (Debts / Equity Capital) |
52.12 |
71.18 |
116.08 |
|
|
|
|
|
|
G-Score Ratio Liquidity (Total Current Assets / Total Current Liabilities) |
1.55 |
1.45 |
1.61 |
Total Liability = Short-term Debt + Long-term Debt + Current Maturities of Long-term debts
STOCK
PRICES
|
Face Value |
INR 1.00/- |
|
Market Value |
INR 557.35 /- |
FINANCIAL ANALYSIS
[all figures are
in INR Million]
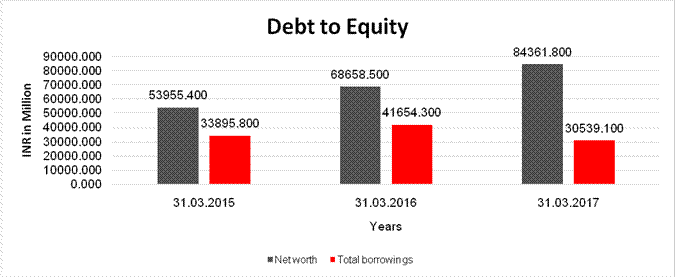
DEBT EQUITY RATIO
|
Particular |
31.03.2015 |
31.03.2016 |
31.03.2017 |
|
|
INR
In Million |
INR
In Million |
INR
In Million |
|
Share Capital |
292.000 |
585.200 |
585.900 |
|
Reserves & Surplus |
53663.400 |
68073.300 |
83775.900 |
|
Money received against
share warrants |
0.000 |
0.000 |
0.000 |
|
Share Application money
pending allotment |
0.000 |
0.000 |
0.000 |
|
Net worth |
53955.400 |
68658.500 |
84361.800 |
|
|
|
|
|
|
Long-term borrowings |
6871.600 |
3347.600 |
1189.000 |
|
Short term borrowings |
23142.200 |
34171.500 |
27512.700 |
|
Current Maturities of
Long term debt |
3882.000 |
4135.200 |
1837.400 |
|
Total borrowings |
33895.800 |
41654.300 |
30539.100 |
|
Debt/Equity ratio |
0.628 |
0.607 |
0.362 |
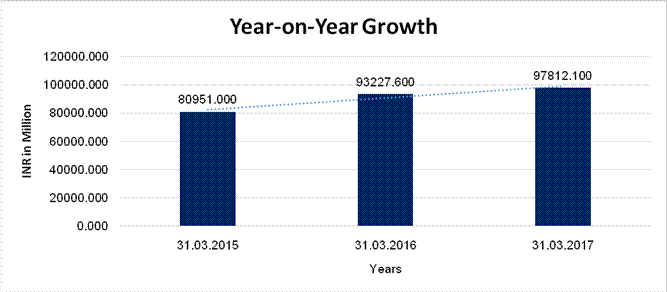
YEAR-ON-YEAR GROWTH
|
Year on Year Growth |
31.03.2015 |
31.03.2016 |
31.03.2017 |
|
|
INR
In Million |
INR
In Million |
INR
In Million |
|
Sales |
80951.000 |
93227.600 |
97812.100 |
|
|
|
15.165 |
4.918 |
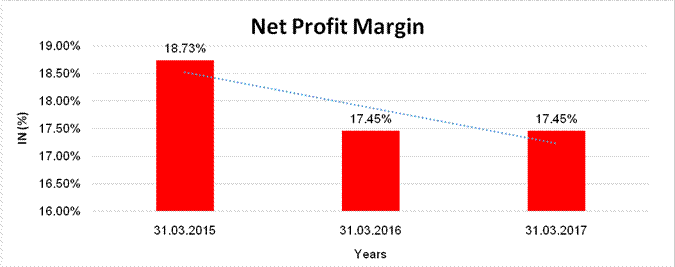
NET PROFIT MARGIN
|
Net Profit Margin |
31.03.2015 |
31.03.2016 |
31.03.2017 |
|
|
INR
In Million |
INR
In Million |
INR
In Million |
|
Sales |
80951.000 |
93227.600 |
97812.100 |
|
Profit/ (Loss) |
15163.500 |
16267.000 |
17067.600 |
|
|
18.73% |
17.45% |
17.45% |
ABRIDGED
BALANCE SHEET (CONSOLIDATED)
|
SOURCES OF FUNDS |
|
31.03.2017 |
31.03.2016 |
|
I.
EQUITY
AND LIABILITIES |
|
|
|
|
(1)Shareholders' Funds |
|
|
|
|
(a) Share Capital |
|
585.900 |
585.200 |
|
(b) Reserves & Surplus |
|
93133.200 |
72287.800 |
|
(c) Money
received against share warrants |
|
0.000 |
0.000 |
|
|
|
|
|
|
(2) Share Application money pending
allotment |
|
0.000 |
0.000 |
|
(3) Minority Interest |
|
20.800 |
25.500 |
|
Total
Shareholders’ Funds (1) + (2) |
|
93739.900 |
72898.500 |
|
|
|
|
|
|
(3)
Non-Current Liabilities |
|
|
|
|
(a) long-term borrowings |
|
1814.000 |
7428.100 |
|
(b) Deferred tax liabilities (Net) |
|
493.000 |
239.600 |
|
(c) Other long term
liabilities |
|
0.000 |
0.000 |
|
(d) long-term
provisions |
|
224.100 |
233.600 |
|
Total Non-current
Liabilities (3) |
|
2531.100 |
7901.300 |
|
|
|
|
|
|
(4)
Current Liabilities |
|
|
|
|
(a) Short
term borrowings |
|
29027.200 |
36726.800 |
|
(b) Trade
payables |
|
24882.700 |
24570.300 |
|
(c) Other
current liabilities |
|
11679.100 |
16595.900 |
|
(d) Short-term
provisions |
|
634.200 |
509.200 |
|
Total Current
Liabilities (4) |
|
66223.200 |
78402.200 |
|
|
|
|
|
|
TOTAL |
|
162494.200 |
159202.000 |
|
|
|
|
|
|
II.
ASSETS |
|
|
|
|
(1)
Non-current assets |
|
|
|
|
(a) Fixed
Assets |
|
|
|
|
(i)
Tangible assets |
|
40830.500 |
33803.900 |
|
(ii)
Intangible Assets |
|
3444.300 |
3930.000 |
|
(iii) Capital
work-in-progress |
|
12373.600 |
8359.000 |
|
(iv)
Intangible assets under development |
|
2207.200 |
122.400 |
|
(v) Goodwill Consolidation |
|
4063.000 |
4063.000 |
|
(b) Non-current Investments |
|
2458.500 |
1229.400 |
|
(c) Deferred tax assets (net) |
|
1677.600 |
2062.800 |
|
(d)
Long-term Loan and Advances |
|
57.000 |
54.500 |
|
(e) Other
Non-current assets |
|
3320.700 |
2633.500 |
|
Total Non-Current
Assets |
|
70432.400 |
56258.500 |
|
|
|
|
|
|
(2) Current
assets |
|
|
|
|
(a)
Current investments |
|
0.200 |
0.200 |
|
(b)
Inventories |
|
43305.400 |
40561.400 |
|
(c) Trade
receivables |
|
27653.300 |
46066.800 |
|
(d) Cash
and cash equivalents |
|
5134.800 |
8002.600 |
|
(e) Short-term
loans and advances |
|
108.900 |
103.100 |
|
(f) Other
current assets |
|
15859.200 |
8209.400 |
|
Total
Current Assets |
|
92061.800 |
102943.500 |
|
|
|
|
|
|
TOTAL |
|
162494.200 |
159202.000 |
PROFIT
& LOSS ACCOUNT (CONSOLIDATED)
|
|
PARTICULARS |
|
31.03.2017 |
31.03.2016 |
|
|
SALES |
|
|
|
|
|
Income |
|
150898.600 |
139552.200 |
|
|
Other Income |
|
1158.900 |
2038.000 |
|
|
TOTAL |
|
152057.500 |
141590.200 |
|
|
|
|
|
|
|
Less |
EXPENSES |
|
|
|
|
|
Cost of Materials Consumed |
|
51849.200 |
48255.600 |
|
|
Purchases of Stock-in-Trade |
|
15585.400 |
14294.200 |
|
|
Changes in inventories of
finished goods, work-in-progress and Stock-in-Trade |
|
(3092.000) |
(928.800) |
|
|
Employees benefits expense |
|
17677.600 |
15426.200 |
|
|
Share of Profit of Joint
Venture |
|
(50.200) |
(14.500) |
|
|
Other expenses |
|
34535.600 |
30623.800 |
|
|
TOTAL
|
|
116505.600 |
107656.500 |
|
|
|
|
|
|
|
Less |
PROFIT/
(LOSS) BEFORE INTEREST, TAX,
DEPRECIATION AND AMORTISATION |
|
35551.900 |
33933.700 |
|
|
|
|
|
|
|
Less |
FINANCIAL
EXPENSES |
|
667.200 |
2567.000 |
|
|
|
|
|
|
|
|
PROFIT
/ (LOSS) BEFORE TAX, DEPRECIATION AND AMORTISATION |
|
34884.700 |
31366.700 |
|
|
|
|
|
|
|
Less/
Add |
DEPRECIATION/
AMORTISATION |
|
4276.300 |
3923.700 |
|
|
|
|
|
|
|
|
PROFIT/ (LOSS)
BEFORE TAX |
|
30608.400 |
27443.000 |
|
|
|
|
|
|
|
Less |
TAX |
|
7596.500 |
7207.100 |
|
|
|
|
|
|
|
|
PROFIT/ (LOSS)
AFTER TAX |
|
23011.900 |
20235.900 |
|
|
|
|
|
|
|
Less
|
NON-CONTROLLING
INTEREST |
|
(4.700) |
(15.000) |
|
|
|
|
|
|
|
|
PROFIT
FOR THE YEAR |
|
23016.600 |
20250.900 |
|
|
|
|
|
|
|
|
Earnings
/ (Loss) Per Share (INR) |
|
39.33 |
34.67 |
LOCAL AGENCY FURTHER INFORMATION
|
Sr. No. |
Check list by
info agents |
Available in
Report (Yes/No) |
|
1 |
Year of establishment |
Yes |
|
2 |
Constitution of the entity -Incorporation
details |
Yes |
|
3 |
Locality of the entity |
Yes |
|
4 |
Premises details |
No |
|
5 |
Buyer visit details |
- |
|
6 |
Contact numbers |
Yes |
|
7 |
Name of the person contacted |
No |
|
8 |
Designation of contact person |
No |
|
9 |
Promoter’s background |
Yes |
|
10 |
Date of Birth of Proprietor / Partners /
Directors |
Yes |
|
11 |
Pan Card No. of Proprietor / Partners |
Yes |
|
12 |
Voter Id Card No. of Proprietor / Partners |
No |
|
13 |
Type of business |
Yes |
|
14 |
Line of Business |
Yes |
|
15 |
Export/import details (if applicable) |
No |
|
16 |
No. of employees |
Yes |
|
17 |
Details of sister concerns |
Yes |
|
18 |
Major suppliers |
No |
|
19 |
Major customers |
No |
|
20 |
Banking Details |
Yes |
|
21 |
Banking facility details |
Yes |
|
22 |
Conduct of the banking account |
-- |
|
23 |
Financials, if provided |
Yes |
|
24 |
Capital in the business |
Yes |
|
25 |
Last accounts filed at ROC, if applicable |
Yes |
|
26 |
Turnover of firm for last three years |
Yes |
|
27 |
Reasons for variation <> 20% |
-- |
|
28 |
Estimation for coming financial year |
No |
|
29 |
Profitability for last three years |
Yes |
|
30 |
Major shareholders, if available |
Yes |
|
31 |
External Agency Rating, if available |
Yes |
|
32 |
Litigations that the firm/promoter involved
in |
--- |
|
33 |
Market information |
--- |
|
34 |
Payments terms |
No |
|
35 |
Negative Reporting by Auditors in the
Annual Report |
No |
PERFORMANCE REVIEW
The Company delivered yet another year of consistent and profitable growth, continued to build a robust pipeline of products, stepped up market development, significantly expanded manufacturing capacities, invested to further improve reliability of deliverables and enhanced the level of execution across the Company. There was a sustained focus on research and development, employee engagement, competency enhancement and relentless pursuit of excellence in quality.
The team at Aurobindo demonstrated resilience despite several challenges, especially in dealing with competitive pricing pressures in the market as well as hardening of the rupee in the latter half of the financial year. At the market place, the momentum was maintained by improving the product mix with high value, differentiated oral solid and injectable products. The Company was quick to respond to the challenges of the market environment by executing improvements in operations. There was considerable work done to scale up volumes, improve efficiencies, optimize costs and fine-tune supply chain and logistics in order to expand the bottom line.
Productivity enhancements and process improvements were proactively undertaken to benchmark the manufacturing systems with the best in the industry. The standalone revenue at INR 97812.100 million increased by 4.9% in 2016-17. Operating profit including forex and other income was higher by 1.4% over the previous year. Profit before tax was higher by 1.4% at INR 21785.700 million over the previous year. Net profit for the year 2016-17 at INR 17067.600 million was an increase of 4.9% compared to 2015-16. The diluted earnings per share for 2016-17 is INR 29.16 as compared to INR 27.84 in 2015-16. At the consolidated level, the Company delivered solid financial results fuelled by strong performance in US and European markets. The revenues increased to INR 150898.600 million, a growth of 8.1% over the previous year with an EBITDA margin of 22.8%. EBITDA for the year was INR 34342.800 million as against INR 31881.200 million in the previous year and the diluted earnings per share grew by 13.5% to INR 39.33. US formulations business contributed 57% to the overall formulations revenue during the year, as against 55% in the previous year. The revenue generated from the US business grew by 12.3% at INR 68272.300 million in 2016-17 over the previous year sales of INR 60785.500 million.
The Company maintained its momentum of growth with the launch of several new oral and injectable products in the US, led by final approvals for 61 products from the US FDA during the financial year. Aurobindo continues to witness strong pace of approvals from US FDA, which helped to maintain growth momentum in sales and improve market presence. In keeping with the past track record, speed to the market and effectiveness in execution has remained the hallmark of Aurobindo's performance.
As a result of customer centric approach and relationship oriented marketing, the Company has become the 6th ranked Rx supplier of prescriptions dispensed (as per IMS National Prescription Audit, April 2017). In the highly competitive US market, the Company has diversified product basket in oral solids including controlled substances and injectable.
OUTLOOK
The Company's endeavor has been to invest in reliability; ensure patient safety through high-end quality of products and processes; develop alternate API and excipient sources to deliver larger volumes, in line with customer expectations on-time-in-full; possess high value, differentiated portfolio of complex molecules; build state-of-theart manufacturing facilities that meet compliance standards; ensure safe working environment to protect the health of the employees; minimize waste and maximize recycling of materials; reduce the risk in operations; and be a preferred partner to all the stakeholders. In effect, strive for execution excellence and be a responsible corporate citizen.
Aurobindo has made significant progress in all these areas, but the team works with the belief that there is room for improvement. Operational excellence is often reviewed closely to further improve productivity, become cost effective and be reliable in every transaction. The market environment is changing rapidly, with newer challenges and newer opportunities. The Company has been conscious to work ahead of time and has invested in several technologies and platforms such as biotechnology, oncology, hormones, steroids, biocatalysts, peptides, vaccines, penems, depot injections, dermatology, inhalers, nasal, patches and films to sustain the growth.
Aurobindo is striving to stay ahead of the curve. A large portfolio is being built of differentiated products which would act as a moat against competitive pressures; new manufacturing facilities are under construction to cater to the growing portfolio; several of the newer technologies would help enhance margins; process improvements and better logistics management are expected to strengthen competitiveness. The Company's product portfolio and pipeline for the US market have significant potential for sustainable volume growth. This is a quality conscious, knowledge driven market and the Company is far better positioned with offers in several therapeutic segments. Pricing pressures in US markets are expected to stay and there is the risk of the ability to maintain current margins. Price sensitivities will test all the players in a crowded market where price tends to sag while volume business gets done.
This threat does not affect Aurobindo significantly, because of its large portfolio of products, control over raw material sourcing and lower product concentration. The Company is a dominant player in the active ingredients business and has been able to control its quality, improve on timelines, be competitive on its costs and has the ability to deliver at short notice. This is a unique advantage that Aurobindo enjoys over competing manufacturers across the world. Competitive pressures and resultant price erosion in US markets has galvanized the Company to focus on the organizational strength, leverage the full capabilities and competencies of the cadre of dedicated and highly experienced professionals. The Company will continue to work to protect the revenues and bottom line, to turn challenges and opportunities into successes.
Research and development (R&D) activity being undertaken is focused on difficult to manufacture, differentiated products, with possible low competitive pressure. Work is on-going in differentiated molecules, both for oral and injectable products. For instance, the Company successfully developed and filed 4 penems for the US market and received approval for one product, an injection drug, at the end of the financial year. This product was successfully launched in April 2017.
Penems are difficult to develop products. The development and filing is in-line with the strategy of moving towards complex/specialty products. The recent acquisition of 5 biosimilar molecules is in furtherance of the same strategy. These are complex biosimilars, and the plans are to take a lead molecule from this transaction for clinical trials in 2017. This is an anti-antiogenesis drug used in treatment of multiple-cancers including metastatic colon or rectal cancer, non-squamous and non-small cell lung cancers. Apart from these molecules, the Company is working on development of biosimilars in therapeutic segments such as oncology, auto immune disorders and ophthalmology. In keeping with the need to manufacture a growing product pipeline, the Company, in addition to commissioning the formulations manufacturing facility at Unit XV referred earlier, has initiated significant improvements in capacities to boost volumes, as given below:
_ Unit X: The Company is building a US FDA compliant oral manufacturing facility at Naidupet, Andhra Pradesh, which will be commissioned in 2017-18. It is presently at project stage, where validation batches are being taken and is being got ready for regulatory inspection;
_ Unit XVI: The Company is building another US FDA compliant Betalactum injectables manufacturing facility at Jedcherla, near Hyderabad. It is planned to get the facility commissioned in 2017-18;
_ The Company is in the processes of tripling the capacity at AuroLife manufacturing facility, which will significantly boost volumes for the US market.
In order to sustain future growth and spread the geographical risk, Aurobindo has been steadily expanding its European footprint since 2006, via acquisitions across several key markets and building a diversified product basket. Most notably, in 2014 the acquisition of Actavis's commercial operations in seven Western European countries added traction to the Company's presence in these developed markets. The acquisition of Generis, referred to earlier, builds upon an already successful growth strategy. Members will recall, Arrow Generiques S.A.S., the French subsidiary of Aurobindo acquired select commercial products in Calcium and Calcium Vitamin D3, including the use of the Orocal trademark. This acquisition enables Arrow Generiques to continue to increase its branded products portfolio and leverage its position as a key player in the market, with focus on selling generics in the retail and hospital markets in France. The Company is well balanced between generics/branded products/biosimilars in the retail and hospital markets. A dedicated Business Unit with sales and marketing team has been set up specifically to enhance this business. Arrow Generiques has continued developing the brand awareness among prescribers through promotion of mature products and launch of specialties for patients care. The present arrangement will boost the position of Arrow Generiques and open new opportunities for the future.
Aurobindo is committed to the larger cause of bringing affordable HIV drugs to millions of patients. Today, in addition to its existing powerful portfolio of products, the Company is bringing a one-of-its-kind generic version of a valuable drug, as also developing a fixed dose combination drug, to help achieve the UNAIDS 90-90-90 goals and an AIDS-free generation. The UNAIDS 90-90-90 goal is an ambitious treatment target aimed at goals briefly described below:
_ By 2020, 90% of all people living with HIV will know their HIV status.
_ By 2020, 90% of all people with diagnosed HIV infection will receive sustained antiretroviral therapy.
_ By 2020, 90% of all people receiving antiretroviral therapy will have viral suppression.
The Company is striving to help achieve these goals by proactively meeting the needs by offering products in several countries. The World Health Organization has recommended Aurobindo's firstline therapy against HIV which is expected to see rapid growth in demand now that a cost-effective generic product is made available to the market. The Company's products have the potential to improve the lives of millions of patients.
MANAGEMENT DISCUSSION AND ANALYSIS
ECONOMIC BACKGROUND
India has emerged as the fastest growing major economy in the world as per the Central Statistics Organisation and International Monetary Fund. The Government of India has forecasted that the Indian economy will grow by 7.1% in 2016-17. As per the Economic Survey 2016-17, the economy should grow between 6.75% and 7.5% in 2017-18. The improvement in India's economic fundamentals has accelerated since 2014 with the combined impact of strong government reforms, Reserve Bank of India's inflation focus supported by benign global commodity prices. India's consumer confidence index stood at 136 in the fourth quarter of 2016, topping the global list of countries on the same parameter, as a result of strong consumer sentiment, according to market research agency, Nielsen. Corporate earnings in India are expected to grow by over 20% in 2017-18 supported by normalization of profits, especially in sectors like automobiles and banks, while GDP is expected to grow by 7.5% during the same period, according to Bloomberg consensus.
Moody's has affirmed the Government of India's Baa3 rating with a positive outlook stating that the reforms by the government will enable the country perform better compared to its peers over the medium term. With the expected normal monsoon in 2017, and the implementation of GST, there is considerable optimism across all sections of the economy.
INDUSTRY PERSPECTIVE
According to the Ministry of Commerce and Industry, Government of India, Indian pharmaceuticals market is the third largest in terms of volume and thirteenth largest in terms of value. India is the largest provider of generic drugs globally with the accounting for 20% of global exports in terms of volume. India enjoys an important position in the global pharmaceuticals industry. The country also has a large pool of scientists and engineers who have the potential to steer the industry ahead to an even higher level. The Indian pharma industry, which is expected to grow by over 15% per annum between 2015 and 2020, will outperform the global pharma industry, which is set to grow at an annual rate of 5% in the same period. The market is expected to grow to USD 55 billion by 2020, thereby emerging as the sixth largest pharmaceutical market globally by absolute size (Source: www.ibef.org of Ministry of Commerce & Industry, Government of India, updated June 2017). India has maintained its lead over China in pharmaceutical exports with a year-on-year growth of 11.4% to USD 12.9 billion in 2015-16, according to data from the Ministry of Commerce and Industry. In addition, Indian pharmaceutical exports are estimated to have grown between 8-10% in 2016-17. Imports of pharmaceutical products rose marginally by 0.8% year-on-year to USD 1,641.1 million.
Overall drug approvals given by the US Food and Drug Administration (USFDA) to Indian companies had nearly doubled to 201 in 2015-16 from 109 in 2014-15. The country accounts for around 30% (by volume) and about 10% (by value) in the USD 70-80 billion US generics market. India's biotechnology industry comprising biopharmaceuticals, bio-services, bio-agriculture, bio-industry and bioinformatics is expected grow at an average growth rate of around 30% a year and reach USD 100 billion by 2025. The Government of India unveiled 'Pharma Vision 2020' aimed at making India a global leader in end-to-end drug manufacture. Approval time for new facilities has been reduced to boost investments. The government proposes to set up chemical hubs across the country, provide early environment clearances in existing clusters and help create adequate infrastructure. Some of the major initiatives taken by the government to promote the pharmaceutical sector in India are as follows:
_ The Government of India plans to set up eight mini drug-testing laboratories across major ports and airports in the country, which is expected to improve the drug regulatory system and infrastructure facilities by monitoring the standards of imported and exported drugs and reduce the overall time spent on quality assessment.
_ India is expected to rank among the top five global pharmaceutical innovation hubs by 2020, based on Government of India's decision to allow 50% public funding in the pharmaceuticals sector through its Public Private Partnership (PPP) model.
_ Indian Pharmaceutical Association (IPA), the professional association of pharmaceutical companies in India, plans to prepare data integrity guidelines which will help to measure and benchmark the quality of Indian companies with global peers.
_ The Government of India plans to incentivize bulk drug manufacturers, including both state-run and private companies, to encourage 'Make in India' program and reduce dependence on imports of Active Pharmaceutical Ingredients (API), nearly 85% of which come from China (Source: www.ibef.org of Ministry of Commerce & Industry, Government of India, updated June 2017).
_ The Department of Pharmaceuticals has set up an inter-ministerial co-ordination committee, which would periodically review, coordinate and facilitate the resolution of the issues and constraints faced by the Indian pharmaceutical companies.
The Indian pharmaceutical market size is expected to grow to USD 100 billion by 2025, driven by increasing consumer spending, rapid urbanization, and raising healthcare insurance among others (Source: www.ibef.org of Ministry of Commerce & Industry, Government of India, updated June 2017). Across the world, speedy introduction of generic drugs into the market has remained in focus and is expected to benefit the Indian pharmaceutical companies. In addition, the thrust on rural health programs, lifesaving drugs and preventive vaccines also augurs well for the pharmaceutical companies. Traditionally pharmaceutical companies undergo an expensive and time consuming process to develop new drugs. It often entails many years and billions of dollars in research and development, as well as an arduous approval process by the US FDA. In exchange, the FDA generally provides drug companies with 12 years of patent protection and, in effect, a monopoly on sales during that period. This allows the drug companies to recoup their costs, earn a profit, and start the process anew for the next wave of new and innovative drugs. Demand for generic drugs will continue to rise as payers take advantage of patent expiries to reduce costs. A generic drug can only be introduced once patent protection on the associated brand-name drug has expired, and exclusivity is granted only to the companies which challenge patents. Generally generics sold at steep discounts to their branded counterparts, and often gain large portions of market share from branded drugs in a short amount of time following patent expirations.
According to the IMS and the DCAT, generics remain the biggest contributor to the pharmaceutical industry's global growth. According to the IMS report 'Global Outlook for Medicines through 2018,' generics will be responsible for 52% of growth in global medicine spending between 2014 and 2018. The scope of generics is immense, and it's only set to grow as more drugs come off their patents. A generic drug (commonly referred as generics) is a drug defined as 'a drug product that is comparable to a brand/reference listed drug product in dosage form, strength, quality and performance characteristics and intended use.' Generic drugs are subject to the regulations of the governments of countries where they are dispensed. A generic drug must contain the same active ingredients as the original formulation. According to the US FDA, generic drugs are identical or within an acceptable bioequivalent range to the brand-name counterpart with respect to pharmacokinetic and pharmacodynamics properties. By extension, therefore, generics are considered (by the FDA) identical in dose, strength, route of administration, safety, efficacy, and intended use. In most cases, generic products are available once the patent protections afforded o the original developer have expired.
In contrast, generic drug companies operate very differently. The amount of money they need to invest to produce a generic is much less than the traditional branded drug companies, since the drug has already been formulated and approved. In order to protect the traditional branded drug companies and allow them to earn a return on investment, generic drugs can only be introduced after the patent protection has expired, if the patent protection has not been further extended by the FDA. Even then, the FDA-approved copies of branded drugs are only given a short 180-day exclusivity period if their generic is the first to launch in the market (Para IV). Once generic drugs are introduced to the market, they are typically sold at steep discounts (50% to 90%) compared to the branded alternative and, as a result, on average, branded drugs typically lose 70% or more of their market share to generics in a short amount of time.
As per IMS report, today in the US, generic drugs account for 90% of all prescriptions filled and may account for 92% of prescription volumes by 2021. According to a report by IMS Health, generic drugs are expected to account for 52% of global pharmaceutical spending growth, compared to 35% for branded drugs. It was reported by IMS in Q2 2015, the U.S. healthcare to the availability of low cost generics. Major factors driving the growth of generics include popular branded drugs losing their patent protection (known as a 'patent cliff'), support for generics from governments, new complex generics coming into the market, and industry consolidation. As per the 'Outlook for Global Medicines through 2021' published by Quintiles IMS Institute in December 2016, total volume of medicines consumed globally will increase by about 3% annually through 2021, only modestly faster than the population and demographic shifts, but driven by very different factors around the world. Spending on medicines will grow by 4-7%, primarily driven by newer medicines in developed markets and increased volume in emerging markets.
Developed markets will offset increased costs from new medicines with the use of generics, and a greater focus on pricing and access measures, while emerging markets will struggle to live up to the promised access expansions made when their economic outlooks were stronger. Global medicine spending is estimated to reach nearly USD 1.5 trillion by 2021 on an invoice price basis, up nearly USD 370 billion from the 2016 estimated spending level. Most global spending growth, particularly in developed markets, will be driven by oncology, autoimmune and diabetes treatments where significant innovations are expected. The U.S. will continue as the world's largest pharmaceutical market and emerging markets will make up 9 of the top 20 markets. China will continue as the #2 market, a rank it has held since 2012. Developed market spending growth will be driven by original brands, while emerging markets will continue to be fueled by nonoriginal products that make up an average 91% of emerging market volume and 78% of spending.
The US nutraceutical market is at around USD 38 billion and is expected to grow to USD 46 billion by 2018 as per 2015 Nutrition Business Journal. According to a market report published by Transparency Market Research 'Nutraceuticals Market - Global Industry Analysis, Size, Share, Growth and Forecast, 2015-21', global nutraceuticals market was valued at USD 165.6 billion in 2014 and is expected to reach USD 278.9 billion by 2021, growing at a CAGR of 7.3% from 2015 to 2021. North America is expected to dominate the global nutraceuticals market in terms of demand over the forecast period. The growth drivers for the US nutraceutical market is ageing population, increased consumer focus on proactive health management, increase/ perceived increase in condition-specific disorders, growing consumer interest in drug-free/natural health solutions that are proven effective. Key macro segments in the US nutraceutical category is vitamins, specialty supplements, herbs & botanicals, sports nutrition, meal replacements and minerals. Seen from India perspective, the country has over 10,500 manufacturing units and over 3,000 pharma companies and exports all forms of pharmaceuticals from active pharmaceutical ingredients (APIs) to formulations, both in modern medicine and traditional Indian medicines.
The Indian pharma industry stands diversified into various spheres of activities including research and development (R&D), manufacturing of branded, generic and branded generic drugs, manufacturing APIs, laboratory testing and clinical research. Indian pharmaceutical manufacturing facilities registered with US FDA as on April 2015 was the highest at 605 (Source: Pharmexcil annual report 2016) for any country outside the US. In a study of the top 20 countries that have filed Drug Master Files, India stood first with 3,820 DMFs that are currently active, filed as at December 2015. The trend continues even today. Indian pharma exports have consistently improved over the years. India exports over USD 16.9 billion worth of medicines, of which more than 50% were to regulated markets. The US with 33% is the India's largest pharma export destination followed by Africa and the European Union. (Source: Pharmexcil annual report 2015-16). These figures indicate that Indian medicines have established themselves as affordable and reliable, across the world. India's exports of generics have been growing at a rate of nearly 22% annually over the past four years. It is estimated that around 40% of the generic drugs in the US come from India. The growth momentum is likely to continue. The drugs and pharmaceuticals sector attracted cumulative foreign direct investment (FDI) inflows worth USD 14,706.9 million between April 2000 and March 2017, according to data released by the Department of Industrial Policy and Promotion (DIPP).
Several state governments are encouraging the pharmaceutical industry. For instance, the State of Telangana has proposed to set up India's largest integrated pharmaceutical city spread over 11,000 acres near Hyderabad, complete with effluent treatment plants and a township for employees, in a bid to attract investment of INR 300 billion in phases. The enormous opportunity for the industry can be best illustrated from the projected human resource requirement of the Indian pharma sector, estimated to be about 2.15 million by 2020. The Indian pharmaceuticals market increased at a CAGR of 17.5% during 2005-16 with the market increasing from USD 6 billion in 2005 to USD 36.7 billion in 2016 and is expected to expand at a CAGR of 15.9% to USD 55 billion by 2020. More important, by 2020, India is likely to be among the top three pharmaceutical markets by incremental growth and sixth largest market globally in absolute size. There are however estimates which suggest more aggressive growth. As per India Ratings, a Fitch Group company, the industry is estimated to grow at 20% compounded annual growth rate (CAGR) over the next five years.
CORPORATE INFORMATION
Subject the Company is a public company domiciled in India and was incorporated under the provisions of the Companies Act applicable in India. The registered office of the Company is located at Plot No.2, Maithri Vihar, Ameerpet, Hyderabad - 500038, India and the Corporate office is located at The Water Mark Building, Plot No. 11, Survey No. 9, Hi-tech City, Hyderabad - 500084, India. Its shares are listed on two recognized stock exchanges in India. The Company is principally engaged in manufacturing and marketing of active pharmaceutical ingredients, generic pharmaceuticals and related services. These financial statements were authorized for issue in accordance with the resolution of the Directors on May 29, 2017.
UNSECURED LOAN
|
PARTICULARS |
31.03.2017 (INR
in Million) |
31.03.2016 (INR
in Million) |
|
Long-term Borrowings |
|
|
|
Long-term maturities of deferred sales tax loan |
0.000 |
255.700 |
|
Short-term
borrowings |
|
|
|
Long-term maturities of deferred sales tax loan |
0.000 |
160.000 |
|
Working capital demand loan (Unsecured) |
750.000 |
0.000 |
|
Packing credit loans (Unsecured) |
24100.300 |
22785.700 |
|
Bill discounting (Unsecured) |
702.300 |
8425.800 |
|
Total |
25552.600 |
31627.200 |
CONTINGENT
LIABILITIES:
(INR in Million)
|
PARTICULARS |
31.03.2017 |
|
Outstanding bank guarantees |
877.600 |
|
Corporate guarantees for loans taken by wholly owned subsidiaries |
2339.500 |
|
Claims arising from disputes not acknowledged as debts - indirect taxes (excise duty and service tax)*# |
320.400 |
|
Claims arising from disputes not acknowledged as debts - direct taxes |
586.300 |
|
Claims against the Company not acknowledged as debts - other duties/claims* |
150.300 |
INDEX OF CAHREGS:
|
SNo |
SRN |
Charge Id |
Charge Holder Name |
Date of Creation |
Date of Modification |
Date of Satisfaction |
Amount |
Address |
|
1 |
C47353040 |
10402714 |
DBS Bank Ltd |
08/01/2013 |
20/02/2015 |
- |
1640000000.0 |
Salarpuria WindsorNo.3, Ulsoor RoadBengaluruKA560042IN |
|
2 |
B41106147 |
10359357 |
IFCI Limited |
25/05/2012 |
- |
- |
1560000000.0 |
IFCI Tower5-9-13, Taramandal Complex, SaifabadHyderabadAP500004IN |
|
3 |
C03655834 |
10215595 |
ICICI Bank Limited |
26/04/2010 |
19/03/2014 |
- |
2050000000.0 |
ICICI Bank Tower, Plot No.12, Nanakram Guda6th Floor, Tower II, North WingHyderabadAP500032IN |
|
4 |
C21711395 |
10044936 |
HDFC BANK LIMITED |
16/02/2007 |
25/08/2014 |
- |
1640000000.0 |
HDFC BANK HOUSESENAPATI BAPAT MARGLOWER PAREL WMUMBAIMH400013IN |
|
5 |
G12805800 |
10030551 |
IDBI Bank Limited |
05/12/2006 |
14/09/2016 |
- |
2000000000.0 |
5-9-89/1 & 2, Chapel RoadPB No.370HYDERABADTG500001IN |
|
6 |
G48227177 |
80022102 |
Canara Bank |
21/09/2000 |
29/06/2017 |
- |
500000000.0 |
Prime Corporate Branch, TSR Complex, 2nd Floor1-7-1, Sardar Patel RoadSECUNDERABADTG500003IN |
|
7 |
G47365028 |
80013577 |
ANDHRA BANK |
01/04/2000 |
22/06/2017 |
- |
2000000000.0 |
Somajiguda Branch, 6-3-352/2, Astra HeightsRoad No.1, Banjara HillsHYDERABADTG500034IN |
|
8 |
C40060543 |
80013576 |
State Bank of India |
05/06/1997 |
15/12/2014 |
- |
8200000000.0 |
Corporate Account Group BranchOZONE, 2nd Floor, # 6-3-669, PanjaguttaHyderabadTG500082IN |
|
9 |
B96933965 |
80026247 |
Standard Chartered Bank |
29/11/1995 |
18/02/2014 |
- |
1640000000.0 |
6-3-1090, TSR Towers, Raj Bhavan RoadSomajigudaHyderabadAP500082IN |
|
10 |
G78881646 |
80013053 |
State Bank of Hyderabad |
29/11/1995 |
05/01/2015 |
08/03/2018 |
12750000000.0 |
Industrial Finance BranchTopaz Building, Amrutha Hills, PanjaguttaHyderabadTG500082IN |
FIXED ASSETS
- Leasehold Land
- Freehold Land
- Leasehold buildings
- Freehold buildings
- Plant and Machinery
- Furniture and fittings
- Vehicles
- Office Equipment
UNAUDITED FINANCIAL RESULTS FOR THE QUARTER ENDED AND NINE MONTHS
DECEMBER 31, 2017
[INR
IN MILLION]
|
PARTICULARS |
3 Months |
9 Months |
|
|
|
31.12.2017 |
30.09.2017 |
31.03.2017 |
|
|
[Unaudited] |
[Unaudited] |
[Unaudited] |
|
1. Income from Operations |
|
|
|
|
Net Sales/income from
operations |
26862.800 |
30738.600 |
79484.300 |
|
Other Operating Income |
337.900 |
288.300 |
742.900 |
|
Total income from operations (net) |
27200.700 |
31026.900 |
80227.200 |
|
|
|
|
|
|
Expenses |
|
|
|
|
Cost of materials consumed |
12414.900 |
12623.900 |
37017.800 |
|
Purchases of stock-in trade |
16.000 |
6.000 |
33.000 |
|
Changes in inventories of finished goods. work-in-progress and stock
in trade |
111.000 |
(388.200) |
(2436.900) |
|
Employee benefits expense |
2831.800 |
2713.100 |
8142.500 |
|
Depreciation and Amortization Expenses |
902.800 |
896.100 |
2632.800 |
|
Other Expenses |
4846.100 |
5391.400 |
14806.600 |
|
Finance Costs |
132.100 |
120.200 |
354.800 |
|
Total expenses |
21254.700 |
21362.500 |
60550.600 |
|
Profit/ (Loss) from ordinary activities after finance cost but before
exceptional items |
5946.000 |
9664.400 |
19676.600 |
|
Exceptional items |
0.000 |
0.000 |
0.000 |
|
Profit/ (Loss) from ordinary activities before tax |
5946.000 |
9664.400 |
19676.600 |
|
Tax expenses |
1502.800 |
2136.100 |
4631.800 |
|
Net Profit / (Loss) from ordinary activities after tax |
4443.200 |
7528.300 |
15044.800 |
|
Extraordinary item (net of tax expense) |
0.000 |
0.000 |
0.000 |
|
Net Profit / (Loss) for the period |
4443.200 |
7528.300 |
15044.800 |
|
Comprehensive Income |
(13.700) |
(12.000) |
(41.100) |
|
Net Profit/ (Loss) after taxes, minority interest and share of
profit/(loss) of associates |
4429.500 |
7516.300 |
15003.700 |
|
|
|
|
|
|
Paid up equity share capital (Face Value of INR 1/-each) |
585.900 |
585.900 |
585.900 |
|
Reserve excluding Revaluation Reserve as per Balance Sheet of previous
accounting year |
- |
- |
- |
|
Earnings per share (before extraordinary items) of INR 10/- each (not
annualized): |
- |
- |
- |
|
(a) Basic |
7.58 |
12.85 |
25.68 |
|
(b) Diluted |
7.58 |
12.85 |
25.68 |
Note:
- The financial results of the Company have been prepared in accordance with the Indian Accounting Standards (Ind AS) prescribed under Section 133 of the Companies Act, 2013, rend with the relevant rules issued thereunder.
- The above standalone financial results as reviewed by the audit committee have been approved by Board of Directors at its meeting held on February 7, 2018.
- Post implementation of Goods and Services Tax {'GST') with effect from July 1, 2017, revenues from operations is disclosed net of GST Revenue from operations for the earlier periods included excise duty which is now subsumed In the GST Revenue from operations for the nine months ended December 31, 2017 includes excise duty upto June 30, 2017 Accordingly, revenue from operations for the quarter and nine months ended December 31, 2017 are not comparable with those of the previous periods presented.
- During the quarter i) Aurobindo Mac Sanayi Ve Ticaret Limited Sirketi, Turkey has been liquidated effective October 31, 2017, ii)Auro Phama India Private Limited, India has been incorporated as a wholly owned subsidiary of Aurobindo Pharma Limited on December 20, 2017, iii)Aurovitas Pharma Ceska republika s.r.o, Czech Republic has been incorporated on December 23, 2017 as a subsidiary of Agile Pharma B.V.
- The Company operates in only one segment viz., 'Pharmaceutical Products'.
- Sales of standalone for current quarter include exports of INR 22313.800 million (December 31, 2016: INR 20471.900 million).
- The Board has approved second interim dividend @100% i.e., INR 1 (Rupee One only) per equity share of INR 1 (Rupee One only) for the year 2017-18.
- Previous period figures have been regrouped/ rearranged wherever considered necessary to conform to the current period presentation.
CMT REPORT (Corruption, Money Laundering & Terrorism]
The Public Notice information has been collected from various sources
including but not limited to: The Courts,
1] INFORMATION ON
DESIGNATED PARTY
No records exist designating subject or any of its beneficial owners,
controlling shareholders or senior officers as terrorist or terrorist
organization or whom notice had been received that all financial transactions
involving their assets have been blocked or convicted, found guilty or against
whom a judgement or order had been entered in a proceedings for violating
money-laundering, anti-corruption or bribery or international economic or
anti-terrorism sanction laws or whose assets were seized, blocked, frozen or
ordered forfeited for violation of money laundering or international
anti-terrorism laws.
2] Court Declaration :
No records exist to suggest that subject is
or was the subject of any formal or informal allegations, prosecutions or other
official proceeding for making any prohibited payments or other improper
payments to government officials for engaging in prohibited transactions or
with designated parties.
3] Asset Declaration :
No records exist to suggest that the property or assets of the subject
are derived from criminal conduct or a prohibited transaction.
4] Record on Financial
Crime :
Charges or conviction
registered against subject: None
5] Records on Violation of
Anti-Corruption Laws :
Charges or
investigation registered against subject: None
6] Records on Int’l
Anti-Money Laundering Laws/Standards :
Charges or
investigation registered against subject: None
7] Criminal Records
No
available information exist that suggest that subject or any of its principals have
been formally charged or convicted by a competent governmental authority for
any financial crime or under any formal investigation by a competent government
authority for any violation of anti-corruption laws or international anti-money
laundering laws or standard.
8] Affiliation with
Government :
No record
exists to suggest that any director or indirect owners, controlling
shareholders, director, officer or employee of the company is a government
official or a family member or close business associate of a Government
official.
9] Compensation Package :
Our market
survey revealed that the amount of compensation sought by the subject is fair
and reasonable and comparable to compensation paid to others for similar
services.
10] Press Report :
No press reports / filings exists on
the subject.
CORPORATE GOVERNANCE
MIRA INFORM as part of its Due Diligence do provide comments on
Corporate Governance to identify management and governance. These factors often
have been predictive and in some cases have created vulnerabilities to credit
deterioration.
Our Governance Assessment focuses principally on the interactions
between a company’s management, its Board of Directors, Shareholders and other
financial stakeholders.
CONTRAVENTION
Subject is not known to have contravened any existing local laws,
regulations or policies that prohibit, restrict or otherwise affect the terms
and conditions that could be included in the agreement with the subject.
FOREIGN EXCHANGE RATES
|
Currency |
Unit
|
INR |
|
US Dollar |
1 |
INR 64.91 |
|
|
1 |
INR 91.87 |
|
Euro |
1 |
INR 80.26 |
INFORMATION DETAILS
|
Information
Gathered by : |
|
|
|
|
|
Analysis Done by
: |
|
|
|
|
|
Report Prepared
by : |
MTN |
SCORE FACTORS
|
DEMERIT POINTS |
|
|
|
--BANK CHARGES |
YES/NO |
YES |
|
--LITIGATION |
YES/NO |
NO |
|
--OTHER ADVERSE INFORMATION |
YES/NO |
NO |
|
MERIT POINTS |
|
|
|
--SOLE DISTRIBUTORSHIP |
YES/NO |
NO |
|
--EXPORT ACTIVITIES |
YES/NO |
NO |
|
--AFFILIATION |
YES/NO |
YES |
|
--LISTED |
YES/NO |
YES |
|
--OTHER MERIT FACTORS |
YES/NO |
YES |
RATING EXPLANATIONS
|
Credit Rating |
Explanation |
Rating Comments |
|
A++ |
Minimum Risk |
Business dealings permissible with minimum
risk of default |
|
A+ |
Low Risk |
Business dealings permissible with low
risk of default |
|
A |
Acceptable Risk |
Business dealings permissible with
moderate risk of default |
|
B |
Medium Risk |
Business dealings permissible on a regular
monitoring basis |
|
C |
Medium High Risk |
Business dealings permissible preferably
on secured basis |
|
D |
High Risk |
Business dealing not recommended or on
secured terms only |
|
NB |
New Business |
No recommendation can be done due to
business in infancy stage |
|
NT |
No Trace |
No recommendation can be done as the business is not
traceable |
NB is stated where there is insufficient information to facilitate rating. However, it is not to be considered as unfavourable.
This score serves as a reference to assess
SC’s credit risk and to set the amount of credit to be extended. It is
calculated from a composite of weighted scores obtained from each of the major
sections of this report. The assessed factors are as follows:
·
Financial
condition covering various ratios
·
Company
background and operations size
·
Promoters
/ Management background
·
Payment
record
·
Litigation
against the subject
·
Industry
scenario / competitor analysis
·
Supplier
/ Customer / Banker review (wherever available)
This report is issued at
your request without any risk and responsibility on the part of MIRA INFORM
PRIVATE LIMITED (MIPL) or its officials.

